Lime and Phosphate Amendment Can Significantly Reduce Uptake of Cd and Pb by Field-Grown Rice
Abstract
:1. Introduction
2. Materials and Methods
2.1. Field Experiment
2.2. Sampling and Analysis
2.3. Data Analysis
3. Results
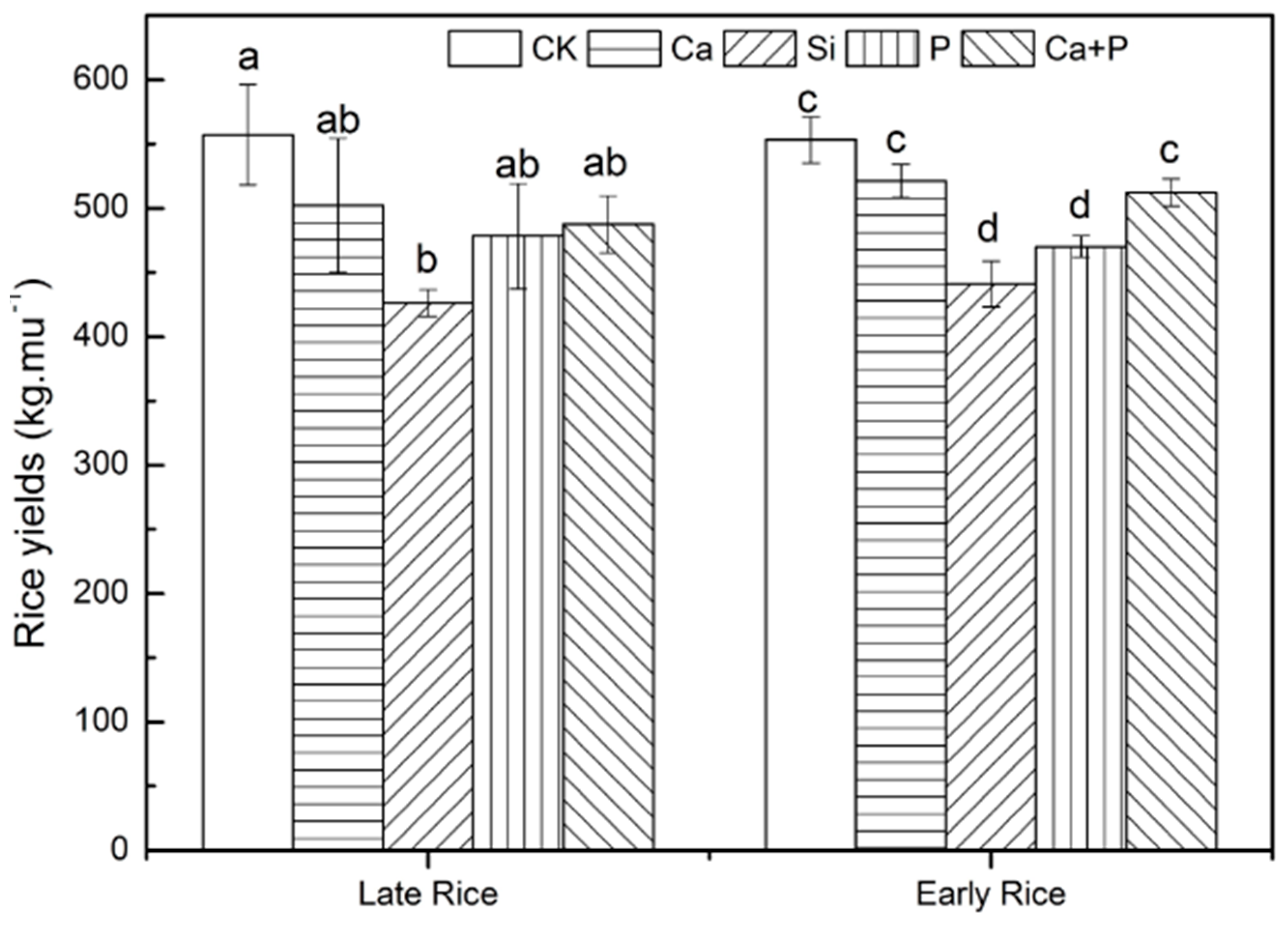
3.1. Basic Information and Rice Yields
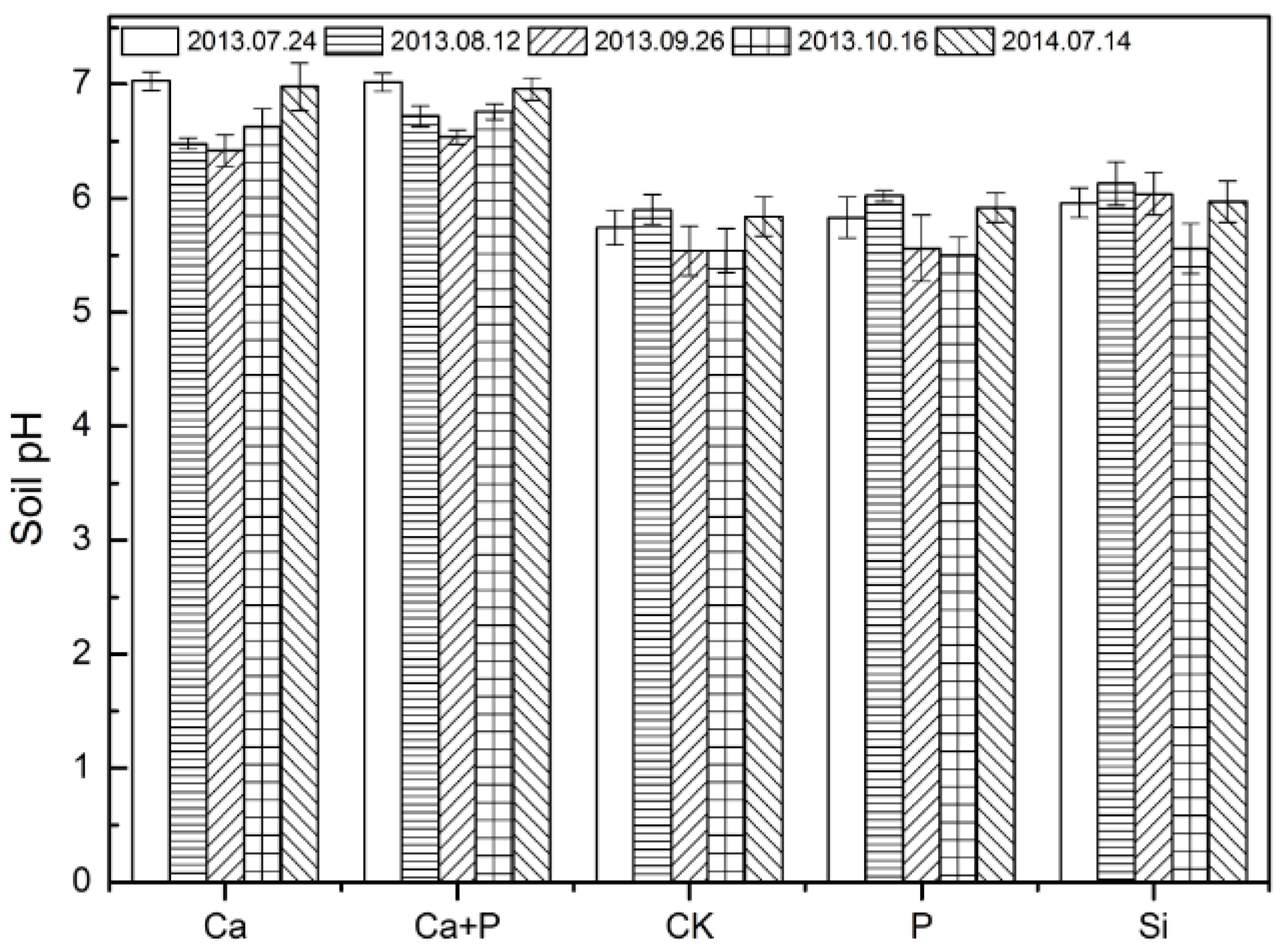
3.2. Soil pH and Metal Availability
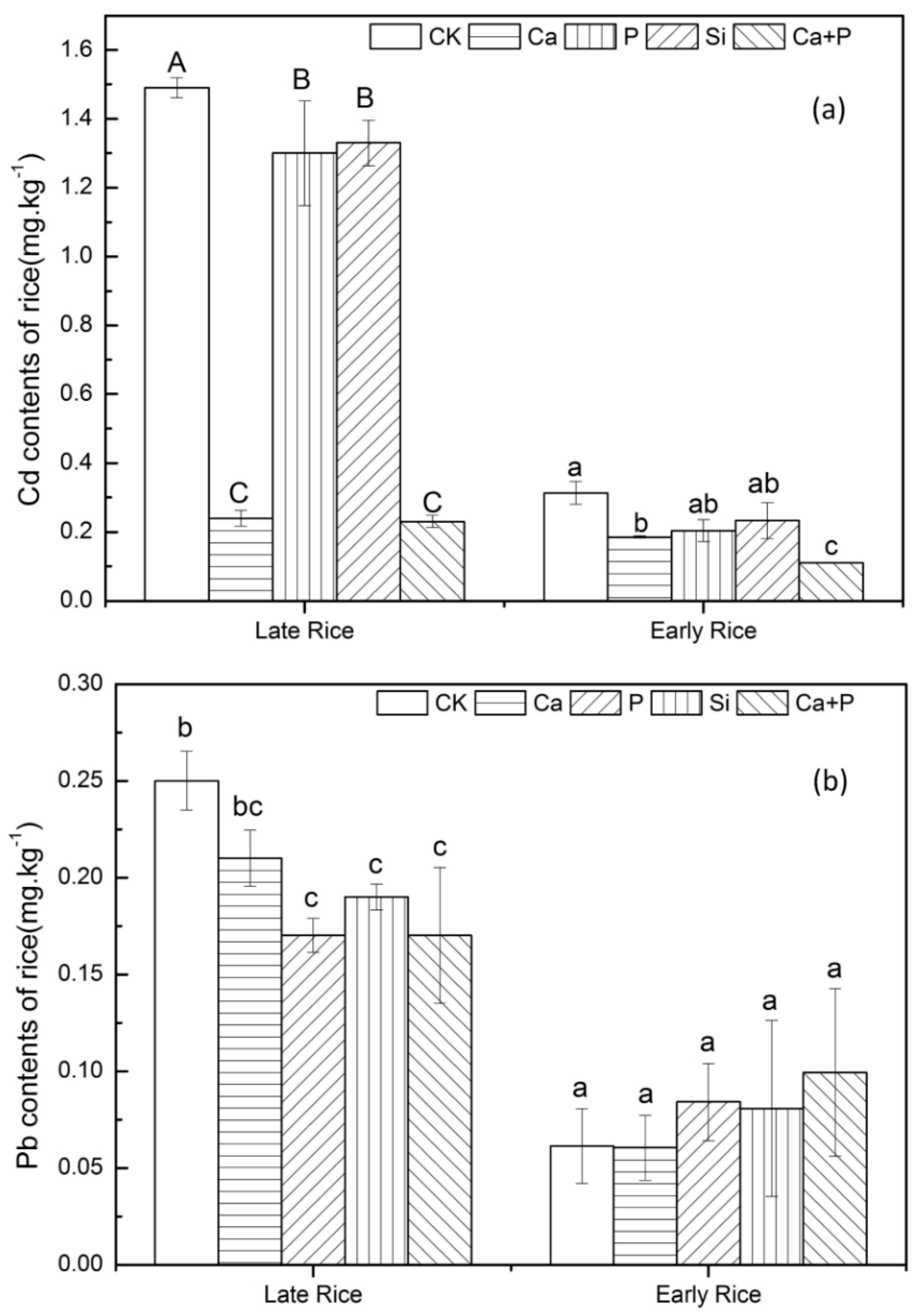
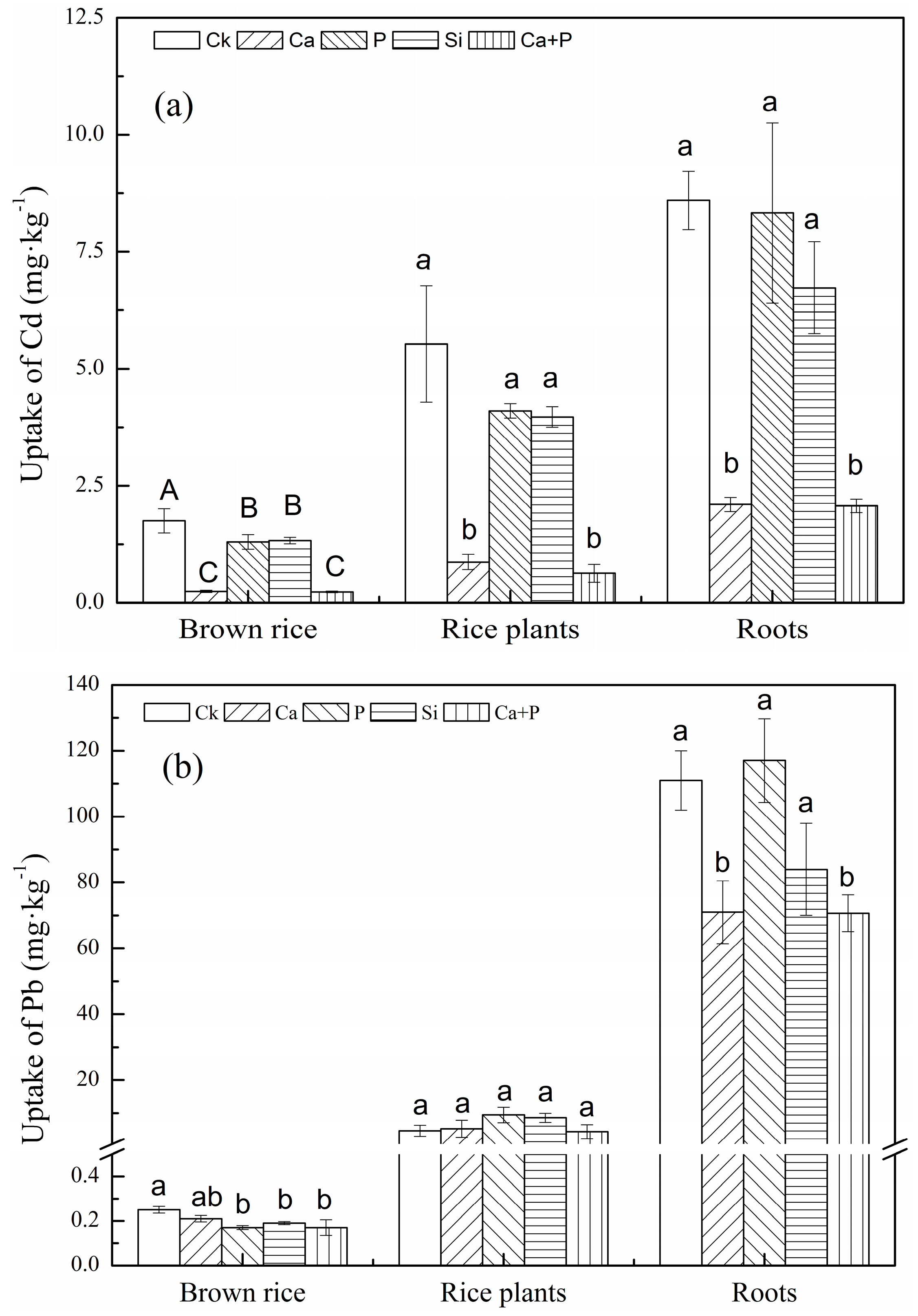
3.3. Uptake of Cd and Pb by Rice
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Zheng, N.; Liu, J.S.; Wang, Q.C.; Liang, Z.Z. Health risk assessment of heavy metal exposure to street dust in the zinc smelting district, Northeast of China. Sci. Total Environ. 2010, 408, 726–733. [Google Scholar] [CrossRef] [PubMed]
- Niu, L.L.; Yang, F.X.; Xu, C.; Yang, H.Y.; Liu, W.P. Status of metal accumulation in farmland soils across China: From distribution to risk assessment. Environ. Pollut. 2013, 176, 55–62. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Zhang, X.H.; Tran, H.; Wang, D.Q.; Zhu, Y.N. Heavy metal contamination and risk assessment in water, paddy soil, and rice around an electroplating plant. Environ. Sci. Pollut. Res. 2011, 18, 1623–1632. [Google Scholar] [CrossRef] [PubMed]
