Analysis of Rare Earth Elements in Pteridophytes from the Historical Mining Area, Southwestern Japan
Abstract
1. Introduction
1.1. REEs in the Mine Area
1.2. Athyrium yokoscense
2. Materials and Methods
2.1. Study Area
2.2. Methods
3. Results and Discussion
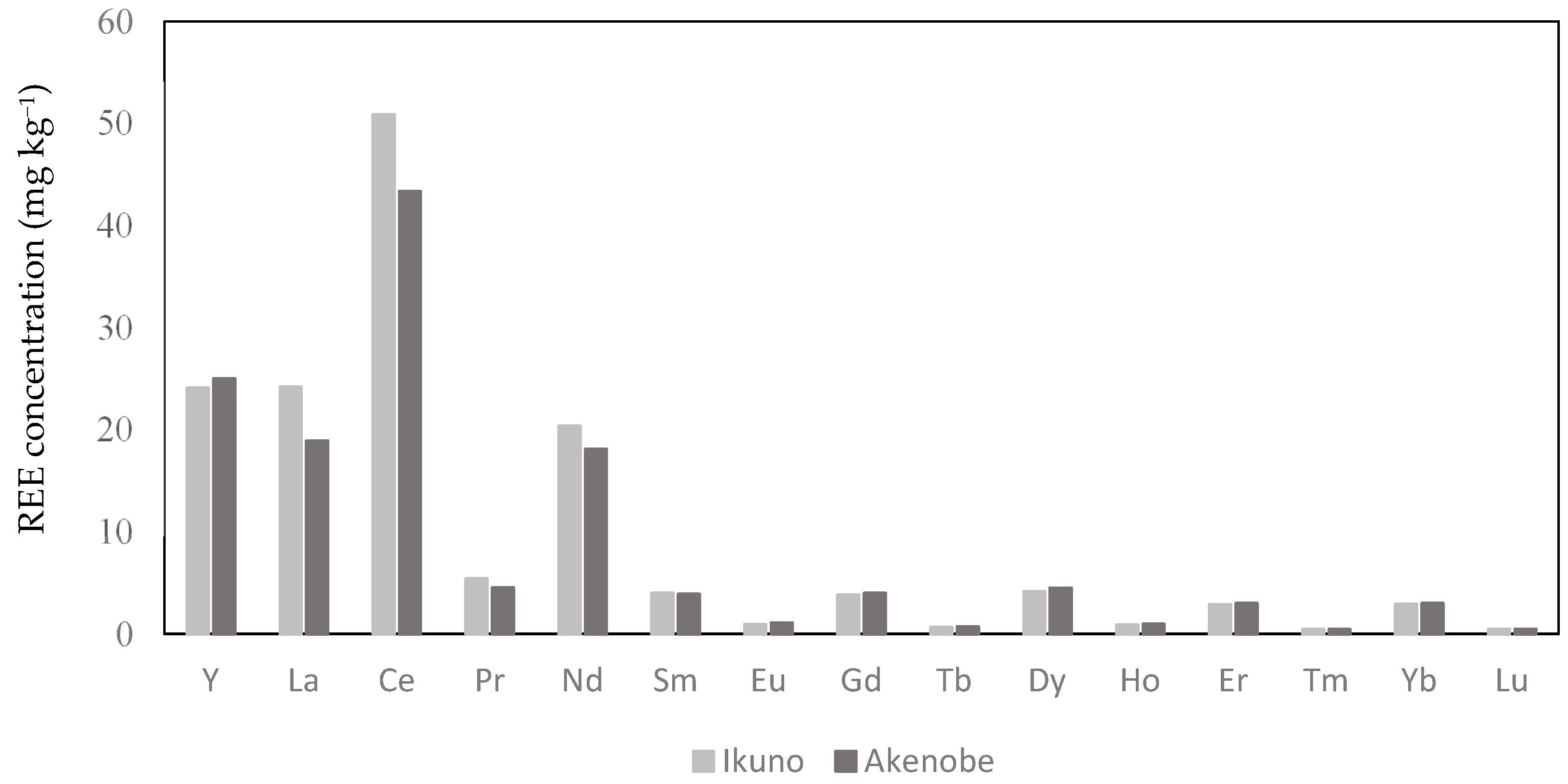
3.1. REEs in Sediment
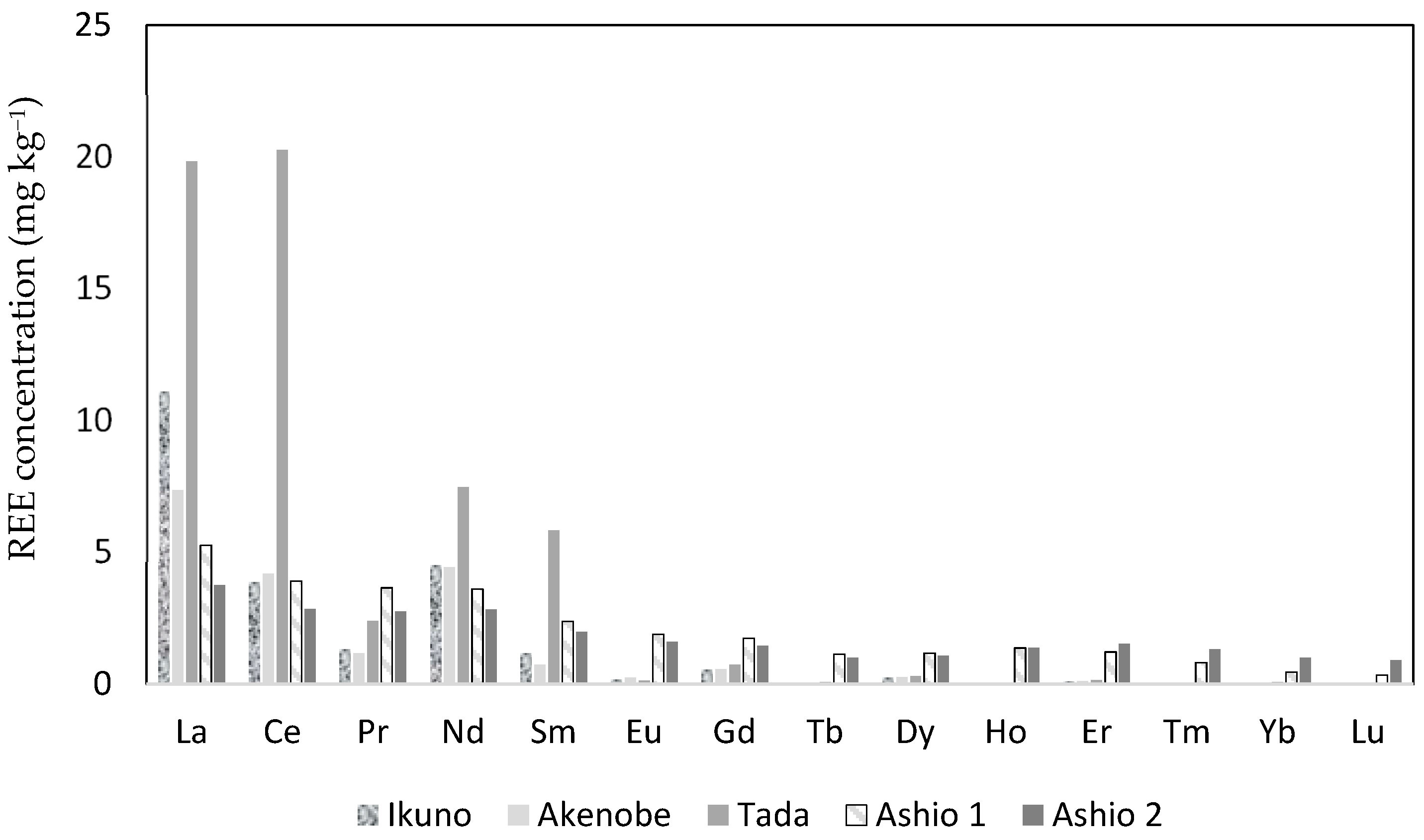
