Leaching Runoff Fraction for Nitrate and Herbicides on Sugarcane Fields: Implications for Grey Water Footprint
Abstract
1. Introduction
- (a)
- (b)
- Management practices can further reduce the fraction of diffuse solutes loaded into the environment through the leaching and runoff processes.
2. Materials and Methods
2.1. Trial Description and Used Treatments
2.2. Field and Lab Measurements
2.3. Runoff and Leaching Assessment
2.3.1. Leaching Runoff Fraction and Total Pollutant Losses from Field Experiments
2.3.2. Leaching Runoff Fraction According to Franke et al. [25]—Tier 1 Approach
2.3.3. Leaching Runoff Fraction According to PestLCI [26] Approach
2.4. Grey Water Footprint Assessment
2.5. Statistical Analysis
3. Results
3.1. Nitrate and Herbicide Pollutant Load on the Environment—Measurement
3.2. Leaching Runoff Fraction and GWF
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- CONAB. Acompanhamento da Safra Brasileira de Cana-de-Açúcar. In Segundo Levantamento—Safra 2021/2022; National Supply Company (CONAB): Brasilia, Brazil, 2023. Available online: http://www.conab.gov.br (accessed on 14 February 2023).
- Aquino, G.S.; Medina, C.C.; Shahab, M.; Santiago, A.D.; Cunha, A.C.B.; Kussaba, D.A.O.; Carvalho, J.B.; Moreira, A. Does straw mulch partial-removal from soil interfere in yield and industrial quality sugarcane? A long term study. Ind. Crops Prod. 2018, 111, 573–578. [Google Scholar] [CrossRef]
- Moutinho, M.F.; de Almeida, E.A.; Espindola, E.L.G.; Daam, M.A.; Schiesari, L. Herbicides employed in sugarcane plantations have lethal and sublethal effects to larval Boana pardalis (Amphibia, Hylidae). Ecotoxicology 2020, 29, 1043–1051. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Ma, L.; Ma, W.; Wu, Z.; Cui, Z.; Hou, Y.; Zang, F. What has caused the use of fertilizers to skyrocket in China? Nutr. Cycl. Agroecosyst. 2018, 110, 241–255. [Google Scholar] [CrossRef]
- Heffer, P.; Gruère, A.; Roberts, T. Assessment of Fertilizer Use by Crop at the Global Level; International Fertilizer Association and International Plant Nutrition Institute: Paris, France, 2013; pp. 1–18. [Google Scholar]
- Thorburn, P.J.; Meier, E.A.; Probert, M.E. Modelling nitrogen dynamics in sugarcane systems: Recent advances and applications. Field Crop Res. 2005, 92, 337–351. [Google Scholar] [CrossRef]
- Kaur, S.; Kaur, R.; Chauhan, B.S. Understanding crop-weed-fertilizer-water interactions and their implications for weed management in agricultural systems. J. Crop Prot. 2018, 103, 65–72. [Google Scholar] [CrossRef]
- Vale, R.L.; Maciel Netto, A.; Xavier, B.T.L.; Barreto, M.L.P.; Silva, J.P.S. Assessment of the gray water footprint of the pesticide mixture in a soil cultivated with sugarcane in the northern area of the State of Pernambuco, Brazil. J. Clean. Prod. 2019, 234, 925–932. [Google Scholar] [CrossRef]
- Reis, F.C.; Victória Filho, R.; Andrade, M.T.; Barroso, A.A.M. Use of herbicides in sugarcane in the São Paulo State. Planta Daninha 2019, 37, e019184227. [Google Scholar] [CrossRef]
- Martinelli, L.A.; Filoso, S. Expansion of sugarcane ethanol production in Brazil: Environmental and social challenges. Ecol. Appl. 2008, 18, 885–898. [Google Scholar] [CrossRef]
- Guimarães, A.C.D.; Mendes, K.F.; Campion, T.F.; Christoffoleti, P.J.; Tornisielo, V.L. Leaching of herbicides commonly applied to sugarcane in five agricultural soils. Planta Daninha 2019, 37, e019181505. [Google Scholar] [CrossRef]
- Santos, E.A. Contaminação por Herbicidas em Corpos Hídricos da Microbacia do Córrego Rico (SP) e Aspectos Toxicológicos de Atrazine a Juvenis de Piaractus Mesopotamicus. Master’s Thesis, University of São Paulo, Jaboticabal, Brazil, 3 May 2013. [Google Scholar]
- Queiroz, S.C.N.; Ferracini, V.L.; Gomes, M.A.F.; Rosa, M.A. Comportamento do herbicida hexazinone em área de recarga do aqüífero Guarani cultivada com cana-de-açúcar. Quím. Nova 2009, 32, 378–381. [Google Scholar] [CrossRef]
- Bordonal, R.O.; Carvalho, J.L.N.; Lal, R.; Figueiredo, E.B.; Oliveira, B.G.; Scala, N., Jr. Sustainability of sugarcane production in Brazil. A review. Agron. Sustain. Dev. 2018, 38, e13. [Google Scholar] [CrossRef]
- Marin-Morales, M.A.; Ventura-Camargo, B.C.; Hoshina, M.M. Toxicity of herbicides: Impact on aquatic and soil biota and human health. In Herbicides-Current Research and Case Studies in Use; Price, A., Kelton, J.A., Eds.; In Tech: Zagreb, Croatia, 2013; pp. 399–443. [Google Scholar] [CrossRef]
