Fully Integrated Hybrid Solid Oxide Fuel Cell–Rankine Cycle System with Carbon Capture, Utilisation, and Storage for Sustainable Combined Heat and Power Production
Abstract
1. Introduction
- The process can operate at times when variable renewable solutions like solar cannot provide enough constant energy supply. In this way, the process provides a solution to balance out the peaks of high energy demand and increases energy reliability.
- A common practice of biogas plants is to provide heat and power [31]. The adoption of the system would help to raise the power output of the biogas plants, since biogas is commonly burned on site through various types of engines, gas, and steam turbines [47], which have lower efficiencies. Replacing those would increase the efficiency of existing biogas power plants.
- Biogas plants are commonly placed in more rural areas, which usually do not have, or have a smaller, industry sector. The adoption of process would bring additional job opportunities and provide hot sanitary water to smaller local districts.
- There is a potential to couple the process with synthetic fuel production in periods of low energy demand, e.g., synthetic natural gas, which could be used as a green alternative for local fuel demand, contributing to sustainable energy solutions.
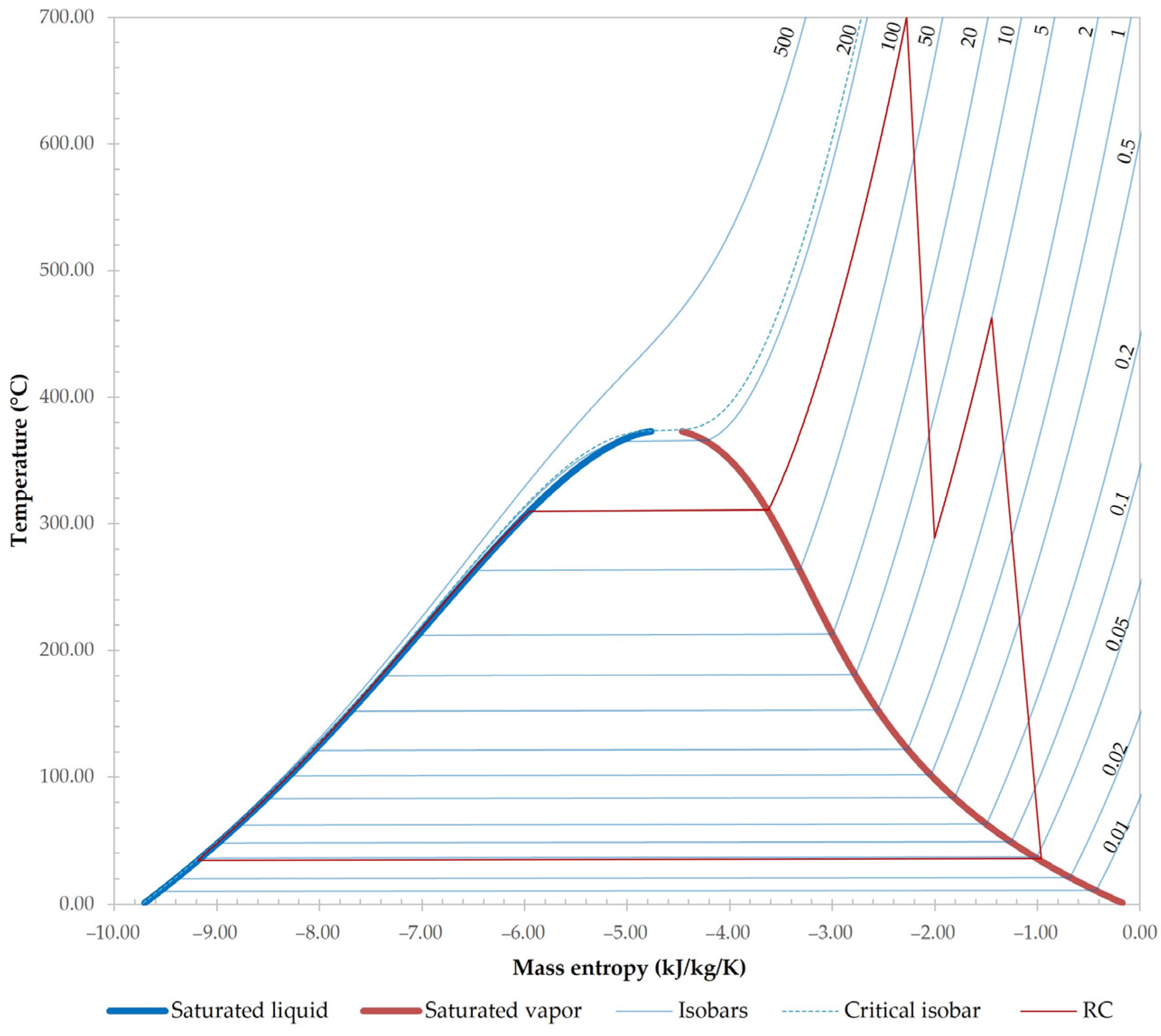
- An improved and modified version of a classic RC is adopted in a current study, increasing the efficiency of the RC, due to steam reheating between a high- and low-pressure turbine.
- The process is fully integrated, making it self-sufficient and decreasing the reliability on outside heat sources, where heat losses can occur between transferring the heat to the specific process units. This also increases the efficiency and lowers the environmental impact and operating costs.
- As the last step of the process, CC was considered, making the current proposed process carbon negative.
- The proposed system uses biogas as a green alternative to fossil fuels.
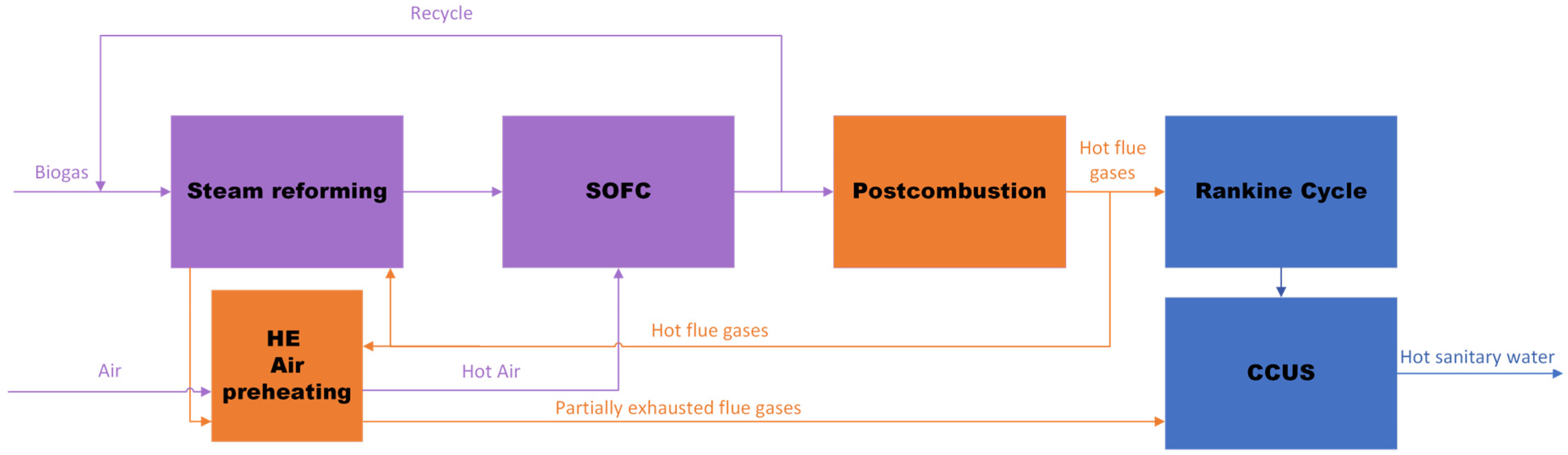
2. Materials and Methods
- A range of operating parameters was chosen by the review of literature data.
- The process is a conceptual design, and therefore not optimised.
- A workaround to simulate SOFC was made in Aspen Plus.
- Heat and pressure losses were neglected.
