Seafood Waste-Based Materials for Sustainable Food Packing: From Waste to Wealth
Abstract
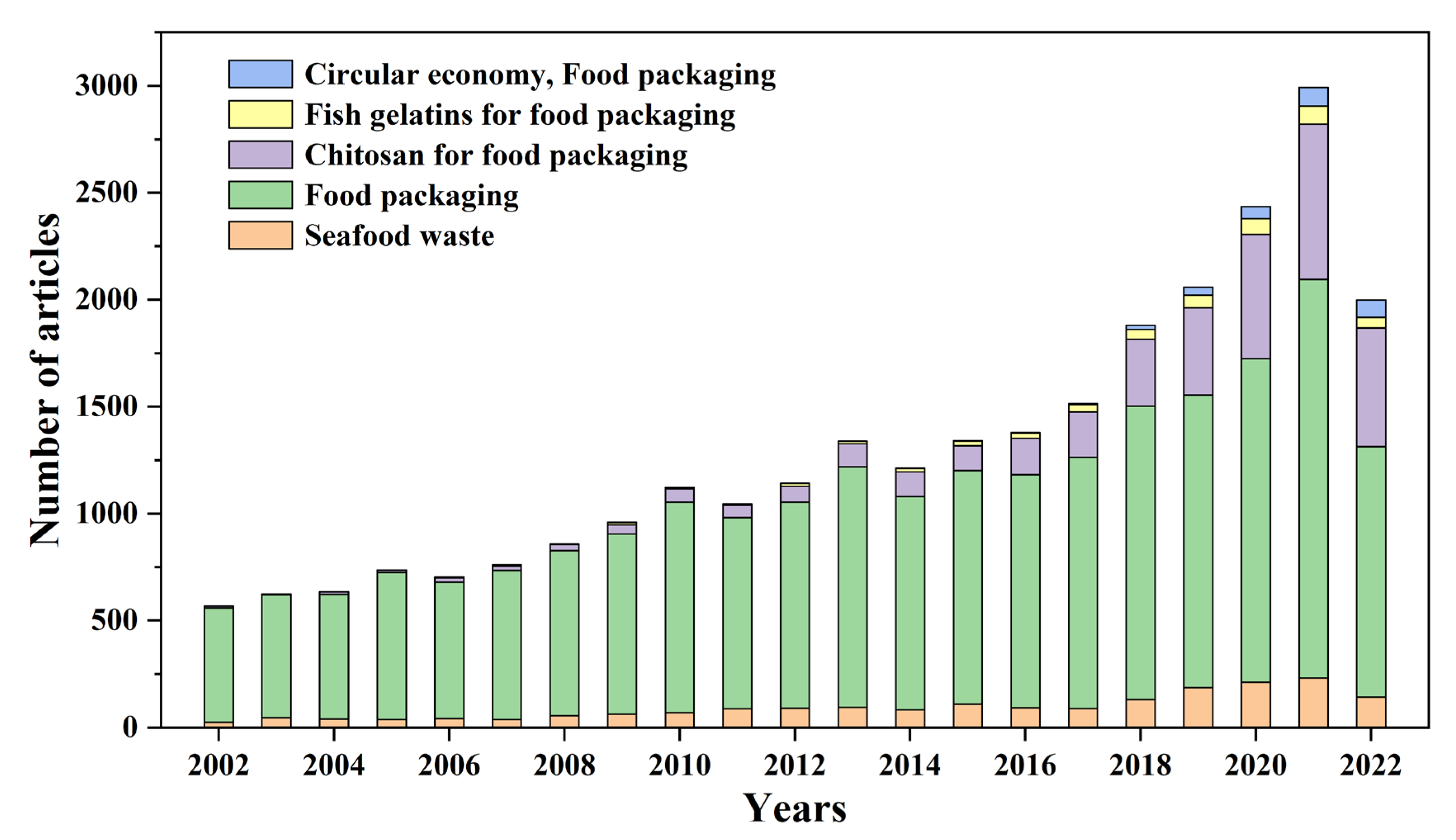
1. Introduction
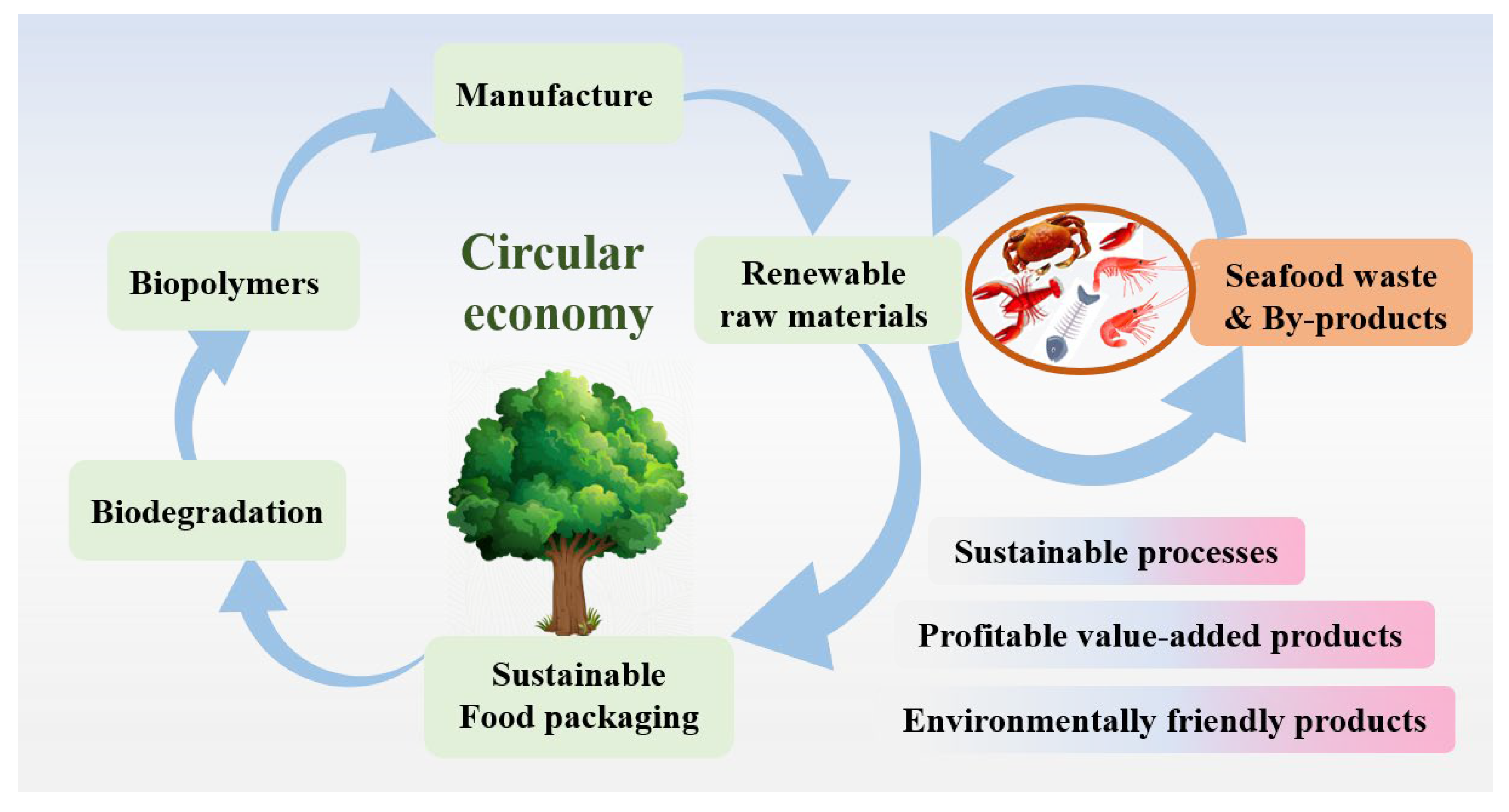
2. Seafood Waste and Utilization
2.1. Global Seafood Production in 2020
2.2. Seafood Waste
2.3. Seafood Waste and By-Product Utilization
3. Demand for Sustainable Biopolymer Based-Food Packaging Materials
4. Seafood Waste-Based Materials for Food Packing
4.1. Fish Gelatins as Antioxidant Antimicrobial Films for Food Packaging
4.2. Chitosan-Based films for Food Packaging
5. Conclusions and Future Prospect
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Xu, C.; Nasrollahzadeh, M.; Selva, M.; Issaabadi, Z.; Luque, R. Waste-to-wealth: Biowaste valorization into valuable bio(nano)materials. Chem. Soc. Rev. 2019, 48, 4791–4822. [Google Scholar] [CrossRef] [PubMed]
- Caldeira, C.; Vlysidis, A.; Fiore, G.; De Laurentiis, V.; Vignali, G.; Sala, S. Sustainability of food waste biorefinery: A review on valorisation pathways, techno-economic constraints, and environmental assessment. Bioresour. Technol. 2020, 312, 123575. [Google Scholar] [CrossRef]
- Tsuruwaka, Y.; Shimada, E. Reprocessing seafood waste: Challenge to develop aquatic clean meat from fish cells. NPJ Sci. Food 2022, 6, 7. [Google Scholar] [CrossRef] [PubMed]
- De la Caba, K.; Guerrero, P.; Trung, T.S.; Cruz-Romero, M.; Kerry, J.P.; Fluhr, J.; Maurer, M.; Kruijssen, F.; Albalat, A.; Bunting, S.; et al. From seafood waste to active seafood packaging: An emerging opportunity of the circular economy. J. Clean. Prod. 2019, 208, 86–98. [Google Scholar] [CrossRef]
- Ma, Y.; Liu, Y. Turning food waste to energy and resources towards a great environmental and economic sustainability: An innovative integrated biological approach. Biotechnol. Adv. 2019, 37, 107414. [Google Scholar] [CrossRef]
- Gregg, J.S.; Jürgens, J.; Happel, M.K.; Strøm-Andersen, N.; Tanner, A.N.; Bolwig, S.; Klitkou, A. Valorization of bio-residuals in the food and forestry sectors in support of a circular bioeconomy: A review. J. Clean. Prod. 2020, 267, 122093. [Google Scholar] [CrossRef]
