Dietary Fiber and Metabolic Syndrome: A Meta-Analysis and Review of Related Mechanisms
Abstract
1. Introduction
2. Methods
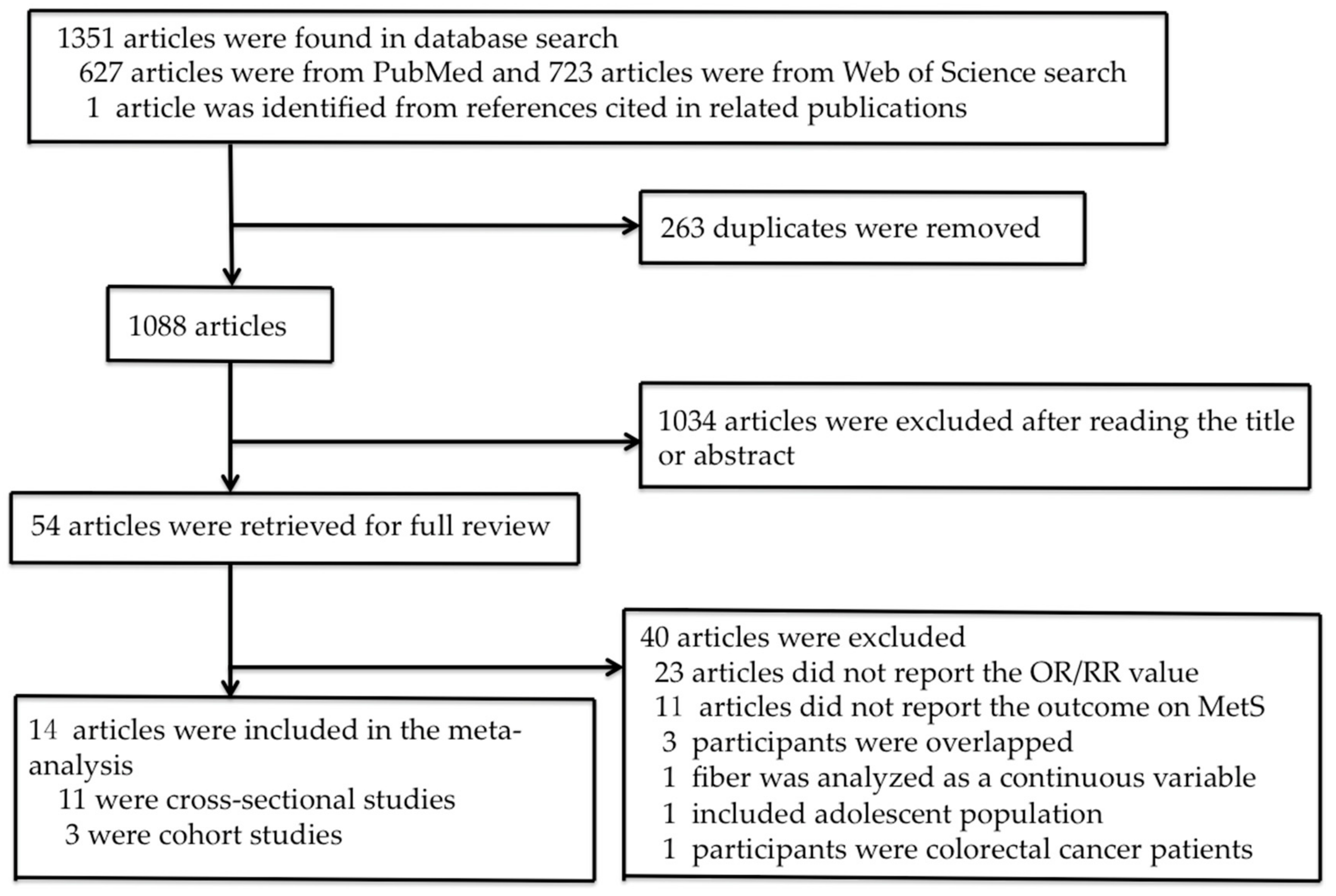
2.1. Literature Search
2.2. Study Selection
2.3. Data Extraction
2.4. Quality Assessment
2.5. Statistical Analysis
3. Results
3.1. Study Characteristics
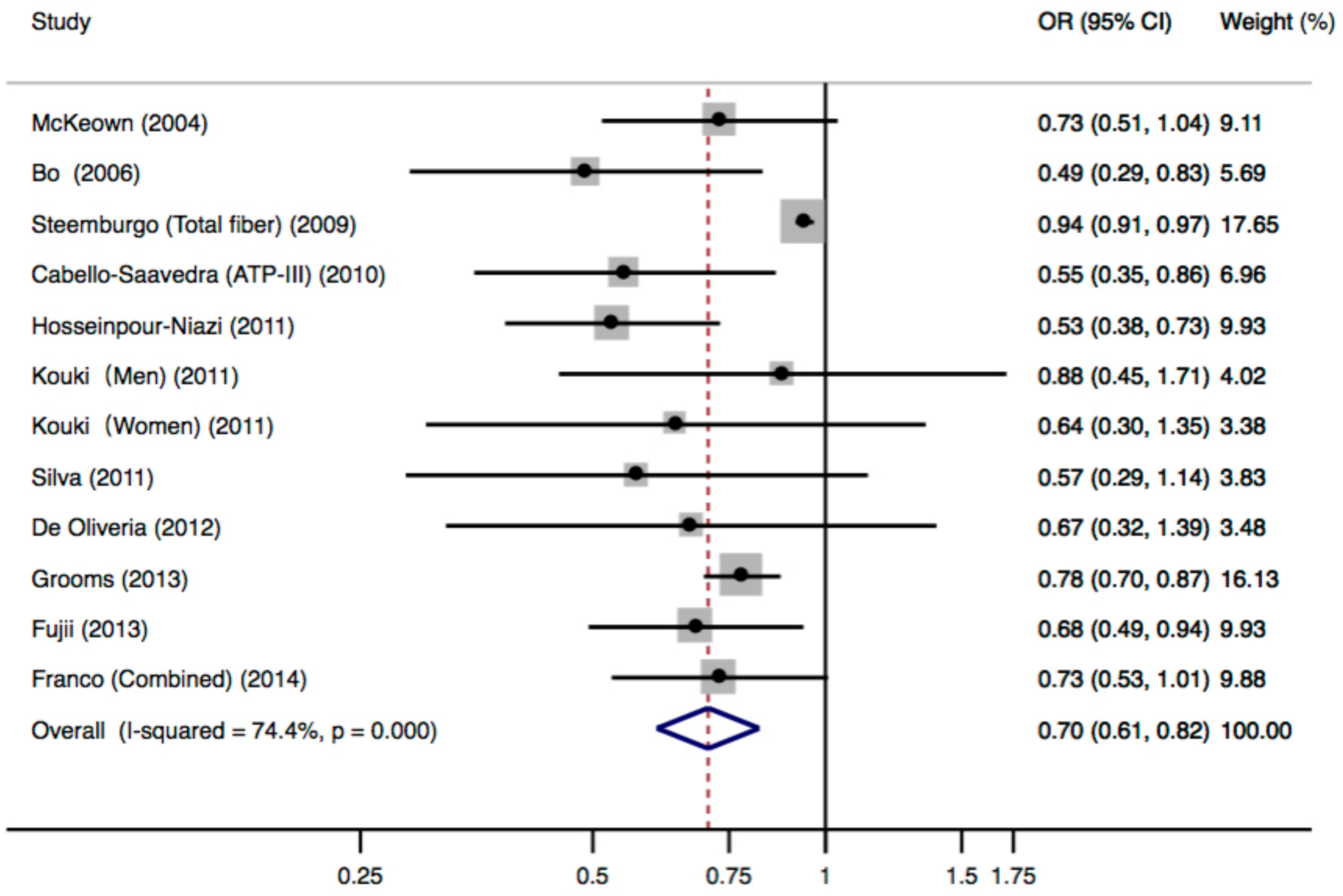
3.2. Meta-Analysis of Cross-Sectional Studies
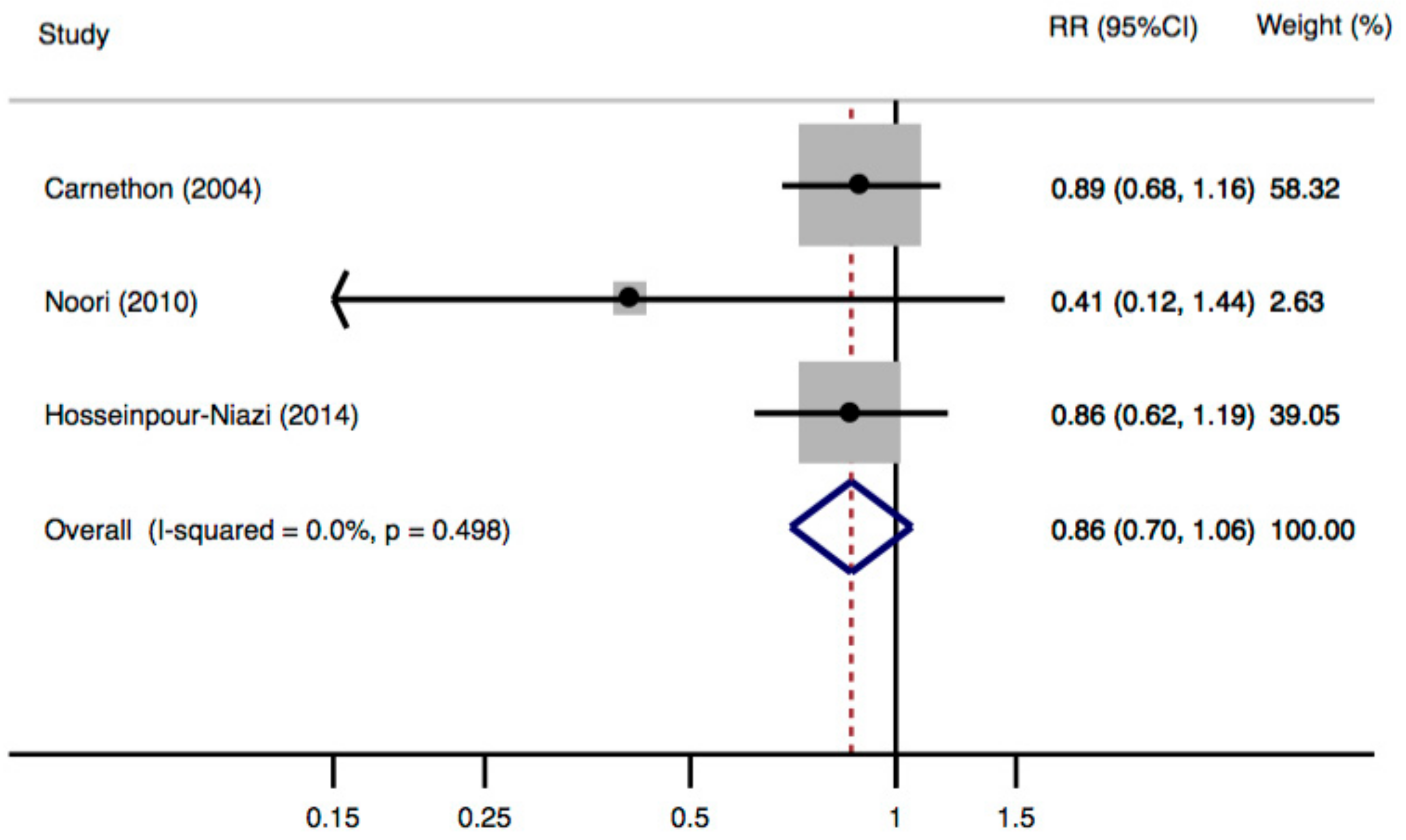
3.3. Meta-Analysis of Cohort Studies
4. Discussion
4.1. Discussion on the Meta-Analysis
4.2. Mechanisms Involved in Dietary Fiber Consumption and MetS
4.2.1. Dietary Fiber and Obesity
4.2.2. Dietary Fiber and Insulin Resistance
4.2.3. Dietary Fiber and Dyslipidemia
4.2.4. Dietary Fiber and Hypertension
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Appendix A
| Database | Search Strategy |
|---|---|
| PubMed | Input query:(fiber OR fibre OR food component) AND (metabolic syndrome OR insulin resistance syndrome) AND (cohort OR prospective OR follow-up OR incidence OR cross-sectional OR case-control)PubMed query translation:((“dietary fiber”[MeSH Terms] OR (“dietary”[All Fields] AND “fiber”[All Fields]) OR “dietary fiber”[All Fields] OR “fiber”[All Fields]) OR (“dietary fiber”[MeSH Terms] OR (“dietary”[All Fields] AND “fiber”[All Fields]) OR “dietary fiber”[All Fields] OR “fibre”[All Fields]) OR ((“food”[MeSH Terms] OR “food”[All Fields]) AND component[All Fields])) AND ((“metabolic syndrome”[MeSH Terms] OR (“metabolic”[All Fields] AND “syndrome”[All Fields]) OR “metabolic syndrome”[All Fields]) OR (“metabolic syndrome”[MeSH Terms] OR (“metabolic”[All Fields] AND “syndrome”[All Fields]) OR “metabolic syndrome”[All Fields] OR (“insulin”[All Fields] AND “resistance”[All Fields] AND “syndrome”[All Fields]) OR “insulin resistance syndrome”[All Fields] OR “insulin resistance”[MeSH Terms] OR (“insulin”[All Fields] AND “resistance”[All Fields]) OR “insulin resistance”[All Fields] OR (“insulin”[All Fields] AND “resistance”[All Fields] AND “syndrome”[All Fields]))) AND ((“cohort studies”[MeSH Terms] OR (“cohort”[All Fields] AND “studies”[All Fields]) OR “cohort studies”[All Fields] OR “cohort”[All Fields]) OR (“longitudinal studies”[MeSH Terms] OR (“longitudinal”[All Fields] AND “studies”[All Fields]) OR “longitudinal studies”[All Fields] OR “prospective”[All Fields]) OR follow-up[All Fields] OR (“epidemiology”[Subheading] OR “epidemiology”[All Fields] OR “incidence”[All Fields] OR “incidence”[MeSH Terms]) OR (“case-control studies”[MeSH Terms] OR (“case-control”[All Fields] AND “studies”[All Fields]) OR “case-control studies”[All Fields] OR (“case”[All Fields] AND “control”[All Fields]) OR “case control”[All Fields]) OR (“cross-sectional studies”[MeSH Terms] OR (“cross-sectional”[All Fields] AND “studies”[All Fields]) OR “cross-sectional studies”[All Fields] OR (“cross”[All Fields] AND “sectional”[All Fields]) OR “cross sectional”[All Fields]))Results: 627 |
| Web of Science | Search details:TS = ((“fiber” OR “fibre” OR food component) AND (“metabolic syndrome” OR insulin resistance syndrome) AND (“cohort” OR “prospective” OR “follow-up”OR “incidence” OR “cross-sectional” OR “case-control”))Refined by: [excluding] DOCUMENT TYPES: (EDITORIAL OR LETTER OR MEETING OR BOOK OR REVIEW)Timespan: All years.Search language = AutoResults: 723 |
References
- Kassi, E.; Pervanidou, P.; Kaltsas, G.; Chrousos, G. Metabolic syndrome: Definitions and controversies. BMC Med. 2011, 9, 48. [Google Scholar] [CrossRef] [PubMed]
- Wilson, P.W.; D’Agostino, R.B.; Parise, H.; Sullivan, L.; Meigs, J.B. Metabolic syndrome as a precursor of cardiovascular disease and type 2 diabetes mellitus. Circulation 2005, 112, 3066–3072. [Google Scholar] [CrossRef] [PubMed]
- Alberti, K.G.; Zimmet, P.; Shaw, J. The metabolic syndrome—A new worldwide definition. Lancet 2005, 366, 1059–1062. [Google Scholar] [CrossRef]
- Eckel, R.H.; Grundy, S.M.; Zimmet, P.Z. The metabolic syndrome. Lancet 2005, 365, 1415–1428. [Google Scholar] [CrossRef]
- Alberti, K.G.M.M.; Zimmet, P.; Shaw, J. Metabolic syndrome—A new world-wide definition. A consensus statement from the International Diabetes Federation. Diabet. Med. 2006, 23, 469–480. [Google Scholar] [CrossRef] [PubMed]
- Expert panel on detection, evaluation and treatment of high blood cholesterol in adults executive summary of the third report of the national cholesterol education program (NCEP) expert panel on detection, evaluation, and treatment of high blood cholesterol in adults (Adult Treatment Panel III). JAMA 2001, 285, 2486–2497.
