Remote Sensing of Shallow Coastal Benthic Substrates: In situ Spectra and Mapping of Eelgrass (Zostera marina) in the Gulf Islands National Park Reserve of Canada
Abstract
:1. Introduction
- 1.
- Can submerged eelgrass be spectrally distinguished from other submerged substrates? If so, what spectral variables (bands and/or band slopes/ratios) offer the most accurate separation among substrates with the least redundancy?
- 2.
- How effective are these spectral variables for detecting eelgrass in a hyperspectral airborne image?
- 3.
- What is the wavelength-dependent depth limit of eelgrass spectral detection in situ and in the image?
2. Methods
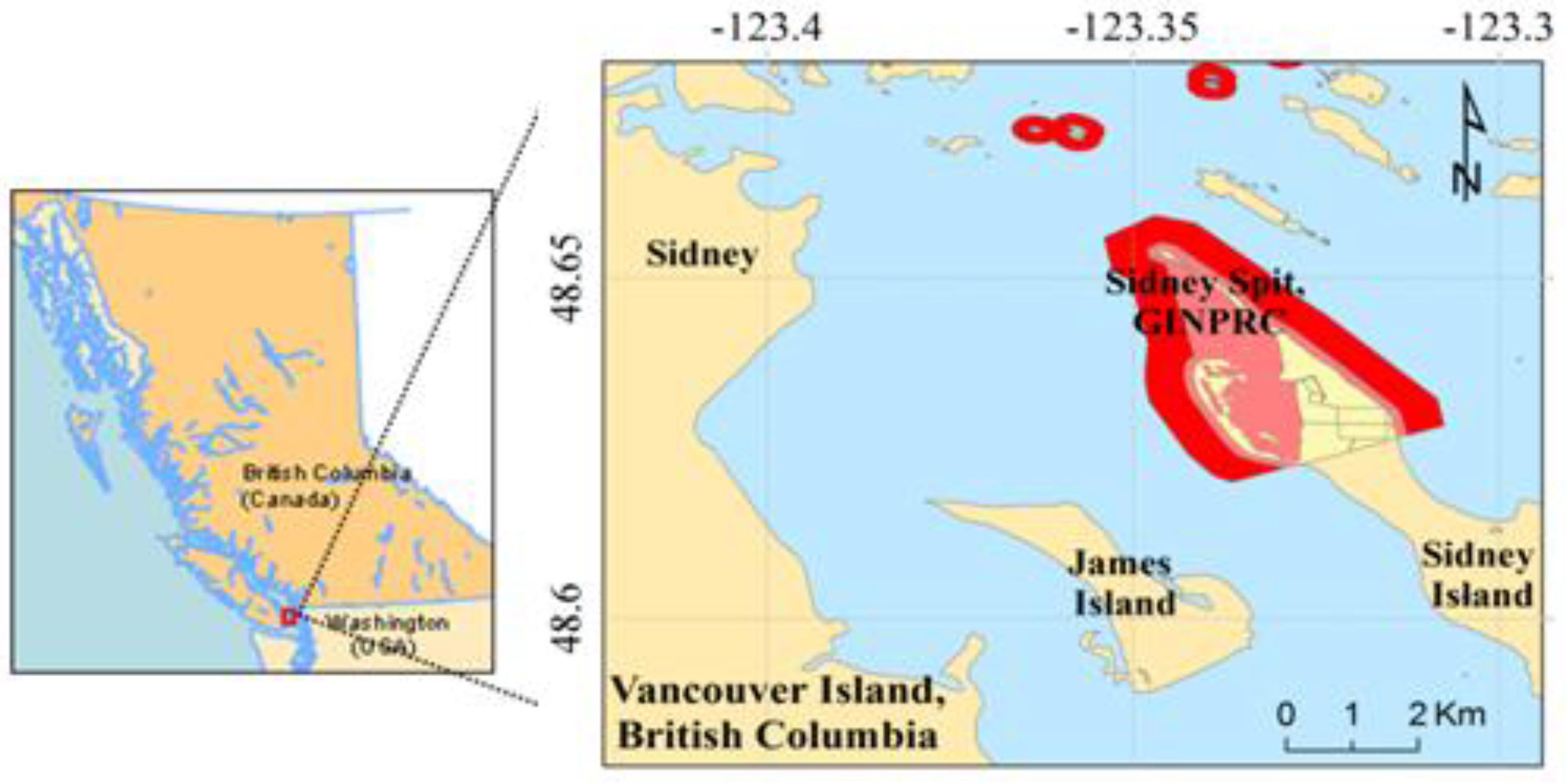
2.1. Study Area
| Average | Range | |
|---|---|---|
| Temperature | 11.5 °C | 11.3–12.2 °C |
| Salinity | 29 ppt | 28.9–29.4 ppt |
| aCDOM(440) | 0.24 m−1 | 0.18–0.33 m−1 |
| TSM | 4.03 g m−3 | 2.25–6.08 g m−3 |
| TOC | 47.60% | 32.7–65.3% |
| Chl-a | 2.44 mg m−3 | 1.28–7.23 mg m−3 |
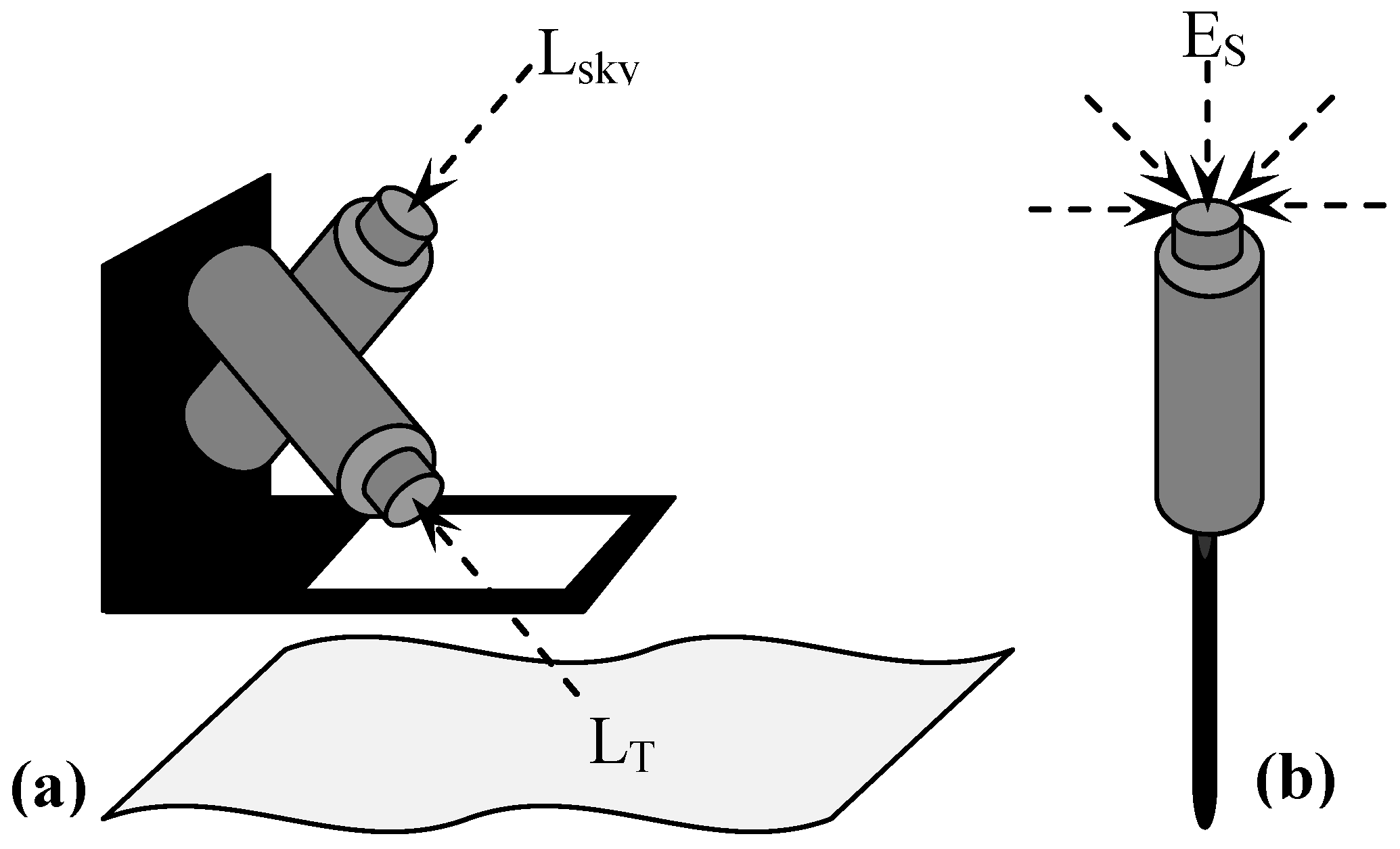
2.2. Data Acquisition
| Field photo | Common name | Latin name | Abbr. | Above-water spectra (n) |
|---|---|---|---|---|
