Monitoring Rice Phenology Based on Backscattering Characteristics of Multi-Temporal RADARSAT-2 Datasets
Abstract
1. Introduction
2. Materials and Methods
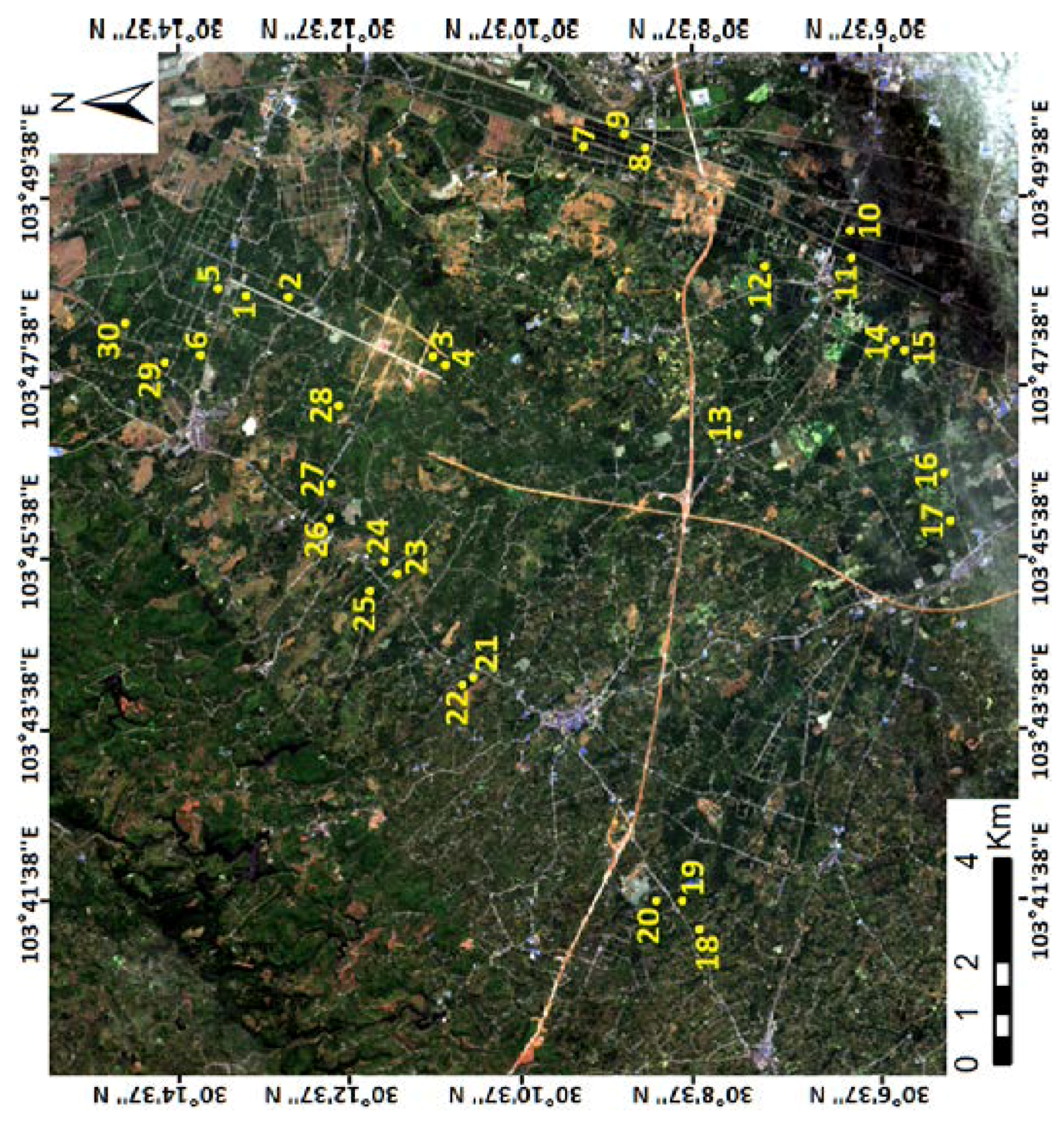
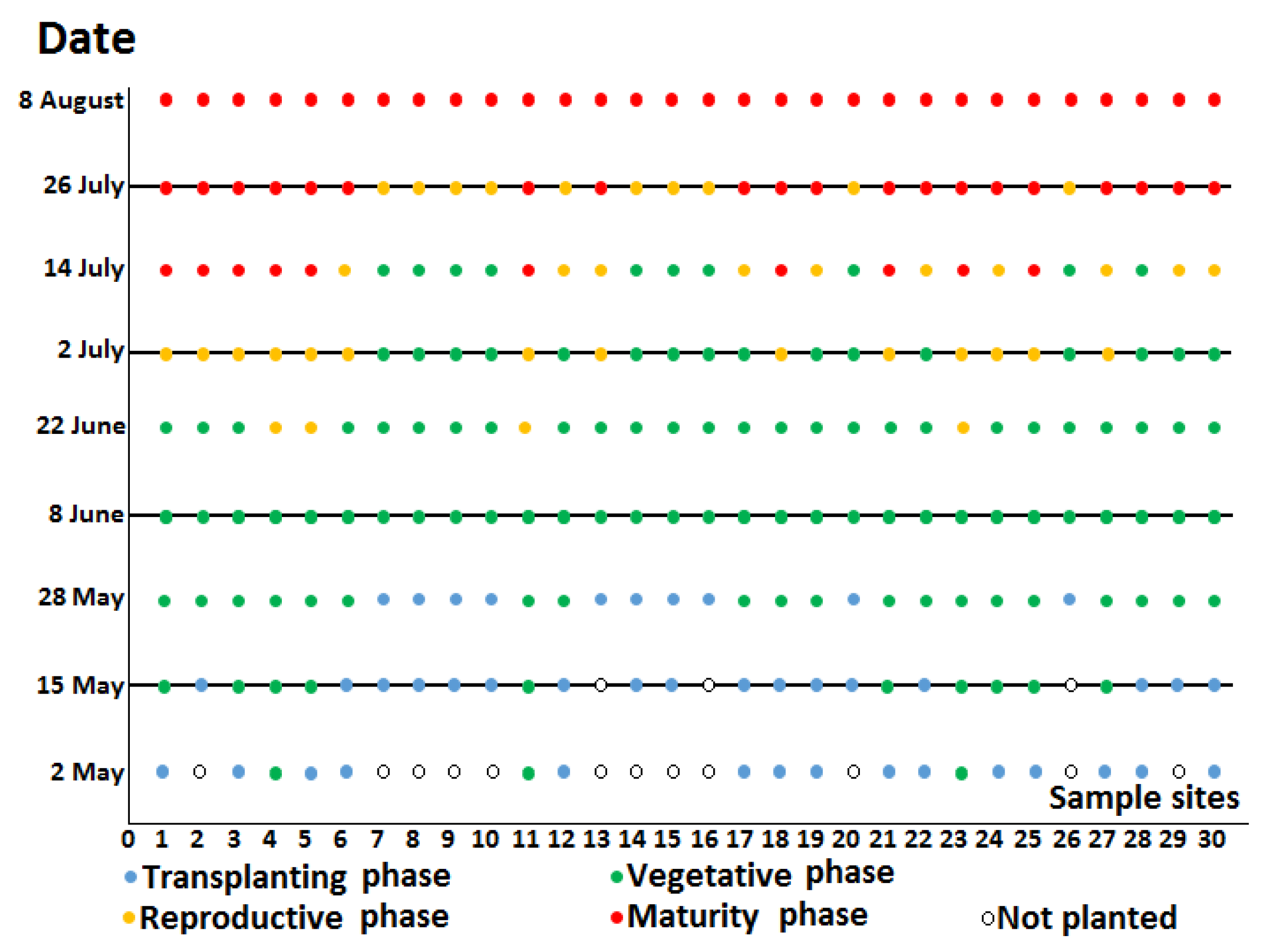
2.1. Study Area and Field Observation
2.2. Rice Phenology
2.3. RADARSAT-2 Data Preprocessing
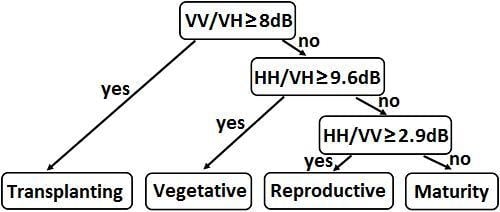
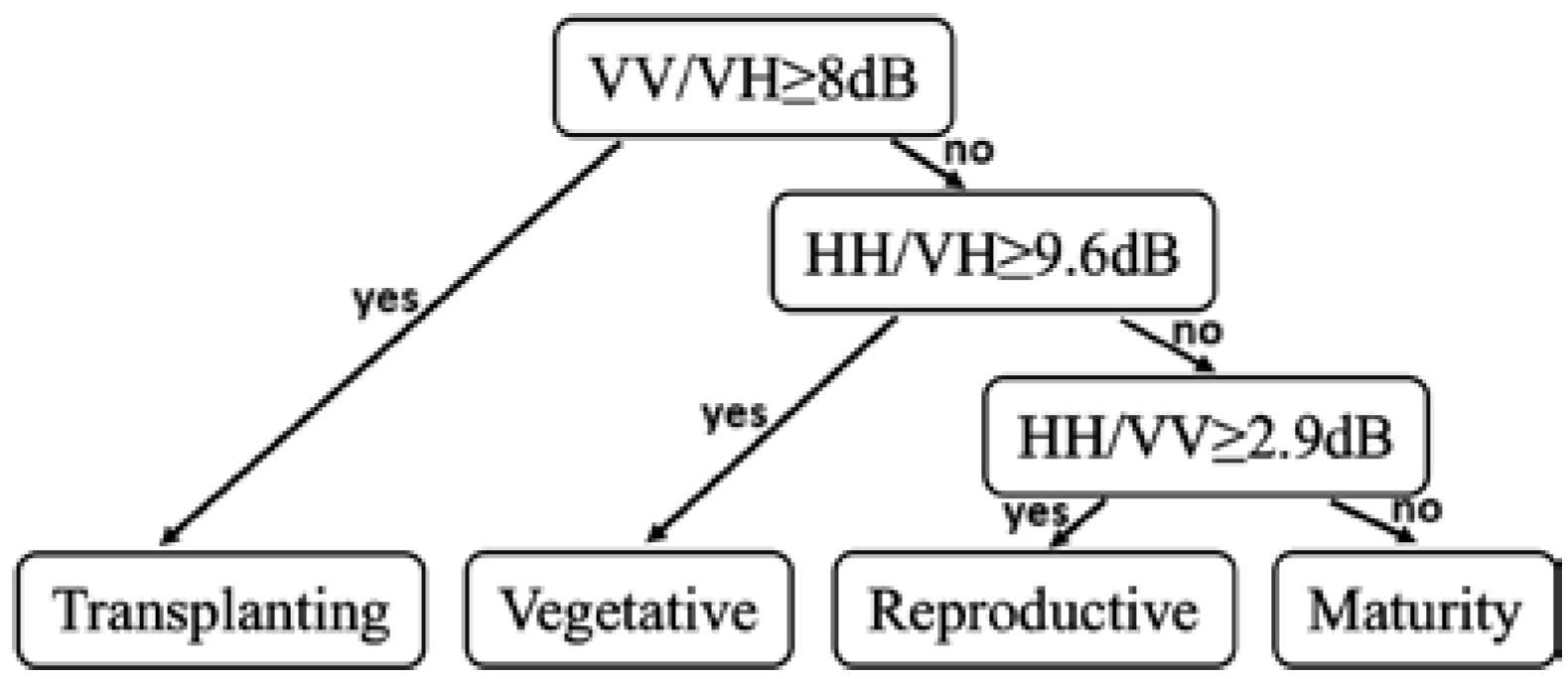
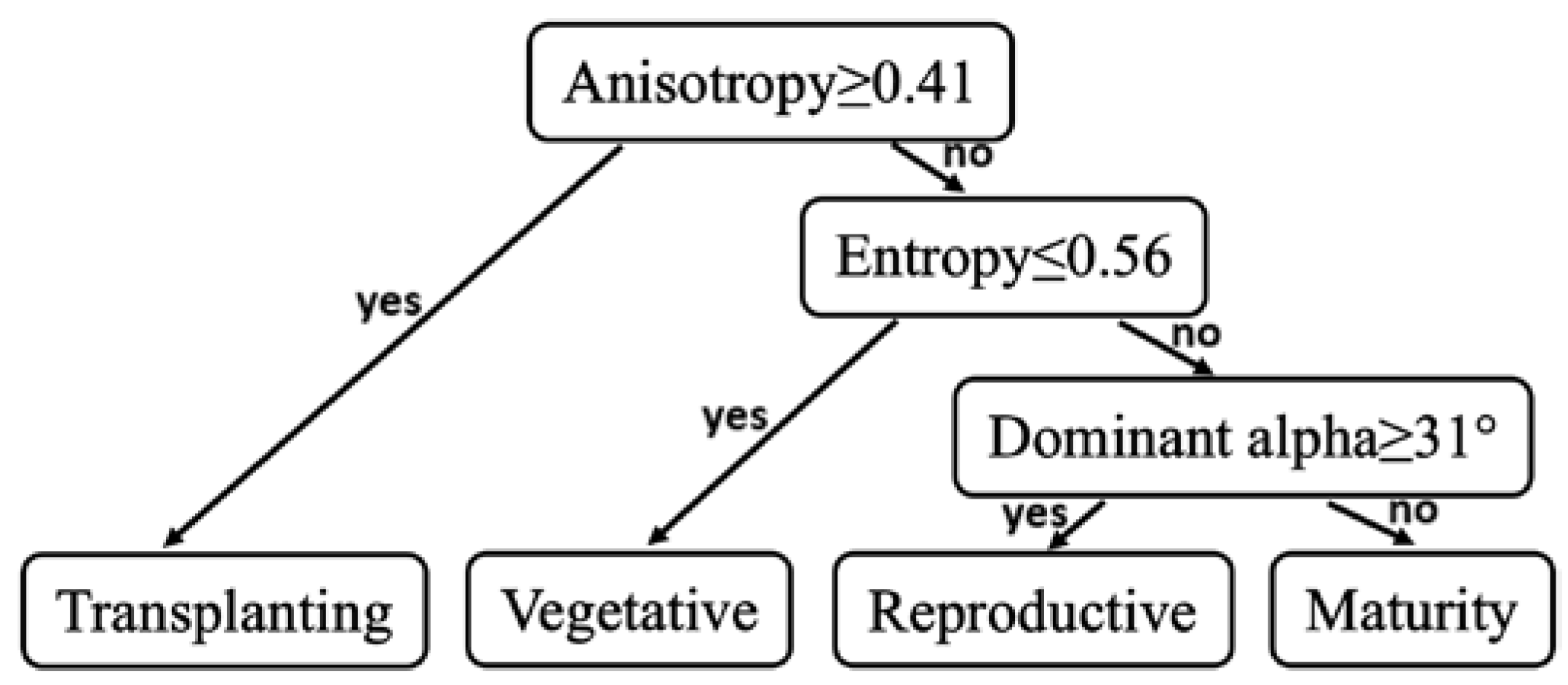
2.4. Decision Tree Method for Phenology Retrieval
3. Results
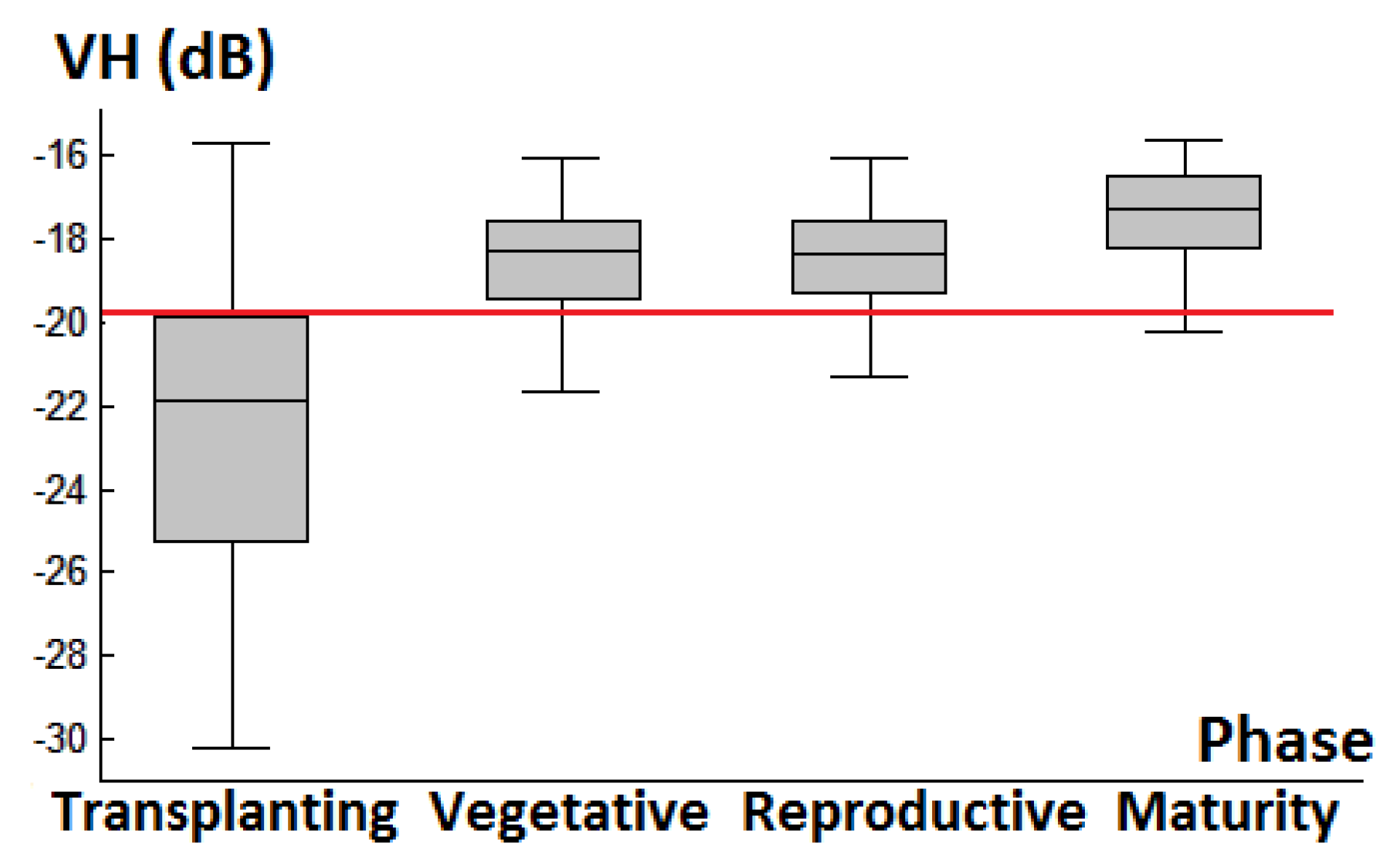
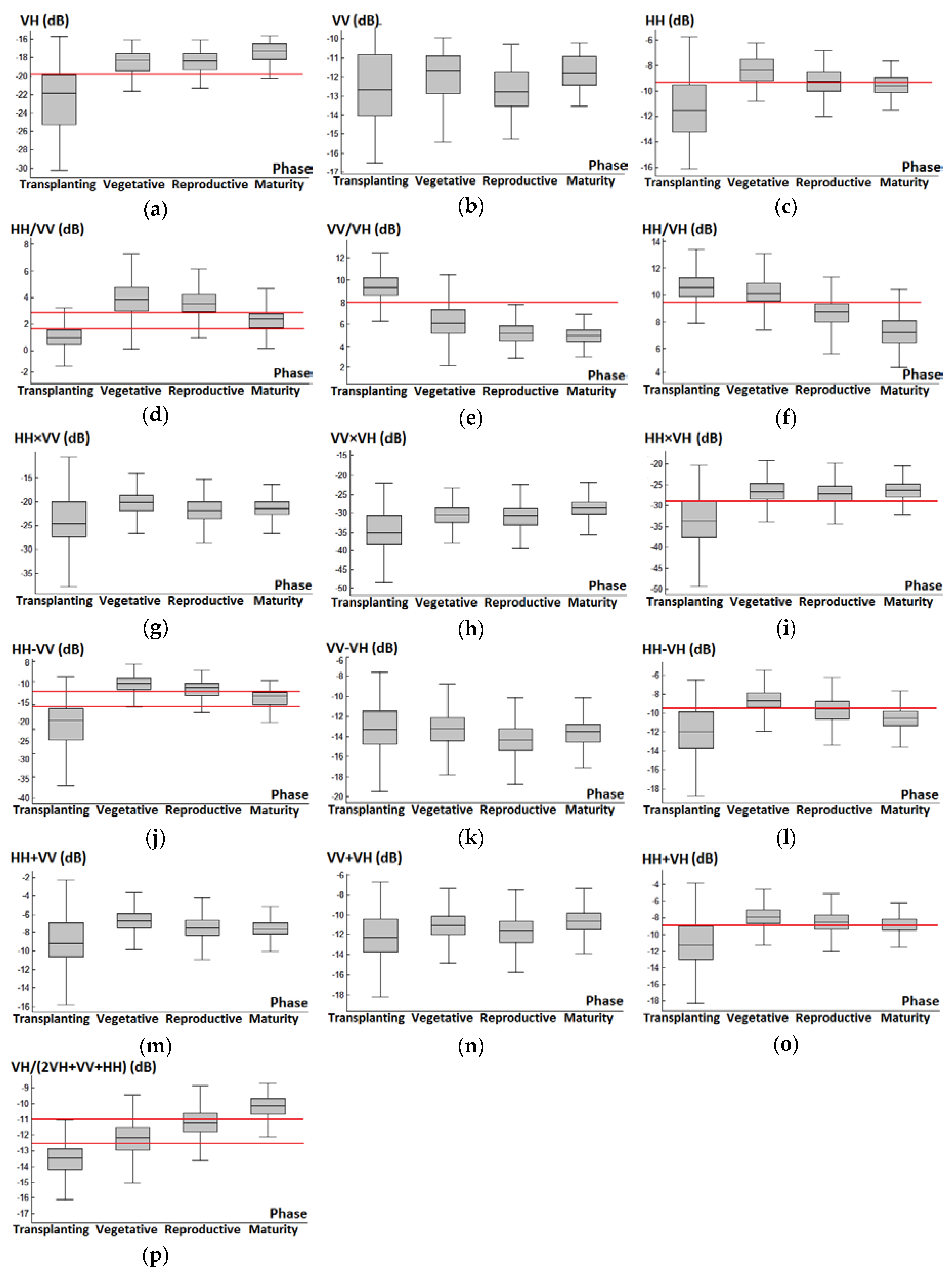
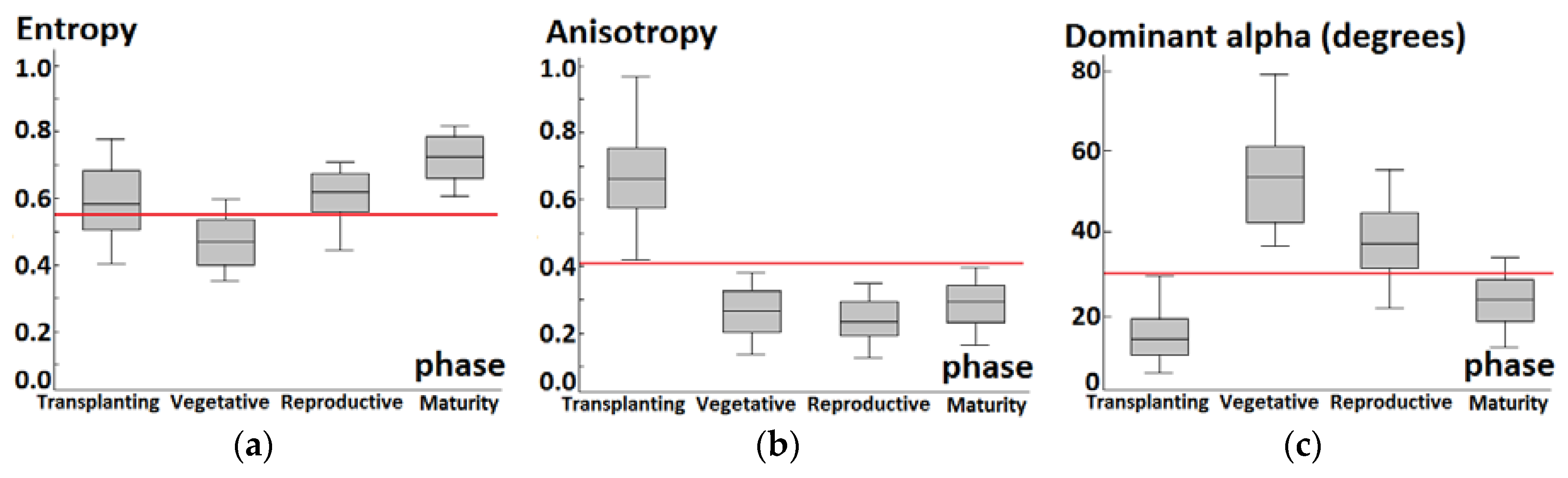
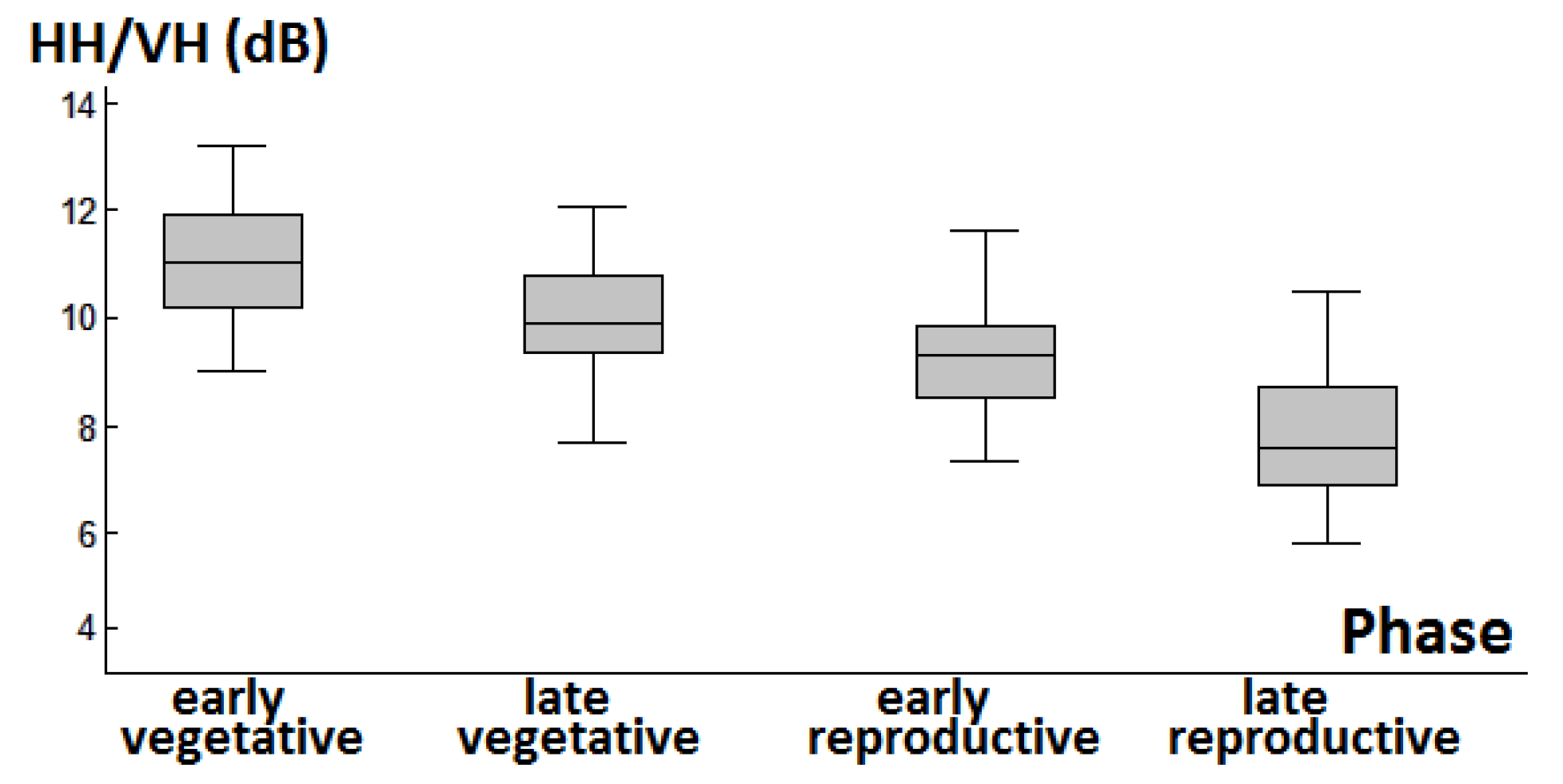
3.1. Backscattering Coefficient Analysis and Decision Tree Development
3.3. Validation
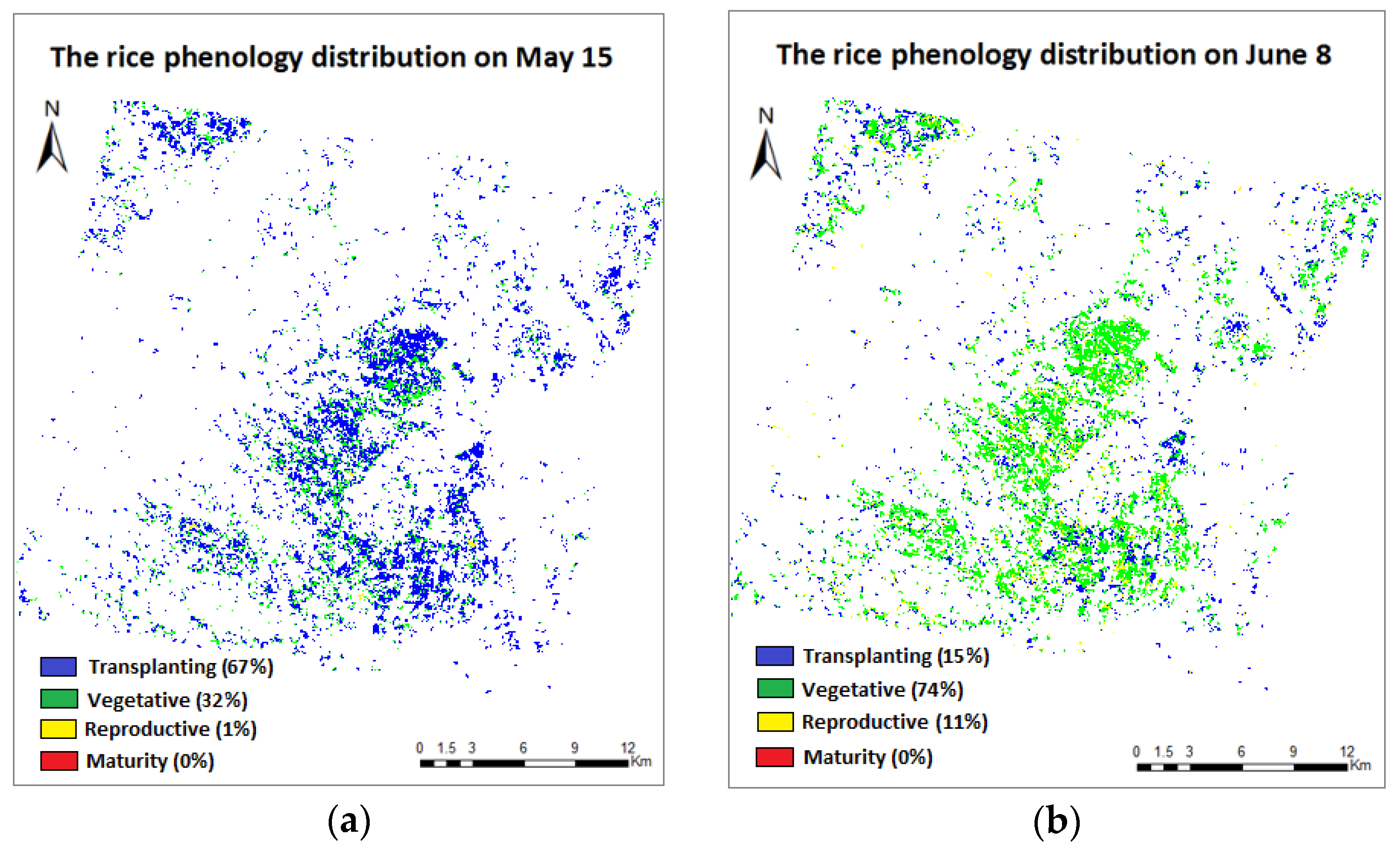
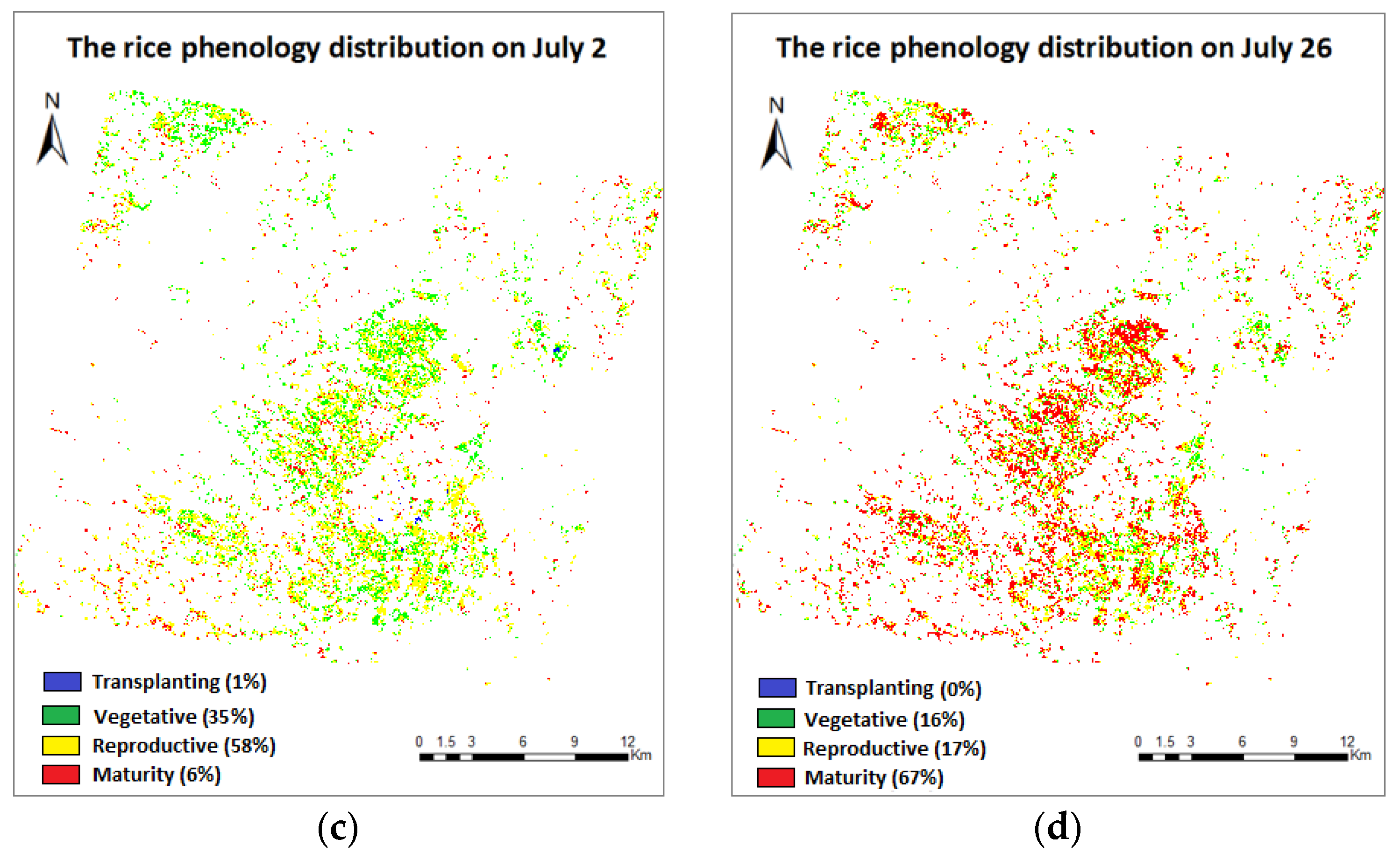
3.4. Phenology Extraction and Mapping
4. Discussion
4.1. Phenology Extraction Comparison between Backscattering Coefficients and Decomposition Parameters
4.2. Response of Backscattering Coefficients in Phenology Retrieval
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Chen, C.; Son, N.T.; Chang, L. Monitoring of rice cropping intensity in the upper Mekong Delta, Vietnam using time-series MODIS data. Adv. Space Res. 2012, 49, 292–301. [Google Scholar] [CrossRef]
- McLean, J.; Hardy, B.; Hettel, G. Rice Almanac, 4th ed.; International Rice Research Institute: Los Banos, Philippines, 2013; ISBN 978-971-22-0300-8. [Google Scholar]
- Sakamoto, T. A crop phenology detection method using time-series MODIS data. Remote Sens. Environ. 2005, 96, 366–374. [Google Scholar] [CrossRef]
