Outplants of the Threatened Coral Acropora cervicornis Promote Coral Recruitment in a Shallow-Water Coral Reef, Culebra, Puerto Rico
Abstract
:1. Introduction
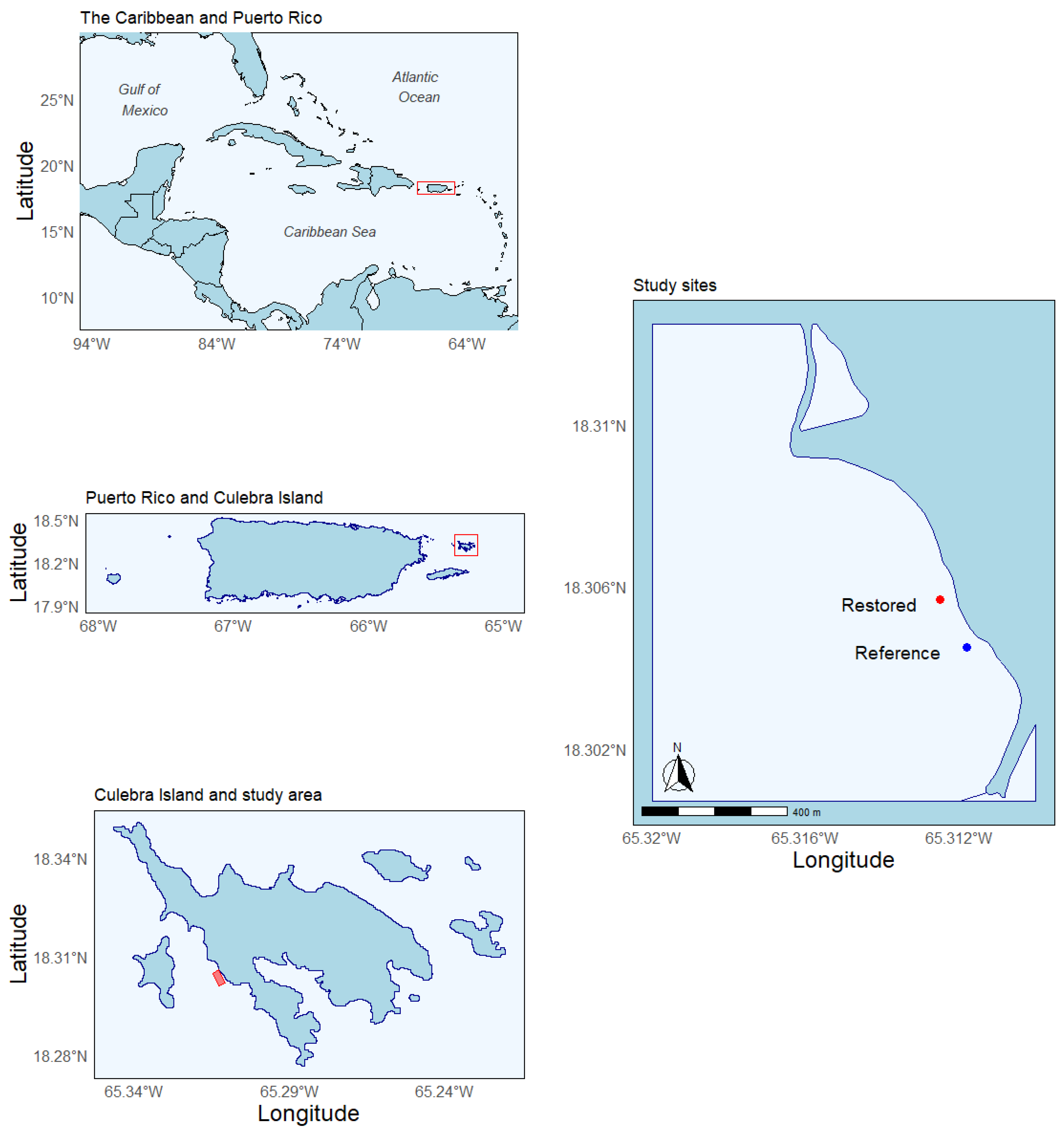
2. Study Site
3. Data and Methods
3.1. Experimental Approach
3.2. Statistical Analyses
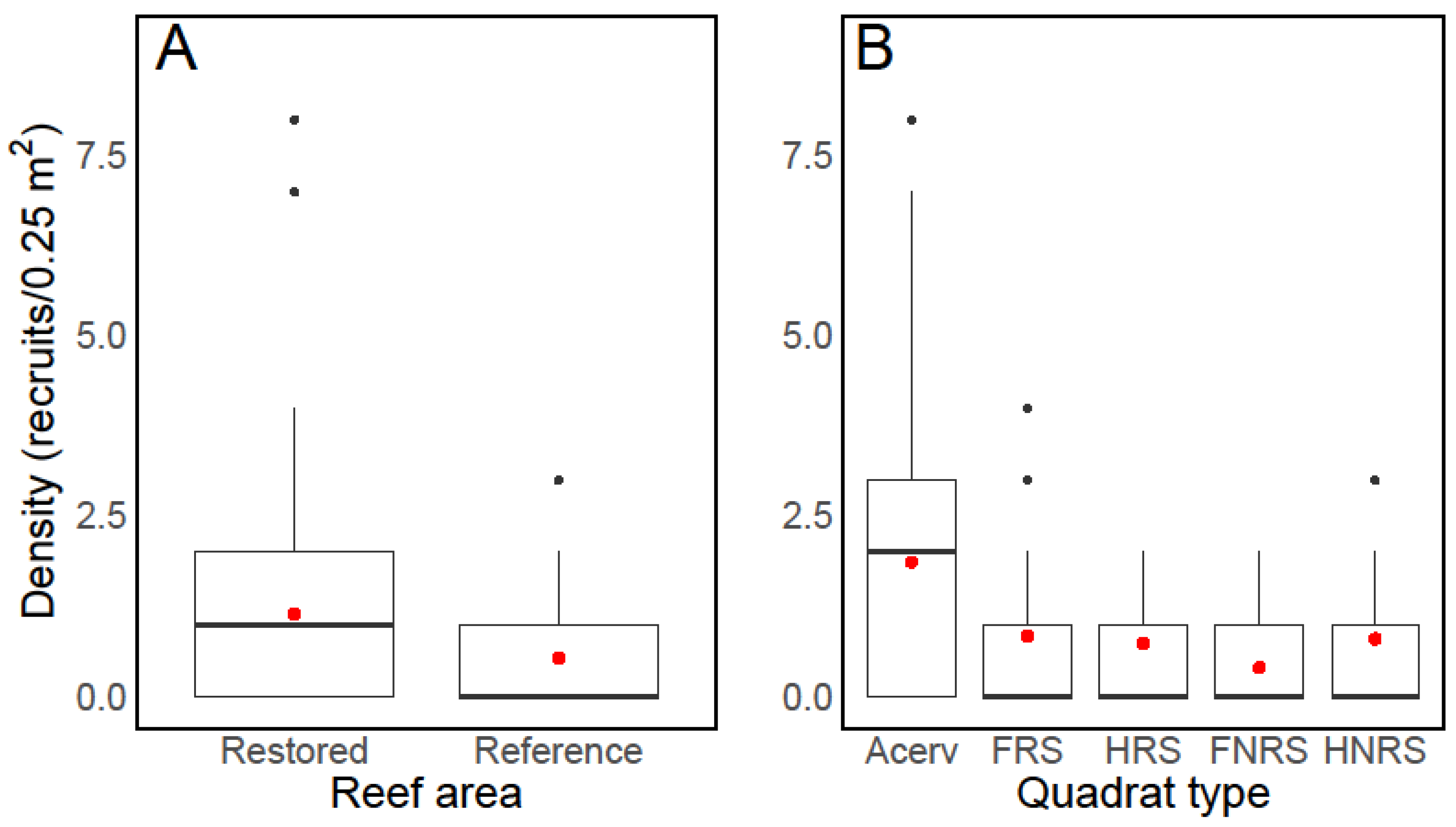
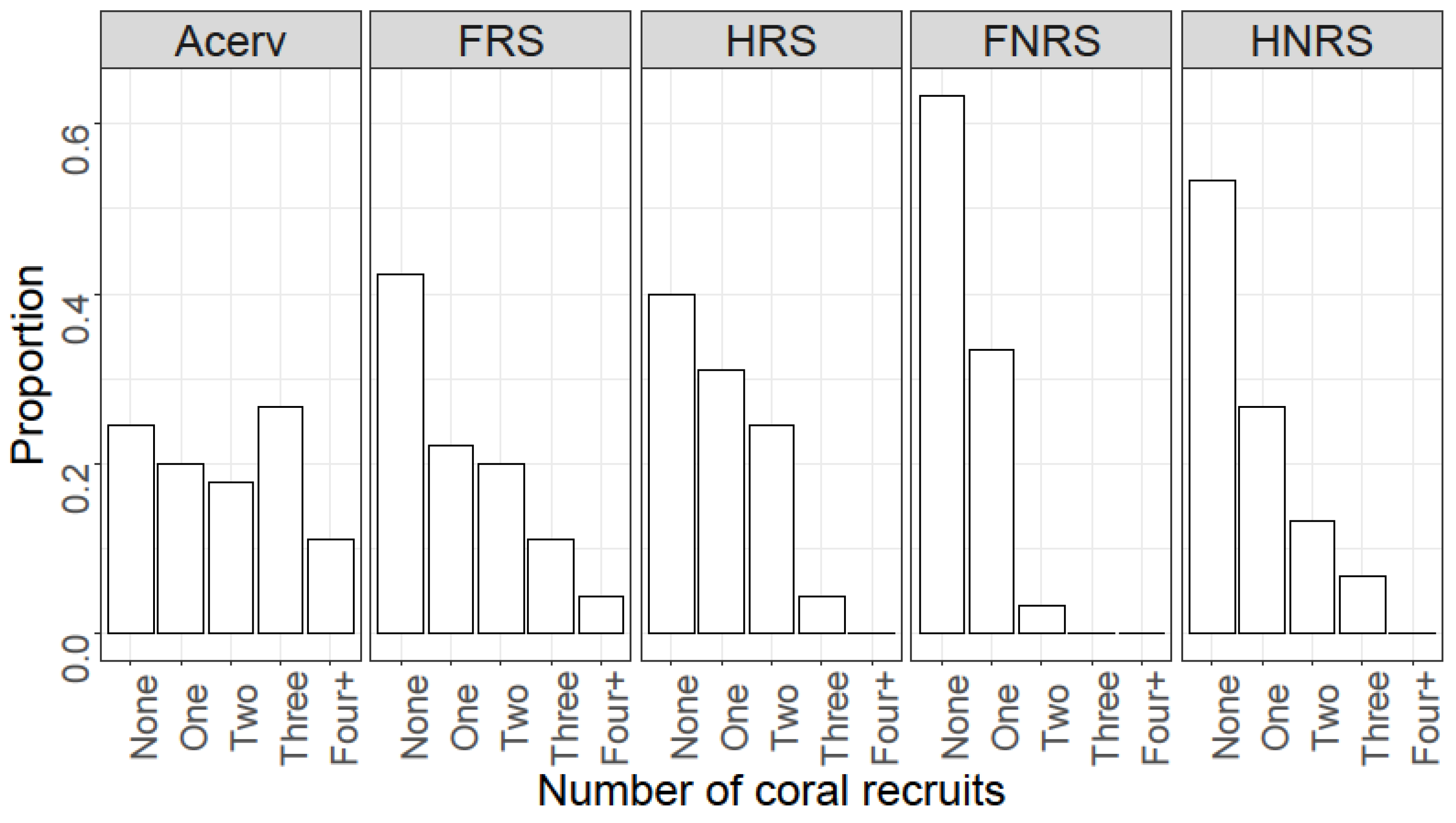
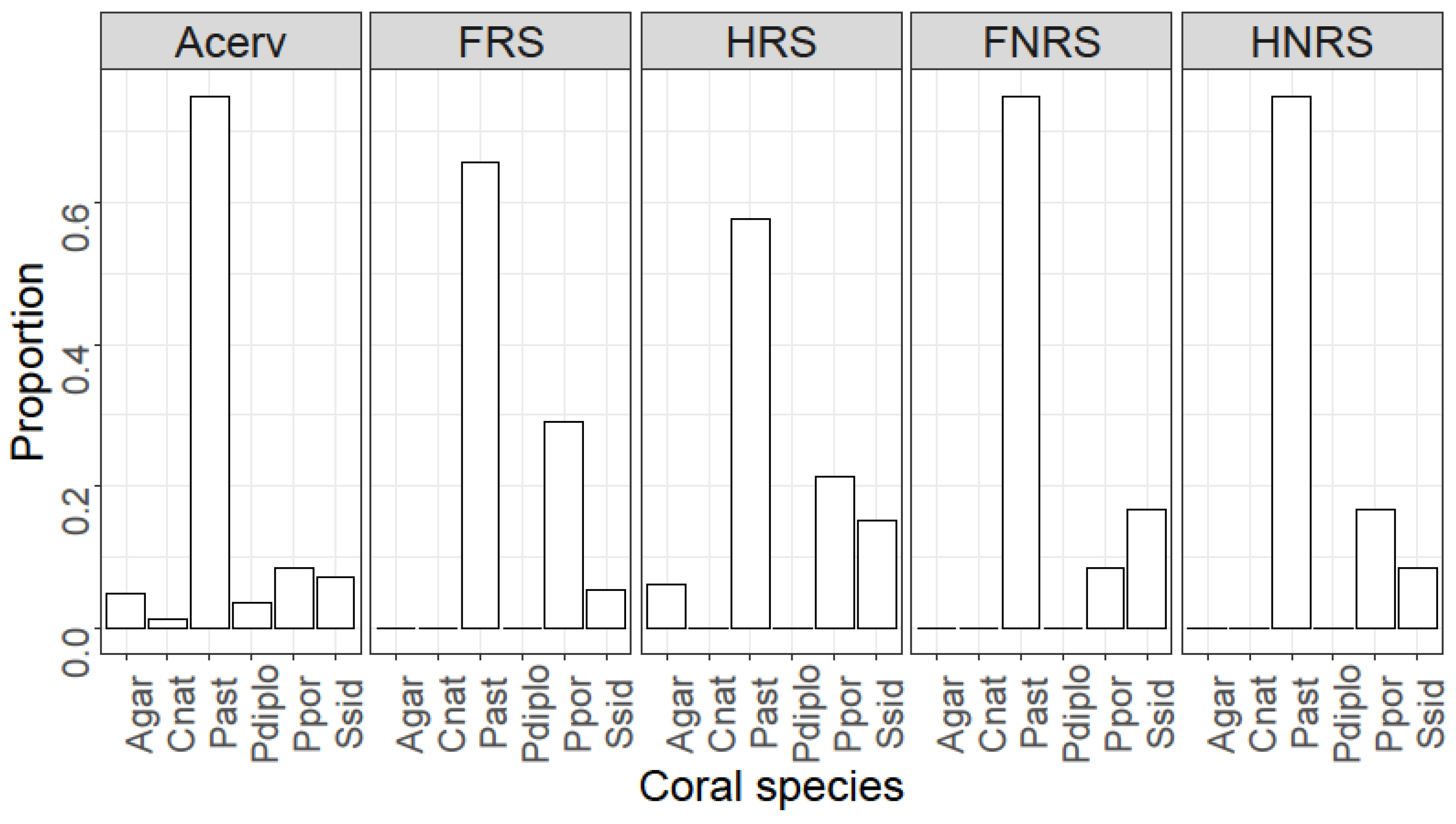
4. Results
5. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Eddy, T.D.; Lam, V.W.; Reygondeau, G.; Cisneros-Montemayor, A.M.; Greer, K.; Palomares, M.L.D.; Bruno, J.F.; Ota, Y.; Cheung, W.W. Global decline in capacity of coral reefs to provide ecosystem services. One Earth 2021, 4, 1278–1285. [Google Scholar] [CrossRef]
- Roff, G.; Mumby, P.J. Global disparity in the resilience of coral reefs. Trends Ecol. Evol. 2012, 27, 404–413. [Google Scholar] [CrossRef] [PubMed]
- Hernández-Delgado, E.A. The emerging threats of climate change on tropical coastal ecosystem services, public health, local economies and livelihood sustainability of small islands: Cumulative impacts and synergies. Mar. Pollut. Bull. 2015, 101, 5–28. [Google Scholar] [CrossRef] [PubMed]
- Fezzi, C.; Ford, D.J.; Oleson, K.L. The economic value of coral reefs: Climate change impacts and spatial targeting of restoration measures. Ecol. Econ. 2023, 203, 107628. [Google Scholar] [CrossRef]
- Barton, J.A.; Willis, B.L.; Hutson, K.S. Coral propagation: A review of techniques for ornamental trade and reef restoration. Rev. Aquac. 2017, 9, 238–256. [Google Scholar] [CrossRef]
