Automatic Wheat Ear Counting Using Thermal Imagery
Abstract
1. Introduction
2. Materials and Methods
2.1. Plant Material and Experimental Setup
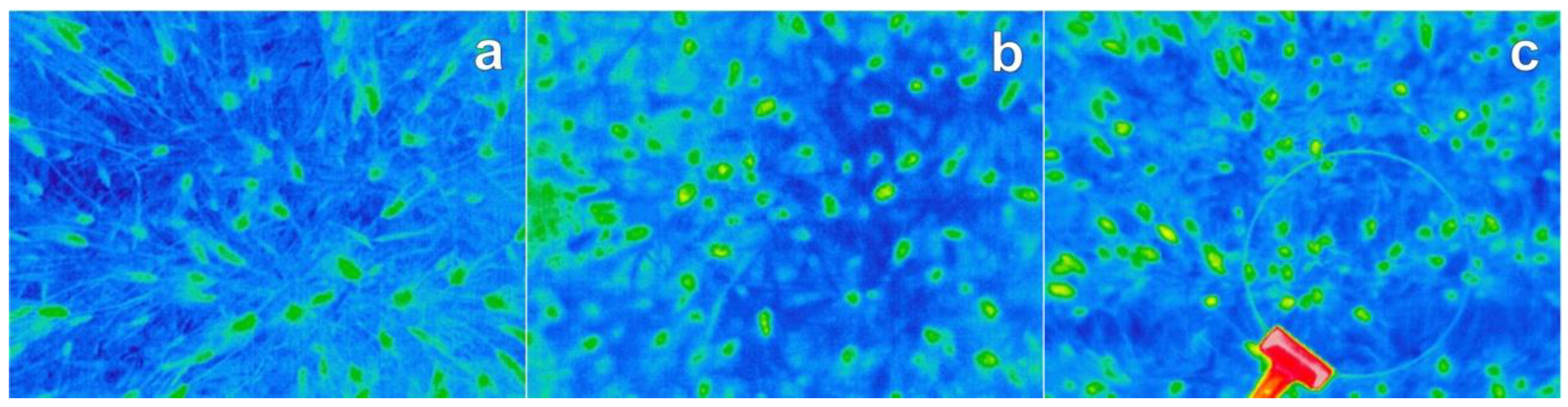
2.2. Thermal Images
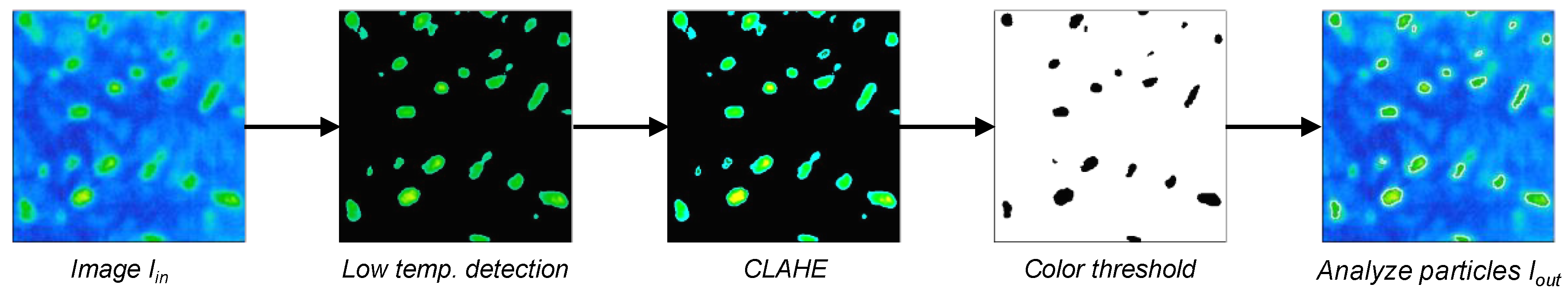
2.3. Automatic Thermal Ear Counting System
2.4. Algorithm Validation
Manual In Situ Counting and RGB Images
2.5. Statistical Analysis
3. Results
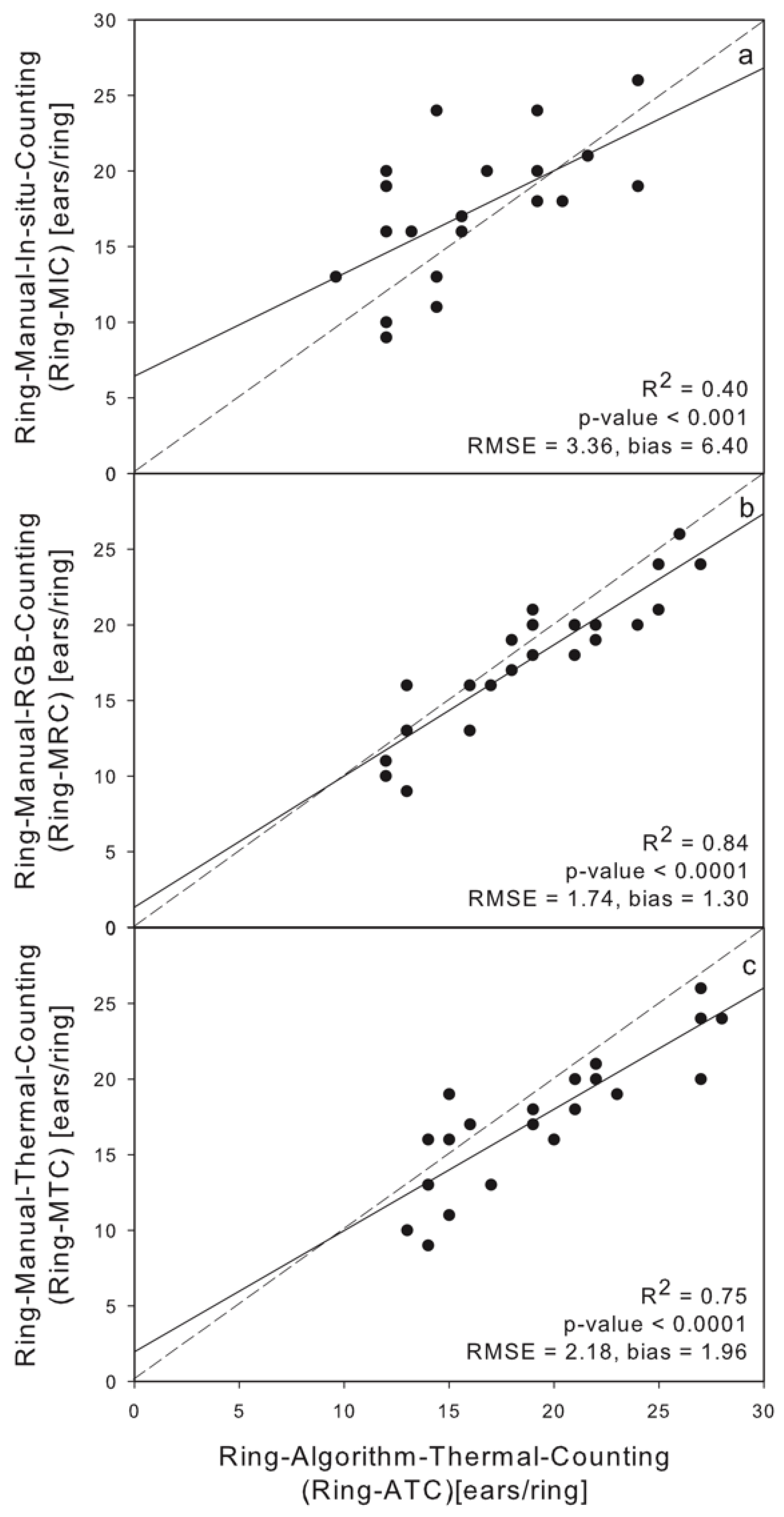
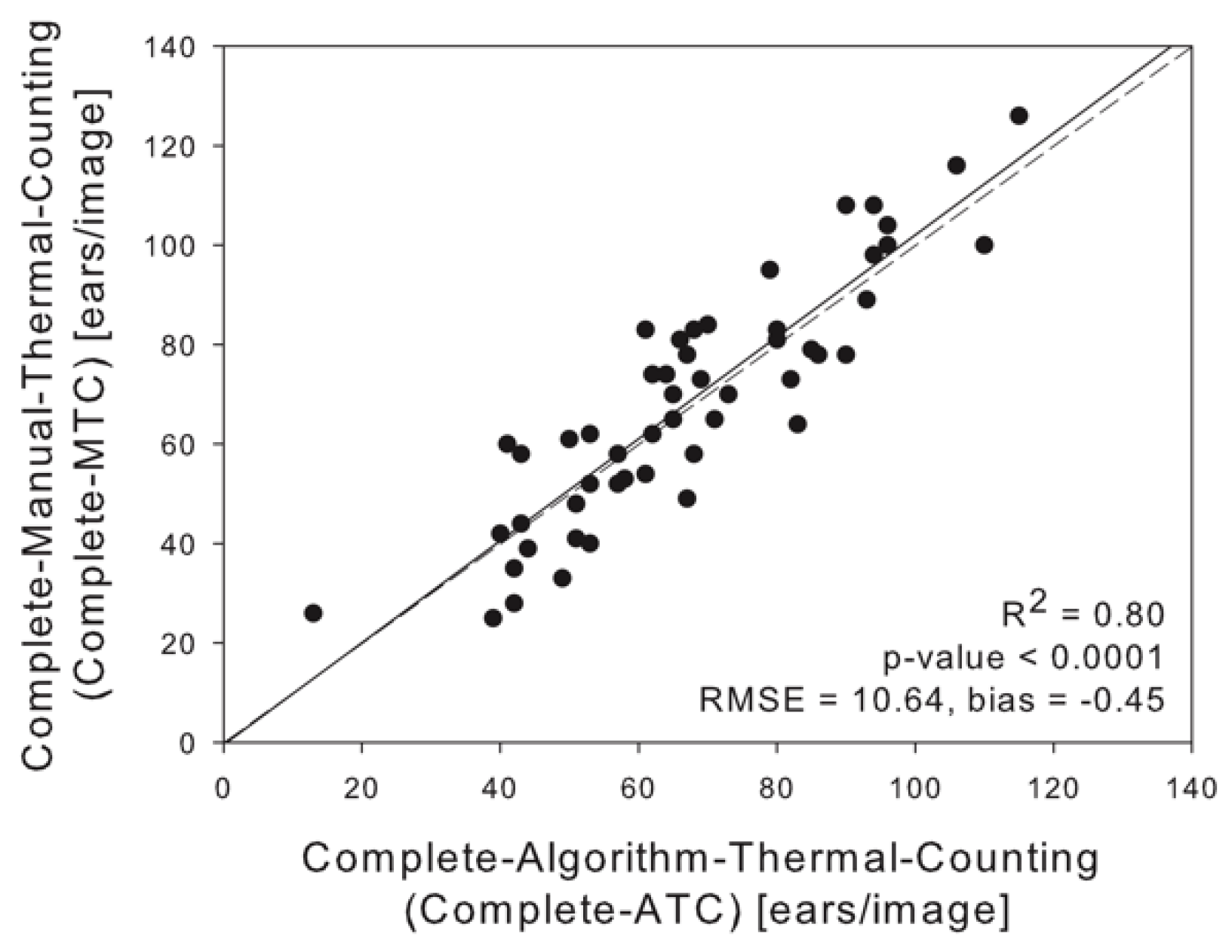
3.1. Linear Regression between Thermal, RGB, In Situ and Algorithm Counting
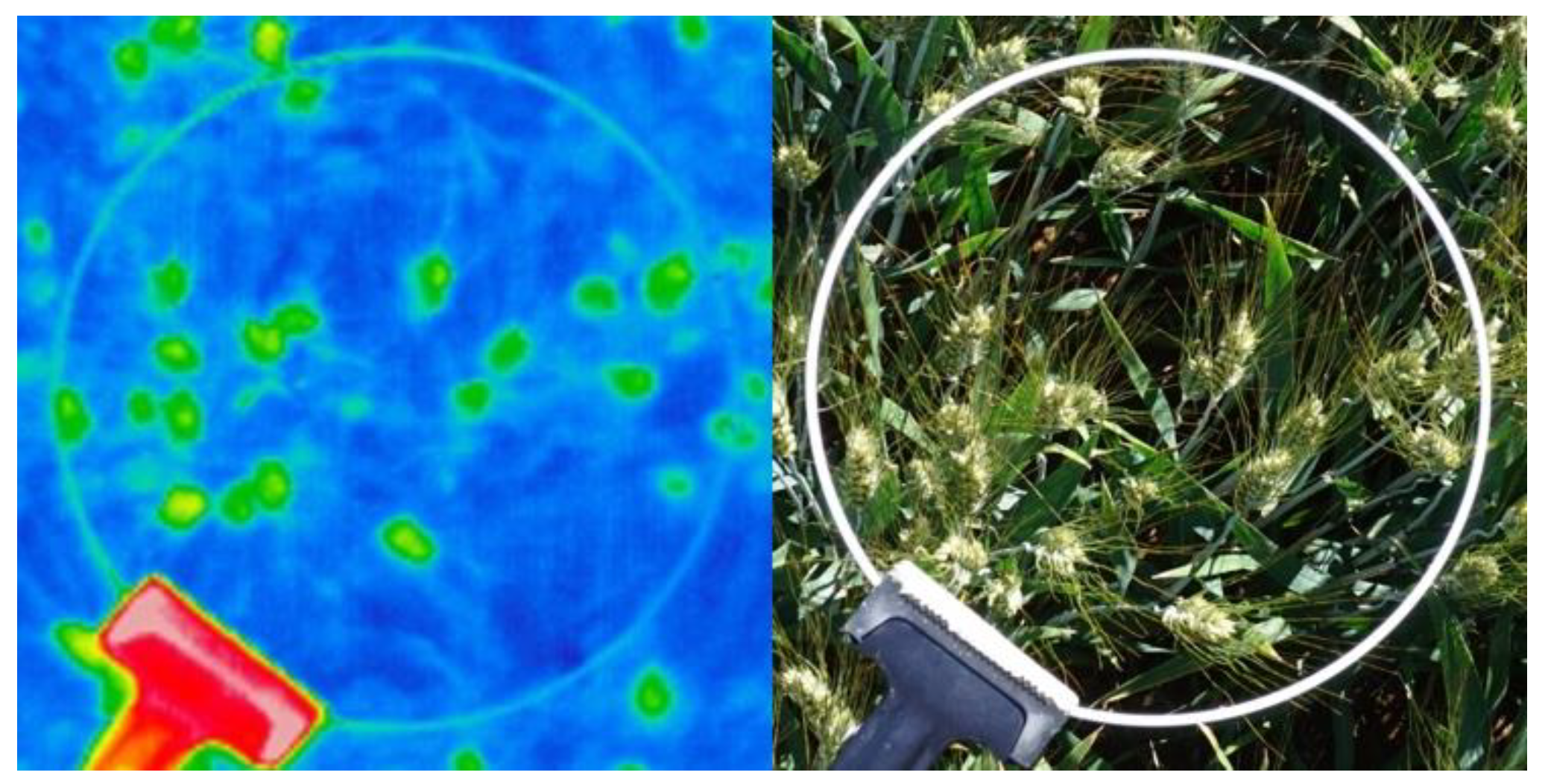
3.2. Understanding Acquisition and Algorithm Errors
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Walter, A.; Liebisch, F.; Hund, A. Plant phenotyping: From bean weighing to image analysis. Plant Methods 2015, 11, 14. [Google Scholar] [CrossRef] [PubMed]
- Araus, J.L.; Kefauver, S.C. Breeding to adapt agriculture to climate change: Affordable phenotyping solutions. Curr. Opin. Plant Biol. 2018, 45 Pt B, 237–247. [Google Scholar] [CrossRef]
- Araus, J.L.; Cairns, J.E. Field high-throughput phenotyping: The new crop breeding frontier. Trends Plant Sci. 2014, 19, 52–61. [Google Scholar] [CrossRef] [PubMed]
- Pask, A.; Pietragalla, J.; Mullan, D.; Reynolds, M. (Eds.) Physiological Breeding II: A Field Guide to Wheat Phenotyping; CIMMYT: Mexico D.F., Mexico, 2012; ISBN 9789706481825. [Google Scholar]
- Slafer, G.A.; Calderini, D.F.; Miralles, D.J. Yield Components and Compensation in Wheat: Opportunities for Further Increasing Yield Potencial. In Increasing Yield Potential in Wheat: Breaking the Barriers; CIMMYT International Symposium: Mexico D.F., Mexico, 1996; pp. 101–133. [Google Scholar]
