Degradation Potential of Metakaolin-Based Geopolymer Composites Immersed in Real and Simulated Acidic Environments
Abstract
1. Introduction
2. Experimental Methodology
2.1. Sample Preparation
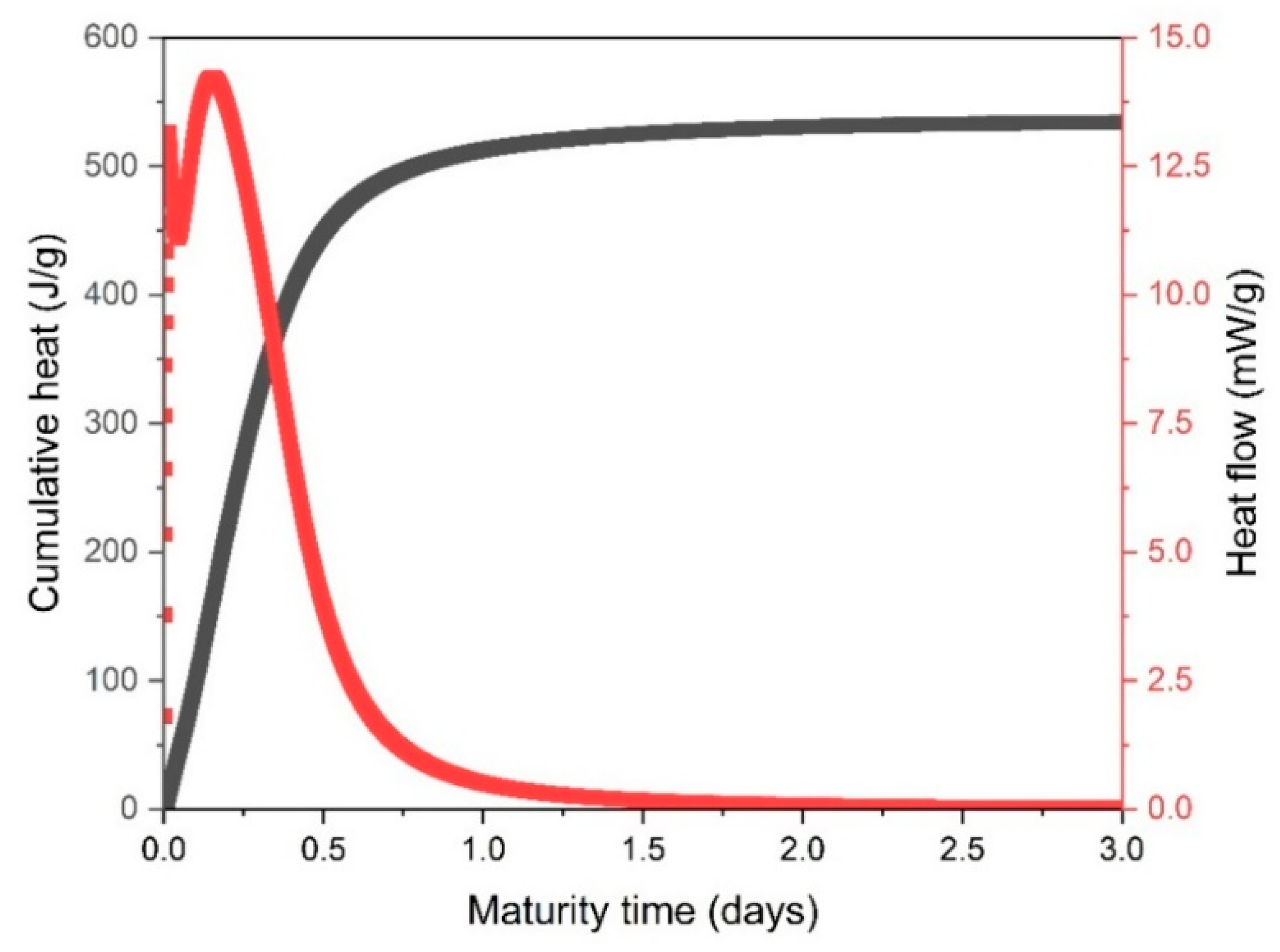
2.2. Isothermal Calorimetry and Compressive Strength Test
