Polymer Recovery from Auto Shredder Residue by Projectile Separation Method
Abstract
:1. Introduction
2. Review of ASR Recovery Process

| Material/component | Composition (% by weight) |
|---|---|
| Paper | 2% |
| Wood | 3% |
| Non ferrous metal | 4% |
| Wire harnesses | 5% |
| Rubber | 7% |
| Glass | 7% |
| Iron | 8% |
| Fabric | 15% |
| Urethane foam | 16% |
| Resins | 33% |
| TOTAL | 100% |
3. Current Segregation Methods
3.1. Float Sink Tank
3.2. Cyclonic Air Separator
3.3. Froth Flotation
3.4. Manual Sorting
3.5. Mid-Infra-Red (MIR) Spectroscopy
3.6. Electrostatic Separation
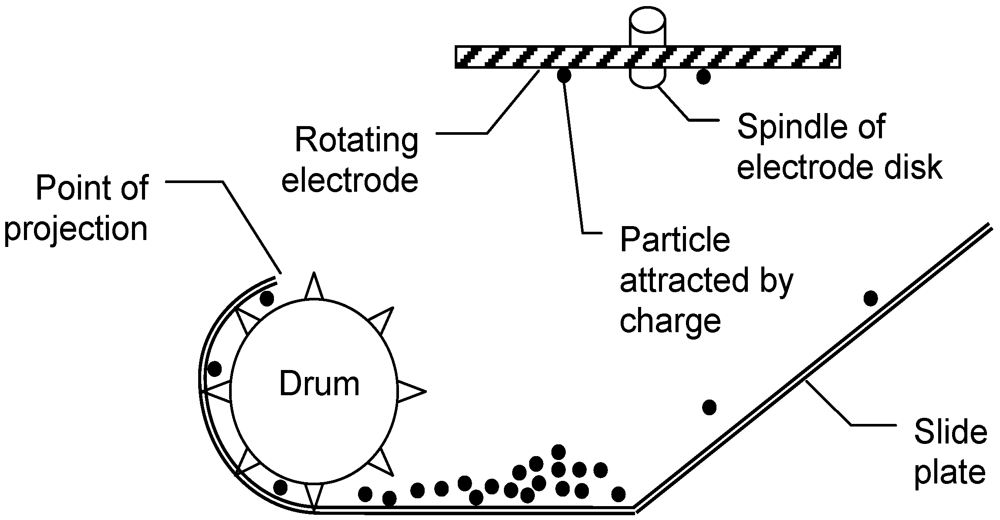
4. The Projectile Separation Method
4.1. Calculations for Prototype Footprint

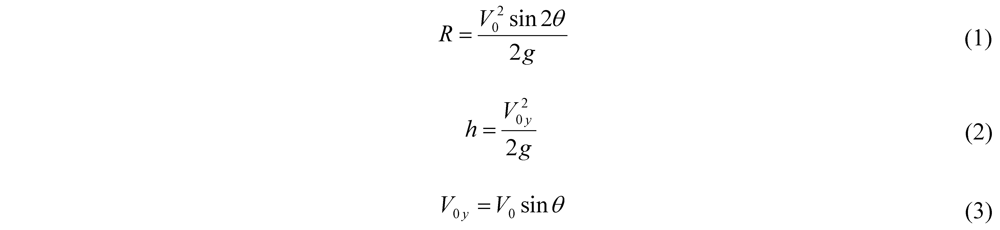
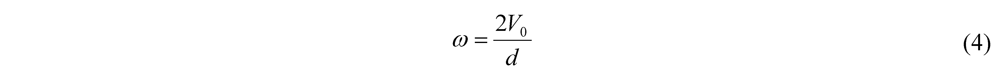

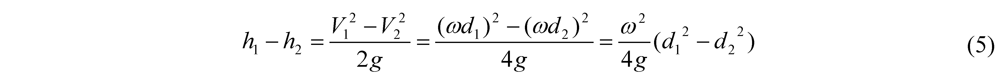
5. Experimental Prototype
6. Conclusions
References
- Stuadinger, J.; Keoleian, G.A. Management of End-of-Life Vehicles (ELVs) in the U.S.; Report CSS01-01; University of Michigan: Ann Arbor, MI, USA, March 2001. [Google Scholar]
- Australian Bureau of Statistics (ABS). 9309.0 Motor Vehicle Census 1996, Australia. Available online: http://www.abs.gov.au/AUSSTATS/abs@.nsf/DetailsPage/9309.031%20October%201996 (accessed on 7 April 2012).
