Commencing Nutrient Supplements before Full Enteral Feed Volume Achievement Is Beneficial for Moderately Preterm to Late Preterm Low Birth Weight Babies: A Prospective, Observational Study
Abstract
:1. Introduction
2. Materials and Methods
2.1. Overview
2.2. Participation
2.3. Outcomes and Feeding Intolerance
2.4. Feeding Protocol
2.5. Data Collection and Calculation Assumptions
2.6. Statistical Methods
3. Results
3.1. Study Population and Demographic Characteristics
3.2. Anthropometric Findings
3.3. Nutrition and Feeding Outcomes
3.4. Blood Urea as a Marker of Protein Intake Adequacy
3.5. Clinical Outcomes
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Engle, W.A.; Tomashek, K.M.; Wallman, C. Committee on Fetus and Newborn, American Academy of Pediatrics. “Late-preterm” infants: A population at risk. Pediatrics 2007, 120, 1390–1401. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.L.; Dorer, D.J.; Fleming, M.P.; Catlin, E.A. Clinical outcomes of near-term infants. Pediatrics 2004, 114, 372–376. [Google Scholar] [CrossRef] [PubMed]
- Escobar, G.J.; Gonzales, V.M.; Armstrong, M.A.; Folck, B.F.; Xiong, B.; Newman, T.B. Rehospitalization for neonatal dehydration: A nested case-control study. Arch. Pediatr. Adolesc. Med. 2002, 156, 155–161. [Google Scholar] [CrossRef] [PubMed]
- Gewolb, I.H.; Vice, F.L. Maturational changes in the rhythms, patterning, and coordination of respiration and swallow during feeding in preterm and term infants. Dev. Med. Child Neurol. 2006, 48, 589–594. [Google Scholar] [CrossRef] [PubMed]
- Adamkin, D.H. Feeding problems in the late preterm infant. Clin. Perinatol. 2006, 33, 831–837. [Google Scholar] [CrossRef] [PubMed]
- Whyte, R.K. Canadian Paediatric Society, Fetus and Newborn Committee. Safe discharge of the late preterm infant. Paediatr. Child Health 2010, 15, 655–660. [Google Scholar] [CrossRef] [PubMed]
- Canberra Hospital and Health Services, Clinical Guideline, Neonatal Nutrition Guide. Available online: http://health.act.gov.au/research-data-and-publications/policy-and-plans-0 (accessed on 7 July 2018).
