What Is the Impact of Dexamethasone and Prednisolone Glucocorticoids on the Structure of Meiobenthic Nematode Communities?
Abstract
1. Introduction
2. Material and Methods
2.1. Collecting Site and Sediment Manipulation
2.2. Sediment Contamination and Experimental Set-Up
2.3. Structural and Functional Traits of Nematode Communities
2.4. Data Processing
3. Results
3.1. Taxonomic Composition
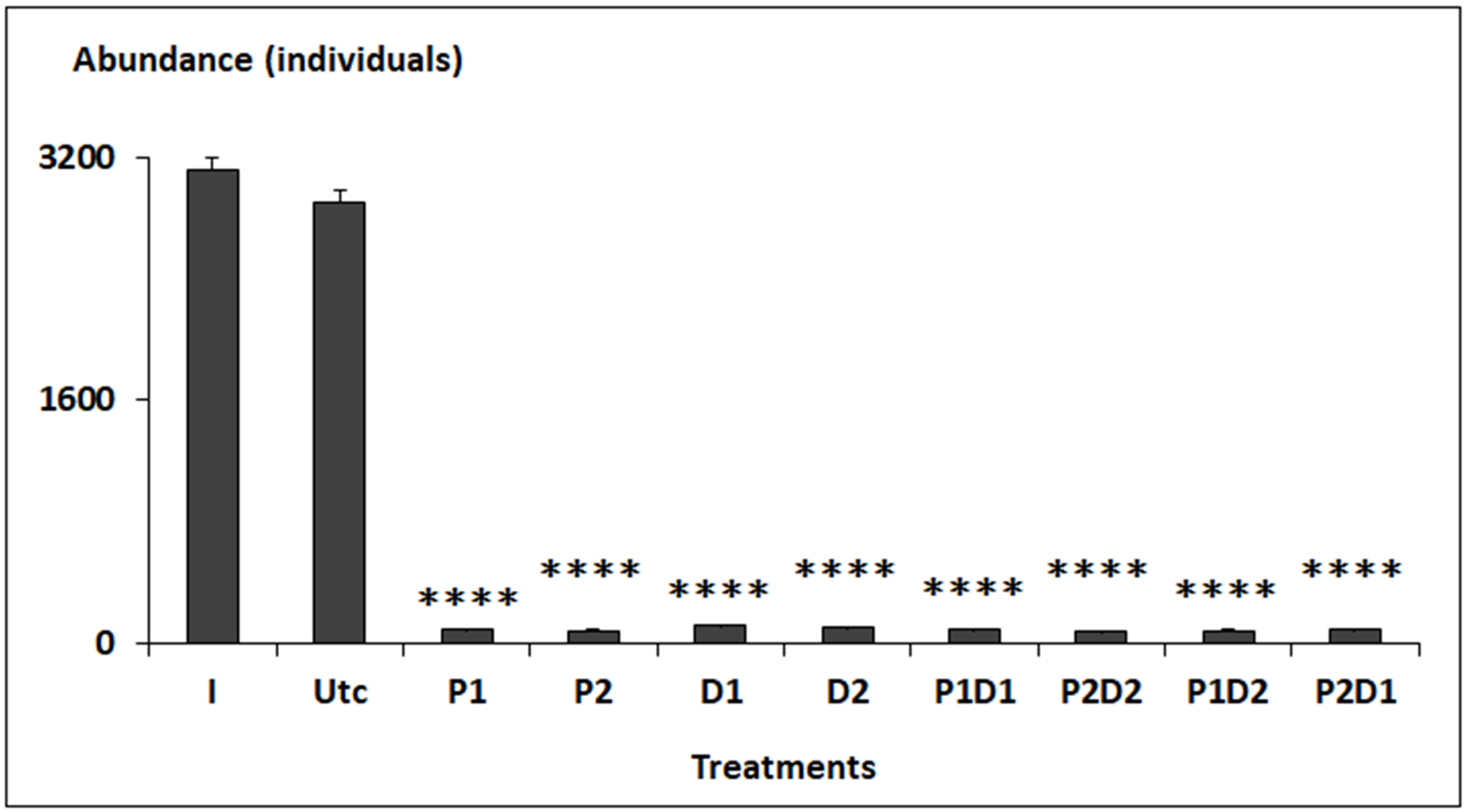
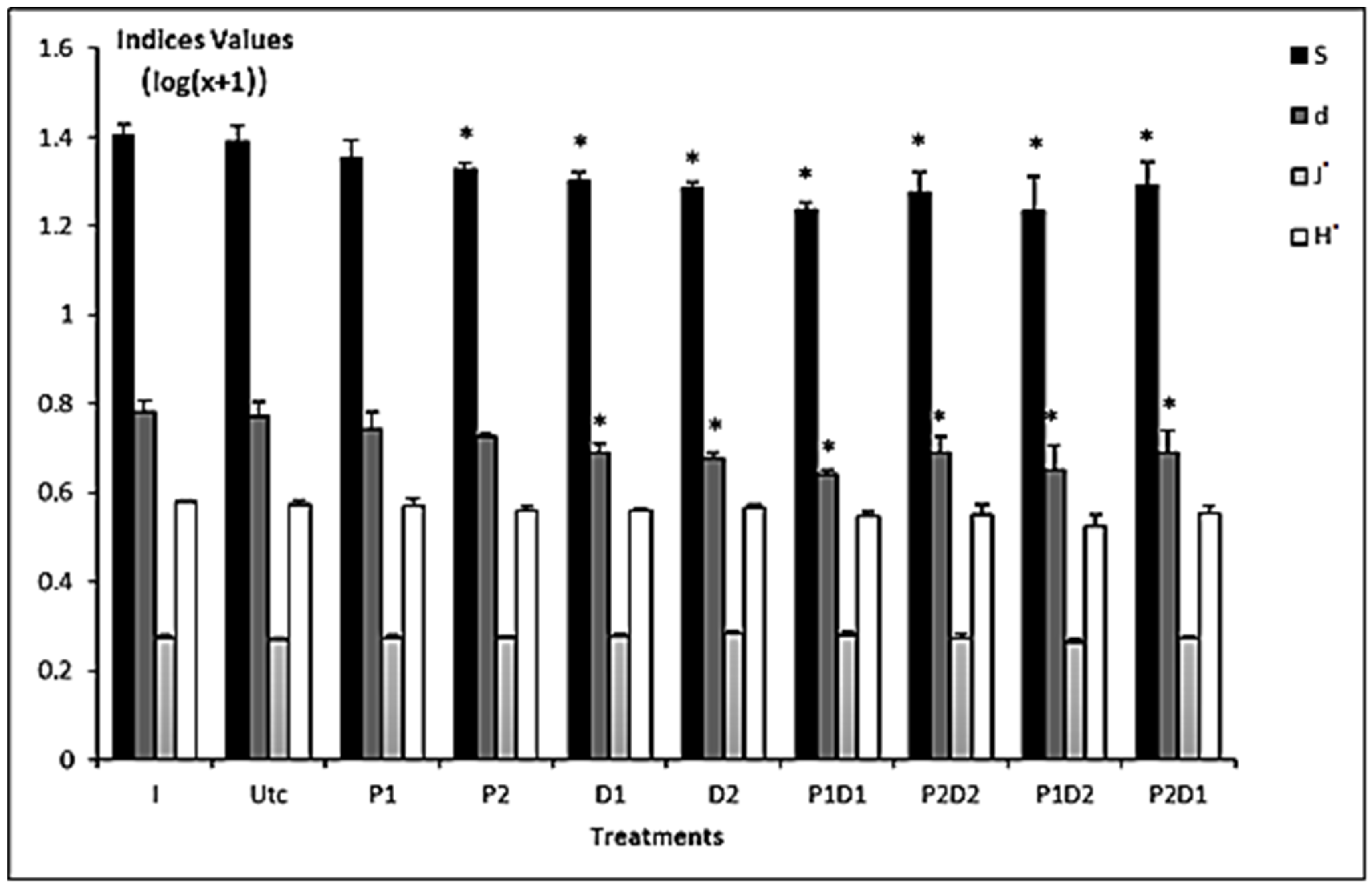
3.2. Abundance and Diversity
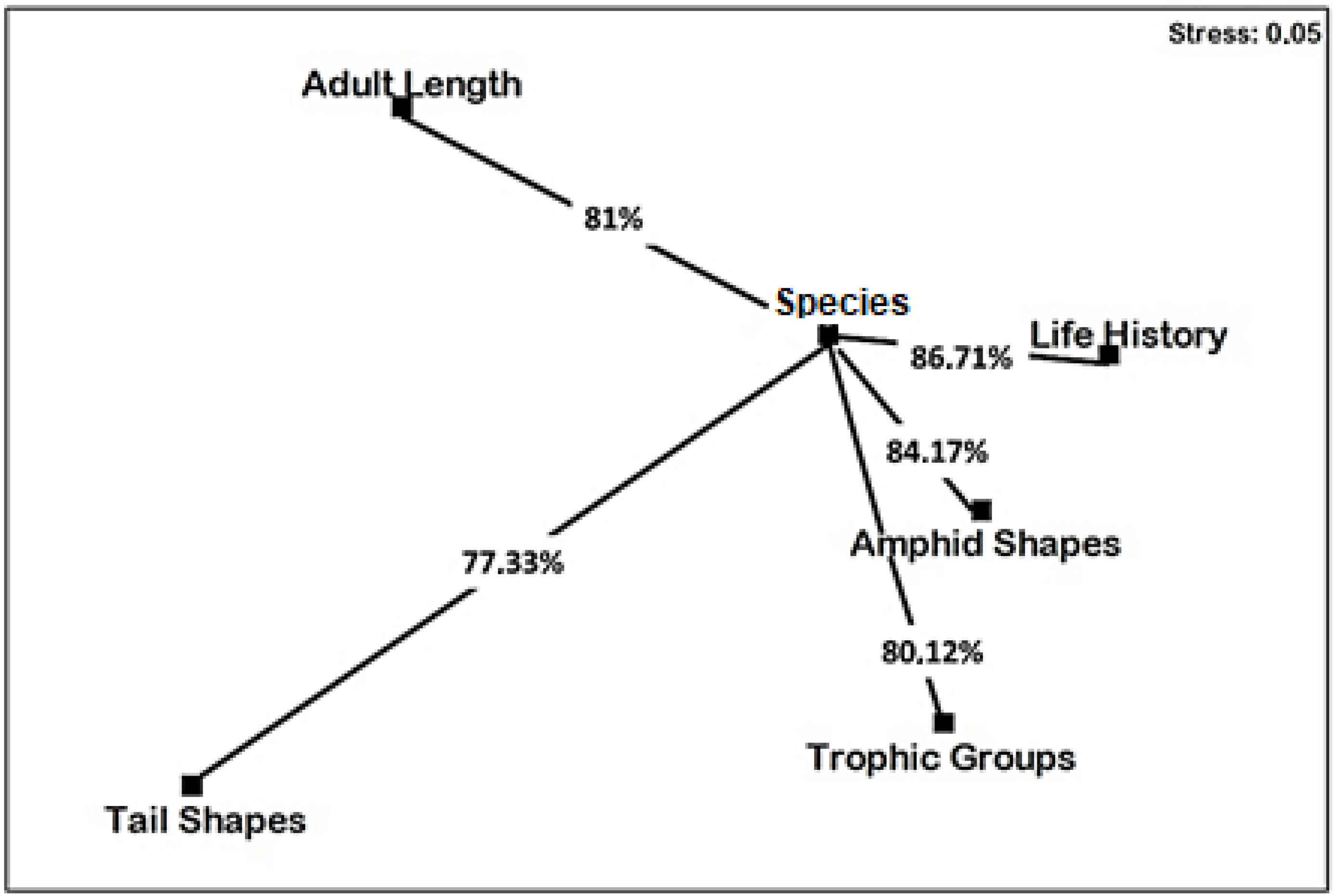
3.3. Multivariate Analyses
3.4. Functional Traits
- The feeding groups of the initial communities were dominated by omnivorous/carnivores (2B) and epistratum-feeders (2A), comprising 36.3 ± 3.05% and 32.3 ± 2.3% of the nematofauna. The 1B group significantly increased in final control, D2, P1D1 and P2D2, respectively. Conversely, the 2A group decreased in P, P2, D2, P1D1, P2D2 and P1D2, whereas the 2B group increased in D1 and P2D1. The nMDS results indicated that D2 and P2D2 groups were furthest from initial control. Conversely, the treatments D1 and P2D1 were situated closer to initial and final control, respectively.
- Amphid shapes of the initial community were dominated by circular (cr) and pocket-like (pk) amphids, comprising 47 ± 1.7% and 32.3 ± 3.5% of the nematofauna. The cr amphid shape types decreased in P2, D1, P2D2 and P1D2, whereas the id amphid shape significantly increased in P1, P2, P1D1, P2D2, and P1D2. The pk amphid shape significantly increased in P1, D1, P1D1, P2D2, and P2D1. The nMDS results indicated that P1D2 was furthest away from initial control, but P2D1 and final were the closest to initial control.
- Tail shapes were dominated by conical (co) and clavate (cla) types, comprising 47.3 ± 1.1% and 46 ± 3.4% of the initial nematofauna, respectively. The contamination induced a significant decrease of co tail shapes in P2, P2D2 and P1D2, as well as of cla tails shape in D1. The co tail shape increased significantly in D1. The nMDS results indicated that D1 was situated the furthest from initial control, but treatments P1 and P1D1 were the closest.
