Drought Impacts on Vegetation in Southeastern Europe
Abstract
1. Introduction
2. Materials and Methods
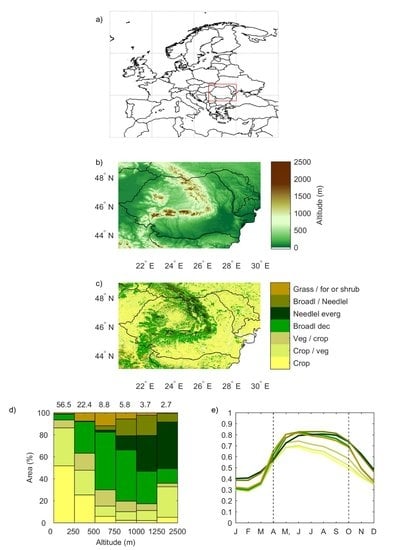
2.1. Land Cover and Elevation Data
2.2. Vegetation Index
2.3. Drought Index
2.4. Analysis
2.4.1. Correlation Analysis
2.4.2. Occurrence of Vegetation Stress and Drought
2.4.3. Drought Event of 2000/2001
3. Results
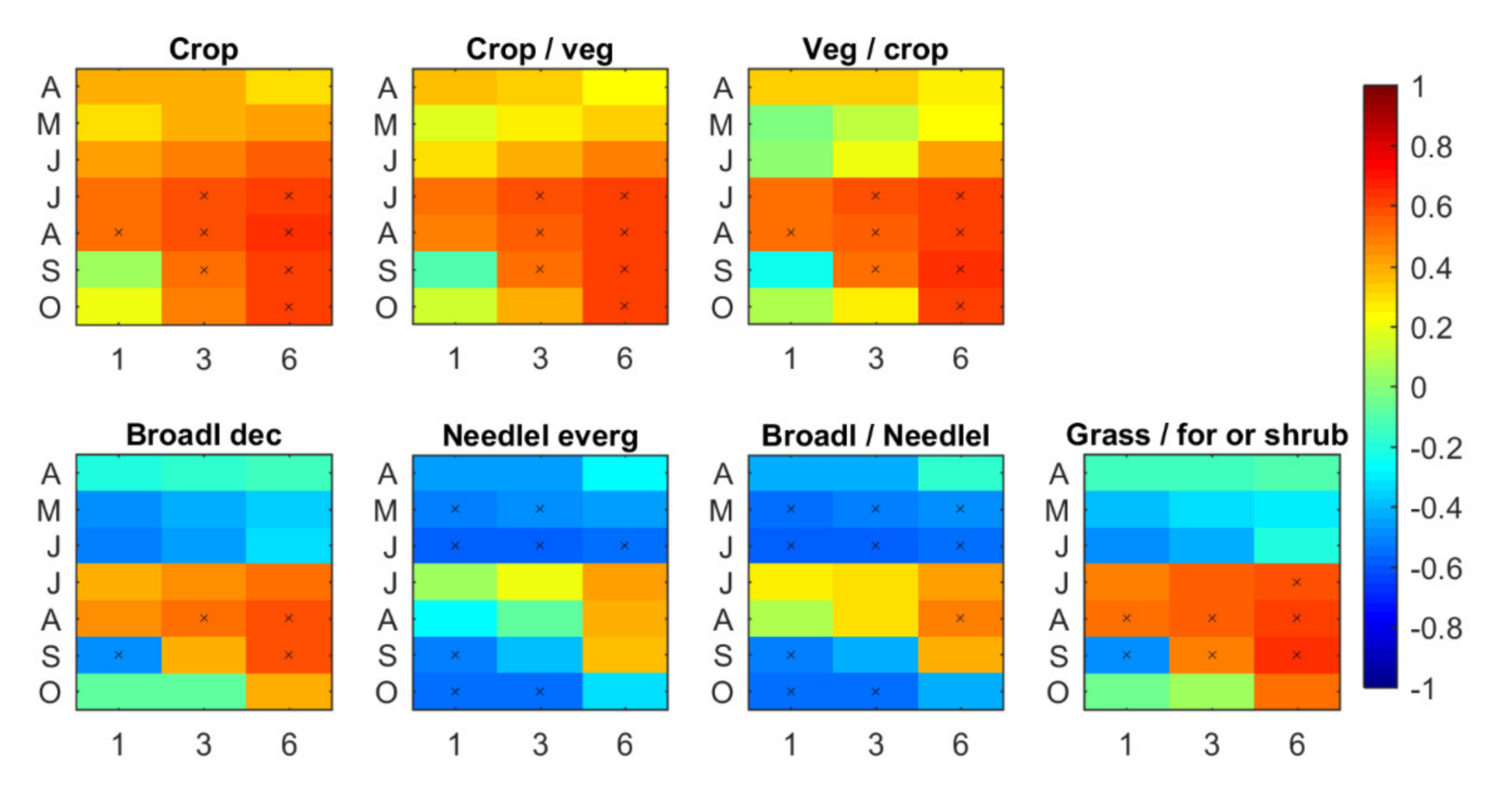
3.1. Spatial Distribution of Vegetation Response to drought Conditions
