Airborne Hyperspectral Images and Ground-Level Optical Sensors As Assessment Tools for Maize Nitrogen Fertilization
Abstract
:1. Introduction
2. Material and Methods
2.1. Experimental Site and Crop Management
2.2. Maize Analysis
2.3. Ground-Level Optical Determinations
2.4. Airborne Campaigns
2.5. Airborne Hyperspectral Index Calculation
2.6. Application to N Fertilizer Recommendation
2.7. Statistical Analysis
3. Results and Discussion
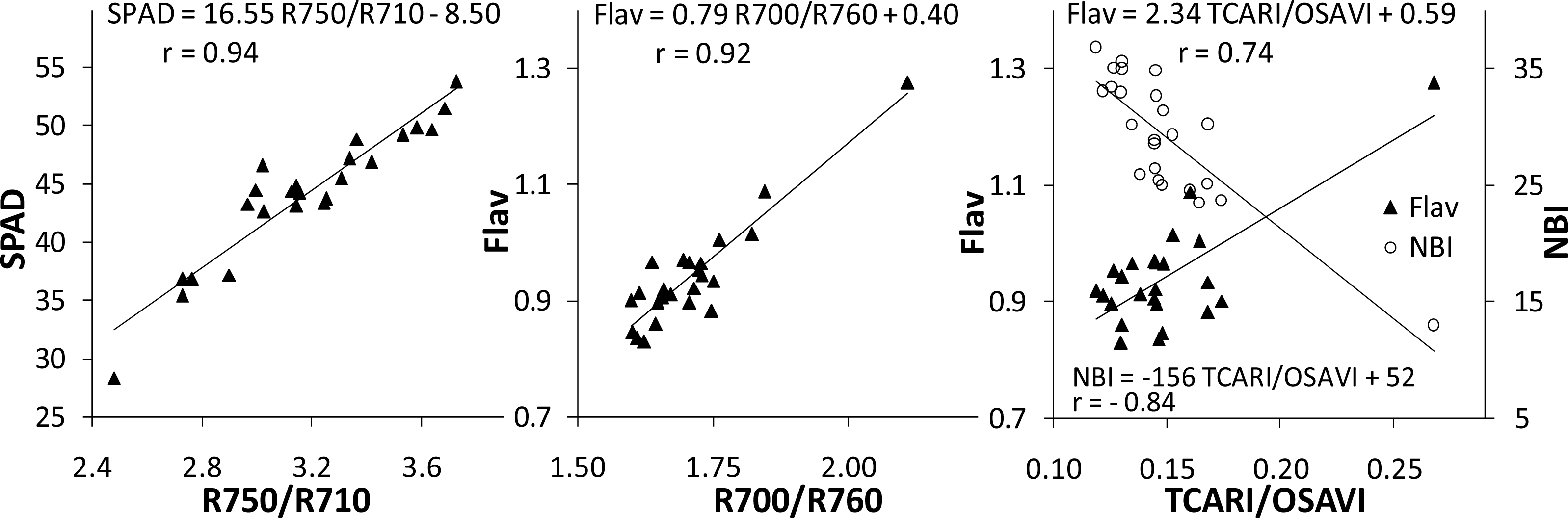
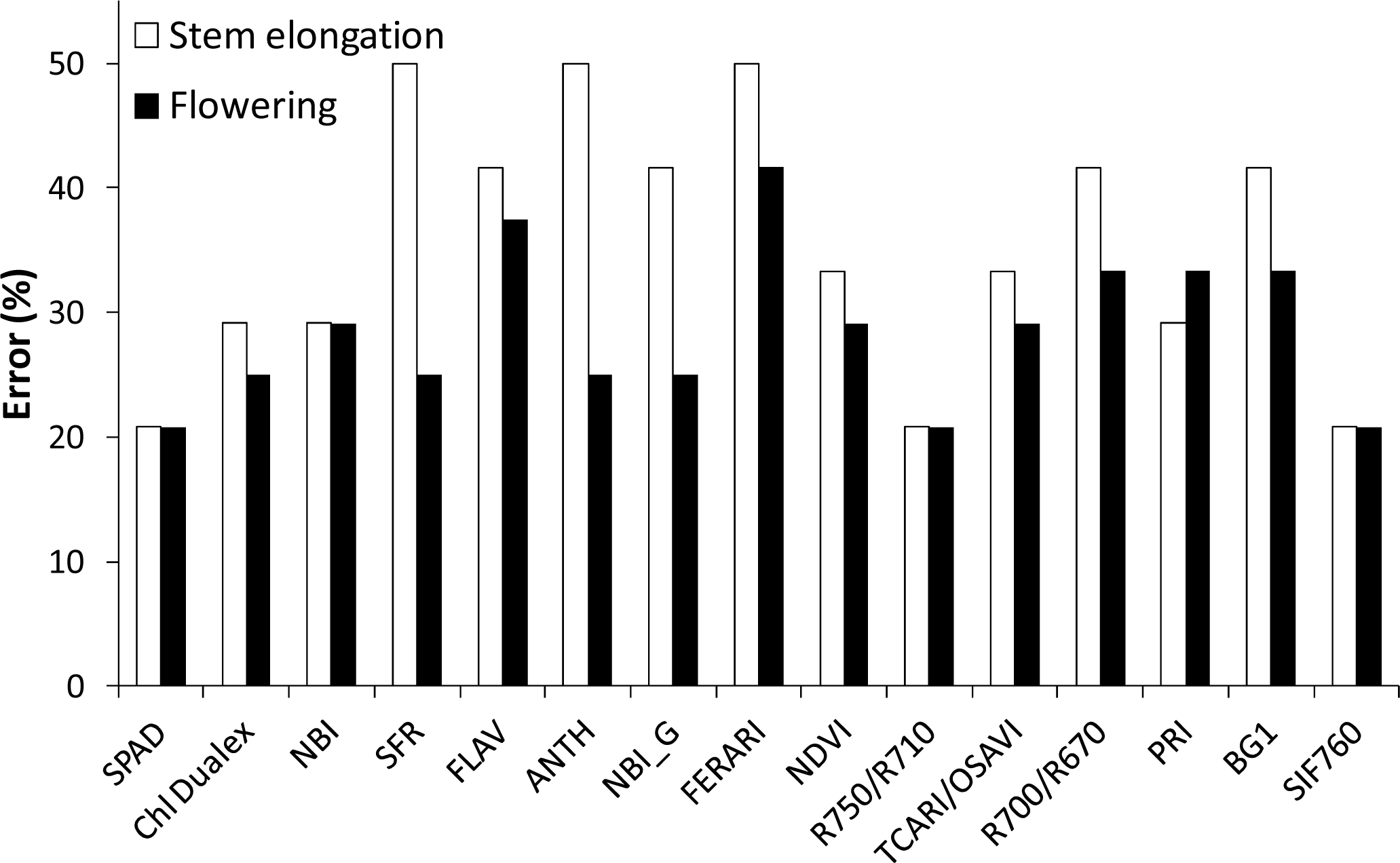
3.1. Optical Ground-Level Measurements
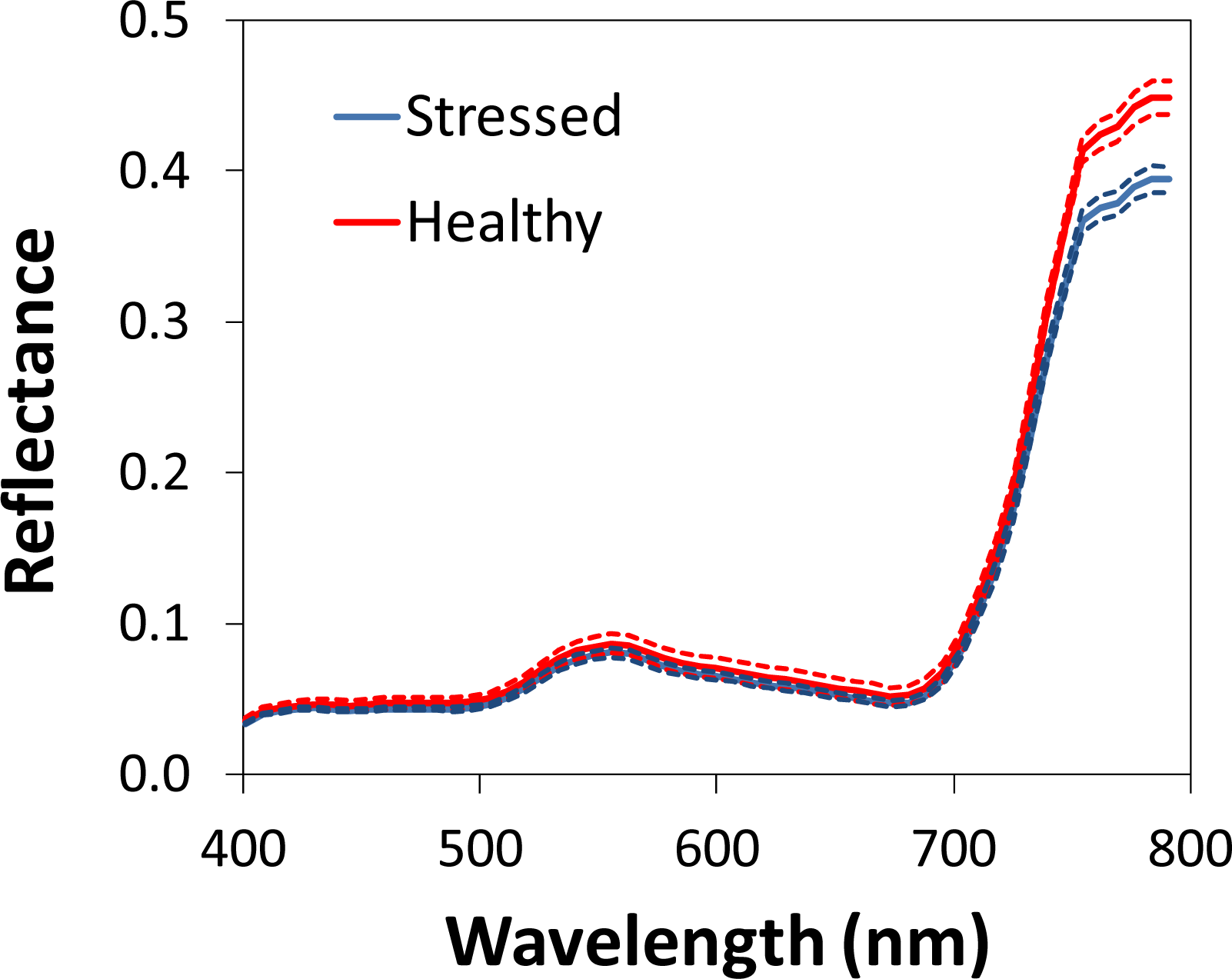
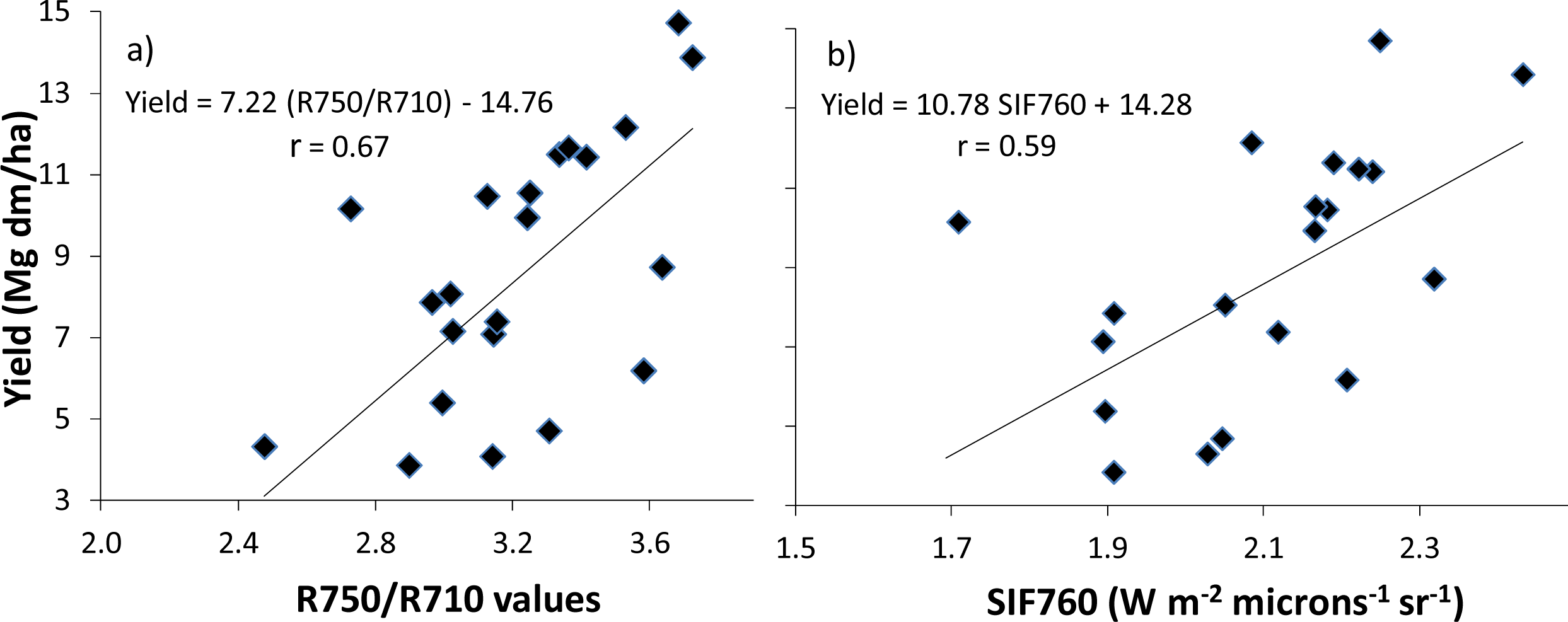
3.2. Optical Ground-Level vs. Airborne Measurements
3.2.1. Flowering
3.2.2. Stem Elongation
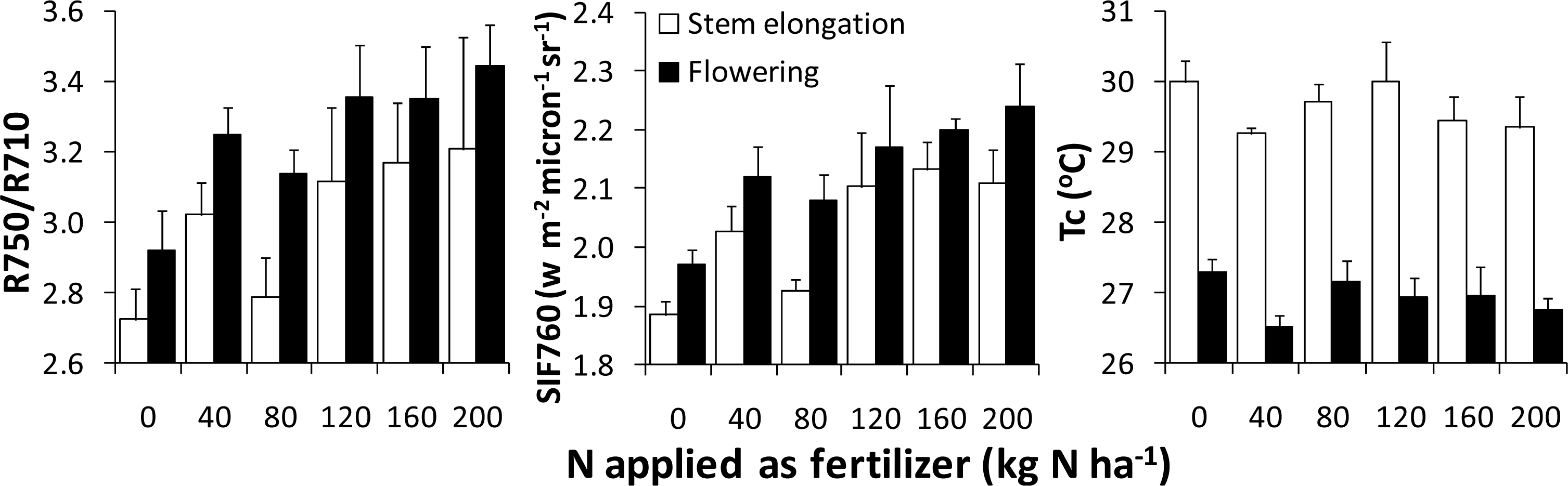
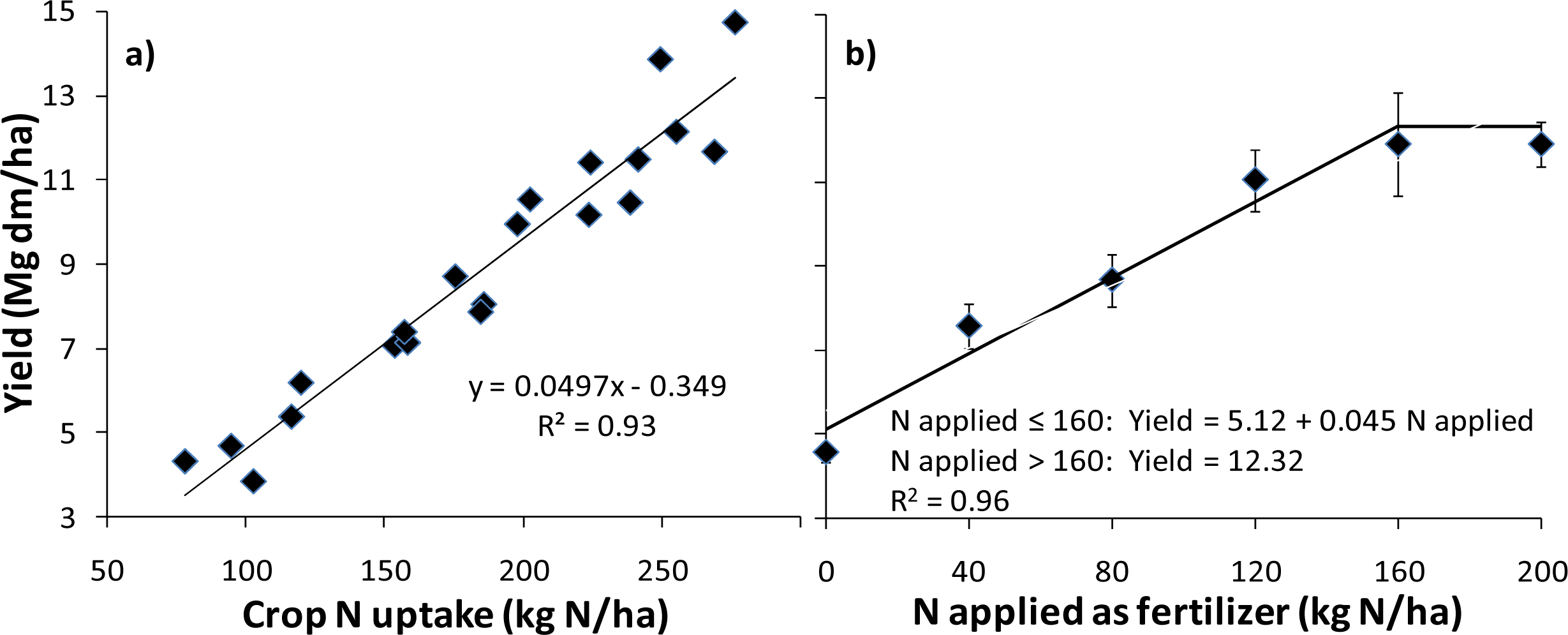
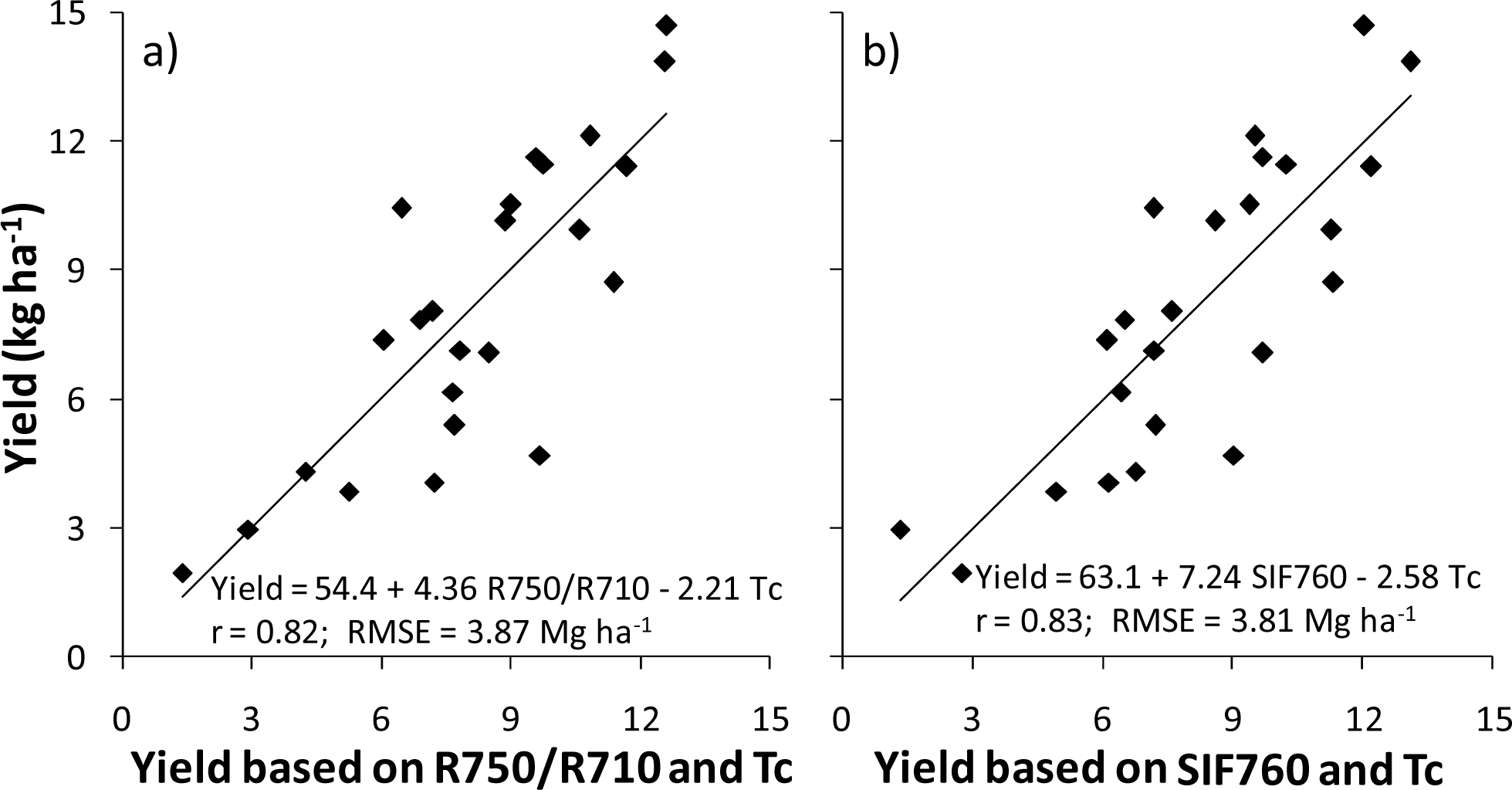
3.3. Application to N Fertilizer Recommendation
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Tilman, D.; Cassman, K.G.; Matson, P.A.; Naylor, R.; Polasky, S. Agricultural sustainability and intensive production practices. Nature 2002, 418, 671–677. [Google Scholar]
- Arregui, L.M.; Lasa, B.; Lafarga, A.; Irañeta, I.; Baroja, E.; Quemada, M. Evaluation of chlorophyll meters as tools for N fertilization in winter wheat under humid Mediterranean conditions. Eur. J. Agron 2006, 24, 140–148. [Google Scholar]
- Samborski, S.M.; Tremblay, N.; Fallon, E. Strategies to make use of plant sensors-based diagnostic information for nitrogen recommendations. Agron. J 2009, 101, 800–816. [Google Scholar]
- Martínez, D.E.; Guimat, J.J. Distortion of the SPAD 502 chlorophyll meter readings by changes in irradiance and leaf water status. Agronomie 2004, 24, 41–46. [Google Scholar]
- Fox, R.H.; Walthall, C.L. Crop Monitoring Technologies to Assess Nitrogen Status. In Nitrogen in Agricultural Systems, Agronomy Monograph 49; Schepers, J.S., Raun, W.R., Eds.; ASA, CSSA, SSSA: Madison, WI, USA, 2008; Chapter 16; pp. 647–674. [Google Scholar]
- Piekielek, W.P.; Fox, R.H.; Toth, J.D.; Macneal, K.E. Use of a chlorophyll meter at the early dent stage of corn to evaluate nitrogen sufficiency. Agron. J 1995, 87, 403–408. [Google Scholar]
- Monje, O.A.; Bugbee, B. Inherent limitations of nondestructive chlorophyll meters: A comparison of two types of meters. HortScience 1992, 27, 69–71. [Google Scholar]
- Cerovic, Z.G.; Ounis, A.; Cartelat, A.; Latouche, G.; Goulas, Y.; Meyer, S.; Moya, I. The use of chlorophyll fluorescence excitation spectra for the non-destructive in situ assessment of UV absorbing compounds in leaves. Plant Cell Environ 2002, 25, 1663–1676. [Google Scholar]
