Life Cycle Assessment of Carbon Capture by an Intelligent Vertical Plant Factory within an Industrial Park
Abstract
1. Introduction
2. Materials and Methods
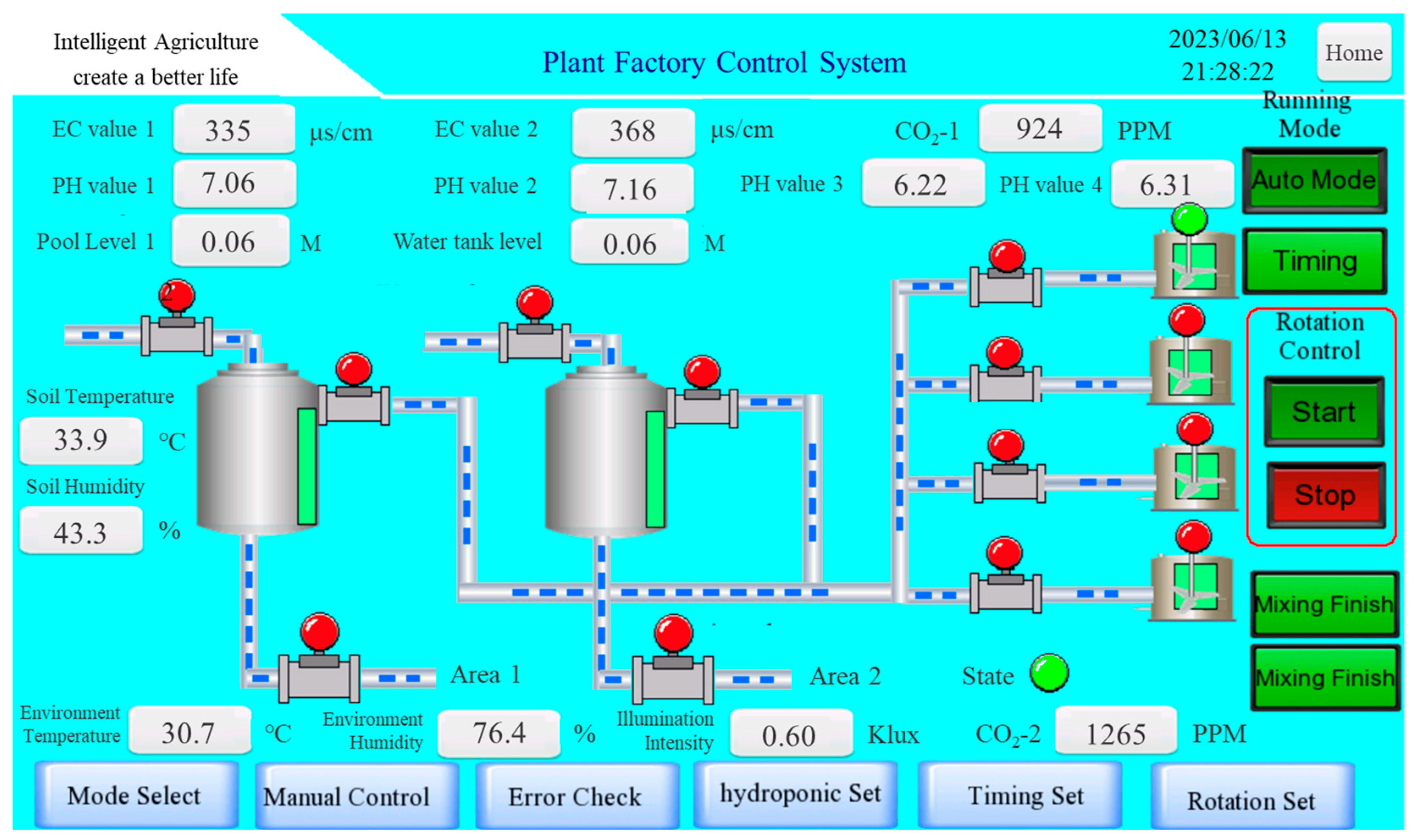
2.1. Design of an Intelligent Plant Factory
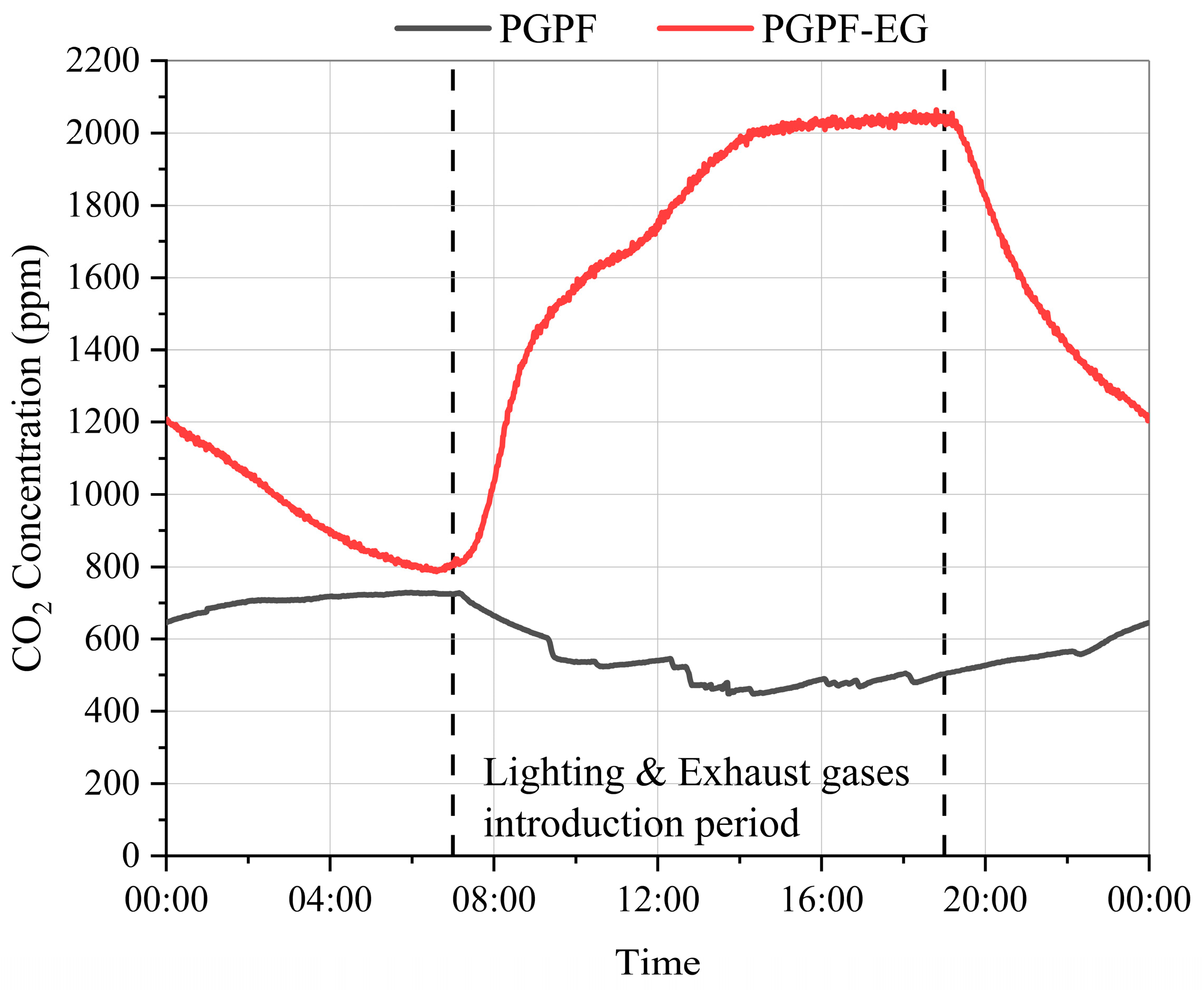
2.2. Establishment of Plant Factory with Exhaust Gases Introduction
2.3. Selection of Plants
2.4. Cultivation of Pennisetum giganteum in and out of Plant Factory
2.4.1. Planting Experiment and Measurement
2.4.2. Statistical Analysis of Planting Experiments
2.4.3. Experiments and Results for Outdoor Plantation
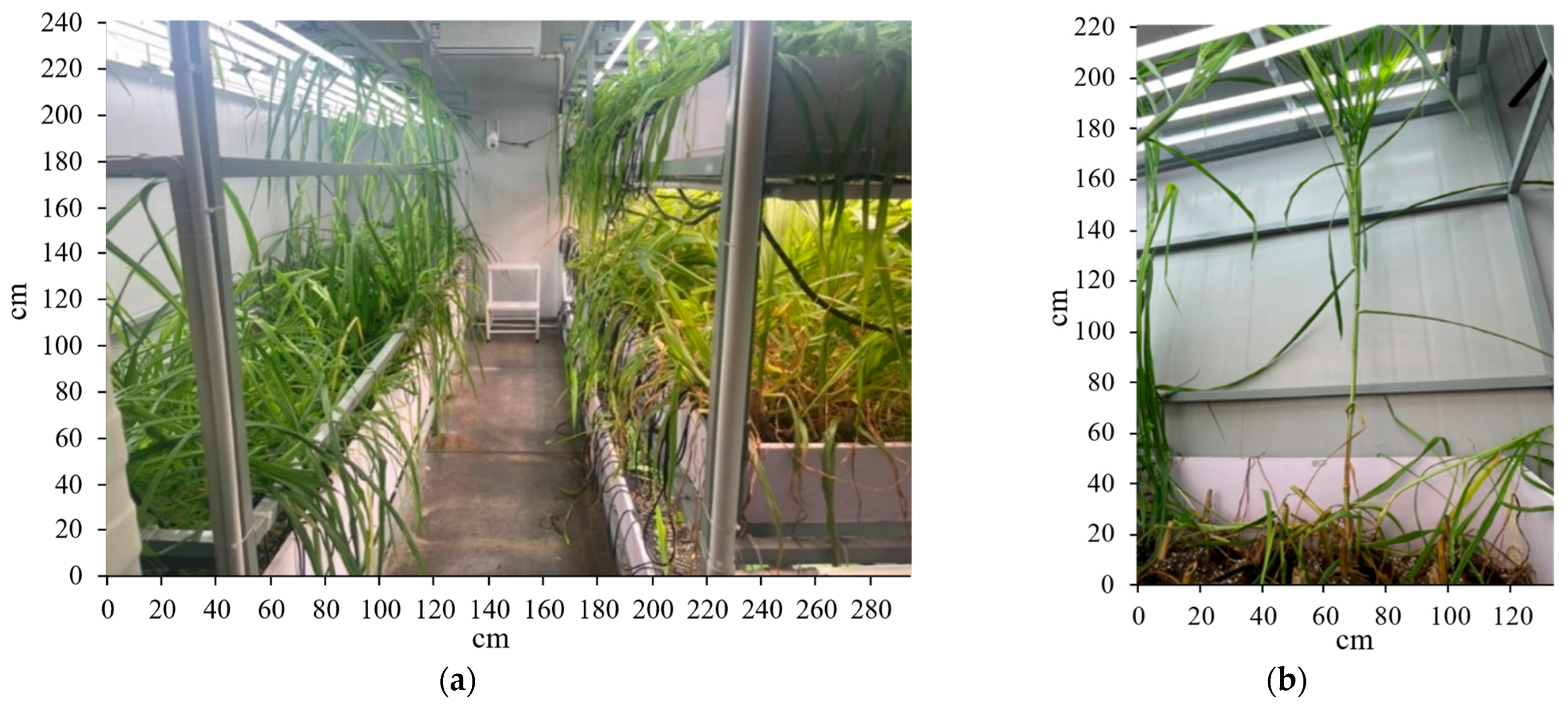
2.4.4. Experiments and Results for Plant Factory
2.5. Carbon Capture Capability of the Pennisetum giganteum Plant Factory
3. Life Cycle Assessment of the Pennisetum giganteum Plant Factory
3.1. Life Cycle Assessment Method
3.2. Goal and Scope Definition
3.2.1. Goal
3.2.2. System Boundary
3.2.3. Functional Unit
3.3. Life Cycle Inventory (LCI)
3.4. Life Cycle Impact Assessment (LCIA)
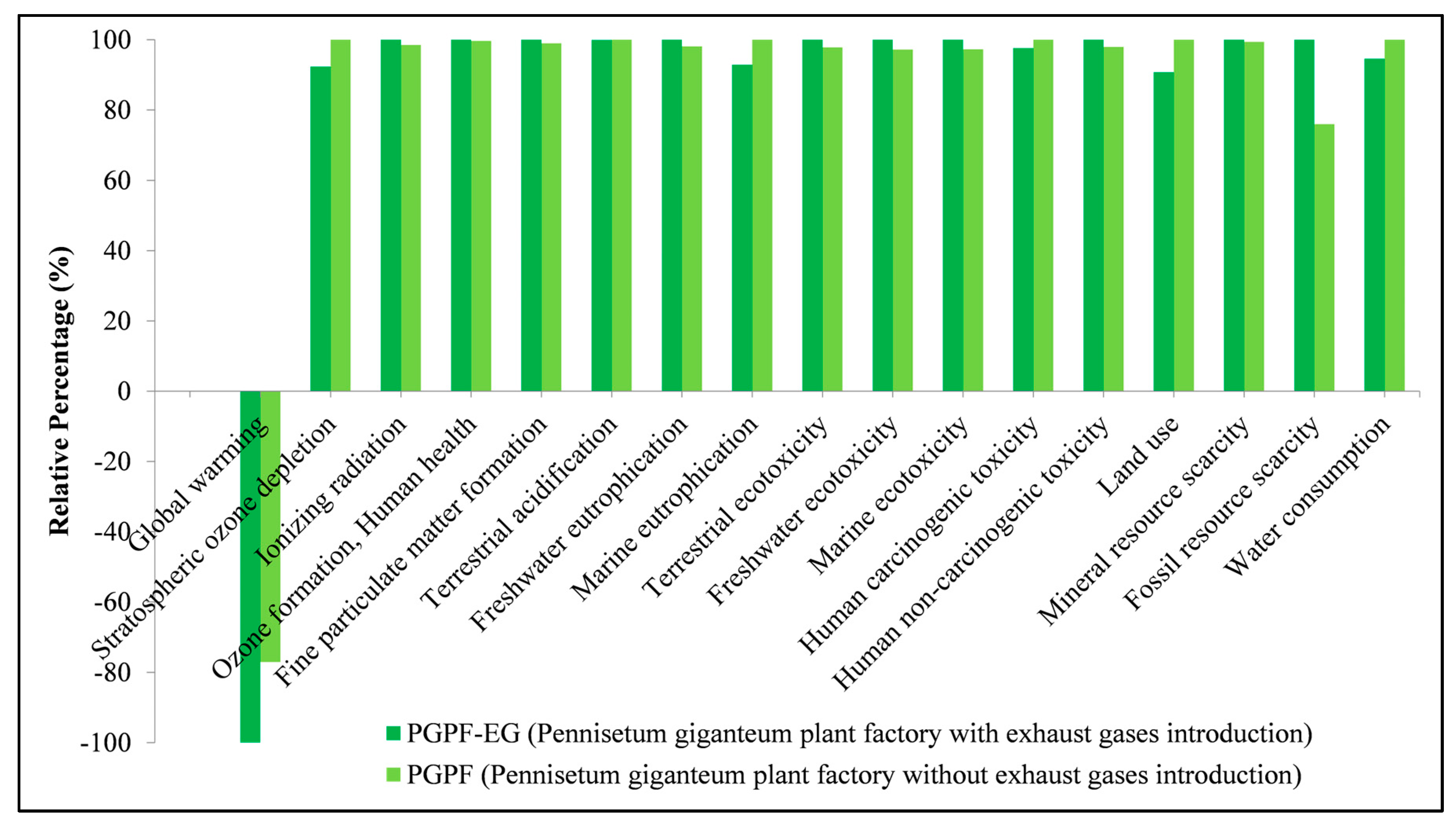
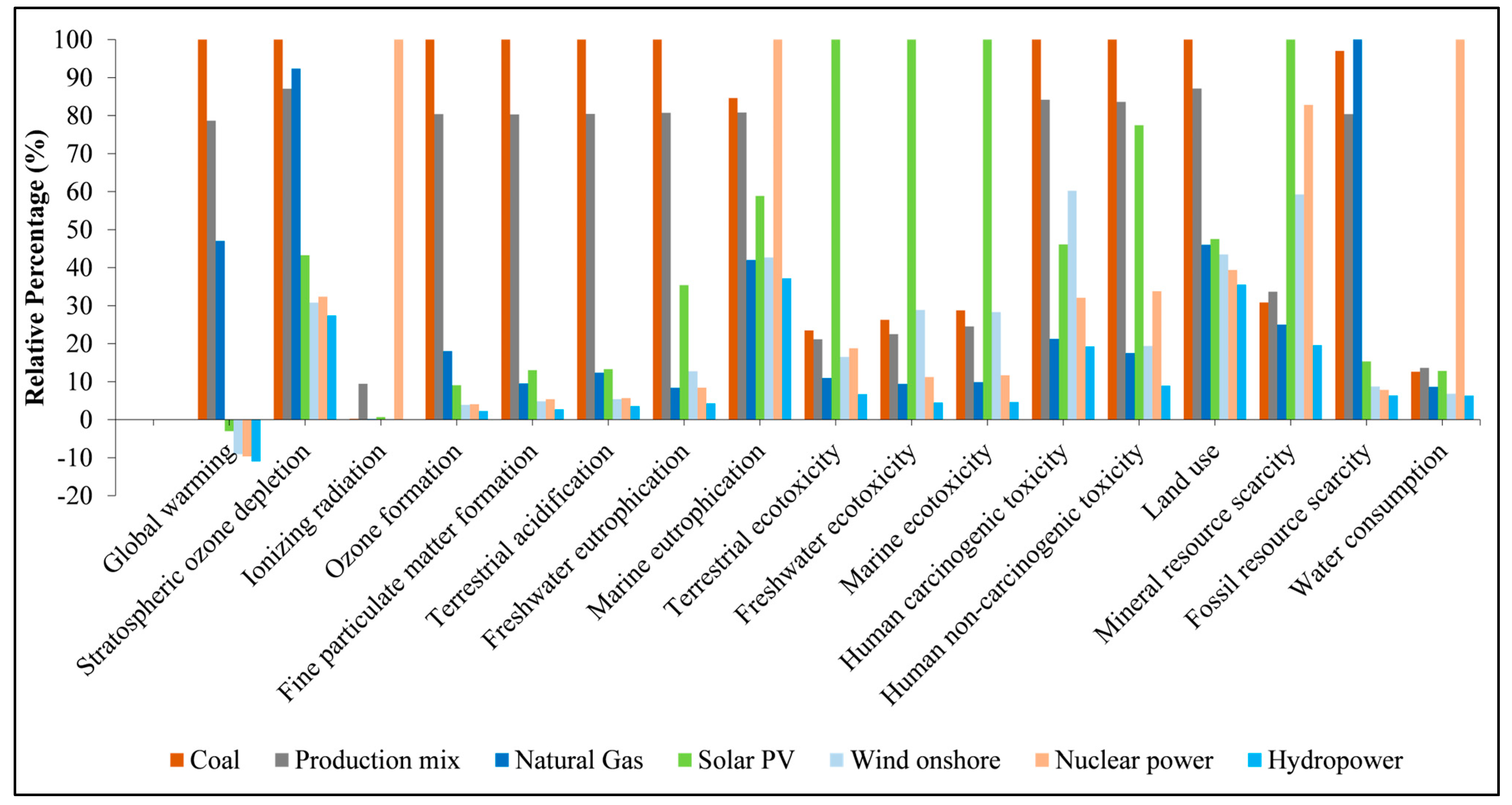
3.5. Interpretation of Results
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Appendix A
| Material | Quantity | Unit | Adapted Quantity | Functional Unit | Explanation |
|---|---|---|---|---|---|
| Stem segments | 0.22 | kg | 3.06 × 10−2 | kg/kg | Stem segments of Pennisetum giganteum |
| CO2 absorption | 10.8 | kg | 1.50 | kg/kg | Direct absorption from atmospheric environment |
| Fertilizer | 0.02 | kg | 2.78 × 10−3 | kg/kg | NPK (26-15-15) fertilizer |
| Irrigation | 0.51 | kg | 7.08 × 10−2 | kg/kg | The water used for irrigating the plants |
| Electricity consumption | 2.78 | kWh | 3.86 × 10−1 | kWh/kg | Powered by production mix electricity for operation including planting, pumping, and harvesting |
| Land-use change | 1 | m2 | 1.39 × 10−1 | m2/kg | Land-use change from industrial land to agricultural land for perennial crop |
| Transportation | 2.68 | tkm | 3.72 × 10−1 | tkm/kg | Transportation of Pennisetum giganteum to processing sites |
Appendix B
| Impact Category | Unit | Infrastructure | Stem Segments | Reclaimed Water | CO2 Absorption | Lighting | Other Processes | Fertilizer | Transport, Lorry | Total |
|---|---|---|---|---|---|---|---|---|---|---|
| Global warming | kg CO2 eq | 1.1 × 10−1 | 3.4 × 10−2 | 1.1 × 10−2 | −1.5 | 7.8 × 10−1 | 1.7 × 10−1 | 7.9 × 10−3 | 4.0 × 10−2 | −3.5 × 10−1 |
| Stratospheric ozone depletion | kg CFC11 eq | 3.7 × 10−8 | 5.4 × 10−7 | 9.7 × 10−10 | 1.2 × 10−8 | 3.6 × 10−7 | 7.8 × 10−8 | 9.4 × 10−8 | 2.6 × 10−8 | 1.1 × 10−6 |
| Ionizing radiation | kBq Co-60 eq | 4.0 × 10−3 | 8.5 × 10−4 | 6.8 × 10−5 | 1.8 × 10−3 | 5.6 × 10−2 | 1.2 × 10−2 | 6.6 × 10−5 | 7.1 × 10−4 | 7.6 × 10−2 |
| Ozone formation, human health | kg NOx eq | 2.8 × 10−4 | 1.3 × 10−4 | 1.2 × 10−5 | 8.5 × 10−5 | 2.2 × 10−3 | 4.8 × 10−4 | 1.2 × 10−5 | 1.1 × 10−4 | 3.3 × 10−3 |
| Fine particulate matter formation | kg PM2.5 eq | 2.0 × 10−4 | 1.5 × 10−4 | 6.3 × 10−6 | 8.6 × 10−5 | 2.0 × 10−3 | 4.4 × 10−4 | 8.8 × 10−6 | 4.4 × 10−5 | 2.9 × 10−3 |
| Terrestrial acidification | kg SO2 eq | 2.8 × 10−4 | 1.3 × 10−4 | 1.2 × 10−5 | 8.9 × 10−5 | 2.3 × 10−3 | 5.0 × 10−4 | 1.2 × 10−5 | 1.2 × 10−4 | 3.4 × 10−3 |
