Biochar Utilization as a Forestry Climate-Smart Tool
Abstract
1. Introduction
2. Historic Atmospheric Conditions
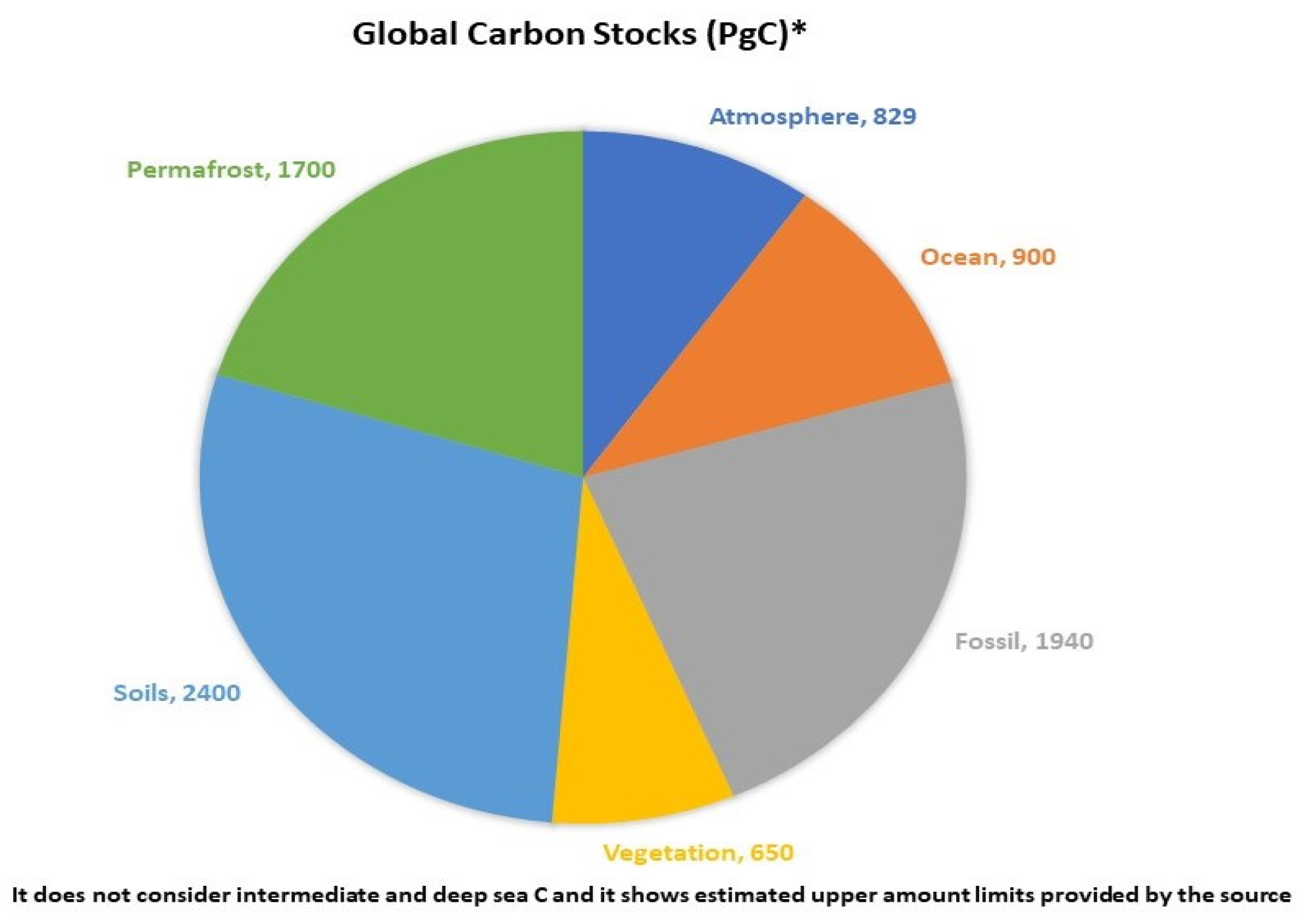
Terrestrial and Atmospheric C Exchange
3. Forest Vegetation and Soil
4. Biochar from Low-Value Woody Residues
5. Discussion
- ▪
- Manage stand density to decrease competition for space, water, and nutrients, and to increase resilience to droughts, insects, and disease, thereby decreasing mortality, wildland fire risk, and vulnerability to extreme weather events.
- ▪
- Where possible, control pest outbreaks when they occur outside the norm.
- ▪
- Restore forest ecosystems impacted by catastrophic disturbances such as wildland fires, hurricanes, and other disturbances, consistent with land management laws and regulations.
- ▪
- Develop programs that enhance C sequestration potential through afforestation, reforestation, and practices that increase and maintain vegetation productivity and ecosystem health.
- ▪
- Encourage cities to retain green space and to plant and maintain trees.
- ▪
- Use available tools to understand management impacts on C stocks and fluxes, such as exploring the possibilities to increase rotation length, and the implementation of best management practices to decrease impacts on C soil pools to protect biodiversity and decrease erosion.
- ▪
- Educate landowners on the value of producing and using biochar on-site.