 | Eelgrass | Zostera marina | sE dE | 21 shallow 4 deep |
 | Green algae: Sea Lettuce | Ulva fenestrata | sAg | 6 shallow 0 deep |
 | Green algae: Filamentous | Enteromorpha spp. | sAg | 2 shallow 0 deep |
 | Sand | - | sS dS | 6 shallow 3 deep |
 | Deep water (>30 m) | - | dW | 7 |
2.3. Data Reduction and Variable Selection
Identifying Major Spectral Variables
| Wavelengths | Slope(s) Ratio(r) | Description | Reference |
|---|---|---|---|
| 480, 490 | s,r | Chl blue absorption | This study |
| 500, 530 | s,r | Green peak: Ascending edge to 530 nm shoulder | This study |
| 536, 566 | s,r | Greek peak: Ascending edge shoulder to max | This study |
| 500, 566 | r | Max blue absorption: Max green reflectance | This study |
| 566, 600 | s,r | Green peak: Descending edge | This study |
| 620, 640 | s,r | Chl-b & water red absorption: Descending slope | This study |
| 630, 640 | s,r | Detect diatom epiphytes | This study |
| 650, 660 | s,r | Chl red absorption: Descending edge | This study |
| 566, 670 | r | Chl max green reflectance: Max red absorption | [63] (672:550) |
| 566, 694 | r | Green peak: Sand NIR peak | This study |
