Increasing the Circularity of Packaging along Pharmaceuticals Value Chain
Abstract
:1. Introduction
2. Materials and Methods
2.1. PhP Materials and Range
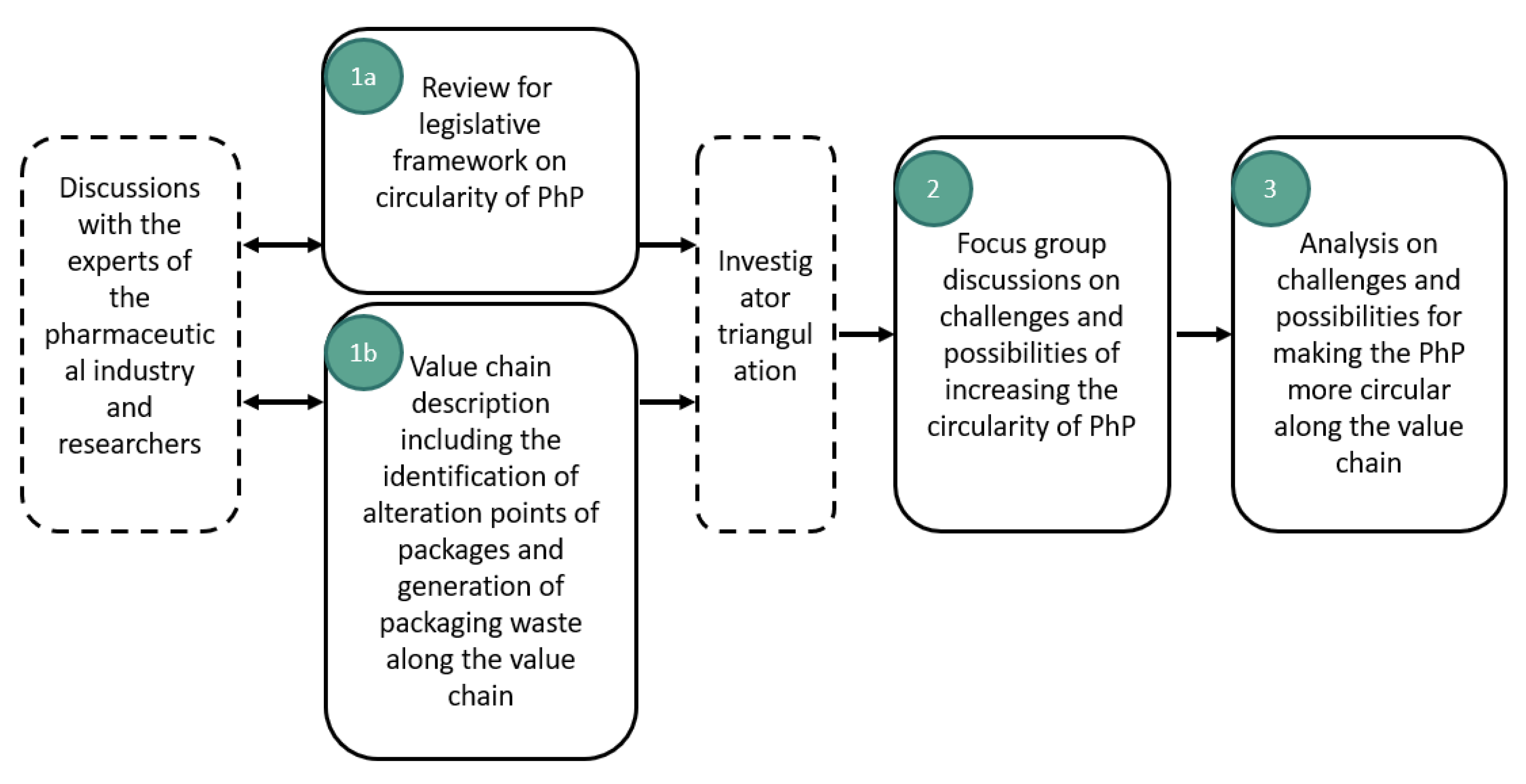
2.2. The Methodology
3. Results
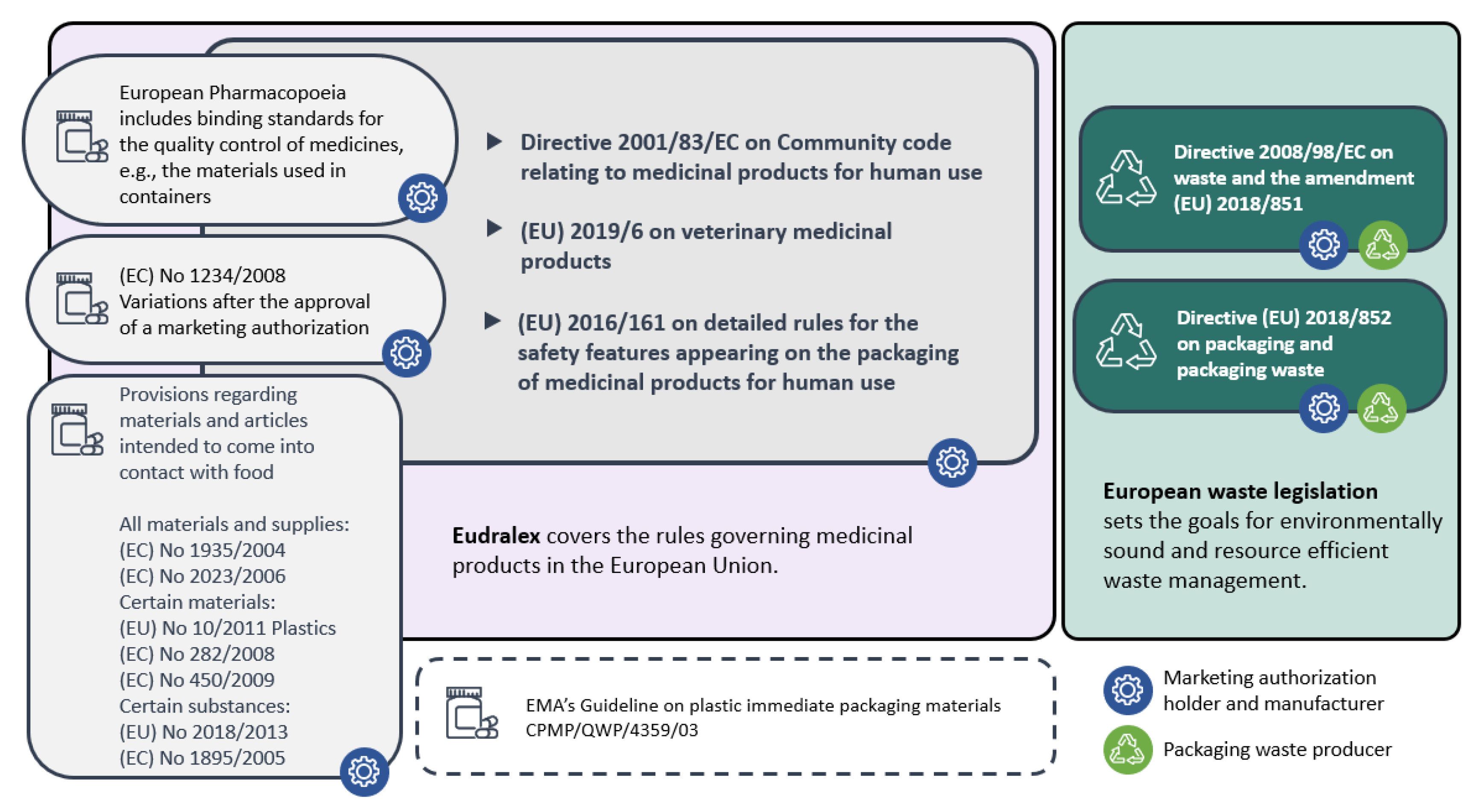
3.1. The Legislative Framework
3.1.1. Legislation on Pharmaceutical Packaging
‘It is necessary to exercise control over the entire chain of distribution of medicinal products, from their manufacture or import into the community through to supply to the public, so as to guarantee that such products are stored, transported and handled in suitable conditions.’