6. Conclusions and Future Directions
- •
- There is strong evidence that climate-smart forest management practices are an efficient option for managing C, increasing C stocks, decreasing impacts of climate change, and promoting sustainable forest ecosystems with other associated benefits.
- •
- Forest management actions, such as afforestation, reforestation, restoration, and utilization of woody biomass residues for biochar, are efficient C management approaches with high mitigation potential.
- •
- Biochar is a climate-smart tool that contributes to climate change mitigation, increasing soil C sequestration, reducing soil GHG emissions, storing water, and providing other associated benefits.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Disclaimer
References
- LibreTexts. General Biology (Boundless). 867 Webpages. California State University Affordable Learning Solutions Program. 2024. Available online: https://bio.libretexts.org/Bookshelves/Introductory_and_General_Biology/Book%3A_General_Biology_(Boundless)/zz%3A_Back_Matter/21%3A_Detailed_Licensing (accessed on 3 January 2024).
- Britannica. The Editors of Encyclopedia. “Carbon”. Encyclopedia Britannica, 15 November 2023. Available online: https://www.britannica.com/science/carbon-chemical-element (accessed on 3 January 2024).
- IPCC. Summary for Policymakers. In Climate Change 2022: Mitigation of Climate Change; Contribution of Working Group III to the Sixth Assessment Report of the Intergovernmental Panel on Climate Change; Shukla, P.R., Skea, J., Slade, R., Al Khourdajie, A., van Diemen, R., McCollum, D., Pathak, M., Some, S., Vyas, P., Fradera, R., et al., Eds.; Cambridge University Press: Cambridge, UK; New York, NY, USA, 2022. [Google Scholar] [CrossRef]
- Lal, R. Soil carbon sequestration to mitigate climate change. Geoderma 2004, 123, 1–22. [Google Scholar] [CrossRef]
- Bonan, G.B. Forests and climate change: Forcings, feedbacks, and the climate benefit of forests. Science 2007, 320, 1444–1451. [Google Scholar] [CrossRef]
- United Nations Climate Change. The Paris Agreement. What Is the Paris Agreement? Process and Meetings. The Paris Agreement. 2024. Available online: https://unfccc.int/process-and-meetings/the-paris-agreement (accessed on 9 January 2024).
- Kasting, J.F. The evolution of the prebiotic atmosphere. Orig. Life Evol. Biosph. 1984, 14, 75–82. [Google Scholar] [CrossRef] [PubMed]
- Kasting, J.F. Theoretical constraints on oxygen and carbon dioxide concentrations in the Precambrian atmosphere. Precambrian Res. 1987, 34, 205–229. [Google Scholar] [CrossRef] [PubMed]
- Payne, C.R.; Brownlee, D.; Kasting, F.J. Oxidized micrometeorites suggest either high pCO2 or low pN2 during the Neoarchean. Proc. Natl. Acad. Sci. USA 2020, 117, 1360–1366. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Momohara, A.; Wang, L.; Lebreton-Anberrée, J.; Zhou, Z. Evolutionary History of Atmospheric CO2 during the Late Cenozoic from Fossilized Metasequoia Needles. PLoS ONE 2015, 10, e0130941. [Google Scholar] [CrossRef]
- Rae, W.B.J.; Zhang, G.Y.; Liu, X.; Foster, L.G.; Stoll, M.H.; Whiteford, D.M.R. Atmospheric CO2 over the Past 66 Million Years from Marine Archives. Annu. Rev. Earth Planet. Sci. 2021, 49, 609–641. [Google Scholar] [CrossRef]
- Dumitru, O.A.; Austermann, J.; Polyak, V.J.; Fornós, J.J.; Asmerom, Y.; Ginés, J.; Ginés, A.; Onac, B.P. Constraints on global mean sea level during Pliocene warmth. Nature 2019, 574, 233–236. [Google Scholar] [CrossRef] [PubMed]
- Lüthi, D.; Le Floch, M.; Bereiter, B.; Blunier, T.; Barnola, J.-M.; Siegenthaler, U.; Raynaud, D.; Jouzel, J.; Fischer, H.; Kawamura, K.; et al. High-resolution carbon dioxide concentration record 650,000–800,000 years before present. Nature 2008, 453, 379–382. [Google Scholar] [CrossRef]
- Siegenthaler, U.; Stocker, T.F.; Monnin, E.; Lu, D.; Schwander, J.; Stauffer, B.; Raynaud, D.; Barnola, J.-M.; Fischer, H.; Masson-Delmotte, V.; et al. Stable Carbon Cycle Climate Relationship During the Late Pleistocene. Science 2005, 310, 1313–1317. [Google Scholar] [CrossRef]
- Masson-Delmotte, V.; Schulz, M.; Abe-Ouchi, A.; Beer, J.; Ganopolski, A.; Rouco, J.F.G.; Jansen, E.; Lambeck, K.; Luterbacher, J.; Naish, T.; et al. Information from Paleoclimate Archives. In Climate Change 2013: The Physical Science Basis; Contribution of Working Group I to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change; Stocker, T.F., Qin, D., Plattner, G.-K., Tignor, M., Allen, S.K., Boschung, J., Nauels, A., Xia, Y., Bex, V., Midgley, P.M., Eds.; Cambridge University Press: Cambridge, UK; New York, NY, USA, 2013; Available online: https://www.ipcc.ch/site/assets/uploads/2018/02/WG1AR5_Chapter05_FINAL.pdf (accessed on 10 October 2023).