- Zou, W.; Li, J.; Wang, R.; Ma, J.; Chen, Z.; Duan, L.; Mi, H.; Chen, H. Hydroxylamine mediated Fenton-like interfacial reaction dynamics on sea urchin-like catalyst derived from spent LiFePO(4) battery. J. Hazard. Mater. 2022, 431, 128590. [Google Scholar] [CrossRef]
- Tao, M.; Lu, D.; Shi, Y.; Wu, C. Utilization and life cycle assessment of low activity solid waste as cementitious materials: A case study of titanium slag and granulated blast furnace slag. Sci. Total Environ. 2022, 849, 157797. [Google Scholar] [CrossRef]
- Chen, Z.; Zou, W.; Zheng, R.; Wei, W.; Wei, W.; Ni, B.-J.; Chen, H. Synergistic recycling and conversion of spent Li-ion battery leachate into highly efficient oxygen evolution catalysts. Green Chem. 2021, 23, 6538–6547. [Google Scholar] [CrossRef]
- Chen, Z.; Zheng, R.; Zou, W.; Wei, W.; Li, J.; Wei, W.; Ni, B.-J.; Chen, H. Integrating high-efficiency oxygen evolution catalysts featuring accelerated surface reconstruction from waste printed circuit boards via a boriding recycling strategy. Appl. Catal. B Environ. 2021, 298, 120583. [Google Scholar] [CrossRef]
- Chen, Z.; Zheng, R.; Wei, W.; Wei, W.; Zou, W.; Li, J.; Ni, B.-J.; Chen, H. Recycling spent water treatment adsorbents for efficient electrocatalytic water oxidation reaction. Resour. Conserv. Recycl. 2022, 178, 106037. [Google Scholar] [CrossRef]
- Chen, Z.; Wei, W.; Chen, H.; Ni, B.-J. Recent advances in waste-derived functional materials for wastewater remediation. Eco-Environ. Health 2022, 1, 86–104. [Google Scholar] [CrossRef]
- Chen, Z.; Wei, W.; Zou, W.; Li, J.; Zheng, R.; Wei, W.; Ni, B.-J.; Chen, H. Integrating electrodeposition with electrolysis for closed-loop resource utilization of battery industrial wastewater. Green Chem. 2022, 24, 3208–3217. [Google Scholar] [CrossRef]
- European Commission, 2015. Closing the Loop—An EU Action Plan for the Circular Economy, Communication from the Commission to the European Parliament, the Council, the European Economic and Social Committee and the Committee of the Regions. Brussels, Belgium, Document 52015DC0614. Available online: https://eur-lex.europa.eu/legal-content/EN/TXT/?uri=CELEX:52015DC0614 (accessed on 25 October 2022).
- Debeaufort, F. Active biopackaging produced from by-products and waste from food and marine industries. FEBS Open Bio 2021, 11, 984–998. [Google Scholar] [CrossRef]
