Chemical Recycling of Plastic Marine Litter: First Analytical Characterization of The Pyrolysis Oil and of Its Fractions and Comparison with a Commercial Marine Gasoil
Abstract
:1. Introduction
2. Materials and Methods
2.1. Plastic Wastes Feedstock
2.2. Pyrolysis Process
2.3. Pyrolysis Oil Distillation
2.4. GC–MS Analysis
2.5. Fourier Transform Infrared (FTIR) Spectroscopy
2.6. ISO8217:2017 Analysis
3. Results and Discussion
3.1. ISO Compliance of MGO (ml-py)
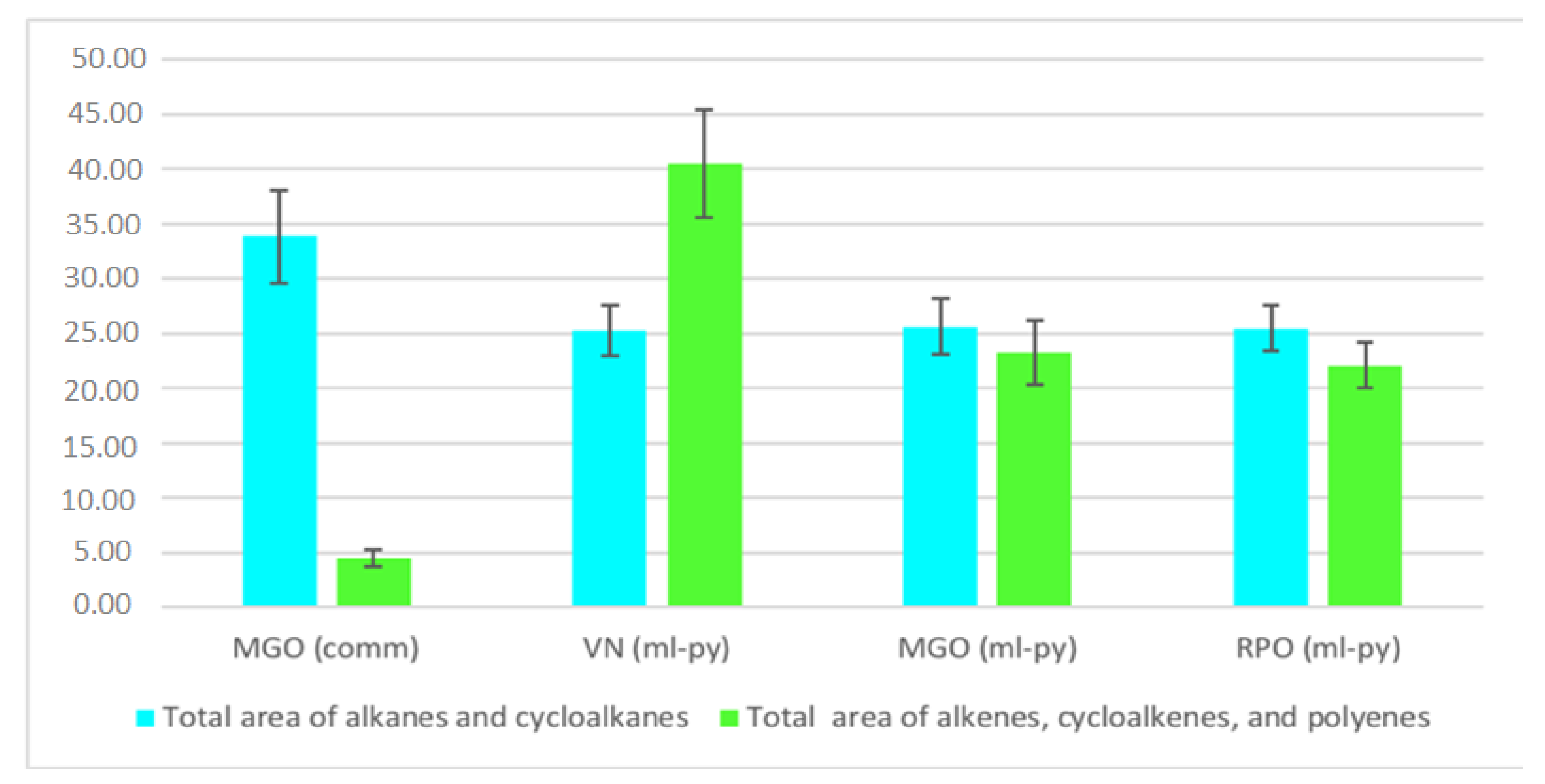
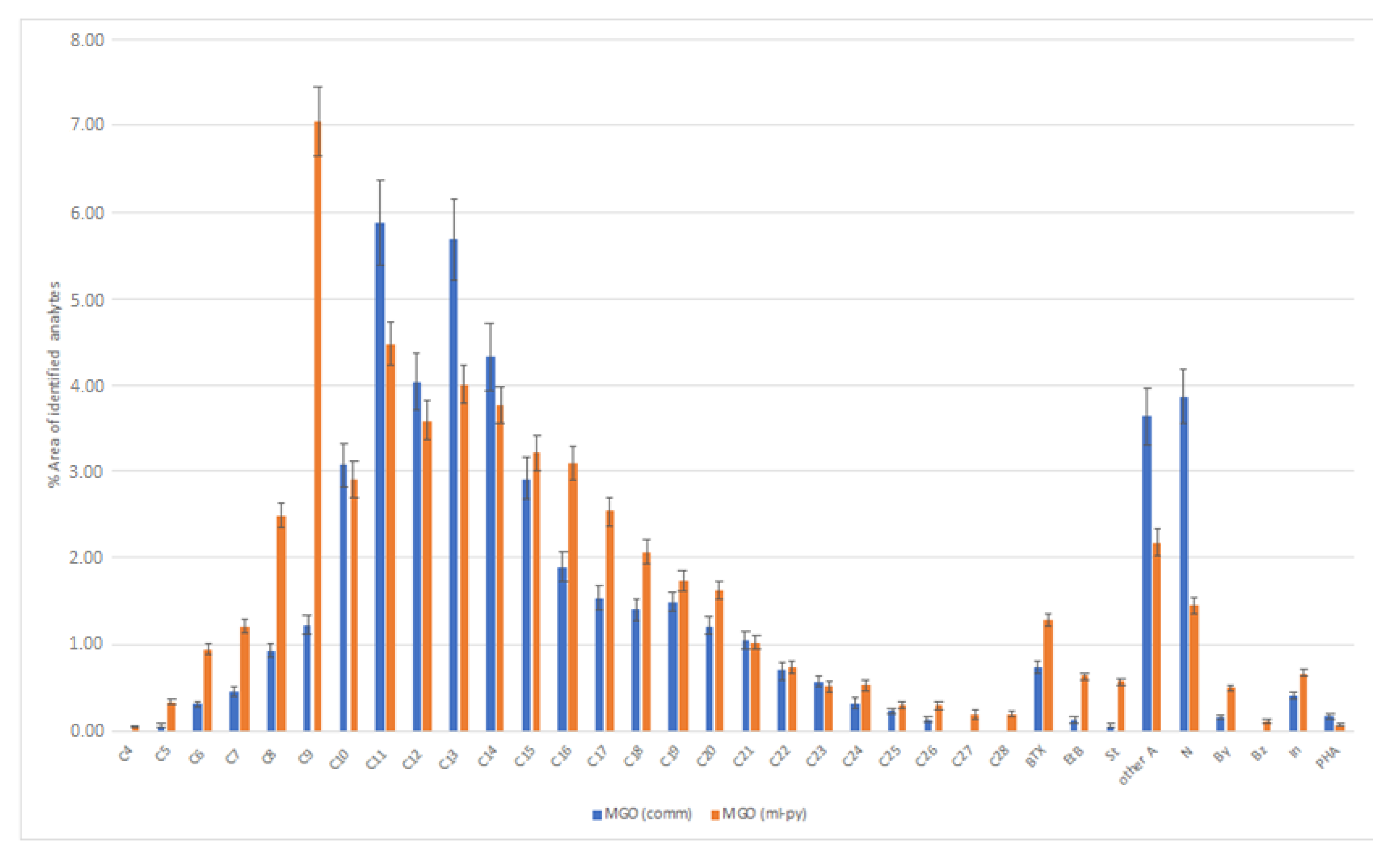
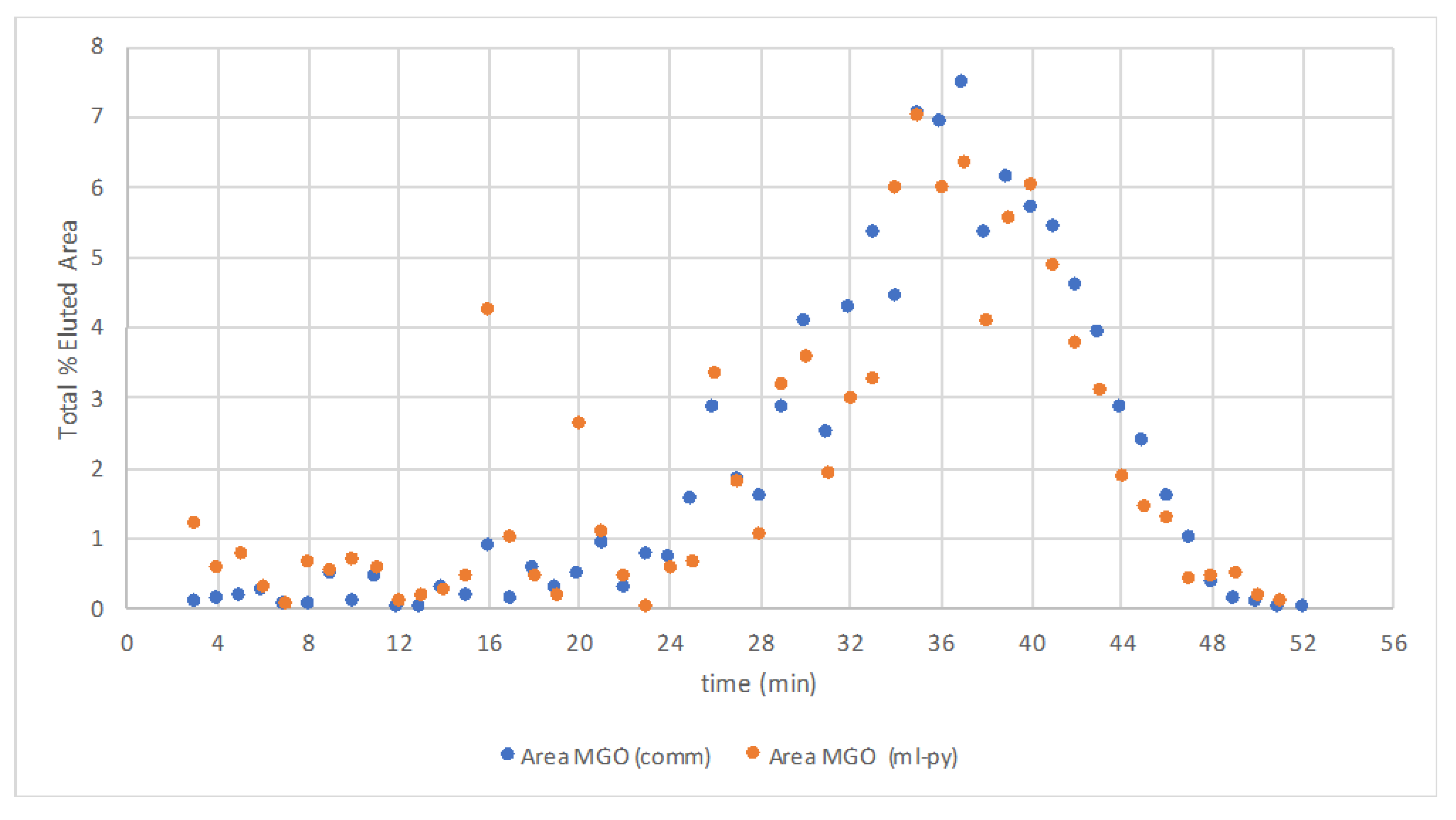
3.2. GC-MS Analysis of the Liquid Samples
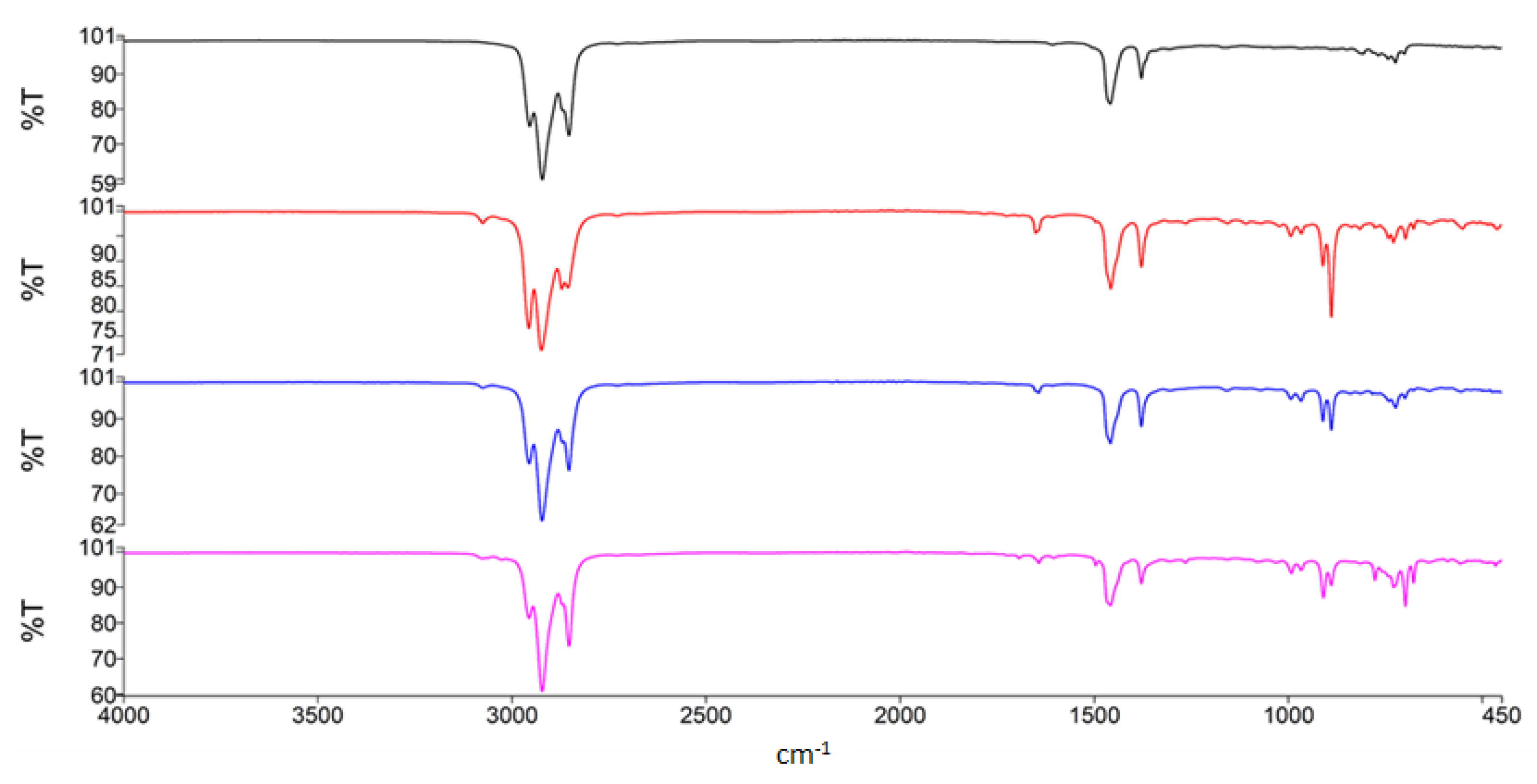
3.3. FTIR Analysis
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| MGO (comm) | commercial marine gasoil |
| VN (ml-py) | virgin naphtha from marine litter pyrolysis |
| MGO (ml-py) | marine gasoil from marine litter pyrolysis |
| RPO (ml-py) | raw oil from marine litter pyrolysis |
| GC-MS | Gas Chromatography-Mass Spectrometry |
References
- Wayman, C.; Niemann, H. The fate of plastic in the ocean environment—A minireview. Environ. Sci. Process. Impacts 2021, 23, 198–212. [Google Scholar] [CrossRef]
- Canning-Clode, J.; Sepúlveda, P.; Almeida, S.; Monteiro, J. Will COVID-19 Containment and Treatment Measures Drive Shifts in Marine Litter Pollution? Front. Mar. Sci. 2020, 7. [Google Scholar] [CrossRef]
- Baena-González, J.; Santamaria-Echart, A.; Aguirre, J.L.; González, S. Chemical recycling of plastic waste: Bitumen, solvents, and polystyrene from pyrolysis oil. Waste Manag. 2020, 118, 139–149. [Google Scholar] [CrossRef]
- Ragaert, K.; Delva, L.; Van Geem, K. Mechanical and chemical recycling of solid plastic waste. Waste Manag. 2017, 69, 24–58. [Google Scholar] [CrossRef] [PubMed]
- Iñiguez-Cantos, M.E.; Conesa, J.; Fullana, A. Marine debris occurrence and treatment: A review. Renew. Sustain. Energy Rev. 2016, 64, 394–402. [Google Scholar] [CrossRef] [Green Version]
- Aguado, J.; Serrano, D.P.; Escola, J.M. Catalytic Upgrading of Plastic Wastes. In Feedstock Recycling and Pyrolysis of Waste Plastics: Converting Waste Plastics into Diesel and Other Fuels; Royal Society of Chemistry: London, UK, 2006; pp. 73–110. [Google Scholar] [CrossRef]
- Toraman, H.E.; Dijkmans, T.; Djokic, M.R.; Van Geem, K.M.; Marin, G.B. Detailed compositional characterization of plastic waste pyrolysis oil by comprehensive two-dimensional gas-chromatography coupled to multiple detectors. J. Chromatogr. A 2014, 1359, 237–246, Erratum in Arab. J. Geosci. 2020, 13, 504. [Google Scholar] [CrossRef]
- Larrain, M.; Van Passel, S.; Thomassen, G.; Kresovic, U.; Alderweireldt, N.; Moerman, E.; Billen, P. Economic performance of pyrolysis of mixed plastic waste: Open-loop versus closed-loop recycling. J. Clean. Prod. 2020, 270, 122442. [Google Scholar] [CrossRef]
