Spatial Solutions and Their Impacts When Reshuffling Coastal Management Priorities in Small Islands with Limited Diversification Opportunities
Abstract
1. Introduction
2. Materials and Methods
2.1. Pearl Oyster Farming in French Polynesia
2.2. Specificities of the Gambier
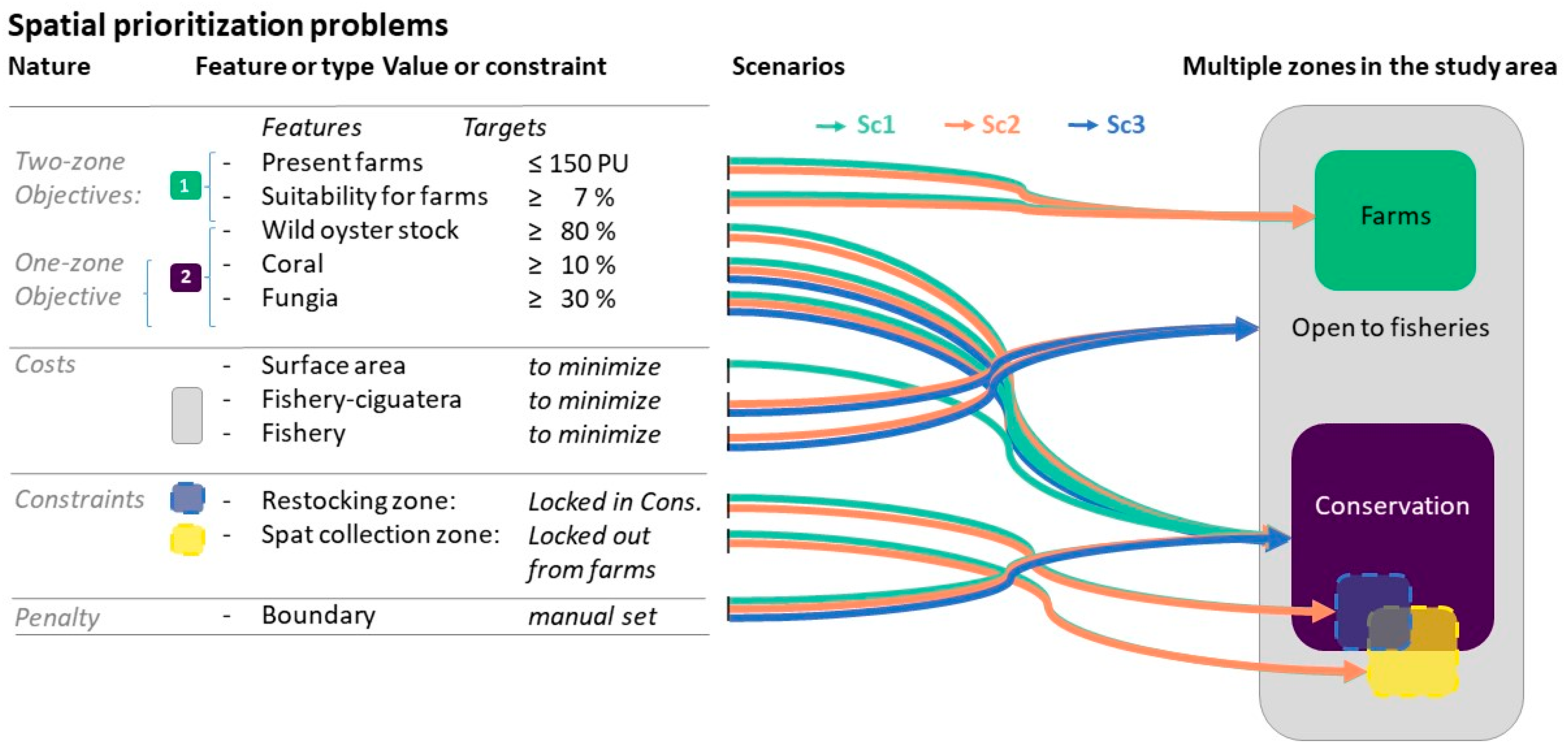
2.3. Diversification Objectives and Study Design
2.4. Collection of Spatial Data and Aggregation by Planning Unit
- Layer 1: A habitat map of high thematic resolution with 77 geomorphologic and benthic classes, from resampled QuickBird and IKONOS imagery at 4 m. resolution spatial imagery. This product is an original work made for this study following recent benthic surveys performed in 2019–2020. It was built following the Millennium Coral Reef Mapping Project principles [39,40], with a hierarchical classification scheme describing the first three levels of detailed geomorphological features, followed by, when suitable for our scenario, benthic information, in particular the type of coral communities. This Gambier habitat map included 7 classes of coarse geomorphological description at level 1 and up to 77 classes at level 5, the finest level of description mixing fine geomorphology and benthic coral information;
- Layer 2: A refined map of fishing grounds established from a map-based questionnaire survey completed by 42 artisanal fishers conducted in November 2019. The sampling strategy was conducted according to [41], and key informants were identified among the most active fishers, recommended by other fishers and the town hall staff, or randomly met all around the main island in order to represent the inhabited area distribution. As a conservative estimation of the representativity of the sampling, the 42 interviews among the 377 households thus represented at 11% sampling, based on the general pattern of one main active fisher/fishery activity per household, or less, in the case of Gambier pearl farming oriented context. Each fishing ground was characterized by an annual level of catch. Using GIS tools, each fishing ground delimitation was then refined according to the limits of the geomorphological features that were compatible with a given fishing gear, following the methodology detailed in [41] (for example, all speargun fishing grounds were redrawn more precisely by eliminating soft-bottom and areas > 30 m deep);
- Layer 3: A ciguatera risk map established from local knowledge on poisoning events and areas regarded at risk of ciguatera. This information was collected during the same survey as the fishery survey. This also followed the methods described in [41] by giving an increasing risk coefficient to the zones identified by fishers, from areas simply avoided by precaution (coefficient = 1) to areas with proven risk, i.e., reported in actual ciguatera poisoning cases, with areas involved in the most recent poisoning events being assigned the highest coefficient (i.e., 10). Another series of ciguatera data derived from a fish toxicity survey on fish samples conducted in 2017 in various Gambier fishing areas by Institut Louis Malardé (HT. Darius, unpublished data);
- Layer 4: a wild oyster stock abundance map, achieved from a density survey conducted between 0 and 30 m depth in 64 stations monitored by SCUBA in November 2019 and February 2020 (Andréfouët, Liao, unpublished data), then generalized to the whole lagoon according to the distribution of densities per type of mapped habitats (from Layer 1) and depth (as in [42]);
- Layer 5: a map of the area reserved for spat collection by legal decree, from the DRM GIS database;
- Layer 6: a map of the existing concessions authorized for pearl oyster farming as of 2019, from the DRM GIS database;
- Layer 7: a map of the boundaries of the four traditionally managed sectors, provided by DRM.