- The process was simulated in a steady-state and continuous mode.
2.1. Biogas Flowrate
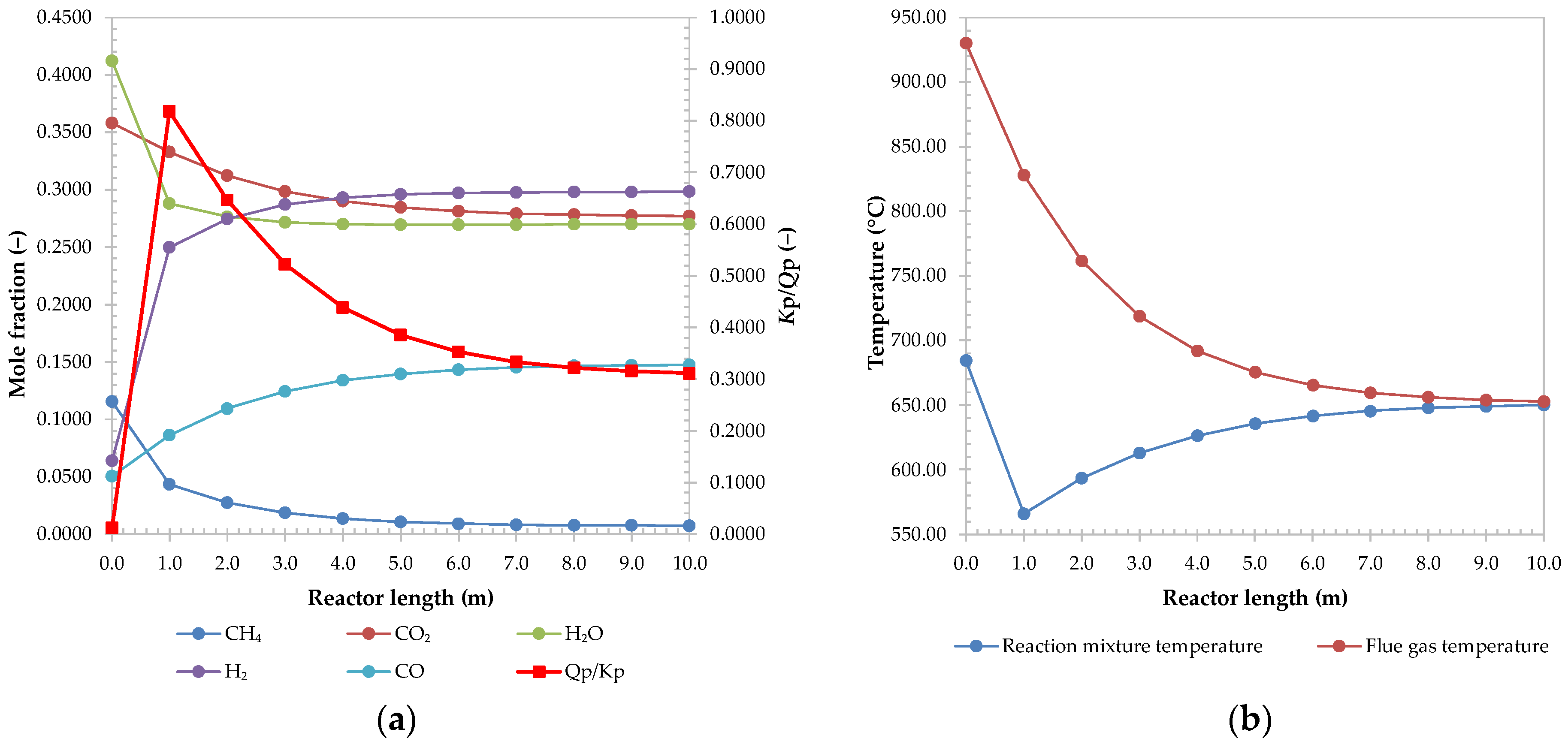
2.2. Reactor and Reaction Kinetics
2.3. SOFC and Combustion
2.4. Base Case Simulation
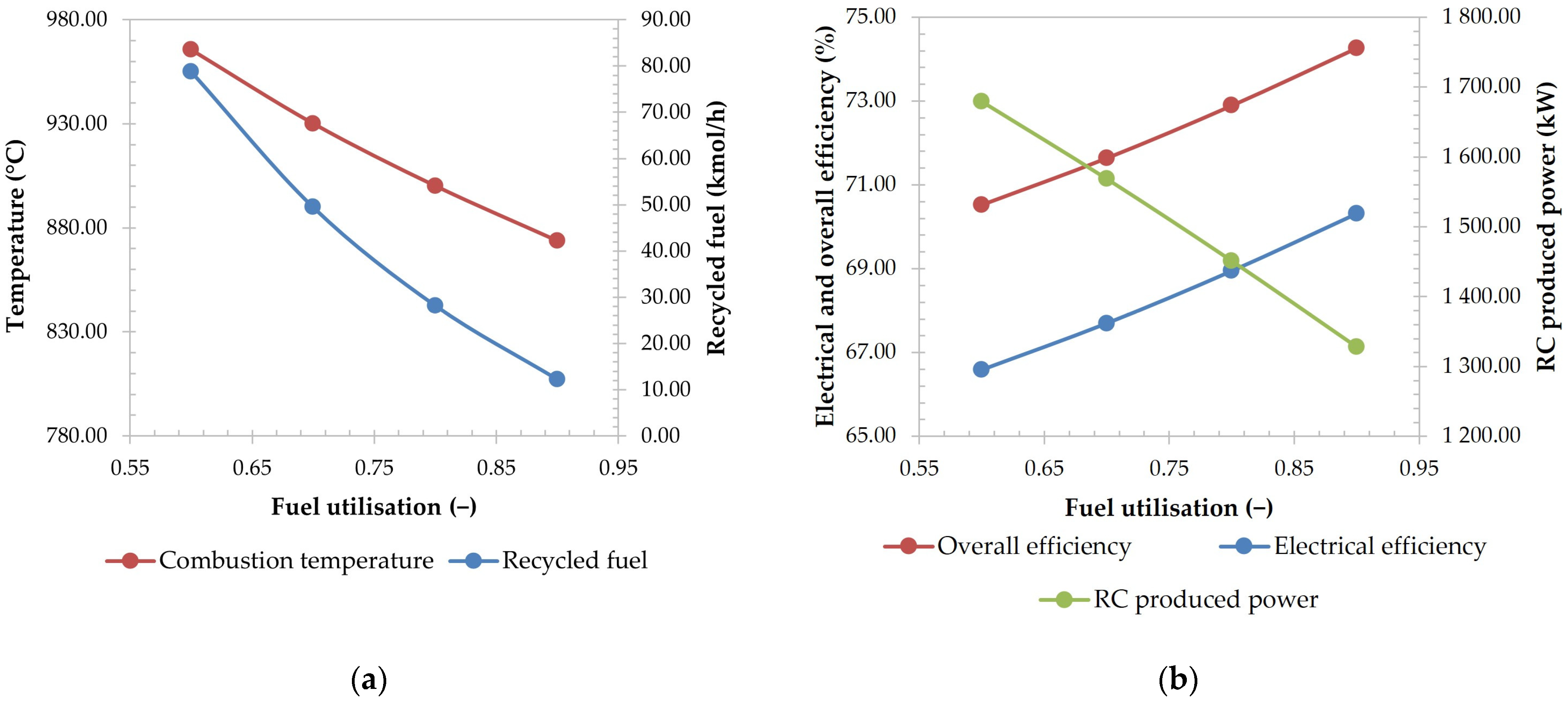
2.5. Sensitivity Analysis
2.6. Results Analysis
2.6.1. Performance Indicators
2.6.2. Coking Potential and Methane Conversion
2.6.3. Hot Sanitary Water
3. Results
3.1. Results of Base Case Simulation
3.2. Sensitivity Analysis
3.2.1. Effect of Reformer Temperature at Different SOFC Operating Temperatures
3.2.2. Effect of Fuel Utilisation
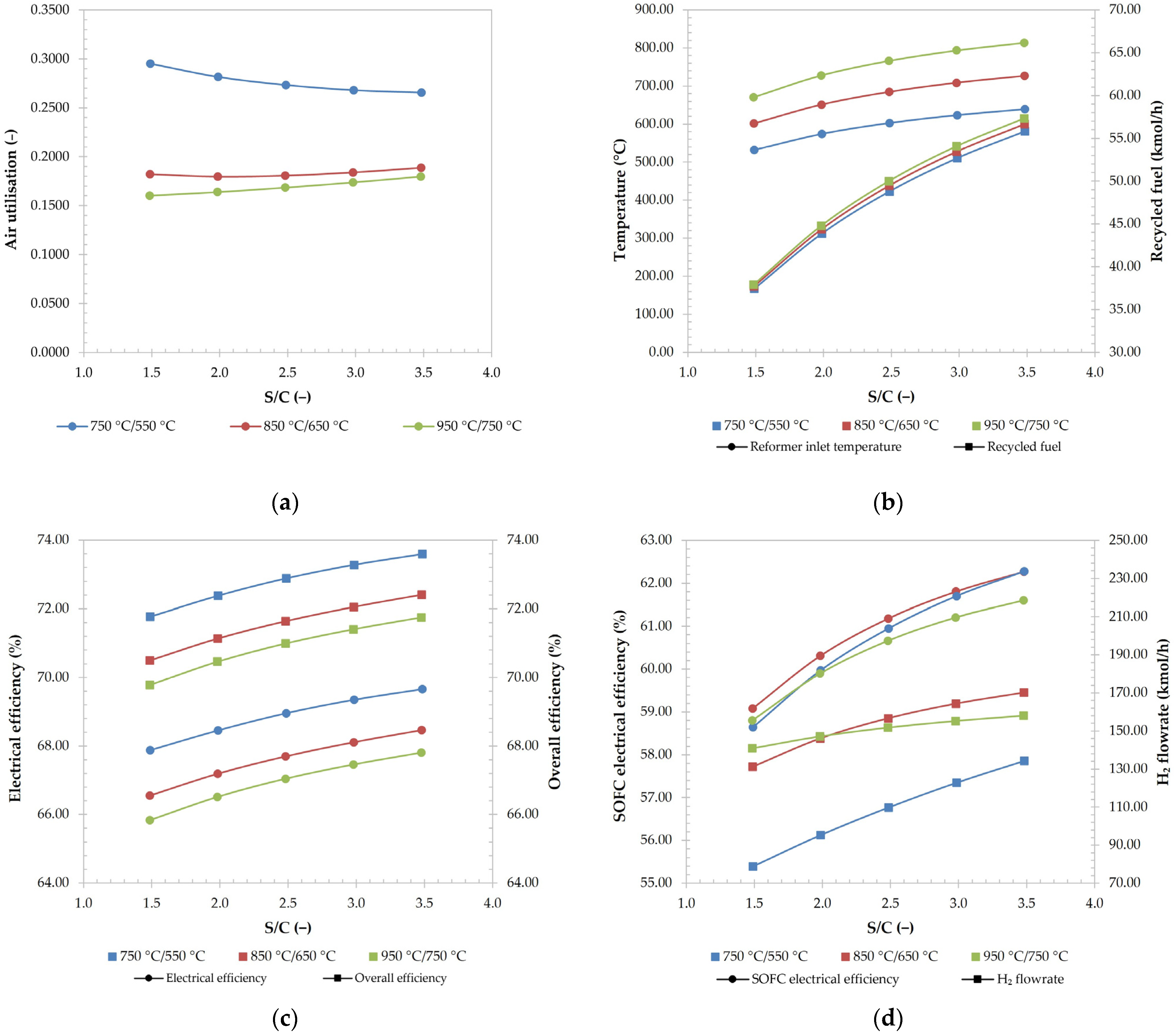
3.2.3. Effect of S/C Ratio
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| AFC | Alkaline Fuel Cell |
| CCUS | Carbon Capture, Utilisation, and Storage |
| CFC | Chlorofluorocarbons |
| CHP | Combined Heat and Power |
| COP | Coefficient of Performance |
| DMFC | Direct Methanol Fuel Cell |
| DSMR | Direct Steam Methane Reforming |
| JRC | Joint Research Centre |
| LHHW | Langmuir–Hinshelwood–Hougen–Watson |
| LHV | Lower Heating Value |
| GHG | Greenhouse Gases |
| MCFC | Molten Carbonate Fuel Cell |
| PAFC | Phosphoric Acid Fuel Cell |
| PEMFC | Polymer Electrolyte Membrane Fuel Cell |
| PFR | Plug Flow Reactor |
| RC | Rankine Cycle |
| SMR | Steam Methane Reforming |
| SOFC | Solid Oxide Fuel Cell |
| SOFC-GT | Solid Oxide Fuel Cell Gas Turbine |
| VSA | Vacuum Swing Adsorption |
| WGS | Water Gas Shift |
Appendix A
| Stream Name | Temperature (°C) | Pressure (bar) | Molar Vapour Fraction (−) | Mole Flow (kmol/h) | Mole Fraction of Component | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| CH4 | CO2 | H2 | H2O | N2 | CO | O2 | NH3 | ||||||
| Fuel streams | FS-1 | 35.00 | 1.01 | 0.9969 | 80.97 | 0.6175 | 0.3325 | 0 | 0.0500 | 0 | 0 | 0 | 0 |
| FS-2 | 35.00 | 1.01 | 1.0000 | 80.72 | 0.6194 | 0.3335 | 0 | 0.0470 | 0 | 0 | 0 | 0 | |
| FS-3 | 50.01 | 1.20 | 1.0000 | 80.72 | 0.6194 | 0.3335 | 0 | 0.0470 | 0 | 0 | 0 | 0 | |
| A-EXH-11 | 850.00 | 1.20 | 1.0000 | 352.11 | 0 | 0.3636 | 0.0784 | 0.4960 | 0 | 0.0621 | 0 | 0 | |
| FS-4 | 684.42 | 1.20 | 1.0000 | 432.82 | 0.1155 | 0.3580 | 0.0638 | 0.4122 | 0 | 0.0505 | 0 | 0 | |
| FS-5 | 649.99 | 1.20 | 1.0000 | 525.15 | 0.0073 | 0.2770 | 0.2983 | 0.2698 | 0 | 0.1475 | 0 | 0 | |
| A-EXH-1 | 850.00 | 1.20 | 1.0000 | 532.82 | 0 | 0.3636 | 0.0784 | 0.4960 | 0 | 0.0621 | 0 | 0 | |
| A-EXH-21 | 850.00 | 1.20 | 1.0000 | 180.72 | 0 | 0.3636 | 0.0784 | 0.4960 | 0 | 0.0621 | 0 | 0 | |
| Air streams | AS-1 | 25.00 | 1.00 | 1.0000 | 2301.76 | 0 | 0 | 0 | 0 | 0.7900 | 0 | 0.2100 | 0 |
| AS-2 | 44.87 | 1.20 | 1.0000 | 2301.76 | 0 | 0 | 0 | 0 | 0.7900 | 0 | 0.2100 | 0 | |
| AS-3 | 650.00 | 1.20 | 1.0000 | 2301.76 | 0 | 0 | 0 | 0 | 0.7900 | 0 | 0.2100 | 0 | |
| SOFC-M | 650.00 | 1.20 | 1.0000 | 87.31 | 0 | 0 | 0 | 0 | 0 | 0 | 1.0000 | 0 | |
| C-EXH-1 | 650.00 | 1.20 | 1.0000 | 2214.49 | 0 | 0 | 0 | 0 | 0.8211 | 0 | 0.1789 | 0 | |
| C-EXH-2 | 850.00 | 1.20 | 1.0000 | 2214.49 | 0 | 0 | 0 | 0 | 0.8211 | 0 | 0.1789 | 0 | |
| Flue gas streams | FG-1 | 930.12 | 1.20 | 1.0000 | 2382.52 | 0 | 0.0323 | 0 | 0.0436 | 0.7632 | 0 | 0.1609 | 0 |
| FG-11 | 930.12 | 1.20 | 1.0000 | 1859.18 | 0 | 0.0323 | 0 | 0.0436 | 0.7632 | 0 | 0.1609 | 0 | |
| FG-12 | 930.12 | 1.20 | 1.0000 | 1043.57 | 0 | 0.0323 | 0 | 0.0436 | 0.7632 | 0 | 0.1609 | 0 | |
| FG-13 | 930.12 | 1.20 | 1.0000 | 815.61 | 0 | 0.0323 | 0 | 0.0436 | 0.7632 | 0 | 0.1609 | 0 | |
| FG-14 | 652.76 | 1.20 | 1.0000 | 1043.57 | 0 | 0.0323 | 0 | 0.0436 | 0.7632 | 0 | 0.1609 | 0 | |
| FG-15 | 776.02 | 1.20 | 1.0000 | 1859.18 | 0 | 0.0323 | 0 | 0.0436 | 0.7632 | 0 | 0.1609 | 0 | |
| FG-16 | 54.87 | 1.20 | 1.0000 | 1859.18 | 0 | 0.0323 | 0 | 0.0436 | 0.7632 | 0 | 0.1609 | 0 | |
| FG-21 | 930.12 | 1.20 | 1.0000 | 523.34 | 0 | 0.0323 | 0 | 0.0436 | 0.7632 | 0 | 0.1609 | 0 | |
| FG-22 | 727.65 | 1.20 | 1.0000 | 523.34 | 0 | 0.0323 | 0 | 0.0436 | 0.7632 | 0 | 0.1609 | 0 | |
| FG-23 | 654.15 | 1.20 | 1.0000 | 523.34 | 0 | 0.0323 | 0 | 0.0436 | 0.7632 | 0 | 0.1609 | 0 | |