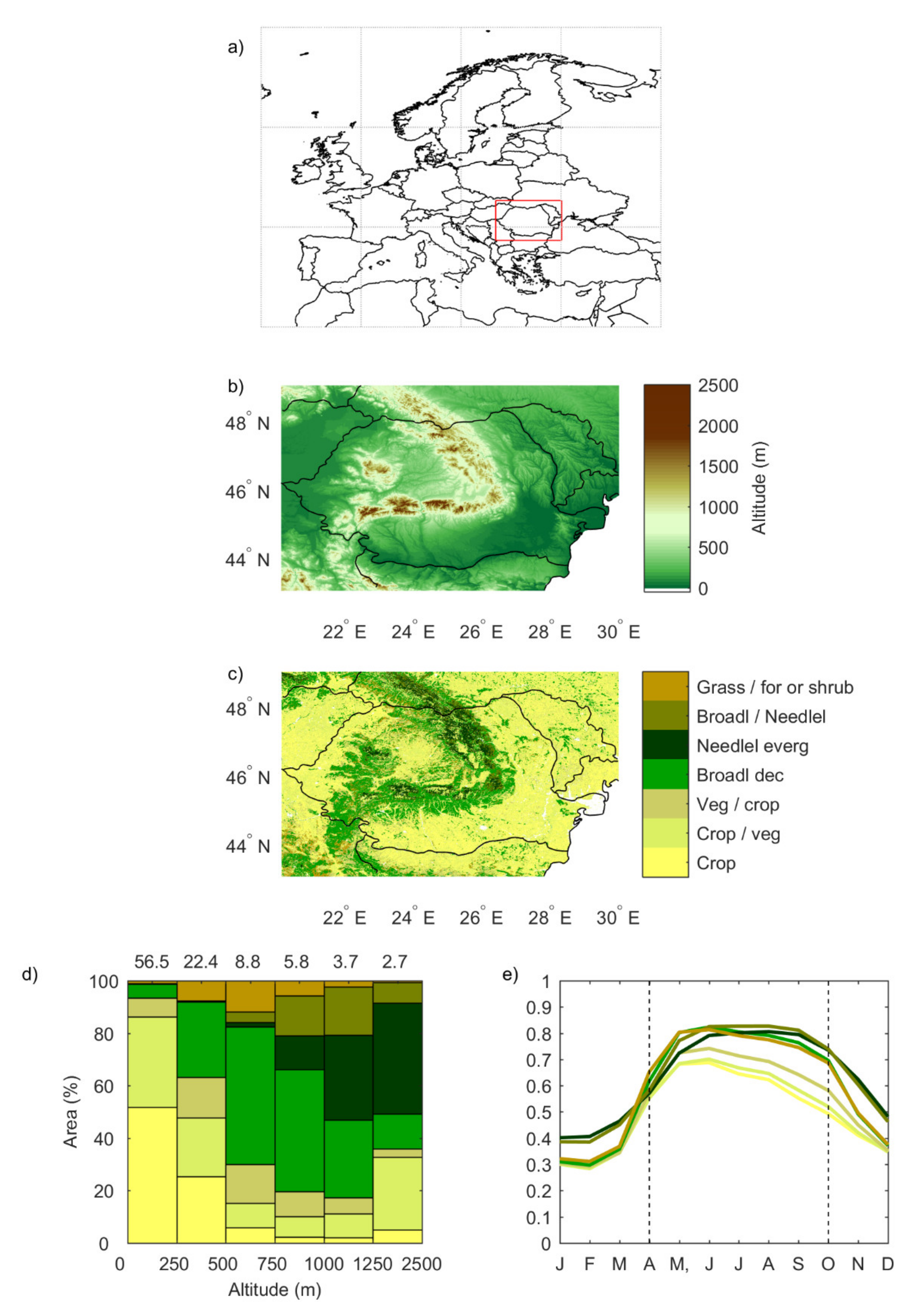
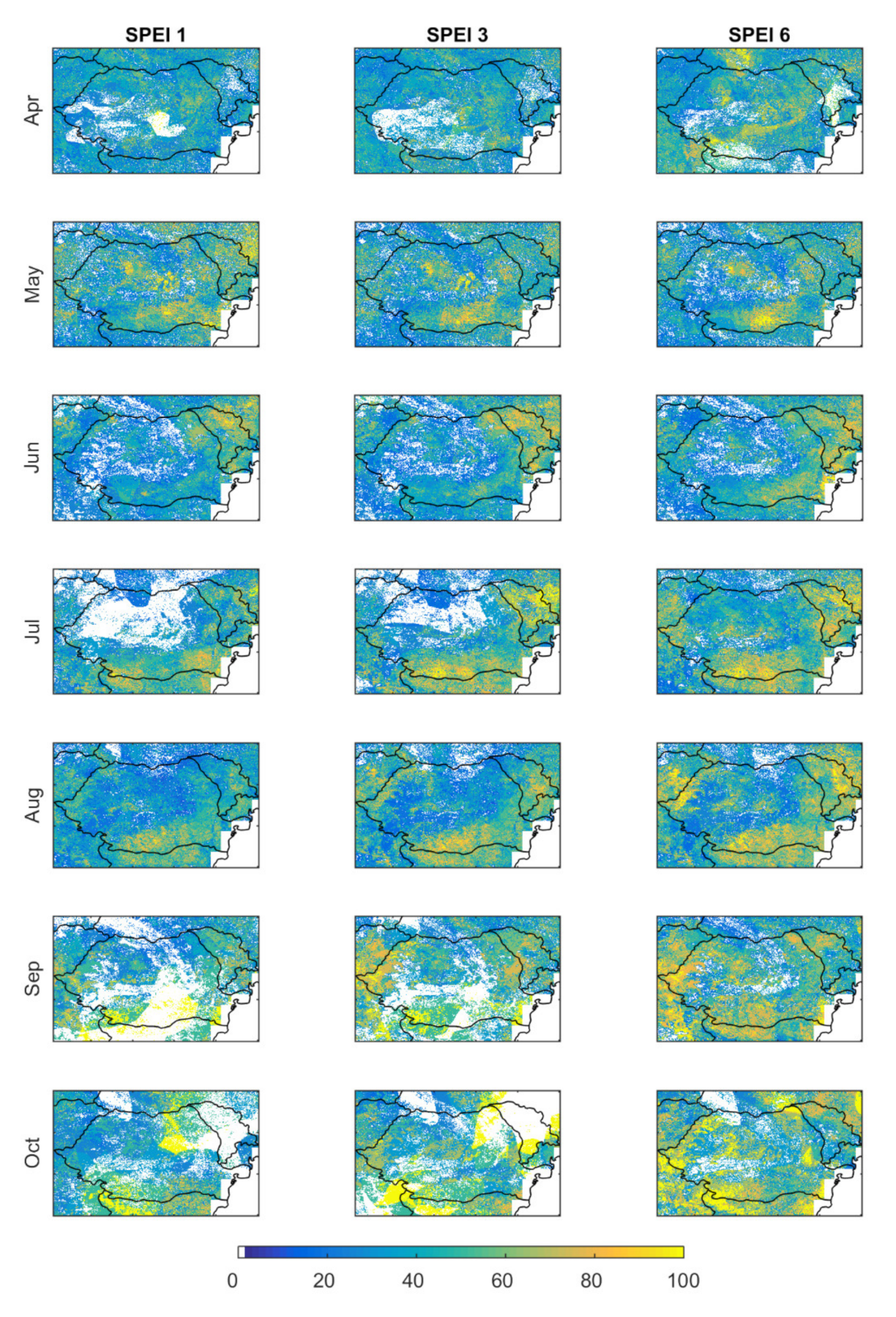
3.1.1. Correlation between SPEI and NDVI
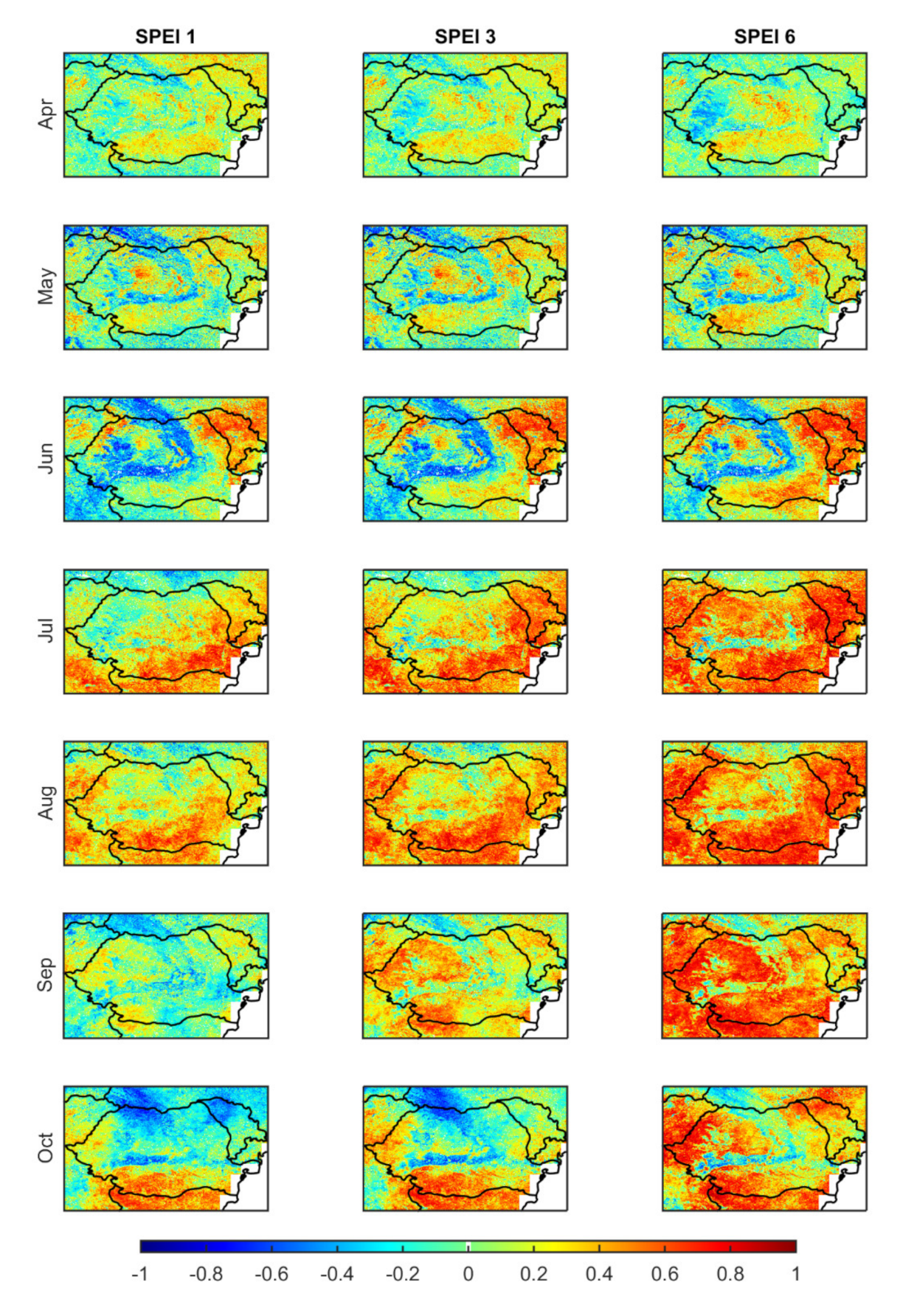
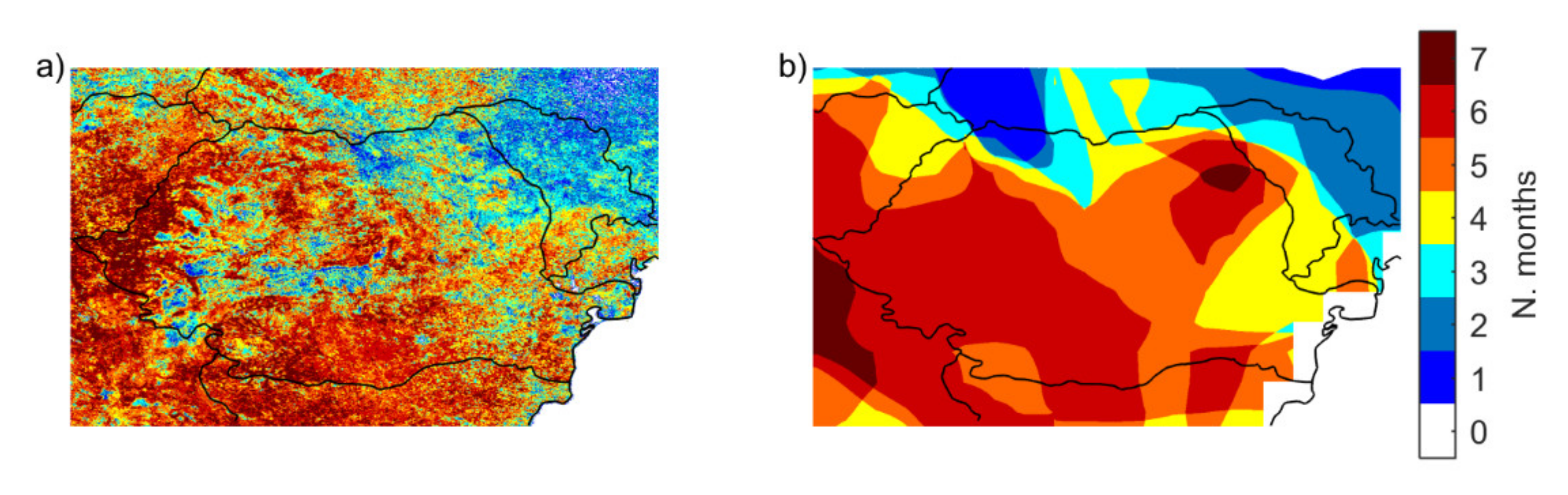
3.1.2. Occurrence of Vegetation Stress and Drought
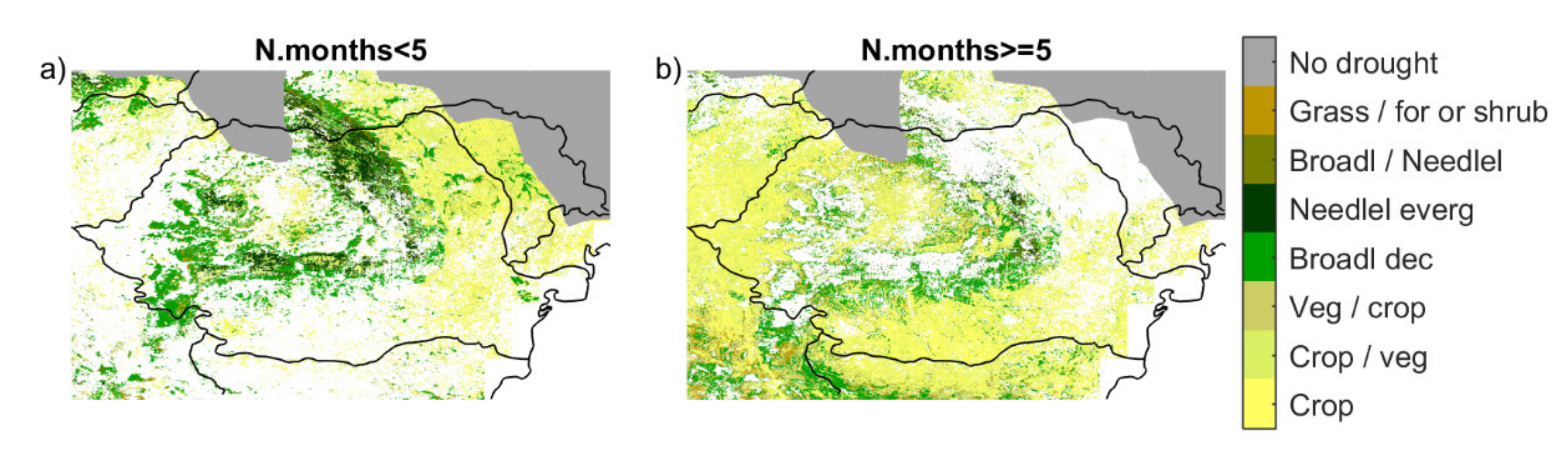
3.2. Response of Land Cover Classes to Drought Conditions
3.3. Impact of the Drought Event of 2000/2001
4. Discussion
5. Conclusions
- (i)
- during the first months of the growing season (April to June), areas with significant correlation values (both positive and negative) increased at all time scales;
- (ii)
- the middle period of the growing season (July and August) showed almost only positive correlations at all time scales;
- (iii)
- finally, positive correlations start disappearing from September onwards, starting at shorter time scales.