- Layer 9: A combined ‘fishery–ciguatera’ index map, used as a cost layer. This layer was based on the fishery catch map (Layer 8) modulated by the ciguatera risk map as in André et al. [41]. In short, for a given level of catch found in a given PU, its fishery opportunity cost was lowered proportionally to the increased risk of ciguatera for that same PU. Amount of catch and coefficient of ciguatera risk per PU were first normalized over the domain and then combined following the method developed in [41], with a relative coefficient weight for ciguatera of a = 0.25;
- Layer 10: Oyster-restocking zones were identified within the four traditionally managed (rāhui) sectors. The areas for oyster restocking were constrained to (i) remain below 10 % of the surface area of each sector (Layer 7), (ii) include habitat typically favorable for oysters (i.e., hard-bottom geomorphological unit, according to the habitat map (Layer 1) at geomorphological resolution level 2), and (iii) avoid existing farm concessions (Layer 6). The restocking areas result from a simple layer superposition, eventually representing 160 to 760 ha per traditional zone;
- Layer 11: A farm suitability index aiming at guiding the selection of optimal places to relocate farming concessions taking into account the farming history. For each PU (i), the farm suitability index (S) was defined as the surface area of geomorphological features suitable for oyster concessions (Fs) (i.e., all features classes except shallow barrier reef exposed to oligotrophic oceanic water and shallow fringing reef, see class list in Supplementary Materials). Since existing concessions are easy to access (a short distance from shore) and benefit from already built infrastructures, the surface area of existing concessions (Es) was also considered, following Equation (1):
2.5. Detailed Description of Prioritization Scenarios
- Optimal farm concession zone in suitable environments (using Layer 11), with a maximum representation target set at 1500 ha, as per DRM specifications;
- Conservation zone, including at least 30% of the surface area of remarkable coral communities, namely the Fungia spp. community and at least 10% of the surface area of the remaining live coral assemblage (resolution level 5 from Layer 1). Similar habitat representation objectives are recommended to protect biodiversity (e.g., in the Aichi Target 11) and sustain fisheries. Depending on the scenarios, the conservation zone can also include wild oyster stock areas, protecting at least 80% of the habitat where wild stocks occur (Layer 4);
- Oyster restocking zones. Using Layer 10, the four predefined restocking areas are locked in the overall conservation zone. In the optimization process, they are also allowed to overlap potential Fungia and other live coral features, and wild oyster stock as well, and incidentally contribute to the conservation targets;
- Spat collection zone, already delimited by decree. It is considered open to fishers. We do not aim to completely redesign its boundary here, but small changes in its boundaries are allowed if it contributes to the general optimization. In particular, identification of conservation areas is authorized within that zone, and if so, these zones become closed to fisheries. The spat collection zone was locked out from the farm concession zone as these uses are mutually exclusive;
- Fishery zone, which is the remaining available area. It is not defined here by a precise target to reach (which could be possible), but the different farming or conservation zones above are computed to minimize the loss of ciguatera-free zones for fishers (using Layer 9) and minimize the loss of productive fishing grounds (using Layer 8) that would be closed if included in other zones.
2.6. Comparison of the Solutions
- The surface area of each solution zone;
- The value of each solution zone in terms of the fishery–ciguatera index value (overlapping the solution with Layer 9);
- The value of each solution zone in terms of fishery catch value (Layer 8);
- The type of solution zones and their surface areas that overlap the zone initially dedicated to concessions as in BAU (Layer 6); and
- The type of solution zones and their surface areas that overlap the Fungia and coral communities (from Layer 1).
2.7. Sensitivity Analyses
- The use of either the fishery catch or the fishery–ciguatera index layers as input for Scenarios 2 and 3;
- The boundary penalty applied to the conservation zone. For the latter, an optimal boundary penalty value was sought for each scenario following Ardron et al. [43], based on iterations with a factor-10 increased penalty value. For each value, when plotting the total final cost of the solution against the sum of the perimeters of the solution zones, the optimal boundary penalty value was the point that defined the inflection point of the curve.
