4.1. Fish Performance
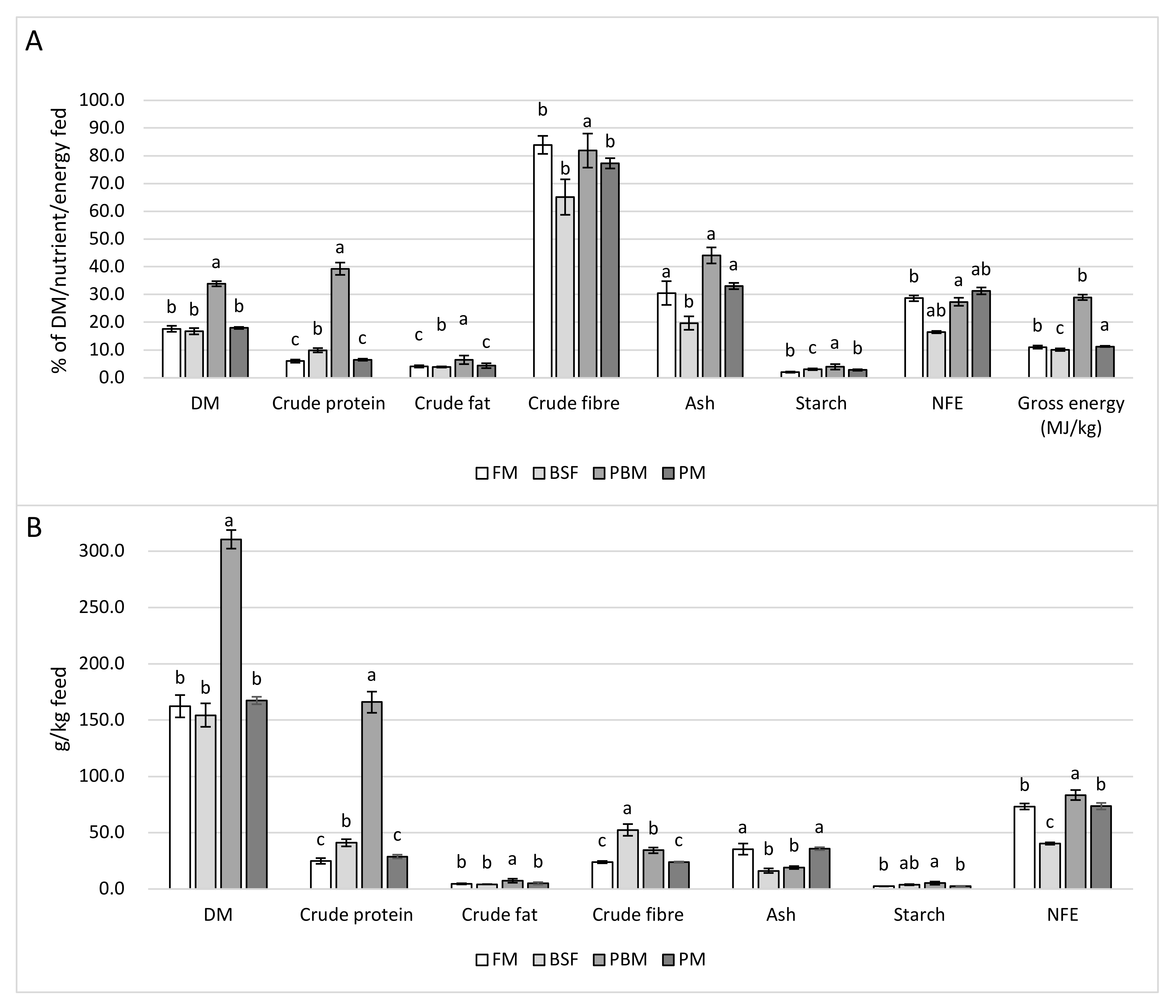
All relevant fish performance indicators, including final body weight and total length, BWG, LG, FCR, SGR, and TGC, paint a clear picture in terms of the suitability of the different protein sources as supporters of growth in Nile tilapia considering the evident rearing conditions and the dietary formulation approach applied.
In a meta-analysis, Galkanda-Arachchige et al. [
17] found that many freshwater fish species can tolerate up to 100% FM replacement and the majority accept > 50% FM replacement with poultry by-product meal without difficulty, which agrees with the findings of this study. Although fish fed the PM diet on average performed slightly less well than fish fed the FM diet with regard to all indicators mentioned above, differences were minor and non-significant. However, PER for fish fed the PM diet was significantly lower compared to fish fed the FM diet, which in combination with the overall slightly reduced growth response suggests yet a slight superiority of FM for growth in Nile tilapia when used as the sole main protein source. Nevertheless, the results of this trial, especially considering the low FCRs observed for the FM and PM diet, indicate the high suitability of PM as a sole protein source and 100% replacement of FM in the diet of juvenile Nile tilapia.
