Labile and Stable Carbon Pools in Antarctic Soils of the Arctowski Region, King George Island
Abstract
1. Introduction
2. Materials and Methods
2.1. Research Area and Study Sites
2.2. Soil Analyses
2.3. Statistical Analyses
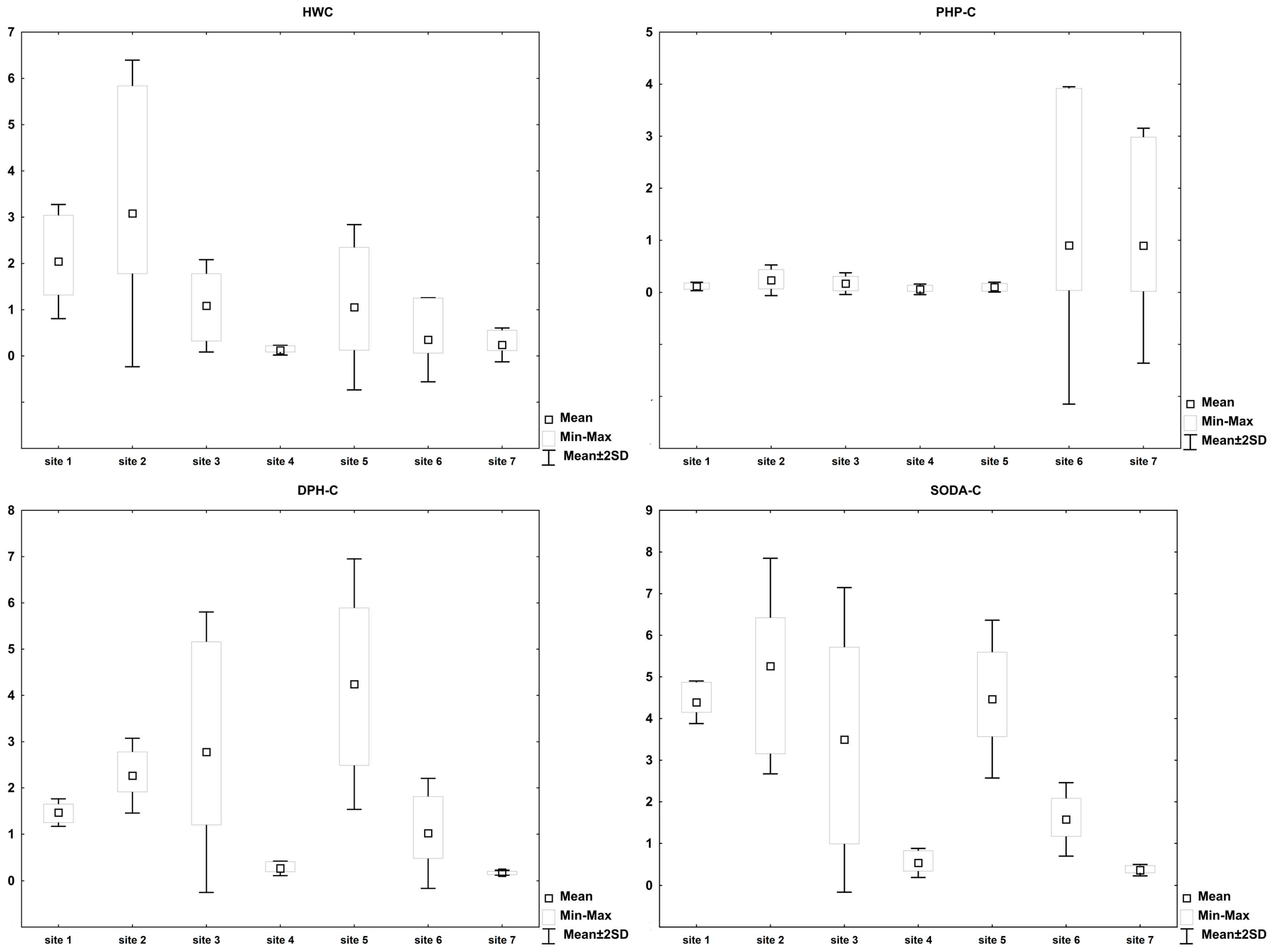
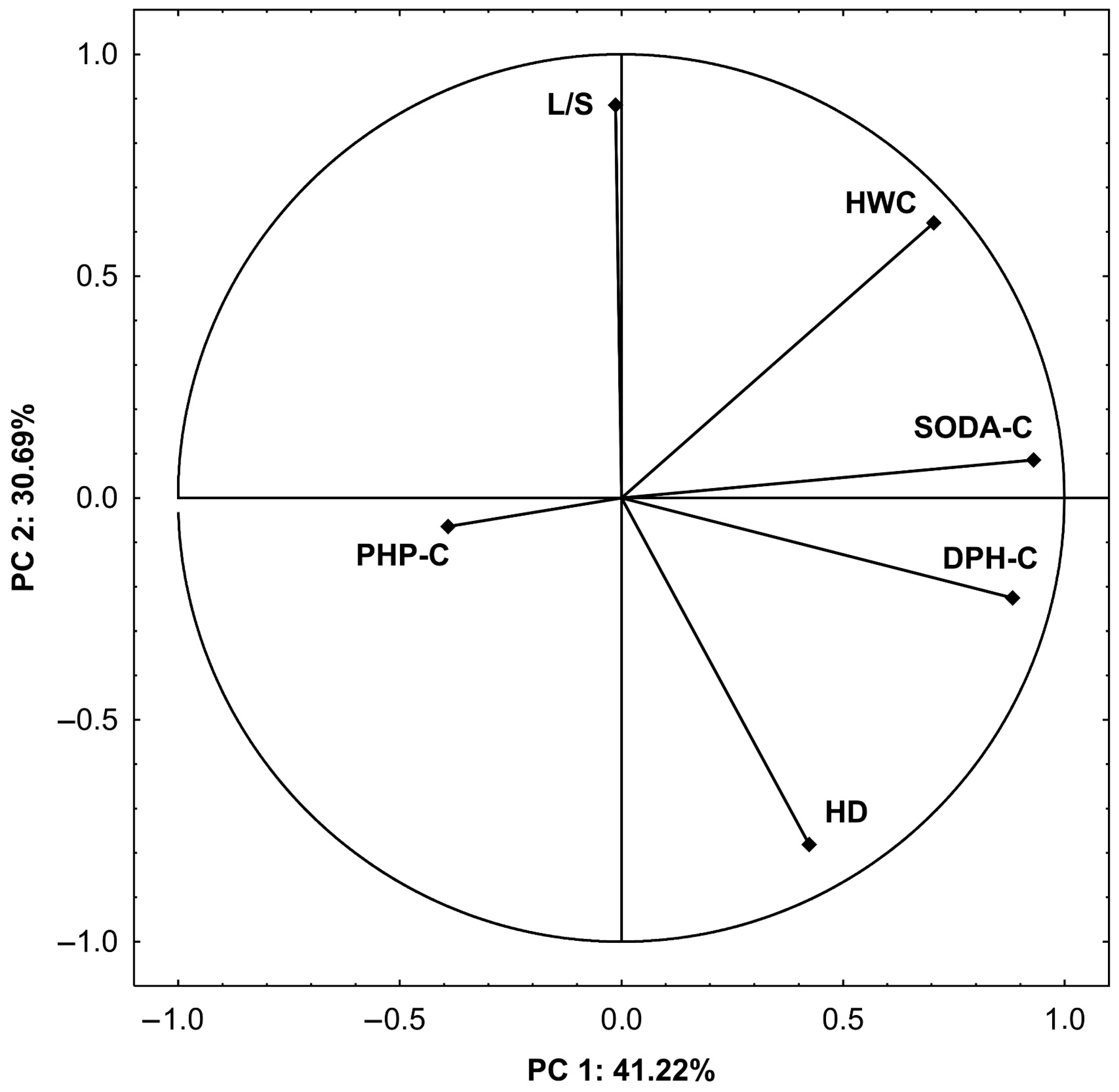
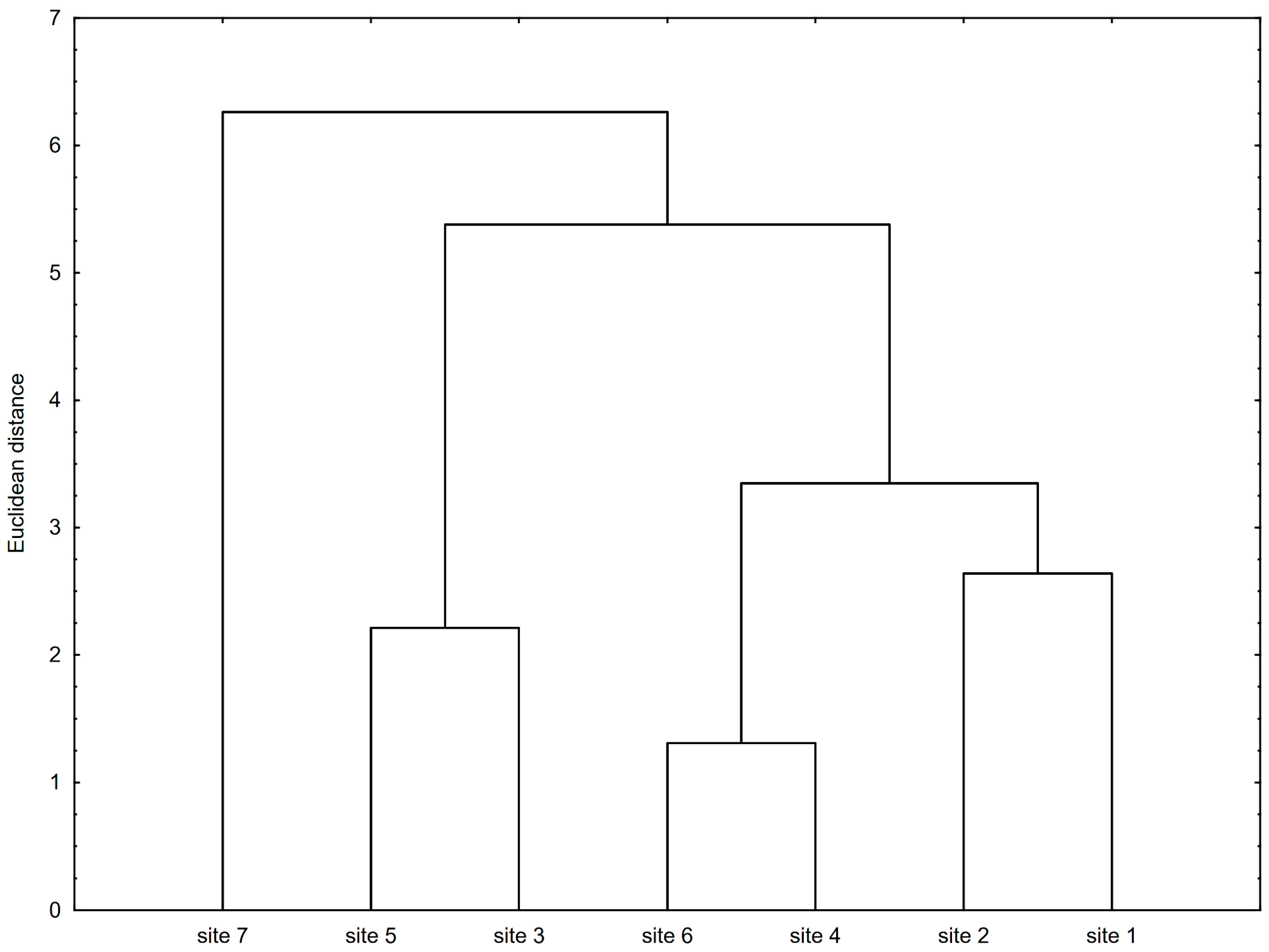
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| DPH-C | humus compounds bound to cations |
| HD | humification degree |
| HWC | hot water-extractable carbon |
| L/S | ratio of labile to stable carbon fraction |
| LOI | loss-on-ignition |
| PHP-C | unbound organic carbon compounds |
| SODA-C | humus compounds that form larger associations or those with higher molecular weight, also associated with cations |
| SOM | soil organic matter |
| TN | total nitrogen |
| TOC | total organic carbon |
| TVC | total vegetation cover |
References
- Bockheim, J.G. Soil-Forming Factors in Antarctica, In The Soils of Antarctica; Bockheim, J.G., Ed.; World Soil Book Series; Springer International Publishing: Cham, Switzerland, 2015. [Google Scholar]
- Glina, B.; Mendyk, L.; Piernik, A.; Nowak, M.; Maier, A.; Inselsbacher, E.; Glatzel, S. Local weather conditions determine DOC production and losses from agricultural fen soils affected by open-pit lignite mining. Catena 2022, 211, 106012. [Google Scholar] [CrossRef]
- Kalisz, B.; Łachacz, A.; Długosz, J.; Żuk-Gołaszewska, K. Effect of TOC and depth on soil chemical properties and labile carbon pool in the topsoil of peatlands. J. Elementol. 2025, 30, 221–240. [Google Scholar] [CrossRef]
- Voltr, V.; Mensik, L.; Hlisnikovsky, L.; Hruska, M.; Pokorny, E.; Pospisilova, L. The soil organic matter in connection with soil properties and soil inputs. Agronomy 2021, 11, 779. [Google Scholar] [CrossRef]
- Lehmann, J.; Kleber, M. The contentious nature of soil organic matter. Nature 2015, 528, 60–68. [Google Scholar] [CrossRef]
- Schmidt, M.W.I.; Torn, M.S.; Abiven, S.; Dittmar, T.; Guggenberger, G.; Janssens, I.A.; Kleber, M.; Kögel-Knabner, I.; Lehmann, J.; Manning, D.A.C.; et al. Persistence of soil organic matter as an ecosystem property. Nature 2011, 478, 49–56. [Google Scholar] [CrossRef]
- Sutton, R.; Sposito, G. Molecular structure in soil humic substances: The new view. Environ. Sci. Technol. 2005, 39, 9009–9015. [Google Scholar] [CrossRef]
- Lal, R. Restoring soil quality to mitigate soil degradation. Sustainability 2015, 7, 5875–5895. [Google Scholar] [CrossRef]