- The initial life history composition was c-p4, followed by c-p3 and c-p2, comprising 41.6 ± 3.7%, 26.3 ± 0.5% and 25 ± 4.58% of the nematofauna, respectively. The c-p2 types increased significantly in most treatments, excepting P1D2 and P2D2, as well as c-p5 in P1, P2 and P1D2. Conversely, the results showed a significant decrease of c-p3 in most treatments, except for final control, D1 and P2D1, and of c-p4 in most treatments except for final control and D1. The nMDS results indicated that P1D2 and P2D2 were situated the furthest from initial control, whereas the treatments P2D1 and final control were the closest.
- Body sizes were dominated by 2–4 mm and 1–2 mm species, comprising 57.6 ± 3.2% and 23.6 ± 2.5% of the initial nematofauna. The species smaller than 1 mm increased in D1, same for those between 1 and 2 mm interval in D2 and P1D1. Conversely, the species with body sizes between 2 and 4 mm intervals decreased in D1, D2, P1D1 and P2D2. The nMDS ordination indicated that D2 was situated the furthest from initial control, whereas D1 was the closest.
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A
| Tl | Am | FG | c-p | AL | I | Utc | P1 | P2 | D1 | D2 | P1D1 | P2D2 | P1D2 | P2D1 | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Bathylaimus sp. | cla | sp | 1B | 2 | 1–2 mm | 1.33 ± 1.15 | 1.66 ± 1.52 | 0.74 ± 0.64 | 1.33 ± 1.52 | 3.66 ± 1.15 | 0.38 ± 0.67 | 1.82 ± 1.21 | |||
| Cyartonema germanicum | co | cr | 1A | 4 | <1 mm | 6.33 ± 1.52 | 7 ± 2.64 | 3.64 ± 1.41 | 0.71 ± 1.21 | 11.33 ± 1.52 | 4 ± 2 | 5.76 ± 1.29 | 1.73 ± 0.81 | 2.25 ± 1.81 | 8.92 ± 4.21 |
| Daptonema fallax | cla | cr | 1B | 2 | 1–2 mm | 0.66 ± 0.57 | 0.66 ± 0.57 | 0.66 ± 1.15 | 5.33 ± 2.08 | 0.45 ± 0.79 | 1.45 ± 1.63 | ||||
| Enoplolaimus longicaudatus | cla | id | 2B | 5 | 2–4 mm | 3 ± 1 | 0.66 ± 1.15 | 15.08 ± 3.85 | 14.01 ± 3.45 | 12.64 ± 1.93 | 13.21 ± 0.81 | 16.69 ± 0.53 | 1.81 ± 2.22 | ||
| Halalaimus gracilis | e/f | REL | 1A | 4 | 2–4 mm | 3 ± 1.73 | 1.33 ± 0.57 | 1.04 ± 1.06 | 1.53 ± 0.64 | 0.33 ± 0.57 | 1.09 ± 0.07 | 1.61 ± 1.76 | 0.76 ± 1.32 | 1.11 ± 1.08 | |
| Longicyatholaimus longicandatus | e/f | sp | 2A | 3 | 2–4 mm | 3 ± 1 | 3.33 ± 2.08 | 3.13 ± 1.94 | 2.93 ± 2.07 | 0.66 ± 0.57 | 3.99 ± 1.21 | 4.73 ± 4.12 | 1.53 ± 2.65 | 5.44 ± 2.77 | |
| Metoncholaimus pristiurus | cla | pk | 2B | 4 | >4 mm | 3.33 ± 057 | 0.66 ± 1.15 | 0.66 ± 0.57 | 8.33 ± 1.52 | ||||||
| Microlaimus cyatholaimoïdes | co | cr | 2A | 2 | 2–4 mm | 5.66 ± 3.05 | 7 ± 1.73 | 8.95 ± 1.36 | 7.31 ± 2.41 | 11.33 ± 4.93 | 2 ± 1 | 7.22 ± 3.01 | 5.66 ± 1.88 | 10.54 ± 1.13 | 8.15 ± 3.09 |
| Microlaimus honestus | co | cr | 2A | 3 | 2–4 mm | 15 ± 2 | 14.66 ± 3.51 | 15 ± 2 | 5.33 ± 0.57 | 13.91 ± 4.15 | |||||
| Nudora gerlachi | co | cr | 2A | 3 | <1 mm | 2.66 ± 2.08 | 1 ± 1.73 | 1.12 ± 1.16 | 1.71 ± 2.96 | 7.66 ± 2.51 | 2 ± 2 | 0.68 ± 1.19 | 0.45 ± 0.79 | 2.26 ± 2.2 | |
| Odontophora villoti | co | REL | 1B | 2 | 1–2 mm | 0.33 ± 0.57 | 1.33 ± 0.57 | 1.85 ± 1.41 | 1.96 ± 0.81 | 3 ± 2 | 0.33 ± 0.57 | 2.25 ± 2.35 | 3.86 ± 0.33 | 0.77 ± 0.67 | |
| Oncholaimellus calvadocicus | cla | pk | 2B | 4 | 1–2 mm | 3.33 ± 0.57 | 2 ± 1 | 1.73 ± 1.61 | 2.41 ± 2.1 | 1 ± 1 | 5.33 ± 1.52 | 1.71 ± 2.97 | 1.93 ± 3.35 | 1.16 ± 1.14 | 0.71 ± 1.24 |
| Oncholaimus campylocercoïdes | cla | pk | 2B | 4 | 2–4 mm | 19.33 ± 2.51 | 18.66 ± 4.72 | 16.96 ± 3.32 | 20.15 ± 5.83 | 13 ± 2.64 | 14 ± 2.64 | 15.31 ± 3.94 | 16.07 ± 4.43 | 24.52 ± 3.11 | 17.54 ± 4.26 |
| Paramonohystera proteus | cla | cr | 1B | 2 | 1–2 mm | 7.33 ± 0.57 | 7.33 ± 2.08 | 8.69 ± 2.67 | 7.46 ± 3.63 | 4 ± 1 | 8 ± 2 | 7.88 ± 2.76 | 7.28 ± 0.82 | 5.67 ± 2.69 | 7.363 ± 5.46 |
| Parasphaerolaimus paradoxus | cla | cr | 2B | 3 | 1–2 mm | 2.33 ± 0.57 | 2.33 ± 1.52 | 5.09 ± 1.63 | 3.01 ± 1.04 | 2 ± 2 | 1 ± 1 | 8.85 ± 2.79 | 4.66 ± 2.93 | 4.01 ± 2.29 | 2.58 ± 1.31 |
| Phanoderma sp. | s/r | pk | 2A | 4 | 2–4 mm | 0.66 ± 0.57 | 1.33 ± 1.15 | 1.12 ± 1.16 | 1.11 ± 1.13 | 0.33 ± 0.57 | 0.77 ± 1.34 | 1.19 ± 2.06 | |||
| Prochromadorella longicaudata | co | id | 2A | 2 | <1 mm | 1.33 ± 0.57 | 2.66 ± 1.52 | 2.46 ± 1.12 | 2.66 ± 0.45 | 1.66 ± 1.52 | 2.66 ±2.08 | 1.73 ± 2.13 | 1.36 ± 2.37 | 3.71 ± 0.93 | 2.64 ± 2.9 |
| Rhabditis sp. | co | id | 1B | 1 | <1 mm | 1.66 ± 0.57 | 1.66 ± 1.15 | 2.12 ± 1.01 | 3.023 ± 2.33 | 4.66 ± 1.15 | 8.66 ± 1.52 | 3.65 ± 1.66 | 5.63 ± 3.56 | 2.32 ± 2.29 | 0.71 ± 1.24 |
| Sabatiera splendens | cla | sp | 1B | 2 | 1–2 mm | 1.33 ± 1.52 | 1.66 ± 0.57 | 1.46 ± 0.074 | 1.08 ± 1.05 | 1 ± 1 | 4 ± 2 | 0.34 ± 0.59 | 0.77 ± 1.34 | 0.49 ± 084 | 2.18 ± 1.03 |
| Spirinia parasitifera | co | REL | 2A | 3 | 2–4 mm | 4 ± 1 | 2.33 ± 1.52 | 3.48 ± 2.05 | 1.12 ± 1.06 | 1.66 ± 1.52 | 0.33 ± 0.57 | 1.51 ± 1.3 | 0.39 ± 0.68 | 3.29 ± 1.85 | |