- Cointault, F.; Guerin, D.; Guillemin, J.; Chopinet, B. In-field Triticum aestivum ear counting using colour-texture image analysis. N. Z. J. Crop Hortic. Sci. 2008, 36, 117–130. [Google Scholar] [CrossRef]
- Liu, T.; Sun, C.; Wang, L.; Zhong, X.; Zhu, X.; Guo, W. In-field wheatear counting based on image processing technology. Nongye Jixie Xuebao/Trans. Chin. Soc. Agric. Mach. 2014, 45, 282–290. [Google Scholar] [CrossRef]
- Zhou, C.; Liang, D.; Yang, X.; Yang, H.; Yue, J.; Yang, G. Wheat Ears Counting in Field Conditions Based on Multi-Feature Optimization and TWSVM. Front. Plant Sci. 2018, 9, 1024. [Google Scholar] [CrossRef]
- Zhu, Y.; Cao, Z.; Lu, H.; Li, Y.; Xiao, Y. In-field automatic observation of wheat heading stage using computer vision. Biosyst. Eng. 2016, 143, 28–41. [Google Scholar] [CrossRef]
- Sadeghi-Tehran, P.; Sabermanesh, K.; Virlet, N.; Hawkesford, M.J. Automated Method to Determine Two Critical Growth Stages of Wheat: Heading and Flowering. Front. Plant Sci. 2017, 8, 252. [Google Scholar] [CrossRef]
- Madec, S.; Jin, X.; Lu, H.; De Solan, B.; Liu, S.; Duyme, F.; Heritier, E.; Baret, F. Ear density estimation from high resolution RGB imagery using deep learning technique. Agric. For. Meteorol. 2019, 264, 225–234. [Google Scholar] [CrossRef]
- Fernandez-Gallego, J.A.; Kefauver, S.C.; Aparicio Gutiérrez, N.; Nieto-Taladriz, M.T.; Araus, J.L. Wheat ear counting in-field conditions: High throughput and low-cost approach using RGB images. Plant Methods 2018, 14, 22. [Google Scholar] [CrossRef] [PubMed]