- Lausch, A.; Salbach, C.; Schmidt, A.; Doktor, D.; Merbach, I.; Pause, M. Deriving phenology of barley with imaging hyperspectral remote sensing. Ecol. Model. 2015, 295, 123–135. [Google Scholar] [CrossRef]
- Lopez-Sanchez, J.M.; Cloude, S.R.; Ballester-Berman, J.D. Rice phenology monitoring by means of SAR polarimetry at X-band. IEEE Trans. Geosci. Remote Sens. 2012, 50, 2695–2709. [Google Scholar] [CrossRef]
- Lopez-Sanchez, J.M.; Vicente-Guijalba, F.; Ballester-Berman, J.D.; Cloude, S.R. Polarimetric response of rice fields at C-band: Analysis and phenology retrieval. IEEE Trans. Geosci. Remote Sens. 2014, 52, 2977–2993. [Google Scholar] [CrossRef]
- Vicente-Guijalba, F.; Martinez-Marin, T.; Lopez-Sanchez, J.M. Crop phenology estimation using a multitemporal model and a Kalman filtering strategy. IEEE Geosci. Remote Sens. Lett. 2014, 11, 1081–1085. [Google Scholar] [CrossRef]
- Xu, D.; Fu, M. Detection and modeling of vegetation phenology spatiotemporal characteristics in the middle part of the Huai river region in China. Sustainability 2015, 7, 2841–2857. [Google Scholar] [CrossRef]
- Yang, Z.; Li, K.; Liu, L.; Shao, Y.; Brisco, B.; Li, W. Rice growth monitoring using simulated compact polarimetric C band SAR. Radio Sci. 2014, 49, 1300–1315. [Google Scholar] [CrossRef]
- Zhang, Y.; Li, L.; Wang, H.; Zhang, Y.; Wang, N.; Chen, J. Land surface phenology of Northeast China during 2000-2015: temporal changes and relationships with climate changes. Environ Monit Assess. 2017, 189. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Xiao, J.; Ni, P.; Zhang, J.; Wang, H.; Wang, J. Monitoring paddy rice phenology using time series MODIS data over Jiangxi Province, China. Int. J. Agric. & Biol. Eng. 2014, 7, 28–36. [Google Scholar]