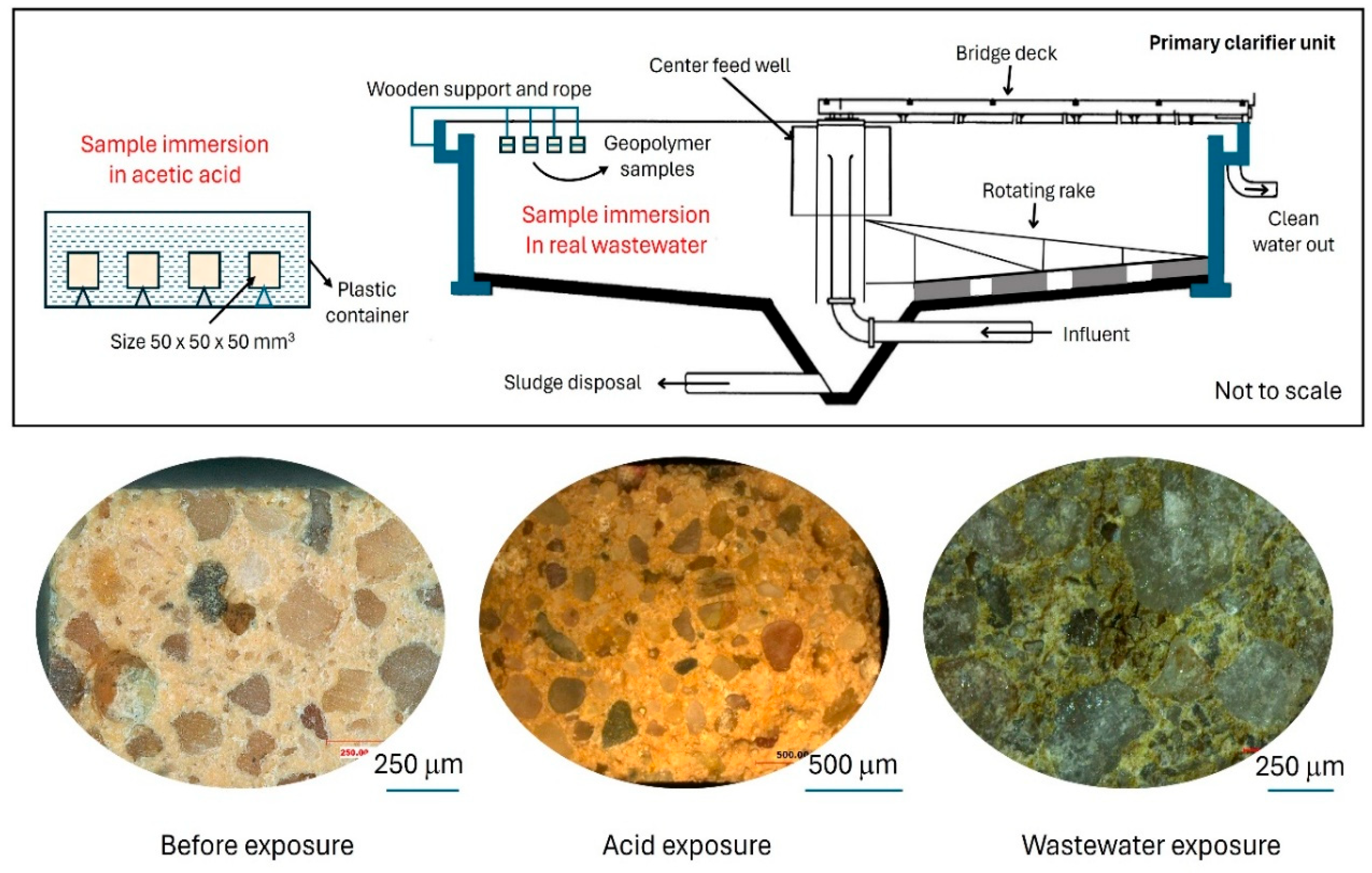
2.3. Simulated Acid Attack Test
2.4. Wastewater Attack (Actual)
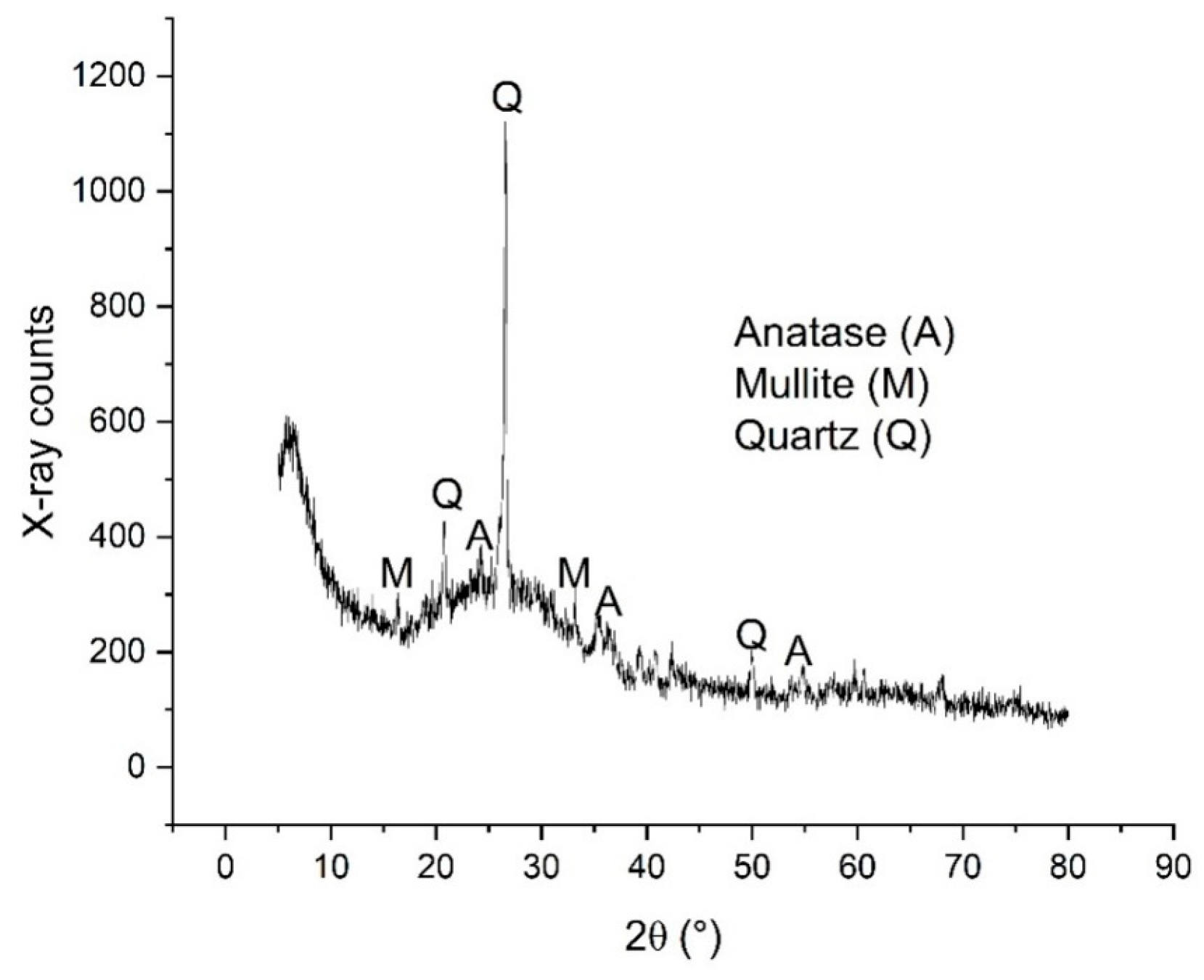
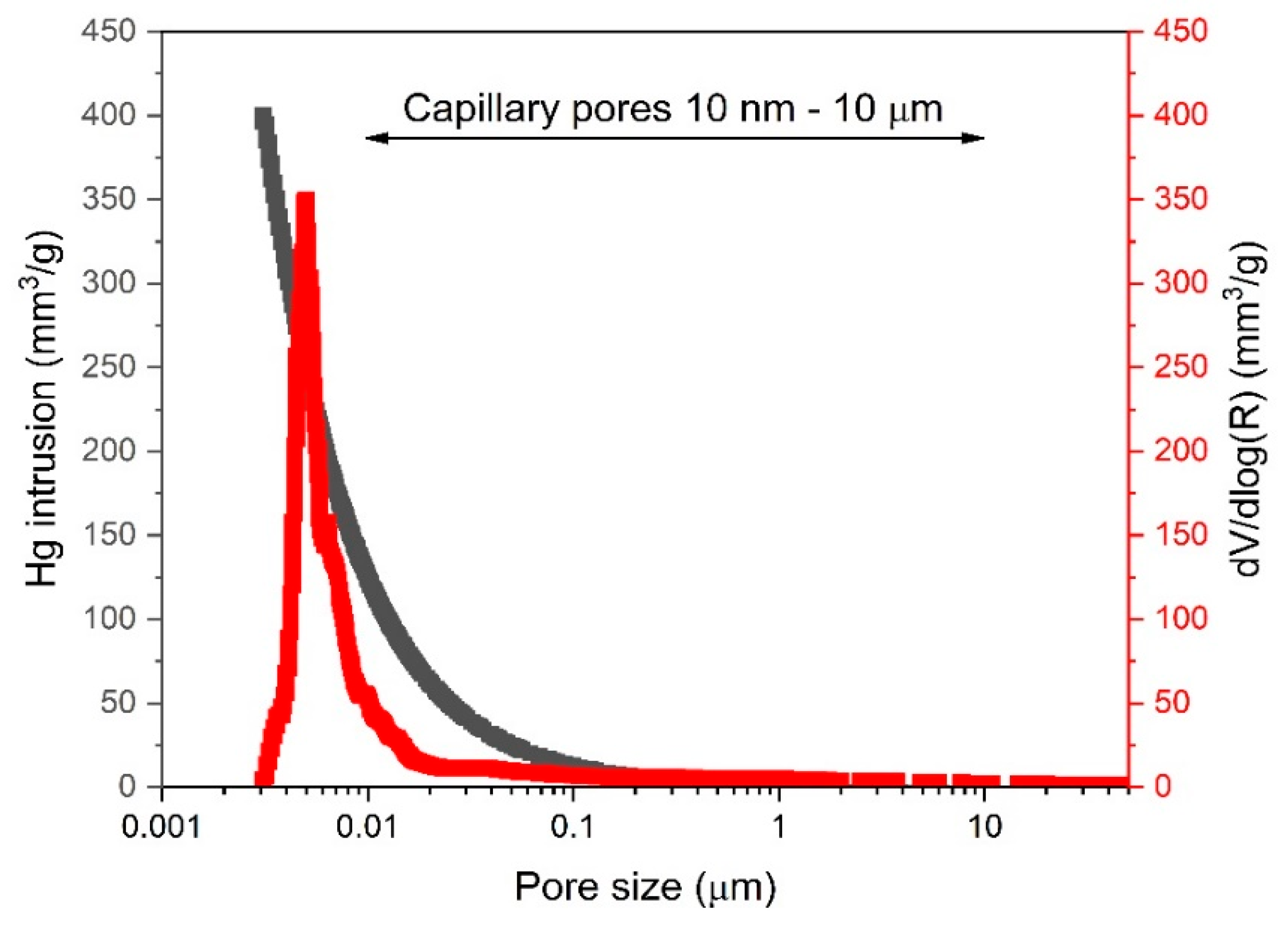
2.5. Characterization Studies
3. Results and Discussion
