1. Introduction
Common reed (
Phragmites Australis), a native helophyte in coastal areas of the Baltic Sea, has significantly spread on the Finnish coast during the last decades raising ecological issues and concerns due to the important role it plays in the ecosystem dynamics of shallow coastal areas [
1]. In addition to biodiversity there are other ecological and economic issues such as water protection, bioenergy, construction, farming and landscape. Reed beds have proven to be effective in the treatment of waste waters such as domestic sewage and industrial discharge containing heavy metal wastes [
2]. Reed biomass can be used as an energy source in three ways, namely by combustion, biogas production and biofuel production [
3]. There are currently studies aiming to develop economical and sustainable methods to harvest reed for bioenergy production in Finland. Reed can be used as a soil conditioner in agriculture thus substituting the use of fertilizers [
4].
Recent developments in sensor technology and data processing methods have led to an increase in the use hyperspectral imagery for environmental applications. High spectral and spatial resolution imagery provides researchers the potential to map vegetation at species’ level, provided the plant species under study are spectrally distinct [
5]. Species discrimination using remote sensing is based on the assumption that each species is characterized by a set of unique biophysical features and biochemical composition that control the variability in its spectral signature [
6].
The fundamental problem in vegetation discrimination using remote sensing is that there is an overall qualitative similarity in the spectral reflectance of green plant species. Furthermore, the assumption that individual plant species have unique spectral signatures may be wrong. Price [
7] has argued that several species may actually have quantitatively similar spectra due to the spectral signature variation present within a species.
The capability of discriminating plant species using hyperspectral imagery has been demonstrated in many studies. Clark
et al. [
8] successfully discriminated tropical rain forest tree species. Schmidt and Skidmore [
9] studied the discrimination of vegetation types in coastal wetlands. Thenkabail [
10] used hyperspectral data to discriminate agricultural crops. Vahtmäe
et al. [
11] demonstrated the feasibility of hyperspectral remote sensing for mapping benthic microalgae cover. The remote sensing of wetlands does, to some extent, differ from remote sensing based mapping of other terrestrial features. Differences exist because wetlands occupy a unique interface between aquatic and terrestrial ecosystems [
12]. In addition, the reflectance spectra of wetland vegetation canopies are often very similar and are combined with reflectance spectra of underlying soil [
13]. The frequent and rapid changes of water depth and salinity add to the complexity of analyzing wetland environment using remote sensing.
The use of remote sensing in reed bed discrimination has been studied in several publications. Pengra
et al. [
14] evaluated the use of the spaceborne Hyperion sensor. The classification of reed beds showed good overall accuracy of 81.4%. It was found, however, that the small size and spatial arrangement of
Phragmites stands was less than optimal considering Hyperion’s spatial resolution of 30 m. Lopez
et al. [
15] studied the discrimination of
Phragmites using airborne hyperspectral data collected by the Probe-1 sensor. The study produced
Phragmites maps showing an estimated accuracy of 80%. Onojeghuo and Blackburn [
16] proposed the use of airborne hyperspectral and LiDAR data in reed bed discrimination. A comprehensive set of methods such as Principal Component Analysis (PCA), Spectrally Segmented PCA (SSPCA) and Minimum Noise Fraction (MNF) were applied to the hyperspectral data and combined with LiDAR derived measures including those based on texture analysis. A significant improvement (+11%) in the accuracy of reed bed delineation was achieved when a LiDAR-derived Canopy Height Map (CHM) was used together with the optimal SSPCA data set. Stratoulias
et al. [
17] used airborne AISA Eagle data in order to derive narrow band spectral indices used to characterize reed beds’ ecological status. Seasonal time-series studies can provide important information on spectral variability. Given the dynamic character of vegetation cover, a snapshot in time is not nearly as revealing as a time sequence [
18]. Ouyang
et al. [
19] studied the spectral characteristics of
Phragmites and two other wetland species using time-series analysis. The results showed that differences among saltmarsh communities’ spectral characteristics were affected by their phenological stages. Artigas and Yang [
20] published a field-collected seasonal time-series of
Phragmites spectra. The measured spectra were used to determine the vigor gradient of plants in marshlands.
Several measures have been proposed to quantify spectral similarity or separability of targets [
21], Euclidean Distance (ED) being probably the most well-known measure used. Spectral Angle Mapper (SAM) is another well-established similarity measure in remote sensing applications. SAM is related to Pearson’s Correlation Coefficient, sometimes also called Spectral Correlation Mapper (SCM) in remote sensing literature. An advantage of Pearson’s Correlation Coefficient over SAM is its ability to distinguish between negative and positive correlation. Spectral Information Divergence (SID) classifier represents an information theoretic approach to hyperspectral classification. SID compares the similarity between two spectra by measuring the probabilistic discrepancy between two corresponding spectral signatures. In principle, the similarity between two spectra has two components: similarity in absolute level of reflectance and similarity in spectral shape. In this study, both of these components of similarity are assessed using the Euclidean Distance and Spectral Angle measures. Jeffries–Matusita distance (JM) was used as a statistical separability measure.