| Synonchiella edax | cla | sp | 2B | 3 | 2–4 mm | 1.66 ± 1.15 | 2.66 ± 1.15 | 2.84 ± 0.046 | 2.28 ± 1.03 | 1 ± 1.73 | 2.66 ± 0.57 | 1.82 ± 2.22 | 2.07 ± 1.83 | 2.32 ± 2.29 | 2.65 ± 3.71 |
| Thalassironus britannicus | co | pk | 2B | 4 | >4 mm | 3.33 ± 0.57 | 1.66 ± 1.15 | 2.81 ± 1.55 | 3.14 ± 1.99 | 5 ± 1 | 6.33 ± 1.52 | 4.81 ± 3.37 | 4.46 ± 3.55 | 2.34 ± 3.11 | 2.55 ± 2.49 |
| Theristus modicus | co | cr | 1B | 2 | 1–2 mm | 1.66 ± 0.57 | 3.66 ± 1.52 | 3.55 ± 1.46 | 4.14 ± 2.03 | 1.66 ± 1.52 | 4 ± 1.73 | 3.54 ± 2.09 | 2.06 ± 1.23 | 2.92 ± 0.53 | 2.95 ± 0.7 |
| Theristus pertenuis | co | cr | 1B | 2 | 1–2 mm | 2 ± 1.73 | 2 ± 1 | 3.18 ± 0.85 | 2.34 ± 1.19 | 0.66 ± 1.15 | 0.66 ± 1.15 | 2.57 ± 2.69 | 2.27 ± 2.85 | 0.383 ± 0.66 | 1.1 ± 1.07 |
| Thoonchus inermis | cla | pk | 2B | 4 | 2–4 mm | 2.33 ± 0.08 | 1.33 ± 1.15 | 1.12 ± 1.16 | 3.07 ± 1.29 | 1 ± 1.73 | 2.063 ± 1.23 | 1.65 ± 0.57 | |||
| Trichotheristus mirabilis | co | cr | 1B | 2 | 1–2 mm | 2 ± 1 | 7.33 ± 3.21 | 6.32 ± 2.8 | 9.81 ± 2.54 | 5.33 ± 2.88 | 11.33 ± 1.51 | 12.57 ± 4.74 | 14.71 ± 5.81 | 11.64 ± 4.14 | 7.37 ± 3.57 |
| Valvaelaimus maior | co | cr | 1B | 2 | 1–2 mm | 1.33 ± 1.15 | 2 ± 1 | 1.41 ± 1.63 | 2.96 ± 1.51 | 4 ± 1 | 1.72 ± 2.05 | 0.39 ± 0.68 | 3.66 ± 1.61 |
| I vs. P1D1 | I vs. P2D2 | I vs. P1D2 | I vs. P2D1 | |
|---|---|---|---|---|
| Species | 44.87% | 48.32% | 44.44% | 30.72% |
| Microlaimus honestus (17.45%) elim | Microlaimus honestus (17.46%) elim | Microlaimus honestus (18.83%) elim | Trichotheristus mirabilis (7.98%) − | |
| Trichotheristus mirabilis (10.95%) + | Trichotheristus mirabilis (10.91%) + | Enoplolaimus longicaudatus (12.87%) + | Oncholaimus campylocercoïdes (6.9%) − | |
| Enoplolaimus longicaudatus (10.02%) + | Enoplolaimus longicaudatus (8.49%) + | Trichotheristus mirabilis (8.88%) + | Paramonohystera proteus (6.41%) − | |
| Parasphaerolaimus paradoxus (6.64%) + | Oncholaimus campylocercoïdes (7.86%) − | Cyartonema germanicum (5.8%) − | Microlaimus honestus (6.3%) − | |
| Oncholaimus campylocercoïdes (6.21%) − | Cyartonema germanicum (5.81%) − | Spirinia parasitifera (4.61%) − | Metoncholaimus pristiurus (5.69%) + | |
| Metoncholaimus pristiurus (3.88%) elim | Spirinia parasitifera (4.66%) elim | Metoncholaimus pristiurus (4.18%) elim | Microlaimus cyatholaimoïdes (5.15%) − | |
| Microlaimus cyatholaimoïdes (3.47%) + | Metoncholaimus pristiurus (3.88%) elim | Microlaimus cyatholaimoïdes (3.9%) + | Oncholaimellus calvadosicus (4.57%) + | |
| Oncholaimellus calvadosicus (3.25%) − | Oncholaimellus calvadosicus (3.26%) − | Paramonohystera proteus (3.45%) − | Nudora gerlachi (4.55%) + | |
| Spirinia parasitifera (3.08%) − | Rhabditis sp. (3.12%) + | Oncholaimellus calvadosicus (2.97%) − | Longicyatholaimus longigicandatus (4.53%) − | |
| Thalassironus britanicus (2.96%) + | Odontophora villoti (3.1%) + | Halalaimus gracilis (2.96%) − | Cyartonema germanicum (4.42%) − | |
| Thoonchus inermis (3.99%) + | ||||
| Synonchiella edax (3.83%) − | ||||
| Thalassironus britannicus (3.61%) − | ||||
| Feeding groups | 19.87% | 27.48% | 17.41% | 14.95% |
| 2A − | 2A − | 2A − | 2B − | |
| 1B + | ||||
| Tail shape | 6.91% | 16.71% | 16% | 9.6% |
| cla − | co − | co − | cla − | |
| Amphid shape | 16.62% | 24.2% | 25.36% | 14.5% |
| pk − | cr − | cr − | pk − | |
| pk − | id + | cr + | ||
| Adult length | 18.89% | 21.26% | 12.77% | 11.92% |
| 2–4 mm − | 2–4 mm − | 2–4 mm − | 2–4 mm − | |
| > 4 mm − | 1–2 mm + | |||
| c-p score | 30.13% | 32.86% | 30.16% | 16.92% |
| c-p3 − | c-p3 − | c-p3 − | c-p4 − | |
| c-p4 − | c-p4 − | c-p4 − | c-p2 + | |
| Utc vs. P1 | Utc vs. P2 | Utc vs. D1 | Utc vs. D2 | |
| Species | 34.62% | 36.76% | 32% | 41.67% |
| Microlaimus honestus (21.97%) elim | Microlaimus honestus (21.37%) elim | Nudora gerlachi (10.42%) + | Microlaimus honestus (11.2%) − | |
| Enoplolaimus longicaudatus (19.95%) + | Enoplolaimus longicaudatus (16.85%) + | Oncholaimus campylocercoïdes (9.2%) − | Metoncholaimus pristiurus(9.2%) + | |
| Oncholaimus campylocercoïdes (6.12%) − | Cyartonema germanicum (9.27%) − | Cyartonema germanicum (6.77%) + | Rhabditis sp. (8.4%) + | |
| Cyartonema germanicum (5.46%) − | Oncholaimus campylocercoïdes (6.42%) − | Microlaimus cyatholaimoïdes (6.77%) + | Oncholaimus campylocercoïdes (6.13%) − | |
| Trichotheristus mirabilis (4.69%) − | Trichotheristus mirabilis (4.48%) − | Thalassironus britannicus (5.21%) + | Microlaimus cyatholaimoïdes (6%) − | |
| Parasphaerolaimus paradoxus (3.53%) + | Paramonohystera proteus (3.39%) − | Trichotheristus mirabilis (5.21%) − | Thalassironus britannicus (5.6%) + | |
| Paramonohystera proteus (3.02%) + | Longicyatholaimus longicaudatus (2.93%) − | Paramonohystera proteus (5.21%) + | Daptonema fallax (5.6%) + | |