- Monnin, E.; Indermühle, A.; Dällenbach, A.; Flückiger, J.; Stauffer, B.; Stocker, T.F.; Raynaud, D.; Barnola, J.M. Atmospheric CO2 Concentrations over the Last Glacial Termination. Science 2001, 291, 112–114. [Google Scholar] [CrossRef]
- National Oceanic and Atmospheric Administration. Climate Change: Atmospheric Carbon Dioxide. 2023. Available online: https://www.climate.gov/news-features/understanding-climate/climate-change-atmospheric-carbon-dioxide#:~:text=In%20May%202023%2C%20carbon%20dioxide,people%20are%20burning%20for%20energy (accessed on 10 October 2023).
- National Oceanic and Atmospheric Administration. Carbon Cycle Greenhouse Gases, Trends in CO2. Global Monitoring Laboratory. 2024. Available online: https://gml.noaa.gov/ccgg/trends/weekly.html (accessed on 8 January 2024).
- Copernicus Climate Change Service. Global Climate Highlights 2023. Europe Commission Programme. 2024. Available online: https://climate.copernicus.eu/global-climate-highlights-2023 (accessed on 9 January 2024).
- British Geological Survey. The Carbon Story. 2023. Available online: https://www.bgs.ac.uk/discovering-geology/climate-change/the-carbon-story/#:~:text=More%20than%2099%20per%20cent,in%20their%20skeletons%20and%20shells (accessed on 19 October 2023).
- Hayes, J.M. Evolution of the Atmosphere. Britannica. (Archived on 16 November 2020). 2020. Available online: https://www.britannica.com/topic/evolution-of-the-atmosphere-1703862 (accessed on 25 September 2023).
- Bowyer, J.; Bratkovich, S.; Frank, M.; Howe, J.; Stai, S.; Fernholz, K. Carbon 101: Understanding the Carbon Cycle and the Forest Carbon Debate; Dovetail Partners, Inc.: Minneapolis, MN, USA, 2012; 13p, Available online: https://www.dovetailinc.org/upload/tmp/1581622918.pdf (accessed on 13 October 2023).
- Dong, Y.; Cui, Y.; Wang, J.; Chen, H.; Zhang, F.; Wu, Y.; Li, Z.; Zhu, P.; Hijun Jiang, S. Paleozoic carbon cycle dynamics: Insights from stable carbon isotopes in marine carbonates and C3 land plants. Earth-Sci. Rev. 2021, 222, 103813. [Google Scholar] [CrossRef]
- Bruhwiler, L.; Michalak, A.M.; Birdsey, R.; Fisher, J.B.; Houghton, R.A.; Huntzinger, D.N.; Miller, J.B. Chapter 1: Overview of the global carbon cycle. In Second State of the Carbon Cycle Report (SOCCR2): A Sustained Assessment Report; Cavallaro, N., Shrestha, G., Birdsey, R., Mayes, M.A., Najjar, R.G., Reed, S.C., Romero-Lankao, P., Zhu, Z., Eds.; U.S. Global Change Research Program: Washington, DC, USA, 2018; pp. 42–70. [Google Scholar] [CrossRef]
- Carlson, C.A.; Bates, N.R.; Hansell, D.A.; Steinberg, D.K. Carbon Cycle. In Encyclopedia of Ocean Sciences, 2nd ed.; Steele, J.H., Ed.; Academic Press: Cambridge, MA, USA, 2001; pp. 477–486. ISBN 9780123744739. [Google Scholar] [CrossRef]
- Stephenson, M. Chapter 1—The Carbon Cycle, Fossil Fuels and Climate Change. In Energy and Climate Change: An Introduction to Geological Controls, Interventions and Mitigations; Energy and Climate Change; Stephenson, M., Ed.; Elsevier: Amsterdam, The Netherlands, 2018; pp. 1–26. ISBN 9780128120217. [Google Scholar] [CrossRef]
- Wuebbles, D.J.; Fahey, D.W.; Hibbard, K.A.; DeAngelo, B.; Doherty, S.; Hayhoe, K.; Horton, R.; Kossin, J.P.; Taylor, P.C.; Waple, A.M.; et al. Executive summary. In Climate Science Special Report: Fourth National Climate Assessment, Volume I; Wuebbles, D.J., Fahey, D.W., Hibbard, K.A., Dokken, D.J., Stewart, B.C., Maycock, T.K., Eds.; U.S. Global Change Research Program: Washington, DC, USA, 2017; pp. 12–34. [Google Scholar] [CrossRef]
