Depth Profiling (ICP-MS) Study of Toxic Metal Buildup in Concrete Matrices: Potential Environmental Impact
Abstract
1. Introduction
| Element | Symbol | Approximate concentration (ppm) in sea water | Average concentration (ppm) in cement |
|---|---|---|---|
| Arsenic | As | 1.8 × 10−3 | 9 |
| Cadmium | Cd | 1.2 × 10−4 | 1 |
| Lead | Pb | 3.6 × 10−5 | 40 |
| Mercury | Hg | 2.0 × 10−6 | Not available |
2. Results and Discussion
2.1. Trace Toxic Elemental Buildup in Concrete Matrices
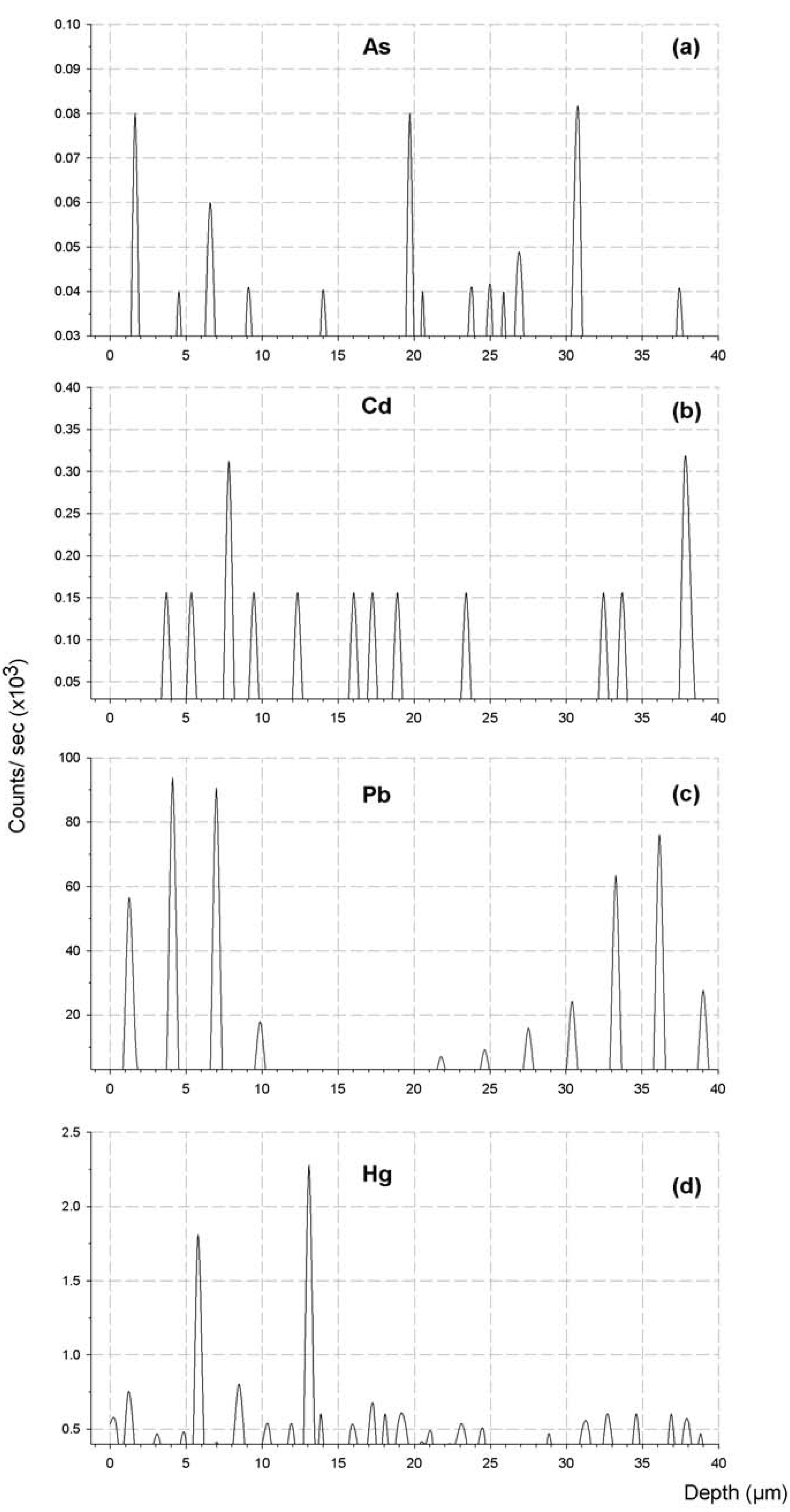
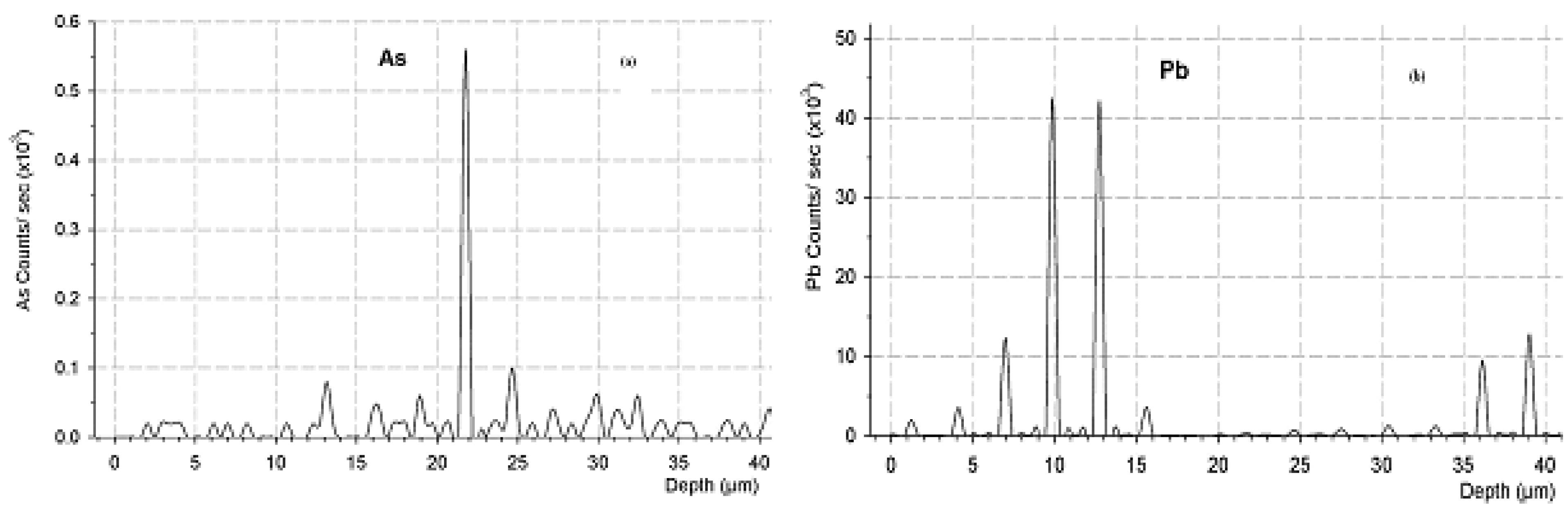
2.2. Potential Environmental Impact
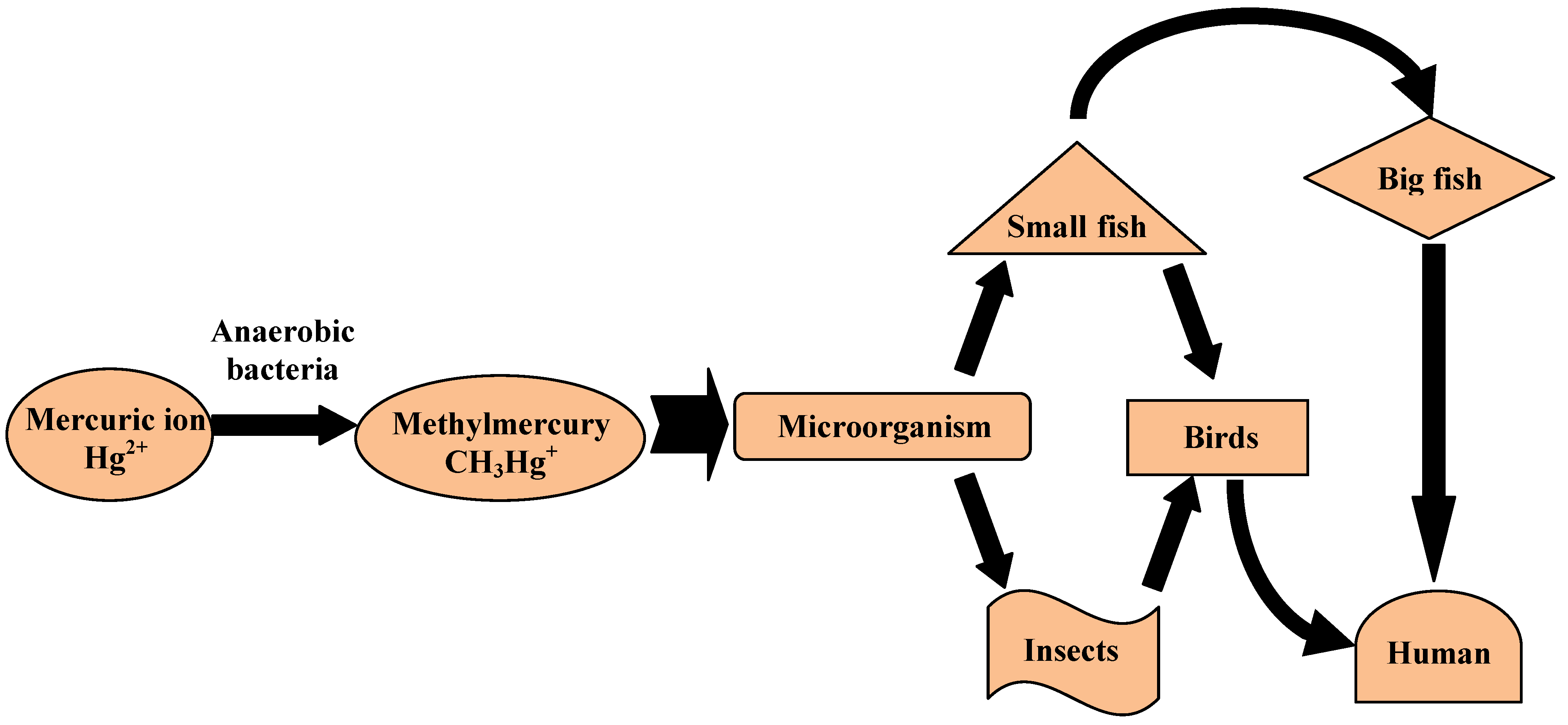
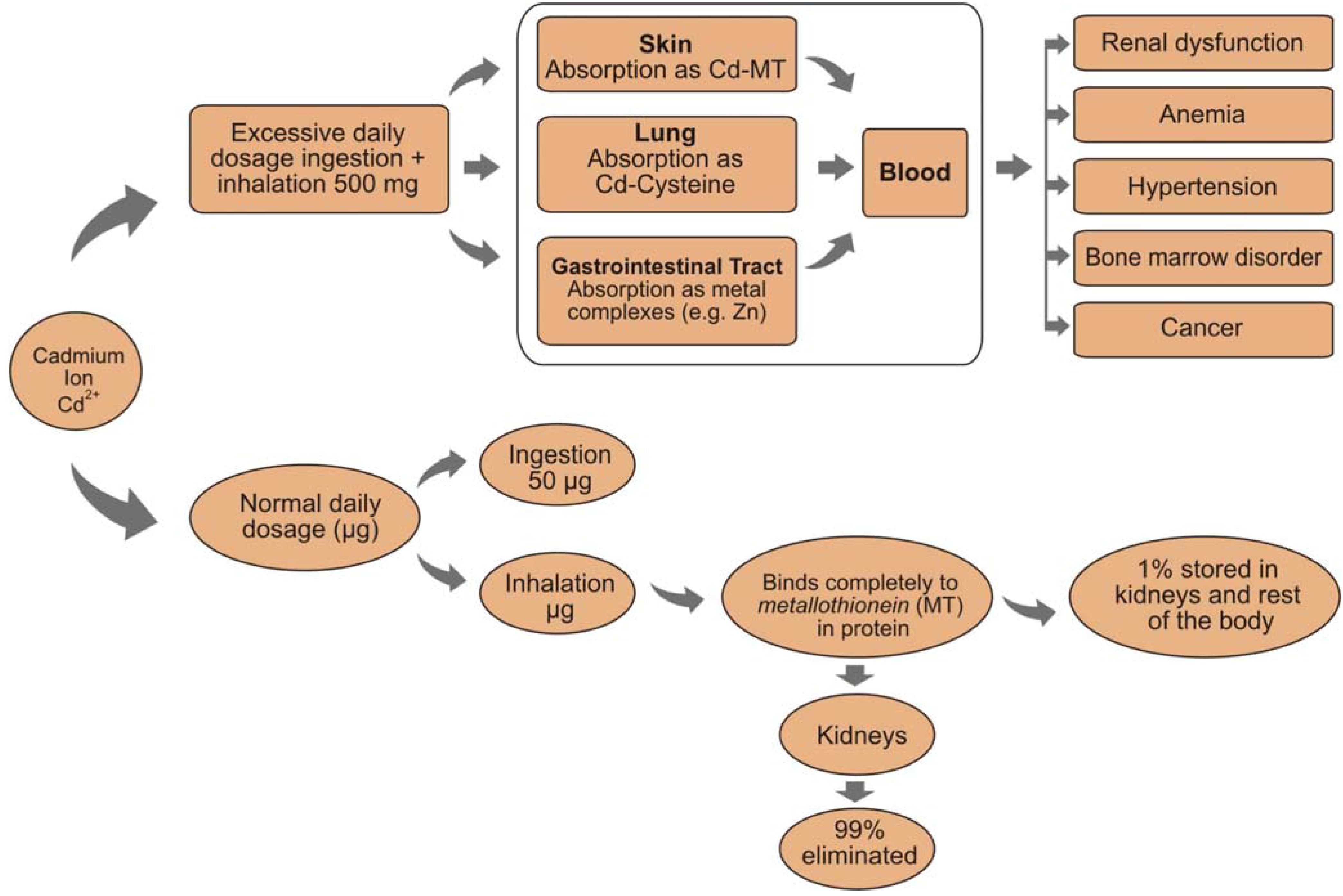
2.3. Detection of Beryllium and Osmium in Concrete
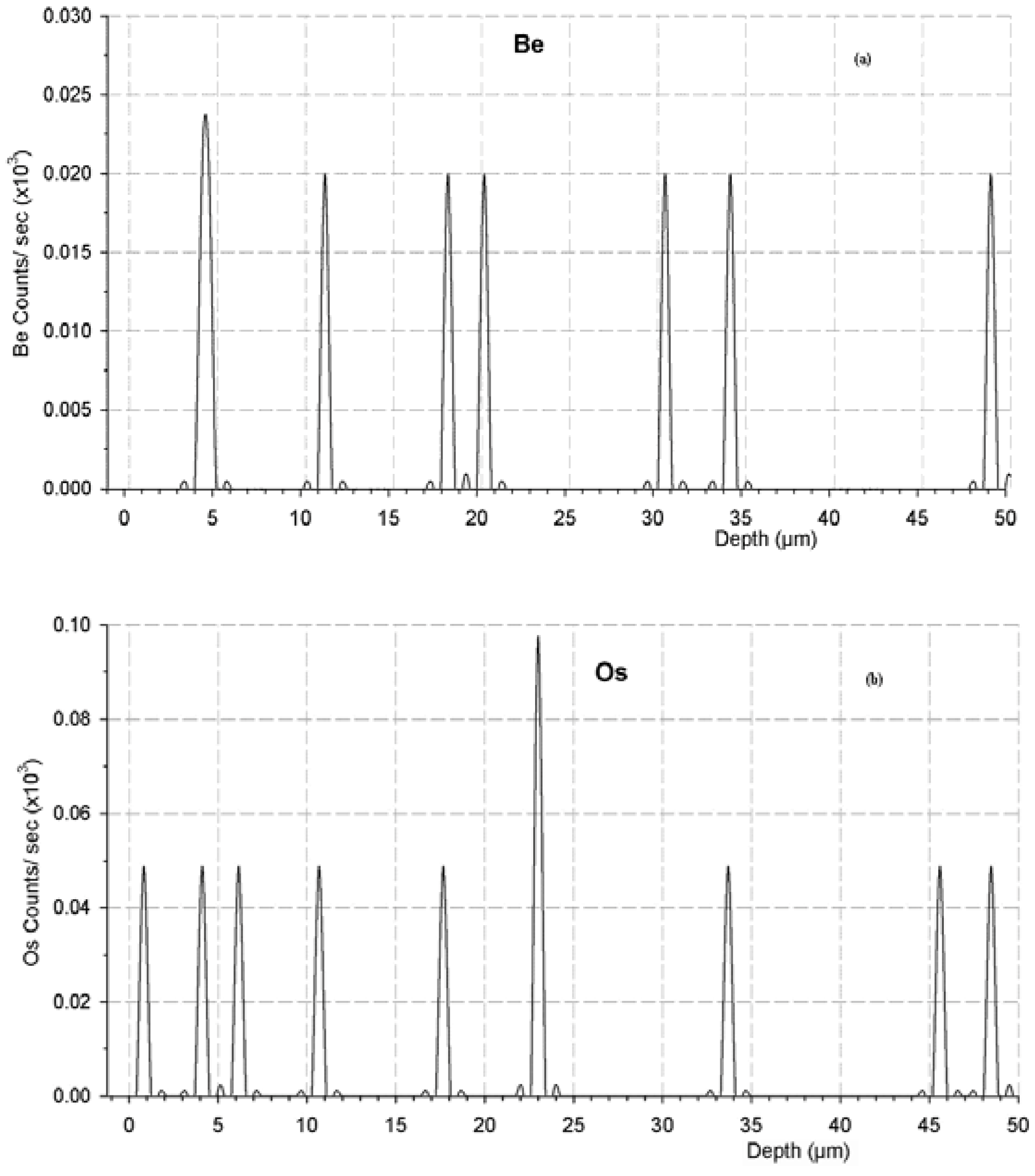
2.4. Suggested Remedial Measures
3. Materials and Methods
3.1.Sample Preparation/Sample Handling

3.2. Instrumentation/Spectral Characteristics
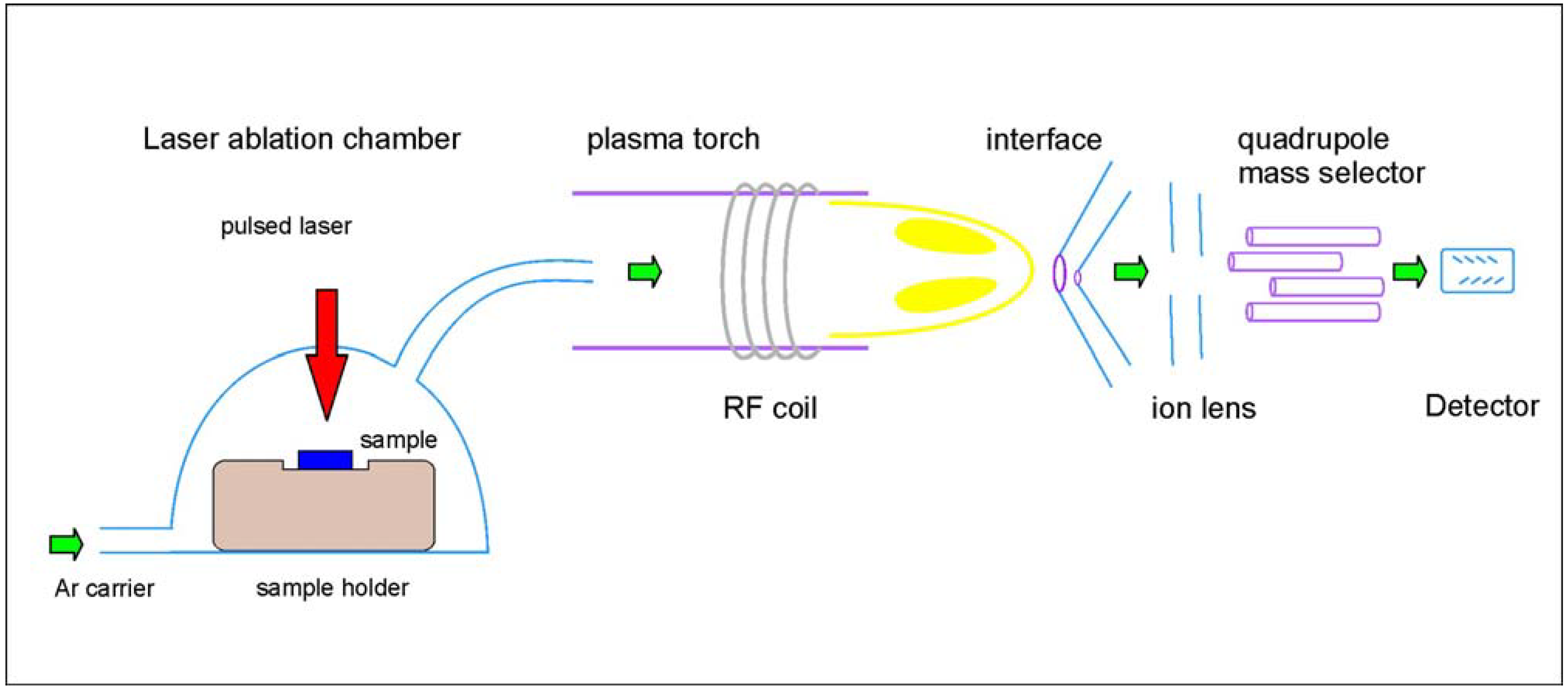
