Diel and Spatial Dependence of Humpback Song and Non-Song Vocalizations in Fish Spawning Ground
Abstract
:1. Introduction
2. Materials and Methods
2.1. The Gulf of Maine 2006 Experiment Data Collection
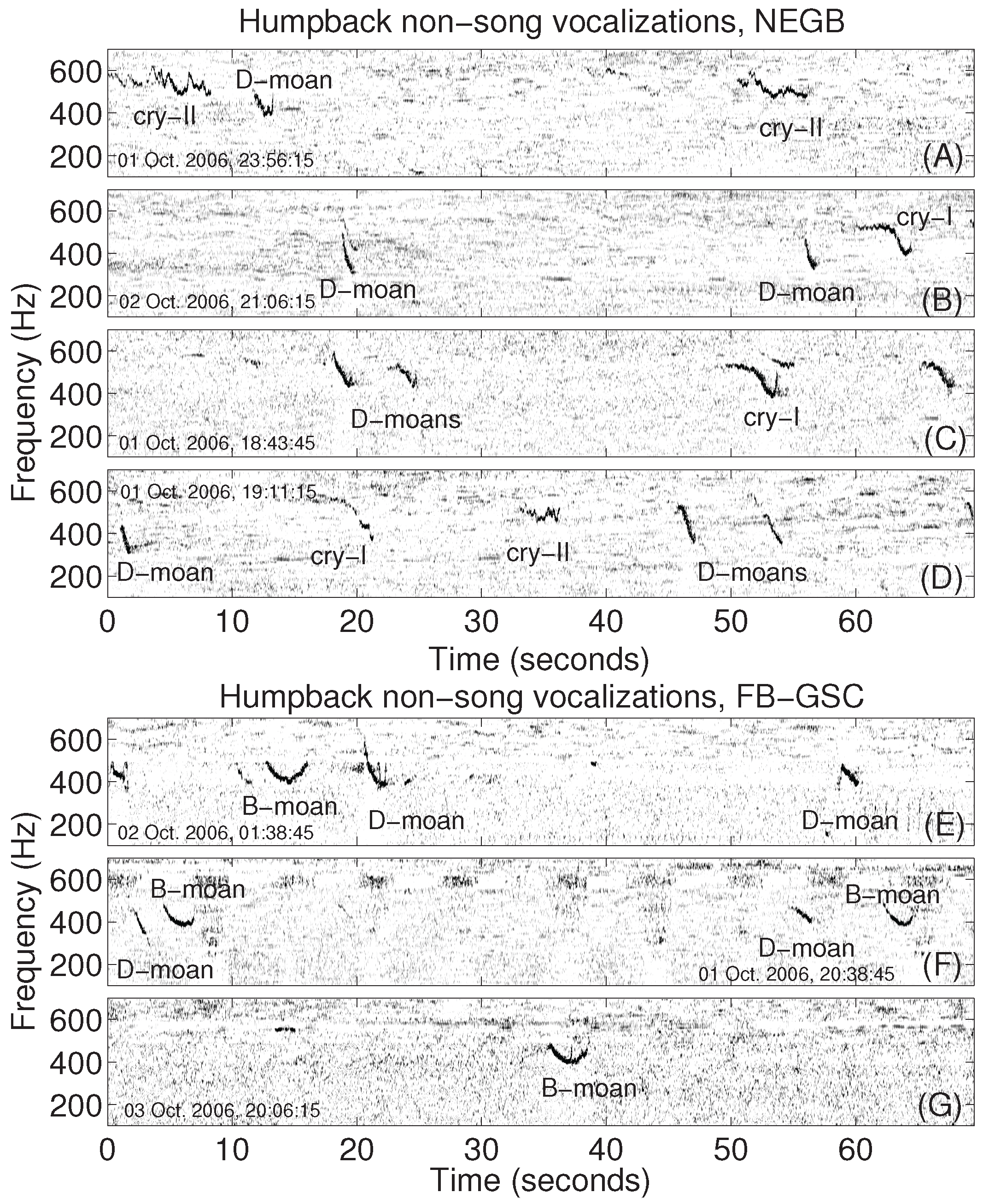
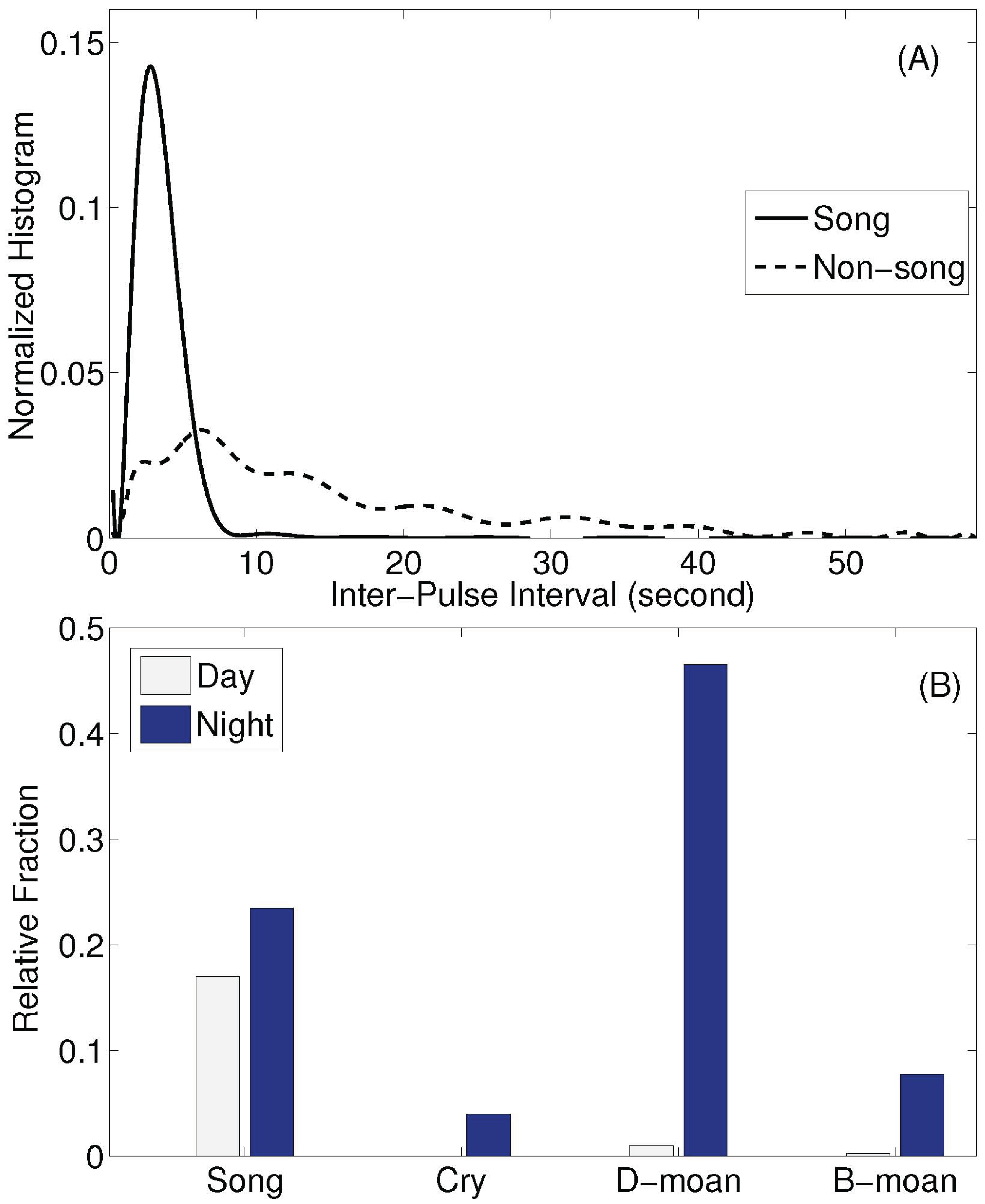
2.2. Analysis of Humpback Song and Non-Song Vocalizations
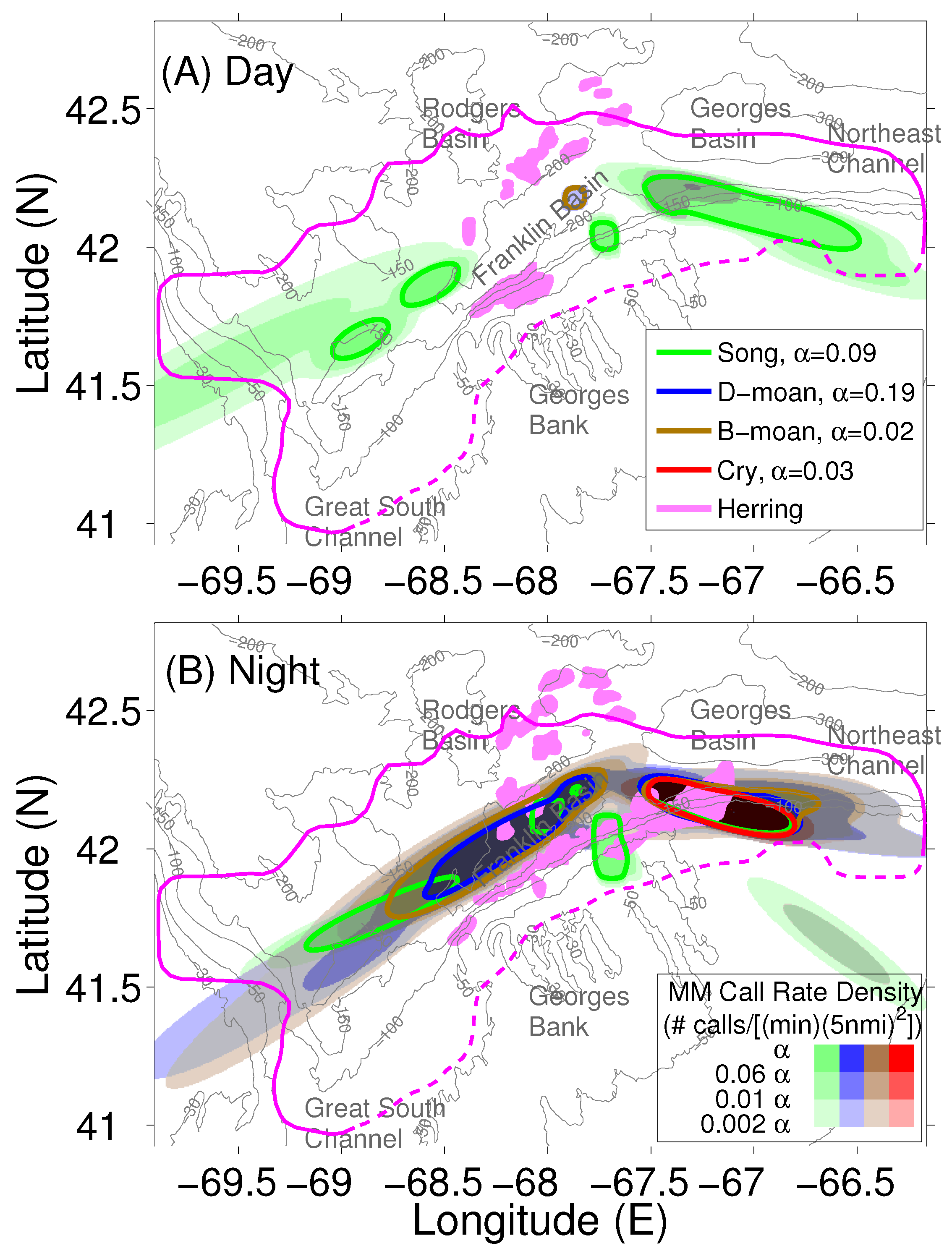
2.3. Localization and Call Rate Spatial Distributions of Humpback Song and Non-Song Vocalization Types
2.4. Atlantic Herring Areal Population Density Distribution
2.5. Determination of Humpback Cummulative Call Rate Spatial Distributions for Different Vocalization Types
2.6. Source Levels of Humpback Song and Non-Song Vocalization Types
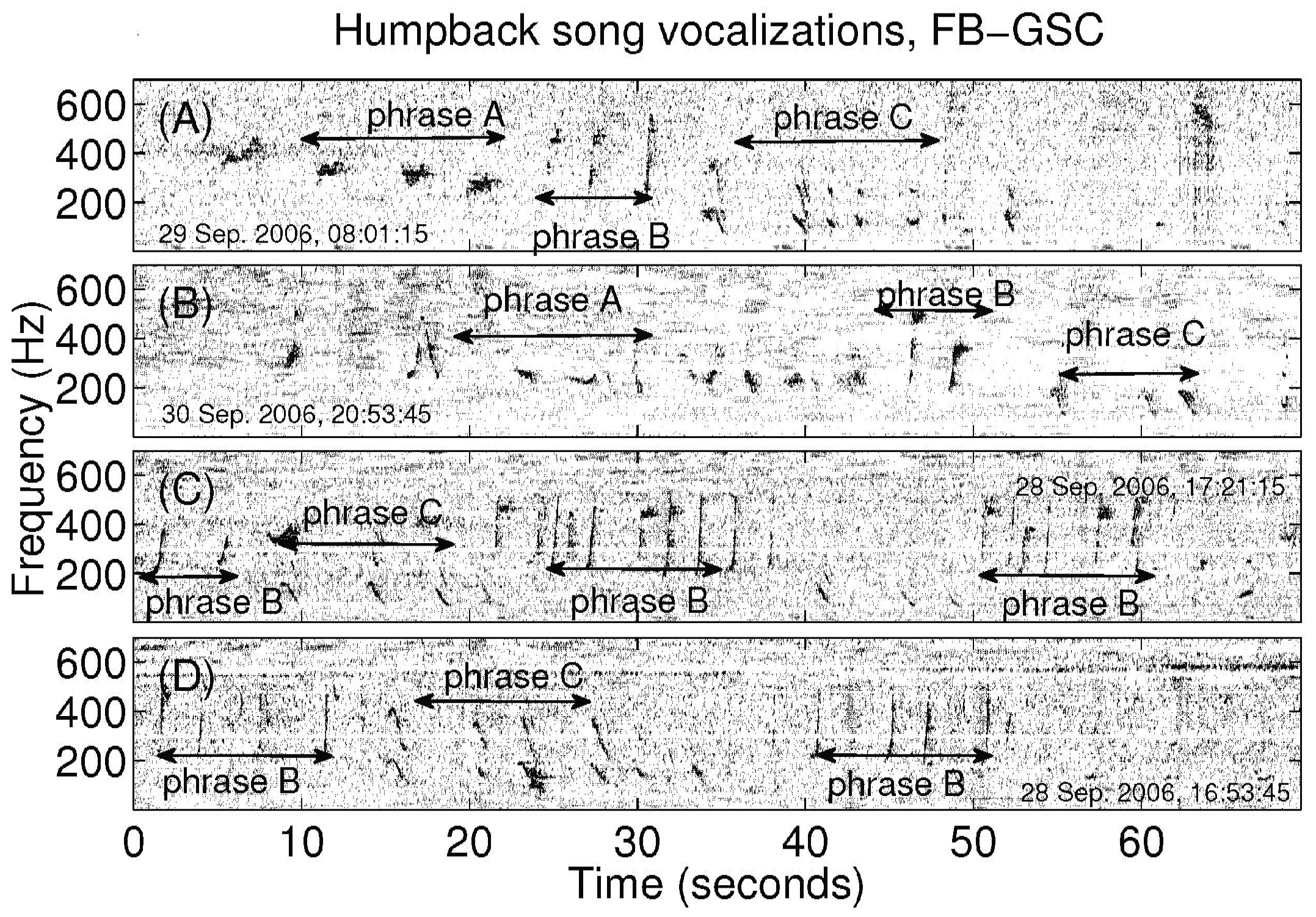
3. Results
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| POAWRS | Passive Ocean Acoustic Waveguide Remote Sensing |
| OAWRS | Ocean Acoustic Waveguide Remote Sensing |
| D-moan | Downsweep moan or meow |
| B-moan | Bow-shaped moan |
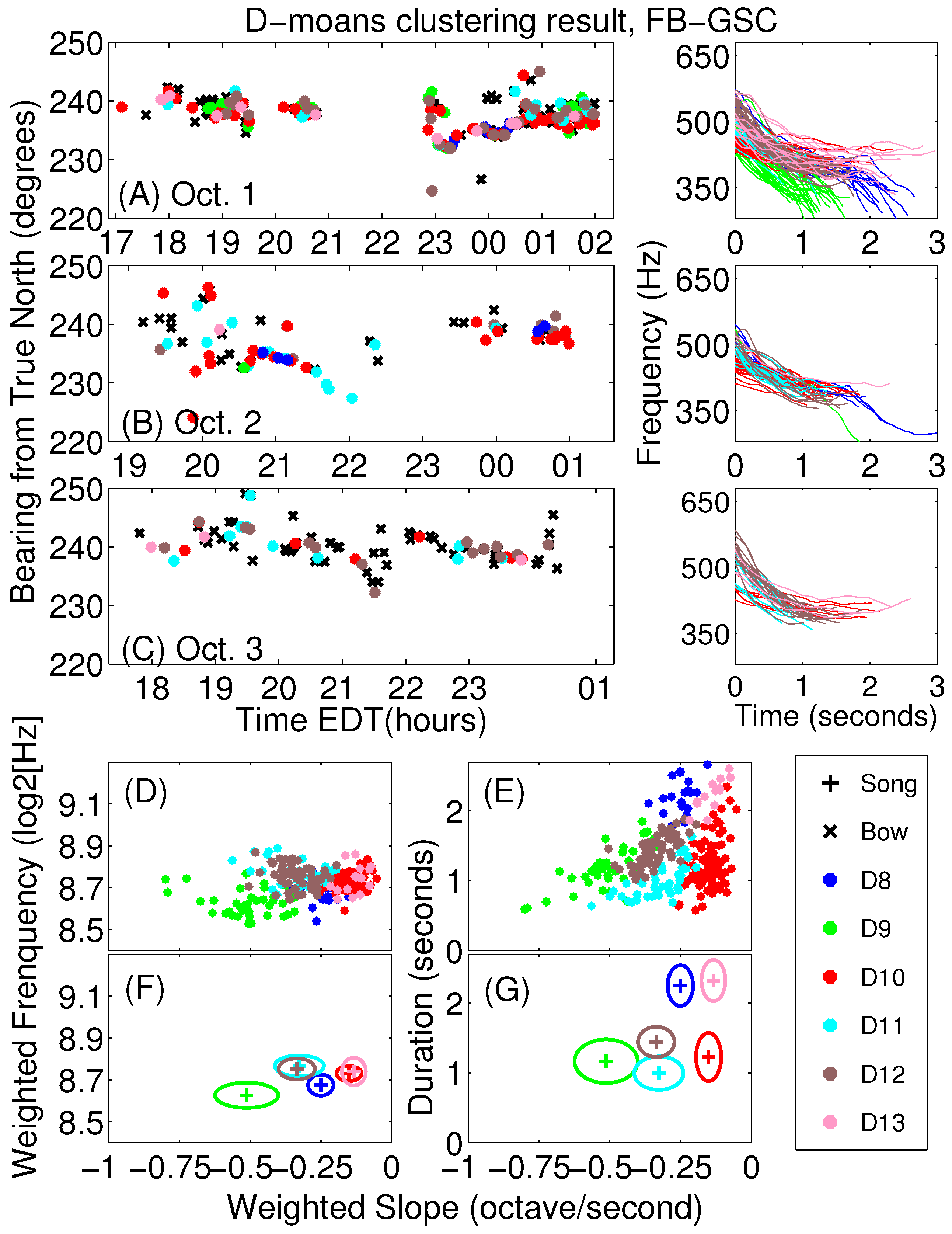
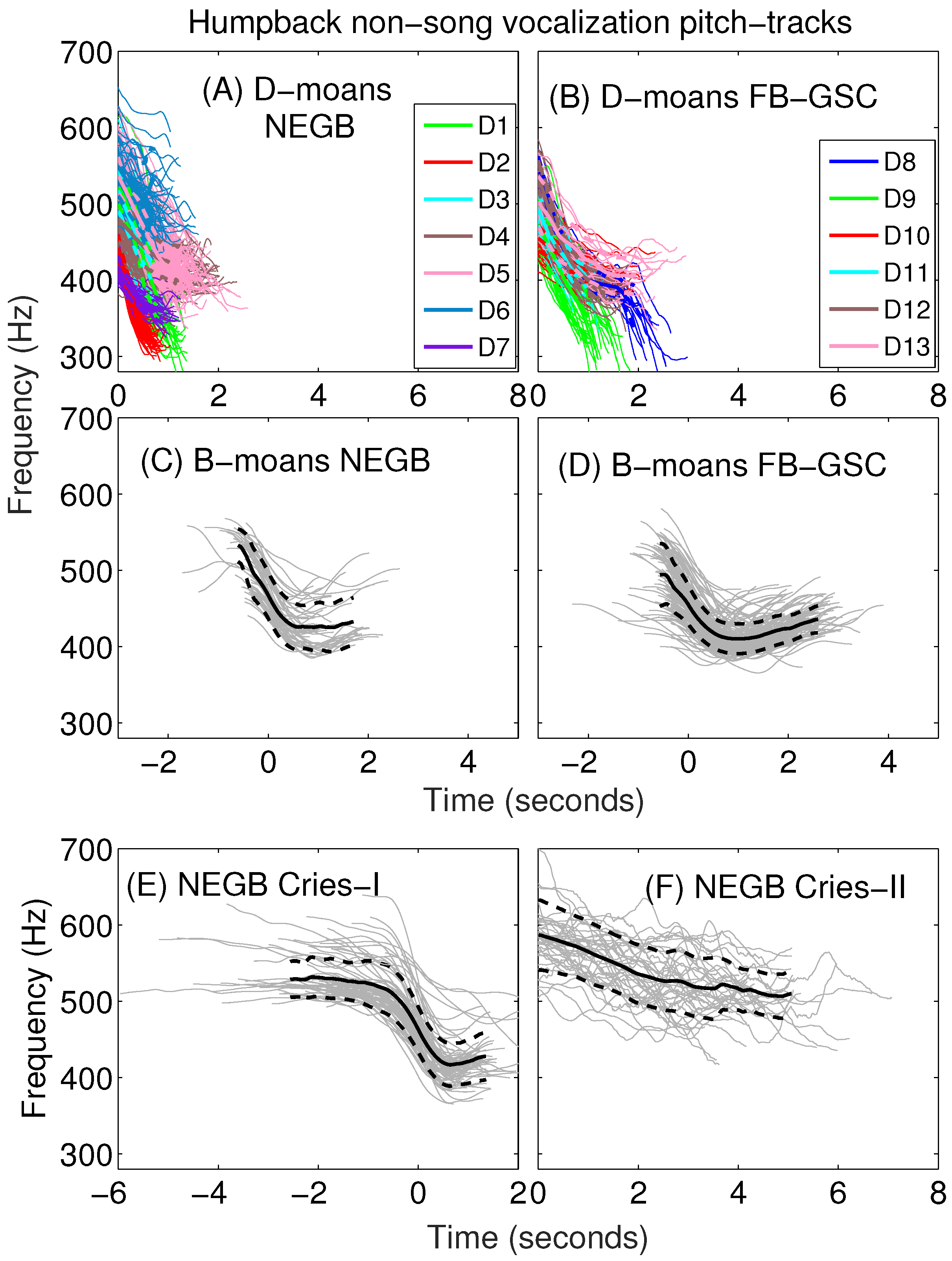
Appendix A. Sub-Classification of Humpback Non-Song D-Moan Vocalizations
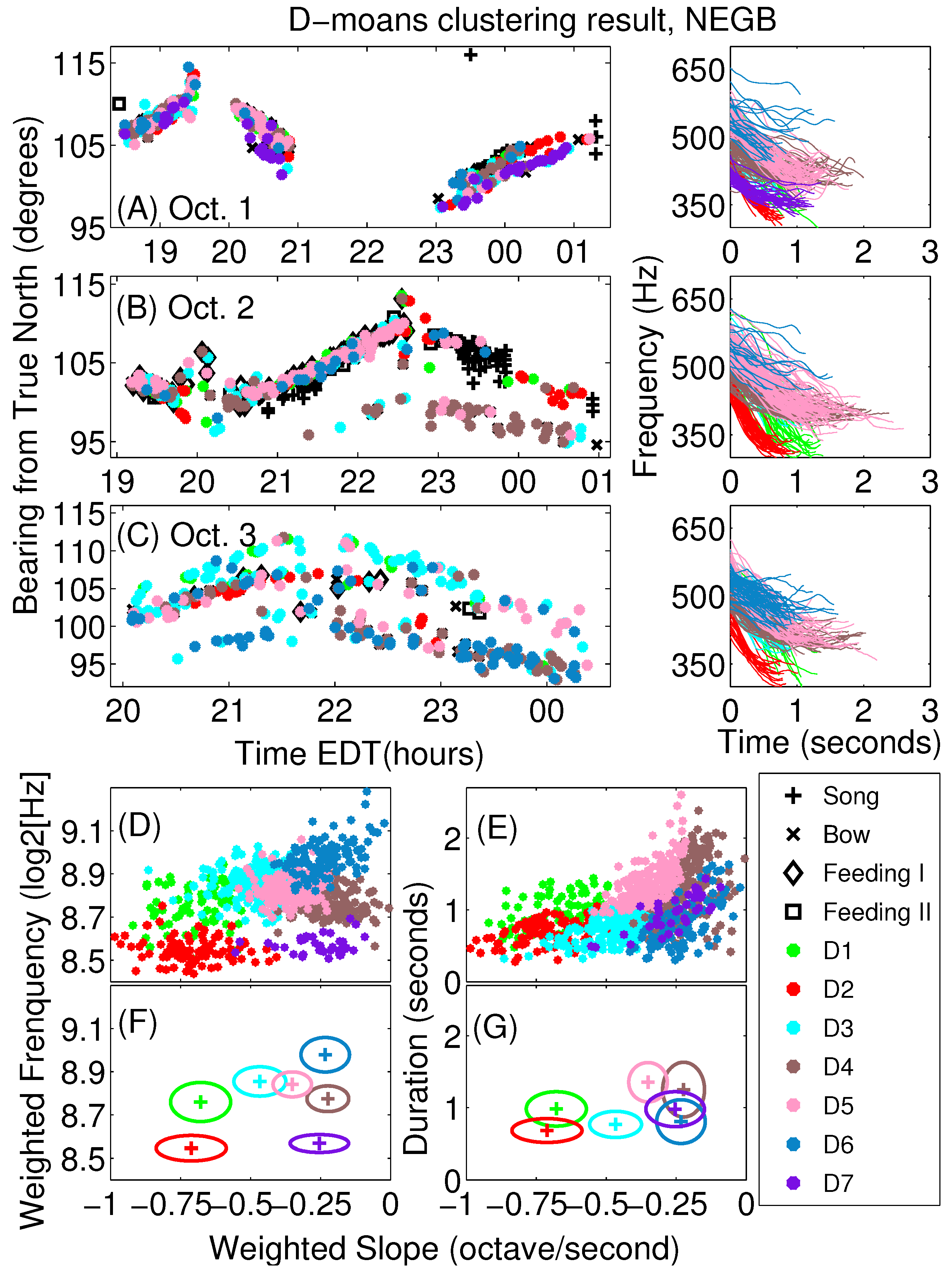
| β | τ | ν | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| D1 | 346 (31) | 534 (30) | 189 (35) | −0.68 (0.11) | 8.76 (0.09) | 0.30 (0.07) | 0.20 (0.04) | 0.98 (0.24) | −203 (31) | −22 (148) |
| D2 | 328 (19) | 452 (21) | 124 (30) | −0.71 (0.13) | 8.55 (0.06) | 0.21 (0.05) | 0.15 (0.03) | 0.68 (0.17) | −188 (34) | 192 (122) |
| D3 | 415 (24) | 527 (28) | 112 (24) | −0.47 (0.10) | 8.85 (0.07) | 0.24 (0.05) | 0.11 (0.02) | 0.77 (0.18) | −151 (30) | 60 (137) |
| D4 | 404 (18) | 492 (26) | 88 (25) | −0.22 (0.07) | 8.77 (0.06) | 0.38 (0.11) | 0.09 (0.03) | 1.25 (0.38) | −69 (23) | 50 (82) |
| D5 | 402 (23) | 548 (26) | 146 (22) | −0.35 (0.07) | 8.84 (0.06) | 0.41 (0.08) | 0.14 (0.02) | 1.35 (0.27) | −113 (22) | 58 (55) |
| D6 | 475 (32) | 537 (31) | 63 (22) | −0.23 (0.09) | 8.98 (0.08) | 0.25 (0.09) | 0.06 (0.02) | 0.81 (0.30) | −81 (29) | −5 (110) |
| D7 | 354 (14) | 424 (17) | 70 (16) | −0.25 (0.10) | 8.57 (0.04) | 0.29 (0.07) | 0.08 (0.02) | 0.98 (0.24) | −68 (28) | 86 (68) |
| Average | 400 (45) | 514 (44) | 114 (43) | −0.39 (0.19) | 8.80 (0.13) | 0.31 (0.11) | 0.12 (0.05) | 1.02 (0.38) | −121 (54) | 57 (119) |
| β | τ | ν | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| D8 | 323 (30) | 533 (26) | 210 (43) | −0.25 (0.04) | 8.68 (0.05) | 0.69 (0.03) | 0.18 (0.03) | 2.25 (0.29) | −73 (13) | 25 (17) |