- Sebilo, M.; Mayer, B.; Nicolardot, B.; Pinay, G.; Mariotti, A. Long-term fate of nitrate fertilizer in agricultural soils. Proc. Natl. Acad. Sci. USA 2013, 110, 18185–18189. [Google Scholar] [CrossRef] [PubMed]
- Van der Laan, M.; Annandale, J.G.; Bristow, K.L.; Stirzaker, R.J.; Preez, C.C.du.; Thorburn, P.J. Modelling nitrogen leaching: Are we getting the right answer for the right reason? Agric. Water Manag. 2014, 133, 74–80. [Google Scholar] [CrossRef]
- Stewart, L.K.; Charlesworth, P.B.; Bristow, K.L.; Thorburn, P.J. Estimating deep drainage and nitrate leaching from the root zone under sugarcane using APSIM-SWIM. Agric. Water Manag. 2006, 81, 315–334. [Google Scholar] [CrossRef]
- Barreto, M.L.P.; Maciel Netto, A.; Silva, J.P.S.; Amaral, A.; Borges, E.; França, E.J.; Vale, R.L. Gray water footprint assessment for pesticide mixtures applied to a sugarcane crop in Brazil: A comparison between two models. J. Clean. Prod. 2020, 276, e124254. [Google Scholar] [CrossRef]
- Rodriguez, R.; Scanlon, B.R.; King, C.W.; Scarpare, F.V.; Xavier, A.C.; Pruski, F.F. Biofuel-water-land nexus in the last agricultural frontier region of the Brazilian Cerrado. Appl. Energy 2018, 231, 1330–1345. [Google Scholar] [CrossRef]
- Scarpare, F.V.; Hernandes, T.A.D.; Ruiz-Corrêa, S.T.; Kolln, O.T.; Gava, G.J.C.; Santos, L.N.S.; Victoria, R.L. Sugarcane water footprint under different management practices in Brazil: Tietê/Jacaré watershed assessment. J. Clean. Prod. 2016, 112, 4576–4584. [Google Scholar] [CrossRef]
- Mekonnen, M.M.; Hoekstra, A.Y. The green, blue, and grey water footprint of crops and derived crop products. Hydrol. Earth Syst. Sci. 2010, 15, 1577–1600. [Google Scholar] [CrossRef]
- Hoekstra, A.Y. Virtual Water Trade. In Proceedings of the International Expert Meeting on Virtual Water Trade, Delft, The Netherlands, 12–13 December 2002; IHE—International Institute for Hydraulic and Environmental Engineering: Deft, The Netherlands, 2003. [Google Scholar]
- Jamshidi, S. Grey Water Footprint Accounting, Challenges, and Problem-Solving. In Agroecological Footprints Management for Sustainable Food System; Banerjee, A., Meena, R.S., Jhariya, M.K., Yadav, D.K., Eds.; Springer: Singapore, 2021. [Google Scholar] [CrossRef]
- Franke, N.A.; Boyacioglu, H.; Hoekstra, A.Y. Grey Water Footprint Accounting: Tier 1 Supporting Guidelines; UNESCO-IHE Institute for Water Education: Delft, The Netherlands, 2013; pp. 1–57. [Google Scholar]
- Dijkman, T.; Birkved, M.; Hauschild, M. PestLCI 2.0: A second generation model for estimating emissions of pesticides from arable land in LCA. Int. J. Life Cycle Assess. 2012, 17, 973–986. [Google Scholar] [CrossRef]
- CONAB (Companhia Nacional de Abastecimento). Acompanhamento da Safra Brasileira de Cana-de-Açúcar na Safra 2019/20. Quarto Levantamento; 2020. Available online: https://www.conab.gov.br/info-agro/safras/cana (accessed on 9 September 2020).
- CANASAT. Cane Mapping via Earth Observation Satellite Images (Mapeamento de Cana via Imagens de Satélite de Observação da Terra). Tables. Available online: http://www.dsr.inpe.br/laf/canasat/en/ (accessed on 16 May 2015.).
- Castro, S.G.Q.; Decaro, S.T.; Franco, H.C.J.; Magalhães, P.S.G.; Garside, A.; Mutton, M.A. Best practices of nitrogen fertilization management for sugarcane under green cane trash blanket in Brazil. Sugar Tech. 2016, 19, 51–56. [Google Scholar] [CrossRef]
- Van Raij, B.; Cantarella, H.; Quaggio, J.A.; Furlani, A.M.C. Recomendações de Adubacão e Calagem para o Estado de São Paulo, 2nd ed.; Instituto Agronômico/Fundação IAC: Campinas, Brazil, 2007.
- Scarpare, F.V.; Jong van Lier, Q.; Camargo, L.; Pires, R.C.M.; Ruiz-Corrêa, S.T.; Gava, G.J.C.; Bezerra, A.H.F.; Dias, C.T.S. Tillage assessment on soil physical properties associated with sugarcane water availability. Soil Tillage Res. 2019, 187, 110–118. [Google Scholar] [CrossRef]
- CETESB (Companhia Ambiental do Estado de São Paulo). Guia Nacional de Coleta e Preservação de Amostras; CETESB/ANA: São Paulo, Brasília, 2011. Available online: https://cetesb.sp.gov.br/wp-content/uploads/2021/10/Guia-nacional-de-coleta-e-preservacao-de-amostras-2012.pdf (accessed on 24 February 2016).
