Effect of Inoculum Concentration on the Degradation of Diesel 2 by a Microbial Consortium
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Material
2.2. Methods
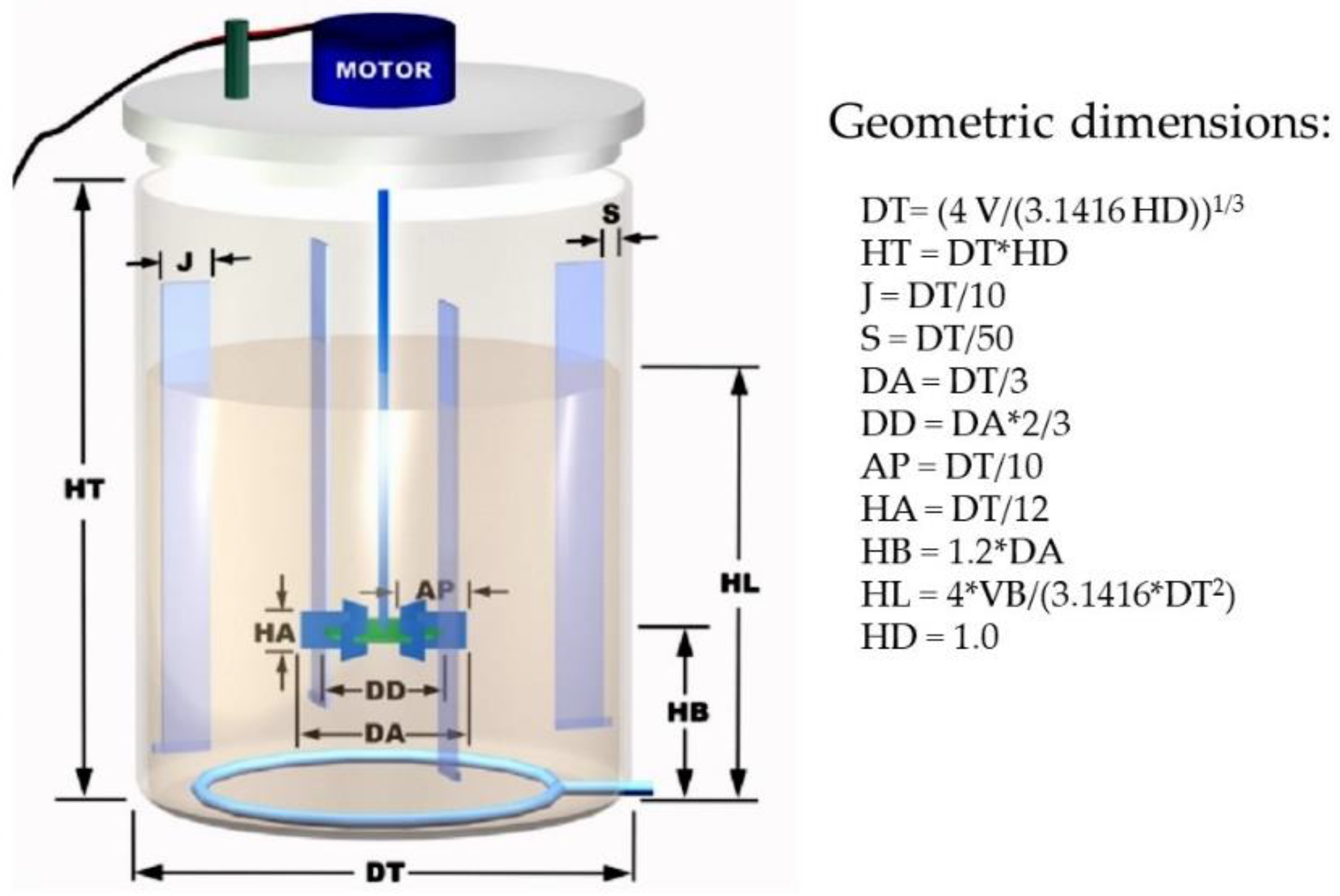
2.2.1. Inoculum Preparation
2.2.2. Preparation of Evaluation Systems
2.2.3. Indirect Determination of Degradation of Diesel 2
2.2.4. Determination of Bioprocess Efficiency
- MOc = Diesel 2 consumed (% oil v/v)
- MOi = Initial concentration of Diesel 2 (% oil v/v)
- MOf = Final concentration of Diesel 2 (% of oil v/v)
- * Diesel 2 concentrations were determined by the Gerber method for total fats
2.3. Analysis of Data
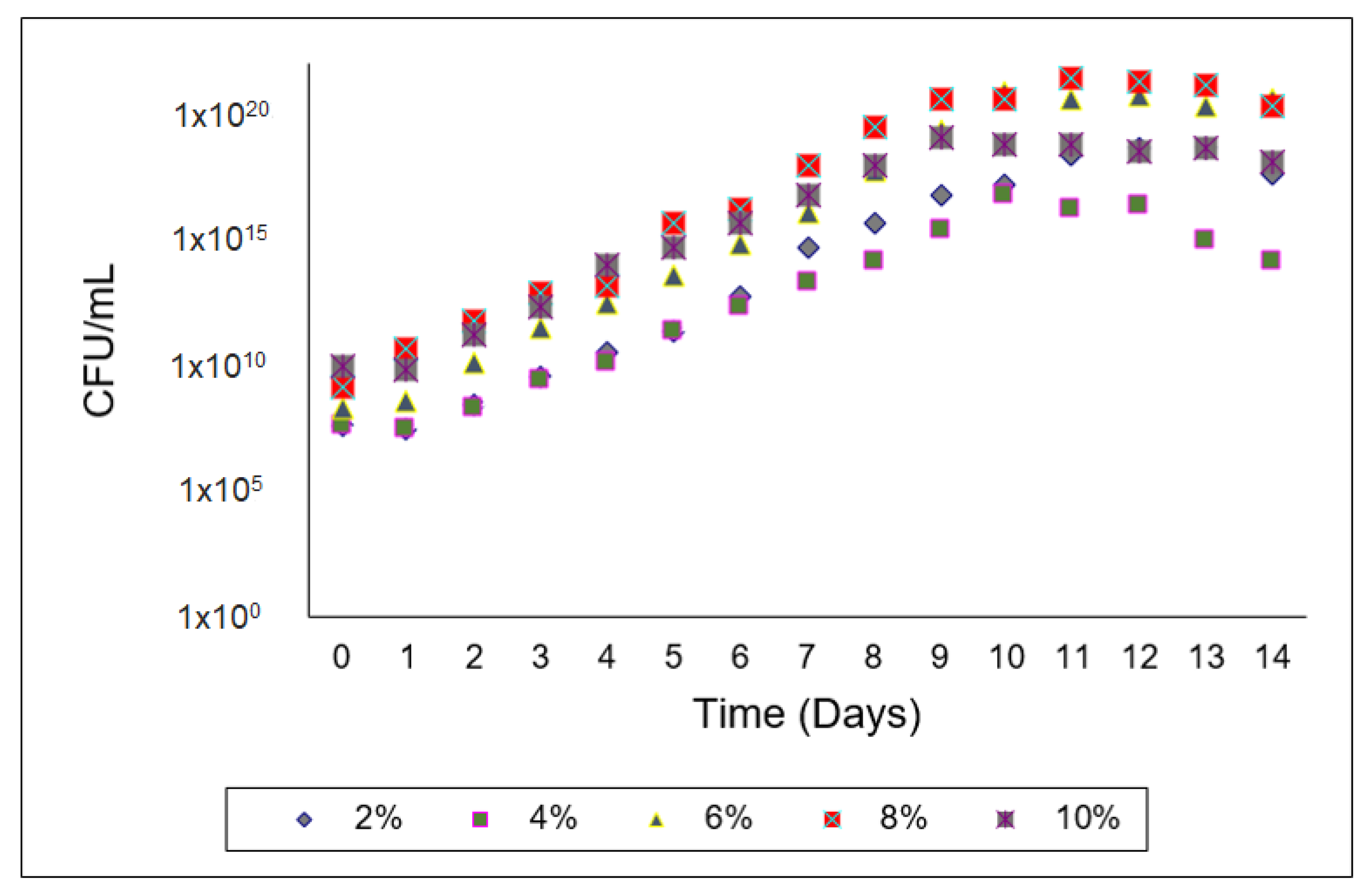
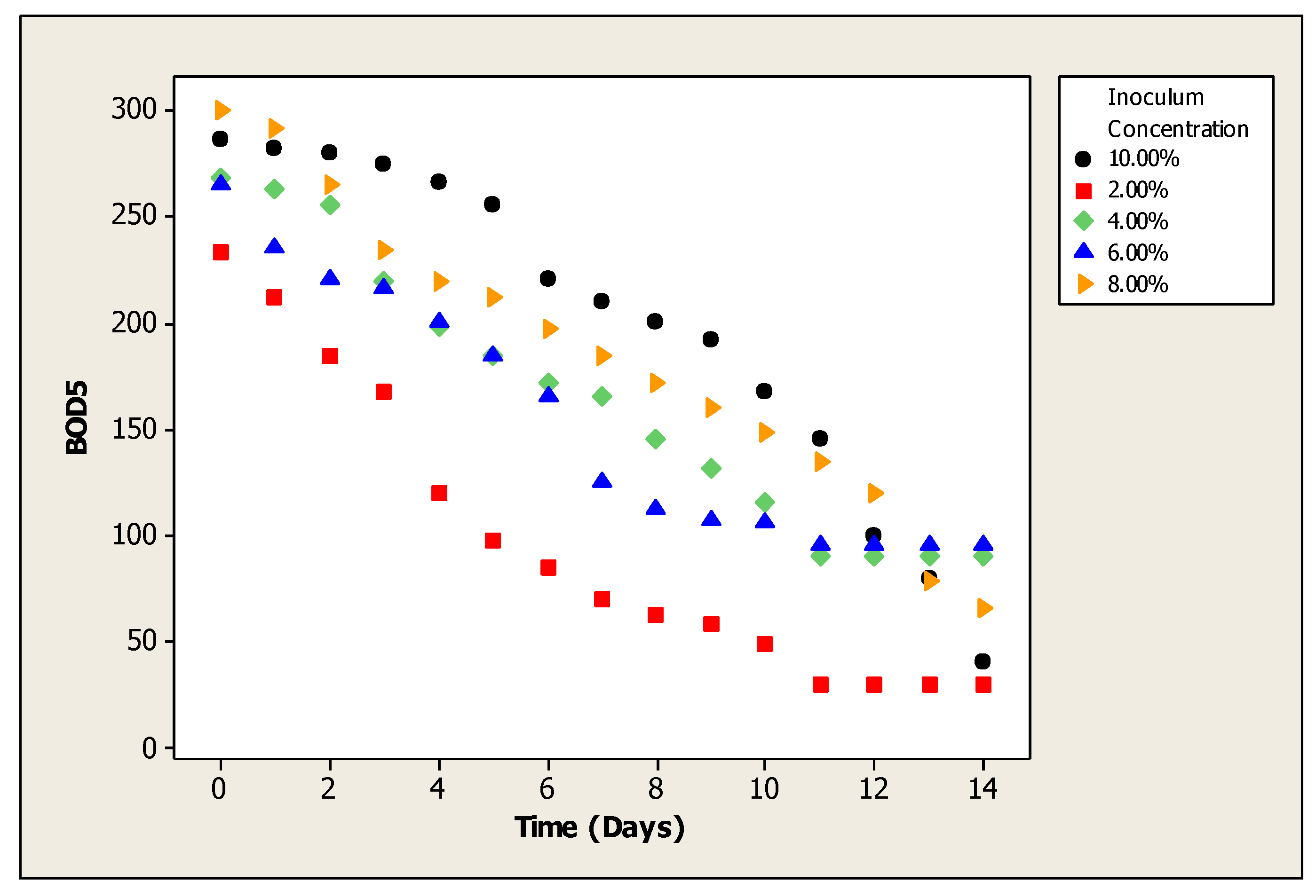
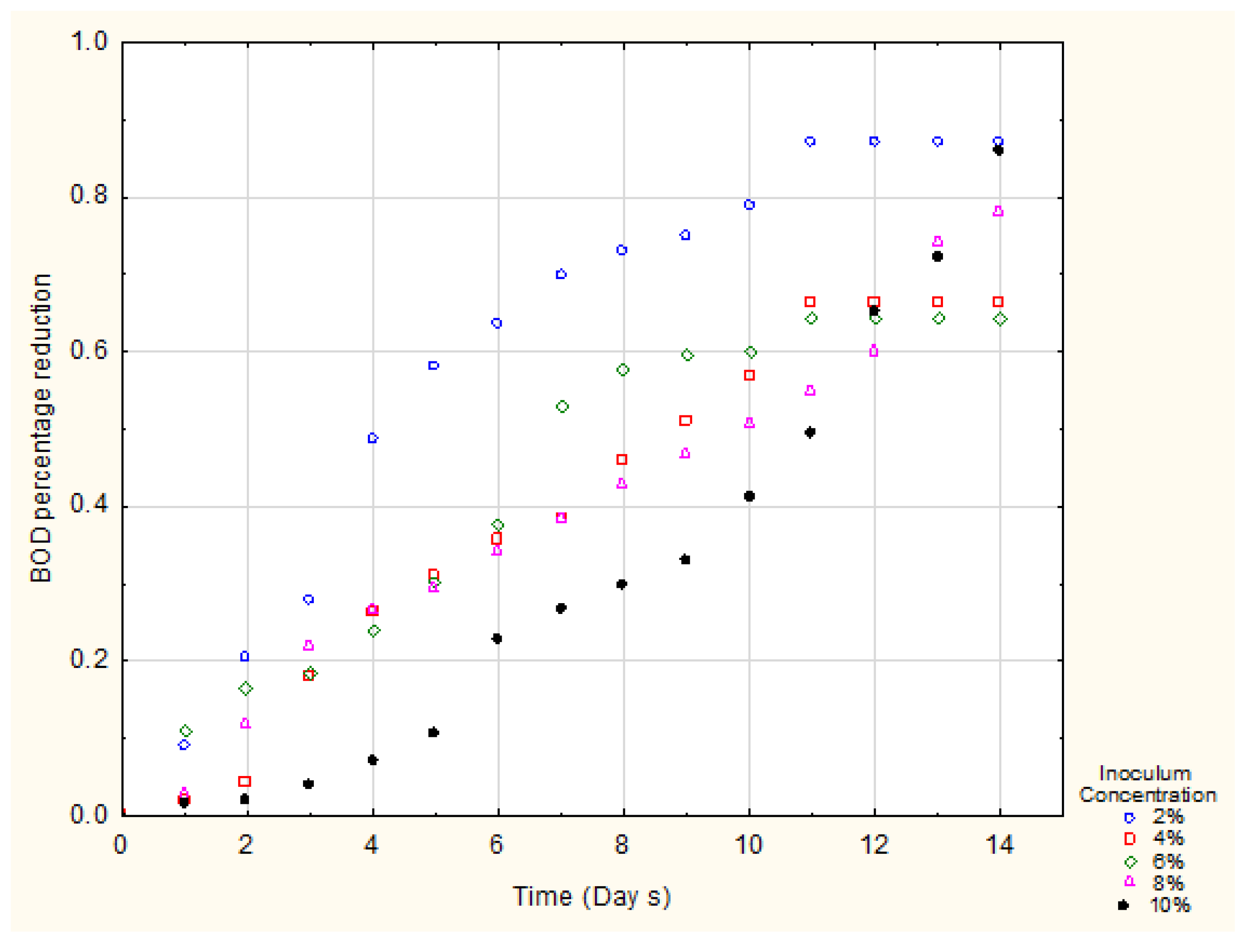
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Yang, Y.; Zhang, Z.-W.; Liu, R.-X.; Ju, H.-Y.; Bian, X.-K.; Zhang, W.-Z.; Zhang, C.-B.; Yang, T.; Guo, B.; Xiao, C.-L.; et al. Research Progress in Bioremediation of Petroleum Pollution. Environ. Sci. Pollut. Res. Int. 2021, 28, 46877–46893. [Google Scholar] [CrossRef] [PubMed]
- Plan Energético Nacional 2014–2025. Available online: https://www.minem.gob.pe/_detalle.php?idSector=10&idTitular=6397 (accessed on 8 October 2022).
- Khalid, F.E.; Lim, Z.S.; Sabri, S.; Gomez-Fuentes, C.; Zulkharnain, A.; Ahmad, S.A. Bioremediation of Diesel Contaminated Marine Water by Bacteria: A Review and Bibliometric Analysis. J. Mar. Sci. Eng. 2021, 9, 155. [Google Scholar] [CrossRef]