3.1.2. Waste Legislation on Packages
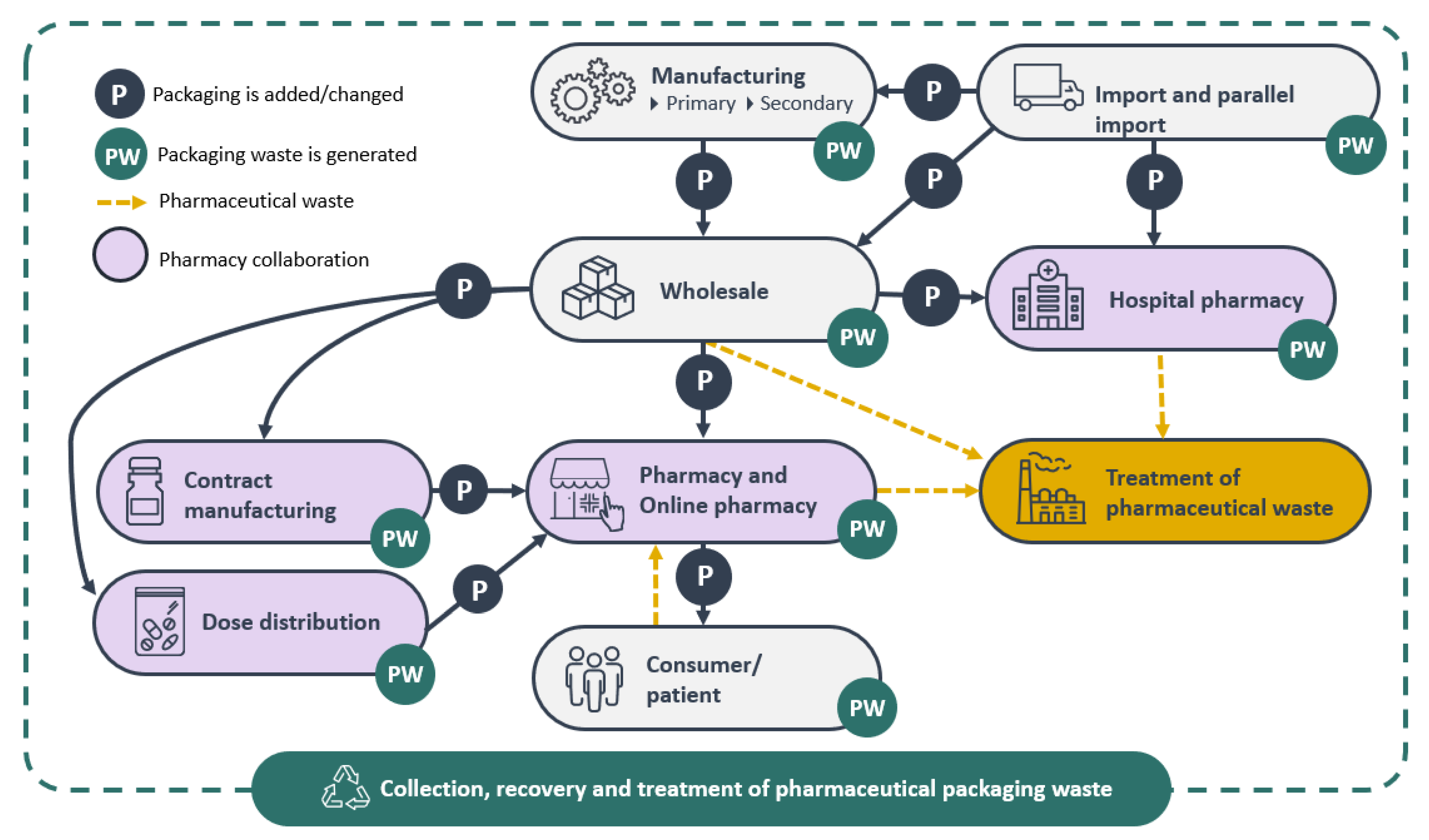
3.2. The Pharmaceutical Packaging Value Chain
3.3. Focus Group Discussions
3.3.1. Challenges in the Development of Pharmaceutical Packaging from a Circular Economy Perspective
3.3.2. Possible Changes in Pharmaceutical Packaging
3.3.3. Categorized Barriers and Possibilities
4. Discussion
5. Conclusions
Author Contributions
Funding
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A
| Number of Participants in Groups | Organisation |
|---|---|
| Group 1 | |
| Participant 1#, | Pharmacy |
| Participant 2#, | Pharmaceutical company |
| Participant 3#, | Distributor of pharmaceuticals |
| Participant 4#, | University |
| Participant 5#, | Parallel import of medicines |
| Participant 6#, | Governmental research institute |
| Participant 7#, | University |
| Facilitator | |
| Group 2 | |
| Participant 8#, | Dose dispending |
| Participant 9#, | Research and communication |
| Participant 10#, | State administration |
| Participant 11#, | Plastic waste recycling company |
| Participant 12#, | Pharmaceutical information centre |
| Facilitator | |
| Group 3 | |
| Participant 13#, | Dose dispending |
| Participant 14#, | University |
| Participant, 15#, | Municipal waste management company |