| Longicyatholaimus longicaudatus (2.84%) − | Microlaimus cyatholaimoïdes (2.59%) − | Rhabditis sp. (4.69%) + | Trichotheristus mirabilis (5.33%) − | |
| Theristus modicus (2.57%) = | Microlaimus honestus (4.34%) + | Cyartonema germanicum (4.13%) elim | ||
| Longicyatholaimus longicaudatus (4.17%) − | Longicyatholaimus longicaudatus (4%) + | |||
| Theristus modicus (3.47%) − | Oncholaimellus calvadosicus (4%) + | |||
| Synonchiella edax (3.3%) + | ||||
| Feeding groups | 16.9% | 21.78% | 10.3% | 25.52% |
| 2A − | 2A − | 2B − | 2A − | |
| 2A + | ||||
| Tail Shape | 15.3% | 15.25% | 18.78% | 12.89% |
| co − | co − | co + | cla + | |
| Amphid Shape | 20.32% | 23.47% | 12.44% | 17% |
| cr − | cr − | cr + | cr − | |
| sp − | pk + | |||
| Adult Length | 7.9% | 10.39% | 17.89% | 29% |
| 1–2 mm + | 2–4 mm − | < 1 mm + | 2–4 mm − | |
| 2–4 mm − | 1–2 mm − | 2–4 mm − | ||
| c-p score | 22.75% | 24.35% | 8% | 16.39% |
| c-p3 − | c-p3 − | c-p2 − | c-p3 − | |
| c-p5 + | c-p5 + | c-p4 − | c-p1 + | |
| Utc vs. P1D1 | Utc vs. P2D2 | Utc vs. P1D2 | Utc vs. P2D1 | |
| Species | 40.94% | 44.68% | 40.54% | 27.28% |
| Microlaimus honestus (18.7%) elim | Microlaimus honestus (18.46%) elim | Microlaimus honestus (20.18%) elim | Oncholaimus campylocercoïdes (9.45%) − | |
| Enoplolaimus longicaudatus (13.96%) + | Enoplolaimus longicaudatus (12.12%) + | Enoplolaimus longicaudatus (17.32%) + | Microlaimus honestus (7.72%) − | |
| Parasphaerolaimus paradoxus (7.28%) + | Oncholaimus campylocercoïdes (7.93%) − | Cyartonema germanicum (7.28%) − | Paramonohystera proteus (7.65%) − | |
| Oncholaimus campylocercoïdes (6.51%) − | Cyartonema germanicum (7.13%) − | Oncholaimus campylocercoïdes (5.19%) + | Trichotheristus mirabilis (6.4%) − | |
| Trichotheristus mirabilis (6.02%) + | Trichotheristus mirabilis (6.19%) + | Paramonohystera proteus (4.38%) − | Cyartonema germanicum (6.23%) − | |
| Thalassironus britannicus (3.99%) + | Rhabditis sp. (3.66%) + | Trichotheristus mirabilis (4.16%) + | Longicyatholaimus longicaudatus (5.32%) − | |
| Cyartonema germanicum (3.27%) − | Microlaimus cyatholaimoïdes (3.32%) − | Longicyatholaimus longicaudatus (3.71%) − | Synonchiella edax (4.93%) − | |
| Microlaimus cyatholaimoïdes (3.24%) − | Longicyatholaimus longicaudatus (3.24%) + | Spirinia parasitifera (2.76%) − | Microlaimus cyatholaimoïdes (4.07%) − | |
| Oncholaimellus calvadosicus (2.96%) − | Spirinia parasitifera (2.94%) elim | Parasphaerolaimus paradoxus (2.58%) + | Prochromadorella longicaudata (4.06%) − | |
| Paramonohystera proteus (2.86%) = | Oncholaimellus calvadosicus (2.92%) − | Theristus pertenuis (2.31%) − | Enoplolaimus longicaudatus (3.19%) − | |
| Oncholaimellus calvadosicus (3%) + | ||||
| Spirinia parasitifera (2.98%) − | ||||
| Thalassironus britannicus (2.98%) + | ||||
| Feeding groups | 16.81% | 21% | 24.96% | 9.84% |
| 2A − | 2A − | 2A − | 1B − | |
| 2B + | 2A − | |||
| Tail Shape | 10.91% | 19.24% | 20.43% | 7.81% |
| co − | co − | co − | cla − | |
| Amphid Shape | 17.69% | 26.28% | 26.71% | 10.63% |
| id + | cr − | cr − | pk − | |
| cr − | cr − | |||
| Adult Length | 13.69% | 17.1% | 13.3% | 9.82% |
| 2–4 mm − | 2–4 mm − | 1–2 mm − | 1–2 mm − | |
| 2–4 mm − | ||||
| c-p score | 23.55% | 28.67% | 28.06% | 9.82% |
| c-p3 − | c-p3 − | c-p3 − | c-p4 − | |
| c-p5 + | c-p4 − | c-p5 + | c-p2 − |
References
- Han, G.H.; Hur, H.G.; Kim, S.D. Ecotoxicological risk of pharmaceuticals from wastewater treatment plants in Korea: Occurrence and toxicity to Daphnia magna. Environ. Toxicol. Chem. 2006, 25, 265–271. [Google Scholar] [CrossRef] [PubMed]
- Kasprzyk-Hordern, B.; Dinsdale, R.M.; Guwy, A.J. The removal of pharmaceuticals, personal care products, endocrine disruptors and illicit drugs during wastewater treatment and its impact on the quality of receiving waters. Water Res. 2009, 43, 363–380. [Google Scholar] [CrossRef] [PubMed]
- Santos, L.H.M.L.M.; Araújo, A.N.; Fachini, A.; Pena, A.; Delerue-Matos, C.; Montenegro, M.C.B.S.M. Ecotoxicological aspects related to the presence of pharmaceuticals in the aquatic environment. J. Hazard. Mater. 2010, 175, 45–95. [Google Scholar] [CrossRef] [PubMed]
- Godoy, A.A.; Oliveira, Á.C.D.; Silva, J.G.M.; Azevedo, C.C.D.J.; Domingues, I.; Nogueira, A.J.A.; Kummrow, F. Single and mixture toxicity of four pharmaceuticals of environmental concern to aquatic organisms, including a behavioral assessment. Chemosphere 2019, 235, 373–382. [Google Scholar] [CrossRef] [PubMed]
- Halling-Sorensen, B.; Nors Nielsen, S.; Lanzky, P.F.; Ingerslev, F.; Holten Loutzhoft, H.C.; Jorgensen, S.E. Occurrence, fate and effects of pharmaceutical substances in the environment––A review. Chemosphere 1998, 36, 357–393. [Google Scholar] [CrossRef]
- Daughton, C.G.; Ternes, T.A. Special report: Pharmaceuticals and personal care products in the environment: Agents of subtle change? Environ. Health Perspect. 1999, 107, 907–938. [Google Scholar] [CrossRef] [PubMed]