| FG-24 | 367.64 | 1.20 | 1.0000 | 523.34 | 0 | 0.0323 | 0 | 0.0436 | 0.7632 | 0 | 0.1609 | 0 | |
| FG-25 | 44.94 | 1.20 | 1.0000 | 523.34 | 0 | 0.0323 | 0 | 0.0436 | 0.7632 | 0 | 0.1609 | 0 | |
| FG-2 | 52.69 | 1.20 | 1.0000 | 2382.52 | 0 | 0.0323 | 0 | 0.0436 | 0.7632 | 0 | 0.1609 | 0 | |
| FG-3 | 52.69 | 1.20 | 1.0000 | 2382.52 | 0 | 0.0323 | 0 | 0.0436 | 0.7632 | 0 | 0.1609 | 0 | |
| CC and liquefication | CC-1 | 52.69 | 1.20 | 1.0000 | 75.61 | 0 | 0.9970 | 0 | 0.0001 | 0.0024 | 0 | 0.0005 | 0 |
| CC-2 | 147.25 | 3.30 | 1.0000 | 75.61 | 0 | 0.9970 | 0 | 0.0001 | 0.0024 | 0 | 0.0005 | 0 | |
| CC-3 | 40.00 | 3.30 | 1.0000 | 75.61 | 0 | 0.9970 | 0 | 0.0001 | 0.0024 | 0 | 0.0005 | 0 | |
| CC-4 | 132.88 | 9.10 | 1.0000 | 75.61 | 0 | 0.9970 | 0 | 0.0001 | 0.0024 | 0 | 0.0005 | 0 | |
| CC-5 | 40.00 | 9.10 | 1.0000 | 75.61 | 0 | 0.9970 | 0 | 0.0001 | 0.0024 | 0 | 0.0005 | 0 | |
| CC-6 | 134.35 | 25.06 | 1.0000 | 75.61 | 0 | 0.9970 | 0 | 0.0001 | 0.0024 | 0 | 0.0005 | 0 | |
| CC-7 | 40.00 | 25.06 | 1.0000 | 75.61 | 0 | 0.9970 | 0 | 0.0001 | 0.0024 | 0 | 0.0005 | 0 | |
| CC-8 | 137.39 | 69.00 | 1.0000 | 75.61 | 0 | 0.9970 | 0 | 0.0001 | 0.0024 | 0 | 0.0005 | 0 | |
| CC-9 | 40.00 | 69.00 | 1.0000 | 75.61 | 0 | 0.9970 | 0 | 0.0001 | 0.0024 | 0 | 0.0005 | 0 | |
| CC-10 | 18.00 | 69.00 | 1.0000 | 75.61 | 0 | 0.9970 | 0 | 0.0001 | 0.0024 | 0 | 0.0005 | 0 | |
| HP cycle streams | HP-1 | 8.01 | 5.68 | 0.1975 | 8.80 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 1.0000 |
| HP-2 | 8.01 | 5.68 | 1.0000 | 8.80 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 1.0000 | |
| HP-3 | 145.72 | 23.86 | 1.0000 | 8.80 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 1.0000 | |
| HP-4 | 56.46 | 23.86 | 0 | 8.80 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 1.0000 | |
| Hot water for sanitary use | SHW-C-1 | 30.00 | 1.00 | 0 | 135.14 | 0 | 0 | 0 | 1.0000 | 0 | 0 | 0 | 0 |
| SHW-H-1 | 60.00 | 1.00 | 0 | 135.14 | 0 | 0 | 0 | 1.0000 | 0 | 0 | 0 | 0 | |
| SHW-C-2 | 30.00 | 1.00 | 0 | 118.96 | 0 | 0 | 0 | 1.0000 | 0 | 0 | 0 | 0 | |
| SHW-H-2 | 60.00 | 1.00 | 0 | 118.96 | 0 | 0 | 0 | 1.0000 | 0 | 0 | 0 | 0 | |
| SHW-C-3 | 30.00 | 1.00 | 0 | 129.83 | 0 | 0 | 0 | 1.0000 | 0 | 0 | 0 | 0 | |
| SHW-H-3 | 60.00 | 1.00 | 0 | 129.83 | 0 | 0 | 0 | 1.0000 | 0 | 0 | 0 | 0 | |
| SHW-C-4 | 30.00 | 1.00 | 0 | 184.14 | 0 | 0 | 0 | 1.0000 | 0 | 0 | 0 | 0 | |
| SHW-H-4 | 60.00 | 1.00 | 0 | 184.14 | 0 | 0 | 0 | 1.0000 | 0 | 0 | 0 | 0 | |
| SHW-C-5 | 30.00 | 1.00 | 0 | 79.13 | 0 | 0 | 0 | 1.0000 | 0 | 0 | 0 | 0 | |
| SHW-H-5 | 60.00 | 1.00 | 0 | 79.13 | 0 | 0 | 0 | 1.0000 | 0 | 0 | 0 | 0 | |
| SHW-1 | 60.00 | 1.00 | 0 | 647.20 | 0 | 0 | 0 | 1.0000 | 0 | 0 | 0 | 0 | |
| SHW-2 | 60.04 | 6.00 | 0 | 647.20 | 0 | 0 | 0 | 1.0000 | 0 | 0 | 0 | 0 | |
| RC streams | HPW-1 | 34.94 | 100.00 | 0 | 200.51 | 0 | 0 | 0 | 1.0000 | 0 | 0 | 0 | 0 |
| HPW-2 | 34.94 | 100.00 | 0 | 200.51 | 0 | 0 | 0 | 1.0000 | 0 | 0 | 0 | 0 | |
| HPW-3 | 310.09 | 100.00 | 0 | 200.51 | 0 | 0 | 0 | 1.0000 | 0 | 0 | 0 | 0 | |
| HPS-1 | 310.09 | 100.00 | 1.0000 | 200.51 | 0 | 0 | 0 | 1.0000 | 0 | 0 | 0 | 0 | |
| SHPS-1 | 700.00 | 100.00 | 1.0000 | 200.51 | 0 | 0 | 0 | 1.0000 | 0 | 0 | 0 | 0 | |
| LPS-1 | 288.79 | 5.00 | 1.0000 | 200.51 | 0 | 0 | 0 | 1.0000 | 0 | 0 | 0 | 0 | |
| SLPS-1 | 463.00 | 5.00 | 1.0000 | 200.51 | 0 | 0 | 0 | 1.0000 | 0 | 0 | 0 | 0 | |
| VPS-1 | 35.68 | 0.05 | 1.0000 | 200.51 | 0 | 0 | 0 | 1.0000 | 0 | 0 | 0 | 0 | |
| VPW-1 | 34.20 | 0.05 | 0 | 200.51 | 0 | 0 | 0 | 1.0000 | 0 | 0 | 0 | 0 | |
| CW-1 | 9.00 | 1.00 | 0 | 37,577.17 | 0 | 0 | 0 | 1.0000 | 0 | 0 | 0 | 0 | |
| CW-2 | 9.02 | 5.00 | 0 | 37,577.17 | 0 | 0 | 0 | 1.0000 | 0 | 0 | 0 | 0 | |
| CW-3 | 12.00 | 5.00 | 0 | 37,577.17 | 0 | 0 | 0 | 1.0000 | 0 | 0 | 0 | 0 | |
| Waste streams | WS-1 | 35.00 | 1.01 | 0 | 0.25 | 0 | 0 | 0 | 1.0000 | 0 | 0 | 0 | 0 |
| WS-2 | 0.00 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | ||||
