The Role of the Japanese Traditional Diet in Healthy and Sustainable Dietary Patterns around the World
Abstract
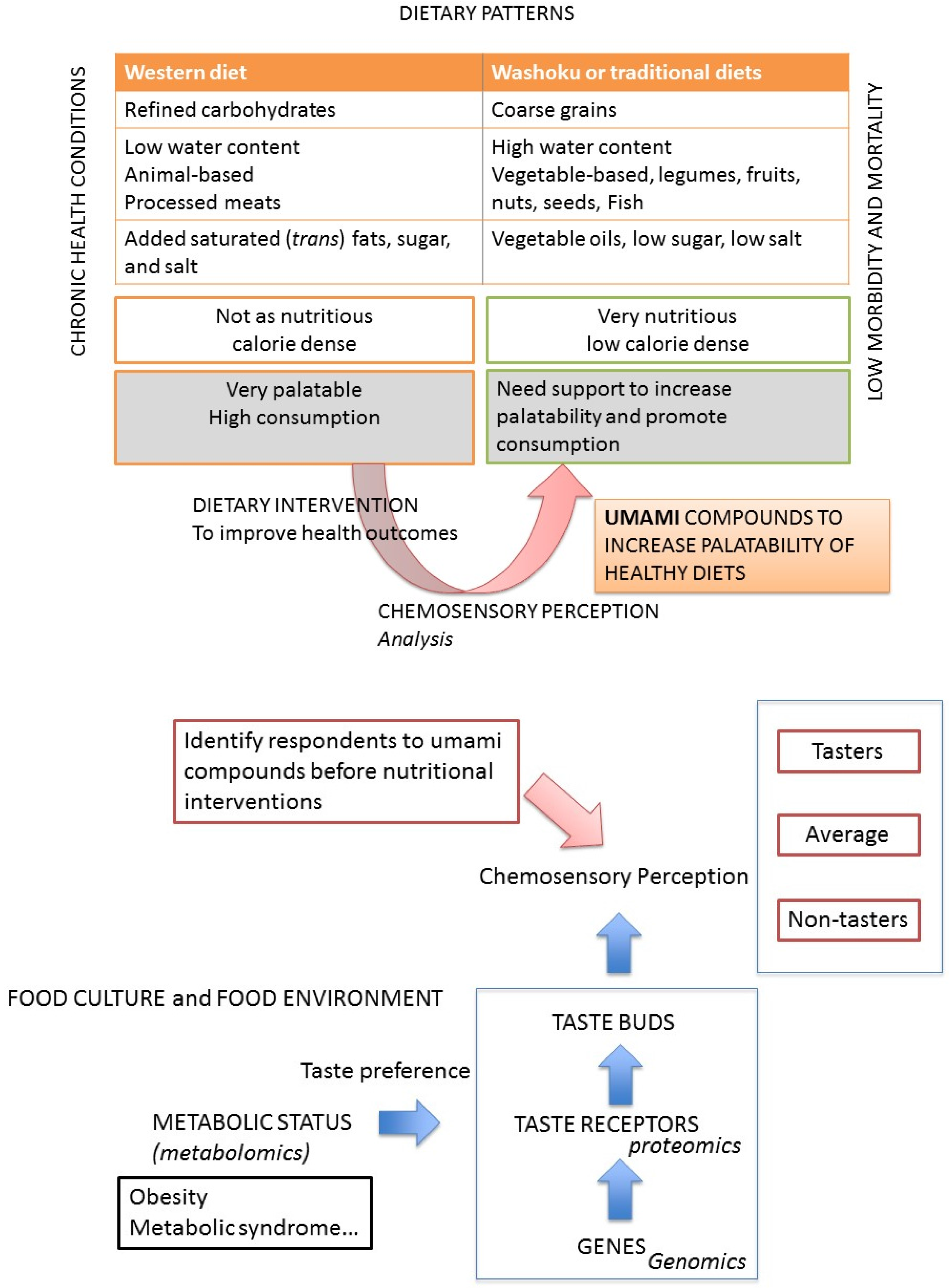
:1. The Traditional Japanese Diet and Its Potential Health Benefits
1.1. The Importance of Umami Taste in Foods and Its Application
1.2. How Does Umami Enhances the Palatability of Foods?
1.3. The Traditional Japanese Cuisine, Washoku: Why Is It Thought to Be Healthy?
2. Food Polyphenols and Their Sensory Properties
3. Why Taste Matters?
3.1. Genetic Variation of Taste Receptors: Bitter Taste
3.2. Other Taste Receptor Variants and Taste Perception
4. Is the Current Diet of the Japanese People Healthy?
5. Sustainability of Healthy Diets
6. Summary and Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Bachmanov, A.A.; Beauchamp, G.K. Taste receptor genes. Annu. Rev. Nutr. 2007, 27, 389–414. [Google Scholar] [CrossRef] [PubMed]
- San Gabriel, A.; Uneyama, H.; Yoshie, S.; Torii, K. Clonning and characterization of a novel mGluR1 variant from vallate papillae that functions as a receptor for l-glutamate stimuli. Chem. Senses 2005, 30 (Suppl. 1), i25–i26. [Google Scholar] [CrossRef] [PubMed]
- Yasuo, T.; Kusuhara, Y.; Yasumatsu, K.; Ninomiya, Y. Multiple receptor systems for glutamate detection in the taste organ. Biol. Pharm. Bull. 2008, 31, 1833–1837. [Google Scholar] [CrossRef] [PubMed]
- Lindemann, B.; Ogiwara, Y.; Ninomiya, Y. The Discovery of umami. Chem. Senses 2002, 27, 843–844. [Google Scholar] [CrossRef] [PubMed]
- Vazquez, M.; Pearson, P.B.; Beauchamp, G.K. Flavor preferences in malnourished Mexican infants. Physiol. Behav. 1982, 28, 513–519. [Google Scholar] [CrossRef]
- Masic, U.; Yeomans, M.R. Does acute or habitual protein deprivation influence liking for monosodium glutamate. Physiol. Behav. 2017, 15, 79–86. [Google Scholar] [CrossRef] [PubMed]
- Pepino, M.Y.; Finkbeiner, S.; Beauchamp, G.K.; Mennella, J.A. Obese women have lower monosodium glutamate taste sensitivity and prefer higher concentrations than do normal-weight women. Obesity 2010, 18, 959–965. [Google Scholar] [CrossRef] [PubMed]
- Yamaguchi, S.; Ninomiya, K. Umami and food palatability. J. Nutr. 2000, 130 (4S Suppl.), 921S–926S. [Google Scholar] [CrossRef] [PubMed]
- Kumakura, I.; Japanese Culinary Academy. What is Japanese Cuisine. In Introduction to Japanese Cuisine, Nature, History and Culture; Kiyota Junji, Shuhari Initiative: Tokyo, Japan, 2015. [Google Scholar]
- Ninomiya, K. Science of umami taste: Adaptation to gastronomic culture. Flavour 2015, 4, 1–5. [Google Scholar] [CrossRef]
- Steiner, J. Umami: A Basic Taste; Kawamura, Y., Kare, M.R., Eds.; Marcel Dekker: New York, NY, USA, 1987; pp. 97–123. [Google Scholar]
- Beauchamp, G.K.; Pearson, P. Human development and umami taste. Physiol. Behav. 1991, 49, 1009–1012. [Google Scholar] [CrossRef]
- Prescott, J. Effects of added glutamate on liking for novel food flavors. Appetite 2004, 42, 143–150. [Google Scholar] [CrossRef] [PubMed]
- Yamaguchi, S.; Takahashi, C. Interactions of monosodium glutamate and sodium chloride on saltiness and palatability. J. Food Sci. 1984, 49, 82–85. [Google Scholar] [CrossRef]
