Diamondback Moth, Plutella xylostella (L.) in Southern Africa: Research Trends, Challenges and Insights on Sustainable Management Options
Abstract
:1. Introduction
2. Horticulture and DBM in Southern Africa
3. Why is Southern Africa Hard Hit by the DBM Scourge?
3.1. Vulnerability to Effects of Climate Change
3.2. Farmers’ Behaviour and Insecticide Use
3.3. Lack of Insecticide Resistance Research
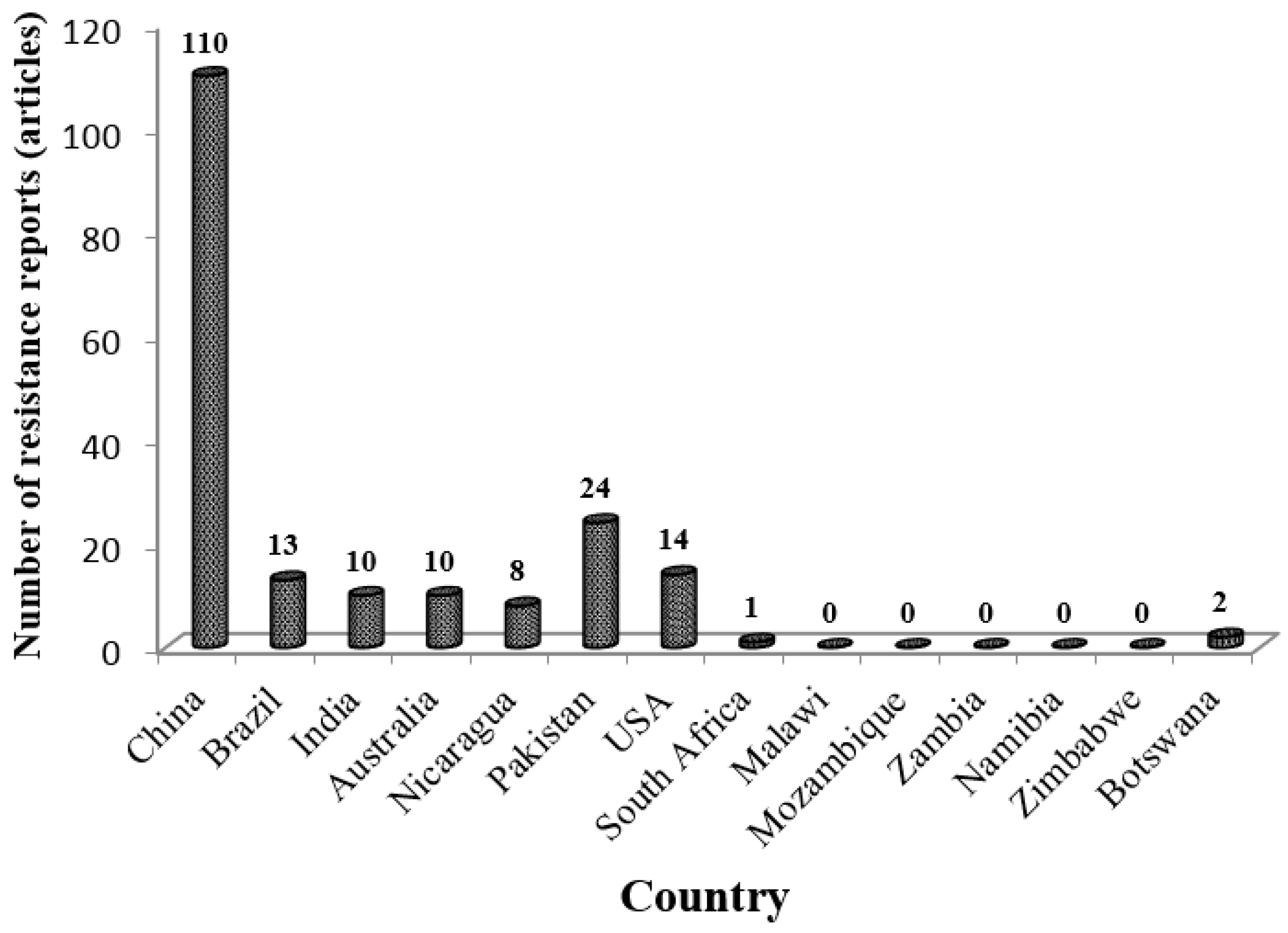
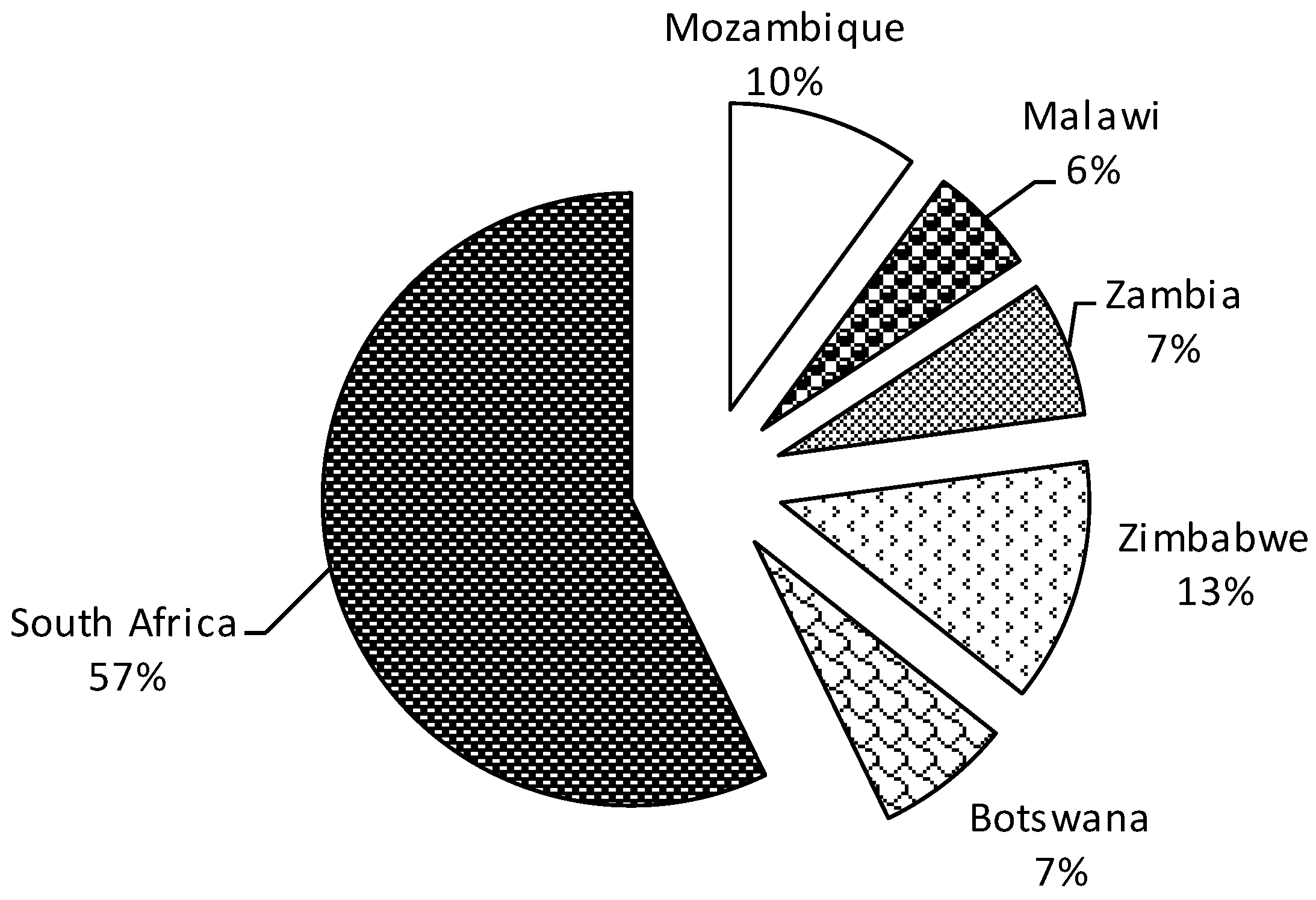
3.4. Low Research Attention
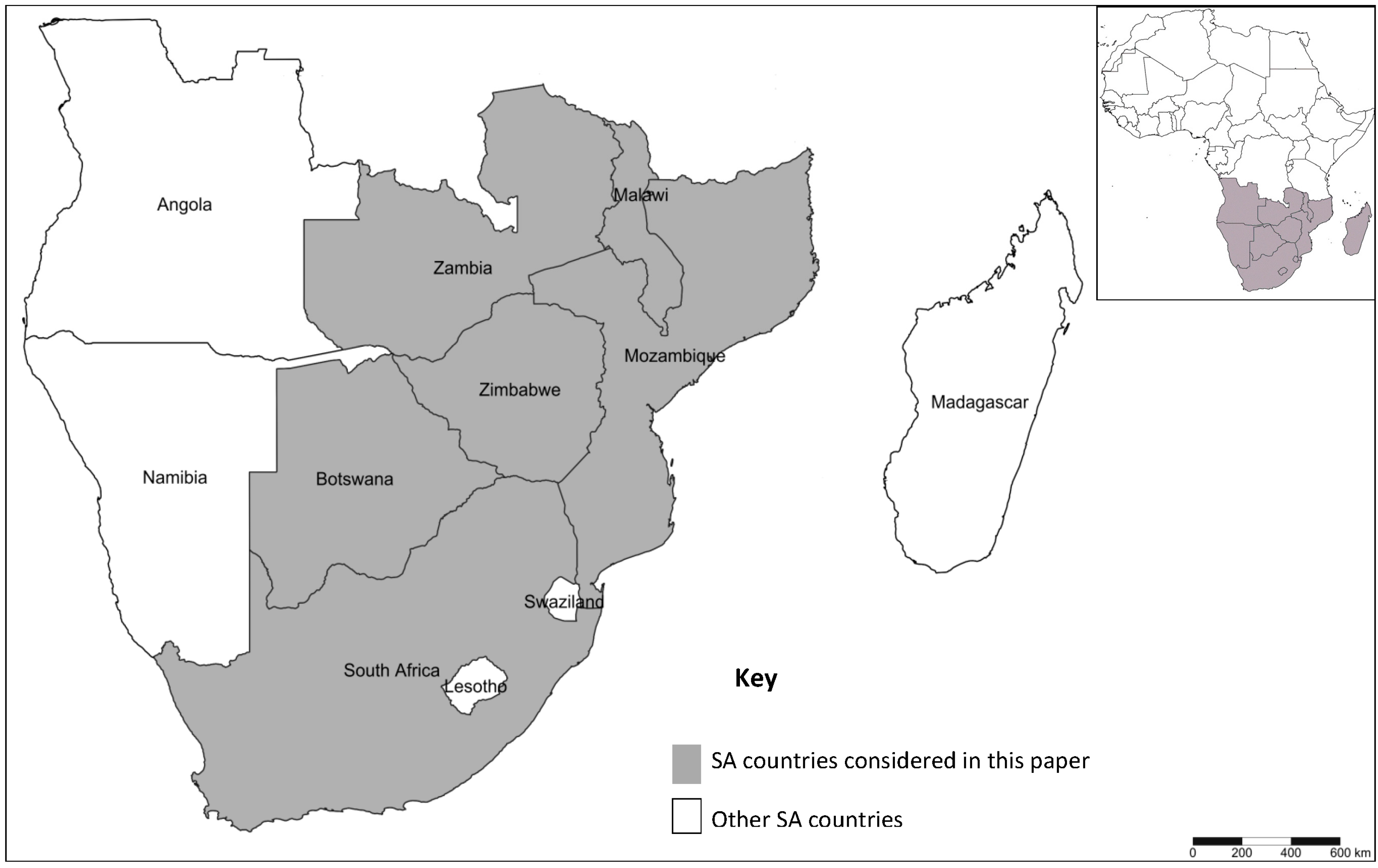