3. Results
3.1. Spatial Data Collection and Aggregation
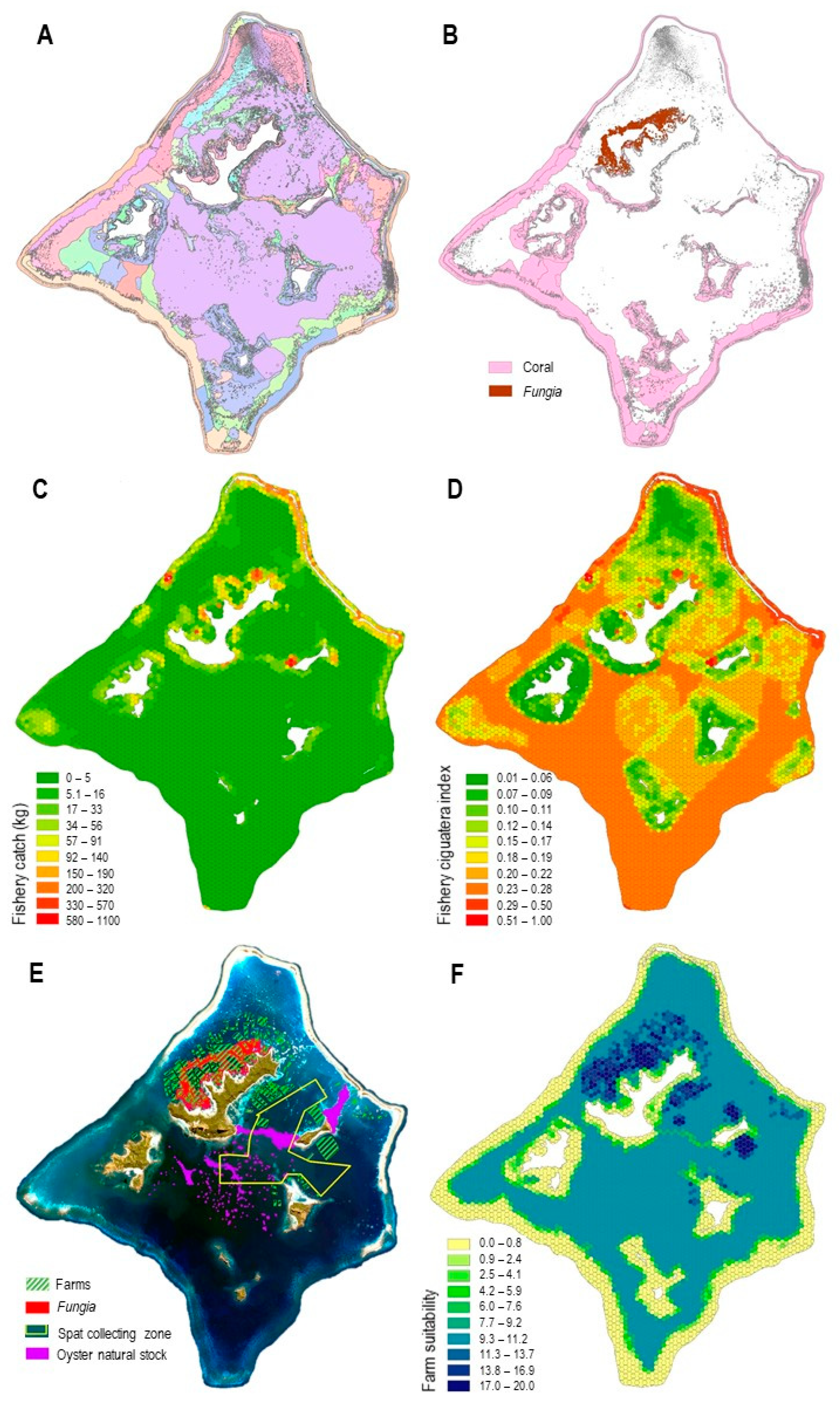
- A high diversity of habitats (Figure 3A) distributed in the overall 48,560 ha lagoon, including (i) 5920 ha of coral-dominated benthos, preferentially found on fringing reef, patch reef, and lagoon floor geomorphological units, and (ii) 747 ha of remarkable Fungia spp. communities, which were distributed on the northwest of the main island Mangareva (Figure 3B);
- An extensive fishery activity, estimated at 63.5 t. annual catch based on the 42 fishers surveys, who identified 252 overlapping individual fishing grounds covering 33,845 ha, which corresponds to 70% of the lagoon surface area, a spatially representative depiction of the fishery in the lagoon. A total of 128 individual zones of moderate to a high level of ciguatera risk were identified based on the fishers’ knowledge, completed with five locations of significant risk of ciguatera, identified following the fish toxicity survey conducted by ILM. The resulting risk map, combined with the fishery catch map, reflected the most favorable areas for fishing activity at a lower risk of ciguatera (Figure 3C,D). These favorable areas for fishing were mainly located on the north coast of Mangareva, the west end of Aukena, in a specific location in the middle of the northwest barrier reef near a small pass and, to a lesser extent, all along the northeast barrier reef.
- A well-established black pearl oyster-farming activity shown by the BAU situation, with 2253 ha of maritime concessions, mainly on the northwest of Mangareva Island and, to a lesser extent, in the south and near Aukena Island (Figure 3E and Figure 4A). Concessions overlapped 358 ha of Fungia spp. communities (48%) (Figure 3E). The lagoon also has 780 ha of abundant wild oyster stock, which is mainly distributed on the shallow terraces between Mangareva and Aukena Islands, between Aukena and the eastern barrier reef, and on the summit of deep patch reefs in the center of the lagoon. The collecting zone overlapped some of these oyster stock habitats (Figure 3E). The farm suitability index highlighted the suitable farming areas (Figure 3F).
3.2. Scenarios Solutions
3.3. Comparison of the Respective Impacts of Solution Options
3.4. Sensitivity Analyses
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Cinner, J.E.; McClanahan, T.R.; Graham, N.A.J.; Daw, T.M.; Maina, J.; Stead, S.M.; Wamukota, A.; Brown, K.; Bodin, Ö. Vulnerability of Coastal Communities to Key Impacts of Climate Change on Coral Reef Fisheries. Glob. Environ. Chang. 2012, 22, 12–20. [Google Scholar] [CrossRef]
- Lau, J.D.; Cinner, J.E.; Fabinyi, M.; Gurney, G.G.; Hicks, C.C. Access to Marine Ecosystem Services: Examining Entanglement and Legitimacy in Customary Institutions. World Dev. 2020, 126, 104730. [Google Scholar] [CrossRef]
- Jupiter, S.; Mangubhai, S.; Kingsford, R.T. Conservation of Biodiversity in the Pacific Islands of Oceania: Challenges and Opportunities. Pac. Conserv. Biol. 2014, 20, 206–220. [Google Scholar] [CrossRef]
- He, Q.; Silliman, B.R. Climate Change, Human Impacts, and Coastal Ecosystems in the Anthropocene. Curr. Biol. 2019, 29, R1021–R1035. [Google Scholar] [CrossRef]
- Payri, C.E.; Vidal, E. Biodiversity, a Pressing Need for Action in Oceania; Payri, C.E., Vidal, E., Eds.; Presses Universitaires de la Nouvelle-Calédonie (PUNC): Nouméa, New Caldonia, 2019. [Google Scholar]
- Birch, T.; Reyes, E. Forty Years of Coastal Zone Management (1975–2014): Evolving Theory, Policy and Practice as Reflected in Scientific Research Publications. Ocean Coast. Manag. 2018, 153, 1–11. [Google Scholar] [CrossRef]
- Campbell, S.J.; Jakub, R.; Valdivia, A.; Setiawan, H.; Setiawan, A.; Cox, C.; Kiyo, A.; Darman; Djafar, L.F.; de la Rosa, E.; et al. Immediate Impact of COVID-19 across Tropical Small-Scale Fishing Communities. Ocean Coast. Manag. 2021, 200, 105485. [Google Scholar] [CrossRef]
- Fenichel, E.P.; Levin, S.A.; McCay, B.; St. Martin, K.; Abbott, J.K.; Pinsky, M.L. Wealth Reallocation and Sustainability under Climate Change. Nat. Clim. Chang. 2016, 6, 237–244. [Google Scholar] [CrossRef]
- Barrett, C.B.; Bezuneh, M.; Clay, D.C.; Reardon, T. Heterogeneous Constraints, Incentives and Income Diversification Strategies in Rural Africa; Working Paper; Cornell University: Ithaca, NY, USA, 2001; p. 46. Available online: https://ageconsearch.umn.edu/record/14761 (accessed on 22 February 2022).