- Fraser, B. Oil in the Forest. Science 2016, 353, 641–643. [Google Scholar] [CrossRef] [PubMed]
- Yusta-García, R.; Orta-Martínez, M.; Mayor, P.; González-Crespo, C.; Rosell-Melé, A. Water Contamination from Oil Extraction Activities in Northern Peruvian Amazonian Rivers. Environ. Pollut. 2017, 225, 370–380. [Google Scholar] [CrossRef] [PubMed]
- Jernelöv, A. The Threats from Oil Spills: Now, Then, and in the Future. Ambio 2010, 39, 353–366. [Google Scholar] [CrossRef]
- Grados, C.; Pacheco, E. El Impacto de La Actividad Extractiva Petrolera En El Acceso al Agua: El Caso de Dos Comunidades Kukama Kukamiria de La Cuenca Del Marañón (Loreto, Perú). Anthropológica 2016, 34, 33–59. [Google Scholar] [CrossRef]
- Capurro, V.P.; Martínez, J.C.; Bustamante, C.A.; Carhuaz, E.O. Environmental damage to the Peruvian marine littoral caused by the oil spill (January 2022) at the La Pampilla refinery. Manglar 2022, 19, 67–75. [Google Scholar] [CrossRef]
- Vasquez-Velásquez, C.; Ordóñez-Aquino, C.; Gonzales, G.F. Derrame de Petróleo y Sus Efectos Sobre La Salud. Acta Médica Peru 2022, 39, 96–98. [Google Scholar] [CrossRef]
- Mega, E.R. Unprecedented Oil Spill Catches Researchers in Peru off Guard. Nature 2022. [Google Scholar] [CrossRef]
- Stepanova, A.Y.; Gladkov, E.A.; Osipova, E.S.; Gladkova, O.V.; Tereshonok, D.V. Bioremediation of Soil from Petroleum Contamination. Processes 2022, 10, 1224. [Google Scholar] [CrossRef]