- Houghton, R.A. 8.10—The Contemporary Carbon Cycle. In Treatise on Geochemistry; Holland, H.D., Turekian, K.K., Eds.; Pergamon: Oxford, UK, 2003; pp. 473–513. ISBN 9780080437514. [Google Scholar] [CrossRef]
- Hannah, L. Chapter 2—The Climate System and Climate Change. In Climate Change Biology; Hannah, L., Ed.; Academic Press: Cambridge, MA, USA, 2011; pp. 13–52. ISBN 9780123741820. [Google Scholar] [CrossRef]
- United Nations Environment Programme. Executive summary. In Emissions Gap Report 2023: Broken Record—Temperatures Hit New Highs, Yet World Fails to Cut Emissions (Again); United Nations Environment Programme: Nairobi, Kenya, 2023. [Google Scholar] [CrossRef]
- Birdsey, R.; Pan, Y. Trends in management of the world’s forests and impacts on carbon stocks. For. Ecol. Manag. 2015, 355, 83–90. [Google Scholar] [CrossRef]
- Keenan, J.R.; Reams, A.G.; Achard, F.; de Freitas, V.J.; Grainger, A.; Lindquist, E. Dynamics of global forest area: Results from the FAO Global Forest Resources Assessment 2015. For. Ecol. Manag. 2015, 352, 9–20. [Google Scholar] [CrossRef]
- Bastin, J.F.; Berrahmouni, N.; Grainger, A.; Maniatis, D.; Mollicone, D.; Moore, R.; Patriarca, C.; Picard, N.; Sparrow, B.; Abraham, E.M.; et al. The extent of forest in dryland biomes. Science 2017, 356, 635–638. [Google Scholar] [CrossRef]
- FAO. Global Forest Resources Assessment 2020: Main Report; FAO: Rome, Italy, 2020. [Google Scholar] [CrossRef]
- Kayler, Z.; Janowiak, M.; Swanston, C. Global Carbon. (June 2017). U.S. Department of Agriculture, Forest Service, Climate Change Resource Center. 2017. Available online: https://www.fs.usda.gov/ccrc/topics/global-carbon (accessed on 27 November 2023).
- Mäkipää, R.; Abramoff, R.; Adamczyk, B.; Baldy, V.; Biryol, C.; Bosela, M.; Casals, P.; Yuste, C.J.; Dondini, M.; Filipek, S.; et al. How does management affect soil C sequestration and greenhouse gas fluxes in boreal and temperate forests?—A review. For. Ecol. Manag. 2023, 529, 120637. [Google Scholar] [CrossRef]
- Pugh, T.A.; Lindeskog, M.; Smith, B.; Poulter, B.; Arneth, A.; Haverd, V.; Calle, L. Role of forest regrowth in global carbon sink dynamics. Proc. Natl Acad. Sci. USA 2019, 116, 4382–4387. [Google Scholar] [CrossRef] [PubMed]
- Harris, N.L.; Gibbs, D.A.; Baccini, A.; Birdsey, R.A.; de Bruin, S.; Farina, M.; Fatoyinbo, L.; Hansen, M.C.; Herold, M.; Houghton, R.A.; et al. Global maps of twenty-first century forest carbon fluxes. Nat. Clim. Chang. 2021, 11, 234–240. [Google Scholar] [CrossRef]
- Mayer, M.; Prescott, C.E.; Abaker, W.E.; Augusto, L.; Cécillon, L.; Ferreira, G.W.; James, J.; Jandl, R.; Katzensteiner, K.; Laclau, J.P.; et al. Tamm Review: Influence of forest management activities on soil organic carbon stocks: A knowledge synthesis. For. Ecol. Manag. 2020, 466, 118127. [Google Scholar] [CrossRef]
- Woodall, C.W.; Coulston, J.W.; Domke, G.M.; Walters, B.F.; Wear, D.N.; Smith, J.E.; Andersen, E.H.; Clough, B.J.; Cohen, W.B.; Griffith, D.M.; et al. The U.S. Forest Carbon Accounting Framework: Stocks and Stock Change, 1990–2016; General Technical Report NRS-154; USDA Forest Service: Newtown Square, PA, USA, 2015; p. 50. Available online: https://www.fs.fed.us/nrs/pubs/gtr/gtr_nrs154.pdf (accessed on 20 October 2023).