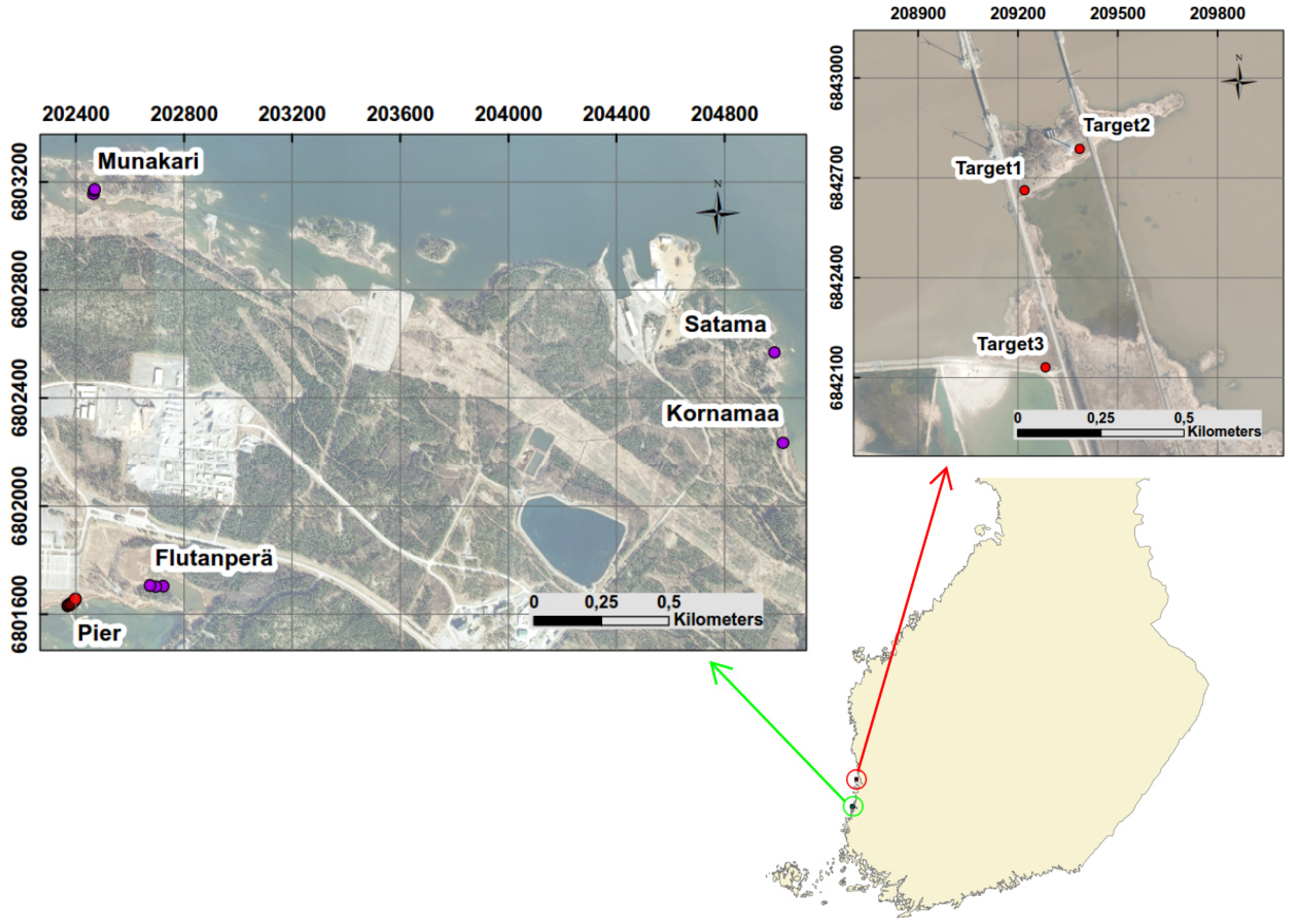
Reed beds cover significant part of the shoreline of the Olkiluoto Island. Reed can be found on gyttja, clay, till or stone bottoms [
22]. The extent and vitality of the reed beds varies significantly, depending on the soil type and degree of shelter [
22]. A repository site for spent nuclear fuel is currently under construction in Olkiluoto. The disposal is planned to begin in 2022. The results of reed bed studies will be used as input data to the biosphere assessment exercise for the safety analysis of the spent nuclear fuel repository at Olkiluoto [
23]. Common reed is a major producer of biomass among wetland species in Olkiluoto and it has significant potential to store and transport radionuclides. Therefore, quantitative information on the extent and biomass of reed stands is an integral part of long-term biosphere assessment. The overall aim of this study is to determine temporal and spatial spectral variability of reed beds in the Olkiluoto Island. More specifically, the objectives of this study are: (1) to characterize the spectral properties of the dominant wetland species
Phragmites Australis in different phenological stages and to identify the most suitable time to discriminate it from other green vegetation; (2) to study the spatial variability of reed spectra and evaluate the effects of this variability on reed bed mapping; and (3) to suggest promising methods to be used in reed bed mapping.