- Tiralerdpanich, P.; Sonthiphand, P.; Luepromchai, E.; Pinyakong, O.; Pokethitiyook, P. Potential Microbial Consortium Involved in the Biodegradation of Diesel, Hexadecane and Phenanthrene in Mangrove Sediment Explored by Metagenomics Analysis. Mar. Pollut. Bull. 2018, 133, 595–605. [Google Scholar] [CrossRef] [PubMed]
- Leahy, J.G.; Colwell, R.R. Microbial Degradation of Hydrocarbons in the Environment. Microbiol. Rev. 1990, 54, 305–315. [Google Scholar] [CrossRef] [PubMed]
- Xu, X.; Liu, W.; Tian, S.; Wang, W.; Qi, Q.; Jiang, P.; Gao, X.; Li, F.; Li, H.; Yu, H. Petroleum Hydrocarbon-Degrading Bacteria for the Remediation of Oil Pollution under Aerobic Conditions: A Perspective Analysis. Front. Microbiol. 2018, 9, 2885. [Google Scholar] [CrossRef] [PubMed]
- Abdel-Shafy, H.I.; Mansour, M.S.M. Microbial Degradation of Hydrocarbons in the Environment: An Overview. In Microbial Action on Hydrocarbons; Springer: Singapore, 2018; pp. 353–386. ISBN 9789811318399. [Google Scholar]
- Boopathy, R. Factors Limiting Bioremediation Technologies. Bioresour. Technol. 2000, 74, 63–67. [Google Scholar] [CrossRef]
- Bartha, R. Biotechnology of Petroleum Pollutant Biodegradation. Microb. Ecol. 1986, 12, 155–172. [Google Scholar] [CrossRef] [PubMed]
- Cervantes, F.J.; Dijksma, W.; Duong-Dac, T.; Ivanova, A.; Lettinga, G.; Field, J.A. Anaerobic Mineralization of Toluene by Enriched Sediments with Quinones and Humus as Terminal Electron Acceptors. Appl. Environ. Microbiol. 2001, 67, 4471–4478. [Google Scholar] [CrossRef]