- Thoene, M.; Hanson, C.; Lyden, E.; Dugick, L.; Ruybal, L.; Anderson-Berry, A. Comparison of the Effect of Two Human Milk Fortifiers on Clinical Outcomes in Premature Infants. Nutrients 2014, 6, 261–275. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Valley, M.A.; Heard, K.J.; Ginde, A.A.; Lezotte, D.C.; Lowenstein, S.R. Observational studies of patients in the emergency department: A comparison of 4 sampling methods. Ann. Emerg. Med. 2012, 60, 139–145. [Google Scholar] [CrossRef] [PubMed]
- Yang, W.; Zilov, A.; Soewondo, P.; Bech, O.M.; Sekkal, F.; Home, P.D. Observational studies: Going beyond the boundaries of randomized controlled trials. Diabetes Res. Clin. Pract. 2010, 88, S3–S9. [Google Scholar] [CrossRef]
- Gregory, K.E.; Deforge, C.E.; Natale, K.M.; Phillips, M.; Van Marter, L.J. Necrotizing enterocolitis in the premature infant: Neonatal nursing assessment, disease pathogenesis, and clinical presentation. Adv. Neonatal Care 2011, 11, 155–164. [Google Scholar] [CrossRef] [PubMed]
- Fenton, T.R.; Kim, J.H. A systematic review and meta-analysis to revise the Fenton growth chart for preterm infants. BMC Pediatr. 2013, 13, 59. [Google Scholar] [CrossRef] [PubMed]
- Bauer, J.; Gerss, J. Longitudinal analysis of macronutrients and minerals in human milk produced by mothers of preterm infants. Clin. Nutr. 2011, 30, 215–220. [Google Scholar] [CrossRef] [PubMed]
- Arslanoglu, S.; Moro, G.E.; Ziegler, E.E. Adjustable fortification of human milk fed to preterm infants: Does it make a difference? J. Perinatol. 2006, 26, 614–621. [Google Scholar] [CrossRef] [PubMed]
- Bueva, A.; Guignard, J.P. Renal Function in Preterm Neonates. Pediatr. Res. 1994, 36, 572–577. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Moro, G.E.; Minoli, I.; Ostrom, M.; Jacobs, J.R. Fortification of human milk: Evaluation of a novel fortification scheme and of a new fortifier. J. Pediatr. Gastroenterol. Nutr. 1995, 20, 162–172. [Google Scholar] [CrossRef] [PubMed]