Author Contributions
Funding
Conflicts of Interest
References
- Kelman, I.; Gaillard, J.; Lewis, J.; Mercer, J. Learning from the history of disaster vulnerability and resilience research and practice for climate change. Nat. Hazards 2016, 82, 129–143. [Google Scholar] [CrossRef]
- Below, R.; Grover-Kopec, E.; Dilley, M. Documenting Drought-Related Disasters: A Global Reassessment. J. Environ. Dev. 2007, 16, 328–344. [Google Scholar] [CrossRef]
- Carrão, H.; Naumann, G.; Barbosa, P. Mapping global patterns of drought risk: An empirical framework based on sub-national estimates of hazard, exposure and vulnerability. Glob. Environ. Chang. 2016, 39, 108–124. [Google Scholar] [CrossRef]
- Wilhite, D.A.; Pulwarty, R.S. Drought as a hazard: Understanding the natural and the social context. In Drought and Water Crisis. Integrating Science, Management, and Policy; CRC Press: Boca Raton, FL, USA, 2017; pp. 3–20. [Google Scholar]
- Wilhite, D.A. Drought as a natural hazard: Concepts and definitions. In Drought: A Global Assessment; Routledge: New York, NY, USA, 2000; Volume 1, pp. 3–18. [Google Scholar]
- Li, Y.; Ye, W.; Wang, M.; Yan, X. Climate change and drought: A risk assessment of crop-yield impacts. Clim. Res. 2009, 39, 31–46. [Google Scholar] [CrossRef]
- Lesk, C.; Rowhani, P.; Ramankutty, N. Influence of extreme weather disasters on global crop production. Nature 2016, 529, 84–87. [Google Scholar] [CrossRef] [PubMed]
- Stagge, J.H.; Kohn, I.; Tallaksen, L.M.; Stahl, K. Modeling drought impact occurrence based on meteorological drought indices in Europe. J. Hydrol. 2015, 530, 37–50. [Google Scholar] [CrossRef]
- De Wit, M.; Stankiewicz, J. Changes in Surface Water Supply Across Africa with Predicted Climate Change. Science 2006, 311, 1917. [Google Scholar] [CrossRef]
- Rajagopalan, B.; Nowak, K.; Prairie, J.; Hoerling, M.; Harding, B.; Barsugli, J.; Ray, A.; Udall, B. Water supply risk on the Colorado River: Can management mitigate? Water Resour. Res. 2009, 45. [Google Scholar] [CrossRef]
- Allen, C.D.; Breshears, D.D. Drought-induced shift of a forest–woodland ecotone: Rapid landscape response to climate variation. Proc. Natl. Acad. Sci. USA 1998, 95, 14839. [Google Scholar] [CrossRef]
- Zeng, N.; Qian, H.; Roedenbeck, C.; Heimann, M. Impact of 1998–2002 midlatitude drought and warming on terrestrial ecosystem and the global carbon cycle. Geophys. Res. Lett. 2005, 32. [Google Scholar] [CrossRef]
- Fauset, S.; Baker, T.R.; Lewis, S.L.; Feldpausch, T.R.; Affum-Baffoe, K.; Foli, E.G.; Hamer, K.C.; Swaine, M.D. Drought-induced shifts in the floristic and functional composition of tropical forests in Ghana. Ecol. Lett. 2012, 15, 1120–1129. [Google Scholar] [CrossRef]
- Lloyd-Hughes, B. The impracticality of a universal drought definition. Theor. Appl. Climatol. 2014, 117, 607–611. [Google Scholar] [CrossRef]
- Martínez-Alonso, C.; Valladares, F.; Camarero, J.J.; Arias, M.L.; Serrano, M.; Rodríguez, J.A. The uncoupling of secondary growth, cone and litter production by intradecadal climatic variability in a mediterranean scots pine forest. For. Ecol. Manag. 2007, 253, 19–29. [Google Scholar] [CrossRef]
- Bennett, A.C.; McDowell, N.G.; Allen, C.D.; Anderson-Teixeira, K.J. Larger trees suffer most during drought in forests worldwide. Nat. Plants 2015, 1, 15139. [Google Scholar] [CrossRef] [PubMed]
- Allen, C.D.; Macalady, A.K.; Chenchouni, H.; Bachelet, D.; McDowell, N.; Vennetier, M.; Kitzberger, T.; Rigling, A.; Breshears, D.D.; Hogg, E.T.; et al. A global overview of drought and heat-induced tree mortality reveals emerging climate change risks for forests. For. Ecol. Manag. 2010, 259, 660–684. [Google Scholar] [CrossRef]
- Smit, H.J.; Metzger, M.J.; Ewert, F. Spatial distribution of grassland productivity and land use in Europe. Agric. Syst. 2008, 98, 208–219. [Google Scholar] [CrossRef]
- Chaves, M.M.; Maroco, J.P.; Pereira, J.S. Understanding plant responses to drought—From genes to the whole plant. Funct. Plant Biol. 2003, 30, 239–264. [Google Scholar] [CrossRef]
- McDowell, N.; Pockman, W.T.; Allen, C.D.; Breshears, D.D.; Cobb, N.; Kolb, T.; Plaut, J.; Sperry, J.; West, A.; Williams, D.G.; et al. Mechanisms of plant survival and mortality during drought: Why do some plants survive while others succumb to drought? New Phytol. 2008, 178, 719–739. [Google Scholar] [CrossRef] [PubMed]
- Gazol, A.; Camarero, J.J.; Vicente-Serrano, S.M.; Sánchez-Salguero, R.; Gutiérrez, E.; de Luis, M.; Sangüesa-Barreda, G.; Novak, K.; Rozas, V.; Tíscar, P.A.; et al. Forest resilience to drought varies across biomes. Glob. Chang. Biol. 2018, 24, 2143–2158. [Google Scholar] [CrossRef]
- Kannenberg, S.A.; Schwalm, C.R.; Anderegg, W.R.L. Ghosts of the past: How drought legacy effects shape forest functioning and carbon cycling. Ecol. Lett. 2020, 23, 891–901. [Google Scholar] [CrossRef]
- Vicente-Serrano, S.M.; Gouveia, C.; Camarero, J.J.; Beguería, S.; Trigo, R.; López-Moreno, J.I.; Azorín-Molina, C.; Pasho, E.; Lorenzo-Lacruz, J.; Revuelto, J.; et al. Response of vegetation to drought time-scales across global land biomes. Proc. Natl. Acad. Sci. USA 2013, 110, 52. [Google Scholar] [CrossRef] [PubMed]
- Vicente-Serrano, S.M.; Camarero, J.J.; Azorin-Molina, C. Diverse responses of forest growth to drought time-scales in the Northern Hemisphere. Glob. Ecol. Biogeogr. 2014, 23, 1019–1030. [Google Scholar] [CrossRef]
- Gouveia, C.M.; Trigo, R.M.; Beguería, S.; Vicente-Serrano, S.M. Drought impacts on vegetation activity in the Mediterranean region: An assessment using remote sensing data and multi-scale drought indicators. Glob. Planet. Chang. 2017, 151, 15–27. [Google Scholar] [CrossRef]
- Anderegg, W.R.; Berry, J.A.; Smith, D.D.; Sperry, J.S.; Anderegg, L.D.; Field, C.B. The roles of hydraulic and carbon stress in a widespread climate-induced forest die-off. Proc. Natl. Acad. Sci. USA 2012, 109, 233–237. [Google Scholar] [CrossRef] [PubMed]
- Choat, B.; Jansen, S.; Brodribb, T.J.; Cochard, H.; Delzon, S.; Bhaskar, R.; Bucci, S.J.; Feild, T.S.; Gleason, S.M.; Hacke, U.G.; et al. Global convergence in the vulnerability of forests to drought. Nature 2012, 491, 752–755. [Google Scholar] [CrossRef]