| D9 | 316 (25) | 476 (33) | 160 (35) | −0.51 (0.11) | 8.63 (0.06) | 0.36 (0.03) | 0.18 (0.04) | 1.17 (0.31) | −140 (34) | 7 (96) |
| D10 | 399 (14) | 455 (16) | 56 (16) | −0.15 (0.04) | 8.73 (0.04) | 0.37 (0.10) | 0.06 (0.02) | 1.23 (0.34) | −44 (13) | 1 (26) |
| D11 | 393 (18) | 492 (21) | 99 (21) | −0.33 (0.09) | 8.77 (0.05) | 0.31 (0.07) | 0.10 (0.02) | 1.00 (0.24) | −99 (28) | 31 (78) |
| D12 | 381 (16) | 535 (21) | 155 (21) | −0.34 (0.06) | 8.75 (0.05) | 0.44 (0.07) | 0.15 (0.02) | 1.44 (0.22) | −103 (20) | 83 (37) |
| D13 | 393 (25) | 505 (30) | 112 (21) | −0.13 (0.04) | 8.74 (0.07) | 0.70 (0.08) | 0.10 (0.02) | 2.32 (0.30) | −42 (12) | 28 (15) |
| Average | 376 (37) | 491 (40) | 115 (56) | −0.28 (0.14) | 8.72 (0.07) | 0.42 (0.15) | 0.11 (0.05) | 1.38 (0.49) | −82 (41) | 28 (62) |
References
- Wang, D.; Garcia, H.; Huang, W.; Tran, D.D.; Jain, A.D.; Yi, D.H.; Gong, Z.; Jech, J.M.; Godoe, O.R.; Makris, N.C.; Ratilal, P. Vast assembly of vocal marine mammals from diverse species on fish spawning ground. Nature 2016, 531, 366–370. [Google Scholar] [CrossRef] [PubMed]
- Gong, Z.; Jain, A.D.; Tran, D.D.; Yi, D.H.; Wu, F.; Zorn, A.; Ratilal, P.; Makris, N.C. Ecosystem scale acoustic sensing reveals humpback whale behavior synchronous with herring spawning processes and re-evaluation finds no effect of sonar on humpback song occurrence in the Gulf of Maine in Fall 2006. PLoS ONE 2014, 9, e104733. [Google Scholar] [CrossRef] [PubMed]
- Tran, D.D.; Huang, W.; Bohn, A.C.; Wang, D.; Gong, Z.; Makris, N.C.; Ratilal, P. Using a coherent hydrophone array for observing sperm whale range, classification, and shallow-water dive profiles. J. Acoust. Soc. Am. 2014, 135, 3352–3363. [Google Scholar] [CrossRef] [PubMed]
- Kay, S.M. Fundamentals of statistical signal processing, Vol. II: Detection Theory. In Signal Processing; Prentice-Hall: Upper Saddle River, NJ, USA, 1998; Volume 7. [Google Scholar]
- Payne, R.S.; McVay, S. Songs of Humpback Whales. Science 1971, 173, 585–597. [Google Scholar] [CrossRef] [PubMed]
- Noad, M.J.; Cato, D.H.; Bryden, M.M.; Jenner, M.N.; Jenner, K.C.S. Cultural revolution in whale songs. Nature 2000, 408, 537–537. [Google Scholar] [CrossRef] [PubMed]
- Winn, H.E.; Thompson, T.J.; Cummings, W.C.; Hain, J.; Hudnall, J.; Hays, H.; Steiner, W.W. Song of the humpback whale—Population comparisons. Behav. Ecol. Sociobiol. 1981, 8, 41–46. [Google Scholar] [CrossRef]