- Ishida-Castañeda, J.; Pizarro, V.; López-Victoria, M.; Zapata, F.A. Coral reef restoration in the Eastern Tropical Pacific: Feasibility of the coral nursery approach. Restor. Ecol. 2020, 28, 22–28. [Google Scholar] [CrossRef]
- Combillet, L.; Fabregat-Malé, S.; Mena, S.; Marín-Moraga, J.A.; Gutierrez, M.; Alvarado, J.J. Pocillopora spp. growth analysis on restoration structures in an Eastern Tropical Pacific upwelling area. PeerJ 2022, 10, e13248. [Google Scholar] [CrossRef] [PubMed]
- Calle-Triviño, J.; Rivera-Madrid, R.; León-Pech, M.G.; Cortés-Useche, C.; Sellares-Blasco, R.I.; Aguilar-Espinosa, M.; Arias-González, J.E. Assessing and genotyping threatened staghorn coral Acropora cervicornis nurseries during restoration in southeast Dominican Republic. PeerJ 2020, 8, e8863. [Google Scholar] [CrossRef]
- Bayraktarov, E.; Banaszak, A.T.; Montoya Maya, P.; Kleypas, J.; Arias-González, J.E.; Blanco, M.; Frías-Torres, S. Coral reef restoration efforts in Latin American countries and territories. PLoS ONE 2020, 15, e0228477. [Google Scholar] [CrossRef]
- Mercado-Molina, A.E.; Ruiz-Diaz, C.P.; Pérez, M.E.; Rodríguez-Barreras, R.; Sabat, A.M. Demography of the threatened coral Acropora cervicornis: Implications for its management and conservation. Coral Reefs 2015, 34, 1113–1124. [Google Scholar] [CrossRef]
- Hernández-Delgado, E.A.; Mercado-Molina, A.E.; Suleimán-Ramos, S.E.; Lucking, M.A. Multi-Disciplinary Lessons Learned from Low-Tech Coral Farming Reef Rehabilitation: II. Coral Demography and Social-Ecological Benefits. In Corals in a Changing World; IntechOpen: London, UK, 2018; pp. 245–268. [Google Scholar]