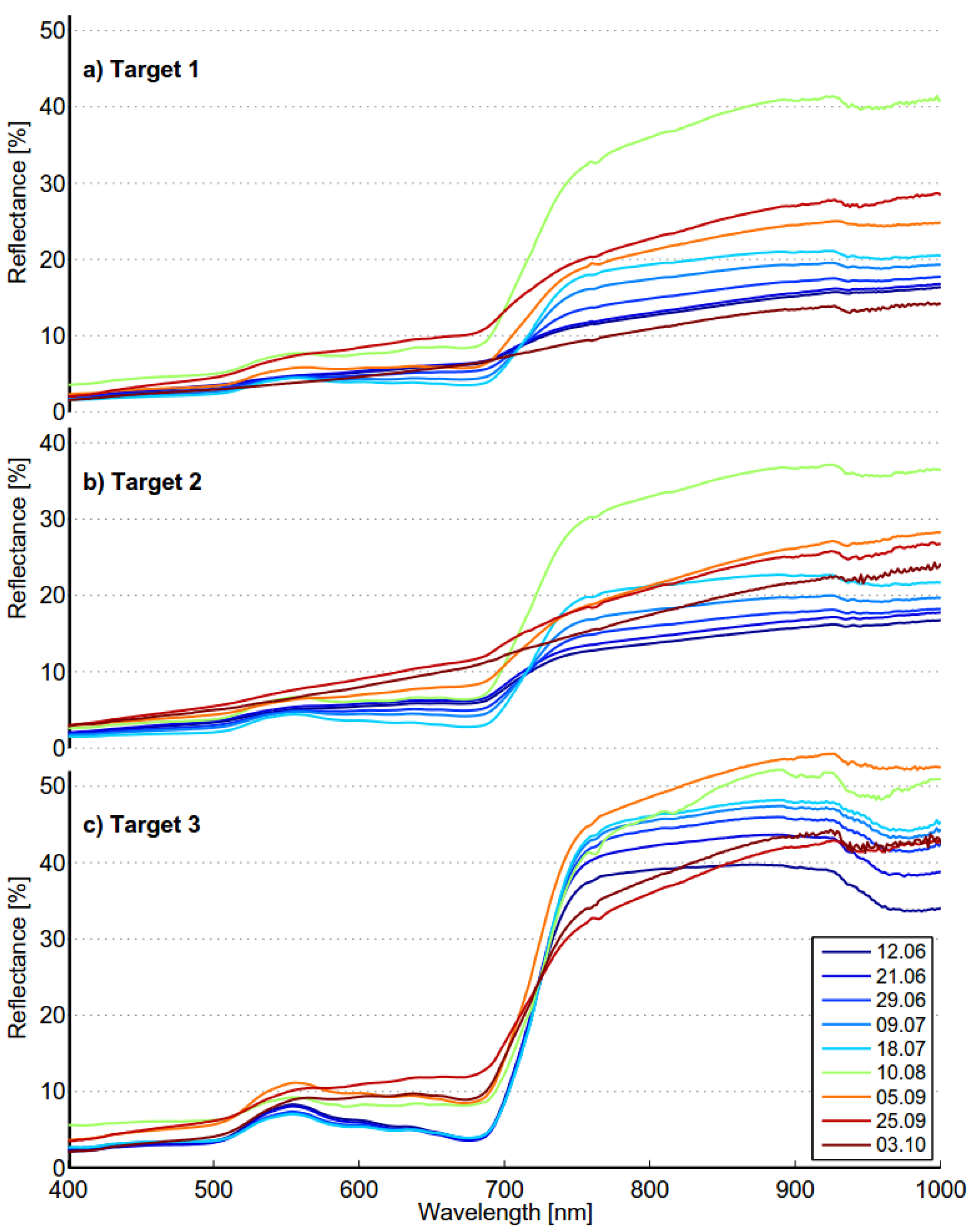
4. Discussion
Several seasonal time-series spectra of
Phragmites have been published in the literature. Seasonal spectra are dependent on local conditions, but spectral features reflecting phenological stages should be found in time-series spectra anywhere. The seasonal spectra of targets 1 and 2 are quite consistent with those published by Ouyang
et al. [
19]. The shape of the spectra is quite similar at the beginning and the end of the season as well as in the “full vigor” state. The growth period is naturally somewhat shorter in Finland than in Dongtan, China. The seasonal spectra of target 3 differ from those in [
19] at the beginning and end of the season. The seasonal spectra of targets 1 and 2 show similar trends as those found in time-series spectra of
Phragmites measured in New Jersey Meadowlands [
20]. When seasonal spectra were compared to those published by Gilmore
et al. [
26], the situation was quite the opposite: the published spectra were very similar to those of target 3 in our study. The results show that the optimal time for data acquisition is dependent on classification method to be used. For distance based classifier optimal time for old reed bed is in the middle of June when it is the beginning of September for new reed bed. Optimal times are different when shape based classifier is