| Freshwater eutrophication | kg P eq | 4.3 × 10−4 | 9.6 × 10−4 | 1.4 × 10−5 | 2.2 × 10−4 | 4.4 × 10−3 | 9.7 × 10−4 | 2.5 × 10−5 | 1.0 × 10−4 | 7.1 × 10−3 |
| Marine eutrophication | kg N eq | 3.3 × 10−5 | 1.4 × 10−5 | 5.5 × 10−6 | 2.4 × 10−5 | 6.3 × 10−4 | 1.4 × 10−4 | 1.6 × 10−6 | 9.2 × 10−6 | 8.6 × 10−4 |
| Terrestrial ecotoxicity | kg 1,4-DCB | 2.5 × 10−6 | 1.0 × 10−4 | 5.4 × 10−6 | 2.9 × 10−6 | 5.6 × 10−5 | 1.2 × 10−5 | 1.8 × 10−7 | 3.6 × 10−7 | 1.8 × 10−4 |
| Freshwater ecotoxicity | kg 1,4-DCB | 8.0 × 10−1 | 7.0 × 10−2 | 6.0 × 10−3 | 9.2 × 10−1 | 29 | 6.3 | 2.2 × 10−2 | 4.5 × 10−1 | 37 |
| Marine ecotoxicity | kg 1,4-DCB | 3.7 × 10−3 | 9.5 × 10−4 | 1.0 × 10−4 | 1.3 × 10−2 | 3.8 × 10−1 | 8.4 × 10−2 | 2.2 × 10−4 | 1.2 × 10−3 | 4.9 × 10−1 |
| Human carcinogenic toxicity | kg 1,4-DCB | 5.1 × 10−3 | 1.2 × 10−3 | 1.4 × 10−4 | 1.6 × 10−2 | 4.9 × 10−1 | 1.1 × 10−1 | 2.9 × 10−4 | 1.9 × 10−3 | 6.2 × 10−1 |
| Human non-carcinogenic toxicity | kg 1,4-DCB | 5.3 × 10−2 | 1.5 × 10−3 | 1.1 × 10−3 | 3.7 × 10−3 | 1.1 × 10−1 | 2.5 × 10−2 | 2.5 × 10−4 | 2.5 × 10−3 | 2.0 × 10−1 |
| Land use | m2a crop eq | 7.2 × 10−2 | 2.4 × 10−1 | 1.0 × 10−2 | 1.4 × 10−1 | 3.4 | 7.6 × 10−1 | 4.6 × 10−3 | 3.7 × 10−2 | 4.7 |
| Mineral resource scarcity | kg Cu eq | 1.7 × 10−3 | 7.4 × 10−2 | 9.7 × 10−5 | 7.9 × 10−4 | 2.5 × 10−2 | 5.5 × 10−3 | 1.2 × 10−4 | 1.5 × 10−3 | 1.1 × 10−1 |
| Fossil resource scarcity | kg oil eq | 2.1 × 10−3 | 8.7 × 10−5 | 5.9 × 10−6 | 4.3 × 10−4 | 1.3 × 10−2 | 3.0 × 10−3 | 4.5 × 10−5 | 1.2 × 10−4 | 1.9 × 10−2 |
| Water consumption | m3 | 3.3 × 10−2 | 5.0 × 10−3 | 8.9 × 10−4 | 1.0 × 10−1 | 2.0 × 10−1 | 4.5 × 10−2 | 1.1 × 10−3 | 1.3 × 10−2 | 4.1 × 10−1 |
References
- Rogelj, J.; den Elzen, M.; Höhne, N.; Fransen, T.; Fekete, H.; Winkler, H.; Schaeffer, R.; Sha, F.; Riahi, K.; Meinshausen, M. Paris Agreement Climate Proposals Need a Boost to Keep Warming Well below 2 °C. Nature 2016, 534, 631–639. [Google Scholar] [CrossRef]
- Rissman, J.; Bataille, C.; Masanet, E.; Aden, N.; Morrow, W.R.; Zhou, N.; Elliott, N.; Dell, R.; Heeren, N.; Huckestein, B.; et al. Technologies and Policies to Decarbonize Global Industry: Review and Assessment of Mitigation Drivers through 2070. Appl. Energy 2020, 266, 114848. [Google Scholar] [CrossRef]
- Sifat, N.S.; Haseli, Y. A Critical Review of CO2 Capture Technologies and Prospects for Clean Power Generation. Energies 2019, 12, 4143. [Google Scholar] [CrossRef]
- Daneshvar, E.; Wicker, R.J.; Show, P.-L.; Bhatnagar, A. Biologically-Mediated Carbon Capture and Utilization by Microalgae towards Sustainable CO2 Biofixation and Biomass Valorization—A Review. Chem. Eng. J. 2022, 427, 130884. [Google Scholar] [CrossRef]
- Wang, X.; Song, C. Carbon Capture from Flue Gas and the Atmosphere: A Perspective. Front. Energy Res. 2020, 8, 560849. [Google Scholar] [CrossRef]
- Nocito, F.; Dibenedetto, A. Atmospheric CO2 Mitigation Technologies: Carbon Capture Utilization and Storage. Curr. Opin. Green Sustain. Chem. 2020, 21, 34–43. [Google Scholar] [CrossRef]
- Singh, J.; Dhar, D.W. Overview of Carbon Capture Technology: Microalgal Biorefinery Concept and State-of-the-Art. Front. Mar. Sci. 2019, 6, 417505. [Google Scholar] [CrossRef]
