Abstract
The North-Colombian region has enormous potential for producing bioproducts and bioenergy from agricultural residues. Yet, scaling bioproducts and bioenergy to industrial practice requires further investigation, especially for environmental impact minimization and improved process safety. This work assesses two alternatives for valorizing cassava residues via acetone, butanol, and ethanol (ABE) fermentation. Two ABE fermentation routes are assessed. In Route 1, pretreatment and purification involve dilute-acid pretreatment and multi-effect distillation and decantation operations, while Route 2 includes steam explosion and reactive distillation. Hazard Identification and Risk Assessment (HIRA) and Waste Reduction Algorithm (WAR) were applied to assess ABE fermentation. Simulation results indicate butanol yields of 0.10–0.12 kg/kg feedstock and net energy ratio (NER) <1. Route 2 shows the highest total output of Potential Environmental Impacts (PEI) with 5.56 PEI/kg butanol. Both ABE fermentation routes obtained Fire and Explosion Damage Index (FEDI) values above 300 for acetone and ethanol recovery/purification stages. Both routes are classified as “hazardous” considering the flammability of handled substances, and their relative safety performance is remarkably similar. These results pave the way toward deploying both routes for adding value to the cassava residues in North Colombia by applying safe, efficient, and environmentally friendly transformation technologies.
1. Introduction
Globally, agriculture accounts for about 26% of CO2 emissions, 50% of habitable land, and 70% of freshwater withdrawals [1,2]. Since agroindustry generates significant environmental impacts, there is a need to transition toward a more sustainable circular bioeconomy. Using agri-food waste as feedstock in biorefineries enables the production of a wide range of bioproducts and bioenergy, representing a step in the right direction [3]. Colombia has considerable potential for agroindustry development due to its biodiversity, high-yield crops, and favorable climatic conditions. For example, Colombia is among the top ten world producers, and the second in Latin America behind Brazil, of sugar cane, oil palm, and cassava crops, with a substantial availability of related side streams [4,5]. Compared to other crops, cassava can grow in low-nutrient soils and with fewer rainfalls. The cassava plant is composed of 45%wt. tuber, 10%wt. rhizome, 35%wt. stem and 10%wt. leaves [6]. However, only the edible part (i.e., tuber) is used for food industries, and the remaining portions are considered fibrous residues. The production rates of this solid waste is estimated at 200 kg/ton of processed tubers [7]. One of the most important by-products of the cassava processing industry is the bagasse, which contains around 40–75% starch on a dry weight basis [8]. Due to its composition, cassava bagasse has vast potential as feedstock for various high-value products [9].
Even though Colombia has a boundless availability of agricultural biomass, there is an absence of knowledge related to the optimal exploitation of these residues and the scaling-up of biomass conversion technologies [4]. Processes transforming agricultural residues into bioenergy and biochemicals need to consider sustainability issues, environmental impacts, and hazard identification. Hence, bioenergy and bio-based production face many challenges in reaching commercialization due to technological inefficiencies and raw material costs, among others [10]. Applying CAPE tools, environmental assessments, and process safety assessments provides valuable insights into bioenergy and bioproduct production systems. The ABE fermentation routes assessed in this work were determined by applying a process synthesis methodology developed by the authors [11] for a case study of ABE fermentation. As a result, two optimal ABE fermentation process configurations were synthesized as alternatives for valorizing cassava residues.
Valorizing cassava residues is essential to reduce health issues and environmental impacts from firewood and charcoal combustion and energy paucity [12]. In the bioenergy and agriculture sectors, this may help enhance food security and the need for expanding this crop’s land [8]. Previously, much research has been conducted on cassava residues, including bioconversion into high-value products such as biomethane, biohydrogen, lactic acid, and acetone-butanol-ethanol (ABE) [13,14]. ABE fermentation has gained scientific attention as an attractive alternative for biofuel production from starchy substrates and anaerobic bacteria [15]. Moreover, the generation of by-products can reduce the butanol production cost [16,17]. However, advances remain insufficient to face some drawbacks of large-scale production [18]. Different studies seek optimal process configuration [19], improved fermentation [20], or more efficient downstream processing [21]. Upstream processes include pretreatment and fermentation units, while downstream processes involve product recovery and purification [22]. Pretreatment is vital for cost-effectiveness [23] and environmental sustainability [24].
Conversely, improving process performance involves applying process analysis tools, such as environmental and process safety assessments. Kumar et al. [17] performed a techno-economic analysis of biobutanol production from ABE fermentation using cellulosic (bagasse, barley straw, wheat straw, corn stover, and switchgrass) and non-cellulosic (glucose, sugarcane, corn, and sago) feedstocks. They assessed the sensitivity of biobutanol price when varying design/process parameters and reported economic feasibility for sugarcane and cellulosic materials. Patrascu et al. [22] considered energy-based aspects of biobutanol separation after ABE fermentation, and developed a novel intensified separation process to reduce the energy usage of downstream units. Table 1 summarizes recent contributions focused on the simulation, environmental assessment, and process safety evaluation of ABE fermentation in either upstream or downstream processing.

Table 1.
Application of process analysis for environmental and process safety aspects in ABE fermentation.
Table 1 shows a lack of knowledge regarding the safety assessment of biobutanol production via ABE fermentation, considering technical, environmental, and safety aspects. Previous works have assessed economic, energy, and technical aspects, whereas environmental impacts and, more remarkably, process safety issues have not been included. Modern chemical and biochemical industries are now more aware of emerging hazards, keeping them under control and minimizing the environmental impact to an acceptable minimum [33]. This work aims to understand process engineering and specific knowledge of safety and environmental technologies. Specifically, this work compares viable, safe, and environmentally friendly alternatives at a large scale for biomass valorization in Northern Colombia. The novelty of this work lies in the application of the engineering design principles, simulation, and assessment of two ABE fermentation systems at a large scale. For the first time, Waste Reduction Algorithm and Hazard Identification and Risk Assessment methodologies are used to analyze the performance of integrated upstream-downstream processing technologies to obtain bioenergy sources from cassava (Manihot esculenta Crantz) residues via ABE fermentation.