- Kalisz, B.; Łachacz, A. Relations between labile and stable pool of soil organic carbon in peatlands. J. Elementol. 2023, 28, 263–278. [Google Scholar] [CrossRef]
- Lehmann, M.F.; Carstens, D.; Deek, A.; McCarthy, M.; Schubert, C.J.; Zopfi, J. Amino acid and amino sugar compositional changes during In Vitro degradation of algal organic matter indicate rapid bacterial re-synthesis. Geochim. Cosmochim. Acta 2020, 283, 67–84. [Google Scholar] [CrossRef]
- Simas, F.N.B.; Schaefer, C.E.G.R.; Filho, M.R.A.; Francelino, M.R.; Filho, E.I.F.; da Costa, L.M. Genesis, properties and classification of Cryosols from Admiralty Bay, maritime Antarctica. Geoderma 2008, 144, 116–122. [Google Scholar] [CrossRef]
- Elberling, B.; Gregorich, E.G.; Hopkins, D.W.; Sparrow, A.D.; Novis, P.; Greenfield, L.G. Distribution and dynamics of soil organic matter in an Antarctic dry valley. Soil Biol. Biochem. 2006, 38, 3095–3106. [Google Scholar] [CrossRef]
- Claridge, G.G.C.; Campbell, I.B.; Sheppard, D.S. Global Climate Change and Cold Regions Ecosystems. In Carbon Pools in Antarctica and Their Significance for Global Climate Change; Lal, R., Kimble, J.M., Stewart, B.H., Eds.; Lewis Publishers: Boca Raton, FL, USA, 2000. [Google Scholar]
- Scharlemann, J.P.W.; Tanner, E.V.J.; Hiederer, R.; Kapos, V. Global soil carbon: Understanding and managing the largest terrestrial carbon pool. Carbon Manag. 2014, 5, 81–91. [Google Scholar] [CrossRef]
- Tatur, A. Ornitogenic soils of the Maritime Antarctic. Pol. Polar Res. 1989, 10, 481–532. [Google Scholar]
- Nędzarek, A.; Rakusa-Suszczewski, S. Decomposition of macroalgae and the release of nutrients in Admiralty Bay, King George Island, Antarctica. Polar Biosci. 2004, 17, 26–35. [Google Scholar]
- Nędzarek, A. Sources, diversity and circulation of biogenic compounds in Admiralty Bay, King George Island, Antarctica. Antarct. Sci. 2008, 20, 135–145. [Google Scholar] [CrossRef]
- Bonan, G.B.; Hartman, M.D.; Parton, W.J.; Wieder, W.R. Evaluating litter decomposition in earth system models with long-term litterbag experiments: An example using the Community Land Model version 4 (CLM4). Glob. Change Biol. 2013, 19, 957–974. [Google Scholar] [CrossRef]
- Michel, R.F.M.; Schaefer, C.E.G.R.; Dias, L.E.; Simas, F.N.B.; de Melo Benites, V.; de Sá Mendonça, E. Ornithogenic Gelisols (Cryosols) from Maritime Antarctica: Pedogenesis, Vegetation, and Carbon Studies. Soil Sci. Soc. Am. J. 2006, 70, 1370–1376. [Google Scholar] [CrossRef]