| Participant 16#, | Distributor of pharmaceuticals |
| Participant 17#, | Pharmacy |
| Participant 18#, | State administration |
| Facilitator | |
| Group 4 | |
| Participant 19#, | Pharmaceutical company |
| Participant 20#, | Governmental research institute |
| Participant 21#, | Dose dispensing |
| Participant 22#, | Producer responsibility of packaging |
| Participant 23#, | Distribution of pharmaceuticals |
| Participant 24#, | University |
| Facilitator |
References
- WHO Guidelines on Packaging for Pharmaceutical Products, Annex 9; World Health Organization: Geneva, Switzerland, 2002; p. 156.
- Zadbuke, N.; Shahi, S.; Gulecha, B.; Padalkar, A.; Thube, M. Recent trends and future of pharmaceutical packaging technology. J. Pharm. Bioallied Sci. 2013, 5, 98. [Google Scholar] [CrossRef] [PubMed]
- European Commission. A New Circular Economy Action Plan for a Cleaner and More Competitive Europe; European Commission: Brussels, Belgium, 2020; p. 19. [Google Scholar]
- Eurostat Packaging Waste Statistics. Data Extracted on 18 October 2021. Available online: https://ec.europa.eu/eurostat/statistics-explained/index.php?title=Packaging_waste_statistics#Time_series_of_packaging_waste (accessed on 7 February 2022).
