A Comparison of the Signal from Diverse Optical Sensors for Monitoring Alpine Grassland Dynamics
Abstract
:1. Introduction
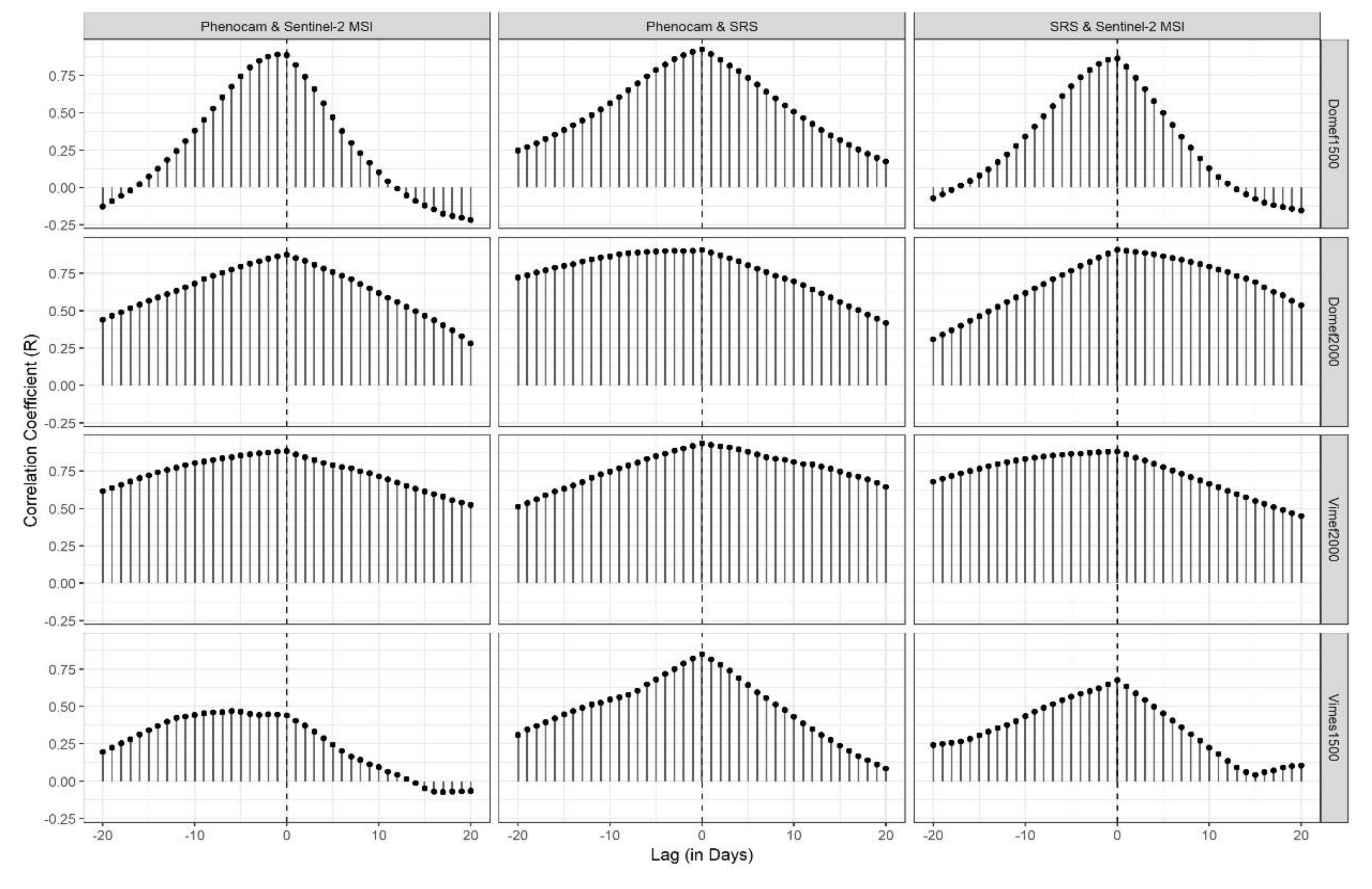
- The similarity of NDVI signal among sensors for each meadow site by visually interpreting the NDVI signatures as well as by calculating linear correlation and cross-correlation among sensors;
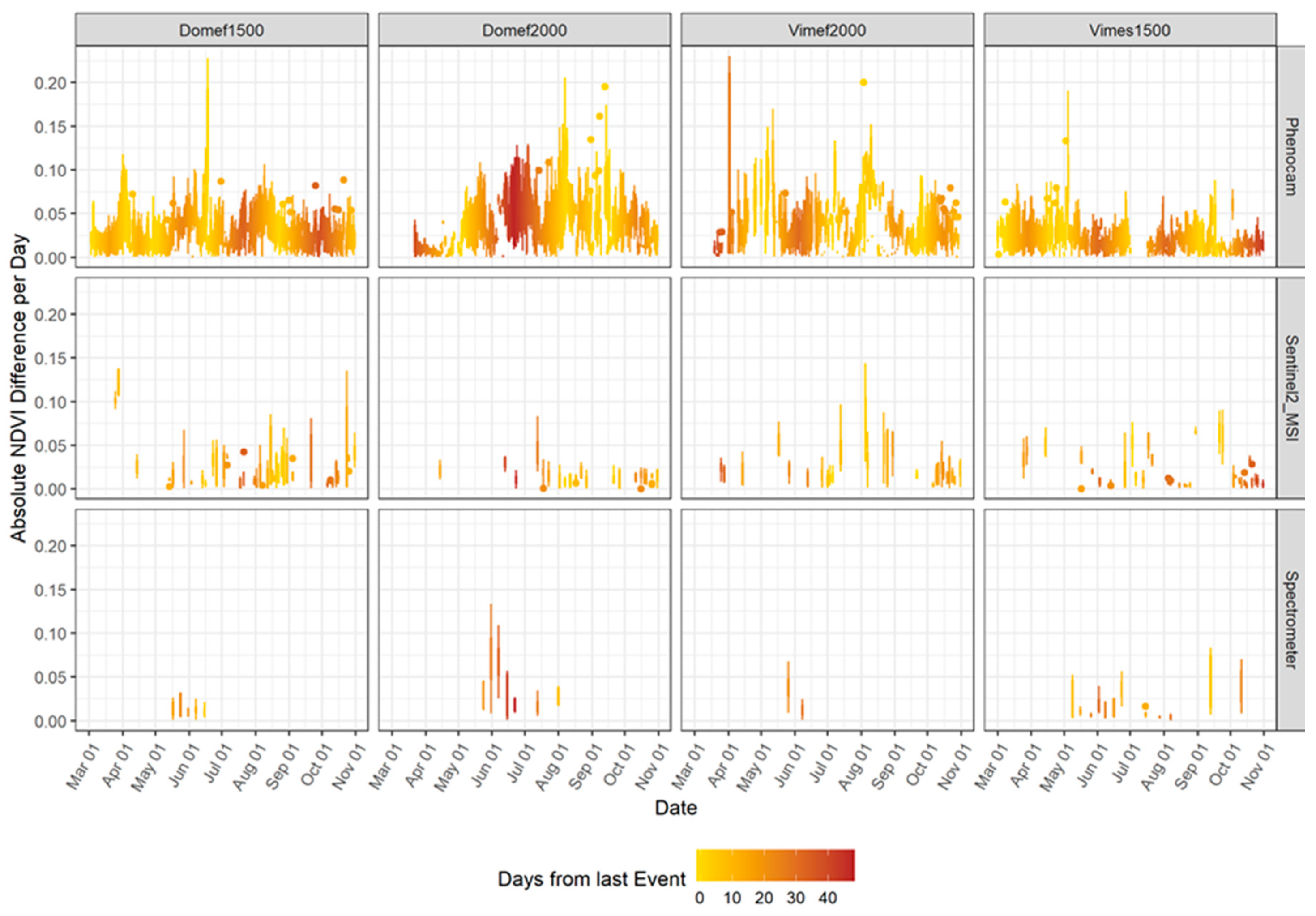
- the suitability of each sensor for detecting events with short-term impacts on the vegetation cover, such as harvests and snow coverage, by analyzing short time spectral changes in each sensor; and