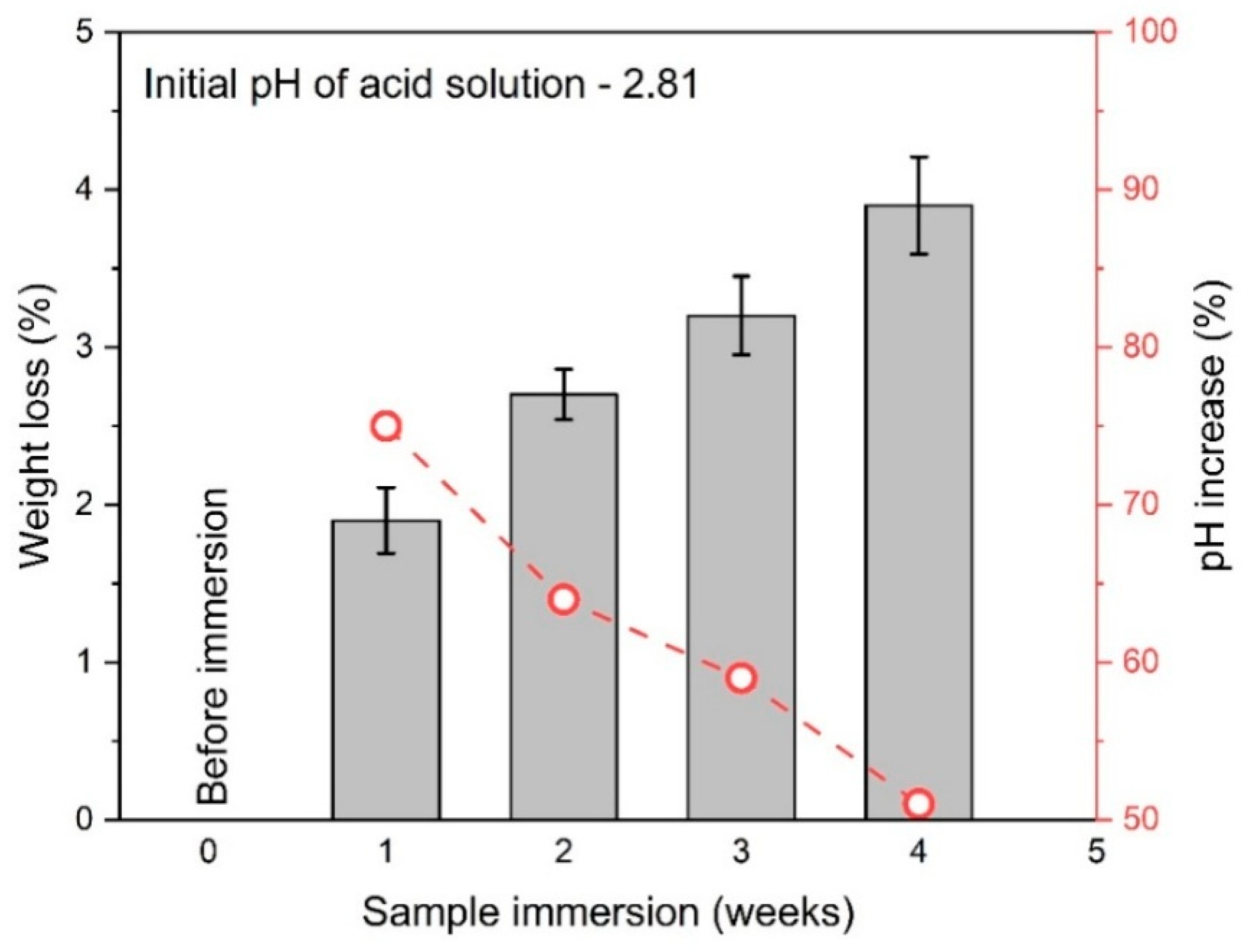
3.1. Sample Degradation
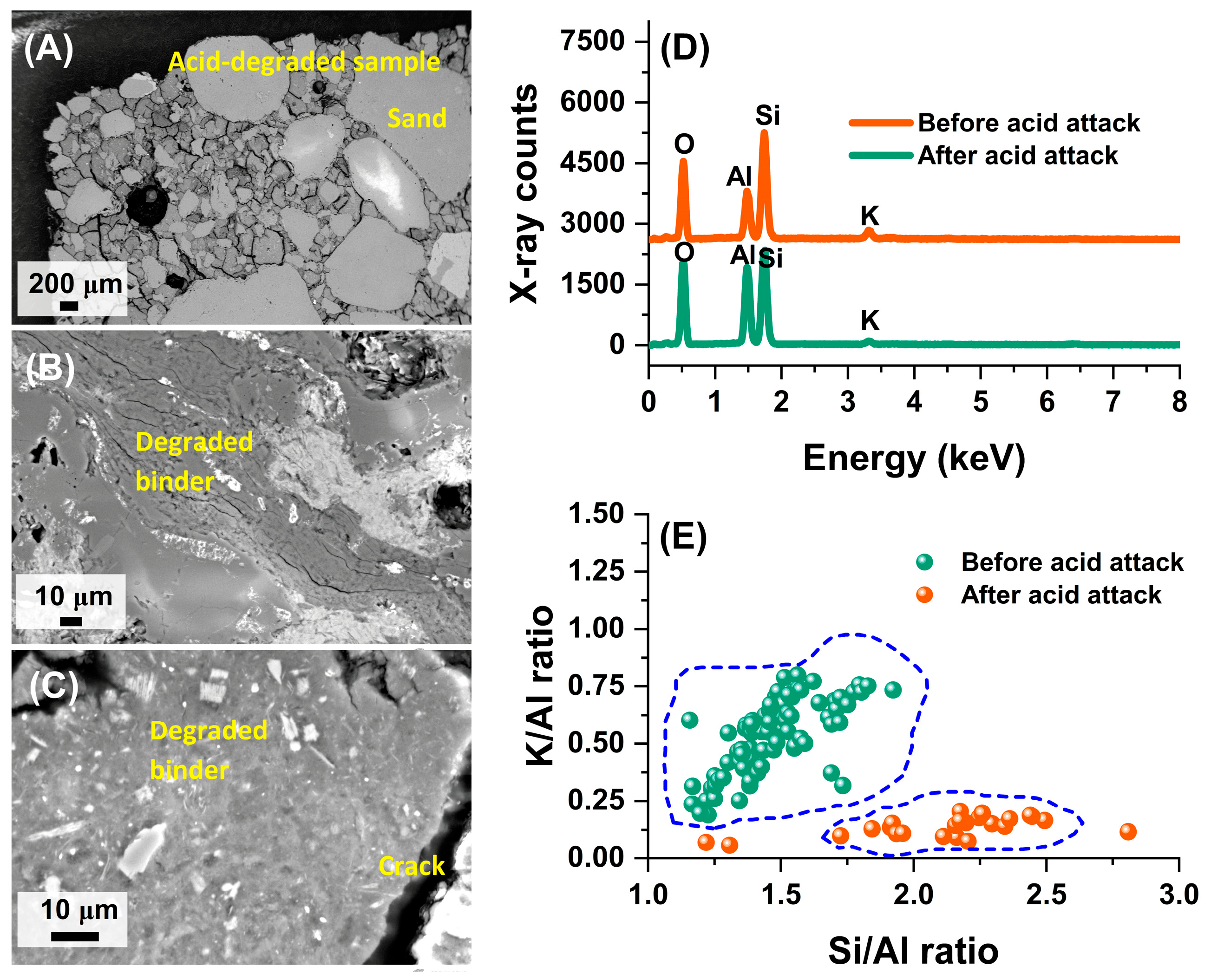
3.2. Microstructural Assessment
4. Conclusions
- The metakaolin GP samples exhibited substantial initial compressive strength, averaging 79.4 MPa after 28 days of curing, indicating their potential as a robust construction material. Conversely, the isothermal calorimetry results indicated a cumulative heat generation of 534 J/g, demonstrating the rapid setting time and energy efficiency of the GP binder compared to its traditional OPC counterparts.
- The GP samples immersed in a 0.50 molar acetic acid solution demonstrated a strength reduction of 13% and a weight loss of 3.90% after four weeks, highlighting the impact of acidic environments on material integrity. In contrast, the GP samples exposed to actual wastewater conditions experienced an 18% reduction in strength and a weight loss of 5.60%. This suggests that real-world conditions may lead to more severe degradation due to factors such as microbial activity and fluid movement.
