Unmanned Aerial Vehicles (UAVs) for Monitoring Macroalgal Biodiversity: Comparison of RGB and Multispectral Imaging Sensors for Biodiversity Assessments
Abstract
1. Introduction
2. Materials and Methods
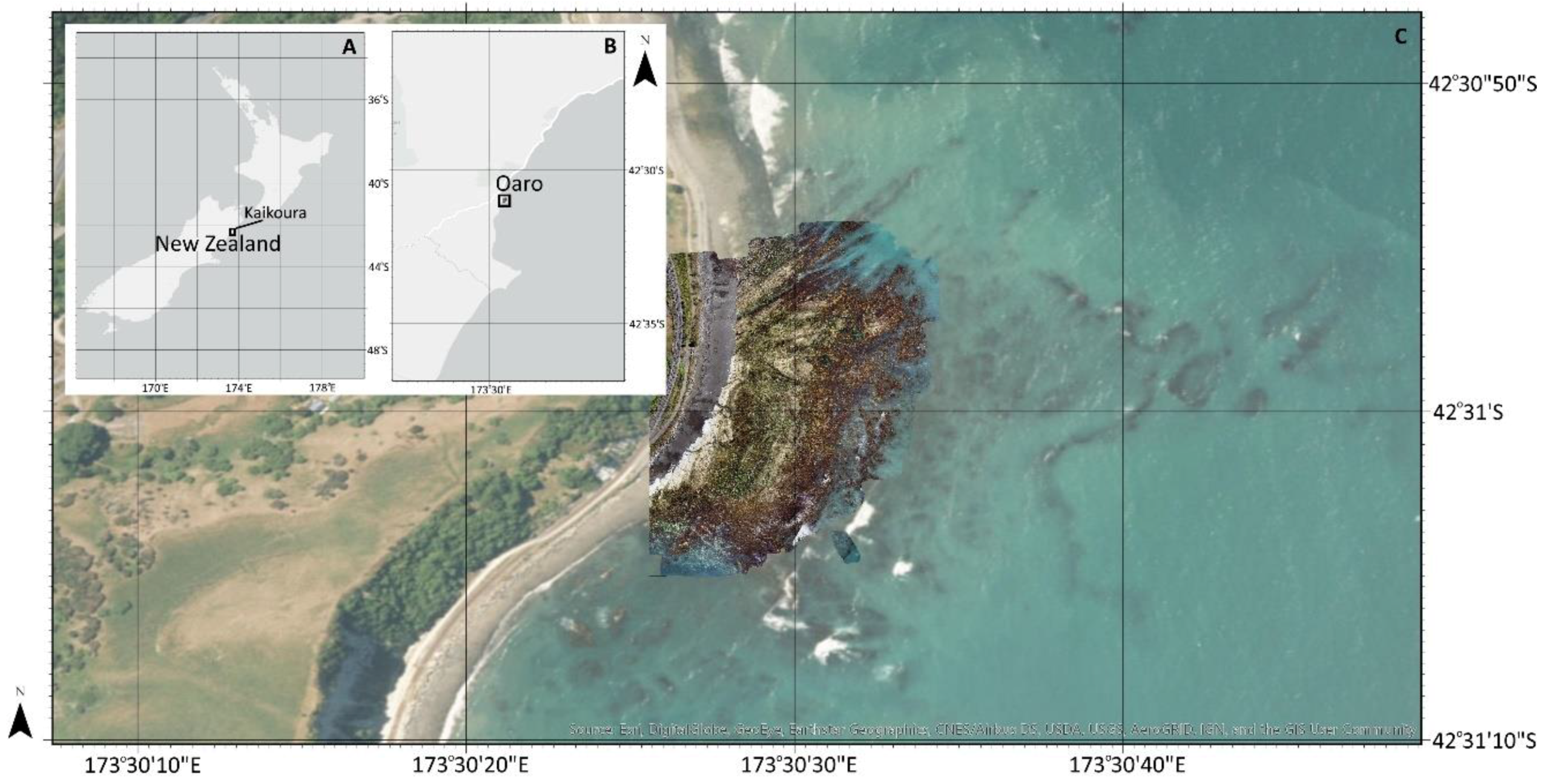
2.1. Study Site
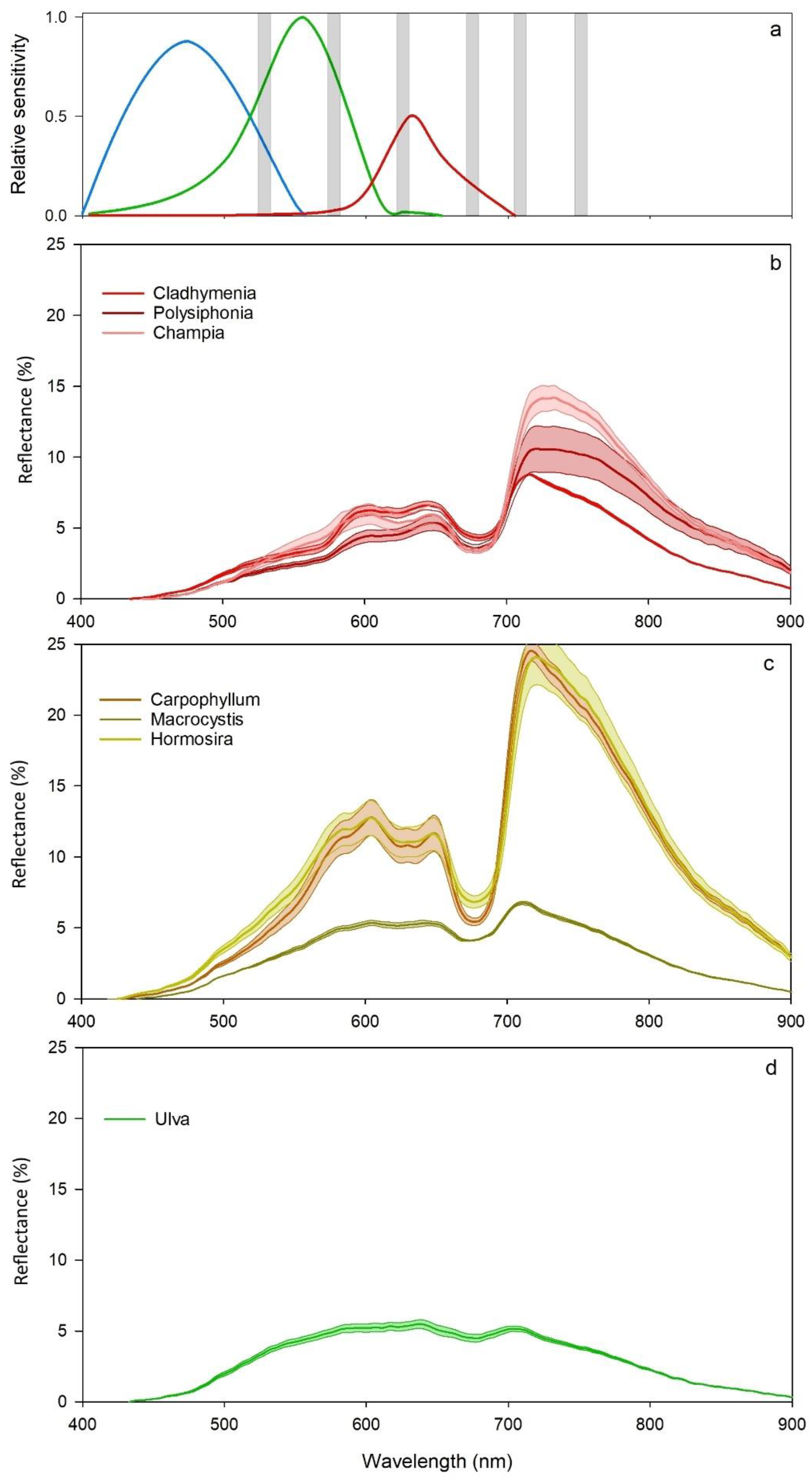
2.2. Macroalgal Richness and Spectral Profiles
2.3. UAV Mapping
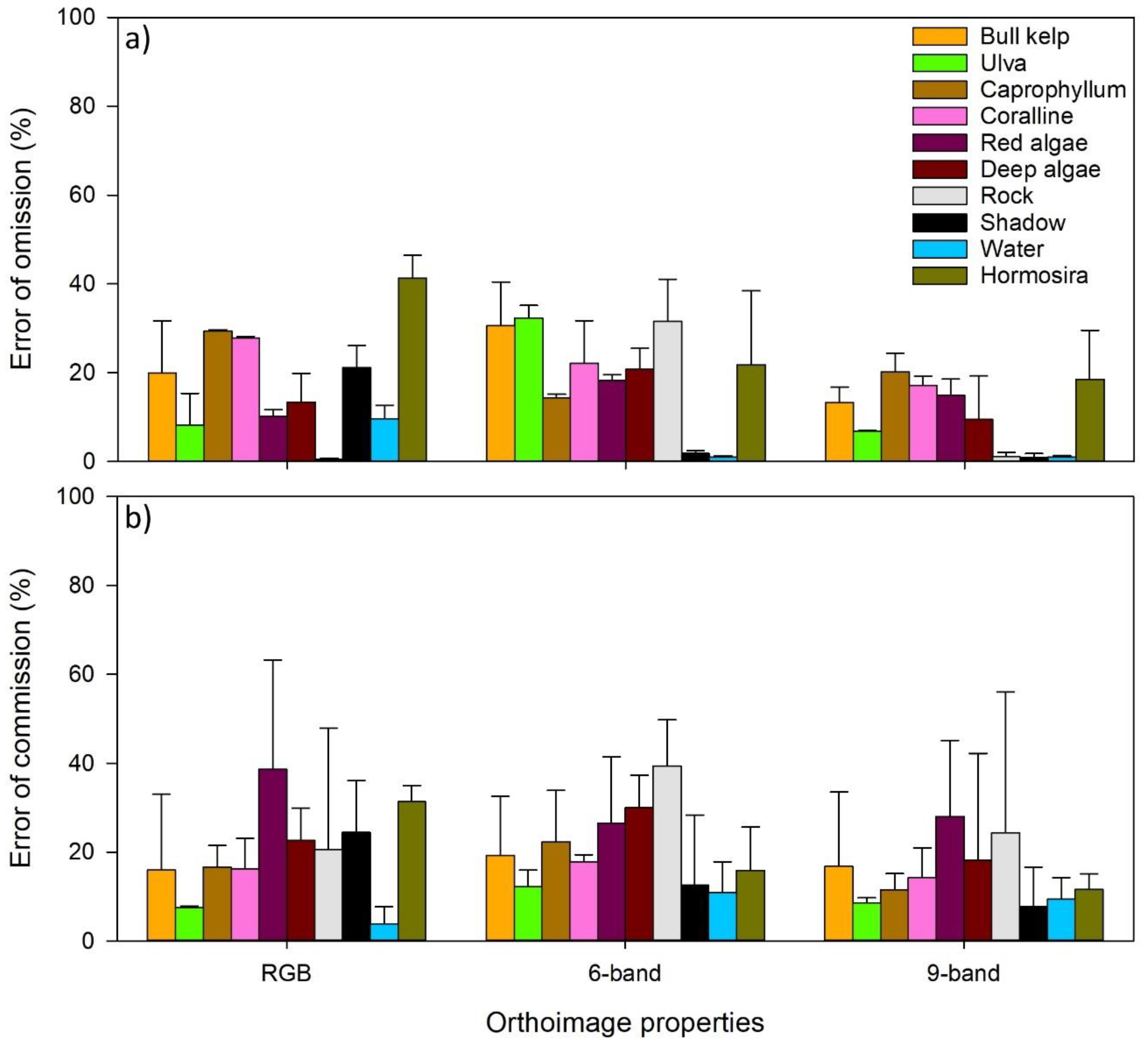
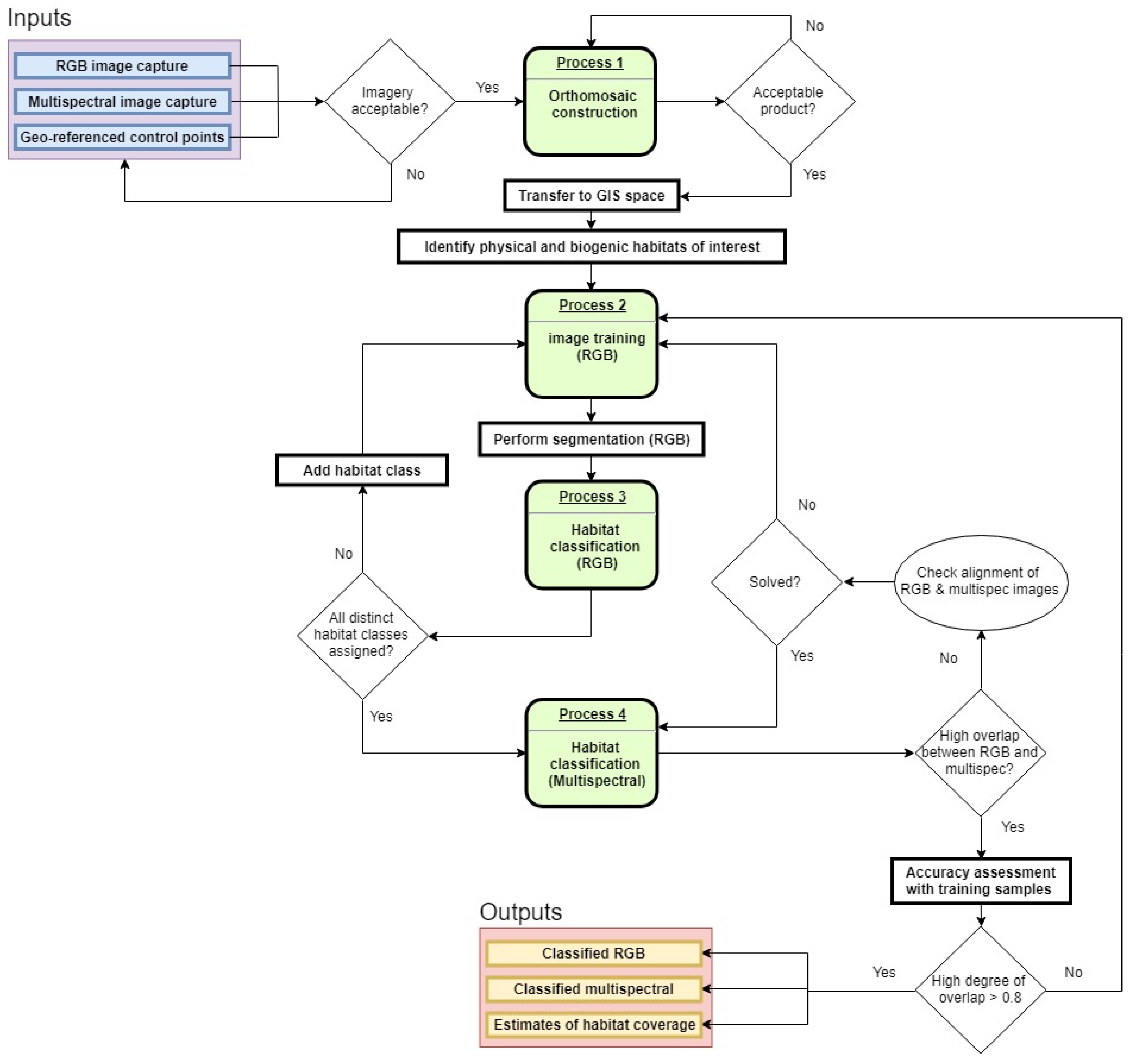
2.4. Analysis and Validation
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A
| Low Shore | Mid-Shore | ||||
|---|---|---|---|---|---|
| Phylum | Species/Genus | Mean % Cover | SD | Mean % Cover | SD |
| Chlorophyta | Bryopsis sp. | 0.0 | 0.0 | 0.1 | 0.3 |
| Codium fragile | 0.0 | 0.0 | 0.4 | 1.3 | |
| Ulva sp. | 10.6 | 9.6 | 16.4 | 18.3 | |
| Ochrophyta | Adenocystis utricularis | 0.0 | 0.0 | 0.1 | 0.3 |
| Carpophyllum maschalocarpum | 16.4 | 17.6 | 1.1 | 2.1 | |
| Colpomenia complex | 0.0 | 0.0 | 0.2 | 0.3 | |
| Colpomenia bullosa | 0.0 | 0.0 | 0.1 | 0.3 | |
| Cystophora scalaris | 8.9 | 13.3 | 0.7 | 1.3 | |
| Cystophora torulosa | 0.0 | 0.0 | 0.1 | 0.3 | |
| Dictyota spp. | 0.2 | 0.4 | 0.1 | 0.2 | |
| Durvillaea poha adult | 32.0 | 34.4 | 0.0 | 0.0 | |
| Durvillaea willana adult | 0.1 | 0.3 | 0.0 | 0.0 | |
| Halopteris sp. | 0.6 | 0.8 | 0.2 | 0.6 | |
| Hormosira banksii | 0.0 | 0.0 | 12.3 | 9.4 | |
| Marginariella boryana | 0.5 | 1.6 | 0.0 | 0.0 | |
| Notheia anomala | 0.0 | 0.0 | 0.3 | 0.6 | |
| Ralfsia verrucosa | 0.4 | 1.0 | 0.0 | 0.0 | |
| Zonaria | 0.0 | 0.0 | 0.1 | 0.3 | |
| Rhodophyta | Coralline turf | 18.7 | 21.4 | 51.2 | 36.1 |
| Coralline Paint | 20.0 | 20.4 | 0.7 | 0.8 | |
| Jania sp. | 0.0 | 0.0 | 0.1 | 0.1 | |
| Ceramium spp. | 0.5 | 1.3 | 0.6 | 1.1 | |
| Champia | 1.2 | 1.5 | 1.7 | 1.9 | |
| Chondria macrocarpa | 15.8 | 17.7 | 0.3 | 0.7 | |
| Cladhymenia spp. | 1.9 | 3.1 | 0.1 | 0.2 | |
| Curdiea flabellata/Gig leathery | 0.1 | 0.2 | 0.0 | 0.0 | |
