Laboratory Visible and Near-Infrared Spectroscopy with Genetic Algorithm-Based Partial Least Squares Regression for Assessing the Soil Phosphorus Content of Upland and Lowland Rice Fields in Madagascar
Abstract
:1. Introduction
2. Materials and Methods
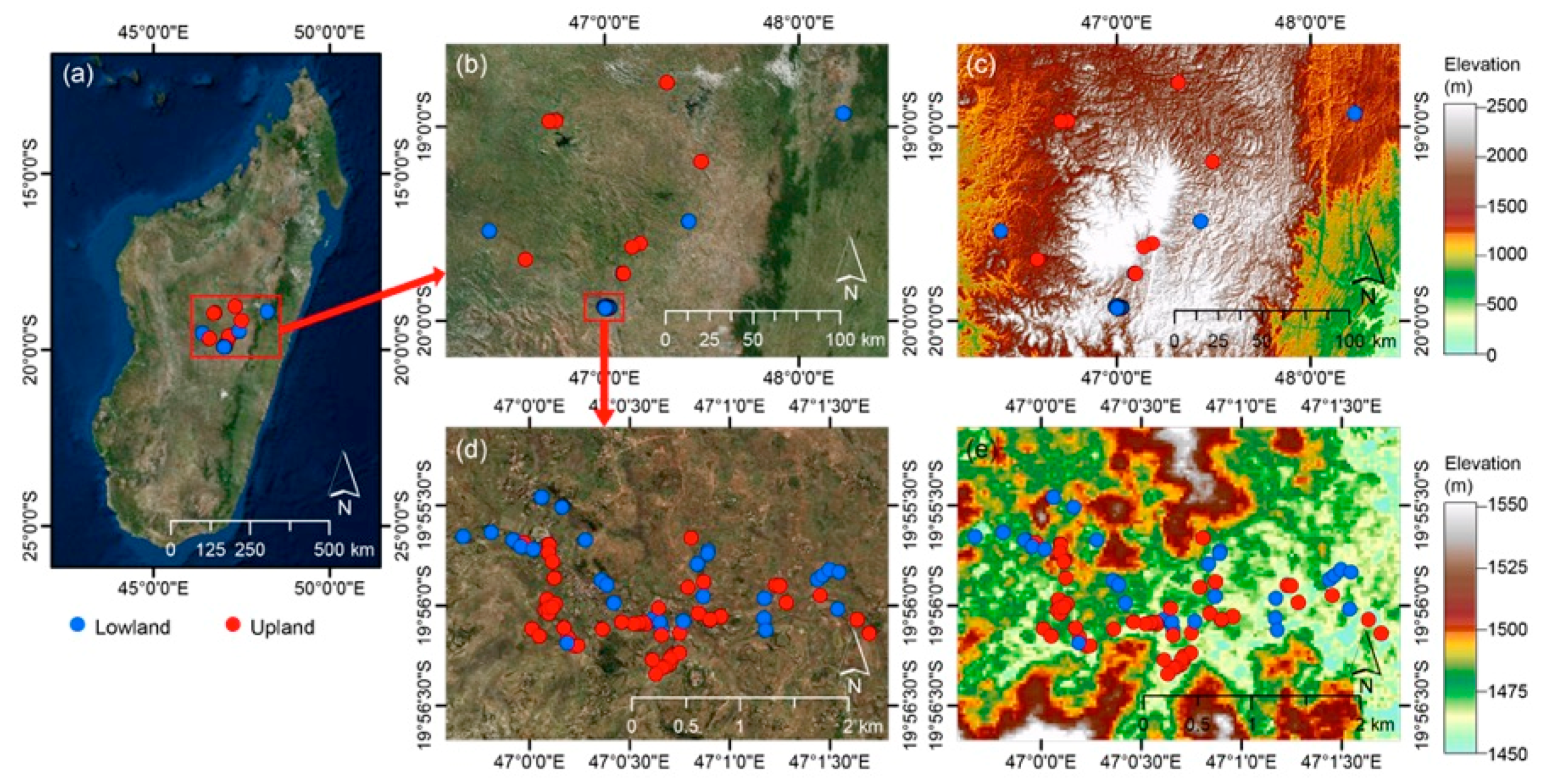
2.1. Study Site and Soil Sampling and Chemical Analyses
2.2. Vis-NIR Diffuse Reflectance Measurement
2.3. Overview of Data Processing
2.4. Preprocessing of Spectral Data
2.5. Standard Full-Spectrum Partial Least Squares (FS-PLS) Regression
2.6. Iterative Stepwise Elimination Partial Least Squares (ISE-PLS) Regression
2.7. Genetic Algorithm Partial Least Squares (GA-PLS) Regression
2.8. Predictive Ability of the PLS Models
2.9. Assessing Significant Wavelengths
3. Results and Discussion
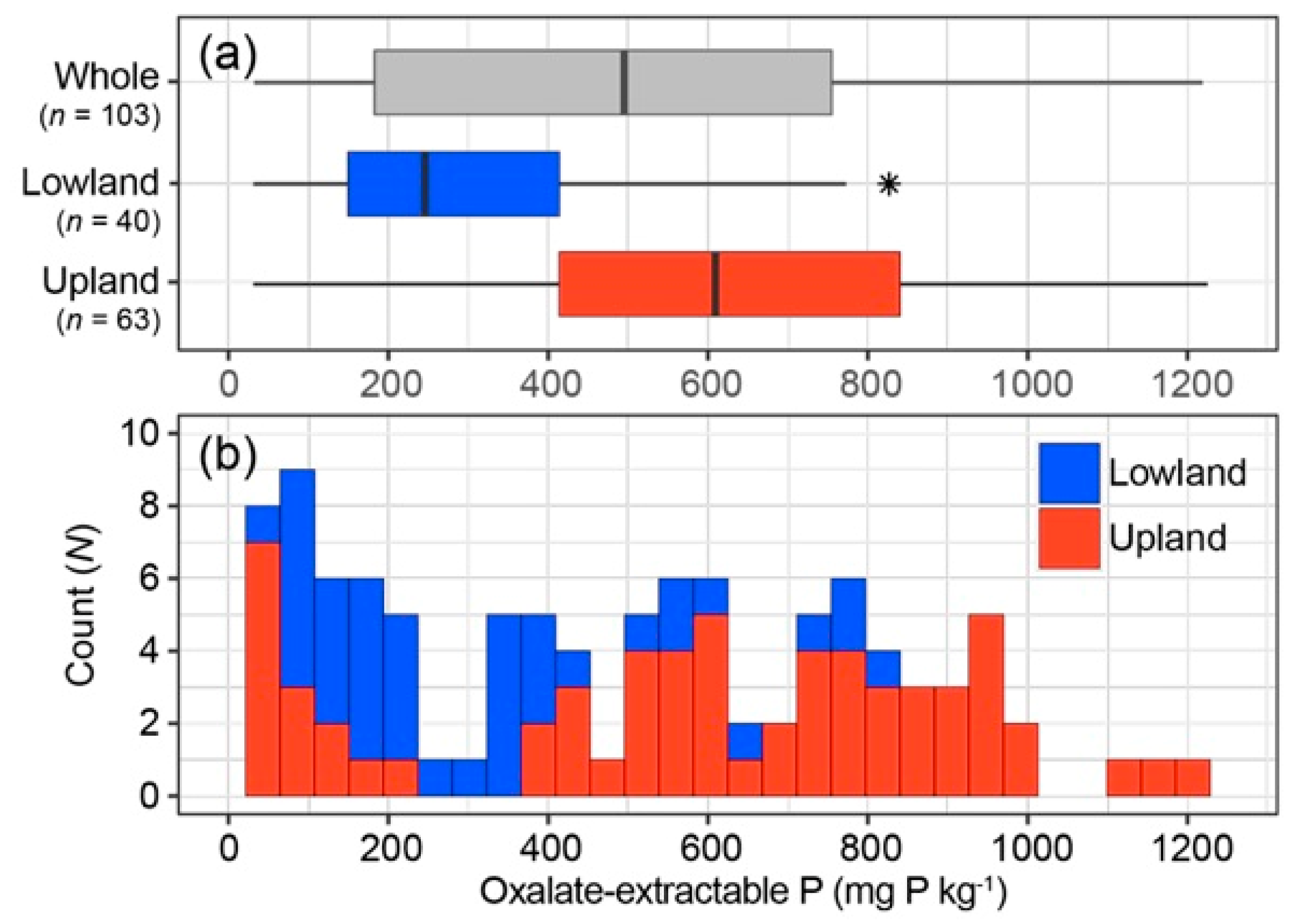
3.1. A Wide Range of Soil Oxalate-Extractable P Contents in Upland and Lowland Rice Fields
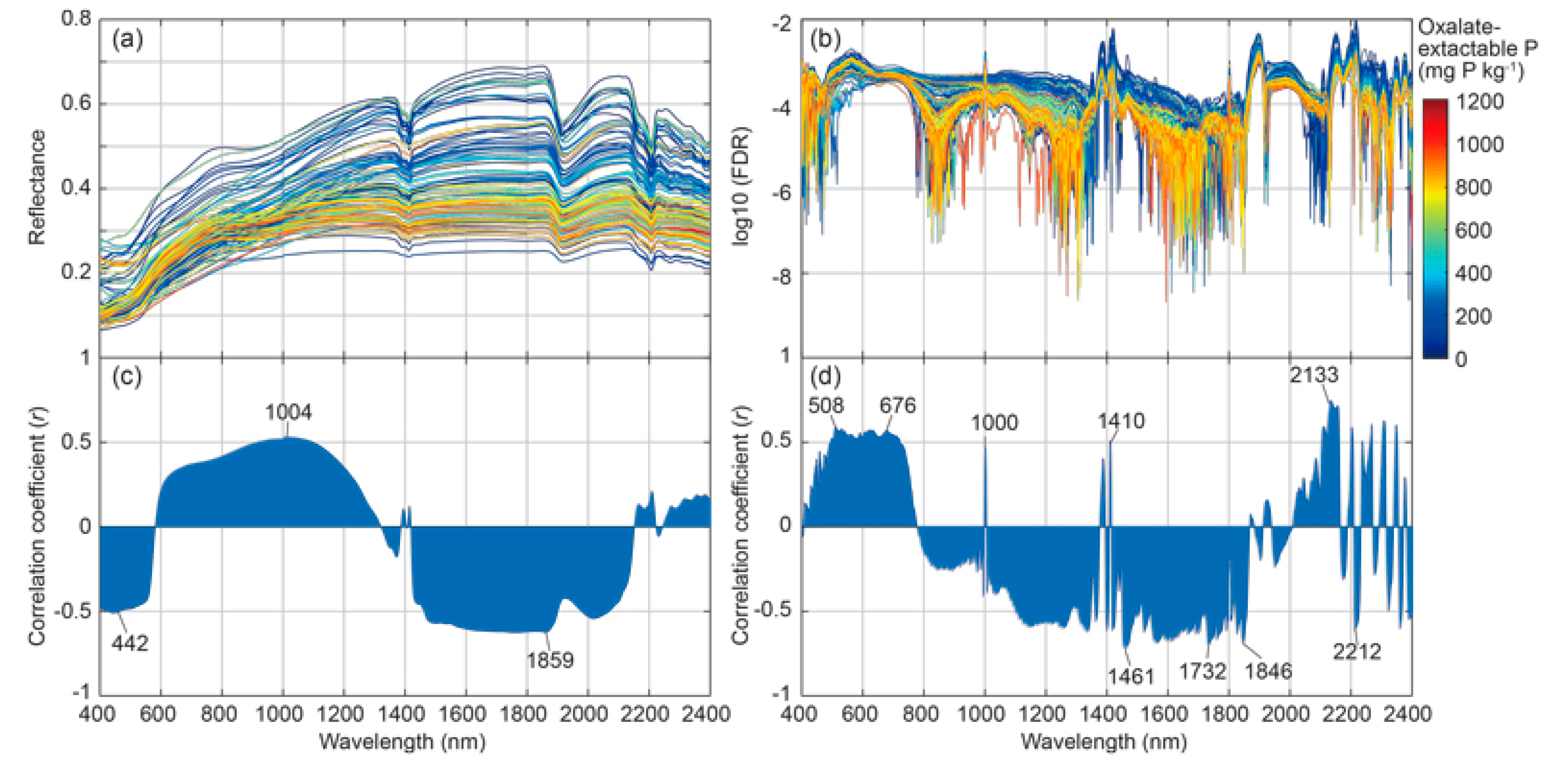
3.2. Soil Spectral Response and Its Correlation to Oxalate-Extractable P in Soil
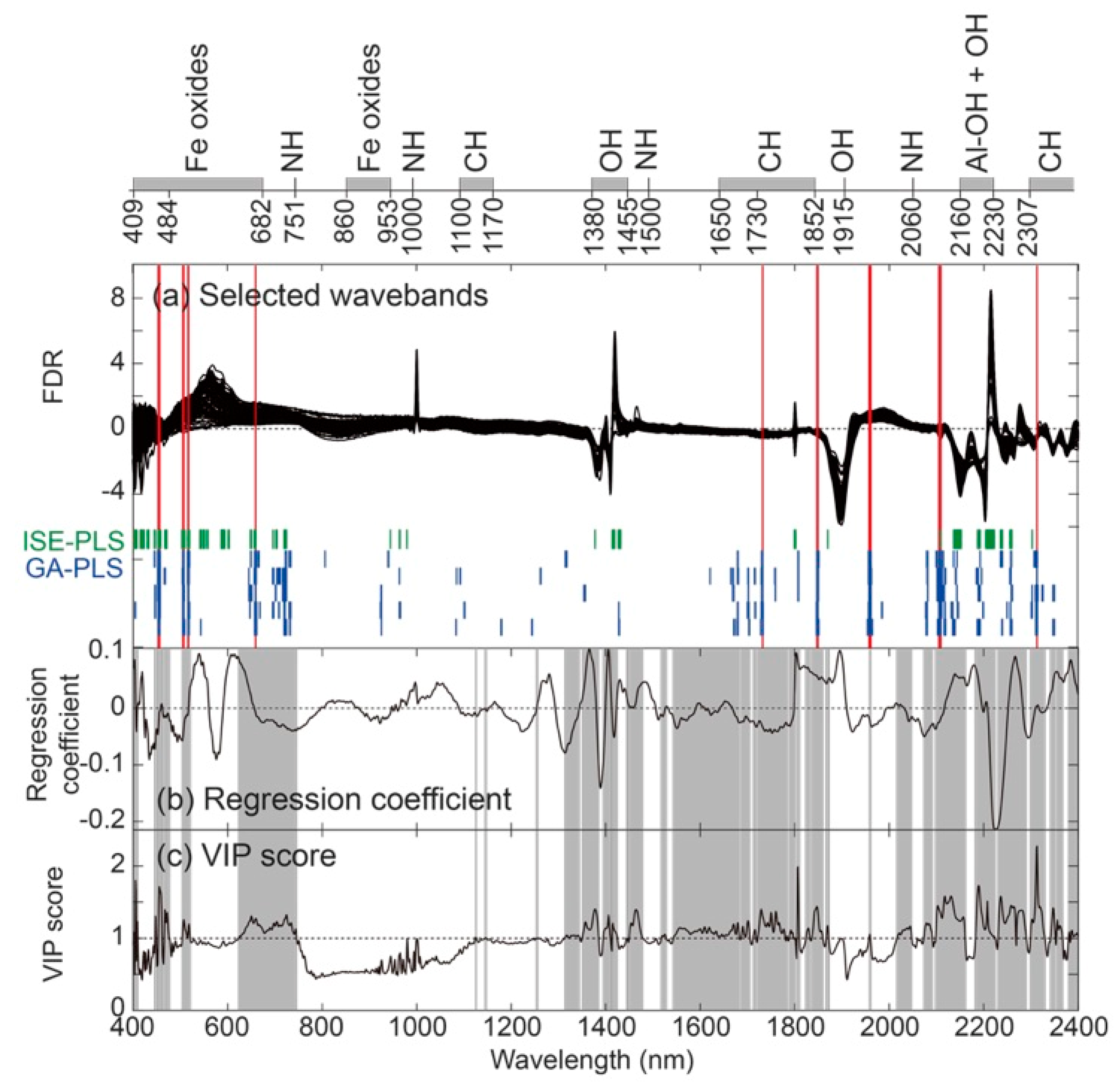
3.3. Selected Wavebands from ISE-PLS and GA-PLS Models
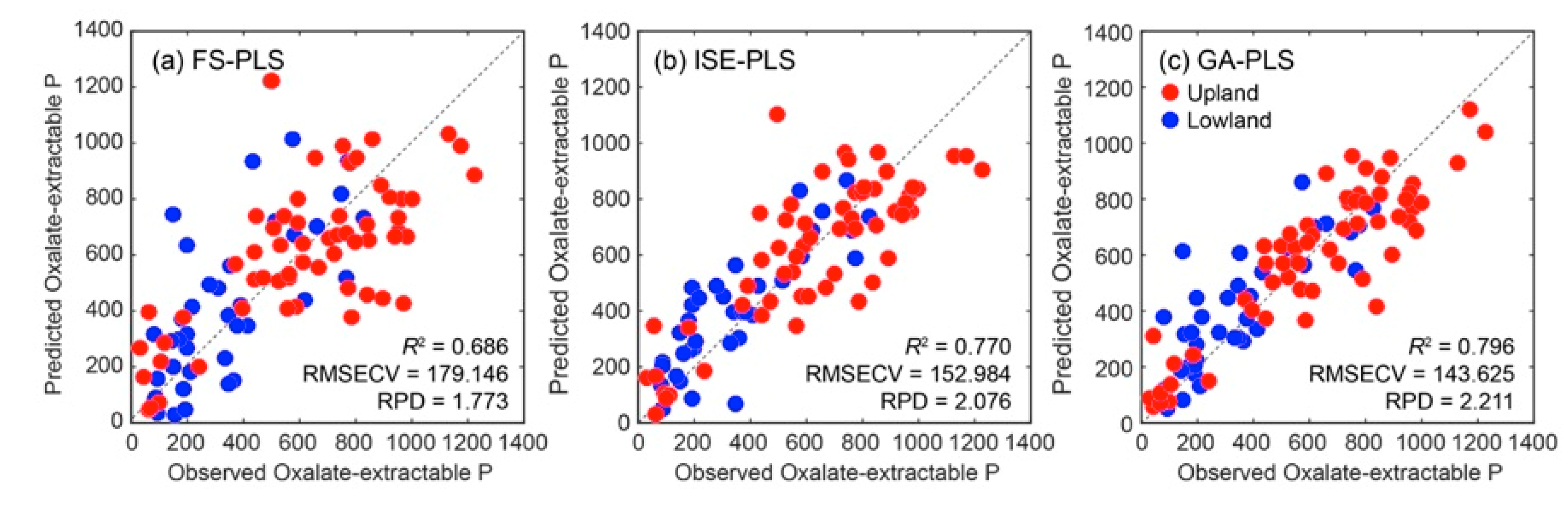
3.4. Waveband Selection with Cross-Validated Calibration Results
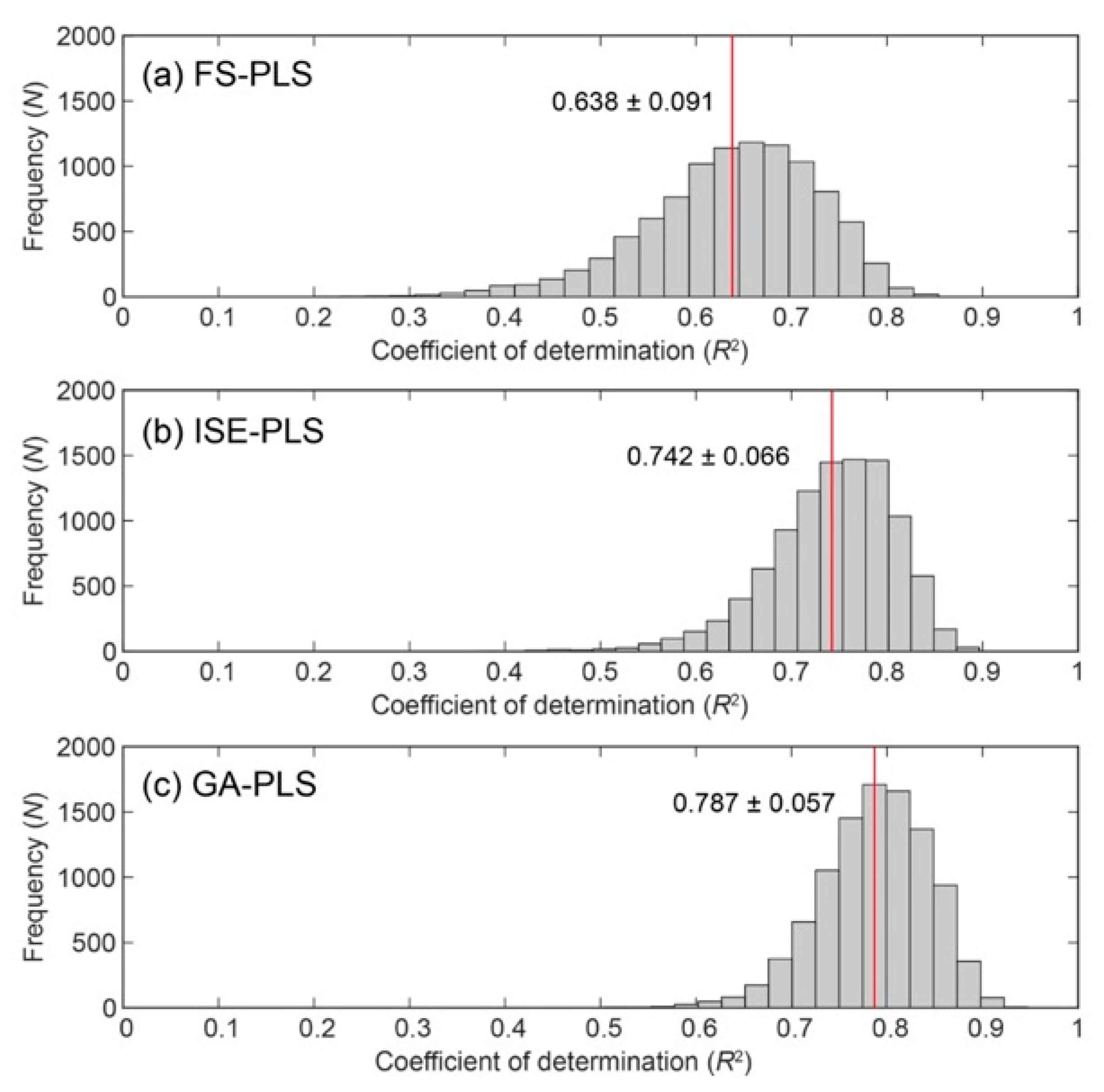
3.5. Evaluation of Predictive Ability Using Modified Bootstrapping
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Dogbe, W.; Sogbedji, J.M.; Buah, S.S.J. Site-specific Nutrient Management for Lowland Rice in the Northern Savannah Zones of Ghana. Curr. Agric. Res. J. 2015, 3, 109–117. [Google Scholar] [CrossRef]
- Kone, B.; Fofana, M.; Sorho, F.; Diatta, S.; Ogunbayo, A.; Sie, M. Nutrient constraint of rainfed rice production in foot slope soil of Guinea Forest in Côte d’Ivoire. Arch. Agron. Soil Sci. 2014, 60, 735–746. [Google Scholar] [CrossRef]
- Koné, B.; Amadji, G.L.; Aliou, S.; Diatta, S.; Akakpo, C. Nutrient constraint and yield potential of rice on upland soil in the south of the Dahoumey gap of West Africa. Arch. Agron. Soil Sci. 2011, 57, 763–774. [Google Scholar] [CrossRef]
- Tamburini, F.; Bernasconi, S.M.; Paytan, A. Phosphorus in the environment. In Eos; White, P.J., Hammond, J.P., Eds.; Springer Netherlands: Dordrecht, The Netherlands, 2012; Volume 93, p. 405. ISBN1 978-1-4020-8434-8. ISBN2 978-1-4020-8435-5. [Google Scholar]
- Balemi, T.; Negisho, K. Management of soil phosphorus and plant adaptation mechanisms to phosphorus stress for sustainable crop production: A review. J. Soil Sci. Plant Nutr. 2012, 12, 547–562. [Google Scholar] [CrossRef]
- Nishigaki, T.; Tsujimoto, Y.; Rinasoa, S.; Rakotoson, T.; Andriamananjara, A.; Razafimbelo, T. Phosphorus uptake of rice plants is affected by phosphorus forms and physicochemical properties of tropical weathered soils. Plant Soil 2018, 435, 27–38. [Google Scholar] [CrossRef]