3.3. Validation of the Technique
4. Conclusions
Acknowledgements
References and Notes
- Spanka, G.; Schneider, M. Release of inorganic and organic substances from cement bound materials. Struct. Concr. 2001, 2, 145–153. [Google Scholar]
- Fairly, H.F. Reef Alchemy: What is Seawater? Available online: http://reefkeeping.com/issues/2005-11/rhf/index.php#4 (accessed on 13 October 2010).
- Achternboch, M.; Brätigam, K.R.; Gleis, M.; Hartlieb, N.; Kupsch, C.; Richers, U.; Stemmermann, P. Heavy Metals in Cement and Concrete Resulting from the Co-incineration of Wastes in Cement Kilns with Regards to the Legitimacy of Waste Utilization; Forschungszentrum Karlsruhe: Karlsruhe, Germany, 2003; p. 23. [Google Scholar]
- Robinson, J.G. The limits to caring: Sustainable living and the loss to biodiversity. Conserv. Biol. 1993, 7, 20–28. [Google Scholar] [CrossRef]
- Binqiu, Z.; Wong, C.S.; Johnson, K.W. Distribution of dissolved and particulate trace metals in anoxic seawater. Chin. J. Oceanol. Limnol. 1992, 10, 75–83. [Google Scholar] [CrossRef]
- Akan, J.C.; Abdulrahman, F.I.; Sodipo, O.A.; Akandu, P.I. Bioaccumulation of some heavy metals of six fresh water fishes caught from Lake Chad in Dohron Buhari, Maiduguri, Borno State, Nigeria. J. Appl. Sci. Environ. Sanit. Sby. 2009, 4, 103–114. [Google Scholar]
- Čelechovská, O.; Malota, L.; Zima, S. Entry of heavy metals into food chains: A 20-year comparison study in northern Moravia. Acta Vet. Brno. 2008, 77, 645–652. [Google Scholar] [CrossRef]
- Donsbach, K.W. Toxic metal elimination by mineral substitution. Dyn. Chiropractic 1990, 8, 26. [Google Scholar]
- Clarkson, T.W. Mercury: An element of mystery. N. Engl. J. Med. 1990, 323, 1137–1139. [Google Scholar] [CrossRef] [PubMed]
- Kumar, A. Chemical Toxicity Environmental Chemistry, 3rd ed.; Wiley Eastern: New Delhi, India, 1994; pp. 75–88. [Google Scholar]
- Mercury in the Environment; US Department of the Interior: Washington, DC, USA, 2000.