used; beginning of October for old reed bed and the middle of June for new reed bed. The separability of old reed bed is almost as high in the middle of June as in October. Acquisition in the middle of June could give good results for both reed bed types.
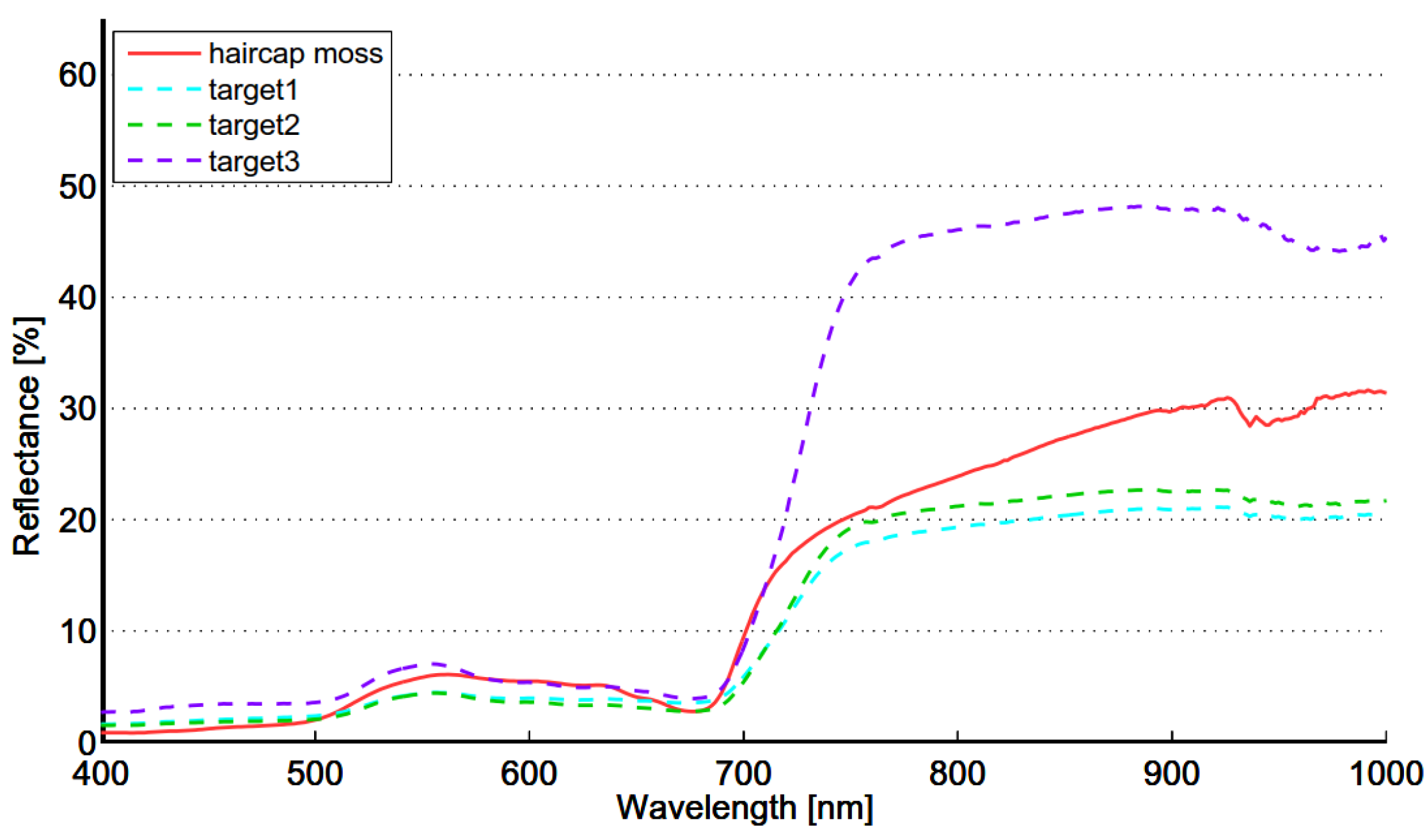
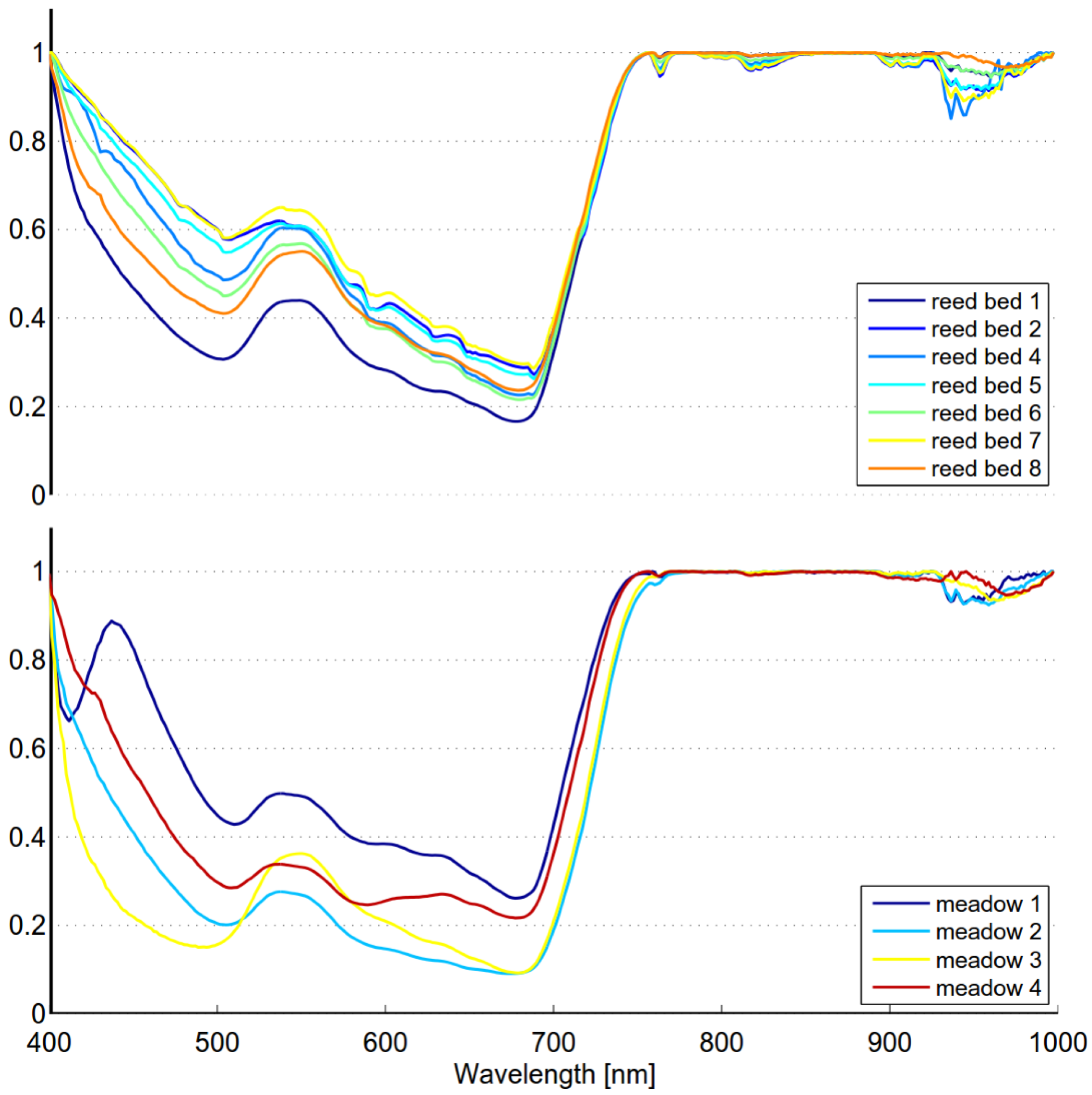
The seasonal spectral changes of targets 1, 2 and 3 are largely determined by two variables: density of the reed bed and the ratio of dead and live biomass. Density defines if the soil or water is visible. Dark soil and water have low reflectance, which lowers the mixed spectra. Dead biomass has low and flat spectrum, whereas live biomass has distinct features of green vegetation. In the beginning of season old reed beds have only a small fraction on live biomass and the soil is partly visible because of low density. The spectrum is low and flat and therefore separable from moss. In the beginning of October old reed beds are full of dead biomass, resulting similar flat spectrum and high separability from green vegetation. The spectra of moss show signs of moderate chlorophyll content, modest local maximum near 560 nm and gentle red-edge. Target 3 is in “full vigor” state on 5 September 2012, the reflectance in green and NIR region is at the season’s highest level and therefore the separability from moss is optimal.