- Patel, H.A.; Byun, J.; Yavuz, C.T. Carbon Dioxide Capture Adsorbents: Chemistry and Methods. ChemSusChem 2017, 10, 1303–1317. [Google Scholar] [CrossRef]
- Lai, J.Y.; Ngu, L.H.; Hashim, S.S. A Review of CO2 Adsorbents Performance for Different Carbon Capture Technology Processes Conditions. Greenh. Gases Sci. Technol. 2021, 11, 1076–1117. [Google Scholar] [CrossRef]
- Khalilpour, R.; Mumford, K.; Zhai, H.; Abbas, A.; Stevens, G.; Rubin, E.S. Membrane-Based Carbon Capture from Flue Gas: A Review. J. Clean. Prod. 2015, 103, 286–300. [Google Scholar] [CrossRef]
- De Silva, G.; Ranjith, P.G.; Perera, M. Geochemical Aspects of CO2 Sequestration in Deep Saline Aquifers: A Review. Fuel 2015, 155, 128–143. [Google Scholar] [CrossRef]
- Song, J.; Zhang, D. Comprehensive Review of Caprock-Sealing Mechanisms for Geologic Carbon Sequestration. Environ. Sci. Technol. 2013, 47, 9–22. [Google Scholar] [CrossRef] [PubMed]
- Aminu, M.D.; Nabavi, S.A.; Rochelle, C.A.; Manovic, V. A Review of Developments in Carbon Dioxide Storage. Appl. Energy 2017, 208, 1389–1419. [Google Scholar] [CrossRef]
- Zhou, W.; Wang, J.; Chen, P.; Ji, C.; Kang, Q.; Lu, B.; Li, K.; Liu, J.; Ruan, R. Bio-Mitigation of Carbon Dioxide Using Microalgal Systems: Advances and Perspectives. Renew. Sustain. Energy Rev. 2017, 76, 1163–1175. [Google Scholar] [CrossRef]
- Peng, Y.; Thomas, S.C.; Tian, D. Forest Management and Soil Respiration: Implications for Carbon Sequestration. Environ. Rev. 2008, 16, 93–111. [Google Scholar] [CrossRef]
- Nayak, N.; Mehrotra, R.; Mehrotra, S. Carbon Biosequestration Strategies: A Review. Carbon Capture Sci. Technol. 2022, 4, 100065. [Google Scholar] [CrossRef]
- Fernández, F.G.A.; Reis, A.; Wijffels, R.H.; Barbosa, M.; Verdelho, V.; Llamas, B. The Role of Microalgae in the Bioeconomy. New Biotechnol. 2021, 61, 99–107. [Google Scholar] [CrossRef]
- Agostini, F.; Gregory, A.S.; Richter, G.M. Carbon Sequestration by Perennial Energy Crops: Is the Jury Still Out? Bioenergy Res. 2015, 8, 1057–1080. [Google Scholar] [CrossRef]
- Lemus, R.; Lal, R. Bioenergy Crops and Carbon Sequestration. Crit. Rev. Plant Sci. 2005, 24, 1–21. [Google Scholar] [CrossRef]
- Stewart, C.; Hessami, M.-A. A Study of Methods of Carbon Dioxide Capture and Sequestration––the Sustainability of a Photosynthetic Bioreactor Approach. Energy Convers. Manag. 2005, 46, 403–420. [Google Scholar] [CrossRef]
- Song, J.; Yang, W.; Higano, Y.; Wang, X. Modeling the Development and Utilization of Bioenergy and Exploring the Environmental Economic Benefits. Energy Convers. Manag. 2015, 103, 836–846. [Google Scholar] [CrossRef]
- Ramaraj, R.; Tsai, D.D.-W.; Chen, P.H. Carbon Dioxide Fixation of Freshwater Microalgae Growth on Natural Water Medium. Ecol. Eng. 2015, 75, 86–92. [Google Scholar] [CrossRef]
- Judd, S.; van den Broeke, L.J.P.; Shurair, M.; Kuti, Y.; Znad, H. Algal Remediation of CO2 and Nutrient Discharges: A Review. Water Res. 2015, 87, 356–366. [Google Scholar] [CrossRef] [PubMed]
- Wei, C.; Xu, Y.; Xu, L.; Liu, J.; Chen, H. Comparative Life-Cycle Assessment of Various Harvesting Strategies for Biogas Production from Microalgae: Energy Conversion Characteristics and Greenhouse Gas Emissions. Energy Convers. Manag. 2023, 289, 117188. [Google Scholar] [CrossRef]