- Fernandez-Gallego, J.A.; Kefauver, S.C.; Gutiérrez, N.A.; Nieto-Taladriz, M.T.; Araus, J.L. Automatic wheat ear counting in-field conditions: Simulation and implication of lower resolution images. In Proceedings of the Remote Sensing for Agriculture, Ecosystems, and Hydrology XX, Berlin, Germany, 10–13 September 2018; p. 23. [Google Scholar] [CrossRef]
- Zhou, C.; Liang, D.; Yang, X.; Xu, B.; Yang, G. Recognition of wheat spike from field based phenotype platform using multi-sensor fusion and improved maximum entropy segmentation algorithms. Remote Sens. 2018, 10, 246. [Google Scholar] [CrossRef]
- Jafri, M.Z.M.; Tan, S.C. Feature selection from hyperspectral imaging for guava fruit defects detection. In Proceedings of the SPIE Digital Optical Technologies, Munich, Germany, 25–29 June 2017. [Google Scholar]
- Bhakta, I.; Phadikar, S.; Majumder, K. Importance of Thermal Features in the Evaluation of Bacterial Blight in Rice Plant. In Annual Convention of the Computer Society of India; Springer: Singapore, 2018; pp. 300–313. ISBN 9781489911827. [Google Scholar]
- Bellvert, J.; Zarco-Tejada, P.J.; Girona, J.; Fereres, E. Mapping crop water stress index in a “Pinot-noir” vineyard: Comparing ground measurements with thermal remote sensing imagery from an unmanned aerial vehicle. Precis. Agric. 2014, 15, 361–376. [Google Scholar] [CrossRef]
- Mangus, D.L.; Sharda, A.; Zhang, N. Development and evaluation of thermal infrared imaging system for high spatial and temporal resolution crop water stress monitoring of corn within a greenhouse. Comput. Electron. Agric. 2016, 121, 149–159. [Google Scholar] [CrossRef]
- Buitrago, M.F.; Groen, T.A.; Hecker, C.A.; Skidmore, A.K. Changes in thermal infrared spectra of plants caused by temperature and water stress. ISPRS J. Photogramm. Remote Sens. 2016, 111, 22–31. [Google Scholar] [CrossRef]