- Roininen, K.; Lahteenmaki, L.; Tuorila, H. Effect of umami taste on pleasantness of low-salt soups during repeated testing. Physiol. Behav. 1996, 60, 953–958. [Google Scholar] [CrossRef]
- Ball, P.; Woodward, D.; Beard, T.; Shoobridge, A.; Ferrier, M. Calcium diglutamate improves taste characteristics of lower-salt soup. Eur. J. Clin. Nutr. 2002, 56, 519–523. [Google Scholar] [CrossRef] [PubMed]
- Carter, B.E.; Monsivais, P.; Drewnowski, A. The sensory optimum of chicken broths supplemented with calcium di-glutamate: A possibility for reducing sodium while maintaining taste. Food Qual. Prefer. 2011, 22, 699–703. [Google Scholar] [CrossRef]
- Leong, J.; Kasamatsu, C.; Ong, E.; Hoi, J.T.; Loong, M.N. A study on sensory properties of sodium reduction and replacement in Asian food using difference-from-control test. Food Sic. Nutr. 2015, 4, 469–478. [Google Scholar] [CrossRef] [PubMed]
- Prescott, J.; Young, A. Does information about MSG (monosodium glutamate) content influence consumer ratings of soups with and without added MSG? Appetite 2002, 39, 25–33. [Google Scholar] [CrossRef] [PubMed]
- Bellisle, F.; Monneuse, M.O.; Chabert, M.; Laure-Achagiotis, C.; Lanteaume, M.T.; Louis-Sylvestre, J. Monosodium glutamate as a palatability enhancer in the European diet. Physiol. Behav. 1991, 49, 869–873. [Google Scholar] [CrossRef]
- Yamamoto, S.; Tomoe, M.; Toyama, K.; Kawai, M.; Uneyama, H. Can dietary supplementation of monosodium glutamate improve the health of the elderly? Am. J. Clin. Nutr. 2009, 90, 844S–849S. [Google Scholar] [CrossRef] [PubMed]
- Beyreuther, K.; Biesalski, H.K.; Fernstrom, J.D.; Grimm, P.; Hammes, W.P.; Heinemann, U.; Kempski, O.; Stehle, P.; Steinhart, H.; Walker, R. Consensus meeting: Monosodium glutamate—And update. Eur. J. Clin. Nutr. 2007, 61, 304–313. [Google Scholar] [CrossRef] [PubMed]
- Henry-Unaeze, H.N. Update on food safety of monosodium l-glutamate (MSG). Pathophysiology 2017, 24, 243–249. [Google Scholar] [CrossRef] [PubMed]
- Okiyama, A.; Beuchamp, G.K. Taste dimensions of monosodium glutamate (MSG) in a food system: Role of glutamate in young American subjects. Physiol. Behav. 1998, 65, 177–181. [Google Scholar] [CrossRef]
- Wilkie, L.M.; Capaldi Phillips, E.D. Heterogeneous binary interactions of taste primaries: Perceptual outcomes, physiology, and future directions. Neurosci. Biobehav. Rev. 2014, 47, 70–86. [Google Scholar] [CrossRef] [PubMed]
- Kawasaki, H.; Sekizaki, Y.; Hirota, M.; Sekine-Hayakawa, Y.; Nonaka, M. Analysis of binary taste-taste interaction of MSG, lactic acid, and NaCl by temporal dominance of sensations. Food Qual. Pref. 2016, 52, 1–10. [Google Scholar] [CrossRef]
- Hodson, N.A.; Linden, R.W. The effect of monosodium glutamate on parotid salivary flow in comparison to response to representatives of the other four basic tastes. Physiol. Behav. 2006, 89, 711–717. [Google Scholar] [CrossRef] [PubMed]