- Mattila, D.K.; Guinee, L.N.; Mayo, C.A. Humpback whale songs on a North Atlantic feeding ground. J. Mammal. 1987, 68, 880–883. [Google Scholar] [CrossRef]
- Gong, Z.; Tran, D.D.; Ratilal, P. Comparing passive source localization and tracking approaches with a towed horizontal receiver array in an ocean waveguide. J. Acoust. Soc. Am. 2013, 134, 3705–3720. [Google Scholar] [CrossRef] [PubMed]
- Gong, Z.; Ratilal, P.; Makris, N.C. Simultaneous localization of multiple broadband non-impulsive acoustic sources in an ocean waveguide using the array invariant. J. Acoust. Soc. Am. 2015, 138, 2649–2667. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.; Makris, N.C. The array invariant. J. Acoust. Soc. Am. 2006, 119, 336–351. [Google Scholar] [CrossRef] [PubMed]
- Makris, N.C.; Ratilal, P.; Jagannathan, S.; Gong, Z.; Andrews, M.; Bertsatos, I.; Godø, O.R.; Nero, R.W.; Jech, J.M. Critical Population Density Triggers Rapid Formation of Vast Oceanic Fish Shoals. Science 2009, 323, 1734–1737. [Google Scholar] [CrossRef] [PubMed]
- Gong, Z.; Andrews, M.; Jagannathan, S.; Patel, R.; Jech, J.M.; Makris, N.C.; Ratilal, P. Low-frequency target strength and abundance of shoaling Atlantic herring (Clupea harengus) in the Gulf of Maine during the Ocean Acoustic Waveguide Remote Sensing 2006 Experiment. J. Acoust. Soc. Am. 2010, 127, 104–123. [Google Scholar] [CrossRef] [PubMed]
- Makris, N.C.; Ratilal, P.; Symonds, D.T.; Jagannathan, S.; Lee, S.; Nero, R.W. Fish population and behavior revealed by instantaneous continental shelf-scale imaging. Science 2006, 311, 151–160. [Google Scholar] [CrossRef] [PubMed]
- Jagannathan, S.; Bertsatos, I.; Symonds, D.; Chen, T.; Nia, H.T.; Jain, A.D.; Jech, M. Ocean acoustic waveguide remote sensing (OAWRS) of marine ecosystems. MEPS 2009, 395, 137–160. [Google Scholar] [CrossRef]
- Jech, J.M.; Stroman, F. Aggregative patterns of pre-spawning Atlantic herring on Georges Bank from 1999–2010. Aquat. Living Resour. 2012, 25, 1–14. [Google Scholar] [CrossRef]
- Klaer, N. CIE Reviewer’s Independent on 54th Northeast Regional Stock Assessment Workshop; Center for Independent Experts: Wood’s Hole, MA, USA, 2012. [Google Scholar]
- Garland, E.C.; Goldizen, A.W.; Rekdahl, M.L.; Constantine, R.; Garrigue, C.; Hauser, N.D.; Poole, M.M.; Robbins, J.; Noad, M.J. Dynamic horizontal cultural transmission of humpback whale song at the ocean basin scale. Curr. Biol. 2011, 21, 687–691. [Google Scholar] [CrossRef] [PubMed]