- De Carvalho Vidigal, F.; Bressan, J.; Babio, N.; Salas-Salvado, J. Prevalence of metabolic syndrome in Brazilian adults: A systematic review. BMC Public Health 2013, 13, 1198. [Google Scholar] [CrossRef] [PubMed]
- Li, R.; Li, W.; Lun, Z.; Zhang, H.; Sun, Z.; Kanu, J.S.; Qiu, S.; Cheng, Y.; Liu, Y. Prevalence of metabolic syndrome in Mainland China: A meta-analysis of published studies. BMC Public Health 2016, 16, 296. [Google Scholar] [CrossRef] [PubMed]
- Tabatabaie, A.H.; Shafiekhani, M.; Nasihatkon, A.A.; Rastani, I.H.; Tabatabaie, M.; Borzoo, A.R.; Hojat, F. Prevalence of metabolic syndrome in adult population in Shiraz, southern Iran. Diabetes Metab. Syndr. 2015, 9, 153–156. [Google Scholar] [CrossRef] [PubMed]
- Viteri Holguin, M.P.; Castro Burbano, J. Prevalence of metabolic syndrome among the staff of universidad internacional del ecuador, headquarters, period 2014-2015. Nutr. Hosp. 2015, 32, 2684–2691. [Google Scholar] [CrossRef] [PubMed]
- Aguilar, M.; Bhuket, T.; Torres, S.; Liu, B.; Wong, R.J. Prevalence of the metabolic syndrome in the United States, 2003-2012. JAMA 2015, 313, 1973–1974. [Google Scholar] [CrossRef] [PubMed]
- Lim, E.S.; Ko, Y.K.; Ban, K.O. Prevalence and risk factors of metabolic syndrome in the Korean population—Korean National Health Insurance Corporation Survey 2008. J. Adv. Nurs. 2013, 69, 1549–1561. [Google Scholar] [CrossRef] [PubMed]
- Goodman, D.; Fraga, M.A.; Brodine, S.; Ibarra, M.-L.; Garfein, R.S. Prevalence of diabetes and metabolic syndrome in a migrant Mixtec population, Baja California, Mexico. J. Immigr. Minor. Heal. 2013, 15, 93–100. [Google Scholar] [CrossRef] [PubMed]
- Mozaffarian, D. Dietary and policy priorities for cardiovascular disease, diabetes, and obesity. Circulation 2016, 133, 187–225. [Google Scholar] [CrossRef] [PubMed]
- DeVries, J.W. The definition of dietary fibre. Cereal Foods World 2001, 46, 112–129. [Google Scholar] [CrossRef]
- Tucker, L.A.; Thomas, K.S. Increasing total fiber intake reduces risk of weight and fat gains in women. J. Nutr. 2009, 139, 576–581. [Google Scholar] [CrossRef] [PubMed]
- Chandalia, M.; Garg, A.; Lutjohann, D.; von Bergmann, K.; Grundy, S.M.; Brinkley, L.J. Beneficial effects of high dietary fiber intake in patients with type 2 diabetes mellitus. N. Engl. J. Med. 2000, 342, 1392–1398. [Google Scholar] [CrossRef] [PubMed]
- Evans, C.E.L.; Greenwood, D.C.; Threapleton, D.E.; Cleghorn, C.L.; Nykjaer, C.; Woodhead, C.E.; Gale, C.P.; Burley, V.J. Effects of dietary fibre type on blood pressure: A systematic review andmeta-analysis of randomized controlled trials of healthy individuals. J. Hypertens. 2015, 33, 897–911. [Google Scholar] [CrossRef] [PubMed]
- Jenkins, D.J.A.; Kendall, C.W.C.; Vuksan, V.; Vidgen, E.; Parker, T.; Faulkner, D.; Mehling, C.C.; Garsetti, M.; Testolin, G.; Cunnane, S.C.; et al. Soluble fiber intake at a dose approved by the US Food and Drug Administration for a claim of health benefits: Serum lipid risk factors for cardiovascular disease assessed in a randomized controlled crossover trial. Am. J. Clin. Nutr. 2002, 75, 834–839. [Google Scholar] [PubMed]
- Brown, L.; Rosner, B.; Willett, W.W.; Sacks, F.M. Cholesterol-lowering effects of dietary fiber: A meta-analysis. Am. J. Clin. Nutr. 1999, 69, 30–42. [Google Scholar] [CrossRef] [PubMed]
- Bruscato, N.; da Costa Vieira, J.; do Nascimento, N.; Canto, M.; Stobbe, J.; Gottlieb, M.; Wagner, M.; Dalacorte, R. Dietary intake is not associated to the metabolic syndrome in elderly women. N. Am. J. Med. Sci. 2010, 2, 182–188. [Google Scholar] [CrossRef] [PubMed]
- Hutton, B.; Salanti, G.; Caldwell, D.M.; Chaimani, A.; Schmid, C.H.; Cameron, C.; Ioannidis, J.P.A.; Straus, S.; Thorlund, K.; Jansen, J.P.; et al. The PRISMA extension statement for reporting of systematic reviews incorporating network meta-analyses of health care interventions: Checklist and explanations. Ann. Intern. Med. 2015, 162, 777–784. [Google Scholar] [CrossRef] [PubMed]
- Rostom, A.; Dubé, C.; Cranney, A.; Saloojee, N.; Sy, R.; Garritty, C.; Sampson, M.; Zhang, L.; Yazdi, F.; Mamaladze, V.; et al. Cross-sectional/prevalence study quality. In Celiac Disease; Agency for Healthcare Research and Quality: Rockville, MD, USA, 2004. [Google Scholar]
- Wells, G.A.; Shea, B.; O’Connell, D.; Peterson, J.; Welch, V.; Losos, M.; Tugwell, P. The Newcastle-Ottawa Scale (NOS) for assessing the quality of nonrandomized studies in meta-analyses. Ottawa Hosp. Res. Inst. 2013, 1–4. [Google Scholar] [CrossRef]