- Dash, J.; Jeganathan, C.; Atkinson, P.M. The use of MERIS Terrestrial Chlorophyll Index to study spatio-temporal variation in vegetation phenology over India. Remote Sens. Environ. 2010, 114, 1388–1402. [Google Scholar] [CrossRef]
- Corcione, V.; Nunziata, F.; Mascolo, L.; Migliaccio, M. A study of the use of COSMO-SkyMed SAR PingPong polarimetric mode for rice growth monitoring. Int. J. Remote Sens. 2016, 37, 633–647. [Google Scholar] [CrossRef]
- Peng, D.; Huete, A.R.; Huang, J.; Wang, F.; Sun, H. Detection and estimation of mixed paddy rice cropping patterns with MODIS data. Int. J. Appl. Earth Obs. Geoinf. 2011, 13, 13–23. [Google Scholar] [CrossRef]
- Motohka, T.; Nasahara, K.N.; Miyata, A.; Mano, M.; Tsuchida, S. Evaluation of optical satellite remote sensing for rice paddy phenology in monsoon Asia using a continuous in situ dataset. Int. J. Remote Sens. 2009, 30, 4343–4357. [Google Scholar] [CrossRef]
- Boschetti, L.; Roy, D.P. Strategies for the fusion of satellite fire radiative power with burned area data for fire radiative energy derivation. J. Geophys. Res. Atmos. 2009, 114, 215–216. [Google Scholar] [CrossRef]
- Wang, H.; Chen, J.; Wu, Z.; Lin, H. Rice heading date retrieval based on multi-temporal MODIS data and polynomial fitting. Int. J. Remote Sens. 2012, 33, 1905–1916. [Google Scholar] [CrossRef]
- Wang, J.; Huang, J.; Wang, X.; Jin, M.; Zhou, Z.; Guo, Q.; Zhao, Z.; Huang, W.; Zhang, Y.; Song, X. Estimation of rice phenology date using integrated HJ-1 CCD and Landsat-8 OLI vegetation indices time-series images. J. Zhejiang Univ. Sci. B 2015, 16, 832–844. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Liu, X.; Su, S.; Wang, C. Retrieving canopy height and density of paddy rice from Radarsat-2 images with a canopy scattering model. Int. J. Appl. Earth Obs. Geoinf. 2014, 28, 170–180. [Google Scholar] [CrossRef]
