Evaluating the Flow Accelerated Corrosion and Erosion–Corrosion Behavior of a Pipeline Grade Carbon Steel (AISI 1030) for Sustainable Operations
Abstract
:1. Introduction
2. Materials and Methods
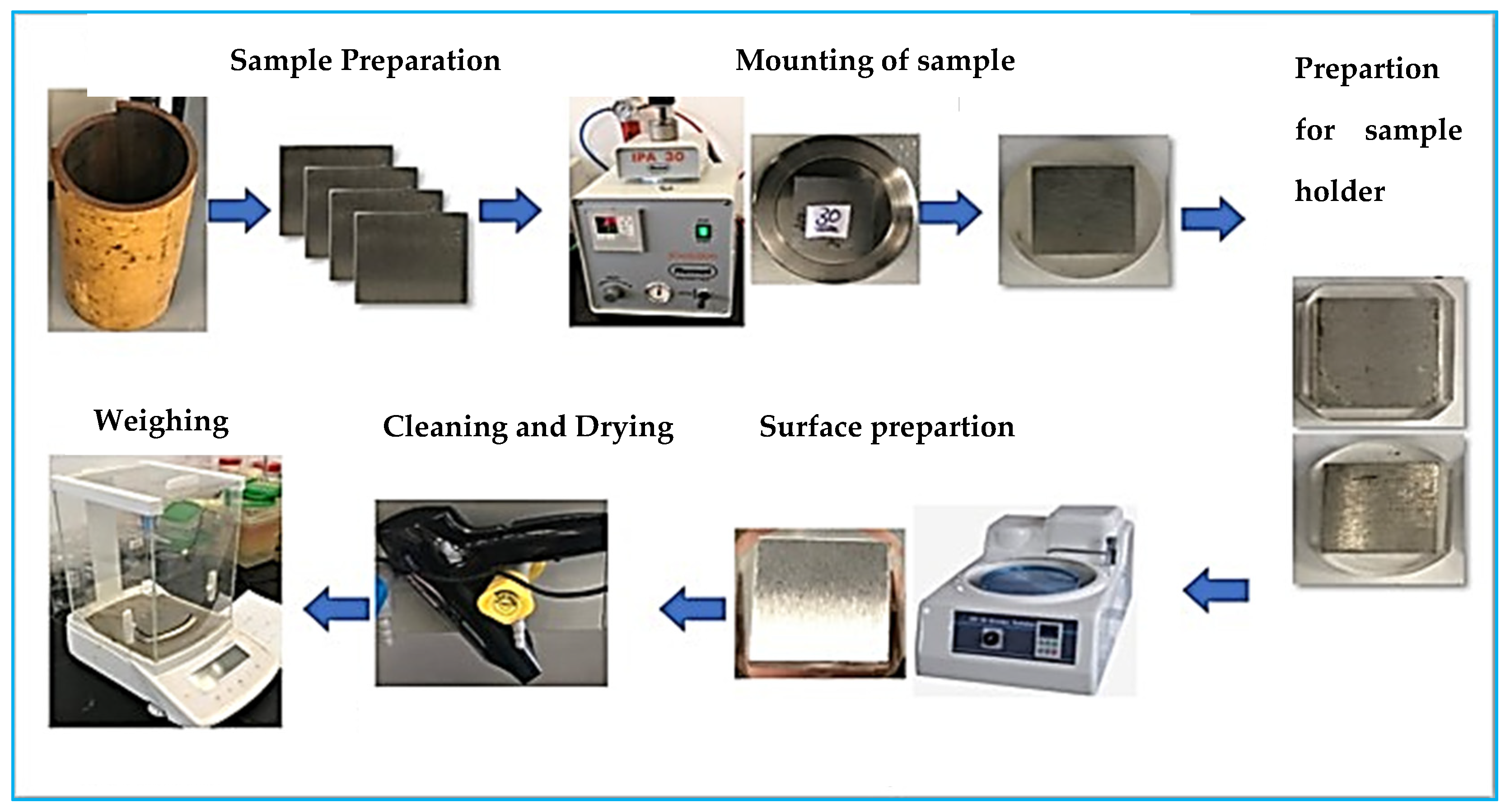