- American Public Health Association (APHA). Standard Methods for the Examination of Water and Wastewater, 22nd ed.; Rice, E.W., Baird, R.B., Eaton, A.D., Clesceri, L.S., Eds.; American Public Health Association (APHA): Washington, DC, USA; American Water Works Association (AWWA): Washington, DC, USA; Water Environment Federation (WEF): Washington, DC, USA, 2012. [Google Scholar]
- Armstrong, F.A.; Stearns, C.R.; Strickland, J.D. The measurement of upwelling and subsequent biological processes by means of the Technicon Autoanalyzer and associated equipment. Deep-Sea Res. 1967, 14, 381–389. [Google Scholar] [CrossRef]
- Van Genuchten, M.T. A closed-form equation for predicting the hydraulic conductivity of unsatured soils. Soil Sci. Soc. Am. J. 1980, 44, 892–898. [Google Scholar] [CrossRef]
- Van Dam, J.C.; Groenendijk, P.; Hendriks, R.F.A.; Kroes, J.G. Advances of modeling water flow in variably saturated soils with SWAP. Vadose Zone J. 2008, 7, 640–653. [Google Scholar] [CrossRef]
- Kroes, J.G.; van Dam, J.C.; Groenendijk, P.; Hendriks, R.F.A.; Jacobs, C.M.J. SWAP Version 3.2. Theory Description and User Manual; Alterra: Wageningen, The Netherlands, 2008; pp. 147–172. [Google Scholar]
- van Diepen, C.A.; Rappoldt, C.; Wolf, J.; van Keulen, H. Crop Growth Simulation Model WOFOST. Documentation Version 4.1; Centre for World Food Studies: Wageningen, The Netherlands, 1988; 299p. [Google Scholar]
- Scarpare, F.V. Simulação do crescimento da cana-de-açúcar pelo modelo agrohidrológico SWAP/WOFOST. Ph.D. Thesis, University of São Paulo, Piracicaba, Brazil, 31 August 2011. [Google Scholar]
- Bezerra, A.H.F.; Scarpare, F.V.; de Jong van Lier, Q. Optimizing hydraulic conductivity parameters with field measurements. In Proceedings of the 4th Brazilian Soil Physics Meeting, Piracicaba, Brazil, 30 May 2017. [Google Scholar]
- Doherty, J. PEST: Model Independent Parameter Estimation; Watermark Numerical Computing: Corinda, Australia, 2002. [Google Scholar]
- Hoekstra, A.Y.; Chapagain, A.K.; Aldaya, M.M.; Mekonnen, M.M. The Water Footprint Assessment Manual: Setting the Global Standard; Earthscan: London, UK, 2011; pp. 19–68. [Google Scholar]