- Cerchio, S.; Jacobsen, J.K.; Norris, T.F. Temporal and geographical variation in songs of humpback whales, Megaptera novaeangliae: synchronous change in Hawaiian and Mexican breeding assemblages. Anim. Behav. 2001, 62, 313–329. [Google Scholar] [CrossRef]
- McSweeney, D.J.; Chu, K.C.; Dolphin, W.F.; Guinee, L.N. North Pacific humpback whale songs: A comparison of southeast Alaskan feeding ground songs with Hawaiian wintering ground songs. Mar. Mam. Sci. 1989, 5, 139–148. [Google Scholar] [CrossRef]
- Magnúsdóttir, E.E.; Rasmussen, M.H.; Lammers, M.O.; Svavarsson, J. Humpback whale songs during winter in subarctic waters. Polar Biol. 2014, 37, 427–433. [Google Scholar] [CrossRef]
- Cerchio, S.; Dahlheim, M. Variation in feeding vocalizations of humpback whales Megaptera novaeangliae from southeast Alaska. Bioacoustics 2001, 11, 277–295. [Google Scholar] [CrossRef]
- Dunlop, R.A.; Cato, D.H.; Noad, M.J. Non-song acoustic communication in migrating humpback whales (Megaptera novaeangliae). Mar. Mam. Sci. 2008, 24, 613–629. [Google Scholar] [CrossRef]
- Dunlop, R.A.; Noad, M.J.; Cato, D.H.; Stokes, D. The social vocalization repertoire of east Australian migrating humpback whales (Megaptera novaeangliae). J. Acoust. Soc. Am. 2007, 122, 2893–2905. [Google Scholar] [CrossRef] [PubMed]
- Thompson, P.O.; Cummings, W.C.; Ha, S.J. Sounds, source levels, and associated behavior of humpback whales, Southeast Alaska. J. Acoust. Soc. Am. 1986, 80, 735–740. [Google Scholar] [CrossRef] [PubMed]
- Parks, S.E.; Cusano, D.A.; Stimpert, A.K.; Weinrich, M.T.; Friedlaender, A.S.; Wiley, D.N. Evidence for acoustic communication among bottom foraging humpback whales. Sci. Rep. 2014, 4, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Zoidis, A.M.; Smultea, M.A.; Frankel, A.S.; Hopkins, J.L.; Day, A.; McFarland, A.S.; Whitt, A.D.; Fertl, D. Vocalizations produced by humpback whale (Megaptera novaeangliae) calves recorded in Hawaii. J. Acoust. Soc. Am. 2008, 123, 1737–1746. [Google Scholar] [CrossRef] [PubMed]
- Rekdahl, M.L.; Dunlop, R.A.; Goldizen, A.W.; Garland, E.C.; Biassoni, N.; Miller, P.; Noad, M.J. Non-song social call bouts of migrating humpback whales. J. Acoust. Soc. Am. 2015, 137, 3042–3053. [Google Scholar] [CrossRef] [PubMed]
- Fournet, M.E.; Szabo, A.; Mellinger, D.K. Repertoire and classification of non-song calls in Southeast Alaskan humpback whales (Megaptera novaeangliae). J. Acoust. Soc. Am. 2015, 137, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Stimpert, A.K.; Au, W.W.L.; Parks, S.E.; Hurst, T.; Wiley, D.N. Common humpback whale (Megaptera novaeangliae) sound types for passive acoustic monitoring. J. Acoust. Soc. Am. 2011, 129, 476–482. [Google Scholar] [CrossRef] [PubMed]