- Bowser, W.; Nelson, C.H. Land Institutions, Investments, and Income Diversification: Pathways to Economic Development for Brazil’s Quilombo Communities; IFPRI Discussion Paper 1179; International Food Policy Research Institute (IFPRI): Washington, DC, USA, 2012; Available online: http://ebrary.ifpri.org/cdm/ref/collection/p15738coll2/id/126905 (accessed on 22 February 2022).
- Kasperski, S.; Holland, D.S. Income Diversification and Risk for Fishermen. Proc. Natl. Acad. Sci. USA 2013, 110, 2076–2081. [Google Scholar] [CrossRef]
- Andréfouët, S.; Dewantama, I.M.I.; Ampou, E.E. Seaweed Farming Collapse and Fast Changing Socio-Ecosystems Exacerbated by Tourism and Natural Hazards in Indonesia: A View from Space and from the Households of Nusa Lembongan Island. Ocean Coast. Manag. 2021, 207, 1–8. [Google Scholar] [CrossRef]
- Bell, J.D.; Adams, T.J.; Johnson, J.E.; Hobday, A.J.; Gupta, A. Sen. Chap. 1. Pacific Communities, Fisheries, Aquaculture and Climate Change: An Introduction. In Vulnerability of Tropical Pacific Fisheries and Aquaculture to Climate Change; Bell, J.D., Johnson, J.E., Hobday, A.J., Eds.; Secretariat of the Pacific Community: Noumea, New Caledonia, 2011; pp. 1–48. [Google Scholar]
- UN—United Nations General Assembly. Transforming Our World: The 2030 Agenda for Sustainable Development; Cambridge University Press: Cambridge, UK, 2015; p. 35. [Google Scholar] [CrossRef]
- André, L.V.; Van Wynsberge, S.; Chinain, M.; Andréfouët, S. An Appraisal of Systematic Conservation Planning for Pacific Ocean Tropical Islands Coastal Environments. Mar. Pollut. Bull. 2021, 165, 20. [Google Scholar] [CrossRef]
- Chinain, M.; Gatti, C.M.I.; Darius, H.T.; Quod, J.P.; Tester, P.A. Ciguatera Poisoning: A Global Review of Occurrences and Trends. Harmful Algae 2020, 22. [Google Scholar] [CrossRef]
- Ehler, C.N.; Douvere, F. Marine Spatial Planning, A Step-by-Step Approach toward Ecosystem-Based Management; Intergovernmental Oceanographic Commission and Man and the Biosphere Programme. iOC Manual and Guides no. 53, iCaM Dossier no. 6; UneSCO: Paris, France, 2009; (In English). [Google Scholar] [CrossRef]
- Margules, C.R.; Pressey, R.L. Systematic Conservation Planning. Nature 2000, 405, 243–253. [Google Scholar] [CrossRef] [PubMed]
- Ball, I.R.; Possingham, H.P.; Watts, M.E. Marxan and Relatives: Software for Spatial Conservation Prioritization. In Spatial Conservation Prioritization. Quantitative Methods & Computational Tools; Moilanen, A., Wilson, K.A., Possingham, H.P., Eds.; Oxford university Press: Oxford, UK, 2009; pp. 185–195. [Google Scholar]
- Watts, M.E.; Ball, I.R.; Stewart, R.S.; Klein, C.J.; Wilson, K.; Steinback, C.; Lourival, R.; Kircher, L.; Possingham, H.P. Marxan with Zones: Software for Optimal Conservation Based Land- and Sea-Use Zoning. Environ. Model. Softw. 2009, 24, 1513–1521. [Google Scholar] [CrossRef]
- Moilanen, A.; Franco, A.M.A.; Early, R.I.; Fox, R.; Wintle, B.; Thomas, C.D. Prioritizing Multiple-Use Landscapes for Conservation: Methods for Large Multi-Species Planning Problems. Proc. R. Soc. B Biol. Sci. 2005, 272, 1885–1891. [Google Scholar] [CrossRef] [PubMed]
- Pressey, R.L.; Watts, M.E.; Barrett, T.W.; Ridges, M.J. The C-Plan Conservation Planning System: Origins, Applications, and Possible Futures. In Spatial Conservation Prioritization; Moilanen, A., Wilson, K.A., Possingham, H.P., Eds.; Oxford Biology: Oxford, UK, 2009; pp. 211–234. [Google Scholar]
- Hanson, J.O.; Schuster, R.; Morrell, N.; Strimas-Mackey, M.; Watts, M.E.; Arcese, P.; Bennett, J.; Possingham, H.P. Prioritizr: Systematic Conservation Prioritization in R. R Package Version 7.0.1. 2021. Available online: https://CRAN.R-project.org/package=prioritizr (accessed on 31 August 2021).
