Comparative Physicochemical and Microbiological Qualities of Source and Stored Household Waters in Some Selected Communities in Southwestern Nigeria
Abstract
:1. Introduction
2. Materials and Methods
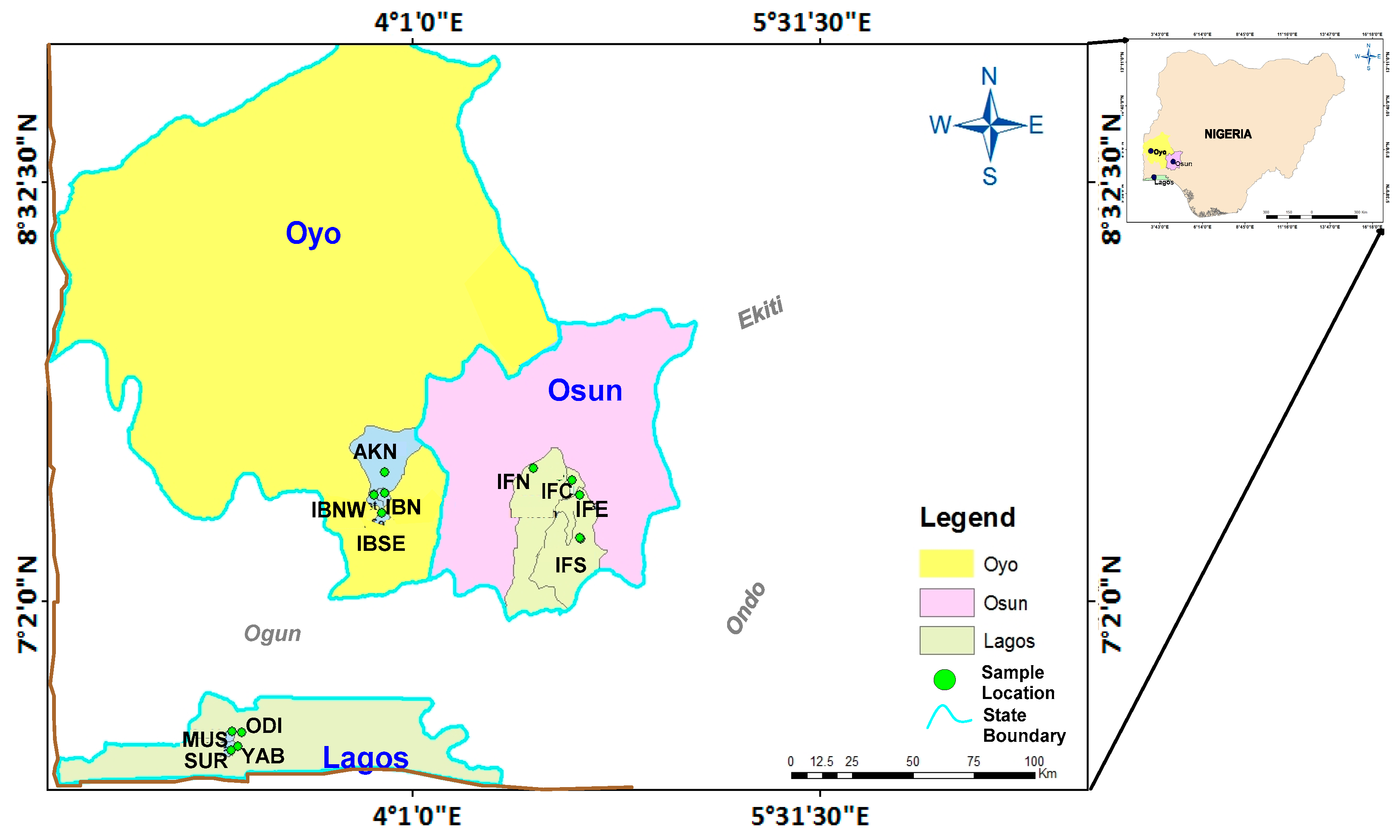
2.1. Study Area and Sampling
2.2. Physicochemical Analyses of Collected Samples
2.3. Microbiological Analyses of Collected Samples
2.4. Statistical Analysis
3. Results
3.1. Physicochemical Analysis of Source and Stored Samples
3.2. Microbial Analysis of Source and Stored Samples
4. Discussion
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- The United Nations International Children’s Emergency Fund (UNICEF). World Water Day 2005: 4000 Children Die Each Day from a Lack of Safe Water. Available online: http://www.unicef.org/wash/index_25637.html (accessed on 12 December 2016).