3.5. Lack of Regional Coordination in DBM Research
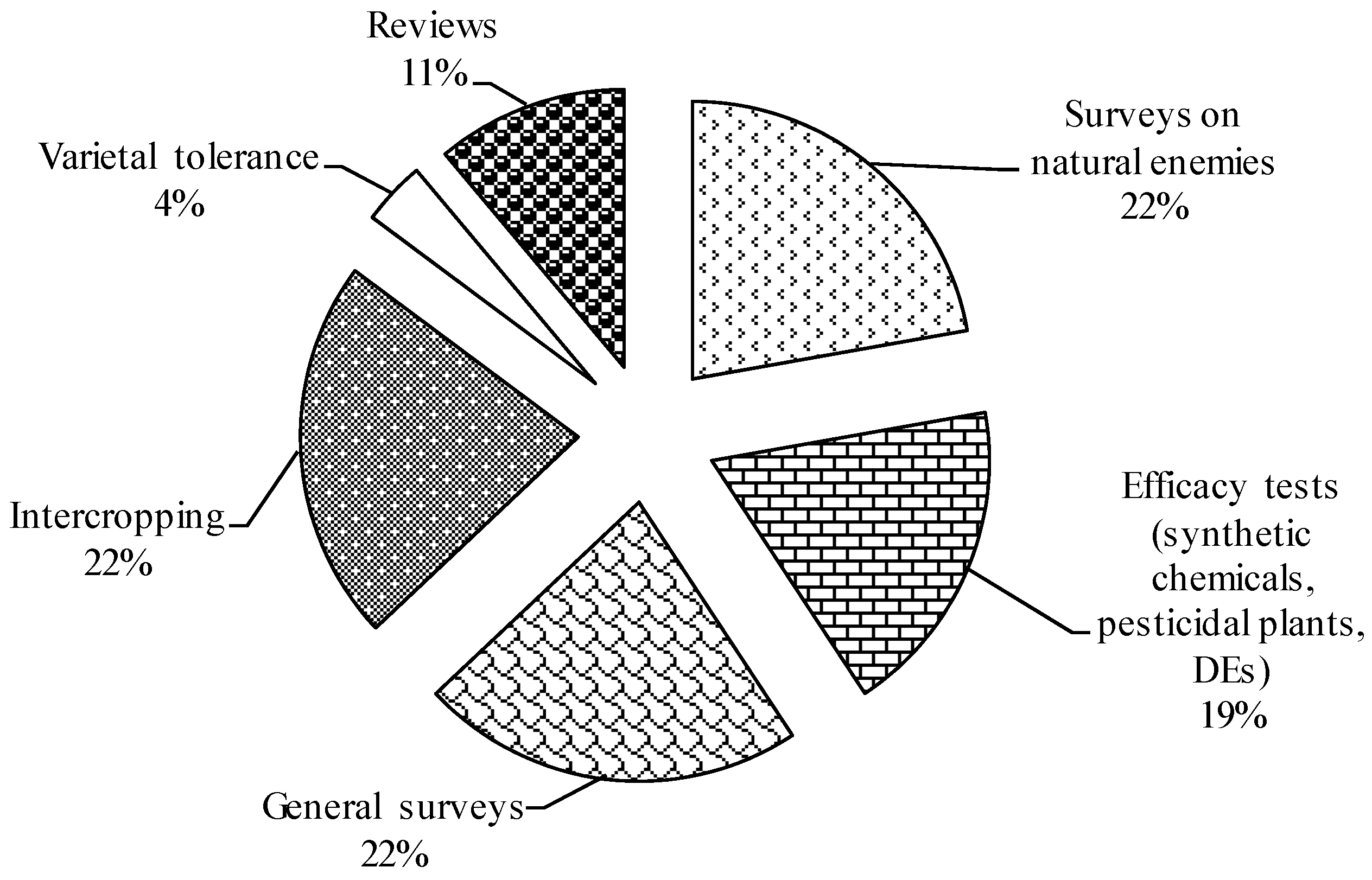
4. Possible Novel DBM Control Options
5. Constraints to IPM Implementation and Adoption of Novel Sustainable Control Methods
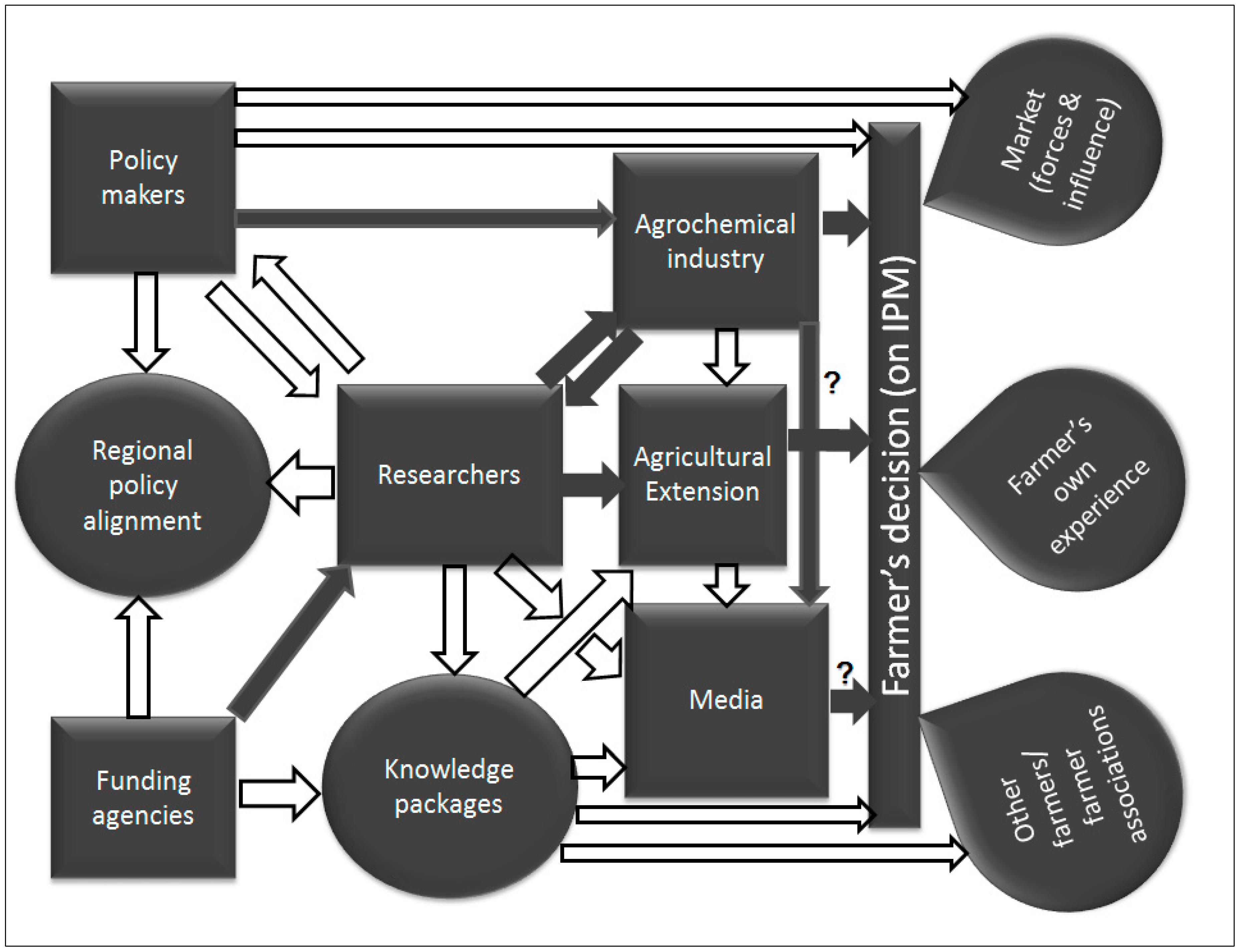
5.1. Poor Understanding of the IPM Concept and Information Flow among DBM Management Actors
5.2. Weak Links between the Players in the Agroindustry
5.3. Lack of Locally-Developed Well-Packaged IPM Practices and Procedures
5.4. Lack of Policy Support
5.5. Taxonomic Confusion and Insufficient Adaptation of Biocontrol Agents to Release-Sites Climate and Bio-Ecological Conditions
5.6. Limited Alternative Control Options
6. Future Prospects and Research Needs
- (1)
- ‘Do no harm’—the use of biologically- and environmentally-safe pest control methods with no or selective soft and safe insecticide use.
- (2)
- ‘Do good’—Improving farmer knowledge, consumer, agrochemical industry, and policy-maker awareness; policy reforms and regional policy harmonization; strengthening regulatory frameworks; and national and regional institutional capacitation.
7. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Sibanda, T.; Dobson, H.M.; Cooper, J.F.; Manyangarirwa, W.; Chiimba, W. Pest management challenges for small-holder vegetable farmers in Zimbabwe. Crop Prot. 2000, 19, 807–815. [Google Scholar] [CrossRef]
- Saka, A.R.; Mtukuso, A.P.; Mbale, B.J.; Phiri, I.M.G. The role of research-extension-farmer linkages in vegetable production and development in Malawi. In Vegetable Research and Development in Malawi. Review and Planning Workshop Proceedings, Lilongwe, Malawi, 23–24 September 2003; Chadha, M.L., Oluoch, M.O., Saka, A.R., Mtukuso, A.P., Daudi, A., Eds.; World Vegetable Center (AVRDC): Shanhua, Taiwan, 2003. [Google Scholar]
- Munthali, D.C. Evaluation of cabbage varieties to cabbage aphid. Afr. Entomol. 2009, 17, 1–7. [Google Scholar] [CrossRef]
- Food and Agriculture Organization (FAO). Evolving a Plant Breeding and Seed System in Sub-Saharan Africa in an Era of Donor Dependence; FAO Plant Production and Protection Paper 210; FAO: Rome, Italy, 2011. [Google Scholar]
- Canico, A.; Santos, L.; Massing, R. Development and adult longevity of diamondback moth and its parasitoids Cotesia plutellae and Diadegma semiclausum in uncontrolled conditions. Afr. Crop Sci. Conf. Proc. 2013, 11, 257–262. [Google Scholar]
- Khonje, A.A. Research trends in horticultural crops in Malawi. J. Crop Weed 2013, 9, 13–25. [Google Scholar]
- Ekesi, S.; Chabi-Olaye, A.; Subramanian, S.; Borgeimeister, C. Horticultural pest management and African Economy: Successes, Challenges and Opportunities in a changing global environment. Acta Hortic. 2009, 911, 165–183. [Google Scholar] [CrossRef]
- Stathers, T.; Lamboll, R.; Mvumi, B.M. Postharvest agriculture in changing climate: Its importance to African smallholder farmers. Food Secur. 2013, 5, 361–392. [Google Scholar] [CrossRef]
- Intergovernmental Panel on Climate Change (IPCC). Climate Change 2014: Synthesis Report; Fifth Assessment Report (AR5); Contribution of Working Groups I, II and III to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change; IPCC: Geneva, Switzerland, 2014. [Google Scholar]
- Maniania, N.K.; Takasu, K. Development of microbial control agents at the International Centre of Insect Physiology and Ecology. Bull. Inst. Trop. Agric. 2006, 29, 1–9. [Google Scholar]
- Food and Agriculture Organization (FAO). Growing Greener Cities in Africa; First Status Report on Urban and Peri-Urban Horticulture in Africa; Food and Agriculture Organisation of the United Nations: Rome, Italy, 2009. [Google Scholar]
- Katsaruware, R.D.; Dubiwa, M. Onion (Allium cepa) and garlic (Allium sativum) as pest control intercrops in cabbage based intercrop system in Zimbabwe. J. Agric. Vet. Sci. 2014, 7, 13–17. [Google Scholar]
- Abate, T.; van Huis, A.; Ampofo, J.K.O. Pest Management Strategies in traditional agriculture: An African Perspective. Ann. Rev. Entomol. 2000, 45, 631–659. [Google Scholar] [CrossRef] [PubMed]
- Momanyi, D.; Lagat, K.J.; Ayuya, O.I. Determinants of smallholder African indigenous leafy vegetables farmers’ market participation behaviour in Nyamira County, Kenya. J. Econ. Sustain. 2015, 16, 212–217. [Google Scholar]
- Department of Agriculture, Forestry and Fisheries. A Profile of the South African Cabbage Market Value Chain; Department of Agriculture, Forestry and Fisheries: Arcadia, South Africa, 2014.
- Lubinga, M.; Ogundeji, A.; Jordaan, H. East African community trade potential and performance with European Union: A perspective of selected fruit and vegetable commodities. ESJ 2014, 1, 430–443. [Google Scholar]
- Talekar, N.S.; Shelton, A.M. Biology, ecology and management of the diamondback moth. Ann. Rev. Entomol. 1993, 38, 275–301. [Google Scholar] [CrossRef]
- Furlong, M.J.; Wright, D.J.; Dosdall, L.M. Diamondback moth ecology and management: Problems, progress and prospects. Ann. Rev. Entomol. 2013, 58, 517–554. [Google Scholar] [CrossRef] [PubMed]
- Sithole, R. Life History Parameters of Diadegma mollipla (Holmgren), Competition with Diadegma semiclausum Hellen (Hymenoptera: Ichneumonidae) and Spatial and Temporal Distribution of the Host, Plutella xylostella (L.) and Its Indigenous Parasitoids in Zimbabwe. Ph.D. Thesis, University of Zimbabwe, Harare, Zimbabwe, 2005. [Google Scholar]
- Obopile, M.; Munthali, D.C.; Matilo, B. Farmers’ knowledge, perceptions and management of vegetable pests and diseases in Botswana. Crop Prot. 2008, 27, 1220–1224. [Google Scholar] [CrossRef]
- Nofemela, R.; Kfir, R. The pest status of Diamondback moth (Lepidoptera: Plutellidae) in South Africa: The role of parasitoids in suppressing the pest populations. In Proceedings of the Fifth International Workshops on the Management of Diamondback Moth and other Crucifer Pests, Beijing, China, 21–24 October 2008.
- Manyangarirwa, W.; Zehnder, G.W.; McCutcheon, G.S.; Smith, J.P.; Adler, P.H.; Mphuru, A.N. Parasitoids of the diamondback moth on brassicas in Zimbabwe. Afr. Crop Sci. Conf. Proc. 2009, 9, 565–570. [Google Scholar]
- Nyambo, B.; Sevgan, S.; Chabi-Olaye, A.; Ekesi, S. Management of alien invasive insect pest species and diseases of fruits of vegetables: Experiences from East Africa. Acta Hort. 2009, 911. [Google Scholar] [CrossRef]
- Tibugari, H.; Mandumbu, R.; Jowah, P.; Karavina, C. Farmer knowledge, attitude and practice on cotton (Gossypium hirsutum L.) pest resistance and management practices in Zimbabwe. Arch. Phytopathol. Pflanzenschutz. 2012, 45, 2395–2405. [Google Scholar] [CrossRef]
- Gryzwacz, D.; Rosbach, A.; Rauf, A.; Russel, D.A.; Srivansan, R.; Shelton, A.M. Current control methods for diamondback moth and other brassica insect pests and the prospexcts for improved management with lepidopteran resistant Bt vegetable brassicas in Asia and Africa. J. Crop Prot. 2010, 29, 68–79. [Google Scholar] [CrossRef]
- Chidawanyika, F.; Mudavanhu, P.; Nyamukondiwa, C. Biologically based methods for pest management under changing climates: Challenges and future directions. Insects 2012, 3, 1171–1189. [Google Scholar] [CrossRef] [PubMed]
- Zalucki, M.P.; Shabbir, A.; Silva, R.; Adamson, D.; Shu-shen, L.; Furlong, M.J. Estimating the Economic Cost of One of the World’s Major Insect Pests, Plutella xylostella (Lepidoptera: Plutellidae): Just How Long Is a Piece of String? J. Econ. Entomol. 2012, 105, 1115–1129. [Google Scholar] [CrossRef] [PubMed]
- Macharia, I.; Mithofer, D.; Waibel, H. Health effects of pesticide use among vegetable farmers in Kenya. In Proceedings of the 4th International Conference of the African Association of Agricultural Economists, Hammamet, Tunisia, 22–25 September 2013.