- Magris, R.A.; Pressey, R.L.; Mills, M.; Vila-nova, D.A.; Floeter, S. Integrated Conservation Planning for Coral Reefs: Designing Conservation Zones for Multiple Conservation Objectives in Spatial Prioritisation. Glob. Ecol. Conserv. 2017, 11, 53–68. [Google Scholar] [CrossRef]
- Venier, C.; Menegon, S.; Possingham, H.P.; Gissi, E.; Zanella, A.; Depellegrin, D.; Sarretta, A.; Barbanti, A.; McGowan, J. Multi-Objective Zoning for Aquaculture and Biodiversity. Sci. Total Environ. 2021, 785, 146997. [Google Scholar] [CrossRef] [PubMed]
- Delavenne, J. Conservation of Marine Habitats under Multiple Human Uses: Methods, Objectives and Constraints to Optimize a Marine Protected Areas Network in the Eastern English Channel; Université du Littoral Côte d’Opale: Dunkerque, France, 2012. [Google Scholar]
- Krueck, N.C.; Ahmadia, G.N.; Possingham, H.P.; Riginos, C.; Treml, E.A.; Mumby, P.J. Marine Reserve Targets to Sustain and Rebuild Unregulated Fisheries. PLoS Biol. 2017, 15, 1–20. [Google Scholar] [CrossRef] [PubMed]
- Cinner, J.E. Coral Reef Livelihoods. Curr. Opin. Environ. Sustain. 2014, 7, 65–71. [Google Scholar] [CrossRef]
- Hurtado, A.Q.; Gerung, G.S.; Yasir, S.; Critchley, A.T. Cultivation of Tropical Red Seaweeds in the BIMP-EAGA Region. J. Appl. Phycol. 2014, 26, 707–718. [Google Scholar] [CrossRef]
- Le Pennec, M.; Anastas, M.; Bichet, H.; Buestel, D.; Cochard, J.-C.; Cochennec-Laureau, N.; Coeroli, M.; Conte, E.; Correia, P.; Fougerousse-Tsing, A.; et al. Huître perlière et perle de Tahiti; Université de la Polynésie française: Faaa, French Polynesia, 2009; 204p, ISBN 978-2-9534554-2-7. [Google Scholar]
- Sangare, N.; Lo-Yat, A.; Le Moullac, G.; Pecquerie, L.; Thomas, Y.; Lefebvre, S.; Le Gendre, R.; Beliaeff, B.; Andréfouët, S. Impact of Environmental Variability on Pinctada Margaritifera Life-History Traits: A Full Life Cycle Deb Modeling Approach. Ecol. Modell. 2020, 423, 109006. [Google Scholar] [CrossRef]
- Andréfouët, S.; Dutheil, C.; Menkes, C.E.; Bador, M.; Lengaigne, M. Mass Mortality Events in Atoll Lagoons: Environmental Control and Increased Future Vulnerability. Glob. Chang. Biol. 2015, 21, 195–205. [Google Scholar] [CrossRef] [PubMed]
- Hoeksema, B.W.; Benzoni, F. Multispecies Aggregations of Mushroom Corals in the Gambier Islands, French Polynesia. Coral Reefs 2013, 32, 1041. [Google Scholar] [CrossRef]
- Chevalier, J.P. Aperçu sur les Scléractiniaires des Iles Gambier. Cahier du Pacifique 1974, 18, 615–627. [Google Scholar]
- DRM—Direction des Ressources Marines. Zones Maritimes Réglementées; Direction des Ressources Marines: Papeete, French Polynesia, 2020.
- Yasumoto, T.; Inoue, A.; Bagnis, R.; Adaci, R. Finding of a dinoflagellate as a likely culprit of ciguatera. Bull. Jap. Soc. Sci. Fish 1977, 43, 1021–1026. [Google Scholar] [CrossRef]
- Bambridge, T. The Rahui: Legal Pluralism in Polynesian Traditional Management of Resources and Territories; Bambridge, T., Ed.; ANU-Press: Camberra, Australia, 2016; Available online: http://press.anu.edu.au/?p=337293 (accessed on 22 February 2022).
- Naidoo, R.; Balmford, A.; Ferraro, P.J.; Polasky, S.; Ricketts, T.H.; Rouget, M. Integrating Economic Costs into Conservation Planning. Trends Ecol. Evol. 2006, 21, 681–687. [Google Scholar] [CrossRef]
- Andréfouët, S.; Muller-Karger, F.E.; Robinson, J.A.; Kranenburg, C.; Torres-Pulliza, D.; Spraggins, S.A.; Murch, B. Global Assessment of Modern Coral Reef Extent and Diversity for Regional Science and Management Applications: A View from Space. In Proceedings of the 10th International Coral Reef Symposium, Japanese Coral Reef Society, Okinawa, Japan, 28 June–2 July 2004; Suzuki, Y., Nakamori, T., Hidaka, M., Kayanne, H., Casareto, B.E., Nadaoka, K., Yamano, H., Tsuchiya, M., Yamazato, K., Eds.; Japanese Coral Reef Society: Okinawa, Japan, 2006; pp. 1732–1745. [Google Scholar]
- Andréfouët, S.; Bionaz, O. Lessons from a Global Remote Sensing Mapping Project. A Review of the Impact of the Millennium Coral Reef Mapping Project for Science and Management. Sci. Total Environ. 2021, 776, 145987. [Google Scholar] [CrossRef]
- André, L.V.; Van Wynsberge, S.; Chinain, M.; Gatti, C.M.I.; Dempsey, A.; Andréfouët, S. A Framework for Mapping Local Knowledge on Ciguatera and Artisanal Fisheries to Inform Systematic Conservation Planning. ICES J. Mar. Sci. 2021, 78, 1357–1371. [Google Scholar] [CrossRef]
- Andréfouët, S.; Thomas, Y.; Dumas, F.; Lo, C. Revisiting Wild Stocks of Black Lip Oyster Pinctada Margaritifera in the Tuamotu Archipelago: The Case of Ahe and Takaroa Atolls and Implications for the Cultured Pearl Industry. Estuar. Coast. Shelf Sci. 2016, 182, 243–253. [Google Scholar] [CrossRef]
- Ardron, J.A.; Possingham, H.P.; Klein, C.J. Marxan Good Practices Handbook, Version 2; Ardron, J.A., Possingham, H.P., Klein, C.J., Eds.; Pacific Marine Analysis and Research Association: Victoria, BC, Canada, 2010; Volume 2010. [Google Scholar]
- Van Wynsberge, S.; Andréfouët, S.; Gaertner-Mazouni, N.; Remoissenet, G. Conservation and Resource Management in Small Tropical Islands: Trade-Offs between Planning Unit Size, Data Redundancy and Data Loss. Ocean Coast. Manag. 2015, 116, 37–43. [Google Scholar] [CrossRef]
