Exploratory Analysis of Dengue Fever Niche Variables within the Río Magdalena Watershed
Abstract
:1. Introduction
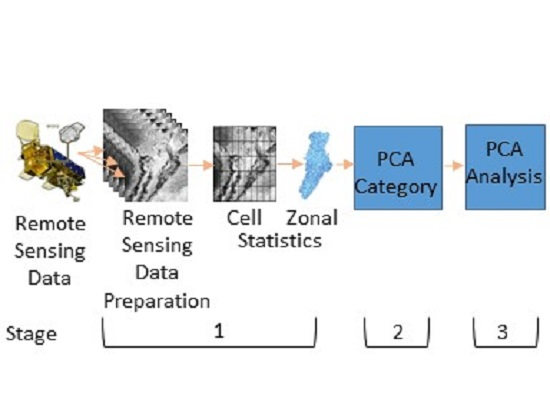
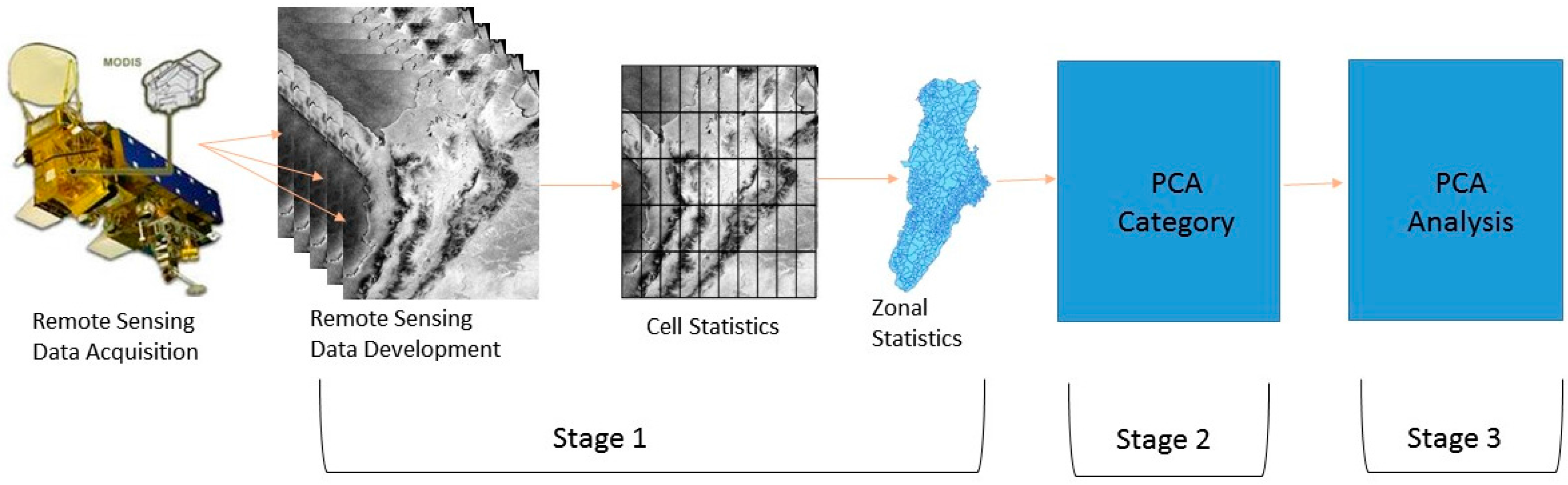
2. Materials and Methods
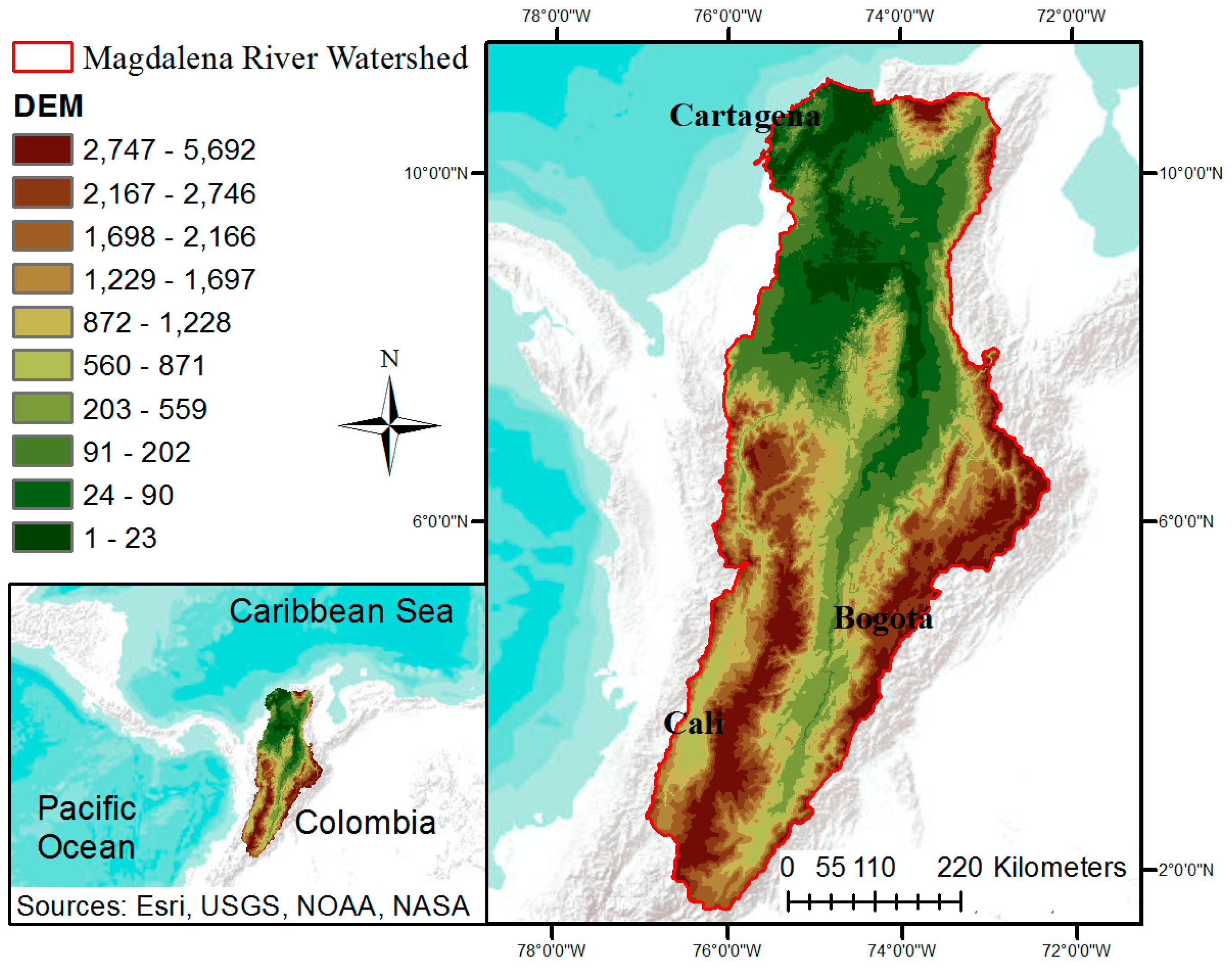
2.1. Study Site
2.2. Population Data
2.3. Remote Sensing Data
2.4. Health Data
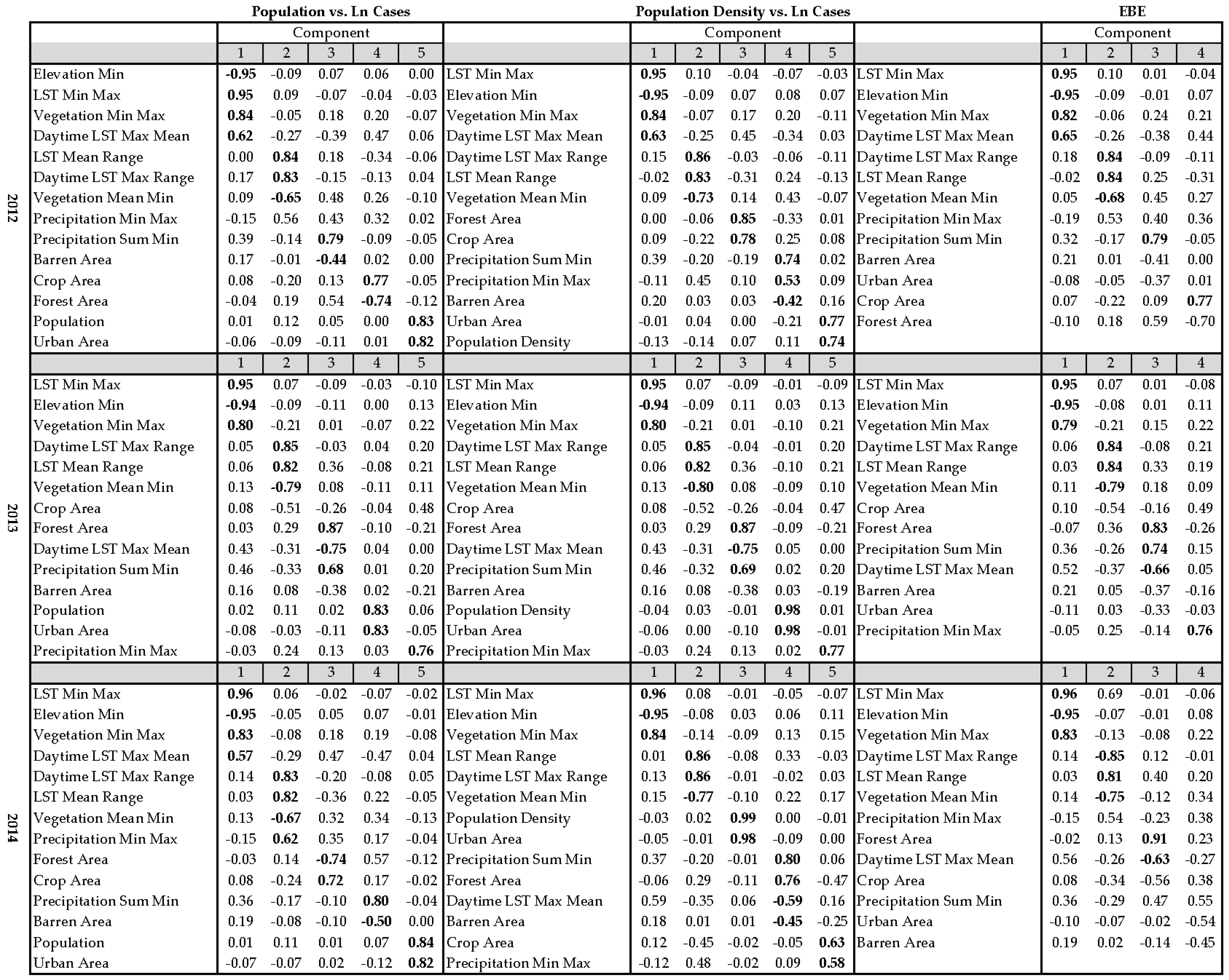
2.5. Principal Component Analysis
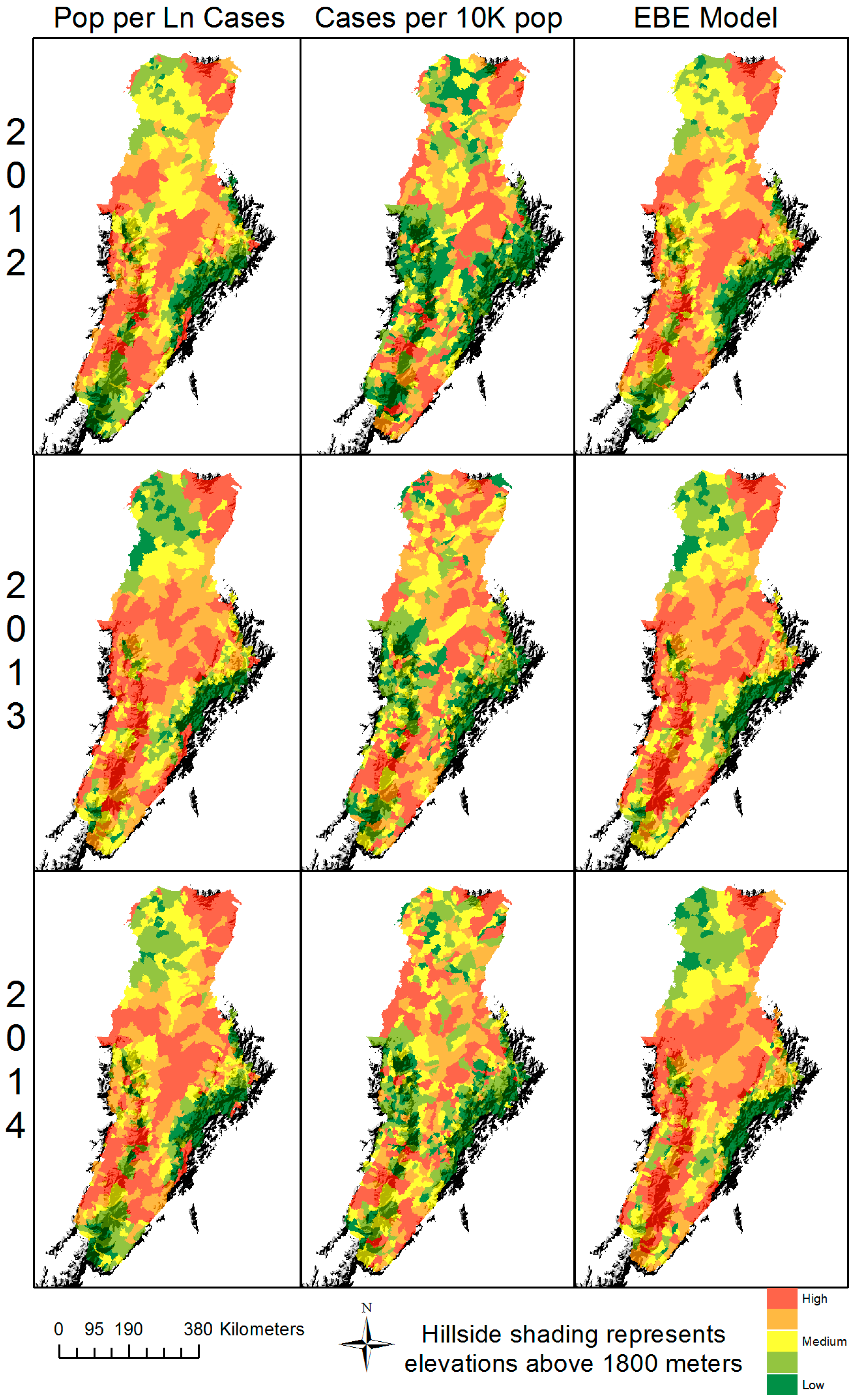
3. Results
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Gubler, D.J. Dengue and dengue hemorrhagic fever. Clin. Microbiol. Rev. 1998, 11, 480–496. [Google Scholar] [PubMed]
- Ceccato, P.; Connor, S.J.; Jeanne, I.; Thomson, M.C. Application of geographical information systems and remote sensing technologies for assessing and monitoring malaria risk. Parassitologia 2005, 47, 81–96. [Google Scholar] [PubMed]
