Continuous Pest Surveillance and Monitoring Constitute a Tool for Sustainable Agriculture: Case of Xylella fastidiosa in Morocco
Abstract
:1. Introduction
2. Materials and Methods
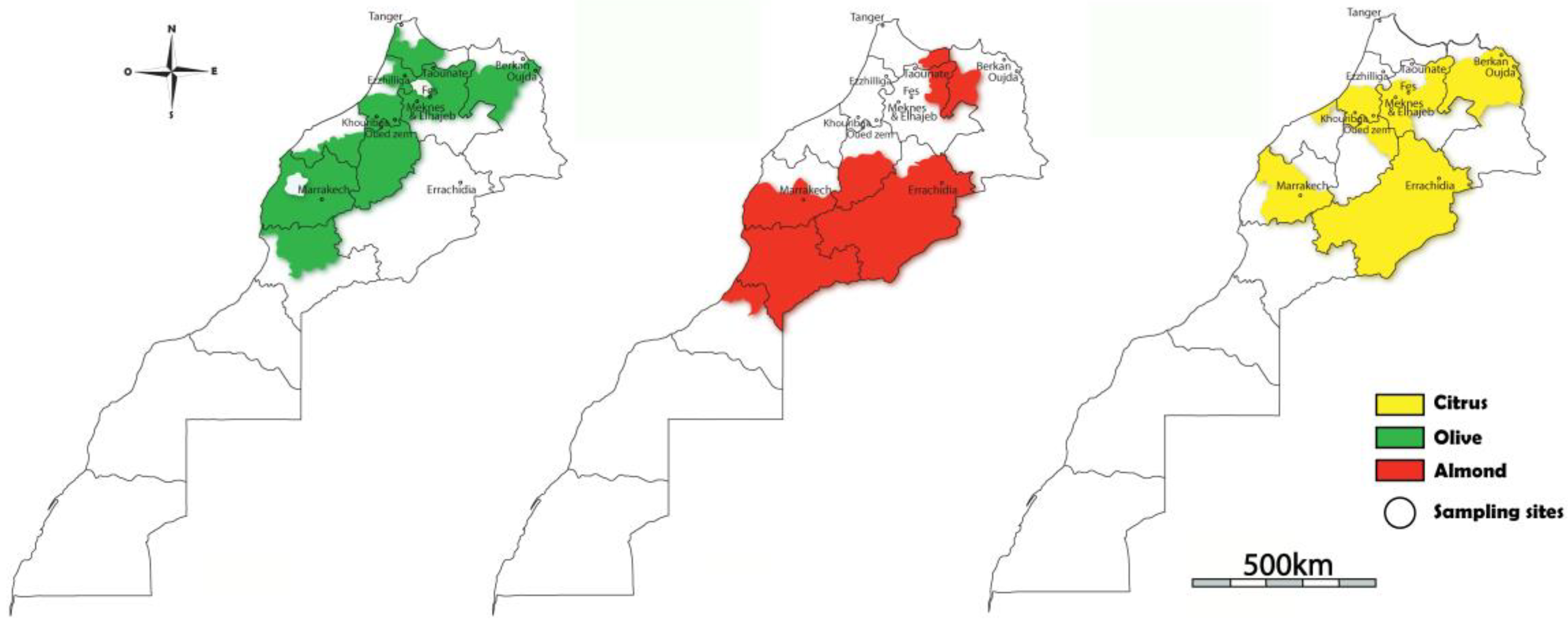
2.1. Study Areas and Collection of Samples
2.2. Sample Preparation
2.3. Testing Techniques
2.3.1. Serological Assay
2.3.2. Molecular Assay
- -
- DNA isolation
- -
- PCR Primers and Cycling Conditions
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Rossi, A. Etude de La Prévalence des Vecteurs Potentiels Naturellement Infectés Par Xylella fastidiosa subsp. pauca dans Les Oliveraies du Sud de L’italie. Master’s Thesis, Université de Liège, Liege, Belgium, September 2017. [Google Scholar]
- Frem, M.; Chapman, D.; Fucilli, V.; Choueiri, E.; El Moujabber, M.; La Notte, P.; Nigro, F. Xylella fastidiosa invasion of new countries in Europe, the Middle East and North Africa: Ranking the potential exposure scenarios. NeoBiota 2020, 59, 77–97. [Google Scholar] [CrossRef]
- Del Coco, L.; Migoni, D.; Girelli, C.R.; Angilè, F.; Scortichini, M.; Fanizzi, F.P. Soil and Leaf Ionome Heterogeneity in Xylella fastidiosa subsp. pauca-Infected, Non-Infected and Treated Olive Groves in Apulia, Italy. Plants 2020, 9, 760. [Google Scholar] [CrossRef]
- Giampetruzzi, A.; Baptista, P.; Morelli, M.; Cameirao, C.; Lino Neto, T.; Costa, D.; D’Attoma, G.; Abou Kubaa, R.; Altamura, G.; Saponari, M.; et al. Differences in the Endophytic Microbiome of Olive Cultivars Infected by Xylella fastidiosa across Seasons. Pathogens 2020, 9, 723. [Google Scholar] [CrossRef]
- Abou Kubaa, R.; Giampetruzzi, A.; Altamura, G.; Saponari, M.; Saldarelli, P. Infections of the Xylella fastidiosa subsp. pauca Strain “De Donno” in Alfalfa (Medicago sativa) Elicits an Overactive Immune Response. Plants 2019, 8, 335. [Google Scholar] [CrossRef] [Green Version]
- Marcelletti, S.; Scortichini, M. Genome-wide comparison and taxonomic relatedness of multiple Xylella fastidiosa strains reveal the occurrence of three subspecies and a new Xylella species. Arch. Microbiol. 2016, 198, 803–812. [Google Scholar] [CrossRef]
- Cendoya, M.; Martínez-Minaya, J.; Dalmau, V.; Ferrer, A.; Saponari, M.; Conesa, D.; López-Quílez, A.; Vicent, A. Spatial Bayesian Modeling Applied to the Surveys of Xylella fastidiosa in Alicante (Spain) and Apulia (Italy). Front. Plant Sci. 2020, 11, 1204. [Google Scholar] [CrossRef]
- Frem, M.; Santeramo, F.G.; Lamonaca, E.; El Moujabber, M.; Choueiri, E.; La Notte, P.; Nigro, F.; Bozzo, F.; Fucilli, V. Landscape restoration due to Xylella fastidiosa invasion in Italy: Assessing the hypothetical public’s preferences. NeoBiota 2021, 66, 31–54. [Google Scholar] [CrossRef]
- Afechtal, M.; Vicent, A.; Saponari, M.; D’Onghia, A.M. Pest risk analysis on Xylella fastidiosa in Morocco. J. Plant Prot. Res. 2018, 58, 215–219. [Google Scholar]