- Tomar, M.; Jain, A.; Pujari, P.C.; Dewal, H.; Kumar, N. Potentials of waste plastic pyrolysis oil as an extender fuel for diesel engine. Arab. J. Geosci. 2020, 13, 1–10. [Google Scholar] [CrossRef]
- Papari, S.; Bamdad, H.; Berruti, F. Pyrolytic Conversion of Plastic Waste to Value-Added Products and Fuels: A Review. Materials 2021, 14, 2586. [Google Scholar] [CrossRef]
- Anuar Sharuddin, S.D.; Abnisa, F.; Wan Daud, W.M.A.; Aroua, M.K. A review on pyrolysis of plastic wastes. Energy Convers. Manag. 2016, 115, 308–326. [Google Scholar] [CrossRef]
- Abbas-Abadi, M.S.; Nekoomanesh-Haghighi, M.; Yeganeh, H.; McDonald, A.G. Evaluation of pyrolysis process parameters on polypropylene degradation products. J. Anal. Appl. Pyrolysis 2014, 109, 272–277. [Google Scholar] [CrossRef]
- Ahmad, I.; Khan, M.I.; Khan, H.; Ishaq, M.; Tariq, R.; Gul, K.; Ahmad, W. Pyrolysis Study of Polypropylene and Polyethylene Into Premium Oil Products. Int. J. Green Energy 2014, 12, 663–671. [Google Scholar] [CrossRef]
- López, A.; de Marco, I.; Caballero, B.; Laresgoiti, M.; Adrados, A. Pyrolysis of municipal plastic wastes: Influence of raw material composition. Waste Manag. 2010, 30, 620–627. [Google Scholar] [CrossRef]
- Onwudili, J.; Insura, N.; Williams, P. Composition of products from the pyrolysis of polyethylene and polystyrene in a closed batch reactor: Effects of temperature and residence time. J. Anal. Appl. Pyrolysis 2009, 86, 293–303. [Google Scholar] [CrossRef]
- Amjad, U.; Ishaq, M.; Rehman, H.U.; Ahmad, N.; Sherin, L.; Hussain, M.; Mustafa, M. Diesel and gasoline like fuel production with minimum styrene content from catalytic pyrolysis of polystyrene. Environ. Prog. Sustain. Energy 2020, 40, e13493. [Google Scholar] [CrossRef]
- Sogancioglu, M.; Yel, E.; Ahmetli, G. Investigation of the Effect of Polystyrene (PS) Waste Washing Process and Pyrolysis Temperature on (PS) Pyrolysis Product Quality. Energy Procedia 2017, 118, 189–194. [Google Scholar] [CrossRef]
- Rehan, M.; Miandad, R.; Barakat, M.A.; Ismail, I.M.I.; Almeelbi, T.; Gardy, J.; Hassanpour, A.; Khan, M.Z.; Demirbas, A.; Nizami, A.S. Effect of zeolite catalysts on pyrolysis liquid oil. Int. Biodeterior. Biodegrad. 2017, 119, 162–175. [Google Scholar] [CrossRef]
- Mangesh, V.L.; Padmanabhan, S.; Tamizhdurai, P.; Ramesh, A. Experimental investigation to identify the type of waste plastic pyrolysis oil suitable for conversion to diesel engine fuel. J. Clean. Prod. 2020, 246, 119066. [Google Scholar] [CrossRef]
- Gaurh, P.; Pramanik, H. Production of benzene/toluene/ethyl benzene/xylene (BTEX) via multiphase catalytic pyrolysis of hazardous waste polyethylene using low cost fly ash synthesized natural catalyst. Waste Manag. 2018, 77, 114–130. [Google Scholar] [CrossRef] [PubMed]