- Simas, F.N.B.; Schaefer, C.R.; Melo, V.F.; Albuquerqu-Filho, M.R.; Muchel, R.F.M.; Pereria, V.V.; Gomes, M.R.M.; da Costa, L.M. Ornithogenic cryosols from Maritime Antarctica: Phosphatization as a soil forming process. Geoderma 2007, 138, 191–203. [Google Scholar] [CrossRef]
- Abakumov, E.V.; Trubetskoj, O.; Demin, D.; Trubetskaya, O. Electrophoretic evaluation of initial humification in organic horizons of soils of western Antarctica. Polarforschung 2013, 83, 73–82. [Google Scholar]
- Zdanowski, M.K.; Żmuda-Baranowska, M.J.; Borsuk, P.; Świątecki, A.; Górniak, D.; Wolicka, D.; Jankowska, K.M.; Grzesiak, J. Culturable bacteria community development in postglacial soils of Ecology Glacier, King George Island, Antarctica. Polar Biol. 2013, 36, 511–527. [Google Scholar] [CrossRef]
- Świtoniak, M.; Melke, J.; Bartmiński, P. The differences in cellulolytic activity of the Arctic soils of Calypsostranda, Spitsbergen. Polar Rec. 2014, 50, 199–208. [Google Scholar] [CrossRef]
- Androsiuk, P.; Chwedorzewska, K.; Szandar, K.; Giełwanowska, I. Genetic variation of Colobanthus quitensis from King George Island (Antarctic). Pol. Polar Res. 2015, 36, 281–295. [Google Scholar] [CrossRef]
- Poelking, E.L.; Schaefer, C.E.R.; Fernandes Filho, E.I.; de Andrade, A.M.; Spielmann, A.A. Soil–landform–plant-community relationships of a periglacial landscape on Potter Peninsula, maritime Antarctica. Solid Earth 2015, 6, 583–594. [Google Scholar] [CrossRef]
- Abakumov, E.V. The sources and composition of humus in some soils of West Antarctica. Eur. Soil Sci. 2010, 43, 499–508. [Google Scholar] [CrossRef]
- Mu, C.C.; Zhang, T.J.; Zhao, Q.; Guo, H.; Zhong, W.; Su, H.; Wu, Q.B. Soil organic carbon stabilization by iron in permafrost regions of the Qinghai-Tibet Plateau. Geophys. Res. Lett. 2016, 43, 286–294. [Google Scholar] [CrossRef]
- Natali, S.M.; Schuur, E.A.; Webb, E.E.; Pries, C.E.H.; Crummer, K.G. Permafrost degradation stimulates carbon loss from experimentally warmed tundra. Ecology 2014, 95, 602–608. [Google Scholar] [CrossRef]
- Burke, E.J.; Hartley, I.P.; Jones, C.D. Uncertainties in the global temperature change caused by carbon release from permafrost thawing. Cryosphere 2012, 6, 1063–1076. [Google Scholar] [CrossRef]
- Beyer, L.; Vogt, B.; Blume, H.P.; Erlenkeuser, H. Soil organic matter composition and transformation in a swamp soil from algae of coastal continental Antarctica. Polarforschung 1995, 65, 117–122. [Google Scholar]