- Bahadar, A.; Bilal Khan, M. Progress in Energy from Microalgae: A Review. Renew. Sustain. Energy Rev. 2013, 27, 128–148. [Google Scholar] [CrossRef]
- Kozai, T.; Niu, G. Chapter 2—Role of the Plant Factory with Artificial Lighting (PFAL) in Urban Areas. In Plant Factory, 2nd ed.; Kozai, T., Niu, G., Takagaki, M., Eds.; Academic Press: Cambridge, MA, USA, 2020; pp. 7–34. ISBN 978-0-12-816691-8. [Google Scholar]
- Hunt, R. Basic Growth Analysis: Plant Growth Analysis for Beginners; Springer Science & Business Media: Berlin/Heidelberg, Germany, 2012; ISBN 94-010-9117-X. [Google Scholar]
- Gan, P.; Liu, F.; Li, R.; Wang, S.; Luo, J. Chloroplasts—Beyond Energy Capture and Carbon Fixation: Tuning of Photosynthesis in Response to Chilling Stress. Int. J. Mol. Sci. 2019, 20, 5046. [Google Scholar] [CrossRef] [PubMed]
- Martin, M.; Poulikidou, S.; Molin, E. Exploring the Environmental Performance of Urban Symbiosis for Vertical Hydroponic Farming. Sustainability 2019, 11, 6724. [Google Scholar] [CrossRef]
- Pesch, H.; Louw, L. Exploring the Industrial Symbiosis Potential of Plant Factories during the Initial Establishment Phase. Sustainability 2023, 15, 1240. [Google Scholar] [CrossRef]
- Liang, L.; Tian, H.; Ning, P. Artificial Light LED Planting System Design. In Proceedings of the 2017 14th China International Forum on Solid State Lighting: International Forum on Wide Bandgap Semiconductors China (SSLChina: IFWS), Beijing, China, 1–3 November 2017; pp. 88–90. [Google Scholar]
- Zhang, R.; Liu, T.; Ma, J. Plant Factory: A New Method for Reducing Carbon Emissions. AIP Conf. Proc. 2017, 1820, 040016. [Google Scholar] [CrossRef]
- Chowdhury, M.; Kiraga, S.; Islam, M.N.; Ali, M.; Reza, M.N.; Lee, W.-H.; Chung, S.-O. Effects of Temperature, Relative Humidity, and Carbon Dioxide Concentration on Growth and Glucosinolate Content of Kale Grown in a Plant Factory. Foods 2021, 10, 1524. [Google Scholar] [CrossRef]
- Chen, D.; Mei, Y.; Liu, Q.; Wu, Y.; Yang, Z. Carbon Dioxide Enrichment Promoted the Growth, Yield, and Light-Use Efficiency of Lettuce in a Plant Factory with Artificial Lighting. Agron. J. 2021, 113, 5196–5206. [Google Scholar] [CrossRef]
- Tian, Z.; Ma, W.; Yang, Q.; Duan, F. Research Status and Prospect of Energy-Saving Technology of LED Light Source in Plant Factory. In Proceedings of the 2020 17th China International Forum on Solid State Lighting & 2020 International Forum on Wide Bandgap Semiconductors China (SSLChina: IFWS), Shenzhen, China, 23–25 November 2020; pp. 152–158. [Google Scholar]
- Kikuchi, Y.; Kanematsu, Y.; Yoshikawa, N.; Okubo, T.; Takagaki, M. Environmental and Resource Use Analysis of Plant Factories with Energy Technology Options: A Case Study in Japan. J. Clean. Prod. 2018, 186, 703–717. [Google Scholar] [CrossRef]
- Santiteerakul, S.; Sopadang, A.; Yaibuathet Tippayawong, K.; Tamvimol, K. The Role of Smart Technology in Sustainable Agriculture: A Case Study of Wangree Plant Factory. Sustainability 2020, 12, 4640. [Google Scholar] [CrossRef]