- The SEM results revealed significant microstructural changes in the geopolymer samples after exposure, including the formation of biofilms and air voids, indicating interactions between the GP matrix and its environment that could affect long-term structural resilience.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Alexander, M.; Bertron, A.; De Belie, N. Performance of Cement-Based Materials in Aggressive Aqueous Environments; Springer: Berlin/Heidelberg, Germany, 2013; Volume 10, ISBN 9400754124. [Google Scholar]
- Von Sperling, M. Wastewater Characteristics, Treatment and Disposal; IWA Publishing: London, UK, 2007. [Google Scholar]
- Woyciechowski, P.; Łukowski, P.; Szmigiera, E.; Adamczewski, G.; Chilmon, K.; Spodzieja, S. Concrete Corrosion in a Wastewater Treatment Plant—A Comprehensive Case Study. Constr. Build. Mater. 2021, 303, 124388. [Google Scholar] [CrossRef]
- Kiliswa, M.W.; Scrivener, K.L.; Alexander, M.G. The Corrosion Rate and Microstructure of Portland Cement and Calcium Aluminate Cement-Based Concrete Mixtures in Outfall Sewers: A Comparative Study. Cem. Concr. Res. 2019, 124, 105818. [Google Scholar] [CrossRef]
- Pramanik, S.K.; Bhuiyan, M.; Robert, D.; Roychand, R.; Gao, L.; Cole, I.; Pramanik, B.K. Bio-Corrosion in Concrete Sewer Systems: Mechanisms and Mitigation Strategies. Sci. Total Environ. 2024, 921, 171231. [Google Scholar] [CrossRef] [PubMed]
- Sheshadri, A.; Marathe, S.; Sadowski, Ł. Development of Sustainable, High Strength Slag Based Alkali Activated Pavement Quality Concrete Using Agro-Industrial Wastes: Properties and Life Cycle Analysis. Int. J. Pavement Eng. 2024, 25, 2401953. [Google Scholar] [CrossRef]
- Marathe, S.; Martyna, N.; Gronostajska, B. Alkali-Activated Permeable Concretes with Agro-Industrial Wastes for a Sustainable Built Environment. Materials 2025, 18, 87. [Google Scholar] [CrossRef]
- Khan, H.A.; Castel, A.; Khan, M.S.H.; Mahmood, A.H. Durability of Calcium Aluminate and Sulphate Resistant Portland Cement Based Mortars in Aggressive Sewer Environment and Sulphuric Acid. Cem. Concr. Res. 2019, 124, 105852. [Google Scholar] [CrossRef]
- Davidovits, J. False Values on CO2 Emission for Geopolymer Cement/Concrete Published in Scientific Papers. Tech. Pap. 2015, 24, 1–9. [Google Scholar]
- McLellan, B.C.; Williams, R.P.; Lay, J.; Van Riessen, A.; Corder, G.D. Costs and Carbon Emissions for Geopolymer Pastes in Comparison to Ordinary Portland Cement. J. Clean. Prod. 2011, 19, 1080–1090. [Google Scholar] [CrossRef]
- Sheshadri, A.; Marathe, S.; Bettadapura Manjunath, M.; Jayasimhan, A.; Sadowski, Ł. Effective Utilization of Foundry Waste as Aggregates in Developing Eco-Friendly Alkali-Activated and Conventional Concretes for Sustainable Pavement Infrastructure. Pract. Period. Struct. Des. Constr. 2024, 29, 04024018. [Google Scholar] [CrossRef]
- Weil, M.; Dombrowski, K.; Buchwald, A. 10—Life-Cycle Analysis of Geopolymers. In Woodhead Publishing Series in Civil and Structural Engineering; Provis, J.L., van Deventer, J.S.J.B.T.-G., Eds.; Woodhead Publishing: Sawston, UK, 2009; pp. 194–210. ISBN 978-1-84569-449-4. [Google Scholar]
- Zhang, M.; Zhao, N.; Wang, S.; Quan, X.; Liu, K.; Xu, J.; Wang, Z.; Ying, H.; Liu, B. Mechanical and Durability Performance of Polyvinyl Alcohol Fiber Hybrid Geopolymer-Portland Cement Concrete under Freeze–Thaw Cycles. Boletín Soc. Española Cerámica Vidr. 2024, 63, 222–235. [Google Scholar] [CrossRef]
- Gao, X.X.; Michaud, P.; Joussein, E.; Rossignol, S. Behavior of Metakaolin-Based Potassium Geopolymers in Acidic Solutions. J. Non. Cryst. Solids 2013, 380, 95–102. [Google Scholar] [CrossRef]
- Khaled, Z.; Mohsen, A.; Soltan, A.; Kohail, M. Optimization of Kaolin into Metakaolin: Calcination Conditions, Mix Design and Curing Temperature to Develop Alkali Activated Binder. Ain Shams Eng. J. 2023, 14, 102142. [Google Scholar] [CrossRef]
- Pelisser, F.; Guerrino, E.L.; Menger, M.; Michel, M.D.; Labrincha, J.A. Micromechanical Characterization of Metakaolin-Based Geopolymers. Constr. Build. Mater. 2013, 49, 547–553. [Google Scholar] [CrossRef]
- Wang, T.; Wu, K.; Kan, L.; Wu, M. Current Understanding on Microbiologically Induced Corrosion of Concrete in Sewer Structures: A Review of the Evaluation Methods and Mitigation Measures. Constr. Build. Mater. 2020, 247, 118539. [Google Scholar] [CrossRef]
- Li, X.; O’Moore, L.; Song, Y.; Bond, P.L.; Yuan, Z.; Wilkie, S.; Hanzic, L.; Jiang, G. The Rapid Chemically Induced Corrosion of Concrete Sewers at High H2S Concentration. Water Res. 2019, 162, 95–104. [Google Scholar] [CrossRef] [PubMed]
- Gutiérrez-Padilla, M.G.D.; Bielefeldt, A.; Ovtchinnikov, S.; Hernandez, M.; Silverstein, J. Biogenic Sulfuric Acid Attack on Different Types of Commercially Produced Concrete Sewer Pipes. Cem. Concr. Res. 2010, 40, 293–301. [Google Scholar] [CrossRef]
- Grengg, C.; Mittermayr, F.; Baldermann, A.; Böttcher, M.E.; Leis, A.; Koraimann, G.; Grunert, P.; Dietzel, M. Microbiologically Induced Concrete Corrosion: A Case Study from a Combined Sewer Network. Cem. Concr. Res. 2015, 77, 16–25. [Google Scholar] [CrossRef]
- ASTM C1702-23; Standard Test Method for Measurement of Heat of Hydration of Hydraulic Cementitious Materials Using Isothermal Conduction Calorimetry. ASTM International: West Conshohocken, PA, USA, 2023; Volume i, pp. 1–8.