- Higgins, J.; Thompson, S. Quantifying heterogeneity in a meta-analysis. Stat. Med. 2002, 21, 1539–1558. [Google Scholar] [CrossRef] [PubMed]
- Higgins, J.; Thompson, S.; Deeks, J.; Altman, D. Measuring inconsistency in meta-analyses. Br. Med. J. 2003, 327, 557–560. [Google Scholar] [CrossRef] [PubMed]
- Franco, B.M.; María, E.; Esteban, A.; Ordovás, J.M.; Casasnovas, J.A.; Peñalvo, J.L.; Srsxodwlrq, L.Q.D.Z.; Zlwklq, R.O.G.; Udjrq, W.K.H. Soluble and insoluble dietary fiber intake and risk factors for metabolic syndrome and cardiovascular disease in middle-aged adults:the AWHS cohort. Nutr. Hosp. 2014, 30, 1279–1288. [Google Scholar] [CrossRef]
- Cabello-Saavedra, E.; Bes-Rastrollo, M.; Martinez, J.A.; Diez-Espino, J.; Buil-Cosiales, P.; Serrano-Martinez, M.; Martinez-Gonzalez, M.A. Macronutrient intake and metabolic syndrome in subjects at high cardiovascular risk. Ann. Nutr. Metab. 2010, 56, 152–159. [Google Scholar] [CrossRef] [PubMed]
- Fujii, H.; Iwase, M.; Ohkuma, T.; Ogata-Kaizu, S.; Ide, H.; Kikuchi, Y.; Idewaki, Y.; Joudai, T.; Hirakawa, Y.; Uchida, K.; et al. Impact of dietary fiber intake on glycemic control, cardiovascular risk factors and chronic kidney disease in Japanese patients with type 2 diabetes mellitus: The Fukuoka Diabetes Registry. Nutr. J. 2013, 12, 159. [Google Scholar] [CrossRef] [PubMed]
- De Oliveira, E.P.; McLellan, K.C.; Vaz de Arruda Silveira, L.; Burini, R.C. Dietary factors associated with metabolic syndrome in Brazilian adults. Nutr. J. 2012, 11, 13–20. [Google Scholar] [CrossRef] [PubMed]
- Kouki, R.; Schwab, U.; Hassinen, M.; Komulainen, P.; Heikkila, H.; Lakka, T.A.; Rauramaa, R. Food consumption, nutrient intake and the risk of having metabolic syndrome: The DR’s EXTRA Study. Eur. J. Clin. Nutr. 2011, 65, 368–377. [Google Scholar] [CrossRef] [PubMed]
- Egger, M.; Davey Smith, G.; Schneider, M.; Minder, C.; Mulrow, C.; Egger, M.; Smith, G.D.; Eysenck, H.; Egger, M.; Zellweger-Zähner, T.; et al. Bias in meta-analysis detected by a simple, graphical test. Br. Med. J. (Clin. Res. Ed.) 1997, 315, 629–634. [Google Scholar] [CrossRef]
- Nafar, M.; Noori, N.; Jalali-Farahani, S.; Hosseinpanah, F.; Poorrezagholi, F.; Ahmadpoor, P.; Samadian, F.; Firouzan, A.; Einollahi, B. Mediterranean diets are associated with a lower incidence of metabolic syndrome one year following renal transplantation. Kidney Int. 2009, 76, 1199–1206. [Google Scholar] [CrossRef] [PubMed]
- Esmaillzadeh, A.; Mirmiran, P.; Azizi, F. Whole-grain consumption and the metabolic syndrome: A favorable association in Tehranian adults. Eur. J. Clin. Nutr. 2005, 59, 353–362. [Google Scholar] [CrossRef] [PubMed]
- Mirmiran, P.; Noori, N.; Azizi, F. A prospective study of determinants of the metabolic syndrome in adults. Nutr. Metab. Cardiovasc. Dis. 2008, 18, 567–573. [Google Scholar] [CrossRef] [PubMed]
- Bahadoran, Z.; Mirmiran, P.; Delshad, H.; Azizi, F. White rice consumption is a risk factor for metabolic syndrome in Tehrani adults: A prospective approach in Tehran lipid and glucose study. Arch. Iran. Med. 2014, 17, 435–440. [Google Scholar] [PubMed]
- Carlson, J.J.; Eisenmann, J.C.; Norman, G.J.; Ortiz, K.A.; Young, P.C. Dietary fiber and nutrient density are inversely associated with the metabolic syndrome in US adolescents. J. Am. Diet. Assoc. 2011, 111, 1688–1695. [Google Scholar] [CrossRef] [PubMed]
- Lim, H.-S.; Shin, E.-J.; Yeom, J.-W.; Park, Y.-H.; Kim, S.-K. Association between nutrient intake and metabolic syndrome in patients with colorectal cancer. Clin. Nutr. Res. 2017, 6, 38. [Google Scholar] [CrossRef] [PubMed]
- Silva, F.M.; Steemburgo, T.; de Mello, V.D.F.; Tonding, S.F.; Gross, J.L.; Azevedo, M.J. High dietary glycemic index and low fiber content are associated with metabolic syndrome in patients with type 2 diabetes. J. Am. Coll. Nutr. 2011, 30, 141–148. [Google Scholar] [CrossRef] [PubMed]
- McKeown, N.M.; Meigs, J.B.; Liu, S.; Saltzman, E.; Wilson, P.W.F.; Jacques, P.F. Carbohydrate nutrition, insulin resistance, and the prevalence of the metabolic syndrome in the Framingham Offspring Cohort. Diabetes Care 2004, 27, 538–546. [Google Scholar] [CrossRef] [PubMed]
- Bo, S.; Durazzo, M.; Guidi, S.; Carello, M.; Sacerdote, C.; Silli, B.; Rosato, R.; Cassader, M.; Gentile, L.; Pagano, G. Dietary magnesium and fiber intakes and inflammatory and metabolic indicators in middle-aged subjects from a population-based cohort. Am. J. Clin. Nutr. 2006, 84, 1062–1069. [Google Scholar] [PubMed]