- Kaur, G.; Upadhyaya, P.; Chawla, P. Comparative Analysis of IoT-Based Controlled Environment and Uncontrolled Environment Plant Growth Monitoring System for Hydroponic Indoor Vertical Farm. Environ. Res. 2023, 222, 115313. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.; Xiang, C.; Xu, L.; Cui, J.; Fu, S.; Chen, B.; Yang, S.; Wang, P.; Xie, Y.; Wei, M.; et al. SMRT Sequencing of a Full-Length Transcriptome Reveals Transcript Variants Involved in C18 Unsaturated Fatty Acid Biosynthesis and Metabolism Pathways at Chilling Temperature in Pennisetum Giganteum. BMC Genom. 2020, 21, 52. [Google Scholar] [CrossRef]
- Tong, X.Y.; Zheng, S.H.; Ye, D.P.; Xie, J.J.; Zhu, Z. The Research Progress and Prospect of Pennisetum Sp.(Giant Juncao) Planting Working Mechanism. Appl. Mech. Mater. 2014, 641, 1093–1098. [Google Scholar] [CrossRef]
- Qiu, Y.; Lei, Y.; Zhao, H.; He, X.; Liu, B.; Huang, Y. Mesophilic Anaerobic Digestion of Arundo Donax Cv. Lvzhou No. 1 and Pennisetum Giganteum for Biogas Production: Structure and Functional Analysis of Microbial Communities. BioEnergy Res. 2023, 16, 1205–1216. [Google Scholar] [CrossRef]
- Ye, L.; Zhang, J.; Xu, R.; Yu, J.; Cao, M.; Yu, Y.; Liu, S. Gasification of Organic Solid Waste for Syngas: Physicochemical and Conversion Mechanism Investigation. Int. J. Hydrog. Energy 2024, 49, 384–397. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhang, H.; Lee, D.-J.; Zhang, T.; Jiang, D.; Zhang, Z.; Zhang, Q. Effect of Enzymolysis Time on Biohydrogen Production from Photo-Fermentation by Using Various Energy Grasses as Substrates. Bioresour. Technol. 2020, 305, 123062. [Google Scholar] [CrossRef]
- Boone, L.; Van Linden, V.; De Meester, S.; Vandecasteele, B.; Muylle, H.; Roldán-Ruiz, I.; Nemecek, T.; Dewulf, J. Environmental Life Cycle Assessment of Grain Maize Production: An Analysis of Factors Causing Variability. Sci. Total Environ. 2016, 553, 551–564. [Google Scholar] [CrossRef]
- Parajuli, R.; Knudsen, M.T.; Djomo, S.N.; Corona, A.; Birkved, M.; Dalgaard, T. Environmental Life Cycle Assessment of Producing Willow, Alfalfa and Straw from Spring Barley as Feedstocks for Bioenergy or Biorefinery Systems. Sci. Total Environ. 2017, 586, 226–240. [Google Scholar] [CrossRef]
- Kazlauskas, M.; Bručienė, I.; Savickas, D.; Naujokienė, V.; Buragienė, S.; Steponavičius, D.; Romaneckas, K.; Šarauskis, E. Life Cycle Assessment of Winter Wheat Production Using Precision and Conventional Seeding Technologies. Sustainability 2023, 15, 14376. [Google Scholar] [CrossRef]
- Martin, M.; Molin, E. Environmental Assessment of an Urban Vertical Hydroponic Farming System in Sweden. Sustainability 2019, 11, 4124. [Google Scholar] [CrossRef]
- Hallikainen, E. Life Cycle Assessment on Vertical Farming. Master’s Thesis, Aalto University, Otaniemi, Finland, 2019. [Google Scholar]
- Verma, D.; Senal, I. 6—Natural Fiber-Reinforced Polymer Composites: Feasibiliy Study for Sustainable Automotive Industries. In Biomass, Biopolymer-Based Materials, and Bioenergy; Verma, D., Fortunati, E., Jain, S., Zhang, X., Eds.; Woodhead Publishing: Cambridge, UK, 2019; pp. 103–122. ISBN 978-0-08-102426-3. [Google Scholar]