- Maya, P.H.M.; Smit, K.P.; Burt, A.J.; Frias-Torres, S. Large-scale coral reef restoration could assist natural recovery in Seychelles, Indian Ocean. Nat. Conserv. 2016, 16, 1–17. [Google Scholar] [CrossRef]
- Goergen, E.A.; Schopmeyer, S.; Moulding, A.L.; Moura, A.; Kramer, P.; Viehman, T.S. Coral Reef Restoration Monitoring Guide: Methods to Evaluate Restoration Success from Local to Ecosystem Scales; NOAA Technical Memorandum NOS NCCOS 279: Silver Spring, MD, USA, 2020; p. 145. [Google Scholar] [CrossRef]
- Tunnicliffe, V. Breakage and propagation of the stony coral Acropora cervicornis. Proc. Natl. Acad. Sci. USA 1981, 78, 2427–2431. [Google Scholar] [CrossRef] [PubMed]
- Gilmore, M.D.; Hall, B.R. Life history, growth habits, and constitutional roles of Acropora cervicornis in the patch reef environment. J. Sediment. Res. 1976, 46, 519–522. [Google Scholar]
- Agudo-Adriani, E.A.; Cappelletto, J.; Cavada-Blanco, F.; Croquer, A. Colony geometry and structural complexity of the endangered species Acropora cervicornis partly explains the structure of their associated fish assemblage. PeerJ 2016, 4, e1861. [Google Scholar] [CrossRef] [PubMed]
- Fabregat-Malé, S.; Mena, S.; Alvarado, J.J. Nursery-reared coral outplanting success in an upwelling-influenced area in Costa Rica. Rev. Biol. Trop. 2023, 71, e54879. [Google Scholar] [CrossRef]
- Calle-Triviño, J.; Muñiz-Castillo, A.I.; Cortés-Useche, C.; Morikawa, M.; Sellares-Blasco, R.; Arias-González, J.E. Approach to the Functional Importance of Acropora cervicornis in Outplanting Sites in the Dominican Republic. Front. Mar. Sci. 2021, 8, 668325. [Google Scholar] [CrossRef]