- Amiri, H.; Aghbashlo, M.; Sharma, M.; Gaffey, J.; Manning, L.; Moosavi Basri, S.M.; Kennedy, J.F.; Gupta, V.K.; Tabatabaei, M. Chitin and chitosan derived from crustacean waste valorization streams can support food systems and the UN Sustainable Development Goals. Nat. Food 2022, 3, 822–828. [Google Scholar] [CrossRef]
- Food and Agriculture Organization of the United Nations (FAO). The State of World Fisheries and Aquaculture 2022. Sustain. Action 2022. Available online: https://doi.org/10.4060/cc0461en (accessed on 25 October 2022).
- Shahidi, F.; Varatharajan, V.; Peng, H.; Senadheera, R. Utilization of marine by-products for the recovery of value-added products. J. Food Bioact. 2019, 6, 10–61. [Google Scholar] [CrossRef]
- Teixeira-Costa, B.E.; Andrade, C.T. Chitosan as a Valuable Biomolecule from Seafood Industry Waste in the Design of Green Food Packaging. Biomolecules 2021, 11, 1599. [Google Scholar] [CrossRef]
- Chisenga, S.M.; Tolesa, G.N.; Workneh, T.S. Biodegradable Food Packaging Materials and Prospects of the Fourth Industrial Revolution for Tomato Fruit and Product Handling. Int. J. Food Sci. 2020, 2020, 8879101. [Google Scholar] [CrossRef]
- Beltran, M.; Tjahjono, B.; Bogush, A.; Julião, J.; Teixeira, E.L.S. Food Plastic Packaging Transition towards Circular Bioeconomy: A Systematic Review of Literature. Sustainability 2021, 13, 3896. [Google Scholar] [CrossRef]
- Markevičiūtė, Z.; Varžinskas, V. Smart Material Choice: The Importance of Circular Design Strategy Applications for Bio-Based Food Packaging Preproduction and End-of-Life Life Cycle Stages. Sustainability 2022, 14, 6366. [Google Scholar] [CrossRef]
- Wang, H.; Qian, J.; Ding, F. Emerging Chitosan-Based Films for Food Packaging Applications. J. Agric Food Chem. 2018, 66, 395–413. [Google Scholar] [CrossRef] [PubMed]
- European Commission. Sustainable Technologies for the Production of Biodegradable Materials Based on Natural Chitin-Nanofibrils Derived by Waste of Fish Industry, to Produce Food Grade Packaging. 2015. Available online: https://cordis.europa.eu/article/id/151596-food-packaging-from-shellfish-waste (accessed on 25 October 2022).