- Léopold, M.; Guillemot, N.; Rocklin, D.; Chen, C. A Framework for Mapping Small-Scale Coastal Fisheries Using Fishers’ Knowledge. ICES J. Mar. Sci. 2014, 17, 12. [Google Scholar] [CrossRef]
- Beyer, H.L.; Dujardin, Y.; Watts, M.; Possingham, H.P. Solving Conservation Planning Problems with Integer Linear Programming (Appendices). Ecol. Modell. 2016, 2010, 1–16. [Google Scholar]
- Possingham, H.P.; Ball, I.R.; Andelman, S. Mathematical Methods for Identifying Representative Reserve Networks. In Quantitative Methods for Conservation Biology; Ferson, S., Burgman, M., Eds.; Springer: New York, NY, USA, 2000; pp. 291–305. [Google Scholar] [CrossRef]
- Justeau-Allaire, D. Constraint-Based Systematic Conservation Planning, A Generic and Expressive Approach; Université Montpellier 2: Montpellier, France, 2020. [Google Scholar]
- André, L.V. Systematic Spatial Planning for the Management of Black Pearl Farming Islands in the Pacific, with A Multi-Criteria Approach (Biodiversity, Resources, Uses, Ciguatera); Sorbonne Université ED 129: Paris, France, 2021. [Google Scholar]
- Cheok, J.; Pressey, R.L.; Weeks, R.; Andréfouët, S.; Moloney, J. Sympathy for the Devil: Detailing the Effects of Planning-Unit Size, Thematic Resolution of Reef Classes, and Socioeconomic Costs on Spatial Priorities for Marine Conservation. PLoS ONE 2016, 11, e0164869. [Google Scholar] [CrossRef] [PubMed]
- Kabbadj, L.; Van Wynsberge, S.; Andréfouët, S. Scaling Tropical Island Conservation Planning to the Regional Level Can Lead to Unbalanced Ecological Representation and Poor Social Equity among Islands. Mar. Policy 2018, 93, 31–39. [Google Scholar] [CrossRef]
- Reisser, C.M.O.; Le Gendre, R.; Chupeau, C.; Lo-Yat, A.; Planes, S.; Andréfouët, S. Population Connectivity and Genetic Assessment of Exploited and Natural Populations of Pearl Oysters within a French Polynesian Atoll Lagoon. Genes 2020, 11, 426. [Google Scholar] [CrossRef] [PubMed]
- Thomas, Y.; Dumas, F.; Andréfouët, S. Larval Connectivity of Pearl Oyster through Biophysical Modelling; Evidence of Food Limitation and Broodstock Effect. Estuar. Coast. Shelf Sci. 2016, 182, 283–293. [Google Scholar] [CrossRef]
- Dutheil, C.; Andréfouët, S.; Jullien, S.; Le Gendre, R.; Aucan, J.; Menkes, C. Characterization of South Central Pacific Ocean Wind Regimes in Present and Future Climate for Pearl Farming Application. Mar. Pollut. Bull. 2020, 160. [Google Scholar] [CrossRef]
- André, L.V.; Chinain, M.; Gatti, C.M.I.; Liao, V.; Van Wynsberge, S.; Tedesco, P.; Andréfouët, S. A Systematic Prioritization Approach for Identifying Suitable Pearl Oyster Restocking Zones Following a Mass Mortality Event in Takaroa Atoll, French Polynesia. Mar. Pollut. Bull. 2022, 176, 113472. [Google Scholar] [CrossRef]
- Jacquet, J.; Pauly, D. Funding Priorities: Big Barriers to Small-Scale Fisheries. Conserv. Biol. 2008, 22, 832–835. [Google Scholar] [CrossRef]
- FAO. The State of the World Fisheries and Aquaculture 2018—Meeting the Sustainable Development Goals; Licence: CC BY-NC-SA 3.0 IGO; FAO: Rome, Italy, 2018; Volume 35. [Google Scholar]
- Grafeld, S.; Oleson, K.L.L.; Teneva, L.; Kittinger, J.N. Follow That Fish: Uncovering the Hidden Blue Economy in Coral Reef Fisheries. PLoS ONE 2017, 12, e0182104. [Google Scholar] [CrossRef]
- Zeller, D.S.; Booth, S.; Pauly, D. Fisheries Contributions to the Gross Domestic Product: Underestimating Small-Scale Fisheries in the Pacific. Mar. Resour. Econ. 2007, 21, 355–374. [Google Scholar] [CrossRef]
- Bell, J.D.; Kronen, M.; Vunisea, A.; Nash, W.J.; Keeble, G.; Demmke, A.; Pontifex, S.; Andréfouët, S. Planning the Use of Fish for Food Security in the Pacific. Mar. Policy 2009, 33, 64–76. [Google Scholar] [CrossRef]
- Kronen, M.; Pinca, S.; Magron, F.; McArdle, B.; Vunisea, A.; Vigliola, L.; Kulbicki, M.; Andréfouët, S. Socio-Economic and Fishery Indicators to Identify and Monitor Artisanal Finfishing Pressure in Pacific Island Countries and Territories. Ocean Coast. Manag. 2012, 55, 63–73. [Google Scholar] [CrossRef]
- Thiault, L.; Collin, A.; Chlous, F.; Gelcich, S.; Claudet, J. Combining Participatory and Socioeconomic Approaches to Map Fishing Effort in Smallscale Fisheries. PLoS ONE 2017, 12, e0176862. [Google Scholar] [CrossRef] [PubMed]
- Aswani, S.; Lauer, M. Incorporating Fishermen’s Local Knowledge and Behavior into Geographical Information Systems (GIS) for Designing Marine Protected Areas in Oceania. Hum. Organ. 2006, 65, 81–102. [Google Scholar] [CrossRef]
- Bell, J.D.; Albert, J.; Andréfouët, S.; Andrew, N.L.; Blanc, M.; Bright, P.; Brogan, D.; Campbell, B.; Govan, H.; Hampton, J.; et al. Optimising the Use of Nearshore Fish Aggregating Devices for Food Security in the Pacific Islands. Mar. Policy 2015, 56, 98–105. [Google Scholar] [CrossRef]
- Bell, J.D.; Cisneros-Montemayor, A.; Hanich, Q.; Johnson, J.E.; Lehodey, P.; Moore, B.R.; Pratchett, M.S.; Reygondeau, G.; Senina, I.; Virdin, J.; et al. Adaptations to Maintain the Contributions of Small-Scale Fisheries to Food Security in the Pacific Islands. Mar. Policy 2018, 88, 303–314. [Google Scholar] [CrossRef]
- DRM. 2020. Available online: http://www.ressources-marines.gov.pf/cdi/bulletin-statistique/chiffres-cles-peche-aquaculture-et-perliculture-en-polynesie-francaise/ (accessed on 31 August 2021).