- Ghisellini, P.; Cialani, C.; Ulgiati, S. A review on circular economy: The expected transition to a balanced interplay of environmental and economic systems. J. Clean. Prod. 2016, 114, 11–32. [Google Scholar] [CrossRef]
- Alajärvi, L.; Timonen, J.; Lavikainen, P.; Martikainen, J. Attitudes and Considerations towards Pharmaceuticals-Related Environmental Issues among Finnish Population. Sustainability 2021, 13, 12930. [Google Scholar] [CrossRef]
- De Jesus, A.; Mendonça, S. Lost in transition? Drivers and barriers in the eco-innovation road to the circular economy. Ecol. Econ. 2018, 145, 75–89. [Google Scholar] [CrossRef] [Green Version]
- Kirchherr, J.; Piscicelli, L.; Bour, R.; Kostense-Smit, E.; Muller, J.; Huibrechtse-Truijens, A.; Hekkert, M. Barriers to the circular economy: Evidence from the European Union (EU). Ecol. Econ. 2018, 150, 264–272. [Google Scholar] [CrossRef] [Green Version]
- Salmenperä, H.; Pitkänen, K.; Kautto, P.; Saikku, L. Critical factors for enhancing the circular economy in waste management. J. Clean. Prod. 2021, 280, 124339. [Google Scholar] [CrossRef]
- Tura, N.; Hanski, J.; Ahola, T.; Ståhle, M.; Piiparinen, S.; Valkokari, P. Unlocking circular business: A framework of barriers and drivers. J. Clean. Prod. 2019, 212, 90–98. [Google Scholar] [CrossRef]
- Kirchherr, J.; Reike, D.; Hekkert, M. Conceptualizing the circular economy: An analysis of 114 definitions. Resour. Conserv. Recycl. 2017, 127, 221–232. [Google Scholar] [CrossRef]
- Korhonen, J.; Honkasalo, A.; Seppälä, J. Circular economy: The concept and its limitations. Ecol. Econ. 2018, 143, 37–46. [Google Scholar] [CrossRef]
- Geissdoerfer, M.; Savaget, P.; Bocken, N.M.; Hultink, E.J. The Circular Economy–A new sustainability paradigm? J. Clean. Prod. 2017, 143, 757–768. [Google Scholar] [CrossRef] [Green Version]
- MacArthur, E. Towards a circular economy: Business rationale for an accelerated transition. Accessed Oct. 2015, 25, 2016. [Google Scholar]
- Antonopoulos, I.; Faraca, G.; Tonini, D. Recycling of post-consumer plastic packaging waste in the EU: Recovery rates, material flows, and barriers. Waste Manag. 2021, 126, 694–705. [Google Scholar] [CrossRef] [PubMed]
- Eriksen, M.K.; Christiansen, J.; Daugaard, A.E.; Astrup, T.F. Closing the loop for PET, PE and PP waste from households: Influence of material properties and product design for plastic recycling. Waste Manag. 2019, 96, 75–85. [Google Scholar] [CrossRef] [PubMed]
- Silvenius, F.; Grönman, K.; Katajajuuri, J.M.; Soukka, R.; Koivupuro, H.K.; Virtanen, Y. The role of household food waste in comparing environmental impacts of packaging alternatives. Packag. Technol. Sci. 2014, 27, 277–292. [Google Scholar] [CrossRef]
- Verghese, K.; Lewis, H.; Lockrey, S.; Williams, H. Packaging’s role in minimizing food loss and waste across the supply chain. Packag. Technol. Sci. 2015, 28, 603–620. [Google Scholar] [CrossRef]
- Shukla, S.; Halli, P.; Khalid, M.K.; Lundström, M. Waste Pharmaceutical Blister Packages as a Source of Secondary Aluminum. JOM 2022, 74, 612–621. [Google Scholar] [CrossRef]
- HPRC. Advanced Recycling Pilot Project White Paper—24 January 2022; The Healthcare Plastics Recycling Council (HPRC): St. Paul, MN, USA, 2022; p. 16. [Google Scholar]
- Ernstoff, A.; Niero, M.; Muncke, J.; Trier, X.; Rosenbaum, R.K.; Hauschild, M.; Fantke, P. Challenges of including human exposure to chemicals in food packaging as a new exposure pathway in life cycle impact assessment. Int. J. Life Cycle Assess. 2019, 24, 543–552. [Google Scholar] [CrossRef] [Green Version]