- Vlasov, D.Y.; Abakumov, E.V.; Nadporozhskaya, M.A.; Kovsh, N.V.; Krylenkov, V.A.; Lukin, V.V.; Safronova, E.V. Lithosols of King George Island, Western Antarctica. Eur. Soil Sci. 2005, 38, 681–687. [Google Scholar]
- Wu, L.; Sheng, M.; Liu, X.; Zheng, Z.; Emslie, S.; Yang, N.; Wang, X.; Nie, Y.; Jin, J.; Xie, Q.; et al. Molecular transformation of organic nitrogen in Antarctic penguin guano-affected soil. Environ. Int. 2023, 172, 107796. [Google Scholar] [CrossRef]
- Alekseev, I.; Abakumov, E. Soil organic matter and biogenic–abiogenic interactions in soils of Larsemann Hills and Bunger Hills, East Antarctica. Polar Sci. 2024, 40, 101040. [Google Scholar] [CrossRef]
- Guglielmin, M.; Ellis Evans, C.J.; Cannone, N. Active layer thermal regime under different vegetation conditions in permafrost areas at Signy Island (Maritime Antarctic). Geoderma 2008, 144, 73–85. [Google Scholar] [CrossRef]
- Meier, L.A.; Krauze, P.; Prater, I.; Horn, F.; Schaefer, C.E.G.R.; Scholten, T.; Wagner, D.; Mueller, C.W.; Kühn, P. Pedogenic and microbial interrelation in initial soils under semiarid climate on James Ross Island, Antarctic Peninsula region. Biogeosciences 2019, 16, 2481–2499. [Google Scholar] [CrossRef]
- Ortiz, M.; Bosch, J.; Coclet, C.; Johnson, J.; Lebre, P.; Salawu-Rotimi, A.; Vikram, S.; Makhalanyane, T.; Cowan, D. Microbial nitrogen cycling in Antarctic soils. Microorganisms 2020, 8, 1442. [Google Scholar] [CrossRef]
- Chwedorzewska, K.J.; Bednarek, P.T. Genetic and epigenetic studies on populations of Deschampsia antarctica Desv. from contrasting environments at King George Island (Antarctic). Pol. Polar Res. 2011, 32, 15–26. [Google Scholar] [CrossRef]
- Nędzarek, A.; Tórz, A.; Drost, A. Selected elements in surface waters of Antarctica and their relations with the natural environment. Polar Res. 2014, 33, 21417. [Google Scholar] [CrossRef]
- Ghani, A.; Dexter, M.; Perrott, K.W. Hot water carbon as an integrated indicator of soil quality. In Proceedings of the 17th World Congress of Soil Science (WCCS), Bangkok, Thailand, 14–21 August 2002. [Google Scholar]
- Valladares, G.S.; Pereira, M.G.; dos Anjos, L.H.C.; Benites, V.M.; Ebeling, A.G.; Mouta, R.O. Humic substance fractions and attributes of histosols and related high-organic-matter soils from Brazil. Commun. Soil Sci. Plant Anal. 2007, 38, 763–777. [Google Scholar] [CrossRef]
- Weber, J.; Chen, Y.; Jamroz, E.; Miano, T. Preface: Humic substances in the environment. J. Soils Sediments 2018, 18, 2665–2667. [Google Scholar] [CrossRef]