- Australian Bureau of Statistics (ABS). 9309.0 Motor Vehicle Census 2006, Australia. Available online: http://www.abs.gov.au/AUSSTATS/abs@.nsf/DetailsPage/9309.031%20Mar%202006 (accessed on 7 April 2012).
- Australian Bureau of Statistics (ABS). 9314.0—Sales of new motor vehicles, Australia, October 2007. Available online: http://www.abs.gov.au/ausstats/abs@.nsf/ProductsbyReleaseDate/7976EE54AC6BE1D9CA2573B70011C49C?OpenDocument (accessed on 5 April 2012).
- Allan, P. End-of-Life motor vehicles: Market snapshot 2007. Available online: http://www.resourcesmart.vic.gov.au/documents/End_of_life_motor_vehicles.pdf (accessed on 7 April 2012).
- European Commission. 2000 directive 2000/53/EC of the European parliament and of the council of 18 September, 2000 on End-of-Life Vehicles. Official J. European Communities 2000, L269, 34–42.
- Australian Government, Environmental impact of End-of-Life Vehicles: An information paper. Department of the Environment and Heritage: Sydney, Australia, 2002; ISBN 0642547513.
- Mark, F.E.; Kamprath, A.E. End-of-Life Vehicles recovery and recycling polyurethane seat cushions recycling options analysis. Presented at SAE 2004 World Congress; SAE Technical Paper Series 2004-01-0249. SAE: Detroit, MI, USA, 2004; ISBN 0-7680-1319-4 2004. [Google Scholar]
- Keoleian, G.A.; Kar, K.; Manion, M.M.; Bulkley, J.W. Industrial Ecology of the Automobile: A Life Cycle Perspective; Society of Automotive Engineers Inc.: Warrendale, PA, USA, 1997. [Google Scholar]
- Sakai, S.-I.; Noma, Y.; Kida, A. End-of-Life vehicle recycling and automobile shredder residue management in Japan. J. Mater. Cycles Waste Manag. 2007, 9, 151–158. [Google Scholar]
- Jeong, K.M.; Hong, S.J.; Lee, J.Y.; Hur, T. Life cycle assessment on end-of life vehicle treatment system in korea. J. Ind. Eng. Chem. 2007, 13, 624–630. [Google Scholar]
- Isaacs, J.A.; Gupta, S.M. Economic consequences of increasing polymer content for the U.S. automobile recycling infrastructure. J. Industr. Ecol. 1998, 1, 19–33. [Google Scholar] [CrossRef]
- Choi, J.-K.; Stuart, J.A.; Ramini, K. Modelling of automobile recycling planning in the United States. Int. J. Auto. Tech. 2005, 6, 413–419. [Google Scholar]
- Lee, H.-Y.; Oh, J.-K. A study on the shredding of End-of Life Vehicles and materials separation. Geosystem Eng. 2003, 6, 100–105. [Google Scholar]
- Castro, M.B.G.; Remmerwaal, J.A.M.; Reuter, M.A. Life cycle impact assessment of the average passenger vehicle in the Netherland. Int. J. Life Cycle Ass. 2003, 8, 297–304. [Google Scholar]
- Ciacci, L.; Morselli, L.; Passarini, F.; Santini, A.; Vassura, I. A comparison among different automotive shredder residue treatment processes. Int. J. Life Cycle Ass. 2010, 15, 896–906. [Google Scholar]
- Nourreddine, M. Recycling of auto shredder residue. J. Hazard. Mater. 2006, 139, 481–490. [Google Scholar] [CrossRef]
- Kanari, N.; Pineau, J.-L.; Shallari, S. End-of-life vehicle recycling in the European Union. J. Miner. Met. Mater. Soc. 2003, 55, 15–19. [Google Scholar]
- Hoffman, J.M. New life for shredded plastic waste. Machine Design. Available online: http://machinedesign.com/article/new-life-for-shredded-plastic-waste-0207 (accessed on 7 February 2008).