- Du, Y.; Hu, X.; Wu, X.; Shu, Y.; Jiang, Y.; Yan, X. Affects of mining activities on Cd pollution to the paddy soils and rice grain in Hunan province: Central south China. Environ. Monit. Assess. 2013, 185, 9843–9856. [Google Scholar] [CrossRef] [PubMed]
- Gall, J.E.; Boyd, R.S.; Rajakaruna, N. Transfer of heavy metals through terrestrial food webs: A review. Environ. Monit. Assess. 2015, 187, 201. [Google Scholar] [CrossRef] [PubMed]
- Li, W.L.; Xu, B.B.; Song, Q.J.; Liu, X.M.; Xu, J.M.; Brookes, P.C. The identification of ‘hotspots’ of heavy metal pollution in soil–rice systems at a regional scale in eastern China. Sci. Total Environ. 2014, 472, 407–420. [Google Scholar] [CrossRef] [PubMed]
- Neilson, S.; Nishanta, R. Phytoremediation of Agricultural Soils: Using Plants to Clean Metal-Contaminated Arable Land; Spring International Publishing: Berlin, Germany, 2015; pp. 159–168. [Google Scholar]
- Contaminated Land Rehabilitation Network for Environmental Technologies (CLARINET). Remediation of Contaminated Land Technology Implementation in Europe. 2002. Available online: http://www.commonforum.eu/Documents/DOC/Clarinet/WG7_Final_Report.pdf (accessed on 26 June 2015).
- Dermont, G.; Bergeron, M.; Mercier, G.; Richer-Laflèche, M. Metal-contaminated soils: Remediation practices and treatment technologies. Pract. Period. Hazard. Toxic Radioact. Waste Manag. 2008, 12, 188–209. [Google Scholar] [CrossRef]