- Wuenscher, R.; Unterfrauner, H.; Peticzka, R.; Zehetner, F. A comparison of 14 soil phosphorus extraction methods applied to 50 agricultural soils from Central Europe. Plant Soil Environ. 2015, 61, 86–96. [Google Scholar] [CrossRef]
- Helfenstein, J.; Tamburini, F.; von Sperber, C.; Massey, M.S.; Pistocchi, C.; Chadwick, O.A.; Vitousek, P.M.; Kretzschmar, R.; Frossard, E. Combining spectroscopic and isotopic techniques gives a dynamic view of phosphorus cycling in soil. Nat. Commun. 2018, 9, 3226. [Google Scholar] [CrossRef] [PubMed]
- Rabeharisoa, L.; Razanakoto, O.R.; Razafimanantsoa, M.P.; Rakotoson, T.; Amery, F.; Smolders, E. Larger bioavailability of soil phosphorus for irrigated rice compared with rainfed rice in Madagascar: Results from a soil and plant survey. Soil Use Manag. 2012, 28, 448–456. [Google Scholar] [CrossRef]
- Sims, J.T.; Sharpley, A.N.; Condron, L.M.; Turner, B.L.; Cade-Menun, B.J. Chemistry and Dynamics of Soil Organic Phosphorus. Phosphorus Agric. Environ. 2005, 87–121. [Google Scholar] [CrossRef]
- Viscarra Rossel, R.A.; Walvoort, D.J.J.; McBratney, A.B.; Janik, L.J.; Skjemstad, J.O. Visible, near infrared, mid infrared or combined diffuse reflectance spectroscopy for simultaneous assessment of various soil properties. Geoderma 2006, 131, 59–75. [Google Scholar] [CrossRef]
- Miller, C.E. Chemical principles of near-infrared technology. In Near Infrared Technology in the Agricultural and Food Industries; Williams, P.C., Horris, K.H., Eds.; American Association of Cereal Chemists: St. Paul, MN, USA, 2001; pp. 19–37. [Google Scholar]
- Cañasveras, J.C.; Barrón, V.; del Campillo, M.C.; Torrent, J.; Gómez, J.A. Estimation of aggregate stability indices in Mediterranean soils by diffuse reflectance spectroscopy. Geoderma 2010, 158, 78–84. [Google Scholar] [CrossRef] [Green Version]
- Cañasveras Sánchez, J.C.; Barrón, V.; del Campillo, M.C.; Viscarra Rossel, R.A. Reflectance spectroscopy: A tool for predicting soil properties related to the incidence of Fe chlorosis. Span. J. Agric. Res. 2012, 10, 10. [Google Scholar]
- Viscarra Rossel, R.A.; Behrens, T.; Ben-Dor, E.; Brown, D.J.; Demattê, J.A.M.; Shepherd, K.D.; Shi, Z.; Stenberg, B.; Stevens, A.; Adamchuk, V.; et al. A global spectral library to characterize the world’s soil. Earth-Sci. Rev. 2016, 155, 198–230. [Google Scholar] [CrossRef]
- Yang, H.; Kuang, B.; Mouazen, A.M. Quantitative analysis of soil nitrogen and carbon at a farm scale using visible and near infrared spectroscopy coupled with wavelength reduction. Eur. J. Soil Sci. 2012, 63, 410–420. [Google Scholar] [CrossRef]
- Vohland, M.; Ludwig, M.; Thiele-Bruhn, S.; Ludwig, B. Determination of soil properties with visible to near- and mid-infrared spectroscopy: Effects of spectral variable selection. Geoderma 2014, 223, 88–96. [Google Scholar] [CrossRef]