- Onabolu, B.; Jimoh, O.D.; Igboro, S.B.; Sridhar, M.K.C.; Onyilo, G.; Gege, A.; Ilya, R. Source to point of use drinking water changes and knowledge, attitude and practices in Katsina State, Northern Nigeria. Phys. Chem. Earth 2011, 36, 1189–1196. [Google Scholar] [CrossRef]
- Wateraid.org. WaterAid—Water Charity News. 2016. Available online: http://www.wateraid.org/ng#sthash.qBgz2PhY.dpuf (accessed on 20 December 2016).
- Karavoltsosa, S.; Sakellaria, A.; Mihopoulosb, N.; Dassenakisa, M.; Scoullosa, M.J. Evaluation of the quality of drinking water in regions of Greece. Desalination 2008, 224, 317–329. [Google Scholar] [CrossRef]
- Manish, U.; Bed, L.; Om Prakas, P. Degradation of water quality due to heavy pollution in industrial area of Korba, Chhattisgarh. Recent Res. Sci. Technol. 2013, 5, 37–39. [Google Scholar]
- U.S. Environmental Protection Agency (USEPA). Protecting Water Quality from Agricultural Runoff. Fact Sheet No.EPA-841-F-05-001. Available online: http://www.epa.gov./owow/nps/Ag_Runoff Fact_Sheet.pdf (accessed on 6 November 2016).
- Manish, U.; Sudhir, P. Analysis of surface and industrial waste water in Sipat industrial area in Bilaspur district, Chhattisgarh, India. Int. J. Pharm. 2016, 6, 74–77. [Google Scholar]
- Schueler, T.R. Microbes and Urban Watershed: Concentration, Source and Pathway. 2000. Reprinted in The Practice of Watershed Protection. Available online: http//www.cwp.org/resource_library/center_docs (accessed on 7 November 2016).
- World Health Organization (WHO). Guidelines for Drinking-Water Quality. 2011. Available online: http://www.who.int/water_sanitation_health/publications/2011/dwq_guidelines/en/ (accessed on 5 November 2016).
- Hogan, C.M. Water Pollution. Encyclopedia of Earth; National Council on Science and the Environment: Washington, DC, USA, 2010; Available online: http//www.oearth.org/article/Water_pollution (accessed on 20 November 2016).
- U.S. Environmental Protection Agency (EPA). Report to Congress: Impacts and Control of CSOs and SSOs. August 2004; Document No.EPA-833-R-04-001; 2004. Available online: http://cfpub.epa.gov/npdes/cso/cpolicy_report 2004.cfm (accessed on 6 November 2016).