- Moreno-Madriñán, M.J.; Crosson, W.L.; Eisen, L.; Estes, S.M.; Estes, M.G., Jr.; Hayden, M.; Hemmings, S.N.; Irwin, D.E.; Lozano-Fuentes, S.; Monaghan, A.J.; et al. Correlating remote sensing data with the abundance of pupae of the dengue virus mosquito vector, Aedes aegypti, in Central Mexico. Int. J. Geo Inf. 2014, 3, 732–749. [Google Scholar] [CrossRef]
- Cleckner, H.; Allen, T.R. Dasymetric mapping and spatial modeling of mosquito vector exposure, Chesapeake, Virginia, USA. Int. J. Geo Inf. 2014, 3, 891–913. [Google Scholar] [CrossRef]
- Li, Z.; Roux, E.; Dessay, N.; Girod, R.; Stefani, A.; Nacher, M.; Moiret, A.; Seyler, F. Mapping a knowledge-based malaria hazard index related to landscape using remote sensing: Application to the cross-border area between French Guiana and Brazil. Remote Sens. 2016, 8, 319. [Google Scholar] [CrossRef]
- Beebe, N.W.; Cooper, R.D.; Mottram, P.; Sweeney, A.W. Australia’s dengue risk driven by human adaptation to climate change. PLoS Negl. Trop. Dis. 2009, 3, e429. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Weng, Q.; Gaines, D. Spatio-temporal analysis of the relationship between WNV dissemination and environmental variables in Indianapolis, USA. Int. J. Health Geogr. 2008, 7, 66. [Google Scholar] [CrossRef] [PubMed]
- Pinto, E.; Coelho, M.; Oliver, L.; Massad, E. The influence of climate variables on dengue in Singapore. Int. J. Environ. Health Res. 2011, 21, 415–426. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Zhao, J. Spatio-temporal epidemiology of hand, foot and mouth disease in Liaocheng City, North China. Exp. Ther. Med. 2015, 9, 811–816. [Google Scholar] [CrossRef] [PubMed]
- Harlan, S.L.; Brazela, A.J.; Prashada, L.; Stefanovb, W.L.; Larsenc, L. Neighborhood microclimates and vulnerability to heat stress. Soc. Sci. Med. 2006, 63, 2847–2863. [Google Scholar] [CrossRef] [PubMed]
- NASA. MODIS. 2016. Available online: http://www.modis.gsfc.nasa.gov/ (accessed on 4 September 2016). [Google Scholar]
- Fuller, D.; Troyo, A.; Beier, J. El nino southern oscillation and vegetation dynamics as predictors of Dengue Fever cases in Costa Rica. Environ. Res. Lett. 2009, 4, 140111–140118. [Google Scholar] [CrossRef] [PubMed]
- Famine Early Warning Sysems Network - FEWS NET. Available online: http://www.fews.net (accessed on 14 September 2016).