- Ding, J.; Shi, G.; Zhao, X.; Zhao, Q. Planting Techniques of Pennisetum Giganteum in Huanghuai Area. Agric. Biotechnol. 2021, 10, 42–58. [Google Scholar]
- Tan, F.; He, L.; Zhu, Q.; Wang, Y.; Chen, C.; He, M. Pennisetum Hydridum: A Potential Energy Crop with Multiple Functions and the Current Status in China. BioEnergy Res. 2022, 15, 850–862. [Google Scholar] [CrossRef]
- Zhu, S.; Zhang, Q.; Yang, R.; Chen, B.; Zhang, B.; Yang, Z.; Chen, X.; Wang, X.; Du, M.; Tang, L. Typical JUNCAO Overwintering Performance and Optimized Cultivation Conditions of Pennisetum Sp. in Guizhou, Southwest China. Sustainability 2022, 14, 4086. [Google Scholar] [CrossRef]
- Zakikhani, P.; Zahari, R.; Sultan, M.T.H.; Majid, D.L. Extraction and Preparation of Bamboo Fibre-Reinforced Composites. Mater. Des. 2014, 63, 820–828. [Google Scholar] [CrossRef]
- Kuluev, B.; Minchenkov, N.; Gumerova, G. Russian Dandelion (Taraxacum Kok-Saghyz Rodin): Rubber Extraction Methods and Prospects for Biotechnological Methods Application. Plant Biotechnol. Breed. 2019, 2, 33–43. [Google Scholar] [CrossRef]
- Chakraborty, I.; Chatterjee, K. Polymers and Composites Derived from Castor Oil as Sustainable Materials and Degradable Biomaterials: Current Status and Emerging Trends. Biomacromolecules 2020, 21, 4639–4662. [Google Scholar] [CrossRef]
- Vandelook, S.; Elsacker, E.; Van Wylick, A.; De Laet, L.; Peeters, E. Current State and Future Prospects of Pure Mycelium Materials. Fungal Biol. Biotechnol. 2021, 8, 20. [Google Scholar] [CrossRef]
- Raman, J.; Kim, D.-S.; Kim, H.-S.; Oh, D.-S.; Shin, H.-J. Mycofabrication of Mycelium-Based Leather from Brown-Rot Fungi. J. Fungi 2022, 8, 317. [Google Scholar] [CrossRef] [PubMed]
- Deshmukh, G.S. Advancement in Hemp Fibre Polymer Composites: A Comprehensive Review. J. Polym. Eng. 2022, 42, 575–598. [Google Scholar] [CrossRef]
- Asim, M.; Abdan, K.; Jawaid, M.; Nasir, M.; Dashtizadeh, Z.; Ishak, M.R.; Hoque, M.E. A Review on Pineapple Leaves Fibre and Its Composites. Int. J. Polym. Sci. 2015, 2015, 950567. [Google Scholar] [CrossRef]
- Suárez, L.; Ortega, Z.; Barczewski, M.; Cunningham, E. Use of Giant Reed (Arundo donax L.) for Polymer Composites Obtaining: A Mapping Review. Cellulose 2023, 30, 4793–4812. [Google Scholar] [CrossRef]
- Wjunow, C.; Moselewski, K.-L.; Huhnen, Z.; Sultanova, S.; Sabantina, L. Sustainable Textiles from Unconventional Biomaterials—Cactus Based. Eng. Proc. 2023, 37, 58. [Google Scholar] [CrossRef]
- ISO 14040; Environmental Management—Life Cycle Assessment—Principles and Framework. International Organization for Standardization: Basel, Switzerland, 1997.
- Jeroen, B. Guinee Handbook on Life Cycle Assessment Operational Guide to the ISO Standards. Int. J. Life Cycle Assess. 2002, 7, 311–313. [Google Scholar] [CrossRef]
- Parajuli, R.; Kristensen, I.S.; Knudsen, M.T.; Mogensen, L.; Corona, A.; Birkved, M.; Peña, N.; Graversgaard, M.; Dalgaard, T. Environmental Life Cycle Assessments of Producing Maize, Grass-Clover, Ryegrass and Winter Wheat Straw for Biorefinery. J. Clean. Prod. 2017, 142, 3859–3871. [Google Scholar] [CrossRef]
- Consequential LCA. The Functional Unit 2022. Available online: https://consequential-lca.org/clca/the-functional-unit/ (accessed on 9 October 2023).