- Bouvet, A.; Le Toan, T.; Lam-Dao, N. Monitoring of the rice cropping system in the Mekong delta using ENVISAT/ASAR dual polarization data. IEEE Trans. Geosci. Remote Sens. 2009, 47, 517–526. [Google Scholar] [CrossRef]
- Yuzugullu, O.; Erten, E.; Hajnsek, I. Rice Growth monitoring by means of X-Band co-polar SAR: Feature clustering and BBCH scale. IEEE Geosci. Remote Sens. Lett. 2015, 12, 1218–1222. [Google Scholar] [CrossRef]
- Ҫağlar, K.; Gülşen, T.; Erten, E. Paddy-rice phenology classification based on machine-learning methods using multi-temporal co-polar X-Band SAR images. IEEE J. Sel. Top. Appl. Earth Observ. Remote Sens. 2016, 9, 2509–2519. [Google Scholar]
- De Bernardis, C.G.; Vicente-Guijalba, F.; Martinez-Marin, T.; Lopez-Sanchez, J.M. Estimation of key dates and stages in rice crops using dual-polarization SAR time series and a particle filtering approach. IEEE J. Sel. Top. Appl. Earth Observ. Remote Sens. 2015, 8, 1008–1018. [Google Scholar] [CrossRef]
- Erten, E.; Lopez-Sanchez, J.M.; Yuzugullu, O.; Hajnsek, I. Retrieval of agricultural crop height from space: A comparison of SAR techniques. Remote Sens. Environ. 2017, 187, 130–144. [Google Scholar] [CrossRef]
- Koppe, W.; Gnyp, M.L.; Hütt, C.; Yao, Y.; Miao, Y.; Chen, X.; Bareth, G. Rice monitoring with multi-temporal and dual-polarimetric TerraSAR-X data. Int. J. Appl. Earth Obs. Geoinf. 2013, 21, 568–576. [Google Scholar] [CrossRef]