- World Health Organization (WHO); The United Nations International Children’s Emergency Fund (UNICEF). Joint Monitoring Programme for Water Supply and Sanitation. In Progress on Drinking Water and Sanitation: 2014 Update; UNICEF: New York, NY, USA, 2014. [Google Scholar]
- World Health Organization (WHO). Pathogenic Mycobacteria in Water: A Guide to Public Health Consequences, Monitoring and Management; Pedley, S., Batram, J., Rees, G., Dufuor, A., Cotruvo, J., Eds.; IWA Publishing: London, UK, 2004. [Google Scholar]
- Roohul-Amin, S.A.; Jabar, Z.K. Microbial analysis of drinking water distribution in New Urban Peshawar. Curr. Res. J. Biol. Sci. 2012, 4, 731–737. [Google Scholar]
- Xu, H.; Yang, L.; Zhao, G.; Jiao, J.; Yin, S.; Liu, Z. Anthropogenic Impact on Surface Water Quality in Taihu Lake Region, China. Pedosphere 2009, 19, 765–778. [Google Scholar] [CrossRef]
- Azrina, M.Z.; Yap, C.K.; Rahim-Ismail, A.; Ismail, A.; Tan, S.G. Anthropogenic impacts on the distribution and biodiversity of benthic macroinvertebrates and water quality of the Langat River, Peninsular Malaysia. Ecotoxicol. Environ. Saf. 2006, 64, 337–347. [Google Scholar] [CrossRef] [PubMed]
- Annalakshmi, G.; Amsath, A. Nutrient status of Arasalar River, a tributary of Cauvery river at Tanjore district of Tamilnadu, India. Int. J. Plant Anim. Environ. Sci. 2012, 2, 214–222. [Google Scholar]
- Okonko, I.O.; Ogunnusi, T.A.; Adejoye, O.D.; Shittu, O.B. Microbiological and Physicochemical Analysis of Different Water Samples Used for Domestic Purposes in Abeokuta, Ogun State and Ojota, Lagos State, Nigeria. Afr. J. Biotechnol. 2008, 7, 617–621. [Google Scholar]
- Okonko, I.O.; Ogunjobi, A.A.; Adejoye, O.D.; Ogunnusi, T.A.; Olasogba, M.C. Comparative studies and Microbial risk assessment of different water samples used for processing frozen sea-foods in Ijora-Olopa, Lagos State, Nigeria. Afr.J. Biotechnol. 2008, 7, 2902–2929. [Google Scholar]
- Patil, V.T.; Patil, P.R. Physicochemical Analysis of Selected Groundwater Samples of Amalner Town in Jalgaon District, Maharashtra, India. Int. J. Adv. Earth Sci. Eng. 2010, 7, 111–116. [Google Scholar]
- Grisey, E.; Belle, E.; Dat, J.; Mudry, J.; Aleya, L. Survival of pathogenic and indicator organisms in groundwater and landfill leachate through coupling bacterial enumeration with tracer tests. Desalination 2010, 261, 162–168. [Google Scholar] [CrossRef]
- Julie, D.; Solen, L.; Antoine, V.; Jaufrey, C.; Annick, D.; Dominique, H.H. Ecology of pathogenic and non-pathogenic Vibrio parahaemolyticus on the French Atlantic coast. Effects of temperature, salinity, turbidity and chlorophyll. Environ. Microbiol. 2010, 12, 929–937. [Google Scholar] [CrossRef] [PubMed]
- Eniola, K.I.T.; Olayemi, A.B.; Adegoke, A.; Abolade, O.O.; Kayode-Ishola, T.M. Effect of storage on the bacteriological quality of well water. Afr. J. Clin. Exp. Microbiol. 2006, 2, 27–32. [Google Scholar]
- Eniola, K.I.T.; Obafemi, D.Y.; Awe, S.F.; Yusuf, I.I.; Falaiye, O.A. Effect of container and storage conditions on the bacteriological quality of borehole water in Nigeria. J. Microbiol. 2007, 21, 1578–1585. [Google Scholar]
- Wright, J.; Gundry, S.; Conroy, R. Household drinking water in developing countries: A systematic review of microbiological contamination between source and point-of-use. Trop. Med. Health 2004, 9, 106–117. [Google Scholar] [CrossRef]
- Trevett, A.F.; Carter, R.C.; Tyrrel, S.F. Mechanisms leading to post-supply water quality deterioration in rural Honduran communities. Int. J. Hyg. Environ. Health 2005, 208, 153–161. [Google Scholar] [CrossRef] [PubMed]
- Ravichandran, P.; Subha, K.; Sugumaran, P.; Unnamalai, N. Effect of Storage Containers on Coliforms in Household Drinking Water. Int. J. Microbiol. App. Sci. 2016, 5, 461–477. [Google Scholar]
- Jensen, P.P.; Ensink, J.H.J.; Jayasinghe, G.; van der Hoek, W.; Cairncross, S.; Dalsgaard, A. Domestic transmission routes of pathogens: The problem of in-house contamination of drinking water during storage in developing countries. Trop. Med. Int. Health 2002, 7, 604–609. [Google Scholar] [CrossRef] [PubMed]
- Onigbogi, O.; Ogunyemi, O. Effect of Storage Containers on Quality of Household Drinking Water in Urban Communities in Ibadan, Nigeria. Int.J. Public Health Sci. 2014, 3, 253–258. [Google Scholar] [CrossRef]
- World Health Organization (WHO); The United Nations International Children’s Emergency Fund (UNICEF). Progress on Drinking Water and Sanitation: 2012. Update; World Health Organization, Geneva and United Nations Children’s Fund: New York, NY, USA, 2012. Available online: https://www.unicef.org/media/files/JMPreport2012.pdf (accessed on 5 November 2016).