This finding is generally supported by the published literature on PM in Nile tilapia diets with similar results recorded by Yones and Metwalli [
60] and Hernández et al. [
58], who found no significant reduction in growth performance and feed conversion at 100% FM replacement compared to the control diet. El-Sayed [
74] also recorded a similar growth response for Nile tilapia at 100% FM replacement, while Fasakin et al. [
75] successfully replaced 66% of the FM in the control diet without adversely affecting growth performance and feed conversion of hybrid tilapia (
Oreochromis niloticus ×
Oreochromis mossambicus).
PM often has a lower content of the EAA lysine, methionine, and histidine compared to FM, and in some fish species, the reduced growth performance at high PM inclusion levels has been ascribed to this lack [
17]. In this study, although lysine, methionine, and histidine were lower in the PM diet than in the FM diet, they were above published requirements for juvenile Nile tilapia and likely not limiting. However, tryptophan was on the low end compared to the other diets and just at or above requirements, potentially indicating a slight lack [
63,
64]. On the mineral side, no obvious insufficiencies were apparent for the PM diet.
However, in this study, the overall CP content of the diets was generally higher than in previous work on poultry meal in tilapia diets [
58,
60,
74,
75], and the CP content of the PM diet with 43.7% was above the targeted 40% CP and the 40.3% CP of the FM diet. This could have had an effect on the growth performance of the fish fed the PM diet. Alongside differing protein digestibility, the slightly higher CP in the PM diet could have perhaps contributed to the higher recorded CP as well as EAA/NEAA content in the PM feces. Nevertheless, although a diet with such a high inclusion of expensive FM is very unlikely in a commercial context, especially for omnivorous Nile tilapia, the PM diet resulted in a 30% lower cost per kg of body weight gain (0.67 €/kg BWG) than the FM diet (0.96 €/kg) and incurred the significantly lowest cost among all of the tested diets.
While resulting in significantly better growth performance as well as feed and protein conversion in comparison to the PBM diet, the BSF diet still produced inferior fish performance compared to the FM and PM diet. Various authors have replaced FM to a certain extent with BSFM in tilapia diets without adversely affecting growth performance.
While Agbohessou et al. [
76] and Tippayadara et al. [
77] found no adverse effects on the performance of tilapia when entirely replacing the FM in their control diets with BSFM, the findings of Taufek et al. [
78], Rana et al. [
79], Muin et al. [
80], and Devic et al. [
53] are in line with this study and show reduced performance above a certain level of FM replacement with 50% replacement or, in the case of [
53], a BSFM inclusion of 80 g/kg diet being optimal. In this study as well, the BSF diet resulted in reduced growth and feed conversion compared to the FM diet. However, all of the above-cited studies have incorporated a fairly substantial amount of soybean meal or soybean products as a secondary protein source in their experimental diets and thus did not incorporate BSFM as the sole protein source as was performed in the present study. Furthermore, these authors opted for a considerably lower inclusion of the BSFM (8–30%) compared to this study (61.6%). This could have reduced potentially detrimental effects of high absolute BSF inclusion and moderated the differences in fish performance between FM control and BSF test diets even when replacing a majority of FM.
Although BSFM is overall regarded as a quality protein source for fish feeds due to its high protein content and relatively balanced AA profile [
24,
81], generally resembling the EAA pattern of FM [
82], the BSF diet in this trial did have lower arginine, lysine, methionine, phenylalanine, threonine, methionine + cysteine levels than the other experimental diets as well as the lowest sum of EAAs. Especially, arginine, methionine, threonine, and methionine + cysteine were merely at or even below published requirements depending on the source [
63,
64], which could be one reason for the suppression of growth performance and feed conversion in comparison to our FM control diet. Depending on the digestibility of P in the BSF diet, also the low P content of 12.1 g/kg in comparison to the FM and PM diet could have affected growth performance. Furthermore, despite Nile tilapia generally being thought to have some advantages in digesting chitin [
83], the elevated chitin levels likely to have resulted from the high BSFM inclusion opted for in this study may have interfered with protein digestibility as suggested by other authors [
79,
84,
85]. It should, however, be noted that the nutrient composition of BSF larvae, especially protein, fatty acid, and mineral composition and to a lesser extent EAA pattern, can be altered through diet modification [
24,
86,
87,
88] as well as harvest timing [
89], which may present avenues for optimizing BSFM for use in tilapia and other aquaculture diets. In terms of cost per kg body weight gain, the BSF diet did not perform well compared to the other diets with 2.21 €/kg, which primarily stresses the importance of further scaling and industrializing BSF larvae production in order to make prices more competitive (however, it should be noted that, due to low volume, the feed manufacturer paid a higher price than would be expected for large industrial volume orders).