- Hosseinpour-Niazi, S.; Mirmiran, P.; Sohrab, G.; Hosseini-Esfahani, F.; Azizi, F. Inverse association between fruit, legume, and cereal fiber and the risk of metabolic syndrome: Tehran Lipid and Glucose Study. Diabetes Res. Clin. Pract. 2011, 94, 276–283. [Google Scholar] [CrossRef] [PubMed]
- Grooms, K.N.; Ommerbor, M.J.; Quyen, D.; Luc, D.; Clark, C.R. Dietary fiber intake and cardiometabolic risks among US adults, NHANES 1999–2010. Am. J. Med. 2013, 126, 130–134. [Google Scholar] [CrossRef] [PubMed]
- Steemburgo, T.; Dall’Alba, V.; Almeida, J.C.; Zelmanovitz, T.; Gross, J.L.; de Azevedo, M.J. Intake of soluble fibers has a protective role for the presence of metabolic syndrome in patients with type 2 diabetes. Eur. J. Clin. Nutr. 2009, 63, 127–133. [Google Scholar] [CrossRef] [PubMed]
- Hosseinpour-Niazi, S.; Mirmiran, P.; Mirzaei, S.; Azizi, F. Cereal, fruit and vegetable fibre intake and the risk of the metabolic syndrome: A prospective study in the Tehran Lipid and Glucose Study. J. Hum. Nutr. Diet. 2014, 28, 236–245. [Google Scholar] [CrossRef] [PubMed]
- Noori, N.; Nafar, M.; Poorrezagholi, F.; Ahmadpoor, P.; Samadian, F.; Firouzan, A.; Einollahi, B. Dietary intakes of fiber and magnesium and incidence of metabolic syndrome in first year after renal transplantation. J. Ren. Nutr. 2010, 20, 101–111. [Google Scholar] [CrossRef] [PubMed]
- Carnethon, M.R.; Loria, C.M.; Hill, J.O.; Sidney, S.; Savage, P.J.; Liu, K. Coronary Artery Risk Development in Young Adults Study. Risk factors for the metabolic syndrome: The Coronary Artery Risk Development in Young Adults (CARDIA) study, 1985–2001. Diabetes Care 2004, 27, 2707–2715. [Google Scholar] [CrossRef] [PubMed]
- Aleixandre, A.; Miguel, M. Dietary fiber in the prevention and treatment of metabolic syndrome: A review. Crit. Rev. Food Sci. Nutr. 2008, 48, 905–912. [Google Scholar] [CrossRef] [PubMed]
- Femenia, A.; Selvendran, R.R.; Ring, S.G.; Robertson, J.A. Effects of heat treatment and dehydration on properties of cauliflower fiber. J. Agric. Food Chem. 1999, 47, 728–732. [Google Scholar] [CrossRef] [PubMed]
- Kerckhoffs, D.A.J.M.; Hornstra, G.; Mensink, R.P. Cholesterol-lowering effect of beta-glucan from oat bran in mildly hypercholesterolemic subjects may decrease when beta-glucan is incorporated into bread and cookies. Am. J. Clin. Nutr. 2003, 78, 221–227. [Google Scholar] [PubMed]
- Wei, B.; Liu, Y.; Lin, X.; Fang, Y.; Cui, J.; Wan, J. Dietary fiber intake and risk of metabolic syndrome: A meta-analysis of observational studies. Clin. Nutr. 2017. [Google Scholar] [CrossRef] [PubMed]
- Ye, E.Q.; Chacko, S.A.; Chou, E.L.; Kugizaki, M.; Liu, S.M. Greater whole-grain intake is associated with lower risk of type 2 diabetes, cardiovascular disease, and weight gain. J. Nutr. 2012, 142, 1304–1313. [Google Scholar] [CrossRef] [PubMed]
- Kranz, S.; Brauchla, M.; Slavin, J.L.; Miller, K.B. What do we know about dietary fiber intake in children and health ? The effects of fiber intake on constipation, obesity, and diabetes in children. Am. Soc. Nutr. Adv. Nutr. 2012, 3, 47–53. [Google Scholar] [CrossRef] [PubMed]
- Du, H.; van der A, D.L.; Boshuizen, H.C.; Forouhi, N.G.; Wareham, N.J.; Halkjaer, J.; Tjonneland, A.; Overvad, K.; Jakobsen, M.U.; Boeing, H.; et al. Dietary fiber and subsequent changes in body weight and waist circumference in European men and women. Am. J. Clin. Nutr. 2010, 91, 329–336. [Google Scholar] [CrossRef] [PubMed]
- Davis, J.N.; Alexander, K.E.; Ventura, E.E.; Toledo-Corral, C.M.; Goran, M.I. Inverse relation between dietary fiber intake and visceral adiposity in overweight Latino youth. Am. J. Clin. Nutr. 2009, 90, 1160–1166. [Google Scholar] [CrossRef] [PubMed]
- Murakami, K.; Sasaki, S.; Okubo, H.; Takahashi, Y.; Hosoi, Y.; Itabashi, M.; Freshmen in Dietetic Courses Study II Group. Dietary fiber intake, dietary glycemic index and load, and body mass index: A cross-sectional study of 3931 Japanese women aged 18-20 years. Eur. J. Clin. Nutr. 2007, 61, 986–995. [Google Scholar] [CrossRef] [PubMed]
- Koh-Banerjee, P.; Rimm, E.B. Whole grain consumption and weight gain: A review of the epidemiological evidence, potential mechanisms and opportunities for future research. Proc. Nutr. Soc. 2003, 62, 25–29. [Google Scholar] [CrossRef] [PubMed]
- Kromhout, D.; Bloemberg, B.; Seidell, J.C.; Nissinen, A.; Menotti, A.; Seven Countries Study, G. Physical activity and dietary fiber determine population body fat levels: The Seven Countries Study. Int. J. Obes. 2001, 25, 301–306. [Google Scholar] [CrossRef] [PubMed]