- Ayalew, G. Comparison of yield losses on cabbage from diamondback moth, Plutella xylostella L. (Lepidoptera: Plutellidae) using two insecticides. Crop Prot. 2006, 25, 915–919. [Google Scholar] [CrossRef]
- Safraz, M.; Keddie, A.B.; Dosdall, L.M. Biological control of the diamondback moth, Plutella xylostella. A Review. Biocontrol Sci. Technol. 2005, 15, 763–789. [Google Scholar] [CrossRef]
- Marchioro, C.A.; Foerster, L.A. Development and survival of diamondback moth, Plutella xylostella L. (Lepidoptera: Plutellidae) as a function of temperature: Effect on the number of generations in tropical and sub-tropical regions. J. Neotrop. Entomol. 2011, 40, 533–541. [Google Scholar]
- Chown, S.L.; Nicolson, S. Insect Physiological Ecology: Mechanisms and Patterns; Oxford University Press: Oxford, UK, 2004. [Google Scholar]
- Denlinger, D.L.; Lee, R.E., Jr. Low Temperature Biology of Insects; Cambridge University Press: Cambridge, UK, 2010. [Google Scholar]
- Bahar, M.H.; Hegedus, D.; Soroka, J.; Coutu, C.; Bekkaoui, D. Survival and Hsp70 gene expression in Plutella xylostella and its larval parasitoid Diadegma insulare varied between slowly ramping and abrupt extreme temperature regimes. PLoS ONE 2013, 8, e73901. [Google Scholar] [CrossRef] [PubMed]
- Hill, M.P.; Bertelsmeier, C.; Clusella-Trullas, S.; Garnas, J.R.; Robertson, M.P.; Terblanche, J.S. Predicted decrease in global climate suitability masks regional complexity of invasive fruit fly species response to climate change. Biol. Invasions 2016, 18, 1105–1119. [Google Scholar] [CrossRef]
- Andrew, N.R.; Hill, S.J.; Binns, M.; Bahar, M.H.; Ridley, E.V.; Jung, M.P.; Fyfe, C.; Yates, M.; Khusro, M. Assessing insect responses to climate change: What are we testing for? Where should we be heading? Peer J. 2013, 1, e11. [Google Scholar] [CrossRef] [PubMed]
- Shirai, Y. Temperature tolerance of diamondback moth, Plutella xylostella (Lepidoptera: Yponomeutidae) in tropical and temperate regions of Asia. Bull. Entomol. Res. 2000, 90, 357–364. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, C.; Bahar, M.H.; Baker, G.; Andrew, N.R. Thermal tolerance limits of DBM in ramping and plunging assays. PLoS ONE 2014, 9, e87535. [Google Scholar] [CrossRef] [PubMed]
- Kuntashula, E.; Silesh, G.; Mafongoya, P.L.; Bond, J. Farmer participatory evaluation of the potential for organic vegetable production in the wetlands of Zambia. Outlook Agric. 2006, 35, 299–305. [Google Scholar] [CrossRef]
- Nyirenda, S.P.; Sileshi, G.W.; Belmain, S.R.; Kamanula, J.F.; Mvumi, B.M.; Sola, P.; Nyirenda, G.K.C.; Stevenson, P.C. Farmers’ ethno-ecological knowledge of vegetable pests and pesticidal plant use in Northern Malawi and Eastern Zambia. Afr. J. Agric. Res. 2011, 6, 1525–1537. [Google Scholar]
- Madisa, M.E.; Obopile, M.; Assefa, Y. Analysis of horticultural production trends in Botswana. J. Plant Stud. 2012, 1. [Google Scholar] [CrossRef]
- Wandaat, E.Y.; Kugbe, J.X. Pesticide misuse in rural-urban agriculture: A case study of vegetable production in Tano South of Ghana. AJAFS 2015, 3, 343–360. [Google Scholar]
- Williamson, S.; Ball, A.; Pretty, J. Trends in pesticide use and drivers for safer pest management in four African countries. Crop Prot. 2008, 27, 1327–1334. [Google Scholar] [CrossRef]
- Timprasert, S.; Datta, A.; Ranamukhaarachchi, S.L. Factors determining adoption of integrated pest management by vegetable growers in Nakhon Ratchasima Province, Thailand. Crop Prot. 2014, 62, 32–39. [Google Scholar] [CrossRef]
- Lobell, D.B.; Burke, M.B.; Tebaldi, C.; Mastrandrea, M.D.; Falcon, W.P.; Naylor, R.L. Prioritizing climate change adaptation needs for food security in 2030. Science 2008, 319, 607–610. [Google Scholar] [CrossRef] [PubMed]
- Jack, C. Climate projections for United Republic of Tanzania; Climate Systems Analysis Group (CSAG)—University of Cape Town: Cape Town, South Africa, 2010. [Google Scholar]
- Steynour, A.; Jack, C.; Taylor, A. Information on Zimbabwe’s Climate and How It Is Changing; Climate Systems Analysis Group—University of Cape Town: Cape Town, South Africa, 2012. [Google Scholar]
- Arthropod Pesticide Resistance Database (IRAC). Michigan State University. 2015. Available online: http://www.pesticideresistance.com/display.php?page=speciesarId=571 (accessed on 22 August 2015).
- Mosiane, S.M.; Kfir, R.; Villet, M.H. Seasonal phenology of the diamondback moth, Plutella xylostella L. (Lepidoptera: Plutellidae) and its parasitoids on canola Brassica napus (L.) in Gauteng Province, South Africa. Afr. Entomol. 2010, 11, 277–285. [Google Scholar]
- Dosdall, L.M.; Zalucki, M.P.; Tansey, J.A.; Furlong, M.J. Developmental responses of the diamondback moth parasitoid Diadegma semiclausum (Hellén) (Hymenoptera: Ichneumonidae) to temperature and host plant species. Bull. Entomol. Res. 2012, 102, 373–384. [Google Scholar] [CrossRef] [PubMed]
- Sgrò, C.M.; Lowe, A.J.; Hoffmann, A.A. Building evolutionary resilience for conserving biodiversity under climate change. Evol. Appl. 2010, 4, 326–337. [Google Scholar] [CrossRef] [PubMed]
- Kopper, B.J.; Lindroth, R. Effects of elevated carbon dioxide and ozone on the phytochemistry of aspen performance of an herbivore. Oecologia 2003, 134, 95–103. [Google Scholar] [CrossRef] [PubMed]
- Sanders, N.T.; Belote, R.T.; Weltzin, K.F. Multitrophic effects of elevated carbon dioxide on understory plant and arthropod communities. Environ. Entomol. 2004, 33, 1609–1616. [Google Scholar] [CrossRef]
- Gill, H.K.; Garg, H. Pesticide: Environmental impacts and management strategies. In Pesticides—Toxic Effects; Solenski, S., Larramenday, M.L., Eds.; Intech: Rijeka, Croatia, 2014; pp. 187–230. [Google Scholar]
- Polson, K.A.; Brogdon, W.G.; Rawlins, S.C.; Chadee, D.D. Impact of environmental temperatures on resistance to organophosphate insecticides in Aedes aegypti from Trinidad. Rev. Panam. Salud Publ. 2012, 32, 1–8. [Google Scholar] [CrossRef]
- Liu, F.; Miyata, T.; Wu, Z.J.; Li, C.W.; Wu, G.; Zhao, S.X.; Xie, L.H. Effects of temperature and fitness costs, insecticide susceptibility and heat shock protein in insecticide resistant and susceptible Plutella xylostella. Pestic. Biochem. Physiol. 2008, 91, 45–52. [Google Scholar] [CrossRef]