- Benjamin, A.P.; Brown, R. Encyclopaedia of Food Science and Nutrition, 2nd ed.; Academic Press: Kent, UK, 2003; Volume 10. [Google Scholar]
- The American Public Health Association (APHA); the American Water Works Association (AWWA); the Water Environment Federation (WEF). Standard Methods for the Examination of Water and Wastewater; APHA, AWWA and WEF: Washington, DC, USA, 2005. [Google Scholar]
- Food and Agricultural Organization (FAO). Chemical Analysis Manual for Food and Water, 5th ed.; FAO: Rome, Italy, 1997; Volume 1, pp. 20–26. [Google Scholar]
- Ademoroti, C.M.A. Standard Methods for Water and Effluent Analysis; Foludex Press, Ltd.: Ibadan, Nigeria, 1996; p. 182. [Google Scholar]
- Buchanan, R.E.; Gibbons, N.E. Bergey’s Manual of Determinative Bacteriology, 8th ed.; The Williams and Wilkins Company: Baltimore, MD, USA, 1994. [Google Scholar]
- Watanabe, T. Pictorial Atlas of Soil and Seed Fungi; Kluwer Academic Publishers: Dordrecht, The Netherlands, 2002. [Google Scholar]
- Pommepuy, M.; Guillaud, J.F.; Dupray, E.; Derrien, A.; Le Guyader, F.; Cormier, M. Enteric bacteria survival factors. Water Sci. Technol. 1992, 25, 93–103. [Google Scholar]
- World Health Organization (WHO). Guidelines for Drinking Water Quality, Incorporating 1st and 2nd Addenda, Vol. 1, Recommendations, 3rd ed.; WHO: Geneva, Switzerland, 2008. [Google Scholar]
- Rogbesan, A.A.; Eniola, K.I.T.; Olayemi, A.B. Bacteriological Examination of some Boreholes within University of Ilorin (PS). Niger. J. Pure Appl. Sci. 2002, 17, 1223–1226. [Google Scholar]
- Sibanda, T.; Chigor, V.N.; Koba, S.; Obi, C.L.; Okoh, A.I. Characterisation of the physicochemical qualities of a typical rural-based river: Ecological and public health implications. Int. J. Environ. Sci. Technol. 2014, 11, 1771–1780. [Google Scholar] [CrossRef]
- Oluyemi, E.A.; Adekunle, A.S.; Adenuga, A.A.; Makinde, W.O. Physico-chemical properties and heavy metal content of water sources in Ife North Local Government Area of Osun State, Nigeria. Afr. J. Environ. Sci. Technol. 2010, 4, 691–697. [Google Scholar]
- U.S. Environmental Protection Agency (USEPA). Volunteer Stream Monitoring: A Methods Manual. USEPA 841-B-97-003. U.S. Environmental Protection Agency. Methods for Volunteer Monitoring of Streams. 1997. Available online: http://www.usepa.gov/owow/monitoring/ volunteer (accessed on 5 November 2016). [Google Scholar]
- Shittu, O.B.; Olaitan, J.O.; Amusa, T.S. Physico-Chemical and Bacteriological Analysis of Water Used for Drinking and Swimming Purpose. Afr. J. Biol. Res. 2008, 11, 285–290. [Google Scholar]