Fish fed the PBM diet performed poorly compared to the other diets with strongly depressed feed conversion and growth. This result is consistent with the findings of other studies in which 50% total dietary blood meal inclusion [
90], as well as 50–100% replacement of the FM in the control diets by PBM, resulted in reduced growth performance of tilapia [
74,
91]. However, Aladetohun and Sogbesan [
92] recorded better growth of tilapia at 100% FM replacement by PBM compared to the FM control diet, demonstrating the inconsistent assessment of PBM as a protein source for tilapia [
93].
The inferior growth performance observed in the present study for fish fed the PBM diet has likely to do with the low Ca (5.7 g/kg) and P content (7.8 g/kg) which are both below requirements [
65,
66]. Ca and P are both essential for bone and scale mineralization as well as various other physiological functions [
66,
94]. Ca plays an important role in nerve transmission, muscle contraction, maintenance of cell membrane integrity, and blood clotting. P is critical in, e.g., amino acid, lipid, and carbohydrate metabolism, as well as an integral component of ATP, phospholipids, and nucleic acids. Deficiencies in these two minerals can lead to reduced growth, as observed for the PBM diet in this trial. From the perspective of the EAA content, however, the PBM diet compares most favorably to the other diets, with the sum of EAAs actually being the highest. However, methionine was recorded slightly below the published requirement [
63], and also methionine + cysteine barely matched requirements [
64]. Nevertheless, Ca and P deficiency most probably caused the inferior fish performance, which is supported by the exceedingly high CP, as well as EAA/NEAA content, detected in the recovered feces as well as the low TIN accumulation in the process water, indicating reduced digestibility of otherwise valuable growth-supporting nutrients. On the one hand, these results clearly show the unsuitability of the PBM diet in the current form. However, on the other hand, the comparably favorable AA profile of the PBM diet in combination with the deficiencies in Ca and P perhaps present the opportunity to improve growth performance in such fish meal-free PBM diets for tilapia by increasing Ca and P either through mineral supplementation or strategic supplementation of the PBM with protein ingredients higher in Ca and P such as bone meals or PM.
4.2. Water—Implications for Plant Production
The second objective of this study was to evaluate the potential of alternative protein sources to address some of the nutrient imbalances and deficiencies often encountered in aquaponic waters [
47,
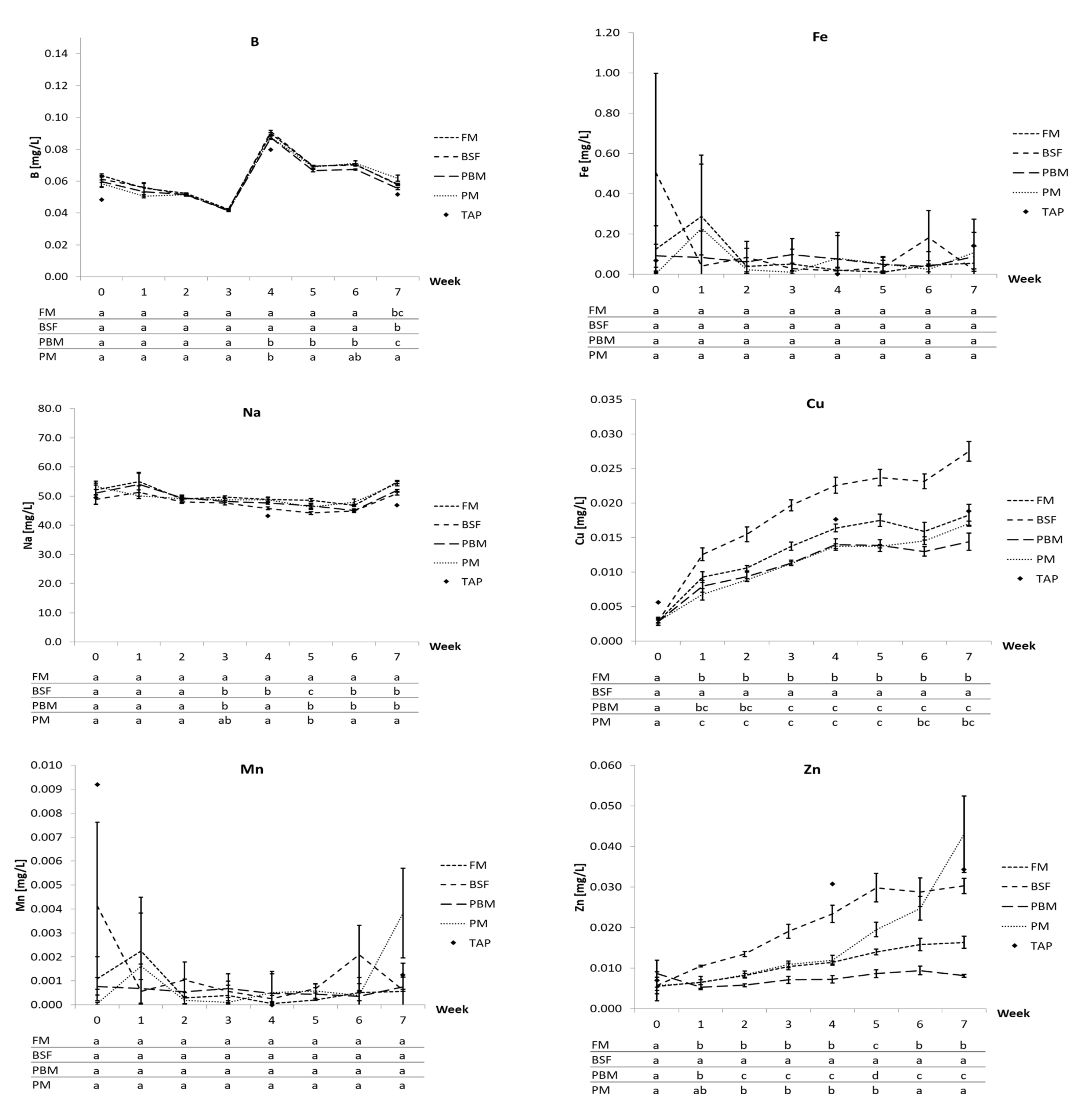