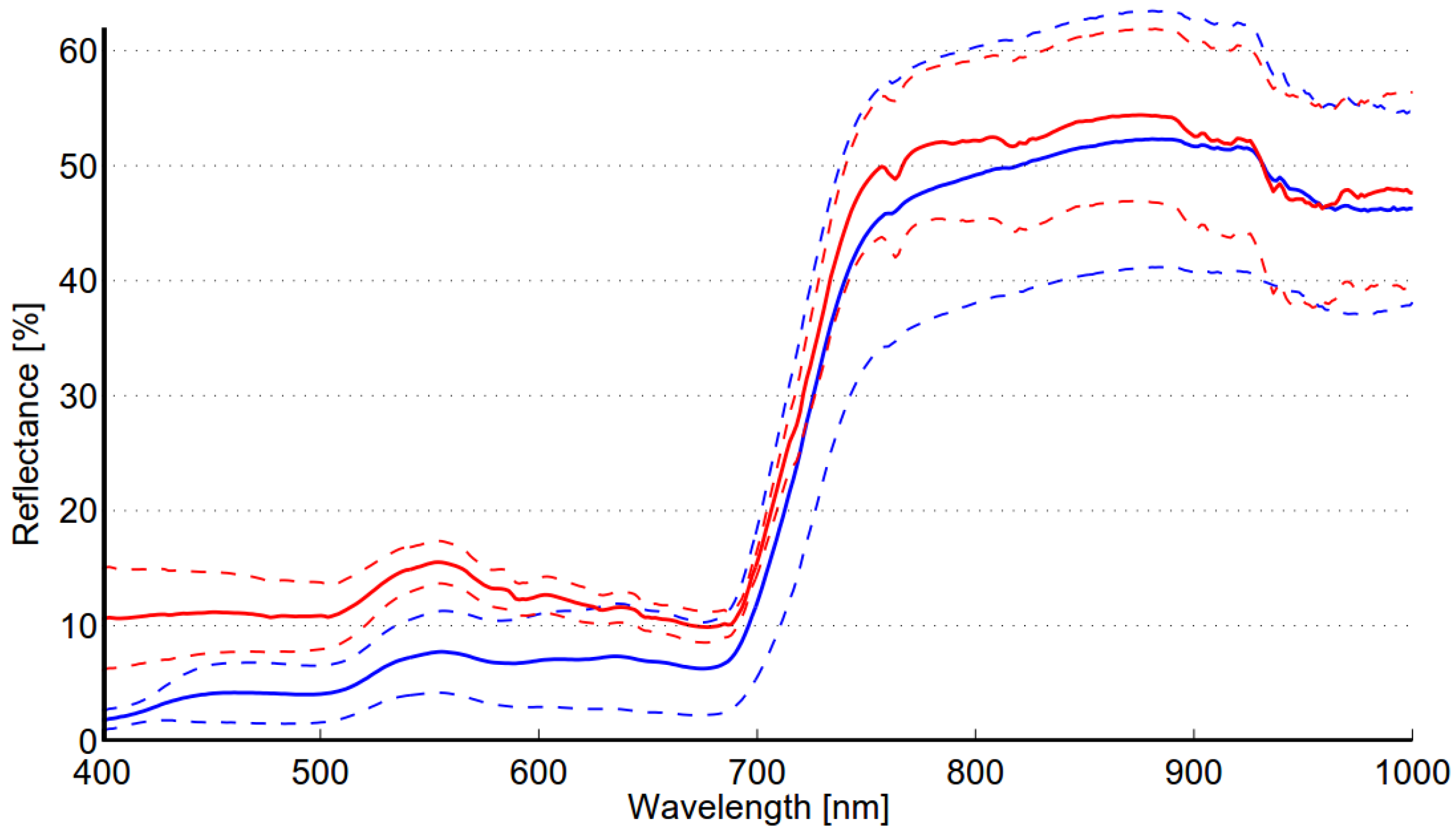
The within-class variability of reed is somewhat lower than that of the meadow class (see
Table 8). This can be expected as the meadow class contains several species,
i.e., grasses, weeds and flowers. The well-kept grass field was visually very homogenous; however, the spectral variability obtained for this class is higher than could be expected. This is most likely due the structural changes in grass. Results indicate that a single target (R1) significantly contributes to the within-class variability of the reed class. Debba
et al. [
31] studied the spectral within-class variability of different savannah trees. The average variability of seven tree species was lower than that of reed in this study. The published reflectance spectra of tree species showed that the within-class variability of some species (
Combretum apiculatum,
Terminalia sericia) was at the same level as obtained here for reed, although the average variability over all species was lower. Based on the analysis of the mean and standard deviation of the spectra obtained for the reed and meadow classes (see
Figure 5) it is fair to argue that the two classes are extremely difficult to discriminate due to high within-class variability of both classes as well as spectral similarity of the classes. Several published studies have proposed fusion of hyperspectral and LiDAR data in wetlands mapping applications [
16]. LiDAR can complement the spectral information of optical imagery and thus improve classification results. As the height of reed beds clearly differs from that of meadows, such approach could be highly beneficial. In addition, using textural features together with spectral information has been shown to enhance the classification accuracy of remotely sensed data [
32].