- Nag, M.; Lahiri, D.; Dey, A.; Sarkar, T.; Pati, S.; Joshi, S.; Bunawan, H.; Mohammed, A.; Edinur, H.A.; Ghosh, S.; et al. Seafood Discards: A Potent Source of Enzymes and Biomacromolecules with Nutritional and Nutraceutical Significance. Front. Nutr. 2022, 9, 879929. [Google Scholar] [CrossRef] [PubMed]
- Ozogul, F.; Cagalj, M.; Šimat, V.; Ozogul, Y.; Tkaczewska, J.; Hassoun, A.; Kaddour, A.A.; Kuley, E.; Rathod, N.B.; Phadke, G.G. Recent developments in valorisation of bioactive ingredients in discard/seafood processing by-products. Trends Food Sci. Technol. 2021, 116, 559–582. [Google Scholar] [CrossRef]
- Pauly, D.Z.D.; Palomares, M.L.D. (Eds.) Sea around Us Concepts, Design and Data (seaaroundus.org). Available online: http://www.seaaroundus.org/ (accessed on 25 October 2022).
- Mathew, G.M.; Mathew, D.C.; Sukumaran, R.K.; Sindhu, R.; Huang, C.C.; Binod, P.; Sirohi, R.; Kim, S.H.; Pandey, A. Sustainable and eco-friendly strategies for shrimp shell valorization. Environ. Pollut. 2020, 267, 115656. [Google Scholar] [CrossRef]
- Da Trindade Alfaro, A.; Balbinot, E.; Weber, C.I.; Tonial, I.B.; Machado-Lunkes, A. Fish Gelatin: Characteristics, Functional Properties, Applications and Future Potentials. Food Eng. Rev. 2014, 7, 33–44. [Google Scholar] [CrossRef]
- Siddik, M.A.B.; Howieson, J.; Fotedar, R.; Partridge, G.J. Enzymatic fish protein hydrolysates in finfish aquaculture: A review. Rev. Aquac. 2020, 13, 406–430. [Google Scholar] [CrossRef]
- Suntornsaratoon, P.; Charoenphandhu, N.; Krishnamra, N. Fortified tuna bone powder supplementation increases bone mineral density of lactating rats and their offspring. J. Sci. Food Agric. 2018, 98, 2027–2034. [Google Scholar] [CrossRef]
- Simat, V.; Vlahovic, J.; Soldo, B.; Skroza, D.; Ljubenkov, I.; Generalic Mekinic, I. Production and Refinement of Omega-3 Rich Oils from Processing By-Products of Farmed Fish Species. Foods 2019, 8, 125. [Google Scholar] [CrossRef]
- Elsabee, M.Z.; Abdou, E.S. Chitosan based edible films and coatings: A review. Mater Sci. Eng. C Mater Biol. Appl. 2013, 33, 1819–1841. [Google Scholar] [CrossRef]
- Sayari, N.; Sila, A.; Abdelmalek, B.E.; Abdallah, R.B.; Ellouz-Chaabouni, S.; Bougatef, A.; Balti, R. Chitin and chitosan from the Norway lobster by-products: Antimicrobial and anti-proliferative activities. Int. J. Biol. Macromol. 2016, 87, 163–171. [Google Scholar] [CrossRef]
- van den Broek, L.A.; Knoop, R.J.; Kappen, F.H.; Boeriu, C.G. Chitosan films and blends for packaging material. Carbohydr. Polym. 2015, 116, 237–242. [Google Scholar] [CrossRef]
- UNEP, 2015. Transforming our World: The 2030 Agenda for Sustainable Development. United Nations Resolution A/RES/70/1. Available online: https://sdgs.un.org/2030agenda (accessed on 25 October 2022).