48]. For Ca, S, B, Fe, Na, and Mn, no meaningful differences were found between the experimental diets, and concentrations were closely related to tap water concentrations, i.e., exchange water, mostly corroborating the findings of other authors [
47]. Delaide et al. [
50] found the majority of the dissolved Ca, S, B, and Na originating from the tap water, while Strauch et al. [
95] also found notable tap water influence on Ca, S, and Mg (they did not measure B and Na). These authors both report Fe and Mn predominantly originating from feed input. However, Strauch et al. [
95] only removed the sludge from the system clarifier once a week while feces were removed daily in the present study, and water pH was substantially below what was recorded in this trial. Both may have increased the mineralization and solubilization of Fe, Mn, and other elements compared to the present study. Nevertheless, due to the considerable differences in Ca, S, B, Fe, Na, and Mn content between the experimental diets used in this study, the likelihood of substantially influencing the dissolved concentration of these nutrients by mere ingredient manipulation appears low under the experimental conditions faced (physico-chemical conditions, water exchange rate, feeding rate, fish species, sludge removal rate, etc.).
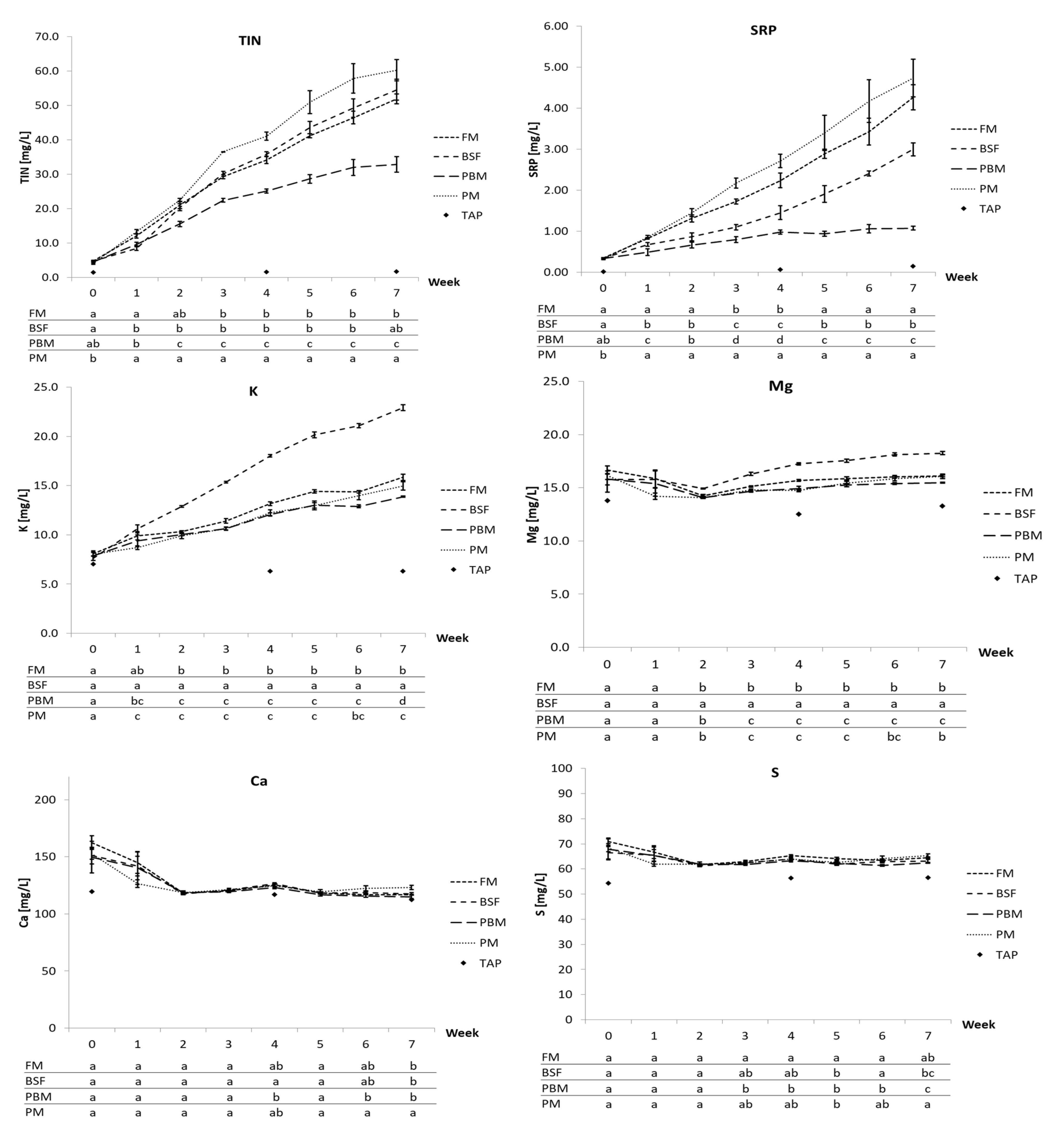
The most pronounced uptrend for all diets was recorded for TIN, SRP, and K, with partly significant differences between diets. As suggested, on the one hand, by the lower levels of P and K in the PBM diet and on the other hand by the poor diet digestibility and substantially higher recovery of CP in the feces (among other nutrients), especially TIN and SRP but also K, even though to a lesser extent, accumulated more slowly and reached lower concentrations. It is likely, however, that by fostering protein digestibility through alleviating the Ca and P deficiency in the PBM diet as discussed in
Section 4.1, branchial nitrogen excretion and thereby process water TIN concentration could be increased. TIN accumulation for the FM and BSF diet were similar, whereas the PM diet resulted in consistently higher TIN concentrations, which may have been a direct consequence of its elevated protein level alluded to in
Section 4.1. This will lead to absolutely more nitrogen added to the system through the feed and probably increased amino acid catabolism, eventually resulting in higher branchial nitrogen excretion [
8,
96]. Differences in the accumulation of SRP reflect the P contents in the diets, illustrating that not only FM and PM are superior SRP suppliers in diets for aquaponics but also highlighting that SRP, a mostly deficient plant nutrient in aquaponics [
47,
49,
50,
