Quantifying Chlorophyll Fluorescence Parameters from Hyperspectral Reflectance at the Leaf Scale under Various Nitrogen Treatment Regimes in Winter Wheat
Abstract
1. Introduction
2. Materials and Methods
2.1. Experimental Design
2.2. Data Acquisition
2.2.1. Measurement of ChlF Parameters
2.2.2. Measurement of Leaf Physiological Parameters
2.2.3. Measurement of Leaf Reflectance
2.2.4. Optical Measurements of Pigments in Laboratory
2.3. Data Analysis and Utilization
2.3.1. Calculation of VIs
2.3.2. Methods for REP Extractions
2.3.3. Continuous Wavelet Analysis (CWA)
2.4. Statistical Analysis
3. Results
3.1. Relationships among Leaf Pigments, Physiological Indices, and ChlF Parameters
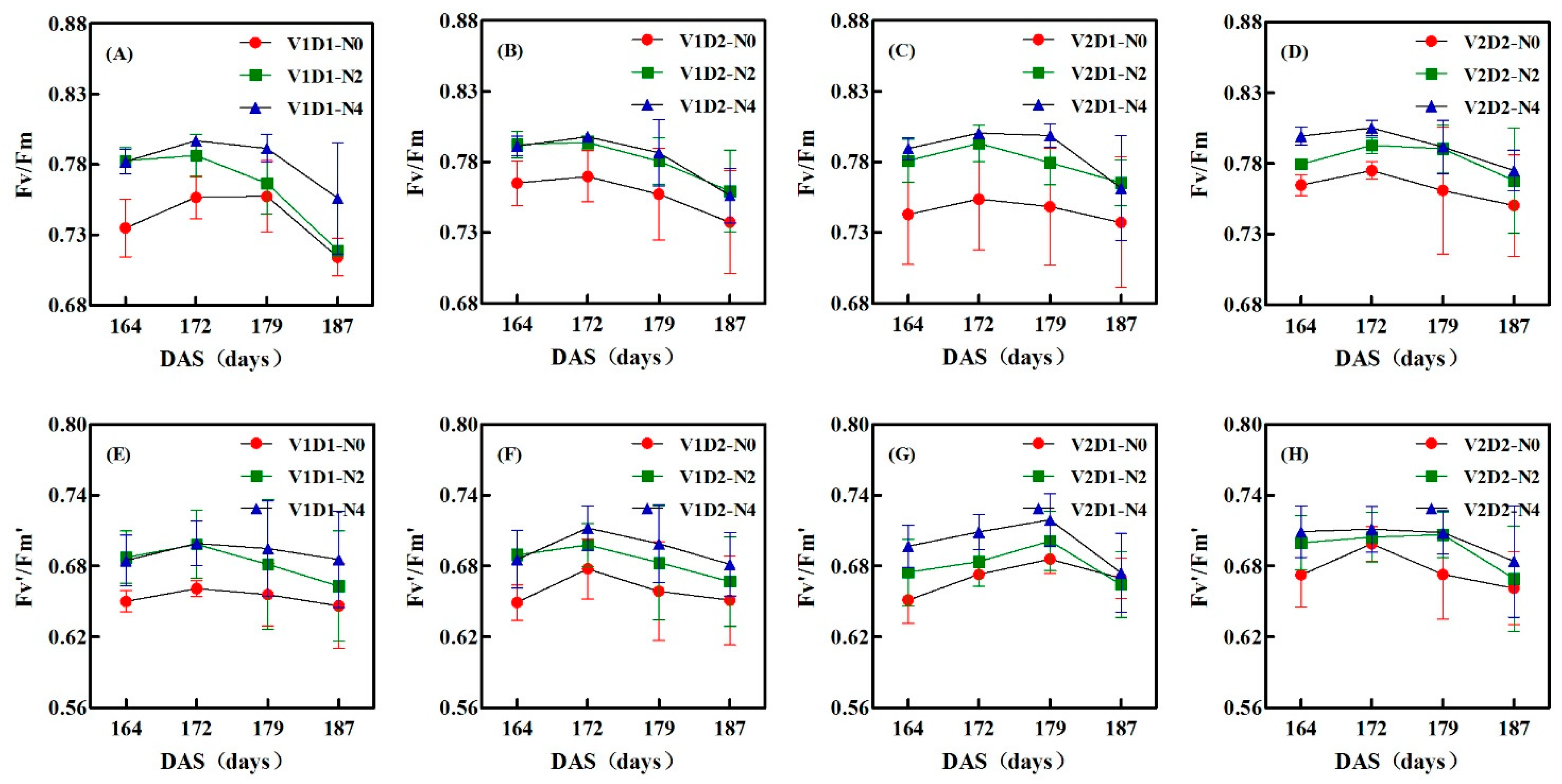
3.2. Dynamic Changes of Fv/Fm And Fv’/Fm’ under Different N Treatments during the Growing Season
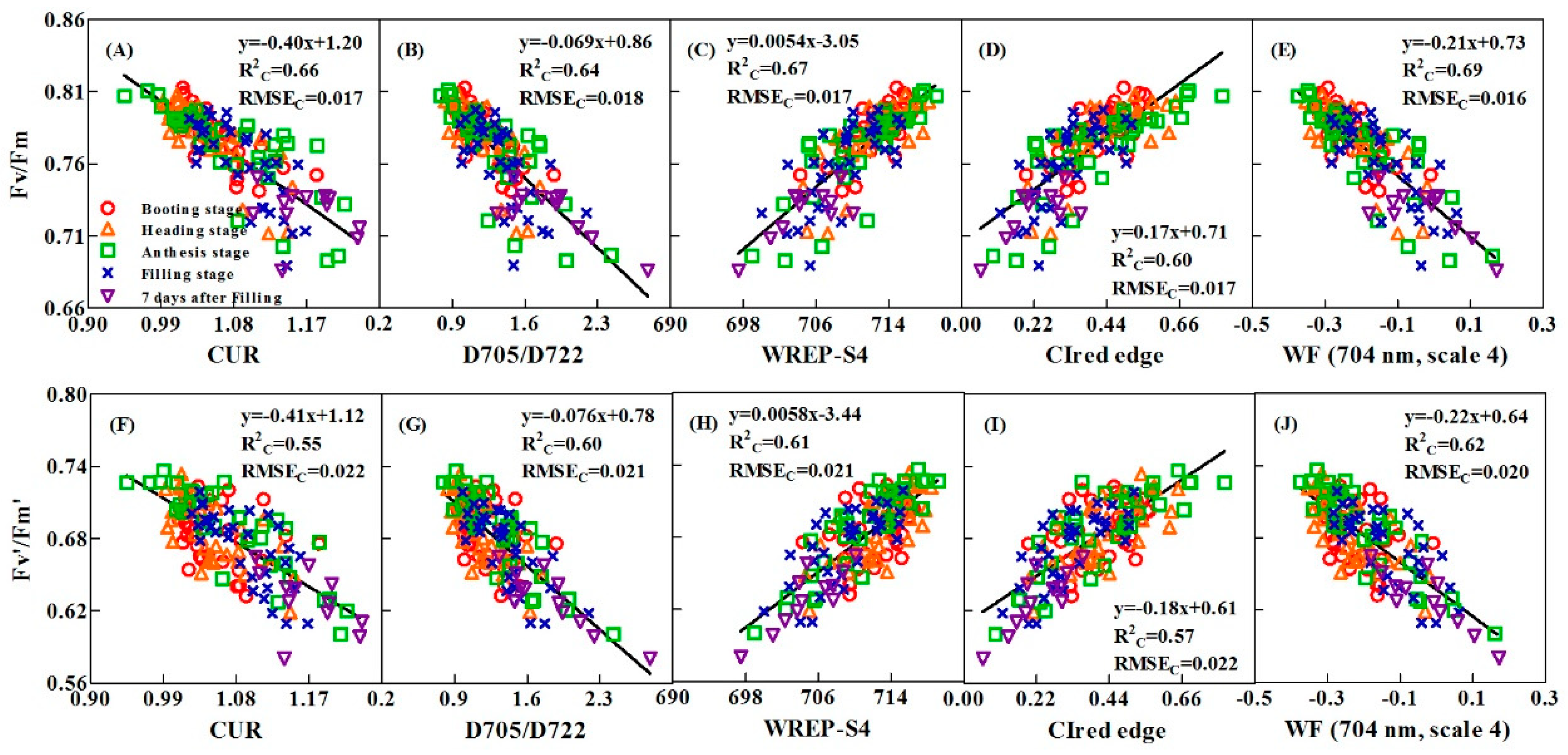
3.3. Semi-empirical Models for Estimating the Leaf Fv/Fm And Fv’/Fm’ Using Spectral Features
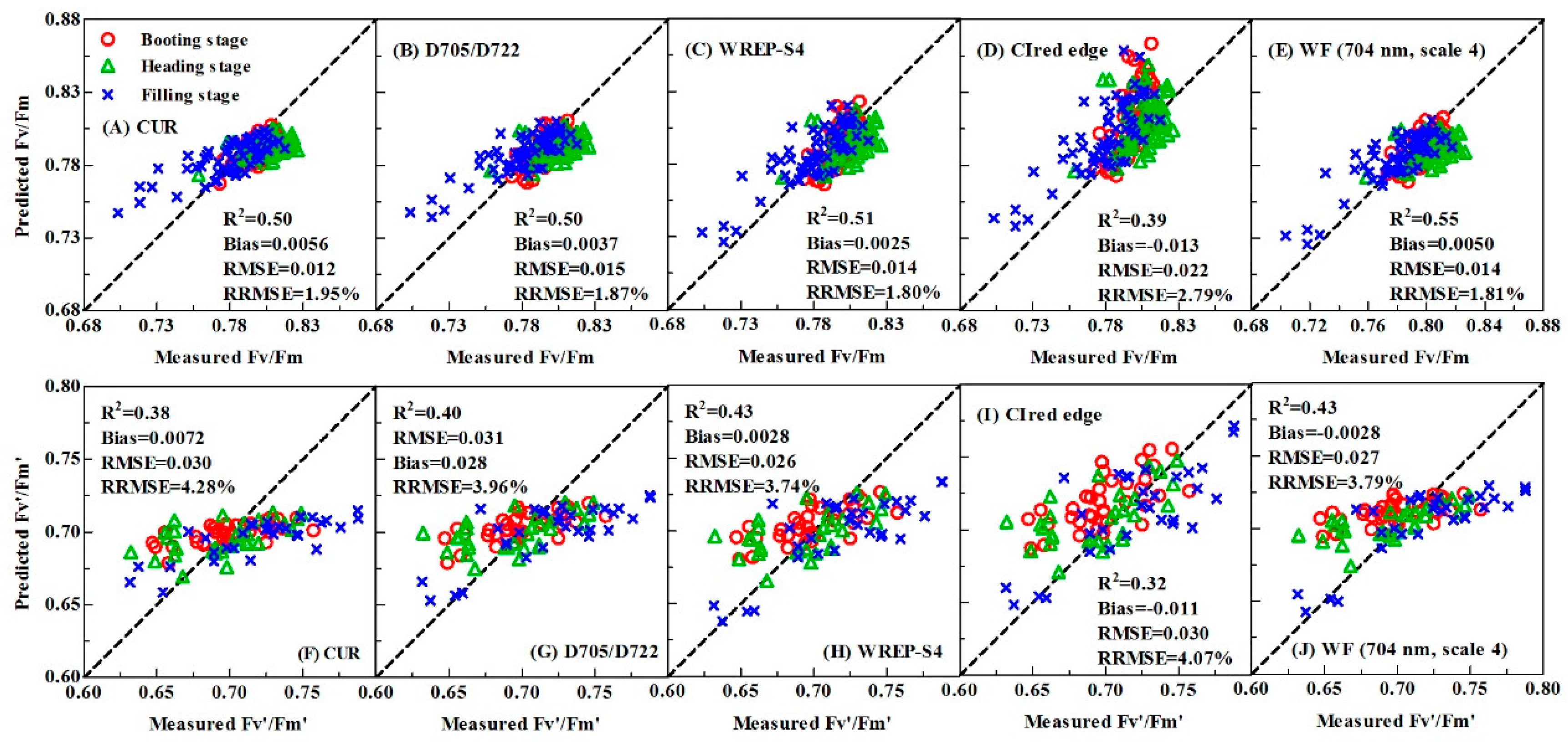
3.3.1. Performance of VIs-based Fv/Fm and Fv’/Fm’ Models in Calibration and Validation Datasets
3.3.2. Performance of REP for Predicting Leaf Fv/Fm and Fv’/Fm’ in Calibration and Validation
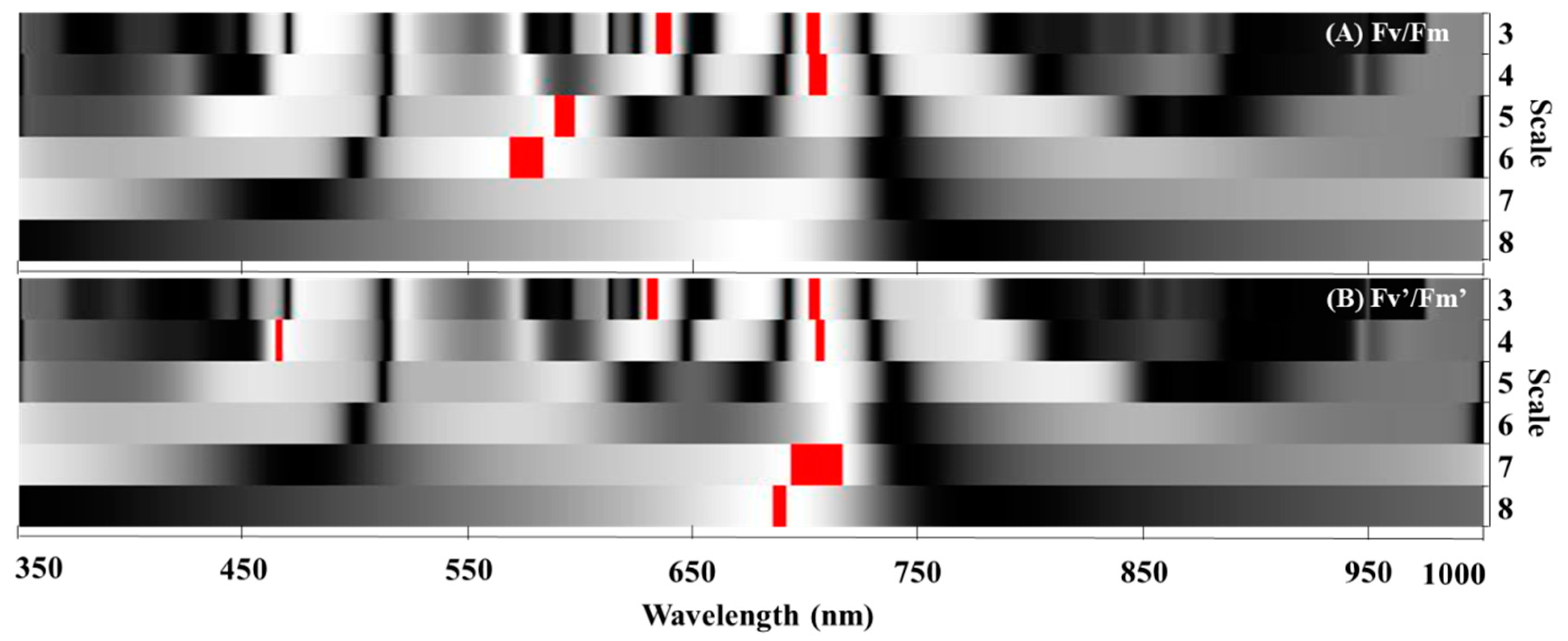
3.3.3. Models for Estimating the Leaf Fv/Fm and Fv’/Fm’ Based on Wavelet Features
4. Discussion
4.1. Signature Components of Leaf Chlf Parameters and Their Variation with N Regimes
4.2. Comparing VIs and REPs for Quantitatively Retrieving Fv/Fm and Fv’/Fm’
4.3. Comparison between CWA and Other Spectral Features for Estimating Fv/Fm and Fv’/Fm’
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Krause, G.H.; Weis, E. Chlorophyll fluorescence as a tool in plant physiology: II. Interpretation of fluorescence signals. Photosynth. Res. 1984, 5, 139–157. [Google Scholar] [CrossRef] [PubMed]
- Schreiber, U. Chlorophyll fluorescence as a tool in plant physiology I. The measuring systems. Photosynth. Res. 1983, 4, 361–373. [Google Scholar] [CrossRef] [PubMed]
- Portable Fluorometer PAM-2000 and Data Acquisition Software DA-2000; Heinz-Walz-GmbH: Effeltrich, Germany, 1993.