3.2. REEs in Athyrium yokoscense
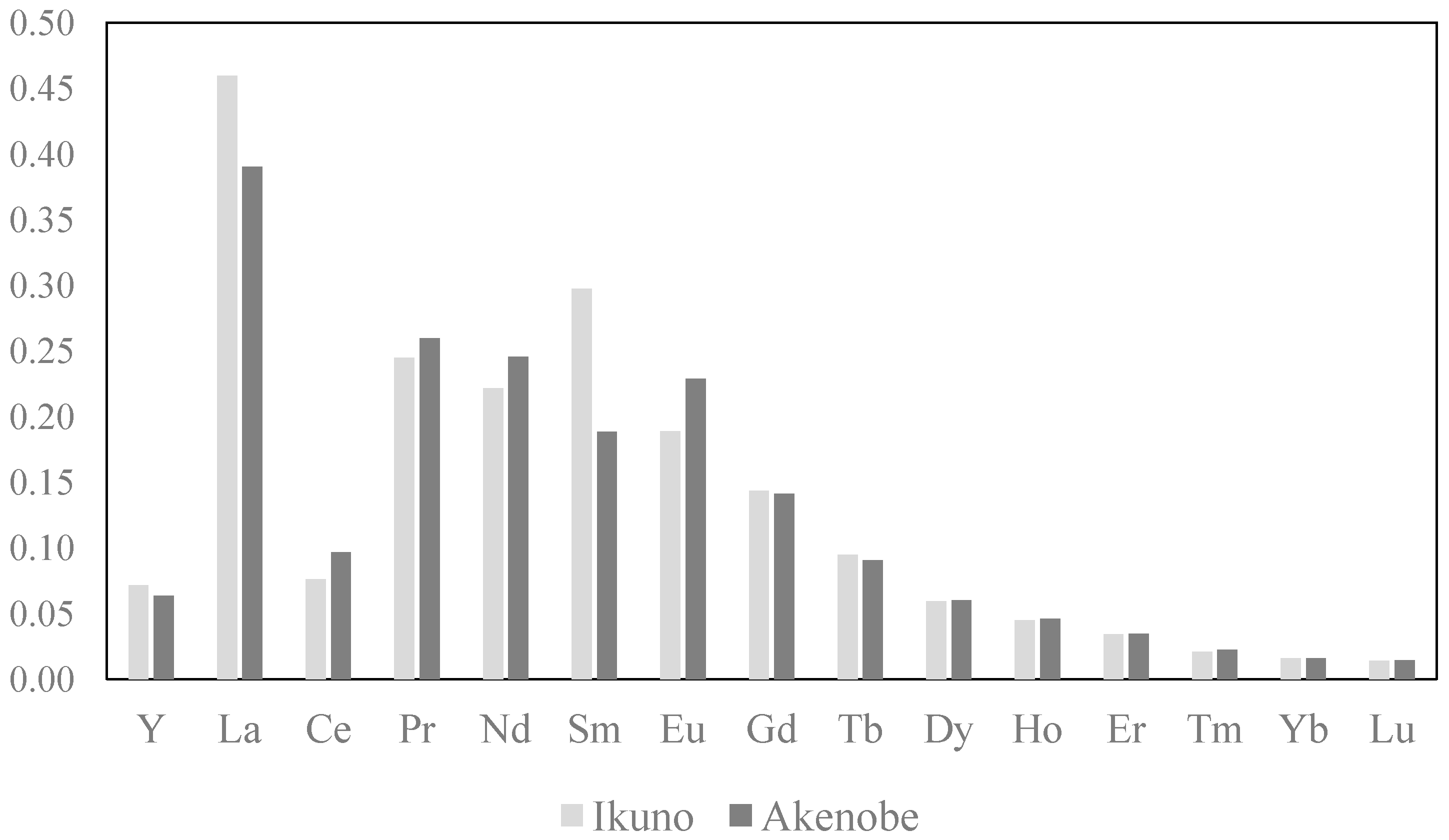
3.3. Bioconcentration Factor
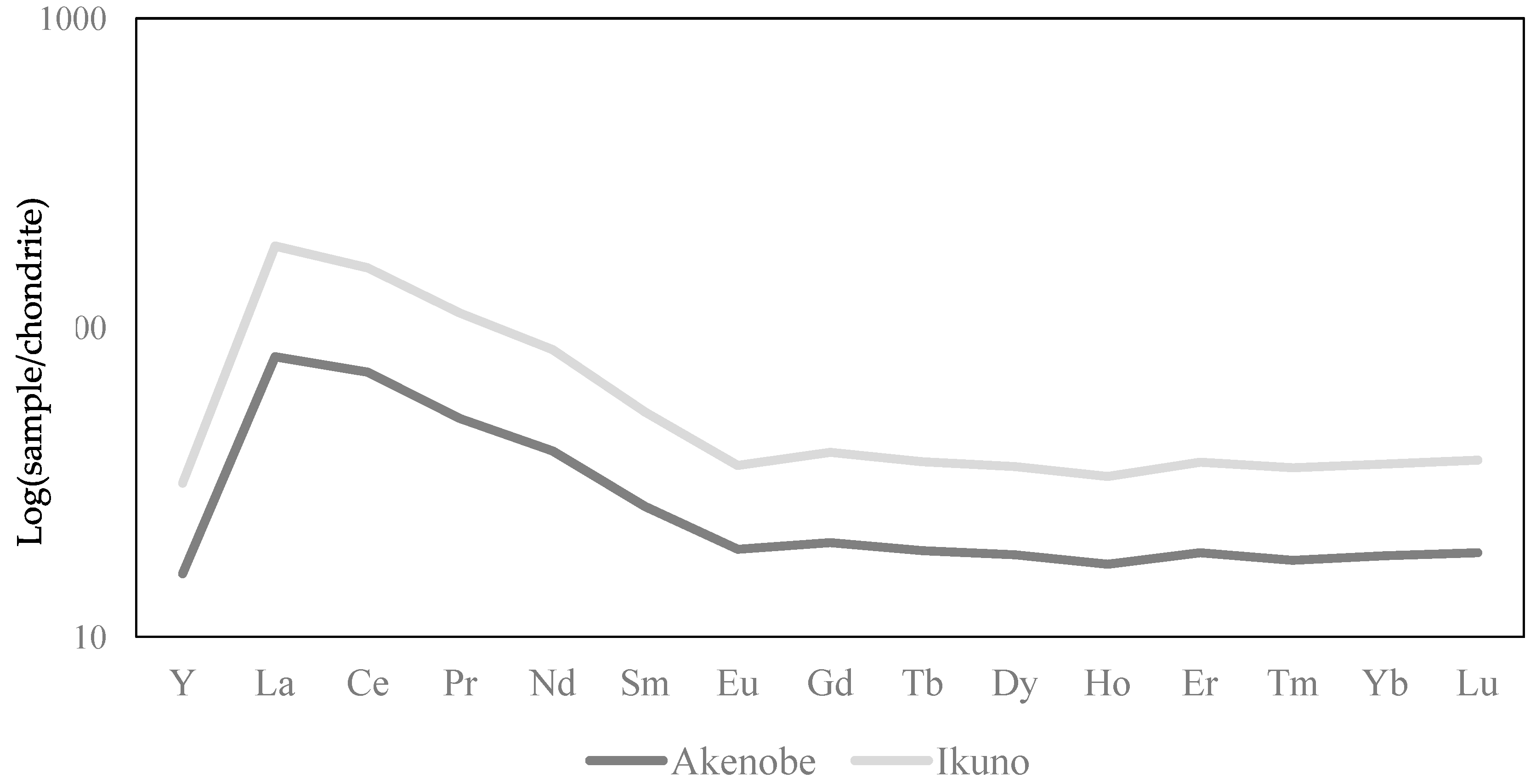
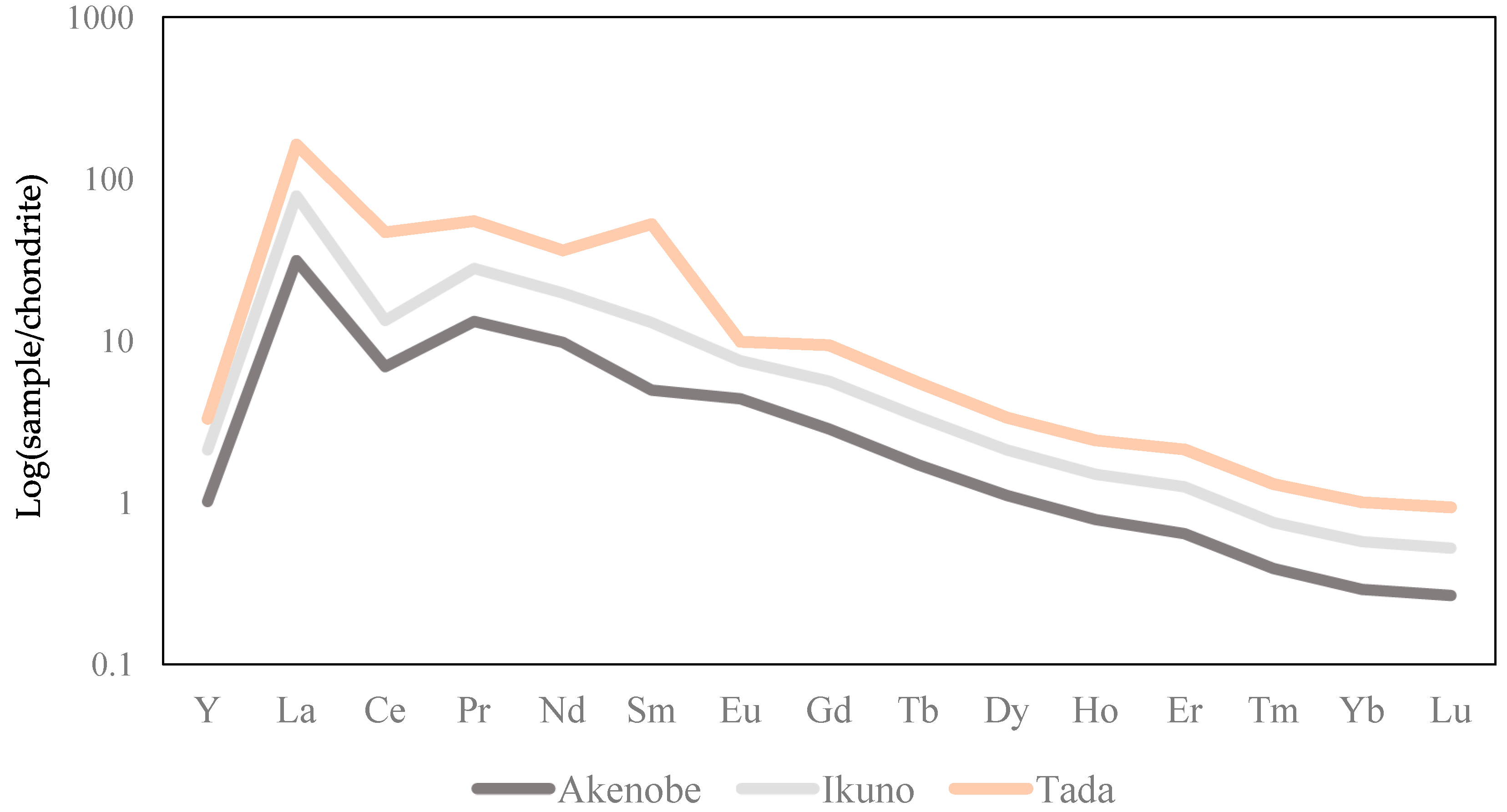
3.4. Chondrite-Normalized REEs Pattern
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- IUPAC. Nomenclature of Inorganic Chemistry; IUPAC Recommendations2005 (“Red Book”); RSC Publishing: Cambridge, UK, 2005; ISBN 0-85404-438-8. [Google Scholar]
- Walters, A.; Lusty, P.; Chetwyn, C.; Hill, A. Rare Earth Elements; Mineral Profile Series British Geological Survey: Keyworth, UK, 2010; 45p. [Google Scholar]
- Pourbaix, M. Atlas of Electrochemical Equilibria in Aqueous Solutions; Pergamon: New York, NY, USA, 1966. [Google Scholar]
- Ni, Y.; Hunghes, J.M.; Mariano, A.N. Crystal chemistry of the monatize and xenotime structures. Am. Mineral. 1995, 80, 21–26. [Google Scholar] [CrossRef]
- Rare Earth Elements; A Briefing Note by the Geological Society of London. Available online: https://www.geolsoc.org.uk/ (accessed on 28 July 2023).