- Carless, H.A.J. ChemInform Abstract: The Use of Cyclohexa-3,5-Diene-1,2-Diols in Enantiospecific Synthesis. ChemInform 2010, 23, 795–826. [Google Scholar] [CrossRef]
- Ahmad, J. Bioremediation of Petroleum Sludge Using Effective Microorganism (EM) Technology. Pet. Sci. Technol. 2017, 35, 1515–1522. [Google Scholar] [CrossRef]
- Elumalai, P.; Parthipan, P.; AlSalhi, M.S.; Huang, M.; Devanesan, S.; Karthikeyan, O.P.; Kim, W.; Rajasekar, A. Characterization of Crude Oil Degrading Bacterial Communities and Their Impact on Biofilm Formation. Environ. Pollut. 2021, 286, 117556. [Google Scholar] [CrossRef]
- Ebadi, K.; Zarei, M.; Sanati, A.M. Optimization of Crude Oil Biodegradation by Brevibacterium sp. Isolated from the Native Sponges of the Persian Gulf. Iran. J. Biotechnol. 2021, 19, e2690. [Google Scholar] [CrossRef]
- Oba, A.; John, B.; Garba, J.; John, K.V.; Allamin, I.A.; Hambali, I.U.; Musa, J.A. Hydrocarbon Degradation Competence of Bacterial Consortium Isolated from Oil Polluted Soil in Azuabie Town, Port Harcourt, Nigeria. J. Biochem. Microbiol. Biotechnol. 2022, 10, 36–40. [Google Scholar] [CrossRef]
- Naloka, K.; Polrit, D.; Muangchinda, C.; Thoetkiattikul, H.; Pinyakong, O. Bioballs Carrying a Syntrophic Rhodococcus and Mycolicibacterium Consortium for Simultaneous Sorption and Biodegradation of Fuel Oil in Contaminated Freshwater. Chemosphere 2021, 282, 130973. [Google Scholar] [CrossRef] [PubMed]
