Enzymatic Degradation of Lignin in Soil: A Review
Abstract
:1. Introduction
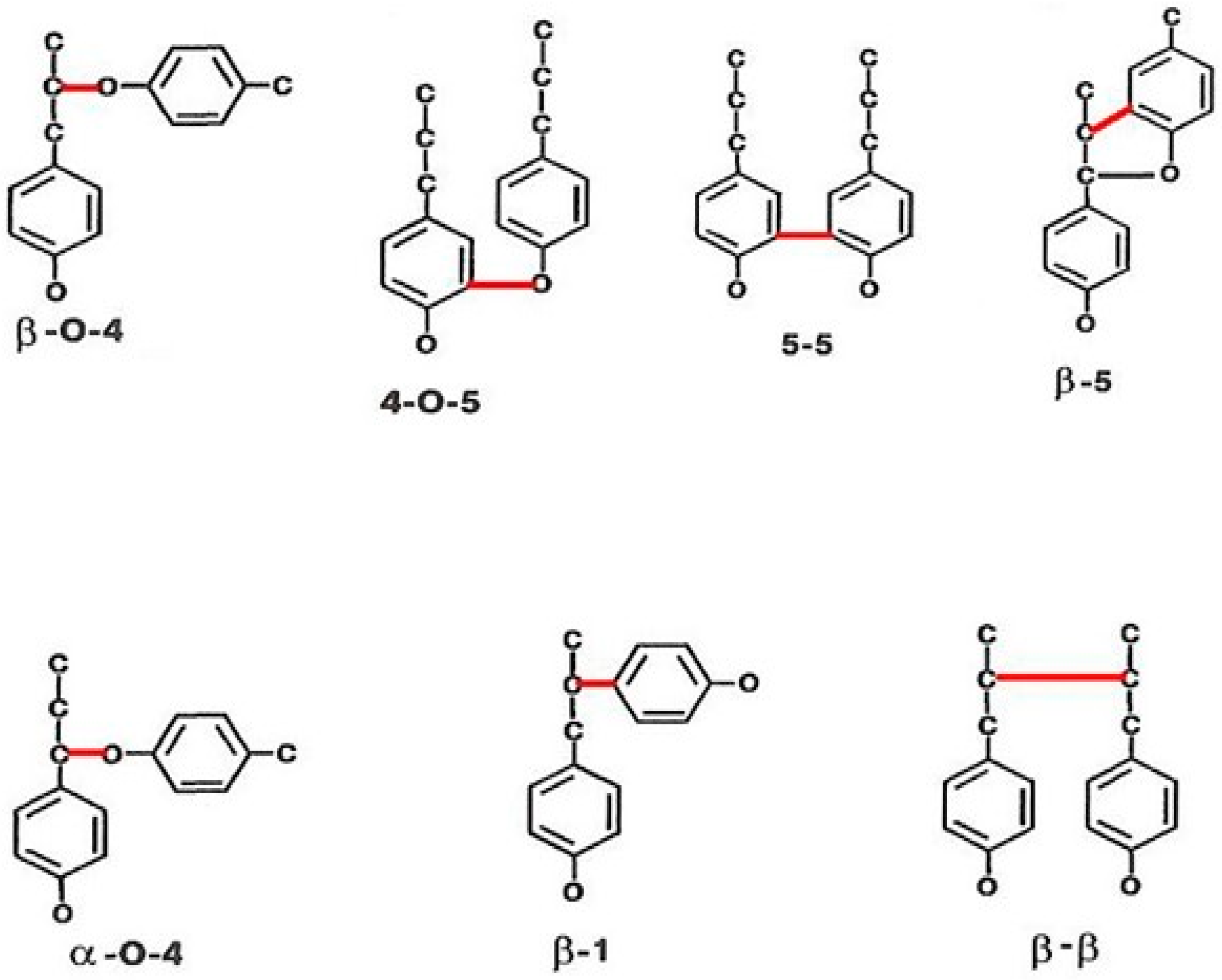
2. Lignin Structure and Its Biosynthesis
- p-Coumaryl alcohol;
- Coniferyl alcohol;
- Sinapyl alcohol.
3. Lignin in Soil
4. Lignin Distribution in Different Soil Horizons
5. Lignin Degradation in Soil
6. Steps in Lignin Degradation
- Oxidation of β–O–4 linkages to arylglycerol compounds;
- Aromatic rings cleavage, mostly follows the β–ketoadipate pathway [53];
- Cleaved aromatic rings coupled with β–O–4 oxidation leads to the formation of cyclic carbonate structures.
7. Humification