- Kugathas, S.; Williams, R.J.; Sumpter, J.P. Prediction oenvironmental concentrations of glucocorticoids: The River Thames, UK, as an example. Environ. Int. 2012, 40, 15–23. [Google Scholar] [CrossRef]
- Heberer, T. Occurrence, fate, and removal of pharmaceutical residues in the aquatic environment: A review of recent research data. Toxicol. Lett. 2002, 131, 5–17. [Google Scholar] [CrossRef]
- Chang, H.; Hu, J.; Shao, B. Occurrence of natural and synthetic glucocorticoids in sewage treatment plants and receiving river waters. Environ. Sci. Technol. 2007, 41, 3462–3468. [Google Scholar] [CrossRef]
- Chang, H.; Wan, Y.; Hu, J. Determination and source apportionment of five classes of steroid hormones in urban rivers. Environ. Sci. Technol. 2009, 43, 7691–7698. [Google Scholar] [CrossRef]
- Schriks, M.; van Leerdam, J.A.; van der Linden, S.C.; van der Burg, B.; van Wezel, A.P.; de Voogt, P. High-Resolution Mass Spectrometric Identification and Quantification of Glucocorticoid Compounds in Various Wastewaters in The Netherlands. Environ. Sci. Technol. 2010, 44, 4766–4774. [Google Scholar] [CrossRef] [PubMed]
- Runnalls, T.J.; Margiotta-Casaluci, L.; Kugathas, S.; Sumpter, J.P. Pharmaceuticals in the aquatic environment: Steroids and antisteroids as high priorities for research. Hum. Ecol. Risk Assess. 2010, 16, 1318–1338. [Google Scholar] [CrossRef]
- McNeil, P.L.; Nebot, C.; Cepeda, A.; Sloman, K.A. Environmental concentrations of prednisolone alter visually mediated responses during early life stages of zebrafish (Danio rerio). Environ. Pollut. 2016, 218, 981–987. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Kugathas, S.; Sumpter, J.P. Synthetic glucocorticoids in the environment: First results on their potential impacts on fish. Environ. Sci. Technol. 2011, 45, 2377–2383. [Google Scholar] [CrossRef] [PubMed]
- Maayan, R.; Segal, R.; Feuerman, E.J.; Sandbank, M.; Kaufman, H. Simple methods for estimation of prednisone intake and metabolism. Biomed. Pharmacother. 1988, 42, 409–414. [Google Scholar]
- Hillegass, J.M.; Villano, C.M.; Cooper, K.R.; White, L.A. Matrix metalloproteinase-13 is required for zebra fish (Danio rerio) development and is a target for glucocorticoids. Toxicol. Sci. 2007, 100, 168–179. [Google Scholar] [CrossRef]
- Guo, Y.; Somerfield, P.J.M.; Zhang, Z. Large-scale patterns in the community structure and biodiversity of free living nematodes in the Bohai Sea, China. J. Mar. Biol. Assoc. UK 2001, 81, 755–763. [Google Scholar] [CrossRef]
- Balsamo, M.; Albertelli, G.; Ceccherelli, V.U.; Coccionia, R.; Colangeloc, M.A.; Curini-Gallettid, M.; Danovaroe, R.; D’Addabbof, R.; De Leonardisf, C.; Fabianob, M.; et al. Meiofauna of the Adriatic Sea: Present knowledge and future perspectives. Chem. Ecol. 2010, 26, 45–63. [Google Scholar] [CrossRef]
- Moreno, M.; Semprucci, F.; Vezzulli, L.; Balsamo, M.; Fabiano, M.; Albertelli, G. The use of nematodes in assessing ecological quality status in the Mediterranean coastal ecosystems. Ecol. Indic. 2011, 11, 328–336. [Google Scholar] [CrossRef]
- Ben Ali, M.; Hedfi, A.; Almalki, M.; Karachle, P.K.; Boufahja, F. Toxicity of hydroxychloroquine, a potential treatment for COVID-19, on free-living marine nematodes. Mar. Pollut. Bull. 2021, 167, 112361. [Google Scholar] [CrossRef]
- Mahmoudi, E.; Essid, E.; Beyrem, H.; Hedfi, A.; Boufahja, F.; Vitiello, P.; Aïssa, P. Individual and combined effects of lead and zinc of a free living marine nematode community: Results from microcosm experiments. J. Exp. Mar. Biol. Ecol. 2007, 343, 217–226. [Google Scholar] [CrossRef]
- Semprucci, F.; Balsamo, M. Key role of free-living nematodes in the marine ecosystem. In Nematodes: Morphology, Functions and Management Strategies; Boeri, F., Jordan, A.C., Eds.; NOVA Science Publishers, Inc.: Hauppauge, NY, USA, 2012; pp. 109–134. [Google Scholar]
- DellaGreca, M.; Fiorentino, A.; Isidori, M.; Lavorgna, M.; Previtera, L.; Rubino, M.; Temussi, F. Toxicity of prednisolone, dexamethasone and their photochemical derivatives on aquatic organisms. Chemosphere 2004, 54, 629–637. [Google Scholar] [CrossRef]
- Salgado, R.; Marques, R.; Noronha, J.P.; Mexia, J.T.; Carvalho, G.; Oehmen, A.; Reis, M.A.M. Assessing the diurnal variability of pharmaceutical and personal care products in a full-scale activated sludge plant. Environ. Pollut. 2011, 159, 2359–2367. [Google Scholar] [CrossRef] [PubMed]
- Wieser, W. Benthic studies in buzzards bay. II. The meiofauna. Limnol. Oceanogr. 1960, 5, 121–137. [Google Scholar] [CrossRef]
- Schratzberger, M.; Whomersley, P.; Warr, K.; Bolam, S.G.; Rees, H.L. Colonisation of various types of sediment by estuarine nematodes via lateral infaunal migration: A laboratory study. Mar. Biol. 2004, 145, 69–78. [Google Scholar] [CrossRef]