| WS-3 | 52.69 | 1.20 | 1.0000 | 2306.90 | 0 | 0.0007 | 0 | 0.0450 | 0.7882 | 0 | 0.1662 | 0 | |
| Process Unit (−) | Heat or Work (kW) | |
|---|---|---|
| Consumed power | C-1 | 12.99 |
| C-2 | 390.09 | |
| C-3 | 84.06 | |
| C-4 | 80.02 | |
| C-5 | 77.73 | |
| C-6 | 71.45 | |
| C-7 | 11.96 | |
| RC-P-1 | 13.40 | |
| RC-P-2 | 98.06 | |
| DH-P | 2.22 | |
| Generated power | SOFC | 6821.81 |
| RC-HPT | 770.84 | |
| RC-LPT | 797.88 | |
| Integrated heat | R-1 | 2772.07 |
| SOFC—Air heating | 4085.11 | |
| AS-HE-1 | 11,905.43 | |
| CC-HE-5 | 42.41 | |
| RC-HPWH | 1431.95 | |
| RC-HPSG | 1359.18 | |
| RC-SHPSG | 1021.27 | |
| RC-LPSR | 362.10 | |
| Utilities | RC-COND | 2535.94 |
| Useful heat | CC-HE-1 | 91.83 |
| CC-HE-2 | 80.84 | |
| CC-HE-3 | 88.22 | |
| CC-HE-4 | 125.12 | |
| CC-HE-6 | 53.77 |
References
- Jiang, Z.; Rahman Mahmud, A.; Maneengam, A.; Nassani, A.A.; Haffar, M.; The Cong, P. Non Linear Effect of Biomass, Fossil Fuels and Renewable Energy Usage on the Economic Growth: Managing Sustainable Development through Energy Sector. Fuel 2022, 326, 124943. [Google Scholar] [CrossRef]
- Mirza, Z.T.; Anderson, T.; Seadon, J.; Brent, A. A Thematic Analysis of the Factors That Influence the Development of a Renewable Energy Policy. Renew. Energy Focus 2024, 49, 100562. [Google Scholar] [CrossRef]
- Sheng, M.S.; Sharp, B.; Yi, M.; Wen, L.; Suomalainen, K. A Cointegration Analysis of New Zealand’s Economic Development, Fossil Fuel Usage and Transport Emissions. Case Stud. Transp. Policy 2022, 10, 2497–2505. [Google Scholar] [CrossRef]
- Liu, G.; Ofori, C.; Ampong, S.A.; Appiah-Twum, F.; Alhassan, E.A. Towards a Sustainable Environment: Examining the Spatial VARIATIONS of Renewable Energy, Environmental Pollution, and Economic Growth in Europe. Energy Strategy Rev. 2023, 50, 101231. [Google Scholar] [CrossRef]
- Herwartz, H.; Theilen, B.; Wang, S. Unraveling the Structural Sources of Oil Production and Their Impact on CO2 Emissions. Energy Econ. 2024, 132, 107488. [Google Scholar] [CrossRef]
- Cary, M.; Stephens, H.M. Economic, Environmental, and Technical Gains from the Kyoto Protocol: Evidence from Cement Manufacturing. Resour. Policy 2024, 91, 104926. [Google Scholar] [CrossRef]
- Nyangchak, N. Assessing Renewable Energy Efficiency and Policies: A Combined Analysis of LMDI, Super-SBM, and Fieldwork in Qinghai, China. Energy Sustain. Dev. 2024, 80, 101420. [Google Scholar] [CrossRef]
- Xu, G.; Yang, M.; Li, S.; Jiang, M.; Rehman, H. Evaluating the Effect of Renewable Energy Investment on Renewable Energy Development in China with Panel Threshold Model. Energy Policy 2024, 187, 114029. [Google Scholar] [CrossRef]
- Nath, F.; Mahmood, M.N.; Yousuf, N. Recent Advances in CCUS: A Critical Review on Technologies, Regulatory Aspects and Economics. Geoenergy Sci. Eng. 2024, 238, 212726. [Google Scholar] [CrossRef]
- Zhang, Q.; Liu, J.; Wang, G.; Gao, Z. A New Optimization Model for Carbon Capture Utilization and Storage (CCUS) Layout Based on High-Resolution Geological Variability. Appl. Energy 2024, 363, 123065. [Google Scholar] [CrossRef]
- Gupta, N.C.; Tanwar, R.; Dipesh; Kaushik, A.; Singh, R.; Patra, A.K.; Sar, P.; Khakharia, P. Perspectives on CCUS Deployment on Large Scale in India: Insights for Low Carbon Pathways. Carbon Capture Sci. Technol. 2024, 12, 100195. [Google Scholar] [CrossRef]
- Petrovič, A.; Stergar, J.; Škodič, L.; Rašl, N.; Cenčič Predikaka, T.; Čuček, L.; Goričanec, D.; Urbancl, D. Thermo-Kinetic Analysis of Pyrolysis of Thermally Pre-Treated Sewage Sludge from the Food Industry. Therm. Sci. Eng. Prog. 2023, 42, 101863. [Google Scholar] [CrossRef]
- Trop, P.; Agrez, M.; Urbancl, D.; Goricanec, D. Co-Gasification of Torrefied Wood Biomass and Sewage Sludge. In Computer Aided Chemical Engineering; Elsevier: Portorož, Slovenia, 2016; Volume 38, pp. 2229–2234. ISBN 978-0-444-63428-3. [Google Scholar]
- Goričanec, D.; Ivanovski, I.; Krope, J.; Urbancl, D. The Exploitation of Low-Temperature Hot Water Boiler Sources with High-Temperature Heat Pump Integration. Energies 2020, 13, 6311. [Google Scholar] [CrossRef]