- Rigling, A.; Bräker, O.; Schneiter, G.; Schweingruber, F. Intra-annual tree-ring parameters indicating differences in drought stress of Pinus sylvestris forests within the Erico-Pinion in the Valais (Switzerland). Plant. Ecol. 2002, 163, 105–121. [Google Scholar] [CrossRef]
- Bigler, C.; Bräker, O.U.; Bugmann, H.; Dobbertin, M.; Rigling, A. Drought as an Inciting Mortality Factor in Scots Pine Stands of the Valais, Switzerland. Ecosystems 2006, 9, 330–343. [Google Scholar] [CrossRef]
- Hlavinka, P.; Trnka, M.; Semerádová, D.; Dubrovský, M.; Žalud, Z.; Možný, M. Effect of drought on yield variability of key crops in Czech Republic. Agric. For. Meteorol. 2009, 149, 431–442. [Google Scholar] [CrossRef]
- Páscoa, P.; Gouveia, C.M.; Russo, A.; Trigo, R.M. The role of drought on wheat yield interannual variability in the Iberian Peninsula from 1929 to 2012. Int. J. Biometeorol. 2017, 61, 439–451. [Google Scholar] [CrossRef]
- Bernal, M.; Estiarte, M.; Peñuelas, J. Drought advances spring growth phenology of the Mediterranean shrub Erica multiflora. Plant. Biol. 2011, 13, 252–257. [Google Scholar] [CrossRef]
- Ivits, E.; Horion, S.; Fensholt, R.; Cherlet, M. Drought footprint on European ecosystems between 1999 and 2010 assessed by remotely sensed vegetation phenology and productivity. Glob. Chang. Biol. 2014, 20, 581–593. [Google Scholar] [CrossRef] [PubMed]
- Gouveia, C.; Trigo, R.M.; DaCamara, C.C. Drought and vegetation stress monitoring in Portugal using satellite data. Nat. Hazards Earth Syst. Sci. 2009, 9, 185–195. [Google Scholar] [CrossRef]
- Gouveia, C.M.; Bastos, A.; Trigo, R.M.; DaCamara, C.C. Drought impacts on vegetation in the pre- and post-fire events over Iberian Peninsula. Nat. Hazards Earth Syst. Sci. 2012, 12, 3123–3137. [Google Scholar] [CrossRef]
- Trigo, R.M.; Gouveia, C.M.; Barriopedro, D. The intense 2007–2009 drought in the Fertile Crescent: Impacts and associated atmospheric circulation. Agric. For. Meteorol. 2010, 150, 1245–1257. [Google Scholar] [CrossRef]
- Kerr, J.T.; Ostrovsky, M. From space to species: Ecological applications for remote sensing. Trends Ecol. Evol. 2003, 18, 299–305. [Google Scholar] [CrossRef]
- Gu, Y.; Brown, J.F.; Verdin, J.P.; Wardlow, B. A five-year analysis of MODIS NDVI and NDWI for grassland drought assessment over the central Great Plains of the United States. Geophys. Res. Lett. 2007, 34. [Google Scholar] [CrossRef]
- Vicente-Serrano, S.M. Evaluating the Impact of Drought Using Remote Sensing in a Mediterranean, Semi-arid Region. Nat. Hazards 2007, 40, 173–208. [Google Scholar] [CrossRef]
- Mkhabela, M.S.; Bullock, P.; Raj, S.; Wang, S.; Yang, Y. Crop yield forecasting on the Canadian Prairies using MODIS NDVI data. Agric. For. Meteorol. 2011, 151, 385–393. [Google Scholar] [CrossRef]
- Xu, X.; Piao, S.; Wang, X.; Chen, A.; Ciais, P.; Myneni, R.B. Spatio-temporal patterns of the area experiencing negative vegetation growth anomalies in China over the last three decades. Environ. Res. Lett. 2012, 7, 035701. [Google Scholar] [CrossRef]
- Vicente-Serrano, M.S.; Cabello, D.; Tomás-Burguera, M.; Martín-Hernández, N.; Beguería, S.; Azorin-Molina, C.; Kenawy, E.A. Drought Variability and Land Degradation in Semiarid Regions: Assessment Using Remote Sensing Data and Drought Indices (1982–2011). Remote Sens. 2015, 7. [Google Scholar] [CrossRef]
- Bento, V.A.; Gouveia, C.M.; DaCamara, C.C.; Trigo, I.F. A climatological assessment of drought impact on vegetation health index. Agric. For. Meteorol. 2018, 259, 286–295. [Google Scholar] [CrossRef]
- McKee, T.B.; Doesken, N.J.; Kleist, J. The relationship of drought frequency and duration to time-scales. In Proceedings of the 8th Conference on Applied Climatology, Anaheim, CA, USA, 17–22 January 1993. [Google Scholar]
- Vicente-Serrano, S.M.; Beguería, S.; López-Moreno, J.I. A Multiscalar Drought Index Sensitive to Global Warming: The Standardized Precipitation Evapotranspiration Index. J. Clim. 2010, 23, 1696–1718. [Google Scholar] [CrossRef]
- Pasho, E.; Camarero, J.J.; de Luis, M.; Vicente-Serrano, S.M. Factors driving growth responses to drought in Mediterranean forests. Eur. J. For. Res. 2012, 131, 1797–1807. [Google Scholar] [CrossRef]
- Lévesque, M.; Saurer, M.; Siegwolf, R.; Eilmann, B.; Brang, P.; Bugmann, H.; Rigling, A. Drought response of five conifer species under contrasting water availability suggests high vulnerability of Norway spruce and European larch. Glob. Chang. Biol. 2013, 19, 3184–3199. [Google Scholar] [CrossRef] [PubMed]
- Horvat, I.; Glavac, V.; Ellenberg, H. Vegetation Südosteuropas; Gustav Fischer Verlag: Stuttgart, Germany, 1974; Volume 4. [Google Scholar]
- Doniţă, N.; Ivan, D.; Coldea, G.; Sanda, V.; Popescu, A.; Chifu, T.; Păucă-Comănescu, M.; Mititelu, D.; Boşcaiu, N. Vegetaţia României, Editura Tehnică Agricolă; Editura Tehnică Agricolă: Bucharest, Romania, 1992. [Google Scholar]
- Bohn, U.; Neuhäusl, R.; Gollub, G.; Hettwer, C.; Neuhäuslová, Z.; Raus, T.; Schluter, H.; Weber, H. Karte der natürlichen Vegetation Europas/Map of the natural vegetation of Europe. Maßstab/Scale 1: 2500000. Available online: https://is.muni.cz/el/1431/podzim2012/Bi9420/um/Bohn_etal2004_Map-Nat-Veg-Europe.pdf (accessed on 25 May 2020).
- Badea, O.; Biriş, I.A. Ancient beech forests of Romania-the preliminary identification of potential nomination areas for the World Heritage List. Available online: https://scholar.google.com/scholar?q=1.+51.+Badea%2C+O.%3B+Biri%C5%9F%2C+I.A.+Ancient+beech+forests+of+Romania-the+preliminary+identification+of+potential+nomination+areas+for+the+World+Heritage+List.+Proj.+Rep.+%28Contract+No.+8789%2F02.05.+2012+Bundesamt+f%C3%BCr+Nat.+ICAS+Bucure%C8%99ti+2012. (accessed on 25 May 2020).
- Knorn, J.; Kuemmerle, T.; Radeloff, V.C.; Keeton, W.S.; Gancz, V.; Biriş, I.-A.; Svoboda, M.; Griffiths, P.; Hagatis, A.; Hostert, P. Continued loss of temperate old-growth forests in the Romanian Carpathians despite an increasing protected area network. Environ. Conserv. 2013, 40, 182–193. [Google Scholar] [CrossRef]
- Kuemmerle, T.; Levers, C.; Erb, K.; Estel, S.; Jepsen, M.R.; Müller, D.; Plutzar, C.; Stürck, J.; Verkerk, P.J.; Verburg, P.H.; et al. Hotspots of land use change in Europe. Environ. Res. Lett. 2016, 11, 064020. [Google Scholar] [CrossRef]