- Tremblay, N.; Wang, Z.; Cerovic, Z.G. Sensing crop nitrogen status with fluorescence indicators. A review. Agron. Sustain. Dev 2011, 32, 451–464. [Google Scholar]
- Hamilton, J.; Zangerl, A.; De Lucia, E.; Berenbaum, M. The carbon-nutrient balance hypothesis: Its rise and fall. Ecol. Lett 2001, 4, 86–95. [Google Scholar]
- Cartelat, A.; Cerovic, Z.G.; Goulas, Y.; Meyer, S.; Lelarge, C.; Prioul, J.L.; Barbottin, A.; Jeuffroy, M.H.; Gate, P.; Agati, G.; et al. Optically assessed contents of leaf polyphenolics and chlorophyll as indicators of nitrogen deficiency in wheat (Triticum. aestivum L.). Field Crop. Res 2005, 91, 35–49. [Google Scholar]
- Zarco-Tejada, P.J.; Miller, J.R.; Mohammed, G.H.; Noland, T.L.; Sampson, P.H. Scaling-up and model inversion methods with narrow-band optical indices for chlorophyll content estimation in closed forest canopies with hyperspectral data. IEEE Trans. Geosci. Remote Sens 2001, 39, 1491–1507. [Google Scholar]
- Kyveryga, P.M.; Blackmer, T.M.; Pearson, R. Normalization of uncalibrated late-season digital aerial imagery for evaluating corn nitrogen status. Prec. Agric 2012, 13, 2–16. [Google Scholar]
- Ritchie, G.L.; Sullivan, D.G.; Vencill, W.K.; Bednarz, C.W.; Hook, J.E. Sensitivities of normalized difference vegetation index and a green/red ratio index to cotton ground cover fraction. Crop Sci 2010, 50, 1000–1010. [Google Scholar]
- Rouse, J.W.; Haas, R.H.; Schell, J.A.; Deering, D.W.; Harlan, J.C. Monitoring the Vernal Advancements and Retrogradation of Natural Vegetation; NASA/GSFC: Greebelt, MD, USA, 1974. [Google Scholar]
- Pinter, P.J., Jr.; Hatfield, J.L.; Schepers, J.S.; Barnes, E.M.; Moran, M.S.; Daughtry, C.S.; Upchurch, D.R. Remote sensing for crop management. Photogramm. Eng. Remote Sens 2003, 69, 647–664. [Google Scholar]
- Reed, J.J.; Tarpley, L.; McKinion, J.M.; Reddy, K.R. Narrow-waveband reflectance ratios for remote estimation of nitrogen status in cotton. J. Environ. Qual 2002, 31, 1442–1452. [Google Scholar]
- Zarco-Tejada, P.J.; González Dugo, V.; Berni, J.A.J. Fluorescence, temperature and narrowband indices acquired from a UAV platform for water stress detection using a micro-hyperspectral imager and a thermal camera. Remote Sens. Environ 2012, 117, 322–337. [Google Scholar]
- Zarco-Tejada, P.J.; Morales, A.; Testi, L.; Villalobos, F.J. Spatio-temporal patterns of chlorophyll fluorescence and physiological and structural indices acquired from hyperspectral imagery as compared with carbon fluxes measured with eddy covariance. Remote Sens. Environ 2013, 133, 102–115. [Google Scholar]