- sensor-specific characteristics of the grassland sites by collecting multiple NDVI measurements on each of the four sites. The different plots are compared visually and with linear correlation analysis to assess the stability of the NDVI signal over time.
2. Materials and Methods
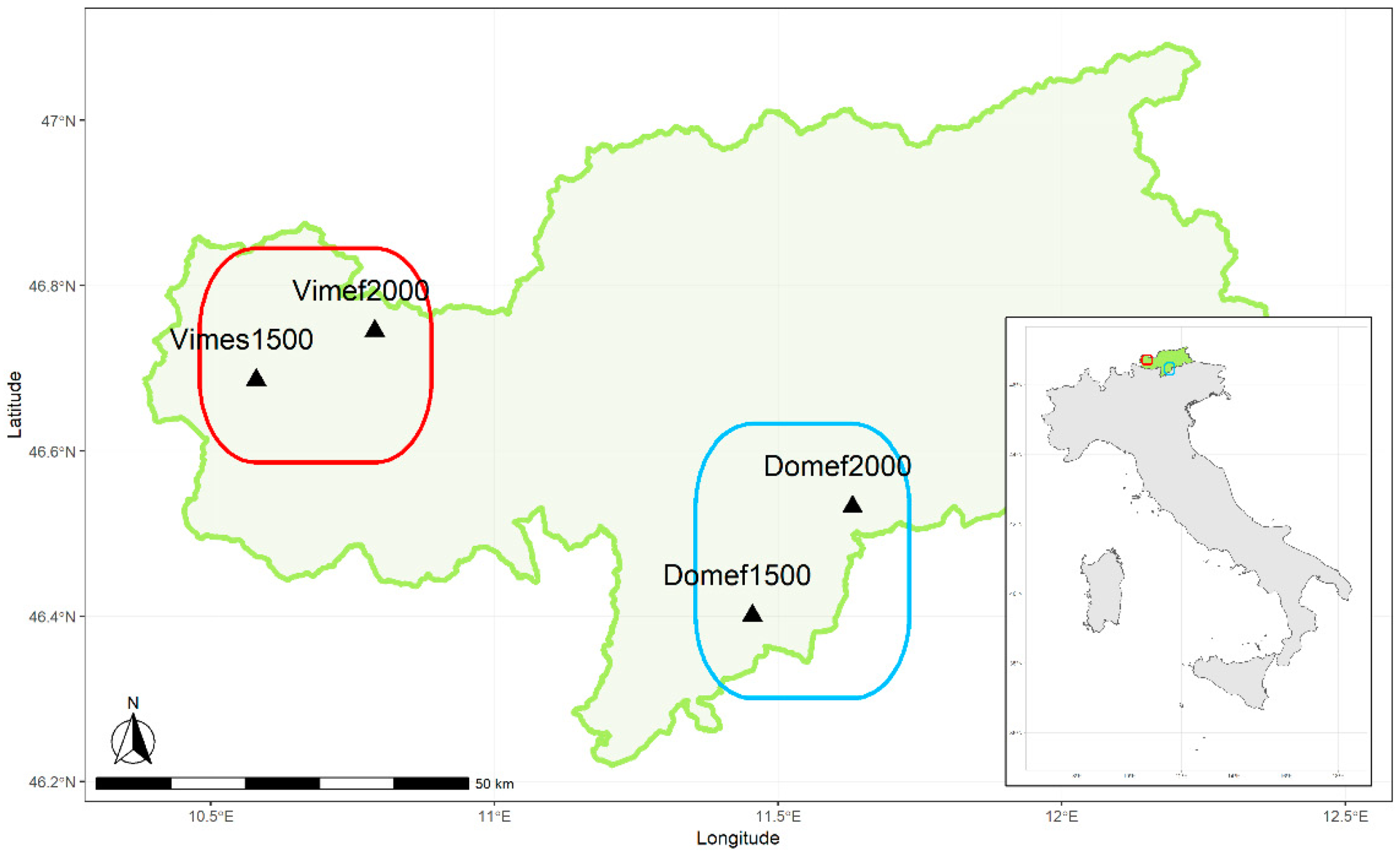
2.1. Study Site
2.2. Vegetation Index Selection
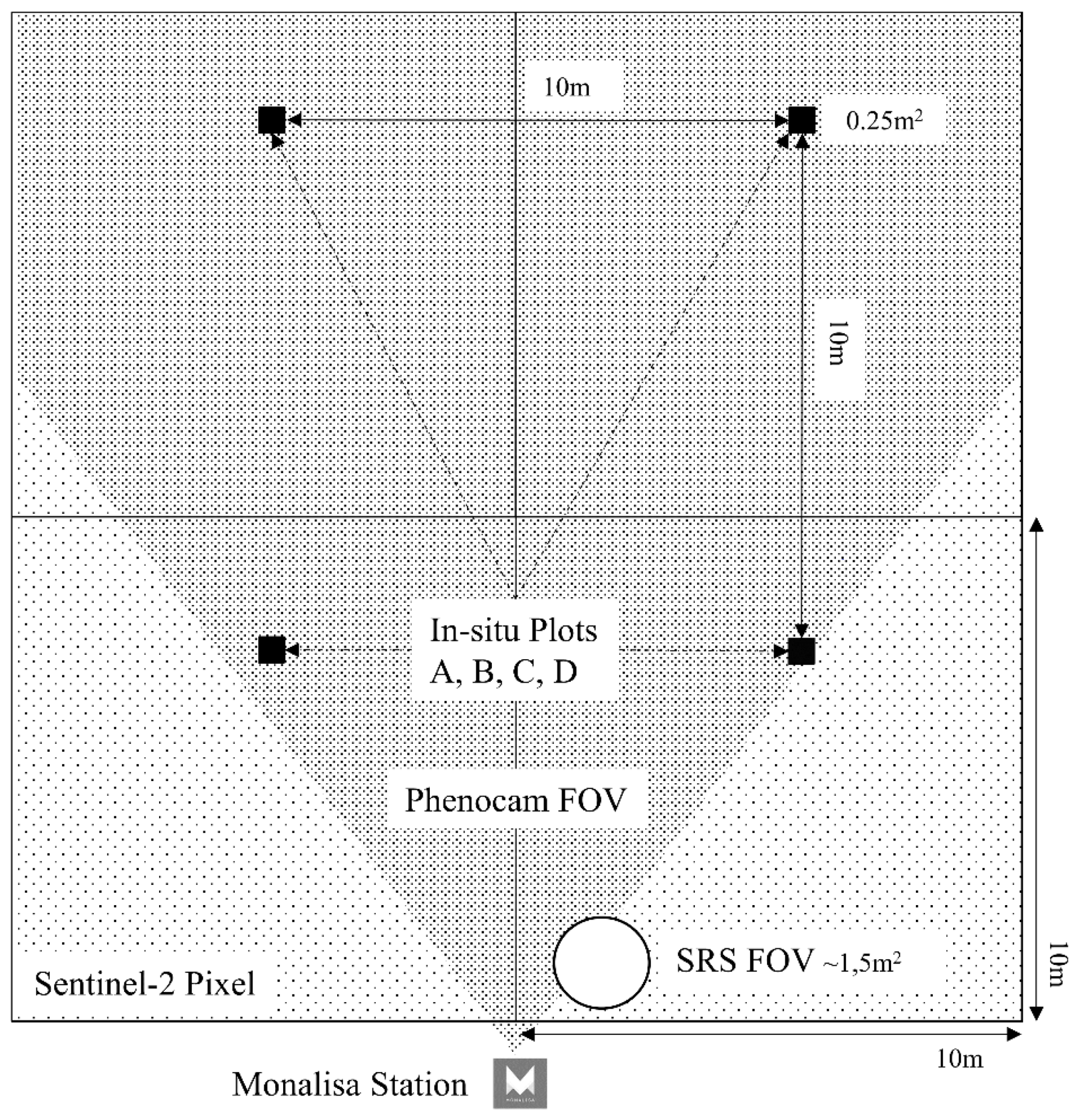
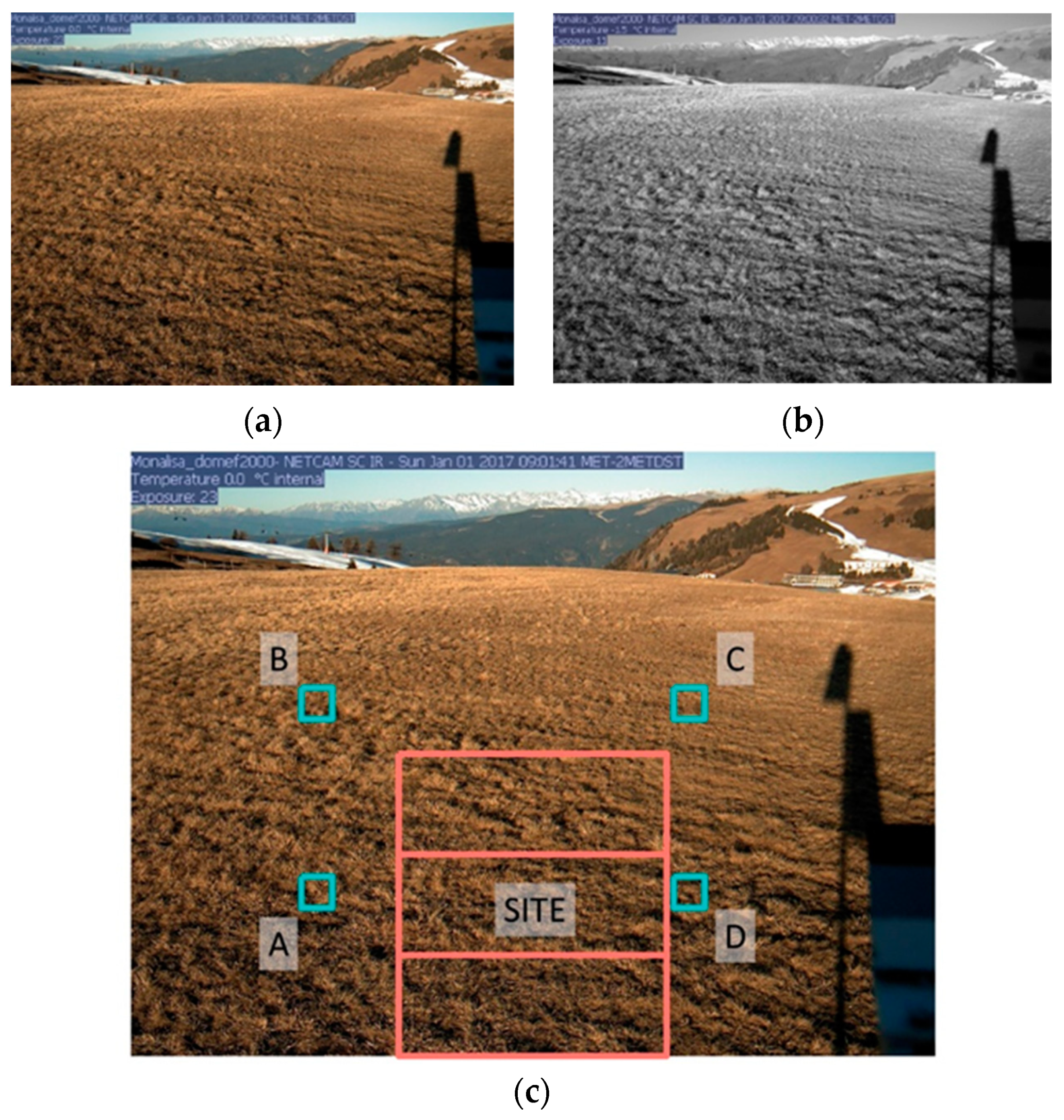