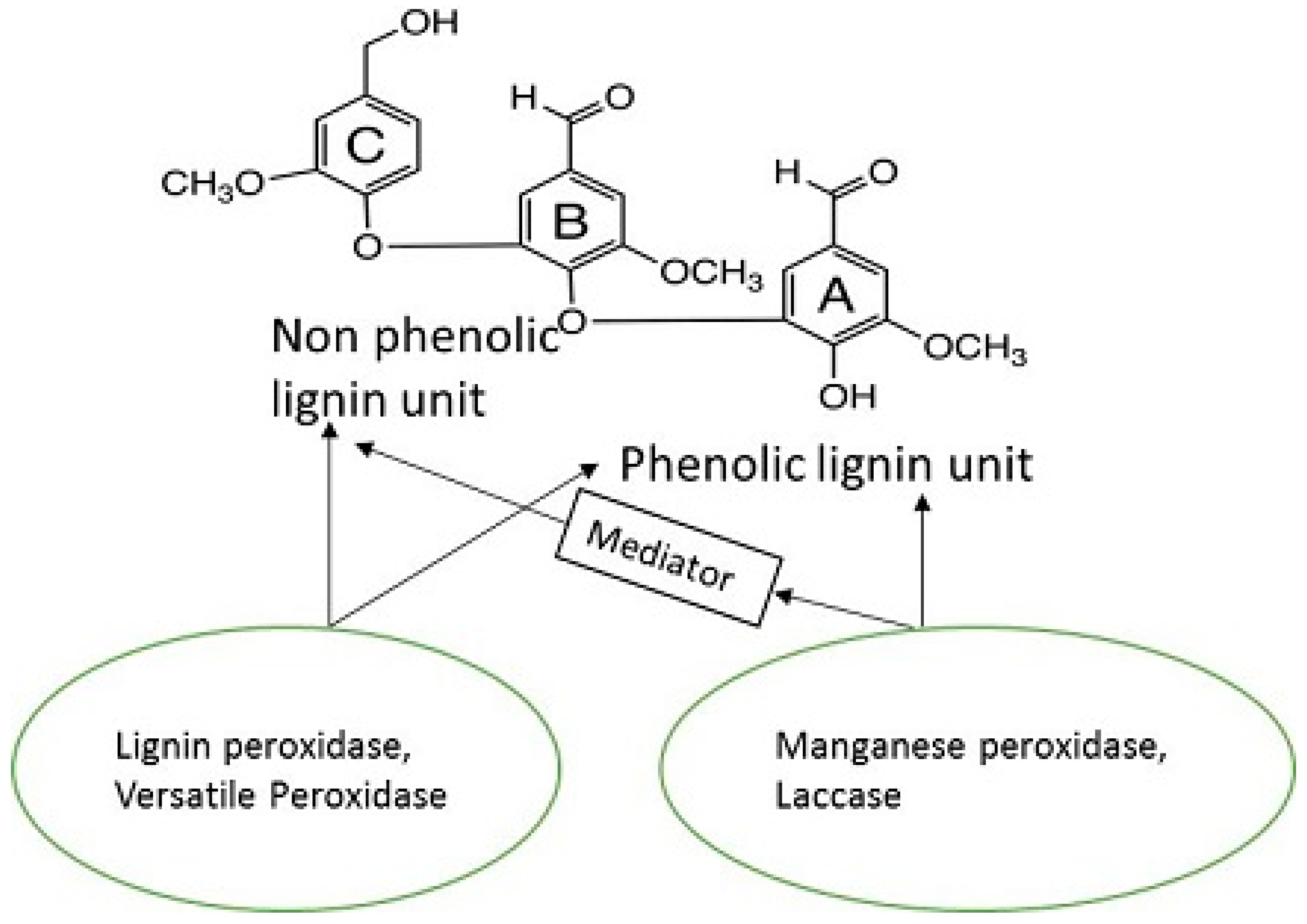
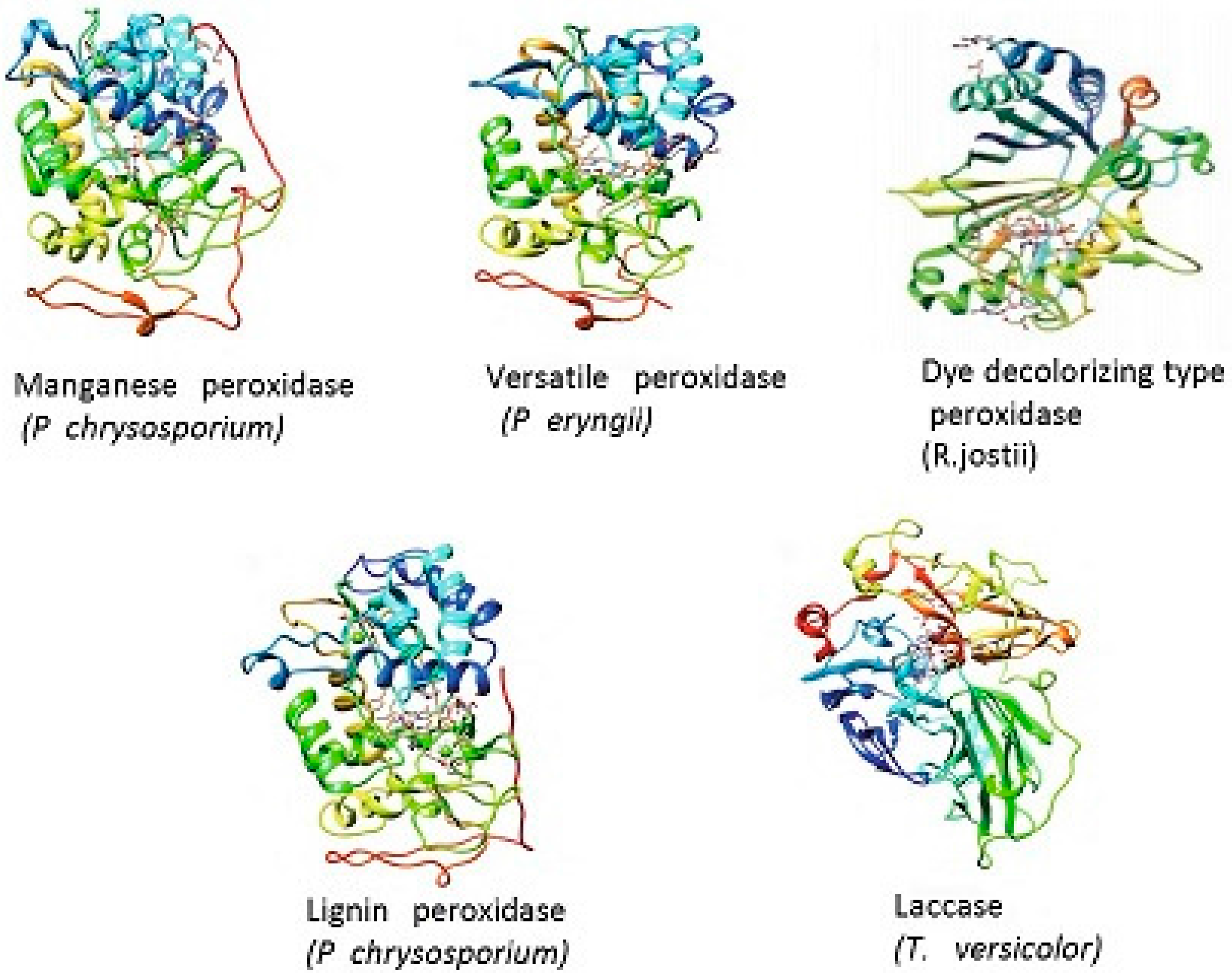
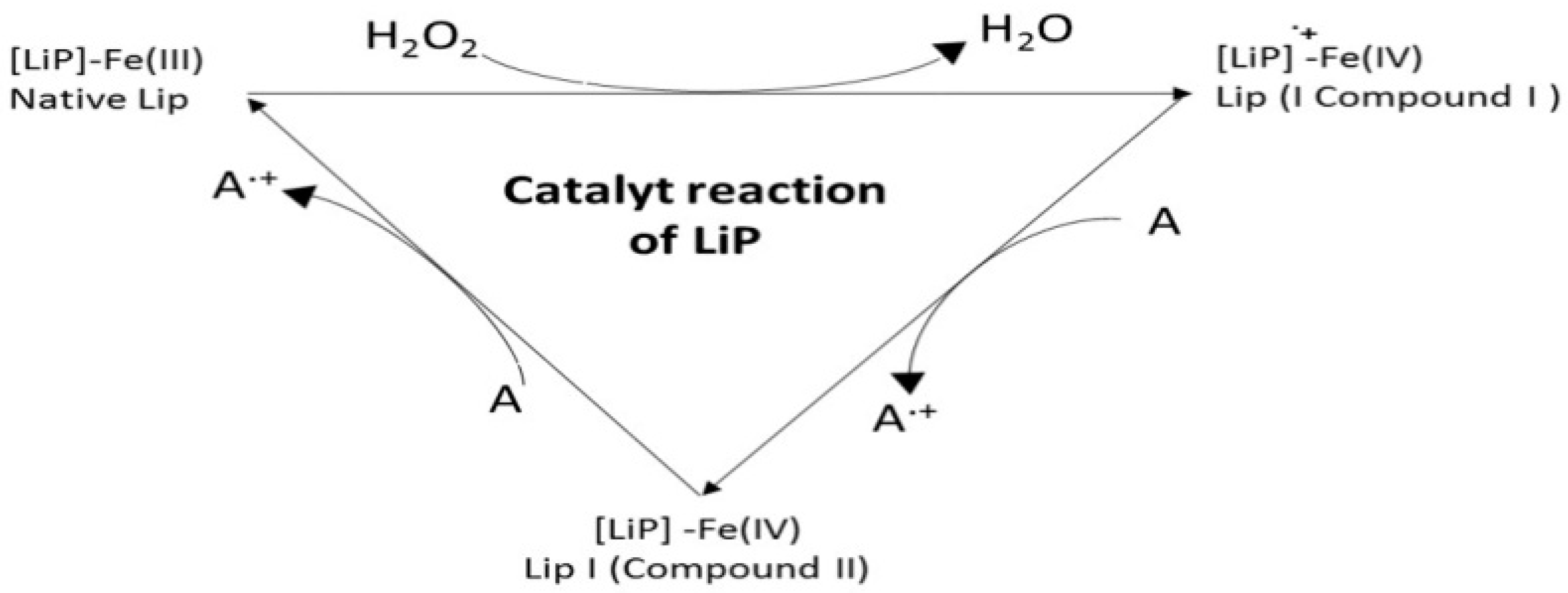
8. Lignin Peroxidase (LiP, EC 1.11.1.14)
- Step 1
- Two-electron oxidation of the resting (native) ferric enzyme ([LiP]-Fe(III)) by H2O2 to form the Compound I oxo-ferryl intermediate [Fe(IV)];
- Step 2
- Reduction of Compound I by the non-phenolic aromatic reducing substrate (A) to form Compound II by gaining one electron;
- Step 3
- Finally, the oxidation cycle ends when Compound II is returned to the resting ferric state with a gain of one more electron from the reducing substrate A.