- Babaharra, O.; Choukairy, K.; Faraji, H.; Hamdaoui, S. Improved Heating Floor Thermal Performance by Adding PCM Microcapsules Enhanced by Single and Hybrid Nanoparticles. Heat Trans. 2023, 52, 3817–3838. [Google Scholar] [CrossRef]
- Kahraman, H.; Akın, Y. Recent Studies on Proton Exchange Membrane Fuel Cell Components, Review of the Literature. Energy Convers. Manag. 2024, 304, 118244. [Google Scholar] [CrossRef]
- Guo, X.; Hu, X.; Zhang, S. Application Status of Variable-Frequency Drive in Hydrogen Fuel Cell Air Compressors from an Industrial Viewpoint: A Review. Sustain. Energy Technol. Assess. 2024, 64, 103716. [Google Scholar] [CrossRef]
- Tariq, A.H.; Kazmi, S.A.A.; Hassan, M.; Muhammed Ali, S.A.; Anwar, M. Analysis of Fuel Cell Integration with Hybrid Microgrid Systems for Clean Energy: A Comparative Review. Int. J. Hydrogen Energy 2024, 52, 1005–1034. [Google Scholar] [CrossRef]
- Sinha, A.A.; Srivastava, K.; Rajpoot, A.S.; Choudhary, T.; Pandey, S.P.; Sanjay. A Thermodynamic Approach to Analyze Energy, Exergy, Emission, and Sustainability (3E-S) Performance by Utilizing Low Temperature Waste Heat in SOFC–CHP-TEG System. Int. J. Hydrogen Energy 2024, 63, 1088–1104. [Google Scholar] [CrossRef]
- Zhang, X.; Liu, W.; Pan, J.; Zhao, B.; Yi, Z.; He, X.; Liu, Y.; Li, H. Comprehensive Performance Assessment of a Novel Biomass-Based CCHP System Integrated with SOFC and HT-PEMFC. Energy 2024, 295, 131112. [Google Scholar] [CrossRef]
- Wang, N.; Wang, D.; Xing, Y.; Shao, L.; Afzal, S. Application of Co-Evolution RNA Genetic Algorithm for Obtaining Optimal Parameters of SOFC Model. Renew. Energy 2020, 150, 221–233. [Google Scholar] [CrossRef]
- Salam, A.; Zholobko, O.; Wu, X.-F. Roles of Functionalized Nanoparticles in the Performance Improvement of Proton-Exchange Membranes Used in Low- and Intermediate-Temperature Hydrogen Fuel Cells: A Review. Prog. Nat. Sci. Mater. Int. 2024; in press. [Google Scholar] [CrossRef]
- Liang, W.; Han, J.; Zhu, W.; Yang, J.; Lv, W.; Liu, C. A Novel Pathway for Achieving Efficient Integration of SOFC/SOEC and Addressing Photovoltaic Duck Curve Challenge. Energy Convers. Manag. 2024, 306, 118326. [Google Scholar] [CrossRef]
- Hagen, A.; Caldogno, R.; Sun, X. Direct Ammonia SOFC—A Potential Technology for Green Shipping. Fuel 2024, 365, 131238. [Google Scholar] [CrossRef]
- Yang, P.; Zhang, Y.; Yang, C.; Chen, J.; Liu, Z.; Deng, C.; Yang, S. Thermodynamic Performance Comparison of a SOFC System Integrated with Steam Reforming and Dry Reforming by Utilizing Different Fuels. Energy Convers. Manag. 2024, 300, 117981. [Google Scholar] [CrossRef]
- Liu, L.; Duan, L.; Zheng, N.; Wang, Q.; Zhang, M.; Xue, D. Thermodynamic Performance Evaluation of a Novel Solar-Assisted Multi-Generation System Driven by Ammonia-Fueled SOFC with Anode Outlet Gas Recirculation. Energy 2024, 294, 130845. [Google Scholar] [CrossRef]
- Kim, H.; Lee, J.; Gokbayrak, A.A.; Seo, Y.; Oh, S.; Oh, M.J.; Jun, Y.; Son, J.-W.; Yang, S. Characterization of Direct-Ammonia Solid Oxide Fuel Cells (DA-SOFCs) at 650–750 °C in a Single-Repeating Unit Stack: Effects of Metallic Components and Residual Ammonia. Int. J. Hydrogen Energy 2024, 68, 1312–1321. [Google Scholar] [CrossRef]
- Narayanan, M.; Mengedoht, G.; Commerell, W. Evaluation of SOFC-CHP’s Ability to Integrate Thermal and Electrical Energy System Decentrally in a Single-Family House with Model Predictive Controller. Sustain. Energy Technol. Assess. 2021, 48, 101643. [Google Scholar] [CrossRef]
- Ismael, I.; El-Fergany, A.A.; Gouda, E.A.; Kotb, M.F. Cooperation Search Algorithm for Optimal Parameters Identification of SOFCs Feeding Electric Vehicle at Steady and Dynamic Modes. Int. J. Hydrogen Energy 2024, 50, 1395–1407. [Google Scholar] [CrossRef]
- Zarabi Golkhatmi, S.; Asghar, M.I.; Lund, P.D. A Review on Solid Oxide Fuel Cell Durability: Latest Progress, Mechanisms, and Study Tools. Renew. Sustain. Energy Rev. 2022, 161, 112339. [Google Scholar] [CrossRef]