- Bellman, K.; Khare, A. European response to issues in recycling car plastics. Technovation 1999, 19, 721–734. [Google Scholar]
- Lofti, A. Automotive recycling. Available online: http://www.lotfi.net/recycle/ (accessed 29 September 2011).
- Malmgren-Hansen, B.; Picot, P. Position Paper ELV; Danish Technological Institute, 2005. Available online: http://www.wastexchange.co.uk/documenti/elv/C%20_bmhfiler_tekstbeh_word97dti1_proj05_verc_wp3_virtual_discussion_platforms_position_papers_Position_paper_ELVv4.pdf (accessed on 7 April 2012).
- Pagenkopf, I.; Allies, M. Process for separating solid mixture of different density, separating liquid and device for implementing the process. U.S. Patent 5,738,222, 14 April 1998. [Google Scholar]
- Müller, P.; von Heyking, A.; Leitzke, J. Method and device for recovering pure PVC from plastic waste containing PVC. World Intellectual Property Organisation Publication Number WO/2000/010787, 3 February 2000. [Google Scholar]
- Jung, W.-H.; Choi, Y.-S.; Moon, J.-M.; Tortorella, N.; Beatty, C.L.; Lee, J.-O. Nonreactive processing of recycled polycarbonate/acrylonitrile–butadiene–styrene. Environ. Eng. Sci. 2010, 27, 365–376. [Google Scholar] [CrossRef]
- Greely, R.H. Rotary Drum Solid Waste Air Classifier. U.S. Patent 5,022,982, 6 November 1991. [Google Scholar]
- Al-Salem, S.M.; Lettieri, P.; Baeyens, J. Recycling and recovery routes of plastic solid waste(PSW): A review. Waste Manag. 2009, 29, 2625–2643. [Google Scholar]
- Karlsson, S. Recycled polyolefins material properties and mean for quality determination. Adv. Polym. Sci. 2004, 169, 201–230. [Google Scholar] [CrossRef]
- Stückrad, B.; Löhr, K. Method for sorting plastics from a particle mixture composed of different plastics. U.S. Patent 5,566,832, 22 October 1996. [Google Scholar]
- Jody, B.J.; Pomykala, J.A., Jr.; Spangenberger, J.S. Recycling-End-of-Life-Vehicles of the Future Energy Systems Division; ANL/ES-C0201801. Argonne National Laboratory: Illinois, IL, 1 December 2009. [Google Scholar]
- Jody, B.J.; Bayram, A.; Karveles, D.; Pomykala, J.A., Jr.; Daniels, E.J. Method for the separation of high impact polystyrene (HIPS) and acrylonitrile butadiene styrene (ABS) plastics. U.S. Patent 5,653,867, 5 August 1996. [Google Scholar]
- American Chemistry Council. Plastic Packaging Resins. Available online: http://plastics.americanchemistry.com/Plastic-Resin-Codes-PDF (accessed on 7 April 2012).
- Börjeson, L.; Löfvenius, G.; Hjelt, M.; Johansson, S.; Marklund, S. Characterization of automotive shredder residues from two shredding facilities with different refining processes in Sweden. Waste Manag. Res. 2000, 18, 358–366. [Google Scholar]
- USA Strategies Inc. Understanding the current trends in plastics recycling. 2007. Available online: http://www.foodandbeveragepackaging.com/FDP/Home/Files/PDF/PlasticRecyclingTrends.pdf (accessed on 7 April 2012).