| 566, 710 | r | Green peak: Vegetation NIR peak | [64] |
| 570, 710 | r | Green peak: Vegetation NIR peak | This study |
| 670, 700 | s,r | Red edge: Correlated with chlorophyll-a | [65] |
| 668, 710 | r | Red edge | [66] (RVI) |
| 690, 710 | r | NIR peak position | [64] |
Variable Selection: M-Statistic and Discriminant Analysis
2.4. Case Study: Classification of Hyperspectral Airborne Image
- Image 1:
- Full spectral resolution above-water image
- Image 1R:
- Reduced variable above-water image (Set 2 from previous)
- Image 2:
- Full spectral resolution water-corrected image
- Image 2R:
- Reduced variable water-corrected image (Set 2 from previous)
| Substrate Classes | Substrate Abbr. | Ground-truth Survey Sites | AISA Training/Validation Sites (~20%/80%) |
|---|---|---|---|
| Shallow Eelgrass | (sE) | 265 | 51/214 |
| Deep Eelgrass | (dE) | 19 | 4/15 |
| Green Algae | (sAg) | 129* | 24/105 |
| Shallow Sand | (sS) | 49 | 10/39 |
| Deep Sand | (dS) | 32 | 6/26 |
| Brown Algae | (sAb) | 7 | 2/5 |
| Exposed Sea Asparagus | (eAsp) | 6 | 2/4 |
3. Results
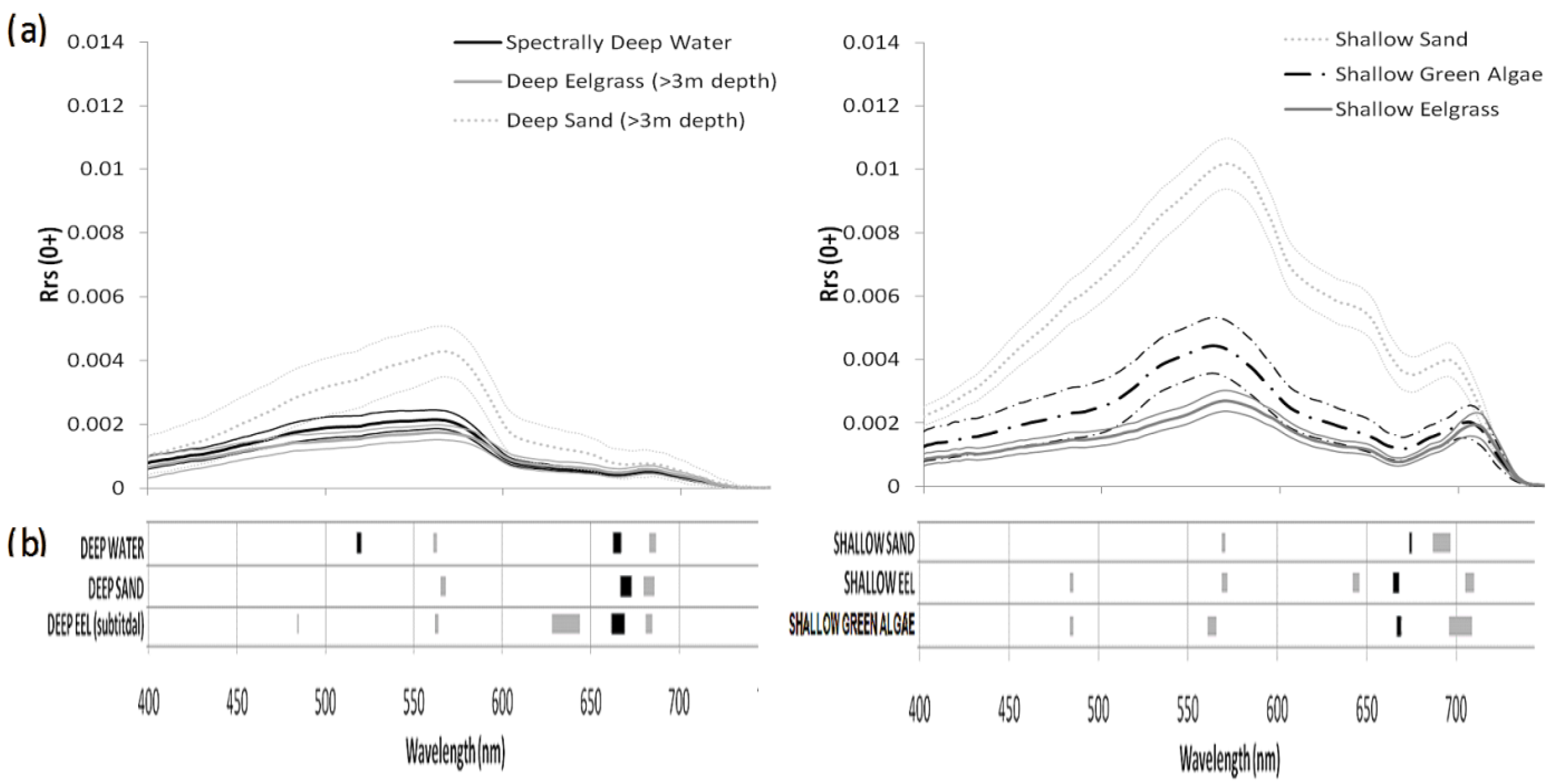
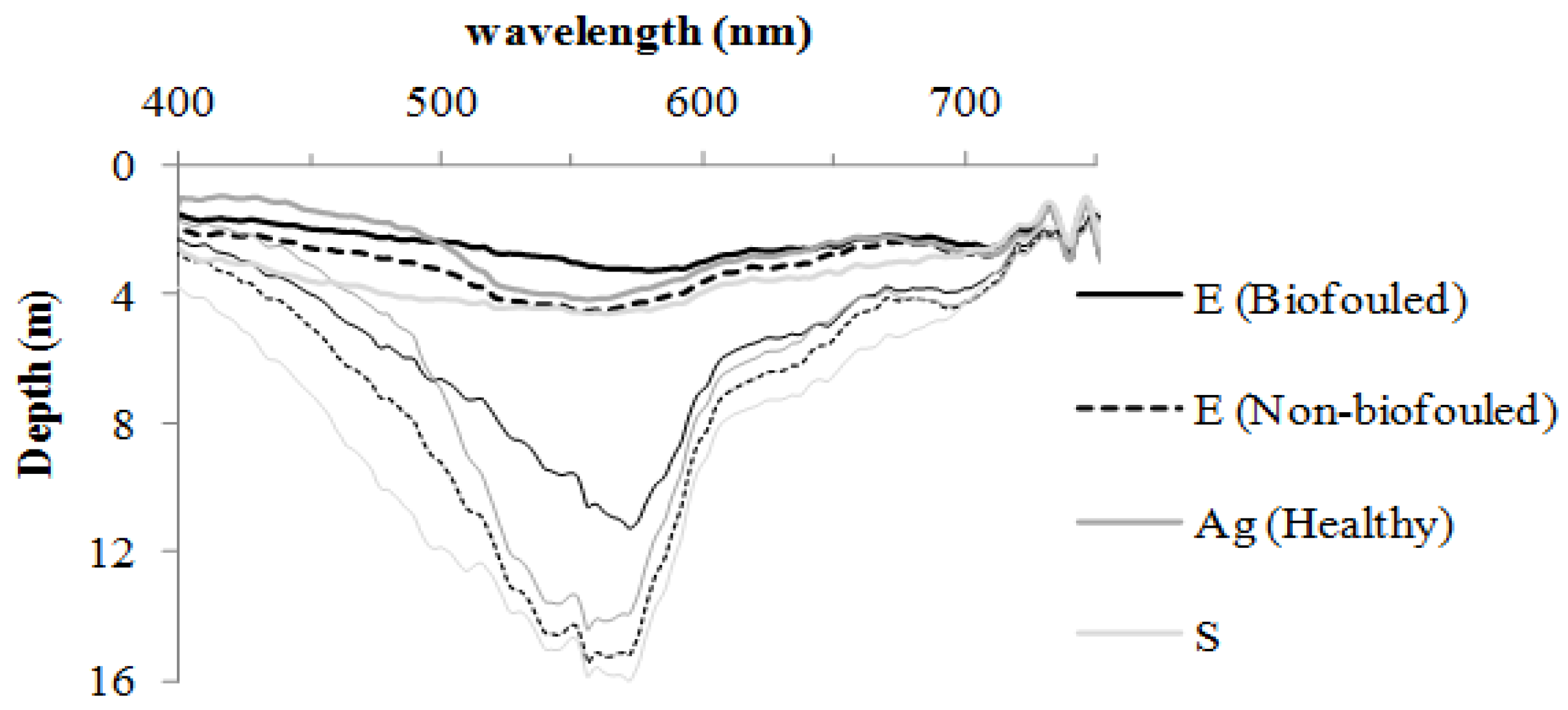