- Mumby, P.J.; Wolff, N.H.; Bozec, Y.M.; Chollett, I.; Halloran, P. Operationalizing the resilience of coral reefs in an era of climate change. Conserv. Lett. 2014, 7, 176–187. [Google Scholar] [CrossRef]
- Clark, S.; Edwards, A.J. Coral transplantation as an aid to reef rehabilitation: Evaluation of a case study in the Maldive Islands. Coral Reefs 1995, 14, 201–213. [Google Scholar] [CrossRef]
- Ferse, S.C.A.; Nugues, M.M.; Romatzki, S.B.C.; Kunzmann, A. Examining the use of mass transplantation of brooding and spawning corals to support natural coral recruitment in Sulawesi/Indonesia. Restor. Ecol. 2023, 21, 745–754. [Google Scholar] [CrossRef]
- Hernández-Delgado, E.A.; Ortiz-Flores, M.F. The Long and Winding Road of Coral Reef Recovery in the Anthropocene: A Case Study from Puerto Rico. Diversity 2022, 14, 804. [Google Scholar] [CrossRef]
- Mercado-Molina, A.E.; Sabat, A.M.; Hernández-Delgado, E.A. Population Dynamics of Diseased Corals: Effects of a Shut Down Reaction Outbreak in Puerto Rican Acropora cervicornis. In Advances in Marine Biology; Academic Press: Cambridge, MA, USA, 2020; Volume 87, pp. 61–82. [Google Scholar]
- Toledo-Hernández, C.; Ruiz-Diaz, C.P.; Hernández-Delgado, E.A.; Suleimán-Ramos, S.E. Devastation of 15-year old community-based coral farming and reef-restoration sites in Puerto Rico by major hurricanes Irma and María. Caribb. Nat. 2018, 53, 1–6. [Google Scholar] [CrossRef]
- Santiago-Padua, P.; Velázquez-Alvarado, J.; López-Pérez, A.D.M.; Nevárez-Mélendez, J.; Díaz-Druet, L.E.; Suleimán-Ramos, S.E.; Mercado-Molina, A.E. Demographic and population response of the threatened coral Acropora cervicornis (Scleractinia, Acroporidae) to fireworm corallivory. Rev. Biol. Trop. 2023, 71 (Suppl. S1), e54912. [Google Scholar] [CrossRef]