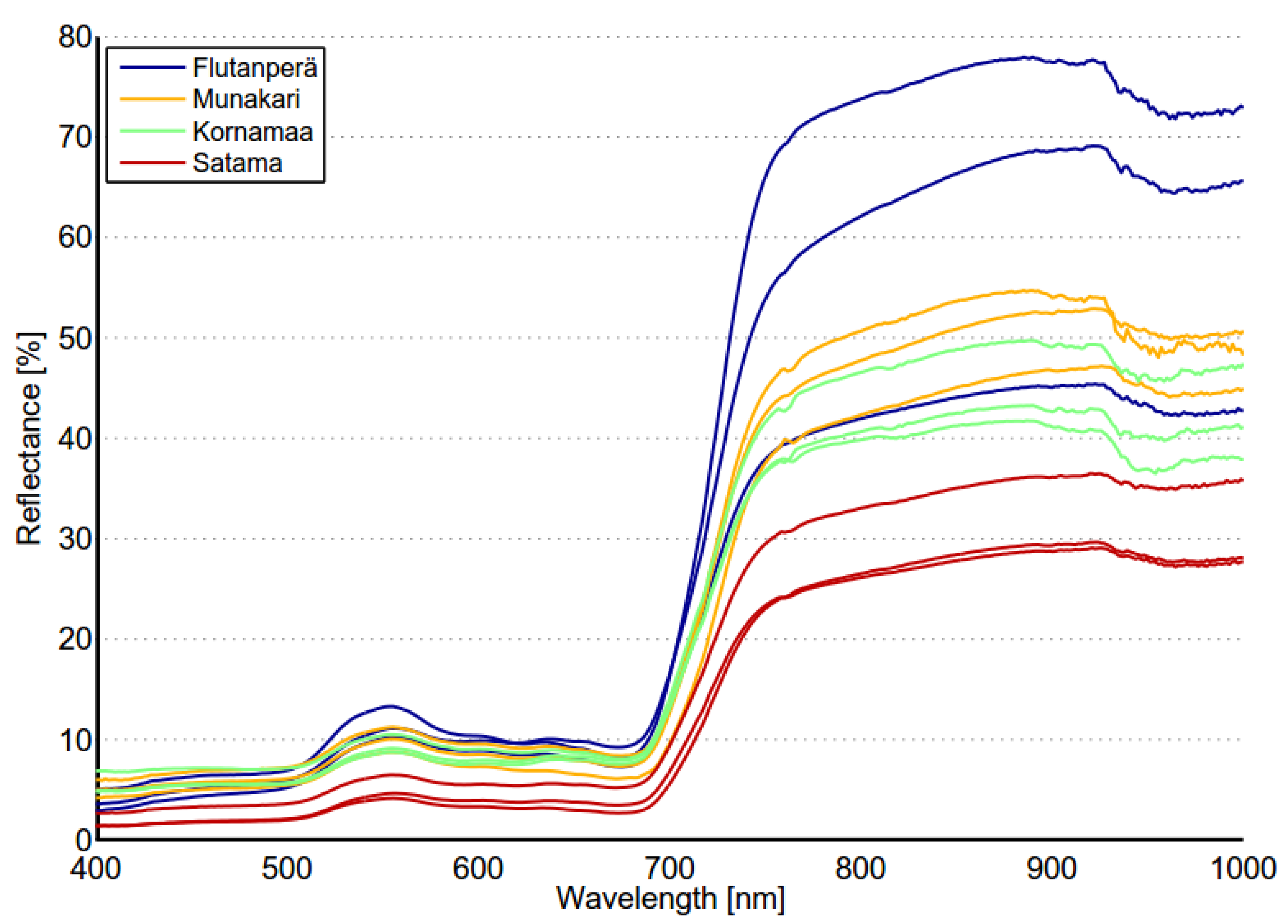
Spectral variability of reed beds within the Olkiluoto Island is significant (see
Figure 7). Zomer
et al. [
33] studied the reflectance spectra of dominant wetland species. The measured results of common reed (
Phragmites Australis) showed similar shape and high variability as in
Figure 7. This suggests that high spectral variability could be common characteristics for reed beds. In order to study spectral variability between reed sites, each site was assigned to a separate class and between-class variability was calculated for each pair of sites. Surprisingly the between-class variability of sites Munakari and Kornamaa (3.85) was lower than local spatial inter-class variability of reed bed at site Pier (15.95). The between-class variability of these two sites was also lower than the variability between spectrally similar Savannah trees [
31]. The highest variability was measured between sites Flutanperä and Satama. This is likely due to remarkable reflectance differences in the NIR region shown in