- Gaurh, P.; Pramanik, H. A novel approach of solid waste management via aromatization using multiphase catalytic pyrolysis of waste polyethylene. Waste Manag. 2018, 71, 86–96. [Google Scholar] [CrossRef]
- Venkatesan, H.; Sivamani, S.; Bhutoria, K.; Vora, H.H. Assessment of waste plastic oil blends on performance, combustion and emission parameters in direct injection compression ignition engine. Int. J. Ambient Energy 2019, 40, 170–178. [Google Scholar] [CrossRef]
- Areeprasert, C.; Asingsamanunt, J.; Srisawat, S.; Kaharn, J.; Inseemeesak, B.; Phasee, P.; Khaobang, C.; Siwakosit, W.; Chiemchaisri, C. Municipal Plastic Waste Composition Study at Transfer Station of Bangkok and Possibility of its Energy Recovery by Pyrolysis. Energy Procedia 2017, 107, 222–226. [Google Scholar] [CrossRef]
- Sarker, M.; Rashid, M.M.; Molla, M. Waste polypropylene plastic conversion into liquid hydrocarbon fuel for producing electricity and energies. Environ. Technol. 2012, 33, 2709–2721. [Google Scholar] [CrossRef]
- Yan, G.; Jing, X.; Wen, H.; Xiang, S. Thermal Cracking of Virgin and Waste Plastics of PP and LDPE in a Semibatch Reactor under Atmospheric Pressure. Energy Fuels 2015, 29, 2289–2298. [Google Scholar] [CrossRef]
- Huang, W.-C.; Huang, M.-S.; Huang, C.-F.; Chen, C.-C.; Ou, K.-L. Thermochemical conversion of polymer wastes into hydrocarbon fuels over various fluidizing cracking catalysts. Fuel 2010, 89, 2305–2316. [Google Scholar] [CrossRef]
- Cho, M.-H.; Jung, S.-H.; Kim, J.-S. Pyrolysis of Mixed Plastic Wastes for the Recovery of Benzene, Toluene, and Xylene (BTX) Aromatics in a Fluidized Bed and Chlorine Removal by Applying Various Additives. Energy Fuels 2010, 24, 1389–1395. [Google Scholar] [CrossRef]
- Ateş, F.; Miskolczi, N.; Borsodi, N. Comparision of real waste (MSW and MPW) pyrolysis in batch reactor over different catalysts. Part I: Product yields, gas and pyrolysis oil properties. Bioresour. Technol. 2013, 133, 443–454. [Google Scholar] [CrossRef]
- Singh, R.K.; Ruj, B.; Sadhukhan, A.K.; Gupta, P. Thermal degradation of waste plastics under non-sweeping atmosphere: Part 2: Effect of process temperature on product characteristics and their future applications. J. Environ. Manag. 2020, 261, 110112. [Google Scholar] [CrossRef]
- Allison, E.H.; Cheung, W.W.L.; Dey, M.M.; Halpern, B.S.; Mccauley, D.J.; Smith, M.; Vaitla, B.; Zeller, D.; Myers, S.S.; Nilsson, M.; et al. The State of World Fisheries and Aquaculture (SOFIA) 2010. FAO Proc. Nat. Acad. Sci. 2013, 382. [Google Scholar]
- Galgani, F.; Hanke, G.; Maes, T. Global Distribution, Composition and Abundance of Marine Litter. In Marine Anthropogenic Litter; Springer: Cham, Switzerland, 2015; pp. 29–56. [Google Scholar] [CrossRef] [Green Version]