- Elarbaoui, S.; Richard, M.; Boufahja, F.; Mahmoudi, E.; Thomas-Guyonc, H. Effect of crude oil exposure and dispersant applicationon meiofauna: An intertidal mesocosm experiment. Environ. Sci. Process. Impacts 2015, 17, 997–1004. [Google Scholar] [CrossRef]
- Seinhorst, J.W. A rapid method for the transfer of nematodes from fixative to anhydrous glycerine. Nematologica 1959, 4, 67–69. [Google Scholar] [CrossRef]
- Platt, H.M.; Warwick, R.M. Free-Living Marine Nematodes. Part I. British Enoploids; Cambridge University: London, UK, 1983; 307p. [Google Scholar]
- Platt, H.M.; Warwick, R.M. Free-Living Marine Nematodes. Part II. British Chromadorids; Synopsis of the British Fauna (New Series); Cambridge University: London, UK, 1988; p. 38. [Google Scholar]
- Warwick, R.M.; Platt, H.M.; Somerfield, P.J. Free-Living Marine Nematodes. Part III. British Monohysterids; Synopsis of British Fauna (New Series No. 53); Field Studies Council: London, UK, 1998. [Google Scholar]
- Bezerra, T.N.; Decraemer, W.; Eisendle-Flockner, U.; Hodda, M.; Holovachov, O.; Leduc, D.; Miljutin, D.; Mokievsky, V.; Santiago, P.R.; Sharma, J.; et al. Nemys: World Database of Nematodes. 2020. Available online: http://nemys.ugent.be (accessed on 20 February 2021).
- Semprucci, F.; Balsamo, M.; Appolloni, L.; Sandulli, R. Assessment of ecological quality status along the Apulian coasts (eastern Mediterranean Sea) based on meiobenthic and nematode assemblages. Mar. Biodivers. 2018, 48, 105–115. [Google Scholar] [CrossRef]
- Thistle, D.; Lambshead, P.J.D.; Sherman, K.M. Nematode tail-shape groups respond to environmental differences in the deep-sea. Vie Milieu 1995, 45, 107–115. [Google Scholar]
- Wieser, W. Die Beziehung zwischen Mundhöhlengestalt, Ernäh rungsweiseund Vorkommen bei freilebenden marinen Nematoden. Arkiv. För. Zool. 1953, 2, 439–484. [Google Scholar]
- Bongers, T.; Alkemade, R.; Yeates, G.W. Interpretation of disturbance-induced maturity decrease in marine nematode assemblages by means of the maturity index. Mar. Ecol. Prog. Ser. 1991, 76, 135–142. [Google Scholar] [CrossRef]
- Bongers, T.; de Goede, R.G.M.; Korthals, G.W.; Yeates, G.W. An update to the cprating of nematode genera can be found in proposed changes of c-p classification for nematodes. Russ. J. Nematol. 1995, 3, 61–62. [Google Scholar]
- Clarke, K.R. Non-parametric multivariate analyses of changes in community structure. Aust. J. Ecol. 1993, 18, 117–143. [Google Scholar] [CrossRef]
- Clarke, K.R.; Warwick, R.M. Change in Marine Communities: An Approach to Statistical Analysis and Interpretation, 2nd ed.; PRIMER-E., Ltd., Plymouth Marine Laboratory: London, UK, 2001. [Google Scholar]
- Clarke, K.R.; Gorley, R.N. PRIMER v5: User Manual/Tutorial; PRIMER-E: Plymouth, UK, 2001; p. 91. [Google Scholar]
- Bray, J.R.; Curtis, J.T. An Ordination of the Upland Forest Communities of Southern Wisconsin. Ecol. Monogr. 1957, 27, 325–349. [Google Scholar] [CrossRef]
- Mahmoudi, E.; Essid, N.; Beyrem, H.; Hedfi, A.; Boufahja, F.; Vitiello, P.; Aissa, P. Effects of hydrocarbon contamination on a free-living marine nematode community: Results from microcosm experiments. Mar. Pollut. Bull. 2005, 50, 1197–1204. [Google Scholar] [CrossRef] [PubMed]
- Hedfi, A.; Mahmoudi, E.; Boufahja, F.; Beyrem, H.; Aïssa, P. Effects of increasing levels of nickel contamination on structure of offshore nematode communities in experimental microcosms. Bull. Environ. Contam. Toxicol. 2007, 79, 345–349. [Google Scholar] [CrossRef] [PubMed]
- Boufahja, F.; Sellami, B.; Dellali, M.; Aïssa, P.; Mahmoudi, E.; Beyrem, H. A microcosm experiment on the effects of permethrin on a free-living nematode assemblage. Nematology 2011, 13, 901–909. [Google Scholar]
- Wilson, K.S.; Matrone, G.; Livingstone, D.E.W.; Al-Dujaili, E.A.S.; Mullins, J.J.; Tucker, C.S.; Hadoke, P.W.F.; Kenyon, C.J.; Denvir, M.A. Physiological roles of glucocorticoids during early embryonic development of the zebrafish (Danio rerio). J. Physiol. 2013, 15, 6209–6220. [Google Scholar] [CrossRef]
- Barrett, R.; Chappell, C.; Quick, M.; Fleming, A. A rapid, high content, in vivo model of glucocorticoid-induced osteoporosis. Biotechnol. J. 2006, 1, 651–655. [Google Scholar] [CrossRef]
- Mesut, S. Blue Economy and Blue Ocean Strategy. J. Ecol. Nat. Resour. 2021, 5, 000263. [Google Scholar]
- Mesut, S. The Effects of the Ports and Water Transportation on the Aquatic Ecosystem. J. Biogen. Sci. Res. 2021, 10, 1–8. [Google Scholar]