- Cuzco, A.; Lorenzo, D. Aislamiento y Selección de un Cultivo Bacteriano Puro Degradador de Hidrocarburos a Partir de Botaderos de Residuos de Petróleo en Talara en Enero del 2016; Universidad Nacional de Trujillo: Trujillo, Peru, 2016. [Google Scholar]
- Okabe, S.; Kuroda, H.; Watanabe, Y. Significance of Biofilm Structure on Transport of Inert Particulates into Biofilms. Water Sci. Technol. 1998, 38, 163–170. [Google Scholar] [CrossRef]
- Otiniano-García, M. Eficiencia de Pseudomonas sp, Rhodopseudomonas sp sp, Micrococcus sp y Bacillus sp Empleados como Cultivos Individuales y en Consorcio, en la Degradación de Diesel II. Ph.D. Thesis, Universidad Nacional de Trujillo, Trujillo, Peru, 2009. [Google Scholar]
- McGrath, J.E.; Harfoot, C.G. Reductive Dehalogenation of Halocarboxylic Acids by the Phototrophic Genera Rhodospirillum and Rhodopseudomonas. Appl. Environ. Microbiol. 1997, 63, 3333–3335. [Google Scholar] [CrossRef]
- Li, X.; Li, H.; Qu, C. A Review of the Mechanism of Microbial Degradation of Petroleum Pollution. IOP Conf. Ser. Mater. Sci. Eng. 2019, 484, 012060. [Google Scholar] [CrossRef]
- Márquez-Rocha, F.J.; Hernández-Rodrí, V.; Lamela, M.T. Biodegradation of Diesel Oil in Soil by a Microbial Consortium. Water Air Soil Pollut. 2001, 128, 313–320. [Google Scholar] [CrossRef]
- Flores, G.P.; Argüello, G.B.; Howard, A.M.M. biodegradación de asfaltenos por un consorcio microbiano aislado de petróleo crudo MAYA. Rev. Int. Contam. Ambient. 2002, 18, 67–73. [Google Scholar]