- Birkenmajer, K. Retreat of the Ecology Glacier, Admiralty Bay, King George Island (South Shetland Islands, West Antarctica), 1956–2001. Bull. Pol. Acad. Sci. Earth Sci. 2002, 50, 15–29. [Google Scholar]
- Bölter, M. Soil development and soil biology on King George Island, Maritime Antarctic. Pol. Polar Res. 2011, 32, 105–116. [Google Scholar] [CrossRef]
- Rakusa-Suszczewski, S. King George Island—South Shetland Islands, Maritime Antarctic. In Geoecology of Antarctic Ice-Free Coastal Landscapes; Beyer, L., Bölter, M., Eds.; Ecological Studies; Springer: Berlin/Heidelberg, Germany, 2002; Volmue 154, pp. 23–39. [Google Scholar]
- Miranda, C.O.; Lima Neto, E.; Schaefer, C.E.G.R. Anthropogenic effect on the pedochemical variability of potentially toxic elements at the vicinity of an Antarctic research station. An. Acad. Bras. Cienc. 2024, 96, e20230724. [Google Scholar] [CrossRef]
- Convey, P. Antarctic terrestrial biodiversity in a changing world. Polar Biol. 2011, 34, 1629–1641. [Google Scholar] [CrossRef]
- Schmitz, D.; Schaefer, C.E.R.G.; Putzke, J.; Francelino, M.R.; Ferrari, F.R.; Correa, G.R.; Villa, P.M. How does the pedoenvironmental gradient shape non-vascular species assemblages and community structures in Maritime Antarctica? Ecol. Indic. 2020, 108, 105726. [Google Scholar] [CrossRef]
- Ferrari, F.R.; Schaefer, C.E.G.R.; Pereira, A.B.; Thomazini, A.; Schmitz, D.; Francelino, M.R. Coupled soil–vegetation changes along a topographic gradient on King George Island, Maritime Antarctica. Catena 2021, 198, 105038. [Google Scholar] [CrossRef]
- Matos, P.; Rocha, B.; Pinho, P.; Miranda, V.; Pina, P.; Goyanes, G.; Vieira, G. Microscale is key to model current and future Maritime Antarctic vegetation. Sci. Total Environ. 2024, 946, 174171. [Google Scholar] [CrossRef]
- Olech, M.; Chwedorzewska, K.J. The first appearance and establishment of an alien vascular plant in natural habitats on the forefield of a retreating glacier in Antarctica. Antarct. Sci. 2011, 23, 153–154. [Google Scholar] [CrossRef]
- IUSS Working Group WRB. World Reference Base for Soil Resources: International Soil Classification System for Naming Soils and Creating Legends for Soil Maps; International Union of Soil Sciences (IUSS): Vienna, Austria, 2022; p. 234. [Google Scholar]
- ISO 13317-2: 2001; Determination of Particle Size Distribution by Gravitational Liquid Sedimentation Methods. ISO: Geneva, Switzerland, 2001.
