Nitrogen and Sediment Capture of a Floating Treatment Wetland on an Urban Stormwater Retention Pond—The Case of the Rain Project
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Site and Water Quality Monitoring
2.2. FTW Construction
2.3. Plant Selection and Setup
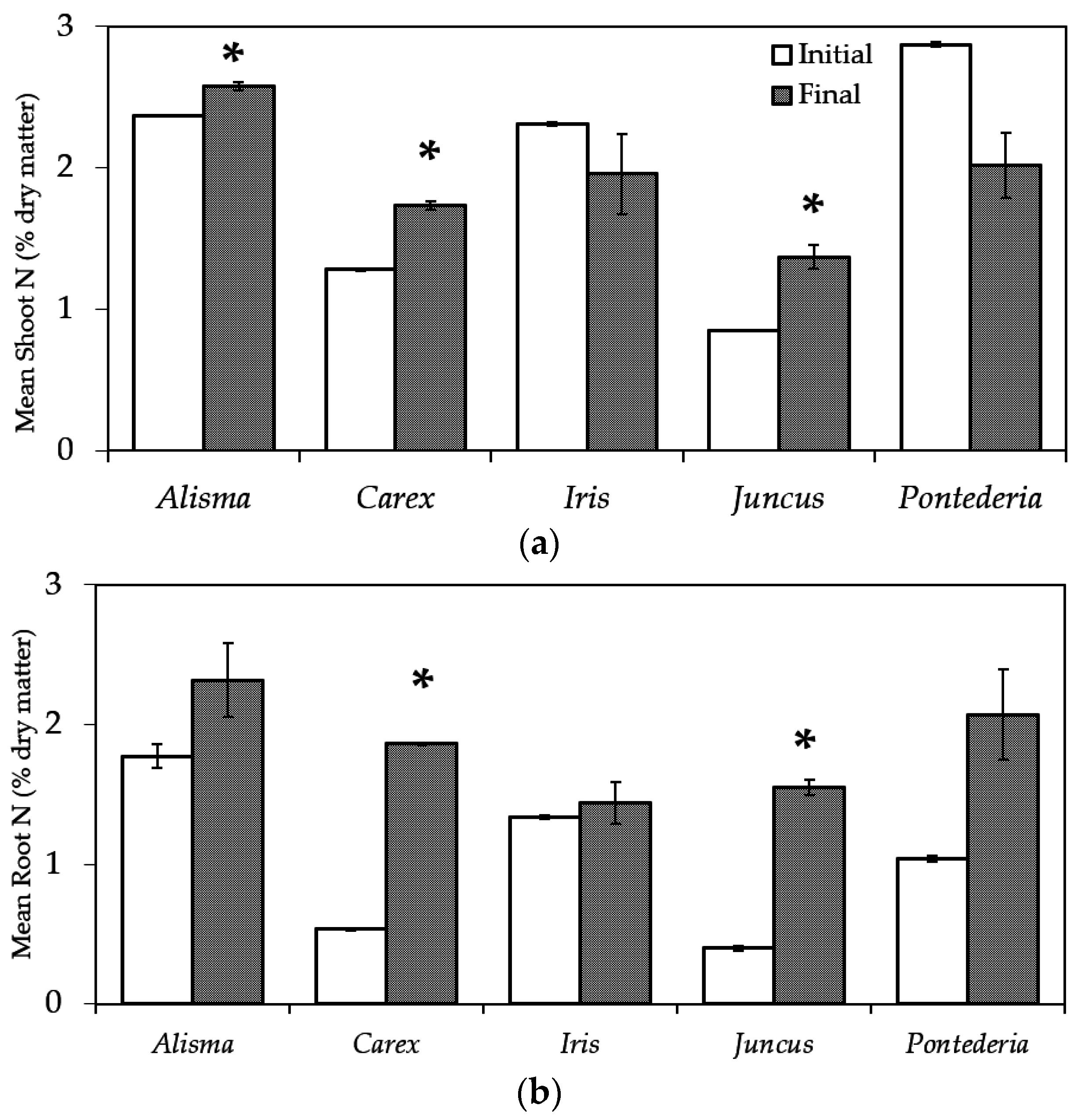
2.4. Plant Biomass Measurement and Tissue Analysis
2.5. Sediment Analysis
2.6. Data Analysis
3. Results and Discussion
3.1. Mason Pond Water Quality
3.2. Plant Biomass Production and Nitrogen Uptake
3.3. Sediment and Sediment N Capture
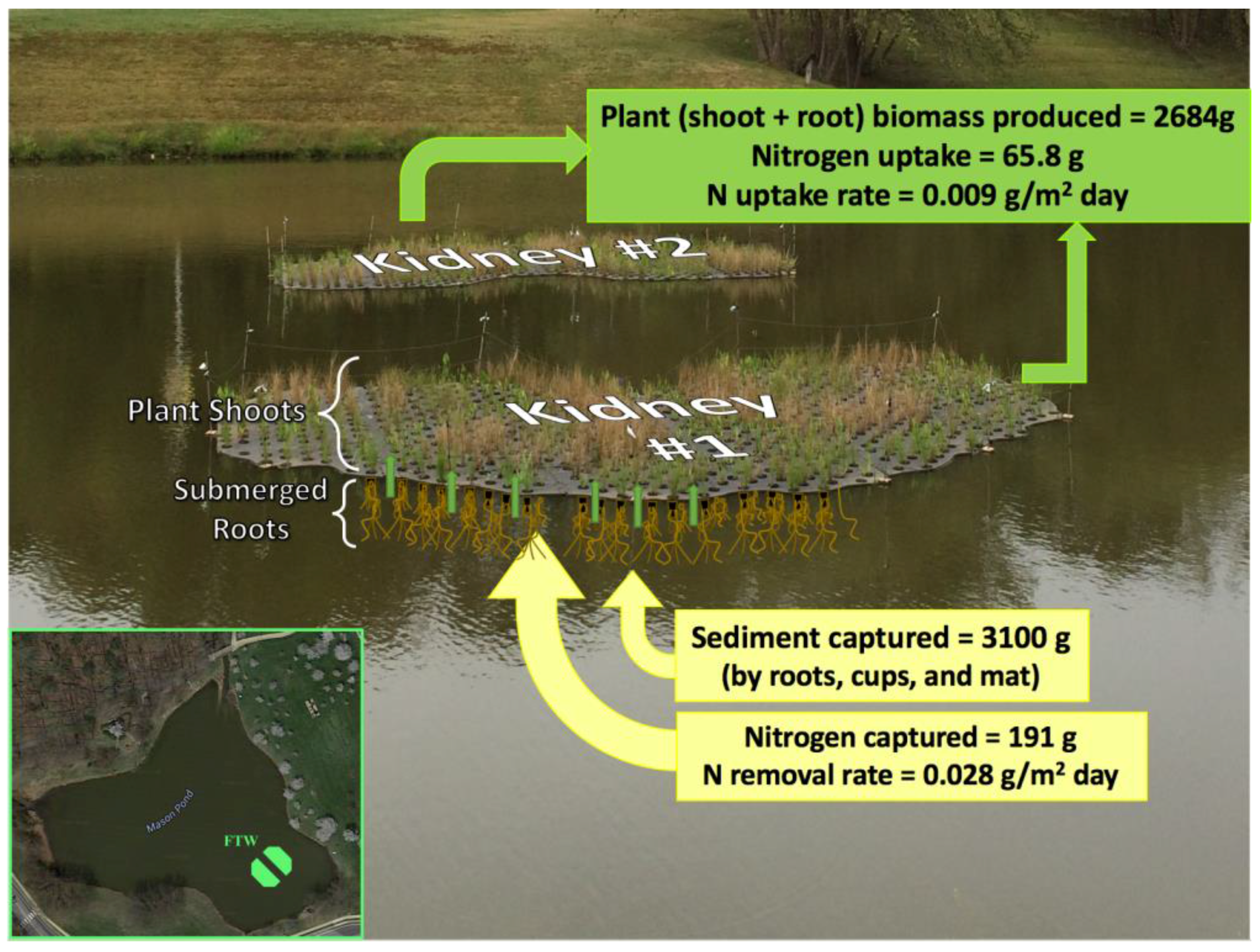
3.4. Nitrogen Budget
3.5. Implications of the Study and Recommendations
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Fletcher, T.; Duncan, H.; Poelsma, P.; Lloyd, S. Stormwater Flow and Quality, and the Effectiveness of Non-Proprietary Stormwater Treatment Measures: A Review and Gap Analysis; Cooperative Research Centre for Catchment Hydrology: Clayton, Australia, 2004. [Google Scholar]
- Yang, G.; Bowling, L.C.; Cherkauer, K.A.; Pijanowski, B.C. The impact of urban development on hydrologic regime from catchment to basin scales. Landsc. Urban Plan. 2011, 103, 237–247. [Google Scholar] [CrossRef]