- Rola, K.; Gruber, S.; Urbancl, D.; Goričanec, D. Utilisation of Renewable Electricity to Produce Synthetic Methane. Energies 2023, 16, 6871. [Google Scholar] [CrossRef]
- Energy Security–Topics. Available online: https://www.iea.org/topics/energy-security (accessed on 15 May 2024).
- Corigliano, O.; Pagnotta, L.; Fragiacomo, P. On the Technology of Solid Oxide Fuel Cell (SOFC) Energy Systems for Stationary Power Generation: A Review. Sustainability 2022, 14, 15276. [Google Scholar] [CrossRef]
- Dell, R.M.; Moseley, P.T.; Rand, D.A.J. Hydrogen, Fuel Cells and Fuel Cell Vehicles. In Towards Sustainable Road Transport; Elsevier: Oxford, UK, 2014; pp. 260–295. ISBN 978-0-12-404616-0. [Google Scholar]
- Zhou, J.; Wang, Z.; Han, M.; Sun, Z.; Sun, K. Optimization of a 30 kW SOFC Combined Heat and Power System with Different Cycles and Hydrocarbon Fuels. Int. J. Hydrogen Energy 2022, 47, 4109–4119. [Google Scholar] [CrossRef]
- Alaedini, A.H.; Tourani, H.K.; Saidi, M. A Review of Waste-to-Hydrogen Conversion Technologies for Solid Oxide Fuel Cell (SOFC) Applications: Aspect of Gasification Process and Catalyst Development. J. Environ. Manag. 2023, 329, 117077. [Google Scholar] [CrossRef] [PubMed]
- Meng, T.; Cui, D.; Shi, Y.; Ji, Y.; Cheng, M.; Tu, B.; Lan, Z. Performance Evaluation of High-Efficiency SOFC-PEMFC Hybrid System Fueled by Liquid Ammonia. Int. J. Hydrogen Energy 2023, 48, 30887–30898. [Google Scholar] [CrossRef]
- Tan, L.; Chen, C.; Gong, Z.; Xia, L. Performance Evaluation on a Novel Combined Cool/Heat and Power (CCP/CHP) System Integrating an SOFC-GT Plant with a Solar-Assisted LiBr Absorption Cooling/Heating Unit. Energy 2023, 283, 129102. [Google Scholar] [CrossRef]
- Liang, W.; Han, J.; Ge, Y.; Zhu, W.; Yang, J.; Lv, W.; Liu, C. Investigation on Combining Multi-Effect Distillation and Double-Effect Absorption Refrigeration Cycle to Recover Exhaust Heat of SOFC-GT System. Energy Convers. Manag. 2024, 301, 118054. [Google Scholar] [CrossRef]
- Roy, D.; Samanta, S.; Roy, S.; Smallbone, A.; Roskilly, A.P. Techno-Economic Analysis of Solid Oxide Fuel Cell-Based Energy Systems for Decarbonising Residential Power and Heat in the United Kingdom. Green Chem. 2024, 26, 3979–3994. [Google Scholar] [CrossRef]
- Gruber, S.; Rola, K.; Urbancl, D.; Goričanec, D. Carbon-Free Heat Production for High-Temperature Heating Systems. Sustainability 2023, 15, 15063. [Google Scholar] [CrossRef]
- Yang, S.; Wang, G.; Liu, Z.; Deng, C.; Xie, N. Energy, Exergy and Exergo-Economic Analysis of a Novel SOFC Based CHP System Integrated with Organic Rankine Cycle and Biomass Co-Gasification. Int. J. Hydrogen Energy 2024, 53, 1155–1169. [Google Scholar] [CrossRef]
- Sun, S.; Guo, X.; Zhang, H.; Li, Y.; He, Z.; Li, C. Design and Sensitivity Analysis of a Multistage Solid Oxide Fuel Cell Hybrid System with an Inter-Cooled-Recuperated Gas Turbine and Organic Rankine Cycle. Energy 2024, 286, 129561. [Google Scholar] [CrossRef]
- Nourpour, M.; Khoshgoftar Manesh, M.H. Availability and 6E Assessment and Optimal Design of a Novel Cogeneration System Based on Integrated Turbo Compressor Station-SOFC-Solar-Geothermal-Steam and Organic Rankine Cycles with Machine Learning. Renew. Energy 2023, 215, 118908. [Google Scholar] [CrossRef]
- Singh, U.R.; Bhogilla, S. Exergy Analysis of Reversible Sofc Coupled with Organic Rankine Cycle and Hydrogen Storage for Renewable Energy Storage. Int. J. Hydrogen Energy 2023, 48, 39169–39181. [Google Scholar] [CrossRef]
- Wang, J.; Tian, H.; Wang, X.; Li, L.; Sun, R.; Bian, X.; Shu, G.; Liang, X. Process Design Methodology for Rankine Cycle Based on Heat Matching. Renew. Sustain. Energy Rev. 2024, 193, 114295. [Google Scholar] [CrossRef]
- Kabeyi, M.J.B.; Olanrewaju, O.A. Biogas Production and Applications in the Sustainable Energy Transition. J. Energy 2022, 2022, 8750221. [Google Scholar] [CrossRef]
- AspenTech. Available online: https://www.aspentech.com/ (accessed on 10 March 2024).