- Sparling, G.; Vojvodić-Vuković, M.; Schipper, L.A. Hot-water-soluble C as a simple measure of labile soil organic matter: The relationship with microbial biomass C. Soil Biol. Biochem. 1998, 30, 1469–1472. [Google Scholar] [CrossRef]
- Pansu, M.; Gautheyrou, J. Handbook of Soil Analysis: Mineralogical, Organic and Inorganic Methods; Springer: Berlin/Heidelberg, Germany, 2006. [Google Scholar]
- Tatur, A.; Myrcha, A.; Niegodzisz, J. Formation of abandoned penguin rookery ecosystems in the maritime Antarctic. Polar Biol. 1997, 17, 405–417. [Google Scholar] [CrossRef]
- Łachacz, A.; Kalisz, B.; Giełwanowska, I.; Olech, M.; Chwedorzewska, K.J.; Kellmann-Sopyła, W. Nutrient abundance and variability from soils in the coast of King George Island. J. Soil Sci. Plant Nutr. 2018, 18, 294–311. [Google Scholar]
- De Souza Carvalho, J.V.; Mendonça, E.; La Scala, N., Jr.; Reis, C.; Reis, E.; Schaefer, C. CO2-C losses and carbon quality of selected Maritime Antarctic soils. Antarct. Sci. 2013, 25, 11–18. [Google Scholar] [CrossRef]
- Mergelov, N.; Dolgikh, A.; Shorkunov, I.; Zazovskaya, E.; Soina, V.; Yakushev, A.; Fedorov-Davydov, D.; Pryakhin, S.; Dobryansky, A. Hypolithic communities shape soils and organic matter reservoirs in the ice-free landscapes of East Antarctica. Sci. Rep. 2020, 10, 10277. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Changchun, S.; Wenyan, Y. Land use effects on the distribution of labile organic carbon fractions through soil profiles. Soil Sci. Soc. Am. J. 2006, 70, 660–667. [Google Scholar]
- Barrett, J.E.; Virginia, R.A.; Parsons, A.N.; Wall, D.H. Potential soil organic matter turnover in Taylor Valley, Antarctica. Arct. Antarct. Alp. Res. 2005, 37, 108–117. [Google Scholar] [CrossRef]
- Nadporozhskaya, M.; Abakumov, E.; Khoras’kina, Y.S.; Bykhovets, S.; Shanin, V.N.; Komarov, A. Assessment of the possible dynamics of organic matter in soil in Antarctica under conditions of climate change using the ROMUL mathematical model. Earth’s Cryosphere 2017, 21, 52–59. [Google Scholar]
- Znój, A.; Gawor, J.; Gromadka, R.; Chwedorzewska, K.J.; Grzesiak, J. Root-associated bacteria community characteristics of Antarctic plants—Deschampsia antarctica and Colobanthus quitensis—A comparison. Microb. Ecol. 2022, 84, 808–820. [Google Scholar] [CrossRef]
- Simpson, J.A.; Simpson, M.; Smith, E.; Kelleher, B. Microbially derived inputs to soil organic matter: Are current estimates too low? Environ. Sci. Technol. 2007, 41, 8070–8076. [Google Scholar] [CrossRef]
- Abakumov, E.V.; Parnikoza, I.Y.; Zhianski, M.; Yaneva, R.; Lupachev, A.V.; Andreev, M.P.; Vlasov, D.Y.; Riano, J.; Jaramillo, N. Ornithogenic factor of soil formation in Antarctica: A review. Eurasian Soil Sci. 2021, 54, 528–540. [Google Scholar] [CrossRef]