| Echinothamnion spp. | 3.0 | 2.3 | 1.2 | 2.5 | |
| Euptilota | 0.1 | 0.2 | 0.0 | 0.0 | |
| Gelidium caulacantheum | 1.5 | 3.2 | 11.7 | 8.4 | |
| Gigartina circumcincta | 3.3 | 5.5 | 0.0 | 0.0 | |
| Gigartina clavifera | 1.0 | 2.0 | 0.3 | 0.6 | |
| Gigartina chapmanii/Caulachantus | 0.0 | 0.0 | 0.1 | 0.3 | |
| Gigartina decipiens | 0.4 | 0.7 | 0.4 | 0.7 | |
| Gigartina lanceolata | 4.4 | 5.0 | 1.0 | 1.0 | |
| Gigartina livida | 0.5 | 1.6 | 0.0 | 0.0 | |
| Gigartina multibranched | 0.1 | 0.3 | 0.0 | 0.0 | |
| Gigartina stripy | 0.2 | 0.6 | 0.0 | 0.0 | |
| Laurencia thysifera | 0.4 | 1.3 | 2.0 | 2.2 | |
| Lophothamnion hirtum | 0.3 | 0.4 | 0.9 | 1.9 | |
| Lophurella caespitosa | 0.1 | 0.3 | 0.0 | 0.0 | |
| Plocamium microcladioides | 0.0 | 0.0 | 0.1 | 0.3 | |
| Polysiphonia mullerii | 20.4 | 29.4 | 0.0 | 0.0 | |
| Polysiphonia spp. | 3.3 | 5.9 | 0.3 | 0.4 | |
| Polysiphonia strictissima | 0.8 | 1.3 | 5.1 | 7.7 | |
| Pterocladia lucida | 1.0 | 1.2 | 0.0 | 0.0 | |
Appendix B
References
- Schiel, D.R.; Foster, M.S. The Biology and Ecology of Giant Kelp Forests; University of California Press: California, CA, USA, 2015. [Google Scholar]
- Graham, M.H.; Vasquez, J.A.; Buschmann, A.H. Global ecology of the giant kelp Macrocystis: From ecotypes to ecosystems. Oceanogr. Mar. Biol. 2007, 45, 39. [Google Scholar]
- Krumhansl, K.A.; Okamoto, D.K.; Rassweiler, A.; Novak, M.; Bolton, J.J.; Cavanaugh, K.C.; Connell, S.D.; Johnson, C.R.; Konar, B.; Ling, S.D.; et al. Global patterns of kelp forest change over the past half-century. Proc. Natl. Acad. Sci. USA 2016, 113, 13785–13790. [Google Scholar] [CrossRef] [PubMed]
- Krause-Jensen, D.; Duarte, C.M. Substantial role of macroalgae in marine carbon sequestration. Nat. Geosci. 2016, 9, 737–742. [Google Scholar] [CrossRef]
- Edgar, G.J.; Samson, C.R.; Barrett, N.S. Species Extinction in the Marine Environment: Tasmania as a Regional Example of Overlooked Losses in Biodiversity. Conserv. Boil. 2005, 19, 1294–1300. [Google Scholar] [CrossRef]