2.3. Sampling Design
2.4. Data
2.4.1. Sentinel-2 MSI
2.4.2. Phenocam
2.4.3. Spectral Reflectance Sensor (SRS)
2.4.4. Spectroradiometer
2.5. NDVI Signal Comparison
3. Results
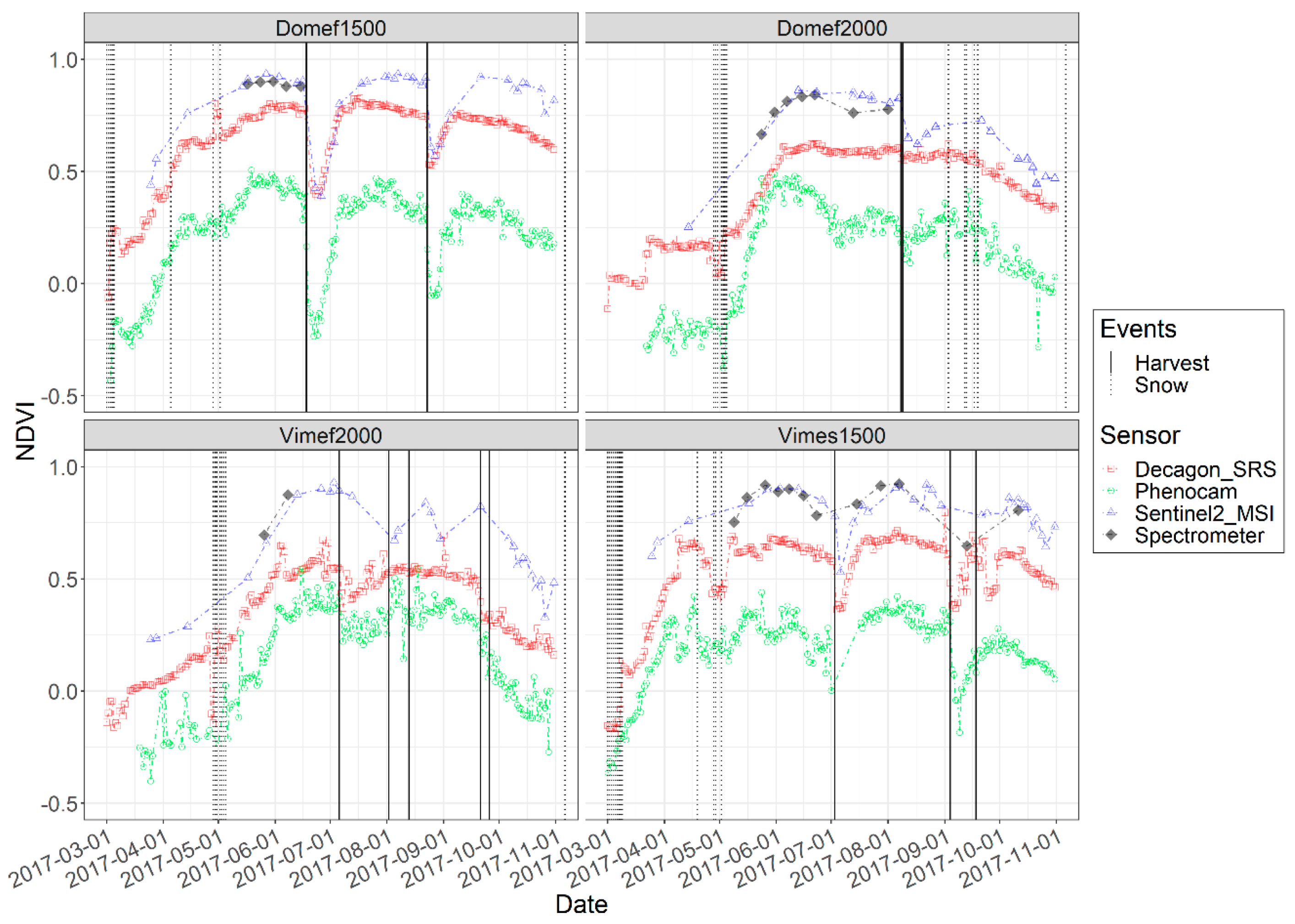
3.1. Optical Responses by Site
3.2. Optical Responses by Plot
4. Discussion
4.1. Sensor Specifications and Geometry
4.2. Temporal and Spectral Resolution
4.3. Potential for Combined Sensor Appoach
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- White, R.P.; Murray, S.; Rohweder, M. Pilot Analysis of Global Ecosystems: Grassland Ecosystems; World Resources Institute: Washington, DC, USA, 2000. [Google Scholar]
- Ali, I.; Cawkwell, F.; Dwyer, E.; Barrett, B.; Green, S. Satellite remote sensing of grasslands: From observation to management. J. Plant Ecol. 2016, 9, 649–671. [Google Scholar] [CrossRef]
- European Economic Area (EEA). CLC Addendum to CLC2006 Technical Guidelines—Final Draft. Available online: https://land.copernicus.eu/user-corner/technical-library/Addendum_finaldraft_v2_August_2014.pdf (accessed on 29 January 2019).