- Jariyasakoolroj, P.; Leelaphiwat, P.; Harnkarnsujarit, N. Advances in research and development of bioplastic for food packaging. J. Sci. Food Agric. 2020, 100, 5032–5045. [Google Scholar] [CrossRef]
- Du Preez, M.; Van der Merwe, D.; Wyma, L.; Ellis, S.M. Assessing Knowledge and Use Practices of Plastic Food Packaging among Young Adults in South Africa: Concerns about Chemicals and Health. Int. J. Environ. Res. Public Health 2021, 18, 10576. [Google Scholar] [CrossRef]
- Flórez, M.; Guerra-Rodríguez, E.; Cazón, P.; Vázquez, M. Chitosan for food packaging: Recent advances in active and intelligent films. Food Hydrocoll. 2022, 124, 107328. [Google Scholar] [CrossRef]
- Tongnuanchan, P.; Benjakul, S.; Prodpran, T. Physico-chemical properties, morphology and antioxidant activity of film from fish skin gelatin incorporated with root essential oils. J. Food Eng. 2013, 117, 350–360. [Google Scholar] [CrossRef]
- Lu, Y.; Luo, Q.; Chu, Y.; Tao, N.; Deng, S.; Wang, L.; Li, L. Application of Gelatin in Food Packaging: A Review. Polymers 2022, 14, 436. [Google Scholar] [CrossRef]
- Zhang, Q.; Wang, Q.; Lv, S.; Lu, J.; Jiang, S.; Regenstein, J.M.; Lin, L. Comparison of collagen and gelatin extracted from the skins of Nile tilapia (Oreochromis niloticus) and channel catfish (Ictalurus punctatus). Food Biosci. 2016, 13, 41–48. [Google Scholar] [CrossRef]
- Benbettaïeb, N.; Tanner, C.; Cayot, P.; Karbowiak, T.; Debeaufort, F. Impact of functional properties and release kinetics on antioxidant activity of biopolymer active films and coatings. Food Chem. 2018, 242, 369–377. [Google Scholar] [CrossRef]
- Bermudez-Oria, A.; Rodriguez-Gutierrez, G.; Rubio-Senent, F.; Fernandez-Prior, A.; Fernandez-Bolanos, J. Effect of edible pectin-fish gelatin films containing the olive antioxidants hydroxytyrosol and 3,4-dihydroxyphenylglycol on beef meat during refrigerated storage. Meat Sci. 2019, 148, 213–218. [Google Scholar] [CrossRef]
- Bermudez-Oria, A.; Rodriguez-Gutierrez, G.; Vioque, B.; Rubio-Senent, F.; Fernandez-Bolanos, J. Physical and functional properties of pectin-fish gelatin films containing the olive phenols hydroxytyrosol and 3,4-dihydroxyphenylglycol. Carbohydr. Polym. 2017, 178, 368–377. [Google Scholar] [CrossRef] [PubMed]
- Ge, Y.; Li, Y.; Bai, Y.; Yuan, C.; Wu, C.; Hu, Y. Intelligent gelatin/oxidized chitin nanocrystals nanocomposite films containing black rice bran anthocyanins for fish freshness monitorings. Int. J. Biol. Macromol. 2020, 155, 1296–1306. [Google Scholar] [CrossRef] [PubMed]
- Guerrero, P.; Zugasti, I.; Etxabide, A.; Bao, H.N.D.; Trang Si, T.; Penalba, M.; de la Caba, K. Effect of Fructose and Ascorbic Acid on the Performance of Cross-Linked Fish Gelatin Films. Polymers 2020, 12, 570. [Google Scholar] [CrossRef] [PubMed]
- Hu, H.; Yao, X.; Qin, Y.; Yong, H.; Liu, J. Development of multifunctional food packaging by incorporating betalains from vegetable amaranth (Amaranthus tricolor L.) into quaternary ammonium chitosan/fish gelatin blend films. Int. J. Biol. Macromol. 2020, 159, 675–684. [Google Scholar] [CrossRef] [PubMed]
- Huang, S.; Tu, Z.; Sha, X.; Hu, Y.; Chen, N.; Wang, H. Fabrication and performance evaluation of pectin-fish gelatin-resveratrol preservative films. Food Chem. 2021, 361, 129832. [Google Scholar] [CrossRef] [PubMed]
- Le, T.; Maki, H.; Okazaki, E.; Osako, K.; Takahashi, K. Influence of Various Phenolic Compounds on Properties of Gelatin Film Prepared from Horse Mackerel Trachurus japonicus Scales. J. Food Sci. 2018, 83, 1888–1895. [Google Scholar] [CrossRef]
- Limpisophon, K.; Schleining, G. Use of Gallic Acid to Enhance the Antioxidant and Mechanical Properties of Active Fish Gelatin Film. J. Food Sci. 2017, 82, 80–89. [Google Scholar] [CrossRef]
- Valdes, A.; Garcia-Serna, E.; Martinez-Abad, A.; Vilaplana, F.; Jimenez, A.; Garrigos, M.C. Gelatin-Based Antimicrobial Films Incorporating Pomegranate (Punica granatum L.) Seed Juice By-Product. Molecules 2019, 25, 166. [Google Scholar] [CrossRef]
- Mousavi, Z.; Naseri, M.; Babaei, S.; Hosseini, S.M.H.; Shekarforoush, S.S. The effect of cross-linker type on structural, antimicrobial and controlled release properties of fish gelatin-chitosan composite films incorporated with epsilon-poly-l-lysine. Int. J. Biol. Macromol. 2021, 183, 1743–1752. [Google Scholar] [CrossRef]