- Zarco-Tejada, P.J.; Catalina, A.; González, M.R.; Martín, P. Relationships between net photosynthesis and steady-state chlorophyll fluorescence retrieved from airborne hyperspectral imagery. Remote Sens. Environ 2013, 136, 247–258. [Google Scholar]
- Soil Survey Staff. Keys to Soil Taxonomy, 9th ed.; USDA, Natural Resources Conservation Service: Washington, DC, USA, 2003. [Google Scholar]
- Papadakis, J. Climates of the World and Their Agricultural Potentialities; DAPCO: Rome, Italy, 1966. [Google Scholar]
- Gabriel, J.L.; Quemada, M. Replacing bare fallow with cover crops in a maize cropping system: Yield, N uptake and fertiliser fate. Eur. J. Agron 2011, 34, 133–143. [Google Scholar]
- Allen, R.G.; Pereira, L.S.; Raes, D.; Smith, M. FAO 56 Irrigation and Drainage Paper: Crop Evapotranspiration; Food and Agriculture Organization: Rome, Italy, 1998. [Google Scholar]
- Martínez-Cob, A. Use of thermal units to estimate corn crop coefficients under semiarid climatic conditions. Irrig. Sci 2008, 26, 335–345. [Google Scholar]
- Lancashire, P.D.; Bleiholder, H.; Langelüddecke, P.; Stauss, R.; van Den Boom, T.; Weber, E.; Witzen-Berger, A. An uniform decimal code for growth stages of crops and weeds. Ann. Appl. Biol 1991, 119, 561–601. [Google Scholar]
- Yadava, U.L. A rapid and nondestructive method to determine chlorophyll in intact leaves. Hortscience 1986, 21, 1449–1450. [Google Scholar]
- Tremblay, N.; Wang, Z.; Bélec, C. Evaluation of the Dualex for the assessment of corn nitrogen status. J. Plant Nutr 2007, 30, 1355–1369. [Google Scholar]
- Berni, J.A.J.; Zarco-Tejada, P.J.; Suárez, L.; Fereres, E. Thermal and narrow-band multispectral remote sensing for vegetation monitoring from an unmanned aerial vehicle. IEEE Trans. Geosci. Remote Sens 2003, 47, 722–738. [Google Scholar]
- Rougean, J.L.; Breon, F.M. Estimating PAR absorbed by vegetation from bidirectional reflectance measurements. Remote Sens. Environ 1995, 51, 375–384. [Google Scholar]
- Haboudane, D.; Miller, J.R.; Tremblay, N.; Zarco-Tejada, P.J.; Dextraze, L. Integrated narrow-band vegetation indices for prediction of crop chlorophyll content for application to precision agriculture. Remote Sens. Environ 2002, 81, 416–426. [Google Scholar]
- Gamon, J.A.; Peñuelas, J.; Field, C.B. A narrow-wave band spectral index that tracks diurnal changes in photosynthetic efficiency. Remote Sens. Environ 1992, 41, 35–44. [Google Scholar]