- U.S. Environmental Protection Agency (EPA). Volunteer Lake Monitoring: A Methods Manual; EPA 440/4-91-002; Office of Water US Environ-Mental Protection Agency: Washington, DC, USA, 1999; p. 65. [Google Scholar]
- Moore, A.C.; Herwaldt, B.L.; Craun, G.F.; Calderon, A.K. Waterborne disease in the United States, 1991 and 1992. J. Am. Water Work. Assoc. 1994, 86, 87–99. [Google Scholar]
- Pickering, A.J.; Davis, J.; Walters, S.P.; Horak, H.M..; Keymer, D.P.; Mushi, D.; Rachelle, S.; Joshua, S.C.; Jesse, L.; Annalise, B.; et al. Hands, water, and health: Fecal contamination in Tanzanian communities with improved, non-networked water supplies. Environ. Sci. Technol. 2010, 44, 3267–3272. [Google Scholar] [CrossRef] [PubMed]
- Pickering, A.J.; Julian, T.R.; Mamuya, S.; Boehm, A.B.; Davis, J. Bacterial hand contamination among Tanzanian mothers varies temporally and following household activities. Trop. Med. Int. Health 2011, 16, 233–239. [Google Scholar] [CrossRef] [PubMed]
- Schets, F.M.; During, M.; Italiaander, R.; Heijnen, L.; Rutjes, S.A.; van der Zwaluw, W.K.; de RodaHusman, A.M. Escherichia coli O157:H7 in drinking water from private water supplies in the Netherlands. Water Res. 2005, 39, 4485–4493. [Google Scholar] [CrossRef] [PubMed]
- Göttlich, E.; van der Lubbe, W.; Lange, B.; Fiedler, S.; Melchert, I.; Reifenrath, M.; Flemming, H.C.; de Hoog, S. Fungal flora in groundwater-derived public drinking water. Int. J. Hyg. Environ. Health 2002, 205, 269–279. [Google Scholar] [CrossRef] [PubMed]
- Paterson, R.R.M.; Hageskal, G.; Skaar, I.; Lima, N. Occurrence, problems, analysis and removal of filamentous fungi in drinking water. In Fungicides: Chemistry, Environmental Impacts and Health Effects; De Costa, P., Bezerra, P., Eds.; Nova Science Publishers, Inc.: Hauppaugen, NY, USA, 2009. [Google Scholar]
- Anaissie, E.J.; Stratton, S.L.; Dignani, M.C.; Summerbell, R.C.; Rex, J.H.; Monson, T.P.; Spencer, T.; Kasai, M.; Francesconi, A.; Walsh, T.J. Pathogenic Aspergillus species recovered from a hospital water system: A 3-year prospective study. Clin. Infect. Dis. 2002, 34, 780–789. [Google Scholar] [CrossRef] [PubMed]
- De Rosa, F.G.; Garazzino, S.; Pasero, D.; Di Perri, G.; Ranieri, V.M. Invasive candidiasis and candidemia: New guidelines. Minerva Anestesiol. 2009, 75, 453–458. [Google Scholar] [PubMed]
- Hageskal, G.; Lima, N.; Skaar, I. The study of fungi in drinking water. Mycol. Res. 2009, 113, 165–172. [Google Scholar] [CrossRef] [PubMed]