- Solah, V.A.; Kerr, D.A.; Hunt, W.J.; Johnson, S.K.; Boushey, C.J.; Delp, E.J.; Meng, X.; Gahler, R.J.; James, A.P.; Mukhtar, A.S.; et al. Effect of fibre supplementation on body weight and composition, frequency of eating and dietary choice in overweight individuals. Nutrients 2017, 9, 149. [Google Scholar] [CrossRef] [PubMed]
- Abete, I.; Astrup, A.; Martinez, J.A.; Thorsdottir, I.; Zulet, M.A. Obesity and the metabolic syndrome: Role of different dietary macronutrient distribution patterns and specific nutritional components on weight loss and maintenance. Nutr. Rev. 2010, 68, 214–231. [Google Scholar] [CrossRef] [PubMed]
- Parnell, J.A.; Reimer, R.A. Weight loss during oligofructose supplementation is associated with decreased ghrelin and increased peptide YY in overweight and obese adults. Am. J. Clin. Nutr. 2009, 89, 1751–1759. [Google Scholar] [CrossRef] [PubMed]
- Trigueros, L.; Pena, S.; Ugidos, A.V.; Sayas-Barbera, E.; Perez-Alvarez, J.A.; Sendra, E. Food ingredients as anti-obesity agents: A review. Crit. Rev. Food Sci. Nutr. 2013, 53, 929–942. [Google Scholar] [CrossRef] [PubMed]
- Brownlee, I.A.; Chater, P.I.; Pearson, J.P.; Wilcox, M.D. Dietary fibre and weight loss: Where are we now? Food Hydrocoll. 2017, 68, 186–191. [Google Scholar] [CrossRef]
- Slavin, J.L. Dietary fiber and body weight. Nutrition 2005, 21, 411–418. [Google Scholar] [CrossRef] [PubMed]
- Pereira, M.; Ludwig, D. Dietary fiber and body-weight regulation. Observations and mechanisms. Pediatr. Clin. N. Am. 2001, 969–980. [Google Scholar] [CrossRef]
- Howarth, N.C.; Saltzman, E.; Roberts, S.B. Dietary fiber and weight regulation. Nutr. Rev. 2001, 59, 129–139. [Google Scholar] [CrossRef] [PubMed]
- Eastwood, M.A.; Morris, E.R. Physical properties of dietary fiber that influence physiological function: A model for polymers along the gastrointestinal tract. Am. J. Clin. Nutr. 1992, 55, 436–442. [Google Scholar] [PubMed]
- Lee, I.; Shi, L.; Webb, D.-L.; Hellstrom, P.M.; Riserus, U.; Landberg, R. Effects of whole-grain rye porridge with added inulin and wheat gluten on appetite, gut fermentation and postprandial glucose metabolism: A randomised, cross-over, breakfast study. Br. J. Nutr. 2016, 116, 2139–2149. [Google Scholar] [CrossRef] [PubMed]
- Ye, Z.; Arumugam, V.; Haugabrooks, E.; Williamson, P.; Hendrich, S. Soluble dietary fiber (Fibersol-2) decreased hunger and increased satiety hormones in humans when ingested with a meal. Nutr. Res. 2015, 35, 393–400. [Google Scholar] [CrossRef] [PubMed]
- Lafond, D.W.; Greaves, K.A.; Maki, K.C.; Leidy, H.J.; Romsos, D.R. Effects of two dietary fibers as part of ready-to-eat cereal (RTEC) breakfasts on perceived appetite and gut hormones in overweight women. Nutrients 2015, 7, 1245–1266. [Google Scholar] [CrossRef] [PubMed]
- Byrne, C.S.; Chambers, E.S.; Morrison, D.J.; Frost, G. The role of short chain fatty acids in appetite regulation and energy homeostasis. Int. J. Obes. 2015, 39, 1331–1338. [Google Scholar] [CrossRef] [PubMed]
- Chambers, E.S.; Morrison, D.J.; Frost, G. Control of appetite and energy intake by SCFA: What are the potential underlying mechanisms? Proc. Nutr. Soc. 2015, 74, 328–336. [Google Scholar] [CrossRef] [PubMed]
- Sanchez, D.; Miguel, M.; Aleixandre, A.; Sánchez, D.; Miguel, M.; Aleixandre, A. Dietary fiber, gut peptides, and adipocytokines. J. Med. Food 2012, 15, 223–230. [Google Scholar] [CrossRef] [PubMed]
- Benítez-Páez, A.; Gómez Del Pulgar, E.M.; Kjølbæk, L.; Brahe, L.K.; Astrup, A.; Larsen, L.H.; Sanz, Y. Impact of dietary fiber and fat on gut microbiota re-modeling and metabolic health. Trends Food Sci. Technol. 2016, 57, 201–212. [Google Scholar] [CrossRef]
- Schroeder, N.; Marquart, L.F.; Gallaher, D.D. The role of viscosity and fermentability of dietary fibers on satiety- and adiposity-related hormones in rats. Nutrients 2013, 5, 2093–2113. [Google Scholar] [CrossRef] [PubMed]
- Kristensen, M.; Jensen, M.G. Dietary fibres in the regulation of appetite and food intake. Importance of viscosity. Appetite 2011, 56, 65–70. [Google Scholar] [CrossRef] [PubMed]
- Isken, F.; Klaus, S.; Osterhoff, M.; Pfeiffer, A.F.H.; Weickert, M.O. Effects of long-term soluble vs. insoluble dietary fiber intake on high-fat diet-induced obesity in C57BL/6J mice. J. Nutr. Biochem. 2010, 21, 278–284. [Google Scholar] [CrossRef] [PubMed]