2. Materials and Methods
The first step of the method involves modeling and simulating the upstream and downstream technologies for obtaining ABE products from agricultural biomass under two different routes. The input data for performing the first task relate to the processing technology, feedstock specifications, product specifications, and plant capacity. The operating variables for process technologies are defined according to recent contributions at the lab scale available in the literature. The feedstock selection and production capacity are described below. The process simulation is performed using the Aspen Plus v.10 software. The next task is the application of technical, environmental, and process safety assessments. For this task, the input data consist of the mass and energy balances and equipment provided by process simulation of ABE fermentation routes. The technical analysis determines the plants’ Net Energy Ratio and production yield. The WARGUI software is selected to quantify potential environmental impacts following the Waste Reduction algorithm [34]. The Hazard Identification and Risk Assessment (HIRA) methodology calculates the Fire and Explosion Damage Index (FEDI) [35]. Finally, the analysis will provide insights into the environmental, sustainability, and technical performance of ABE fermentation routes. Figure 1 depicts the schematic representation of the methodological procedure followed in this work.
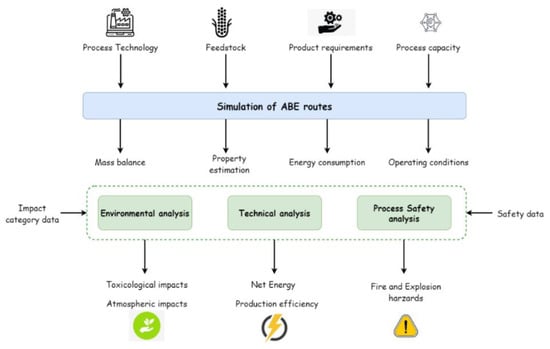
Figure 1.
Schematic representation of the methodological procedure.
2.1. Feedstock Selection
The feedstock selection was based on the availability of agricultural residues in northern Colombia and feedstock characteristics. The Supplementary Materials section shows data for crop production in Northern Colombia. The waste generation in North Colombia is considerably high for banana and cassava crops. However, the yield per hectare is lower for banana crops. As cassava yield could be increased up to 400% using improved varieties, which represent an attractive option for bioenergy sources [36,37]. The estimated energy potential of cassava waste is another criterion for selecting cassava waste as feedstock for bioenergy and bioproduct production. Aso [12] stated that up to 105,790.56 MWh/y could be provided if the total Colombian cassava output is processed, proving that producer countries such as Colombia can satisfy a portion of their energy requirements by converting such waste into bioenergy.
The processing capacity was set to be 5% of the total cassava waste generation in North Colombia (i.e., 3.1 Mt/y or 3.54 t/h). Table 2 summarizes the chemical composition of this feedstock gathered from previous contributions [38]. The production capacity is set according to local/regional demands and considering the high availability of the feedstock in the region. Moreover, the governments in this country have prioritized biofuel production in the energy matrix to shift to a more sustainable energy generation. From a global perspective, the current production rate is within reported values for big producers, such as the US, Brazil, or the European Union [39]. Cellulose and hemicellulose were modeled as glucan and xylan in the software. The thermodynamic modeling was performed using the nonrandom two-liquid (NRTL) modified with Redlich-Kwong (RK) equation. This selection was based on the accuracy of NRTL-RK to estimate activity coefficients of the handled liquid and vapor phases, respectively [34].

Table 2.
Chemical composition of cassava waste (Manihot esculenta Crantz).
2.2. Processes Description
The biomass is subjected to mechanical pretreatment by reducing particle size to 5 mm, reducing sugar recovery [40]. For route 1, the small-size biomass particles continue the pretreatment with dilute acid (H2SO4) at 1.1% w/w to make soluble sugars (i.e., xylose and glucose) accessible. The yield for hemicellulose to pentoses is 0.9 g/g, and for cellulose to hexoses, 0.07 g/g [41]. A lignin portion in the biomass is also solubilized during the pretreatment. As a subproduct of dilute acid pretreatment, the residual lignin can potentially block enzyme accessibility to cellulose for sugar synthesis [42]. This could mean the formation of inhibitors that may slow down the fermentation process accompanying these reactions, such as furfural due to pentose and hexose sugar degradation [38]. From this step, the resulting hydrolysate slurry feeds into the ammonia-based neutralization to keep the required pH to 5–6 for further hydrolysis [29]. An additional detoxification step (over-liming) is conducted to neutralize the acid [43]. For route 2, steam explosion technology is applied during biomass pretreatment under defined conditions reported by [44]. Both pretreatment technologies render the hemicellulose soluble with the formation of C5 sugars that can be quickly recovered [45]. The dissolved sugar stream is directly sent to fermentation, while the pretreated biomass is fed into enzymatic hydrolysis [46]. The yield for steam explosion indicates yields for hemicellulose to pentoses of 0.75 g/g, and for cellulose to hexoses of 0.08 g/g [47]. Figure 2 depicts route 1 block diagram 1, and Figure 3 shows route 2, including upstream and downstream units.
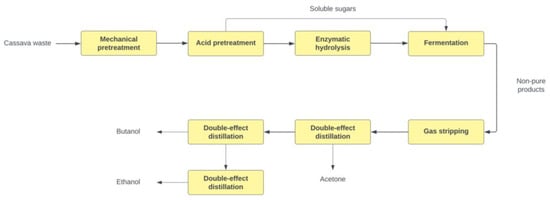
Figure 2.
Process block diagram for ABE route 1.
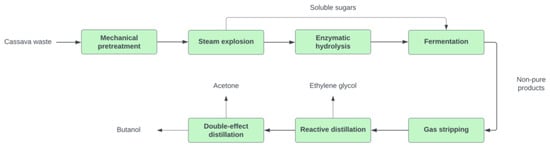
Figure 3.
Process block diagram for ABE route 2.
During hydrolysis, cellulose is transformed into fermentable sugars by the cellulase enzyme [48]. This enzyme was assumed to contain endoglucanases, exoglucanases, and b-glucosidase for polymer size reduction, chemical hydrolysis, and glucose production [49]. This step includes lignin solubilization and enzymatic saccharification, in which glucose is produced. The liquor-rich glucose is sent to fermentation while lignin-rich solid residue passes through filtration [34]. The fermentation was simulated in a continuous setting. Continuous fermentation requires fresh inoculum culture with lower residence time (compared to batch fermentation) and higher productivity due to reduced sterilization and inoculation time [50]. The inoculum preparation involved tryptone-yeast extract-acetate media comprising 20 g/L glucose, 2 g/L yeast extract, 6 g/L tryptone, and other sulfate and phosphate compounds, as performed by [51]. ABE Fermentation converts sugars into acetone, butanol, ethanol, CO2, H2, N-butyric acid, and acetic acid [29,52]. The Supplementary Materials section describes in more detail the upstream operation. A stoichiometric approach was used in all reactors based on the conversion yields reported for pretreatment [38], hydrolysis [41], and fermentation [25,53].