- Wernberg, T.; Smale, D.A.; Tuya, F.; Thomsen, M.S.; Langlois, T.J.; De Bettignies, T.; Bennett, S.; Rousseaux, C.S. An extreme climatic event alters marine ecosystem structure in a global biodiversity hotspot. Nat. Clim. Chang. 2013, 3, 78. [Google Scholar]
- Thomsen, M.S.; Mondardini, L.; Alestra, T.; Gerrity, S.; Tait, L.; South, P.M.; Lilley, S.A.; Schiel, D.R. Local Extinction of Bull Kelp (Durvillaea spp.) Due to a Marine Heatwave. Front. Mar. Sci. 2019, 6, 84. [Google Scholar] [CrossRef]
- Kogan, F.; Kogan, F. Application of vegetation index and brightness temperature for drought detection. Adv. Space Res. 1995, 15, 91–100. [Google Scholar] [CrossRef]
- Chen, J.M. Evaluation of Vegetation Indices and a Modified Simple Ratio for Boreal Applications. Can. J. Remote. Sens. 1996, 22, 229–242. [Google Scholar] [CrossRef]
- Brown, M.E.; Pinzón, J.E.; Didan, K.; Morisette, J.T.; Tucker, C.J. Evaluation of the consistency of long-term NDVI time series derived from AVHRR, SPOT-vegetation, SeaWiFS, MODIS, and Landsat ETM+ sensors. IEEE Trans. Geosci. Remote Sens. 2006, 44, 1787–1793. [Google Scholar] [CrossRef]
- Cavanaugh, K.; Siegel, D.; Reed, D.; Dennison, P. Environmental controls of giant-kelp biomass in the Santa Barbara Channel, California. Mar. Ecol. Prog. Ser. 2011, 429, 1–17. [Google Scholar] [CrossRef]
- Bell, T.W.; Cavanaugh, K.C.; Reed, D.C.; Siegel, D.A. Geographical variability in the controls of giant kelp biomass dynamics. J. Biogeogr. 2015, 42, 2010–2021. [Google Scholar] [CrossRef]
- Bell, T.W.; Allen, J.G.; Cavanaugh, K.C.; Siegel, D.A. Three decades of variability in California’s giant kelp forests from the Landsat satellites. Remote Sens. Environ. 2018. [Google Scholar] [CrossRef]
- Nijland, W.; Reshitnyk, L.; Rubidge, E. Satellite remote sensing of canopy-forming kelp on a complex coastline: A novel procedure using the Landsat image archive. Remote. Sens. Environ. 2019, 220, 41–50. [Google Scholar] [CrossRef]