- Abed, J.; Rayburg, S.; Rodwell, J.; Neave, M. A Review of the Performance and Benefits of Mass Timber as an Alternative to Concrete and Steel for Improving the Sustainability of Structures. Sustainability 2022, 14, 5570. [Google Scholar] [CrossRef]
- National Academies of Sciences, Engineering, and Medicine. Negative Emissions Technologies and Reliable Sequestration: A Research Agenda; The National Academies Press: Washington, DC, USA, 2019. [Google Scholar] [CrossRef]
- Janowiak, M.; Connelly, W.J.; Dante-Wood, K.; Domke, G.M.; Giardina, C.; Kayler, Z.; Marcinkowski, K.; Ontl, T.; Rodriguez-Franco, C.; Swanston, C.; et al. Considering Forest and Grassland Carbon in Land Management; General Technical Report WO-95; USDA, Forest Service: Washington, DC, USA, 2017; 69p. [CrossRef]
- Chadwick, D.O.; Larson, B. Forest Stand Dynamics; John Wiley and Sons Inc.: Hoboken, NJ, USA, 1996; p. 520. [Google Scholar]
- Scharlemann, P.W.J.; Tanner, V.J.E.; Hiederer, R.; Kapos, V. Global soil carbon: Understanding and managing the largest terrestrial carbon pool. Carbon Manag. 2014, 5, 81–91. [Google Scholar] [CrossRef]
- Grassi, G.; House, J.; Dentener, F.; Federici, S.; den Elzen, M.; Penman, J. The key role of forests in meeting climate targets requires science for credible mitigation. Nat. Clim. Chang. 2017, 7, 220–226. [Google Scholar] [CrossRef]
- Pan, Y.; Birdsey, A.R.; Fang, J.; Houghton, R.; Kauppi, E.P.; Kurz, A.W.; Phillips, L.O.; Shvidenko, A.; Lewis, L.S.; Canadell, G.J.; et al. A Large and Persistent Carbon Sink in the World’s Forests. Science 2011, 333, 988–993. [Google Scholar] [CrossRef] [PubMed]
- Jandl, R.; Lindner, M.; Vesterdal, L.; Bauwens, B.; Baritz, R.; Hagedorn, F.; Johnson, D.W.; Minkkinen, K.; Byrne, K.A. How strongly can forest management influence soil carbon sequestration? Geoderma 2007, 137, 253–268. [Google Scholar] [CrossRef]
- Noormets, A.; Epron, D.; Domec, J.C.; McNulty, S.G.; Fox, T.; Sun, G.; King, J.S. Effects of forest management on productivity and carbon sequestration: A review and hypothesis. For. Ecol. Manag. 2015, 355, 124–140. [Google Scholar] [CrossRef]
- Failey, E.L.; Dilling, L. Carbon stewardship: Land management decisions and the potential for carbon sequestration in Colorado, USA. Environ. Res. Lett. 2010, 5, 024005. [Google Scholar] [CrossRef]
- Mahapatra, A.K.; Shackleton, C.M. Exploring the relationships between trade in natural products, cash income and livelihoods in tropical forest regions of Eastern India. Int. For. Rev. 2012, 14, 62–73. [Google Scholar] [CrossRef]
- Bárcena, T.G.; Kiær, P.L.; Vesterdal, L.; Stefánsdóttir, H.; Gundersen, P.; Sigurdsson, B. Soil carbon stock change following afforestation in Northern Europe: A meta-analysis. Glob. Chang. Biol. 2014, 20, 2393–2405. [Google Scholar] [CrossRef]
- Nave, L.; Swanston, C.; Mishra, U.; Nadelhoffer, K. Afforestation effects on soil carbon storage in the United States: A synthesis. Soil Sci. Soc. Am. J. 2013, 77, 1035–1047. [Google Scholar] [CrossRef]
- Laganiere, J.; Angers, D.A.; Pare, D. Carbon accumulation in agricultural soils after afforestation: A meta-analysis. Glob. Chang. Biol. 2010, 16, 439–453. [Google Scholar] [CrossRef]
- 55. Shi, S.W.; Zhang, W.; Zhang, P.; Yu, Y.Q.; Ding, F. A synthesis of change in deep soil organic carbon stores with afforestation of agricultural soils. For. Ecol. Manag 2013, 296, 53–63. [Google Scholar] [CrossRef]
- Nave, E.L.; Vance, D.E.; Swanston, W.C.; Curtis, S.P. Harvest impacts on soil carbon storage in temperate forests. For. Ecol. Manag. 2010, 259, 857–866. [Google Scholar] [CrossRef]
- Zhang, J.; Page-Dumroese, D.S.; Jurgensen, M.F.; Busse, M.; Mattson, K.G. Coarse Woody Debris and Carbon Stocks in Pine Forests after 50 Years of Recovery from Harvesting in Northeastern California. Forests 2023, 14, 623. [Google Scholar] [CrossRef]
- Magnússon, R.Í.; Tietema, A.; Cornelissen, J.H.; Hefting, M.M.; Kalbitz, K. Tamm Review: Sequestration of carbon from coarse woody debris in forest soils. For. Ecol. Manag. 2016, 377, 1–15. [Google Scholar] [CrossRef]
- FAO. Forestry for a Low-Carbon Future: Integrating Forests and Wood Products in Climate Change Strategies; FAO Forestry Paper 177; FAO: Rome, Italy, 2016; 180p, Available online: https://www.fao.org/3/i5857e/I5857E.pdf (accessed on 9 January 2024).