97], is influenceable by protein source choice. This agrees with efforts to reduce phosphorus excretion through appropriate feed formulation and ingredient choice, i.e., promoting diets with higher energy density and P digestibility as well as reducing the inclusion of protein sources high in P such as FM and other animal meals [
6,
98,
99,
100]. Consequentially, with the low price and the support of growth in Nile tilapia, PM appears to be a highly suitable protein source for aquaponic feeds that could spare marine ingredients while increasing SRP availability for plants and thereby reduce the need for additional phosphorus fertilization.
K accumulation, while being similar for the FM, PBM, and PM diet, was substantially elevated for the BSF diet, which is in line with its higher K content compared to the other diets. At the end of the trial, the mean K concentration in the process water of the BSF fed systems was 22.9 mg/L, roughly 45% higher than the 15.9 mg/L in the FM treatment, the next highest K concentration among the treatments. This shows that higher dietary K levels may translate well into higher dissolved K concentrations in the process water and that, in this case, the BSFM considerably added to the dietary K level in comparison to the other protein sources. The mineral content of BSF larvae is considered high among animal feed ingredients [
24] and can be subject to considerable variability depending on the mineral composition of the feed substrate [
82,
88,
101,
102] as well as the life stage [
89]. Due to this variability, it should not generally be inferred that BSFM is an appropriate protein source for increasing dissolved K excretion. However, if provided with feed high in K, seaweed being one example [
88], BSF larvae seem capable of increasing dissolved K excretion if incorporated in diets at sufficiently high levels and could thus help to alleviate K deficiencies encountered in aquaponics [
47,
49,
50,
97].
From week 2 (first feed ration increase), Mg concentrations also increased steadily for all diets, although the accumulation rate was lower than for K. While differences in Mg concentration between the FM, PM, and PBM diets were minor, again the BSF diet stood out with a faster increase, leading to mean concentration of 18.3 mg/L Mg in week 7, 13.4% higher compared to the FM diet. This is in line with the higher Mg content measured in the BSF diet versus the other diets. Yet, in contrast to K, Mg concentrations are more governed by tap water concentration as illustrated by the 13.3 mg/L Mg measured in the tap water in week 7, which agrees with Delaide et al. [
50] and Strauch et al. [
95]. However, Strauch et al. [
95] and Seawright et al. [
49] found increased Mg accumulation at higher fish production intensities, supporting the result of the present study that Mg concentration can likely be manipulated through feed amount and composition.