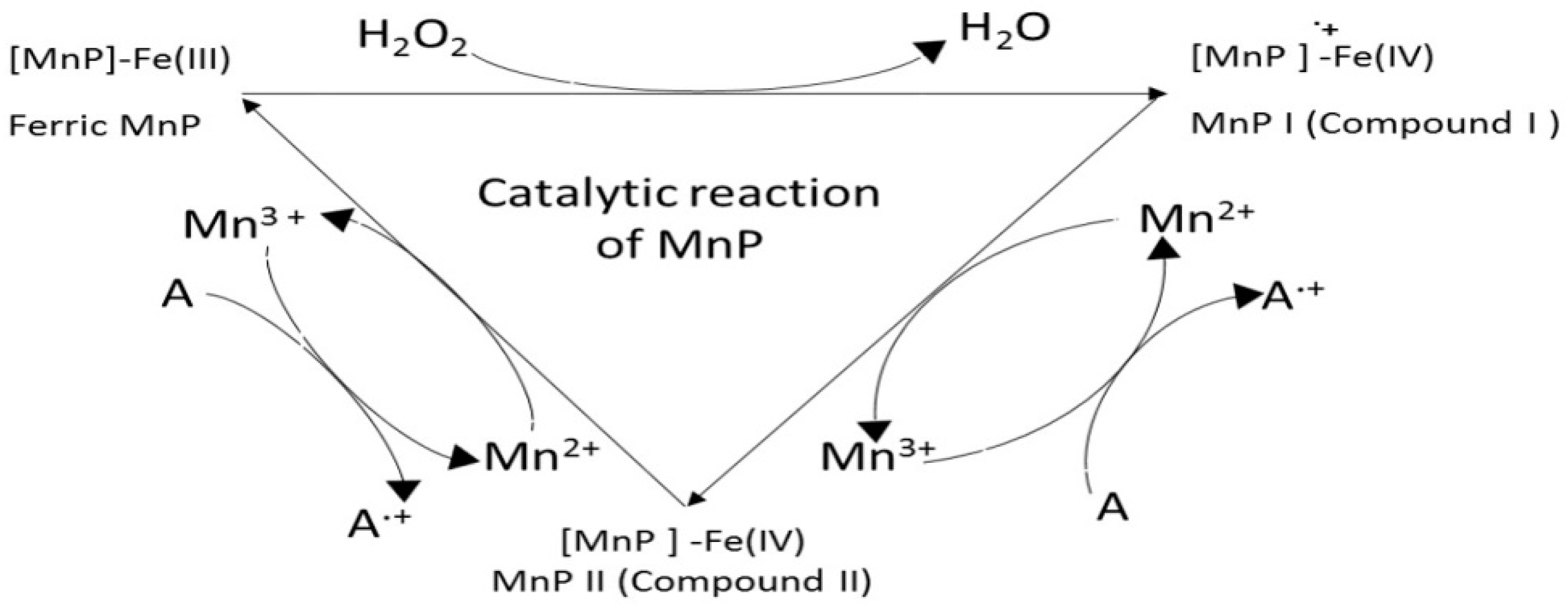
9. Manganese Peroxidase (MnP, EC 1.11.1.13)
10. Versatile Peroxidase (VP, EC 1.11.1.16)
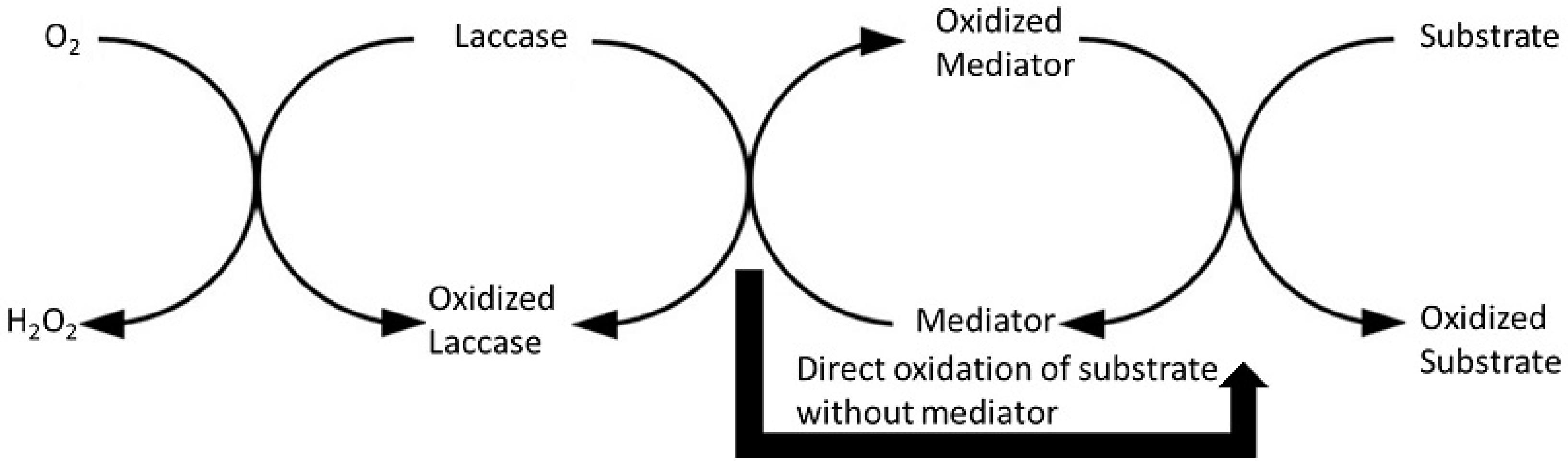
11. Laccase (Lac, E.C. 1.10.3.2)
12. Dye-Decolorizing Peroxidase (DyP, EC 1.11.1.19)
13. Fenton Chemistry in Lignin Degradation
14. Low Molecular Weight Compounds Involved in Lignin Degradation
14.1. Manganese
14.2. Veratryl Alcohol
14.3. Oxalate
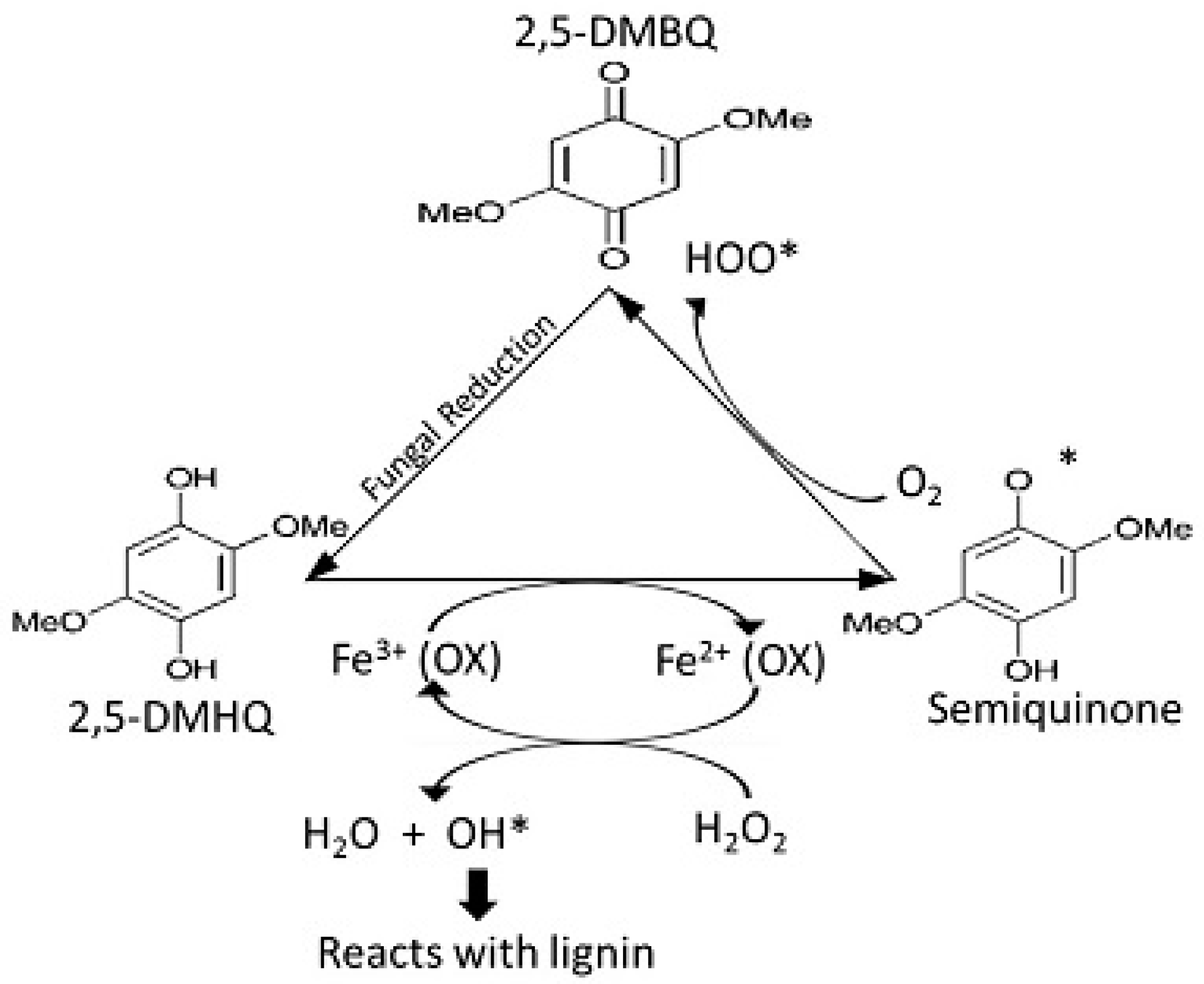
14.4. 2-Chloro-1,4-dimethoxybenzene
15. Discovery of New Lignin-Degrading Bacteria
16. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Mishra, P.K.; Wimmer, R. Aerosol assisted self-assembly as a route to synthesize solid and hollow spherical lignin colloids and its utilization in layer by layer deposition. Ultrason. Sonochem. 2016, 35, 40–50. [Google Scholar] [CrossRef] [PubMed]
- Klímek, P.; Wimmer, R.; Kumar Mishra, P.; Kúdela, J. Utilizing brewer’s-spent-grain in wood-based particleboard manufacturing. J. Clean. Prod. 2017, 141, 812–817. [Google Scholar] [CrossRef]
- Mishra, P.K.; Gregor, T.; Wimmer, R. Utilising brewer’s spent grain as a source of cellulose nanofibres following separation of protein-based biomass. Bioresources 2016, 12, 107–116. [Google Scholar] [CrossRef]
- Barder, M.J.; Crawford, D.L. Effects of carbon and nitrogen supplementation on lignin and cellulose decomposition by a Streptomyces. Can. J. Microbiol. 1981, 27, 859–863. [Google Scholar] [CrossRef] [PubMed]
- Gleixner, G.; Czimczik, C.J.; Kramer, C.; Lühker, B.; Schmidt, M.W.I. Plant compounds and their turnover and stabilization as soil organic matter. Glob. Biogeochem. Cycles Clim. Syst. 2001, 201–215. [Google Scholar] [CrossRef]
- Conesa, A.; Punt, P.J.; van den Hondel, C.A. Fungal peroxidases: Molecular aspects and applications. J. Biotechnol. 2002, 93, 143–158. [Google Scholar] [CrossRef]
- Zhao, X.; Zhang, L.; Liu, D. Biomass recalcitrance. Part I: The chemical compositions and physical structures affecting the enzymatic hydrolysis of lignocellulose. Biofuels Bioprod. Biorefin. 2012, 6, 465–482. [Google Scholar] [CrossRef]
- Sharma, P.; Jha, A.B.; Dubey, R.S.; Pessarakli, M. Reactive oxygen species, oxidative damage, and antioxidative defense mechanism in plants under stressful conditions. J. Bot. 2012, 2012, 217037. [Google Scholar] [CrossRef]
- Kubicek, C.P. The Plant Biomass. In Fungi and Lignocellulosic Biomass; Kubicek, C.P., Ed.; Wiley-Blackwell: Oxford, UK, 2012; pp. 1–28. [Google Scholar]
- Brown, M.E.; Chang, M.C.Y. Exploring bacterial lignin degradation. Curr. Opin. Chem. Biol. 2014, 19, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Kellner, H.; Vandenbol, M. Fungi unearthed: Transcripts encoding lignocellulolytic and chitinolytic enzymes in forest soil. PLoS ONE 2010, 5, e10971. [Google Scholar] [CrossRef]
- Faix, O. Classification of lignins from different botanical origins by ft-ir spectroscopy. Holzforschung 1991, 45, 21–28. [Google Scholar] [CrossRef]
- Wong, D.W.S. Structure and action mechanism of ligninolytic enzymes. Appl. Biochem. Biotechnol. 2008, 157, 174–209. [Google Scholar] [CrossRef] [PubMed]
- Parthasarathi, R.; Romero, R.A.; Redondo, A.; Gnanakaran, S. Theoretical study of the remarkably diverse linkages in lignin. J. Phys. Chem. Lett. 2011, 2, 2660–2666. [Google Scholar] [CrossRef]
- Kai, D.; Tan, M.J.; Chee, P.L.; Chua, Y.K.; Yap, Y.L.; Loh, X.J. Towards lignin-based functional materials in a sustainable world. Green Chem. 2016, 18, 1175–1200. [Google Scholar] [CrossRef]
- Stevenson, F.J. Humus Chemistry: Genesis, Composition, Reactions, 2nd ed.; John Wiley & Sons: Hoboken, NJ, USA, 1994. [Google Scholar]
- Kirk, T.K.; Connors, W.J.; Zeikus, J.G. Requirement for a growth substrate during lignin decomposition by two wood-rotting fungi. Appl. Environ. Microbiol. 1976, 32, 192–194. [Google Scholar] [PubMed]
- Haider, K. Problems related to the humification processes in soils of temperate climates. Soil Biochem. 1992, 7, 55–94. [Google Scholar]
- Guggenberger, G.; Christensen, B.T.; Zech, W. Land-use effects on the composition of organic matter in particle-size separates of soil: I. Lignin and carbohydrate signature. Eur. J. Soil Sci. 1994, 45, 449–458. [Google Scholar] [CrossRef]
- Guggenberger, G.; Zech, W. Dissolved organic carbon in forest floor leachates: Simple degradation products or humic substances? Sci. Total Environ. 1994, 152, 37–47. [Google Scholar] [CrossRef]
- Otto, A.; Simpson, M.J. Sources and composition of hydrolysable aliphatic lipids and phenols in soils from western canada. Org. Geochem. 2006, 37, 385–407. [Google Scholar] [CrossRef]
- Wiesmeier, M.; Dick, D.P.; Rumpel, C.; Dalmolin, R.S.D.; Hilscher, A.; Knicker, H. Depletion of soil organic carbon and nitrogen under Pinus taeda plantations in southern brazilian grasslands (campos). Eur. J. Soil Sci. 2009, 60, 347–359. [Google Scholar] [CrossRef]
- Sanger, L.J.; Anderson, J.M.; Little, D.; Bolger, T. Phenolic and carbohydrate signatures of organic matter in soils developed under grass and forest plantations following changes in land use. Eur. J. Soil Sci. 1997, 48, 311–317. [Google Scholar] [CrossRef]
- Feng, X.; Simpson, M.J. The distribution and degradation of biomarkers in Alberta grassland soil profiles. Org. Geochem. 2007, 38, 1558–1570. [Google Scholar] [CrossRef]
- Mason, S.L.; Filley, T.R.; Abbott, G.D. The effect of afforestation on the soil organic carbon (SOC) of a peaty gley soil using on-line thermally assisted hydrolysis and methylation (THM) in the presence of 13C-labelled tetramethylammonium hydroxide (TMAH). J. Anal. Appl. Pyrolysis 2009, 85, 417–425. [Google Scholar] [CrossRef]
- Schulten, H.-R.; Leinweber, P. Influence of long-term fertilization with farmyard manure on soil organic matter: Characteristics of particle-size fractions. Biol. Fertil. Soils 1991, 12, 81–88. [Google Scholar] [CrossRef]
- Guggenberger, G.; Zech, W.; Thomas, R.J. Lignin and carbohydrate alteration in particle-size separates of an oxisol under tropical pastures following native savanna. Soil Biol. Biochem. 1995, 27, 1629–1638. [Google Scholar] [CrossRef]