- French, K.E.; Tkacz, A.; Turnbull, L.A. Conversion of grassland to arable decreases microbial diversity and alters community composition. Appl. Soil Ecol. 2017, 110, 43–52. [Google Scholar] [CrossRef]
- Lüth, C.; Tasser, E.; Niedrist, G.; Via, J.D.; Tappeiner, U. Plant communities of mountain grasslands in a broad cross-section of the Eastern Alps. Flora 2011, 206, 433–443. [Google Scholar] [CrossRef]
- Xu, D.; Guo, X. Some insights on grassland health assessment based on remote sensing. Sensors 2015, 15, 3070–3089. [Google Scholar] [CrossRef] [PubMed]
- Ali, I.; Greifeneder, F.; Stamenkovic, J.; Neumann, M.; Notarnicola, C. Review of Machine Learning Approaches for Biomass and Soil Moisture Retrievals from Remote Sensing Data. Remote Sens. 2015, 7, 16398–16421. [Google Scholar] [CrossRef]
- Jankowska-Huflejt, H. The function of permanent grasslands in water resources protection. J. Water Land Dev. 2006, 10, 233. [Google Scholar] [CrossRef]
- Theurillat, J.P.; Guisan, A. Potential Impact of Climate Change on Vegetation in the European Alps: A Review. Clim. Chang. 2001, 50, 77–109. [Google Scholar] [CrossRef]
- Della Chiesa, S.; Bertoldi, G.; Niedrist, G.; Obojes, N.; Endrizzi, S.; Albertson, J.D.; Wohlfahrt, G.; Hörtnagl, L.; Tappeiner, U. Modelling changes in grassland hydrological cycling along an elevational gradient in the Alps. Ecohydrology 2014, 7, 1453–1473. [Google Scholar] [CrossRef]
- Niedrist, G.; Tasser, E.; Bertoldi, G.; Della Chiesa, S.; Obojes, N.; Egarter-Vigl, L.; Tappeiner, U. Down to future: Transplanted mountain meadows react with increasing phytomass or shifting species composition. Flora 2016, 224, 172–182. [Google Scholar] [CrossRef]
- Gitelson, A.A.; Merzlyak, M.N.; Lichtenthaler, H.K. Detection of Red Edge Position and Chlorophyll Content by Reflectance Measurements near 700 nm. J. Plant Physiol. 1996, 148, 501–508. [Google Scholar] [CrossRef]
- Horler, D.N.H.; Dockray, M.; Barber, J. The red edge of plant leaf reflectance. Int. J. Remote Sens. 1983, 4, 273–288. [Google Scholar] [CrossRef]
- Rouse, J.W.; Haas, R.H.; Schell, J.A.; Deering, D.W. Monitoring Vegetation Systems in the Great Plains with ERTS. In Proceedings of the 3rd ERTS Symposium, Washington, DC, USA, 10–14 December 1973; pp. 309–317. [Google Scholar]
- Browning, D.; Karl, J.; Morin, D.; Richardson, A.; Tweedie, C. Phenocams Bridge the Gap between Field and Satellite Observations in an Arid Grassland Ecosystem. Remote Sens. 2017, 9, 1071. [Google Scholar] [CrossRef]
- Enciso, J.; Maeda, M.; Landivar, J.; Jung, J.; Chang, A. A ground based platform for high throughput phenotyping. Comput. Electron. Agric. 2017, 141, 286–291. [Google Scholar] [CrossRef]
- Gamon, J.A.; Kovalchuck, O.; Wong, C.Y.S.; Harris, A.; Garrity, S.R. Monitoring seasonal and diurnal changes in photosynthetic pigments with automated PRI and NDVI sensors. Biogeosciences 2015, 12, 4149–4159. [Google Scholar] [CrossRef]
- Richardson, A.D.; Braswell, B.H.; Hollinger, D.Y.; Jenkins, J.P.; Ollinger, S.V. Near-surface remote sensing of spatial and temporal variation in canopy phenology. Ecol. Appl. 2009, 19, 1417–1428. [Google Scholar] [CrossRef] [PubMed]
- Filippa, G.; Cremonese, E.; Migliavacca, M.; Galvagno, M.; Sonnentag, O.; Humphreys, E.; Hufkens, K.; Ryu, Y.; Verfaillie, J.; Di Morra Cella, U.; et al. NDVI derived from near-infrared-enabled digital cameras: Applicability across different plant functional types. Agric. For. Meteorol. 2018, 249, 275–285. [Google Scholar] [CrossRef]
- Brown, T.B.; Hultine, K.R.; Steltzer, H.; Denny, E.G.; Denslow, M.W.; Granados, J.; Henderson, S.; Moore, D.; Nagai, S.; SanClements, M.; et al. Using phenocams to monitor our changing Earth: Toward a global phenocam network. Front. Ecol. Environ. 2016, 14, 84–93. [Google Scholar] [CrossRef]
- Srbinovska, M.; Gavrovski, C.; Dimcev, V.; Krkoleva, A.; Borozan, V. Environmental parameters monitoring in precision agriculture using wireless sensor networks. J. Clean. Prod. 2015, 88, 297–307. [Google Scholar] [CrossRef]
- Aqeel-ur-Rehman; Abbasi, A.Z.; Islam, N.; Shaikh, Z.A. A review of wireless sensors and networks’ applications in agriculture. Comput. Stand. Interfaces 2014, 36, 263–270. [Google Scholar] [CrossRef]
- Reed, C.; Botts, M.; Davidson, J.; Percivall, G. Ogc® sensor web enablement: Overview and high level achhitecture. In Proceedings of the 2007 IEEE Autotestcon, Baltimore, MD, USA, 10–20 September 2007; pp. 372–380. [Google Scholar]
- Kulmala, M. Build a Global Earth observatory. Nature 2018, 553, 21–23. [Google Scholar] [CrossRef]
- Filippa, G.; Cremonese, E.; Migliavacca, M.; Galvagno, M.; Forkel, M.; Wingate, L.; Tomelleri, E.; Di Morra Cella, U.; Richardson, A.D. Phenopix: A R package for image-based vegetation phenology. Agric. For. Meteorol. 2016, 220, 141–150. [Google Scholar] [CrossRef]
- Petach, A.R.; Toomey, M.; Aubrecht, D.M.; Richardson, A.D. Monitoring vegetation phenology using an infrared-enabled security camera. Agric. For. Meteorol. 2014, 195–196, 143–151. [Google Scholar] [CrossRef]
- Cremonese, E.; Filippa, G.; Galvagno, M.; Siniscalco, C.; Oddi, L.; Di Morra Cella, U.; Migliavacca, M. Heat wave hinders green wave—The impact of climate extreme on the phenology of a mountain grassland. Agric. For. Meteorol. 2017, 247, 320–330. [Google Scholar] [CrossRef]