- Consoli, P.; Falautano, M.; Sinopoli, M.; Perzia, P.; Canese, S.; Esposito, V.; Battaglia, P.; Romeo, T.; Andaloro, F.; Galgani, F.; et al. Composition and abundance of benthic marine litter in a coastal area of the central Mediterranean Sea. Mar. Pollut. Bull. 2018, 136, 243–247. [Google Scholar] [CrossRef]
- PlasticsEurope. The Facts 2018; PlasticsEurope: Brussels, Belgium, 2018. [Google Scholar]
- Okuwaki, A.; Yoshioka, T.; Asai, M.; Tachibana, H.; Wakai, K.; Tada, K. The Liquefaction of Plastic Containers and Packaging in Japan. In Feedstock Recycling and Pyrolysis of Waste Plastics: Converting Waste Plastics into Diesel and Other Fuels; John Wiley & Sons: Hoboken, NJ, USA, 2006; pp. 663–708. [Google Scholar] [CrossRef]
- Ripari, V.; Tomassetti, M.; Cecchi, T.; Enrico, B. Recipe, volatiles profile, sensory analysis, physico-chemical and microbial characterization of acidic beers from both sourdough yeasts and lactic acid bacteria. Eur. Food Res. Technol. 2018, 244, 2027–2040. [Google Scholar] [CrossRef]
- Ripari, V.; Cecchi, T.; Berardi, E. Microbiological characterisation and volatiles profile of model, ex-novo, and traditional Italian white wheat sourdoughs. Food Chem. 2016, 205, 297–307. [Google Scholar] [CrossRef] [PubMed]
- Benucci, I.; Cecchi, T.; Lombardelli, C.; Maresca, D.; Mauriello, G.; Esti, M. Novel microencapsulated yeast for the primary fermentation of green beer: Kinetic behavior, volatiles and sensory profile. Food Chem. 2020, 340, 127900. [Google Scholar] [CrossRef]
- Cecchi, T.; Alfei, B. Volatile profiles of Italian monovarietal extra virgin olive oils via HS-SPME–GC–MS: Newly identified compounds, flavors molecular markers, and terpenic profile. Food Chem. 2013, 141, 2025–2035. [Google Scholar] [CrossRef]
- Cherfaoui, M.; Cecchi, T.; Keciri, S.; Boudriche, L. Volatile compounds of Algerian extra-virgin olive oils: Effects of cultivar and ripening stage. Int. J. Food Prop. 2018, 21, 36–49. [Google Scholar] [CrossRef]
- Madricardo, F.; Ghezzo, M.; Nesto, N.; Mc Kiver, W.J.; Faussone, G.C.; Fiorin, R.; Riccato, F.; Mackelworth, P.C.; Basta, J.; De Pascalis, F.; et al. How to Deal With Seafloor Marine Litter: An Overview of the State-of-the-Art and Future Perspectives. Front. Mar. Sci. 2020, 7, 830. [Google Scholar] [CrossRef]
- Faussone, G.C.; Kržan, A.; Grilc, M. Conversion of Marine Litter from Venice Lagoon into Marine Fuels via Thermochemical Route: The Overview of Products, Their Yield, Quality and Environmental Impact. Sustainability 2021, 13, 9481. [Google Scholar] [CrossRef]
- IMO Annex 14, Resolution MEPC. 320(74) (adopted 17 May2019). 2019 Guidelines for Consistent Implementation ofthe 0.50% Sulphur Limit Under Marpol Annex VI. Available online: https://wwwcdn.imo.org/localresources/en/OurWork/Environment/Documents/Resolution%20MEPC.320%2874%29.pdf. (accessed on 1 November 2021).