- Phinn, S.; Roelfsema, C.; Dekker, A.; Brando, V.; Anstee, J.; Brando, V. Mapping seagrass species, cover and biomass in shallow waters: An assessment of satellite multi-spectral and airborne hyper-spectral imaging systems in Moreton Bay (Australia). Remote. Sens. Environ. 2008, 112, 3413–3425. [Google Scholar] [CrossRef]
- Hedley, J.D.; Russell, B.J.; Randolph, K.; Pérez-Castro, M.Á.; Vásquez-Elizondo, R.M.; Dierssen, H.M.; Enríquez, S. Remote Sensing of Seagrass Leaf Area Index and Species: The Capability of a Model Inversion Method Assessed by Sensitivity Analysis and Hyperspectral Data of Florida Bay. Front. Mar. Sci. 2017, 4, 362. [Google Scholar] [CrossRef]
- Knudby, A.; Nordlund, L. Remote sensing of seagrasses in a patchy multi-species environment. Int. J. Remote. Sens. 2011, 32, 2227–2244. [Google Scholar] [CrossRef]
- Menge, B.A.; Farrell, T.M.; Oison, A.M.; Van Tamelen, P.; Turner, T. Algal recruitment and the maintenance of a plant mosaic in the low intertidal region on the Oregon coast. J. Exp. Mar. Boil. Ecol. 1993, 170, 91–116. [Google Scholar] [CrossRef]
- Van Genne, B.; Heaven, C.S.; Scrosati, R.A.; Watt, C.A. Species richness and diversity in different functional groups across environmental stress gradients: A model for marine rocky shores. Ecography 2011, 34, 151–161. [Google Scholar]
- Murfitt, S.L.; Allan, B.M.; Bellgrove, A.; Rattray, A.; Young, M.A.; Ierodiaconou, D. Applications of unmanned aerial vehicles in intertidal reef monitoring. Sci. Rep. 2017, 7, 10259. [Google Scholar] [CrossRef]
- Koh, L.P.; Wich, S.A. Dawn of Drone Ecology: Low-Cost Autonomous Aerial Vehicles for Conservation. Trop. Conserv. Sci. 2012, 5, 121–132. [Google Scholar] [CrossRef]
- Chirayath, V.; Earle, S.A. Drones that see through waves—Preliminary results from airborne fluid lensing for centimetre-scale aquatic conservation. Aquat. Conserv. Mar. Freshw. Ecosyst. 2016, 26, 237–250. [Google Scholar] [CrossRef]