- Sasano, T.; Satoh-Kuriwada, S.; Shoji, N.; Sekine-Hayakawa, Y.; Kawaki, M.; Uneyama, H. Application of umami taste stimulation to remedy hypogeusia based on reflex salivation. Biol. Pharm. Bull. 2010, 33, 1791–1795. [Google Scholar] [CrossRef] [PubMed]
- Mese, H.; Matsuo, R. Salivary secretion, taste and hyposalivation. J. Oral Rehabil. 2007, 34, 711–723. [Google Scholar] [CrossRef] [PubMed]
- Sasano, T.; Satoh-Kuriwada, S.; Shoji, N.; Iikubo, M.; Kawai, M.; Uneyama, H.; Sakamoto, M. Important role of umami taste sensitivity in oral and overall health. Curr. Pharm. Des. 2014, 20, 2750–2754. [Google Scholar] [CrossRef] [PubMed]
- San Gabriel, A.M.; Maekawa, T.; Uneyama, H.; Yoshie, S.; Torii, K. mGluR1 in the fundic glands of rat stomach. FEBS Lett. 2007, 582, 1119–1123. [Google Scholar] [CrossRef] [PubMed]
- Young, R.L.; Sutherland, K.; Pezos, N.; Brierley, S.M.; Horowitz, M.; Rayner, C.K.; Blackshaw, L.A. Expression of taste molecules in the upper gastrointestinal tract in humans with and without type 2 diabetes. Gut 2009, 58, 337–346. [Google Scholar] [CrossRef] [PubMed]
- Uneyama, H.; Niijima, A.; San Gabriel, A.; Torii, K. Luminal amino acid sensing in the rat gastric mucosa. Am. J. Physiol. Gastrointest. Liver Physiol. 2006, 291, G1163–G1170. [Google Scholar] [CrossRef] [PubMed]
- San Gabriel, A.; Uneyama, H. Amino acid sensing in the gastrointestinal tract. Amino Acids 2013, 45, 451–461. [Google Scholar] [CrossRef] [PubMed]
- Nishimura, T.; Goto, S.; Miura, K.; Takakura, Y.; Egusa, A.S.; Wakabayashi, H. Umami compounds enhance the intensity of retronasal sensation of aroma from model chicken soups. Food Chem. 2016, 196, 577–583. [Google Scholar] [CrossRef] [PubMed]
- Prescott, J. Taste hedonics and the role of umami. Food Aust. 2001, 53, 550–554. [Google Scholar]
- Brondel, L.; Lauraine, G.; Van Wymerbeke, V.; Romer, M.; Schaal, B. Alternation between foods within a meal. Influence on satiation and consumption in humans. Appetite 2009, 53, 203–209. [Google Scholar] [CrossRef] [PubMed]
- Ello-Martin, J.A.; Ledikwe, J.H.; Rolls, B.J. The influence of food portion size and energy density on energy intake: Implications for weight management. Am. J. Clin. Nutr. 2005, 82 (1 Suppl.), 236S–241S. [Google Scholar] [PubMed]
- Brunstrom, J.M.; Jarvstad, A.; Griggs, R.L.; Potter, C.; Evans, N.R.; Martin, A.A.; Brooks, J.C.; Rogers, P.J. Large portions encourage the selection of palatable rather than filling foods. J. Nutr. 2016, 146, 2117–2123. [Google Scholar] [CrossRef] [PubMed]
- Kuroda, M.; Ohta, M.; Okufuji, T.; Takigami, C.; Eguchi, M.; Hayabuchi, H.; Ikeda, M. Frequency of soup intake is inversely associated with body mass index, waist circumference, and waist-to-hip ratio, but not with other metabolic risk factors in Japanese men. J. Am. Diet. Assoc. 2011, 111, 137–142. [Google Scholar] [CrossRef] [PubMed]
- Rolls, B.J.; Fedoroff, I.C.; Guthrie, J.F.; Laster, L.J. Foods with different satiating effects in humans. Appetite 1990, 15, 115–126. [Google Scholar] [CrossRef]
- Clegg, M.E.; Ranawana, V.; Shafat, A.; Henry, C.J. Soups increase satiety through delayed gastric emptying yet increased glycaemic response. Eur. J. Clin. Nutr. 2013, 67, 8–11. [Google Scholar] [CrossRef] [PubMed]