- Available online: https://resourcetrade.earth/data (accessed on 12 December 2021).
- Haddad, N.; Afechtal, M.; Streito, J.C.; Ouguas, Y.; Benkirane, R.; Lhomme, P.; Smaili, M.C. Occurrence in Morocco of potential vectors of Xylella fastidiosa that may contribute to the active spread of the bacteria. Ann. De La Société Entomol. De Fr. 2021, 57, 359–371. [Google Scholar] [CrossRef]
- Cardone, G.; Digiaro, M.; Djelouah, K.; El Bilali, H.; Michel FR, E.M.; Fucilli, V.; Yaseen, T. Potential socio-economic impact of Xylella fastidiosa in the Near East and North Africa (NENA): Risk of introduction and spread, risk perception and socio-economic effects. New Mediterr. J. Econ. Agric. Environ. 2021, 20, 27. [Google Scholar]
- Nunney, L.; Schuenzel, E.L.; Scally, M.; Bromley, R.E.; Stouthamer, R. Large-Scale Intersubspecific Recombination in the Plant-Pathogenic Bacterium Xylella fastidiosa Is Associated with the Host Shift to Mulberry. Appl. Environ. Microbiol. 2014, 80, 3025–3033. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Loreti, S.; Pucci, N.; Loconsole, G.; Modesti, V.; Potere, O.; Lucchesi, S.; Saponari, M. Organization of ring tests on diagnostic methods among Italian laboratories. Xylella fastidiosa & the Olive Quick Decline Syndrome (OQDS). Options Méditerranéennes 2017, 121, 69. [Google Scholar]
- Sherald, J.L.; Lei, J.D. Evaluation of a rapid ELISA test kit for detection of Xylella fastidiosa in landscape trees. Plant Dis. 1991, 75, 200–203. [Google Scholar] [CrossRef]
- Marchi, G.; Rizzo, D.; Ranaldi, F.; Ghelardini, L.; Ricciolini, M.; Scarpelli, I.; Surico, G. First detection of Xylella fastidiosa subsp. multiplex DNA in Tuscany (Italy). Phytopathol. Mediterr. 2018, 57, 363–364. [Google Scholar]
- Djelouah, K.; Frasheri, D.; Valentini, F.; D’Onghia, A.M.; Digiaro, M. Direct tissue blot immunoassay for detection of Xylella fastidiosa in olive trees. Phytopathol. Mediterr. 2014, 53, 559–564. [Google Scholar]
- Minsavage, G.V.; Thompson, C.M.; Hopkins, D.L.; Leite, R.M.V.B.C.; Stall, R.E. Development of a polymerase chain reaction protocol for detection of Xylella fastidiosa in plant tissue. Phytopathology 1994, 84, 456–461. [Google Scholar] [CrossRef]
- Francis, M.; Lin, H.; Cabrera-La Rosa, J.; Doddapaneni, H.; Civerolo, E.L. Genome-based PCR primers for specific and sensitive detection and quantification of Xylella fastidiosa. Eur. J. Plant Pathol. 2006, 115, 203–213. [Google Scholar] [CrossRef]
- Harper, S.J.; Ward, L.I.; Clover GR, G. Development of LAMP and real-time PCR methods for the rapid detection of Xylella fastidiosa for quarantine and field applications. Phytopathology 2010, 100, 1282–1288. [Google Scholar] [CrossRef]