- Glaser, B.; Haumaier, L.; Guggenberger, G.; Zech, W. The Terra Preta phenomenon—A model for sustainable agriculture in the humid tropics. Aturwissenschaften 2001, 88, 37–41. [Google Scholar] [CrossRef]
- Lehmann, J.; Gaunt, J.; Rondon, M. Bio-char sequestration in terrestrial ecosystems: A review. Mitig. Adapt. Strateg. Glob. Chang. 2006, 11, 403–427. [Google Scholar] [CrossRef]
- Lefebvre, D.; Fawzy, S.; Aquije, C.A.; Osman, A.I.; Draper, K.T.; Trabold, T.A. Biomass residue to carbon dioxide removal: Quantifying the global impact of biochar. Biochar 2023, 5, 65. [Google Scholar] [CrossRef]
- Joseph, S.; Cowie, A.; Zwieten, L.; Bolan, N.; Budai, A.; Buss, W.; Cayuela, M.; Graber, E.; Ippolito, J.; Kuzyakov, Y.; et al. How biochar works, and when it doesn’t: A review of mechanisms controlling soil and plant responses to biochar. Glob. Chang. Biol. Bioenergy 2021, 13, 1731–1764. [Google Scholar] [CrossRef]
- Rodriguez, F.C.; Page-Dumroese, S.D.; Archuleta, J. Forest management and biochar for continued ecosystem services. J. Soil Water Conserv. 2022, 77, 60A–64A. [Google Scholar] [CrossRef]
- Rodriguez-Franco, C.; Page-Dumroese, D.S. Woody biochar potential for abandoned mine land restoration in the U.S.: A review. Biochar 2021, 3, 7–22. [Google Scholar] [CrossRef]
- Jia, J.; Cheng, P.; Yu, Y.; Chen, S.; Wang, C.; He, L.; Nie, H.; Wang, J.; Zhang, J.; Fan, B.; et al. Regeneration mechanism of a novel high-performance biochar mercury adsorbent directionally modified by multimetal multilayer loading. J. Environ. Manag. 2023, 326 Pt B, 116790. [Google Scholar] [CrossRef]
- Lehmann, J.; Joseph, S. Biochar for environmental management: An introduction. In Biochar for Environmental Management: Science, Technology and Implementation, 2nd ed.; Lehmann, J., Joseph, S., Eds.; Earthscan: London, UK, 2015; 1214p, Available online: https://www.book2look.com/embed/9781134489602 (accessed on 11 August 2023).
- Lehmann, J.; Joseph, S. Biochar for Environmental Management: An Introduction. In Biochar for Environmental Management; Lehmann, J., Joseph, S., Eds.; Earthscan: London, UK, 2009; 438p, Available online: https://www.taylorfrancis.com/books/edit/10.4324/9781849770552/biochar-environmental-management-johannes-lehmann-stephen-joseph (accessed on 3 October 2023).
- Greco, G.; Gonzalez, B.; Manya, J.J. Operating conditions affecting char yield and its potential stability during slow pyrolysis of biomass: A review. In Advanced Carbon Materials from Biomass: An Overview; Manyà, J.J., Ed.; GreenCarbon Project and Consortium: Zaragoza, Spain, 2019. [Google Scholar] [CrossRef]
- McHenry, P.M. Chapter 26—Biochar Processing for Sustainable Development in Current and Future Bioenergy Research. In Bioenergy Research: Advances and Applications; Gupta, V.K., Tuohy, M.G., Kubicek, C.P., Saddler, J., Xu, F., Eds.; Elsevier: Amsterdam, The Netherlands, 2014; pp. 447–456. [Google Scholar] [CrossRef]
- Blackwell, P.; Riethmuller, G.; Collins, M. Biochar application to soil. In Biochar for Environmental Management: Science and Technology; Lehmann, J., Joseph, S., Eds.; Earthscan: London, UK, 2009; pp. 207–226. Available online: https://www.taylorfrancis.com/books/edit/10.4324/9781849770552/biochar-environmental-management-johannes-lehmann-stephen-joseph (accessed on 3 October 2023).
- Spokas, A.K. Review of the stability of biochar in soils: Predictability of O:C molar ratios. Carbon Manag. 2010, 1, 289–303. [Google Scholar] [CrossRef]
- Ali, M.; Javeed, H.M.R.; Tariq, M.; Khan, A.A.; Qamar, R.; Nawaz, F.; Masood, N.; Ditta, A.; Abbas, T.; Zamir, M.S.I.; et al. Use of Biochar for Biological Carbon Sequestration. In Climate Change Impacts on Agriculture; Jatoi, W.N., Mubeen, M., Hashmi, M.Z., Ali, S., Fahad, S., Mahmood, K., Eds.; Springer: Cham, Switzerland, 2023. [Google Scholar] [CrossRef]
- Amonette, E.J.; Blanco-Canqui, H.; Hassebrook, C.; Laird, A.D.; Lal, R.; Lehmann, J.; Page-Dumroese, D. Integrated biochar research: A roadmap. J. Soil Water Conserv. 2021, 76, 24A–29A. [Google Scholar] [CrossRef]
- Wang, C.; Tu, Q.; Dong, D.; Strong, P.J.; Wang, H.; Sun, B.; Wu, W. Spectroscopic evidence for biochar amendment promoting humic acid synthesis and intensifying humification during composting. J. Hazard Mater. 2014, 280, 409–416. [Google Scholar] [CrossRef]
- Sarauer, J.L.; Page-Dumroese, D.S.; Coleman, M.D. Soil greenhouse gas, carbon content, and tree growth response to biochar amendment in western United States forests. Glob. Chang. Biol. Bioenergy 2019, 11, 660–671. [Google Scholar] [CrossRef]
- National Academies of Sciences, Engineering, and Medicine. Land Management Practices for Carbon Dioxide Removal and Reliable Sequestration: Proceedings of a Workshop—In Brief; The National Academies Press: Washington, DC, USA, 2018. [Google Scholar] [CrossRef]
- Prentice, I.C.; Farquhar, G.D.; Fasham, M.J.R.; Goulden, M.L.; Heimann, M.; Jaramillo, V.J.; Kheshgi, H.S.; Le Quéré, C.; Scholes, R.J.; Wallace, D.W.R. The Carbon Cycle and Atmospheric Carbon Dioxide. In Climate Change 2001: The Scientific Basis; Contribution of Working Group I to the Third Assessment Report of the Intergovernmental Panel on Climate Change; Houghton, J.T., Ding, Y., Griggs, D.J., Noguer, M., van der Linden, P.J., Dai, X., Maskell, K., Johnson, C.A., Eds.; Cambridge University Press: Cambridge, UK; New York, NY, USA, 2001; 881p, Available online: https://www.ipcc.ch/site/assets/uploads/2018/02/TAR-03.pdf (accessed on 9 January 2024).