- Harbottle, M.J.; Al-Tabbaa, A.; Evans, C.W. A comparison of the technical sustainability of in situ stabilisation/solidification with disposal to landfill. J. Hazard. Mater. 2007, 141, 430–440. [Google Scholar] [CrossRef] [PubMed]
- Akcil, A.; Erust, C.; Ozdemiroglu, S.; Fontib, V.; Beolchinib, F. A review of approaches and techniques used in aquatic contaminated sediments: Metal removal and stabilization by chemical and biotechnological processes. J. Clean. Prod. 2015, 86, 24–36. [Google Scholar] [CrossRef]
- Chen, Z.; Tie, B.Q.; Lei, M.; Liu, X.L.; Ye, C.C.; Luo, M.M.; Mao, Y.D. Phytoexclusion potential studies of Si fertilization modes on rice cadmium. Environ. Sci. 2014, 35, 2762–2770. [Google Scholar]
- Huang, Z.; Pan, X.; Wu, P.; Han, J.; Chen, Q. Heavy metals in vegetables and the health risk to population in Zhejiang, China. Food Control 2014, 36, 248–252. [Google Scholar] [CrossRef]
- Luo, Y.H.; Gu, X.Y.; Wu, Y.G. In-situ remediation of cadmium-polluted agriculture land using stabilizing amendments. J. Agro-Environ. Sci. 2014, 33, 890–897. [Google Scholar]
- Wang, K.R.; Zhang, Y.Z.; Hu, R.G. Effects of different types of soil amelioration materials on reducing concentrations of Pb and Cd in brown rice in heavy metal polluted paddy soils. J. Agro-Environ. Sci. 2007, 26, 476–481. [Google Scholar]