- Ventura, D.; Bruno, M.; Lasinio, G.J.; Belluscio, A.; Ardizzone, G. A low-cost drone based application for identifying and mapping of coastal fish nursery grounds. Estuar. Coast. Shelf Sci. 2016, 171, 85–98. [Google Scholar] [CrossRef]
- Konar, B.; Iken, K. The use of unmanned aerial vehicle imagery in intertidal monitoring. Deep. Sea Res. Part II: Top. Stud. Oceanogr. 2018, 147, 79–86. [Google Scholar] [CrossRef]
- Manfreda, S.; McCabe, M.F.; Miller, P.E.; Lucas, R.; Madrigal, V.P.; Mallinis, G.; Ben Dor, E.; Helman, D.; Estes, L.; Ciraolo, G.; et al. On the Use of Unmanned Aerial Systems for Environmental Monitoring. Remote. Sens. 2018, 10, 641. [Google Scholar] [CrossRef]
- Ventura, D.; Bonifazi, A.; Gravina, M.F.; Belluscio, A.; Ardizzone, G. Mapping and Classification of Ecologically Sensitive Marine Habitats Using Unmanned Aerial Vehicle (UAV) Imagery and Object-Based Image Analysis (OBIA). Remote. Sens. 2018, 10, 1331. [Google Scholar] [CrossRef]
- Nahirnick, N.K.; Reshitnyk, L.; Campbell, M.; Hessing-Lewis, M.; Costa, M.; Yakimishyn, J.; Lee, L. Mapping with confidence; delineating seagrass habitats using Unoccupied Aerial Systems (UAS). Remote Sens. Ecol. Conserv. 2019, 5, 121–135. [Google Scholar] [CrossRef]
- Anderson, K.; Gaston, K.J. Lightweight unmanned aerial vehicles will revolutionize spatial ecology. Front. Ecol. Environ. 2013, 11, 138–146. [Google Scholar] [CrossRef]
- Elvidge, C.D.; Chen, Z. Comparison of broad-band and narrow-band red and near-infrared vegetation indices. Remote. Sens. Environ. 1995, 54, 38–48. [Google Scholar] [CrossRef]
- Enríquez, S.; Agustí, S.; Duarte, C.M. Light absorption by marine macrophytes. Oecologia 1994, 98, 121–129. [Google Scholar] [CrossRef] [PubMed]
- Tait, L.W.; Hawes, I.; Schiel, D.R. Shining Light on Benthic Macroalgae: Mechanisms of Complementarity in Layered Macroalgal Assemblages. PLoS ONE 2014, 9, e114146. [Google Scholar] [CrossRef]