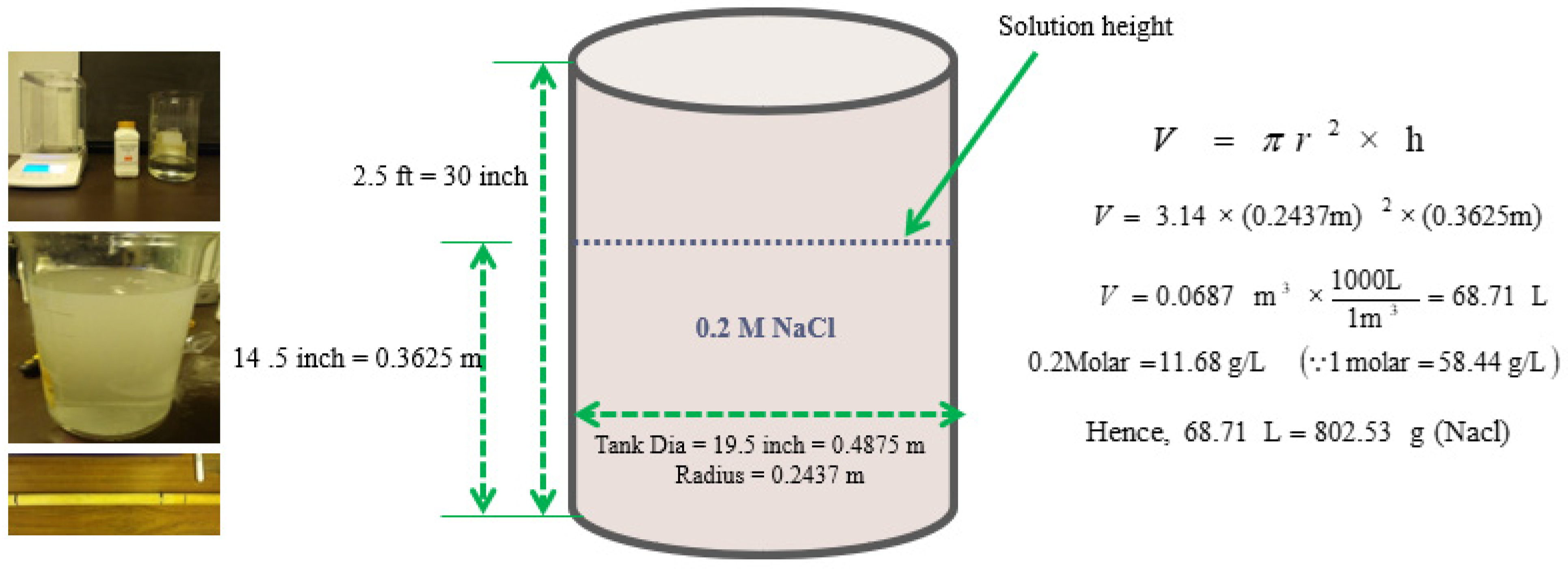
2.1. Erosion–Corrosion Test Apparatus and Material Preparation
2.2. Test Procedure
3. Results
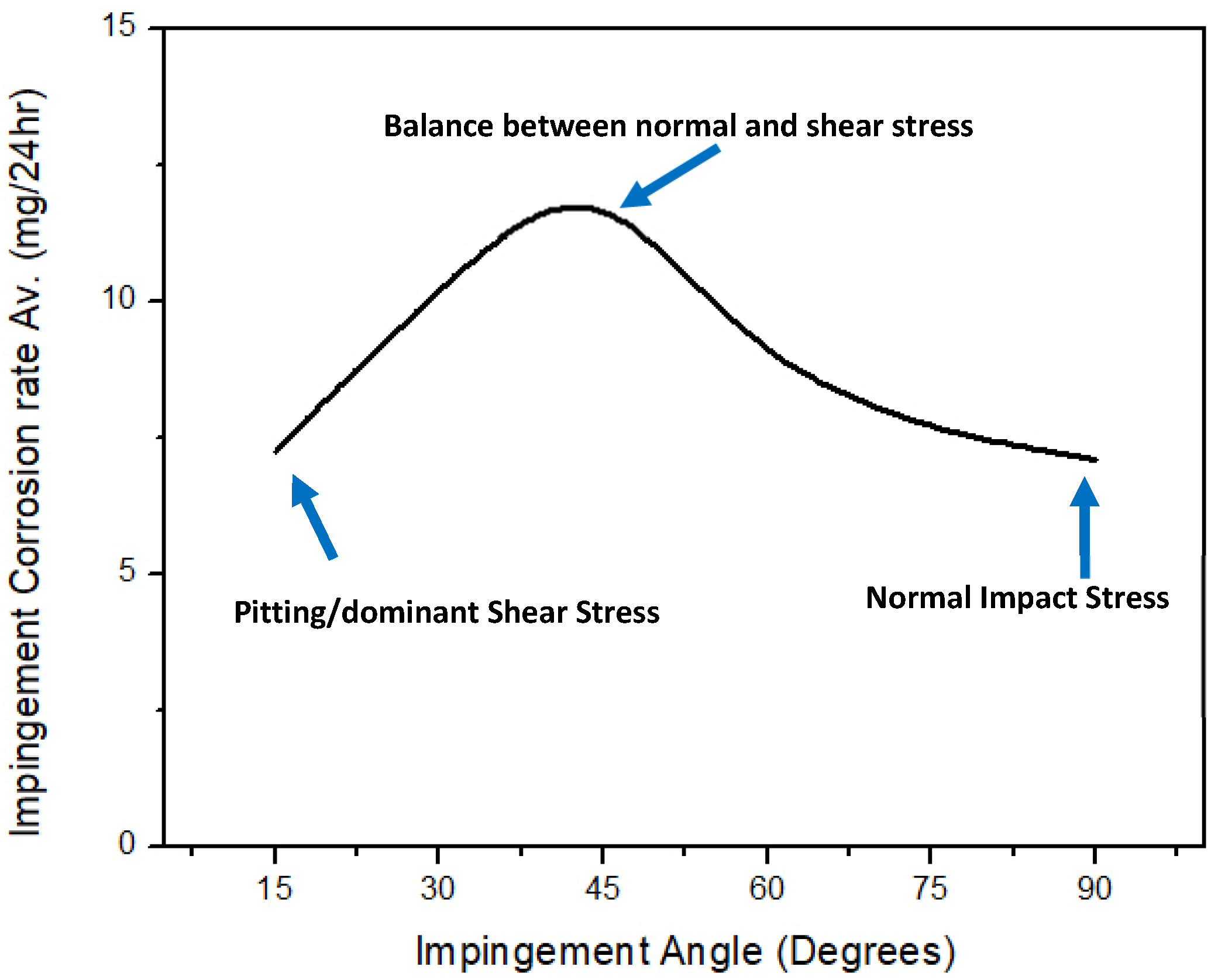
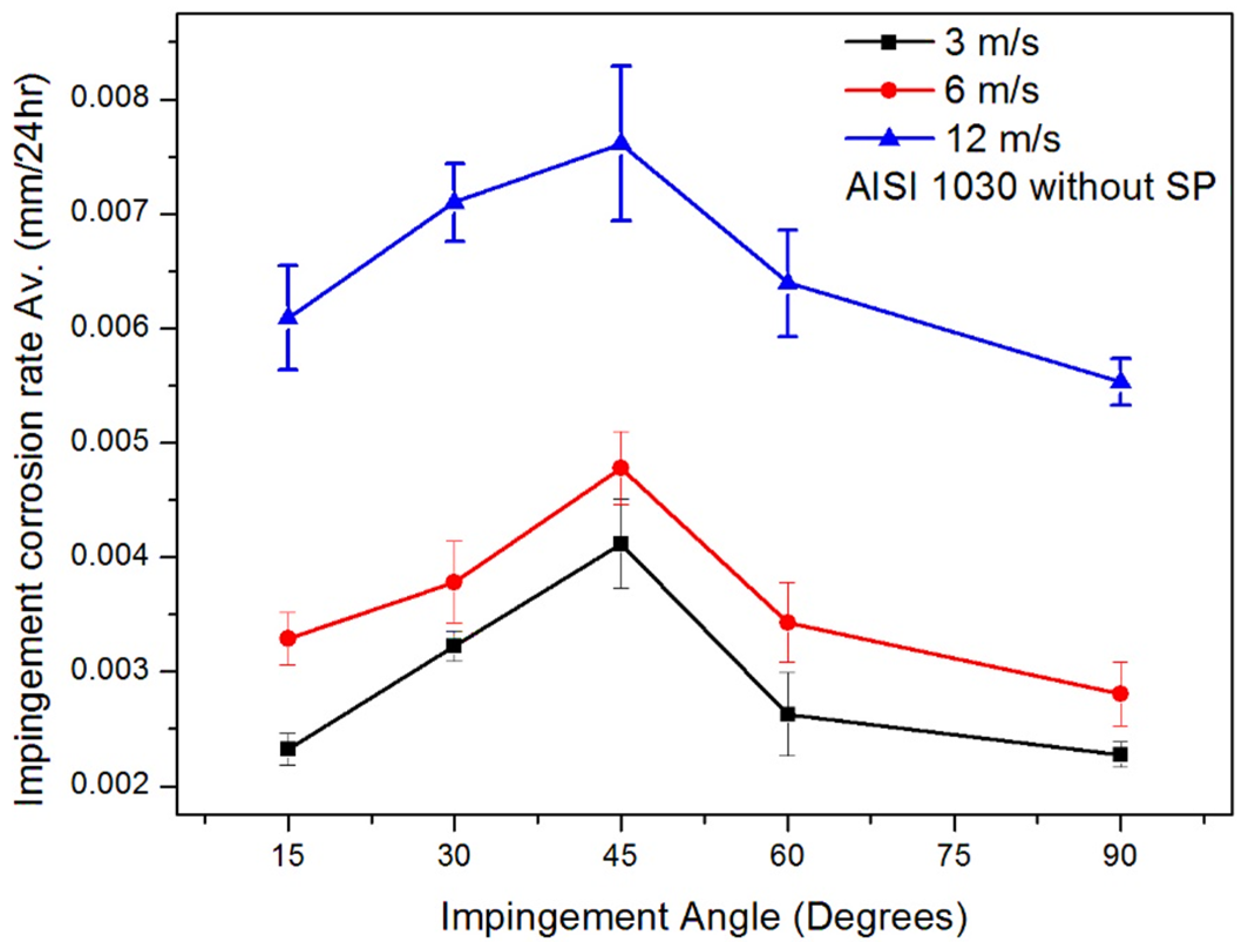