- Zarco-Tejada, P.J.; Berjón, A.; López-Lozano, R.; Miller, J.R.; Marin, P.; Cachorro, V.; González, M.R.; de Frutos, A. Assessing vineyard condition with hyperspectral indices: Leaf and canopy reflectance simulation in a row-structured discontinuous canopy. Remote Sens. Environ 2005, 99, 271–287. [Google Scholar]
- Varvel, G.E.; Schepers, J.S.; Francis, D.D. Ability for in season correction of nitrogen deficiency in corn using chlorophyll meter. Soil Sci. Soc. Am. J 1997, 61, 1233–1239. [Google Scholar]
- Chen, P.; Haboudane, D.; Tremblay, N.; Wang, J.; Vigneault, P.; Li, B. New spectral indicator assesing the efficiency of crop nitrogen treatment in corn and wheat. Remote Sens. Environ 2010, 114, 1987–1997. [Google Scholar]
- Blackmer, T.M.; Schepers, J.S.; Varvel, G.E. Light reflectance compared with other nitrogen stress measurements in corn leaves. Agron. J 1996, 6, 934–938. [Google Scholar]
- Tarpley, L; Reddy, K.R.; Sassenrath-Cole, G.F. Reflectance indices with precision and accuracy in predicting cotton leaf nitrogen concentration. Crop Sci 2002, 40, 1814–1819. [Google Scholar]
- Rodriguez, D.; Fitzgerald, G.J.; Belford, R. Detection of nitrogen deficiency in wheat from spectral reflectance indices and basic crop eco-physiological concepts. Aust. J. Agric. Res 2006, 57, 781–789. [Google Scholar]
- Moran, M.S.; Clarke, T.R.; Inoue, Y.; Vidal, A. Estimating crop water deficit using the relationship between surface air temperature and spectral vegetation index. Remote Sens. Environ 1994, 46, 246–263. [Google Scholar]
- Fitzgerald, G.J.; Rodriguez, D.; Christenssen, L.K.; Belford, R.; Sadras, V.O.; Clarke, T.R. Spectral and thermal sensing for nitrogen and water status in rainfed and irrigated wheat environments. Prec. Agric 2006, 1, 1–16. [Google Scholar]
- Kostrzewski, M.; Waller, P.; Guertin, P.; Haberland, J.; Colaizzi, P.; Barnes, E.; Thompson, T.; Clarke, T.; Riley, E.; Choi, C. Ground-based remote sensing of water and nitrogen stress. Trans. ASAE 2003, 46, 29–38. [Google Scholar]
- El-Shikha, D.M.; Waller, P.; Hunsaker, D.; Clarke, T.; Barnes, E. Ground-based remote sensing for assessing water and nitrogen status of broccoli. Agric. Water Manag 2007, 92, 183–193. [Google Scholar]
- Arregui, L.M.; Quemada, M. Strategies to improve nitrogen-use efficiency in winter cereal crops under rainfed Mediterranean conditions. Agron. J 2008, 100, 277–284. [Google Scholar]
- Quemada, M.; Baranski, M.; de Lange, M.N.J.; Vallejo, A.; Cooper, J.M. Meta-analysis of strategies to control nitrate leaching in irrigated agricultural systems and their effects on crop yield. Agric. Ecosyst. Environ 2013, 174, 1–10. [Google Scholar]