- Artiss, J.D.; Brogan, K.; Brucal, M.; Moghaddam, M.; Jen, K.L.C. The effects of a new soluble dietary fiber on weight gain and selected blood parameters in rats. Metab. Exp. 2006, 55, 195–202. [Google Scholar] [CrossRef] [PubMed]
- Gemen, R.; de Vries, J.J.F.F.; Slavin, J.L. Relationship between molecular structure of cereal dietary fiber and health effects: Focus on glucose/insulin response and gut health. Nutr. Rev. 2011, 69, 22–33. [Google Scholar] [CrossRef] [PubMed]
- Cho, S.S.; Qi, L.; Fahey, G.C.; Klurfeld, D.M. Consumption of cereal fiber, mixtures of whole grains and bran, and whole grains and risk reduction in type 2 diabetes, obesity, and cardiovascular disease. Am. J. Clin. Nutr. 2013, 98, 594–619. [Google Scholar] [CrossRef] [PubMed]
- Garcia, A.L.; Otto, B.; Reich, S.-C.; Weickert, M.O.; Steiniger, J.; Machowetz, A.; Rudovich, N.N.; Möhlig, M.; Katz, N.; Speth, M.; et al. Arabinoxylan consumption decreases postprandial serum glucose, serum insulin and plasma total ghrelin response in subjects with impaired glucose tolerance. Eur. J. Clin. Nutr. 2007, 61, 334–341. [Google Scholar] [CrossRef] [PubMed]
- Pereira, M.A.; Jacobs, J.D.R.; Joel, J.P.; Susan, K.R.; Myron, D.G.; Joanne, L.S.; Elizabeth, R.S. Effect of whole grains on insulin sensitivity in overweight hyperinsulinemic adults. Am. J. Clin. Nutr. 2002, 75, 848–855. [Google Scholar] [PubMed]
- Delzenne, N.M.; Cani, P.D. A place for dietary fibre in the management of the metabolic syndrome. Curr. Opin. Clin. Nutr. Metab. Care 2005, 8, 636–640. [Google Scholar] [CrossRef] [PubMed]
- Hallfrisch, J.; Facn; Behall, K.M. Mechanisms of the effects of grains on insulin and glucose responses. J. Am. Coll. Nutr. 2000, 19, 320S–325S. [Google Scholar] [CrossRef] [PubMed]
- Consortium, I. Dietary fibre and incidence of type 2 diabetes in eight European countries: The EPIC-InterAct Study and a meta-analysis of prospective studies. Diabetologia 2015, 58, 1394–1408. [Google Scholar] [CrossRef] [PubMed]
- Galisteo, M.; Duarte, J.; Zarzuelo, A. Effects of dietary fibers on disturbances clustered in the metabolic syndrome. J. Nutr. Biochem. 2008, 19, 71–84. [Google Scholar] [CrossRef] [PubMed]
- Weickert, M.O.; Pfeiffer, A.F.H. Metabolic effects of dietary fiber consumption and prevention of diabetes. J. Nutr. 2008, 138, 439–442. [Google Scholar] [PubMed]
- Dandona, P.; Aljada, A.; Chaudhuri, A.; Mohanty, P.; Garg, R. Metabolic syndrome: A comprehensive perspective based on interactions between obesity, diabetes, and inflammation. Circulation 2005, 111, 1448–1454. [Google Scholar] [CrossRef] [PubMed]
- Leclère, C.; Champ, M.; Boillot, J.; Guille, G.; Lecannu, G.; Molis, C.; Bornet, F.; Krempf, M.; Delort-Laval, J.; Galmiche, J. Role of viscous response after guar gums in lowering the glycemic response after a solid meal. Am. J. Clin. Nutr. 1994, 59, 914–921. [Google Scholar] [PubMed]
- Jenkins, D.J.; Wolever, T.M.; Leeds, A.R.; Gassull, M.A.; Haisman, P.; Dilawari, J.; Goff, D.V.; Metz, G.L.; Alberti, K.G. Dietary fibres, fibre analogues, and glucose tolerance: Importance of viscosity. Br. Med. J. 1978, 1, 1392–1394. [Google Scholar] [CrossRef] [PubMed]
- Torsdottir, I.; Alpsten, M.; Andersson, H.; Einarsson, S. Dietary guar gum effects on postprandial blood glucose, insulin and hydroxyproline in humans. J. Nutr. 1989, 119, 1925–1931. [Google Scholar] [PubMed]
- Zhou, Q.; Wu, J.; Tang, J.; Wang, J.-J.; Lu, C.-H.; Wang, P.-X. Beneficial effect of higher dietary fiber intake on plasma HDL-C and TC/HDL-C ratio among Chinese rural-to-urban migrant workers. Int. J. Environ. Res. Public Health 2015, 12, 4726–4738. [Google Scholar] [CrossRef] [PubMed]
- Shinozaki, K.; Okuda, M.; Sasaki, S.; Kunitsugu, I.; Shigeta, M. Dietary fiber consumption decreases the risks of overweight and hypercholesterolemia in Japanese children. Ann. Nutr. Metab. 2015, 67, 58–64. [Google Scholar] [CrossRef] [PubMed]
- Hollaender, P.L.B.; Ross, A.B.; Kristensen, M. Whole-grain and blood lipid changes in apparently healthy adults: A systematic review and meta-analysis of randomized controlled studies. Am. J. Clin. Nutr. 2015, 102, 556–572. [Google Scholar] [CrossRef] [PubMed]
- Zhu, X.; Sun, X.; Wang, M.; Zhang, C.; Cao, Y.; Mo, G.; Liang, J.; Zhu, S. Quantitative assessment of the effects of beta-glucan consumption on serum lipid profile and glucose level in hypercholesterolemic subjects. Nutr. Metab. Cardiovasc. Dis. 2015, 25, 714–723. [Google Scholar] [CrossRef] [PubMed]