Cu showed an accumulative trend over the entire trial for all diets as well, yet followed tap water concentrations very closely with levels for the most part being at or even below levels measured in the tap water, which agrees with Delaide et al. [
50] and Strauch et al. [
95]. However, the higher Cu content in the BSF diet in contrast to the other diets directly translated into higher Cu concentrations above tap water levels throughout the trial. Zn also showed an upward trajectory for all diets, yet primarily caused by tap water concentrations. However, from week 4, a comparably sharp rise up and above the tap water concentration was recorded for the systems fed the PM diet, which cannot be attributed to the higher Zn content of the diet. This rise coincided with a faster drop in water pH observed in the PM treatment, with the mean pH reaching 6.4 at the end of the trial versus 7.2 (FM), 7.9 (PBM), and 7.4 (BSF), which may have contributed to increased solubility of Zn.
For a long-term trial, it would be very insightful to compare nutrient concentrations and ratios to hydroponic standard solutions. However, due to the short trial duration not allowing for an equilibrium between feed input, water exchange rate, and nutrient accumulation to develop, nutrient concentrations recorded in the present study are considerably lower than in any kind of mature stabilized system (as also confirmed by the still rising concentrations of many of the dissolved nutrients at the end of the trial). In addition, nutrient ratios were not compared to hydroponic standard solutions since accumulation rates and initial concentrations differed between nutrients (e.g., as discussed for K and Mg), confirming the differential accumulation rates also reported by Seawright et al. [
49].
4.3. Feces—Implications for Insect Larvae Production
In a tri-trophic agricultural system that interdependently integrates fish, plant, and insect larvae production [
52], it is important to identify resource inputs that are either beneficial on all three production levels or beneficial on one without compromising the other two. Therefore, this study not only set out to compare fish performance and dissolved nutrient excretion with the objective of aquaponic diets in mind but also aimed to evaluate solid nutrient excretion by the fish with regard to potential nutritional suitability for insect larvae production (e.g., BSF larvae). Higher value insect larvae proteins, lipids, and minerals could then again be recycled back into the fish diets, further closing internal nutrient cycles and thereby minimizing waste and maximizing the use of initial resource inputs.
4.3.1. Feces Composition and Nutrient Recovery
Mineral compositions found for the feces in this study were comparable to the ranges found for Nile tilapia published by other authors who identified the dominant mineral constituents to be Ca (24–66 g/kg DM) > P (13–33 g/kg DM) > S (4.7–6.6 g/kg DM) > Mg (2.2–3.8 g/kg DM) and reported reasonably similar ranges for other minerals as well [
37,
103,
104]. Data published for African catfish sludge also suggests comparable ranges with Ca (27–38 g/kg DM) > P (15–19 g/kg DM > S (5 g/kg DM) > Mg (1.2–2 g/kg DM) [
95,
104]. Assuming a nitrogen-to-CP conversion factor of 6.25, the nitrogen measurements published for Nile tilapia sludge (equating to 22.7–25.6% CP in DM) [
37,
103,
104] and African catfish sludge (equating to 11.1–25.9% CP in DM) [
95,
104] support the measurements made in the present study. With the exception of the PBM diet, showing strongly diminished growth performance and protein digestibility, which resulted in high DM recovery through feces collection, the other three feeds resulted in similar DM recovery, suggesting that the FM, PM, and BSF diet in a production setting would produce a similar raw material output that could then be provided downstream as a feed source for insect larvae production. Quantity and quality of the raw material collected will, however, likely be affected not only by feed composition but also by production-related factors such as uneaten feed, residence time of the feces in the water, the solids removal method, and the subsequent processing of the sludge.
The experimental diets resulted in feces with low CF and starch content. This, in combination with the low percentage recovery of the fed CF and starch through feces collection, indicates high digestibility of both of these nutrients in all four experimental diets. In line with the similar growth performance, FCR, and PER of the fish fed the FM and PM diet, the respective feces reflected a similar nutrient profile regarding all measured gross nutrients; however, they also had the lowest P/E ratio and CP content among all feces types. The low percentage recovery of the fed CP compared to the BSF and PBM diet further corroborates the effective use of the FM and PM diet and suggests higher CP digestibility. However, the weak fish performance achieved by the PBM diet is clearly reflected in the high CP content, GE content, and P/E ratio of the feces. The slightly less superior growth performance achieved by the BSF diet in contrast to the FM and PM diet is also reflected in the higher CP content of the BSF feces as well as the higher percentage recovery of fed CP. The higher ash content, as well as total ash, recovered per kg of feed of the FM and PM feces reflect the higher ash content of the FM and PM diet. On the contrary, the low ash content of the PBM diet resulted in the lowest ash content and recovery of ash per kg of feed, whereas the percentage recovery of the ash fed was highest for the PBM diet. This suggests that despite already limited mineral availability in the diet, biological availability appeared even more constrained, likely due to the previously discussed mineral/nutrient imbalances (Ca, P and methionine).