An assertive non-ideal behavior characterizes the stream containing ABE products because of polar compounds and azeotropic systems [54]. This system requires an accurate description of equilibria in downstream processing. The recovery stage involved modeling and simulating the liquid-liquid (LL) and vapor-liquid (VP) equilibria for the compounds involved. Henry’s Law was also applied in modeling gas solubilities in liquid phases during downstream processing (for the gas stripping). The validation of the property set is then conducted by comparing simulation results with the data found in the literature for the main product, i.e., biobutanol. The non-pure ABE products from upstream processing are subjected to in situ gas stripping involving fermentation gas (CO2 and H2) recycling via a condenser, as suggested by Outram et al. [55]. The light stream from gas stripping contains carbon dioxide as a by-product, while the bottoms contain unreacted compounds. In route 1, double-effect distillation columns were used to recover ABE products because of the azeotrope formation. The RadFrac model was used for the rigorous calculation of these columns. The Supplementary Materials section gives more details on process modeling for both upstream and downstream operations.
2.3. Technical Assessment
The net energy ratio (NER) was also calculated according to the energy balance and the higher heating value (HHV) found in the literature for cassava waste and products [56,57]. The caloric content and energy consumption were then considered to estimate the energy inputs [58], while the energy inputs included ABE products. This indicator provides information on whether the energy given for biomass conversion into biofuels is higher than the energy obtained from products. The NER analysis was completed with a Sankey diagram to determine the primary sources of energy inefficiencies. Finally, the technical assessment was complemented by analyzing the overall production yield product-feedstock for the ABE fermentation routes.
2.4. Environmental Assessment
The environmental performance of both bioenergy systems was analyzed using the computer-aided tool WAR algorithm under atmospheric and toxicological categories [59]. The atmospheric ones include global warming potential (GWP), ozone depletion potential (ODP), acidification potential (AP), and photochemical oxidation potential (PCOP). The toxicological ones are human toxicity potential by ingestion (HTPI), human toxicity potential by exposure (HTPE), aquatic toxicity potential (ATP), and terrestrial toxicity potential (TTP) [60]. The potential environmental impacts for each category were calculated by the software WARGUI using mass and energy balances. Detailed equations behind the estimation of these impacts were reported by [61]. In addition, the missing toxicological data were collected and included in the software using safety data sheets and the literature. As the core of this work is to evaluate environmental aspects of bioenergy production from cassava residues through ABE fermentation, special attention was paid to the effect of energy consumption and product nature on potential impact quantification. To this end, the following four different approaches were considered:
- Case 1. Route 1, only energy flow;
- Case 2. Route 2, only energy flow;
- Case 3. Route 1, both energy and product flows;
- Case 4. Route 2, both energy and product flows.
2.5. Process Safety Assessment
The safety aspects of ABE fermentation routes were analyzed via HIRA, which facilitates identifying operations with the highest fire and explosion risks [35]. Besides, the methodology provides a systematic procedure for the quantification of FEDI that consists of the following steps: (i) unit classification, (ii) energy factors estimation, (iii) assignment of penalties, (iv) damage potential estimation, and (v) FEDI estimation. The detailed mathematical formulation behind the application of HIRA to assess chemical processes is reported in [62]. The general equations of damage potential and FEDI are given by Equations (1) and (2), respectively, where corresponds to penalties and is the energy factor. The safety data, such as the National Fire Protection Association (NFPA) ranking, flash point, and autoignition temperature of chemical compounds handled in routes 1 and 2 were gathered from Material Safety Data Sheets (MSDSs) and contributions available in the literature [63,64,65]. The fire point was assumed to be 50–70 °F higher than the open flashpoint, as stated by [66].
HIRA and FEDI estimation include four factors and eight penalties considering the potential damage from the heat of the reactions, flammability, pressure/system stress, and safety linked with the unit operation. Four elements are derived from thermodynamics, and their designation is calculation-oriented, making them suitable for integration with process simulation and CAPE. Compared to other methodologies, the HIRA-FEDI is analogous to the Dow Fire and Explosion Index, with enhanced sensitivity to variations in operational conditions [67]. Values of penalties depend on unit classification. An intermediate step is calculating damage potential, which is used for FEDI evaluation. Again, an assumption of the distance between equipment is required to calculate one of the penalties.
3. Results
A process simulation was performed, generating mass and energy balances. Additionally, technical information about the equipment and process unit was obtained. This information was used to develop the technical, environmental, and process safety assessment of ABE fermentation routes. The economic analysis of the assessed processes was not included in this work since the authors published those results elsewhere [31]. The techno-economic assessment indicated 1.06 USD/kg ABE for route 1 and 1.165 USD/kg ABE for route 2. The results are described in more detail in the following subsections.
3.1. Process Simulation and Technical Results
Process simulation provides the mass and energy balances of ABE routes based on the described configuration. This step also requires inputs for property estimation and the process of thermodynamics. The property validation of the main products involved in the simulation (butanol, ethanol, and acetone) are described in more detail in the Supplementary Materials section. An assertive non-ideal behavior characterizes the ABE product stream because of polar compounds and azeotropic systems [54]. The recovery stage involved modeling and simulating the liquid-liquid (LL) and vapor-liquid (VP) equilibria for the compounds involved. Henry’s Law was also applied in modeling gas solubilities in liquid phases during downstream processing (for the gas stripping). The validation of the property set is then conducted by comparing simulation results with the data found in the literature for the main product, i.e., biobutanol (check Supplementary Materials). The non-pure ABE products from upstream processing are subjected to in situ gas stripping involving fermentation gas (CO2 and H2) recycling via a condenser, as suggested by [55]. The light stream from gas stripping contains carbon dioxide as a by-product, while the bottoms contain unreacted compounds. In route 1, double-effect distillation columns were used to recover ABE products because of the azeotrope formation. The RadFrac model was used for the rigorous calculation of these columns. Table S3 in the Supplementary Materials section summarizes operational information about separation units in process route 1.