- Carey, R.O.; Hochmuth, G.J.; Martinez, C.J.; Boyer, T.H.; Dukes, M.D.; Toor, G.S.; Cisar, J.L. Evaluating nutrient impacts in urban watersheds: Challenges and research opportunities. Environ. Pollut. 2013, 173, 138–149. [Google Scholar] [CrossRef] [PubMed]
- U.S. Environmental Protection Agency. Preliminary Data Summary of Urban Storm Water Best Management Practices; EPA: Washington, DC, USA, 1999.
- Fletcher, T.D.; Andrieu, H.; Hamel, P. Understanding, management and modelling of urban hydrology and its consequences for receiving waters: A state of the art. Adv. Water Resour. 2013, 51, 261–279. [Google Scholar] [CrossRef]
- Collins, K.A.; Lawrence, T.J.; Stander, E.K.; Jontos, R.J.; Kaushal, S.S.; Newcomer, T.A.; Grimm, N.B.; Cole Ekberg, M.L. Opportunities and challenges for managing nitrogen in urban stormwater: A review and synthesis. Ecol. Eng. 2010, 36, 1507–1519. [Google Scholar] [CrossRef]
- Winston, R.J.; Hunt, W.F.; Kennedy, S.G.; Merriman, L.S.; Chandler, J.; Brown, D. Evaluation of floating treatment wetlands as retrofits to existing stormwater retention ponds. Ecol. Eng. 2013, 54, 254–265. [Google Scholar] [CrossRef]