- Baumgartner, M.F.; Fratantoni, D.M. Diel periodicity in both sei whale vocalization rates and the vertical migration of their copepod prey observed from ocean gliders. Limnol. Oceanogr. 2008, 353, 2197–2209. [Google Scholar] [CrossRef]
- Mayo, C.A.; Marx, M.K. Surface foraging behaviour of the North Atlantic right whale, Eubalaena glacialis, and associated zooplankton characteristics. Can. J. Zool. 1990, 68, 2214–2220. [Google Scholar] [CrossRef]
- Sims, D.W.; Quayle, V.A. Selective foraging behaviour of basking sharks on zooplankton in a small-scale front. Nature 1998, 393, 460–464. [Google Scholar] [CrossRef]
- Jain, A.D.; Ignisca, A.; Yi, D.H.; Ratilal, P.; Makris, N.C. Feasibility of Ocean Acoustic Waveguide Remote Sensing (OAWRS) of Atlantic Cod with Seafloor Scattering Limitations. Remote Sens. 2013, 6, 180–208. [Google Scholar] [CrossRef]
- Haedrich, R.L. Bigelow and Schroeder’s Fishes of the Gulf of Maine. Copeia 2003, 2, 422–423. [Google Scholar] [CrossRef]
- Nelson, G.A.; Ross, M.R. Biology and population changes of northern sand lance (Ammodytes dubius) from the Gulf of Maine to the Middle Atlantic Bight. J. Northwest Atl. Fish. Sci. 1991, 11, 11–27. [Google Scholar] [CrossRef]
- Payne, P.M.; Wiley, D.N.; Young, S.B.; Pittman, S.; Clapham, P.J.; Jossi, J.W. Recent fluctuations in the abundance of baleen whales in the southern Gulf of Maine in relation to changes in selected prey. Fish. Bull. 1990, 88, 687–696. [Google Scholar]
- Overholtz, W.J.; Link, J.S. Consumption impacts by marine mammals, fish, and seabirds on the Gulf of Maine Georges Bank Atlantic herring (Clupea Harengus) complex during the years 1977–2002. ICES J. Mar. Sci. 2007, 64, 83–96. [Google Scholar]
- Overholtz, W.J.; Jacobson, L.D.; Melvin, G.D.; Cieri, M.; Power, M.; Libby, D.; Clark, K. Stock assessment of the Gulf of Maine-Georges Bank Atlantic herring complex, 2003. Northeast Fish. Sci. Center Ref. Doc. 2004, 4, 1–290. [Google Scholar]
- Melvin, G.D.; Stephenson, R.L. The dynamics of a recovering fish stock: Georges Bank herring. ICES J. Mar. Sci. 2007, 64, 6982. [Google Scholar]
- Read, A.J.; Brownstein, C.R. Considering other consumers: fisheries, predators, and Atlantic herring in the Gulf of Maine. Conserv. Ecol. 2003, 7, 2. [Google Scholar]