Even though both routes differ significantly in the technologies used during upstream and downstream processing, the butanol production yield reached similar values, according to Table 3. The energy consumption increased by 7.8% from route 1 to route 2. The NER values for both routes were determined as part of the technical analysis. The value of NER < 1 for both routes (see Figure 4) suggests that the systems lose net energy because the energetic potential of cassava waste is higher than the energy provided by these biofuels.

Table 3.
Technical results for ABE fermentation routes.
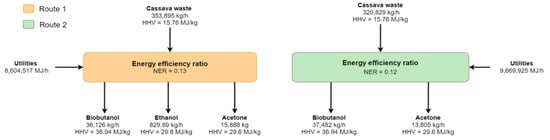
Figure 4.
Global energy balance for routes 1 and 2.
Process integration tools are strongly recommended to reduce energy consumption and gain net energy [68]. These technical results left the decision open between bioenergy systems and other process performance criteria, such as economics, that must be analyzed.
3.2. Environmental Assessment Results
Figure 5 shows the overall performance for routes 1 and 2 from an environmental point of view. The cases, including product flow, reported an increase of 0.5 and 2.9 times the output impacts estimation when considering only energy flows for routes 1 and 2, respectively. The highest total output rate of PEI was calculated at 2.08 × 105 PEI/h for route 2, followed by route 1 with 1.51 × 105 PEI/h. These results are causally related to the ABE production yields as a higher quantity of products increases the output rate. The environmental impacts per product mass were lower for cases 2 and 4 (route 2) than cases 1 and 3 (route 1), suggesting that route 2 is more environmentally benign than route 1. This outcome may be attributed to smaller loads of chemicals requiring steam explosion technology than the dilute acid pretreatment and further neutralization. The generation of PEI also confirmed these results because of the negative values for cases 2 and 4, which indicates that the conversion/purification technologies in upstream and downstream processing generate chemicals with less environmental impact than the raw materials entering the system. The total output and generation of PEI were compared with other contributions to assess the bioenergy production from biomasses.
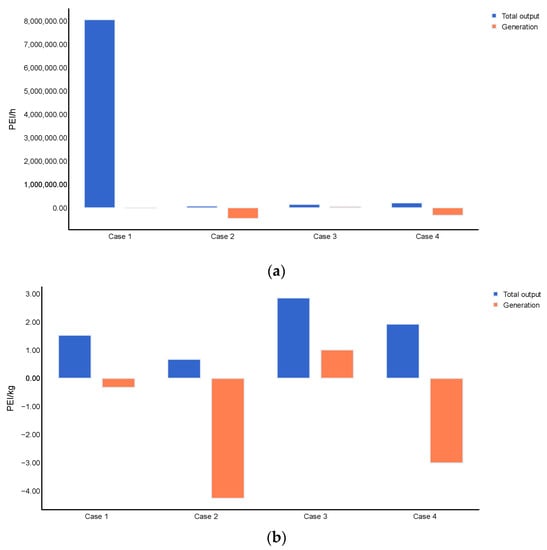
Figure 5.
Global environmental performance of routes 1 and 2, (a) PEI/h and (b) PEI/kg of product.
Figure 6 shows the estimation of the toxicological impacts of ABE fermentation. Results revealed significant contributions to the output rate of PEI from categories of HTPI and TTP. For route 1, these PEI values (cases 1 and 3) are attributed mainly to furfural and ammonia, substances handled during upstream processing which leave the system through waste streams 15, 21, 49, 71, 74, and 79. Route 2 reached lower values than route 1 for both impact categories because ammonia, sulfuric acid, and calcium hydroxide are not needed in the biomass pretreatment, leading to less generation of these related waste products. However, ethylene oxide is introduced in this route with similar normalized impact scores for HTPI and TTP compared to furfural, which affected the output PEI results. The generation of PEI was negative for most of the cases analyzed, indicating that the components of output streams are less contaminating than the feedstock [69]. These results also revealed the excellent environmental performance of this biomass conversion to bioenergy. Unlike route 2, route 1 showed positive values of PEI generation for HTPI and TTP categories when considering energy and product flows, suggesting that route 1 generates more potential environmental toxicological issues. Rincon et al. [70] found similar trends in toxicological categories for biodiesel production from five feedstocks. They reported output values below 7.00 × 10−2 PEI/kg for HTPI and TTP categories, significant contributors.
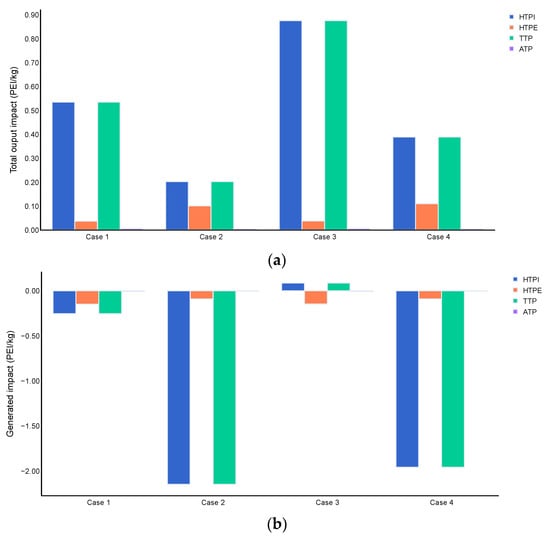
Figure 6.
Toxicological impacts of routes 1 and 2, (a) Total output impact (PEI/kg), and (b) generated impact (PEI/kg).
Figure 7 depicts the environmental performance of routes for atmospheric categories of GWP, ODP, PCOP, and AP. The highest output of PEI was reported for the PCOP category when considering energy and product flows (cases 3 and 4), standing for 3.78 and 16.6 times the values reached for cases 1 and 2, respectively. These results reveal that the nature of bioenergy products is primarily responsible for emissions causing photochemical oxidation, while the energy sources affect the performance of route 2 less than route 1. The AP category showed similar results for cases, regardless of whether the analysis counts the contribution of product flow, which suggested that the energy source is the largest contributor to acid generation from the combustion of natural gas. Route 1 reported higher values for this category than route 2, which was attributed to the higher consumption of high-pressure steam, whose production affects the acidification potential by around 2.35–8.29% [71]. The total generation of PEI was positive for all atmospheric categories. This is an expected finding if the ABE products such as butanol and ethanol deliver emissions to the environment when employing bioenergy production, which affects global warming, ozone depletion, and acidification indicators.