- Ascenzi, P.; Bettinelli, M.; Boffi, A.; Botta, M.; De Simone, G.; Luchinat, C.; Marengo, E.; Mei, H.; Aime, S. Rare earth elements (REE) in biology and medicine. Rend. Lincei. Sci. Fis. Nat. 2020, 31, 821–833. [Google Scholar] [CrossRef]
- Mehmood, M. Rare Earth Elements—A review. J. Ecol. Nat. Resour. 2018, 2, 000128. [Google Scholar] [CrossRef]
- Dostal, J. Rare earth element deposits alkaline igneous rocks. Resources 2017, 6, 34. [Google Scholar] [CrossRef]
- Brioschi, L.; Steinmann, M.; Lucot, E.; Pierret, M.C.; Stille, P.; Prunier, J.; Badot, P.M. Transfer of rare earth elements (REE) from natural soil to plant systems: Implications for the environmental availability of anthropogenic REE. Plant Soil 2012, 366, 143–163. [Google Scholar] [CrossRef]
- Aguilar, J.; Dorronsoro, C.; Fernández, E.; Fernández, J.; García, I.; Martín, F. Soil pollution by a pyrite mine spill in Spain: Evolution in time. Environ. Pollut. 2004, 132, 395–401. [Google Scholar] [CrossRef]
- Klukanová, A.; Rapant, S. Impact of mining activities upon the environment of the Slovak Republic: Two case studies. J. Geochem. Explor. 1999, 66, 299–306. [Google Scholar] [CrossRef]
- Liu, J.Y.; Chang, X.Y.; Tu, X.L. Review on heavy metal pollution in mine exploitation. Miner. Resour. Geol. 2006, 12, 645–650. [Google Scholar]
- Luís, A.T.; Teixeira, P.; Almeida SF, P.; Matos, J.X.; Silva, E.F. Environmental impact of mining activities in the Lousal area (Portugal): Chemical and diatom characterization of metal-contaminated stream sediments and surface water of Corona stream. Sci. Total Environ. 2011, 409, 4312–4325. [Google Scholar] [CrossRef]
- Salomons, W. Environmental impact of metals derived from mining activities: Processes, predictions, prevention. J. Geochem. Explor. 1995, 52, 5–23. [Google Scholar] [CrossRef]
- Bhattacharya, A.; Joyanto, R.A.; Gunnar, J.B.; Prosun, B.B.; Magnus, M.A. Environmental assessment of abandoned mine tailings in Adak, Västerbotten district, northern Sweden. Appl. Geochem. 2006, 21, 1760–1780. [Google Scholar] [CrossRef]
- Sánchez España, J.; Pamo, E.L.; Pastor, E.S.; Ercilla, M.D. The acidic mine pit lakes of the Iberian Pyrite Belt: An approach to their physical limnology and hydrogeochemistry. Appl. Geochem. 2008, 23, 1260–1287. [Google Scholar] [CrossRef]
- Medas, D.; Cidu, R.; de Giudici, G.; Podda, F. Geochemistry of rare Earth elements in water and solid materials at abandoned mines in SW Sardinia (Italy). J. Geochem. Explor. 2013, 133, 149–159. [Google Scholar] [CrossRef]
- Zhang, W.; Chen, W.T.; Gao, J.-F.; Chen, H.-K.; Li, J.-H. Two episodes of REE mineralization in the Qinling Orogenic Belt, Central China: In-situ U-Th-Pb dating of bastnäsite and monazite. Miner. Depos. 2019, 54, 1265–1280. [Google Scholar] [CrossRef]
- Grawunder, A.; Merten, D. Rare Earth Elements in Acidic Systems—Biotic and Abiotic Impacts. In Bio-Geo Interactions in Metal-Contaminated Soils; Springer: Berlin/Heidelberg, Germany, 2012; pp. 81–97. [Google Scholar]
- Hu, Z.Y.; Haneklaus, S.; Sparovek, G.; Schnug, E. Rare earth elements in soils. Commun. Soil Sci. Plant 2006, 37, 1381–1420. [Google Scholar] [CrossRef]
- Jones, D.L. Trivalent metal (Ce, Y, Rh, La, Pr, Gd) sorption in two acid soils and its consequences for bioremediation. Eur. J. Soil Sci. 1997, 48, 697–702. [Google Scholar] [CrossRef]
- Abraham, J.; Dowling, K.; Florentine, S. Assessment of potentially toxic metal contamination in the soils of a legacy mine site in Central Victoria. Chemosphere 2018, 192, 122–132. [Google Scholar] [CrossRef]
- Li, Z.; Hadioui, M.; Wilkinson, K.J. Conditions affecting the release of thorium and uranium from the tailings of a niobium mine. Environ. Pollut. 2019, 247, 206–215. [Google Scholar] [CrossRef]
- Campaner, V.P.; Silva, W.L.; Machado, W. Geochemistry of acid mine drainage from a coal mining area and processes controlling metal attenuation in stream water, southern Brazil. Ann. Braz. Acad. Sci. 2014, 86, 539–554. [Google Scholar] [CrossRef]
- Prudêncio, M.I.; Valente, T.; Marques, R.; Sequeira Braga, M.A.; Pamplona, J. Geochemistry of rare earth elements in a passive treatment system built for acid mine drainage remediation. Chemosphere 2015, 138, 691–700. [Google Scholar] [CrossRef]