- Thomson, M.C.; Conn, S.J. Malaria Early Warning Systems—A Framework for Field Research in Africa; World Health Organization: Geneva, Switzerland, 2001. [Google Scholar]
- Brown, M.E.; Brickley, E.B. Evaluating the use of remote sensing data in the US agency for international development famine early warning systems network. J. Appl. Remote Sens. 2012, 6, 063511. [Google Scholar]
- NASA Applied Sciences Projects Improve Famine Predictions Worldwide. Available online: http://www.nasa.gov/topics/earth/features/famine.html (accessed on 14 September 2016).
- CDC. Dengue. 19 January 2016. Available online: http://www.cdc.gov/dengue/ (accessed on 2 February 2016). [Google Scholar]
- Lozano-Fuentes, S.; Hayden, M.H.; Welsh-Rodriguez, C.; Ochoa-Martinez, C.; Tapia-Santos, B.; Kobylinski, K.C.; Uejio, C.K.; Zielinski-Gutierrez, E.; Monache, L.D.; Monaghan, A.J.; et al. The dengue virus mosquito vector Aedes aegypti at high elevation in Mexico. Am. J. Trop. Med. Hyg. 2012, 87, 902–909. [Google Scholar] [CrossRef] [PubMed]
- Dengue and Severe Dengue. Available online: http://www.who.int/mediacentre/factsheets/fs117/en/ (accessed on 2 February 2016).
- Delmelle, E.; Dony, C.; Casas, I.; Jia, M.; Tang, W. Visualizing the impact of space-time uncertainties on Dengue Fever patterns. Int. J. Geogr. Inf. Sci. 2014, 28, 1107–1127. [Google Scholar] [CrossRef]
- Brunkard, J.M.; Cifuentes, E.; Rothenberg, S.J. Assessing the roles of temperature, precipitation, and ENSO in dengue re-emergence on the Texas-Mexico border region. Salud Pública Méx. 2008, 50, 227–234. [Google Scholar] [CrossRef] [PubMed]
- Kraemer, M.U.; Sinka, M.E.; Duda, K.A.; Mylne, A.Q.N.; Shearer, F.M.; Barker, C.M.; Moore, C.G.; Carvalho, R.G.; Coelho, G.E.; van Bortel, W.; et al. The global distribution of the arbovirus vectors Aedes aegypti and Ae. albopictus. Elife 2015, 4, e08347. [Google Scholar] [CrossRef] [PubMed]
- Mylne, A.Q.; Pigott, D.M.; Longbottom, J.; Shearer, F.; Duda, K.A.; Messina, J.P.; Weiss, D.J.; Moyes, C.L.; Golding, N.; Hay, S.I. Mapping the zoonotic niche of lassa fever in Africa. Trans. R. Soc. Trop. Med. Hyg. 2015, 109, 483–492. [Google Scholar] [CrossRef] [PubMed]
- Pigott, D.M.; Golding, N.; Mylne, A.; Huang, Z.; Henry, A.J.; Weiss, D.J.; Brady, O.J.; Kraemer, M.U.G.; Smith, D.L.; Moyes, C.L.; et al. Mapping the zoonotic niche of Ebola virus disease in Africa. Elife 2014, 3, e04395. [Google Scholar] [CrossRef] [PubMed]
- Pigott, D.M.; Golding, N.; Mylne, A.; Huang, Z.; Weiss, D.J.; Brady, O.J.; Kraemer, M.U.; Hay, S.I. Mapping the zoonotic niche of Marburg virus disease in Africa. Trans. R. Soc. Trop. Med. Hyg. 2015, 109, 366–378. [Google Scholar] [CrossRef] [PubMed]
- Eastin, M.D.; Delmelle, E.; Casas, I.; Wexler, J.; Self, C. Intra-and interseasonal autoregressive prediction of dengue outbreaks using local weather and regional climate for a tropical environment in Colombia. Am. J. Trop. Med. Hyg. 2014, 91, 598–610. [Google Scholar] [CrossRef] [PubMed]
- Lambrechts, L.; Paaijmans, K.P.; Fansiri, T.; Carrington, L.B.; Kramer, L.D.; Thomas, M.B.; Scott, T.W. Impact of daily temperature fluctuations on dengue virus transmission by Aedes aegypti. Proc. Natl. Acad. Sci. USA 2011, 108, 7460–7465. [Google Scholar] [CrossRef] [PubMed]