- Richardson, A.D.; Hufkens, K.; Milliman, T.; Aubrecht, D.M.; Chen, M.; Gray, J.M.; Johnston, M.R.; Keenan, T.F.; Klosterman, S.T.; Kosmala, M.; et al. Tracking vegetation phenology across diverse North American biomes using PhenoCam imagery. Sci. Data 2018, 5, 1–24. [Google Scholar] [CrossRef] [PubMed]
- Loveland, T.R.; Irons, J.R. Landsat 8—The plans, the reality, and the legacy. Remote Sens. Environ. 2016, 185, 1–6. [Google Scholar] [CrossRef]
- Deilami, K.; Hashim, M. Very High Resolution Optical Satellites for DEM Generation: A Review. Eur. J. Sci. Res. 2011, 49, 542–554. [Google Scholar]
- Malenovský, Z.; Rott, H.; Cihlar, J.; Schaepman, M.E.; García-Santos, G.; Fernandes, R.; Berger, M. Sentinels for science: Potential of Sentinel-1, -2, and -3 missions for scientific observations of ocean, cryosphere, and land. Remote Sens. Environ. 2012, 120, 91–101. [Google Scholar] [CrossRef]
- Li, J.; Roy, D. A Global Analysis of Sentinel-2A, Sentinel-2B and Landsat-8 Data Revisit Intervals and Implications for Terrestrial Monitoring. Remote Sens. 2017, 9, 902. [Google Scholar] [CrossRef]
- Drusch, M.; Del Bello, U.; Carlier, S.; Colin, O.; Fernandez, V.; Gascon, F.; Hoersch, B.; Isola, C.; Laberinti, P.; Martimort, P.; et al. Sentinel-2: ESA’s Optical High-Resolution Mission for GMES Operational Services. Remote Sens. Environ. 2012, 120, 25–36. [Google Scholar] [CrossRef]
- Lessio, A.; Fissore, V.; Borgogno-Mondino, E. Preliminary Tests and Results Concerning Integration of Sentinel-2 and Landsat-8 OLI for Crop Monitoring. J. Imaging 2017, 3, 49. [Google Scholar] [CrossRef]
- Stenzel, S.; Fassnacht, F.E.; Mack, B.; Schmidtlein, S. Identification of high nature value grassland with remote sensing and minimal field data. Ecol. Indic. 2017, 74, 28–38. [Google Scholar] [CrossRef]
- Lange, M.; Dechant, B.; Rebmann, C.; Vohland, M.; Cuntz, M.; Doktor, D. Validating MODIS and Sentinel-2 NDVI Products at a Temperate Deciduous Forest Site Using Two Independent Ground-Based Sensors. Sensors 2017, 17, 1855. [Google Scholar] [CrossRef] [PubMed]
- Jordan, C.F. Derivation of Leaf-Area Index from Quality of Light on the Forest Floor. Ecology 1969, 50, 663–666. [Google Scholar] [CrossRef]
- Peñuelas, J.; Filella, I. Visible and near-infrared reflectance techniques for diagnosing plant physiological status. Trends Plant Sci. 1998, 3, 151–156. [Google Scholar] [CrossRef]
- Gao, B.-C. NDWI—A normalized difference water index for remote sensing of vegetation liquid water from space. Remote Sens. Environ. 1996, 58, 257–266. [Google Scholar] [CrossRef]
- Gamon, J.A.; Peñuelas, J.; Field, C.B. A narrow-waveband spectral index that tracks diurnal changes in photosynthetic efficiency. Remote Sens. Environ. 1992, 41, 35–44. [Google Scholar] [CrossRef]
- Huete, A.; Liu, H.Q.; Batchily, K.; van Leeuwen, W. A comparison of vegetation indices over a global set of TM images for EOS-MODIS. Remote Sens. Environ. 1997, 59, 440–451. [Google Scholar] [CrossRef]
- Huete, A. A soil-adjusted vegetation index (SAVI). Remote Sens. Environ. 1988, 25, 295–309. [Google Scholar] [CrossRef]
- Kauth, R.J.; Thomas, G.S. The Tasselled Cap—A Graphic Description of the Spectral-Temporal Development of Agricultural Crops as Seen by LANDSAT. In Proceedings of the Symposium on Machine Processing of Remotely Sensed Data, West Lafayette, IN, USA, 29 June–1 July 1976; pp. 4B41–4B51. [Google Scholar]
- Wang, Y.; Wu, G.; Deng, L.; Tang, Z.; Wang, K.; Sun, W.; Shangguan, Z. Prediction of aboveground grassland biomass on the Loess Plateau, China, using a random forest algorithm. Sci. Rep. 2017, 7, 6940. [Google Scholar] [CrossRef]
- Mänd, P.; Hallik, L.; Peñuelas, J.; Nilson, T.; Duce, P.; Emmett, B.A.; Beier, C.; Estiarte, M.; Garadnai, J.; Kalapos, T. Responses of the reflectance indices PRI and NDVI to experimental warming and drought in European shrublands along a north–south climatic gradient. Remote Sens. Environ. 2010, 114, 626–636. [Google Scholar] [CrossRef]
- Gu, Y.; Wylie, B.K.; Howard, D.M.; Phuyal, K.P.; Ji, L. NDVI saturation adjustment: A new approach for improving cropland performance estimates in the Greater Platte River Basin, USA. Ecol. Indic. 2013, 30, 1–6. [Google Scholar] [CrossRef]
- Menzel, A.; Sparks, T.H.; Estrella, N.; Koch, E.; Aasa, A.; Ahas, R.; Als-Kübler, K.; Bissolli, P.; Braslavska, O.; Briede, A.; et al. European phenological response to climate change matches the warming pattern. Glob. Chang. Biol. 2006, 12, 1969–1976. [Google Scholar] [CrossRef]
- Cornelius, C.; Estrella, N.; Franz, H.; Menzel, A. Linking altitudinal gradients and temperature responses of plant phenology in the Bavarian Alps. Plant Biol. 2013, 15 (Suppl. 1), 57–69. [Google Scholar] [CrossRef]
- Julitta, T.; Cremonese, E.; Migliavacca, M.; Colombo, R.; Galvagno, M.; Siniscalco, C.; Rossini, M.; Fava, F.; Cogliati, S.; Di Morra Cella, U.; et al. Using digital camera images to analyse snowmelt and phenology of a subalpine grassland. Agric. For. Meteorol. 2014, 198–199, 116–125. [Google Scholar] [CrossRef]
- Gómez, C.; White, J.C.; Wulder, M.A. Optical remotely sensed time series data for land cover classification: A review. ISPRS J. Photogramm. Remote Sens. 2016, 116, 55–72. [Google Scholar] [CrossRef]
- Xue, J.; Su, B. Significant Remote Sensing Vegetation Indices: A Review of Developments and Applications. J. Sens. 2017, 2017, 1353691. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing. R Foundation for Statistical Computing. Available online: https://www.R-project.org/ (accessed on 29 January 2019).