- Wildeman, R. Vertical Farming: A Future Perspective or a Mere Conceptual Idea? A Comprehensive Life Cycle Analysis on the Environmental Impact of a Vertical Farm Compared to Rural Agriculture in the US. Bachelor’s Thesis, University of Twente, Twente, The Netherlands, 2020. [Google Scholar]
- Graamans, L.; Baeza, E.; van den Dobbelsteen, A.; Tsafaras, I.; Stanghellini, C. Plant Factories versus Greenhouses: Comparison of Resource Use Efficiency. Agric. Syst. 2018, 160, 31–43. [Google Scholar] [CrossRef]
- Huijbregts, M.A.J.; Steinmann, Z.J.N.; Elshout, P.M.F.; Stam, G.; Verones, F.; Vieira, M.; Zijp, M.; Hollander, A.; van Zelm, R. ReCiPe2016: A Harmonised Life Cycle Impact Assessment Method at Midpoint and Endpoint Level. Int. J. Life Cycle Assess. 2017, 22, 138–147. [Google Scholar] [CrossRef]











| Technology | Advantages | Disadvantages | Reference | |
|---|---|---|---|---|
| Absorption | Physical absorption | High capacity at low temperature and high pressure Cheaper solvent | Low capacity at high temperature and low pressure High energy consumption | [3,5] |
| Chemical absorption | High capacity at low CO2 pressure Thermally stable Mature technology | High solvent loss due to evaporation Absorbent degradation High operating cost | [5,7] | |
| Adsorption | Chemical adsorbents | Work at high temperature High capacity | Negative effect of moisture High energy consumption | [5,8] |
| Physical adsorbents | High capacity at low temperature and high pressure Low waste generation | Low CO2 selectivity Capacity decreases with temperature Normally require high pressure | [5,9] | |
| Membrane technology | High separation efficiency Low waste generation Relatively low operation cost | High manufacturing cost Relatively low separation selectivity Negative effect of moisture | [6,10] | |
| CO2 storage | Geological sequestration | Huge storage capacity Replenish depleted oil/gas reserves | High operational cost Risk of CO2 leakage and environmental contamination Specific geomorphic structure requirement | [11,12] |
| Oceanic injection | Huge CO2 storage capacity | Cost intensive Potential threat to marine life | [13,14] | |
| Biological absorption | Forestation | Huge CO2 storage capacity No hazards of chemicals | Long time requirement Large area requirement May affect biological diversity | [15,16] |
| Microalgae | Highly efficient Faster growth rate than plants | Large facility requirement Sensitive to other flue gas components and contamination | [7,17] | |
| Energy crop | Low cost input Combined with industrial production Co-production of feed, biofuel, and value-added products | Low carbon sequestration efficiency Low land utilization rate Immaturity of crop energy utilization | [18,19] | |
| Instrument | Measurement Range | Resolution | Accuracy |
|---|---|---|---|
| Air Temperature Sensor | −20~50 °C | 0.1 °C | ±0.5 °C |
| Air Humidity Sensor | 0~100% RH | 0.1% RH | ±0.5% (at 60% RH, 25 °C) |
| CO2 Concentration Sensor | 0–5000 ppm | 1 ppm | ±(50 ppm + 3% F·S) |
| Light Intensity Sensor | 0.1–10 klux | 0.01 klux | ±4% (at 25 °C) |
| Soil Temperature Sensor | −40~80 °C | 0.1 °C | ±0.5 °C |
| Soil Humidity Sensor | 0~100% RH | 0.1% RH | ±3% (at 60% RH, 25 °C) |
| Soil pH Sensor | 3~9 | 0.01 | ±0.5 |
| Electrical Conductivity Sensor | 0–2000 μs/cm | 1 μs/cm | ±5% |
| Liquid Level Sensor | — | 1 cm | — |
| Water Meter | — | 1 L | — |
| Electricity Meter | — | 0.1 kWh | — |
| Electronic Scale | 0–50 kg | 1 g | ±10 g |
| Items | Emission Concentration (mg/m3) | Emission Limit (mg/m3) |
|---|---|---|
| Particulate Matter | 1.5 | ≤30 |
| 1,3,5-Trimethylbenzene | <0.01 | - |
| 1,2,4-Trimethylbenzene | <0.01 | - |
| Non-Methane Total Hydrocarbons (as Carbon) | 2.9 | ≤60 |
| Toluene | <0.004 | - |
| Xylene | 0.014 | - |
| Ethyl Acetate | <0.005 | - |
| CO2 | 2034 ppm |
| Plant | Carbon Sequestration Capacity | Temp Range (°C) | Min Harvest Height (m) | Growth Cycle (month) | Harvest Frequency (time/year) | Regeneration Capacity | Applied Organ | Product | Industrial Application |
|---|---|---|---|---|---|---|---|---|---|
| Pennisetum giganteum [51,52] | C4 plant | 10–35 | 0.5 | 1–3 | 3–4 | Perennial | Stem leaf | Cellulosic ethanol/polymer composite | Processed for cellulose ethanol or used in the manufacturing of composite materials. |
| Bamboo [53] | C3 plant with high carbon sequestration efficiency | 8–36 | 2 | >12 | 1 | Perennial | Stem | Bamboo fiber composite | Extracted from culms using mechanical or chemical methods for composite production. |
| Russian dandelion [54] | C3 plant | 15–35 | 0.2 | 6 | 1 | Perennial | Root | Rubber | Produce a milky fluid in roots, containing a high-quality rubber. |
| Castor oil plant [55] | C3 plant | 14–36 | 1.5 | 6 | 1 | Perennial | Seed | Polymer composite | Castor oil can be a source for polymers such as polyurethanes, polyesters, polyamides, and epoxy-polymers. |
| Fungi [56,57] | - | 5–35 | 0.2 | 1 | 8–12 | Annual | Mycelium | Polymer composite or mycelium-based leather | Transform into a resilient natural composite with controlled properties through chemical and heat treatments. |
| Hemp [58] | C3 plant | 19–25 | 1 | 3–4 | 1 | Annual | Stem | Hemp fiber composite | Wide-ranging applications in the automotive, electrical, construction, and packaging sectors. |