- Friesl, W.; Friedl, J.; Platzer, K.; Horak, O.; Gerzabek, M.H. Remediation of contaminated agricultural soils near a former Pb/Zn smelter in Austria: Batch, pot and field experiments. Environ. Pollut. 2006, 144, 40–50. [Google Scholar] [CrossRef] [PubMed]
- Hong, C.O.; Lee, D.K.; Chung, D.Y.E.A. Liming effects on cadmium stabilization in upland soil affected by gold mining activity. Arch. Environ. Contam. Toxicol. 2007, 52, 496–502. [Google Scholar] [CrossRef] [PubMed]
- Kibria, M.G.; Osman, K.T.; Ahammad, M.J.; Alamgir, M.D. Effects of farm yard manure and lime on cadmium uptake by rice grown in two contaminated soils of Chittagong. J. Agric. Sci. Technol. 2011, 5, 352–358. [Google Scholar]
- Lu, R.K. Agricultural Chemical Analysis of the Soil, 1st ed.; China Agriculture Science and Technology Press: Beijing, China, 1999. [Google Scholar]
- Li, R.M.; Wang, G.; Fang, L. Effects of Lime Complexation Organic On Uptake of Cd, Pb by Crops. J. Agro-Environ. Sci. 2003, 22, 293–296. [Google Scholar]
- Sun, Y.B.; Zhou, Q.X.; Guo, G.L. Phytoremediation and strengthening measures for soil contaminated by heavy metals. Chin. J. Environ. Eng. 2007, 1, 103–110. [Google Scholar]
- Wang, L.; Xu, Y.M.; Liang, X.F.; Sun, G.H.; Sun, Y.B.; Lin, D.S. Remediation of contaminated paddy soil by immobilization of pollutants in the Diaojiang Catchment, Guangxi Province. J. Ecol. Rural Environ. 2012, 5, 563–568. [Google Scholar]