- Feurdean, A.; Munteanu, C.; Kuemmerle, T.; Nielsen, A.B.; Hutchinson, S.M.; Ruprecht, E.; Parr, C.L.; Perşoiu, A.; Hickler, T. Long-term land-cover/use change in a traditional farming landscape in Romania inferred from pollen data, historical maps and satellite images. Reg. Environ. Chang. 2017, 17, 2193–2207. [Google Scholar] [CrossRef]
- Jepsen, M.R.; Kuemmerle, T.; Müller, D.; Erb, K.; Verburg, P.H.; Haberl, H.; Vesterager, J.P.; Andrič, M.; Antrop, M.; Austrheim, G.; et al. Transitions in European land-management regimes between 1800 and 2010. Land Use Policy 2015, 49, 53–64. [Google Scholar] [CrossRef]
- Hajnalová, M.; Dreslerová, D. Ethnobotany of einkorn and emmer in Romania and Slovakia: Towards interpretation of archaeological evidence. Památky Archeol. 2010, 101, 169–202. [Google Scholar]
- Sarbu, A.; Coldea, G.; Negrean, G.; Cristea, V.; Hanganu, J.; Veen, P. Grasslands of Romania—Final Report on National Grasslands Inventory 2000–2003; University of Bucharest: Bucharest, Romania, 2004. [Google Scholar]
- Kuemmerle, T.; Müller, D.; Griffiths, P.; Rusu, M. Land use change in Southern Romania after the collapse of socialism. Reg. Environ. Chang. 2009, 9, 1. [Google Scholar] [CrossRef]
- Stoate, C.; Báldi, A.; Beja, P.; Boatman, N.D.; Herzon, I.; van Doorn, A.; de Snoo, G.R.; Rakosy, L.; Ramwell, C. Ecological impacts of early 21st century agricultural change in Europe—A review. J. Environ. Manag. 2009, 91, 22–46. [Google Scholar] [CrossRef]
- United Nations Environment Programme; Division of Early Warning and Assessment. Carpathians Environment Outlook; United Nations Environment Programme: Geneva, Switzerland, 2007. [Google Scholar]
- Noss, R.F. Beyond Kyoto: Forest Management in a Time of Rapid Climate Change. Conserv. Biol. 2001, 15, 578–590. [Google Scholar] [CrossRef]
- Loos, J.; Turtureanu, P.D.; von Wehrden, H.; Hanspach, J.; Dorresteijn, I.; Frink, J.P.; Fischer, J. Plant diversity in a changing agricultural landscape mosaic in Southern Transylvania (Romania). Agric. Ecosyst. Environ. 2015, 199, 350–357. [Google Scholar] [CrossRef]
- Cremene, C.; Groza, G.; Rakosy, L.; Schileyko, A.A.; Baur, A.; Erhardt, A.; Baur, B. Alterations of Steppe-Like Grasslands in Eastern Europe: A Threat to Regional Biodiversity Hotspots. Conserv. Biol. 2005, 19, 1606–1618. [Google Scholar] [CrossRef]
- Cheval, S.; Dumitrescu, A.; Birsan, M.-V. Variability of the aridity in the South-Eastern Europe over 1961–2050. CATENA 2017, 151, 74–86. [Google Scholar] [CrossRef]
- Paltineanu, C.; Mihailescu, I.F.; Seceleanu, I.; Dragota, C.; Vasenciuc, F. Using aridity indices to describe some climate and soil features in Eastern Europe: A Romanian case study. Theor. Appl. Climatol. 2007, 90, 263–274. [Google Scholar] [CrossRef]
- Dumitrescu, A.; Bojariu, R.; Birsan, M.-V.; Marin, L.; Manea, A. Recent climatic changes in Romania from observational data (1961–2013). Theor. Appl. Climatol. 2015, 122, 111–119. [Google Scholar] [CrossRef]
- Marin, L.; Birsan, M.-V.; Bojariu, R.; Dumitrescu, A.; Micu, D.M.; Manea, A. An overview of annual climatic changes in Romania: Trends in air temperature, precipitation, sunshine hours, cloud cover, relative humidity and wind speed during the 1961–2013 period. Carpathian J. Earth Environ. Sci. 2014, 9, 253–258. [Google Scholar]
- Croitoru, A.-E.; Piticar, A.; Dragotă, C.S.; Burada, D.C. Recent changes in reference evapotranspiration in Romania. Glob. Planet. Chang. 2013, 111, 127–136. [Google Scholar] [CrossRef]
- Pravalie, R.; Sîrodoev, I.; Peptenatu, D. Detecting climate change effects on forest ecosystems in Southwestern Romania using Landsat TM NDVI data. J. Geogr. Sci. 2014, 24, 815–832. [Google Scholar] [CrossRef]
- Bojariu, R.; Birsan, M.-V.; Cica, R.; Velea, L.; Burcea, S.; Dumitrescu, A.; Dascalu, S.I.; Gothard, M.; Dobrinescu, A.; Carbunaru, F.; et al. Schimbările Climatice—de la Bazele Fizice la Riscuri şi Adaptare; Printech: Bucharest, Romania, 2015. [Google Scholar]
- Ciais, P.; Reichstein, M.; Viovy, N.; Granier, A.; Ogée, J.; Allard, V.; Aubinet, M.; Buchmann, N.; Bernhofer, C.; Carrara, A.; et al. Europe-wide reduction in primary productivity caused by the heat and drought in 2003. Nature 2005, 437, 529–533. [Google Scholar] [CrossRef] [PubMed]
- Bréda, N.; Huc, R.; Granier, A.; Dreyer, E. Temperate forest trees and stands under severe drought: A review of ecophysiological responses, adaptation processes and long-term consequences. Ann. For. Sci. 2006, 63, 625–644. [Google Scholar] [CrossRef]
- Giorgi, F.; Lionello, P. Climate change projections for the Mediterranean region. Glob. Planet. Chang. 2008, 63, 90–104. [Google Scholar] [CrossRef]
- Zhao, T.; Dai, A. Uncertainties in historical changes and future projections of drought. Part II: Model-simulated historical and future drought changes. Clim. Chang. 2017, 144, 535–548. [Google Scholar] [CrossRef]
- Samaniego, L.; Thober, S.; Kumar, R.; Wanders, N.; Rakovec, O.; Pan, M.; Zink, M.; Sheffield, J.; Wood, E.F.; Marx, A. Anthropogenic warming exacerbates European soil moisture droughts. Nat. Clim. Chang. 2018, 8, 421–426. [Google Scholar] [CrossRef]
- Bontemps, S.; Defourny, P.; van Bogaert, E.; Arino, O.; Kalogirou, V.; Perez, J.R. GlobCover 2009: Products Description and Validation Report; ESA GlobCover Project: London, UK, 2011; p. 53. [Google Scholar]
- GLOBE; Hastings, D.A.; Dunbar, P.K.; Elphingstone, G.M.; Bootz, M.; Murakami, H. The Global Land One-Kilometer Base Elevation (GLOBE) Digital Elevation Model; National Oceanic and Atmospheric Administration, National Geophysical Data Center: Boulder, CO, USA, 1999; pp. 80305–83328. [Google Scholar]
- Gamon, J.A.; Field, C.B.; Goulden, M.L.; Griffin, K.L.; Hartley, A.E.; Joel, G.; Penuelas, J.; Valentini, R. Relationships Between NDVI, Canopy Structure, and Photosynthesis in Three Californian Vegetation Types. Ecol. Appl. 1995, 5, 28–41. [Google Scholar] [CrossRef]
- Grace, J.; Nichol, C.; Disney, M.; Lewis, P.; Quaife, T.; Bowyer, P. Can we measure terrestrial photosynthesis from space directly, using spectral reflectance and fluorescence? Glob. Chang. Biol. 2007, 13, 1484–1497. [Google Scholar] [CrossRef]