- Irizarry-Soto, E.; Weil, E. Spatial and temporal variability in juvenile coral densities, survivorship and recruitment in La Parguera, southwestern Puerto Rico. Caribb. J. Sci. 2009, 45, 269–281. [Google Scholar] [CrossRef]
- Hernández-Delgado, E.A.; González-Ramos, C.M.; Alejandro-Camis, P.J. Large-scale coral recruitment patterns on Mona Island, Puerto Rico: Evidence of a transitional community trajectory after massive coral bleaching and mortality. Rev. Biol. Trop. 2014, 62, 283–298. [Google Scholar]
- Wulff, J.L. Asexual fragmentation, genotype success, and population dynamics of erect branching sponges. J. Exp. Mar. Biol. Ecol. 1991, 149, 227–247. [Google Scholar] [CrossRef]
- Pearson, R.K. Exploring Data in Engineering, the Sciences, and Medicine; Oxford University Press: New York, NY, USA, 2011. [Google Scholar]
- Ware, M.; Garfield, E.N.; Nedimyer, K.; Levy, J.; Kaufman, L.; Precht, W.; Miller, S.L. Survivorship and growth in staghorn coral (Acropora cervicornis) outplanting projects in the Florida Keys National Marine Sanctuary. PLoS ONE 2020, 15, e0231817. [Google Scholar] [CrossRef] [PubMed]
- Fisk, D.A.; Harriott, V.J. Spatial and temporal variation in coral recruitment on the Great Barrier Reef: Implications for dispersal hypotheses. Mar. Biol. 1990, 107, 485–490. [Google Scholar] [CrossRef]
- Lange, I.D.; Perry, C.T.; Stuhr, M. Recovery trends of reef carbonate budgets at remote coral atolls 6 years post-bleaching. Limnol. Oceanogr. 2023, 68, S8–S22. [Google Scholar] [CrossRef]