- Polberger, S.K.; Axelsson, I.E.; Räihä, N.C. Urinary and serum urea as indicators of protein metabolism in very low birthweight infants fed varying human milk protein intakes. Acta Paediatr. Scand. 1990, 79, 737–742. [Google Scholar] [CrossRef] [PubMed]
- Villar, J.; Giuliani, F.; Bhutta, Z.A.; Bertino, E.; Ohuma, E.O. Postnatal growth standards for preterm infants: The Preterm Postnatal Follow-up Study of the INTERGROWTH-21(st) Project. Lancet Glob. Health 2015, 3, e681–e691. [Google Scholar] [CrossRef]
- Cooke, R.J. Improving growth in preterm infants during initial hospital stay: Principles into practice. Arch. Dis. Child Fetal Neonatal Ed. 2016, 101, F366–F370. [Google Scholar] [CrossRef] [PubMed]
- Thureen, P.; Heird, W.C. Protein and energy requirements of the preterm/low birthweight (LBW) infant. Pediatr. Res. 2005, 57, 95R–98R. [Google Scholar] [CrossRef] [PubMed]
- Kalhan, S.C. Rates of urea synthesis in the human newborn: Effect of maternal diabetes and small size for gestational age. Pediatr. Res. 1993, 34, 801–804. [Google Scholar] [CrossRef] [PubMed]
- Zeigler, E.E. Meeting the Nutritional Needs of the Low-Birth-Weight Infant. Ann. Nutr. Metab. 2011, 58, 8–18. [Google Scholar] [CrossRef] [PubMed]
- Fanaro, S. Feeding intolerance in the preterm infant. Early Hum. Dev. 2013, 89, S13–S20. [Google Scholar] [CrossRef] [PubMed]
- Indrio, F.; Riezzo, G.; Cavallo, L.; Di Mauro, A.; Francavilla, R. Physiological basis of food intolerance in VLBW. J. Matern. Fetal Neonatal Med. 2011, 24, 64–66. [Google Scholar] [CrossRef] [PubMed]
- Forsgren, M.; Isolauri, E.; Salminen, S.; Rautava, S. Late preterm birth has direct and indirect effects on infant gut microbiota development during the first six months of life. Acta Paediatr. 2017, 106, 1103–1109. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Warner, B.B.; Tarr, P.I. Necrotizing enterocolitis and preterm infant gut bacteria. Semin. Fetal Neonatal Med. 2016, 21, 394–399. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Terrin, G.; Canani, B.C.; Di Chiara, M.; Pietravalle, A.; Aleandri, V.; Conte, F.; De Curtis, M. Zinc in Early Life: A Key Element in the Fetus and Preterm Neonate. Nutrients 2015, 7, 10427–10446. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Starke, I.C.; Pieper, R.; Neumann, K.; Zentek, J.; Vahjen, W. The impact of high dietary zinc oxide on the development of the intestinal microbiota in weaned piglets. FEMS Microbiol. Ecol. 2014, 87, 416–427. [Google Scholar] [CrossRef] [PubMed]
- Sahni, R.; Polin, R.A. Physiologic underpinnings for clinical problems in moderately preterm and late preterm infants. Clin. Perinatol. 2013, 40, 645–663. [Google Scholar] [CrossRef] [PubMed]
- Ou, X.; Glasier, C.M.; Ramakrishnaiah, R.H.; Kanfi, A.; Rowell, A.C.; Pivik, R.T.; Andres, A.; Cleves, M.A.; Badger, T.M. Gestational Age at Birth and Brain White Matter Development in Term-Born Infants and Children. AJNR Am. J. Neuroradiol. 2017, 38, 2373–2379. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dabydeen, L.; Thomas, J.E.; Aston, T.J.; Hartley, H.; Sinha, S.K.; Eyre, J.A. Highenergy and -protein diet increases brain and corticospinal tract growth in term and preterm infants after perinatal brain injury. Pediatrics 2008, 121, 148–156. [Google Scholar] [CrossRef] [PubMed]
- Allin, M.P.; Kontis, D.; Walshe, M.; Wyatt, J.; Barker, G.J.; Kanaan, R.A.; McGuire, P.; Rifkin, L.; Murray, R.M.; Nosarti, C. White matter and cognition in adults who were born preterm. PLoS ONE 2011, 6, e24525. [Google Scholar] [CrossRef] [PubMed]
- Keunen, K.; van Elburg, R.M.; van Bel, F.; Benders, M.J. Impact of nutrition on brain development and its neuroprotective implications following preterm birth. Pediatr. Res. 2015, 77, 148–155. [Google Scholar] [CrossRef] [PubMed]
- Crump, C.; Winkleby, M.A.; Sundquist, K.; Sundquist, J. Risk of hypertension among young adults who were born preterm: A Swedish national study of 636,000 births. Am. J. Epidemiol. 2011, 173, 797–803. [Google Scholar] [CrossRef] [PubMed]