- Barriopedro, D.; Gouveia, C.M.; Trigo, R.M.; Wang, L. The 2009/10 drought in China: Possible causes and impacts on vegetation. J. Hydrometeorol. 2012, 13, 1251–1267. [Google Scholar] [CrossRef]
- Toté, C.; Swinnen, E.; Sterckx, S.; Clarijs, D.; Quang, C.; Maes, R. Evaluation of the SPOT/VEGETATION Collection 3 reprocessed dataset: Surface reflectances and NDVI. Remote Sens. Environ. 2017, 201, 219–233. [Google Scholar] [CrossRef]
- Holben, B.N. Characteristics of maximum-value composite images from temporal AVHRR data. Int. J. Remote Sens. 1986, 7, 1417–1434. [Google Scholar] [CrossRef]
- Chmielewski, F.-M.; Rötzer, T. Response of tree phenology to climate change across Europe. Agric. For. Meteorol. 2001, 108, 101–112. [Google Scholar] [CrossRef]
- Mishra, A.K.; Singh, V.P. A review of drought concepts. J. Hydrol. 2010, 391, 202–216. [Google Scholar] [CrossRef]
- Vicente-Serrano, S.M.; Beguería, S.; Lorenzo-Lacruz, J.; Camarero, J.J.; López-Moreno, J.I.; Azorin-Molina, C.; Revuelto, J.; Morán-Tejeda, E.; Sanchez-Lorenzo, A. Performance of Drought Indices for Ecological, Agricultural, and Hydrological Applications. Earth Interact. 2012, 16, 1–27. [Google Scholar] [CrossRef]
- Huang, L.; He, B.; Chen, A.; Wang, H.; Liu, J.; Lű, A.; Chen, Z. Drought dominates the interannual variability in global terrestrial net primary production by controlling semi-arid ecosystems. Sci. Rep. 2016, 6, 24639. [Google Scholar] [CrossRef] [PubMed]
- Labudová, L.; Labuda, M.; Takáč, J. Comparison of SPI and SPEI applicability for drought impact assessment on crop production in the Danubian Lowland and the East Slovakian Lowland. Theor. Appl. Climatol. 2017, 128, 491–506. [Google Scholar] [CrossRef]
- Harris, I.; Jones, P.D.; Osborn, T.J.; Lister, D.H. Updated high-resolution grids of monthly climatic observations—The CRU TS3.10 Dataset. Int. J. Climatol. 2014, 34, 623–642. [Google Scholar] [CrossRef]
- Agnew, C.T. Using the SPI to Identify Drought; University College London: London, UK, 2000. [Google Scholar]
- Sousa, P.M.; Trigo, R.M.; Aizpurua, P.; Nieto, R.; Gimeno, L.; Garcia-Herrera, R. Trends and extremes of drought indices throughout the 20th century in the Mediterranean. Nat. Hazards Earth Syst. Sci. 2011, 11, 33–51. [Google Scholar] [CrossRef]
- Dai, A. Increasing drought under global warming in observations and models. Nat. Clim. Chang. 2013, 3, 52–58. [Google Scholar] [CrossRef]
- Calderini, D.F.; Slafer, G.A. Changes in yield and yield stability in wheat during the 20th century. Field Crop. Res. 1998, 57, 335–347. [Google Scholar] [CrossRef]
- Hafner, S. Trends in maize, rice, and wheat yields for 188 nations over the past 40 years: A prevalence of linear growth. Agric. Ecosyst. Environ. 2003, 97, 275–283. [Google Scholar] [CrossRef]
- Piao, S.; Friedlingstein, P.; Ciais, P.; Zhou, L.; Chen, A. Effect of climate and CO2 changes on the greening of the Northern Hemisphere over the past two decades. Geophys. Res. Lett. 2006, 33. [Google Scholar] [CrossRef]
- Donohue, R.J.; Roderick, M.L.; McVicar, T.R.; Farquhar, G.D. Impact of CO2 fertilization on maximum foliage cover across the globe’s warm, arid environments. Geophys. Res. Lett. 2013, 40, 3031–3035. [Google Scholar] [CrossRef]
- Berndt, C.; Haberlandt, U. Spatial interpolation of climate variables in Northern Germany—Influence of temporal resolution and network density. J. Hydrol. Reg. Stud. 2018, 15, 184–202. [Google Scholar] [CrossRef]
- Spinoni, J.; Antofie, T.; Barbosa, P.; Bihari, Z.; Lakatos, M.; Szalai, S.; Szentimrey, T.; Vogt, J. An overview of drought events in the Carpathian Region in 1961–2010. Adv. Sci. Res. 2013, 10, 21–32. [Google Scholar] [CrossRef]
- Ionita, M.; Scholz, P.; Chelcea, S. Assessment of droughts in Romania using the Standardized Precipitation Index. Nat. Hazards 2016, 81, 1483–1498. [Google Scholar] [CrossRef]
- Sepulcre-Canto, G.; Horion, S.; Singleton, A.; Carrao, H.; Vogt, J. Development of a Combined Drought Indicator to detect agricultural drought in Europe. Nat. Hazards Earth Syst. Sci. 2012, 12, 3519–3531. [Google Scholar] [CrossRef]
- Spinoni, J.; Naumann, G.; Vogt, J.V.; Barbosa, P. The biggest drought events in Europe from 1950 to 2012. J. Hydrol. Reg. Stud. 2015, 3, 509–524. [Google Scholar] [CrossRef]
- Colesca, S.E.; Ciocoiu, C.N. An overview of the Romanian renewable energy sector. Renew. Sustain. Energy Rev. 2013, 24, 149–158. [Google Scholar] [CrossRef]
- Potopová, V.; Boroneanţ, C.; Boincean, B.; Soukup, J. Impact of agricultural drought on main crop yields in the Republic of Moldova. Int. J. Climatol. 2016, 36, 2063–2082. [Google Scholar] [CrossRef]
- Levanič, T.; Popa, I.; Poljanšek, S.; Nechita, C. A 323-year long reconstruction of drought for SW Romania based on black pine (Pinus Nigra) tree-ring widths. Int. J. Biometeorol. 2013, 57, 703–714. [Google Scholar] [CrossRef]
- Koleva, E.; Alexandrov, V. Drought in the Bulgarian low regions during the 20th century. Theor. Appl. Climatol. 2008, 92, 113–120. [Google Scholar] [CrossRef]
- Cheval, S.; Baciu, M.; Dumitrescu, A.; Breza, T.; Legates, D.R.; Chendeş, V. Climatologic adjustments to monthly precipitation in Romania. Int. J. Climatol. 2011, 31, 704–714. [Google Scholar] [CrossRef]
- Spinoni, J.; Szalai, S.; Szentimrey, T.; Lakatos, M.; Bihari, Z.; Nagy, A.; Németh, Á.; Kovács, T.; Mihic, D.; Dacic, M.; et al. Climate of the Carpathian Region in the period 1961–2010: Climatologies and trends of 10 variables. Int. J. Climatol. 2015, 35, 1322–1341. [Google Scholar] [CrossRef]
- Dascălu, S.I.; Gothard, M.; Bojariu, R.; Birsan, M.-V.; Cică, R.; Vintilă, R.; Adler, M.-J.; Chendeș, V.; Mic, R.-P. Drought-related variables over the Bârlad basin (Eastern Romania) under climate change scenarios. CATENA 2016, 141, 92–99. [Google Scholar] [CrossRef]
- Nemani, R.R.; Keeling, C.D.; Hashimoto, H.; Jolly, W.M.; Piper, S.C.; Tucker, C.J.; Myneni, R.B.; Running, S.W. Climate-Driven Increases in Global Terrestrial Net Primary Production from 1982 to 1999. Science 2003, 300, 1560. [Google Scholar] [CrossRef]
- Karnieli, A.; Agam, N.; Pinker, R.T.; Anderson, M.; Imhoff, M.L.; Gutman, G.G.; Panov, N.; Goldberg, A. Use of NDVI and Land Surface Temperature for Drought Assessment: Merits and Limitations. J. Clim. 2010, 23, 618–633. [Google Scholar] [CrossRef]