- Hammer, O.; Harper, D.A.T.; Ryan, P.D. PAST: Paleontological Statistics Software Package for Education and Data Analysis. Palaeontol. Electron. 2001, 4, 1–9. [Google Scholar]
- Box, G.E.; Cox, D.R. An analysis of transformations. J. R. Stat. Soc. Ser. B Stat. Methodol. 1964, 26, 211–252. [Google Scholar] [CrossRef]
- CONAMA (National Environmental Council). Resolução CONAMA 430, de 17 de Março de 2005. 2005. Available online: http://www2.mma.gov.br/port/conama/legiabre.cfm?codlegi=459 (accessed on 2 February 2016).
- Ghiberto, P.J.; Libardi, P.L.; Trivelin, P.C.O. Nutrient leaching in an Ultisol cultivated with sugarcane. Agric. Water Manag. 2015, 148, 141–149. [Google Scholar] [CrossRef]
- Blum, J.; Melfi, A.J.; Montes, C.R. Nutrição mineral da cana-de-açúcar irrigada com efluente de esgoto tratado, em área com aplicação de fosfogesso. Pesq. Agropec. Bras. 2012, 47, 593–602. [Google Scholar]
- Tomer, M.D.; Burkart, M.R. Long-term effects of nitrogen fertilizer use on ground water nitrate in two small watersheds. J. Environ. Qual. 2003, 32, 2158–2171. [Google Scholar] [CrossRef]
- Ghanem, M.; Jebreen, H.; Lange, J. Qualitative spring water management in the central western catchment of the West Bank from hydrochemical and environmental isotopes approaches. Arab. J. Geosci. 2016, 9, 283. [Google Scholar] [CrossRef]
- Nachimuthu, G.; Halpin, N.V.; Bell, M.J. Effect of sugarcane cropping systems on herbicide losses in surface runoff. Sci. Total Environ. 2016, 557–558, 773–784. [Google Scholar] [CrossRef] [PubMed]
- Masters, B.; Rohde, K.; Gurner, N.; Reid, D. Reducing the risk of herbicide runoff in sugarcane farming through controlled traffic and early-banded application. Agric. Ecosyst. Environ. 2013, 180, 29–39. [Google Scholar] [CrossRef]
- Lewis, K.A.; Tzilivakis, J.; Warner, D.; Green, A. An international database for pesticide risk assessments and management. Hum. Ecol. Risk. Assess. 2016, 22, 1050–1064. [Google Scholar] [CrossRef]