- Fairfax County. Stormwater Management Pond Quick Reference Guide. Available online: http://www.fairfaxcounty.gov/dpwes/environmental/swm_quickref.htm (accessed on 22 May 2016).
- U.S. Environmental Protection Agency. Storm Water Technology Fact Sheet—Wet Detention Ponds; EPA: Washington, DC, USA, 1999.
- Borne, K.E.; Fassman, E.A.; Tanner, C.C. Floating treatment wetland retrofit to improve stormwater pond performance for suspended solids, copper and zinc. Ecol. Eng. 2013, 54, 173–182. [Google Scholar] [CrossRef]
- Hunt, W.F. Maintenance of Stormwater Wetlands and Wet Ponds; North Carolina Cooperative Extension Service: Raleigh, NC, USA, 2006; Available online: https://brunswick.ces.ncsu.edu/wp-content/uploads/2013/04/Wetland-and-Pond-Maintenance-2006.pdf (accessed on 2 June 2016).
- Rollins, S. Neighborhood Stormwater Pond Maintenance Log and Resources; Clemson University: Clemson, SC, USA, 2016; Available online: https://www.clemson.edu/extension/natural_resources/water/stormwater_ponds/resources/files/final_binder_pond_maintenance.pdf (accessed on 2 June 2016).
- EPA Program Overview: Total Maximum Daily Loads (TMDL). Available online: https://www.epa.gov/tmdl/program-overview-total-maximum-daily-loads-tmdl (accessed on 16 September 2016).