- Li, W.; Xi, B.; Yang, W.; Hawkins, M.; Schubart, U.K. Complex DNA melting profiles of small PCR products revealed using SYBR® Green I. BioTechniques 2003, 35, 702–706. [Google Scholar] [CrossRef]
- Yaseen, T.; Drago, S.; Valentini, F.; Elbeaino, T.; Stampone, G.; Digiaro, M.; D’Onghia, A.M. On-site detection of Xylella fastidiosa in host plants and in “spy insects” using the real-time loop-mediated isothermal amplification method. Phytopathol. Mediterr. 2015, 54, 488–496. [Google Scholar]
- European Food Safety Authority (EFSA); Vos, S.; Camilleri, M.; Diakaki, M.; Lázaro, E.; Parnell, S.; Vicent, A. Pest survey card on Xylella fastidiosa. EFSA Supporting Publ. 2019, 16, 1667E. [Google Scholar]
- Loconsole, G.; Potere, O.; Boscia, D.; Altamura, G.; Djelouah, K.; Elbeaino, T.; Saponari, M. Detection of Xylella fastidiosa in olive trees by molecular and serological methods. J. Plant Pathol. 2014, 96, 7–14. [Google Scholar]
- Rodrigues, C.M.; de Souza, A.A.; Takita, M.A.; Kishi, L.T.; Machado, M.A. RNA-Seq analysis of Citrus reticulata in the early stages of Xylella fastidiosa infection reveals auxin-related genes as a defense response. BMC Genom. 2013, 14, 676. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- European and Mediterranean Plant Protection Organization (EPPO). Diagnostic protocols for regulated pests. Xylella fastidiosa. Bull. OEPP/EPPO Bull. 2004, 34, 187–192. [Google Scholar] [CrossRef]
- El Handi, K.; Hafidi, M.; Habbadi, K.; El Moujabber, M.; Ouzine, M.; Benbouazza, A.; Achbani, E.H. Assessment of Ionomic, Phenolic and Flavonoid Compounds for a Sustainable Management of Xylella fastidiosa in Morocco. Sustainability 2021, 13, 7818. [Google Scholar] [CrossRef]
- Saponari, M.; D’Attoma, G.; Abou Kubaa, R.; Loconsole, G.; Altamura, G.F.; Zicca, S.; Rizzo, D.; Boscia, D. A new variant of Xylella fastidiosa subspecies multiplex detected in different host plants in the recently emerged outbreak in the region of Tuscany, Italy. Eur. J. Plant Pathol. 2019, 154, 1195–1200. [Google Scholar] [CrossRef] [Green Version]
- D’Attoma, G.; Morelli, M.; Saldarelli, P.; Saponari, M.; Giampetruzzi, A.; Boscia, D.; Cobine, P.A. Ionomic differences between susceptible and resistant olive cultivars infected by Xylella fastidiosa in the outbreak area of Salento, Italy. Pathogens 2019, 8, 272. [Google Scholar] [CrossRef] [Green Version]
- Afechtal, M.; Ait Friha, A.; Bibi, I. A preliminary survey on the presence of Xylella fastidiosa in olive, citrus and grapevine groves in Morocco. Rev. Maroc. Des. Sci. Agron. Et Vétérinaires 2017, 6, 6–9. [Google Scholar]
- EPPO. European and Mediterranean Plant Protection Organization Reporting Service No. 05. Nr. 2016/102. 2017. Available online: https://gd.eppo.int/reporting/article-6070 (accessed on 12 December 2021).