- Metzger, M.J.; Bunce, R.G.H.; Trabucco, A.; Sayre, R.; Jangman, R.H.G.; Zomer, R.J. A high resolution bioclimate map for the world: A unifying framework for global biodiversity research and monitoring. Global Ecol. Biogeogr. 2013, 22, 630–638. [Google Scholar] [CrossRef]
- Food and Agriculture Organization (FAO). Identifying Opportunities for Climate-Smart Agriculture Investment in Africa; Economics & Policy Innovations for Climate-Smart Agriculture, FAO: Rome, Italy, 2012. [Google Scholar]
- Harvey, C.D. Integrated Pest Management in temperate horticulture: Seeing the wood for trees. CAB Rev. 2015, 10, 028. [Google Scholar] [CrossRef]
- Ngowi, A.V.; Maeda, D.W.; Partanen, T.J. Knowledge, Attitudes and Practices (KAP) among agricultural extension workers concerning the reduction of the adverse impact in agricultural areas in Tanzania. Crop Prot. 2007, 26, 1617–1624. [Google Scholar] [CrossRef] [PubMed]
- Baliga, S.S.; Repetto, R. Pesticides and the Immune System: The Public Health Risks; World Resources Institute: Washington, DC, USA, 1996. [Google Scholar]
- Tsimbiri, P.F.; Moturi, W.N.; Sawe, J.; Henley, P.; Bend, J.R. Health impact of pesticides on residents and horticultural workers in the Lake Naivasha Region, Kenya. Occup. Dis. Environ. Med. 2015, 3, 24–34. [Google Scholar] [CrossRef]
- Magauzi, R.; Mabaera, B.; Rusakaniko, S.; Chimusoro, A.; Ndlovu, N.; Tshimanga, M.; Shambira, G.; Chadambuka, A.; Gombe, N. Health effects of agrochemicals among farm workers in commercial farms of Kwekwe district, Zimbabwe. Pan Afr. Med. J. 2011, 9, 26. [Google Scholar] [CrossRef] [PubMed]
- Khoza, S.; Nhachi, C.F.B.; Chikumo, O.; Murambiwa, W.; Ndudzo, A.; Bwakura, E.; Mhonda, M. Organophosphate and organochlorine poisoning in selected horticultural farms in Zimbabwe. JASSA 2003, 9, 7–15. [Google Scholar]
- Mvumi, B.M.; Giga, D.P.; Chiuswa, D.V. The maize (Zea mays L.) post-production practices of smallholder farmers in Zimbabwe: Findings from surveys. JASSA 1995, 1, 115–130. [Google Scholar] [CrossRef]
- Horna, D.; Falk-Zepeda, J.; Timpo, S.E. Insecticide Use on Vegetables in Ghana. Would GM Seeds Benefit Farmers; International Food Policy Research Institute (IFPRI), Environment and Production Technology Division: Accra, Ghana, 2008. [Google Scholar]
- Abang, A.; Kouame, C.M.; Abang, M.M.; Hanna, R.; Kuate, A.F. Vegetable growers perception of pesticide use practices, cost and health effects in the tropical region of Cameroon. Int. J. Agron. Plant Prod. 2013, 4, 873–883. [Google Scholar]
- Mudimu, G.D.; Waibel, H.; Fleischer, S. Pesticide Policies in Zimbabwe: Status and Implications for Change; Pesticide Policy Project; Special Issue Publication Series 1; Institute of Horticultural Economics: Hannover, Germany, 1999. [Google Scholar]
- Tabashnik, B.E.; Malvar, T.; Liu, Y.B.; Finson, N.; Borthakur, D.; Shin, B.S.; Parck, S.H.; Masson, L.; Maard, R.A.; Bosch, D. Cross resistance of the diamondback moth indicates altered interactions with domain II of Bacillus thuringiensis Toxins. J. Appl. Environ. Microbiol. 1996, 62, 2839–2844. [Google Scholar]
- Xia, Y.; Lu, Y.; Shen, J.; Gao, X.; Qiu, H.; Li, J. Resistance monitoring for eight insecticides in Plutella xylostella in central China. Crop Prot. 2014, 63, 131–137. [Google Scholar] [CrossRef]
- Legwaila, M.M.; Munthali, D.C.; Kwerepe, B.C.; Obopile, M. Effectiveness of cypermethrin against diamondback moth (Plutella xylostella L.) eggs and larvae in cabbage under Botswana conditions. Afr. J. Agric. Res. 2014, 9, 3704–3710. [Google Scholar]
- Legwaila, M.M.; Munthali, D.C.; Kwerepe, B.C.; Obopile, M. Efficacy of Bacillus thuringiensis (var. kurstaki) against diamondback moth Plutella xylostella (L.) Eggs and larvae on cabbage under semi controlled greenhouse conditions. Int. J. Trop. Insect Sci. 2015, 7, 39–45. [Google Scholar]
- Kfir, R. Effect of parasitoid elimination on populations of diamondback moth in cabbage. In The Management of Diamondback Moth and Other Crucifer Pests, Proceedings of the 4th International Workshop, Melbourne, Australia, 26–29 November 2001; Endersby, N.M., Ridland, P.M., Eds.; The Regional Institute Ltd.: Gosford, Australia, 2004. [Google Scholar]
- Lӧhr, B. Toward biocontrol based IPM for the diamondback moth in Eastern and Southern Africa. In The Management of Diamondback Moth and Other Crucifer Pests, Proceedings of the 4th International Workshop. Melbourne, Australia, 26–29 November 2001; Endersby, N.M., Ridland, P.M., Eds.; The Regional Institute Ltd.: Gosford, Australia, 2004; pp. 197–206. [Google Scholar]
- Nyambo, B.; Löhr, B. The role and significance of farmer participation in biological control based IPM for brassica crops in East Africa. In Proceedings of the Second International Symposium on Biological Control of Arthropods, Davos, Switzerland, 12–16 September 2005.
- Tonnang, N.E.Z.; Nodorezov, L.V.; Owino, O.; Ochanda, H.; Lӧhr, B. Evaluation of Discrete host-parasitoid models for diamondback moth and Diadegma semiclausum field time population densities. Ecol. Model. 2009, 220, 1735–1744. [Google Scholar] [CrossRef]
- Bopape, M.J. The Management of Diamondback Moth, Plutella xylostella (L.) (Lepidoptera: Plutellidae), Population Density on Cabbage Using Chemical and Biological Control Methods. Master’s Thesis, University of South Africa, Pretoria, South Africa, 2013. [Google Scholar]
- Luchen, S.W.S. Effects of Intercropping Cabbage with Alliums and Tomato on the Incidences of Diamondback Moth Plutella xylostella (L.). Master’s Thesis, University of Zambia, Lusaka, Zambia, 2001. [Google Scholar]
- Karavina, C.; Mandumbu, R.; Zivenge, E.; Munetsi, T. Use of garlic Allium sativum as a repellent crop to control diamondback moth (Plutella xylostella) in cabbage (Brassica oleracia var. capitata). J. Agric. Res. 2014, 52, 615–622. [Google Scholar]
- Opena, R.T.; Kyomo, M.L. Vegetable Research and Development in SADCC Countries. Available online: http://trove.nla.gov.au/work/7530097?selectedversion=NBD7890552 (accessed on 5 January 2017).