- Brady, O.J.; Johansson, M.A.; Guerra, C.A.; Bhatt, S.; Golding, N.; Pigott, D.M.; Delatte, H.; Grech, M.G.; Leisnham, P.T.; Maciel-de-Freitas, R.; et al. Modelling adult Aedes aegypti and Aedes albopictus survival at different temperatures in laboratory and field settings. Parasites Vectors 2013, 6, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Miron, I.J.; Montero, J.C.; Criado-Alvarez, J.J.; Díaz, J.; Linares, C. Effects of temperature extremes on daily mortality in Castile-La Mancha (Spain): Trends from 1975 to 2003. Gac. Sanit. 2010, 24, 117–122. [Google Scholar] [PubMed]
- Rueda, L.; Patel, K.J.; Axtell, R.C.; Stinner, R.E. Temperature-dependent development and survival rates of Culex quinquefasciatus and Aedes aegypti (Diptera: Culicidae). J. Med. Entomol. 1990, 27, 892–898. [Google Scholar] [CrossRef] [PubMed]
- Ibanez-Bernal, S. New altitudinal record of Aedes(Stegomyia) aegypti(Linnaeus, 1762)(Diptera: Culicidae) in Mexico. Folia Entomol. Mex. 1987, 72, 163–164. [Google Scholar]
- Kanaroglou, P.; Delmelle, E. Spatial Analysis in Health Geography; Routledge: New York, NY, USA, 2015; p. 344. [Google Scholar]
- Suárez, M.F.; Nelson, M.J. Registro de altitud del Aedes aegypti en Colombia. Biomédica 1981, 1, 225. [Google Scholar] [CrossRef]
- Stanforth, A.C. Identifying Variations of Socio-Spatial Vulnerability to Heat-Related Mortality during the 1995 Extreme Heat Event in Chicago, IL, USA; Indiana University: Bloomington, IN, USA, 2011. [Google Scholar]
- StatSoft. Electronic Statistics Textbook; StatSoft: Tulsa, OK, USA, 2015. [Google Scholar]
- DeCoster, J. Data Analysis in SPSS. 21 February 2004. Available online: https://msu.edu/course/psy/818/snapshot.afs/deshon/Projects/Misc%20files/spss%20overview.pdf (accessed on 18 September 2016).
- Kaiser, H.F. The application of electronic computers to factor analysis. Educ. Psychol. Meas. 1960, 20, 141–150. [Google Scholar] [CrossRef]
- Idre. Annotated SPSS Output—Principal Components Analysis. 2016. Available online: http://www.ats.ucla.edu/stat/spss/output/principal_components.htm (accessed on 19 September 2016).
- Lin, C.-H.; Wen, T.-H. Using geographically weighted regression (GWR) to explore spatial varying relationships of immature mosquitoes and human densities with the incidence of dengue. Int. J. Environ. Res. Public Health 2011, 8, 2798–2815. [Google Scholar] [CrossRef] [PubMed]
- UCLA, Introduction to SAS. UCLA: Statistcal Consulting Group. Available online: http://www.ats.ucla.edu/stat/SPSS/output/principal_components.htm (accessed on 14 September 2016).
- Morin, C.W.; Comrie, A.C.; Ernst, K. Climate and dengue transmission: Evidence and implications. Environ. Health Perspect. 2013, 121, 1264–1272. [Google Scholar] [CrossRef] [PubMed]
- Venkatesan, M.; Rasgon, J.L. Population genetic data suggest a role for mosquito-mediated dispersal of west nile virus across the Western United States. Mol. Ecol. 2010, 19, 1573–1584. [Google Scholar] [CrossRef] [PubMed]
- DANE, Colombia. Proyecciones De Población Por Área 2005–2020. 2010; El Departamento Administrativo Nacional de Estadística. Available online: http://www.dane.gov.co/index.php/poblacion-y-demografia/proyecciones-de-poblacion (accessed on 10 September 2016). [Google Scholar]
- ArcGIS, version 10.3; Esri: Redlands, CA, USA, 2014.
- Earth Explorer. Available online: http://earthexplorer.usgs.org (accessed on 14 September 2016).
- NASA. Satellite Data was obtained from NASA. Available online: http://disc2.nascom.nasa.gov (accessed on 1 November 2015).
- U.S.G. Survey (USGS). Remote Sensing Data; U.S.G. Survey: Reston, VA, USA, 2016.