- Tall, S. Recycling of mixed plastic waste—Is separation worthwhile? PhD Thesis, Department of Polymer Technology, Royal Institute of Technology, Stockholm, Sweden, 2000. [Google Scholar]
- Scheirs, J. Polymer Recycling: Science, Technology and Applications; Wiley: Hoboken, NY, USA, 1998. [Google Scholar]
- Graham, J.; Hendra, P.J.; Mucci, P. Rapid identification of plastic components recovered from scrap automobiles. Plast. Rubber Compos. Process. Appl. 1995, 24, 55–67. [Google Scholar]
- Kenny, G.; Al-Ali, A.; Morgan, D. Method and apparatus for classifying and separation of plastic containers. European Patent 554,850, 7 June 1994. [Google Scholar]
- Beck, M.H.; Rolend, G.F.; Nichols, R.C.; Muszynski, J. Electrostatic separation of plastic materials. U.S. Patent 5,118 407, 2 June 1992. [Google Scholar]
- Stencel, J.M.; Schaefer, J.I.; Ban, H.; Neathery, J.K.; Li, T. Triboelectric separator with mixing chamber and pre-separator. U.S. Patent 5,944,875, 31 August 1999. [Google Scholar]
- Osing, D. Treatment of waste material. U.S. Patent 7,445,591, 4 November 2008. [Google Scholar]
- Geisler, I.; Knauer, H.-J.; Stahl, I. Electrostatic separator for classifying triboelectrically charged substance mixtures. U.S. Patent 6,011,229, 4 January 2000. [Google Scholar]
- Inoue, T.; Daiku, H.; Arai, H.; Machata, H.; Kato, T. Plastic sorter. U.S. Patent 6,720,514, 13 April 2004. [Google Scholar]
- Mankosa, M.J.; Luttrell, G.H. Plastic material having enhanced magnetic susceptibility, method of making and method of separating. U.S. Patent 6,920,982, 26 July 2005. [Google Scholar]
- Allen, L.E., III; Rilse, B.L. Mediating electrostatic separation. U.S. Patent 7,063,213, 20 June 2006. [Google Scholar]
- Dascalescu, L.; Fati, O.; Bilici, M.; Rahou, F.; Dragan, C.; Samuila, A.; Iuga, A. Factors that influence the efficiency of a fluidized-bed-type tribo-electrostatic separator for mixed granular plastics. In 13th International Conference on Electrostatics, Bangor, UK, 10–14 April 2011.
- Miloudi, M.; Medles, K.; Tilmatine, A.; Brahami, M.; Dascalescu, L. 2011 optimisation of belt-type electrostatic separation of granular plastic mixtures tribocharged in a propeller-type device. In 13th International Conference on Electrostatics, Bangor, UK, 10–14 April 2011.
- Tilmatine, A.; Medles, K.; Bendimerad, S.-E.; Boukholda, F.; Dascalescu, L. Electrostatic separators of particles: Application to plastic/metal, metal/metal, plastic/plastic mixtures. Waste Manag. 2009, 29, 228–232. [Google Scholar]
- Ciacci, L.; Morselli, L.; Passarini, F.; Santini, A.; Vassura, I. A comparism among different automotive shredder residue treatment processes. Int. J. Life Cycle Ass. 2010, 15, 896–906. [Google Scholar]
- Xiao, C.; Allen, L.; Biddle, M. Electrostatic separation and recovery of mixed plastics. In Proceedings of the Annual Recycling Conference, Detroit, MI, USA, 9–11 November 1999; Society of Plastics Engineers.
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Lee, J.J.S.; Mo, J.P.T.; Wu, D.Y. Polymer Recovery from Auto Shredder Residue by Projectile Separation Method. Sustainability 2012, 4, 643-655. https://doi.org/10.3390/su4040643
Lee JJS, Mo JPT, Wu DY. Polymer Recovery from Auto Shredder Residue by Projectile Separation Method. Sustainability. 2012; 4(4):643-655. https://doi.org/10.3390/su4040643
Chicago/Turabian StyleLee, Jason J. S., John P. T. Mo, and Dong Yang Wu. 2012. "Polymer Recovery from Auto Shredder Residue by Projectile Separation Method" Sustainability 4, no. 4: 643-655. https://doi.org/10.3390/su4040643
APA StyleLee, J. J. S., Mo, J. P. T., & Wu, D. Y. (2012). Polymer Recovery from Auto Shredder Residue by Projectile Separation Method. Sustainability, 4(4), 643-655. https://doi.org/10.3390/su4040643