- Goldenberg, E.D. Outplanted Acropora Cervicornis Enhances the Fish Assemblages of Southeast Florida. Master’s Thesis, NOVA Southeastern University, Fort Lauderdale, FL, USA, May 2019. [Google Scholar]
- Holbrook, S.J.; Brooks, A.J.; Schmitt, R.J.; Stewart, H.L. Effects of sheltering fish on growth of their host corals. Mar. Biol. 2008, 155, 521–530. [Google Scholar] [CrossRef]
- Shantz, A.A.; Ladd, M.C.; Schrack, E.; Burkepile, D.E. Fish-derived nutrient hotspots shape coral reef benthic communities. Ecol. Appl. 2015, 25, 2142–2152. [Google Scholar] [CrossRef]
- Birrell, C.L.; McCook, L.J.; Willis, B.L. Effects of algal turfs and sediment on coral settlement. Mar. Pollut. Bull. 2005, 51, 408–414. [Google Scholar] [CrossRef]
- Speare, K.E.; Duran, A.; Miller, M.W.; Burkepile, D.E. Sediment associated with algal turfs inhibits the settlement of two endangered coral species. Mar. Pollut. Bull. 2019, 144, 189–195. [Google Scholar] [CrossRef]
- Dixson, D.L.; Hay, M.E. Corals chemically cue mutualistic fishes to remove competing seaweeds. Science 2012, 338, 804–807. [Google Scholar] [CrossRef] [PubMed]
- Brakel, W.H. Small-scale spatial variation in light available to coral reef benthos: Quantum irradiance measurements from a Jamaican reef. Bull. Mar. Sci. 1979, 29, 406–413. [Google Scholar]
- Gleason, D.F.; Edmunds, P.J.; Gates, R.D. Ultraviolet radiation effects on the behavior and recruitment of larvae from the reef coral Porites astreoides. Mar. Biol. 2006, 148, 503–512. [Google Scholar] [CrossRef]