- Marimon, Z.A.; Xuan, Z.; Chang, N. System dynamics modeling with sensitivity analysis for floating treatment wetlands in a stormwater wet pond. Ecol. Model. 2013, 267, 66–79. [Google Scholar] [CrossRef]
- Mallin, M.A.; Ensign, S.H.; Wheeler, T.L.; Mayes, D.B. Pollutant Removal Efficacy of Three Wet Detention Ponds Michael. J. Environ. Qual. 2002, 31, 654–660. [Google Scholar] [CrossRef] [PubMed]
- VDEQ. Maintaining Your BMP: A Guidebook for Private Owners and Operators. 2000. Available online: http://www.deq.virginia.gov/Portals/0/DEQ/Water/Publications/BMPHandbook.pdf (accessed on 7 May 2016). [Google Scholar]
- Headley, T.R.; Tanner, C.C. Application of Floating Wetlands for Enhanced Stormwater Treatment: A Review; National Institute of Water & Atmospheric Research Ltd.: Hamilton, New Zealand, 2006. Available online: http://www.aucklandcity.govt.nz/council/documents/technicalpublications/TP324%20-%20Floating%20Wetland%20Review-Final.pdf (accessed on 16 May 2016).
- Sample, D.J.; Wang, C.-Y.; Fox, L.J. Innovative Best Management Fact Sheet No. 1: Floating Treatment Wetlands; Virginia Cooperative Extension: Blacksburg, VA, USA, 2013; Available online: https://pubs.ext.vt.edu/BSE/BSE-76/BSE-76-PDF.pdf (accessed on 4 April 2016).
- Wanielista, M.P.; Chang, N.-B.; Chopra, M.; Xuan, Z.; Islam, K.; Marimon, Z. Floating Wetland Systems for Nutrient Removal in Stormwater Ponds; Florida Department of Environmental Protection: Tallahassee, FL, USA, 2012; Available online: https://www.dep.state.fl.us/water/nonpoint/docs/nonpoint/final-report-ucf-fdot-beemats.pdf (accessed on 8 July 2016).
- Tanner, C.C.; Headley, T.R. Components of floating emergent macrophyte treatment wetlands influencing removal of stormwater pollutants. Ecol. Eng. 2011, 37, 474–486. [Google Scholar] [CrossRef]
- Kadlec, R.H.; Wallace, S. Treatment Wetlands, 2nd ed.; CRC Press: Boca Raton, FL, USA, 2009. [Google Scholar]
- Osem, Y.; Chen, Y.; Levinson, D.; Hadar, Y. The effects of plant roots on microbial community structure in aerated wastewater-treatment reactors. Ecol. Eng. 2007, 29, 133–142. [Google Scholar] [CrossRef]
- Borne, K.E.; Fassman-Beck, E.A.; Winston, R.J.; Hunt, W.F.; Tanner, C.C. Implementation and maintenance of floating treatment wetlands for urban stormwater management. J. Environ. Eng. 2015. [Google Scholar] [CrossRef]
- Headley, T.R.; Tanner, C.C. Constructed wetlands with floating emergent macrophytes: An innovative stormwater treatment technology. Crit. Rev. Environ. Sci. Technol. 2012, 42, 2261–2310. [Google Scholar] [CrossRef]
- Wang, C.Y.; Sample, D.J. Assessment of the nutrient removal effectiveness of floating treatment wetlands applied to urban retention ponds. J. Environ. Manag. 2014, 137, 23–35. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.-Y.; Sample, D.J.; Bell, C. Vegetation effects on floating treatment wetland nutrient removal and harvesting strategies in urban stormwater ponds. Sci. Total Environ. 2014, 499, 384–393. [Google Scholar] [CrossRef] [PubMed]
- Marschner, H. Mineral Nutrition of Higher Plants; Academic Press: London, UK; San Diego, CA, USA, 1995. [Google Scholar]
- Williamson, L.C. Phosphate availability regulates root system architecture in Arabidopsis. Plant Physiol. 2001, 126, 875–882. [Google Scholar] [CrossRef] [PubMed]
- Meuleman, A.F.M.; Beekman, J.P.; Verhoeven, J.T.A. Nutrient retention and nutrient-use efficiency in Phragmites australis stands after wasterwater application. Wetlands 2002, 22, 712–721. [Google Scholar] [CrossRef]
- Ahn, C. A creative collaboration between the science of ecosystem restoration and art for sustainable stormwater management on an urban college campus. Restor. Ecol. 2016, 24, 291–297. [Google Scholar] [CrossRef]
- George Mason University (GMU). MS4 Annual Report; George Mason University: Fairfax, VA, USA, 2015. [Google Scholar]
- George Mason University. Mason SWMP Draft 2013; George Mason University: Fairfax, VA, USA, 2013. [Google Scholar]
- National Oceanic and Atmospheric Administration (NOAA). Climate Data Inventories; Vienna Station: Fairfax, VA, USA, 2016.