CFB content was higher in the BSF diet compared to the other diets, while the inclusion of cellulose, lignin, and hemicellulose from wheat bran and corn meal was lowest, indicating that a substantial amount of the CFB measured originated from the chitin in the BSFM. The high CFB content of the BSF diet was also reflected in the CFB content and CFB recovery per kg of feed of the BSF feces, being highest among dietary treatments. However, recovery of CFB as a percentage of CFB fed was, in fact, significantly lower compared to the other feces’ types. This suggests the higher digestibility of the chitin portion of the CFB in Nile tilapia compared to the cellulose/lignin/hemicellulose portion, corroborating the findings of other authors with regard to the ability of Nile tilapia to digest chitin [
83].
In line with the weak fish performance and the high feces CP content, AA levels found in the PBM feces were above those found in the BSF, FM, and PM feces. While total AA content of the PBM feces was equal to its CP content, total AA content for the FM, BSF, and PM feces was only 65%, 45%, and 83%, respectively, indicating that a substantial portion of the CP measured in the latter three feces types may actually have originated from non-protein/non-AA nitrogen. This not only corroborates the poor protein and AA digestibility of the PBM diet but furthermore highlights the insufficiency of mere CP analysis for judging protein availability in fish feces and reveals the high variability in the ratio of CP to AAs that has to be expected.
4.3.2. Feces as Feed for Insect Larvae
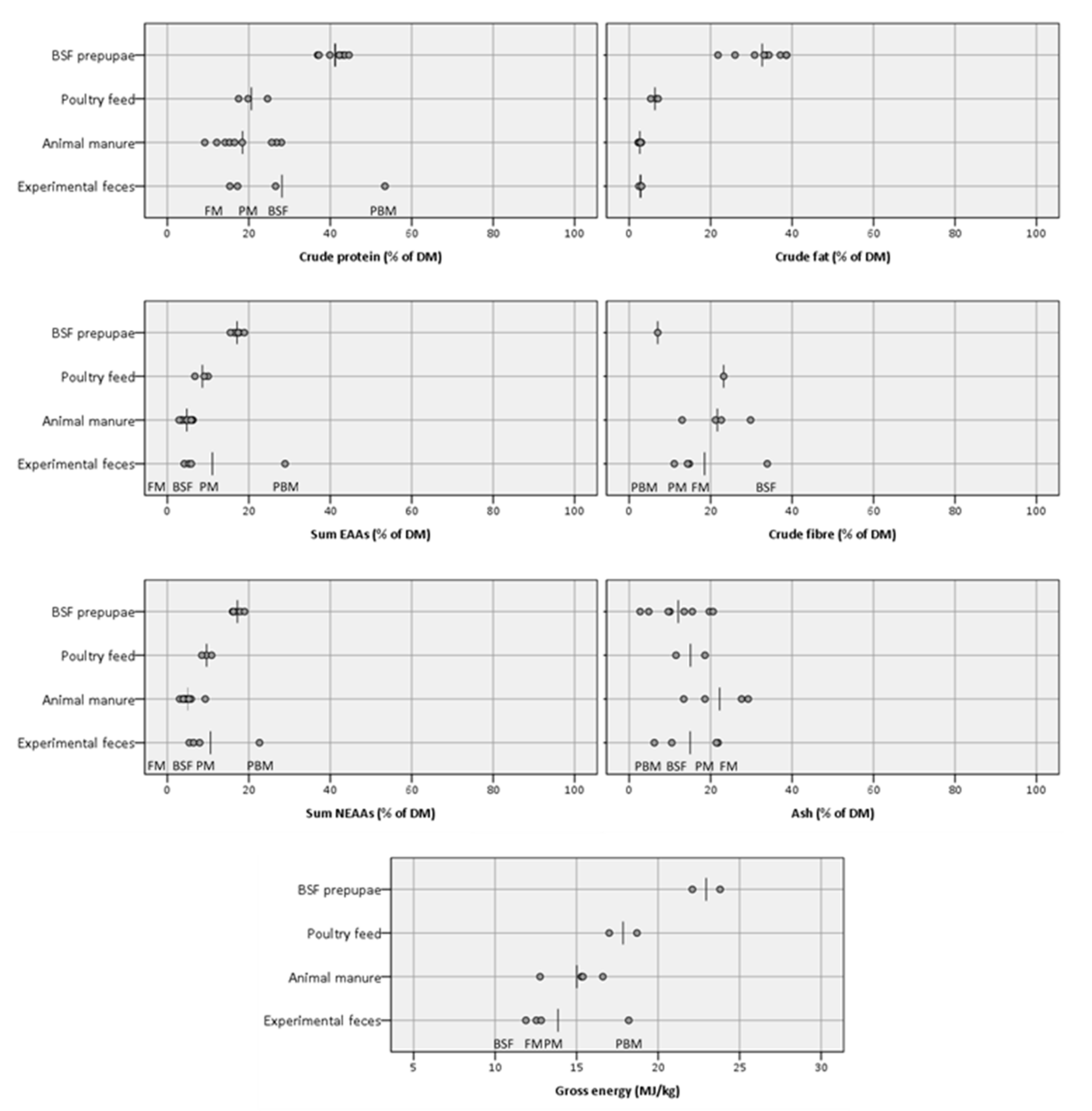
With the above details on feces composition and recovery in mind, important macronutrients were compared between the experimental feces and other relevant raw materials (
Figure 5), including animal manures (often suggested as waste streams to be used for BSF larvae production [
23,
105,
106,
107,
108]), poultry feed (a common reference diet in BSF trials [
109,
110,
111]) as well as BSF larvae themselves. Disregarding the PBM feces, the other experimental feces appear approximately similar to other animal manures in terms of CP, CF as well as EAA, and ash content (except for the BSF feces with lower ash content) with perhaps slightly higher content of NEAAs, yet somewhat reduced GE content. The BSF feces shows a higher CFB content compared to animal manures, whereas the FM and PM show a lower CFB content. CP and ash content in poultry feed are in a similar range to what was found in the experimental feces, whereas the levels of CF and EAA/NEAA were somewhat higher, and GE was considerably higher in poultry feed. BSF larvae show substantially higher levels of CP, CF, EAAs/NEAAs, and GE (again excl. PBM), yet a more similar ash and a lower CFB content in comparison with the experimental feces. However, it should be noted that BSF larvae show a comparably wide range of ash and CF content, underpinning the tendency to more strongly reflect dietary characteristics in their body composition with respect to mineral and CF versus, e.g., CP or AA content [
82,
88,
89,
101,
102,
105].
Since protein content and protein quality of feed substrates, i.e., EAA composition, are likely to be of major importance for BSF larvae growth and development [
105,