- Giacomazzi, S.; Cochet, N. Environmental impact of diuron transformation: A review. Chemosphere 2004, 56, 1021–1032. [Google Scholar] [CrossRef]
- Birkved, M.; Hauschild, M.Z. PestLCI: A model for estimating field emissions of pesticides in agricultural LCA. Ecol. Model. 2006, 198, 433–451. [Google Scholar] [CrossRef]
- Fantin, V.; Buscaroli, A.; Djikman, T.; Zamagni, A.; Garavini, G.; Bonoli, A.; Righi, S. PestLCI 2.0 sensitivity to soil variations for the evaluation of pesticide distribution in Life Cycle Assessment studies. Sci. Total Environ. 2019, 656, 1021–1031. [Google Scholar] [CrossRef]
- Gentil, C.; Fantke, P.; Mottes, C.; Basset-Mens, C. Challenges and ways forward in pesticide emission and toxicity characterization modeling for tropical conditions. Int. J. Life Cycle Assess. 2020, 25, 1290–1306. [Google Scholar] [CrossRef]
- Otto, R.; Castro, S.A.Q.; Mariano, E.; Castro, S.G.Q.; Franco, H.C.J.; Trivelin, P.C.O. Nitrogen use efficiency for sugarcane-biofuel production: What is next? Bioenerg. Res. 2016, 9, 1272–1289. [Google Scholar] [CrossRef]
- Muratoglu, A. Grey water footprint of agricultural production: An assessment based on nitrogen surplus and high-resolution leaching runoff fractions in Turkey. Sci. Total Environ. 2020, 2020, 742. [Google Scholar] [CrossRef]
- Gmach, M.R.; Scarpare, F.V.; Cherubin, M.R.; Lisboa, I.P.; Santos, A.K.B.; Cerri, C.E.P.; Cerri, C.C. Sugarcane straw removal effects on soil water storage and drainage in Southeastern Brazil. J. Soil Water Conserv. 2019, 74, 466–476. [Google Scholar] [CrossRef]
- Ruiz-Corrêa, S.T.; Barbosa, L.C.; Menandro, L.M.S.; Scarpare, F.V.; Reichardt, K.; Moraes, L.O.; Hernandes, T.A.D.; Franco, H.J.; Carvalho, J.L.N. Straw removal effects on soil water dynamics, soil temperature, and sugarcane yield in south-central Brazil. BioEnergy Res. 2019, 12, 749–763. [Google Scholar] [CrossRef]
- Motasim, A.M.; Samsuri, A.W.; Sukor, A.S.A.; Adibah, A.M. Nitrogen dynamics in tropical soils treat with liquid and granular urea fertilizers. Agriculture 2021, 11, 546. [Google Scholar] [CrossRef]