- Overholtz, W.J. The Gulf of Maine-Georges Bank Atlantic herring (Clupea Harengus): Spatial pattern analysis of the collapse and recovery of a large marine fish complex. Fish. Res. 2002, 57, 237–254. [Google Scholar] [CrossRef]
- Jagannathan, S.; Kusel, E.T.; Ratilal, P.; Makris, N.C. Scattering from extended targets in range-dependent fluctuating ocean-waveguides with clutter from theory and experiments. J. Acoust. Soc. Am. 2012, 132, 680–693. [Google Scholar] [CrossRef] [PubMed]
- Tran, D.; Andrews, M.; Ratilal, P. Probability distribution for energy of saturated broadband ocean acoustic transmission: Results from Gulf of Maine 2006 experiment. J. Acoust. Soc. Am. 2012, 132, 3659–3672. [Google Scholar] [CrossRef] [PubMed]
- Ratilal, P.; Lai, Y.S.; Symonds, D.T.; Ruhlmann, L.A.; Preston, J.R.; Scheer, E.K.; Garr, M.T.; Holland, C.W.; Goff, J.A.; Makris, N.C. Long range acoustic imaging of the continental shelf environment: The Acoustic Clutter Reconnaissance Experiment 2001. J. Acoust. Soc. Am. 2005, 1117, 1977–1998. [Google Scholar] [CrossRef]
- Johnson, D.H.; Dudgeon, D.E. Array Signal Processing: Concepts and Techniques; Simon & Schuster: New York, NY, USA, 1992; pp. 1–512. [Google Scholar]
- Baumgartner, M.F.; Mussoline, S.E. A generalized baleen whale call detection and classification system. J. Acoust. Soc. Am. 2011, 129, 2889–2902. [Google Scholar] [CrossRef] [PubMed]
- Shapiro, A.D.; Wang, C. A versatile pitch tracking algorithm: From human speech to killer whale vocalizations. J. Acoust. Soc. Am. 2009, 126, 451–459. [Google Scholar] [CrossRef] [PubMed]
- Jech, J.M.; Price, V.; Chavez-Rosales, S.; Michaels, W. Atlantic herring (Clupea Harengus) demographics in the Gulf of Maine from 1998 to 2012. J. Northwest Atl. Fish. Sci. 2015, 47, 57–74. [Google Scholar] [CrossRef]
- Kinsler, L.E.; Frey, A.R.; Coppens, A.B.; Sanders, J.V. Fundamentals of Acoustics, 4th ed.; Wiley-VCH: New York, NY, USA, 1999; p. 448. [Google Scholar]
- Urick, R.J. Principles of Underwater Sound, 3th ed.; McGraw Hill: New York, NY, USA, 1983; pp. 29–65, 343–366. [Google Scholar]
- Collins, M.D. A split-step Padé solution for the parabolic equation method. J. Acoust. Soc. Am. 1993, 93, 1736–1742. [Google Scholar] [CrossRef]
- Andrews, M.; Chen, T.; Ratilal, P. Empirical dependence of acoustic transmission scintillation statistics on bandwidth, frequency and range in New Jersey continental shelf. J. Acoust. Soc. Am. 2009, 125, 111–124. [Google Scholar] [CrossRef] [PubMed]