- Crump, C.; Winkleby, M.A.; Sundquist, K.; Sundquist, J. Risk of diabetes among young adults born preterm in Sweden. Diabetes Care 2011, 34, 1109–1113. [Google Scholar] [CrossRef] [PubMed]
- Bloomfield, F.H.; Harding, J.E.; Meyer, M.P.; Alsweiler, J.M.; Jiang, Y.; Wall, C.R.; Alexander, T.; DIAMOND Study Group. The DIAMOND trial—DIfferent Approaches to MOderate & late preterm Nutrition: Determinants of feed tolerance, body composition and development: Protocol of a randomised trial. BMC Pediatr. 2018, 7, 220. [Google Scholar] [CrossRef]
| F80 (n = 45) | F160 (n = 42) | p Value | |
|---|---|---|---|
| Birth Weight (g) ± sd | 2028 ± 235 | 2032 ± 297 | 0.95 |
| Birth Weight Z-score ± sd | −0.737 ± 0.91 | −0.578 ± 0.97 | 0.43 |
| Birth Gestation (weeks) ± sd | 34.4 ± 2.1 | 34.2 ± 1.6 | 0.74 |
| Discharge Weight (g) ± sd | 2312 ± 255 | 2300 ± 263 | 0.83 |
| SGA | 19 (42%) | 11 (26%) | 0.12 |
| Males | 26 (52%) | 20 (48%) | 0.34 |
| Maternal GDM | 9 (20%) | 13 (31%) | 0.24 |
| Caesarean Birth | 29 (64%) | 18 (43%) | 0.07 |
| F80 (n = 45) | F160 (n = 42) | p Value | |
|---|---|---|---|
| Maximum Weight Loss (g) ± sd | 108 ± 69 | 124 ± 59 | 0.95 |
| Weight Loss as % Birth Weight | 5.3% | 6.1% | |
| Weight Loss Δ Z-score ± sd | −0.531 ± 0.28 | −0.639 ± 0.25 | 0.08 |
| Days to Regain Birth Weight ± sd | 7.5 ± 3.2 | 9.4 ± 2.8 | <0.01 |
| Weight Gain Velocity (g/kg/day) | 7.3 ± 5.5 | 7.0 ± 4.2 | 0.76 |
| Weight Gain: Δ Z-Score at 10 Days ± sd | −0.661 ± 0.18 | −0.762 ± 0.22 | 0.03 |
| Weight Gain: Δ Z-Score at Discharge ± sd | −0.620 ± 0.27 | −0.765 ± 0.23 | 0.02 |
| F80 | F160 | p Value | |
|---|---|---|---|
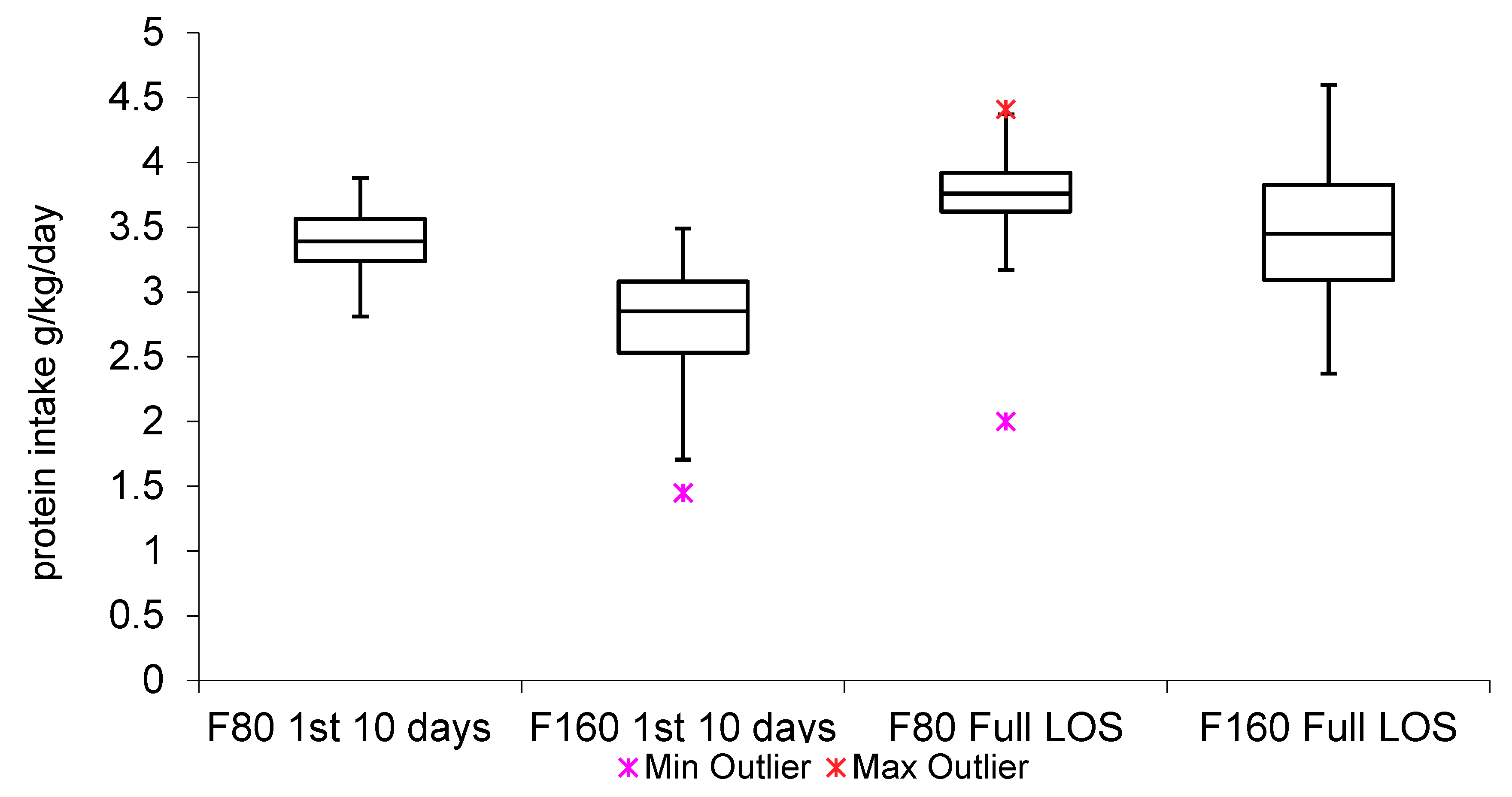
| TP Intake daily 1st 10 days (g/kg/day) ± sd | 3.38 ± 0.26 | 2.74 ± 0.53 | <0.0001 * |
| TP Intake daily 1st 10 days—Variance | 0.07 | 0.28 | <0.0001 ** |
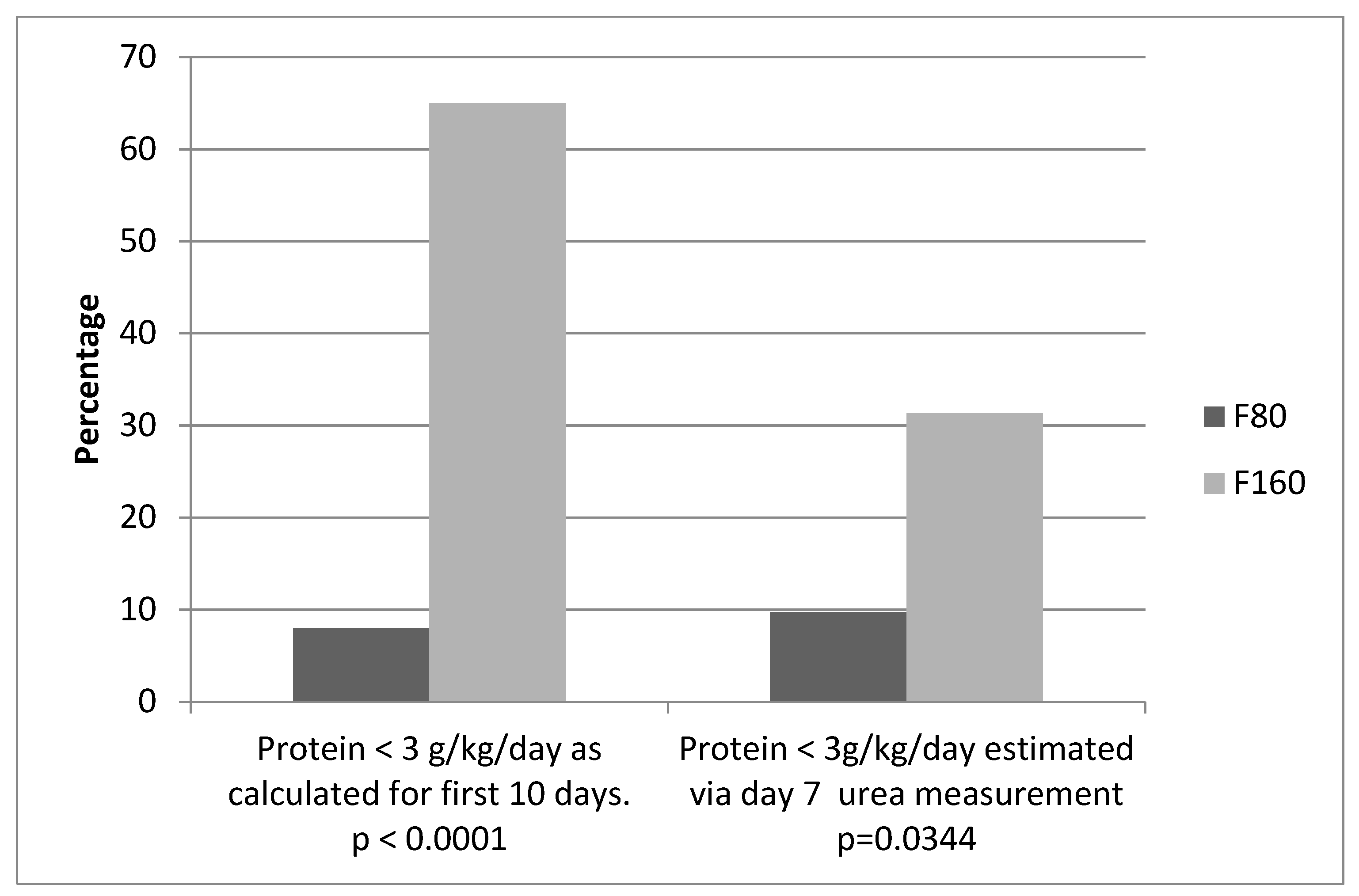
| TP Intake daily 1st 10 days <3 g/day | 8% | 65% | <0.0001 *** |
| TP Intake daily per LOS (g/kg/day) ± sd | 3.69 ± 0.46 | 3.44 ± 0.52 | 0.0245 * |