- Cohen, Y.; Alchanatis, V.; Sela, E.; Saranga, Y.; Cohen, S.; Meron, M.; Bosak, A.; Tsipris, J.; Ostrovsky, V.; Orolov, V.; et al. Crop water status estimation using thermography: Multi-year model development using ground-based thermal images. Precis. Agric. 2015, 16, 311–329. [Google Scholar] [CrossRef]
- Grant, O.M.; Ochagavía, H.; Baluja, J.; Diago, M.P.; Tardáguila, J. Thermal imaging to detect spatial and temporal variation in the water status of grapevine (Vitis vinifera L.). J. Hortic. Sci. Biotechnol. 2016, 91, 44–55. [Google Scholar] [CrossRef]
- Moran, M.S.; Clarke, T.R.; Inoue, Y.; Vidal, A. Estimating crop water deficit using the relation between surface-air temperature and spectral vegetation index. Remote Sens. Environ. 1994, 49, 246–263. [Google Scholar] [CrossRef]
- Gonzalez-Dugo, V.; Zarco-Tejada, P.; Nicolás, E.; Nortes, P.A.; Alarcón, J.J.; Intrigliolo, D.S.; Fereres, E. Using high resolution UAV thermal imagery to assess the variability in the water status of five fruit tree species within a commercial orchard. Precis. Agric. 2013, 14, 660–678. [Google Scholar] [CrossRef]
- Wang, Y.; Zhang, Y.; Zhang, R.; Li, J.; Zhang, M.; Zhou, S.; Wang, Z. Reduced irrigation increases the water use efficiency and productivity of winter wheat-summer maize rotation on the North China Plain. Sci. Total Environ. 2018, 618, 112–120. [Google Scholar] [CrossRef]
- Vicente, R.; Vergara-Díaz, O.; Medina, S.; Chairi, F.; Kefauver, S.C.; Bort, J.; Serret, M.D.; Aparicio, N.; Araus, J.L. Durum wheat ears perform better than the flag leaves under water stress: Gene expression and physiological evidence. Environ. Exp. Bot. 2018, 153, 271–285. [Google Scholar] [CrossRef]