- Lee, K.Y.; Yang, H.J.; Song, K.B. Application of a puffer fish skin gelatin film containing Moringa oleifera Lam. leaf extract to the packaging of Gouda cheese. J. Food Sci. Technol. 2016, 53, 3876–3883. [Google Scholar] [CrossRef]
- Lin, D.; Yang, Y.; Wang, J.; Yan, W.; Wu, Z.; Chen, H.; Zhang, Q.; Wu, D.; Qin, W.; Tu, Z. Preparation and characterization of TiO2-Ag loaded fish gelatin-chitosan antibacterial composite film for food packaging. Int. J. Biol. Macromol. 2020, 154, 123–133. [Google Scholar] [CrossRef]
- Li, X.; Tu, Z.C.; Sha, X.M.; Ye, Y.H.; Li, Z.Y. Flavor, antimicrobial activity, and physical properties of composite film prepared with different surfactants. Food Sci. Nutr. 2020, 8, 3099–3109. [Google Scholar] [CrossRef]
- Salem, A.; Jridi, M.; Abdelhedi, O.; Fakhfakh, N.; Nasri, M.; Debeaufort, F.; Zouari, N. Development and characterization of fish gelatin-based biodegradable film enriched with Lepidium sativum extract as active packaging for cheese preservation. Heliyon 2021, 7, e08099. [Google Scholar] [CrossRef]
- Jeya Shakila, R.; Jeevithan, E.; Varatharajakumar, A.; Jeyasekaran, G.; Sukumar, D. Comparison of the properties of multi-composite fish gelatin films with that of mammalian gelatin films. Food Chem. 2012, 135, 2260–2267. [Google Scholar] [CrossRef]
- Yadav, M.; Goswami, P.; Paritosh, K.; Kumar, M.; Pareek, N.; Vivekanand, V. Seafood waste: A source for preparation of commercially employable chitin/chitosan materials. Bioresour. Bioprocess. 2019, 6, 8. [Google Scholar] [CrossRef]
- Tongwanichniyom, S.; Kitjaruwankul, S.; Phornphisutthimas, S. Production of biomaterials from seafood waste for application as vegetable wash disinfectant. Heliyon 2022, 8, e09357. [Google Scholar] [CrossRef]
- Mujtaba, M.; Morsi, R.E.; Kerch, G.; Elsabee, M.Z.; Kaya, M.; Labidi, J.; Khawar, K.M. Current advancements in chitosan-based film production for food technology; A review. Int. J. Biol. Macromol. 2019, 121, 889–904. [Google Scholar] [CrossRef]
- Gumienna, M.; Gorna, B. Antimicrobial Food Packaging with Biodegradable Polymers and Bacteriocins. Molecules 2021, 26, 3735. [Google Scholar] [CrossRef]
- Affes, S.; Aranaz, I.; Acosta, N.; Heras, A.; Nasri, M.; Maalej, H. Chitosan derivatives-based films as pH-sensitive drug delivery systems with enhanced antioxidant and antibacterial properties. Int. J. Biol. Macromol. 2021, 182, 730–742. [Google Scholar] [CrossRef]
- Amor, G.; Sabbah, M.; Caputo, L.; Idbella, M.; De Feo, V.; Porta, R.; Fechtali, T.; Mauriello, G. Basil Essential Oil: Composition, Antimicrobial Properties, and Microencapsulation to Produce Active Chitosan Films for Food Packaging. Foods 2021, 10, 121. [Google Scholar] [CrossRef]
- Ardebilchi Marand, S.; Almasi, H.; Ardebilchi Marand, N. Chitosan-based nanocomposite films incorporated with NiO nanoparticles: Physicochemical, photocatalytic and antimicrobial properties. Int. J. Biol. Macromol. 2021, 190, 667–678. [Google Scholar] [CrossRef] [PubMed]
- Bi, F.; Qin, Y.; Chen, D.; Kan, J.; Liu, J. Development of active packaging films based on chitosan and nano-encapsulated luteolin. Int. J. Biol. Macromol. 2021, 182, 545–553. [Google Scholar] [CrossRef] [PubMed]
- Bras, T.; Rosa, D.; Goncalves, A.C.; Gomes, A.C.; Alves, V.D.; Crespo, J.G.; Duarte, M.F.; Neves, L.A. Development of bioactive films based on chitosan and Cynara cardunculus leaves extracts for wound dressings. Int. J. Biol. Macromol. 2020, 163, 1707–1718. [Google Scholar] [CrossRef] [PubMed]
- Costa, S.M.; Ferreira, D.P.; Teixeira, P.; Ballesteros, L.F.; Teixeira, J.A.; Fangueiro, R. Active natural-based films for food packaging applications: The combined effect of chitosan and nanocellulose. Int. J. Biol. Macromol. 2021, 177, 241–251. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Bramasco, C.A.; Blancas-Benitez, F.J.; Montano-Leyva, B.; Medrano-Castellon, L.M.; Gutierrez-Martinez, P.; Gonzalez-Estrada, R.R. Influence of Marine Yeast Debaryomyces hansenii on Antifungal and Physicochemical Properties of Chitosan-Based Films. J. Fungi 2022, 8, 369. [Google Scholar] [CrossRef] [PubMed]