- Davis, D.W.; Hirdes, W.; Schaltegger, U.; Nunoo, E.A. U-Pb age constraints on deposition and provenance of Birimian and gold-bearing Tarkwaian sediments in Ghana, West Africa. Precambrian Res. 1994, 67, 89–107. [Google Scholar] [CrossRef]
- Natarajan, K.A.; Radhika, V.; Subramanian, S. Bioremediation of zinc using Desulfotomaculum nigrificans: Bioprecipitation and characterization studies. Water Res. 2006, 40, 3628–3636. [Google Scholar]
- Tao, L.; Zhang, S.; Wang, L.; Kung, H.-T.; Wang, Y.; Hu, A.; Ding, S. Environmental biogeochemical behaviors of rare earth elements in soil–plant systems. Environ. Geochem. Health 2005, 27, 301–311. [Google Scholar] [CrossRef]
- Elias, M.S.; Ibrahim, S.; Samudin, K.; Kantasamy, N.; Rahman, S.A.; Hashim, A. Rare earth elements (REEs) as pollution indicator in sediment of Linggi River, Malaysia. Appl. Radiat. Isot. 2019, 15, 116–123. [Google Scholar] [CrossRef]
- Wang, Z.; Yin, L.; Xiang, H.; Qin, X.; Wang, S. Accumulation patterns and species-specific characteristics of yttrium and rare earth elements (YREEs) in biological matrices from Maluan Bay, China: Implications for biomonitoring. Environ. Res. 2019, 179, 108804. [Google Scholar] [CrossRef]
- Kitagishi, K.; Yamane, I. Heavy Metal Pollution in Soils of Japan; Japan Scientific Societies Press: Tokyo, Japan, 1981. [Google Scholar]
- Van, T.K.; Kang, Y.M.; Fukui, T.; Sakurai, K.; Iwasaki, K.; Aikawa, Y.; Phuong, N.M. Arsenic and heavy metal accumulation by Athyrium yokoscense from contaminated soils. Soil Sci. Plant Nutr. 2006, 52, 701–710. [Google Scholar] [CrossRef]
- Guo, F.Q.; Wang, Y.Q.; Sun, J.X.; Chen, H.M. Preliminary study on rare earth bound proteins in natural plant fern dicranopteris dichotoma. J. Radioanal. Nucl. Chem. 1996, 209, 91–99. [Google Scholar] [CrossRef]
- Adriano, D.C. Arsenic in Trace Elements in Terrestrial Environments: Biochemistry, Bioavailability, and Risks of Metals; Adriano, D.C., Ed.; Springer: New York, NY, USA, 2001; pp. 219–263. [Google Scholar]
- Tu, C.; Ma, L.Q.; Bondada, B. Arsenic accumulation in the hyperaccumulator Chinese brake and its utilization potential for phytoremediation. J. Environ. Qual. 2002, 31, 1671–1675. [Google Scholar] [CrossRef]
- Shen, Z.G.; Liu, Y.L. Progress in the study on the plants that hyper accumulate heavy metal. Plant Physiol. 1998, 34, 133. [Google Scholar]
- Wei, Z.G.; Zhang, H.J.; Li, H.X.; Hu, F. Research trends on rare earth element hyperaccumulator. J. Chin. Rare Earth Soc. 2006, 24, 1–11. (In Chinese) [Google Scholar]
- Li, J.T.; Gurajala, H.K.; Wu, L.; van der Ent, A.; Qiu, R.-L.; Baker AJ, M.; Shu, W. Hyperaccumulator plants from China: A synthesis of the current state of knowledge. Environ. Sci. Technol. 2018, 52, 11980–11994. [Google Scholar] [CrossRef] [PubMed]
- Cunningham, S.D.; Berti, W.R.; Huang, J.W. Phytoremediation of contaminated soils. Trends Biotechnol. 1995, 13, 393–397. [Google Scholar] [CrossRef]
- Ward, O.P.; Singh, A. Soil bioremediation and phytoremediation—An overview. In Applied Bioremediation and Phytoremediation; Singh, A., Ward, O.P., Eds.; Springer: Heidelberg, Germany, 2004; p. 7. [Google Scholar]
- Van der Ent, A.; Baker, A.J.M.; Reeves, R.D.; Pollard, A.J.; Schat, H. Commentary: Toward a more physiologically and evolutionarily relevant definition of metal hyperaccumulation in plants. Front. Plant Sci. 2015, 6, 554. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.-S.; Chen, Y.-Y.; Huot, H.; Liu, C.; Guo, M.-N.; Qiu, R.-L.; Tang, Y.-T. Phytoextraction of rare earth elements from ion-adsorption mine tailings by Phytolacca americana: Effects of organic material and biochar amendment. J. Clean. Prod. 2020, 275, 122959. [Google Scholar] [CrossRef]
- Pagano, G.; Guida, M.; Tommasi, F.; Oral, R. Health effects and toxicity mechanisms of rare earth elements—Knowledge gaps and research prospects. Ecotoxicol. Environ. Saf. 2015, 115, 40–48. [Google Scholar] [CrossRef]
- Ikhlayel, M. Evaluation of the environmental impacts of rare earth elements production. Int. J. Environ. Stud. 2017, 74, 939–957. [Google Scholar] [CrossRef]