- Araus, J.L.; Kefauver, S.C.; Zaman-Allah, M.; Olsen, M.S.; Cairns, J.E. Translating High-Throughput Phenotyping into Genetic Gain. Trends Plant Sci. 2018, 23, 451–566. [Google Scholar] [CrossRef] [PubMed]
- Chandel, A.K.; Khot, L.R.; Osroosh, Y.; Peters, T.R. Thermal-RGB imager derived in-field apple surface temperature estimates for sunburn management. Agric. For. Meteorol. 2018, 253–254, 132–140. [Google Scholar] [CrossRef]
- Gan, H.; Lee, W.S.; Alchanatis, V.; Ehsani, R.; Schueller, J.K. Immature green citrus fruit detection using color and thermal images. Comput. Electron. Agric. 2018, 152, 117–125. [Google Scholar] [CrossRef]
- Camino, C.; Zarco-Tejada, P.J.; Gonzalez-Dugo, V. Effects of heterogeneity within tree crowns on airborne-quantified SIF and the CWSI as indicators of water stress in the context of precision agriculture. Remote Sens. 2018, 10, 4. [Google Scholar] [CrossRef]
- Jiang, Y.; Shuang, L.; Li, C.; Paterson, A.H.; Robertson, J. Deep learning for thermal image segmentation to measure canopy temperature of Brassica oleracea in the field. In Proceedings of the 2018 ASABE Annual International Meeting, Detroit, MI, USA, 29 July–1 August 2018; American Society of Agricultural and Biological Engineers: St. Joseph, MI, USA, 2018; Volume 39, pp. 300–313. [Google Scholar]
- Page, G.F.M.; Liénard, J.F.; Pruett, M.J.; Moffett, K.B. Spatiotemporal dynamics of leaf transpiration quantified with time-series thermal imaging. Agric. For. Meteorol. 2018, 256–257, 304–314. [Google Scholar] [CrossRef]
- Agro-Climatic Information System for Irrigation (Sistema de Información Agroclimática para el Regadío, SIAR). Available online: http://eportal.mapama.gob.es/websiar/Inicio.aspx (accessed on 20 February 2019).