- Athanasiou, C.; Drosakis, C.; Booto, G.K.; Elmasides, C. Economic Feasibility of Power/Heat Cogeneration by Biogas–Solid Oxide Fuel Cell (SOFC) Integrated Systems. Energies 2022, 16, 404. [Google Scholar] [CrossRef]
- He, V.; Gaffuri, M.; Van Herle, J.; Schiffmann, J. Readiness Evaluation of SOFC-MGT Hybrid Systems with Carbon Capture for Distributed Combined Heat and Power. Energy Convers. Manag. 2023, 278, 116728. [Google Scholar] [CrossRef]
- Doherty, W.; Reynolds, A.; Kennedy, D. Computer Simulation of a Biomass Gasification-Solid Oxide Fuel Cell Power System Using Aspen Plus. Energy 2010, 35, 4545–4555. [Google Scholar] [CrossRef]
- Doherty, W. Modelling and Simulation of a Biomass Gasification-Solid Oxide Fuel Cell Combined Heat and Power Plant Using Aspen Plus. In Proceedings of the 22nd International Conference on Efficiency, Cost, Optimization, Simulation and Environmental Impact of Energy Systems, Foz Do Iguaçu, Brazil, 30 August–3 September 2009. [Google Scholar] [CrossRef]
- Rashid, K.; Dong, S.K.; Mehran, M.T.; Lee, D.W. Design and Analysis of Compact Hotbox for Solid Oxide Fuel Cell Based 1 kW-Class Power Generation System. Appl. Energy 2017, 208, 620–636. [Google Scholar] [CrossRef]
- Duan, L.; Zhang, X.; Yang, Y. Exergy Analysis of a Novel SOFC Hybrid System with Zero-CO2 Emission. In Advances in Gas Turbine Technology; Benini, E., Ed.; InTech: London, UK, 2011; ISBN 978-953-307-611-9. [Google Scholar]
- Großmann, K.; Treiber, P.; Karl, J. Steam Methane Reforming at Low S/C Ratios for Power-to-Gas Applications. Int. J. Hydrogen Energy 2016, 41, 17784–17792. [Google Scholar] [CrossRef]
- General Information about CO2. Available online: https://www.ascoco2.com/us/co2-production-and-co2-recovery-plants/general-information-about-co2 (accessed on 24 April 2024).
- ISEP. The 4th Generation District Heating and Japan’s Thermal Policy; Institute for Sustainable Energy Policies: Kyoto, Japan, 2022. [Google Scholar]
- Sommer, T.; Mennel, S.; Sulzer, M. Lowering the Pressure in District Heating and Cooling Networks by Alternating the Connection of the Expansion Vessel. Energy 2019, 172, 991–996. [Google Scholar] [CrossRef]
- Lund, H.; Thorsen, J.E.; Jensen, S.S.; Madsen, F.P. Fourth-Generation District Heating and Motivation Tariffs. ASME Open J. Eng. 2022, 1, 011002. [Google Scholar] [CrossRef]
- Xu, J.; Froment, G.F. Methane Steam Reforming, Methanation and Water-gas Shift: I. Intrinsic Kinetics. AIChE J. 1989, 35, 88–96. [Google Scholar] [CrossRef]
- Pashchenko, D.; Mustafin, R.; Mustafina, A. Steam Methane Reforming in a Microchannel Reformer: Experiment, CFD-Modelling and Numerical Study. Energy 2021, 237, 121624. [Google Scholar] [CrossRef]
- Abbas, S.Z.; Dupont, V.; Mahmud, T. Kinetics Study and Modelling of Steam Methane Reforming Process over a NiO/Al2O3 Catalyst in an Adiabatic Packed Bed Reactor. Int. J. Hydrogen Energy 2017, 42, 2889–2903. [Google Scholar] [CrossRef]
- Hou, K.; Hughes, R. The Kinetics of Methane Steam Reforming over a Ni/α-Al2O Catalyst. Chem. Eng. J. 2001, 82, 311–328. [Google Scholar] [CrossRef]
- Wójcik, M.; Szabłowski, Ł.; Dybiński, O. Comparison of Mathematical Models of Steam Methane Reforming Process for the Needs of Fuel Cells. Int. J. Hydrogen Energy 2024, 52, 965–982. [Google Scholar] [CrossRef]
- Fan, L.; Mokhov, A.; Saadabadi, S.A.; Brandon, N.; Aravind, P.V. Methane Steam Reforming Reaction in Solid Oxide Fuel Cells: Influence of Electrochemical Reaction and Anode Thickness. J. Power Sources 2021, 507, 230276. [Google Scholar] [CrossRef]
- Yun, J.; Kim, Y.; Yu, S. Interactive Heat Transfer Characteristics of 5 kW Class Shell-and-Tube Methane Steam Reformer with Intermediate Temperature Heat Source. Int. J. Hydrogen Energy 2020, 45, 21767–21778. [Google Scholar] [CrossRef]
- Thermodynamics. Available online: https://www.doitpoms.ac.uk/tlplib/fuel-cells/sofc_thermodynamics.php (accessed on 16 April 2024).
- Liu, J.; Ma, C.; Zhu, Q.; Wang, H.; Huo, D.; Zhao, J. Study on Optimization Characteristics of Methanol Combustion Cooker Based on Porous Media. Case Stud. Therm. Eng. 2024, 58, 104269. [Google Scholar] [CrossRef]
- Hossan, Md.N.; Ahmed, M.M.; Siddhpura, M.; Ananno, A.A.; Masud, M.H. Methanol Production From Biogas: Current Status and Future Prospects. In Reference Module in Chemistry, Molecular Sciences and Chemical Engineering; Elsevier: Amsterdam, The Netherlands, 2023; p. B9780443157400000033. ISBN 978-0-12-409547-2. [Google Scholar]
- Wang, J.; Al-attab, K.A.; Yew Heng, T. Techno-Economic and Thermodynamic Analysis of Solid Oxide Fuel Cell Combined Heat and Power Integrated with Biomass Gasification and Solar Assisted Carbon Capture and Energy Utilization System. Energy Convers. Manag. 2023, 280, 116762. [Google Scholar] [CrossRef]
- 11 Gibbs Free Energy and Equilibrium. Available online: https://chem.libretexts.org/Courses/Grand_Rapids_Community_College/CHM_120_-_Survey_of_General_Chemistry(Neils)/7%3A_Equilibrium_and_Thermodynamics/7.11%3A_Gibbs_Free_Energy_and_Equilibrium (accessed on 24 April 2024).