- Rodríguez, Y.C.; Gómez, J.D.; Sánchez-Carnero, N.; Rodríguez-Pérez, D. A comparison of spectral macroalgae taxa separability methods using an extensive spectral library. Algal Res. 2017, 26, 463–473. [Google Scholar] [CrossRef]
- Haxo, F.T.; Blinks, L.R. Photosynthetic Action Spectra of Marine Algae. J. Gen. Physiol. 1950, 33, 389–422. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, K.; Skidmore, A. Spectral discrimination of vegetation types in a coastal wetland. Remote. Sens. Environ. 2003, 85, 92–108. [Google Scholar] [CrossRef]
- Schiel, D.R. Experimental analyses of diversity partitioning in southern hemisphere algal communities. Oecologia 2019, 190, 179–193. [Google Scholar] [CrossRef] [PubMed]
- Schiel, D.R. Biogeographic patterns and long-term changes on New Zealand coastal reefs: Non-trophic cascades from diffuse and local impacts. J. Exp. Mar. Boil. Ecol. 2011, 400, 33–51. [Google Scholar] [CrossRef]
- Tait, L.W.; Schiel, D.R. Ecophysiology of Layered Macroalgal Assemblages: Importance of Subcanopy Species Biodiversity in Buffering Primary Production. Front. Mar. Sci. 2018, 5, 444. [Google Scholar] [CrossRef]
- Filbee-Dexter, K.; Scheibling, R. Sea urchin barrens as alternative stable states of collapsed kelp ecosystems. Mar. Ecol. Prog. Ser. 2014, 495, 1–25. [Google Scholar] [CrossRef]
- Foster, M.S.; Schiel, D.R. Loss of predators and the collapse of southern California kelp forests (?): Alternatives, explanations and generalizations. J. Exp. Mar. Boil. Ecol. 2010, 393, 59–70. [Google Scholar] [CrossRef]
- Tait, L.W. Giant kelp forests at critical light thresholds show compromised ecological resilience to environmental and biological drivers. Estuar. Coast. Shelf Sci. 2019, 219, 231–241. [Google Scholar] [CrossRef]
- Ling, S.D.; Johnson, C.R.; Frusher, S.D.; Ridgway, K.R. Overfishing reduces resilience of kelp beds to climate-driven catastrophic phase shift. Proc. Natl. Acad. Sci. USA 2009, 106, 22341–22345. [Google Scholar] [CrossRef]