- Zadoks, J.; Chang, T.; Konzak, C. A decimal growth code for the growth stages of cereals. Weed Res. 1974, 14, 415–421. [Google Scholar] [CrossRef]
- Schneider, C.A.; Rasband, W.S.; Eliceiri, K.W. NIH Image to ImageJ: 25 years of image analysis. Nat. Methods 2012, 9, 671–675. [Google Scholar] [CrossRef] [PubMed]
- Malacara, D. Uniform Color Systems. In Color Vision and Colorimetry: Theory and Applications, 2nd ed.; SPIE: Bellingham, WA, USA, 2011; pp. 103–129. [Google Scholar]
- Zuiderveld, K. Contrast Limited Adaptive Histogram Equalization. In Graphics Gems; Elsevier: Amsterdam, The Netherlands, 1994; pp. 474–485. ISBN 0-12-336155-9. [Google Scholar]
- Su, C.H.; Chiu, H.S.; Hsieh, T.M. An efficient image retrieval based on HSV color space. In Proceedings of the 2011 International Conference on Electrical and Control Engineering, Yichang, China, 16–18 September 2011; pp. 5746–5749. [Google Scholar] [CrossRef]
- Jimenez-Berni, J.A.; Deery, D.M.; Rozas-Larraondo, P.; Condon, A.; Tony, G.; Rebetzke, G.J.; James, R.A.; Bovill, W.D.; Furbank, R.T.; Sirault, X.R.R. High Throughput Determination of Plant Height, Ground Cover, and Above-Ground Biomass in Wheat with LiDAR. Front. Plant Sci. 2018, 9, 237. [Google Scholar] [CrossRef]
- Tambussi, E.A.; Bort, J.; Guiamet, J.J.; Nogués, S.; Araus, J.L. The Photosynthetic Role of Ears in C3 Cereals: Metabolism, Water Use Efficiency and Contribution to Grain Yield. CRC Crit. Rev. Plant Sci. 2007, 26, 1–16. [Google Scholar] [CrossRef]
- Singh, K.K.; Frazier, A.E. A meta-analysis and review of unmanned aircraft system (UAS) imagery for terrestrial applications. Int. J. Remote Sens. 2018, 39, 5078–5098. [Google Scholar] [CrossRef]