- Kawamura, K.; Tsujimoto, Y.; Rabenarivo, M.; Asai, H.; Andriamananjara, A.; Rakotoson, T. Vis-NIR spectroscopy and PLS regression with waveband selection for estimating the total C and N of paddy soils in Madagascar. Remote Sens. 2017, 9, 1081. [Google Scholar] [CrossRef]
- Bolster, K.L.; Martin, M.E.; Aber, J.D. Determination of carbon fraction and nitrogen concentration in tree foliage by near infrared reflectance: A comparison of statistical methods. Can. J. For. Res. 1996, 26, 590–600. [Google Scholar] [CrossRef]
- Kawamura, K.; Watanabe, N.; Sakanoue, S.; Inoue, Y. Estimating forage biomass and quality in a mixed sown pasture based on PLS regression with waveband selection. Grassl. Sci. 2008, 54, 131–146. [Google Scholar] [CrossRef]
- Boggia, R.; Forina, M.; Fossa, P.; Mosti, L. Chemometric study and validation strategies in the structure-activity relationships of new cardiotonic agents. Quant. Struct. Relatsh. 1997, 16, 201–213. [Google Scholar] [CrossRef]
- Centner, V.; Massart, D.L.; de Noord, O.E.; de Jong, S.; Vandeginste, B.M.; Sterna, C. Elimination of uninformative variables for multivariate calibration. Anal. Chem. 1996, 68, 3851–3858. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Liang, Y.; Xu, Q.; Cao, D. Key wavelengths screening using competitive adaptive reweighted sampling method for multivariate calibration. Anal. Chim. Acta 2009, 648, 77–84. [Google Scholar] [CrossRef] [PubMed]
- Nørgaard, L.; Saudland, A.; Wagner, J.; Nielsen, J.P.; Munck, L.; Engelsen, S.B. Interval partial least-squares regression (iPLS): A comparative chemometric study with an example from near-infrared spectroscopy. Appl. Spectrosc. 2000, 54, 413–419. [Google Scholar] [CrossRef]
- Jiang, J.H.; James, R.; Siesler, B.H.W.; Ozaki, Y. Wavelength interval selection in multicomponent spectral analysis by moving window partial least-squares regression with applications to mid-infrared and near-infrared spectroscopic data. Anal. Chem. 2002, 74, 3555–3565. [Google Scholar] [CrossRef] [PubMed]
- Leardi, R. Application of a genetic algorithm to feature selection under full validation conditions and to outlier detection. J. Chemom. 1994, 8, 65–79. [Google Scholar] [CrossRef]
- Leardi, R. Application of genetic algorithm-PLS for feature selection in spectral data sets. J. Chemom. 2000, 14, 643–655. [Google Scholar] [CrossRef]
- Leardi, R.; González, A.L. Genetic algorithms applied to feature selection in PLS regression: How and when to use them. Chemom. Intell. Lab. Syst. 1998, 41, 195–207. [Google Scholar] [CrossRef]
- Lucasius, C.B.; Kateman, G. Understanding and using genetic algorithms Part 2. Representation, configuration and hybridization. Chemom. Intell. Lab. Syst. 1994, 25, 99–145. [Google Scholar] [CrossRef]