- Yang, Z.; Shao, Y.; Li, K.; Liu, Q.; Liu, L.; Brian, B. An improved scheme for rice phenology estimation based on time-series multispectral HJ-1A/B and polarimetric RADARSAT-2 data. Remote Sens. Environ. 2017, 195, 184–201. [Google Scholar] [CrossRef]
- Francis, C.; Shang, J.; Liu, J.; Huang, X.; Ma, B.; Jiao, X.; Geng, X.; John, M.K.; Dan, W. Tracking crop phenological development using multi-temporal polarimetric Radarsat-2 data. Remote Sens. Environ. 2017. [Google Scholar] [CrossRef]
- Tian, H.; Wu, M.; Wang, L.; Niu, Z. Mapping early, middle and late rice extent using sentinel-1A and Landsat-8 data in the poyang lake plain, China. Sensors 2018, 18, 185. [Google Scholar] [CrossRef] [PubMed]
- Inoue, Y.; Kurosu, T.; Maeno, H.; Uratsuka, S.; Kozu, T.; Dabrowska-Zielinska, K.; Qi, J. Season-long daily measurements of multifrequency (Ka, Ku, X, C, and L) and full-polarization backscatter signatures over paddy rice field and their relationship with biological variables. Remote Sens. Environ. 2002, 81, 194–204. [Google Scholar] [CrossRef]
- Wu, F.; Wang, C.; Zhang, H.; Zhang, B.; Tang, Y. Rice crop monitoring in South China with RADARSAT-2 quad-polarization SAR data. IEEE Geosci. Remote Sens. Lett. 2011, 8, 196–200. [Google Scholar] [CrossRef]
- Li, S.; Ni, P.; Cui, G.; He, P.; Liu, H.; Li, L.; Liang, Z. Estimation of rice biophysical parameters using multitemporal RADARSAT-2 images. In Proceedings of the Symposium of the International Society for Digital Earth (ISDE), Halifax, NS, Canada, 5–9 October 2015; p. 012019. [Google Scholar]
- Yang, S.; Zhao, X.; Li, B.; Hua, G. Interpreting RADARSAT-2 quad-polarization SAR signatures from rice paddy based on experiments. IEEE Geosci. Remote Sens. Lett. 2012, 9, 65–69. [Google Scholar] [CrossRef]