- Goodbody-Gringley, G.; Wong, K.H.; Becker, D.M.; Glennon, K.; De Putron, S.J. Reproductive ecology and early life history traits of the brooding coral, Porites astreoides, from shallow to mesophotic zones. Coral Reefs 2018, 37, 483–494. [Google Scholar] [CrossRef]
- Baria MV, B.; Guest, J.R.; Edwards, A.J.; Aliño, P.M.; Heyward, A.J.; Gómez, E.D. Caging enhances post-settlement survival of juveniles of the Scleractinia coral Acropora tenuis. J. Exp. Mar. Biol. Ecol. 2010, 394, 149. [Google Scholar] [CrossRef]
- Smith, S.R. Patterns of coral recruitment and post-settlement mortality on Bermuda’s reefs: Comparisons to Caribbean and Pacific reefs. Am. Zool. 1992, 32, 663–673. [Google Scholar] [CrossRef]
- Miller, M.W.; Barimo, J. Assessment of juvenile coral populations at two reef restoration sites in the Florida Keys National Marine Sanctuary: Indicators of success? Bull. Mar. Sci. 2001, 69, 395–405. [Google Scholar]
- Perera-Valderrama, S.; Hernández-Arana, H.; Ruiz-Zárate, M.; Alcolado, P.M.; Caballero-Aragón, H.; González-Cano, J.; Vega-Zepeda, A.; Cobián-Rojas, D. Condition assessment of coral reefs of two marine protected areas under different regimes of use in the north-western Caribbean. Ocean. Coast. Manag. 2016, 127, 16–25. [Google Scholar] [CrossRef]
- Olsen, K.; Sneed, J.M.; Paul, V.J. Differential larval settlement responses of Porites astreoides and Acropora palmata in the presence of the green alga Halimeda opuntia. Coral Reefs 2016, 35, 521–525. [Google Scholar] [CrossRef]
- Chornesky, E.A.; Peters, E.C. Sexual reproduction and colony growth in the scleractinian coral Porites astreoides. Biol. Bull. 1987, 172, 161–177. [Google Scholar] [CrossRef]