- Monreal, C.M.; Schnitzer, M.; Schulten, H.-R.; Campbell, C.A.; Anderson, D.W. Soil organic structures in macro and microaggregates of a cultivated brown chernozem. Soil Biol. Biochem. 1995, 27, 845–853. [Google Scholar] [CrossRef]
- Schöning, I.; Morgenroth, G.; Kögel-Knabner, I. O/N-alkyl and alkyl C are stabilised in fine particle size fractions of forest soils. Biogeochemistry 2005, 73, 475–497. [Google Scholar] [CrossRef]
- Kubicek, C.P. Fungi and Lignocellulosic Biomass; John Wiley & Sons: Ames, IA, USA, 2012. [Google Scholar]
- Datta, R.; Anand, S.; Moulick, A.; Baraniya, D.; Pathan, S.I.; Rejsek, K.; Varnova, V.; Sharma, M.; Sharma, D.; Kelkar, A.; et al. How enzymes are adsorbed on soil solid phase and factors limiting its activity: A Review. Int. Agrophys. 2017, 31, 287–302. [Google Scholar] [CrossRef]
- Datta, R.; Varnova, V.; Marian, P.; Rejsek, K.; Formanek, P. Effect of soil sieving on respiration induced by low-molecular-weight substrates. Int. Agrophys. 2013, 28, 119–124. [Google Scholar] [CrossRef]
- Su, Y.; Xian, H.; Shi, S.; Zhang, C.; Manik, S.M.N.; Mao, J.; Zhang, G.; Liao, W.; Wang, Q.; Liu, H. Biodegradation of lignin and nicotine with white rot fungi for the delignification and detoxification of tobacco stalk. BMC Biotechnol. 2016, 16, 81. [Google Scholar] [CrossRef] [PubMed]
- Fakoussa, R.M.; Hofrichter, M. Biotechnology and microbiology of coal degradation. Appl. Microbiol. Biotechnol. 1999, 52, 25–40. [Google Scholar] [CrossRef] [PubMed]
- Kirk, T.K.; Ibach, R.; Mozuch, M.D.; Conner, A.H.; Highley, L. Characteristics of cotton cellulose depolymerized by a brown-rot fungus, by acid, or by chemicaloxidants. Holzforschung 1991, 45, 239–244. [Google Scholar] [CrossRef]
- Kerem, Z.; Bao, W.; Hammel, K.E. Rapid polyether cleavage via extracellular one-electron oxidation by a brown-rot basidiomycete. Proc. Natl. Acad. Sci. USA 1998, 95, 10373–10377. [Google Scholar] [CrossRef] [PubMed]
- Kerem, Z.; Jensen, K.A.; Hammel, K.E. Biodegradative mechanism of the brown rot basidiomycete Gloeophyllum trabeum: Evidence for an extracellular hydroquinone-driven fenton reaction. FEBS Lett. 1999, 446, 49–54. [Google Scholar] [CrossRef]
- Hatakka, A.; Hammel, K.E. Fungal biodegradation of lignocelluloses. In Industrial Applications; Hatakka, A., Hammel, K.E., Eds.; Springer: Berlin, Germany, 2011; Volume 10, pp. 319–340. [Google Scholar]
- Spiker, J.; Crawford, D.; Thiel, E. Oxidation of phenolic and non-phenolic substrates by the lignin peroxidase of Streptomyces viridosporus T7A. Appl. Microbiol. Biotechnol. 1992, 37, 518–523. [Google Scholar] [CrossRef]
- Abdel-Hamid, A.M.; Solbiati, J.O.; Cann, I.K.O. Insights into lignin degradation and its potential industrial applications. Adv. Appl. Microbiol. 2013, 82, 1–28. [Google Scholar] [PubMed]
- Zimmermann, W. Degradation of lignin by bacteria. J. Biotechnol. 1990, 13, 119–130. [Google Scholar] [CrossRef]
- Vrsanska, M.; Buresova, A.; Damborsky, P.; Adam, V. Influence of different inducers on ligninolytic enzyme activities. J. Metastab. Nanocryst. Mater. 2015, 3, 64–70. [Google Scholar]
- Van Bloois, E.; Pazmiño, D.E.T.; Winter, R.T.; Fraaije, M.W. A robust and extracellular heme-containing peroxidase from thermobifida fusca as prototype of a bacterial peroxidase superfamily. Appl. Microbiol. Biotechnol. 2010, 86, 1419–1430. [Google Scholar] [CrossRef] [PubMed]
- Furukawa, T.; Fatai Olumide, B.; Louise, H. Microbial enzyme systems for lignin degradation and their transcriptional regulation. Front. Biol. 2014, 2014, 448–471. [Google Scholar] [CrossRef]
- Bonnen, A.M.; Anton, L.H.; Orth, A.B. Lignin-degrading enzymes of the commercial button mushroom, Agaricus bisporus. Appl. Environ. Microbiol. 1994, 60, 960–965. [Google Scholar] [PubMed]
- Lankinen, P.V.; Bonnen, A.M.; Anton, L.H.; Wood, D.A.; Kalkkinen, N.; Hatakka, A.; Thurston, C.F. Characteristics and N-terminal amino acid sequence of manganese peroxidase from solid substrate cultures of Agaricus bisporus. Appl. Microbiol. Biotechnol. 2001, 55, 170–176. [Google Scholar] [CrossRef] [PubMed]
- Steffen, K.T.; Hatakka, A.; Hofrichter, M. Degradation of humic acids by the litter-decomposing basidiomycete Collybia dryophila. Appl. Environ. Microbiol. 2002, 68, 3442–3448. [Google Scholar] [CrossRef] [PubMed]
- Tagger, S.; Périssol, C.; Gil, G.; Vogt, G.; Le Petit, J. Phenoloxidases of the white-rot fungus Marasmius quercophilus isolated from an evergreen oak litter (Quercus ilex L.). Enzym. Microb. Technol. 1998, 23, 372–379. [Google Scholar] [CrossRef]
- Gramss, G.; Kirsche, B.; Voigt, K.-D.; Günther, T.; Fritsche, W. Conversion rates of five polycyclic aromatic hydrocarbons in liquid cultures of fifty-eight fungi and the concomitant production of oxidative enzymes. Mycol. Res. 1999, 103, 1009–1018. [Google Scholar] [CrossRef]
- Lang, E.; Eller, G.; Zadrazil, F. Lignocellulose decomposition and production of ligninolytic enzymes during interaction of white rot fungi with soil microorganisms. Microb. Ecol. 1997, 34, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Heinzkill, M.; Bech, L.; Halkier, T.; Schneider, P.; Anke, T. Characterization of laccases and peroxidases from wood-rotting fungi (family Coprinaceae). Appl. Environ. Microbiol. 1998, 64, 1601–1606. [Google Scholar] [PubMed]
- Scheibner, K.; Hofrichter, M. Conversion of aminonitrotoluenes by fungal manganese peroxidase. J. Basic Microbiol. 1998, 38, 51–59. [Google Scholar] [CrossRef]
- Harwood, C.S.; Parales, R.E. The β-ketoadipate pathway and the biology of self-identity. Annu. Rev. Microbiol. 1996, 50, 553–590. [Google Scholar] [CrossRef] [PubMed]
- Alianiello, F.; Mascia, M.G.; Valentini, M.; Cozzolino, S. Humification processes of different pure organic sunstances in the soil. In Proceedings of the EGU General Assembly Conference Abstracts, Vienna, Austria, 19–24 April 2009. [Google Scholar]
- Choinowski, T.; Blodig, W.; Winterhalter, K.H.; Piontek, K. The crystal structure of lignin peroxidase at 1.70 Å resolution reveals a hydroxy group on the Cβ of tryptophan 171: A novel radical site formed during the redox cycle. J. Mol. Biol. 1999, 286, 809–827. [Google Scholar] [CrossRef] [PubMed]