- Liu, R.X.; Wang, Y.H.; Chen, B.L.; Guo, W.Q.; Zhou, Z.G. Effects of nitrogen levels on photosynthesis and chlorophyll fluorescence characteristics under drought stress in cotton flowering and boll-forming stage. Acta Agron. Sin. 2008, 34, 675–683. [Google Scholar] [CrossRef]
- Baker, N.R. Chlorophyll fluorescence: A probe of photosynthesis In Vivo. Annu. Rev. Plant Biol. 2008, 59, 89–113. [Google Scholar] [CrossRef] [PubMed]
- Butler, W.L.; Kitajima, M. Fluorescence quenching in photosystem II of chloroplasts. Biochim. Biophys. Acta Bioenerg. 1975, 376, 116–125. [Google Scholar] [CrossRef]
- Feng, W.; He, L.; Zhang, H.Y.; Guo, B.B.; Zhu, Y.J.; Wang, C.Y.; Guo, T.C. Assessment of plant nitrogen status using chlorophyll fluorescence parameters of the upper leaves in winter wheat. Eur. J. Agron. 2015, 64, 78–87. [Google Scholar] [CrossRef]
- Faraloni, C.; Cutino, I.; Petruccelli, R.; Leva, A.R.; Lazzeri, S.; Torzillo, G. Chlorophyll fluorescence technique as a rapid tool for in vitro screening of olive cultivars (Olea europaea L.) tolerant to drought stress. Environ. Exp. Bot. 2011, 73, 49–56. [Google Scholar] [CrossRef]
- Bengtsson, G.B.; Schöner, R.; Lombardo, E.; Schöner, J.; Borge, G.I.A.; Bilger, W. Chlorophyll fluorescence for non-destructive measurement of flavonoids in broccoli. Postharvest Biol. Technol. 2006, 39, 291–298. [Google Scholar] [CrossRef]
- Netto, A.T.; Campostrini, E.; de Oliveira, J.G.; Bressan-Smith, R.E. Photosynthetic pigments, nitrogen, chlorophyll a fluorescence and SPAD-502 readings in coffee leaves. Sci. Hortic. 2005, 104, 199–209. [Google Scholar] [CrossRef]
- Ač, A.; Malenovsky, Z.; Olejnícková, J.; Galle, A.; Rascher, U.; Mohammed, G. Meta-analysis assessing potential of steady-state chlorophyll fluorescence for remote sensing detection of plant water, temperature and nitrogen stress. Remote Sens. Environ. 2015, 168, 420–436. [Google Scholar] [CrossRef]
- Zarco-Tejada, P.J.; Miller, J.R.; Mohammed, G.H.; Noland, T.L. Chlorophyll fluorescence effects on vegetation apparent reflectance: I. Leaf-level measurements and model simulation. Remote Sens Environ. 2000, 74, 582–595. [Google Scholar] [CrossRef]
- Zarco-Tejada, P.J.; Miller, J.R.; Mohammed, G.H.; Noland, T.L.; Sampson, P.H. Chlorophyll Fluorescence Effects on Vegetation Apparent Reflectance: II. Laboratory and Airborne Canopy-Level Measurements with Hyperspectral Data. Remote Sens Environ. 2000, 74, 596–608. [Google Scholar] [CrossRef]
- Zarco-Tejada, P.J.; Pushnik, J.C.; Dobrowski, S.; Ustin, S.L. Steady-state chlorophyll a fluorescence detection from canopy derivative reflectance and double-peak red-edge effects. Remote Sens Environ. 2003, 84, 283–294. [Google Scholar] [CrossRef]
- Dobrowski, S.Z.; Pushnik, J.C.; Zarco-Tejada, P.J.; Ustin, S.L. Simple reflectance indices track heat and water stress-induced changes in steady-state chlorophyll fluorescence at the canopy scale. Remote Sens Environ. 2005, 97, 403–414. [Google Scholar] [CrossRef]
- Pérez-Priego, O.; Zarco-Tejada, P.J.; Miller, J.R.; Sepulcre-Canto, G.; Fereres, E. Detection of water stress in orchard trees with a high-resolution spectrometer through chlorophyll fluorescence in-filling of the O2-A band. IEEE Trans. Geosci. Remote Sens. 2005, 43, 2860–2869. [Google Scholar] [CrossRef]
- Zarco-Tejada, P.J.; Miller, J.R.; Mohammed, G.H.; Noland, T.L.; Sampson, P.H. Estimation of chlorophyll fluorescence under natural illumination from hyperspectral data. Int. J. Appl. Earth Obs. Geoinf. 2001, 3, 321–327. [Google Scholar] [CrossRef]
- Gamon, J.A.; Serrano, L.; Surfus, J.S. The photochemical reflectance index: An optical indicator of photosynthetic radiation-use efficiency across species, functional types, and nutrient levels. Oecologia 1997, 112, 492–501. [Google Scholar] [CrossRef]
- Li, D.; Cheng, T.; Zhou, K.; Zheng, H.B.; Yao, X.; Tian, Y.C.; Zhu, Y.; Cao, W.X. WREP: A wavelet-based technique for extracting the red edge position from reflectance spectra for estimating leaf and canopy chlorophyll contents of cereal crops. ISPRS J. Photogram. Remote Sens. 2017, 129, 103–117. [Google Scholar] [CrossRef]
- Guyot, G.; Baret, F. Utilisation de la haute resolution spectrale pour suivre l’etat des couverts vegetaux. Spect. Signat. Objects Remote Sens. 1988, 287, 279. [Google Scholar]
- Cho, M.A.; Skidmore, A.K. A new technique for extracting the red edge position from hyperspectral data: The linear extrapolation method. Remote Sens. Environ. 2006, 101, 181–193. [Google Scholar] [CrossRef]