- McRorie, J.W.; McKeown, N.M. Understanding the physics of functional fibers in the gastrointestinal tract: An evidence-based approach to resolving enduring misconceptions about insoluble and soluble fiber. J. Acad. Nutr. Diet. 2017, 117, 251–264. [Google Scholar] [CrossRef] [PubMed]
- Mcrorie, J.; Fahey, G. Fiber supplements and cinically meaningful health benefits: Identifying the physiochemical characteristics of fiber that drive specific physiologic effects. In Dietary Supplements in Health Promotion; CRC Press: Florence, Italy, 2015; ISBN 978-1-4822-1034-7. [Google Scholar]
- Chutkan, R.; Fahey, G.; Wright, W.L.; Mcrorie, J. Viscous versus nonviscous soluble fiber supplements: Mechanisms and evidence for fiber-specific health benefits. J. Am. Acad. Nurse Pract. 2012, 24, 476–487. [Google Scholar] [CrossRef] [PubMed]
- Reppas, C.; Swidan, S.Z.; Tobey, S.W.; Turowski, M.; Dressman, J.B. Hydroxypropylmethylcellulose significantly lowers blood cholesterol in mildly hypercholesterolemic human subjects. Eur. J. Clin. Nutr. 2009, 63, 71–77. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Yu, X.; Yuan, Y.; Ling, Y.; Ruan, Y.; Si, T.; Lu, T.; Wu, S.; Gong, X.; Zhu, Z.; et al. Positive association of the human frizzled 3 (FZD3) gene haplotype with schizophrenia in Chinese Han population. Am. J. Med. Genet. B Neuropsychiatr. Genet. 2004, 129, 16–19. [Google Scholar] [CrossRef] [PubMed]
- Gunness, P.; Gidley, M.J. Mechanisms underlying the cholesterol-lowering properties of soluble dietary fibre polysaccharides. Food Funct. 2010, 1, 149. [Google Scholar] [CrossRef] [PubMed]
- Van Bennekum, A.M.; Nguyen, D.V.; Schulthess, G.; Hauser, H.; Phillips, M.C. Mechanisms of cholesterol-lowering effects of dietary insoluble fibres: Relationships with intestinal and hepatic cholesterol parameters. Br. J. Nutr. 2005, 94, 331–337. [Google Scholar] [CrossRef] [PubMed]
- Lelong, H.; Blacher, J.; Baudry, J.; Adriouch, S.; Galan, P.; Fezeu, L.; Hercberg, S.; Kesse-Guyot, E. Individual and combined effects of dietary factors on risk of incident hypertension: Prospective analysis from the nutriNet-Sante Cohort. Hypertension 2017. [Google Scholar] [CrossRef] [PubMed]
- Burke, V.; Hodgson, J.M.; Beilin, L.J.; Giangiulioi, N.; Rogers, P.; Puddey, I.B. Dietary protein and soluble fiber reduce ambulatory blood pressure in treated hypertensives. Hypertension 2001, 38, 821–826. [Google Scholar] [CrossRef] [PubMed]
- He, J.; Streiffer, R.H.; Muntner, P.; Krousel-Wood, M.A.; Whelton, P.K. Effect of dietary fiber intake on blood pressure: A randomized, double-blind, placebo-controlled trial. J. Hypertens. 2004, 22, 73–80. [Google Scholar] [CrossRef] [PubMed]
- Streppel, M.; Arends, L.; van ’t Veer, P.; Grobbee, D.; Geleijnse, J. Dietary fiber and blood pressure: A meta-analysis of randomized placebo-controlled trials. Arch. Intern. Med. 2005, 165, 150–156. [Google Scholar] [CrossRef] [PubMed]
- Whelton, S.; Hyre, A.; Pedersen, B. Effect of dietary fiber intake on blood pressure: A meta-analysis of randomized, controlled clinical trials. J. Hypertens. 2005, 23, 475–481. [Google Scholar] [CrossRef] [PubMed]
- Ferri, C.; Bellini, C.; Desideri, G.; Valenti, M.; de Mattia, G.; Santucci, A.; Hollenberg, N.K.; Williams, G.H. Relationship between insulin resistance and nonmodulating hypertension: Linkage of metabolic abnormalities and cardiovascular risk. Diabetes 1999, 48, 1623–1630. [Google Scholar] [CrossRef] [PubMed]
| Study | Country | No. of Participants | Type of Fiber | Dietary Assessment | Comparison | OR (95% CI) | Variables Accounted for | Score |
|---|---|---|---|---|---|---|---|---|
| McKeown, 2004 [40] | USA | 2834 | Dietary fiber | FFQ | >22.2 vs. <13.5 g/day | ATP-III: 0.73 (0.51–1.03) | Sex, age, cigarette dose, total energy intake, alcohol intake, percentage saturated fat, percentage polyunsaturated fat, multivitamin use and physical activity | 8 |
| Bo, 2006 [41] | Italy | 1653 | Dietary fiber | FFQ | 27.8 (22.6–102.7) vs. 13.2 (3.4–16.4) g/day | ATP-III: 0.49 (0.29–0.83) | Age, sex, BMI, smoking, alcohol intake, level of physical activity, dietary intake of total calories, total percentage of fat, intake of magnesium | 8 |
| Steemburgo, 2009 [44] | Brazil | 214 | Soluble fiberTotal fiber | 3-day weighed-diet record | 6.3 ± 3.0 vs. 5.2 ± 2.1 g/day20.3 ± 7.8 vs. 16.3 ± 7.0 g/day | IDF: 0.86 (0.74–0.98)IDF: 0.94 (0.90–0.95) | Gender, duration of T2DM and total energy intake | 8 |
| Cabello-Saavedra, 2010 [28] | Spain | 967 | Dietary fiber | FFQ | >26 vs. <18 g/day | ATP-III: 0.55 (0.35–0.86)IDF:0.60 (0.38–0.94) | Age, sex, total energy intake, T2DM, smoking, alcohol intake, education level, marital status and physical activity | 9 |