- Andrews, M.; Gong, Z.; Ratilal, P. Effects of multiple scattering, attenuation and dispersion in waveguide sensing of fish. J. Acoust. Soc. Am. 2011, 130, 1253–1272. [Google Scholar] [CrossRef] [PubMed]
- Clark, C.W.; Clapham, P.J. Acoustic monitoring on a humpback whale (Megaptera novaeangliae) feeding ground shows continual singing into late spring. Proc. R. Soc. Lond. B 2004, 271, 1051–1058. [Google Scholar] [CrossRef] [PubMed]
- Vu, E.T.; Risch, D.; Clark, C.W.; Gaylord, S.; Hatch, L.T.; Thompson, M.A.; Van Parijs, S.M. Humpback whale song occurs extensively on feeding grounds in the western North Atlantic Ocean. Aquat. Biol. 2011, 14, 175–183. [Google Scholar] [CrossRef]
- Winn, H.E.; Beamish, P.; Perkins, P.J. Sounds of two entrapped humpback whales (Megaptera novaeangliae) in Newfoundland. Mar. Biol. 1979, 55, 151–155. [Google Scholar] [CrossRef]
- Van Opzeeland, I.; Van Parijs, S.; Kindermann, L.; Burkhardt, E.; Boebel, O. Calling in the cold: Pervasive acoustic presence of humpback whales (Megaptera novaeangliae) in Antarctic coastal waters. PLoS ONE 2013, 8, e73007. [Google Scholar] [CrossRef] [PubMed]
- Cato, D.H. Songs of Humpback Whales: The Australian Perspective. Mem Queensl Mus 1991, 23, 277–290. [Google Scholar]
- Mobley, J.R., Jr.; Herman, L.M.; Frankel, A.S. Responses of wintering humpback whales (Megaptera novaeangliae) to playback of recordings of winter and summer vocalizations and of synthetic sound. Behav. Ecol. Soc. 1988, 23, 211–223. [Google Scholar] [CrossRef]
- Waring, G.T.; Josephson, E.; Maze-Foley, K.; Rosel, P.E. U.S. Atlantic and Gulf of Mexico Marine Mammal Stock Assessments 2014. NOAA Tech. Memo. NMFS NE 231 2014, 228, 02543–1026. [Google Scholar]
- Adi, K.; Johnson, M.T.; Osiejuk, T.S. Acoustic censusing using automatic vocalization classification and identity recognition. J. Acoust. Soc. Am. 2010, 127, 874–883. [Google Scholar] [CrossRef] [PubMed]
- Buck, J.R.; Tyack, P.L. A quantitative measure of similarity for tursiopstruncatus signature whistles. J. Acoust. Soc. Am. 1993, 94, 2497–2506. [Google Scholar] [CrossRef] [PubMed]
- Deecke, V.B.; Ford, J.K.B.; Spong, P. Quantifying complex patterns of bioacoustic variation: Use of a neural network to compare killer whale (Orcinus orca) dialects. J. Acoust. Soc. Am. 1999, 105, 2499–2507. [Google Scholar] [CrossRef] [PubMed]
- Oppenheim, A.V.; Schafer, R.W.; Buck, J.R. Discrete-Time Signal Processing; Prentice-hall Englewood Cliffs: Upper Saddle River, NJ, USA, 1989; pp. 1–879. [Google Scholar]