- Wernberg, T.; Bennett, S.; Babcock, R.C.; De Bettignies, T.; Cure, K.; Depczynski, M.; Dufois, F.; Fromont, J.; Fulton, C.J.; Hovey, R.K.; et al. Climate-driven regime shift of a temperate marine ecosystem. Science 2016, 353, 169–172. [Google Scholar] [CrossRef] [PubMed]
- Helmuth, B.; Broitman, B.R.; Blanchette, C.A.; Gilman, S.; Halpin, P.; Harley, C.D.G.; O’Donnell, M.J.; Hofmann, G.E.; Menge, B.; Strickland, D. Mosaic patterns of thermal stress in the rocky intertidal zone: Implications for climate change. Ecol. Monogr. 2006, 76, 461–479. [Google Scholar] [CrossRef]
- Sorte, C.J.; Bernatchez, G.; Mislan, K.A.S.; Pandori, L.L.; Silbiger, N.J.; Wallingford, P.D. Thermal tolerance limits as indicators of current and future intertidal zonation patterns in a diverse mussel guild. Mar. Boil. 2019, 166, 6. [Google Scholar]
- Oppelt, N.; Schulze, F.; Bartsch, I.; Doernhoefer, K.; Eisenhardt, I. Hyperspectral classification approaches for intertidal macroalgae habitat mapping: A case study in Heligoland. Opt. Eng. 2012, 51, 111703. [Google Scholar] [CrossRef]
- Stuart, M.B.; Mcgonigle, A.J.S.; Willmott, J.R. Hyperspectral Imaging in Environmental Monitoring: A Review of Recent Developments and Technological Advances in Compact Field Deployable Systems. Sensors 2019, 19, 3071. [Google Scholar] [CrossRef] [PubMed]




| Remote Validation | In Situ Validation | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Three-band | Six-band | Nine-band | Three-band | Six-band | Nine-band | |||||||
| Class | U-acc | P-acc | U-acc | P-acc | U-acc | P-acc | U-acc | P-acc | U-acc | P-acc | U-acc | P-acc |
| Durvillaea spp. | 0.88 | 0.71 | 0.76 | 0.71 | 0.89 | 0.78 | 0.72 | 0.96 | 0.63 | 0.91 | 0.79 | 0.96 |
| Ulva | 0.87 | 0.92 | 0.65 | 0.91 | 0.94 | 0.95 | 0.97 | 0.92 | 0.7 | 0.84 | 0.93 | 0.92 |
| Carpophyllum | 0.7 | 0.8 | 0.85 | 0.86 | 0.84 | 0.89 | 0.71 | 0.87 | 0.86 | 0.7 | 0.79 | 0.92 |
| Coralline | 0.71 | 0.89 | 0.84 | 0.81 | 0.84 | 0.83 | 0.72 | 0.79 | 0.7 | 0.84 | 0.81 | 0.9 |