- Dawson, T.P.; Curran, P.J. Technical note: A new technique for interpolating the reflectance red edge position. Int. J. Remote Sens. 1998, 19, 2133–2139. [Google Scholar] [CrossRef]
- Cheng, T.; Rivard, B.; Sánchez-Azofeifa, A.G.; Féret, J.B.; Jacquemoud, S.; Ustin, S.L. Predicting leaf gravimetric water content from foliar reflectance across a range of plant species using continuous wavelet analysis. J. Plant Physiol. 2012, 169, 1134–1142. [Google Scholar] [CrossRef] [PubMed]
- Torrence, C.; Compo, G.P. A practical guide to wavelet analysis. Bull. Am. Meteorol. Soc. 1998, 79, 61–78. [Google Scholar] [CrossRef]
- Cheng, T.; Rivard, B.; Sanchez-Azofeifa, A.G.; Feret, J.B.; Jacquemoud, S.; Ustin, S.L. Deriving leaf mass per area (LMA) from foliar reflectance across a variety of plant species using continuous wavelet analysis. ISPRS J. Photogram. Remote Sens. 2014, 87, 28–38. [Google Scholar] [CrossRef]
- Meroni, M.; Rossini, M.; Guanter, L.; Alonso, L.; Rascher, U.; Colombo, R.; Moreno, J. Remote sensing of solar-induced chlorophyll fluorescence: Review of methods and applications. Remote Sens. Environ. 2009, 113, 2037–2051. [Google Scholar] [CrossRef]
- Lichtenthaler, H.K.; Wellburn, A.R. Determinations of total carotenoids and chlorophyll a and b of leaf extracts in different solvents. Biochem. Soc. Trans. 1983, 603, 591–592. [Google Scholar] [CrossRef]
- Calatayud, Á.; Bautista, A.S.; Pascual, B.; Maroto, J.V.; López-Galarza, S. Use of chlorophyll fluorescence imaging as diagnostic technique to predict compatibility in melon graft. Sci Hortic. 2013, 149, 13–18. [Google Scholar] [CrossRef]
- Sharma, D.K.; Fernández, J.O.; Rosenqvist, E.; Ottosen, C.O.; Andersen, S.B. Genotypic response of detached leaves versus intact plants for chlorophyll fluorescence parameters under high temperature stress in wheat. J. Plant Physiol. 2014, 171, 576–586. [Google Scholar] [CrossRef]
- Cheng, T.; Riaño, D.; Ustin, S.L. Detecting diurnal and seasonal variation in canopy water content of nut tree orchards from airborne imaging spectroscopy data using continuous wavelet analysis. Remote Sens. Environ. 2014, 143, 39–53. [Google Scholar] [CrossRef]
- Li, D.; Wang, X.; Zheng, H.; Zhou, K.; Yao, X.; Tian, Y.; Zhu, Y.; Cao, W.; Cheng, T. Estimation of area- and mass-based leaf nitrogen contents of wheat and rice crops from water-removed spectra using continuous wavelet analysis. Plant Methods 2018, 14, 76. [Google Scholar] [CrossRef]
- Bruce, L.M.; Li, J. Wavelets for computationally efficient hyperspectral derivative analysis. IEEE Trans. Geosci. Remote Sens. 2001, 39, 1540–1546. [Google Scholar] [CrossRef]
- Peñuelas, J.; Baret, F.; Filella, I. Semi-Empirical Indices to Assess Carotenoids/Chlorophyll-a Ratio from Leaf Spectral Reflectance. Photosynthetica 1995, 31, 221–230. [Google Scholar]
- Dash, J.; Curran, P.J. The MERIS terrestrial chlorophyll index. Int. J. Remote Sens. 2004, 25, 5403–5413. [Google Scholar] [CrossRef]
- Gitelson, A.A.; Gritz, Y.; Merzlyak, M.N. Relationships between leaf chlorophyll content and spectral reflectance and algorithms for non-destructive chlorophyll assessment in higher plant leaves. J. Plant Physiol. 2003, 160, 271–282. [Google Scholar] [CrossRef]
- Gitelson, A.A.; Viña, A.; Ciganda, V.; Rundquist, D.C.; Arkebauer, T.J. Remote estimation of canopy chlorophyll content in crops. Geophys. Res. Lett. 2005, 32. [Google Scholar] [CrossRef]
- Rouse, J.W. Monitoring the Vernal Advancement and Retrogradation (Greenwave Effect) of Natural Vegetation. In NASA/GSFCT Technical Report; NTRS: Chicago, IL, USA, 1974. [Google Scholar]
- Jiang, Z.; Huete, A.; Didan, K.; Miura, T. Development of a two-band enhanced vegetation index without a blue band. Remote Sens. Environ. 2008, 112, 3833–3845. [Google Scholar] [CrossRef]
- Gao, B.C. NDWI—A normalized difference water index for remote sensing of vegetation liquid water from space. Remote Sens. Environ. 1996, 58, 257–266. [Google Scholar] [CrossRef]
- Peñuelas, J.; Filella, I.; Biel, C.; Serrano, L.; Save, R. The reflectance at the 950–970 nm region as an indicator of plant water status. Int. J. Remote Sens. 1993, 14, 1887–1905. [Google Scholar] [CrossRef]
- Guyot, G.; Baret, F.; Jacquemoud, S. Imaging spectroscopy for vegetation studies. Imag. Spectrosc. Fundam. Prospect. Appl. 1992, 2, 145–165. [Google Scholar]