| Parameter | Oyo State | Osun State | Lagos State | WHO Limits | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Stored (45) | Source (15) | F | P | Stored (45) | Source (15) | F | P | Stored (45) | Source (15) | F | P | ||
| pH | 6.9 ± 0.1 | 6.8 ± 0.1 | 0.1 | 0.74 | 6.5 ± 0.2 | 6.4 ± 0.2 | 0.3 | 0.58 | 6.9 ± 0.1 | 6.7 ± 0.1 | 2.0 | 0.16 | 6.5–8.5 |
| Temperature(°C) | 16.7 ± 0.4 | 15.8 ± 0.2 | 4.7 | 0.03 | 24.5 ± 0.8 | 25.0 ± 0.9 | 0.2 | 0.66 | 24.5 ± 0.3 | 24.5 ± 0.3 | 0.0 | 1.00 | 25–30 |
| TDS (mg/L) | 355.3 ± 22.4 | 373.2 ± 23.2 | 0.3 | 0.58 | 349.9 ± 29.2 | 376.0 ± 26.0 | 0.5 | 0.51 | 368.3 ± 20.9 | 377.1 ± 21.2 | 0.1 | 0.77 | 500 |
| Turbidity(NTU) | 19.4 ± 2.2 | 24.3 ± 2.6 | 2.1 | 0.30 | 10.4 ± 1.0 | 12.7 ± 1.2 | 2.6 | 0.16 | 17.0 ± 2.1 | 20.8 ± 2.7 | 1.3 | 0.26 | <5 |
| DO(mgO2/L) | 1.2 ± 0.1 | 1.1 ± 0.1 | 0.0 | 1.0 | 3.6 ± 0.3 | 2.9 ± 0.2 | 3.8 | 0.06 | 4.8 ± 0.2 | 4.3 ± 0.4 | 1.1 | 0.30 | ≥5 |
| BOD(mgO2/L) | 0.8 ± 0.1 | 0.8 ± 0.1 | 0.0 | 0.90 | 1.6 ± 0.1 | 1.2 ± 0.1 | 3.8 | 0.05 | 2.7 ± 0.2 | 2.7 ± 0.3 | 0.0 | 0.90 | - |
| Total hardness(mg/L) | 177.3 ± 15.7 | 202.2 ± 18.1 | 1.1 | 0.30 | 134.2 ± 12.1 | 118.2 ± 12.0 | 0.9 | 0.35 | 67.5 ± 12.2 | 51.8 ± 9.5 | 1.0 | 0.31 | 500 |
| Calcium ion (mg/L) | 34.2 ± 3.2 | 38.9 ± 3.8 | 0.9 | 0.35 | 24.5 ± 2.9 | 21.9 ± 3.3 | 0.6 | 0.56 | 13.1 ± 2.2 | 7.6 ± 1.4 | 4.5 | 0.03 | - |
| Magnesium ion(mg/L) | 23.0 ± 2.3 | 25.7 ± 2.5 | 0.6 | 0.44 | 17.9 ± 1.7 | 15.3 ± 1.3 | 1.7 | 0.21 | 9.5 ± 1.3 | 6.4 ± 0.8 | 4.4 | 0.04 | - |
| States | Osun | Lagos | Oyo | WHO Limits | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Points of Collection (No. of Samples) | Stored (45) | Source (15) | F | P | Stored (45) | Source (15) | F | P | Stored (45) | Source (15) | F | P | ||
| Microbial Parameters | Heterotrophic Plate Count (cfu/100mL) | 140.9 ± 13.4 | 82.0 ± 10.7 | 11.8 | 0.00 | 112.3 ± 13.3 | 77.4 ± 7.9 | 5.1 | 0.03 | 152.6 ± 15.2 | 80.9 ± 7.6 | 17.8 | 0.75 | <10 |
| Faecal Coliform Count (cfu/100mL)) | 42.0 ± 7.0 | 9.6 ± 2.5 | 18.8 | 0.00 | 12.8 ± 6.8 | 1.4 ± 0.5 | 2.8 | 0.10 | 14.48 ± 6.07 | 10.0 ± 4.2 | 0.4 | 0.55 | 0 | |
| Enterococci Count (cfu/100mL) | 59.1 ± 8.8 | 24.9 ± 4.7 | 11.8 | 0.00 | 26.3 ± 7.9 | 16.7 ± 5.2 | 1.0 | 0.32 | 49.28 ± 11.34 | 12.0 ± 3.3 | 1.0 | 0.00 | 0 | |
| Total Coliform Count (cfu/100mL) | 46.2 ± 7.1 | 12.3 ± 3.3 | 18.9 | 0.00 | 72.0 ± 15.5 | 49.9 ± 10.5 | 1.4 | 0.24 | 28.40 ± 9.70 | 11.3 ± 4.7 | 2.5 | 0.12 | no limit given | |
| Fungal Count (cfu/100mL) | 109.6 ± 12.5 | 46.0 ± 6.0 | 21.0 | 0.00 | 93.6 ± 8.4 | 111.5 ± 7.1 | 2.7 | 0.11 | 99.58 ± 9.22 | 55.9 ± 3.4 | 19.8 | 0.00 | no limit given | |