- Bazzano, L.A.; He, J.; Ogden, L.G.; Loria, C.M.; Vupputuri, S.; Myers, L.; Whelton, P.K. Fruit and vegetable intake and risk of cardiovascular diseases in US adults: The first national health and nutrition examination survey epidemiologic follow-up study. Am. J. Clin. Nutr. 2002, 76, 93–99. [Google Scholar] [CrossRef] [PubMed]
- Rolls, B.J.; Bell, E.A.; Thorwart, M.L. Water incorporated into a food but not served with a food decreases energy intake in lean women. Am. J. Clin. Nutr. 1999, 70, 448–455. [Google Scholar] [PubMed]
- Calder, P.C. Very long-chain n-3 fatty acids and human health: Fact, fiction and the future. Proc. Nutr. Soc. 2017, 17, 1–21. [Google Scholar] [CrossRef] [PubMed]
- Rivas, M.; Garay, R.P.; Escanero, J.F.; Cia, P., Jr.; Cia, P.; Alda, J.O. Soy milk lowers blood pressure in men and women with mild to moderate essential hypertension. J. Nutr. 2002, 132, 1900–1902. [Google Scholar] [CrossRef] [PubMed]
- Jayagopal, V.; Albertazzi, P.; Kilpatrick, E.S.; Howarth, E.M.; Jennings, P.E.; Hepburn, D.A.; Atkin, S.L. Beneficial effects of soy phytoestrogen intake in postmenopausal women with type 2 diabetes. Diabetes Care 2002, 25, 1709–1714. [Google Scholar] [CrossRef] [PubMed]
- Zhou, B.F.; Stamler, J.; Dennis, B.; Moag-Stahlberg, A.; Okuda, N.; Robertson, C.; Zhao, L.; Chan, Q.; Elliott, P.; INTERMAP Research Group. Nutrient intakes of middle-age men and women in China, Japan, United Kingdom, and United States in the late 1990s: The INTERMAP study. J. Hum. Hypertens. 2003, 17, 623–630. [Google Scholar] [CrossRef] [PubMed]
- Zhang, R.; Wang, Z.; Fei, Y.; Zhou, B.; Zheng, S.; Wang, L.; Huang, L.; Jiang, S.; Liu, Z.; Jiang, J.; et al. The difference in nutrient intakes between Chinese and Mediterranean, Japanese and American diets. Nutrients 2015, 7, 4661–4688. [Google Scholar] [CrossRef] [PubMed]
- Ando, K.; Kawarazaki, H.; Miura, K.; Matsuura, H.; Watanabe, Y.; Yoshita, K.; Kawamura, M.; Kusaka, M.; Kai, H.; Tsuchihashi, T.; et al. [Scientific statement] Report of the Salt Reduction Committee of the Japanese Society of Hypertension (1) Role of salt in hypertension and cardiovascular diseases. Hypertens. Res. 2013, 36, 1009–1019. [Google Scholar] [CrossRef] [PubMed]
- Wakasugi, M.; Kazama, J.; Narita, I. Associations between the intake of miso soup and Japanese pickles and the estimated 24-hour unrinary sodium excretion: A population-based cross-sectional study. Intern. Med. 2015, 54, 903–910. [Google Scholar] [CrossRef] [PubMed]
- Anderson, C.A.; Appel, L.J.; Okuda, N.; Brown, I.J.; Chang, Q.; Zhao, L.; Ueshima, H.; Kesteloot, H.; Miura, K.; Curb, J.D.; et al. Dietary sources of sodium in Chaina, Japan, the United Kingdom, and the United States, women and men aged 40 to 59 years: The INTERMAP study. J. Am. Diet. Assoc. 2010, 110, 736–745. [Google Scholar] [CrossRef] [PubMed]
- Ministry of Agriculture, Forestry and Fisheries. Traditional Dietary Cultures of the Japanese. Available online: http://www.maff.go.jp/e/japan_food/washoku/pdf/wasyoku_english.pdf (accessed on 31 December 2013).