- Ertan, H.; Siddiqui, K.S.; Muenchhoff, J.; Charlton, T.; Cavicchioli, R. Kinetic and thermodynamic characterization of the functional properties of a hybrid versatile peroxidase using isothermal titration calorimetry: Insight into manganese peroxidase activation and lignin peroxidase inhibition. Biochimie 2012, 94, 1221–1231. [Google Scholar] [CrossRef] [PubMed]
- Sugano, Y. DyP-type peroxidases comprise a novel heme peroxidase family. Cell. Mol. Life Sci. 2009, 66, 1387–1403. [Google Scholar] [CrossRef] [PubMed]
- Kirk, T.K.; Farrell, R.L. Enzymatic “Combustion”: The microbial degradation of lignin. Annu. Rev. Microbiol. 1987, 41, 465–501. [Google Scholar] [CrossRef] [PubMed]
- Moilanen, A.M.; Lundell, T.; Vares, T.; Hatakka, A. Manganese and malonate are individual regulators for the production of lignin and manganese peroxidase isozymes and in the degradation of lignin by Phlebia radiata. Appl. Microbiol. Biotechnol. 1996, 45, 792–799. [Google Scholar] [CrossRef]
- Vares, T.; Niemenmaa, O.; Hatakka, A. Secretion of ligninolytic enzymes and mineralization of (14)c-ring-labelled synthetic lignin by three Phlebia tremellosa strains. Appl. Environ. Microbiol. 1994, 60, 569–575. [Google Scholar] [PubMed]
- Sugiura, T.; Yamagishi, K.; Kimura, T.; Nishida, T.; Kawagishi, H.; Hirai, H. Cloning and homologous expression of novel lignin peroxidase genes in the white-rot fungus Phanerochaete sordida YK-624. Biosci. Biotechnol. Biochem. 2009, 73, 1793–1798. [Google Scholar] [CrossRef] [PubMed]
- Glenn, J.K.; Gold, M.H. Purification and characterization of an extracellular Mn(II)-dependent peroxidase from the lignin-degrading basidiomycete, Phanerochaete chrysosporium. Arch. Biochem. Biophys. 1985, 242, 329–341. [Google Scholar] [CrossRef]
- Paice, M.G.; Reid, I.D.; Bourbonnais, R.; Archibald, F.S.; Jurasek, L. Manganese peroxidase, produced by Trametes versicolor during pulp bleaching, demethylates and delignifies kraft pulp. Appl. Environ. Microbiol. 1993, 59, 260–265. [Google Scholar] [PubMed]
- Lobos, S.; Larrain, J.; Salas, L.; Cullen, D.; Vicuna, R. Isoenzymes of manganese-dependent peroxidase and laccase produced by the lignin-degrading basidiomycete Ceriporiopsis subvermispora. Microbiology 1994, 140, 2691–2698. [Google Scholar] [CrossRef] [PubMed]
- Baldrian, P. Fungal laccases—Occurrence and properties. FEMS Microbiol. Rev. 2006, 30, 215–242. [Google Scholar] [CrossRef] [PubMed]
- Martinez, M.J.; Böckle, B.; Camarero, S.; Guillén, F.; Martinez, A.T. MnP isoenzymes produced by two Pleurotus species in liquid culture and during wheat-straw solid-state fermentation. ACS Symp. Ser. 1996, 655, 183–196. [Google Scholar]
- Salame, T.M.; Knop, D.; Levinson, D.; Mabjeesh, S.J.; Yarden, O.; Hadar, Y. Release of Pleurotus ostreatus versatile-peroxidase from Mn2+ repression enhances anthropogenic and natural substrate degradation. PLoS ONE 2012, 7, e52446. [Google Scholar] [CrossRef] [PubMed]
- Grąz, M.; Jarosz-Wilkołazka, A. Oxalic acid, versatile peroxidase secretion and chelating ability of Bjerkandera fumosa in rich and limited culture conditions. World J. Microbiol. Biotechnol. 2011, 27, 1885–1891. [Google Scholar] [CrossRef] [PubMed]
- Perez, J.; Jeffries, T.W. Mineralization of 14C-ring-labeled synthetic lignin correlates with the production of lignin peroxidase, not of manganese peroxidase or laccase. Appl. Environ. Microbiol. 1990, 56, 1806–1812. [Google Scholar] [PubMed]
- Ten Have, R.; Teunissen, P.J.M. Oxidative mechanisms involved in lignin degradation by white-rot fungi. Chem. Rev. 2001, 101, 3397–3414. [Google Scholar] [CrossRef] [PubMed]
- Morgenstern, I.; Klopman, S.; Hibbett, D.S. Molecular evolution and diversity of lignin degrading heme peroxidases in the Agaricomycetes. J. Mol. Evol. 2008, 66, 243–257. [Google Scholar] [CrossRef] [PubMed]
- Wariishi, H.; Valli, K.; Gold, M.H. Oxidative cleavage of a phenolic diarylpropane lignin model dimer by manganese peroxidase from Phanerochaete chrysosporium. Biochemistry 1989, 28, 6017–6023. [Google Scholar] [CrossRef]
- Aehle, W. Enzymes in Industry: Production and Applications; John Wiley and Sons: Hoboken, NJ, USA, 2007. [Google Scholar]
- Rothschild, N.; Levkowitz, A.; Hadar, Y.; Dosoretz, C.G. Manganese deficiency can replace high oxygen levels needed for lignin peroxidase formation by Phanerochaete chrysosporium. Appl. Environ. Microbiol. 1999, 65, 483–488. [Google Scholar] [PubMed]
- Moreira, P.R.; Almeida-Vara, E.; Malcata, F.X.; Duarte, J.C. Lignin transformation by a versatile peroxidase from a novel Bjerkandera sp. strain. Int. Biodeterior. Biodegrad. 2007, 59, 234–238. [Google Scholar] [CrossRef]
- Pérez-Boada, M.; Ruiz-Dueñas, F.J.; Pogni, R.; Basosi, R.; Choinowski, T.; Martínez, M.J.; Piontek, K.; Martínez, A.T. Versatile peroxidase oxidation of high redox potential aromatic compounds: Site-directed mutagenesis, spectroscopic and crystallographic investigation of three long-range electron transfer pathways. J. Mol. Biol. 2005, 354, 385–402. [Google Scholar] [CrossRef] [PubMed]
- Camarero, S.; Sarkar, S.; Ruiz-Duenas, F.J.; Martinez, M.J.; Martinez, A.T. description of a versatile peroxidase involved in the natural degradation of lignin that has both manganese peroxidase and lignin peroxidase substrate interaction sites. J. Biol. Chem. 1999, 274, 10324–10330. [Google Scholar] [CrossRef] [PubMed]
- Wells, A.; Teria, M.; Eve, T. Green oxidations with laccase–mediator systems. Biochem. Soc. Trans. 2006, 34, 304–308. [Google Scholar] [CrossRef] [PubMed]
- Viswanath, B.; Rajesh, B.; Janardhan, A.; Kumar, A.P.; Narasimha, G. Fungal laccases and their applications in bioremediation. Enzym. Res. 2014, 2014, 163242. [Google Scholar] [CrossRef] [PubMed]
- Thurston, C.F. The structure and function of fungal laccases. Microbiology 1994, 140, 19–26. [Google Scholar] [CrossRef]
- De Gonzalo, G.; Colpa, D.I.; Habib, M.H.M.; Fraaije, M.W. Bacterial enzymes involved in lignin degradation. J. Biotechnol. 2016, 236, 110–119. [Google Scholar] [CrossRef] [PubMed]
- Ansari, S.A.; Husain, Q. Potential applications of enzymes immobilized on/in nano materials: A review. Biotechnol. Adv. 2012, 30, 512–523. [Google Scholar] [CrossRef] [PubMed]