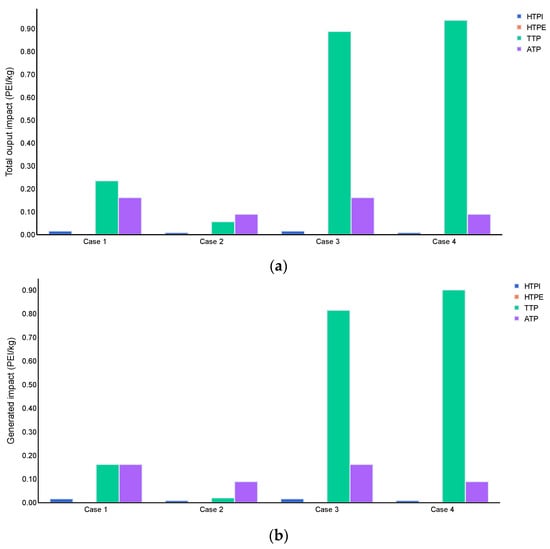
Figure 7.
Atmospheric impacts of routes 1 and 2, (a) Total output impact (PEI/kg), and (b) generated impact (PEI/kg).
3.3. Process Safety Assessment Results
A preliminary examination of fire and explosion hazards due to the active substances suggested downstream processing as the focus of process safety assessment via HIRA methodology. Table 4 lists the process units for routes 1 and 2 with the highest damage potentials, which confirmed that the ABE products and by-products obtained after the fermentation reaction and sent to the downstream processing were the largest contributors to the safety performance of both routes. The estimation of FEDI is depicted in Figure 8 for the selected steps.

Table 4.
Estimation of potential damage for most hazardous processing units in ABE routes.
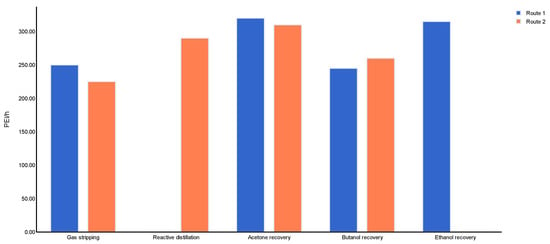
Figure 8.
FEDI results for ABE routes per processing steps.
For route 1, the recovery and purification of acetone and ethanol reached the highest FEDI values (>300), which are within the range of the “hazardous” characterization category. Due to the safety data (flammability) of acetone and ethanol, it was expected that the process would achieve categories such as “highly hazardous” or “extremely hazardous”; however, the operating temperature and pressure were not extreme enough to boost the FEDI value above 400. For route 2, the FEDI value of the acetone recovery and purification step was 301.43, a decrease of 3% from route 1. Route 2 showed fewer fire and explosion risks for most processing steps than route 1. The butanol purification/recovery section obtained a FEDI of 242.5 for route 1 and 264.05 for route 2. This outcome was associated with a higher butanol yield for the second ABE process.
4. Discussion
To analyze more in detail the technical and energy performance, wheel diagrams were drawn in both ABE technologies (see Figure 9). This allows the determination of the primary sources of inefficiencies. In route 1, the pretreatment stage accounts for the highest impact of energy consumption in the system, with a share of 49%. This outcome suggests that energy settings might need improvement strategies to decrease energy usage in this section. Downstream processing comes next, with 29% of the total energy consumption. These results are expected since many distillation towers and separation systems account for this process’s utility consumption. For route 2, the share of consumption in the pretreatment is even higher than in route 1, with 63%. This outcome confirms the energy optimization potential of this process section in both routes.
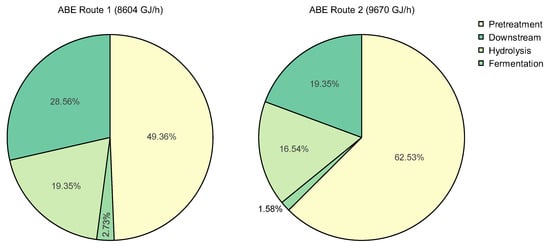
Figure 9.
Energy consumption breakdown per stage of ABE routes.
To benchmark and compare the environmental performance of ABE routes, a comparison with analogous or similar processes was performed. Table 5 summarizes the results reported in previous works. The comparison shows that the output rate of PEI per amount of butanol for routes 1 and 2 was slightly higher than similar biomasses transformation into bioenergy products. Dissimilar results were found considering the compared bioenergy production systems. Compared to hydrogen production from oil palm residues, better environmental performance is obtained in ABE fermentation from cassava waste. Conversely, transformation processes from crude bio-oil and oil palm residues to crude bio-oil, oil blends, and bioethanol. Yet, the difference is not significantly higher, and the impacts evaluated are associated with gate-to-gate boundaries.

Table 5.
Comparison of potential environmental impacts with the literature on bioenergy systems.
In terms of safety performance, other contributions have reported higher magnitudes based on the HIRA methodology for similar flammable substances. For example, Moreno-Sader et al. [74] applied and assessed a hydrogen compressor subsystem to a hydrocracking plant, finding FEDI values above 1000. The operating conditions of the unit were 16,500 kPa and 260 °C. Compared with the ABE fermentation routes for butanol production, the maximum average temperature and pressure were 111°C and 1 atm, respectively. This indicates that the severity of the operational conditions, along with the nature of the components, is strongly related to the performance of processes and units. As there is not much information on bio-based processes’ safety and flammability aspects, more assessments and research are needed in that direction.