- Python, version 2.7; Python Software Foundation: Wilmington, DE, USA, 2010.
- Salano, R.; Didan, K.; Jacobson, A.; Huete, A. MODIS Vegetation Index User’s Guide (MOD13 Series); The University of Arizona: Tucson, AZ, USA, 2010; p. 42. [Google Scholar]
- Jensen, J. Introductory Remote Sensing—A Remote Sensing Perspective, 3rd ed.; Prentice Hall: Upper Saddle River, NJ, USA, 2005. [Google Scholar]
- NASA. TRMM—Tropical Rainfall Measuring Mission. 18 May 2016. Available online: http://www.trmm.gsfc.nasa.gov/ (accessed on 23 May 2016). [Google Scholar]
- Liu-Helmersson, J.; Stenlund, H.; Wilder-Smith, A.; Rocklöv, J. Vectorial capacity of Aedes aegypti: Effects of temperature and implications for global dengue epidemic potential. PLoS ONE 2014, 9, e89783. [Google Scholar] [CrossRef] [PubMed]
- Vigilancia Rutinaria. Available online: http://www.ins.gov.co/lineas-de-accion/Subdireccion-Vigilancia/sivigila/Paginas/vigilancia-rutinaria.aspx (accessed on 14 September 2016).
- SPSS Statistical Software, version 23; IBM Corp.: New Castle, NY, USA, 2016.
- Null, J. El Niño and La Niña Years and Intensities. Available online: http://www.ggweather.com/enso/oni.htm (accessed on 4 August 2016).
- Brandel, J. Empirical Bayes Methods for Missing Data Analysis; Department of Mathematics, Uppsala University: Uppsala, Sweden, 2004. [Google Scholar]
- Woodward, M. Epidemiology: Study Design and Data Analysis; CRC Press: Boca Raton, FL, USA, 2013. [Google Scholar]
- Adjacency For WinBUGS Tool; USGS: Menlo Park, CA, USA, 2012.
- Costello, A.B.; Osborne, J.W. Best practices in exploratory factor analysis: Four recommendations for getting the most from your analysis. Pract. Assess. Res. Eval. 2005, 10, 1. [Google Scholar]
- Cutter, S.L.; Barnes, L.; Berry, M.; Burton, C.; Evans, E.; Tate, E.; Webb, J. A place-based model for understanding community resilience to natural disasters. Glob. Environ. Chang. Hum. Policy Dimens. 2008, 18, 598–606. [Google Scholar] [CrossRef]
- Cutter, S.L.; Boruff, B.J.; Shirley, W. Social vulnerability to environmental hazards. Soc. Sci. Q. 2003, 84, 242–261. [Google Scholar] [CrossRef]
- Cutter, S.L.; Burton, C.G.; Emrich, C.T. Disaster resilience indicators for benchmarking baseline conditions. J. Homel. Secur. Emerg. Manag. 2010, 7, 1547–7355. [Google Scholar] [CrossRef]
- Cutter, S.L.; Finch, C. Temporal and spatial changes in social vulnerability to natural hazards. Proc. Natl. Acad. Sci. USA 2008, 105, 2301–2306. [Google Scholar] [CrossRef] [PubMed]
- Johnson, D.P.; Stanforth, A.; Lulla, V.; Luber, G. Developing an applied extreme heat vulnerability index utilizing socioeconomic and environmental data. Appl. Geogr. 2012, 35, 23–31. [Google Scholar] [CrossRef]
- O’Neill, M.S.; Zanobetti, A.; Schwartz, J. Disparities by race in heat-related mortality in four US cities: The role of air conditioning prevalence. J. Urban Health 2005, 82, 191–197. [Google Scholar] [CrossRef] [PubMed]
- Tape, T.G. Interpreting Diagnostic Tests; University of Nebraska Medical Center: Omaha, NE, USA, 2006. [Google Scholar]
- Carrington, L.B.; Seifert, S.N.; Willits, N.H.; Lambrecht, L.; Scott, T.W. Large diurnal temperature fluctuations negatively influence Aedes aegypti (Diptera: Culicidae) life-history traits. J. Med. Entomol. 2013, 50, 43–51. [Google Scholar] [CrossRef] [PubMed]
- Mohammed, A.; Chadee, D.D. Effects of different temperature regimens on the development of Aedes aegypti (L.) (Diptera: Culicidae) mosquitoes. Acta Trop. 2011, 119, 38–43. [Google Scholar] [CrossRef] [PubMed]