- Firrao, G.; Bazzi, C. Specific identification of Xylella fastidiosa using the polymerase chain reaction. Phytopathol. Mediterr. 1994, 33, 90–92. [Google Scholar]
- AbuObeid, I.; Al-Karablieh, N.; Haddadin, J.; Al Omari, R.; Al-Jabaree, A.M.; Al-Elaumi, L.; Mazahreh, S. Survey on the presence of Xylella fastidiosa, the causal agent of olive quick decline syndrome (OQDS) on olives in Jordan. Arch. Phytopathol. Plant Prot. 2020, 53, 188–197. [Google Scholar] [CrossRef]
| Crop | Location | N of Groves | N of Collected Trees |
|---|---|---|---|
| Olive | Tanger | 4 | 141 |
| Béni Mellal | 5 | 110 | |
| Marrakech | 4 | 120 | |
| Errachidia | 5 | 170 | |
| Meknès | 4 | 116 | |
| Citrus | Azilal | 5 | 80 |
| Meknès | 10 | 90 | |
| Almond | Gharb | 3 | 50 |
| Haouz | 5 | 60 | |
| Meknès | 6 | 70 | |
| Total | 51 | 1007 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
El Handi, K.; Hafidi, M.; Sabri, M.; Frem, M.; El Moujabber, M.; Habbadi, K.; Haddad, N.; Benbouazza, A.; Abou Kubaa, R.; Achbani, E.H. Continuous Pest Surveillance and Monitoring Constitute a Tool for Sustainable Agriculture: Case of Xylella fastidiosa in Morocco. Sustainability 2022, 14, 1485. https://doi.org/10.3390/su14031485
El Handi K, Hafidi M, Sabri M, Frem M, El Moujabber M, Habbadi K, Haddad N, Benbouazza A, Abou Kubaa R, Achbani EH. Continuous Pest Surveillance and Monitoring Constitute a Tool for Sustainable Agriculture: Case of Xylella fastidiosa in Morocco. Sustainability. 2022; 14(3):1485. https://doi.org/10.3390/su14031485
Chicago/Turabian StyleEl Handi, Kaoutar, Majida Hafidi, Miloud Sabri, Michel Frem, Maroun El Moujabber, Khaoula Habbadi, Najat Haddad, Abdellatif Benbouazza, Raied Abou Kubaa, and El Hassan Achbani. 2022. "Continuous Pest Surveillance and Monitoring Constitute a Tool for Sustainable Agriculture: Case of Xylella fastidiosa in Morocco" Sustainability 14, no. 3: 1485. https://doi.org/10.3390/su14031485
APA StyleEl Handi, K., Hafidi, M., Sabri, M., Frem, M., El Moujabber, M., Habbadi, K., Haddad, N., Benbouazza, A., Abou Kubaa, R., & Achbani, E. H. (2022). Continuous Pest Surveillance and Monitoring Constitute a Tool for Sustainable Agriculture: Case of Xylella fastidiosa in Morocco. Sustainability, 14(3), 1485. https://doi.org/10.3390/su14031485