- Hamdi, M.; Nasri, R.; Azaza, Y.B.; Li, S.; Nasri, M. Conception of novel blue crab chitosan films crosslinked with different saccharides via the Maillard reaction with improved functional and biological properties. Carbohydr. Polym. 2020, 241, 116303. [Google Scholar] [CrossRef] [PubMed]
- Hao, Y.; Kang, J.; Guo, X.; Sun, M.; Li, H.; Bai, H.; Cui, H.; Shi, L. pH-responsive chitosan-based film containing oregano essential oil and black rice bran anthocyanin for preserving pork and monitoring freshness. Food Chem. 2022, 403, 134393. [Google Scholar] [CrossRef]
- Hua, L.; Deng, J.; Wang, Z.; Wang, Y.; Chen, B.; Ma, Y.; Li, X.; Xu, B. Improving the functionality of chitosan-based packaging films by crosslinking with nanoencapsulated clove essential oil. Int. J. Biol. Macromol. 2021, 192, 627–634. [Google Scholar] [CrossRef]
- Kaczmarek-Szczepanska, B.; Sosik, A.; Malkowska, A.; Zasada, L.; Michalska-Sionkowska, M. Chitosan-based films enriched by caffeic acid with poly (ethylene glycol)—A physicochemical and antibacterial properties evaluation. Int. J. Biol. Macromol. 2021, 192, 728–735. [Google Scholar] [CrossRef]
- Kayani, A.; Raza, M.A.; Raza, A.; Hussain, T.; Akram, M.S.; Sabir, A.; Islam, A.; Haider, B.; Khan, R.U.; Park, S.H. Effect of Varying Amount of Polyethylene Glycol (PEG-600) and 3-Aminopropyltriethoxysilane on the Properties of Chitosan based Reverse Osmosis Membranes. Int. J. Mol. Sci. 2021, 22, 2290. [Google Scholar] [CrossRef]
- Li, B.; Bao, Y.; Li, J.; Bi, J.; Chen, Q.; Cui, H.; Wang, Y.; Tian, J.; Shu, C.; Wang, Y.; et al. A sub-freshness monitoring chitosan/starch-based colorimetric film for improving color recognition accuracy via controlling the pH value of the film-forming solution. Food Chem. 2022, 388, 132975. [Google Scholar] [CrossRef]
- Lin, W.; Huang, G.; Yang, W.; Zeng, S.; Luo, X.; Huang, J.; Li, Z. A dual-function chitosan packaging film for simultaneously monitoring and maintaining pork freshness. Food Chem. 2022, 392, 133242. [Google Scholar] [CrossRef]
- Oliveira, V.D.S.; Cruz, M.M.D.; Bezerra, G.S.; Silva, N.; Nogueira, F.H.A.; Chaves, G.M.; Sobrinho, J.L.S.; Mendonca-Junior, F.J.B.; Damasceno, B.; Converti, A.; et al. Chitosan-Based Films with 2-Aminothiophene Derivative: Formulation, Characterization and Potential Antifungal Activity. Mar. Drugs 2022, 20, 103. [Google Scholar] [CrossRef]
- Park, K.J.; Lee, J.S.; Jo, H.J.; Kim, E.S.; Lee, H.G. Antimicrobial and indicator properties of edible film containing clove bud oil-loaded chitosan capsules and red cabbage for fish preservation. Int. J. Biol. Macromol. 2022, 196, 163–171. [Google Scholar] [CrossRef]
- Pavlátková, L.; Sedlaříková, J.; Pleva, P.; Peer, P.; Uysal-Unalan, I.; Janalíková, M. Bioactive zein/chitosan systems loaded with essential oils for food-packaging applications. J. Sci. Food Agric. 2022. [Google Scholar] [CrossRef]
- Roy, S.; Rhim, J.W. Genipin-Crosslinked Gelatin/Chitosan-Based Functional Films Incorporated with Rosemary Essential Oil and Quercetin. Materials 2022, 15, 3769. [Google Scholar] [CrossRef]
- Salavati Hamedani, M.; Rezaeigolestani, M.; Mohsenzadeh, M. Optimization of Antibacterial, Physical and Mechanical Properties of Novel Chitosan/Olibanum Gum Film for Food Packaging Application. Polymers 2022, 14, 3960. [Google Scholar] [CrossRef]
- Yadav, S.; Mehrotra, G.K.; Dutta, P.K. Chitosan based ZnO nanoparticles loaded gallic-acid films for active food packaging. Food Chem. 2021, 334, 127605. [Google Scholar] [CrossRef]
- Xu, Y.; Hou, K.; Gao, C.; Feng, X.; Cheng, W.; Wu, D.; Meng, L.; Yang, Y.; Shen, X.; Zhang, Y.; et al. Characterization of chitosan film with cinnamon essential oil emulsion co-stabilized by ethyl-N(alpha)-lauroyl-l-arginate hydrochloride and hydroxypropyl-beta-cyclodextrin. Int. J. Biol. Macromol. 2021, 188, 24–31. [Google Scholar] [CrossRef]
- Zhang, X.; Xiao, G.; Wang, Y.; Zhao, Y.; Su, H.; Tan, T. Preparation of chitosan-TiO2 composite film with efficient antimicrobial activities under visible light for food packaging applications. Carbohydr. Polym. 2017, 169, 101–107. [Google Scholar] [CrossRef]