- Mazhar, S.; Ura, T.; Bahl, R. Vocalization based Individual Classification of Humpback Whales using Support Vector Machine. Proc. IEEE 2007. [Google Scholar] [CrossRef]
- Dahlheim, M.E.; Awbrey, F. A classification and comparison of vocalizations of captive killer whales (Orcinusorca). J. Acoust. Soc. Am. 1982, 72, 661–670. [Google Scholar] [CrossRef]
- Kanungo, T.; Mount, D.M.; Netanyahu, N.S.; Piatko, C.D.; Silverman, R.; Wu, A.Y. An efficient k-means clustering algorithm: Analysis and implementation. IEEE Trans. Pattern Anal. Mach. Intell. 2002, 24, 881–892. [Google Scholar] [CrossRef]
- Jolliffe, I.T. Principle Component Analysis, 2nd ed.; Wiley Online Library: New York, NY, USA, 2002; pp. 11–31. [Google Scholar]
- Hirose, K.; Kawano, S.; Konishi, S.; Ichikawa, M. Bayesian Information Criterion and Selection of the Number of Factors in Factor Analysis Models. J. Data Sci. 2011, 9, 243–259. [Google Scholar]
- Bholowalia, P.; Kumar, A. EBK-Means: A Clustering Technique based on Elbow Method and K-Means in WSN. Int. J. Comput. Appl. 2014, 105, 17–24. [Google Scholar]
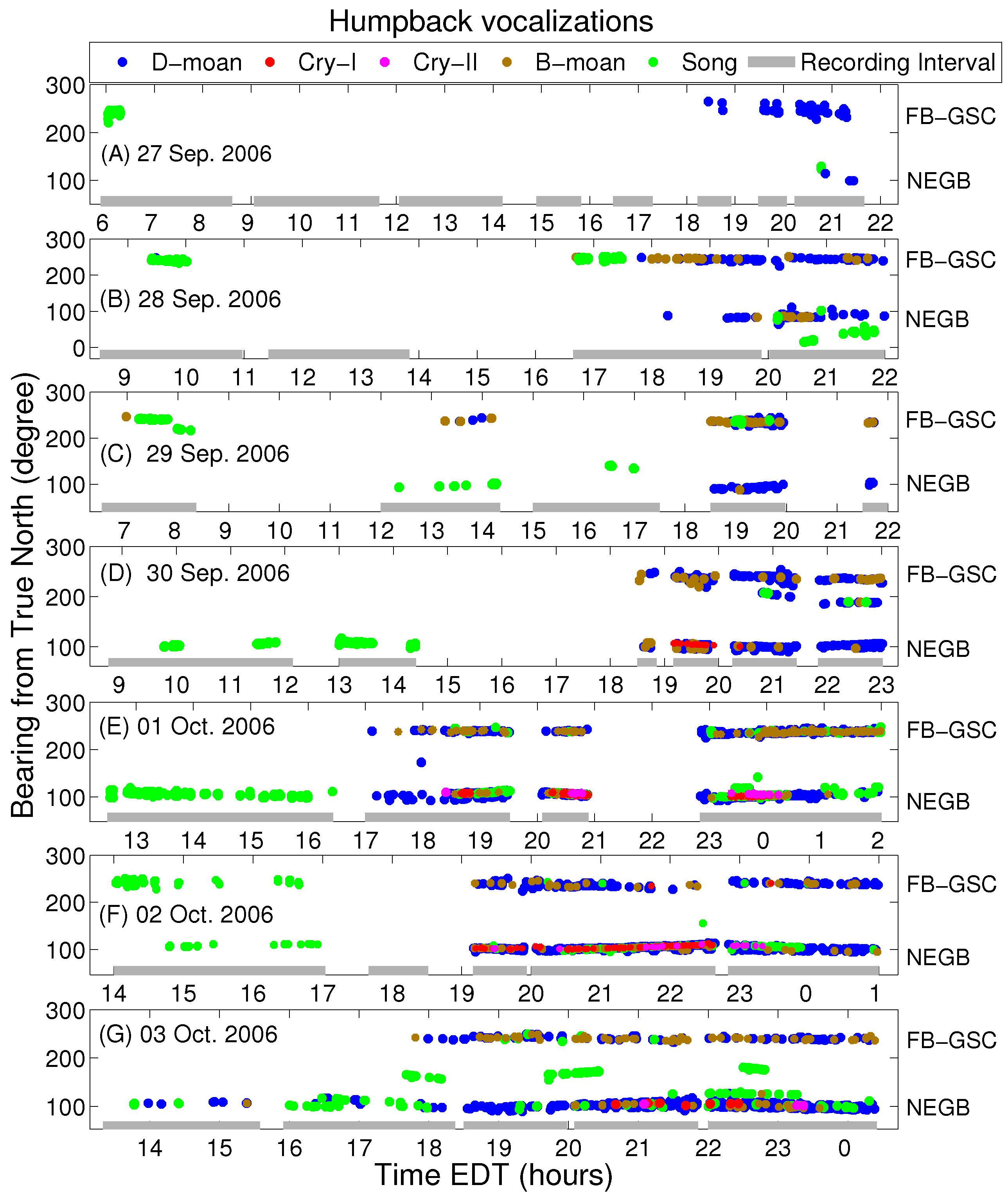

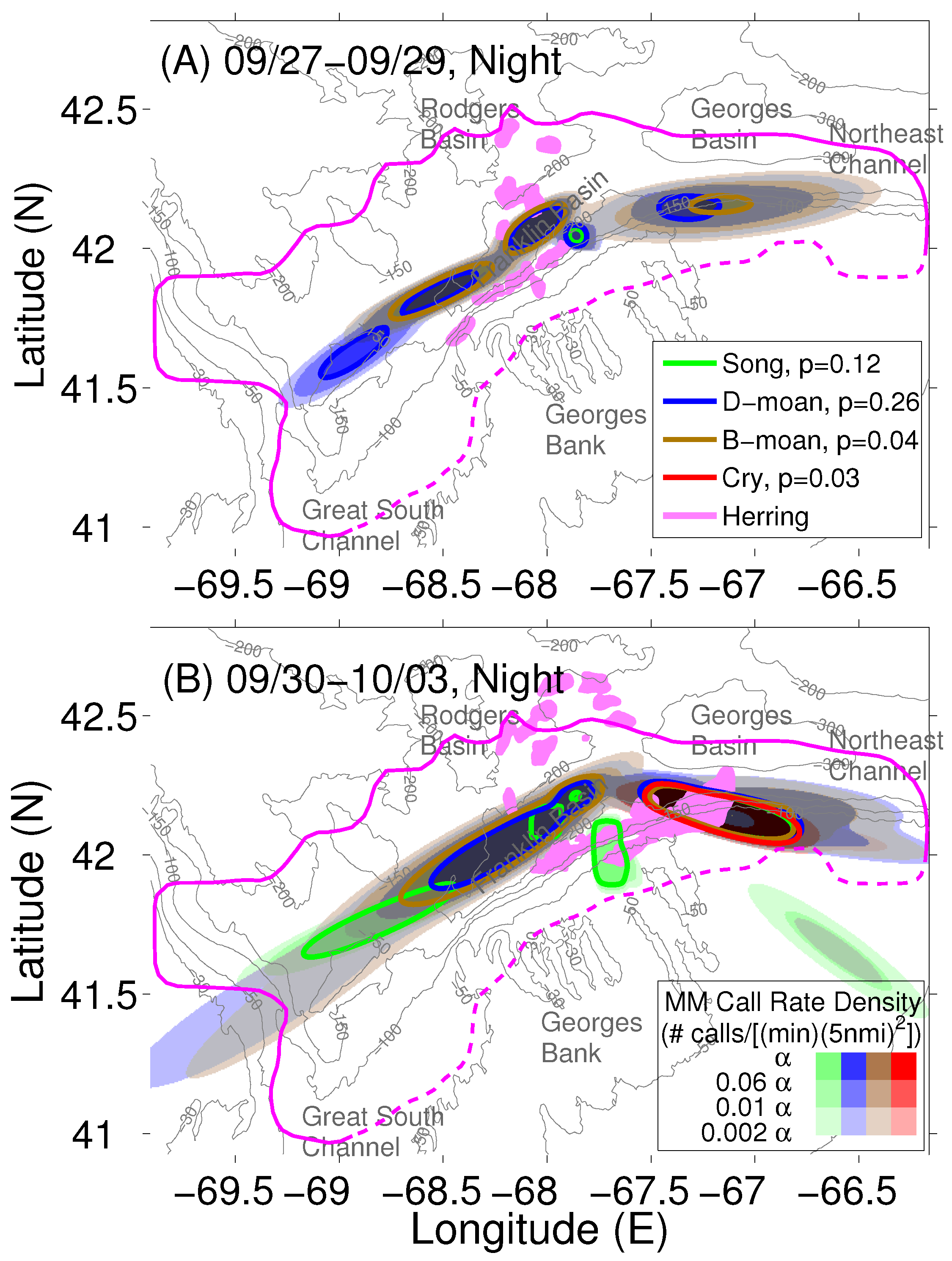
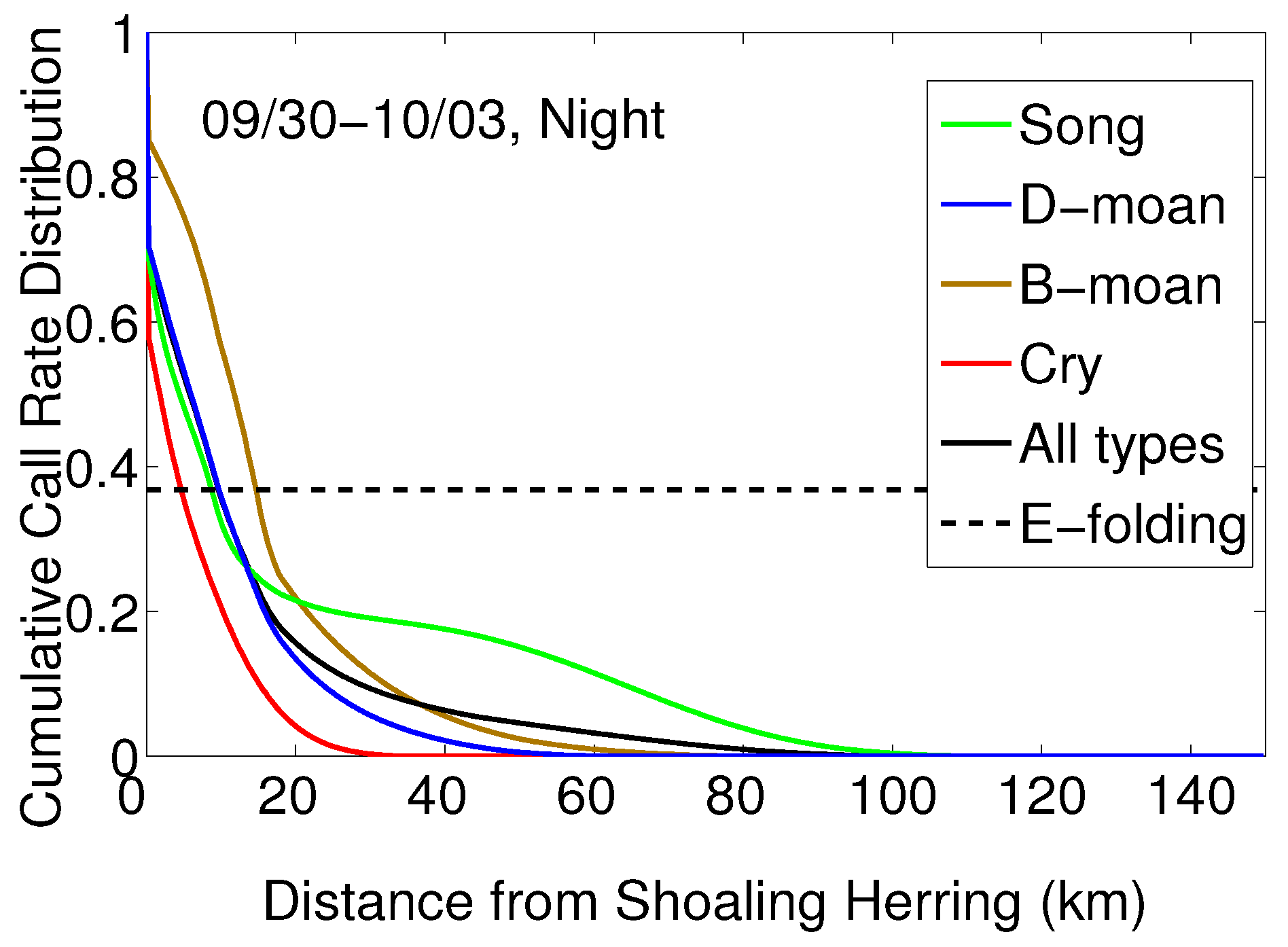
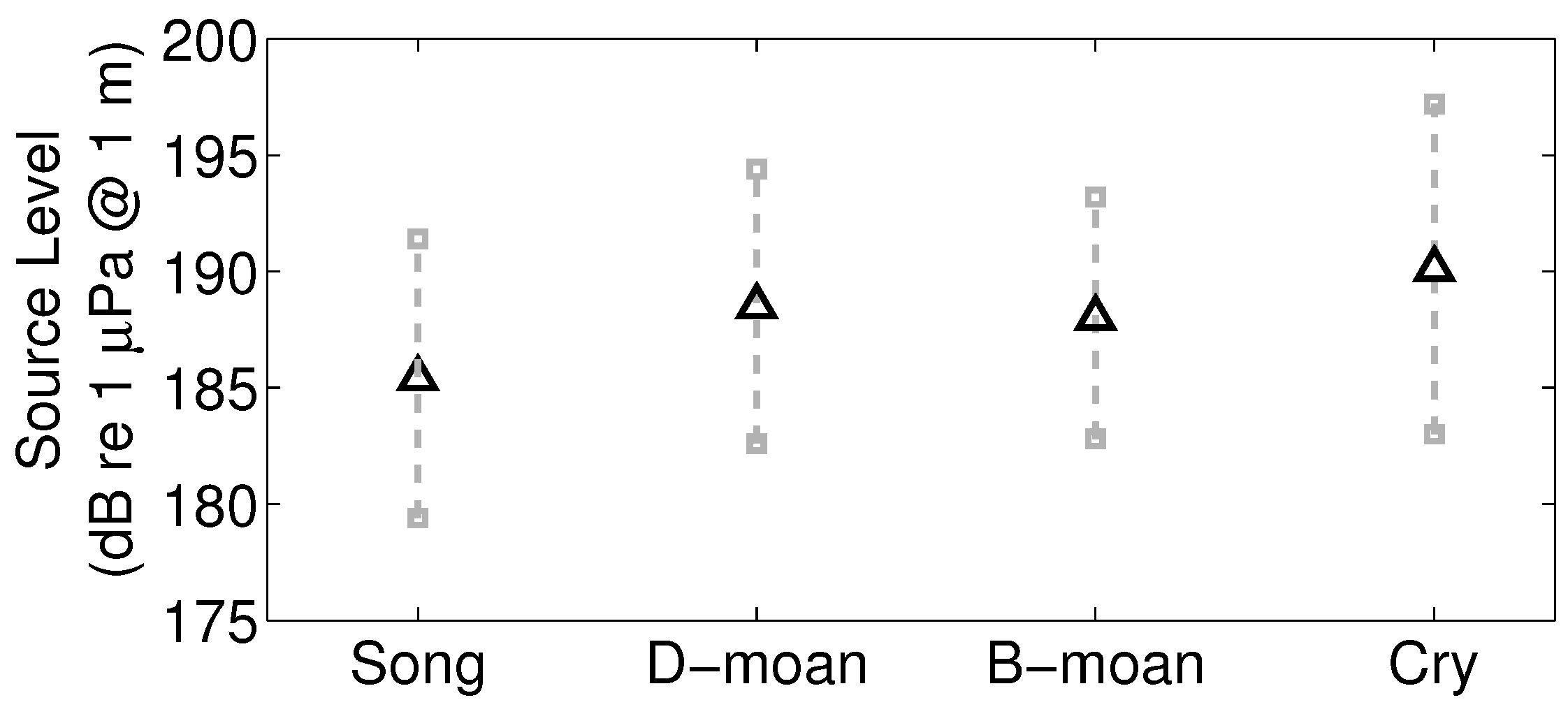
| Types | NEGB(90–130 | FB-GSC (220–250 |
|---|---|---|
| Call rate (calls/day) | 1200 ± 900 | 540 ± 240 |
| Song | 43.2% | 26.5% |
| Non-song | 56.8% | 73.5% |
| Types | NEGB (90–130 | FB-GSC (220–250 |
|---|---|---|
| Cry I | 6.7% | 0.3% |
| Cry II | 3.1% | 0% |
| B-moan | 6.7% | 28.4% |
| D-moan | 83.5% | 71.3% |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Huang, W.; Wang, D.; Ratilal, P. Diel and Spatial Dependence of Humpback Song and Non-Song Vocalizations in Fish Spawning Ground. Remote Sens. 2016, 8, 712. https://doi.org/10.3390/rs8090712
Huang W, Wang D, Ratilal P. Diel and Spatial Dependence of Humpback Song and Non-Song Vocalizations in Fish Spawning Ground. Remote Sensing. 2016; 8(9):712. https://doi.org/10.3390/rs8090712
Chicago/Turabian StyleHuang, Wei, Delin Wang, and Purnima Ratilal. 2016. "Diel and Spatial Dependence of Humpback Song and Non-Song Vocalizations in Fish Spawning Ground" Remote Sensing 8, no. 9: 712. https://doi.org/10.3390/rs8090712
APA StyleHuang, W., Wang, D., & Ratilal, P. (2016). Diel and Spatial Dependence of Humpback Song and Non-Song Vocalizations in Fish Spawning Ground. Remote Sensing, 8(9), 712. https://doi.org/10.3390/rs8090712