- Araujo, I.; Angulo, N.; Cardenas, C.; Méndez, M.; Morante, M.; Machado, M. Biorremediación de Suelos con Consorcio Bacteriano, Compostaje y Fertilización. Bol. Cent. Investig. Biol. 2004, 38, 1–14. [Google Scholar]
- Rodier, J. Análisis de las Aguas; Ediciones Omega: Barcelona, Spain, 1995. [Google Scholar]
- Rafeeq, H.; Afsheen, N.; Rafique, S.; Arshad, A.; Intisar, M.; Hussain, A.; Bilal, M.; Iqbal, H.M.N. Genetically Engineered Microorganisms for Environmental Remediation. Chemosphere 2023, 310, 136751. [Google Scholar] [CrossRef]
- Imron, M.F.; Kurniawan, S.B.; Titah, H.S. Potential of Bacteria Isolated from Diesel-Contaminated Seawater in Diesel Biodegradation. Environ. Technol. Innov. 2019, 14, 100368. [Google Scholar] [CrossRef]
- Maldonado, M.; Baigori, M.; Affranchino, G. Biodegradation of organic compounds and decrease in conductivity by native consortium in effluents from the olive industry. Int. J. Recycl. Org. Waste Agric. 2022, 11, 177–187. [Google Scholar] [CrossRef]
- Food and Drug Administration (FDA). Bacteriological Analytical Manual; FDA: Silver Spring, MD, USA, 1995.
- Mitzel, R. Analysis of Polycyclic Aromatic Hydrocarbons in Utility Ash Using Modified California Air Resources Board Method 429; Alta Analytical Laboratory Inc.: El Dorado Hills, CA, USA, 1999. [Google Scholar]
- Dorozinski, F.; Gigler, G.; Schlager, M. Comparison Study of Different Extraction Methods for Determination of Hydrocarbons in Soil Samples. 2001. Available online: http://www.hblva17.magnet.at/englisch/projekte.htm (accessed on 14 October 2022).