- Adav, S.S.; Ng, C.S.; Arulmani, M.; Sze, S.K. Quantitative iTRAQ secretome analysis of cellulolytic thermobifida fusca. J. Proteome Res. 2010, 9, 3016–3024. [Google Scholar] [CrossRef] [PubMed]
- Koschorreck, K.; Richter, S.M.; Swierczek, A.; Beifuss, U.; Schmid, R.D.; Urlacher, V.B. Comparative characterization of four laccases from Trametes versicolor concerning phenolic C-C coupling and oxidation of PAHs. Arch. Biochem. Biophys. 2008, 474, 213–219. [Google Scholar] [CrossRef] [PubMed]
- Martins, L.O. molecular and biochemical characterization of a highly stable bacterial laccase that occurs as a structural component of the Bacillus subtilis endospore coat. J. Biol. Chem. 2002, 277, 18849–18859. [Google Scholar] [CrossRef] [PubMed]
- Machczynski, M.C.; Vijgenboom, E.; Samyn, B.; Canters, G.W. Characterization of SLAC: A small laccase from Streptomyces coelicolor with unprecedented activity. Protein Sci. 2004, 13, 2388–2397. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, T.; Endo, K.; Ito, M.; Tsujibo, H.; Miyamoto, K.; Inamori, Y. A thermostable laccase from Streptomyces lavendulae REN-7: Purification, characterization, nucleotide sequence, and expression. Biosci. Biotechnol. Biochem. 2003, 67, 2167–2175. [Google Scholar] [CrossRef] [PubMed]
- Margot, J.; Bennati-Granier, C.; Maillard, J.; Blánquez, P.; Barry, D.A.; Holliger, C. Bacterial versus fungal laccase: Potential for micropollutant degradation. AMB Express 2013, 3, 63. [Google Scholar] [CrossRef] [PubMed]
- Claus, H. Laccases: Structure, reactions, distributions. Micron 2004, 35, 93–96. [Google Scholar] [CrossRef] [PubMed]
- Arias, M.E.; Arenas, M.; Rodriguez, J.; Soliveri, J.; Ball, A.S.; Hernandez, M. kraft pulp biobleaching and mediated oxidation of a nonphenolic substrate by laccase from Streptomyces cyaneus cect 3335. Appl. Environ. Microbiol. 2003, 69, 1953–1958. [Google Scholar] [CrossRef] [PubMed]
- Miyazaki, K. A hyperthermophilic laccase from Thermus thermophilus HB27. Extremophiles 2005, 9, 415–425. [Google Scholar] [CrossRef] [PubMed]
- Kersten, P.J.; Stephens, S.K.; Kirk, T.K. Glyoxal oxidase and the extracellular peroxidases of Phanerochaete chrysosporium. Biotechnol. Pulp Pap. Manuf. 1990, 41, 457–463. [Google Scholar]
- Li, K.; Xu, F.; Eriksson, K.-E.L. Comparison of fungal laccases and redox mediators in oxidation of a nonphenolic lignin model compound. Appl. Environ. Microbiol. 1999, 65, 2654–2660. [Google Scholar] [PubMed]
- Call, H.P.; Mücke, I. History, overview and applications of mediated lignolytic systems, especially laccase-mediator-systems (Lignozym®-process). J. Biotechnol. 1997, 53, 163–202. [Google Scholar] [CrossRef]
- Wong, Y.; Yu, J. Laccase-catalyzed decolorization of synthetic dyes. Water Res. 1999, 33, 3512–3520. [Google Scholar] [CrossRef]
- McGuirl, M.A.; Dooley, D.M. Copper-containing oxidases. Curr. Opin. Chem. Biol. 1999, 3, 138–144. [Google Scholar] [CrossRef]
- Sugano, Y.; Muramatsu, R.; Ichiyanagi, A.; Sato, T.; Shoda, M. DyP, a unique dye-decolorizing peroxidase, represents a novel heme peroxidase family: ASP171 replaces the distal histidine of classical peroxidases. J. Biol. Chem. 2007, 282, 36652–36658. [Google Scholar] [CrossRef] [PubMed]
- Singh, R.; Eltis, L.D. The multihued palette of dye-decolorizing peroxidases. Arch. Biochem. Biophys. 2015, 574, 56–65. [Google Scholar] [CrossRef] [PubMed]
- Brown, M.E.; Barros, T.; Chang, M.C.Y. Identification and characterization of a multifunctional dye peroxidase from a lignin-reactive bacterium. ACS Chem. Biol. 2012, 7, 2074–2081. [Google Scholar] [CrossRef] [PubMed]
- Strittmatter, E.; Plattner, D.A.; Piontek, K. Dye-decolorizing peroxidase (DyP). Encycl. Inorg. Bioinorg. Chem. 2011. [Google Scholar] [CrossRef]
- Ogola, H.J.O.; Kamiike, T.; Hashimoto, N.; Ashida, H.; Ishikawa, T.; Shibata, H.; Sawa, Y. Molecular characterization of a novel peroxidase from the cyanobacterium Anabaena sp. strain PCC 7120. Appl. Environ. Microbiol. 2009, 75, 7509–7518. [Google Scholar] [CrossRef] [PubMed]
- Fernández-Fueyo, E.; Linde, D.; Almendral, D.; López-Lucendo, M.F.; Ruiz-Dueñas, F.J.; Martínez, A.T. Description of the first fungal dye-decolorizing peroxidase oxidizing manganese (II). Appl. Microbiol. Biotechnol. 2015, 99, 8927–8942. [Google Scholar] [CrossRef] [PubMed]
- Jensen, K.A.; Houtman, C.J.; Ryan, Z.C.; Hammel, K.E. Pathways for extracellular fenton chemistry in the brown rot basidiomycete Gloeophyllum trabeum. Appl. Environ. Microbiol. 2001, 67, 2705–2711. [Google Scholar] [CrossRef] [PubMed]
- Flournoy, D.S.; Paul, J.A.; Kirk, T.K.; Highley, T.L. Changes in the size and volume of pores in sweetgum wood during simultaneous rot by Phanerochaete chrysosporium Burds. Holzforschung 1993, 47, 297–301. [Google Scholar] [CrossRef]
- Bourbonnais, R.; Paice, M. Demethylation and delignification of kraft pulp by Trametes versicolor laccase in the presence of 2,2′-azinobis-(3-ethylbenzthiazoline-6-sulphonate). Appl. Microbiol. Biotechnol. 1992, 36, 823–827. [Google Scholar] [CrossRef]
- Bourbonnais, R.; Paice, M.G.; Reid, I.D.; Lanthier, P.; Yaguchi, M. Lignin oxidation by laccase isozymes from Trametes versicolor and role of the mediator 2,2′-azinobis(3-ethylbenzthiazoline-6-sulfonate) in kraft lignin depolymerization. Appl. Environ. Microbiol. 1995, 61, 1876–1880. [Google Scholar] [PubMed]
- Valli, K.; Wariishi, H.; Gold, M.H. Degradation of 2,7-dichlorodibenzo-p-dioxin by the lignin-degrading basidiomycete Phanerochaete chrysosporium. J. Bacteriol. 1992, 174, 2131–2137. [Google Scholar] [CrossRef] [PubMed]
- Mester, T.; Swarts, H.J.; i Sole, S.R.; De Bont, J.A.; Field, J.A. Stimulation of aryl metabolite production in the basidiomycete Bjerkandera sp. strain BOS55 with biosynthetic precursors and lignin degradation products. Appl. Environ. Microbiol. 1997, 63, 1987–1994. [Google Scholar] [PubMed]
- Lundquist, K.; Kirk, T.K. De novo synthesis and decomposition of veratryl alcohol by a lignin-degrading basidiomycete. Phytochemistry 1978, 17, 1676. [Google Scholar] [CrossRef]
- De Jong, E.D.; Cazemier, A.E.; Field, J.A.; de Bont, J.A.M. Physiological role of chlorinated aryl alcohols biosynthesized de novo by the white rot fungus Bjerkandera sp. strain BOS55. Appl. Environ. Microbiol. 1994, 60, 271–277. [Google Scholar] [PubMed]