- Kawamura, K.; Watanabe, N.; Sakanoue, S.; Lee, H.J.; Inoue, Y.; Odagawa, S. Testing genetic algorithm as a tool to select relevant wavebands from field hyperspectral data for estimating pasture mass and quality in a mixed sown pasture using partial least squares regression. Grassl. Sci. 2010, 56, 205–216. [Google Scholar] [CrossRef]
- Kawamura, K.; Watanabe, N.; Sakanoue, S.; Lee, H.J.; Lim, J.; Yoshitoshi, R. Genetic algorithm-based partial least squares regression for estimating legume content in a grass-legume mixture using field hyperspectral measurements. Grassl. Sci. 2013, 59, 166–172. [Google Scholar] [CrossRef]
- Bogrekci, I.; Lee, W.S. Spectral soil signatures and sensing phosphorus. Biosyst. Eng. 2005, 92, 527–533. [Google Scholar] [CrossRef]
- Maleki, M.R.; Van Holm, L.; Ramon, H.; Merckx, R.; De Baerdemaeker, J.; Mouazen, A.M. Phosphorus Sensing for Fresh Soils using Visible and Near Infrared Spectroscopy. Biosyst. Eng. 2006, 95, 425–436. [Google Scholar] [CrossRef]
- Kuang, B.; Mouazen, A.M. Calibration of visible and near infrared spectroscopy for soil analysis at the field scale on three European farms. Eur. J. Soil Sci. 2011, 62, 629–636. [Google Scholar] [CrossRef]
- Tsujimoto, Y.; Horie, T.; Randriamihary, H.; Shiraiwa, T.; Homma, K. Soil management: The key factors for higher productivity in the fields utilizing the system of rice intensification (SRI) in the central highland of Madagascar. Agric. Syst. 2009, 100, 61–71. [Google Scholar] [CrossRef]
- IUSS Working Group, WRB. World Reference Base for Soil Resources 2014, Update 2015 International Soil Classification System for Naming Soils and Creating Legends for Soil Maps; World Soil Resources Reports No. 106; Food and Agriculture Organization of the United Nations: Rome, Italy, 2015. [Google Scholar]
- Soil Survey Staff. Keys to Soil Taxonomy, 12th ed.; USDA-Natural Resources Conservation Service: Washington, DC, USA, 2014.
- Schwertmann, U. The differentiation of iron oxides in soils by extraction with ammonium oxalate solution. Z. Pflanz. Bodenkd. 1964, 105, 194–202. [Google Scholar] [CrossRef]
- van Veldhoven, P.P.; Mannaerts, G.P. Inorganic and organic phosphate measurements in the nanomolar range. Anal. Biochem. 1987, 161, 45–48. [Google Scholar] [CrossRef]
- Reeves, J.; McCarty, G.; Mimmo, T. The potential of diffuse reflectance spectroscopy for the determination of carbon inventories in soils. Environ. Pollut. 2002, 116, S277–S284. [Google Scholar] [CrossRef]
- Savitzky, A.; Golay, E.J.M. Smoothing and difference of data by simplified least squares procedures. Anal. Chem. 1964, 36, 1627–1639. [Google Scholar] [CrossRef]