- Nishizono, H.; Ichikawa, H.; Suziki, S.; Ishii, F. The role of the root cell wall in the heavy metal tolerance of Athyrium yokoscense. Plant Soil 1987, 101, 15–20. [Google Scholar] [CrossRef]
- Sakai, Y.; Fukuoka, T.; Honjo, T. Characterization of lead in the tissues of the heavy metal tolerant plant, Athyrium yokoscense (Fr. Et Sav.). Christ. Chem. Soc. 1991, 5, 416–421. [Google Scholar]
- Ashraf, N.; Rodrigues, E.S.; de Almeida, E.; Montanha, G.S.; Abreu-Junior, C.S.; Vítová, M.; Garcia, R.H.L.; Küpper, H.; de Carvalho, H.W.P. Identification of potential plant species hyperaccumulating light rare earth elements (LREE) in a mining area in Minas Gerais, Brazil. Environ. Sci. Pollut. Res. 2022, 29, 90779–90790. [Google Scholar] [CrossRef]
- Bert, V.; Bonnin, I.; Saumitou-Laprade, P.; Laguerie, P.; Petit, D. Do Arabidopsis halleri from nonmetallicolous populations accumulate zinc and cadmium more effectively than those from metallicolous populations. New Phytol. 2002, 155, 47–57. [Google Scholar] [CrossRef]
- Paape, T.; Hatakeyama, M.; Shimizu-Inatsugi, R. Conserved but attenuated parental gene expression in allopolyploids: Constitutive zinc hyperaccumulation in the allotetraploid Arabidopsis kamchatica. Mol. Biol. Evol. 2016, 33, 2781–2800. [Google Scholar] [CrossRef]
- Miyake, K. Hebinonegoza to koushitsutono kankei (Relationship between Aspelnium yokoscense Fr. Et Sav. and mineral). Bot. Mag. Tokyo 1897, 11, 404–406. [Google Scholar]
- Cotton, F.A.; Wilkinson, G.; Murillo, C.A.; Bochmann, M.; Grimes, R. Advanced Inorganic Chemistry, 6th ed.; Wiley: New York, NY, USA, 1999. [Google Scholar]
- Kabata, A.P.; Mukherjee, A.B. Trace Element from Soil to Human; Springer: Berlin/Heidelberg, Germany, 2007; ISBN 978-3-540-32714-1. [Google Scholar]
- Chen, D.; Chen, G. Practical Geochemistry of Rare Earth Elements, 1st ed.; Geological Press: Beijing, China, 1990. [Google Scholar]
- Tyler, G. Rare earth elements in soil and plant systems—A review. Plant Soil 2004, 267, 191–206. [Google Scholar] [CrossRef]
- Šmuc, A.; Tadej, D.A.; Todor, S.B.; Matej, D.A.; Petra, V. Geochemical characteristics of rare earth elements (REEs) in the paddy soil and rice (Oryza sativa L.) system of Kočani Field, Republic of Macedonia. Geoderma 2012, 183–184, 1–11. [Google Scholar] [CrossRef]
- Ozaki, T.; Ambe, S.; Enomoto, S.; Minai, Y.; Yoshida, S.; Makide, Y. Multitracer study on the uptake mechanism of yttrium and rare earth elements by autumn fern. Radiochim. Acta 2002, 90, 303–307. [Google Scholar] [CrossRef]
- Wei, Y.Z. Distribution, transportation and cytolocalization of neodymium in oilseed rape. J. Chin. Rare Earth Soc. 2000, 18, 278–281, (In Chinese with English Abstract). [Google Scholar]
- Gao, Y.; Zeng, F.; Yi, A.; Ping, S.; Jing, L. Research of the entry of rare earth elements Eu3+ and La3+ into the plant cell. Biol. Trace Elem. Res. 2003, 91, 253–265. [Google Scholar] [CrossRef]
- Wei, Z.; Hong, F.; Yin, M.; Li, H.; Hu, F.; Zhao, G.; Wong, J.W. Structural Differences Between Light and Heavy Rare Earth Element Binding Chlorophylls in Naturally, Grown Fern: Dicranopteris linearis. Biol. Trace Elem. Res. 2005, 106, 279–297. [Google Scholar] [CrossRef]
- Hong, F.; Wang, L.; Meng, X.; Wei, Z.; Zhao, G. The effect of cerium (III) on the chlorophyll formation in spinach. Biol. Trace Elem. Res. 2002, 89, 263–276. [Google Scholar]
- Turra, C.; De Fernandes, E.A.N.; Bacchi, M.A.; Sarries, G.A.; Barbosa Junior, F.; Creste, A.L.T.; Reyes, A.E. Effects of Lanthanum on Citrus Plant. Int. J. New Technol. Res. 2015, 1, 48–50. [Google Scholar]
- Miyoshi, K.; Abe, Y.; Takahisa, Y.; Nakai, I. Technical treatise Laser ablation characterization of rare earth elements in Athyrium yokoscense using inductively coupled plasma mass spectrometer. J. Jpn. Soc. Anal. Chem. 2015, 64, 617–624. [Google Scholar]
- Liang, T.; Li, K.; Wang, L. State of rare earth elements in different environmental components in mining areas of China. Environ. Monit. Assess. 2014, 186, 1499–1513. [Google Scholar] [CrossRef] [PubMed]