5. Conclusions
Two ABE fermentation-based bioenergy systems were designed, simulated, and compared by applying process safety and environmental impact assessments. These systems addressed the conversion of cassava waste to ABE products within an integrated downstream and upstream processing. The computer-aided process engineering tools allowed the ABE fermentation technologies to be evaluated under large-scale conditions. From a technical perspective, both routes proved to be quite competitive, considering the yields and product distribution. The performance indicators evaluated in this work (production yield, PEIs, and FEDI) were globally superior for route 2 than route 1. The butanol production yield obtained was within the range of 0.10–0.12 kg/kg feedstock, while the NER reached 0.13–0.12. NER < 1 indicates that the energy performance of both routes requires improvement by applying optimization strategies, such as process integration methodologies. The inclusion of a heat exchange network could reduce utility consumption. According to the global results for environmental analysis, route 2 reached the highest total output of PEI (5.56 PEI/kg butanol), followed by route 1 (4.21 PEI/kg butanol), which had values within the range of similar fermentation routes from a wide variety of lignocellulosic feedstocks.
The process safety assessment showed FEDI values above 300 for acetone and ethanol recovery/purification. Due to the flammability of the handled substances, both routes fall within the “hazardous” characterization category, whereas their relative performance is almost tied. This work demonstrated the possibility of aligning design principles and technical assessments to boost sustainability-driven food waste to bioenergy systems. This work also provides insights into ABE fermentation technology from environmental and process safety perspectives to diagnose emerging hazards, enhance process control, and minimize the environmental impact. Specifically, this work compares viable, safe, and environmentally friendly alternatives at a large scale for biomass valorization in North Colombia. Our results represent a first step toward the sustainable performance of bioenergy systems derived from agricultural biomasses for industrial applications in the field of renewable energy in North Colombia. Future works might include the development of mathematical optimization strategies to reduce the potential impact on the physical environment and minimize the potential risk of biorefineries. In addition, other process analysis tools could be implemented to reach more comprehensive levels of analysis. These can include data analysis, uncertainties, or supply chain analysis, among others, to take the applied assessment to the next level.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/su142316185/s1, Figure S1: Available agricultural residues and yields of local crops in Bolivar; Figure S2: Simulation flowsheet for route 1: (a) hierarchy blocks of upstream and downstream processing, (b) pretreatment, (c) hydrolysis, (d) fermentation and (e) purification; Figure S3: Simulation flowsheet for route 2: (a) hierarchy blocks of upstream and downstream processing, (b) pretreatment, (c) hydrolysis, (d) fermentation and (e) purification; Table S1: Gathered data for upstream processing simulation in route 1; Table S2: Gathered data for upstream processing simulation in route 2; Table S3: Gathered data for downstream processing simulation in route 1; Table S4: Gathered data for downstream processing simulation in route 2; Table S5: Gathered data for downstream processing simulation in route 1; Table S6: Gathered data for downstream processing simulation in route 2; Table S7: Estimation of relevant properties for biobutanol using Aspen Plus; Table S8: Estimation of relevant properties for ethanol using Aspen Plus; Table S9: Estimation of relevant properties for acetone using Aspen Plus; Table S10: Operating data of the main process streams for route 1; Table S11: Operating data of the main process streams for route 2. References [25,27,28,29,31,34,41,46,53,55,63,64,75,76,77,78,79,80,81,82,83,84,85,86,87,88,89,90] are cited in the Supplementary Materials.
Author Contributions
Conceptualization, S.M. and A.G.-D.; methodology, S.M.; software, A.G.-D.; validation, A.G.-Q., A.G.-D. and S.M.; formal analysis, S.M.; investigation, S.M.; resources, A.G.-Q. and A.G.-D.; data curation, S.M.; writing—original draft preparation, S.M.; writing—review and editing, A.G.-Q.; visualization, S.M.; supervision, A.G.-D.; project administration, A.G.-D.; funding acquisition, A.G.-Q. and A.G.-D. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Acknowledgments
The authors thank the University of Cartagena for providing the financial support to conduct this research. The authors are thankful to the Novo Nordisk Center for Biosustainability for supporting the development of this work. A.Q.-G. acknowledges the support from the Universidad del Norte.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Poore, J.; Nemecek, T. Reducing food’s environmental impacts through producers and consumers. Science 2018, 360, 987–992. [Google Scholar] [CrossRef] [PubMed]
- FAO, US. The State of the World’s Land and Water Resources: Managing Systems at Risk. Available online: http://www.fao.org/3/i1688e/i1688e.pdf (accessed on 13 August 2022).
- Yaashikaa, P.R.; Senthil, K.P.; Varjani, S. Valorization of agro-industrial wastes for biorefinery process and circular bioeconomy: A critical review. Bioresour. Technol. 2022, 343, 126126. [Google Scholar] [CrossRef]
- NL Agency. Biomass Opportunities in Colombia; Ministry of Foreign Affairs: The Hague, The Netherlands, 2013. [Google Scholar]
- Caicedo-Ortiz, J.G.; De-la-Hoz-Franco, E.; Morales, R.; Piñeres-Espitia, G.; Combita-Niño, H.; Estévez, F.; Cama-Pinto, A. Monitoring system for agronomic variables based in WSN technology on cassava crops. Comput. Electron. Agric. 2018, 145, 275–281. [Google Scholar] [CrossRef]
- Sivamani, S.; Chandrasekaran, A.; Balajii, M.; Shanmugaprakash, M.; Hosseini-Bandegharaei, A.; Baskar, R. Evaluation of the potential of cassava-based residues for biofuels production. Rev. Env. Sci. BioTechnol. 2018, 17, 553–570. [Google Scholar] [CrossRef]
- Ubalua, A. Cassava wastes: Treatment options and value addition alternatives. Afr. J. Biotechnol. 2007, 6, 2065–2073. [Google Scholar]
- Zhang, M.; Xie, L.; Yin, Z.; Khanal, S.; Zhou, Q. Biorefinery approach for cassava-based industrial wastes: Current status and opportunities. Bioresour. Technol. 2016, 215, 50–62. [Google Scholar] [CrossRef]
- Veiga, J.P.S.; Valle, T.L.; Feltran, J.C.; Bizzo, W. Characterization and productivity of cassava waste and its use as an energy source. Renew Energy 2016, 93, 691–699. [Google Scholar] [CrossRef]