- Choi, Y.J.; Cho, K.W.; Cho, B.W.; Yeo, Y.-K. Optimization of the Sulfolane Extraction Plant Based on Modeling and Simulation. Ind. Eng. Chem. Res. 2002, 41, 5504–5509. [Google Scholar] [CrossRef]
- Bak, A.; Kozik, V.; Dybal, P.; Kus, S.; Swietlicka, A.; Jampilek, J. Sulfolane: Magic Extractor or Bad Actor? Pilot-Scale Study on Solvent Corrosion Potential. Sustainability 2018, 10, 3677. [Google Scholar] [CrossRef] [Green Version]
- Pinto, F.; Costa, P.; Gulyurtlu, I.; Cabrita, I. Pyrolysis of plastic wastes. 1. Effect of plastic waste composition on product yield. J. Anal. Appl. Pyrolysis 1999, 51, 39–55. [Google Scholar] [CrossRef]
- Du, S.; Valla, J.A.; Parnas, R.S.; Bollas, G.M. Conversion of Polyethylene Terephthalate Based Waste Carpet to Benzene-Rich Oils through Thermal, Catalytic, and Catalytic Steam Pyrolysis. ACS Sustain. Chem. Eng. 2016, 4, 2852–2860. [Google Scholar] [CrossRef]
- Kumagai, S.; Grause, G.; Kameda, T.; Takano, T.; Horiuchi, H.; Yoshioka, T. Decomposition of Gaseous Terephthalic Acid in the Presence of CaO. Ind. Eng. Chem. Res. 2011, 50, 1831–1836. [Google Scholar] [CrossRef]
- Kumagai, S.; Grause, G.; Kameda, T.; Takano, T.; Horiuchi, H.; Yoshioka, T. Improvement of the Benzene Yield During Pyrolysis of Terephthalic Acid Using a CaO Fixed-Bed Reactor. Ind. Eng. Chem. Res. 2011, 50, 6594–6600. [Google Scholar] [CrossRef]
- Abbas-Abadi, M.S. The effect of process and structural parameters on the stability, thermo-mechanical and thermal degradation of polymers with hydrocarbon skeleton containing PE, PP, PS, PVC, NR, PBR and SBR. J. Therm. Anal. 2021, 143, 2867–2882. [Google Scholar] [CrossRef]
- Jin, Z.; Chen, D.; Yin, L.; Hu, Y.; Zhu, H.; Hong, L. Molten waste plastic pyrolysis in a vertical falling film reactor and the influence of temperature on the pyrolysis products. Chin. J. Chem. Eng. 2018, 26, 400–406. [Google Scholar] [CrossRef]
- Liu, X.; Li, X.; Liu, J.; Wang, Z.; Kong, B.; Gong, X.; Yang, X.; Lin, W.; Guo, L. Study of high density polyethylene (HDPE) pyrolysis with reactive molecular dynamics. Polym. Degrad. Stab. 2014, 104, 62–70. [Google Scholar] [CrossRef]
- Park, K.-B.; Jeong, Y.-S.; Guzelciftci, B.; Kim, J.-S. Characteristics of a new type continuous two-stage pyrolysis of waste polyethylene. Energy 2018, 166, 343–351. [Google Scholar] [CrossRef]
- Muhammad, C.; Onwudili, J.A.; Williams, P.T. Thermal Degradation of Real-World Waste Plastics and Simulated Mixed Plastics in a Two-Stage Pyrolysis–Catalysis Reactor for Fuel Production. Energy Fuels 2015, 29, 2601–2609. [Google Scholar] [CrossRef]
- Miandad, R.; Barakat, M.; Aburiazaiza, A.S.; Rehan, M.; Nizami, A. Catalytic pyrolysis of plastic waste: A review. Process Saf. Environ. Prot. 2016, 102, 822–838. [Google Scholar] [CrossRef]
- Miandad, R.; Barakat, M.; Aburiazaiza, A.S.; Rehan, M.; Ismail, I.; Nizami, A. Effect of plastic waste types on pyrolysis liquid oil. Int. Biodeterior. Biodegrad. 2017, 119, 239–252. [Google Scholar] [CrossRef]
- Williams, P.; Slaney, E. Analysis of products from the pyrolysis and liquefaction of single plastics and waste plastic mixtures. Resour. Conserv. Recycl. 2007, 51, 754–769. [Google Scholar] [CrossRef]
- Sharuddin, S.D.A.; Abnisa, F.; Daud, W.M.A.W.; Aroua, M.K. Energy recovery from pyrolysis of plastic waste: Study on non-recycled plastics (NRP) data as the real measure of plastic waste. Energy Convers. Manag. 2017, 148, 925–934. [Google Scholar] [CrossRef]
- Das, P.; Tiwari, P. Valorization of packaging plastic waste by slow pyrolysis. Resour. Conserv. Recycl. 2018, 128, 69–77. [Google Scholar] [CrossRef]
- Miskolczi, N.; Angyal, A.; Bartha, L.; Valkai, I. Fuels by pyrolysis of waste plastics from agricultural and packaging sectors in a pilot scale reactor. Fuel Process. Technol. 2009, 90, 1032–1040. [Google Scholar] [CrossRef]
- Williams, E.A.; Williams, P.T. The pyrolysis of individual plastics and a plastic mixture in a fixed bed reactor. J. Chem. Technol. Biotechnol. 1997, 70, 9–20. [Google Scholar] [CrossRef]


| Parameter | Result | Limit | Unit |
|---|---|---|---|
| Kin. Viscosity (50 °C) | 1.848 | Min. 1.4; max. 6 | mm2/s |
| Density (15 °C) | 802.9 | Max. 890 | kg/m3 |
| Cetane index | 61.3 | Min. 40 | - |
| 10% (V/V) recovery | 178.3 | - | °C |
| 50% (V/V) recovery | 257.1 | - | °C |
| 90% (V/V) recovery | 347.0 | - | °C |
| Sulfur content | 196 | Max. 1000 | ppm |
| Flash point | 58.0 | Min. 43 | °C |
| Hydrogen sulfide | <2 | Max. 2 | ppm |
| Acid value | 0.136 | Max. 0.5 | mg KOH/g |
| Sediment content | 0.02 | - | % (m/m) |
| Carbon residue | <0.10 | Max. 0.3 | % (m/m) |
| Pour point (winter quality) | −6 | Max. −6 | °C |
| Pour point (summer quality) | −6 | Max. 0 | °C |
| Water content | 0.01 | - | % (V/V) |