- Remoissenet, G.; Wabnitz, C. Postlarval Capture and Culture of Tridacna Maxima Giant Clams in French Polynesia. SPC Fish. Newsl. 2012, 139, 16–19. [Google Scholar]
- IUCN. French Polynesia Case Study. Sustainable Use, Mariculture and Conservation of Giant Clams in the Marine Regulated Fishing Area Reao Atoll, Tuamotu. Case Study N° 4; IUCN: Gland, Switzerland, 2021. [Google Scholar]
- Wabnitz, C.C.C. SPC ACTIVITIES Commercial Marine Aquarium Surveys in Samoa; SPC Fisheries Newsletter #146: Nouméa, New Caldonia, 2015; pp. 11–13. [Google Scholar]
- Duvat, V. L’évolution de La Recherche Sur Les Systèmes Coralliens (1960–2007). VertigO 2008, 8, 1–17. [Google Scholar] [CrossRef]
- Eger, S.L.; Courtenay, S.C. Integrated Coastal and Marine Management: Insights from Lived Experiences in the Bay of Fundy, Atlantic Canada. Ocean Coast. Manag. 2021, 204, 105457. [Google Scholar] [CrossRef]
- Mills, M.; Weeks, R.; Pressey, R.L.; Gleason, M.G.; Eisma-osorio, R.; Lombard, A.T.; Harris, J.M.; Killmer, A.B.; White, A.; Morrison, T.H. Real-World Progress in Overcoming the Challenges of Adaptive Spatial Planning in Marine Protected Areas. Biol. Conserv. 2015, 181, 54–63. [Google Scholar] [CrossRef]
- Winter, K.B.; Rii, Y.M.; Reppun, F.A.W.L.; Delaforgue Hintzen, K.; Alegado, R.A.; Bowen, B.W.; Bremer, L.L.; Coffman, M.; Deenik, J.L.; Donahue, M.J.; et al. Collaborative Research to Inform Adaptive Comanagement: A Framework for the Heʻeia National Estuarine Research Reserve. Ecol. Soc. 2020, 25, 1–17. [Google Scholar] [CrossRef]
- Gillett, R. Fisheries in the Economies of Pacific Island Countries and Territories; Pacific Community: Nouméa, New Caledonia, 2016; 684p, Available online: https://www.spc.int/resource-centre/publications/benefish-report-by-gillett-fisheries-in-the-economies-of-pacific (accessed on 22 February 2022).
- Cinner, J.E.; Daw, T.; McClanahan, T.R. Socioeconomic Factors That Affect Artisanal Fishers’ Readiness to Exit a Declining Fishery. Conserv. Biol. 2009, 23, 124–130. [Google Scholar] [CrossRef] [PubMed]



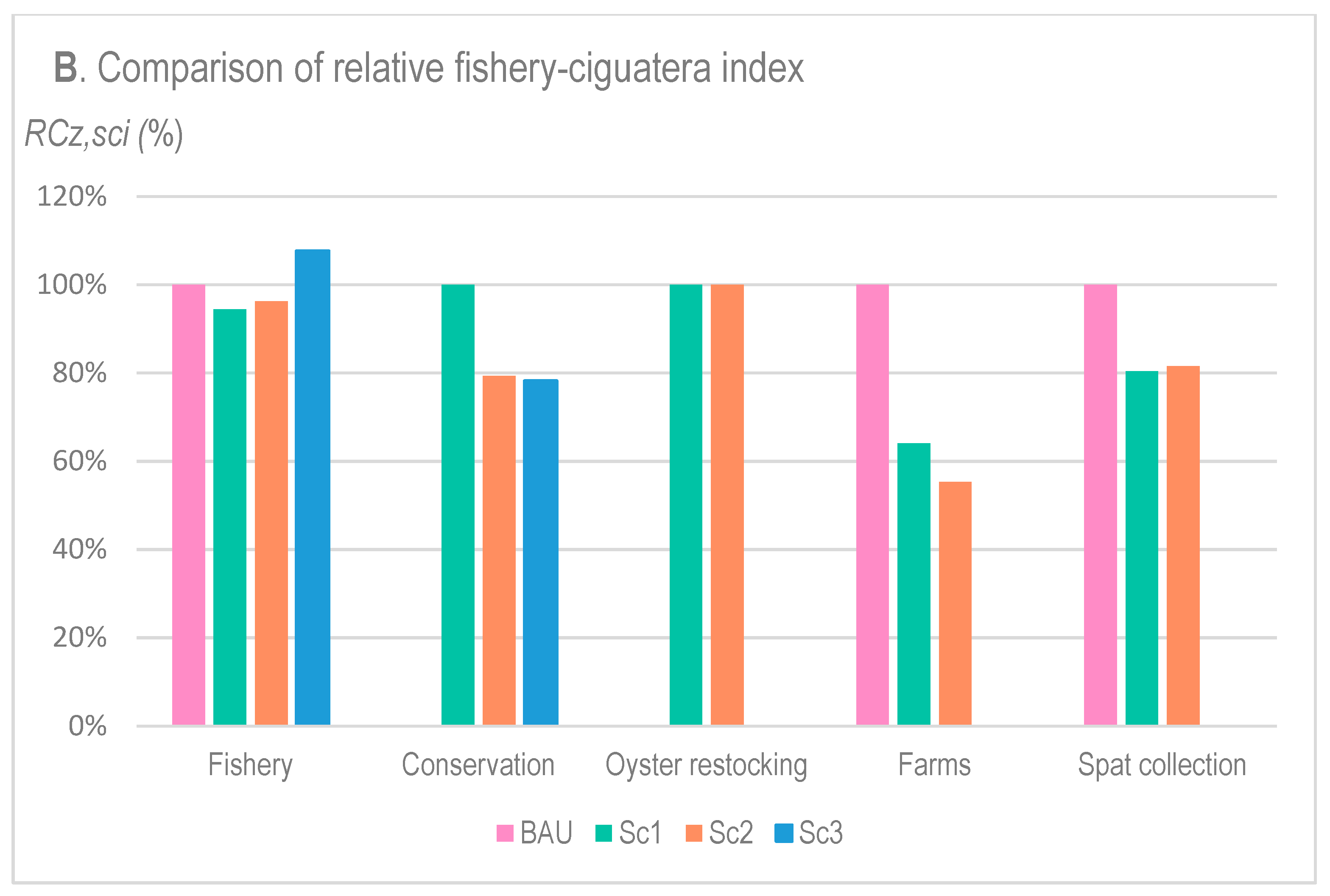


| Scenario or Scheme | Criteria | Goals | |
|---|---|---|---|
| BAU scheme | ‘Business-as-usual’ | The present use of the lagoon with pearl farming & fisheries | - |
| Scenario 1 | Diversification 1 | Pearl farming & conservation | Reach both the revised surface area targeted for the farming objective and conservation objectives while minimizing surface areas |
| Scenario 2 | Diversification 2 | Pearl farming, conservation & fisheries | Reach both the surface area targeted for the farming objective and conservation objectives while minimizing the loss of fishing grounds for fishers |
| Scenario 3 | ‘Back-to-tradition’ | Conservation & fisheries | Reach the conservation objectives while minimizing the loss of fishing grounds for fishers |
| GIS Layers | Object | Resolution | Source | |
|---|---|---|---|---|