- Green, S.K.; Shanmugasundaram, S. AVRDC’s International Networks to Deal with the Tomato Leaf Curl Disease: The Needs for Developing Countries; Springer: Dordrecht, The Netherlands, 2007; pp. 417–439. [Google Scholar]
- Maredia, M.K.; Dakomo, D.; Mota-Sanchez, D. Integrated Pest Management in the Global Arena; Commonwealth Agricultural Bureau International (CABI): Wallingford, UK, 2003. [Google Scholar]
- Devine, G.J.; Furlong, M.J. Insecticide use: Contexts and Ecological Consequences. Agric. Hum. Values 2007, 24, 281–306. [Google Scholar] [CrossRef]
- Walsh, B. Impact of Insecticides on Natural Enemies in Brassica Vegetables; Horticulture Australia Ltd.: Sydney, Australia, 2005. [Google Scholar]
- Kfir, R. Biological control of the diamondback moth Plutella xylostella in Africa. In Biological Control in IPM Systems in Africa; Neuenschwander, P., Borgemeister, J., Langewald, J., Eds.; CABI Publishing: Wallingford, UK; Cambridge, MA, USA, 2003; pp. 363–376. [Google Scholar]
- Safraz, M.; Dosdall, L.M.; Keddie, B.A. Diamondback moth host plant interactions: Implications for pest management. Crop Prot. 2006, 25, 625–639. [Google Scholar] [CrossRef]
- Rossbach, A.; Lӧhr, B.; Vidal, S. Host shift to peas in the diamondback moth Plutella xylostella (Lepidoptera: Plutellidae) and response of its parasitoid Diadegma mollipla (Hymenoptera: Ichneumonidae). Bull. Entomol. Res. 2006, 96, 413–419. [Google Scholar] [CrossRef] [PubMed]
- Silva-Torres, C.S.A.; Torres, J.B.; Barros, R. Can cruciferous agro-ecosystems grown under variable conditions influence biological control of Plutella xylostella (L.) (Lepidoptera: Plutellidae). Biocontrol Sci. Technol. 2011, 21, 625–641. [Google Scholar] [CrossRef]
- Sohati, H.P. Establishment of Cotesia vestalis (Haliday) and Diadromus collaris (Grav.) Parasitoids of the Diamondback Moth Plutella xylostella (L.) and Assessment of the Effectiveness of Cotesia vestalis as a Biological Control Agent in Zambia. Ph.D. Thesis, University of Zambia, Lusaka, Zambia, 2012. [Google Scholar]
- Mazlan, N.; Mumford, J. Insecticide use in cabbage pest management in Cameron highlands, Malaysia. Crop Prot. 2004, 24, 31–39. [Google Scholar] [CrossRef]
- Price, L.L. Demystifying farmers’ entomological and pest management knowledge: A methodology for assessing the impacts on knowledge from IPM-FFS and NES Interventions. Agric. Hum. Values 2001, 18, 153–176. [Google Scholar] [CrossRef]
- Stadlinger, N.; Mmochi, A.J.; Dobo, S.; Glyllback, E.; Kumblad, L. Pesticide use among smallholder rice farmers in Tanzania. Environ. Dev. Sustain. 2011, 13, 641–656. [Google Scholar] [CrossRef]
- Williamson, S. Understanding natural enemies: A review of training and information in the practical use of biological control. Biocontrol News Inf. 1998, 19, 117–126. [Google Scholar]
- Hough, P. The Global Politics of Pesticides: Forging Consensus from Conflicting Interests; Earthscan Publications Ltd.: London, UK, 1998. [Google Scholar]
- Association of Veterinary and Crop Associations of South Africa (AVCASA). Hands off Banned Pesticides; Media Statement; AVCASA: Midrand, South Africa, 2008. [Google Scholar]
- Quinn, L.P.; de Vos, B.J.; Fernandes-Whaley, M.; Roos, C.; Bouwman, H.; Kylin, H.; Pieters, R.; van den Berg, J. Pesticide Use in South Africa: One of the Largest Importers of Pesticides in Africa. 2012. Available online: http://www.intechopen.com/books/pesticides-in-the-modern-world-pesticides-use-and-management/pesticide-use-in-south-africa-one-of-the-largest-importers-of pesticides-in-africa (accessed on 5 January 2017).
- Lӧhr, B.; Kfir, R. Diamond back in Africa: A review with emphasis on Biological Control. In Improving Biocontrol of Plutella xylostella; Kirk, A.A., Bordat, D., Eds.; Agricultural Research for Development: Montpellier, France, 2004. [Google Scholar]
- Momanyi, C.; Lӧhr, B.; Gitonga, L. Biological Impact of the Exotic Parasitoid Diadegma semiclausum (Hellen) of diamondback moth Plutella xylostella (L.) in Kenya. Biol. Control 2006, 38, 254–263. [Google Scholar] [CrossRef]
- Lӧhr, B.; Kfir, R. Diamondback moth Plutella xylostella (L.) in Africa: A review with emphasis on biological control. In Proceedings of the International Symposium, Montpellier, France, 21–24 October 2002.
- Chidawanyika, F.; Terblanche, J.S. Costs and benefits of thermal acclimation for codling moth, Cydia pomonella (Lepidoptera: Tortricidae): Implications for pest control and the sterile insect release programme. Evol. Appl. 2011, 4, 534–544. [Google Scholar] [CrossRef] [PubMed]