- Wickham, H.; Grolemund, G. R for Data Science; O’Reilly Media Inc.: Sebastopol, CA, USA, 2017. [Google Scholar]
- Wickham, H. Tidyverse: Easily Install and Load the ’Tidyverse’. R Package Version 1.2.1. Available online: https://CRAN.R-project.org/package=tidyverse (accessed on 29 January 2019).
- Loius, J.; Debaecker, V.; Pflug, B.; Main-Knorn, M.; Bieniarz, J.; Müller-Wilm, U.; Cadau, E.; Gascon, F. SENTINEL-2 SEN2COR: L2A Processor for Users. Available online: http://elib.dlr.de/107381/1/LPS2016_sm10_3louis.pdf (accessed on 29 January 2019).
- Farr, T.G.; Kobrick, M. Shuttle radar topography mission produces a wealth of data. Eos Trans. Am. Geophys. Union 2000, 81, 583–585. [Google Scholar] [CrossRef]
- Stardot Technologies. NetCam SC H.264 Megapixel Hypbrid IP Camera: User’s Manual. Available online: http://www.stardot.com/manuals (accessed on 12 September 2018).
- Ooms, J. Tesseract: Open Source OCR Engine for R. Available online: https://CRAN.R-project.org/package=tesseract (accessed on 29 January 2019).
- Liu, Y.; Hill, M.J.; Zhang, X.; Wang, Z.; Richardson, A.D.; Hufkens, K.; Filippa, G.; Baldocchi, D.D.; Ma, S.; Verfaillie, J.; et al. Using data from Landsat, MODIS, VIIRS and PhenoCams to monitor the phenology of California oak/grass savanna and open grassland across spatial scales. Agric. For. Meteorol. 2017, 237–238, 311–325. [Google Scholar] [CrossRef]
- Meter Group Inc. SRS Spectral Relflectance Sensor: Operator’s Manual. Available online: http://manuals.decagon.com/Manuals/14597_SRS_Web.pdf (accessed on 12 September 2018).
- Open Geospatial Consortium. OGC® Sensor Observation Service Interface Standard. Available online: http://www.opengeospatial.org/standards/sos (accessed on 2 October 2018).
- Spectra Vista Corporation. SVC HR-1024i/SVC HR-768i User Manual rev. 1.5. Available online: http://twiki.cis.rit.edu/twiki/pub/Main/SepTember19/SVC_HR1024i_HR768i_User_Manual.pdf (accessed on 12 September 2018).
- Hyndman, R.J.; Khandakar, Y. Automatic Time Series Forecasting: The forecast Package for R. J. Stat. Softw. 2008, 27, 1–22. [Google Scholar] [CrossRef]
- Härdle, W.K.; Simar, L. Applied Multivariate Statistics, 3rd ed.; Springer: Heidelberg, Germany, 2012. [Google Scholar]
- Klosterman, S.T.; Hufkens, K.; Gray, J.M.; Melaas, E.; Sonnentag, O.; Lavine, I.; Mitchell, L.; Norman, R.; Friedl, M.A.; Richardson, A.D. Evaluating remote sensing of deciduous forest phenology at multiple spatial scales using PhenoCam imagery. Biogeosciences 2014, 11, 4305–4320. [Google Scholar] [CrossRef]
- Sonnentag, O.; Hufkens, K.; Teshera-Sterne, C.; Young, A.M.; Friedl, M.; Braswell, B.H.; Milliman, T.; O’Keefe, J.; Richardson, A.D. Digital repeat photography for phenological research in forest ecosystems. Agric. For. Meteorol. 2012, 152, 159–177. [Google Scholar] [CrossRef]
- Hufkens, K.; Friedl, M.; Sonnentag, O.; Braswell, B.H.; Milliman, T.; Richardson, A.D. Linking near-surface and satellite remote sensing measurements of deciduous broadleaf forest phenology. Remote Sens. Environ. 2012, 117, 307–321. [Google Scholar] [CrossRef]
- Balzarolo, M.; Vescovo, L.; Hammerle, A.; Gianelle, D.; Papale, D.; Tomelleri, E.; Wohlfahrt, G. On the relationship between ecosystem-scale hyperspectral reflectance and CO2 exchange in European mountain grasslands. Biogeosciences 2015, 12, 3089–3108. [Google Scholar] [CrossRef]
- Van der Werff, H.; van der Meer, F. Sentinel-2A MSI and Landsat 8 OLI Provide Data Continuity for Geological Remote Sensing. Remote Sens. 2016, 8, 883. [Google Scholar] [CrossRef]
- Gatebe, C.K.; King, M.D. Airborne spectral BRDF of various surface types (ocean, vegetation, snow, desert, wetlands, cloud decks, smoke layers) for remote sensing applications. Remote Sens. Environ. 2016, 179, 131–148. [Google Scholar] [CrossRef]