- Brunet, D.; Barthès, B.G.; Chotte, J.-L.; Feller, C. Determination of carbon and nitrogen contents in Alfisols, Oxisols and Ultisols from Africa and Brazil using NIRS analysis: Effects of sample grinding and set heterogeneity. Geoderma 2007, 139, 106–117. [Google Scholar] [CrossRef]
- Forina, M.; Lanteri, S.; Oliveros, M.C.C.; Millan, C.P. Selection of useful predictors in multivariate calibration. Anal. Bioanal. Chem. 2004, 380, 397–418. [Google Scholar] [CrossRef] [PubMed]
- Leardi, R.; Boggia, R.; Terrile, M. Genetic Algorithms as a strategyfor feature selection. J. Chemom. 1992, 6, 267–281. [Google Scholar] [CrossRef]
- Ding, Q.; Small, G.W.; Arnold, M.A. Genetic algorithm-based wavelength selection for the near-infrared determination of glucose in biological matrixes: Initialization strategies and effects of spectral resolution. Anal. Chem. 1998, 70, 4472–4479. [Google Scholar] [CrossRef] [PubMed]
- Leardi, R.; Seasholtz, M.B.; Pell, R.J. Variable selection for multivariate calibration using a genetic algorithm: Prediction of additive concentrations in polymer films from Fourier transform-infrared spectral data. Anal. Chim. Acta 2002, 461, 189–200. [Google Scholar] [CrossRef]
- Wold, S.; Sjöström, M.; Eriksson, L. PLS-regression: A basic tool of chemometrics. Chemom. Intell. Lab. Syst. 2001, 58, 109–130. [Google Scholar] [CrossRef]
- Chong, I.-G.; Jun, C.-H. Performance of some variable selection methods when multicollinearity is present. Chemom. Intell. Lab. Syst. 2005, 78, 103–112. [Google Scholar] [CrossRef]
- Li, B.; Liew, O.W.; Asundi, A.K. Pre-visual detection of iron and phosphorus deficiency by transformed reflectance spectra. J. Photochem. Photobiol. B Biol. 2006, 85, 131–139. [Google Scholar] [CrossRef] [PubMed]
- Ben-Dor, E. Quantitative remote sensing of soil properties. In Advances in Agronomy; Academic Press: New York, NY, USA, 2002; Volume 75, pp. 173–243. ISBN 9780120007936. [Google Scholar]
- Drăguţ, L.; Dornik, A. Land-surface segmentation as a method to create strata for spatial sampling and its potential for digital soil mapping. Int. J. Geogr. Inf. Sci. 2016, 30, 1359–1376. [Google Scholar] [CrossRef]
- Scheinost, A.C.; Chavernas, A.; Barrón, V.; Torrent, J. Use and limitations of second-derivative diffuse reflectance spectroscopy in the visible to near-infrared range to identify and quantify Fe oxide minerals in soils. Clays Clay Miner. 1998, 46, 528–536. [Google Scholar] [CrossRef]
- Mortimore, J.L.; Marshll, L.-J.R.; Almond, J.M.; Hollins, P.; Matthews, W. Analysis of red and yellow ochre samples from Clearwell Caves and Çatalhöyük by vibrational spectroscopy and other techniques. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2004, 60, 1179–1188. [Google Scholar] [CrossRef] [PubMed]
- Shonk, J.L.; Gaultney, L.D.; Schulze, D.G.; Van Scoyoc, G.E. Spectroscopic sensing of soil organic-matter content. Trans. ASAE 1991, 34, 1978–1984. [Google Scholar] [CrossRef]