- George Mason University. MS4 TMDL Action Plan; George Mason University: Fairfax, VA, USA, 2015. [Google Scholar]
- Floating Islands International, Inc. BioHaven® Technology. Available online: http://www.floatingislandinternational.com/products/biohaven-technology/ (accessed on 23 September 2016).
- BeeMats, LLC. BeeMats: Floating Wetlands. Available online: http://beemats.com/home.html (accessed on 23 September 2016).
- De Stefani, G.; Tocchetto, D.; Salvato, M.; Borin, M. Performance of a floating treatment wetland for in-stream water amelioration in NE Italy. Hydrobiologia 2011, 674, 157–167. [Google Scholar] [CrossRef]
- Wang, C.-Y.; Sample, D.J. Assessing floating treatment wetlands nutrient removal performance through a first order kinetics model and statistical inference. Ecol. Eng. 2013, 61, 292–302. [Google Scholar] [CrossRef]
- Henry, J.H.; Henry, E.T. Plant Root Pruning Container 2007. Available online: https://www.google.com/patents/US7210266 (accessed on 23 September 2016).
- NBC 4 Washington. Students Say Pond Class at GMU Really Floats Their Boats. Available online: http://www.nbcwashington.com/news/local/304771321.html (accessed on 25 May 2015).
- Tanner, C.C. Plants for constructed wetland treatment systems—A comparison of the growth and nutrient uptake of eight emergent species. Ecol. Eng. 1996, 7, 59–83. [Google Scholar] [CrossRef]
- Means, M.M.; Ahn, C.; Korol, A.R.; Williams, L.D. Carbon storage potential by four macrophytes as affected by planting diversity in a created wetland. J. Environ. Manag. 2016, 165, 133–139. [Google Scholar] [CrossRef] [PubMed]
- Lynch, J.; Fox, L.J.; Owen, J.S.; Sample, D.J. Evaluation of commercial floating treatment wetland technologies for nutrient remediation of stormwater. Ecol. Eng. 2015, 75, 61–69. [Google Scholar] [CrossRef]
- Wang, C.-Y.; Sample, D.J.; Day, S.D.; Grizzard, T.J. Floating treatment wetland nutrient removal through vegetation harvest and observations from a field study. Ecol. Eng. 2015, 78, 15–26. [Google Scholar] [CrossRef]
- Lorenzen, B.; Brix, H.; Mendelssohn, I.A.; McKee, K.L.; Miao, S.L. Growth, biomass allocation and nutrient use efficiency in Cladium jamaicense and Typha domingensis as affected by phosphorus and oxygen availability. Aquat. Bot. 2001, 70, 117–133. [Google Scholar] [CrossRef]
- Lenhart, H.A.; Hunt, W.F.; Burchell, M.R. Harvestable nitrogen accumulation for five storm water wetland plant species: Trigger for storm water control measure maintenance? J. Environ. Eng. 2012, 138, 972–978. [Google Scholar] [CrossRef]
- Borne, K.E.; Tanner, C.C.; Fassman-Beck, E.A. Stormwater nitrogen removal performance of a floating treatment wetland. Water Sci. Technol. 2013, 68, 1657–1664. [Google Scholar] [CrossRef] [PubMed]


| Parameter | Value |
|---|---|