- Baker, N.R.; Rosenqvist, E. Applications of chlorophyll fluorescence can improve crop production strategies: An examination of future possibilities. J. Exp. Bot. 2004, 55, 1607–1621. [Google Scholar] [CrossRef]
- Zhang, R.; Sharkey, T.D. Photosynthetic electron transport and proton flux under moderate heat stress. Photosynth. Res. 2009, 100, 29–43. [Google Scholar] [CrossRef] [PubMed]
- Kalaji, H.M.; Jajoo, A.; Oukarroum, A.; Brestic, M.; Zivcak, M.; Samborska, I.A.; Cetner, M.D.; Lukasik, I.; Goltsev, V.; Ladle, R.J. Chlorophyll a fluorescence as a tool to monitor physiological status of plants under abiotic stress conditions. Acta Physiol. Plant. 2016, 38, 102. [Google Scholar] [CrossRef]
- Rastogi, A.; Zivcak, M.; Tripathi, D.K.; Yadav, S.; Kalaji, H.M. Phytotoxic effect of silver nanoparticles in Triticum aestivum: Improper regulation of photosystem I activity as the reason for oxidative damage in the chloroplast. Photosynthetica 2019, 57, 209–216. [Google Scholar] [CrossRef]
- Zhou, Y.; Huang, L.F.; Du, Y.S.; Yu, J.Q. Greenhouse and field cucumber genotypes use different mechanisms to protect against dark chilling. Funct. Plant Biol. 2004, 31, 1215–1223. [Google Scholar] [CrossRef]
- Singh, S.K.; Reddy, V.R.; Fleisher, D.H.; Timer, D.J. Relationship between photosynthetic pigments and chlorophyll fluorescence in soybean under varying phosphorus nutrition at ambient and elevated CO2. Photosynthetica 2017, 55, 421–433. [Google Scholar] [CrossRef]
- Lin, Y.C.; Yue-Gao, H.U.; Ren, C.Z.; Guo, L.C.; Wang, C.L.; Jiang, Y.; Wang, X.J.; Hlatshwayo, P.; Zeng, Z.H. Effects of Nitrogen Application on Chlorophyll Fluorescence Parameters and Leaf Gas Exchange in Naked Oat. J. Integr. Agric. 2013, 12, 2164–2171. [Google Scholar] [CrossRef]
- Baret, F.; Jacquemoud, S.; Guyot, G.; Leprieur, C. Modeled analysis of the biophysical nature of spectral shifts and comparison with information content of broad bands. Remote Sens. Environ. 1992, 41, 133–142. [Google Scholar] [CrossRef]
- Broge, N.H.; Leblanc, E. Comparing prediction power and stability of broadband and hyperspectral vegetation indices for estimation of green leaf area index and canopy chlorophyll density. Remote Sens. Environ. 2000, 76, 156–172. [Google Scholar] [CrossRef]
- Mutanga, O.; Skidmore, A.K. Red edge shift and biochemical content in grass canopies. ISPRS J. Photogram. Remote Sens. 2007, 62, 34–42. [Google Scholar] [CrossRef]
- Daumard, F.; Goulas, Y.; Champagne, S.; Fournier, A.; Ounis, A.; Olioso, A.; Moya, I. Continuous Monitoring of Canopy Level Sun-Induced Chlorophyll Fluorescence During the Growth of a Sorghum Field. IEEE Trans. Geosci. Remote 2012, 50, 4292–4300. [Google Scholar] [CrossRef]
| Experiment (Exp.) | Season | Rowledge (cm) | Cultivar | N rate (kg/ha) | Samples (Fv/Fm, Fv’/Fm’) | Sampling Date (Date/Phenological Stages/DAS) /Function |
|---|---|---|---|---|---|---|
| Exp. 1 | 2014 to 2015 | 25, 40 | Shengxuan 6 (V1) Yangmai 18 (V2) | 0 (N0), 150 (N2), 300 (N4) | 36, 35 36, 36 36, 36 33, 33 12, 12 | (9th Apr/booting/164), (16th Apr/heading/172), (26th Apr/anthesis/179), (1st May/filling/187), (7th May/7days after filling/193) (calibration) |
| Exp. 2 | 2015 to 2016 | 20, 30, 40 | Yangmai 18 (V2) | 0 (N0), 80 (N1), 150 (N2), 220 (N3) | 62, 35 62, 30 62, 30 | (9th Apr/booting/160), (18th Apr/heading/167), (5th May/filling/187), (validation) |
| Abbreviation | Description | Equation | |
|---|---|---|---|
| dark-adapted condition | Fo | Minimal fluorescence yield | |
| Fm | Maximal fluorescence yield | ||
| Fv/Fm | Maximal photochemical efficiency of PSII | = (Fm−Fo)/Fm | |
| light-adapted condition | Fs | Steady-state fluorescence | |
| Fo’ | Initial fluorescence in the presence of NPQ | ||
| Fm’ | Maximal fluorescence in the presence of NPQ | ||
| Fv’/Fm’ | Photochemical efficiency of PSII in the light | = (Fm’−Fs’)/Fm’ | |
| Y(II) | Yield of quantum efficiency | ||
| NPQ | Non-photochemical quenching calculated with Fm | = (Fm−Fm’)/Fm’ | |
| ETR | Apparent photosynthetic electron transport rate | =Yield × PFD × 0.5 × 0.8 |
| Spectral Feature Type | Index Name (Abbreviation) | Index Formulation And Reference | ||
|---|---|---|---|---|
| VI | ChlF VI | Reflectance ratio VI | Curvature index (CUR) | (R675 × R691)/R6832 [12] |
| RVI (750,800) | R750/R800 [12] | |||
| RVI (685,655) | R685/R655 [12] | |||
| RVI (680,630) | R680/R630 [13] | |||
| Derivative VI | Double-peak index (DPi) | (D688 × D710)/D6972 [14] | ||
| D705/D722 | D705/D722 [14] | |||
| D730/D706 | D730/D706 [14] | |||