- Lorenzini, G.C.; Mostaghel, R.; Hellström, D. Drivers of pharmaceutical packaging innovation: A customer-supplier relationship case study. J. Bus. Res. 2018, 88, 363–370. [Google Scholar] [CrossRef]
- Pareek, V.; Khunteta, A. Pharmaceutical packaging: Current trends and future. Int J. Pharm. Pharm. Sci. 2014, 6, 480–485. [Google Scholar]
- Kaplinsky, R. Globalisation and unequalisation: What can be learned from value chain analysis? J. Dev. Stud. 2000, 37, 117–146. [Google Scholar] [CrossRef]
- Holopainen, E. Lääkevalmisteiden Pakkausmateriaalivirrat: Kierrätys ja Kierrätyksen Haasteet. Master’s Thesis, Helsingin Yliopisto, Helsinki, Finland, 2021, unpublished. (In Finnish). [Google Scholar]
- Karayılan, S.; Yılmaz, Ö.; Uysal, Ç.; Naneci, S. Prospective evaluation of circular economy practices within plastic packaging value chain through optimization of life cycle impacts and circularity. Resour. Conserv. Recycl. 2021, 173, 105691. [Google Scholar] [CrossRef]
- Meherishi, L.; Narayana, S.A.; Ranjani, K. Sustainable packaging for supply chain management in the circular economy: A review. J. Clean. Prod. 2019, 237, 117582. [Google Scholar] [CrossRef]
- Sohrabpour, V.; Oghazi, P.; Olsson, A. An improved supplier driven packaging design and development method for supply chain efficiency. Packag. Technol. Sci. 2016, 29, 161–173. [Google Scholar] [CrossRef] [Green Version]
- Yin, R.K. Case Study Research: Design and Methods; Sage: New York, NY, USA, 2009; Volume 5. [Google Scholar]
- Honkanen, O. Lääkepakkauksia Koskeva Lainsäädäntö; unpublished presentation 20 January 2021, in Finnish; Fimea: Helsinki, Finland, 2021; p. 15. (In Finnish) [Google Scholar]
- Moretti, F.; van Vliet, L.; Bensing, J.; Deledda, G.; Mazzi, M.; Rimondini, M.; Zimmermann, C.; Fletcher, I. A standardized approach to qualitative content analysis of focus group discussions from different countries. Patient Educ. Couns. 2011, 82, 420–428. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mishra, L. Focus group discussion in qualitative research. Techno Learn 2016, 6, 1. [Google Scholar] [CrossRef]
- Denzin, N.K. The Research Act: A Theoretical Introduction to Sociological Methods; Routledge: London, UK, 2017. [Google Scholar]
- Naderifar, M.; Goli, H.; Ghaljaie, F. Snowball sampling: A purposeful method of sampling in qualitative research. Strides Dev. Med. Educ. 2017, 14, e67670. [Google Scholar] [CrossRef] [Green Version]
- Braun, V.; Clarke, V. Using thematic analysis in psychology. Qual. Res. Psychol. 2006, 3, 77–101. [Google Scholar] [CrossRef] [Green Version]
- Rizos, V.; Behrens, A.; Van der Gaast, W.; Hofman, E.; Ioannou, A.; Kafyeke, T.; Flamos, A.; Rinaldi, R.; Papadelis, S.; Hirschnitz-Garbers, M. Implementation of circular economy business models by small and medium-sized enterprises (SMEs): Barriers and enablers. Sustainability 2016, 8, 1212. [Google Scholar] [CrossRef] [Green Version]
- EDQM. The European Pharmacopoeia: Ensuring the Safety and Good Quality of Medicines (May 2020). Fact Sheet; The European Directorate for the Quality of Medicines and HealthCare (EDQM) & European Commission: Strasbourg, France, 2020; p. 4. [Google Scholar]
- Fimea. European Pharmacopoeia. Available online: https://www.fimea.fi/web/en/supervision/legislation/european_pharmacopoeia (accessed on 12 January 2021).