3.1. Effect of Jet Velocity on Impingement Corrosion (No Solid Particles)
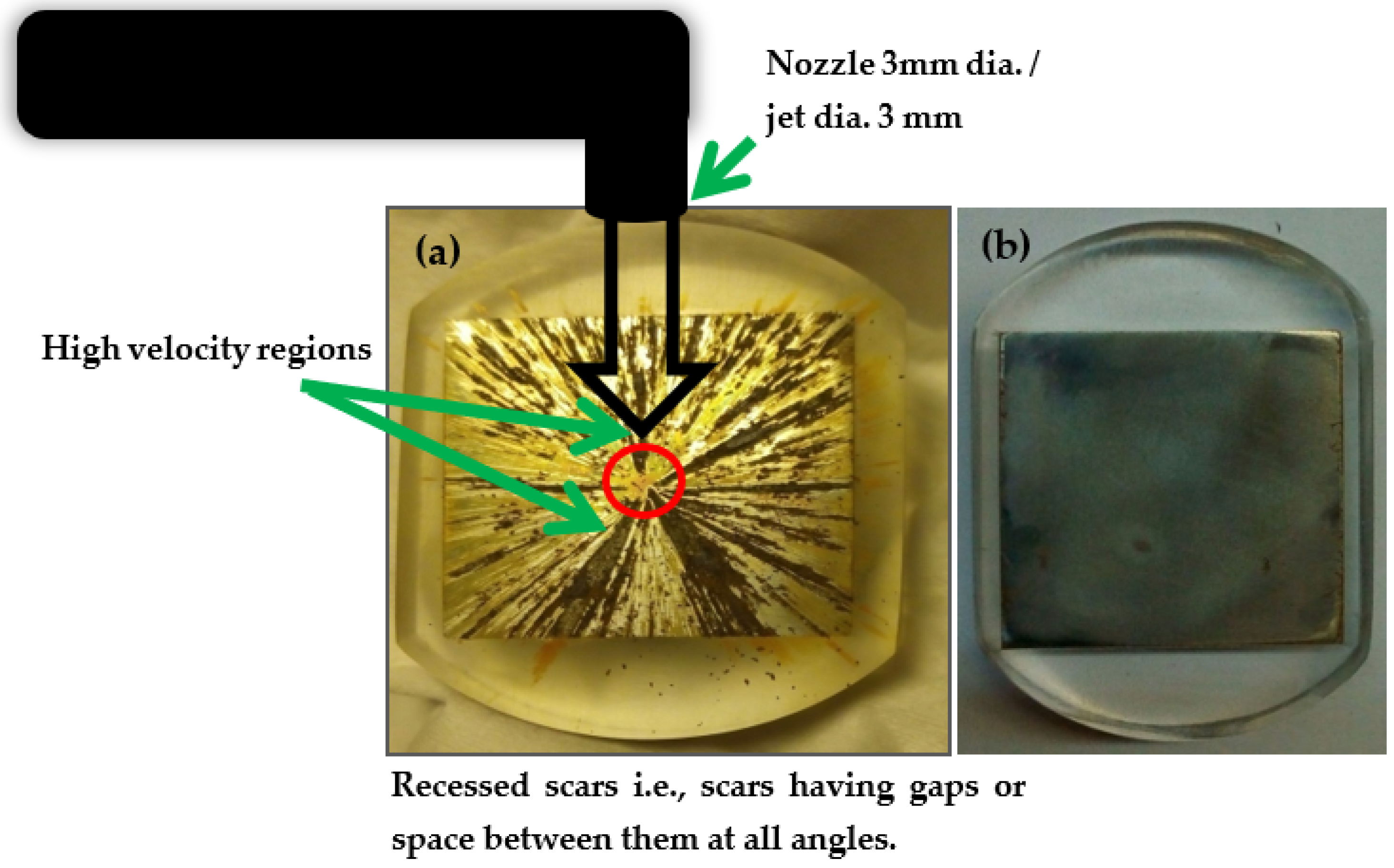
3.2. Surface Morphology and Corrosion Scar Features
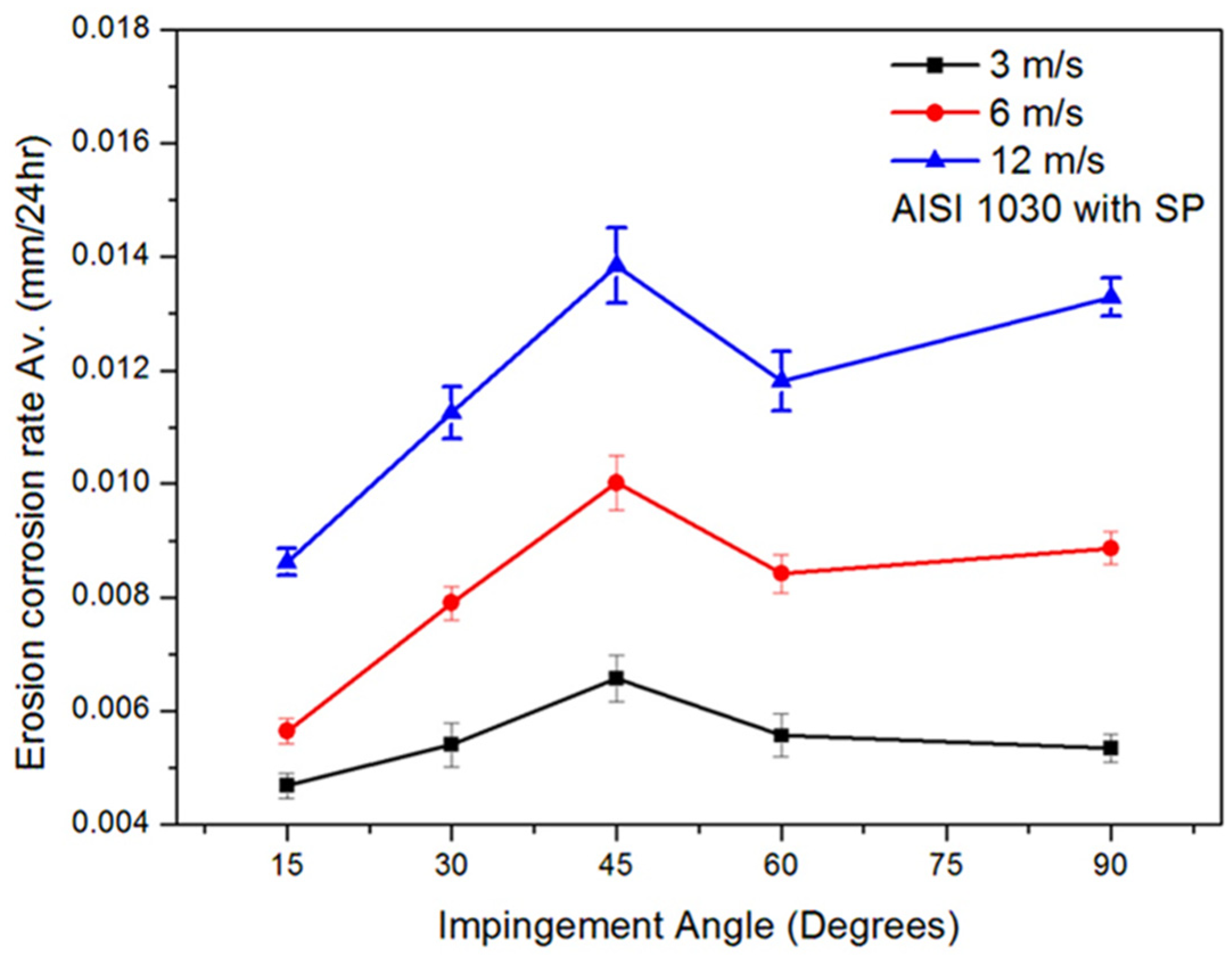