- Castro, M.F.; Neves, J.C.L.; Francelino, M.R.; Schaefer, C.E.G.R.; Oliveira, T.S. Seabirds enrich Antarctic soil with trace metals in organic fractions. Sci. Total Environ. 2021, 785, 147271. [Google Scholar] [CrossRef]
- Bartos, A.; Stolarczyk, M.; Szymański, W. Content and quality of soil organic matter in topsoils under different tundra vegetation in central Spitsbergen (High Arctic). Geoderma Reg. 2024, 39, e00891. [Google Scholar] [CrossRef]

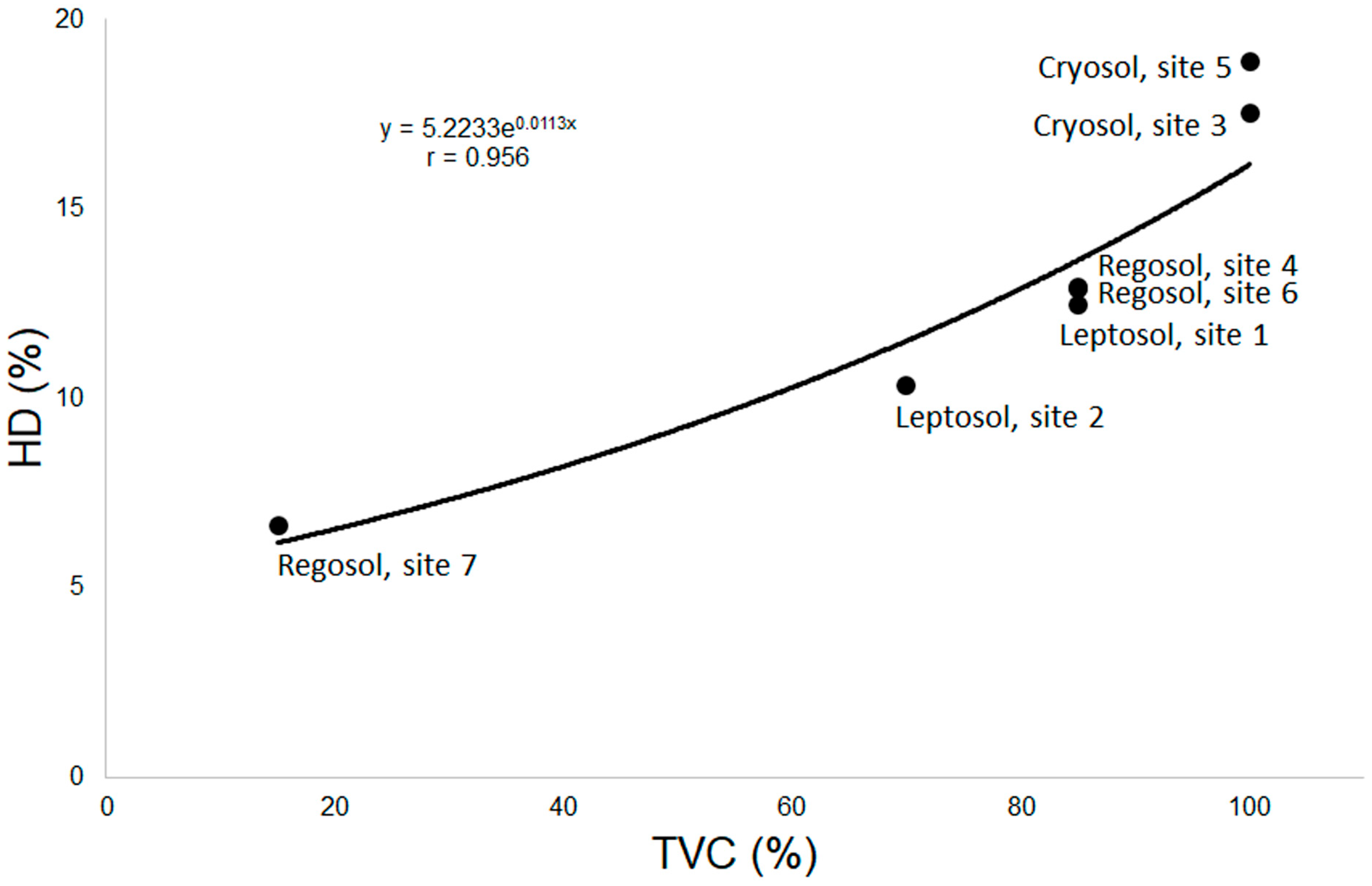
| Site * | Geographical Coordinates | Soil Type | Dominant Plant Species | TVC ** [%] | Distance from the Sea [m] | Altitude [m a.s.l.] |
|---|---|---|---|---|---|---|
| 1 | 62°09′44.7″ S 58°27′45.9″ W | Eutric Skeletic Lithic Leptosols (Humic, Ornithic, Protic) | Ornithocoprophilous lichens: Caloplaca hookeri, C. regalis, Lecania brialmontii, Physcia caesia, Turgidosculum complicatulum and Xanthoria Candelaria; also Prasiola crispa | 85 | 95 | 20 |
| 2 | 62°09′45.0″ S 58°27′25.1″ W | Eutric Skeletic Lithic Leptosols (Humic, Ornithic, Protic) | Caloplaca cirrochrooides, Lecania brialmontii, Ramalina terebrata, Turgidosculum complicatulum, various halophilous species, mainly Verrucaria tesselatula; Prasiola crispa | 70 | 7 | 5 |
| 3 | 62°09′55.8″ S 58°27′40.2″ W | Skeletic Protic Turbic Cryosol (Eutric, Humic, Ornithic) | Flowering plants, moss: Polytrichastrum alpinum and lichens: Bryoria forsteri, Cetraria aculeata, Sphaerophorus globosus and Usnea antarctica, Catharacta maccormicki, C. antarctica | 100 | 800 | 70 |
| 4 | 62°09′56.8″ S 58°28′03.0″ W | Eutric Skeletic Protic Regosol (Turbic) | Flowering plants, lichens: Cetraria aculeata, Leptogium puberulum and Usnea antarctica | 85 | 800 | 60 |
| 5 | 62°09′48.0″ S 58°28′09.0″ W | Dystric Skeletic Protic Cryosol (Humic, Ornithic, Turbic) | Mosses and lichens: Polytrichastrum alpinum, Polytrichum piliferum, Ochrolechia frigida, O. parella | 100 | 350 | 55 |
| 6 | 62°10′07.0″ S 58°28′55.0″ W | Eutric Skeletic Protic Leptic Regosol | Lichens: Cladonia borealis, Himantormia lugubris, Leptogium puberulum, Placopsis parella, Parmelia saxatilis, Stereocaulon glabrum, Umbilicaria antarctica, Usnea antarctica, U. aurantiacoatra | 85 | 900 | 180 |
| 7 | 62°10′05.0″ S 58°27′45.0″ W | Eutric Skeletic Protic Regosol | Colobanthus quitensis, Deschampsia antarctica | 15 | 100–150 (from the lagoon) | 3 |
| Site No. | Statistical Parameter | SOM | TOC | TOC/TN | HWC | PHP-C | DPH-C | SODA-C | HD | L/S | Residuum |
|---|---|---|---|---|---|---|---|---|---|---|---|
| g kg−1 | % TOC | % | % TOC | ||||||||
| 1 | Mean | 72.4 ± 10.6 | 48.7 ± 9.5 | 7.2 ± 1.5 | 4.3 ± 1.6 | 0.24 ± 0.09 | 3.1 ± 0.8 | 9.4 ± 2.2 | 12.5 ± 2.9 | 0.35 ± 0.11 | 87.5 ± 2.9 |
| CV (%) | 145.9 | 19.5 | 20.1 | 36.3 | 39.4 | 24.2 | 23.3 | 23.2 | 31.7 | 3.3 | |