- de Oliveira, D.P.; Costa, J.S.R.; Oliveira-Nascimento, L. Sustainability of blisters for medicines in tablet form. Sustain. Chem. Pharm. 2021, 21, 100423. [Google Scholar] [CrossRef]
- OECD. Extended Producer Responsibility: Updated Guidance for Efficient Waste Management; OECD Publishing: Paris, France, 2016. [Google Scholar]
- Fimea Kotimaiset Lääketehtaat (Domestic Pharmaceutical Companies). Available online: www.fimea.fi/valvonta/laaketehtaat_ja_-tukkukaupat/toimiluvat/kotimaiset_laaketehtaat (accessed on 24 November 2021).
- STM. Lääkkeiden Potilaskohtaisen Annosjakelun Hyvät Toimintatavat; Sosiaali- ja Terveysministeriö: Helsinki, Finland, 2016; p. 31. [Google Scholar]
- Syrjälä, S. Apteekit Lääkejätteen Kerääjinä. Bachelor’s Thesis, Hamk University of Applied Sciences, Forssa, Finland, 2012; p. 85. (In Finnish). [Google Scholar]
- Geueke, B.; Groh, K.; Muncke, J. Food packaging in the circular economy: Overview of chemical safety aspects for commonly used materials. J. Clean. Prod. 2018, 193, 491–505. [Google Scholar] [CrossRef]
- Gripwall, A.-C. Common Waste Sorting Symbols in Nordic Countries; Nordic Council of Ministers: Copenhagen, Denmark, 2021. [Google Scholar]
- Garcia-Muiña, F.E.; González-Sánchez, R.; Ferrari, A.M.; Volpi, L.; Pini, M.; Siligardi, C.; Settembre-Blundo, D. Identifying the equilibrium point between sustainability goals and circular economy practices in an Industry 4.0 manufacturing context using eco-design. Soc. Sci. 2019, 8, 241. [Google Scholar] [CrossRef] [Green Version]
- Yu, J.; Hills, P.; Welford, R. Extended producer responsibility and eco-design changes: Perspectives from China. Corp. Soc. Responsib. Environ. Manag. 2008, 15, 111–124. [Google Scholar] [CrossRef]
- Micheaux, H.; Aggeri, F. Eco-modulation as a driver for eco-design: A dynamic view of the French collective EPR scheme. J. Clean. Prod. 2021, 289, 125714. [Google Scholar] [CrossRef]
- de Mello Soares, C.T.; Ek, M.; Östmark, E.; Gällstedt, M.; Karlsson, S. Recycling of multi-material multilayer plastic packaging: Current trends and future scenarios. Resour. Conserv. Recycl. 2022, 176, 105905. [Google Scholar] [CrossRef]
- Ragaert, K.; Delva, L.; Van Geem, K. Mechanical and chemical recycling of solid plastic waste. Waste Manag. 2017, 69, 24–58. [Google Scholar] [CrossRef] [PubMed]
- Quicker, P.; Seitz, M.; Vogel, J. Chemical recycling: A critical assessment of potential process approaches. Waste Manag. Res. 2022. [Google Scholar] [CrossRef]
- Johansen, M.R.; Christensen, T.B.; Ramos, T.M.; Syberg, K. A review of the plastic value chain from a circular economy perspective. J. Environ. Manag. 2022, 302, 113975. [Google Scholar] [CrossRef]
| Value Chain Phase | Barriers | Possibilities |
|---|---|---|
| Manufacturing | R: Regulations on pharmaceuticals make changes in packaging difficult and slow. R: Lack of incentives to develop PhPs from environmental perspective. R: National requirements for medicine packaging might be stricter compared to European ones. Need for uniformity. T: Knowledge gap in comparison with different packaging materials and their environmental impacts in the different stages of the value chain. T: Lack of recommendations for recycling instructions on the package and information sheet. T: Need for tertiary packaging to seal the package to increase the safety and decrease the use of falsified medicines. T: Maintaining the cold chain of pharmaceuticals and protecting the medicines from freezing can lead to overpacking. E: Customers do not want to keep large stocks, so the number of deliveries and shipments is increasing. S: Lack of dialog between waste management and packaging developers. | Manufacturers need to be challenged to offer circular packaging solutions. Economic support should be allocated for experiments, and authorities should allow testing and piloting. Authorities should join the dialog on the development of the pharmaceutical packaging. Packaging design guidelines from the perspective of circularity. A new electronic packaging leaflet. Implementation of consistent EU pictogram system for all packaging. Use of monomaterials in plastic packaging enhances circularity. Film-coated tablets could reduce drug residues in the packaging. The efficiency of transport system of pharmaceuticals should be improved, and LCA-based knowledge is needed. Instructions also to manufacturers and wholesale on how packaging should be recycled. |