- Diner, B.M.; Brenner, B.E. Toxicity, Mercury; e-medecine: Omaha, NE, USA, 2009; Available online: http://emedicine.medscape.com/article/819872-overview (accessed on 13 October 2010).
- Hansen, J.C.; Gilman, A.P. Exposure of arctic populations to methylmercury from consumption of marine food: An update risk-benefit assessment. Int. J. Circumpolar Health 2005, 64, 121–136. [Google Scholar] [CrossRef] [PubMed]
- Wilson, L. Toxic Metals and Human Health; The Center for Development: Prescott, AZ, USA, 2010. [Google Scholar]
- Hu, H.; McCally, M. Human healthand heavy metals exposure. In Life Support: The Environment and Human Health; MIT: Cambridge, MA, USA, 2002; pp. 65–82. [Google Scholar]
- Suwazono, Y.; Kobayashi, E.; Okubo, Y.; Nogawa, K.; Kido, T.; Nakagawa, H. Renal effects of cadmium exposure in cadmium nonpolluted areas in Japan. Environ. Res. 2000, 84, 44–55. [Google Scholar] [CrossRef]
- Jarup, L.; Hellstrom, L.; Alfven, T.; Carlsson, M.D.; Grubb, A.; Persson, B. Low level exposure to cadmium and early kidney damage: The OSCAR study. Occup. Environ. Med. 2000, 57, 668–672. [Google Scholar] [CrossRef]
- Parsons, T.; Brigham, K. Scientists Discover How Lead Changes the Brain to Impair Learning and Memory; Public Health News Center in John Hopkins University: Washington, DC, USA, 2000; Available online: http://www.jhsph.edu/publichealthnews/press_releases/PR_2000/lead_change.html (accessed on 13 October 2010).
- Cooper, R.G.; Harrison, A.P. The uses and adverse effects of beryllium on health. Indian J. Occup. Environ. Med. 2009, 13, 65–76. [Google Scholar] [CrossRef]
- Skjåk, B.G.; Malnes, D.; Jensen, A. Heavy metal tolerance of marine phytoplankton. J. Exp. Mar. Biol. Ecol. 1980, 42, 39–54. [Google Scholar] [CrossRef]
- Pillay, A.E.; Elkadi, M.; Feghali, F.; Fok, S.; Bassioni, G.; Stephen, S. Tracking chloride and metal diffusion in proofed and unproofed concrete matrices using ablative laser technology (ICP-MS). Nat. Sci. 2010, 2, 809–816. [Google Scholar]
© 2010 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Elkadi, M.; Pillay, A.; Fok, S.C.; Feghali, F.; Bassioni, G.; Stephen, S. Depth Profiling (ICP-MS) Study of Toxic Metal Buildup in Concrete Matrices: Potential Environmental Impact. Sustainability 2010, 2, 3258-3269. https://doi.org/10.3390/su2103258
Elkadi M, Pillay A, Fok SC, Feghali F, Bassioni G, Stephen S. Depth Profiling (ICP-MS) Study of Toxic Metal Buildup in Concrete Matrices: Potential Environmental Impact. Sustainability. 2010; 2(10):3258-3269. https://doi.org/10.3390/su2103258
Chicago/Turabian StyleElkadi, Mirella, Avin Pillay, Sai Cheong Fok, Fadi Feghali, Ghada Bassioni, and Sasi Stephen. 2010. "Depth Profiling (ICP-MS) Study of Toxic Metal Buildup in Concrete Matrices: Potential Environmental Impact" Sustainability 2, no. 10: 3258-3269. https://doi.org/10.3390/su2103258
APA StyleElkadi, M., Pillay, A., Fok, S. C., Feghali, F., Bassioni, G., & Stephen, S. (2010). Depth Profiling (ICP-MS) Study of Toxic Metal Buildup in Concrete Matrices: Potential Environmental Impact. Sustainability, 2(10), 3258-3269. https://doi.org/10.3390/su2103258