| Ash content (775 °C) | <0.001 | Max. 0.01 | % (m/m) |
| Cloud point | 14 | °C | |
| HFRR (Lubricity at 60 °C) | 240 | Max. 520 | µm |
| Oxidation stability | 15.46 | - | min |
| Analyte | Analyte Code | RI Lit | RI Calc | CAS | Average % Area MGO (comm) | Average % Area VN (ml-py) | Average % Area MGO (ml-py) | Average % Area RPO (ml-py) |
|---|---|---|---|---|---|---|---|---|
| 1-propene-2-methyl | 394 | 399 | 115-11-7 | 0.04 | 0.05 | 0.14 | ||
| 2-methylbutane | C5 | 474 | 480 | 78-78-4 | 0.02 | |||
| 1-pentene | C5 | 478 | 483 | 109-67-1 | 0.11 | |||
| pentane | C5 | 500 | 499 | 109-66-0 | 0.03 | 1.14 | 0.16 | 0.37 |
| 2-methyl-1,3-butanediene (isoprene) | C5 | 520 | 515 | 78-79-5 | 0.11 | 0.18 | 0.12 | |
| 2-methyl-2-butene | C5 | 525 | 520 | 513-35-9 | 0.12 | 0.1 | ||
| 1,3-pentadiene | C5 | 542 | 538 | 504-60-9 | 0.04 | |||
| 1,3-Cyclopentadiene | C5 | 540 | 542 | 542-92-7 | 0.04 | |||
| cyclopentene | C5 | 552 | 556 | 142-29-0 | 0.14 | 0.06 | ||
| 2-methylpentane | C6 | 560 | 563 | 107-83-5 | 0.06 | 0.98 | 0.06 | 0.17 |
| 3-methylpentane | C6 | 580 | 580 | 96-14-0 | 0.05 | 0.07 | 0.04 | |
| 1-hexene | C6 | 588 | 588 | 592-41-6 | 0.11 | 2.87 | 0.45 | 0.87 |
| hexane | C6 | 600 | 599 | 110-54-3 | 0.04 | 0.72 | 0.13 | 0.32 |
| 2-methyl-2-pentene, | C6 | 606 | 602 | 625-27-4 | 0.03 | 0.64 | 0.1 | 0.22 |
| 3-methylcyclopentene | C6 | 612 | 612 | 592-48-3 | 0.09 | 0.03 | ||
| 2-hexene, (Z)- | C6 | 617 | 615 | 7688-21-3 | 0.12 | 0.05 | ||
| 3-methyl-2-pentene, (E)- | C6 | 620 | 621 | 616-12-6 | 0.06 | 0.03 | ||
| methylcyclopentane | C6 | 627 | 628 | 96-37-7 | 0.04 | 0.31 | 0.11 | |
| 2,4-hexadiene, (E,Z)- | C6 | 636 | 637 | 5194-50-3 | 0.2 | 0.09 | ||
| 3-methyl-1,3-Pentadiene, (Z) | C6 | 640 | 642 | 2787-45-3 | 0.03 | 0.23 | 0.05 | 0.07 |
| 2,4-dimethyl-1-pentene | C7 | 642 | 646 | 2213-32-3 | 0.82 | 0.06 | 0.11 | |
| 1-methylcyclopentene | C6 | 647 | 648 | 693-89-0 | 0.39 | 0.04 | 0.15 | |
| benzene | Benz | 663 | 663 | 71-43-2 | 0.05 | 0.7 | 0.25 | 1.08 |
| 2-methylhexane | C7 | 667 | 667 | 591-76-4 | 0.04 | 0.17 | ||
| 3-methylhexane | C7 | 677 | 676 | 589-34-4 | 0.03 | 0.13 | 0.03 | |
| cyclohexene | C6 | 678 | 680 | 110-83-8 | 0.28 | 0.05 | 0.1 | |
| 2-methyl-1-hexene | C7 | 683 | 688 | 6094-02-6 | 0.33 | 0.22 | 0.09 | |
| 1-heptene | C7 | 692 | 692 | 592-76-7 | 0.09 | 1.99 | 0.16 | 0.64 |
| heptane | C7 | 700 | 702 | 142-82-5 | 0.13 | 1.8 | 0.34 | 0.59 |
| 2-methyl-2-hexene | C7 | 702 | 705 | 2738-19-4 | 0.51 | 0.1 | 0.14 | |
| 2-heptene, (E)- | C7 | 705 | 707 | 7642-10-6 | 0.23 | 0.05 | 0.09 | |
| 2-heptene, (z)- | C7 | 714 | 716 | 592-77-8 | 0.13 | 0.04 | 0.07 | |
| 3-methylcyclohexene | C7 | 728 | 726 | 591-48-0 | 0.13 | 0.04 | ||
| methylcyclohexane | C7 | 732 | 729 | 108-87-2 | 0.12 | 0.48 | 0.13 | 0.14 |
| ethylcyclopentane | C7 | 739 | 737 | 1640-89-7 | 0.04 | 0.23 | 0.09 | |
| 4-methylcyclohexene | C7 | 742 | 742 | 591-47-9 | 0.28 | 0.06 | 0.08 | |
| methylenecyclohexane | C7 | 745 | 746 | 1192-37-6 | 0.16 | |||
| 2,4-Heptadiene | C7 | 746 | 749 | 628-72-8 | 0.19 | 0.06 | ||
| 1-ethylcyclopentene | C7 | 747 | 751 | 2146-38-5 | 0.22 | 0.05 | 0.08 | |
| 4-methyl-1-heptene | C8 | 748 | 755 | 13151-05-8 | 0.59 | 0.1 | ||
| 4-methyl-2-heptene | C8 | 751 | 754 | 66225-17-0 | 0.38 | 0.08 | ||
| Toluene | Tol | 756 | 759 | 108-88-3 | 0.19 | 1.54 | 0.51 | 1.38 |
| 4-methylheptane | C8 | 768 | 770 | 589-53-7 | 0.15 | 2.3 | 0.55 | 0.45 |
| cis-1,3-dimethylcyclohexane | C8 | 774 | 772 | 638-04-0 | 0.07 | |||
| 2-methyl-1-heptene | C8 | 782 | 784 | 15870-10-7 | 0.4 | 0.07 | 0.11 | |
| 3-methyleneheptane 2-Ethyl-1-hexene | C8 | 785 | 789 | 1632-16-2 | 0.37 | 0.07 | 0.15 | |
| 1-octene | C8 | 790 | 798 | 111-66-0 | 0.1 | 2.2 | 0.57 | 0.68 |
| octane | C8 | 800 | 804 | 111-65-9 | 0.32 | 2.22 | 0.59 | 0.69 |
| 3-octene | C8 | 814 | 814 | 14919-01-8 | 0.03 | 0.21 | 0.08 | |
| 2-octene | C8 | 818 | 820 | 13389-42-9 | 0.14 | |||
| 3-octyne | C8 | 820 | 825 | 15232-76-5 | 0.2 | |||
| 2,4-dimethylheptane | C9 | 822 | 828 | 2213-23-2 | 0.9 | 0.2 | 0.15 | |
| (1α,3α,5α)-1,3,5-trimethylcyclohexane | C9 | 831 | 832 | 1795-27-3 | 0.05 | 0.81 | 0.21 | 0.15 |
| ethylcyclohexane | C8 | 832 | 838 | 1678-91-7 | 0.17 | 0.4 | 0.08 | 0.1 |
| propylcyclopentane | C8 | 833 | 842 | 1678-92-8 | 0.09 | 1.67 | 0.46 | 0.32 |
| 2,4-dimethyl-1-heptene | C9 | 842 | 849 | 19549-87-2 | 16.51 | 4.26 | 2.74 | |
| (1α,3α,5β)- 1,3,5-trimethylcyclohexane | C9 | 850 | 855 | 1795-26-2 | 0.07 | 1.21 | 0.4 | 0.25 |
| ethylbenzene | Etb | 857 | 561 | 100-41-4 | 0.12 | 1.51 | 0.63 | 2.1 |
| m-xylene | X | 862 | 869 | 108-38-3 | 0.38 | 0.58 | 0.34 | 0.18 |
| 1,8-nonadiene | C9 | 879 | 880 | 4900-30-5 | 0.25 | 0.09 | 0.04 | |