3.1. Spectral Characteristics of the Benthic Substrates
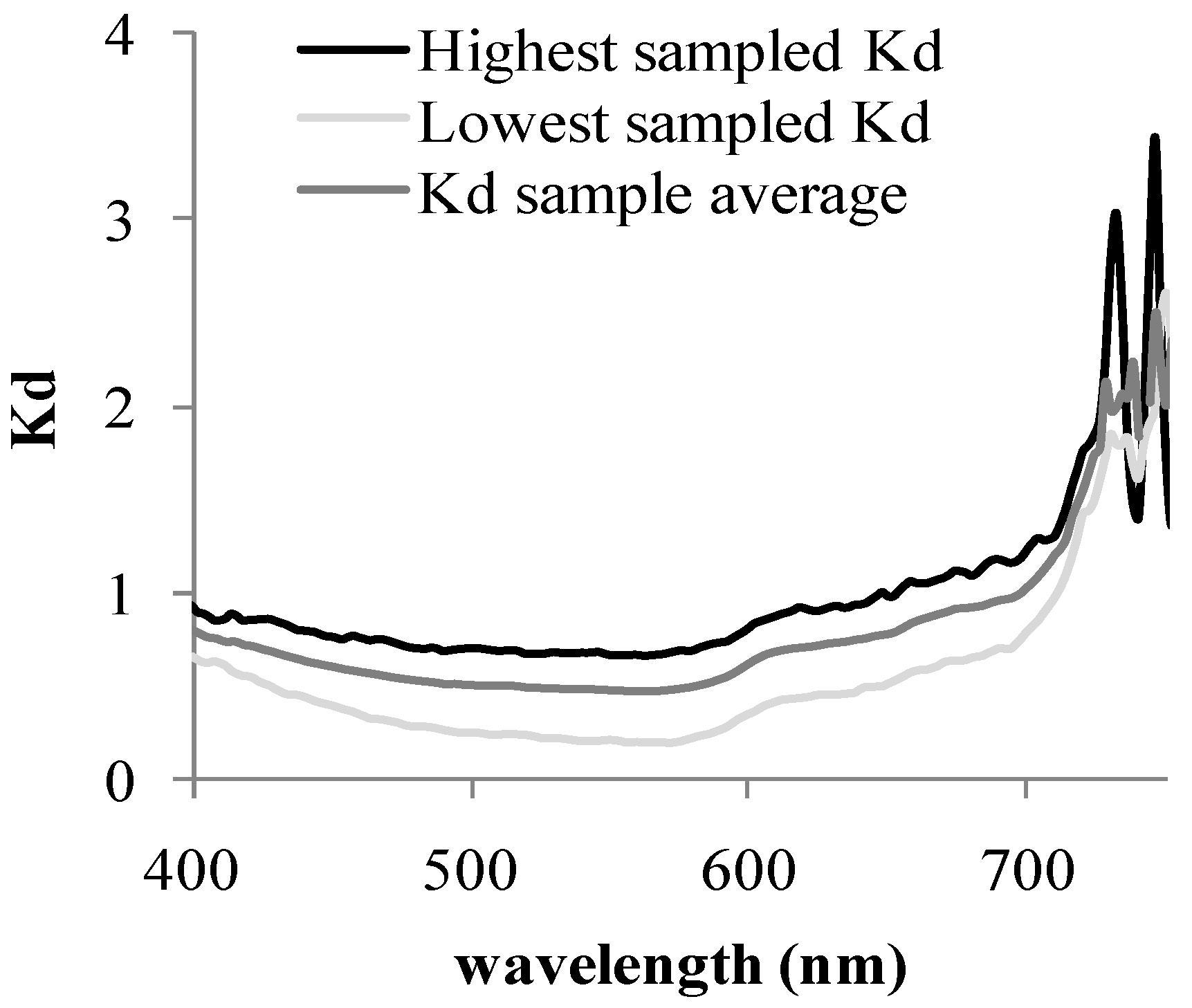
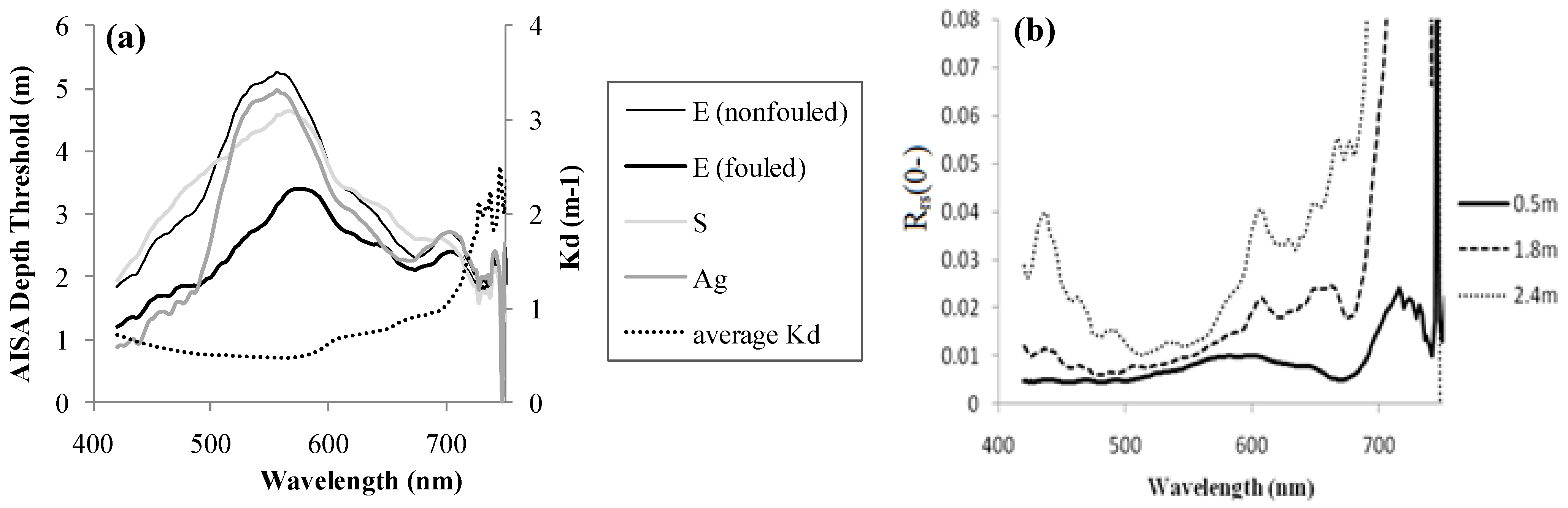
3.2. Water Correction
3.3. Spectral Variable Selection
Indices
| Band | sS | sAg | dE | dS | dW |
|---|---|---|---|---|---|
| s500-530 | 5.73 | 1.63 | 1.90 | 0.49 | 1.76 |
| R'566 | 0.56 | 1.40 | 2.51 | 0.68 | 2.80 |
| R'580 | 2.10 | 1.64 | 0.33 | 3.21 | 1.05 |
| R'602 | 5.70 | 0.64 | 0.56 | 2.46 | 0.25 |
| Band | sS | sAg | sE | dS | dW |
|---|---|---|---|---|---|
| s500-530 | 11.45 | 2.91 | 1.90 | 4.55 | 0.09 |
| R'566 | 1.62 | 0.56 | 2.51 | 1.47 | 1.10 |
| R'580 | 3.86 | 2.93 | 0.33 | 6.60 | 2.10 |
| R'602 | 8.93 | 1.19 | 0.56 | 4.22 | 0.26 |
| Band | sS | sAg |
|---|---|---|
| 566:600 | 0.78 | 1.23 |
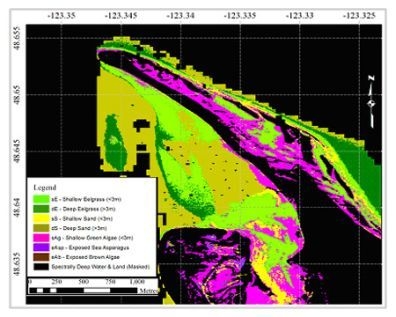
3.4. Case Study: Classification of a Hyperspectral Airborne Image
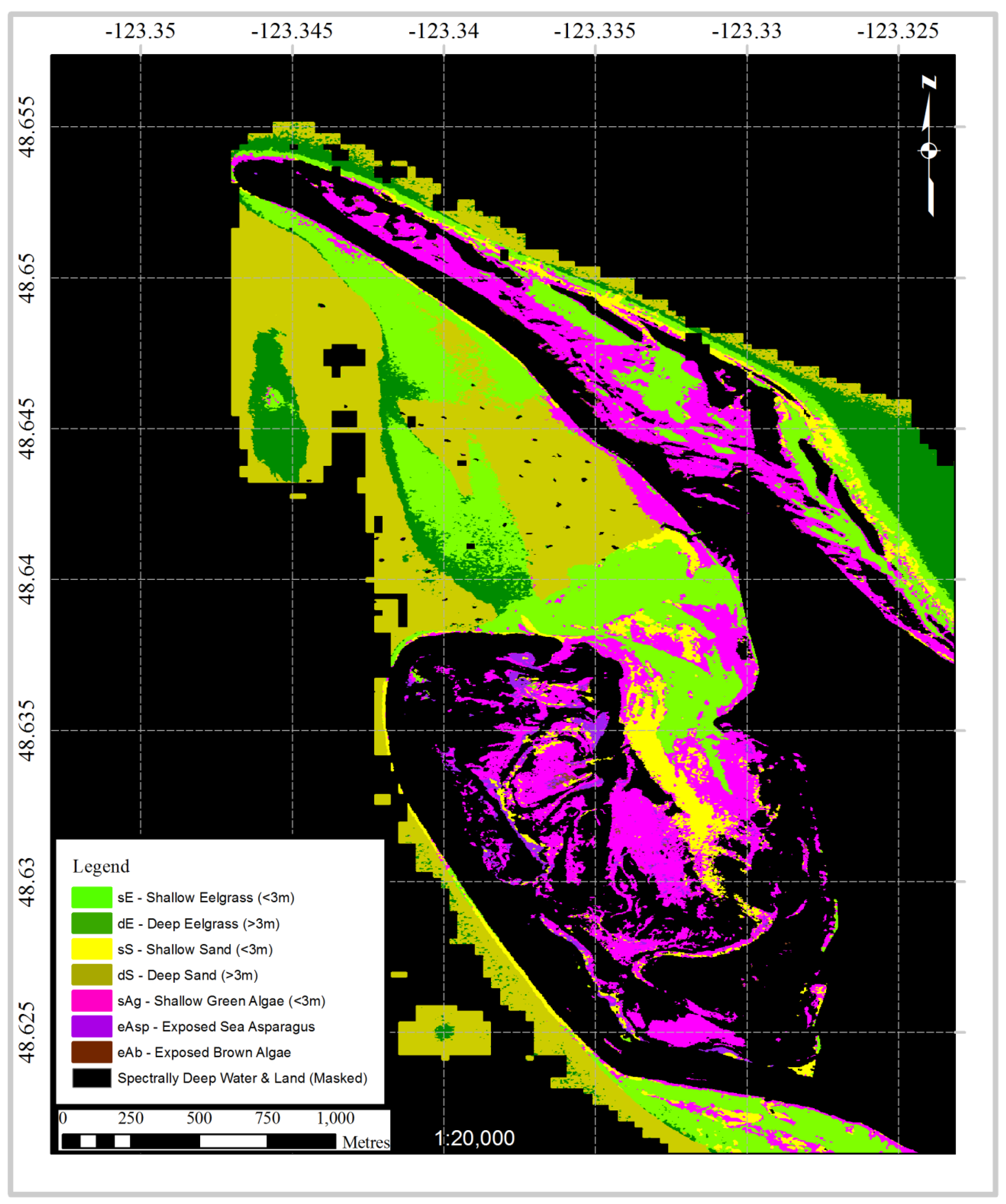
4. Discussion
Can Submerged Eelgrass be spectrally Distinguished from other Submerged Substrates? If so, What Spectral Variables (Bands and/or Band Slopes/Ratios) Offer the Most Accurate Separation among Substrates with the Least Redundancy?