- Daniel, K.W.; Tripathi, N.K.; Honda, K. Artificial neural network analysis of laboratory and in situ spectra for the estimation of macronutrients in soils of Lop Buri (Thailand). Aust. J. Soil Res. 2003, 41, 47–59. [Google Scholar] [CrossRef]
- Knadel, M.; Viscarra Rossel, R.A.; Deng, F.; Thomsen, A.; Greve, M.H. Visible–Near Infrared Spectra as a Proxy for Topsoil Texture and Glacial Boundaries. Soil Sci. Soc. Am. J. 2013, 77, 568. [Google Scholar] [CrossRef]
- Hunt, G.H.; Salisbury, J.W. Visible and Near Infrared Spectra of Minerals and Rocks: XI. Sedimentary Rocks. Mod. Geol. 1976, 5, 211–217. [Google Scholar]
- Katuwal, S.; Knadel, M.; Moldrup, P.; Norgaard, T.; Greve, M.H.; de Jonge, L.W. Visible–Near-Infrared Spectroscopy can predict Mass Transport of Dissolved Chemicals through Intact Soil. Sci. Rep. 2018, 8, 11188. [Google Scholar] [CrossRef] [PubMed]
- Ramaroson, V.H.; Becquer, T.; Sá, S.O.; Razafimahatratra, H.; Delarivière, J.L.; Blavet, D.; Vendrame, P.R.S.; Rabeharisoa, L.; Rakotondrazafy, A.F.M. Mineralogical analysis of ferralitic soils in Madagascar using NIR spectroscopy. CATENA 2018, 168, 102–109. [Google Scholar] [CrossRef]
- Ben-Dor, E.; Inbar, Y.; Chen, Y. The reflectance spectra of organic matter in the visible near-infrared and short wave infrared region (400–2500 nm) during a controlled decomposition process. Remote Sens. Environ. 1997, 61, 1–15. [Google Scholar] [CrossRef]
- Clark, R.N.; King, T.V.V.; Klejwa, M.; Swayze, G.A.; Vergo, N. High spectral resolution reflectance spectroscopy of minerals. J. Geophys. Res. 1990, 95, 12653–12680. [Google Scholar] [CrossRef]
- Clark, R.N. Spectroscopy of rocks and minerals, and principles of spectroscopy. In Manual of Remote Sensing; John Wiley and Sons, Inc.: Chichester, UK, 1999; pp. 3–58. [Google Scholar]
- Turner, B.L. Organic phosphorus in Madagascan rice soils. Geoderma 2006, 136, 279–288. [Google Scholar] [CrossRef] [Green Version]
- Viscarra Rossel, R.A.; Fouad, Y.; Walter, C. Using a digital camera to measure soil organic carbon and iron contents. Biosyst. Eng. 2008, 100, 149–159. [Google Scholar] [CrossRef]
- Hunt, G.R. Spectral Signatures of Particulate Minerals in the Visible and Near Infrared. Geophysics 1977, 42, 501–513. [Google Scholar] [CrossRef]
- Sherman, D.M.; Waite, D.T. Electronic spectra of Fe3+ oxides and oxide hydroxides in the near IR to near UV. Am. Mineral. 1985, 70, 1262–1269. [Google Scholar]
- Stenberg, B.; Viscarra Rossel, R.A.; Mouazen, A.M.; Wetterlind, J. Visible and Near Infrared Spectroscopy in Soil Science. Adv. Agron. 2010, 107, 163–215. [Google Scholar] [Green Version]
- Fourty, T.; Baret, F.; Jacquemoud, S.; Schmuck, G.; Verdebout, J. Leaf optical properties with explicit description of its biochemical composition: Direct and inverse problems. Remote Sens. Environ. 1996, 56, 104–117. [Google Scholar] [CrossRef]