| Wholesale | E: Amount of obsolete medicine waste from wholesale for disposal means also packaging waste. E: Due to competitive reasons, all packaging sizes cannot be kept on the market. E: Finland is a small market area. T: Keeping up the cold chain of pharmaceuticals and protecting the medicines from freezing can lead to overpacking. | Because of the large volumes of PhPs, wholesale is in a key role when developing possibilities to recycle the packaging. Provide instructions also to manufacturers and wholesale on how packaging should be recycled. The efficiency of the transport system of pharmaceuticals should be improved. |
| Dose distribution | R: Regulations hinder the possibility of dose distribution to order larger packs of medicines abroad. T: Keeping up the cold chain of pharmaceuticals and protecting the medicines from freezing can lead to overpacking. | Dose distribution can be developed to use larger packs, which reduces blister amounts. The efficiency of transport system of pharmaceuticals should be improved. |
| Pharmacy and hospital pharmacy | R: The dose delivery packs are not allowed for hospital use because of marketing authorizations. R, E: Delivery obligation restricts the pack sizes available. T, E: Same size packaging is user-friendly for medicine storage and delivery. S: Lack of dialog between waste management and pharmacies. | Pharmacies could follow the amounts of returned PhP by consumers. |
| Consumer | R, E: Restrictions in reimbursements for medicine expenses have an impact on what package size customer chooses. E: Bigger packages can be cheaper for customers. T: Consumers do not necessarily understand the packaging waste sorting instructions. S: Commitment to medication. The medicines are not necessarily used according to the doctor’s advice which creates unnecessary wastes. | Information campaigns are needed. EU pictograms would ease sorting. Consumer research should be carried out on how much medication is left unused and for what reason. |
| Waste management | T: Lack of information on whether the pre-treatment of plastic packages is enough to provide safe cycles. T: Lack of data on PhP waste volumes. T: Recycling of pharmaceutical packages is hindered by certain materials or packaging types, e.g., PVC or multilayer packaging. S: Lack of dialog between waste management and pharmacies. | Chemical recycling is expected to ease the plastic packaging recycling. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Salmenperä, H.; Kauppi, S.; Dahlbo, H.; Fjäder, P. Increasing the Circularity of Packaging along Pharmaceuticals Value Chain. Sustainability 2022, 14, 4715. https://doi.org/10.3390/su14084715
Salmenperä H, Kauppi S, Dahlbo H, Fjäder P. Increasing the Circularity of Packaging along Pharmaceuticals Value Chain. Sustainability. 2022; 14(8):4715. https://doi.org/10.3390/su14084715
Chicago/Turabian StyleSalmenperä, Hanna, Sari Kauppi, Helena Dahlbo, and Päivi Fjäder. 2022. "Increasing the Circularity of Packaging along Pharmaceuticals Value Chain" Sustainability 14, no. 8: 4715. https://doi.org/10.3390/su14084715
APA StyleSalmenperä, H., Kauppi, S., Dahlbo, H., & Fjäder, P. (2022). Increasing the Circularity of Packaging along Pharmaceuticals Value Chain. Sustainability, 14(8), 4715. https://doi.org/10.3390/su14084715