- Gür, M.T. Carbon Dioxide Emissions, Capture, Storage and Utilization: Review of Materials, Processes and Technologies. Prog. Energy Combust. Sci. 2022, 89, 100965. [Google Scholar] [CrossRef]
- Jones, M.W.; Santín, C.; van der Werf, G.R.; Doerr, S.H. Global fire emissions buffered by the production of pyrogenic carbon. Nat. Geosci. 2019, 12, 742–747. [Google Scholar] [CrossRef]
- Zheng, B.; Philippe Ciais, P.; Chevallier, F.; Yang, H.; Canadell, G.J.; Chen, Y.; van der Velde, R.I.; Aben, I.; Chuvieco, E.; Davis, J.S.; et al. Record-high CO2 emissions from boreal fires in 2021. Science 2023, 379, 912–917. [Google Scholar] [CrossRef] [PubMed]
- European Union. Atmosphere Monitoring Service. A Record-Breaking Boreal Wildfire Season. 2023. Available online: https://atmosphere.copernicus.eu/record-breaking-boreal-wildfire-season#:~:text=on%20the%20atmosphere.-,Record%2Dbreaking%20fires%20across%20Canada,total%20for%202023%20to%20date. (accessed on 15 November 2023).
- Ameray, A.; Bergeron, Y.; Valeria, O.; Montoro Girona, M.; Cavard, X. Forest Carbon Management: A Review of Silvicultural Practices and Management Strategies Across Boreal, Temperate and Tropical Forests. Curr. For. Rep 2021, 7, 245–266. [Google Scholar] [CrossRef]
- Depro, B.M.; Brian CMurray, B.C.; Ralph, J.; Alig, R.J.; Shanks, A. Public land, timber harvests, and climate mitigation: Quantifying carbon sequestration potential on U.S. public timberlands. For. Ecol. Manag. 2007, 255, 1122–1134. Available online: https://www.fs.fed.us/pnw/pubs/journals/pnw_2008_depro001.pdf (accessed on 3 October 2023). [CrossRef]
- Ryan, M.G.; Harmon, M.E.; Birdsey, R.A.; Giardina, C.P.; Heath, L.S.; Houghton, R.A.; Jackson, R.B.; McKinley, D.C.; Morrison, J.F.; Murray, B.C.; et al. A Synthesis of the Science on Forests and Carbon for U.S. Forests. In Issues in Ecology; Report 13; Ecological Society of America: Washington, DC, USA, 2010. [Google Scholar]
- McKinley, D.C.; Ryan, M.G.; Birdsey, R.A.; Giardina, C.P.; Harmon, M.E.; Heath, L.S.; Houghton, R.A.; Jackson, R.B.; Morrison, J.F.; Murray, B.C.; et al. A synthesis of current knowledge on forests and carbon storage in the United States. Ecol. Appl. 2010, 21, 1902–1924. Available online: https://www.fs.fed.us/rm/pubs_other/rmrs_2011_mckinley_d001.pdf (accessed on 3 October 2023). [CrossRef] [PubMed]
- Oliver, C.D.; Nassar, N.T.; Lippke, B.R.; McCarter, J.B. Carbon, Fossil Fuel, and Biodiversity Mitigation with Wood and Forests. J. Sustain. For. 2014, 33, 248–275. Available online: https://www.tandfonline.com/action/showCitFormats?doi=10.1080/10549811.2013.839386 (accessed on 8 December 2023). [CrossRef]
- U.S. Forest Stewardship Council. Towards Climate Smart Forestry. Increasing Carbon Storage in the Working Forests of Canada and the United States. 2023. Available online: https://us.fsc.org/download-box.879.htm (accessed on 4 December 2023).
- Grand View Research, Inc. Biochar Market Size, Share & Trend Analysis by Technology (Pyrolysis, Gasification, Others), By Application (Agriculture (Farming, Livestock) By Region & Segment Forecasts, 2012–2025. 2019. Available online: https://www.grandviewresearch.com/industry-analysis/biochar-market (accessed on 21 May 2018).
- Inkwood Research. Global Biochar Market Forecast 2022–2030. 2023. Available online: https://inkwoodresearch.com/reports/biochar-market/#:~:text=Report%20Summary,13.53%25%20during%20the%20forecast%20period. (accessed on 20 October 2023).
- Marland, G.; Marland, S. Should We Store Carbon in Trees? In Natural Sinks of CO2; Wisniewski, J., Lugo., A.E., Eds.; Springer: Dordrecht, The Netherlands, 1992; Available online: https://link.springer.com/content/pdf/10.1007/BF00477101.pdf (accessed on 16 January 2024).
- U.S. Government. Consolidated Appropriations Act, 2018. H.R. 1625. 2018. Available online: https://www.congress.gov/115/bills/hr1625/BILLS-115hr1625enr.pdf (accessed on 16 January 2024).