- Sørensen, J.; Addison, M.; Terblanche, J.S. Mass rearing of insects for pest management: Challenges, synergies and advances from evolutionary physiology. Crop Prot. 2012, 38, 87–94. [Google Scholar] [CrossRef]
- Overgaard, J.; Sørensen, J.G. Rapid thermal adaptation during field temperature variations in Drosophila melanogaster. Cryobiology 2008, 56, 159–162. [Google Scholar] [CrossRef] [PubMed]
- Kristensen, T.N.; Hoffmann, A.A.; Overgaard, J.; Sørensen, J.G.; Hallas, R. Costs and benefits of cold acclimation in field released Drosophila. Proc. Natl. Acad. Sci. USA 2008, 105, 216–221. [Google Scholar] [CrossRef] [PubMed]
- Htun, P.W.; Myint, M. Radiation induced sterility for biological control of diamondback moth Plutella xylostella (L.). Int. J. Adv. Sci. Eng. Technol. 2014, 4, 285–291. [Google Scholar]
- Dyck, V.A.; Hendrichs, J.; Robinson, A.S. Sterile Insect Technique: Principles and Practice in Area-Wide Integrated Pest Management; Dyck, V.A., Hendrichs, J., Robinson, A.S., Eds.; Springer: Dordrecht, The Netherlands, 2005. [Google Scholar]
- Harvey-Samuel, T.; Morrison, N.I.; Walker, A.I.; Marubbi, T.; Yao, J.; Collins, H.L.; Gorman, K.; Davies, T.G.E.; Alphey, N.; Warner, S.; et al. Pest control and resistance management through release of insects carrying a male sterile transgene. BMC Biol. 2015, 13, 49–64. [Google Scholar] [CrossRef] [PubMed]
- Ant, T.; Koukidou, M.; Rempoulakis, P.; Gong, H.F.; Economopoulos, A.; Vontas, J.; Alphey, L. Control of the olive fruit fly using genetics-enhanced sterile insect technique. BMC Biol. 2012, 10, 51. [Google Scholar] [CrossRef] [PubMed]
- Leftwich, P.T.; Koukidou, M.; Rempoulakis, P.; Gong, H.F.; Zacharopoulou, A.; Fu, G.; Chapman, T.; Economopoulos, A.; Vontas, J.; Alphey, L. Genetic elimination of field-cage populations of Mediterranean fruit flies. Proc. R. Soc. Biol. Sci. 2014, 281, 1792. [Google Scholar] [CrossRef] [PubMed]
- De Valdez, M.R.W.; Nimmo, D.; Betz, J.; Gong, H.F.; James, A.A.; Alphey, L. Genetic elimination of dengue vector mosquitoes. Proc. Natl. Acad. Sci. USA 2011, 108, 4772–4775. [Google Scholar] [CrossRef] [PubMed]
- Van Veen, S. The worldbank and pest management. In Integrated Pest Management in the Global Arena; Maredia, K.M., Dakouo, D., Mota-Sanchez, D., Eds.; Commonwealth Agricultural Bureau International (CABI): Wallingford, UK, 2003. [Google Scholar]
- Gillson, L.; Dawson, T.P.; Jack, S.; McGeoch, M.A. Accommodating climate change contingencies in conservation strategy. Trends Ecol. Evol. 2013, 28, 135–142. [Google Scholar] [CrossRef] [PubMed]
- Cook, S.M.; Khan, Z.R.; Pickett, J.A. The use of push-pull strategies in Integrated Pest Management. Ann. Rev. Entomol. 2007, 52, 375–400. [Google Scholar] [CrossRef] [PubMed]
- Khan, R.Z.; Midega, C.A.O.; Bruce, T.J.A.; Hooper, A.M.; Pickett, J.A. Exploiting phyto-chemicals for developing a “push-pull” crop protection strategy for cereal farmers in Africa. J. Exp. Bot. 2010, 10, 1–12. [Google Scholar]
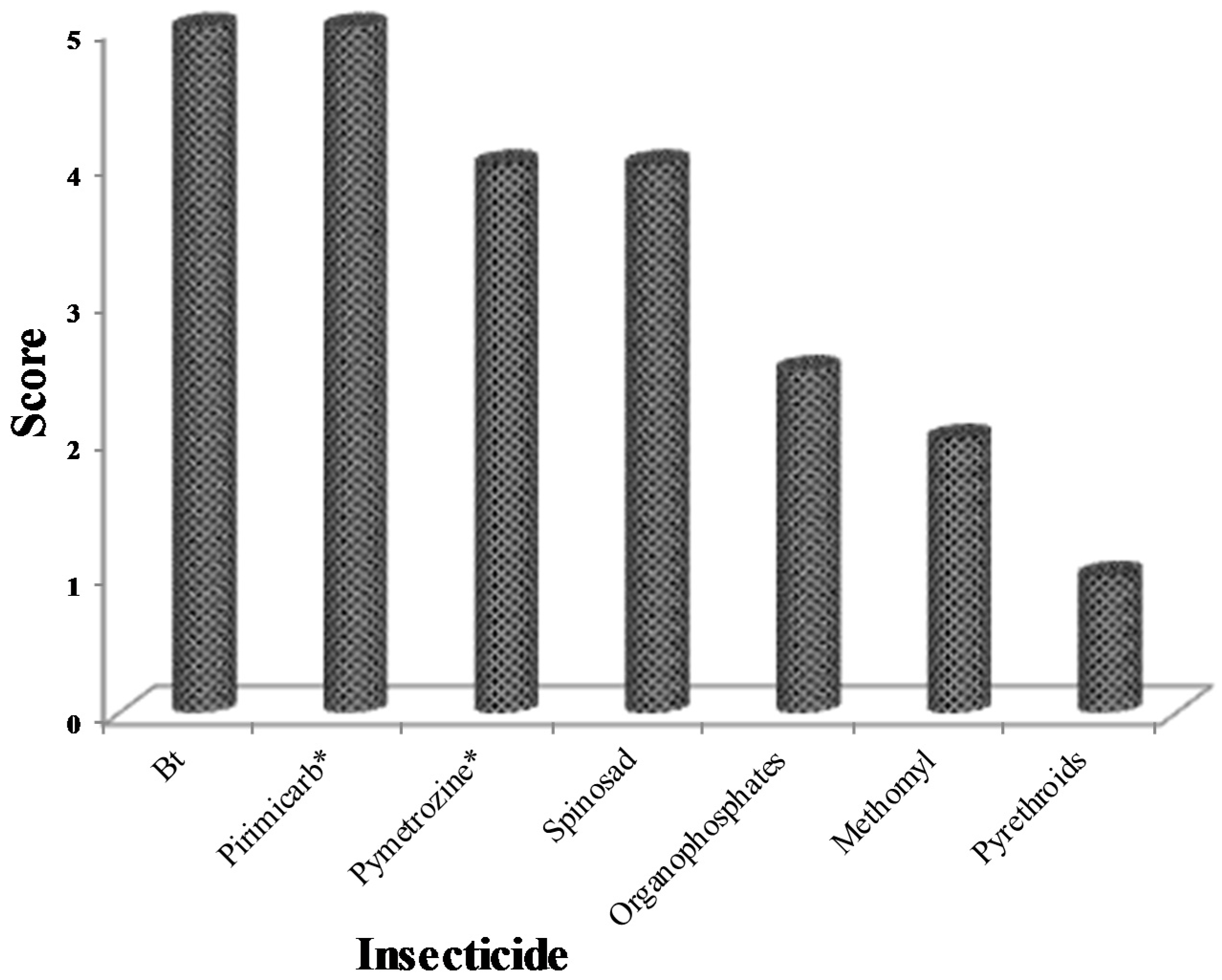
| Country | Farmers Using Pesticides (%) | Reference |
|---|---|---|
| Southern Africa | ||
| Mozambique | 100 | [5] |
| Botswana | 98 | [20] |
| Zimbabwe | No data | |
| Zambia | 75 | [40] |
| Malawi | 75 | [40] |
| Other selected African countries | ||
| Tanzania | 98 | [60] |
| Cameron | 90 | [66] |
| Ghana | 85 | [67] |
| Kenya | No data | |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Machekano, H.; Mvumi, B.M.; Nyamukondiwa, C. Diamondback Moth, Plutella xylostella (L.) in Southern Africa: Research Trends, Challenges and Insights on Sustainable Management Options. Sustainability 2017, 9, 91. https://doi.org/10.3390/su9020091
Machekano H, Mvumi BM, Nyamukondiwa C. Diamondback Moth, Plutella xylostella (L.) in Southern Africa: Research Trends, Challenges and Insights on Sustainable Management Options. Sustainability. 2017; 9(2):91. https://doi.org/10.3390/su9020091
Chicago/Turabian StyleMachekano, Honest, Brighton M. Mvumi, and Casper Nyamukondiwa. 2017. "Diamondback Moth, Plutella xylostella (L.) in Southern Africa: Research Trends, Challenges and Insights on Sustainable Management Options" Sustainability 9, no. 2: 91. https://doi.org/10.3390/su9020091
APA StyleMachekano, H., Mvumi, B. M., & Nyamukondiwa, C. (2017). Diamondback Moth, Plutella xylostella (L.) in Southern Africa: Research Trends, Challenges and Insights on Sustainable Management Options. Sustainability, 9(2), 91. https://doi.org/10.3390/su9020091