| Area (m2) | ~7100 |
| Volume (m3) | ~7810 |
| Mean Depth (m) | 1.10 |
| Mean Water Temperature (°C) | 29.0 ± 2.7 |
| Mean pH | 7.51 ± 0.80 |
| Mean Dissolved Oxygen (mg/L) | 9.71 ± 2.05 |
| Mean TSS (mg/L) | 21.8 ± 10.5 |
| Plant Species | Alisma | Carex | Iris | Juncus | Pontederia |
|---|---|---|---|---|---|
| # of individuals planted | 200 | 350 | 170 | 500 | 290 |
| Below-Mat | Above-Mat | A:B | |||
|---|---|---|---|---|---|
| Plant Species | Biomass (g) | Nitrogen (mg) | Biomass (g) | Nitrogen (mg) | Biomass Ratio |
| Alisma subcordatum | 0.82 ± 0.19 | 18.89 ± 2.15 | 0.47 ± 0.12 | 12.18 ± 0.13 | 0.57 |
| Carex stricta | 2.82 ± 0.38 * | 52.62 ± 0.25 | 1.49 ± 0.13 * | 25.79 ± 0.47 | 0.52 |
| Iris versicolor | 1.77 ± 0.24 | 25.42 ± 2.62 | 0.93 ± 0.14 | 18.23 ± 2.64 | 0.52 |
| Juncus effusus | 2.25 ± 0.35 | 34.85 ± 1.21 | 2.22 ± 0.41 | 30.44 ± 1.89 | 0.99 |
| Pontederia cordata | 0.69 ± 0.19 | 14.36 ± 2.25 | 1.55 ± 0.31 | 31.26 ± 3.57 | 2.24 |
| FTW Component | |||||||
|---|---|---|---|---|---|---|---|
| Plant Species | Mat | ||||||
| Alisma | Carex | Iris | Juncus | Pontederia | Total | ||
| Biomass (g) | |||||||
| per plant | 0.42 | 3.29 | 2.51 | −0.79 * | 2.02 | — | — |
| FTW Total | 84.17 | 1151.79 | 425.94 | −392.80 * | 584.78 | — | 2684.16 |
| Nitrogen—Plant Biomass (g) | |||||||
| per plant | 0.014 | 0.068 | 0.042 | 0.032 | 0.041 | — | — |
| FTW total | 3.03 | 24.77 | 6.82 | 19.11 | 12.06 | — | 65.80 |
| Sediment (g) | |||||||
| per plant-cup | 0.89 | 1.44 | 1.16 | 1.21 | 1.04 | — | — |
| FTW Total | 178.98 | 503.40 | 196.75 | 606.06 | 301.96 | 1312.97 | 3100.12 |
| Nitrogen—Sediment (g) | |||||||
| per plant-cup | 0.03 | 0.04 | 0.04 | 0.03 | 0.04 | — | — |
| FTW Total | 6.79 | 14.00 | 7.28 | 15.55 | 10.48 | 71.47 | 125.57 |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
McAndrew, B.; Ahn, C.; Spooner, J. Nitrogen and Sediment Capture of a Floating Treatment Wetland on an Urban Stormwater Retention Pond—The Case of the Rain Project. Sustainability 2016, 8, 972. https://doi.org/10.3390/su8100972
McAndrew B, Ahn C, Spooner J. Nitrogen and Sediment Capture of a Floating Treatment Wetland on an Urban Stormwater Retention Pond—The Case of the Rain Project. Sustainability. 2016; 8(10):972. https://doi.org/10.3390/su8100972
Chicago/Turabian StyleMcAndrew, Brendan, Changwoo Ahn, and Joanna Spooner. 2016. "Nitrogen and Sediment Capture of a Floating Treatment Wetland on an Urban Stormwater Retention Pond—The Case of the Rain Project" Sustainability 8, no. 10: 972. https://doi.org/10.3390/su8100972
APA StyleMcAndrew, B., Ahn, C., & Spooner, J. (2016). Nitrogen and Sediment Capture of a Floating Treatment Wetland on an Urban Stormwater Retention Pond—The Case of the Rain Project. Sustainability, 8(10), 972. https://doi.org/10.3390/su8100972