- Liu, F.; Chang, W.; Chen, M.; Xu, F.; Ma, J.; Zhong, F. Tailoring physicochemical properties of chitosan films and their protective effects on meat by varying drying temperature. Carbohydr. Polym. 2019, 212, 150–159. [Google Scholar] [CrossRef] [PubMed]
- Chang, W.; Liu, F.; Sharif, H.R.; Huang, Z.; Goff, H.D.; Zhong, F. Preparation of chitosan films by neutralization for improving their preservation effects on chilled meat. Food Hydrocoll. 2019, 90, 50–61. [Google Scholar] [CrossRef]



| Type | Compounds Name | Reference |
|---|---|---|
| Antioxidants | Ferulic acid, Caffeic acid and Tyrosol | [43] |
| Hydroxytyrosol (HT) 3,4-dihydroxyphenylglycol (DHPG) | [44,45] | |
| Black rice bran anthocyanins | [46] | |
| Fructose and Ascorbic acid | [47] | |
| Betalains from vegetable amaranth | [48] | |
| Resveratrol | [49] | |
| Ferulic acid, Caffeic acid, Gallic acid, Catechin, and Rutin | [50] | |
| Gallic Acid | [51] | |
| Antimicrobials | Seed juice by-product | [52] |
| ε-poly-Llysine (ε-PLL) | [53] | |
| Leaf extract | [54] | |
| TiO2-Ag | [55] | |
| Biological polymer | Pectin | [44,45] |
| Oxidized chitin nanocrystals | [46] | |
| Lecithin, Tween-20, and Tween-80 | [56] | |
| Chitosan | [55] |
| Chitosan Concentration | Modified Materials | Tools | Drying Conditions | Reference |
|---|---|---|---|---|
| 1% (w/v) | Glycerol | Plastic Petri dishes | 48 h at 25 °C | [63] |
| 3% (w/v) | Basil essential oil | polypropylene sheet (24 × 18 cm2) | 5 h at RT | [64] |
| 3–9% (w/v) | Nickel oxide nanoparticles | plates (8 cm) | 45 °C for 3 days | [65] |
| 2% (w/v) | Luteolin | Plexiglas plate (24 cm × 24 cm) | 30 °C for 2 days, 50% (RH) | [66] |
| 2% (w/v) | Cynara cardunculus leaves extracts | Petri dishes (90 mm diameter) | 40 °C for 48 h. | [67] |
| 2% (w/v) | cellulose | Petri dishes | 35 °C for 48 h | [68] |
| 0.5–1.5% (w/v) | Marine Yeast Debaryomyces hansenii | Petri dishes (60 × 15 mm2) | 40 h at 40 °C (22% RH) | [69] |
| 2% (w/v) | Glucose, Fructose, Xylose, Arabinose | Petri dishes (13.5 cm diameter) | 48 h at 25 °C | [70] |
| 2% (w/v) | Oregano essential oil Black rice bran anthocyanin | Petri dishes (9 cm diameter) | 25 °C for 3 days, 50% (RH) | [71] |
| 1.5% (w/v) | Clove essential oil | - | 48 h at 25 °C (50% RH) | [72] |
| 1% (w/v) | Caffeic acid poly (ethylene glycol) | Plastic holder 40 mL per 10 × 10 cm2 | - | [73] |
| 3% (w/v) | Glycol (PEG-600) 3-Aminopropyltriethoxysilane | glass plate | 5–6 h at 60 °C | [74] |
| 0.8 (w/v) | potato starch, anthocyanins | Glass plate (15 × 15 × 0.5 cm3) | 45 °C for 12 h | [75] |
| 2 wt% | N-doped carbon dots | Petri dishes (9 cm diameter) | 60 °C for 12 h | [76] |
| 1% (w/v) | 2-amino-4,5,6,7-tetrahydrobenzo[b]thiophene-3-carbonitrile | Petri dishes (47 mm diameter) | 50 °C for 24 h | [77] |
| 0.32 g/cm2 | Red cabbage Clove bud oil | Petri dishes (8 cm diameter | - | [78] |
| 0.5% (w/v) | Zein | Disks (9 mm diameter), | 3 h, 24 h, and 5 days, 12 days | [79] |
| 2 wt% | Gelatin | Flat Teflon film-coated glass plate | RT for 48 h. | [80] |
| 0.5–1.5% (w/v) | Olibanum Gum | 60 °C by a heater–stirrer for 3 h. | [81] | |
| 2%(w/v) | Zinc oxide nanoparticles gallic-acid | Petri-plates | 50 °C for 12 h in an oven. | [82] |
| 1.5% (w/v) | Cinnamon essential oil Ethyl-Nα-lauroyl-L-arginate hydrochloride Hydroxypropyl-β-cyclodextrin | Polyethylene Petri dishes | 45 °C for 12 h | [83] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhao, Z.; Li, Y.; Du, Z. Seafood Waste-Based Materials for Sustainable Food Packing: From Waste to Wealth. Sustainability 2022, 14, 16579. https://doi.org/10.3390/su142416579
Zhao Z, Li Y, Du Z. Seafood Waste-Based Materials for Sustainable Food Packing: From Waste to Wealth. Sustainability. 2022; 14(24):16579. https://doi.org/10.3390/su142416579
Chicago/Turabian StyleZhao, Zezhong, Yajuan Li, and Zhiyang Du. 2022. "Seafood Waste-Based Materials for Sustainable Food Packing: From Waste to Wealth" Sustainability 14, no. 24: 16579. https://doi.org/10.3390/su142416579
APA StyleZhao, Z., Li, Y., & Du, Z. (2022). Seafood Waste-Based Materials for Sustainable Food Packing: From Waste to Wealth. Sustainability, 14(24), 16579. https://doi.org/10.3390/su142416579