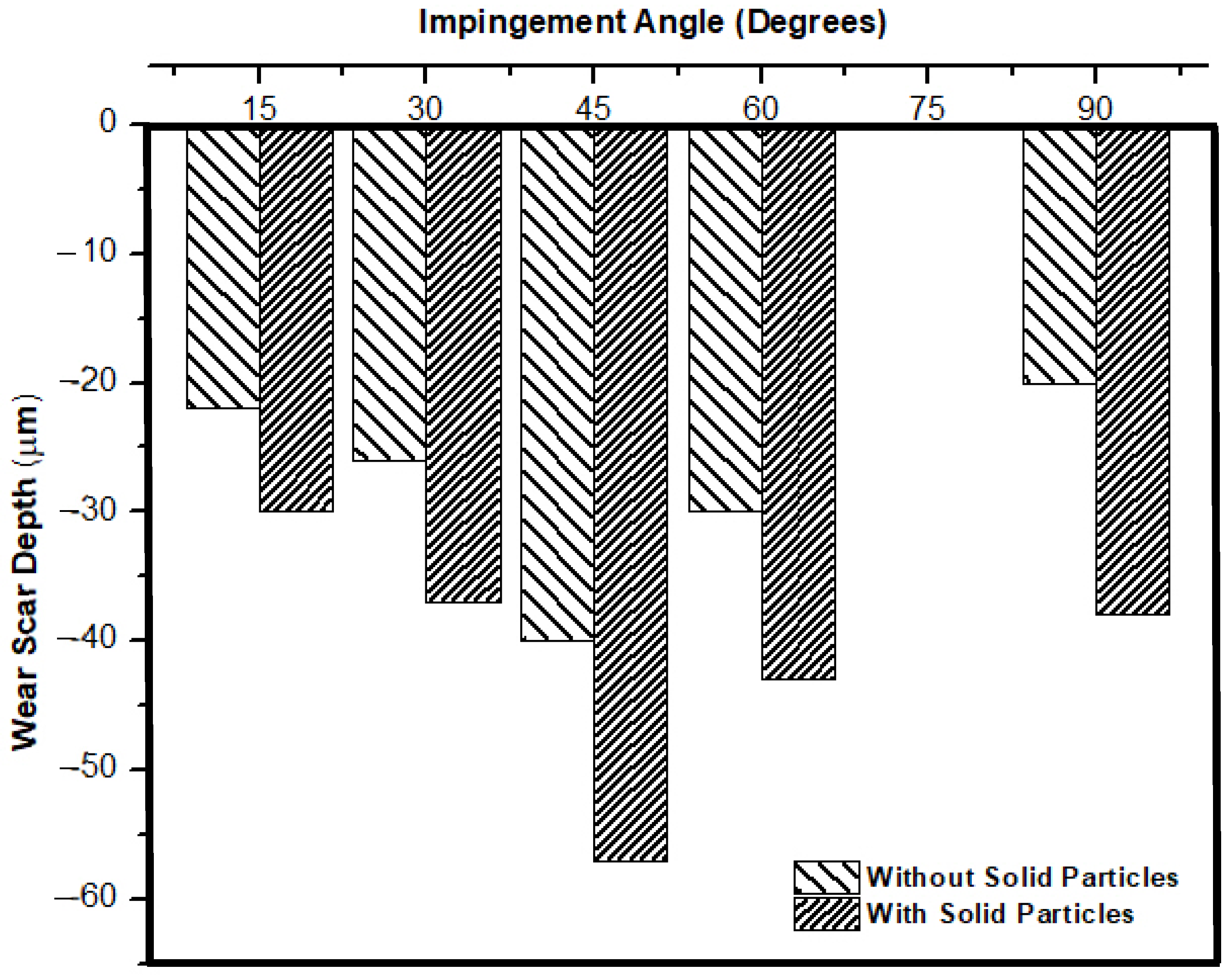
3.3. Effect of Jet Velocity and Angle on Erosion–Corrosion (with Solid Particles)
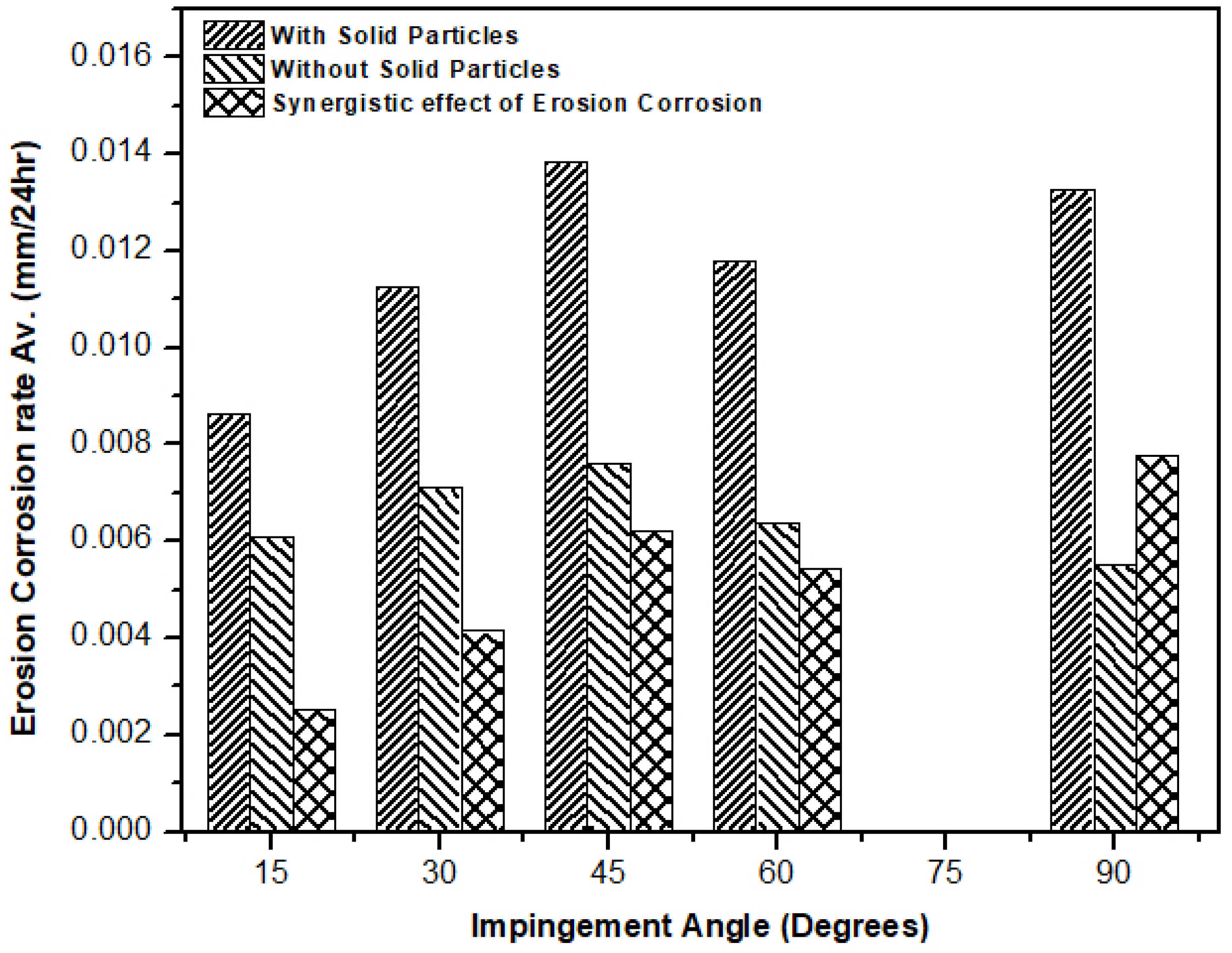
3.4. Effect of Erosion on Corrosion and Vice Versa