- Smith, D.M.; Inman-Bamber, N.G.; Thorburn, P.J. Growth and function of the sugarcane root system. Field Crop. Res. 2005, 92, 169–183. [Google Scholar] [CrossRef]
- Laclau, P.B.; Laclau, J.P. Growth of the whole root system for a plant crop of sugarcane under rainfed and irrigated environments in Brazil. Field Crop. Res. 2009, 114, 351–360. [Google Scholar] [CrossRef]
- Prado, H.; Prado, T.A.B. Pedologia Facil: Aplicaçoes em Solos Tropicais, 6th ed.; Editora UFLA—Universidade Federal de Lavras: Lavras, Brazil, 2022; pp. 229–259. [Google Scholar]
- Silva, J.F.; Carvalho, A.M.; Rein, T.A.; Coser, T.R.; Ribeiro Junior, W.Q.; Vieira, D.L.; Coome, D.A. Nitrous oxide emissions from sugarcane fields in the Brazilian Cerrado. Agric Ecosyst Environ. 2017, 246, 55–56. [Google Scholar] [CrossRef]
- Rachid, C.T.; Piccolo, M.C.; Leite, D.C.A.; Balieiro, F.C.; Coutinho, H.L.C.; van Elsas, J.D.; Peixoto, R.S.; Rosado, A.S. Physical-chemical and microbiological changes in Cerrado soil under differing sugarcane harvest management systems. BMC Microbiol. 2012, 12, 2–11. [Google Scholar] [CrossRef]
- Robinson, N.; Brackin, R.; Vinall, K.; Soper, F.; Holst, J.; Gamage, H.; Paungfoo-Lohienne, C.; Renneberg, H.; Lakshmanan, P.; Schmidt, S. Nitrate paradigm does not hold up for sugarcane. PLoS ONE 2011, 6, e19045. [Google Scholar] [CrossRef]
- Spironello, A.; van Raij, B.; Penatti, C.P.; Cantarella, H.; Morelli, J.L.; Orlando Filho, J.; Landell, M.G.A.; Rossetto, R. Cana-de-açúcar. In Technical Report 100—Fertilization and Liming Recommendations for São Paulo State, 3rd ed.; van Raij, B., Cantarella, H., Quaggio, J., Furlani, A.M.C., Eds.; Instituto Agronômico de Campinas: Campinas, Brazil, 1996; pp. 237–239. [Google Scholar]
- Barbosa, E.A.A.; Matsura, E.E.; dos Santos, L.N.S.; Gonçalves, I.Z.; Nazário, A.A.; Feitosa, D.R.C. Water footprint of sugarcane irrigated with treated sewage and freshwater under subsurface drip irrigation, in Southeast Brazil. J. Clean. Prod. 2017, 153, 448–456. [Google Scholar] [CrossRef]
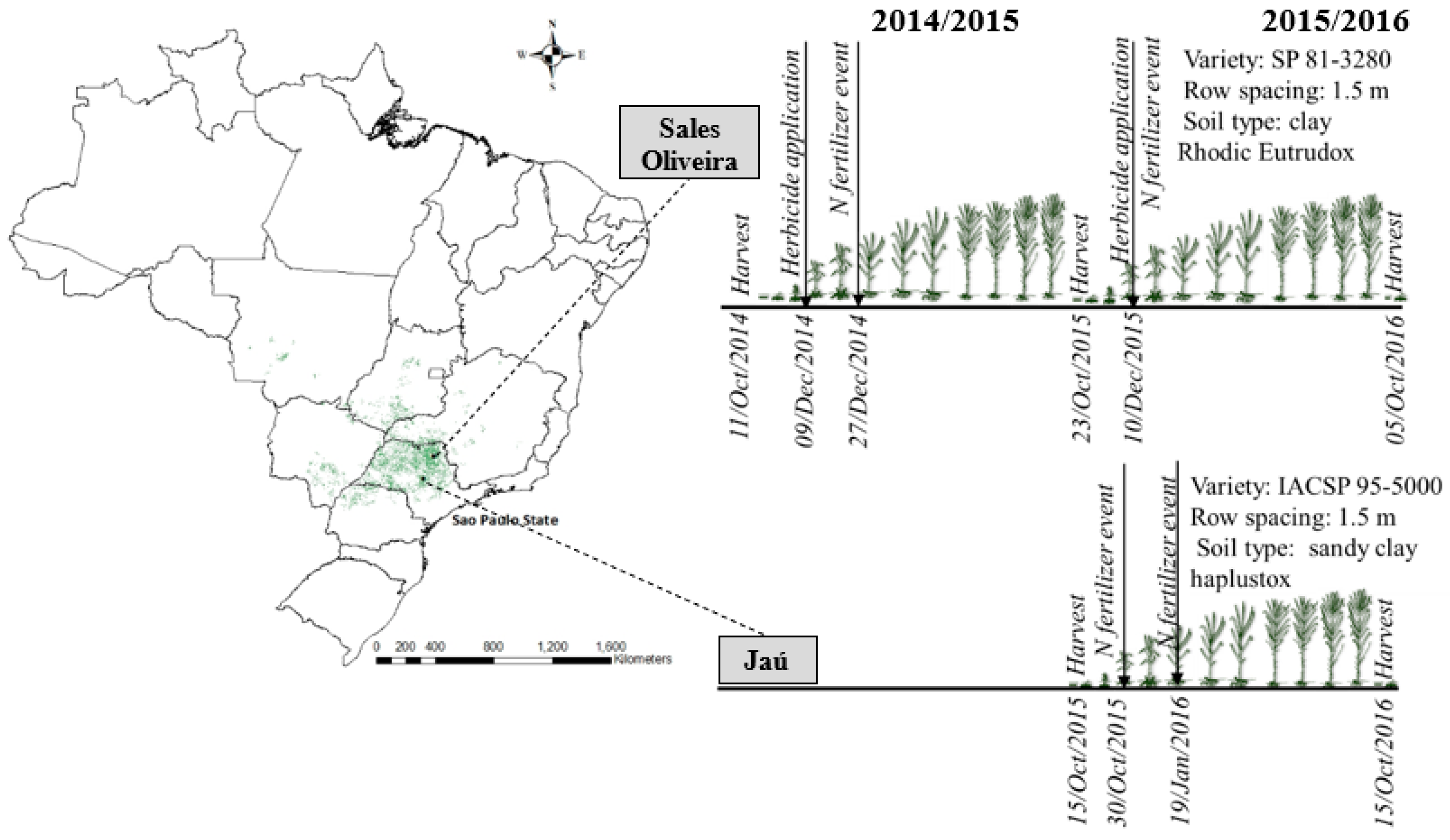

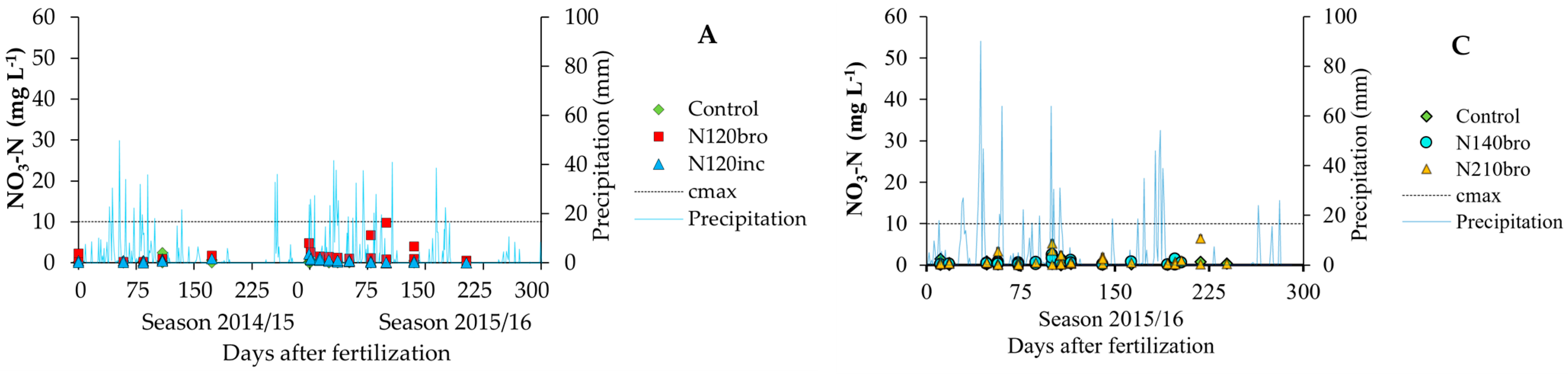
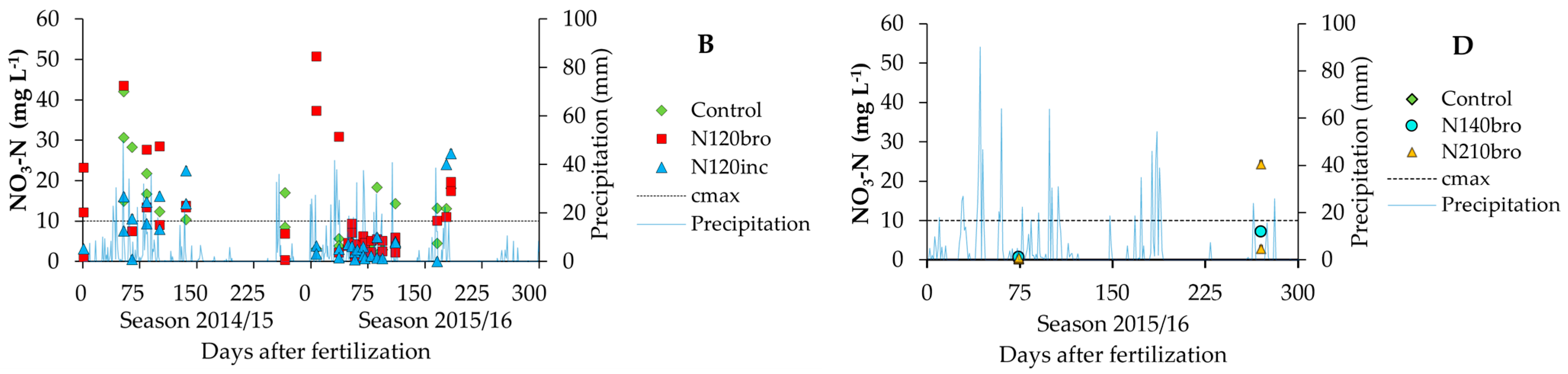