| TP Intake daily per LOS—Variance | 0.21 | 0.27 | 0.2280 ** |
| TP Intake daily per LOS <3 g/day | 11% | 17% | 0.6629 *** |
| Fortifier commencement day ± sd | 2.8 ± 0.8 | 6.7 ± 1.8 | <0.0001 * |
| Length of fortification (days) ± sd | 15.2 ± 6.1 | 12.2 ± 6.5 | 0.0273 * |
| Number babies fed fortified EBM | 42 (93%) | 38 (91%) | 0.9242 *** |
| F80 days to reach 160 mL/kg ± sd | 6.8 ± 1.2 | 6.7 ± 1.8 | 0.8288 * |
| F80 | F160 | p Value | |
|---|---|---|---|
| Urea 1st week (raw data) ± sd | 4.1 ± 1.5 | 3.6 ± 1.9 | 0.2087 * |
| Urea 1st week (corrected) ± sd | 3.6 ± 2.0 | 3.15 ± 2.0 | 0.3642 * |
| Urea (corrected) 1st week <1.6 | 9.7% | 31.3% | 0.0344 ** |
| F80 (n = 45) | F160 (n = 42) | p Value | |
|---|---|---|---|
| Feeding intolerance Jaundice requiring phototherapy | 11 (24.4%) 12 (26.7%) | 20 (47.6%) 18 (42.9%) | 0.0276 * 0.1124 * |
| Presumed sepsis | 17 (40.0%) | 17 (40.5%) | 0.9639 * |
| Hypoglycaemia | 3 (6.7%) | 4 (9.5%) | 0.6244 * |
| NEC | 0 | 0 | 1.0 |
| ALP at 7 days ± sd | 256.7 ± 86.9 | 229.4 ± 80.5 | 0.1659 ** |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fan, W.Q.; Gan, A.; Crane, O. Commencing Nutrient Supplements before Full Enteral Feed Volume Achievement Is Beneficial for Moderately Preterm to Late Preterm Low Birth Weight Babies: A Prospective, Observational Study. Nutrients 2018, 10, 1340. https://doi.org/10.3390/nu10101340
Fan WQ, Gan A, Crane O. Commencing Nutrient Supplements before Full Enteral Feed Volume Achievement Is Beneficial for Moderately Preterm to Late Preterm Low Birth Weight Babies: A Prospective, Observational Study. Nutrients. 2018; 10(10):1340. https://doi.org/10.3390/nu10101340
Chicago/Turabian StyleFan, Wei Qi, Amy Gan, and Olivia Crane. 2018. "Commencing Nutrient Supplements before Full Enteral Feed Volume Achievement Is Beneficial for Moderately Preterm to Late Preterm Low Birth Weight Babies: A Prospective, Observational Study" Nutrients 10, no. 10: 1340. https://doi.org/10.3390/nu10101340