- Smith, M.W.; Vericat, D. From experimental plots to experimental landscapes: Topography, erosion and deposition in sub-humid badlands from Structure-from-Motion photogrammetry. Earth Surf. Process. Landf. 2015, 40, 1656–1671. [Google Scholar] [CrossRef]
- Berdugo, C.A.; Steiner, U.; Dehne, H.W.; Oerke, E.C. Effect of bixafen on senescence and yield formation of wheat. Pestic. Biochem. Physiol. 2012, 104, 171–177. [Google Scholar] [CrossRef]

| Thermal | RGB | |
|---|---|---|
| Temperature of the Ears | Several degrees warmer than leaves [25]. | Irrelevant. |
| Stage growth | From heading to near crop maturity [42]. | From heading to near crop maturity [12]. |
| Accuracy hour of the day and sky conditions | Clear sky conditions. After midday until 18:00, depending on plant water stress conditions. | Depends of the hour of the day, 8:00 to 18:30 [10], 8:00 to 17:00 [14], 9:00 to 16:00 [9], 12:00 to 16:00 and sky (preferably diffuse light) conditions [12]. |
| Position of the camera | Zenithal/nadir. | Zenithal/nadir [6,10,11,12,13]; 45° above the horizontal [14]. |
| Distance of the camera from crop | 0.8–1 m. | 0.85 m [6], 2.5 m [10], 2.9 m [11], 3.5 m [14], 0.8–1 m [12]. |
| Spatial resolution from ground acquired images | Approximately 0.14 cm/pixel, depending on camera and distance from crop. | Ranging 0.01–0.25 cm/pixel [6,10,11,12,13]; depending on camera and distance from crop. |
| Possible algorithm errors | -The algorithm presents errors when the air temperature is too low or high or the sky is too cloudy, or the conditions very windy, which may prevent differences between the canopy and the ear temperature appears. -The camera could be out-of-focus, potentially due to a very short image