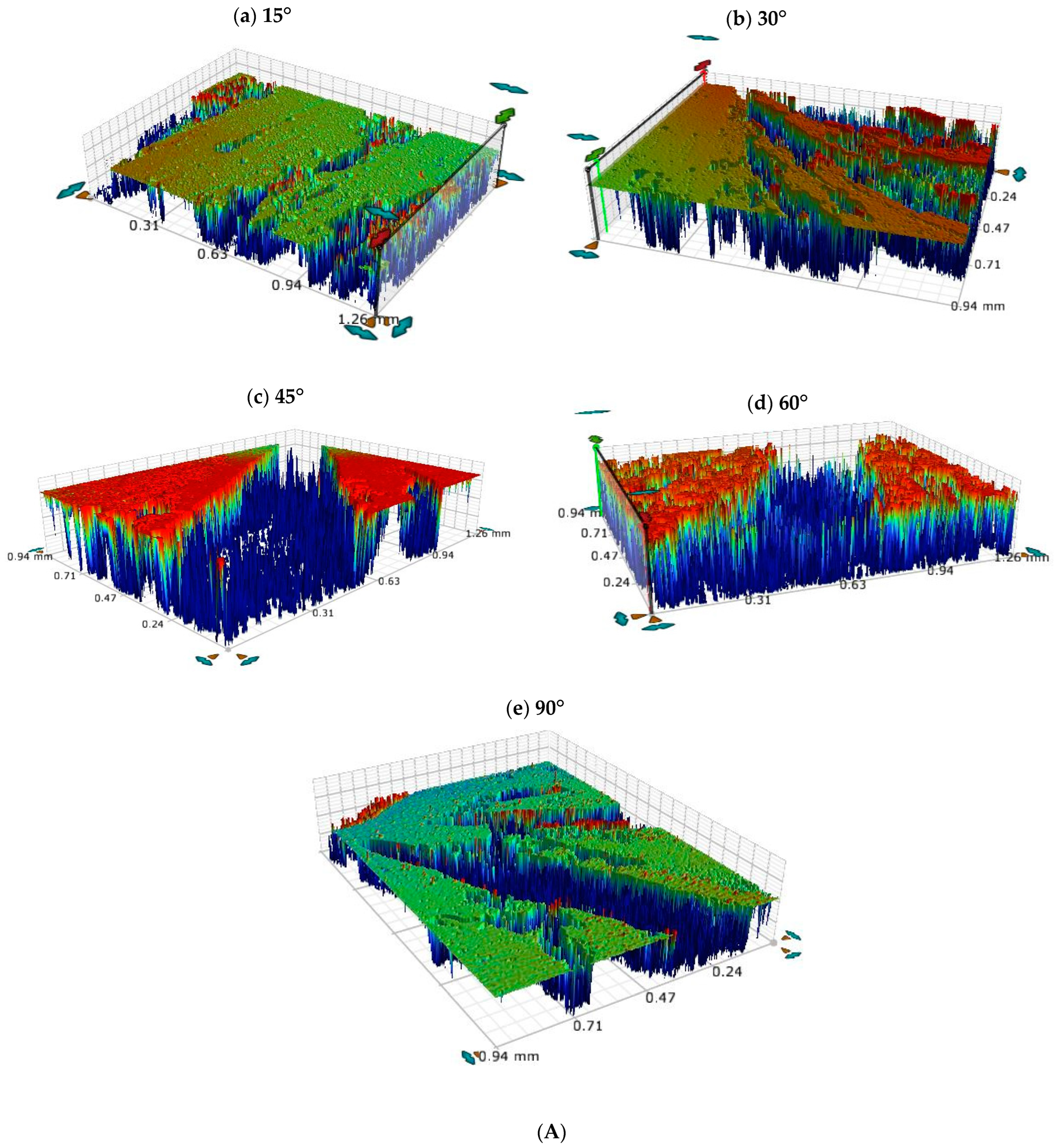
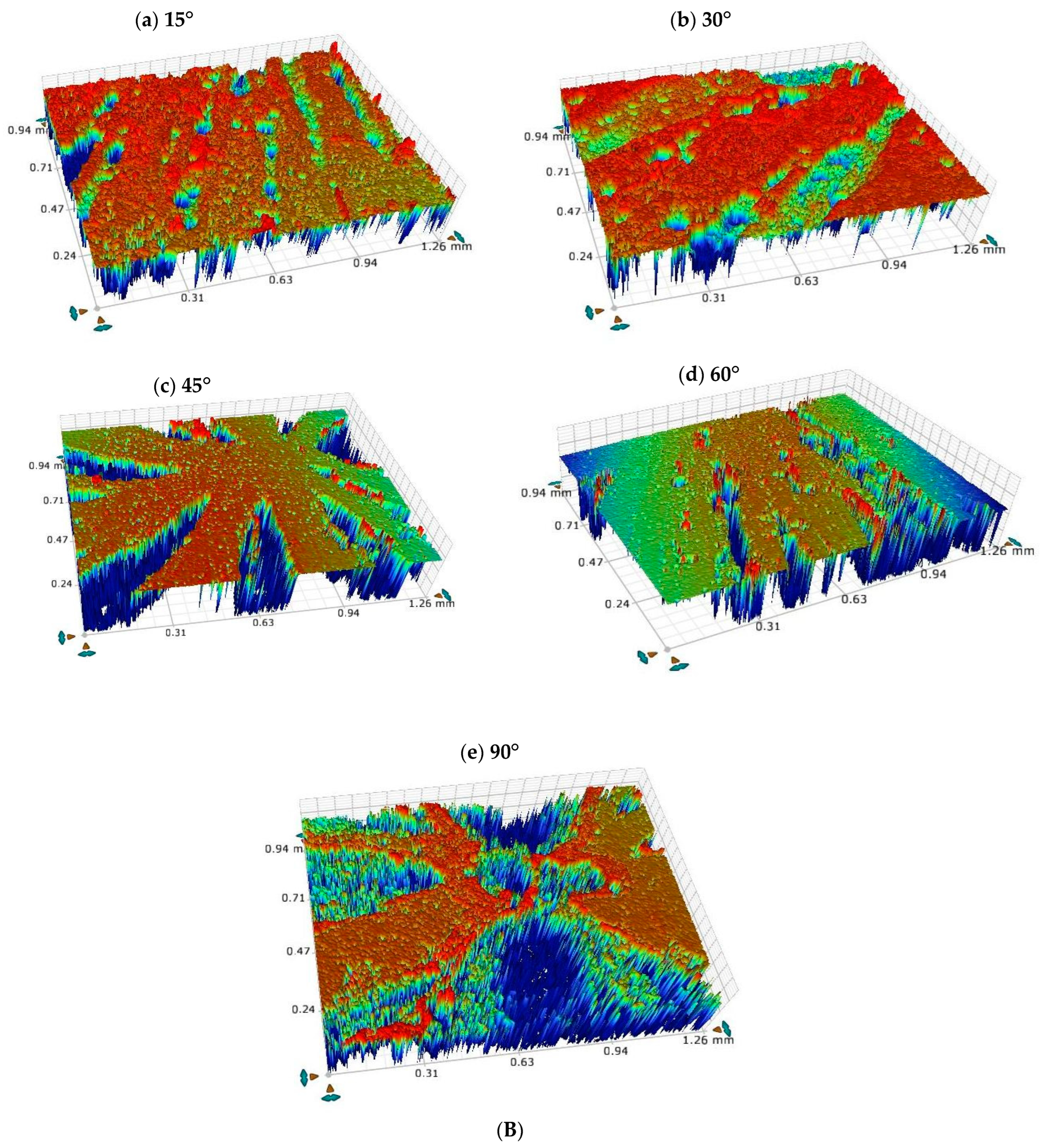
3.5. Optical Profilometric Studies of Corrosion/Wear Scars
4. Conclusions
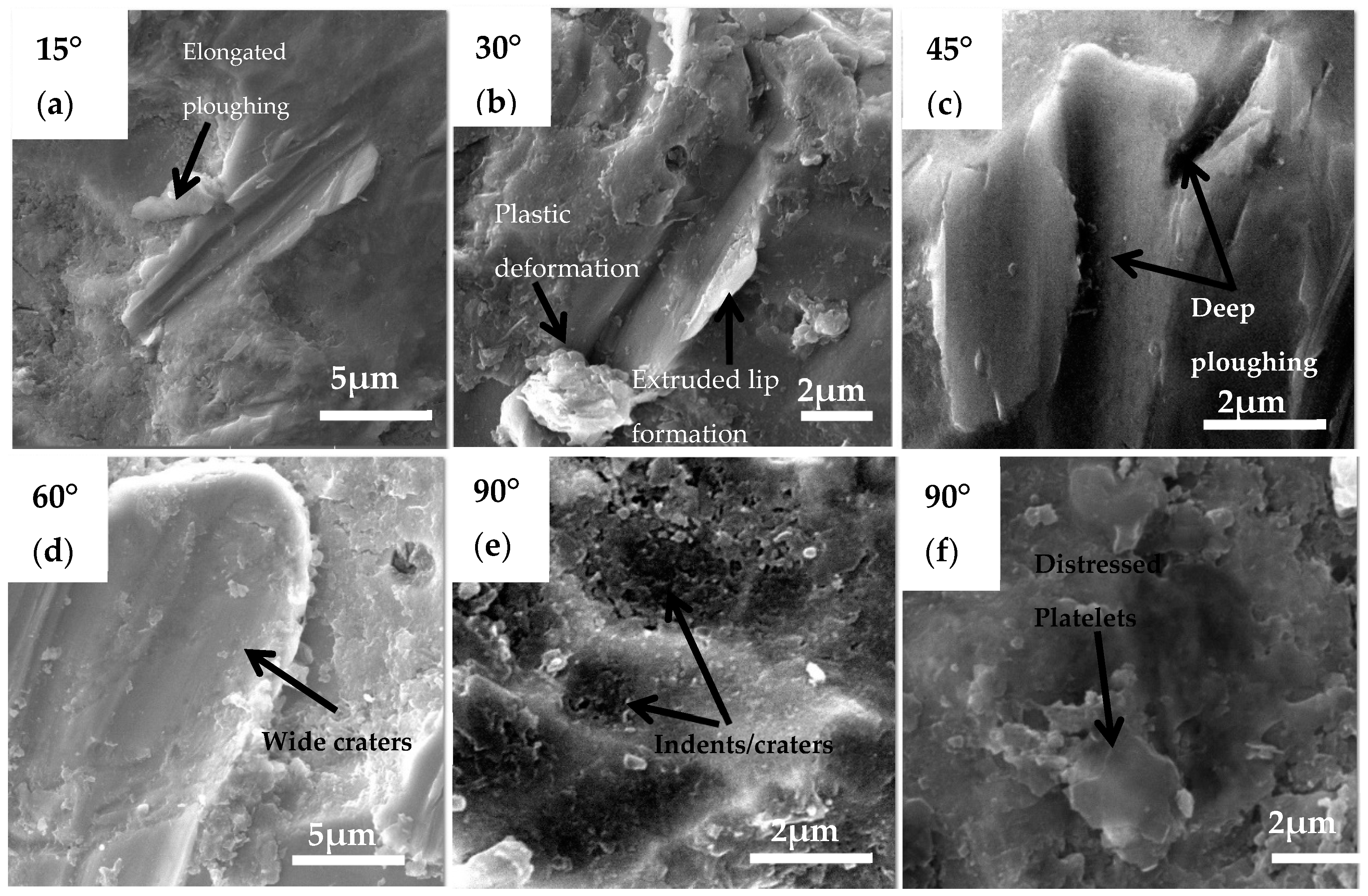
- Ploughing, elongated erosive tracks, and metal cutting were the dominating erosion–corrosion mechanisms at lower impingement angles, while extrusion, flattening of ridges, and fracture were dominant mechanisms at high impact angles.