| Original information | Layer 1 | Habitat map | 77 classes/from 4 m image resolution | This study |
| Layer 2 | Fishing grounds and associated catch | Fishers mapping: ~1 ha precision | This study | |
| Layer 3 | Ciguatera risk map | Fishers mapping: ~1 ha precision | This study | |
| Layer 4 | Wild oyster stock abundance | Habitat resolution | This study | |
| Layer 5 | Spat collection area | Legal unit: 1 ha min | DRM record | |
| Layer 6 | Farm concessions | Legal unit: 1 ha min | DRM record | |
| Layer 7 | Traditionally managed sectors | Context-dependent polygons | DRM record | |
| Information aggregated by PU | Layer 8 | Fishery catch/PU | 10 ha PU | Layer 2 aggregated by PUs |
| Layer 9 | Fishery–ciguatera index/PU | 10 ha PU | Combination of Layers 2 and 3, aggregated by PUs | |
| Layer 10 | Oyster restocking zones/PU | 10 ha PU | Combination of Layers 1, 6, 7, aggregated by PUs | |
| Layer 11 | Farm suitability index/PU | 10 ha PU | Combination of Layers 1, 6, 7, aggregated by PUs |
| Zone | Layer | Objective, Cost, or Locked Zone | Scenario |
|---|---|---|---|
| Optimal farm concession zone | Layer 11 | Farming Objective | 1, 2 |
Conservation zone:
| Layer 1, 4 | Conservation Objective | 1, 2 |
Conservation zone:
| Layer 1 | Conservation Objective | 3 |
| Fishery (and ciguatera free) zone | Layer 8 or 9 | Fishery opportunity Cost | 2, 3 |
| Conservation zone | Layer 1 | Cost of the surface area of the conservation zone | 1 |
| Oyster restocking zone | Layer 10 | Locked in a conservation zone | 1, 2 |
| Spat collection zone | Layer 5 | Locked out from farm zone | 1, 2 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
André, L.V.; Van Wynsberge, S.; Chinain, M.; Gatti, C.M.I.; Liao, V.; Andréfouët, S. Spatial Solutions and Their Impacts When Reshuffling Coastal Management Priorities in Small Islands with Limited Diversification Opportunities. Sustainability 2022, 14, 3871. https://doi.org/10.3390/su14073871
André LV, Van Wynsberge S, Chinain M, Gatti CMI, Liao V, Andréfouët S. Spatial Solutions and Their Impacts When Reshuffling Coastal Management Priorities in Small Islands with Limited Diversification Opportunities. Sustainability. 2022; 14(7):3871. https://doi.org/10.3390/su14073871
Chicago/Turabian StyleAndré, Laure Vaitiare, Simon Van Wynsberge, Mireille Chinain, Clémence Mahana Iti Gatti, Vetea Liao, and Serge Andréfouët. 2022. "Spatial Solutions and Their Impacts When Reshuffling Coastal Management Priorities in Small Islands with Limited Diversification Opportunities" Sustainability 14, no. 7: 3871. https://doi.org/10.3390/su14073871
APA StyleAndré, L. V., Van Wynsberge, S., Chinain, M., Gatti, C. M. I., Liao, V., & Andréfouët, S. (2022). Spatial Solutions and Their Impacts When Reshuffling Coastal Management Priorities in Small Islands with Limited Diversification Opportunities. Sustainability, 14(7), 3871. https://doi.org/10.3390/su14073871