- Bishop, J.L.; Pieters, C.M.; Edwards, J.O. Infrared spectroscopic analyses on the nature of water in montmorillonite. Clays Clay Miner. 1994, 42, 702–716. [Google Scholar] [CrossRef]
- Darvishzadeh, R.; Skidmore, A.; Atzberger, C.; van Wieren, S. Estimation of vegetation LAI from hyperspectral reflectance data: Effects of soil type and plant architecture. Int. J. Appl. Earth Obs. Geoinf. 2008, in press, 358–373. [Google Scholar] [CrossRef]
- Wang, Z.; Kawamura, K.; Sakuno, Y.; Fan, X.; Gong, Z.; Lim, J. Retrieval of chlorophyll-a and total suspended solids using iterative stepwise elimination partial least squares (ISE-PLS) regression based on field hyperspectral measurements in irrigation ponds in Higashihiroshima, Japan. Remote Sens. 2017, 9, 264. [Google Scholar] [CrossRef]
- Holst, J.; Liu, C.; Yao, Z.; Brüggemann, N.; Zheng, X.; Han, X.; Butterbach-Bahl, K. Importance of point sources on regional nitrous oxide fluxes in semi-arid steppe of Inner Mongolia, China. Plant Soil 2007, 296, 209–226. [Google Scholar] [CrossRef]
- Leardi, R.; Nørgaard, L. Sequential application of backward interval partial least squares and genetic algorithms for the selection of relevant spectral regions. J. Chemom. 2004, 18, 486–497. [Google Scholar] [CrossRef]

| Parameter | Condition |
|---|---|
| Population size | 30 chromosomes |
| Regression method | PLS |
| Response | Cross-validated percent explained variance (five deletion group; the number of components is determined by cross-validation) |
| Maximum number of variables selected for the same chromosome | 30 |
| Probability of mutation | 1% |
| Maximum number of latent variables | 15 |
| Number of runs | 100 |
| Window size for smoothing | 3 |
| Data Set | n | Min | Max | Median | Mean | SD | CV |
|---|---|---|---|---|---|---|---|
| Whole | 103 | 30.73 | 1225.16 | 496.84 | 484.15 | 319.10 | 65.91 |
| Upland | 63 | 30.78 | 1225.16 | 609.23 | 588.74 | 324.93 | 55.70 |
| Lowland | 40 | 30.73 | 826.64 | 245.79 | 319.41 | 223.26 | 69.89 |
| Selected Waveband (nm) | Previously Known Waveband and Related Soil Component | ||
|---|---|---|---|
| Waveband (nm) | Soil Component | Reference | |
| 454–457 | 400–700 | organic matter (color) | [11,64] |
| 470 | Fe3+, ferric oxide | [65] | |
| 506–508, 517, 518 | 488–499 | ferrihydrite | [52] |
| 495, 510 | hematite | [66] | |
| 660 | 660 | goethite | [67] |
| 655 | schwertmannite | [52] | |
| 1732 | 1720 | organic matter | [55] |
| 1726 | aliphatic C-H stretch, cellulose, lignin, starch, pectin, wax, humic acid | [60] | |
| 1730 | protein, cellulose, aliphatic C-H stretch, lignin, starch, pectin, wax, humic acid | [68] | |
| 1847–1849 | 1730–1852 | methyl (C-H) | [61,62] |