- The increase in impingement corrosion and erosion–corrosion rates with an increase in impingement velocity was due to the presence of high shear and normal impact stresses. The maximum impingement corrosion and erosion–corrosion rates were found at a 45° impingement angle, as there was a balance between shearing force and normal impact force.
- Erosion had a significant effect on corrosion as particles cut the surface and activate the localized sites, which resulted in accelerated corrosion attack.
- The corrosion layer/oxide layer is continuously removed due to liquid jet impingement under high velocity conditions, as well as with the impact of the solid particles.
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Heidersbach, R. Metallurgy and Corrosion Control in Oil and Gas Production; John Wiley & Sons: Hoboken, NJ, USA, 2018. [Google Scholar]
- Levy, A.V. Solid Particle Erosion and Erosion-Corrosion of Materials; ASM International: Geauga County, OH, USA, 1995. [Google Scholar]
- Pasha, A.; Ghasemi, H.; Neshati, J. Synergistic Erosion–Corrosion Behavior of X-65 Carbon Steel at Various Impingement Angles. J. Tribol. 2017, 139, 011105. [Google Scholar] [CrossRef]
- Al Zoubi, W.; Kamil, M.P.; Fatimah, S.; Nashrah, N.; Ko, Y.G. Recent advances in hybrid organic-inorganic materials with spatial architecture for state-of-the-art applications. Prog. Mater. Sci. 2020, 112, 100663. [Google Scholar] [CrossRef]
- Al Zoubi, W.; Ko, Y.G. Chemical stability of synergistic inorganic materials for enhancing electrochemical performance. Compos. Sci. Technol. 2020, 199, 108383. [Google Scholar] [CrossRef]
- Javaherdashti, R.; Nwaoha, C.; Tan, H. Corrosion and Materials in the Oil and Gas Industries; CRC Press: Boca Raton, FL, USA, 2016. [Google Scholar]
- Neville, A.; Reyes, M.; Xu, H. Examining corrosion effects and corrosion/erosion interactions on metallic materials in aqueous slurries. Tribol. Int. 2002, 35, 643–650. [Google Scholar] [CrossRef]
- Verma, C.; Quraishi, M.; Rhee, K.Y. Aqueous Phase Polymeric Corrosion Inhibitors: Recent Advancements and Future Opportunities. J. Mol. Liq. 2021, 348, 118387. [Google Scholar] [CrossRef]
- Wang, J.; Shirazi, S.A. A CFD based correlation for mass transfer coefficient in elbows. Int. J. Heat Mass Transf. 2001, 9, 1817–1822. [Google Scholar] [CrossRef]
- Efird, K.D. Jet Impingement Testing for Accelerated Corrosion. In Corrosion 2000; NACE International: Houston, TX, USA, 2000; p. 00052. [Google Scholar]
- Fontana, M.G. Corrosion Engineering; McGraw-Hill Education: New York, NY, USA, 2005. [Google Scholar]
- Mazumder, Q.H. Prediction of erosion due to solid particle impact in single-phase and multiphase flows. J. Press. Vessel. Technol. 2007, 129, 576. [Google Scholar] [CrossRef]
- Giourntas, L.; Hodgkiess, T.; Galloway, A. Enhanced approach of assessing the corrosive wear of engineering materials under impingement. Wear 2015, 338, 155–163. [Google Scholar] [CrossRef] [Green Version]
- Khan, M.I.; Yasmin, T. Erosion–Corrosion of Low Carbon (AISI 1008 Steel) Ring Gasket Under Dynamic High Pressure CO2 Environment. J. Fail. Anal. Prev. 2014, 14, 537–548. [Google Scholar] [CrossRef]
- Sasaki, K.; Burstein, G. Erosion–corrosion of stainless steel under impingement by a fluid jet. Corros. Sci. 2007, 49, 92–102. [Google Scholar] [CrossRef]
- Neville, A.; Hodgkiess, T.; Xu, H. An electrochemical and microstructural assessment of erosion–corrosion of cast iron. Wear 1999, 233, 523–534. [Google Scholar] [CrossRef]
- Hu, X.; Neville, A. The effect of an impinging liquid-solid jet on the electrochemical corrosion of stainless steels. Mater. Corros. 2001, 52, 598–606. [Google Scholar] [CrossRef]
- Neville, A.; Hodgkiess, T.; Dallas, J. A study of the erosion-corrosion behaviour of engineering steels for marine pumping applications. Wear 1995, 186, 497–507. [Google Scholar] [CrossRef]
- Cui, Z.; Liu, Z.; Wang, L.; Li, X.; Du, C.; Wang, X. Effect of plastic deformation on the electrochemical and stress corrosion cracking behavior of X70 steel in near-neutral pH environment. Mater. Sci. Eng. A 2016, 677, 259–273. [Google Scholar] [CrossRef]
- Tian, H.; Wang, X.; Cui, Z.; Lu, Q.; Wang, L.; Lei, L.; Li, Y.; Zhang, D. Electrochemical corrosion, hydrogen permeation and stress corrosion cracking behavior of E690 steel in thiosulfate-containing artificial seawater. Corros. Sci. 2018, 144, 145–162. [Google Scholar] [CrossRef]
- Wang, L.; Liang, J.; Li, H.; Cheng, L.; Cui, Z. Quantitative study of the corrosion evolution and stress corrosion cracking of high strength aluminum alloys in solution and thin electrolyte layer containing Cl. Corros. Sci. 2021, 178, 109076. [Google Scholar] [CrossRef]
- Toor, I.U.; Alashwan, Z.; Badr, H.M.; Ben-Mansour, R.; Shirazi, S.A. Effect of Jet Impingement Velocity and Angle on CO2 Erosion–Corrosion with and without Sand for API 5L-X65 Carbon Steel. Materials 2020, 13, 2198. [Google Scholar] [CrossRef]
- Toor, I.U.; Irshad, H.M.; Badr, H.M.; Samad, M.A. The effect of impingement velocity and angle variation on the erosion corrosion performance of API 5L-X65 carbon steel in a flow loop. Metals 2018, 8, 402. [Google Scholar] [CrossRef] [Green Version]
- Abedini, M.; Ghasemi, H. Synergistic erosion–corrosion behavior of Al–brass alloy at various impingement angles. Wear 2014, 319, 49–55. [Google Scholar] [CrossRef]
- Liu, Y.; Zhao, Y.; Yao, J. Synergistic erosion–corrosion behavior of X80 pipeline steel at various impingement angles in two-phase flow impingement. Wear 2021, 466–467, 203572. [Google Scholar] [CrossRef]
- Azarian, N.S.; Ghasemi, H.M.; Monshi, M.R. Synergistic erosion and corrosion behavior of AA5052 aluminum alloy in 3.5 wt% NaCl solution under various impingement angles. J. Bio-Tribo-Corros. 2015, 1, 10. [Google Scholar] [CrossRef] [Green Version]