- Hedfi, A.; Ben Ali, M.; Hassan, M.M.; Albogami, B.; Al-Zahrani, S.S.; Mahmoudi, E.; Karachle, P.K.; Rohal-Lupher, M.; Boufahja, F. Nematode traits after separate and simultaneous exposure to Polycyclic Aromatic Hydrocarbons (anthracene, pyrene and benzo [a] pyrene) in closed and open microcosms. Environ. Pollut. 2021, 276, 116759. [Google Scholar] [CrossRef] [PubMed]
- Semprucci, F.; Moreno, M.; Sbrocca, S.; Rocchi, M.; Albertelli, G.; Balsamo, M. The nematode assemblage as a tool for the assessment of marine ecological quality status: A case-study in the Central Adriatic Sea. Mediterr. Mar. Sci. 2013, 14, 48–57. [Google Scholar] [CrossRef][Green Version]
- Ürkmez, D.; Sezgin, M.; Bat, L. Use of nematode maturity index for the determination of ecological quality status: A case study from the Black Sea. J. Black Sea/Mediterr. Environ. 2014, 20, 96–107. [Google Scholar]
- Bongers, T. The Maturity Index: An ecological measure of environmental disturbance based on nematode species composition. Oecologia 1990, 83, 14–19. [Google Scholar] [CrossRef]
- Bongers, T. The Maturity Index, the evolution of nematode life history traits, adaptive radiation and cp-scaling. Plant Soil 1999, 212, 13–22. [Google Scholar] [CrossRef]
- Armenteros, M.; Pérez-García, J.A.; Ruiz-Abierno, A.; Díaz-Asencio, L.; Helguera, Y.; Vincx, M.; Decraemer, W. Effects of organic enrichment on nematode assemblages in a microcosm experiment. Mar. Environ. Res. 2010, 70, 374–382. [Google Scholar] [CrossRef]
- Losi, V.; Montefalcone, M.; Moreno, M.; Giovannetti, E.; Gaozza, L.; Grondona, M.; Albertelli, G. Nematodes as indicators of environmental quality in seagrass (Posidonia ceanic) meadows of the NW Mediterranean Sea. Adv. Oceanogr. Limnol. 2012, 3, 69–91. [Google Scholar] [CrossRef]
- Patrício, J.; Adão, H.; Neto, J.M.; Alves, A.S.; Traunspurger, W.; Marques, J.C. Do nematode and macrofauna assemblages provide similar ecological assessment information? Ecol. Indic. 2012, 14, 124–137. [Google Scholar] [CrossRef]
- Boufahja, F.; Vitiello, P.; Aïssa, P. More than 35 years of studies on marine nematodes from Tunisia: A checklist of species and their distribution. Zootaxa 2014, 3786, 269–300. [Google Scholar] [CrossRef]
- Rodrigues Da Silva, N.R.; Da Silva, M.C.; Fonseca Genevois, V.; De Esteves, A.M.; De Ley, P.; Decraemer, W.; Rieger, T.T.; Dos Santos Correia, M.T. Marine nematode taxonomy in the age of DNA: The present and future of molecular tools to assess their biodiversity. Nematology 2010, 12, 661–672. [Google Scholar] [CrossRef]
- Derycke, S.; Remerie, T.; Vierstraete, A.; Backeljau, T.; Vanfleteren, J.; Vincx, M.; Moens, T. Mitochondrial DNA variation and cryptic speciation within the free-living marine nematode Pellioditis marina. Mar. Ecol. Prog. Ser. 2005, 300, 91–103. [Google Scholar] [CrossRef]
- Derycke, S.; Hendrickx, F.; Backeljau, T.; D’Hondt, S.; Camphijn, L.; Vincx, M.; Moens, T. Effects of sublethal abiotic stressors on population growth and genetic diversity of Pellioditis marina (Nematoda) from the Westerschelde estuary. Aquat. Toxicol. 2007, 82, 110–119. [Google Scholar] [CrossRef] [PubMed]
- Derycke, S.; Van Vynckt, R.; Vanaverbecke, J.; Vincx, M.; Moens, T. Colonization patterns of nematode on decomposing algae in the estuarine environment: Community assembly and genetic structure of the dominant species Pellioditis marina. Limnol. Oceanogr. 2007, 52, 992–1001. [Google Scholar] [CrossRef]
- De Meester, N.; Derycke, S.; Bonte, D.; Moens, T. Salinity effects on the coexistence of cryptic species: A case study on marine nematodes. Mar. Biol. 2011, 158, 2717–2726. [Google Scholar] [CrossRef]


Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Allouche, M.; Ishak, S.; Nasri, A.; Harrath, A.H.; Alwasel, S.; Beyrem, H.; Pacioglu, O.; Boufahja, F. What Is the Impact of Dexamethasone and Prednisolone Glucocorticoids on the Structure of Meiobenthic Nematode Communities? Sustainability 2022, 14, 5344. https://doi.org/10.3390/su14095344
Allouche M, Ishak S, Nasri A, Harrath AH, Alwasel S, Beyrem H, Pacioglu O, Boufahja F. What Is the Impact of Dexamethasone and Prednisolone Glucocorticoids on the Structure of Meiobenthic Nematode Communities? Sustainability. 2022; 14(9):5344. https://doi.org/10.3390/su14095344
Chicago/Turabian StyleAllouche, Mohamed, Sahar Ishak, Ahmed Nasri, Abdel Halim Harrath, Saleh Alwasel, Hamouda Beyrem, Octavian Pacioglu, and Fehmi Boufahja. 2022. "What Is the Impact of Dexamethasone and Prednisolone Glucocorticoids on the Structure of Meiobenthic Nematode Communities?" Sustainability 14, no. 9: 5344. https://doi.org/10.3390/su14095344
APA StyleAllouche, M., Ishak, S., Nasri, A., Harrath, A. H., Alwasel, S., Beyrem, H., Pacioglu, O., & Boufahja, F. (2022). What Is the Impact of Dexamethasone and Prednisolone Glucocorticoids on the Structure of Meiobenthic Nematode Communities? Sustainability, 14(9), 5344. https://doi.org/10.3390/su14095344