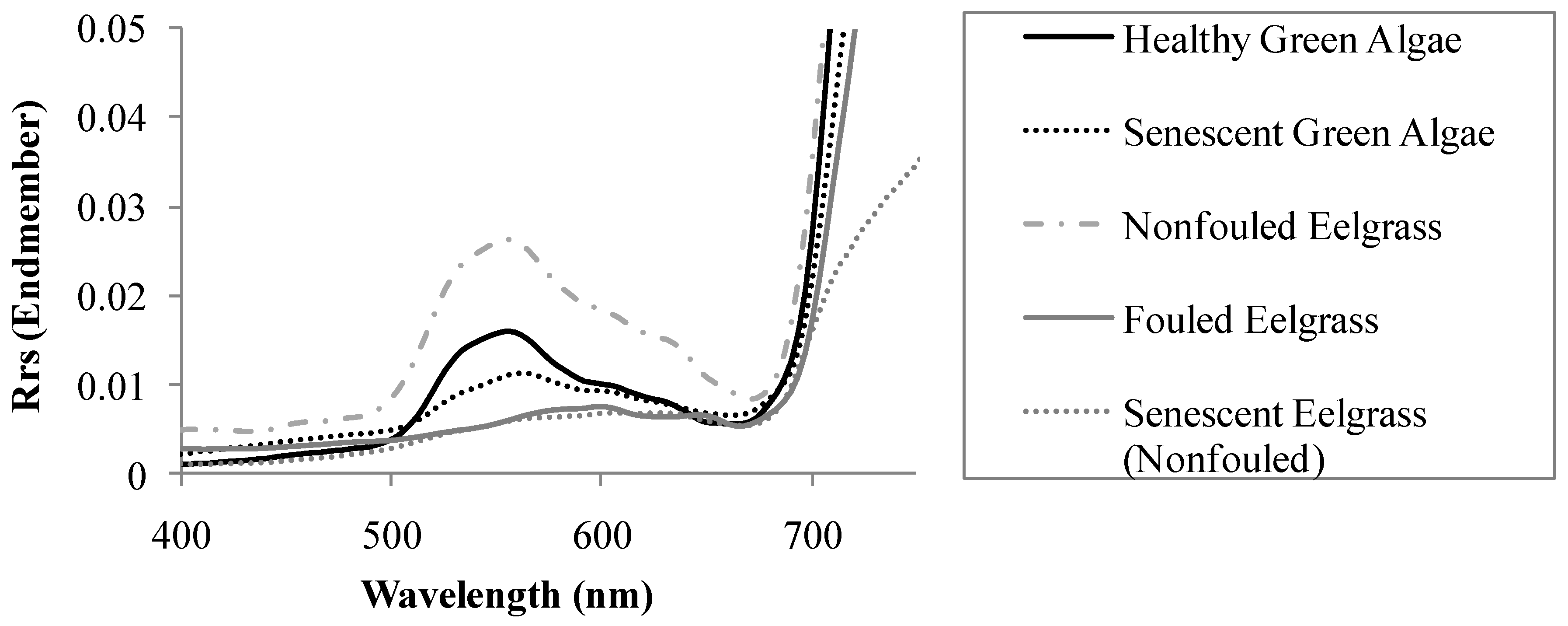
How Effective are these Spectral Variables for Detecting Eelgrass in a Hyperspectral Airborne Image?
What is the Wavelength-Dependent Depth Limit of Eelgrass Detection in situ and in the Image?
5. Conclusions
Acknowledgements
References and Notes
- Hemming, M.A.; Duarte, C.M. Seagrass Ecology; Cambridge University Press: Cambridge, UK, 2000. [Google Scholar]
- Jackson, J.B.C.; Kirby, M.X.; Berger, W.H.; Bjorndal, K.A.; Botsford, L.W.; Bourque, B.J.; Bradbury, R.H.; Cooke, R.; Erlandson, J.; Estes, J.A.; Hughes, T.P.; Kidwell, S.; Lange, C.B.; Lenihan, H.S.; Pandolfi, J.M.; Peterson, C.H.; Steneck, R.S.; Tegner, M.J.; Warner, R.R. Historical overfishing and the recent collapse of coastal ecosystems. Science 2001, 293, 629–638. [Google Scholar] [CrossRef] [PubMed]
- Fonseca, M.; Cahalan, J. A preliminary evaluation of wave attenuation by four species of seagrass. Estuar. Coast. Shelf Sci. 1992, 35, 565–576. [Google Scholar] [CrossRef]
- Apostolaki, E.T.; Marba, N.; Holmer, M.; Karakassis, I. Fish farming enhances biomass and nutrient loss in Posidonia oceanica (L.). Estuar. Coast. Shelf Sci. 2009, 81, 390–400. [Google Scholar] [CrossRef]
- Duarte, C.M.; Chiscano, C.M. Seagrass biomass and production: A reassessment. Aquat. Bot. 1999, 65, 159–174. [Google Scholar] [CrossRef]
- Sewell, A.T.; Norris, J.G.; Wyllie-Echeverria, S.; Skalski, J.R. Eelgrass monitoring in Puget Sound: Overview of the Submerged Vegetation Monitoring Program. In Puget Sound Research 2001; Puget Sound Water Quality Action Team: Olympia, WA, USA, 2001; pp. 98504–0900. [Google Scholar]
- Borg, J.A.; Rowden, A.A.; Attrill, M.J.; Schembri, P.J.; Jones, M.B. Wanted dead or alive: High diversity of macroinvertebrates associated with living and “dead” Posidonia oceanica matte. Mar. Biol. 2006, 149, 667–677. [Google Scholar] [CrossRef]
- Thistle, M.E.; Schneider, D.C.; Gregory, R.S.; Wells, N.J. Fractal measures of habitat structure: Maximum densities of juvenile cod occur at intermediate eelgrass complexity. Mar. Ecol. Progr. Ser. 2010, 405, 39–56. [Google Scholar] [CrossRef]
- Adams, S.M. Feeding ecology of reef fish communities. Trans. Am. Fish. Soc. 1976, 105, 514–519. [Google Scholar] [CrossRef]
- Duarte, C.M. The future of seagrass meadows. Environ. Conser. 2002, 29, 192–206. [Google Scholar] [CrossRef]
- Burkholder, J.; Tomasko, D.; Touchette, B. Seagrasses and eutrophication. J. Exp. Mar. Biol. Ecol. 2007, 350, 46–72. [Google Scholar] [CrossRef]
- Walker, D.I.; Lukatelich, R.J.; Bastyan, G.; McComb, A.J. Effect of boat moorings on seagrass beds near Perth, Western Australia. Aquat. Bot. 1989, 36, 69–77. [Google Scholar] [CrossRef]
- Marba, N.; Santiago, R.; Diazalmela, E.; Alvarez, E.; Duarte, C. Seagrass (Posidonia oceanica) vertical growth as an early indicator of fish farm-derived stress. Estuar. Coast. Shelf Sci. 2006, 67, 475–483. [Google Scholar] [CrossRef]
- Diaz-Almela, E.; Marba, N.; Alvarez, E.; Santiago, R.; Holmer, M.; Grau, A.; Danovaro, R.; Argyrou, M.; Karakassis, I.; Duarte, C.M. Benthic input rates predict seagrass (Posidonia oceanica) fish farm-induced decline. Mar. Pollut. Bull. 2008, 56, 1332–1342. [Google Scholar] [CrossRef] [PubMed]
- Short, F.T.; Wyllie-Echeverria, S. Natural and human-induced disturbance of seagrasses. Environ. Conser. 1996, 23, 17–27. [Google Scholar] [CrossRef]
- Moore, K.; Wetzel, R. Seasonal variations in eelgrass (Zostera marina L.) responses to nutrient enrichment and reduced light availability in experimental ecosystems. J. Exp. Mar. Biol. Ecol. 2000, 244, 1–28. [Google Scholar] [CrossRef]
- Zimmerman, R.C. A biooptical model of irradiance distribution and photosynthesis in seagrass canopies. Limnol. Oceanogr. 2003, 48, 568–585. [Google Scholar] [CrossRef]
- Najjar, R.G.; Pyke, C.R.; Adams, M.B.; Breitburg, D.; Hershner, C.; Kemp, M. Potential climate-change impacts on the Chesapeake Bay. Estuar. Coast. Shelf Sci. 2010, 86, 1–20. [Google Scholar] [CrossRef]
- Dekker, A.G.; Brando, V.E.; Anstee, J.M. Retrospective seagrass change detection in a shallow coastal tidal Australian lake. Remote Sens. Environ. 2005, 97, 415–433. [Google Scholar] [CrossRef]
- Ferwerda, J.G.; Leeuw, J.D.; Atzberger, C.; Vekerdy, Z. Satellite-based monitoring of tropical seagrass vegetation: Current techniques and future developments. Hydrobiologia 2007, 591, 59–71. [Google Scholar] [CrossRef]
- Ackleson, S.G.; Klemas, V. Remote sensing of submerged aquatic vegetation in lower Chesapeake Bay: A comparison of Landsat MSS to TM imagery. Remote Sens. Environ. 1987, 22, 235–248. [Google Scholar] [CrossRef]
- Hedley, J.D.; Mumby, P.J. A remote sensing method for resolving depth and subpixel composition of aquatic benthos. Limnol. Oceanogr. 2003, 48, 480–488. [Google Scholar] [CrossRef]
- Kirk, J.T.O. Light & Photosynthesis in Aquatic Ecosystems; Cambridge University Press: Cambridge, UK, 1994. [Google Scholar]
- Mobley, C.D. Light and Water: Radiative Transfer in Natural Waters; Academic Press: San Diego, CA, USA, 1994. [Google Scholar]
- Drake, L.A.; Dobbs, F.C.; Zimmerman, R.C. Effects of epiphyte load on optical properties and photosynthetic potential of the seagrasses Thalassia testudinum Banks ex Konig and Zostera marina L. Limnol. Oceanogr. 2003, 48, 456–463. [Google Scholar] [CrossRef]
- Fyfe, S.K. Spatial and temporal variation in spectral reflectance: Are seagrass species spectrally distinct? Limnol. Oceanogr. 2003, 48, 464–479. [Google Scholar] [CrossRef]