- Ndeda, L.; Manohar, S. Bioconcentration factor and translocation ability of heavy metals within different habitats of hydrophytes in Nairobi Dam, Kenya. J. Environ. Sci. Toxicol. Food Technol. 2014, 8, 42–45. [Google Scholar]
- Khan, A.M.; Yusoff, I.; Abu Bakar, N.K.; Abu Bakar, A.F.; Alias, Y.; Mispan, M.S. Accumulation, Uptake And Bioavailability of Rare Earth Elements (Rees) in Soil Grown Plants from Ex-Mining Area in Perak, Malaysia. Appl. Ecol. Environ. Res. 2017, 15, 117–133. [Google Scholar] [CrossRef]
- Hugh, S.; O’Neill, C. The Smoothness and Shapes of Chondrite-normalized Rare Earth Element Patterns in Basalts. J. Petrol. 2016, 57, 1463–1508. [Google Scholar]
- Anders, E.; Grevesse, N. The Abundances of the Elements: Meteoritic and Solar. Geochim. Cosmochim. Acta 1988, 53, 197–214. [Google Scholar] [CrossRef]
- Nakadaa, R.; Shibuyac, T.D.; Suzukic, D.K.; Takahashia, Y. Europium Anomaly Variation under Low-Temperature Water-Rock Interaction: A New Thermometer. Geochem. Int. 2017, 55, 822–832. [Google Scholar] [CrossRef]
- Braun, J.J.; Pagel, M.; Muller, J.P.; Bilong, P.; Michard, A.; Guillet, B. Cerium anomalies in lateritic profiles. Geochim. Cosmochim. Acta 1990, 54, 781–795. [Google Scholar] [CrossRef]


| Element | Ikuno | Akenobe | ||
|---|---|---|---|---|
| mg kg−1 | Std | mg kg−1 | Std | |
| Y | 24.09 | 7.15 | 24.99 | 6.12 |
| La | 24.18 | 7.77 | 18.9 | 5.2 |
| Ce | 50.85 | 15.58 | 43.35 | 13.32 |
| Pr | 5.4 | 1.61 | 4.53 | 1.24 |
| Nd | 20.35 | 5.92 | 18.08 | 5.08 |
| Sm | 3.97 | 1.09 | 3.88 | 1.1 |
| Eu | 0.93 | 0.29 | 1.08 | 0.43 |
| Gd | 3.81 | 1.02 | 3.97 | 1.15 |
| Tb | 0.65 | 0.17 | 0.69 | 0.17 |
| Dy | 4.15 | 1.12 | 4.48 | 1.19 |
| Ho | 0.88 | 0.29 | 0.96 | 0.23 |
| Er | 2.86 | 0.78 | 2.98 | 0.73 |
| Tm | 0.42 | 0.11 | 0.43 | 0.09 |
| Yb | 2.91 | 0.78 | 2.98 | 0.65 |
| Lu | 0.45 | 0.11 | 0.46 | 0.1 |
| LREE | 109.49 | 93.79 | ||
| HREE | 36.41 | 37.97 | ||
| REE | 145.9 | 131.76 | ||
| Element | Ikuno | Akenobe | Tada | |||
|---|---|---|---|---|---|---|
| (mg kg−1) | Std | (mg kg−1) | Std | (mg kg−1) | Std | |
| Y | 1.72 | 3.16 | 1.59 | 6.12 | 1.84 | 1.61 |
| La | 11.11 | 13.49 | 7.37 | 5.20 | 19.84 | 16.52 |
| Ce | 3.87 | 4.55 | 4.19 | 13.32 | 20.28 | 29.17 |
| Pr | 1.32 | 1.82 | 1.18 | 1.24 | 2.40 | 1.96 |
| Nd | 4.51 | 6.48 | 4.44 | 5.08 | 7.47 | 5.90 |
| Sm | 1.18 | 2.15 | 0.73 | 1.10 | 5.84 | 5.16 |
| Eu | 0.18 | 0.21 | 0.25 | 0.43 | 0.13 | 0.11 |
| Gd | 0.55 | 0.93 | 0.56 | 1.15 | 0.74 | 0.69 |
| Tb | 0.06 | 0.11 | 0.06 | 0.17 | 0.08 | 0.07 |
| Dy | 0.25 | 0.44 | 0.27 | 1.19 | 0.30 | 0.29 |
| Ho | 0.04 | 0.07 | 0.04 | 0.23 | 0.05 | 0.05 |
| Er | 0.10 | 0.17 | 0.10 | 0.73 | 0.14 | 0.14 |
| Tm | 0.01 | 0.02 | 0.01 | 0.09 | 0.01 | 0.02 |
| Yb | 0.05 | 0.08 | 0.05 | 0.65 | 0.07 | 0.07 |
| Lu | 0.01 | 0.01 | 0.01 | 0.10 | 0.01 | 0.01 |
| Element | Ikuno | Akenobe |
|---|---|---|
| Y | 0.07 | 0.06 |
| La | 0.46 | 0.39 |
| Ce | 0.08 | 0.10 |
| Pr | 0.24 | 0.26 |
| Nd | 0.22 | 0.25 |
| Sm | 0.30 | 0.19 |
| Eu | 0.19 | 0.23 |
| Gd | 0.14 | 0.14 |
| Tb | 0.09 | 0.09 |
| Dy | 0.06 | 0.06 |
| Ho | 0.04 | 0.05 |
| Er | 0.03 | 0.03 |
| Tm | 0.02 | 0.02 |
| Yb | 0.02 | 0.02 |
| Lu | 0.01 | 0.01 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mardiyah, A.; Syahputra, M.R.; Tsuboi, M. Analysis of Rare Earth Elements in Pteridophytes from the Historical Mining Area, Southwestern Japan. Sustainability 2023, 15, 13406. https://doi.org/10.3390/su151813406
Mardiyah A, Syahputra MR, Tsuboi M. Analysis of Rare Earth Elements in Pteridophytes from the Historical Mining Area, Southwestern Japan. Sustainability. 2023; 15(18):13406. https://doi.org/10.3390/su151813406
Chicago/Turabian StyleMardiyah, Ainun, Muhammad Rio Syahputra, and Motohiro Tsuboi. 2023. "Analysis of Rare Earth Elements in Pteridophytes from the Historical Mining Area, Southwestern Japan" Sustainability 15, no. 18: 13406. https://doi.org/10.3390/su151813406
APA StyleMardiyah, A., Syahputra, M. R., & Tsuboi, M. (2023). Analysis of Rare Earth Elements in Pteridophytes from the Historical Mining Area, Southwestern Japan. Sustainability, 15(18), 13406. https://doi.org/10.3390/su151813406