| Pineapple Tree [59] | C3 plant | 20–30 | 0.6 | 18–24 | 1 | Perennial | Leaf | Polymer composite | Pineapple leaf fiber can be applied in the making of reinforced polymer composites. |
| Giant Reed [60] | C3 plant | 10–35 | 1 | >12 | 1 | Perennial | Stem leaf | Polymer composite | Obtained through mechanical processes and used for composite materials production. |
| Cactus [61] | C3 plant | 20–30 | 1 | >12 | 1 | Perennial | Leaf | Cactus-based leather | Employed in the manufacturing of various products including car interiors. |
| Group | Planting Environment | Planting Duration | Light Condition | Nutrient Solution | Height (cm) | Yield (kg/m2) |
|---|---|---|---|---|---|---|
| O-1 | Open Field | 3 months | Sunlight | Compound Fertilizer | 76 ± 7.1 a | 8.3 |
| O-2 | Planters | 3 months | Sunlight | None | 65 ± 3.1 b | 7.2 |
| O-3 | Planters | 3 months | Sunlight | Urea Fertilizer | 74 ± 4.8 a | 8.3 |
| O-4 | Planters | 3 months | Sunlight | Compound Fertilizer | 72 ± 4.9 a | 8.2 |
| O-5 | Planters | 3 months | Semi-shading sunlight | Compound Fertilizer | 42 ± 5.9 c | 4.7 |
| Group | Planting Duration | Irrigation Frequency | Fertilizing Rules | Height (cm) | Yield (kg/m2) |
|---|---|---|---|---|---|
| A | 3 months | 2 min/d | After growing 20 days, every 8 d | 88 ± 7.4 c | 9.6 |
| B | 3 months | 2 min/d | After growing 15 days, every 8 d | 94 ± 3.9 b | 9.9 |
| C | 3 months | 4 min/d | After growing 15 days, every 8 d | 95 ± 5.4 b | 10.1 |
| D | 3 months | 4 min/d | After growing 15 days, every 5 d | 114 ± 6.4 a | 11.6 |
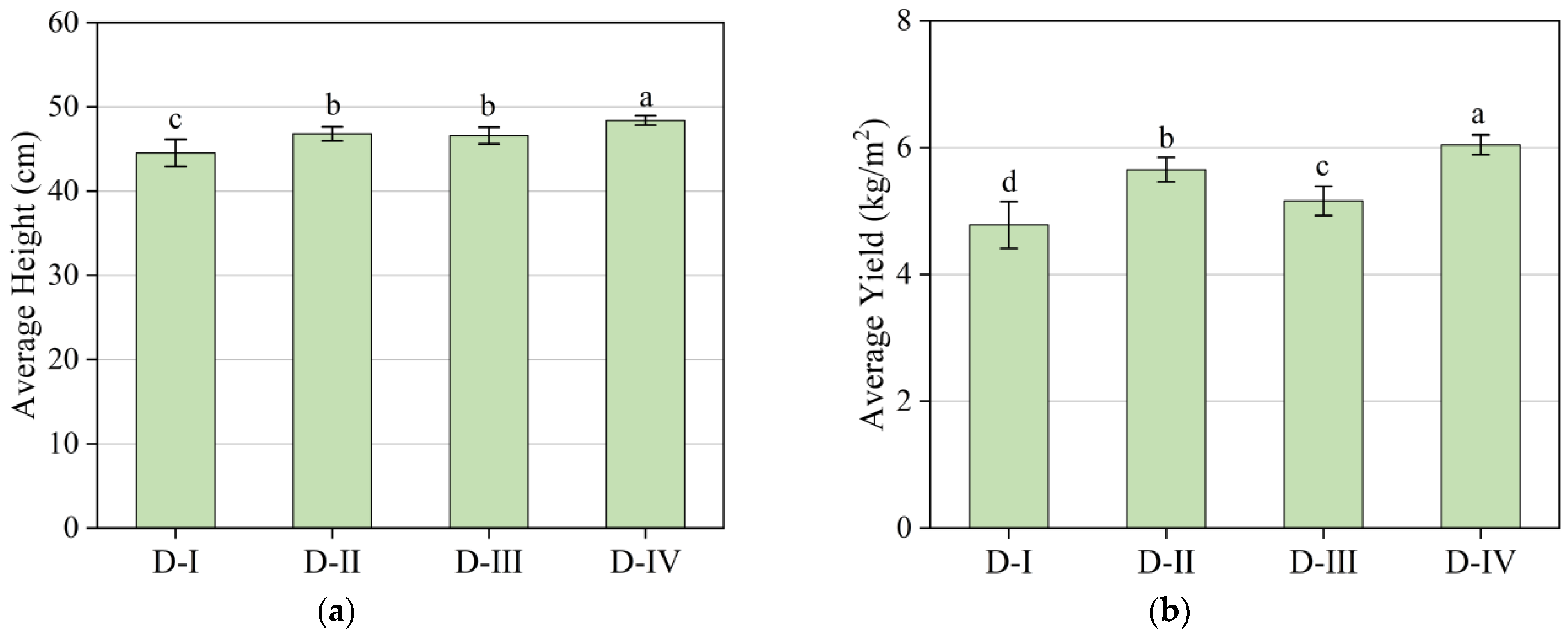
| Group | Planting Duration | Lighting Conditions | Exhaust Gases Introduction | Average Height (cm) | Average Yield (kg/m2) |
|---|---|---|---|---|---|
| D-Ⅰ | 35 days | Full spectrum | no | 45 ± 1.6 c | 4.8 ± 0.4 d |
| D-Ⅱ | 35 days | Full spectrum | yes | 47 ± 0.8 b | 5.7 ± 0.2 b |
| D-Ⅲ | 35 days | Red–blue light | no | 47 ± 1.0 b | 5.2 ± 0.2 c |
| D-Ⅳ | 35 days | Red–blue light | yes | 48 ± 0.6 a | 6.0 ± 0.2 a |
| Sample | N (%) | C (%) | H (%) | S (%) |
|---|---|---|---|---|
| 1 | 3.78 | 40.93 | 5.28 | 0.25 |
| 2 | 3.86 | 40.03 | 5.35 | 0.23 |
| 3 | 3.81 | 41.98 | 5.12 | 0.39 |
| Average | 3.82 | 40.98 | 5.25 | 0.29 |
| Material | Quantity | Unit | Adapted Quantity | Functional Unit | Explanation |
|---|---|---|---|---|---|
| Plant Factory Infrastructure | |||||
| Steel pipe | 75 | kg | 3.77 × 10−3 | kg/kg | Materials for plant racks (40-year life period) |
| Solar glass | 130.6 | kg | 6.57 × 10−3 | kg/kg | Observation windows (40-year life period) |
| PVC calendered sheet | 244 | kg | 2.46 × 10−2 | kg/kg | Materials for planting troughs and pipelines (20-year life period) |
| Light-emitting diode | 0.5 | kg | 1.01 × 10−4 | kg/kg | Illumination equipment (10-year life period) |
| Machinery and computers | 3 | kg | 6.04 × 10−4 | kg/kg | Pumps, valves, control cabinet, and other devices (10-year life period) |
| Transportation | 362.5 | tkm | 1.82 × 10−2 | tkm/kg | Transportation of the plant factory to the industrial park |
| Land-use change | 18 | m2 | 9.06 × 10−4 | m2/kg | Land-use change from industrial land to agricultural land for perennial crop |
| Pennisetum giganteum Cultivating Operation (one-year cultivation period) | |||||
| Soil | 403.2 | kg | 0.812 | kg/kg | Cultivation substrate in planting troughs |
| Stem segments | 10.2 | kg | 2.03 × 10−2 | kg/kg | Stem segments of Pennisetum giganteum |
| CO2 absorption from exhaust gases | 511.3 | kg | 1.03 | kg/kg | Including treatment process for industrial exhaust gases |
| CO2 absorption from atmospheric environment | 235.2 | kg | 0.473 | kg/kg | Direct absorption from atmospheric environment |
| Fertilizer | 1.1 | kg | 2.22 × 10−3 | kg/kg | NPK (26-15-15) fertilizer |
| Reclaim water | 13.1 | m3 | 2.64 × 10−2 | m3/kg | Including water treatment processes |
| Electricity consumption for lighting | 5026 | kWh | 10.1 | kWh /kg | Powered by photovoltaic solar energy from the industrial park. |
| Electricity consumption for other processes | 887 | kWh | 1.79 | kWh /kg | Including irrigation, sensors, and controller operation, introducing exhaust gases, etc. |
| Transportation | 185 | tkm | 0.372 | tkm/kg | Transportation of Pennisetum giganteum to processing sites |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chen, H.; Dong, X.; Lei, J.; Zhang, N.; Wang, Q.; Shi, Z.; Yang, J. Life Cycle Assessment of Carbon Capture by an Intelligent Vertical Plant Factory within an Industrial Park. Sustainability 2024, 16, 697. https://doi.org/10.3390/su16020697
Chen H, Dong X, Lei J, Zhang N, Wang Q, Shi Z, Yang J. Life Cycle Assessment of Carbon Capture by an Intelligent Vertical Plant Factory within an Industrial Park. Sustainability. 2024; 16(2):697. https://doi.org/10.3390/su16020697
Chicago/Turabian StyleChen, Haoyang, Xue Dong, Jie Lei, Ning Zhang, Qianrui Wang, Zhiang Shi, and Jinxing Yang. 2024. "Life Cycle Assessment of Carbon Capture by an Intelligent Vertical Plant Factory within an Industrial Park" Sustainability 16, no. 2: 697. https://doi.org/10.3390/su16020697
APA StyleChen, H., Dong, X., Lei, J., Zhang, N., Wang, Q., Shi, Z., & Yang, J. (2024). Life Cycle Assessment of Carbon Capture by an Intelligent Vertical Plant Factory within an Industrial Park. Sustainability, 16(2), 697. https://doi.org/10.3390/su16020697