| Red algae | 0.89 | 0.79 | 0.8 | 0.84 | 0.82 | 0.84 | 0.91 | 0.44 | 0.8 | 0.62 | 0.88 | 0.9 |
| Submerged algae | 0.82 | 0.73 | 0.75 | 0.65 | 0.84 | 0.98 | - | - | - | |||
| Hormosira | 0.55 | 0.66 | 0.82 | 0.91 | 0.89 | 0.9 | 0.62 | 0.71 | 0.67 | 0.76 | 0.73 | 0.86 |
| Rock | 0.75 | 0.6 | 0.98 | 0.53 | 0.98 | 0.87 | 0.82 | 0.99 | 0.98 | 0.68 | 0.99 | 0.99 |
| Shadow | 0.92 | 0.84 | 0.99 | 0.99 | 0.99 | 0.99 | 0.88 | 0.67 | 0.99 | 0.74 | 0.99 | 0.99 |
| Water | 0.99 | 0.99 | 0.61 | 0.94 | 0.98 | 0.95 | 0.99 | 0.93 | 0.76 | 0.83 | 0.99 | 0.87 |
| Agreement % | 79% | 81% | 90% | 81% | 77% | 88% | ||||||
| Cohen’s Kappa | 0.77 | 0.79 | 0.89 | 0.79 | 0.75 | 0.87 | ||||||
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tait, L.; Bind, J.; Charan-Dixon, H.; Hawes, I.; Pirker, J.; Schiel, D. Unmanned Aerial Vehicles (UAVs) for Monitoring Macroalgal Biodiversity: Comparison of RGB and Multispectral Imaging Sensors for Biodiversity Assessments. Remote Sens. 2019, 11, 2332. https://doi.org/10.3390/rs11192332
Tait L, Bind J, Charan-Dixon H, Hawes I, Pirker J, Schiel D. Unmanned Aerial Vehicles (UAVs) for Monitoring Macroalgal Biodiversity: Comparison of RGB and Multispectral Imaging Sensors for Biodiversity Assessments. Remote Sensing. 2019; 11(19):2332. https://doi.org/10.3390/rs11192332
Chicago/Turabian StyleTait, Leigh, Jochen Bind, Hannah Charan-Dixon, Ian Hawes, John Pirker, and David Schiel. 2019. "Unmanned Aerial Vehicles (UAVs) for Monitoring Macroalgal Biodiversity: Comparison of RGB and Multispectral Imaging Sensors for Biodiversity Assessments" Remote Sensing 11, no. 19: 2332. https://doi.org/10.3390/rs11192332
APA StyleTait, L., Bind, J., Charan-Dixon, H., Hawes, I., Pirker, J., & Schiel, D. (2019). Unmanned Aerial Vehicles (UAVs) for Monitoring Macroalgal Biodiversity: Comparison of RGB and Multispectral Imaging Sensors for Biodiversity Assessments. Remote Sensing, 11(19), 2332. https://doi.org/10.3390/rs11192332