- Mazzella, L.; Alberte, R.S. Light adaptation and the role of autotrophic epiphytes in primary production of the temperate seagrass, Zostera marina L. J. Exp. Mar. Biol. Ecol. 1986, 100, 165–180. [Google Scholar] [CrossRef]
- Werdell, P.J.; Roesler, C.S. Remote assessment of benthic substrate composition in shallow waters using multispectral reflectance. Limnol. Oceanogr. 2003, 48, 557–567. [Google Scholar] [CrossRef]
- Meleder, V.; Barille, L.; Launeau, P.; Carrere, V.; Rince, Y. Spectrometric constraint in analysis of benthic diatom biomass using monospecific cultures. Remote Sens. Environ. 2003, 88, 386–400. [Google Scholar] [CrossRef]
- Murphy, R.J.; Tolhurst, T.J.; Chapman, M.G.; Underwood, A.J. Estimation of surface chlorophyll-a on an immersed mudflat using field spectrometry: Accuracy of ratios and derivative-based approaches. Int. J. Remote Sens. 2005, 26, 1835–1859. [Google Scholar] [CrossRef]
- Richards, J.; Jia, X. Feature reduction. In Remote Sensing Digital Image Analysis: An Introduction, 4th ed.; Chapter 10; Springer: Berlin/Heidelberg, Germany, 2006. [Google Scholar]
- Parks Canada. Sidney Spit (Sidney Island). Gulf Islands National Park Reserve of Canada. 2010. Available online: http://www.pc.gc.ca/pn-np/bc/gulf/carte-map-nfl.aspx#a28 (accessed on 4 January 2011).
- Robinson, C.; Martel, G. The Status of Eelgrass Meadow (Zostera marina) Health during August 2004–2006 in the New Gulf Islands National Park Reserve of Canada; WSNC Technical Report for Western and Northern Canada Service Centre-Parks Canada; Western and Northern Canada Service Centre-Parks Canada: Winnipeg, MB, Canada, 2007; p. 59. [Google Scholar]
- Leatherbarrow, K.E. Monitoring Environmental Impacts of Recreational Boat Anchoring on Eelgrass (Zostera marina L.) and Benthic Invertebrates in the Gulf Islands National Park Reserve of Canada. M.Sc. Thesis, University of Victoria, Victoria, BC, Canada, 2006. [Google Scholar]
- O’Neill, J.D. Mapping of Eelgrass (Zostera marina) at Sidney Spit, Gulf Islands National Park Reserve of Canada, Using High Spatial Resolution Remote Imagery. M.Sc. Thesis, University of Victoria, Victoria, BC, Canada, 2010. [Google Scholar]
- Robinson, C.L.K.; Yakimishyn, J. Monitoring for the Ecological Integrity of Eelgrass Beds (Zostera marina) in Canada’s Coastal National Parks of British Columbia; ParksCanada Resource Conservation Technical Report (Internal Document); Parks Canada: Winnipeg, MB, Canada, 2005; pp. 1–89. [Google Scholar]
- Komick, N.M.; Costa, M.P.F.; Gower, J. Bio-optical algorithm evaluation for MODIS for western Canada coastal waters: An exploratory approach using in situ reflectance. Remote Sens. Environ. 2009, 113, 794–804. [Google Scholar] [CrossRef]
- Rowan, K.S. Photosynthetic Pigments of Algae; Cambridge University Press: New York, NY, USA, 1989. [Google Scholar]
- Harrison, P.J.; Fulton, J.D.; Taylor, F.J.R.; Parsons, T.R. Review of the biological oceanography of the Strait of Georgia: Pelagic environment. Canad. J. Fish. Aquat. Sci. 1983, 40, 1064–1094. [Google Scholar] [CrossRef]
- Antajan, E.; Gasparini, S. Assessment of Cryptophyceae ingestion by copepods using alloxanthin pigment: A caution. Mar. Ecol. Progr. Ser. 2004, 274, 191–198. [Google Scholar] [CrossRef]
- Liedtk, J.; Roberts, A.; Luternauer, J. Practical remote sensing of suspended sediment concentration. Photogramm. Eng. Remote Sensing 1995, 61, 167–175. [Google Scholar]
- Satlantic Inc. Operation Manual for the HyperSAS; Satlantic Document #SAT-DN-00212, Revision A; Satlantic Inc.: Halifax, NS, Canada, 2003. [Google Scholar]
- Satlantic Inc. Operation Manual for the OCR-3000 (MiniSpec); Satlantic Document #SAT-DN-0069, Revision D; Satlantic Inc.: Halifax, NS, Canada, 2003. [Google Scholar]
- Mobley, C.D. Estimation of the remote-sensing reflectance from above-surface measurements. Appl. Opt. 1999, 38, 7441–7455. [Google Scholar] [CrossRef]
- Hooker, S.B.; Zibordi, G.; Brown, J.W. Above-water radiometry in shallow coastal waters. Appl. Opt. 2004, 43, 4254–4268. [Google Scholar] [CrossRef] [PubMed]
- Gordon, H.R. Normalized water-leaving radiance: Revisiting the influence of surface roughness. Appl. Opt. 2005, 44, 241–248. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.H. Effects of ocean surface reflectance variation with solar elevation on normalized water-leaving radiance. Appl. Opt. 2006, 45, 4122–4128. [Google Scholar] [CrossRef] [PubMed]
- Ruddick, K.G.; Cauwer, V.D.; Park, Y.-J.; Moore, G. Seaborne measurements of near infrared water-leaving reflectance: The similarity spectrum for turbid waters. Limnol. Oceanogr. 2006, 51, 1167–1179. [Google Scholar] [CrossRef]
- Mobley, C.D.; Sundman, L.K. HYDROLIGHT 4.1 Users’ Guide; Sequoia Scientific, Inc.: London, WA, USA, 2000; p. 85. [Google Scholar]
- Mobley, C.D.; Sundman, L.K. HYDROLIGHT 4.1 Technical Documentation; Sequoia Scientific, Inc.: London, WA, USA, 2000; p. 76. [Google Scholar]
- Cho, H.J.; Kirui, P.; Natarajan, H. Test of multi-spectral vegetation index for floating and canopy-forming submerged vegetation. Int. J. Environ. Res. Public Health 2008, 5, 477–483. [Google Scholar] [CrossRef] [PubMed]
- Roelfsema, C.; Phinn, S.; Dennison, W.; Dekker, A.; Brando, V. Monitoring toxic cyanobacteria Lyngbya majuscula (Gomont) in Moreton Bay, Australia by integrating satellite image data and field mapping. Harmful Algae 2006, 5, 45–56. [Google Scholar] [CrossRef]
- Brando, V.E.; Dekker, A.G. Satellite hyperspectral remote sensing for estimating estuarine and coastal water quality. IEEE Trans. Geosci. Remote Sens. 2003, 41, 1378–1387. [Google Scholar] [CrossRef]