| 1957–1961 | 1950 | sugar, starch, cellulose, lignin, protein | [68] |
| 1961 | phenolics (C-OH) | [61,62] | |
| 1970 | smectite, shoulder due to absorbed water | [69] | |
| 2105, 2107, 2109 | 2111 | organic matter, cellulose, glucan, pectin | [60] |
| 2312 | 2300 | C-H stretch fundamentals | [61] |
| 2307–2469 | methyl | [62] | |
| 2309 | aliphatic C-H, aromatic stretch, humic acid wax, starch | [60] | |
| 2310 | oil | [54] | |
| Regression Method | Cross-Validation for Whole Data Set (n = 103) | |||||
|---|---|---|---|---|---|---|
| NLV | R2CV | RMSECV | RPD | NW | NW% | |
| FS-PLS | 7 | 0.686 | 179.146 | 1.773 | ||
| ISE-PLS | 7 | 0.770 | 152.984 | 2.076 | 158 | 7.9 |
| GA-PLS | 6 | 0.796 | 143.625 | 2.211 | 94 | 4.7 |
| Regression Method | Training Data Set (n = 69) | Test Data Set (n = 34) | ||||
|---|---|---|---|---|---|---|
| Mean NLV | Mean R2 | Mean RMSECV | Mean R2 | Mean RMSEP | ΔRMSEP 1 | |
| FS-PLS | 7.285 | 0.659 | 188.560 | 0.638 | 197.860 | |
| ISE-PLS | 6.419 | 0.751 | 160.180 | 0.742 | 165.786 | –16.21 |
| GA-PLS | 5.364 | 0.782 | 148.930 | 0.787 | 149.013 | –24.69 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kawamura, K.; Tsujimoto, Y.; Nishigaki, T.; Andriamananjara, A.; Rabenarivo, M.; Asai, H.; Rakotoson, T.; Razafimbelo, T. Laboratory Visible and Near-Infrared Spectroscopy with Genetic Algorithm-Based Partial Least Squares Regression for Assessing the Soil Phosphorus Content of Upland and Lowland Rice Fields in Madagascar. Remote Sens. 2019, 11, 506. https://doi.org/10.3390/rs11050506
Kawamura K, Tsujimoto Y, Nishigaki T, Andriamananjara A, Rabenarivo M, Asai H, Rakotoson T, Razafimbelo T. Laboratory Visible and Near-Infrared Spectroscopy with Genetic Algorithm-Based Partial Least Squares Regression for Assessing the Soil Phosphorus Content of Upland and Lowland Rice Fields in Madagascar. Remote Sensing. 2019; 11(5):506. https://doi.org/10.3390/rs11050506
Chicago/Turabian StyleKawamura, Kensuke, Yasuhiro Tsujimoto, Tomohiro Nishigaki, Andry Andriamananjara, Michel Rabenarivo, Hidetoshi Asai, Tovohery Rakotoson, and Tantely Razafimbelo. 2019. "Laboratory Visible and Near-Infrared Spectroscopy with Genetic Algorithm-Based Partial Least Squares Regression for Assessing the Soil Phosphorus Content of Upland and Lowland Rice Fields in Madagascar" Remote Sensing 11, no. 5: 506. https://doi.org/10.3390/rs11050506
APA StyleKawamura, K., Tsujimoto, Y., Nishigaki, T., Andriamananjara, A., Rabenarivo, M., Asai, H., Rakotoson, T., & Razafimbelo, T. (2019). Laboratory Visible and Near-Infrared Spectroscopy with Genetic Algorithm-Based Partial Least Squares Regression for Assessing the Soil Phosphorus Content of Upland and Lowland Rice Fields in Madagascar. Remote Sensing, 11(5), 506. https://doi.org/10.3390/rs11050506