- Paice, M.G.; Bourbonnais, R.; Reid, I.D.; Archibald, F.S.; Jurasek, L. Oxidative bleaching enzymes: A review. J. Pulp Pap. Sci. 1995, 21, J280–J284. [Google Scholar]
- Hammel, K.E.; Jensen, K.A.; Mozuch, M.D.; Landucci, L.L.; Tien, M.; Pease, E.A. Ligninolysis by a purified lignin peroxidase. J. Biol. Chem. 1993, 268, 12274–12281. [Google Scholar] [PubMed]
- Tuor, U.; Wariishi, H.; Schoemaker, H.E.; Gold, M.H. Oxidation of phenolic arylglycerol .beta.-aryl ether lignin model compounds by manganese peroxidase from Phanerochaete chrysosporium: Oxidative cleavage of an .alpha.-carbonyl model compound. Biochemistry 1992, 31, 4986–4995. [Google Scholar] [CrossRef] [PubMed]
- Wariishi, H.; Valli, K.; Gold, M.H. In vitro depolymerization of lignin by manganese peroxidase of Phanerochaete chrysosporium. Biochem. Biophys. Res. Commun. 1991, 176, 269–275. [Google Scholar] [CrossRef]
- Fengel, D.; Wegener, G. Wood: Chemistry, Ultrastructure, Reactions; Walter de Gruyter: Berlin Germany, 1983. [Google Scholar]
- Blanchette, R.A. Manganese accumulation in wood decayed by white rot fungi. Phytopathology 1984, 74, 725–730. [Google Scholar] [CrossRef]
- Paszczyński, A.; Huynh, V.-B.; Crawford, R. Comparison of ligninase-I and peroxidase-M2 from the white-rot fungus Phanerochaete chrysosporium. Arch. Biochem. Biophys. 1986, 244, 750–765. [Google Scholar] [CrossRef]
- Wariishi, H.; Akileswaran, L.; Gold, M.H. Manganese peroxidase from the basidiomycete Phanerochaete chrysosporium: Spectral characterization of the oxidized states and the catalytic cycle. Biochemistry 1988, 27, 5365–5370. [Google Scholar] [CrossRef] [PubMed]
- Wariishi, H.; Dunford, H.B.; MacDonald, I.D.; Gold, M.H. Manganese peroxidase from the lignin-degrading basidiomycete Phanerochaete chrysosporium. Transient state kinetics and reaction mechanism. J. Biol. Chem. 1989, 264, 3335–3340. [Google Scholar] [PubMed]
- Coassin, M.; Ursini, F.; Bindoli, A. Antioxidant effect of manganese. Arch. Biochem. Biophys. 1992, 299, 330–333. [Google Scholar] [CrossRef]
- Bonnarme, P.; Jeffries, T.W. Mn(II) regulation of lignin peroxidases and manganese-dependent peroxidases from lignin-degrading white rot fungi. Appl. Environ. Microbiol. 1990, 56, 210–217. [Google Scholar] [PubMed]
- Faison, B.D.; Kirk, T.K. Factors involved in the regulation of a ligninase activity in Phanerochaete chrysosporium. Appl. Environ. Microbiol. 1985, 49, 299–304. [Google Scholar] [PubMed]
- Mester, T.; De Jong, E.; Field, J.A. Manganese regulation of veratryl alcohol in white rot fungi and its indirect effect on lignin peroxidase. Appl. Environ. Microbiol. 1995, 61, 1881–1887. [Google Scholar] [PubMed]
- Collins, P.J.; Field, J.A.; Teunissen, P.; Dobson, A.D. Stabilization of lignin peroxidases in white rot fungi by tryptophan. Appl. Environ. Microbiol. 1997, 63, 2543–2548. [Google Scholar] [PubMed]
- Niku-Paavola, M.-L.; Karhunen, E.; Kantelinen, A.; Viikari, L.; Lundell, T.; Hatakka, A. The effect of culture conditions on the production of lignin modifying enzymes by the white-rot fungus Phlebia radiata. J. Biotechnol. 1990, 13, 211–221. [Google Scholar] [CrossRef]
- Harvey, P.J.; Schoemaker, H.E.; Palmer, J.M. Veratryl alcohol as a mediator and the role of radical cations in lignin biodegradation by Phanerochaete chrysosporium. FEBS Lett. 1986, 195, 242–246. [Google Scholar] [CrossRef]
- Dutton, M.V.; Evans, C.S.; Atkey, P.T.; Wood, D.A. Oxalate production by Basidiomycetes, including the white-rot species Coriolus versicolor and Phanerochaete chrysosporium. Appl. Microbiol. Biotechnol. 1993, 39, 5–10. [Google Scholar] [CrossRef]
- Urzúa, U.; Kersten, P.J.; Vicuña, R. Manganese peroxidase-dependent oxidation of glyoxylic and oxalic acids synthesized by Ceriporiopsis subvermispora produces extracellular hydrogen peroxide. Appl. Environ. Microbiol. 1998, 64, 68–73. [Google Scholar] [PubMed]
- Shimazono, H.; Hayaishi, O. Enzymatic decarboxylation of oxalic acid. J. Biol. Chem. 1957, 227, 151–159. [Google Scholar] [PubMed]
- Shimada, M.; Akamtsu, Y.; Tokimatsu, T.; Mii, K.; Hattori, T. Possible biochemical roles of oxalic acid as a low molecular weight compound involved in brown-rot and white-rot wood decays. J. Biotechnol. 1997, 53, 103–113. [Google Scholar] [CrossRef]
- Popp, J.L.; Kalyanaraman, B.; Kirk, T.K. Lignin peroxidase oxidation of Mn2+ in the presence of veratryl alcohol, malonic or oxalic acid, and oxygen. Biochemistry 1990, 29, 10475–10480. [Google Scholar] [CrossRef] [PubMed]
- Akamatsu, Y.; Ma, D.B.; Higuchi, T.; Shimada, M. A novel enzymatic decarboxylation of oxalic acid by the lignin peroxidase system of white-rot fungus Phanerochaete chrysosporium. FEBS Lett. 1990, 269, 261–263. [Google Scholar] [CrossRef]
- De Jong, E.; Field, J.A. Sulfur tuft and turkey tail: Biosynthesis and biodegradation of organohalogens by basidiomycetes. Annu. Rev. Microbiol. 1997, 51, 375–414. [Google Scholar] [CrossRef] [PubMed]
- Field, J.A.; Verhagen, F.J.M.; de Jong, E. Natural organohalogen production by basidiomycetes. Trends Biotechnol. 1995, 13, 451–456. [Google Scholar] [CrossRef]
- Teunissen, P.J.M.; Swarts, H.J.; Field, J.A. The de novo production of drosophilin A (tetrachloro-4-methoxyphenol) and drosophilin A methyl ether (tetrachloro-1,4-dimethoxybenzene) by ligninolytic basidiomycetes. Appl. Microbiol. Biotechnol. 1997, 47, 695–700. [Google Scholar] [CrossRef] [PubMed]
- Teunissen, P.J.M.; Field, J.A. 2-Chloro-1,4-dimethoxybenzene as a novel catalytic cofactor for oxidation of anisyl alcohol by lignin peroxidase. Appl. Environ. Microbiol. 1998, 64, 830–835. [Google Scholar] [PubMed]
- Teunissen, P.J.M.; Sheng, D.; Reddy, G.V.B.; Moënne-Loccoz, P.; Field, J.A.; Gold, M.H. 2-Chloro-1,4-dimethoxybenzene cation radical: Formation and role in the lignin peroxidase oxidation of anisyl alcohol. Arch. Biochem. Biophys. 1998, 360, 233–238. [Google Scholar] [CrossRef] [PubMed]
- Kirk, T.K.; Schultz, E.; Connors, W.J.; Lorenz, L.F.; Zeikus, J.G. Influence of culture parameters on lignin metabolism by Phanerochaete chrysosporium. Arch. Microbiol. 1978, 117, 277–285. [Google Scholar] [CrossRef]