- Reid, C.E.; O’Neill, M.S.; Gronlund, C.J.; Brines, S.J.; Brown, D.G.; Diez-Roux, A.V.; Schwartz, J. Mapping community determinants of heat vulnerability. Environ. Health Perspect. 2009, 117, 1730–1736. [Google Scholar] [CrossRef] [PubMed]
- Curtis, A.; Ye, X.; Heob, E.; Targhetta, J.; Salvato, V.; Reyna, M.; Bueno, R., Jr.; Holmes, L. A comparison of three approaches to identify west nile virus mosquito space-time hotspots in the houston vicinity for the period 2002–2011. Appl. Geogr. 2014, 51, 58–64. [Google Scholar] [CrossRef]
- Lounibos, L.P. Invasions by insect vectors of human disease. Annu. Rev. Entomol. 2002, 47, 233–266. [Google Scholar] [CrossRef] [PubMed]
- Ruiz-López, F.; González-Mazo, A.; Vélez-Mira, A.; Gómez, G.F.; Zuleta, L.; Uribe, S.; Vélez-Bernal, I.D. Presence of Aedes (Stegomyia) aegypti (Linnaeus, 1762) and its natural infection with dengue virus at unrecorded heights in Colombia. Biomédica 2016, 36, 303–308. [Google Scholar] [CrossRef] [PubMed]
- Vogelmann, J.E.; Howard, S.M.; Yang, L.; Larson, C.R.; Wylie, B.K.; van Driel, J.N. Completion of the 1990’s national land cover data set for the conterminous United States. Photogramm. Eng. Remote Sens. 2001, 67, 650–662. [Google Scholar]
- Moreno-Madriñán, M.J.; Turell, M.J. Factors of concern regarding Aedes-transmitted viruses in the U.S. J. Med. Entomology 2016, in press. [Google Scholar]
- Kwan, M.-P. The uncertain geographic context problem. Ann. Assoc. Am. Geogr. 2012, 102, 958–968. [Google Scholar] [CrossRef]
- Kwan, M.-P. How GIS can help address the uncertain geographic context problem in social science research. Ann. GIS 2012, 18, 245–255. [Google Scholar] [CrossRef]
| Projected Population | New Reported Cases | New Cases Rate | |
|---|---|---|---|
| 2012 | 35,375,287 | 15,811 | 0.45/1000 people |
| 2013 | 35,751,512 | 36,977 | 1.03/1000 people |
| 2014 | 36,127,443 | 24,949 | 0.69/1000 people |
| 2012 | 2013 | 2014 | ||
|---|---|---|---|---|
| Population | # Components | 5 | 5 | 5 |
| Cumulative Variance | 74.224 | 73.403 | 74.07 | |
| Population Density | # Components | 5 | 5 | 5 |
| Cumulative Variance | 72.283 | 77.347 | 77.966 | |
| EBE | # Components | 4 | 4 | 4 |
| Cumulative Variance | 69.622 | 68.674 | 69.599 |
| Model | Primary Results AUC | TOP 2 Component AUC | |
|---|---|---|---|
| 2012 | Population | 0.6907276 | N/A |
| Density | 0.6856312 | 0.6733368 | |
| EBE | 0.6656901 | 0.6733368 | |
| 2013 | Population | 0.6612869 | N/A |
| Density | 0.6623720 | 0.6981112 | |
| EBE | 0.6656901 | 0.6981112 | |
| 2014 | Population | 0.6612869 | 0.7389620 |
| Density | 0.7122281 | 0.7341314 | |
| EBE | 0.6587307 | 0.7341314 |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Stanforth, A.; Moreno-Madriñán, M.J.; Ashby, J. Exploratory Analysis of Dengue Fever Niche Variables within the Río Magdalena Watershed. Remote Sens. 2016, 8, 770. https://doi.org/10.3390/rs8090770
Stanforth A, Moreno-Madriñán MJ, Ashby J. Exploratory Analysis of Dengue Fever Niche Variables within the Río Magdalena Watershed. Remote Sensing. 2016; 8(9):770. https://doi.org/10.3390/rs8090770
Chicago/Turabian StyleStanforth, Austin, Max J. Moreno-Madriñán, and Jeffrey Ashby. 2016. "Exploratory Analysis of Dengue Fever Niche Variables within the Río Magdalena Watershed" Remote Sensing 8, no. 9: 770. https://doi.org/10.3390/rs8090770
APA StyleStanforth, A., Moreno-Madriñán, M. J., & Ashby, J. (2016). Exploratory Analysis of Dengue Fever Niche Variables within the Río Magdalena Watershed. Remote Sensing, 8(9), 770. https://doi.org/10.3390/rs8090770