- Zhao, Y.K.; Zhang, W.S.; Wang, Y.N.; Li, K.X.; Jia, H.Z.; Li, X. Research progress in physiology and molecular biology of plant responses to high pH. Chin. J. Eco-Agric. 2008, 16, 783–787. [Google Scholar]
- Zhao, R.F.; Zou, C.Q.; Zhang, F.S. Effects of long-term P fertilization on P and Zn availability in winter wheat rhizoshpere and their nutrition. Plant Nutr. Fertil. Sci. 2007, 13, 368–372. [Google Scholar]
- Cho, U.; Park, J. Mercury-induced oxidative stress in tomato seedlings. Plant Sci. 2000, 156, 1–9. [Google Scholar] [CrossRef]
- Zhou, Q.X.; Song, Y.F. Repair Principles and Methods of Contaminated Soil, 1st ed.; Science Press: Shanghai, China, 2004. [Google Scholar]
- Chen, S.B.; Zhu, Y.G.; Ma, Y.B. Effects of hydroxylapatite on the sorption and desorption of lead in various Chinese soils. Environ. Chem. 2006, 4, 409–413. [Google Scholar]
- Zhang, Xue X.; Zhang, Xiao X.; Zheng, Y.J.; Wang, R.P.; Chen, N.C.; Lu, P.X. Accumulation of S, Fe and Cd in rhizosphere of rice and their uptake in rice with different water managements. Environ. Sci. 2013, 34, 2837–2846. (In Chinese) [Google Scholar]
- Ji, X.H.; Liang, Y.C.; Lu, Y.H.; Liao, Y.L.; Nie, J.; Zheng, S.X.; Li, Z.J. The effect of water management on the mechanism and rate of uptake and accumulation of cadmium by rice grow in gin polluted paddy soil. Acta Ecol. Sin. 2007, 27, 3930–3939. [Google Scholar]