| styrene | STY | 889 | 885 | 100-42-5 | 0.05 | 1.11 | 0.57 | 2.28 |
| o-xylene | X | 891 | 889 | 95-47-6 | 0.12 | 0.46 | 0.19 | 0.16 |
| 1-nonene | C9 | 893 | 891 | 124-11-8 | 0.1 | 2.05 | 0.82 | 0.68 |
| (E)-2-Nonene | C9 | 897 | 893 | 6434-78-2 | 0.11 | 0.05 | 0.05 | |
| nonane | C9 | 900 | 897 | 111-84-2 | 0.72 | 1.83 | 0.81 | 0.67 |
| (Z)-2-Nonene | C9 | 911 | 910 | 6434-77-1 | 0.27 | 0.11 | 0.07 | |
| (1-methylethyl)benzene | AR | 920 | 922 | 98-82-8 | 0.24 | 0.14 | 0.27 | |
| propylcyclohexane | C9 | 923 | 924 | 1678-92-8 | 0.16 | 0.11 | 0.06 | 0.06 |
| butylcyclopentane | C9 | 936 | 938 | 2040-95-1 | 0.12 | 0.09 | 0.05 | 0.03 |
| 2,6-dimethyloctane | C10 | 937 | 940 | 2051-30-1 | 0.24 | |||
| propylbenzene | AR | 955 | 949 | 103-65-1 | 0.05 | 0.12 | 0.08 | 0.11 |
| 1-ethyl-3-methylbenzene | AR | 958 | 951 | 620-14-4 | 0.16 | 0.16 | 0.13 | 0.07 |
| 1-ethyl-4-methylbenzene | AR | 960 | 959 | 622-96-8 | 0.09 | 0.1 | ||
| 4-methylnonane | C10 | 961 | 962 | 17301-94-9 | 0.22 | 0.23 | 0.12 | 0.07 |
| 1,3,5-trimethylbenzene (mesitylene) | AR | 964 | 965 | 108-67-8 | 0.26 | 0.19 | 0.12 | |
| 2-methylnonane | C10 | 965 | 966 | 871-83-0 | 0.36 | |||
| 3-methylnonane | C10 | 970 | 970 | 5911-04-6 | 0.29 | |||
| 1-ethyl-2-methylbenzene | AR | 976 | 972 | 611-14-3 | 0.18 | 0.23 | 0.15 | 0.1 |
| alfa methylstyrene | AR | 978 | 975 | 98-83-9 | 0.17 | 0.1 | 0.41 | |
| benzonitrile | BN | 982 | 989 | 100-47-0 | 0.18 | 0.1 | 0.08 | |
| 2-methyl-1-nonene | C10 | 983 | 990 | 2980-71-4 | 0.26 | 0.12 | 0.09 | |
| 1,2,4-trimethylbenzene | AR | 987 | 993 | 95-63-6 | 0.37 | 0.16 | 0.19 | 0.12 |
| 1-decene | C10 | 993 | 998 | 872-05-9 | 0.22 | 2.07 | 1.37 | 0.97 |
| decane | C10 | 1000 | 1004 | 124-18-5 | 1.75 | 1.45 | 1.04 | 0.79 |
| (1-methylpropyl)benzene | AR | 1006 | 1011 | 135-98-8 | 0.07 | |||
| (Z)-2-decene | C10 | 1011 | 1017 | 20348-51-0 | 0.24 | 0.16 | 0.12 | |
| 2,5-dimethylnonane | C11 | 1015 | 1019 | 17302-27-1 | 0.11 | 0.74 | 0.51 | 0.23 |
| 2,6-dimethylnonane | C11 | 1030 | 1029 | 17302-28-2 | 0.2 | 0.78 | 0.52 | 0.27 |
| 1,2,3-trimethylbenzene | AR | 1032 | 1032 | 526-73-8 | 0.33 | |||
| D-limonene | C10 | 1035 | 1038 | 5989-27-5 | 0.14 | 0.11 | 0.22 | |
| indane | I | 1042 | 1041 | 496-11-7 | 0.12 | 0.2 | 0.29 | 0.3 |
| indene | I | 1050 | 1049 | 95-13-6 | 0.15 | 0.11 | 0.07 | |
| butylbenzene | AR | 1055 | 1058 | 104-51-8 | 0.16 | 0.08 | 0.09 | 0.08 |
| 5-methyldecane | C11 | 1056 | 1060 | 13151-35-4 | 0.37 | 0.35 | 0.34 | 0.17 |
| 4-methyldecane | C11 | 1060 | 1062 | 2847-72-5 | 0.35 | |||
| 2-methyldecane | C11 | 1063 | 1065 | 6975-98-0 | 0.53 | |||
| acetophenone | AcPh | 1066 | 1066 | 98-86-2 | 0.19 | 0.5 | 0.51 | |
| 3-methyldecane | C11 | 1069 | 1069 | 13151-34-3 | 0.41 | |||
| 2-ethyl-1,4-dimethylbenzene | AR | 1074 | 1074 | 1758-88-9 | 0.17 | 0.07 | ||
| 1-ethyl-1,2-dimethylbenzene | AR | 1082 | 1078 | 934-80-5 | 0.22 | 0.1 | ||
| 2-ethyl-1,3-dimethylbenzene | AR | 1087 | 1085 | 2870-04-4 | 0.48 | |||
| 1-undecene | C11 | 1090 | 1087 | 821-95-4 | 0.15 | 1.48 | 1.63 | 1.08 |
| 1-methyl-4-(1-methylpropyl)benzene | AR | 1092 | 1089 | 1595-16-0 | 0.24 | |||
| undecane | C11 | 1100 | 1097 | 1120-21-4 | 2.4 | 1.07 | 1.25 | 0.99 |
| (E)-2-Undecene | C11 | 1104 | 1110 | 821-98-7 | 0.19 | 0.23 | 0.14 | |
| (Z)-2-undecene | C11 | 1110 | 1115 | 821-96-5 | 0.15 | 0.09 | ||
| decahydro-2-methylnaphthalene (trans-2-methyldecalin) | C11 | 1115 | 1117 | 2958-76-1 | 0.36 | |||
| 1,2,4,5-tetramethylbenzene | AR | 1116 | 1118 | 95-93-2 | 0.27 | 0.25 | 0.09 | |
| 1,2,3,5-tetramethylbenzene | AR | 1119 | 1120 | 527-53-7 | 0.44 | 0.2 | ||
| decahydro-1-methylnaphthalene (1-methyldecalin) | C11 | - | 1125 | 2958-75-0 | 0.31 | |||
| 3,7-dimethyldecane | C12 | 1127 | 1129 | 17312-54-8 | 0.31 | |||
| pentylcyclohexane | C11 | 1134 | 1137 | 4292-92-6 | 0.36 | |||
| hexylcyclopentane | C11 | 1136 | 1139 | 4457-00-5 | 0.33 | |||
| 2,3-dihydro-4-methyl-1H-Indene | I | 1147 | 1150 | 824-22-6 | 0.29 | 0.16 | 0.26 | 0.15 |
| pentylbenzene | AR | 1155 | 1152 | 538-68-1 | 0.41 | |||
| 1,2,3,4-tetrahydronaphthalene | N | 1162 | 1161 | 119-64-2 | 0.52 | |||
| 2-methylundecane | C12 | 1167 | 1170 | 7045-71-8 | 0.73 | 0.13 | 0.14 | |
| 3-methylundecane | C12 | 1171 | 1173 | 1002-43-3 | 0.42 | 0.06 | 0.08 | |
| naphthalene | N | 1182 | 1189 | 91-20-3 | 0.15 | 0.18 | 0.4 | 0.24 |
| 1-dodecene | C12 | 1193 | 1190 | 112-41-4 | 0.22 | 0.91 | 1.59 | 1.18 |
| dodecane | C12 | 1200 | 1205 | 112-40-3 | 2.36 | 0.82 | 1.46 | 1.23 |
| (E)-2-dodecene | C12 | 1201 | 1208 | 7206-13-5 | 0.12 | 0.21 | 0.14 | |
| (Z)-2-dodecene | C12 | 1212 | 1216 | 7206-26-0 | 0.07 | 0.14 | 0.11 | |
| 2,6-dimethylundecane | C13 | 1213 | 1215 | 17301-23-4 | 0.55 | |||
| 1,2,3,4-tetrahydro-2-methylnaphthalene | N | - | 1218 | 3877-19-8 | 0.25 | |||