| 2 | Mean | 124.6 ± 47.7 | 89.1 ± 49.1 | 9.8 ± 1.9 | 3.7 ± 1.4 | 0.42 ± 0.52 | 3.2 ± 1.7 | 7.2 ± 2.9 | 10.3 ± 4.3 | 0.40 ± 0.18 | 89.6 ± 4.3 |
| CV (%) | 382.7 | 55.1 | 19.9 | 37.2 | 123.9 | 54.5 | 39.9 | 41.4 | 44.8 | 4.8 | |
| 3 | Mean | 63.6 ± 28.4 | 37.4 ± 23.8 | 19.9 ± 7.1 | 3.5 ± 2.3 | 0.47 ± 0.27 | 8.0 ± 2.8 | 9.6 ± 2.4 | 17.5 ± 4.1 | 0.20 ± 0.15 | 82.5 ± 4.1 |
| CV (%) | 446.6 | 63.7 | 35.7 | 65.9 | 56.8 | 35.8 | 25.4 | 23.1 | 73.5 | 4.9 | |
| 4 | Mean | 27.0 ± 3.4 | 6.3 ± 1.1 | 34.6 ± 5.8 | 2.0 ± 1.2 | 0.93 ± 0.93 | 4.4 ± 2.0 | 8.5 ± 2.3 | 12.9 ± 2.9 | 0.16 ± 0.06 | 87.1 ± 2.9 |
| CV (%) | 124.4 | 17.7 | 16.7 | 58.7 | 99.6 | 46.2 | 27.5 | 22.6 | 40.4 | 3.4 | |
| 5 | Mean | 82.5 ± 26.5 | 49.2 ± 22.9 | 16.2 ± 3.3 | 2.1 ± 1.4 | 0.22 ± 0.12 | 9.1 ± 1.8 | 9.8 ± 2.0 | 18.9 ± 3.5 | 0.12 ± 0.08 | 81.1 ± 3.5 |
| CV (%) | 320.9 | 46.6 | 20.1 | 67.7 | 52.4 | 19.6 | 20.1 | 18.5 | 70.0 | 4.3 | |
| 6 | Mean | 45.0 ± 7.8 | 20.3 ± 6.0 | 20.6 ± 3.2 | 1.5 ± 1.4 | 5.9 ± 10.5 | 4.9 ± 2.4 | 8.0 ± 2.1 | 12.9 ± 2.8 | 0.11 ± 0.11 | 87.1 ± 2.8 |
| CV (%) | 174.2 | 29.6 | 15.7 | 96.3 | 178.2 | 47.7 | 26.5 | 21.9 | 97.5 | 3.2 | |
| 7 | Mean | 23.3 ± 1.7 | 8.5 ± 2.0 | 50.8 ± 11.0 | 2.7 ± 1.7 | 12.3 ± 17.8 | 2.1 ± 0.4 | 4.6 ± 1.8 | 6.6 ± 2.1 | 0.47 ± 0.38 | 93.3 ± 2.1 |
| CV (%) | 71.3 | 23.9 | 21.7 | 62.4 | 144.7 | 16.7 | 40.1 | 32.2 | 81.3 | 2.3 | |
| Variable | TOC | TN | TOC/TN | L/S | HD | Residuum |
|---|---|---|---|---|---|---|
| HWC | 0.855 * | 0.883 * | −0.550 * | 0.471 * | −0.163 | 0.163 |
| PHP-C | −0.185 | 0.190 | 0.247 | −0.041 | −0.087 | 0.087 |
| SODA-C | 0.778 * | 0.726 * | −0.753 * | −0.006 | 0.270 | −0.270 |
| DPH-C | 0.599 * | 0.378 | −0.513 * | −0.154 | 0.506 * | −0.506 * |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kalisz, B.; Łachacz, A.; Giełwanowska, I.; Olech, M.; Chwedorzewska, K.J.; Kellmann-Sopyła, W. Labile and Stable Carbon Pools in Antarctic Soils of the Arctowski Region, King George Island. Sustainability 2025, 17, 7221. https://doi.org/10.3390/su17167221
Kalisz B, Łachacz A, Giełwanowska I, Olech M, Chwedorzewska KJ, Kellmann-Sopyła W. Labile and Stable Carbon Pools in Antarctic Soils of the Arctowski Region, King George Island. Sustainability. 2025; 17(16):7221. https://doi.org/10.3390/su17167221
Chicago/Turabian StyleKalisz, Barbara, Andrzej Łachacz, Irena Giełwanowska, Maria Olech, Katarzyna Joanna Chwedorzewska, and Wioleta Kellmann-Sopyła. 2025. "Labile and Stable Carbon Pools in Antarctic Soils of the Arctowski Region, King George Island" Sustainability 17, no. 16: 7221. https://doi.org/10.3390/su17167221
APA StyleKalisz, B., Łachacz, A., Giełwanowska, I., Olech, M., Chwedorzewska, K. J., & Kellmann-Sopyła, W. (2025). Labile and Stable Carbon Pools in Antarctic Soils of the Arctowski Region, King George Island. Sustainability, 17(16), 7221. https://doi.org/10.3390/su17167221