- Calvin, H.; Loehr, R.; Nyer, E.; Pitrowski, M.; Thomas, M.; Spain, J.; Wilson, J.; Norris, R. Innovate Site Remediation Technology. In Biorremediation; Anderson, W., Ed.; American Academy of Environmental Engineers: Annapolis, MD, USA, 1995. [Google Scholar]
- BADIS, Ibn. Biodegradación de diésel e isomeratos por Pseudomonas aeruginosa y Brevibacillus laterosporus aislados de suelos contaminados con hidrocarburos. Adv. Environ. Biol. 2016, 10, 208–215. [Google Scholar]
- Tao, K.; Liu, X.; Chen, X.; Hu, X.; Cao, L.; Yuan, X. Biodegradation of Crude Oil by a Defined Co-Culture of Indigenous Bacterial Consortium and Exogenous Bacillus subtilis. Bioresour. Technol. 2017, 224, 327–332. [Google Scholar] [CrossRef] [PubMed]
- Birch, H.; Hammershøj, R.; Comber, M.; Mayer, P. Biodegradation of Hydrocarbon Mixtures in Surface Waters at Environmentally Relevant Levels—Effect of Inoculum Origin on Kinetics and Sequence of Degradation. Chemosphere 2017, 184, 400–407. [Google Scholar] [CrossRef] [PubMed]
- Aframian, N.; Eldar, A. A Bacterial Tower of Babel: Quorum-Sensing Signaling Diversity and Its Evolution. Annu. Rev. Microbiol. 2020, 74, 587–606. [Google Scholar] [CrossRef] [PubMed]
- López-Pliego, L.; Castañeda, M. Los sistemas de detección de quorum (quorum sensing) o cómo “socializan” las bacterias. RD-ICUAP 2022, 8, 177–186. [Google Scholar]
- Bajpai, P. Environmental Impact. In Biermann’s Handbook of Pulp and Paper; Elsevier: Amsterdam, The Netherlands, 2018; pp. 325–348. [Google Scholar]
- Cheng, J.; Eyheraguibel, B.; Jacquin, J.; Pujo-Pay, M.; Conan, P.; Barbe, V.; Hoypierres, J.; Deligey, G.; Halle, A.T.; Bruzaud, S.; et al. Biodegradability under Marine Conditions of Bio-Based and Petroleum-Based Polymers as Substitutes of Conventional Microparticles. Polym. Degrad. Stab. 2022, 206, 110159. [Google Scholar] [CrossRef]
- Patiño Hermoza, O.; Robles Castillo, H.; León Mendoza, L. Biodegradación de Petróleo Por Bacillus thuringiensis Como Alternativa Para La Recuperación de Suelos Agrícolas. Arnaldoa 2021, 28, 339–348. [Google Scholar] [CrossRef]
- Abdulrasheed, M.; Zulkharnain, A.; Zakaria, N.N.; Roslee, A.F.A.; Abdul Khalil, K.; Napis, S.; Convey, P.; Gomez-Fuentes, C.; Ahmad, S.A. Response Surface Methodology Optimization and Kinetics of Diesel Degradation by a Cold-Adapted Antarctic Bacterium, Arthrobacter Sp. Strain AQ5-05. Sustainability 2020, 12, 6966. [Google Scholar] [CrossRef]
- Lee, G.L.Y.; Ahmad, S.A.; Yasid, N.A.; Zulkharnain, A.; Convey, P.; Johari, W.L.W.; Alias, S.A.; Gonzalez-Rocha, G.; Shukor, M.Y. Biodegradación de fenol por bacterias adaptadas al frío de suelos antárticos. Polar Biol. 2018, 41, 553–562. [Google Scholar] [CrossRef]
- Aleer, S.; Adetutu, E.M.; Weber, J.; Ball, A.S.; Juhasz, A.L. Potential Impact of Soil Microbial Heterogeneity on the Persistence of Hydrocarbons in Contaminated Subsurface Soils. J. Environ. Manag. 2014, 136, 27–36. [Google Scholar] [CrossRef]
- Souza, E.C.; Vessoni-Penna, T.C.; de Souza Oliveira, R.P. Biosurfactant-Enhanced Hydrocarbon Bioremediation: An Overview. Int. Biodeterior. Biodegrad. 2014, 89, 88–94. [Google Scholar] [CrossRef]
- Morales-Guzmán, G.; Ferrera-Cerrato, R.; Rivera-Cruz, M.d.C.; Torres-Bustillos, L.G.; Arteaga-Garibay, R.I.; Mendoza-López, M.R.; Esquivel-Cote, R.; Alarcón, A. Diesel Degradation by Emulsifying Bacteria Isolated from Soils Polluted with Weathered Petroleum Hydrocarbons. Appl. Soil Ecol. 2017, 121, 127–134. [Google Scholar] [CrossRef]