- Ulaby, F.; Allen, C.; Eger, G.; Kanemasu, E. Relating the microwave backscattering coefficient to leaf area index. Remote Sens. Environ. 1984, 14, 113–133. [Google Scholar] [CrossRef]
- Bouman, B. Crop parameter estimation from ground-based X-band (3-cm wave) radar backscattering data. Remote Sens. Environ. 1991, 37, 193–205. [Google Scholar] [CrossRef]
- Inoue, Y.; Sakaiya, E. Relationship between X-band backscattering coefficients from high-resolution satellite SAR and biophysical variables in paddy rice. Remote Sens. Lett. 2013, 4, 288–295. [Google Scholar] [CrossRef]
- Inoue, Y.; Sakaiya, E.; Wang, C. Capability of C-band backscattering coefficients from high-resolution satellite SAR sensors to assess biophysical variables in paddy rice. Remote Sens. Environ. 2014, 140, 257–266. [Google Scholar] [CrossRef]
- Yuzugullu, O.; Erten, E.; Hajnsek, I. Estimation of rice crop height from X- and C-Band PolSAR by Metamodel-Based optimization. IEEE J. Sel. Top. Appl. Earth Observ. Remote Sens. 2017, 10, 194–204. [Google Scholar] [CrossRef]
- Zadoks, J.C.; Chang, T.T.; Konzak, C.F. A decimal code for the growth stages of cereals. Weed Res. 1974, 14, 415–421. [Google Scholar] [CrossRef]
- Rossi, C.; Erten, E. Paddy-rice monitoring using TanDEM-X. IEEE Trans. Geosci. Remote Sens. 2015, 53, 900–910. [Google Scholar] [CrossRef]
- Pal, M.; Mather, P.M. An assessment of the effectiveness of decision tree methods for land cover classification. Remote Sens. Environ. 2003, 86, 554–565. [Google Scholar] [CrossRef]
- Francis, C.; Richard, F. ALOS PALSAR L-band polarimetric SAR data and in situ measurements for leaf area index assessment. Remote Sens. Lett. 2012, 3, 221–229. [Google Scholar]