- Yang, L.; Wang, X.; Dai, M.; Chen, B.; Qiao, Y.; Deng, H.; Zhang, D.; Zhang, Y.; Villas Bôas de Almeida, C.; Chiu, A.S.F.; et al. Shifting from fossil-based economy to bio-based economy: Status quo, challenges, and prospects. Energy 2021, 228, 120533. [Google Scholar] [CrossRef]
- Meramo-Hurtado, S.-I.; González-Delgado, Á.-D. Biorefinery synthesis and design using sustainability parameters and hierarchical/3D multi-objective optimization. J. Clean Prod. 2019, 240, 118134. [Google Scholar] [CrossRef]
- Aso, S.; Teixeira, A.; Achinewhu, S. Cassava Residues Could Provide Sustainable Bioenergy for Cassava Producing Nations. In Cassava; IntechOpen: London, UK, 2017; pp. 219–240. [Google Scholar]
- Okudoh, V.; Trois, C.; Workneh, T.; Schmidt, S. The potential of cassava biomass and applicable technologies for sustainable biogas production in South Africa: A review. Renew Sustain. Energy Rev. 2014, 39, 1035–1052. [Google Scholar] [CrossRef]
- Pandey, A.; Soccol, C.; Nigam, P.; Soccol, V.; Vandenberghe, L.; Mohan, R. Biotechnological potential of agro-industrial residues. II: Cassava bagasse. Bioresour. Technol. 2000, 74, 81–87. [Google Scholar] [CrossRef]
- Aguado-Deblas, L.; López-Tenllado, F.J.; Luna, D.; Romero, F.; Estevez, R. Advanced Biofuels from ABE (Acetone/Butanol/Ethanol) and Vegetable Oils (Castor or Sunflower Oil) for Using in Triple Blends with Diesel: Evaluation on a Diesel Engine. Materials 2022, 15, 6493. [Google Scholar] [CrossRef] [PubMed]
- Johnravindar, D.; Elangovan, N.; Olaganathan, N.; Muthaiyan, A.; Fei, Q. Biobutanol production from cassava waste residue using Clostridium sp. AS3 in batch culture fermentation. Biofuels 2019, 12, 1259–1266. [Google Scholar] [CrossRef]
- Kumar, M.; Goyal, Y.; Sarkar, A.; Gayen, K. Comparative economic assessment of ABE fermentation based on cellulosic and non-cellulosic feedstocks. Appl. Energy 2012, 93, 193–204. [Google Scholar] [CrossRef]
- Kushwaha, D.; Srivastava, N.; Mishra, I.; Upadhyay, S.N.; Mishra, P. Recent trends in biobutanol production. Rev. Chem. Eng. 2019, 35, 475–504. [Google Scholar] [CrossRef]
- Sarchami, T.; Munch, G.; Johnson, E.; Kießlich, S.; Rehmann, L. A Review of Process-Design Challenges for Industrial Fermentation of Butanol from Crude Glycerol by Non-Biphasic Clostridium pasteurianum. Fermentation 2016, 2, 13. [Google Scholar] [CrossRef]
- Cebreiros, F.; Risso, F.; Cagno, M.; Cabrera, M.; Rochón, E.; Jauregui, G.; Boix, E.; Böthig, S.; Ferrari, M.; Lareo, C. Enhanced production of butanol and xylosaccharides from Eucalyptus grandis wood using steam explosion in a semi-continuous pre-pilot reactor. Fuel 2021, 290, 119818. [Google Scholar] [CrossRef]
- Contreras-Vargas, C.A.; Gómez-Castro, F.I.; Sánchez-Ramírez, E.; Segovia-Hernández, J.; Morales-Rodríguez, R.; Gamiño-Arroyo, Z. Alternatives for the Purification of the Blend Butanol/Ethanol from an Acetone/Butanol/Ethanol Fermentation Effluent. Chem. Eng. Technol. 2019, 42, 1088–1100. [Google Scholar] [CrossRef]
- Patrascu, J.; Sorin, C.; Kiss, A. Eco-efficient Downstream Processing of Biobutanol by Enhanced Process Intensification and Integration. ACS Sustain. Chem. Eng. 2018, 6, 5452–5461. [Google Scholar] [CrossRef]
- Qi, G.; Xiong, L.; Li, H.; Huang, Q.; Luo, M.; Tian, L.; Chen, X.; Huang, C.; Chen, X. Hydrotropic pretreatment on wheat straw for efficient biobutanol production. Biomass Bioenergy 2019, 122, 76–83. [Google Scholar] [CrossRef]
- Kang, S.; Fu, J.; Zhang, G. From lignocellulosic biomass to levulinic acid: A review on acid-catalyzed hydrolysis. Renew Sustain. Energy Rev. 2018, 94, 340–362. [Google Scholar]
- Darkwah, K.; Nokes, S.E.; Seay, J.R.; Knutson, B. Mechanistic simulation of batch acetone–butanol–ethanol (ABE) fermentation with in situ gas stripping using Aspen PlusTM. Bioprocess Biosyst. Eng. 2018, 41, 1283–1294. [Google Scholar] [CrossRef] [PubMed]
- Lodi, G.; Pellegrini, L.A. Recovery of butanol from abe fermentation broth by gas stripping. Chem. Eng. Trans. 2016, 49, 13–18. [Google Scholar]
- Lodi, G.; De Guido, G.; Pellegrini, L.A. Simulation and energy analysis of the ABE fermentation integrated with gas stripping. Biomass Bioenergy 2018, 116, 227–235. [Google Scholar] [CrossRef]
- Kaymak, D.B. Design and Control of an Alternative Process for Biobutanol Purification from ABE Fermentation. Ind. Eng. Chem. Res. 2019, 58, 1957–1965. [Google Scholar] [CrossRef]
- Haigh, K.F.; Petersen, A.M.; Gottumukkala, L.; Mandegari, M.; Naleli, K.; Gorgens, J. Simulation and comparison of processes for biobutanol production from lignocellulose via ABE fermentation. Biofuels Bioprod. Biorefining 2018, 12, 1023–1036. [Google Scholar] [CrossRef]
- Jang, M.O.; Choi, G. Techno-economic analysis of butanol production from lignocellulosic biomass by concentrated acid pretreatment and hydrolysis plus continuous fermentation. Biochem. Eng. J. 2018, 134, 30–43. [Google Scholar] [CrossRef]
- Meramo-Hurtado, S.I.; González-Delgado, Á.; Rehmann, L.; Quinones-Bolanos, E.; Mehvar, M. Comparative analysis of biorefinery designs based on acetone-butanol-ethanol fermentation under exergetic, techno-economic, and sensitivity analyses towards a sustainability perspective. J. Clean Prod. 2021, 298, 126761. [Google Scholar] [CrossRef]
- Martinez-Gomez, J.; Sánchez-Ramírez, E.; Quiroz-Ramírez, J.; Segovia-Hernandez, J.; Ponce-Ortega, J.; El-Halwagi, M. Involving economic, environmental and safety issues in the optimal purification of biobutanol. Process Saf. Env. Prot 2016, 103, 365–376. [Google Scholar] [CrossRef]
- Warnasooriya, S.; Gunasekera, M.Y. Assessing inherent environmental, health and safety hazards in chemical process route selection. Process Saf. Env. Prot. 2017, 105, 224–236. [Google Scholar] [CrossRef]
- Carmona-Garcia, E.; Ortiz-Sánchez, M.; Cardona Alzate, C.A. Analysis of the Coffee Cut Stems as Raw Material for the Production of Sugars for Acetone–Butanol–Ethanol (ABE) Fermentation: Techno-Economic Analysis. Waste Biomass Valorization 2019, 10, 3793–3808. [Google Scholar] [CrossRef]
- Sunaryo; Hamka, M.A. Safety risks assessment on container terminal using hazard identification and risk assessment and fault tree analysis methods. Procedia Eng. 2017, 194, 307–314. [Google Scholar] [CrossRef]
- Cardona, A. Aumenta El Consumo De La Yuca Industrial En Colombia Con Mejoras En Variedades. Agronegocios 2019, 1. Available online: https://www.agronegocios.co/agricultura/aumenta-el-consumo-de-la-yuca-industrial-en-colombia-con-mejoras-en-variedades-2820472#:~:text=Agro-,Aumenta%20el%20consumo%20de%20la%20yuca%20industrial%20en%20Colombia%20con,al%20consumo%20de%20este%20tipo.&text= (accessed on 24 September 2022).