| Treatment | Amendments | Addition |
|---|---|---|
| CK | Control | --- |
| Ca | Lime | 6000 |
| P | phosphate | 165.6 |
| Si | silicon fertilizer | 1500 |
| Ca + P | Lime + phosphate | 6000 + 165.6 |
| Mean | Standard Deviation | |
|---|---|---|
| pH | 4.80 | 0.6 |
| SOM (g·kg−1) | 27.62 | 3.8 |
| CEC (cmol (+)·kg−1) | 8.17 | 0.9 |
| Cd (mg·kg−1) | 0.76 | 0.13 |
| Pb (mg·kg−1) | 136 | 15 |
| Zn (mg·kg−1) | 176 | 17 |
| As (mg·kg−1) | 18 | 3.2 |
| Treatment | Late Rice (n = 15) | Early Rice (n =15) | ||
|---|---|---|---|---|
| Cd | Pb | Cd | Pb | |
| CK | 0.186 ± 0.058 a | 45.1 ± 2.41 a | 0.173 ± 0.06 a | 35.2 ± 3.12 a |
| Ca | 0.169 ± 0.023 a | 42.1 ± 2.44 a | 0.137 ± 0.012 b | 34.2 ± 2.56 a |
| P | 0.182 ± 0.011 a | 47.3 ± 0.41 a | 0.163 ± 0.015 a | 36.5 ± 0.64 a |
| Si | 0.160 ± 0.01 a | 42.2 ± 0.429 a | 0.158 ± 0.31 a | 31.1 ± 1.38 a |
| Ca + P | 0.155 ± 0.06 b | 39.8 ± 1.021 b | 0.130 ± 0.13 b | 30.3 ± 0.98 a |
| Index | Cd in Rice | Pb in Rice | Available Cd | Available Pb | Cd in Roots | Pb in Roots | Cd in Plants | Pb in Plants |
|---|---|---|---|---|---|---|---|---|
| pH | −0.86 ** | −0.173 | −0.234 | −0.401 | −0.764 ** | −0.526 * | −0.825 ** | −0.29 |