- Phinn, S.; Dekker, A.; Brando, V.; Roelfsema, C. Mapping water quality and substrate cover in optically complex coastal and reef waters: An integrated approach. Mar. Pollut. Bull. 2005, 51, 459–469. [Google Scholar] [CrossRef] [PubMed]
- Phinn, S.; Roelfsema, C.; Dekker, A.; Brando, V.; Anstee, J. Remote Sensing of Environment Mapping seagrass species, cover and biomass in shallow waters: An assessment of satellite multi-spectral and airborne hyper-spectral imaging systems in Moreton Bay (Australia). Remote Sens. Environ. 2008, 112, 3413–3425. [Google Scholar] [CrossRef]
- Pasqualini, V.; Pergentmartini, C.; Pergent, G.; Agreil, M.; Skoufas, G.; Sourbes, L. Use of SPOT 5 for mapping seagrasses: An application to Posidonia oceanica. Remote Sens. Environ. 2005, 94, 39–45. [Google Scholar] [CrossRef]
- Maritorena, S.; Morel, A.; Gentili, B. Diffuse reflectance of oceanic shallow waters: Influence of water depth and bottom albedo. Limnol. Oceanogr. 1994, 39, 1689–1703. [Google Scholar] [CrossRef]
- Lee, Z.; Carder, K.L.; Arnone, R.A. Deriving inherent optical properties from water color: A multiband quasi-analytical algorithm for optically deep waters. Appl. Opt. 2002, 41, 5755–5772. [Google Scholar] [CrossRef] [PubMed]
- Brando, V.E.; Anstee, J.M.; Wettle, M.; Dekker, A.G. A physics-based retrieval and quality assessment of bathymetry from suboptimal hyperspectral data. Remote Sens. Environ. 2009, 113, 755–770. [Google Scholar] [CrossRef]
- Wettle, M.; Brando, V.E.; Dekker, A.G. A methodology for retrieval of environmental noise equivalent spectra applied to four Hyperion scenes of the same tropical coral reef. Remote Sens. Environ. 2004, 93, 188–197. [Google Scholar] [CrossRef]
- Becker, B.L.; Lusch, D.P.; Qi, J. Identifying optimal spectral bands from in situ measurements of Great Lakes coastal wetlands using second-derivative analysis. Remote Sens. Environ. 2005, 97, 238–248. [Google Scholar] [CrossRef]
- Vahtmae, E.; Kutser, T.; Martin, G.; Kotta, J. Feasibility of hyperspectral remote sensing for mapping benthic macroalgal cover in turbid coastal waters—A Baltic Sea case study. Remote Sens. Environ. 2006, 101, 342–351. [Google Scholar] [CrossRef]
- Viollier, M.; Belsher, T.; Loubersac, L. Signatures Spectrales des Objets du Littoral. In Proceedings of the 3rd International Colloquium Spectral Signatures of Objects in Remote Sensing, Les Arcs, France, 16–20 December 1985; pp. 253–256.
- Lichtenthaler, H.K.; Gitelson, A.; Lang, M. Non-destructive determination of chlorophyll content of leaves of a green and an aurea mutant of tobacco by reflectance measurements. J. Plant Physiol. 1996, 148, 483–193. [Google Scholar] [CrossRef]
- Gitelson, A.; Garbuzov, G.; Szilagyi, F.; Mittenzwey, K.H.; Karnieli, A.; Kaiser, A. Quantitative remote sensing methods for real-time monitoring of inland waters quality. Int. J. Remote Sens. 1993, 14, 1269–1295. [Google Scholar] [CrossRef]
- Jordan, C.F. Determination of leaf area index from quality of light on the forest floor. Ecology 1969, 50, 663–666. [Google Scholar] [CrossRef]
- Bandos, T.V.; Bruzzone, L.; Camps-Valls, G. Classification of hyperspectral images with regularized linear discriminant analysis. IEEE Trans. Geosci. Remote Sens. 2009, 47, 862–873. [Google Scholar] [CrossRef]
- Kaufman, Y.J.; Remer, L. Detetion of forests using mid-IR reflectance: An application for aerosol studies. IEEE Trans. Geosci. Remote Sens. 1994, 32, 672–683. [Google Scholar] [CrossRef]
- Efron, B. Nonparametric estimates of standard error: The jackknife, the bootstrap and other methods. Biometrika 1981, 68, 589–599. [Google Scholar] [CrossRef]
- Kay, S.; Hedley, J.D.; Lavender, S. Sun glint correction of high and low spatial resolution images of aquatic scenes: A review of methods for visible and near-infrared wavelengths. Remote Sens. 2009, 1, 697–730. [Google Scholar] [CrossRef]
- Teillet, P.M.; Slater, P.N.; Ding, Y.; Santer, R.P.; Jackson, R.D.; Moran, M.S. Three methods for the absolute calibration of the NOAA AVHRR sensors in-flight. Remote Sens. Environ. 1990, 31, 105–120. [Google Scholar] [CrossRef]
- Hedley, J.; Harborne, A.; Mumby, P. Simple and robust removal of sun glint for mapping shallow-water benthos. Int. J. Remote Sens. 2005, 26, 2107–2112. [Google Scholar] [CrossRef]
- Story, M.; Congalton, R.G. Accuracy assessment: A user’s perspective. Photogramm. Eng. Remote Sensing 1986, 52, 397–399. [Google Scholar]
- Morel, A.; Gentili, B.; Claustre, H.; Babin, M.; Bricaud, A.; Ras, J. Optical properties of the “clearest” natural waters. Limnol. Oceanogr. 2000, 52, 217–229. [Google Scholar] [CrossRef]
- Maritorena, S.; Morel, A.; Gentili, B. Determination of the fluorescence quantum yield by oceanic phytoplankton in their natural habitat. Appl. Opt. 2000, 39, 6725–6737. [Google Scholar] [CrossRef] [PubMed]
- Zacharias, M.; Niemann, O.; Borstad, G. An assessment and classification of a multispectral bandset for the remote sensing of intertidal seaweeds. Canad.J. Remote Sens. 1992, 18, 263–274. [Google Scholar] [CrossRef]
- Call, K.A.; Hardy, J.T.; Wallin, D.O. Coral reef habitat discrimination using multivariate spectral analysis and satellite remote sensing. Int. J. Remote Sens. 2003, 24, 2627–2639. [Google Scholar] [CrossRef]
- Dierssen, H.M.; Zimmerman, R.C.; Leathers, R.A.; Downes, T.V.; Davis, C.O.; Davis, C. Ocean color remote sensing of seagrass and bathymetry in the Bahamas Banks by high-resolution airborne imagery. Limnol. Oceanogr. 2003, 48, 444–455. [Google Scholar] [CrossRef]