| DP22 | Dλρ/D720 [14] | |||
| DPRI | Dλρ/D(λρ + 12 nm) [14] | |||
| physiological VI | Photochemical reflectance index (PRI) | (R531 − R570)/(R531 + R570) [33] | ||
| Chl VI | MERIS terrestrial chlorophyll index (MTCI) | (R754 − R709)/(R709 − R681) [34] | ||
| Red edge chlorophyll index (CIred edge) | (R800/R720) − 1 [35,36] | |||
| Structure VI | Normalized difference vegetation index (NDVI) | (R810 − R690)/(R810 + R690) [37] | ||
| Enhanced vegetation index (EVI) | 2.5 × (R810 – R690)/(R810 + 2.4 × R690 + 1) [38] | |||
| Water VI | Normalized difference water index (NDWI) | (R850 − R1240)/(R850 + R1240) [39] | ||
| Water index (WI) | R850/R970 [40] | |||
| REP | Linear interpolation (REPLI) | Explained as below, [41] | ||
| Polynomial fitting (REPPF) | Explained as below, [42] | |||
| Linear extrapolation (REPLE) | Explained as below, [21] | |||
| Wavelet-based REP extraction (WREP-S3) | Explained as below, [19] | |||
| Wavelet-based REP extraction (WREP-S4) | Explained as below, [17] | |||
| Leaf Pigment | Physiological Indices | Dark-adapted ChlF Parameters | Steady-state ChlF Parameters | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Car | Chlab | An | gs | Fo | Fm | Fv/Fm | Fo’ | Fs | Fm’ | Fv’/Fm’ | NPQ | Y(Ⅱ) | ETR | |
| Car | 1 | |||||||||||||
| Chlab | 0.83*** | 1 | ||||||||||||
| An | 0.41*** | 0.32*** | 1 | |||||||||||
| gs | 0.32*** | 0.26** | 0.75 | 1 | ||||||||||
| Fo | 0.0008 | 0.011 | 0.083 | 0.016 | 1 | |||||||||
| Fm | 0.20*** | 0.35*** | 0.12* | 0.087* | 0.54*** | 1 | ||||||||
| Fv/Fm | 0.51*** | 0.60*** | 0.55*** | 0.32*** | 0.017 | 0.54*** | 1 | |||||||
| Fo’ | 0.11** | 0.11* | 0.042 | 0.048 | 0.051 | 0.15* | 0.13* | 1 | ||||||
| Fs | 0.13* | 0.24*** | 0.0059 | 0.0097 | 0.34** | 0.53*** | 0.20** | 0.46*** | 1 | |||||
| Fm’ | 0.32*** | 0.30*** | 0.30** | 0.22** | 0.0014 | 0.20** | 0.52*** | 0.54*** | 0.31** | 1 | ||||
| Fv’/Fm’ | 0.34*** | 0.35*** | 0.47*** | 0.29*** | 0.017 | 0.17** | 0.58*** | 0.0073 | 0.045 | 0.59*** | 1 | |||
| NPQ | 0.035 | 0.0043 | 0.13* | 0.078 | 0.22** | 0.20** | 0.085 | 0.12* | 0.014 | 0.31** | 0.25** | 1 | ||
| Y(Ⅱ) | 0.063 | 0.016 | 0.39** | 0.24** | 0.0009 | 0.066 | 0.33*** | 0.0056 | 0.18** | 0.25** | 0.45*** | 0.53*** | 1 | |
| ETR | 0.063 | 0.016 | 0.40** | 0.24** | 0.37*** | 0.065 | 0.15* | 0.0055 | 0.18** | 0.25** | 0.41*** | 0.53*** | 0.99*** | 1 |
| FvFm | Indices | Calibration | Validation | |||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Equation | Rc2 | RMSEc | Rv2 | Bias | RMSEv | RRMSEv | ||||
| VI | ChlF VI | Reflectance ratio VI | CUR | y = −0.3963x + 1.196 | 0.66 | 0.017 | 0.50 | 0.0056 | 0.012 | 1.95 |
| R680/R630 | y = 0.2209x + 0.5797 | 0.59 | 0.019 | 0.46 | 0.0047 | 0.016 | 1.96 | |||
| R685/R655 | y = 0.3209ln (x) + 0.7388 | 0.50 | 0.021 | 0.25 | 0.021 | 0.027 | 3.43 | |||
| R750/R800 | y = −2.008x + 2.761 | 0.21 | 0.026 | 0.10 | 0.011 | 0.024 | 2.96 | |||
| Derivative VI | D705/D722 | y = −0.06905x + 0.8604 | 0.64 | 0.018 | 0.50 | 0.0037 | 0.015 | 1.87 | ||
| D730/D706 | y = 0.07926ln(x) + 0.8109 | 0.62 | 0.018 | 0.42 | 0.0012 | 0.016 | 1.95 | |||
| Dλρ/D720 | y = -0.08262ln(x) + 0.7919 | 0.64 | 0.018 | 0.28 | 0.0045 | 0.021 | 2.65 | |||
| Dλρ/D(λρ+12 nm) | y = −0.1255x + 0.9238 | 0.43 | 0.022 | 0.03 | 0.012 | 0.028 | 3.49 | |||
| DPi | y = −0.4849x + 0.9379 | 0.38 | 0.023 | 0.27 | 0.023 | 0.029 | 3.69 | |||
| Physiological VI | PRI | y = 1.009 + 0.7439 | 0.50 | 0.021 | 0.23 | 0.015 | 0.022 | 2.80 | ||
| Chl VI | MTCI | y = 0.04633 + 0.7071 | 0.61 | 0.018 | 0.33 | −0.014 | 0.019 | 2.41 | ||
| CIred edge | y = 0.1664 + 0.7056 | 0.60 | 0.018 | 0.39 | −0.013 | 0.022 | 2.79 | |||
| Structure VI | NDVI | y = 0.2546x + 0.5875 | 0.32 | 0.024 | 0.28 | 0.025 | 0.031 | 3.85 | ||
| EVI | y = 0.001713x + 0.7663 | 0.021 | 0.029 | 0.10 | 0.025 | 0.031 | 3.92 | |||
| Water VI | NDWI | y = −0.005260x + 0.7703 | 0.0012 | 0.029 | 0.15 | 0.025 | 0.032 | 4.01 | ||
| WI | y = 0.7209x + 0.02677 | 0.032 | 0.029 | 0.16 | 0.027 | 0.033 | 4.12 | |||
| REP | REPLI | y = 0.005224x − 2.961 | 0.62 | 0.018 | 0.45 | 0.034 | 0.037 | 4.62 | ||
| REPPF | y = 0.004448x − 2.383 | 0.68 | 0.016 | 0.43 | −0.027 | 0.030 | 3.78 | |||
| REPLE | y = 0.003633x − 1.786 | 0.64 | 0.018 | 0.43 | 0.037 | 0.040 | 5.06 | |||
| WREP-S3 | y = 3.155ln(x) − 19.94 | 0.66 | 0.017 | 0.48 | −0.013 | 0.019 | 2.33 | |||
| WREP-S4 | y = 0.005376x − 3.052 | 0.67 | 0.017 | 0.51 | 0.0025 | 0.014 | 1.80 | |||