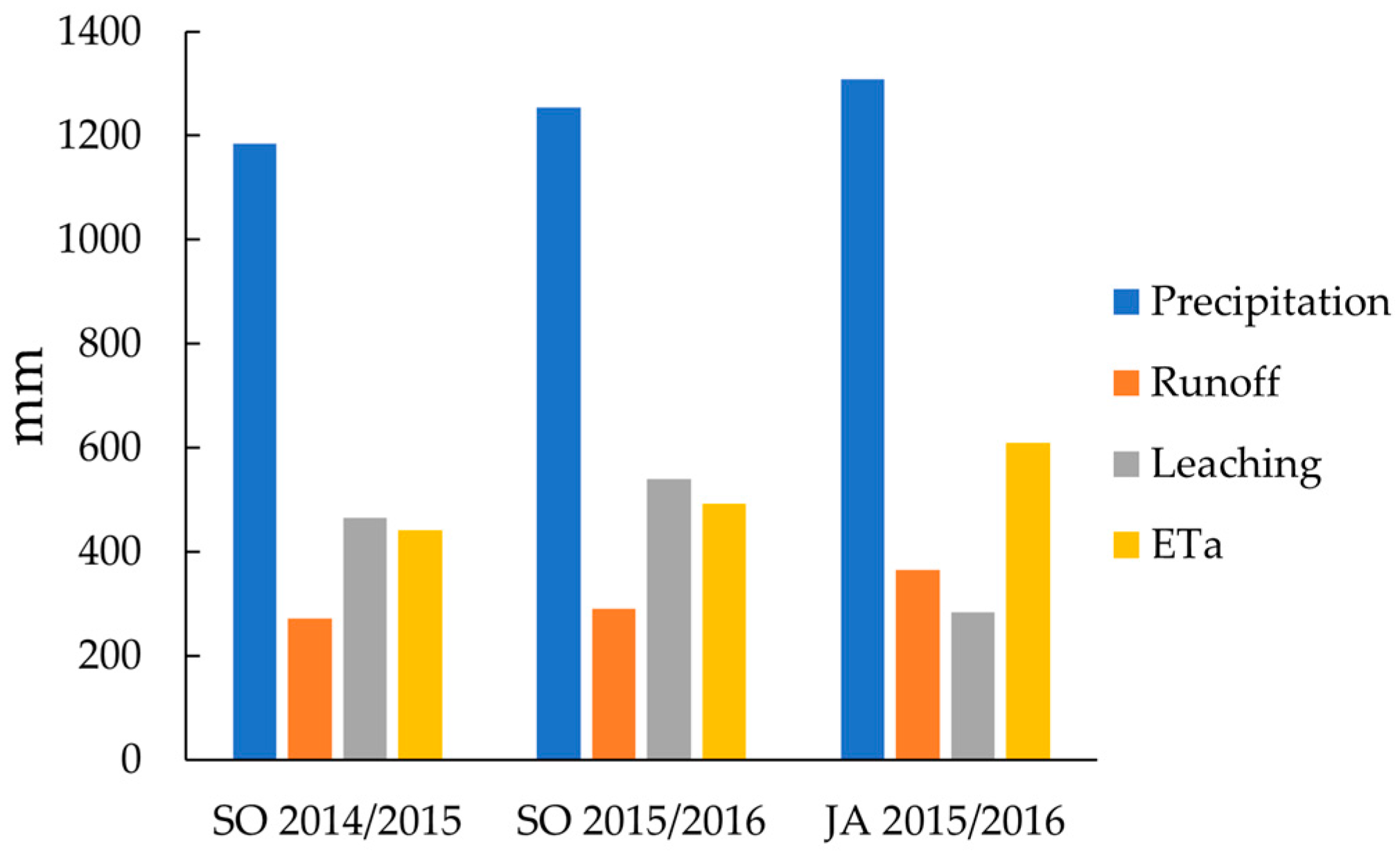
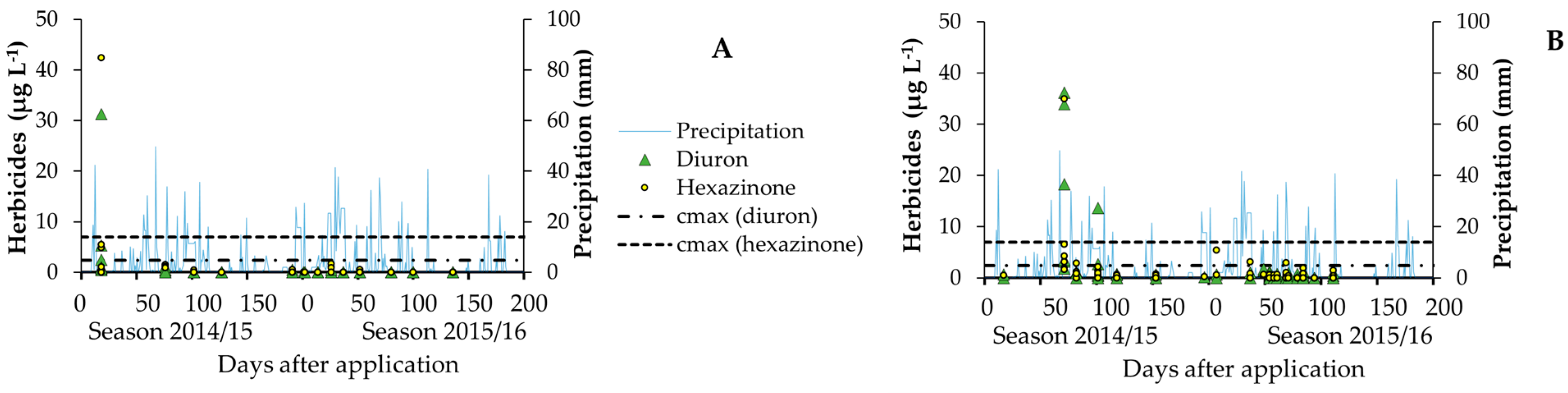
| Soil Layer (m) | SO—Clay Rhodic Eutrudox (Slope 9%) | JA—Sandy-Clay Haplustox (Slope 6%) | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Clay | Silt | Sand | BD * | MaP ** | MiP *** | Clay | Silt | Sand | BD * | MaP ** | MiP *** | |
| g kg−1 | kg m−3 | m3 m−3 | g kg−1 | kg m−3 | m3 m−3 | |||||||
| 0–0.2 | 687 | 244 | 69 | 1.22 | 0.40 | 0.14 | 362 | 58 | 581 | 1.67 | 0.11 | 0.31 |
| 0.2–0.4 | 718 | 214 | 68 | 1.21 | 0.41 | 0.13 | 434 | 55 | 512 | 1.53 | 0.15 | 0.31 |
| 0.4–1.0 | 700 | 235 | 66 | 1.13 | 0.38 | 0.20 | 399 | 49 | 552 | 1.48 | 0.12 | 0.32 |
| Trial sites | SO | JA | ||||
| Treatments | N120bro | N120inc | Nzero | N210bro | N140bro | Nzero |
| N-NO3 losses (kg ha−1) | ||||||
| Leaching | 7.2a ± 2.3 | 1.9b ± 0.3 | 1.0bc ± 0.2 | 1.4A ± 0.3 | 0.7AB ± 0.05 | 0.6B ± 0.2 |
| Runoff | 18.2a ± 2.0 | 10.0a ± 0.7 | 13.8a ± 1.7 | 12.1A ± 0.01 | 7.3A ± 0.01 | 1.8A ± 0.01 |
| Herbicide losses in SO trial (g ha−1) | ||||||
| Leaching | Runoff | Total | ||||
| Diuron | 4.4 ± 4.0 | 4.2 ± 1.7 | 8.6 ± 4.7 | |||
| Hexazinone | 7.1 ± 5.5 | 3.0 ± 1.0 | 10.1 ± 5.7 | |||
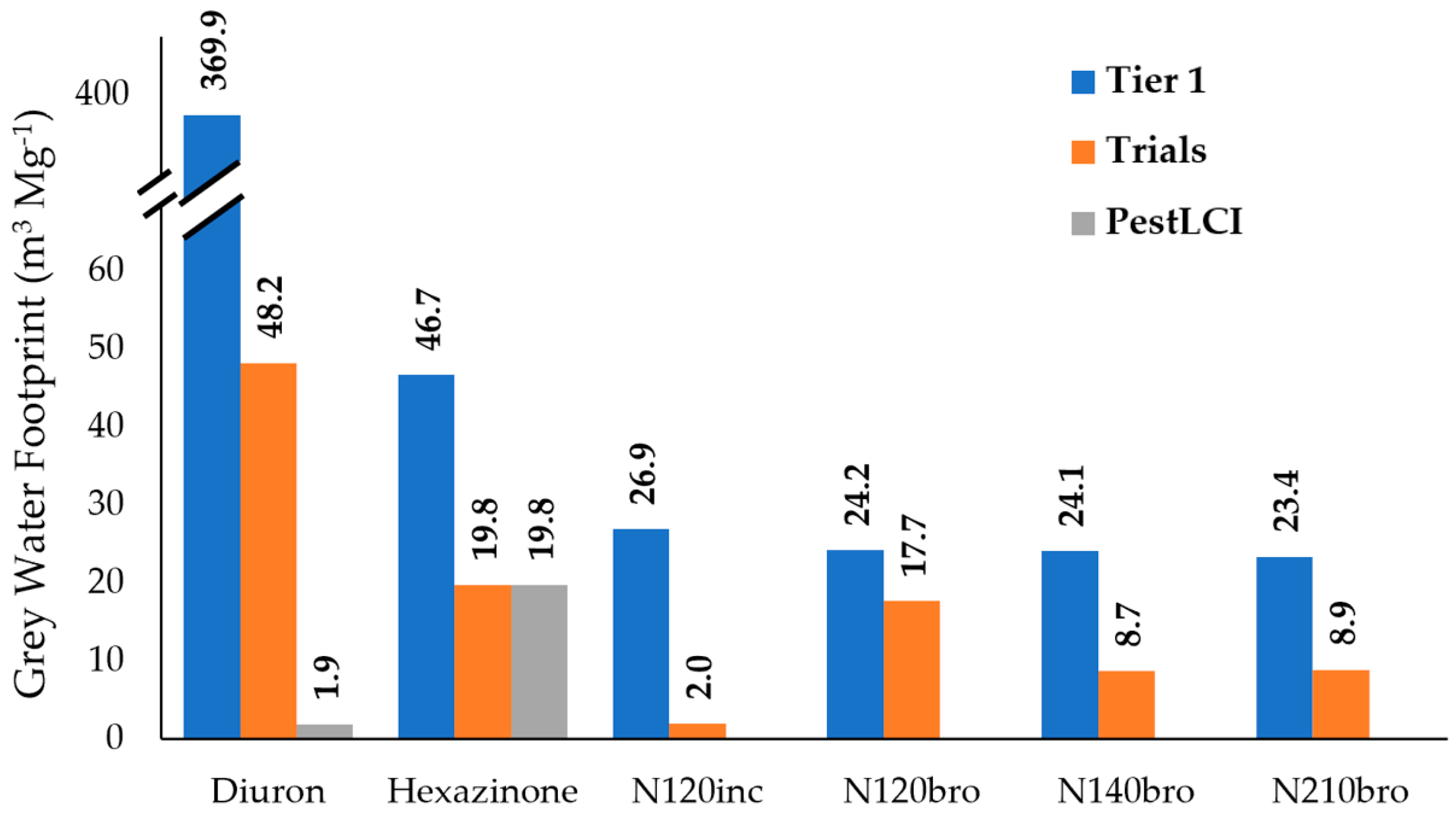
| Field | Season | Treatment | Source/Methodology | αrunoff (%) | αleaching (%) | αtotal (%) |
|---|---|---|---|---|---|---|
| Nitrate | ||||||
| SO | 2014/2015 | N120bro | Field trial | 2.47 | 1.90 | 4.37 |
| SO | 2014/2015 | N120inc | Field trial | 0.00 | 0.50 | 0.50 |
| SO | 2014/2015 | N120bro | Franke et al. [25] | -- | -- | 12.20 |
| SO | 2014/2015 | N120inc | Franke et al. [25] | -- | -- | 12.20 |
| SO | 2015/2016 | N120bro | Field trial | 4.87 | 8.40 | 13.27 |
| SO | 2015/2016 | N120inc | Field trial | 0.34 | 0.93 | 1.27 |
| SO | 2015/2016 | N120bro | Franke et al. [25] | -- | -- | 13.42 |
| SO | 2015/2016 | N120inc | Franke et al. [25] | -- | -- | 13.42 |
| JA | 2015/2016 | N140bro | Field trial | 5.21 | 0.10 | 5.31 |
| JA | 2015/2016 | N210bro | Field trial | 5.78 | 0.38 | 6.16 |
| JA | 2015/2016 | N140bro | Franke et al. [25] | -- | -- | 14.62 |