- Gibbs (Free) Energy. Available online: https://chem.libretexts.org/Bookshelves/Physical_and_Theoretical_Chemistry_Textbook_Maps/Supplemental_Modules_(Physical_and_Theoretical_Chemistry)/Thermodynamics/Energies_and_Potentials/Free_Energy/Gibbs_(Free)_Energy (accessed on 24 April 2024).
- Carbon Monoxide. Available online: https://webbook.nist.gov/cgi/cbook.cgi?ID=C630080&Units=SI&Mask=1#Thermo-Gas (accessed on 17 April 2024).
- Carbon Dioxide. Available online: https://webbook.nist.gov/cgi/cbook.cgi?Source=1927LOW%2FERI2729-2734&Units=SI&Mask=7 (accessed on 17 April 2024).
- Mika, M.T. Synthesis of Carbide Derived Carbon by Reactive Anode Electrolysis in Molten Chloride Salts. Ph.D. Thesis, University of Illinois at Chicago, Chicago, IL, USA, 2018. [Google Scholar]
- Ozbek, B.B.; Aydın, H.; Merey, Ş. Ground Source Cooling to Increase Power Generation from Geothermal Power Plants. Energy 2024, 292, 130649. [Google Scholar] [CrossRef]
- Srivastava, M.; Sarkar, J.; Sarkar, A.; Maheshwari, N.K.; Antony, A. Thermo-Economic Feasibility Study to Utilize ORC Technology for Waste Heat Recovery from Indian Nuclear Power Plants. Energy 2024, 298, 131338. [Google Scholar] [CrossRef]
- Zhang, M.; Chen, H.; Zoghi, M.; Habibi, H. Comparison between Biogas and Pure Methane as the Fuel of a Polygeneration System Including a Regenerative Gas Turbine Cycle and Partial Cooling Supercritical CO2 Brayton Cycle: 4E Analysis and Tri-Objective Optimization. Energy 2022, 257, 124695. [Google Scholar] [CrossRef]
- Xu, L.; Chauhan, B.S.; Salah, B.; Thinh, P.-H. Development of an Innovative High-Performance Poly-Generation Plant for Efficient Multi-Level Heat Recovery of a Biogas-Powered Micro Gas Turbine: Environmental and Multi-Criteria Analysis. Process Saf. Environ. Prot. 2023, 178, 748–764. [Google Scholar] [CrossRef]
- Akay, S.S.; Özcan, O.; Özcan, O.; Yetemen, Ö. Efficiency Analysis of Solar Farms by UAV-Based Thermal Monitoring. Eng. Sci. Technol. Int. J. 2024, 53, 101688. [Google Scholar] [CrossRef]
- Nishinaga, J.; Kamikawa, Y.; Sugaya, T.; Ishizuka, S. Comparison of Polycrystalline and Epitaxial Cu(In, Ga)Se2 Solar Cells with Conversion Efficiencies of More than 21%. Sol. Energy Mater. Sol. Cells 2024, 269, 112791. [Google Scholar] [CrossRef]
- Hao, Q.; Zhu, L.; Fan, J.; Wang, Y.; Yang, Z.; Yang, H.; Huang, Y. Zero-Energy Penalty Carbon Capture and Utilization System Based on CLHG Integrating SOFC for Power and Methanol Cogeneration. Energy Convers. Manag. 2023, 295, 117658. [Google Scholar] [CrossRef]
- Chuahy, F.D.F.; Kokjohn, S.L. Solid Oxide Fuel Cell and Advanced Combustion Engine Combined Cycle: A Pathway to 70% Electrical Efficiency. Appl. Energy 2019, 235, 391–408. [Google Scholar] [CrossRef]
- Lim, T.-W.; Hwang, D.-H.; Choi, Y.-S. Design and Optimization of a Steam Methane Reformer for Ship-Based Hydrogen Production on LNG-Fueled Ship. Appl. Therm. Eng. 2024, 243, 122588. [Google Scholar] [CrossRef]
- Jiao, Y.; Wang, C.; Zhang, L.; An, W.; Zhou, N.; Yang, G.; Wang, W.; Zhou, W.; Li, S. A Steel Slag–Derived Boudouard Reaction Catalyst for Improved Performance of Direct Carbon Solid Oxide Fuel Cells. Int. J. Energy Res. 2019, 43, 6970–6982. [Google Scholar] [CrossRef]




| Kinetic parameters; represented by Arrhenius equation: | |||
| Reaction i | (kmol/(kgcat s)) | (kJ/mol) | |
| SMR | Pa0.5 | 240.10 | |
| WGS | Pa−1 | 67.13 | |
| DSMR | Pa0.5 | 243.90 | |
| Adsorption parameters; represented by Van’t Hoff equation: | |||
| Component j | (kJ/mol) | ||
| CO | Pa−1 | −70.65 | |
| H2 | Pa−1 | −82.90 | |
| CH4 | Pa−1 | −38.28 | |
| H2O | 88.68 | ||
| Reaction equilibria | |||
| Reaction i | Equilibrium equation | ||
| SMR | |||
| WGS | |||
| DSMR | |||
| SOFC Operating Temperature (°C) | Investigated Reformer Temperatures (°C) |
|---|---|
| 750 | 550, 575, 600 |
| 800 | 575, 600, 625, 650 |
| 850 | 600, 625, 650, 675, 700 |
| 900 | 625, 650, 675, 700, 725, 750 |
| 950 | 650, 700, 750, 800 |
| Parameter | Value | Unit |
|---|---|---|
| Biogas flowrate | 80.97 | kmol/h |
| CH4 fraction | 0.6175 | − |
| CO2 fraction | 0.3325 | − |
| H2O fraction | 0.0500 | − |
| Reformer inlet temperature | 684.42 | °C |
| Reformer CH4 conversion | 0.9233 | − |
| S/C | 2.5 | − |
| Reformer operating/SOFC inlet fuel temperature | 649.99 | °C |
| SOFC inlet air temperature | 650 | °C |
| SOFC exhaust temperature | 850 | °C |
| SOFC fuel utilisation | 0.70 | − |
| SOFC air utilisation | 0.1806 | − |
| Produced power in SOFC | 6821.81 | kW |
| Produced power in RC | 1568.72 | kW |
| SOFC electrical efficiency | 61.18 | % |
| System electrical efficiency | 67.70 | % |
| System overall efficiency | 71.64 | % |
| Refrigerant cycle COP | 3.55 | − |
| Flowrate of produced sanitary water | 11.66 | m3 |
| Number of possible consumer households | 231 | − |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gruber, S.; Rola, K.; Goričanec, D.; Urbancl, D. Fully Integrated Hybrid Solid Oxide Fuel Cell–Rankine Cycle System with Carbon Capture, Utilisation, and Storage for Sustainable Combined Heat and Power Production. Sustainability 2024, 16, 4389. https://doi.org/10.3390/su16114389
Gruber S, Rola K, Goričanec D, Urbancl D. Fully Integrated Hybrid Solid Oxide Fuel Cell–Rankine Cycle System with Carbon Capture, Utilisation, and Storage for Sustainable Combined Heat and Power Production. Sustainability. 2024; 16(11):4389. https://doi.org/10.3390/su16114389
Chicago/Turabian StyleGruber, Sven, Klemen Rola, Darko Goričanec, and Danijela Urbancl. 2024. "Fully Integrated Hybrid Solid Oxide Fuel Cell–Rankine Cycle System with Carbon Capture, Utilisation, and Storage for Sustainable Combined Heat and Power Production" Sustainability 16, no. 11: 4389. https://doi.org/10.3390/su16114389
APA StyleGruber, S., Rola, K., Goričanec, D., & Urbancl, D. (2024). Fully Integrated Hybrid Solid Oxide Fuel Cell–Rankine Cycle System with Carbon Capture, Utilisation, and Storage for Sustainable Combined Heat and Power Production. Sustainability, 16(11), 4389. https://doi.org/10.3390/su16114389