- Rajahram, S.; Harvey, T.; Wood, R. Evaluation of a semi-empirical model in predicting erosion–corrosion. Wear 2009, 267, 1883–1893. [Google Scholar] [CrossRef]
- Finnie, I. Erosion of surface by solid particles. Wear 1960, 3, 87–103. [Google Scholar] [CrossRef]
- Liang, G.; Peng, X.; Xu, L.; Cheng, Y.F. Erosion-corrosion of carbon steel pipes in oil sands slurry studied by weight-loss testing and CFD simulation. J. Mater. Eng. Perform. 2013, 22, 3043–3048. [Google Scholar] [CrossRef]
- Jingjun, L.; Yuzhen, L.; Xiaoyu, L. Numerical simulation for carbon steel flow-induced corrosion in high-velocity flow seawater. Anti-Corros. Methods Mater. 2008, 55, 66–72. [Google Scholar] [CrossRef]
- Burstein, G.; Sasaki, K. Effect of impact angle on the erosion–corrosion of 304L stainless steel. Wear 1995, 186, 80–94. [Google Scholar] [CrossRef]
- Islam, M.A.; Alam, T.; Farhat, Z.N.; Mohamed, A.; Alfantazi, A. Effect of microstructure on the erosion behavior of carbon steel. Wear 2015, 332, 1080–1089. [Google Scholar] [CrossRef]
- Neville, A.; Hodgkiess, T. Study of effect of liquid corrosivity in liquid-solid impingement on cast iron and austenitic stainless steel. Br. Corros. J. 1997, 32, 197–205. [Google Scholar] [CrossRef]
- Finnie, I. Some reflections on the past and future of erosion. Wear 1995, 186–187, 1–10. [Google Scholar] [CrossRef]
- Kermani, M.; Morshed, A. Carbon dioxide corrosion in oil and gas productiona compendium. Corrosion 2003, 59, NACE-03080659. [Google Scholar] [CrossRef]
- Meng, H.; Ludema, K. Wear models and predictive equations: Their form and content. Wear 1995, 181, 443–457. [Google Scholar] [CrossRef]
- Nesic, S.; Postlethwaite, J.; Olsen, S. An electrochemical model for prediction of corrosion of mild steel in aqueous carbon dioxide solutions. Corrosion 1996, 52, 280–294. [Google Scholar] [CrossRef]
- Zhou, S.; Stack, M.; Newman, R. Characterization of synergistic effects between erosion and corrosion in an aqueous environment using electrochemical techniques. Corrosion 1996, 52, 934–946. [Google Scholar] [CrossRef]
- Matsumura, M.; Oka, Y.; Hiura, H.; Yano, M. The role of passivating film in preventing slurry erosion-corrosion of austenitic stainless steel. ISIJ Int. 1991, 31, 168–176. [Google Scholar] [CrossRef] [Green Version]
- Postlethwaite, J. Effect of chromate inhibitor on the mechanical and electrochemical components of erosion-corrosion in aqueous slurries of sand. Corrosion 1981, 37, 1–5. [Google Scholar] [CrossRef]
- Li, W.; Pots, B.F.M.; Brown, B.; Kee, K.; Nesic, S. A direct measurement of wall shear stress in multiphase flow—Is it an important parameter in CO2 corrosion of carbon steel pipelines? Corros. Sci. 2016, 110, 35–45. [Google Scholar] [CrossRef]
- Gulbrandsen, E.; Grana, A. Testing of carbon dioxide corrosion inhibitor perfor-mance at high flow velocities in jet impingement geometry. Effects of mass transfer and flow forces. Corrosion 2007, 63, 1009–1020. [Google Scholar] [CrossRef]
- Hou, Y.; Aldrich, C.; Lepkova, K.; Kinsella, B. Detection of under deposit corrosion in a CO2 environment by using electrochemical noise and recurrence quantification analysis. Electrochim. Acta 2018, 274, 160–169. [Google Scholar] [CrossRef]
- Zheng, Z.B.; Zheng, Y.G.; Zhou, X.; He, S.Y.; Sun, W.H.; Wang, J.Q. Determination of the critical flow velocities for erosion-corrosion of passive materials under impingement by NaCl solution containing sand. Corros. Sci. 2014, 88, 187–196. [Google Scholar] [CrossRef]
- Ige, O.; Umoru, L. Effects of shear stress on the erosion-corrosion behaviour of X-65 carbon steel: A combined mass-loss and profilometry study. Tribol. Int. 2016, 94, 155–164. [Google Scholar] [CrossRef]
- Nguyen, Q.; Nguyen, V.; Lim, C.; Trinh, Q.; Sankaranarayanan, S.; Zhang, Y.; Gupta, M. Effect of impact angle and testing time on erosion of stainless steel at higher velocities. Wear 2014, 321, 87–93. [Google Scholar] [CrossRef]





| Material | Angle of Highest Synergistic Rate | Operating Conditions under Consideration | Reference | ||
|---|---|---|---|---|---|
| Velocity of Jet | Testing Time | Testing Angle Range | |||
| X65 Carbon Steel | 25° | 6.5 m/s | 30 min | 20–90° | [3] |
| Al–brass alloy | 20 and 90° | 6 m/s | 30 min | 20–90° | [24] |
| X80 steel | 90° | 12 m/s | 1 h | 30–90 | [25] |
| AA5052 Aluminum Alloy | 30° | 3 m/s | 30 min | 25–90 | [26] |
| AISI 1030 | 45 and 90° | 12 m/s | 24 h (significant time given compared to other studies) | 15–90° (wide range of angles) | Present Study |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Irshad, H.M.; Toor, I.U.; Badr, H.M.; Samad, M.A. Evaluating the Flow Accelerated Corrosion and Erosion–Corrosion Behavior of a Pipeline Grade Carbon Steel (AISI 1030) for Sustainable Operations. Sustainability 2022, 14, 4819. https://doi.org/10.3390/su14084819
Irshad HM, Toor IU, Badr HM, Samad MA. Evaluating the Flow Accelerated Corrosion and Erosion–Corrosion Behavior of a Pipeline Grade Carbon Steel (AISI 1030) for Sustainable Operations. Sustainability. 2022; 14(8):4819. https://doi.org/10.3390/su14084819
Chicago/Turabian StyleIrshad, Hafiz Muzammil, Ihsan Ulhaq Toor, Hassan Mohamed Badr, and Mohammed Abdul Samad. 2022. "Evaluating the Flow Accelerated Corrosion and Erosion–Corrosion Behavior of a Pipeline Grade Carbon Steel (AISI 1030) for Sustainable Operations" Sustainability 14, no. 8: 4819. https://doi.org/10.3390/su14084819
APA StyleIrshad, H. M., Toor, I. U., Badr, H. M., & Samad, M. A. (2022). Evaluating the Flow Accelerated Corrosion and Erosion–Corrosion Behavior of a Pipeline Grade Carbon Steel (AISI 1030) for Sustainable Operations. Sustainability, 14(8), 4819. https://doi.org/10.3390/su14084819