| Wavelet feature | WF(702 nm, scale 3) | y = −0.5217x + 0.7406 | 0.70 | 0.016 | 0.48 | 0.0071 | 0.015 | 1.91 | ||
| WF(637 nm, scale 3) | y = −3.514x + 0.8036 | 0.69 | 0.016 | 0.37 | 0.0089 | 0.016 | 1.99 | |||
| WF(704 nm, scale 4) | y = −0.2112x + 0.7305 | 0.69 | 0.016 | 0.55 | 0.0050 | 0.014 | 1.81 | |||
| WF(592 nm, scale 5) | y = −0.4664x + 0.7930 | 0.69 | 0.016 | 0.40 | 0.0089 | 0.016 | 1.98 | |||
| WF(575 nm, scale 6) | y = −0.1260x + 0.7571 | 0.68 | 0.016 | 0.41 | 0.0047 | 0.016 | 1.99 | |||
| Fv‘Fm’ | Indices | Calibration | Validation | |||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Equation | Rc2 | RMSEc | Rv2 | Bias | RMSEv | RRMSEv | ||||
| VI | ChlF VI | Reflectance ratio VI | CUR | y = -0.4089x + 1.119 | 0.55 | 0.022 | 0.38 | 0.0072 | 0.030 | 4.28 |
| R680/R630 | y = 0.2097x + 0.4987 | 0.42 | 0.025 | 0.35 | 0.0074 | 0.030 | 4.28 | |||
| R685/R655 | y = 0.2811x + 0.3689 | 0.37 | 0.026 | 0.18 | 0.022 | 0.039 | 5.51 | |||
| R750/R800 | y = −2.928x + 3.583 | 0.36 | 0.026 | 0.24 | 0.0050 | 0.031 | 4.40 | |||
| Derivative VI | D705/D722 | y = −0.07581x + 0.7785 | 0.60 | 0.021 | 0.40 | 0.0031 | 0.028 | 3.96 | ||
| D730/D706 | y = 0.1405x + 0.5920 | 0.54 | 0.022 | 0.36 | 0.0034 | 0.030 | 3.96 | |||
| Dλρ/D720 | y = −0.04609x + 0.7422 | 0.52 | 0.023 | 0.26 | 0.0087 | 0.033 | 4.61 | |||
| Dλρ/D(λρ+12 nm) | y = −0.1332x + 0.8426 | 0.38 | 0.026 | 0.09 | 0.019 | 0.045 | 6.41 | |||
| DPi | y = −0.4247x + 0.8264 | 0.22 | 0.029 | 0.02 | 0.026 | 0.044 | 6.22 | |||
| Physiological VI | PRI | y = 1.084x + 0.6512 | 0.45 | 0.024 | 0.08 | 0.010 | 0.037 | 5.21 | ||
| Chl VI | MTCI | y = 0.04936x + 0.6122 | 0.55 | 0.022 | 0.3 | −0.014 | 0.029 | 4.17 | ||
| CIred edge | y = 0.1821x + 0.6088 | 0.57 | 0.022 | 0.32 | −0.011 | 0.030 | 4.07 | |||
| Structure VI | NDVI | y = 0.2999x + 0.4644 | 0.35 | 0.026 | 0.11 | 0.0025 | 0.041 | 5.88 | ||
| EVI | y = −0.000004619x + 0.6792 | 0.0000012 | 0.033 | 0.05 | 0.022 | 0.042 | 5.92 | |||
| Water VI | NDWI | y = 0.008163x + 0.6797 | 0.0022 | 0.033 | 0.06 | 0.022 | 0.041 | 5.82 | ||
| WI | y = 0.8144x − 0.1604 | 0.032 | 0.032 | 0.03 | 0.025 | 0.042 | 6.00 | |||
| REP | REPLI | y = 0.005767x − 3.439 | 0.60 | 0.021 | 0.38 | 0.0069 | 0.029 | 4.15 | ||
| REPPF | y = 0.004753x − 2.690 | 0.61 | 0.021 | 0.40 | 0.0050 | 0.028 | 3.94 | |||
| REPLE | y = 0.003943x − 2.095 | 0.59 | 0.021 | 0.41 | 0.0035 | 0.027 | 3.84 | |||
| WREP-S3 | y = 0.004762x − 2.693 | 0.59 | 0.021 | 0.43 | 0.0016 | 0.027 | 3.76 | |||
| WREP-S4 | y = 0.005792x – 3.438 | 0.61 | 0.021 | 0.43 | 0.0028 | 0.026 | 3.74 | |||
| Wavelet feature | WF(630 nm, scale 3) | y = −5.381x + 0.7119 | 0.59 | 0.021 | 0.24 | −0.0034 | 0.031 | 4.41 | ||
| WF(702 nm, scale 3) | y = −0.5553x + 0.6480 | 0.62 | 0.020 | 0.37 | −0.0009 | 0.028 | 4.03 | |||
| WF(465 nm, scale 4) | y = 4.335x + 0.7136 | 0.58 | 0.021 | 0.31 | −0.0112 | 0.031 | 4.46 | |||
| WF(704 nm, scale 4) | y = −0.2197x + 0.6440 | 0.62 | 0.020 | 0.43 | −0.0028 | 0.027 | 3.79 | |||
| WF(707 nm, scale 7) | y = −0.1804x + 0.7008 | 0.61 | 0.020 | 0.37 | −0.0012 | 0.028 | 3.94 | |||
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jia, M.; Li, D.; Colombo, R.; Wang, Y.; Wang, X.; Cheng, T.; Zhu, Y.; Yao, X.; Xu, C.; Ouer, G.; et al. Quantifying Chlorophyll Fluorescence Parameters from Hyperspectral Reflectance at the Leaf Scale under Various Nitrogen Treatment Regimes in Winter Wheat. Remote Sens. 2019, 11, 2838. https://doi.org/10.3390/rs11232838
Jia M, Li D, Colombo R, Wang Y, Wang X, Cheng T, Zhu Y, Yao X, Xu C, Ouer G, et al. Quantifying Chlorophyll Fluorescence Parameters from Hyperspectral Reflectance at the Leaf Scale under Various Nitrogen Treatment Regimes in Winter Wheat. Remote Sensing. 2019; 11(23):2838. https://doi.org/10.3390/rs11232838
Chicago/Turabian StyleJia, Min, Dong Li, Roberto Colombo, Ying Wang, Xue Wang, Tao Cheng, Yan Zhu, Xia Yao, Changjun Xu, Geli Ouer, and et al. 2019. "Quantifying Chlorophyll Fluorescence Parameters from Hyperspectral Reflectance at the Leaf Scale under Various Nitrogen Treatment Regimes in Winter Wheat" Remote Sensing 11, no. 23: 2838. https://doi.org/10.3390/rs11232838
APA StyleJia, M., Li, D., Colombo, R., Wang, Y., Wang, X., Cheng, T., Zhu, Y., Yao, X., Xu, C., Ouer, G., Li, H., & Zhang, C. (2019). Quantifying Chlorophyll Fluorescence Parameters from Hyperspectral Reflectance at the Leaf Scale under Various Nitrogen Treatment Regimes in Winter Wheat. Remote Sensing, 11(23), 2838. https://doi.org/10.3390/rs11232838