| Kouki, 2011 [31] | Finland | 1334 | Dietary fiber | 4-day food record | Women: 14.6 ± 3.9 vs. 14.1 ± 3.5 g/1000 kcalMen: 13.3 ± 4.0 vs. 12.5 ± 4.3 g/1000 kcal | ATP-III: 0.64 (0.30–1.33)ATP-III: 0.88 (0.44–1.67) | Age, alcohol consumption, smoking, education and VO2max | 8 |
| Hosseinpour-Niazi, 2011 [42] | Iran | 2457 | Dietary fiber | FFQ | ≥20.7 vs. ≤11.5 g/day | ATP-III: 0.53 (0.39–0.74) | Age, gender, physical activity, smoking status, total energy intake, percentage energy from carbohydrate, fat, saturated fatty acid, cholesterol, magnesium and BMI | 8 |
| Silva, 2011 [39] | Brazil | 175 | Dietary fiber | 3-day weighed-diet record | >18.6 g vs. ≤18.6 g/day | IDF: 0.57 (0.29–1.15) | Gender and energy | 7 |
| De Oliveira, 2012 [30] | Brazil | 305 | Dietary fiber | 24 h dietary recall | ≥20 vs. <20 g/day | Modified ATP-III: 0.67 (0.32–1.38) | Gender, age, BMI and TCV | 7 |
| Fujii, 2013 [29] | Japan | 4399 | Dietary fiber | BDHQ | Mean: 7.60 ± 0.03 g/1000 kcal | Harmonized definition: 0.68 (0.49–0.93) | Obesity, age, sex, duration of diabetes, current smoking habits, current drinking habits, total energy intake, fat intake, saturated fatty acid intake, protein intake (only for urinary albumin excretion and eGFR), leisure time physical activity and use of oral hypoglycemic agents or insulin | 7 |
| Grooms, 2013 [43] | USA | 10,473 | Dietary fiber | 24 h dietary recall | 22.5–147.6 vs. 0.0–8.1 g/day | ATP-III: 0.78 (0.70–0.88) | Age, sex, race, educational attainment, smoking status, total energy intake | 8 |
| Franco, 2014 [27] | Spain | 1592 | Insoluble fiberSoluble fiber | FFQ | 20.87–56.04 vs. 3.60–14.2 g/day9.02–18.37 vs. 1.70–6.05 g/day | ATP-III: 0.62 (0.40–0.96)ATP-III: 0.87 (0.55–1.39) | Age, gender, level of studies completed type of work, energy intake, physical activity, smoking status and alcohol intake | 8 |
| Study | Country | Case/Participants | Years of Follow-Up | Type of Fiber | Dietary Assessment | Comparison | RR (95% CI) | Variables Accounted for | Score |
|---|---|---|---|---|---|---|---|---|---|
| Hosseinpour-Niazi, 2014 [45] | Iran | 240/1582 | 3 | Total dietary fiber | FFQ | ≥17.5 vs. ≤12.6 g/1000 kcal | ATP-III: 0.86 (0.63–1.21) | Age, gender, physical activity, smoking status, total energy intake, cholesterol, dairy products, meat, poultry, fish, fat intake, protein intake and BMI | 8 |
| Noori, 2010 [46] | Iran | 62/160 (BMI) | 1 | Dietary fiber | FFQ | 39 ± 10 vs. 13 ± 3 g/day | ATP-III: 0.41 (0.08–0.99) | Age, sex, cigarette smoking, physical activity, dialysis mode and its duration before transplantation, cumulative dose of steroids at 1 year post-transplant, menopausal status, family history of diabetes, stroke, energy intake, BMI and intake of magnesium | 7 |
| 58/160 (WC) | |||||||||
| Carnethon, 2004 [47] | USA | 575/4192 | 15 | Crude fiber | Diet history | F(6.9–33)/M(8.6–29.8) vs. F(0.3–2.7)/M(0.6–3.6) g/day | ATP-III: 0.89 (0.68–1.16) | Age, race, sex, BMI, education, smoking, drinking, physical activity, and intake of carbohydrate and fat | 9 |
| N | OR (95% CI) | p | I2 (%) | |
|---|---|---|---|---|
| Region | ||||
| America | 4 | 0.83 (0.71–0.98) | 0.007 | 75.6% |
| Europe | 6 | 0.64 (0.52–0.78) | 0.692 | 0.0% |
| Asia | 2 | 0.60 (0.47–0.77) | 0.281 | 14.0% |
| MetS assessment criteria | ||||
| ATP-III | 8 | 0.69 (0.60–0.79) | 0.234 | 24.4% |
| IDF | 3 | 0.75 (0.52–1.09) | 0.056 | 65.4% |
| Number of cases | ||||
| ≤300 | 5 | 0.94 (0.91–0.96) | 0.420 | 0.0% |
| >300 | 7 | 0.68 (0.59–0.78) | 0.175 | 33.1% |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chen, J.-P.; Chen, G.-C.; Wang, X.-P.; Qin, L.; Bai, Y. Dietary Fiber and Metabolic Syndrome: A Meta-Analysis and Review of Related Mechanisms. Nutrients 2018, 10, 24. https://doi.org/10.3390/nu10010024
Chen J-P, Chen G-C, Wang X-P, Qin L, Bai Y. Dietary Fiber and Metabolic Syndrome: A Meta-Analysis and Review of Related Mechanisms. Nutrients. 2018; 10(1):24. https://doi.org/10.3390/nu10010024
Chicago/Turabian StyleChen, Jia-Ping, Guo-Chong Chen, Xiao-Ping Wang, Liqiang Qin, and Yanjie Bai. 2018. "Dietary Fiber and Metabolic Syndrome: A Meta-Analysis and Review of Related Mechanisms" Nutrients 10, no. 1: 24. https://doi.org/10.3390/nu10010024
APA StyleChen, J.-P., Chen, G.-C., Wang, X.-P., Qin, L., & Bai, Y. (2018). Dietary Fiber and Metabolic Syndrome: A Meta-Analysis and Review of Related Mechanisms. Nutrients, 10(1), 24. https://doi.org/10.3390/nu10010024