- Sidor, C.G.; Popa, I.; Vlad, R.; Cherubini, P. Different tree-ring responses of Norway spruce to air temperature across an altitudinal gradient in the Eastern Carpathians (Romania). Trees 2015, 29, 985–997. [Google Scholar] [CrossRef]
- Ribeiro, A.F.S.; Russo, A.; Gouveia, C.M.; Páscoa, P. Copula-based agricultural drought risk of rainfed cropping systems. Agric. Water Manag. 2019, 223, 105689. [Google Scholar] [CrossRef]
- Schwalm, C.R.; Williams, C.A.; Schaefer, K.; Arneth, A.; Bonal, D.; Buchmann, N.; Chen, J.; Law, B.E.; Lindroth, A.; Luyssaert, S.; et al. Assimilation exceeds respiration sensitivity to drought: A FLUXNET synthesis. Glob. Chang. Biol. 2010, 16, 657–670. [Google Scholar] [CrossRef]
- Baumbach, L.; Siegmund, J.F.; Mittermeier, M.; Donner, R.V. Impacts of temperature extremes on European vegetation during the growing season. Biogeosciences 2017, 14, 4891–4903. [Google Scholar] [CrossRef]
- Babst, F.; Poulter, B.; Trouet, V.; Tan, K.; Neuwirth, B.; Wilson, R.; Carrer, M.; Grabner, M.; Tegel, W.; Levanic, T. Site-and species-specific responses of forest growth to climate across the E uropean continent. Glob. Ecol. Biogeogr. 2013, 22, 706–717. [Google Scholar] [CrossRef]

| Globcover Class Label | Label | Area (%) |
|---|---|---|
| Rainfed croplands | Crop | 34.8 |
| Mosaic cropland (50–70%) / natural vegetation (grassland/shrubland/forest) (20–50%) | Crop/Veg | 26.2 |
| Mosaic natural vegetation (grassland/shrubland/forest) (50–70%) / cropland (20–50%) | Veg/Crop | 9.6 |
| Closed (>40%) broadleaved deciduous forest (>5 m) | Broadl dec | 18.2 |
| Closed (>40%) needleleaved evergreen forest (>5 m) | Neddlel everg | 3.3 |
| Closed to open (>15%) mixed broadleaved and needleleaved forest (>5 m) | Broadl/Neddlel | 2.2 |
| Mosaic grassland (50–70%) / forest or shrubland (20–50%) | Grass | 3.8 |
| Drought Classification | SPEI Range | Return Period |
|---|---|---|
| Moderate | −0.84 < SPEI | 1 in 5 years |
| Severe | −1.28 < SPEI | 1 in 10 years |
| Extreme | −1.65 < SPEI | 1 in 20 years |
| >50% | >80% | |||||
|---|---|---|---|---|---|---|
| SPEI 1 | SPEI 3 | SPEI 6 | SPEI 1 | SPEI 3 | SPEI 6 | |
| Apr | 22.4 | 24.7 | 38.0 | 0.6 | 0.6 | 2.6 |
| May | 42.0 | 35.4 | 33.2 | 6.0 | 5.4 | 4.6 |
| Jun | 23.8 | 28.5 | 34.4 | 2.0 | 3.5 | 3.6 |
| Jul | 28.7 | 36.8 | 51.4 | 2.9 | 6.1 | 7.6 |
| Aug | 29.3 | 39.8 | 50.8 | 1.4 | 3.9 | 9.2 |
| Sep | 31.2 | 45.8 | 56.6 | 4.7 | 7.0 | 13.2 |
| Oct | 36.1 | 42.7 | 55.2 | 6.4 | 11.7 | 13.5 |
| Time Scale | Crop | Crop/Veg | Veg/Crop | Broadl Dec | Needlel Everg | Broadl/ Needlel | Grass/For or Shrub | |
|---|---|---|---|---|---|---|---|---|
| Apr | SPEI 1 | 6.2 | 6.3 | 3.5 | 1.0 | 0.5 | 0.6 | 0.9 |
| SPEI 3 | 5.4 | 5.6 | 3.7 | 1.3 | 0.5 | 0.8 | 1.1 | |
| SPEI 6 | 3.2 | 3.6 | 4.9 | 2.9 | 1.7 | 2.5 | 2.4 | |
| May | SPEI 1 | 8.8 | 9.1 | 6.5 | 1.7 | 0.9 | 0.4 | 3.1 |
| SPEI 3 | 13.2 | 13.6 | 9.5 | 2.3 | 0.8 | 0.4 | 4.0 | |
| SPEI 6 | 17.0 | 17.2 | 12.0 | 3.5 | 1.0 | 0.5 | 4.8 | |
| Jun | SPEI 1 | 18.1 | 16.9 | 9.7 | 1.5 | 0.1 | 0.0 | 1.0 |
| SPEI 3 | 25.5 | 23.5 | 13.9 | 2.4 | 0.1 | 0.0 | 2.1 | |
| SPEI 6 | 37.8 | 34.4 | 21.1 | 4.3 | 0.1 | 0.1 | 5.2 | |
| Jul | SPEI 1 | 30.9 | 27.5 | 27.0 | 14.5 | 3.6 | 5.4 | 26.7 |
| SPEI 3 | 49.5 | 44.9 | 42.6 | 19.2 | 5.4 | 5.5 | 34.3 | |
| SPEI 6 | 72.7 | 68.6 | 68.2 | 32.1 | 17.3 | 12.9 | 50.9 | |
| Aug | SPEI 1 | 27.9 | 24.4 | 24.2 | 14.6 | 1.3 | 3.3 | 23.4 |
| SPEI 3 | 50.2 | 45.3 | 45.7 | 26.6 | 2.2 | 5.0 | 40.4 | |
| SPEI 6 | 76.4 | 70.0 | 71.2 | 42.8 | 6.0 | 9.2 | 59.5 | |
| Sep | SPEI 1 | 1.5 | 1.4 | 0.8 | 0.1 | 0.0 | 0.0 | 0.1 |
| SPEI 3 | 26.1 | 24.2 | 29.8 | 9.9 | 0.5 | 0.6 | 18.9 | |
| SPEI 6 | 69.0 | 65.9 | 72.3 | 41.5 | 3.7 | 5.4 | 65.3 | |
| Oct | SPEI 1 | 14.9 | 13.4 | 16.4 | 9.4 | 0.1 | 0.2 | 12.9 |
| SPEI 3 | 23.8 | 20.4 | 21.9 | 10.7 | 0.1 | 0.2 | 15.9 | |
| SPEI 6 | 53.7 | 51.0 | 47.5 | 18.9 | 1.0 | 0.7 | 29.9 |
| Time-Scale | Crop | Crop/Veg | Veg/Crop | Broadl Dec | Needlel Everg | Broadl/ Needlel | Grass/For or Shrub | |
|---|---|---|---|---|---|---|---|---|
| Apr | SPEI 1 | 0.6 | 0.9 | 0.7 | 2.4 | 11.8 | 9.6 | 1.7 |
| SPEI 3 | 0.4 | 1.0 | 0.7 | 3.0 | 12.4 | 10.0 | 2.3 | |
| SPEI 6 | 0.8 | 1.3 | 1.5 | 5.2 | 6.1 | 4.8 | 4.2 | |
| May | SPEI 1 | 2.3 | 4.4 | 6.5 | 20.9 | 30.4 | 41.0 | 15.6 |
| SPEI 3 | 1.9 | 3.9 | 5.8 | 20.2 | 26.6 | 35.6 | 15.6 | |
| SPEI 6 | 1.7 | 3.4 | 4.5 | 18.2 | 19.2 | 25.9 | 14.9 | |
| Jun | SPEI 1 | 2.3 | 5.4 | 9.6 | 34.5 | 71.0 | 69.7 | 25.8 |
| SPEI 3 | 1.6 | 4.6 | 7.1 | 29.3 | 69.4 | 66.6 | 20.3 | |
| SPEI 6 | 1.1 | 3.6 | 3.7 | 18.9 | 55.4 | 50.9 | 11.2 | |
| Jul | SPEI 1 | 1.1 | 1.3 | 0.9 | 2.2 | 3.0 | 1.6 | 1.7 |
| SPEI 3 | 0.3 | 0.4 | 0.3 | 1.5 | 2.4 | 1.4 | 1.3 | |
| SPEI 6 | 0.0 | 0.1 | 0.1 | 1.1 | 1.9 | 1.4 | 1.0 | |
| Aug | SPEI 1 | 0.6 | 0.9 | 0.5 | 1.0 | 4.7 | 2.5 | 0.5 |
| SPEI 3 | 0.3 | 0.6 | 0.3 | 0.6 | 3.0 | 1.3 | 0.3 | |
| SPEI 6 | 0.1 | 0.1 | 0.1 | 0.1 | 0.9 | 0.3 | 0.1 | |
| Sep | SPEI 1 | 1.2 | 2.1 | 2.3 | 10.4 | 15.4 | 24.0 | 5.0 |
| SPEI 3 | 0.1 | 0.4 | 0.3 | 1.4 | 4.9 | 6.5 | 0.3 | |
| SPEI 6 | 0.0 | 0.1 | 0.0 | 0.2 | 0.7 | 0.8 | 0.0 | |
| Oct | SPEI 1 | 7.0 | 8.3 | 11.9 | 12.5 | 31.4 | 27.4 | 14.2 |
| SPEI 3 | 1.6 | 4.0 | 8.1 | 14.7 | 36.2 | 37.6 | 13.9 | |
| SPEI 6 | 0.1 | 0.4 | 0.7 | 3.5 | 4.6 | 8.8 | 1.8 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Páscoa, P.; Gouveia, C.M.; Russo, A.C.; Bojariu, R.; Vicente-Serrano, S.M.; Trigo, R.M. Drought Impacts on Vegetation in Southeastern Europe. Remote Sens. 2020, 12, 2156. https://doi.org/10.3390/rs12132156
Páscoa P, Gouveia CM, Russo AC, Bojariu R, Vicente-Serrano SM, Trigo RM. Drought Impacts on Vegetation in Southeastern Europe. Remote Sensing. 2020; 12(13):2156. https://doi.org/10.3390/rs12132156
Chicago/Turabian StylePáscoa, Patrícia, Célia M. Gouveia, Ana C. Russo, Roxana Bojariu, Sergio M. Vicente-Serrano, and Ricardo M. Trigo. 2020. "Drought Impacts on Vegetation in Southeastern Europe" Remote Sensing 12, no. 13: 2156. https://doi.org/10.3390/rs12132156
APA StylePáscoa, P., Gouveia, C. M., Russo, A. C., Bojariu, R., Vicente-Serrano, S. M., & Trigo, R. M. (2020). Drought Impacts on Vegetation in Southeastern Europe. Remote Sensing, 12(13), 2156. https://doi.org/10.3390/rs12132156