- Jacquemoud, S.; Verhoef, W.; Baret, F.; Bacour, C.; Zarco-Tejada, P.J.; Asner, G.P.; François, C.; Ustin, S.L. PROSPECT + SAIL models: A review of use for vegetation characterization. Remote Sens. Environ 2009, 113, S56–S66. [Google Scholar]
- Martens, H.; Naes, T. Multivariate Calibration by Data Compression. In Near-Infrared Technology in Agriculture and Food Industries; Williams, N., Ed.; American Association of Cereal Chemists: St. Paul, MN, USA, 1987; pp. 57–87. [Google Scholar]
- Atzberger, C.; Guérif, M; Baret, F; Werner, W. Comparative analysis of three chemometric techniques for the spectroradiometric assessment of canopy chlorophyll content in winter wheat. Comput. Electron. Agric 2008, 73, 165–173. [Google Scholar]

| Index | Definition |
|---|---|
| SPAD | SPAD-502® |
| Ratio of the transmitted light at the red and infrared wavelengths | |
| Dualex® Scientific | |
| Chl | Ratio of the transmitted light at two different infrared wavelengths |
| FLAV | Log of the fluorescence emission ratio at the red and UV wavelengths |
| NBI | Nitrogen Balance Index = Chl/FLAV |
| Multiplex® | |
| SFR * | Simple fluorescence emission ratio at the red and far-red wavelengths |
| NBI-G † | Nitrogen Balance Index: ratio between the far-red fluorescence under UV excitation (∼ Chl) and the red fluorescence (∼FLAV) |
| ANTH | Log of the fluorescence emission ratio under red and green excitation |
| FERARI | Transformed single-fluorescence excitation anthocyanin relative index |
| Index | Equation |
|---|---|
| Greenness indices | |
| Normalized Difference Vegetation Index (NDVI) | NDVI = (R800 − R670)/(R800 + R670) |
| Reformed Difference Vegetation Index (RDVI) | RDVI = (R800 − R670)/(R800 + R670)°.5 |
| Chlorophyll indices | |
| Red edge optical reflectance | R750/R710 |
| Red to near infrared optical reflectance | R700/R760 |
| Transformed Chlorophyll Absorption in Reflectance Index (TCARI) | TCARI = 3 [(R700 − R670) − 0.2 (R700 − R550)/(R700/R670)] |
| Optimized soil-adjusted vegetation index (OSAVI) | OSAVI = (1 + 0.16) × (R800 − R670)/(R800 + R670 + 0.16) |
| Combined TCARI/OSAVI | TCARI/OSAVI |
| Xanthophyll indices | |
| Photochemical Reflectance Index (PRI) | PRI = (R570 − R539)/(R570 + R539) |
| Normalized Photochemical Reflectance Index (PRI norm) | PRI norm = (R515 − R531)/(R515 + R531) |
| Blue/green/red ratio indices | |
| BGI1 | BGI1 = R400/R550 |
| BGI2 | BGI2 = R450/R550 |
| Fluorescence retrieval | |