- Lesschaeve, I.; Noble, A.C. Polyphenols: Factors influencing their sensory properties and their effects on food and beverage preferences. Am. J. Clin. Nutr. 2005, 81 (1 Suppl.), 330S–335S. [Google Scholar] [PubMed]
- Lee, C.A.; Ismail, B.; Vickers, Z.M. The role of salivary proteins in the mechanism of astringency. J. Food Sci. 2012, 77, C381–C387. [Google Scholar] [CrossRef] [PubMed]
- Breslin, P.A.S.; Gilmore, M.M.; Beauchamp, G.K.; Green, B.G. Psychophysical evidence that oral astringency is a tactile sensation. Chem. Senses 1993, 18, 405–417. [Google Scholar] [CrossRef]
- Gibbins, H.L.; Carpenter, G.H. Alternative mechanisms of astringency—What is the role of saliva? J. Texture Stud. 2013, 44, 364–375. [Google Scholar] [CrossRef]
- Cicerale, S.; Breslin, P.A.; Beauchamp, G.K.; Keast, R.S. Sensory characterization of the irritant properties of oleocanthal, a natural anti-inflammatory agent in extra virgin olive oils. Chem. Senses 2009, 34, 333–339. [Google Scholar] [CrossRef] [PubMed]
- Carluccio, M.A.; Siculella, L.; Ancora, M.A.; Massaro, M.; Scoditti, E.; Storelli, C.; Visioli, F.; Distante, A.; De Caterina, R. Olive oil and red wine antioxidant polyphenols inhibit endothelial activation: Antiatherogenic properties of Mediterranean diet phytochemicals. Arterioscler. Thromb. Vasc. Biol. 2003, 23, 622–629. [Google Scholar] [CrossRef] [PubMed]
- Levine, J.D.; Alessandri-Haber, N. TRP channels: Targets for the relief of pain. Biochim. Biophys. Acta 2007, 1772, 989–1003. [Google Scholar] [CrossRef] [PubMed]
- Peyrot des Gachons, C.; Uchida, K.; Bryant, B.; Shima, A.; Sperry, J.B.; Dankulich-Nagrudny, L.; Tominaga, M.; Smith, A.B., 3rd; Beauchamp, G.K.; Breslin, P.A. Unusual pungency from extra-virgin olive oil is attributable to restricted spatial expression of the receptor of oleocanthal. J. Neurosci. 2011, 31, 999–1009. [Google Scholar] [CrossRef] [PubMed]
- Kim, U.K.; Breslin, P.A.; Reed, D.; Drayna, D. Genetics of human taste perception. J. Dent. Res. 2004, 83, 448–453. [Google Scholar] [CrossRef] [PubMed]
- Tepper, B.J. Nutritional implications of genetic variation: The role of PROP sensitivity and other taste phenotypes. Ann. Rev. Nutr. 2008, 28, 367–388. [Google Scholar] [CrossRef] [PubMed]
- Chamoun, E.; Mutch, D.M.; Allen-Vercoe, E.; Buchholz, A.C.; Duncan, A.M.; Spriet, L.L.; Haines, J.; Ma, D.W.L.; Guelph Family Health Study. A review of the associations between single nucleotide polymorphisms in taste receptors, eating behaviors, and health. Crit. Rev. Food Sci. Nutr. 2016, 31, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Connors, M.; Bisogni, C.A.; Sobal, J.; Devine, C.M. Managing values in personal food systems. Appetite 2001, 36, 189–200. [Google Scholar] [CrossRef] [PubMed]
- Tepper, B.J.; White, E.A.; Koelliker, Y.; Lanzara, C.; d’Adamo, P.; Gasparini, P. Genetic variation in taste sensitivity to 6-n-propylthiouracil and its relationship to taste perception and food selection. Ann. N. Y. Acad. Sci. 2009, 1170, 126–139. [Google Scholar] [CrossRef] [PubMed]
- Hayes, J.E.; Sullivan, B.S.; Duffy, V.B. Explaining variability in sodium intake through oral sensory phenotype, salt and liking. Physiol. Behav. 2010, 100, 369–380. [Google Scholar] [CrossRef] [PubMed]
- Liang, L.C.; Sakimura, J.; May, D.; Breen, C.; Driggin, E.; Tepper, B.J.; Chung, W.K.; Keller, K.L. Fat discrimination: A phenotype with potential implications for studying fat intake behaviors and obesity. Physiol. Behav. 2012, 105, 470–475. [Google Scholar] [CrossRef] [PubMed]