- McGuire, M.P. Timing of larval release by Porites astreoides in the northern Florida Keys. Coral Reefs 1998, 17, 369–375. [Google Scholar] [CrossRef]
- Gleason, D.F.; Danilowicz, B.S.; Nolan, C.J. Reef waters stimulate substratum exploration in planulae from brooding Caribbean corals. Coral Reefs 2009, 28, 549–554. [Google Scholar] [CrossRef]


| Value (s) | |
|---|---|
| Odds ratio | 3.37 |
| 95% CI | 1.58–7.01 |
| Z statistic | 3.18 |
| Significance level (p-value) | 0.0015 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mercado-Molina, A.E.; Suleimán-Ramos, S.E. Outplants of the Threatened Coral Acropora cervicornis Promote Coral Recruitment in a Shallow-Water Coral Reef, Culebra, Puerto Rico. Sustainability 2023, 15, 16548. https://doi.org/10.3390/su152416548
Mercado-Molina AE, Suleimán-Ramos SE. Outplants of the Threatened Coral Acropora cervicornis Promote Coral Recruitment in a Shallow-Water Coral Reef, Culebra, Puerto Rico. Sustainability. 2023; 15(24):16548. https://doi.org/10.3390/su152416548
Chicago/Turabian StyleMercado-Molina, Alex E., and Samuel E. Suleimán-Ramos. 2023. "Outplants of the Threatened Coral Acropora cervicornis Promote Coral Recruitment in a Shallow-Water Coral Reef, Culebra, Puerto Rico" Sustainability 15, no. 24: 16548. https://doi.org/10.3390/su152416548
APA StyleMercado-Molina, A. E., & Suleimán-Ramos, S. E. (2023). Outplants of the Threatened Coral Acropora cervicornis Promote Coral Recruitment in a Shallow-Water Coral Reef, Culebra, Puerto Rico. Sustainability, 15(24), 16548. https://doi.org/10.3390/su152416548