- Fengel, D.; Wegener, G. Wood: Chemistry, ultrastructure, reactions. Walter de Gruyter 1984, 613, 1960–1982. [Google Scholar]
- Périé, F.H.; Gold, M.H. Manganese regulation of manganese peroxidase expression and lignin degradation by the white rot fungus Dichomitus squalens. Appl. Environ. Microbiol. 1991, 57, 2240–2245. [Google Scholar] [PubMed]
- Bonnarme, P.; Jeffries, T.W. Selective production of extracellular peroxidases from Phanerochaete chrysosporium in an airlift bioreactor. J. Ferment. Bioeng. 1990, 70, 158–163. [Google Scholar] [CrossRef]
- Picart, P.; Wiermans, L.; Pérez-Sánchez, M.; Grande, P.M.; Schallmey, A.; Domínguez de María, P. Assessing lignin types to screen novel biomass-degrading microbial strains: Synthetic lignin as useful carbon source. ACS Sustain. Chem. Eng. 2016, 4, 651–655. [Google Scholar] [CrossRef]
- Strachan, C.R.; Singh, R.; VanInsberghe, D.; Ievdokymenko, K.; Budwill, K.; Mohn, W.W.; Eltis, L.D.; Hallam, S.J. Metagenomic scaffolds enable combinatorial lignin transformation. Proc. Natl. Acad. Sci. USA 2014, 111, 10143–10148. [Google Scholar] [CrossRef] [PubMed]
| E.C. | LiP 1.11.1.14 | MnP 1.11.1.13 | Lac 1.10.3.2 | DyP 1.11.1.19 |
|---|---|---|---|---|
| Structure | Monomer, glycoprotein up to 15 | Monomer, glycoprotein | Mono-, di-, or tetramer, glycoprotein, | Dimer |
| Prosthetic group | Heme | Heme | Four Cu atoms per active protein unit | Heme |
| Glycosylation | N– | N– | N– | N–Acetyl-glucosamine and mannose |
| Isoforms | Monomers; up to 15 | Monomers; up to 11 | Mono-, di-, tetramers; several | Dimeric α + β barrel structure |
| pH Range | 2.0–5.0 | 2–6 | 2.0–8.5 | 3.2 (Optimum) |
| C–C Cleavage | Often | Often | No | Yes |
| H2O2 Regulated | Yes | Yes | No | Yes |
| Stability | Low | Immense | Immense | Highly |
| Natural mediators | Unknown mediators | Mn2+; Mn3+ | 3-Hydroxy-anthranilic acid | Mn2+ |
| Specificity | Broad, aromatics, incl. nonphenolics | Mn2+ | Broad, phenolics | Non-phenolics, phenolics, veratryl alcohol. |
| Catalytic center | Fe-protoporphyrin | Fe-protoporphyrin | Four copper atoms | Fe-protoporphyrin |
| Mediators | NO | Thiols, unsaturated fatty acids, organic acids as chelators, Mn3+. | Hydroxyben-zotriazole or ABTS, HBTo, syringaldazine, 3-HAA, RBB. | Chelated Mn3+ |
| Cofactor | H2O2 | H2O2 | O2− | H2O2 |
| Substrate | Halogenated phenolic compounds, polycyclic aromatic compounds | Lignin and other phenolic compounds | Ortho- and para-diphenols, aminophenols, polyphenols, polyamines, lignins, and aryl diamines | Phenols, hydroquinones, dyes, amines, aromatic alcohols and xenobiotics. |
| Fungal Species | Phylum | Family | Mol. Wt (kDa) | Culture Medium | Reference |
|---|---|---|---|---|---|
| Agaricus bisporus | Basidiomycota | Agaricaceae | 40 | Solid | [45,46] |
| Agrocybe praecox | Basidiomycota | Bolbitiaceae | 41–42 | Liquid | [47] |
| Collybia dryophila | Basidiomycota | Tricholomataceae | 43 | Liquid, solid | [47] |
| Marasmius quercophilus | Basidiomycota | Tricholomataceae | ND | Liquid | [48] |
| Phallus impudicus | Basidiomycota | Phlallaceae | ND | Liquid | [49] |
| Pleurotus sp. Bhutan | Basidiomycota | Lentinaceae | ND | Solid | [50] |
| Panaeolus sphinctrinus | Basidiomycota | Strophariaceae | 42 | Liquid | [51] |
| Stropharia aeruginosa | Basidiomycota | Strophariaceae | ND | Liquid | [47] |
| Stropharia coronilla | Basidiomycota | Strophariaceae | 40–43 | Liquid | [47] |
| Stropharia cubensis | Basidiomycota | Strophariaceae | ND | Liquid | [47] |
| Stropharia rugosoannulata | Basidiomycota | Strophariaceae | 41–43 | Liquid, solid | [47,52] |
| Nematoloma frowardii | Basidiomycota | Strophariaceae | 42–44 | liquid | [47] |
| Enzyme | Fungi | Reference |
|---|---|---|
| DyP | Auricularia auricular-judae | [57] |
| LiP | Phanerochaete chrysosporium | [58] |
| Lip | Phlebia radiata | [59] |
| LiP | P. tremellosa | [60] |
| MnP | Phanerochaete sordida | [61] |
| MnP | P.chrysosporium | [62] |
| MnP | Trametes versicolor | [63] |
| MnP | Ceriporiopsis subvermispora | [64] |
| Lac | P. radiata | [65] |
| Lac | C.subvermispora | [65] |
| Lac | Pleurotus eryngii | [65] |
| Lac | T. versicolor | [65] |
| Lac | T.hirsuta | [65] |
| Lac | T. ochracea | [65] |
| VP | P. eryngii | [66] |
| VP | Pleurotus ostreatus | [67] |
| VP | Bjerkandera fumosa | [68] |
| Ligninolytic Enzyme | Bacteria | Reference |
|---|---|---|
| DyP A | Amycolatopsis sp. | [81] |
| DyP A | Escherichia coli | [81] |
| DyP A | Rhodococcus jostii | [82] |
| DyP A | Steptomyces viridosporus | [81] |
| DyP A | S. coelicolor | [81] |
| DyP A | S. viridosporus | [81] |
| DyP A | Thermobifida fusca | [83] |
| DyP A | T. fusca YX | [81] |
| DyP B | Escherichia coli | [81] |
| DyP B | Pseudomonas sp. | [81] |
| Dyp B | Rhodococcus jostii | [82] |
| DyP B | R. jostii | [81] |
| DyP B | S. coelicolor | [81] |
| Laccase | Bacillus atrophaeus | [81] |
| Laccase | B. licheniformis | [84] |
| Laccase | B. pumilus | [81] |
| Laccase | B. subtilis | [85] |
| Laccase | S. coelicolor | [86] |
| Laccase | S. griseus | [87] |
| Laccase | S. ipomoea | [88] |
| Laccase | S. lavendulae | [89] |
| Laccase | Streptomyces cyaneus | [90] |
| Laccase | Thermus thermophilus | [91] |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Datta, R.; Kelkar, A.; Baraniya, D.; Molaei, A.; Moulick, A.; Meena, R.S.; Formanek, P. Enzymatic Degradation of Lignin in Soil: A Review. Sustainability 2017, 9, 1163. https://doi.org/10.3390/su9071163
Datta R, Kelkar A, Baraniya D, Molaei A, Moulick A, Meena RS, Formanek P. Enzymatic Degradation of Lignin in Soil: A Review. Sustainability. 2017; 9(7):1163. https://doi.org/10.3390/su9071163
Chicago/Turabian StyleDatta, Rahul, Aditi Kelkar, Divyashri Baraniya, Ali Molaei, Amitava Moulick, Ram Swaroop Meena, and Pavel Formanek. 2017. "Enzymatic Degradation of Lignin in Soil: A Review" Sustainability 9, no. 7: 1163. https://doi.org/10.3390/su9071163
APA StyleDatta, R., Kelkar, A., Baraniya, D., Molaei, A., Moulick, A., Meena, R. S., & Formanek, P. (2017). Enzymatic Degradation of Lignin in Soil: A Review. Sustainability, 9(7), 1163. https://doi.org/10.3390/su9071163