- Feeney, E.; O’Brien, S.; Scannell, A.; Markey, A.; Gibney, E.R. Genetic variation in taste perception: Does it have a role in healthy eating? Proc. Nutr. Soc. 2011, 70, 135–143. [Google Scholar] [CrossRef] [PubMed]
- Graaf, C. Texture and satiation: The role of oro-sensory exposure time. Physiol. Behav. 2012, 107, 496–501. [Google Scholar] [CrossRef] [PubMed]
- Behrens, M.; Reichling, C.; Batram, C.; Brockhoff, A.; Meyerhof, W. Bitter taste receptors and their cells. Ann. N. Y. Acad. Sci. 2009, 1170, 111–115. [Google Scholar] [CrossRef] [PubMed]
- Blakeslee, A.; Fox, A. Our different taste worlds. J. Hered. 1932, 23, 97–107. [Google Scholar] [CrossRef]
- Bartoshuk, L.M. Bitter taste of saccharin related to the genetic ability to taste the bitter substance 6-n-propylthiouracil. Science 1979, 205, 934–935. [Google Scholar] [CrossRef] [PubMed]
- Keller, K.L.; Adise, S. Variation in the ability to taste bitter thiourea compounds: Implications for food acceptance, dietary intake, and obesity risk in children. Annu. Rev. Nutr. 2016, 36, 157–182. [Google Scholar] [CrossRef] [PubMed]
- Lipchock, S.; Mennella, J.; Spielman, A.; Reed, D. Human bitter perception correlates with bitter receptor messenger RNA expression in taste cells. Am. J. Clin. Nutr. 2013, 98, 1136–1143. [Google Scholar] [CrossRef] [PubMed]
- Running, C.A.; Carig, B.A.; Mattes, R.D. Oleogustus: The unique taste of fat. Chem. Senses 2015, 40, 507–516. [Google Scholar] [CrossRef] [PubMed]
- Stewart, J.E.; Feinle-Bisset, C.; Golding, M.; Delahunty, C.; Clifton, P.M.; Keast, R.S. Oral sensitivity to fatty acids, food consumption and BMI in human subjects. Br. J. Nutr. 2010, 104, 145–152. [Google Scholar] [CrossRef] [PubMed]
- Tucker, R.M.; Matter, R.D. Influences of repeated testing on nonesterified fatty acid taste. Chem. Senses 2013, 38, 325–332. [Google Scholar] [CrossRef] [PubMed]
- Martinez-Ruiz, N.R.; Lopez-Diaz, J.A.; Wall-Medrano, A.; Jimenez-Castro, J.A.; Angulo, O. Oral fat perception is related with body mass index, preference and consumption of high-fat foods. Physiol. Behav. 2014, 129, 36–42. [Google Scholar] [CrossRef] [PubMed]
- Pepino, M.Y.; Love-Gregory, L.; Klein, S.; Abumrad, N.A. The fatty acid translocase gene CD36 and lingual lipase influence oral sensitivity to fat in obese subjects. J. Lipid Res. 2012, 53, 561–566. [Google Scholar] [CrossRef] [PubMed]
- Stewart, J.E.; Seimon, R.V.; Otto, B.; Keast, R.S.; Clifton, P.M.; Feinle-Bisset, C. Marked differences in gustatory and gastrointestinal sensitivity to oleic acid between lean and obese men. Am. J. Clin. Nutr. 2011, 93, 703–711. [Google Scholar] [CrossRef] [PubMed]
- Keller, K.L.; Liang, L.C.; Sakimura, J.; May, D.; van Belle, C.; Breen, C.; Driggin, E.; Tepper, B.J.; Deng, L.; Chung, W.K. Common variants in the CD36 gene are associated with oral fat perception, fat preferences, and obesity in African Americans. Obesity 2012, 20, 1066–1073. [Google Scholar] [CrossRef] [PubMed]
- Tucker, R.M.; Kaiser, K.A.; Parman, M.A.; George, B.J.; Allison, D.B.; Mattes, R.D. Comparison of fatty acid taste detection thresholds in people who are lean vs. overweight or obese: A systematic review and meta-analysis. PLoS ONE 2017, 12, e0169583. [Google Scholar] [CrossRef] [PubMed]
- Kim, U.K.; Wooding, S.; Riaz, N.; Jorde, L.B.; Drayna, D. Variation in the human TAS1R taste receptor genes. Chem. Senses 2006, 31, 599–611. [Google Scholar] [CrossRef] [PubMed]
- Reed, D.R.; Bachmanov, A.A.; Beauchamp, G.K.; Tordoff, M.G.; Price, R.A. Heritable variation in food preferences and their contribution to obesity. Behav. Genet. 1997, 27, 373–387. [Google Scholar] [CrossRef] [PubMed]