112], EAA composition as well as EAA profile of the experimental feces were again contrasted with animal manures, poultry feed, and BSF larvae in order to better evaluate the protein quality of the feces with respect to their suitability to support insect larvae production. The whole-body amino acid composition of animals can be used as a proxy to determine their nutritional needs and to formulate adequate artificial diets [
105,
113,
114]. Therefore, the percentage deviation in EAA content of the experimental feces, animal manures, and poultry feeds from whole body BSF larvae were calculated (
Table 7). Apart from the PBM feces, which had much higher levels for all EAAs than BSF larvae, these results show that the other feces types, including animal manures, on average only reach 26–34% of the levels found in BSF larvae (FM: 26%; BSF: 31%; PM: 34%; animal manures: 31%), whereas the poultry feeds on average reach 48%, indicating its likely superiority as BSF larvae feed substrate with respect to absolute amounts of EAAs.
However, it is known in animal nutrition that not only the quantity of EAAs plays a role in protein quality but also the balance, i.e., ratio, of EAAs [
114,
115,
116,
117]. The ideal protein concept aims to determine the optimal ratios of EAAs in relation to lysine, often the first limiting EAA in animal feedstuffs [
117], by identifying the whole body EAAs ratios with respect to the lysine of the animal in question. If dietary lysine requirement is experimentally determined, then also the requirements of all other EAAs can be specified. Accordingly, EAA ratios relative to lysine were compared between BSF larvae, which according to the ideal protein concept, represent the ideal balance of EAAs for BSF larvae nutrition, and the experimental feces as well as the other feed substrates discussed above (
Table 7). The FM, BSF, and PM feces are on average similarly unbalanced in terms of their amino acid profile with an average absolute deviation of the EAAs ratios from the ratios found in BSF larvae of 66%, 58%, and 65%, respectively. Other animal manures on average deviate by 78% from the ideal ratios, although the standard deviation of this means with 119% much higher primarily due to exceedingly elevated arginine and tryptophan ratios, meaning these two AAs tend to be overrepresented in animal manures relative to lysine. As would be expected, the EAA profile of poultry feed, well-suited BSF larvae feed, is more balanced and closer to the ideal ratios found in BSF larvae with a mean deviation of only 34%. Although for most EAAs an underrepresentation was recorded in relation to lysine in the PBM feces, it had the on-average most balanced EAA profile with a mean absolute deviation of 27%. Nevertheless, the PBM feces is still more abundant in lysine compared to BSF larvae.
With diet quality, diet digestibility, and subsequently fish performance being superior for the BSF and especially the FM and PM diets and allegedly far closer to optimal than the PBM diet, it can be assumed, considering the specific life stage of Nile tilapia and the diet formulation strategy followed in this trial, that the nutritional quality of the FM, PM and BSF feces is what could more likely be expected in a production scenario versus the PBM diet. Overall, the nutritional quality of these fish feces (FM, PM, BSF) appears similar to terrestrial animal manures, whereas both of these raw material types seem similarly inferior to poultry feed, suggesting reduced suitability for BSF larvae production.