- Environmental Protection Agency. EPA’s Treatment of Biogenic Carbon Dioxide (CO2) Emissions from Stationary Sources That Use Forest Biomass for Energy Production. 2018. Available online: https://www.epa.gov/sites/default/files/2018-04/documents/biomass_policy_statement_2018_04_23.pdf (accessed on 16 January 2024).
- Pingoud, K.; Skog, K.; Martino, D.L.; Tonosaki, M.; Xiaoquan, Z.; Ford-Robertson, J. Harvested Wood Products. In IPCC Guidelines for National Greenhouse Gas Inventories; Intergovernmental Panel on Climate Change: Geneva, Switzerland, 2006; Volume 4, Chapter 12; 33p, Available online: https://www.ipcc-nggip.iges.or.jp/public/2006gl/pdf/4_Volume4/V4_12_Ch12_HWP.pdf (accessed on 16 January 2024).
- Chum, H.; Faaij, A.; Moreira, J.; Berndes, G.; Dhamija, P.; Dong, H.; Gabrielle, B.; Eng, A.G.; Lucht, W.; Mapako, M.; et al. Bioenergy. In IPCC Special Report on Renewable Energy Sources and Climate Change Mitigation; Edenhofer, O., Picks-Madruga, R., Sokona, Y., Seyboth, K., Matschoss, P., Kadner, S., Zwickel, T., Eickemeier, P., Hansen, G., Schlomer, S., et al., Eds.; Cambridge University Press: Cambridge, UK; New York, NY, USA, 2011; Available online: https://www.ipcc.ch/site/assets/uploads/2018/03/Chapter-2-Bioenergy-1.pdf (accessed on 16 January 2024).
- de Coninck, H.; Revi, A.; Babiker, M.; Bertoldi, P.; Buckeridge, M.; Cartwright, A.; Dong, W.; Ford, J.; Fuss, S.; Hourcade, J.-C.; et al. Strengthening and Implementing the Global Response. In Global Warming of 1.5 °C; An IPCC Special Report on the Impacts of Global Warming of 1.5 °C above Pre-Industrial Levels and Related Global Greenhouse Gas Emission Pathways, in the Context of Strengthening the Global Response to the Threat of Climate Change, Sustainable Development, and Efforts to Eradicate Poverty; Masson-Delmotte, V., Zhai, P., Portner, H.-O., Roberts, D., Skea, J., Shukla, P.R., Pirani, A., Moufouma-Okia, W., Pean, C., Pidcock, R., et al., Eds.; Cambridge University Press: Cambridge, UK; New York, NY, USA, 2018; pp. 313–444. [Google Scholar] [CrossRef]
- European Commission. Directive 2009/28/EC of the European Parliament and of the Council. on the Promotion of the Use of Energy from Renewable Sources and Amending and Subsequently Repealing Directives 2001/77/EC and 2003/30/EC. 2009. Available online: https://eur-lex.europaeu/legal-content/EN/TXT/PDF/?uri=CELEX:32009L0028&from=EN (accessed on 16 January 2024).
- European Commission. Directive of the European Parliament and of the Council Amending Directive (EU) 2018/2001 of the European Parliament and of the Council, Regulation (EU) 2018/1999 of the European Parliament and of the Council and Directive 98/70/EC of the European Parliament and of the Council as Regards the Promotion of Energy from Renewable Sources, and Repealing Council Directive (EU) 2015/652. 2021. Available online: https://ec.europa.eu/info/sites/default/files/amendment-renewable-energy-directive-2030-climate-target-with-annexes_en.pdf (accessed on 16 January 2024).
- Paris Agreement to the United Nations Framework Convention on Climate Change, Dec. 12, 2015, T.I.A.S. No. 16-1104. 2016 United Nations Framework Convention on Climate Change (UNFCCC), Denmark, 60p. Available online: https://unfccc.int/sites/default/files/resource/parisagreement_publication.pdf (accessed on 16 January 2024).
- Favero, A.; Daigneault, A.; Sohngen, B. Forests: Carbon sequestration, biomass energy, or both? Sci. Adv. 2020, 6, eaay6792. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rodriguez Franco, C.; Page-Dumroese, D.S.; Pierson, D.; Nicosia, T. Biochar Utilization as a Forestry Climate-Smart Tool. Sustainability 2024, 16, 1714. https://doi.org/10.3390/su16051714
Rodriguez Franco C, Page-Dumroese DS, Pierson D, Nicosia T. Biochar Utilization as a Forestry Climate-Smart Tool. Sustainability. 2024; 16(5):1714. https://doi.org/10.3390/su16051714
Chicago/Turabian StyleRodriguez Franco, Carlos, Deborah S. Page-Dumroese, Derek Pierson, and Timothy Nicosia. 2024. "Biochar Utilization as a Forestry Climate-Smart Tool" Sustainability 16, no. 5: 1714. https://doi.org/10.3390/su16051714
APA StyleRodriguez Franco, C., Page-Dumroese, D. S., Pierson, D., & Nicosia, T. (2024). Biochar Utilization as a Forestry Climate-Smart Tool. Sustainability, 16(5), 1714. https://doi.org/10.3390/su16051714