| Fluorescence (SIF760) | FLD3 method using 2 reference bands (750; 762; 780) |
| Treatment * | SPAD® | Dualex® | Multiplex® | |||||
|---|---|---|---|---|---|---|---|---|
| Chl | FLAV | NBI | SFR | NBI-G | ANTH | FERARI | ||
| First sampling date: stem elongation | ||||||||
| 0 | 45.3 (1.2) | 45.7 (1.1) | 1.40 (0.06) | 34.3 (2.2) | 6.7 (0.3) | 5.6 (0.4) | 0.507 (0.010) | 1.46 (0.02) |
| 40 | 47.3 (0.9) | 48.9 (1.0) | 1.39 (0.04) | 36.3 (1.4) | 7.1 (0.5) | 6.6 (0.5) | 0.494 (0.006) | 1.44 (0.02) |
| 80 | 46.6 (0.2) | 47.4 (0.6) | 1.29 (0.04) | 38.3 (1.4) | 6.0 (0.2) | 6.3 (0.5) | 0.486 (0.002) | 1.35 (0.05) |
| 120 | 49.8 (0.5) | 50.3 (0.5) | 1.34 (0.02) | 38.8 (0.6) | 7.4 (0.6) | 6.5 (0.4) | 0.507 (0.006) | 1.43 (0.06) |
| 160 | 49.5 (0.4) | 50.2 (1.1) | 1.25 (0.04) | 42.2 (0.3) | 6.0 (0.2) | 7.3 (0.5) | 0.498 (0.007) | 1.40 (0.04) |
| 200 | 48.0 (1.1) | 50.9 (0.5) | 1.19 (0.15) | 44.3 (5.4) | 6.3 (0.3) | 7.2 (1.7) | 0.478 (0.001) | 1.37 (0.04) |
| Second sampling date: flowering | ||||||||
| 0 | 38.9 (2.6) | 23.7 (1.8) | 1.02 (0.06) | 24.3 (2.6) | 3.7 (0.2) | 5.4 (0.5) | 0.489 (0.009) | 0.68 (0.02) |
| 40 | 44.7 (1.1) | 27.7 (1.4) | 1.00 (0.02) | 27.9 (1.7) | 4.3 (0.2) | 5.7 (0.3) | 0.473 (0.004) | 0.68 (0.02) |
| 80 | 45.3 (0.6) | 27.7 (0.7) | 0.94 (0.02) | 29.7 (0.9) | 4.2 (0.1) | 6.4 (0.1) | 0.471 (0.003) | 0.67 (0.02) |
| 120 | 46.9 (2.1) | 29.0 (1.8) | 0.90 (0.01) | 32.2 (1.6) | 4.3 (0.3) | 6.6 (0.2) | 0.471 (0.007) | 0.68 (0.03) |
| 160 | 46.6 (2.2) | 29.0 (2.7) | 0.92 (0.01) | 31.4 (2.7) | 4.4 (0.3) | 7.0 (0.2) | 0.464 (0.005) | 0.70 (0.03) |
| 200 | 49.1 (0.3) | 30.1 (0.1) | 0.89 (0.04) | 34.0 (1.4) | 4.4 (0.2) | 7.1 (0.4) | 0.464 (0.002) | 0.68 (0.02) |
| SPAD | Chl | FLAV | NBI | SFR | NBI-G | ANTH | FERARI | NDVI | RDVI | R750/R710 | R700/R760 | TCARI | TCARI/OSAVI | PRI | PRI norm | BGI 1 | BGI 2 | SIF 760 | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Chl | 0.90 * | ||||||||||||||||||
| FLAV | −0.27 | −0.40 | |||||||||||||||||
| NBI | 0.58 | 0.73 | −0.90 * | ||||||||||||||||
| SFR | 0.41 | 0.39 | 0.14 | 0.09 | |||||||||||||||
| NBI-G | 0.44 | 0.52 | −0.68 | 0.76 | 0.5 | ||||||||||||||
| ANTH | −0.25 | −0.28 | 0.57 | −0.53 | 0.34 | −0.35 | |||||||||||||
| FERARI | 0.31 | 0.19 | 0.36 | −0.19 | 0.6 | 0.06 | 0.17 | ||||||||||||
| NDVI | 0.34 | 0.28 | 0.2 | −0.01 | 0.48 | 0.09 | 0.07 | 0.67 | |||||||||||
| RDVI | 0.33 | 0.27 | 0.22 | −0.03 | 0.47 | 0.07 | 0.09 | 0.66 | 0.99 * | ||||||||||
| R750/R710 | 0.48 | 0.45 | 0.01 | 0.2 | 0.52 | 0.23 | 0.05 | 0.6 | 0.95 * | 0.95 * | |||||||||