- UNI EN ISO 12390-3; Testing Hardened Concrete-Part 3: Compressive Strength of Test Specimens. iTeh, Inc.: Newark, DE, USA, 2019; Volume 22.
- Aligizaki, K.K. Pore Structure of Cement-Based Materials: Testing, Interpretation and Requirements, 1st ed.; CRC Press: Boca Raton, FL, USA, 2005; ISBN 0429079311. [Google Scholar]
- Rashad, A.M.; Zeedan, S.R. The Effect of Activator Concentration on the Residual Strength of Alkali-Activated Fly Ash Pastes Subjected to Thermal Load. Constr. Build. Mater. 2011, 25, 3098–3107. [Google Scholar] [CrossRef]
- Van Deventer, J.S.J.; Provis, J.L.; Duxson, P.; Lukey, G.C. Reaction Mechanisms in the Geopolymeric Conversion of Inorganic Waste to Useful Products. J. Hazard. Mater. 2007, 139, 506–513. [Google Scholar] [CrossRef] [PubMed]
- Provis, J.L.; Rees, C.A. 7—Geopolymer Synthesis Kinetics. In Woodhead Publishing Series in Civil and Structural Engineering; Provis, J.L., van Deventer, J.S.J.B.T.-G., Eds.; Woodhead Publishing: Sawston, UK, 2009; pp. 118–136. ISBN 978-1-84569-449-4. [Google Scholar]


| Oxides | CaO | Al2O3 | SiO2 | MgO | Fe2O3 | Na2O | SO3 | TiO2 | K2O | Ignition Loss |
|---|---|---|---|---|---|---|---|---|---|---|
| Wt% | 0.02 | 43.87 | 53.11 | 0.03 | 0.43 | 0.23 | 0.03 | 1.71 | 0.19 | 0.38 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Marathe, S.; Szemiot-Jankowska, N.; Kumar, S.; Muthu, M. Degradation Potential of Metakaolin-Based Geopolymer Composites Immersed in Real and Simulated Acidic Environments. Sustainability 2025, 17, 468. https://doi.org/10.3390/su17020468
Marathe S, Szemiot-Jankowska N, Kumar S, Muthu M. Degradation Potential of Metakaolin-Based Geopolymer Composites Immersed in Real and Simulated Acidic Environments. Sustainability. 2025; 17(2):468. https://doi.org/10.3390/su17020468
Chicago/Turabian StyleMarathe, Shriram, Natalia Szemiot-Jankowska, Sanjeev Kumar, and Murugan Muthu. 2025. "Degradation Potential of Metakaolin-Based Geopolymer Composites Immersed in Real and Simulated Acidic Environments" Sustainability 17, no. 2: 468. https://doi.org/10.3390/su17020468
APA StyleMarathe, S., Szemiot-Jankowska, N., Kumar, S., & Muthu, M. (2025). Degradation Potential of Metakaolin-Based Geopolymer Composites Immersed in Real and Simulated Acidic Environments. Sustainability, 17(2), 468. https://doi.org/10.3390/su17020468