| 2-butyl-1,1,3-trimethylcyclohexane | C13 | 1219 | 1222 | 54676-39-0 | 0.36 | |||
| 2,8-dimethylundecane | C13 | 1221 | 1225 | 17301-25-6 | 0.11 | 0.26 | 0.14 | |
| 6-methyldodecane | C13 | 1251 | 1250 | 6044-71-9 | 0.75 | 0.18 | 0.42 | 0.18 |
| hexylbenzene | AR | 1255 | 1253 | 1077-16-3 | 0.04 | 0.18 | 0.09 | |
| 4-methyldodecane | C13 | 1257 | 1254 | 6117-97-1 | 0.29 | |||
| 2-methyldodecane | C13 | 1263 | 1258 | 1560-97-0 | 0.68 | |||
| 1,2,3,4-tetrahydro-5-methylnaphthalene | N | 1279 | 1276 | 2809-64-5 | 0.44 | |||
| 1-tridecene | C13 | 1292 | 1291 | 2437-56-1 | 0.41 | 0.61 | 1.71 | 1.28 |
| tridecane | C13 | 1300 | 1301 | 629-50-5 | 2.65 | 0.67 | 1.62 | 1.65 |
| 1-methylnaphthalene | N | 1306 | 1303 | 90-12-0 | 0.21 | 0.32 | 0.95 | 0.54 |
| 4-methyltridecane | C14 | 1360 | 1357 | 26730-12-1 | 0.34 | 0.1 | ||
| 3-methyltridecane | C14 | 1369 | 1370 | 6418-41-3 | 0.54 | 0.07 | 0.05 | |
| 2,6,10-trimethyldodecane | C15 | 1370 | 1373 | 3891-98-3 | 0.32 | |||
| biphenyl | BY | 1373 | 1376 | 92-52-4 | 0.16 | 0.12 | 0.49 | 0.39 |
| 1-tetradecene | C14 | 1393 | 1393 | 1120-36-1 | 0.52 | 0.37 | 1.6 | 1.26 |
| tetradecane | C14 | 1400 | 1402 | 629-59-4 | 2.28 | 0.38 | 1.6 | 1.52 |
| (E)-2-tetradecene | C14 | 1406 | 1407 | 35953-53-8 | 0.09 | 0.4 | 0.17 | |
| 2,6-dimethylnaphthalene | N | 1409 | 1412 | 581-42-0 | 0.49 | |||
| 1,3-dimethylnaphthalene | N | 1424 | 1428 | 575-41-7 | 0.36 | 0.1 | 0.31 | |
| 1,5-dimethylnaphthalene | N | 1446 | 1442 | 571-61-9 | 0.65 | |||
| 1,8-dimethylnaphthalene | N | 1459 | 1465 | 569-41-5 | 0.21 | |||
| 3-methyltetradecane | C14 | 1472 | 1476 | 18435-22-8 | 0.65 | |||
| 1-pentadecene | C15 | 1493 | 1491 | 13360-61-7 | 0.56 | 0.25 | 1.54 | 1.47 |
| pentadecane | C15 | 1500 | 1507 | 629-62-9 | 2.04 | 0.3 | 1.67 | 1.53 |
| 2,3,6-trimethylnaphthalene | N | 1550 | 1545 | 829-26-5 | 0.37 | |||
| 1,6,7-trimethylnaphthalene | N | 1572 | 1563 | 2245-38-7 | 0.22 | |||
| 1-hexadecene | C16 | 1593 | 1592 | 629-73-2 | 0.43 | 0.15 | 1.26 | 1.05 |
| hexadecane | C16 | 1600 | 1603 | 544-76-3 | 1.47 | 0.22 | 1.83 | 1.55 |
| 1,1’-(1,3-propanediyl)bis-benzene | AR | 1633 | 1640 | 1081-75-0 | 0.31 | 0.61 | ||
| 1-heptadecene | C17 | 1693 | 1692 | 6765-39-5 | 0.28 | 1.02 | 0.9 | |
| heptadecane | C17 | 1700 | 1706 | 629-78-7 | 1.26 | 0.15 | 1.52 | 1.42 |
| 1-octadecene | C18 | 1793 | 1794 | 112-88-9 | 0.31 | 0.06 | 0.78 | 1.02 |
| octadecane | C18 | 1800 | 1806 | 593-45-3 | 1.09 | 0.11 | 1.29 | 1.24 |
| 1-nonadecene | C19 | 1893 | 1893 | 18435-45-5 | 0.43 | 0.03 | 0.63 | 1.24 |
| nonadecane | C19 | 1900 | 1902 | 629-92-5 | 1.06 | 0.08 | 1.11 | 1.41 |
| 1-eicosene | C20 | 1993 | 1992 | 3452-07-1 | 0.16 | 0.43 | 0.54 | |
| eicosane | C20 | 2000 | 1999 | 112-95-8 | 1.05 | 0.06 | 1.2 | 1.53 |
| 1-heneicosene | C21 | 2093 | 2093 | 27400-79-9 | 0.15 | 0.26 | 0.34 | |
| heneicosane | C21 | 2100 | 2104 | 629-94-7 | 0.9 | 0.03 | 0.76 | 1.2 |
| pyrene | PHA | 2126 | 2128 | 129-00-0 | 0.17 | 0.07 | ||
| 1-docosene | C22 | 2193 | 2193 | 1599-67-3 | 0.09 | 0.19 | 0.41 | |
| docosane | C22 | 2200 | 2202 | 629-97-0 | 0.6 | 0.02 | 0.55 | 0.94 |
| 1-tricosene | C23 | 2293 | 2295 | 0.07 | 0.12 | 0.15 | ||
| tricosane | C23 | 2300 | 2301 | 638-67-5 | 0.49 | 0.39 | 0.69 | |
| tetracosane | C24 | 2400 | 2404 | 646-31-1 | 0.32 | 0.53 | 0.7 | |
| pentacosane | C25 | 2500 | 2503 | 629-99-2 | 0.24 | 0.3 | 0.4 | |
| hexacosane | C26 | 2600 | 2601 | 630-01-3 | 0.13 | 0.29 | 0.34 | |
| heptacosane | C27 | 2700 | 2702 | 0.19 | 0.14 | |||
| Octacosane | C28 | 2800 | 2800 | 630-02-4 | 0.19 | 0.13 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Faussone, G.C.; Cecchi, T. Chemical Recycling of Plastic Marine Litter: First Analytical Characterization of The Pyrolysis Oil and of Its Fractions and Comparison with a Commercial Marine Gasoil. Sustainability 2022, 14, 1235. https://doi.org/10.3390/su14031235
Faussone GC, Cecchi T. Chemical Recycling of Plastic Marine Litter: First Analytical Characterization of The Pyrolysis Oil and of Its Fractions and Comparison with a Commercial Marine Gasoil. Sustainability. 2022; 14(3):1235. https://doi.org/10.3390/su14031235
Chicago/Turabian StyleFaussone, Gian Claudio, and Teresa Cecchi. 2022. "Chemical Recycling of Plastic Marine Litter: First Analytical Characterization of The Pyrolysis Oil and of Its Fractions and Comparison with a Commercial Marine Gasoil" Sustainability 14, no. 3: 1235. https://doi.org/10.3390/su14031235
APA StyleFaussone, G. C., & Cecchi, T. (2022). Chemical Recycling of Plastic Marine Litter: First Analytical Characterization of The Pyrolysis Oil and of Its Fractions and Comparison with a Commercial Marine Gasoil. Sustainability, 14(3), 1235. https://doi.org/10.3390/su14031235