- Fushan, A.A.; Simons, C.T.; Slack, J.P.; Manichaikul, A.; Drayna, D. Allelic polymorphism within the TAS1R3 promoter is associated with human taste sensitivity to sucrose. Curr. Biol. 2009, 19, 1288–1293. [Google Scholar] [CrossRef] [PubMed]
- Kulkarni, G.V.; Chng, T.; Eny, K.M.; Nielsen, D.; Wessman, C.; El-Sohemy, A. Association of GLUT2 and TAS1R2 genotypes with risk for dental caries. Caries Res. 2013, 47, 219–225. [Google Scholar] [CrossRef] [PubMed]
- Lugaz, O.; Pillias, A.M.; Faurion, A. A new specific ageusia: Some humans cannot taste L-glutamate. Chem. Senses 2002, 27, 105–115. [Google Scholar] [CrossRef]
- Shigemura, N.; Shirosaki, S.; Sanematsu, K.; Yoshida, R.; Ninomiya, Y. Genetic and molecular basis of individual differences in human umami taste. PLoS ONE 2009, 4, e6717. [Google Scholar] [CrossRef] [PubMed]
- Institute of Medicine (IOM). Sodium Intake in Populations: Assessment of Evidence; The National Academies Press: Washington, DC, USA, 2013. [Google Scholar]
- Yoshida, R.; Horio, N.; Murata, Y.; Yasumatsu, K.; Shigemura, N.; Ninomiya, Y. NaCl responsive taste cells in the mouse fungiform taste buds. Nueroscience 2009, 159, 795–803. [Google Scholar] [CrossRef] [PubMed]
- Dias, A.G.; Rousseau, D.; Duizer, L.; Cockburn, M.; Chiu, W.; Nielsen, D.; El-Sohemy, A. Genetic variation in putative salt taste receptors and salt taste perception in humans. Chem. Senses 2013, 38, 137–145. [Google Scholar] [CrossRef] [PubMed]
- Kurotani, K.; Akter, S.; Kashino, I.; Goto, A.; Mizoue, T.; Noda, M.; Sasazuki, S.; Sawada, N.; Tsugane, S.; Japan Public Health Center based Prospective Study Group. Quality of diet and mortality among Japanese men and women: Japan Public health center prospective study. BMJ 2016, 352, i1209. [Google Scholar] [CrossRef] [PubMed]
- Ikeda, N.; Saito, E.; Kondo, N.; Inoue, M.; Ikeda, S.; Satoh, T.; Wada, K.; Stickley, A.; Katanoda, K.; Mizoue, T.; et al. What has made the population of Japan healthy? Lancet 2011, 378, 1094–1105. [Google Scholar] [CrossRef]
- Nakaji, S.; MacAuley, D.; O’Neill, S.; McNally, O.; Baxter, D.; Sugawara, K. Life expectancies in the United Kingdom and Japan. J. Public Health Med. 2003, 25, 120–124. [Google Scholar] [CrossRef] [PubMed]
- Shimazu, T.; Kuriyama, S.; Hozawa, A.; Ohmori, K.; Sato, Y.; Nakaya, N.; Nishino, Y.; Tsubono, Y.; Tsuji, I. Dietary patterns and cardiovascular disease mortality in Japan: A prospective cohort study. Int. J. Epidemiol. 2007, 63, 600–609. [Google Scholar] [CrossRef] [PubMed]
- Lands, W.E.; Hamazaki, T.; Yamazaki, K.; Okuyama, H.; Sakai, K.; Goto, Y.; Hubbard, V.S. Changing dietary patterns. Am. J. Clin. Nutr. 1990, 51, 991–993. [Google Scholar] [CrossRef] [PubMed]
- Yoshiike, N.; Hayashi, F.; Takemi, Y.; Mizoguchi, K.; Seino, F. A new food guide in Japan: The Japanese food guide Spinning Top. Nutr. Rev. 2007, 65, 149–154. [Google Scholar] [CrossRef] [PubMed]
- Miyoshi, M.; Tsuboyama-Kasaoka, N.; Nishi, N. School-based “Shokuiku” program in Japan: Application to nutrition education in Asian countries. Asia Pac. J. Clin. Nutr. 2012, 21, 159–162. [Google Scholar] [PubMed]
- Kuriyama, N.; Murakami, K.; Livingstone, M.B.E.; Okubo, H.; Kobayashi, S.; Suga, H.; Sasaki, S.; Three-Generation Study of Women on Diets and Health Study Group. Development of a food-based diet quality score for Japanese: Associations of the score with nutrient intakes in young, middle-age and older Japanese women. J. Nutr. Sci. 2016, 5, e41. [Google Scholar] [CrossRef] [PubMed]
- Food and Agriculture Organization of the United Nations. Statistics Yearbook (Food Balance Sheet for the Data of Japan). Available online: http://faostat.fao.org/static/syb/syb_110.pdf (accessed on 31 December 2017).