- Congalton, R.G. Accuracy assessment and validation of remotely sensed and other spatial information. Int. J. Wildland Fire. 2001, 10, 321–328. [Google Scholar] [CrossRef]
- Cloude, S.R.; Pottier, E. An entropy based classification scheme for land applications of polarimetric SAR. IEEE Trans. Geosci. Remote Sens. 1997, 35, 68–78. [Google Scholar] [CrossRef]
- Pacheco, A.; McNairn, H.; Li, Y.; Lampropoulos, G.; Powers, J. Using RADARSAT-2 and TerraSAR-X satellite data for the identification of canola crop phenology. SPIE Remote Sens. 2016, 9998. [Google Scholar] [CrossRef]
- Wang, L.; Kong, J.; Ding, K.; Le Toan, T.; Ribbes-Baillarin, F.; Floury, N. Electromagetic scattering model for rice canopy based on Monte Carlo simulation. Prog. Electromagn. Res. 2005, 52, 153–171. [Google Scholar] [CrossRef]


| Principle Phase | BBCH | Name |
|---|---|---|
| Vegetative | 00–09 | Germination |
| 10–19 | Leaf development | |
| 20–29 | Tillering | |
| 30–39 | Stem elongation | |
| 40–49 | Booting | |
| Reproductive | 50–59 | Heading |
| 60–69 | Flowering | |
| Maturity | 70–79 | Development of fruit |
| 80–89 | Ripening | |
| 90–99 | Senescence | |
| Transplanting | 00–19 | Transplanting, recovery (rice only) |
| Acquisition Dates of SAR Datasets | |||||
|---|---|---|---|---|---|
| 15 May | 8 June | 2 July | 26 July | ||
| Fields hase | Transplanting | 17 | 0 | 0 | 0 |
| Vegetative | 10 | 30 | 16 | 0 | |
| Reproductive | 0 | 0 | 14 | 10 | |
| Maturity | 0 | 0 | 0 | 20 | |
| Decision Tree Parameters | |||
|---|---|---|---|
| VV/VH | HH/VH | HH/VV | |
| Divided Phase | T vs. V, R, or M | V vs. R or M | R vs. M |
| Threshold | 8 dB | 9.6 dB | 2.9 dB |
| Error rate | 10.8% | 19.3% | 23.2% |
| Ground Measured Phase | ||||||