| R700/R760 | 0.14 | 0.05 | 0.42 | −0.28 | 0.44 | −0.11 | 0.24 | 0.75 | 0.92 * | 0.91 * | 0.8 | ||||||||
| TCARI | −0.11 | −0.23 | 0.61 | −0.53 | 0.25 | −0.32 | 0.25 | 0.66 | 0.74 | 0.75 | 0.54 | 0.91 * | |||||||
| TCARI/ OSAVI | −0.42 | −0.53 | 0.72 | −0.74 | 0.02 | −0.51 | 0.29 | 0.46 | 0.33 | 0.35 | 0.08 | 0.63 | 0.88 | ||||||
| PRI | −0.34 | −0.29 | −0.18 | 0.01 | −0.43 | −0.06 | −0.04 | −0.64 | −0.96 * | −0.96 * | −0.95 * | −0.89 | −0.69 | −0.28 | |||||
| PRI norm | −0.27 | −0.20 | −0.27 | 0.1 | −0.41 | −0.01 | −0.04 | −0.66 | −0.99 * | −0.99 * | −0.92* | −0.92* | −0.78 | −0.41 | 0.97 | ||||
| BGI1 | 0.49 | 0.65 | −0.76 | 0.82 | 0 | 0.54 | −0.46 | −0.21 | 0.05 | 0.01 | 0.24 | −0.28 | −0.56 | −0.80 | −0.08 | 0.01 | |||
| BGI2 | −0.04 | 0.06 | −0.51 | 0.39 | −0.42 | 0.2 | −0.32 | −0.74 | −0.83 | −0.83 | −0.67 | −0.96* | −0.95* | −0.74 | 0.76 | 0.83 | 0.44 | ||
| SIF760 | 0.39 | 0.47 | −0.05 | 0.25 | 0.43 | 0.21 | −0.45 | 0.35 | 0.75 | 0.84 | 0.84 | 0.57 | 0.39 | 0.02 | −0.72 | −0.70 | 0.23 | −0.47 | 1 |
| Yield | 0.48 | 0.44 | −0.10 | 0.22 | −0.07 | −0.10 | 0.04 | 0.12 | 0.46 | 0.47 | 0.56 | 0.33 | 0.14 | −0.17 | −0.48 | −0.41 | 0.23 | −0.25 | 0.59 |
| Biomass | 0.43 | 0.38 | −0.18 | 0.26 | −0.09 | −0.04 | −0.08 | 0.07 | 0.43 | 0.44 | 0.51 | 0.28 | 0.1 | −0.20 | −0.43 | −0.38 | 0.27 | −0.21 | 0.49 |
| Grain N | 0.49 | 0.42 | −0.02 | 0.15 | −0.04 | −0.12 | 0.04 | 0.19 | 0.5 | 0.52 | 0.57 | 0.42 | 0.24 | −0.05 | −0.54 | −0.30 | −0.30 | −0.31 | 0.57 |
| N uptake | 0.5 | 0.47 | −0.18 | 0.3 | −0.09 | −0.03 | −0.01 | −0.01 | 0.37 | 0.38 | 0.48 | 0.2 | 0 | −0.29 | −0.38 | −0.31 | 0.3 | −0.12 | 0.15 |
| SPAD | Chl | FLAV | NBI | SFR | NBI-G | ANTH | FERARI | NDVI | RDVI | R750/R710 | R700/R760 | TCARI | TCARI/OSAVI | PRI | PRI norm | BGI1 | BGI2 | SIF 760 | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Chl | 0.91 * | ||||||||||||||||||
| FLAV | −0.37 | −0.28 | |||||||||||||||||
| NBI | 0.89 | 0.93 | −0.60 | ||||||||||||||||
| SFR | 0.88 | 0.88 | −0.05 | 0.73 | |||||||||||||||
| NBI-G | 0.74 | 0.69 | −0.81 | 0.87 | 0.53 | ||||||||||||||
| ANTH | −0.87 | −0.83 | 0.41 | −0.81 | −0.88 | −0.76 | |||||||||||||
| FERARI | 0.25 | 0.24 | 0.51 | 0.02 | 0.58 | −0.09 | −0.36 | ||||||||||||
| NDVI | 0.78 | 0.77 | 0.23 | 0.56 | 0.86 | 0.24 | −0.63 | 0.58 | |||||||||||
| RDVI | 0.65 | 0.63 | 0.36 | 0.39 | 0.8 | 0.14 | −0.56 | 0.75 | 0.93 * | ||||||||||
| R750/R710 | 0.94 * | 0.90 * | −0.18 | 0.8 | 0.94 * | 0.61 | −0.87 | 0.45 | 0.89 | 0.79 | |||||||||
| R700/R760 | −0.32 | −0.23 | 0.92 * | −0.50 | −0.09 | −0.77 | 0.49 | 0.4 | 0.29 | 0.39 | −0.15 | ||||||||