- Liu, S.; Sun, R.; Cai, M.; Kong, Y.; Gao, Y.; Zhang, T.; Xiao, X.; Qiao, Y.; Xue, J.; Huang, G. Petroleum Spill Bioremediation by an Indigenous Constructed Bacterial Consortium in Marine Environments. Ecotoxicol. Environ. Saf. 2022, 241, 113769. [Google Scholar] [CrossRef] [PubMed]
- Fathepure, B.Z. Recent Studies in Microbial Degradation of Petroleum Hydrocarbons in Hypersaline Environments. Front. Microbiol. 2014, 5, 173. [Google Scholar] [CrossRef] [PubMed]
- Dörr de Quadros, P.; Cerqueira, V.S.; Cazarolli, J.C.; Peralba, M.d.C.R.; Camargo, F.A.O.; Giongo, A.; Bento, F.M. Oily Sludge Stimulates Microbial Activity and Changes Microbial Structure in a Landfarming Soil. Int. Biodeterior. Biodegrad. 2016, 115, 90–101. [Google Scholar] [CrossRef]
- Imron, M.F.; Kurniawan, S.B.; Ismail, N.; Abdullah, S.R.S. Future Challenges in Diesel Biodegradation by Bacteria Isolates: A Review. J. Clean. Prod. 2020, 251, 119716. [Google Scholar] [CrossRef]
- Dzionek, A.; Wojcieszyńska, D.; Guzik, U. Natural Carriers in Bioremediation: A Review. Electron. J. Biotechnol. 2016, 23, 28–36. [Google Scholar] [CrossRef]
| Inoculum Concentration (%) | % Efficiency | Growth Rate (Day−1) | |
|---|---|---|---|
| Average | Standard Deviation (SD) | ||
| 2 | 68.46 | 1.94 | 2.61 |
| 4 | 66.67 | 1.53 | 1.80 |
| 6 | 74.10 | 3.59 | 3.03 |
| 8 | 86.67 | 1.86 | 3.84 |
| 10 | 94.77 | 1.78 | 2.58 |
| Sum of Squares | Gl | Root Mean Square | F | Sig. | |
|---|---|---|---|---|---|
| Inter-groups | 1772.05 | 4 | 443.01 | 86.41 | 0.00 |
| Within groups | 51.27 | 10 | 5.13 | ||
| Total | 1823.31 | 14 |
| Concentration | N | Subset for Alpha = 0.05 | |||
|---|---|---|---|---|---|
| 1 | 2 | 3 | 4 | ||
| 4.00 | 3 | 66.67 | |||
| 2.00 | 3 | 68.47 | |||
| 6.00 | 3 | 74.10 | |||
| 8.00 | 3 | 86.67 | |||
| 10.00 | 3 | 94.77 | |||
| Sig. | 0.35 | 1.00 | 1.00 | 1.00 | |
| Sum of Squares | Gl | Root Mean Square | F | Sig. | |
|---|---|---|---|---|---|
| Regression | 1581.23 | 1 | 1581.23 | 84.91 | 0.00 |
| Residue | 242.09 | 13 | 18.62 | ||
| Total | 1823.31 | 14 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Otiniano, N.M.; Rojas-Villacorta, W.; De La Cruz-Noriega, M.; Lora-Cahuas, C.; Mendoza-Villanueva, K.; Benites, S.M.; Gallozzo-Cardenas, M.; Rojas-Flores, S. Effect of Inoculum Concentration on the Degradation of Diesel 2 by a Microbial Consortium. Sustainability 2022, 14, 16750. https://doi.org/10.3390/su142416750
Otiniano NM, Rojas-Villacorta W, De La Cruz-Noriega M, Lora-Cahuas C, Mendoza-Villanueva K, Benites SM, Gallozzo-Cardenas M, Rojas-Flores S. Effect of Inoculum Concentration on the Degradation of Diesel 2 by a Microbial Consortium. Sustainability. 2022; 14(24):16750. https://doi.org/10.3390/su142416750
Chicago/Turabian StyleOtiniano, Nélida Milly, Walter Rojas-Villacorta, Magaly De La Cruz-Noriega, Carmen Lora-Cahuas, Karol Mendoza-Villanueva, Santiago M. Benites, Moises Gallozzo-Cardenas, and Segundo Rojas-Flores. 2022. "Effect of Inoculum Concentration on the Degradation of Diesel 2 by a Microbial Consortium" Sustainability 14, no. 24: 16750. https://doi.org/10.3390/su142416750
APA StyleOtiniano, N. M., Rojas-Villacorta, W., De La Cruz-Noriega, M., Lora-Cahuas, C., Mendoza-Villanueva, K., Benites, S. M., Gallozzo-Cardenas, M., & Rojas-Flores, S. (2022). Effect of Inoculum Concentration on the Degradation of Diesel 2 by a Microbial Consortium. Sustainability, 14(24), 16750. https://doi.org/10.3390/su142416750