Figure 7. This can be explained by the characteristics of reed beds presented in
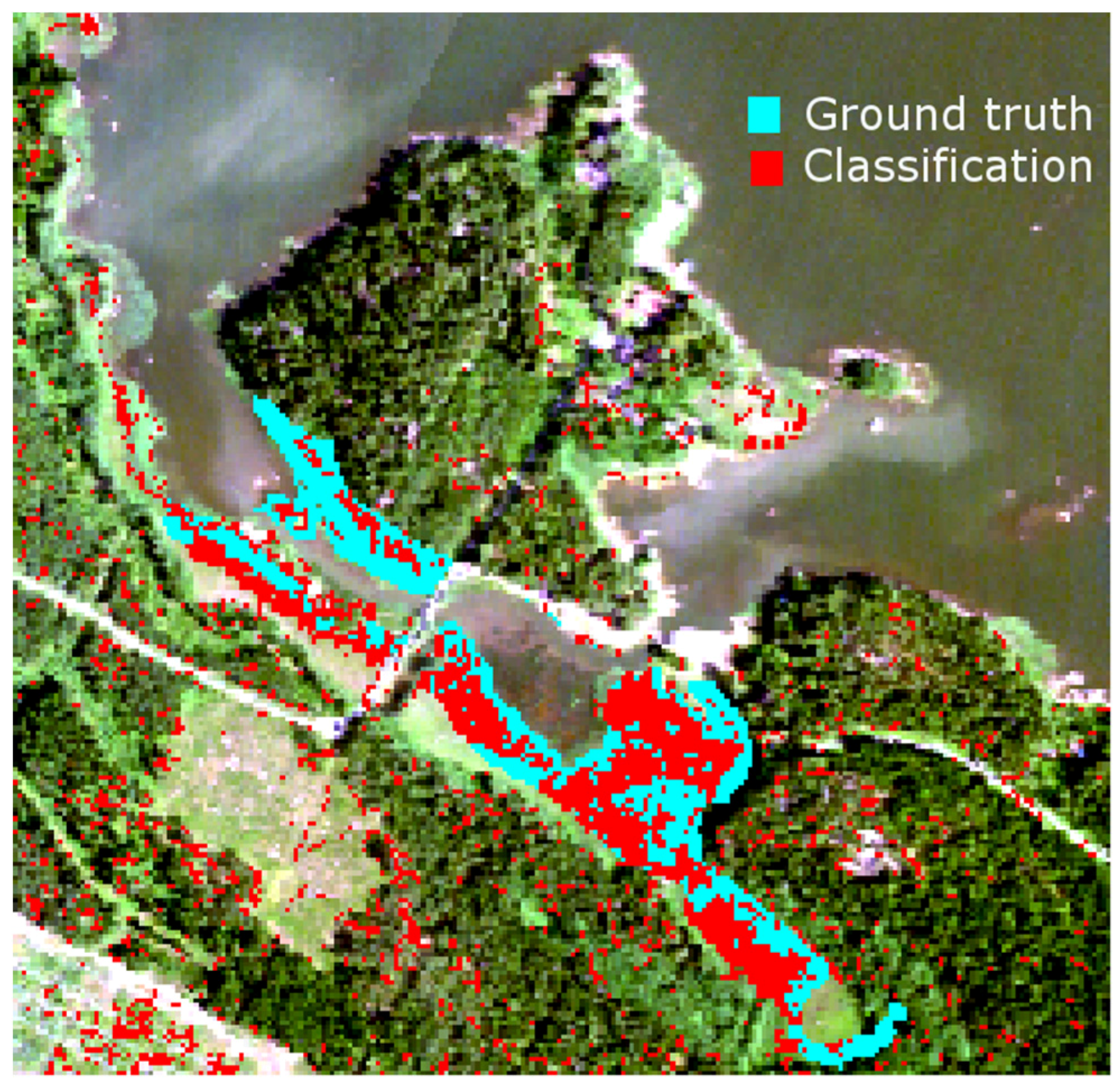
Table 2. The density and height of dead reed stems is clearly higher at Satama than at Flutanperä. The effects of spectral variability to reed bed discrimination within Olkiluoto Island were studied using airborne hyperspectral data. The accuracy of classification was measured using mean spectra of reed bed sites as a target spectra. Overall accuracy was good, mainly because the other class was much larger than reed bed class,
i.e., the relative amount of commission errors stays low. The results of agreement accuracy were modest. This is most likely due to two factors: unfavorable time to separate reed beds from other vegetation and variety of reed bed types present,
i.e., old, new and dry reed beds. Conventional remote sensing schemes use one reference spectrum for each species. The results suggest that the use of multiple-endmember approach might be beneficial, agreement accuracy increases when dedicated spectrum is used for reed bed site instead of using one spectrum for the whole island.