| TCARI | −0.64 | −0.58 | 0.84 | −0.76 | −0.42 | −0.83 | 0.7 | 0.29 | −0.14 | 0.09 | −0.53 | 0.87 | |||||||
| TCARI/OSAVI | −0.79 | −0.72 | 0.74 | −0.84 | −0.60 | −0.85 | 0.82 | 0.12 | −0.36 | −0.15 | −0.71 | 0.77 | 0.97 * | ||||||
| PRI | −0.83 | −0.84 | 0.06 | −0.70 | −0.85 | −0.43 | 0.69 | −0.31 | −0.86 | −0.73 | −0.89 | −0.02 | 0.4 | 0.57 | |||||
| PRI norm | −0.71 | −0.73 | −0.27 | −0.50 | −0.82 | −0.18 | 0.55 | −0.52 | −0.95 * | −0.90 * | −0.82 | −0.35 | 0.05 | 0.26 | 0.92 * | ||||
| BGI1 | 0.66 | 0.61 | −0.66 | 0.73 | 0.52 | 0.72 | −0.69 | −0.17 | 0.29 | 0.03 | 0.62 | −0.70 | −0.94 * | −0.94 * | −0.54 | −0.22 | |||
| BGI2 | 0.46 | 0.39 | −0.90 * | 0.63 | 0.25 | 0.82 | −0.60 | −0.35 | −0.10 | −0.26 | 0.33 | −0.97 * | −0.95 * | −0.88 | −0.18 | 0.17 | 0.85 | ||
| SIF760 | 0.65 | 0.55 | 0.1 | 0.41 | 0.78 | 0.35 | −0.65 | 0.74 | 0.88 | 0.95 | 0.77 | 0.12 | −0.07 | −0.26 | −0.71 | −0.82 | 0.17 | −0.02 | 1 |
| Yield | 0.67 | 0.62 | −0.06 | 0.54 | 0.55 | 0.38 | −0.45 | 0.26 | 0.61 | 0.57 | 0.67 | −0.04 | −0.29 | −0.42 | −0.55 | −0.52 | 0.33 | 0.16 | 0.58 |
| Biomass | 0.65 | 0.56 | −0.05 | 0.49 | 0.52 | 0.34 | −0.42 | 0.26 | 0.6 | 0.56 | 0.63 | −0.04 | −0.27 | −0.40 | −0.53 | −0.51 | 0.3 | 0.15 | 0.49 |
| Grain N | 0.64 | 0.59 | 0 | 0.49 | 0.55 | 0.3 | −0.43 | 0.31 | 0.64 | 0.61 | 0.66 | 0.03 | −0.23 | −0.37 | −0.56 | −0.12 | −0.18 | −0.22 | 0.57 |
| N uptake | 0.65 | 0.57 | −0.17 | 0.55 | 0.47 | 0.42 | −0.42 | 0.15 | 0.53 | 0.47 | 0.61 | −0.14 | −0.35 | −0.46 | −0.47 | −0.41 | 0.34 | 0.24 | −0.07 |
© 2014 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Quemada, M.; Gabriel, J.L.; Zarco-Tejada, P. Airborne Hyperspectral Images and Ground-Level Optical Sensors As Assessment Tools for Maize Nitrogen Fertilization. Remote Sens. 2014, 6, 2940-2962. https://doi.org/10.3390/rs6042940
Quemada M, Gabriel JL, Zarco-Tejada P. Airborne Hyperspectral Images and Ground-Level Optical Sensors As Assessment Tools for Maize Nitrogen Fertilization. Remote Sensing. 2014; 6(4):2940-2962. https://doi.org/10.3390/rs6042940
Chicago/Turabian StyleQuemada, Miguel, Jose Luis Gabriel, and Pablo Zarco-Tejada. 2014. "Airborne Hyperspectral Images and Ground-Level Optical Sensors As Assessment Tools for Maize Nitrogen Fertilization" Remote Sensing 6, no. 4: 2940-2962. https://doi.org/10.3390/rs6042940
APA StyleQuemada, M., Gabriel, J. L., & Zarco-Tejada, P. (2014). Airborne Hyperspectral Images and Ground-Level Optical Sensors As Assessment Tools for Maize Nitrogen Fertilization. Remote Sensing, 6(4), 2940-2962. https://doi.org/10.3390/rs6042940