- Román, M.O.; Gatebe, C.K.; Schaaf, C.B.; Poudyal, R.; Wang, Z.; King, M.D. Variability in surface BRDF at different spatial scales (30 m–500 m) over a mixed agricultural landscape as retrieved from airborne and satellite spectral measurements. Remote Sens. Environ. 2011, 115, 2184–2203. [Google Scholar] [CrossRef]


| Station | Sensor | Raw Acquisitions | Plot | Site | |||
|---|---|---|---|---|---|---|---|
| A | B | C | D | ||||
| Domef1500 | Decagon_SRS | 16,740 | 234 | ||||
| Phenocam | 4283 | 234 | 237 | 237 | 238 | 241 | |
| Sentinel2_MSI | 70 | 37 | 37 | 37 | 37 | 37 | |
| Spectrometer | 5 | 5 | 5 | 5 | 5 | 5 | |
| Domef2000 | Decagon_SRS | 16,895 | 222 | ||||
| Phenocam | 4556 | 202 | 197 | 205 | 203 | 207 | |
| Sentinel2_MSI | 122 | 40 | 39 | 40 | 40 | 40 | |
| Spectrometer | 7 | 7 | 7 | 7 | 7 | 7 | |
| Vimef2000 | Decagon_SRS | 16,190 | 228 | ||||
| Phenocam | 4681 | 188 | 186 | 171 | 166 | 195 | |
| Sentinel2_MSI | 70 | 27 | 27 | 27 | 27 | 27 | |
| Spectrometer | 2 | 2 | 2 | 2 | 2 | 2 | |
| Vimes1500 | Decagon_SRS | 16,615 | 229 | ||||
| Phenocam | 4587 | 215 | 213 | 213 | 207 | 216 | |
| Sentinel2_MSI | 70 | 33 | 33 | 33 | 33 | 33 | |
| Spectrometer | 12 | 12 | 12 | 12 | 12 | 12 | |
| Station | Sensors | Sensors | N | Plot | Site | |||
|---|---|---|---|---|---|---|---|---|
| Scale 1 | Scale 2 | A | B | C | D | |||
| Domef 1500 | Decagon_SRS | Sentinel2 | 219 | 0.74 *** | ||||
| Phenocam | Decagon_SRS | 241 | 0.86 *** | |||||
| Sentinel2 | 219 | 0.76 *** | 0.71 *** | 0.67 *** | 0.81 *** | 0.78 *** | ||
| Spectrometer | Decagon_SRS | 28 | 0.07 | |||||
| Phenocam | 28 | 0.29 *** | 0.03 | 0.04 | 0.04 | 0.34 *** | ||
| Sentinel2 | 28 | 0.38*** | 0.47 *** | 0.31 *** | 0.54 *** | 0.88 *** | ||
| Domef 2000 | Decagon_SRS | Sentinel2 | 199 | 0.82 *** | ||||
| Phenocam | Decagon_SRS | 222 | 0.82 *** | |||||
| Sentinel2 | 199 | 0.76 *** | 0.8 *** | 0.78 *** | 0.6 *** | 0.76 *** | ||
| Spectrometer | Decagon_SRS | 68 | 0.68 *** | |||||
| Phenocam | 68 | 0.19 *** | 0.03 | 0.07 ** | 0.23 *** | 0.07 ** | ||
| Sentinel2 | 68 | 0.38 *** | 0.4 *** | 0.72 *** | 0 | 0.57 *** | ||
| Vimef 2000 | Decagon_SRS | Sentinel2 | 219 | 0.78 *** | ||||
| Phenocam | Decagon_SRS | 223 | 0.87 *** | |||||
| Sentinel2 | 217 | 0.8 *** | 0.82 *** | 0.78 *** | 0.8 *** | 0.78 *** | ||
| Spectrometer | Decagon_SRS | 12 | 0.49 *** | |||||
| Phenocam | 12 | 0.76 *** | 0.76 *** | 0.77 *** | 0.74 *** | 0.76 *** | ||
| Sentinel2 | 12 | 1 *** | 1 *** | 1 *** | 1 *** | 1 *** | ||
| Vimes 1500 | Decagon_SRS | Sentinel2 | 206 | 0.46 *** | ||||
| Phenocam | Decagon_SRS | 230 | 0.72 *** | |||||
| Sentinel2 | 206 | 0.28 *** | 0.31 *** | 0.12 *** | 0.25 *** | 0.19 *** | ||
| Spectrometer | Decagon_SRS | 141 | 0.4 *** | |||||
| Phenocam | 141 | 0.47 *** | 0.52 *** | 0.39 *** | 0.49 *** | 0.5 *** | ||
| Sentinel2 | 141 | 0.54 *** | 0.58 *** | 0.48 *** | 0.44 *** | 0.53 *** | ||
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rossi, M.; Niedrist, G.; Asam, S.; Tonon, G.; Tomelleri, E.; Zebisch, M. A Comparison of the Signal from Diverse Optical Sensors for Monitoring Alpine Grassland Dynamics. Remote Sens. 2019, 11, 296. https://doi.org/10.3390/rs11030296
Rossi M, Niedrist G, Asam S, Tonon G, Tomelleri E, Zebisch M. A Comparison of the Signal from Diverse Optical Sensors for Monitoring Alpine Grassland Dynamics. Remote Sensing. 2019; 11(3):296. https://doi.org/10.3390/rs11030296
Chicago/Turabian StyleRossi, Mattia, Georg Niedrist, Sarah Asam, Giustino Tonon, Enrico Tomelleri, and Marc Zebisch. 2019. "A Comparison of the Signal from Diverse Optical Sensors for Monitoring Alpine Grassland Dynamics" Remote Sensing 11, no. 3: 296. https://doi.org/10.3390/rs11030296
APA StyleRossi, M., Niedrist, G., Asam, S., Tonon, G., Tomelleri, E., & Zebisch, M. (2019). A Comparison of the Signal from Diverse Optical Sensors for Monitoring Alpine Grassland Dynamics. Remote Sensing, 11(3), 296. https://doi.org/10.3390/rs11030296