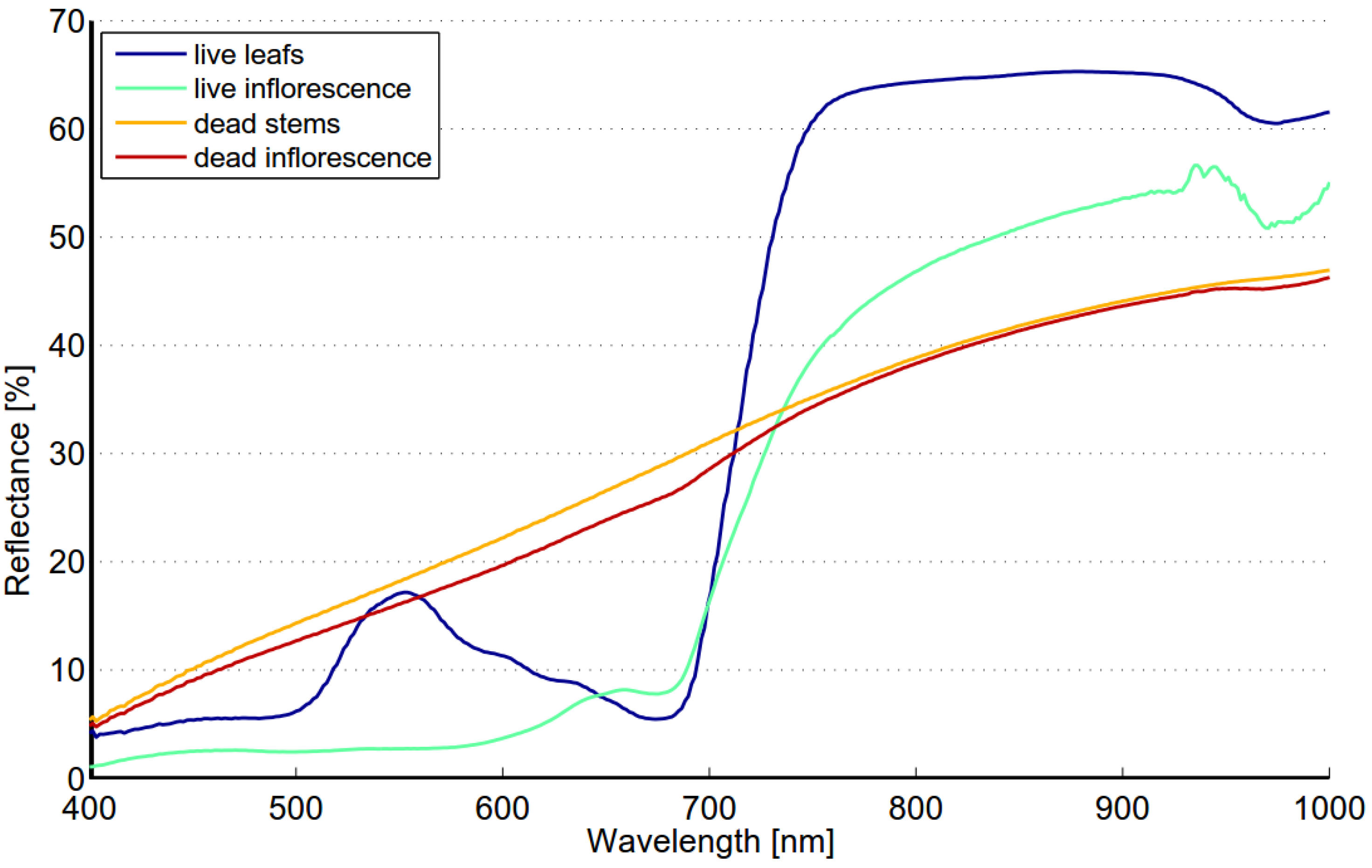
The spectrum of reed bed is largely determined by the density of reed stems and the ratio of live and dead biomass. The reed bed spectra may also include background contributions from water, soil, understory vegetation and shade depending on the density and structure of reed stems. Live leafs have typical spectrum of healthy green vegetation,
i.e., steep red edge and reflectance peak near 560 nm while the spectrum of dead stems is flat and the reflectance level is low (
Figure 9). The reflectance of a dead stem is higher in the NIR region when compared to autumn spectra of targets 1 and 2. This is most likely due to shadows between stems or visible soil/water or both. The spectrum of type “old reed bed” varies depending on the ratio of live and dead stems. Field studies made in August 2012 showed that there is considerable variability in the ratio of live and dead stems. The fraction of dead stems varied from 0 to 83%. The most obvious reason for high spectral variability within Olkiluoto Island is the changes in the ratio of live and dead stems.
5. Conclusions
The temporal and spatial variability of reed bed spectra was evaluated in this study. The temporal variability of reed bed spectra was found to be significant. The main challenge related to temporal variability is that there are two different types of reed beds having different seasonal spectra. The optimal time of data acquisition depends on the reed bed type. When this is combined with the fact that usually the phenological state of the other vegetation has to be considered as well, careful timing of the data acquisition is needed. The spectral within-class variability of both reed bed and meadow in local neighborhood was found to be large when compared to references. Both classes have similar mean spectra, however, all the targets except R1 had lower spectral angle to the mean spectrum of the corresponding class than that of the other class. This gives a positive indication for successful reed bed mapping. The results on within-class spectral variability of reed bed at four test sites within the Olkiluoto Island showed that while the reed spectra from the sites of Kornamaa and Munakari were close to each other, the spectra measured at Satama and Flutanperä differed significantly. This is at least partly due to the variation in the density and height of live and dead reed stems among the four sites. It can be concluded that if features such as reed characteristics, temporal variation and surrounding habitats are known and can be controlled, mapping of reed beds is feasible based on their spectral properties; otherwise LiDAR data or textural features would be needed.
In this study, the optimal times to separate reed beds from other vegetation was determined. Depending on the reed bed types present and classification method to be used, it might be beneficial to use multi-temporal classification, i.e., use several dataset collected at different times. High spectral within-species variability measured advocates the use of multiple-endmember methods. The test using airborne hyperspectral data further supports this conclusion. Scaling from spectral field measurements to airborne/satellite data brings with it new challenges. Poor spatial resolution can lead to significant amount of mixed pixels confusing the classification process. Modest signal-to-noise ratio of satellite sensors can further increase this confusion. The spectral resolution of spaceborne multispectral sensors might be too low to differentiate to subtle differences between reed beds and other wetland vegetation. The study using spectral field measurements showed poor separability between reed beds and meadows when distance and statistical measures where used although better results were obtained using a SAM-measure. The errors in geometric, radiometric and atmospheric correction related to air- and spaceborne sensors can deteriorate this subtle separability. This study provides a sound basis for future research of reed bed discrimination using air- and spaceborne data.