| Cd in rice | −0.181 | 0.388 | 0.466 | 0.853 ** | 0.767 ** | 0.956 ** | 0.158 | |
| Pb in rice | −0.284 | −0.387 | −0.221 | −0.285 | −0.126 | −0.475 | ||
| Available Cd | 0.949 ** | 0.178 | 0.531 * | 0.510 | 0.60 | |||
| Available Pb | 0.244 | 0.547 * | 0.556 * | 0.138 | ||||
| Cd in roots | 0.767 ** | 0.789 ** | 0.237 | |||||
| Pb in roots | 0.714 ** | 0.121 | ||||||
| Cd in plants | 0.135 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Xiao, R.; Huang, Z.; Li, X.; Chen, W.; Deng, Y.; Han, C. Lime and Phosphate Amendment Can Significantly Reduce Uptake of Cd and Pb by Field-Grown Rice. Sustainability 2017, 9, 430. https://doi.org/10.3390/su9030430
Xiao R, Huang Z, Li X, Chen W, Deng Y, Han C. Lime and Phosphate Amendment Can Significantly Reduce Uptake of Cd and Pb by Field-Grown Rice. Sustainability. 2017; 9(3):430. https://doi.org/10.3390/su9030430
Chicago/Turabian StyleXiao, Rongbo, Zehong Huang, Xiaonuo Li, Weiping Chen, Yirong Deng, and Cunliang Han. 2017. "Lime and Phosphate Amendment Can Significantly Reduce Uptake of Cd and Pb by Field-Grown Rice" Sustainability 9, no. 3: 430. https://doi.org/10.3390/su9030430
APA StyleXiao, R., Huang, Z., Li, X., Chen, W., Deng, Y., & Han, C. (2017). Lime and Phosphate Amendment Can Significantly Reduce Uptake of Cd and Pb by Field-Grown Rice. Sustainability, 9(3), 430. https://doi.org/10.3390/su9030430