| JA | 2015/2016 | N210bro | Franke et al. [25] | -- | -- | 16.23 |
| Diuron | ||||||
| SO | 2014/2015 | 1.17 kg ha−1 | Field trial | 0.63 | 0.71 | 1.34 |
| SO | 2014/2015 | 1.17 kg ha−1 | Franke et al. [25] | -- | -- | 4.84 |
| SO | 2014/2015 | 1.17 kg ha−1 | PestLCI [26] | -- | -- | 0.03 |
| SO | 2015/2016 | 1.17 kg ha−1 | Field trial | 0.08 | 0.04 | 0.12 |
| SO | 2015/2016 | 1.17 kg ha−1 | Franke et al. [25] | -- | -- | 5.01 |
| SO | 2015/2016 | 1.17 kg ha−1 | PestLCI [26] | -- | -- | 0.03 |
| Hexazinone | ||||||
| SO | 2014/2015 | 0.33 kg ha−1 | Field trial | 1.25 | 4.22 | 5.47 |
| SO | 2014/2015 | 0.33 kg ha−1 | Franke et al. [25] | -- | -- | 6.34 |
| SO | 2014/2015 | 0.33 kg ha−1 | PestLCI [26] | -- | -- | 0.17 |
| SO | 2015/2016 | 0.33 kg ha−1 | Field trial | 0.57 | 0.10 | 0.67 |
| SO | 2015/2016 | 0.33 kg ha−1 | Franke et al. [25] | -- | -- | 6.52 |
| SO | 2015/2016 | 0.33 kg ha−1 | PestLCI [26] | -- | -- | 0.17 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Scarpare, F.V.; Zotelli, L.d.C.; Barizon, R.; Castro, S.G.Q.d.; Bezerra, A.H.F. Leaching Runoff Fraction for Nitrate and Herbicides on Sugarcane Fields: Implications for Grey Water Footprint. Sustainability 2023, 15, 6990. https://doi.org/10.3390/su15086990
Scarpare FV, Zotelli LdC, Barizon R, Castro SGQd, Bezerra AHF. Leaching Runoff Fraction for Nitrate and Herbicides on Sugarcane Fields: Implications for Grey Water Footprint. Sustainability. 2023; 15(8):6990. https://doi.org/10.3390/su15086990
Chicago/Turabian StyleScarpare, Fabio Vale, Luciana do Carmo Zotelli, Robson Barizon, Sergio Gustavo Quassi de Castro, and Andre Herman Freire Bezerra. 2023. "Leaching Runoff Fraction for Nitrate and Herbicides on Sugarcane Fields: Implications for Grey Water Footprint" Sustainability 15, no. 8: 6990. https://doi.org/10.3390/su15086990
APA StyleScarpare, F. V., Zotelli, L. d. C., Barizon, R., Castro, S. G. Q. d., & Bezerra, A. H. F. (2023). Leaching Runoff Fraction for Nitrate and Herbicides on Sugarcane Fields: Implications for Grey Water Footprint. Sustainability, 15(8), 6990. https://doi.org/10.3390/su15086990