- Ministry of Agriculture, Forestry and Fisheries (MAFF) of Japan. Relationship between Diet, Health, and Life Expectancy. Available online: http://www.maff.go.jp/j/keikaku/syokubunka/culture/eiyo.html (accessed on 31 December 2008). (In Japanese)
- Food and Agriculture Organization of the United Nations. International Scientific Symposium. Biodiversity and Sustainable Diets—United against Hunger. Available online: http://www.fao.org/docrep/016/i3004e/i3004e.pdf (accessed on 31 December 2012).
- Donini, L.M.; Dernini, S.; Lairon, D.; Serra-Majem, L.; Amiot, M.J.; Del Balzo, V.; Giusti, A.M.; Burlingame, B.; Belahsen, R.; Maiani, G.; et al. A consensus proposal for nutritional indicators to assess the sustainability of a healthy diet: The Mediterranean diet as a case study. Food Nutr. 2016, 3, 37. [Google Scholar] [CrossRef] [PubMed]
- Tilman, D.; Clark, M. Global diets link environmental sustainability and human health. Nature 2014, 515, 518–522. [Google Scholar] [CrossRef] [PubMed]

| Dishes | Ingredients | Elements |
|---|---|---|
| Staple food | Grains, mainly rice (noodles or glutinous rice) | Recipes with cooked rice (sushi or curry rice) |
| Soup | Miso Soup (seaweed, shellfish, vegetables) | Dashi soup stock (fermented soybeans) |
| Main dish | Fish, seafood, sometimes meats | Great variety of edible fishes |
| Side dishes | Vegetables, wild plants, mushrooms, seaweed, shellfish | Change with the season and locality |
| Elements | Contents | Description |
|---|---|---|
| Foodstuffs | Seasonal foods | Rice, vegetables, wild plants, mushrooms, variety of fish |
| Dishes | Cooking methods with abundant water, dashi stock, delicious meals, with vegetables and seafood | Steaming, boiling, and stewing |
| Nutrition | Relative low-calorie density, low total fat, high quality protein, variety of ingredients, easy to eat different nutrients | Nutritionally well-balanced |
| Hospitality | Health and family ties | The joy of eating together and caring for one another |
| Element | Effect | Health Consequences |
|---|---|---|
| Small portion size | Smaller meal size | Prevents overeating [38,39]. |
| Soup and dishes with high water content | Lower total energy intake | Lower 1 BMI, waist circumference and waist-to-hip ratio [40,41,42,48,49]. |
| Soy sauce, salted vegetables and fruits, miso soup, and salted fish | High sodium consumption, with a high sodium/potassium ratio | The high vegetable intake seems to protect against CVD [50,51,52]. |
| High 2 EPA and 3 DHA, low animal fat, low total fat | Modulation of the membranes of cells, lipid signaling and gene expression | Supports optimal health, low risk of 1,4 CVD, cancer and inflammation [45] |
| Foods based on beans | Decrease blood pressure and blood glucose | Protects against CVD [46,47] |
| Variety of seasonal vegetables and green tea | Intestinal bulk and protection against inflammation and high blood pressure | Low risk of CVD and all causes of mortality [43]. |
| Umami taste | Enhances flavor, food palatability and salivation | Promotes chewing and swallowing, and maintains adequate taste sensation [27,28,30]. |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gabriel, A.S.; Ninomiya, K.; Uneyama, H. The Role of the Japanese Traditional Diet in Healthy and Sustainable Dietary Patterns around the World. Nutrients 2018, 10, 173. https://doi.org/10.3390/nu10020173
Gabriel AS, Ninomiya K, Uneyama H. The Role of the Japanese Traditional Diet in Healthy and Sustainable Dietary Patterns around the World. Nutrients. 2018; 10(2):173. https://doi.org/10.3390/nu10020173
Chicago/Turabian StyleGabriel, Ana San, Kumiko Ninomiya, and Hisayuki Uneyama. 2018. "The Role of the Japanese Traditional Diet in Healthy and Sustainable Dietary Patterns around the World" Nutrients 10, no. 2: 173. https://doi.org/10.3390/nu10020173
APA StyleGabriel, A. S., Ninomiya, K., & Uneyama, H. (2018). The Role of the Japanese Traditional Diet in Healthy and Sustainable Dietary Patterns around the World. Nutrients, 10(2), 173. https://doi.org/10.3390/nu10020173