acquisition distance between the camera and the canopy. -In sparse canopies, soil temperature may affect the background. -Dry or senescent leaf canopy may affect the background. | -False positives where pixels are labeled as ears correspond to leaves and result in irregularities in the ear counting. -False negatives result in ears that are not detected by the algorithm because the contrast between the ear and soil is not great enough and the segmentation algorithm discarded that region. -The algorithm labeled the area as an ear, where the pixels are soil and noise being a result of background brightness caused by a foreign object [12]. |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fernandez-Gallego, J.A.; Buchaillot, M.L.; Aparicio Gutiérrez, N.; Nieto-Taladriz, M.T.; Araus, J.L.; Kefauver, S.C. Automatic Wheat Ear Counting Using Thermal Imagery. Remote Sens. 2019, 11, 751. https://doi.org/10.3390/rs11070751
Fernandez-Gallego JA, Buchaillot ML, Aparicio Gutiérrez N, Nieto-Taladriz MT, Araus JL, Kefauver SC. Automatic Wheat Ear Counting Using Thermal Imagery. Remote Sensing. 2019; 11(7):751. https://doi.org/10.3390/rs11070751
Chicago/Turabian StyleFernandez-Gallego, Jose A., Ma. Luisa Buchaillot, Nieves Aparicio Gutiérrez, María Teresa Nieto-Taladriz, José Luis Araus, and Shawn C. Kefauver. 2019. "Automatic Wheat Ear Counting Using Thermal Imagery" Remote Sensing 11, no. 7: 751. https://doi.org/10.3390/rs11070751
APA StyleFernandez-Gallego, J. A., Buchaillot, M. L., Aparicio Gutiérrez, N., Nieto-Taladriz, M. T., Araus, J. L., & Kefauver, S. C. (2019). Automatic Wheat Ear Counting Using Thermal Imagery. Remote Sensing, 11(7), 751. https://doi.org/10.3390/rs11070751