| Sampled Area | Isolated Bacteria | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Citrobacter fruendii | Serratia marcescens | Proteus mirabilis | Salmonella sp. | Bacillus sp. | Shigella sp. | Escherichia coli | Vibrio cholerae | Pseudomonas aeruginosa | Enterococcus faecalis | Klebsiella pneumoniae | Aeromonas sp. | Micrococcus sp. | Enterobacter aerogenes | Staphylococcus aureus | ||
| Osun State | ✓ | ★ | ✓ | ✓ | ★ | ✓ | ✓✓ | ★ | ✓✓✓ | ✓ | ✓✓ | ★ | ★ | ✓✓✓ | ✓ | |
| Lagos State | ★ | ✓ | ✓ | ✓✓ | ✓✓ | ✓ | ✓ | ★ | ✓ | ✓✓ | ✓ | ✓ | ✓ | ✓ | ✓ | |
| Oyo State | ✓ | ✓ | ✓ | ✓ | ✓✓ | ✓ | ✓ | ✓ | ✓✓ | ✓ | ✓✓ | ✓ | ★ | ✓ | ✓ | |
| Sampled Area | Isolated Fungi | ||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Candida krusei | Candida parapsilosis | Candida albicans | Rhizopus stolonifer | Mucor janssenii | Trichoderma viridae | Trichoderma harzianum | Rhizoctonia solani | Aspergillus niger | A. brevipes | A. parasiticus | A. wentii | A. fumigatus | Penicillium roqueforti | ||
| Osun State | ✓ | ✓ | ✓ | ✓ | ✓ | ★ | ✓ | ✓ | ✓✓ | ✓✓ | ★ | ★ | ✓✓✓✓ | ✓ | |
| Lagos State | ✓ | ✓ | ✓ | ✓✓ | ✓✓✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | |
| Oyo State | ✓ | ✓ | ✓ | ✓ | ✓✓ | ★ | ★ | ★ | ✓✓ | ✓ | ✓ | ★ | ✓✓✓ | ★ | |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bisi-Johnson, M.A.; Adediran, K.O.; Akinola, S.A.; Popoola, E.O.; Okoh, A.I. Comparative Physicochemical and Microbiological Qualities of Source and Stored Household Waters in Some Selected Communities in Southwestern Nigeria. Sustainability 2017, 9, 454. https://doi.org/10.3390/su9030454
Bisi-Johnson MA, Adediran KO, Akinola SA, Popoola EO, Okoh AI. Comparative Physicochemical and Microbiological Qualities of Source and Stored Household Waters in Some Selected Communities in Southwestern Nigeria. Sustainability. 2017; 9(3):454. https://doi.org/10.3390/su9030454
Chicago/Turabian StyleBisi-Johnson, Mary A., Kehinde O. Adediran, Saheed A. Akinola, Elizabeth O. Popoola, and Anthony I. Okoh. 2017. "Comparative Physicochemical and Microbiological Qualities of Source and Stored Household Waters in Some Selected Communities in Southwestern Nigeria" Sustainability 9, no. 3: 454. https://doi.org/10.3390/su9030454
APA StyleBisi-Johnson, M. A., Adediran, K. O., Akinola, S. A., Popoola, E. O., & Okoh, A. I. (2017). Comparative Physicochemical and Microbiological Qualities of Source and Stored Household Waters in Some Selected Communities in Southwestern Nigeria. Sustainability, 9(3), 454. https://doi.org/10.3390/su9030454