|---|---|---|---|---|---|---|
| Transplanting | Vegetative | Reproductive | Maturity | UA | ||
| Extracted phase | Transplanting | 4 | 1 | 0 | 0 | 80% |
| Vegetative | 0 | 11 | 1 | 0 | 91.7% | |
| Reproductive | 0 | 2 | 5 | 0 | 71.4% | |
| Maturity | 0 | 0 | 0 | 5 | 100% | |
| PA | 100% | 78.6% | 83.3% | 100% | OA = 86.2% | |
| Kappa = 0.802 | ||||||
| Ground Measured Phase | ||||||
|---|---|---|---|---|---|---|
| Transplanting | Vegetative | Reproductive | Maturity | UA | ||
| Extracted hase | Transplanting | 4 | 0 | 0 | 0 | 100% |
| Vegetative | 0 | 13 | 1 | 0 | 92.8 % | |
| Reproductive | 0 | 1 | 5 | 0 | 83.3% | |
| Maturity | 0 | 0 | 0 | 5 | 100% | |
| PA | 100% | 92.8% | 83.3% | 100% | OA = 93.1% | |
| Kappa = 0.89 | ||||||
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
He, Z.; Li, S.; Wang, Y.; Dai, L.; Lin, S. Monitoring Rice Phenology Based on Backscattering Characteristics of Multi-Temporal RADARSAT-2 Datasets. Remote Sens. 2018, 10, 340. https://doi.org/10.3390/rs10020340
He Z, Li S, Wang Y, Dai L, Lin S. Monitoring Rice Phenology Based on Backscattering Characteristics of Multi-Temporal RADARSAT-2 Datasets. Remote Sensing. 2018; 10(2):340. https://doi.org/10.3390/rs10020340
Chicago/Turabian StyleHe, Ze, Shihua Li, Yong Wang, Leiyu Dai, and Sen Lin. 2018. "Monitoring Rice Phenology Based on Backscattering Characteristics of Multi-Temporal RADARSAT-2 Datasets" Remote Sensing 10, no. 2: 340. https://doi.org/10.3390/rs10020340
APA StyleHe, Z., Li, S., Wang, Y., Dai, L., & Lin, S. (2018). Monitoring Rice Phenology Based on Backscattering Characteristics of Multi-Temporal RADARSAT-2 Datasets. Remote Sensing, 10(2), 340. https://doi.org/10.3390/rs10020340