- Kutser, T.; Vahtmäe, E.; Metsamaa, L. Spectral library of macroalgae and benthic substrates in Estonian coastal waters. Proc. Estonian. Acad. Sci. Biol. Ecol. 2006, 55, 329–340. [Google Scholar]
- Andréfouët, S.; Muller-Karger, F.E.; Hochberg, E.J.; Hu, C.; Carder, K.L. Change detection in shallow coral reef environments using Landsat 7 ETM+ data. Remote Sens. Environ. 2001, 78, 150–162. [Google Scholar] [CrossRef]
- Hochberg, E.; Atkinson, M. Capabilities of remote sensors to classify coral,algae and sand as pure and mixed spectra. Remote Sens. Environ. 2003, 85, 174–189. [Google Scholar] [CrossRef]
- Louchard, E.M.; Reid, R.P.; Stephens, F.C.; Davis, C.O.; Leathers, R.A.; Downes, T.V. Optical remote sensing of benthic habitats and bathymetry in coastal environments at Lee Stocking Island, Bahamas: A comparative spectral classification approach. Limnol. Oceanogr. 2003, 48, 511–521. [Google Scholar] [CrossRef]
- Thorhaug, A.; Richardson, A.D.; Berlyn, G.P. Spectral reflectance of the seagrasses: Thalassia testudinum, Halodule wrightii, Syringodium filiforme and five marine algae. Int. J. Remote Sens. 2007, 28, 1487–1501. [Google Scholar] [CrossRef]
- Snodderly, D.M.; Brown, P.K.; Delori, F.C.; Auran, J.D. The Macular Pigment. I. Absorbance spectra, localization, and discrimination from other yellow pigments in primate retinas. Microspectrophotometry 1983, 6, 660–673. [Google Scholar]
- Gausman, H.W. Visible light reflectance, transmittance, and absorptance of differently pigmented cotton leaves. Remote Sens. Environ. 1982, 13, 233–238. [Google Scholar] [CrossRef]
- Richards, J.A. Remote Sensing Digital Image Analysis; Springer-Verlag: Berlin, Germany, 1999. [Google Scholar]
- Conger, C.L.; Hochberg, E.J.; Fletcher, C.H.; Atkinson, M.J. Decorrelating remote sensing color bands from bathymetry in optically shallow waters. IEEE Trans. Geosci. Remote Sens. 2006, 44, 1655–1600. [Google Scholar] [CrossRef]
- Tassan, S. Modified Lyzenga’s method for macroalgae detection in water with non-uniform composition. Int. J. Remote Sens. 1996, 17, 1601–1607. [Google Scholar] [CrossRef]
- Purkis, S.J. A “Reef-Up” approach to classifying coral habitats from IKONOS imagery. IEEE Trans. Geosci. Remote Sens. 2005, 43, 1375–1390. [Google Scholar] [CrossRef]
- Mumby, P.J.; Edwards, A.J. Mapping marine environments with IKONOS imagery: Enhanced spatial resolution can deliver greater thematic accuracy. Remote Sens. Environ. 2002, 82, 248–257. [Google Scholar] [CrossRef]
- Richardson, A.D.; Reeves, J.B.; Gregoire, T.G. Multivariate analyses of Visible/Near Infrared (VIS/NIR) absorbance spectra reveal underlying spectral differences among dried, ground conifer needle samples from different growth environments. New Phytologist 2004, 161, 291–301. [Google Scholar] [CrossRef]
- Glover, F.; Taillard, E.; de Werra, D. A user’s guide to tabu search. Ann. Oper. Res. 1993, 41, 3–28. [Google Scholar] [CrossRef]
- Mumby, P.J.; Green, E.P.; Edwards, A.J.; Clark, C.D. The cost-effectiveness of remote sensing for tropical coastal resources assessment and management. J. Environ. Manag. 1999, 55, 157–166. [Google Scholar] [CrossRef]
- Strittholt, J.R.; Frost, P.A. Determining Abundance and Distribution of Eelgrass (Zostera marina) in Tillamook Bay Estuary, Oregon Using Multispectral Airborne Imagery; Tillamook Bay National Estuary Project: Garibaldi, OR, USA, 1996. [Google Scholar]
- Johannessen, S.; Macdonald, R. Effects of local and global change on an inland sea: The Strait of Georgia, British Columbia, Canada. Climate Res. 2009, 40, 1–21. [Google Scholar] [CrossRef]
- Roelfsema, C.M.; Phinn, S.R.; Udy, N.; Maxwell, P. An integrated field and remote sensing approach for mapping seagrass cover, Moreton Bay, Australia. J. Spatial Sci. 2009, 54, 45–62. [Google Scholar] [CrossRef]
- Pe’eri, S.; Morrison, J.R.; Short, F.; Mathieson, A.; Brook, A.; Trowbridge, P. Macroalgae and Eelgrass Mapping in Great Bay Estuary Using AISA Hyperspectral Imagery; A Final Report to The New Hampshire Estuaries Project; The New Hampshire Estuaries Project: Durham, NH, USA, 2008; p. 148. Available online: http://www.prep.unh.edu/resources/pdf/macroalgae_and_eelgrass-unh-09.pdf (accessed on 16 October 2010).
© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
O’Neill, J.D.; Costa, M.; Sharma, T. Remote Sensing of Shallow Coastal Benthic Substrates: In situ Spectra and Mapping of Eelgrass (Zostera marina) in the Gulf Islands National Park Reserve of Canada. Remote Sens. 2011, 3, 975-1005. https://doi.org/10.3390/rs3050975
O’Neill JD, Costa M, Sharma T. Remote Sensing of Shallow Coastal Benthic Substrates: In situ Spectra and Mapping of Eelgrass (Zostera marina) in the Gulf Islands National Park Reserve of Canada. Remote Sensing. 2011; 3(5):975-1005. https://doi.org/10.3390/rs3050975
Chicago/Turabian StyleO’Neill, Jennifer D., Maycira Costa, and Tara Sharma. 2011. "Remote Sensing of Shallow Coastal Benthic Substrates: In situ Spectra and Mapping of Eelgrass (Zostera marina) in the Gulf Islands National Park Reserve of Canada" Remote Sensing 3, no. 5: 975-1005. https://doi.org/10.3390/rs3050975
APA StyleO’Neill, J. D., Costa, M., & Sharma, T. (2011). Remote Sensing of Shallow Coastal Benthic Substrates: In situ Spectra and Mapping of Eelgrass (Zostera marina) in the Gulf Islands National Park Reserve of Canada. Remote Sensing, 3(5), 975-1005. https://doi.org/10.3390/rs3050975