- Kotoka, F.; Tulashie, S.; Setsoafia, D. Production of bioethanol from liquid waste from cassava dough during gari processing. Biofuels 2017, 10, 1–9. [Google Scholar] [CrossRef]
- Niño, L.; Acosta, A.; Gelves, R. Evaluation of chemical pretreatments for enzymatic hydrolysis of lignocellulosic residues cassava (Manihot esculenta Crantz). Rev. Fac. Ing. Univ. Antioquia 2013, 69, 317–326. [Google Scholar]
- Qureshi, N.; Lin, X.; Liu, S.; Saha, B.C.; Mariano, A.P.; Polaina, J.; Ezeji, T.C.; Friedl, A.; Maddox, I.S.; Klasson, K.T.; et al. Global View of Biofuel Butanol and Economics of Its Production by Fermentation from Sweet Sorghum Bagasse, Food Waste, and Yellow Top Presscake: Application of Novel Technologies. Fermentation 2020, 6, 58. [Google Scholar] [CrossRef]
- Julio-Altamiranda, Y.; Mercado-Pacheco, J.; Sánchez-Tuirán, E.; González-Delgado, A.; Ojeda, K. Evaluation of mechanical-green solvent pretreatment of oil palm wastes for reducing sugars production in North-Colombia. Sustain. Chem. Pharm. 2020, 16, 100256. [Google Scholar] [CrossRef]
- Luo, L.; van der Voet, E.; Huppes, G. Biorefining of lignocellulosic feedstock—Technical, economic and environmental considerations. Bioresour. Technol. 2010, 101, 5023–5032. [Google Scholar] [CrossRef]
- Li, H.; Xiong, L.; Chen, X.; Li, H.; Qi, G.; Huang, C.; Luo, M.; Chen, X. Enhanced enzymatic hydrolysis and acetone-butanol-ethanol fermentation of sugarcane bagasse by combined diluted acid with oxidate ammonolysis pretreatment. Bioresour. Technol. 2017, 228, 257–263. [Google Scholar] [CrossRef]
- Martinez, A.; Rodriguez, M.E.; Wells, M.L.; York, S.W.; Preston, J.; Ingram, L.O. Detoxification of Dilute Acid Hydrolysates of Lignocellulose with Lime. Biotechnol. Prog. 2001, 17, 287–293. [Google Scholar] [CrossRef]
- Procentese, A.; Guida, T.; Raganati, F.; Salatino, P.; Marzocchella, A. Process simulation of biobutanol production from lignocellulosic feedstocks. Chem. Eng. Trans. 2014, 38, 343–348. [Google Scholar]
- Ndaba, B.; Chiyanzu, I.; Marx, S. N-Butanol derived from biochemical and chemical routes: A review. Biotechnol. Rep. 2015, 8, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Jin, Q.; Qureshi, N.; Wang, H.; Huang, H. Acetone-butanol-ethanol (ABE) fermentation of soluble and hydrolyzed sugars in apple pomace by Clostridium beijerinckii P260. Fuel 2019, 244, 536–544. [Google Scholar] [CrossRef]
- Wang, L.; Kong, F.; Chen, H. Steam Explosion Pretreatment and Saccharification of Lignocellulosic Biomass. In Handbook of Biorefinery Research and Technology; Springer: Berlin/Heidelberg, Germany, 2018; pp. 1–14. [Google Scholar]
- Yang, M.; Wang, J.; Nan, Y.; Zhang, J.; Kuittinen, S.; Pappinen, A.; Li, L.; Liu, G. Effect of salts formed by neutralization for the enzymatic hydrolysis of cellulose and acetone–butanol–ethanol fermentation. RSC Adv. 2019, 9, 33755–33760. [Google Scholar] [CrossRef] [PubMed]
- González Quiroga, A.; Bula Silvera, A.; Vasquez Padilla, R.; Da Costa, A.C.; Maciel Filho, R. Continuous and semicontinuous reaction systems for high-solids enzymatic hydrolysis of lignocellulosics. Braz. J. Chem. Eng. 2015, 32, 805–819. [Google Scholar] [CrossRef][Green Version]
- Panpatte, D.G.; Jhala, Y.K. Agricultural Waste: A Suitable Source for Biofuel Production; Springer: Berlin/Heidelberg, Germany, 2019. [Google Scholar] [CrossRef]
- Al-Shorgani, N.K.N.; Al-Tabib, A.I.; Kadier, A.; Zanil, M.F.; Lee, K.M.; Kalil, M. Continuous Butanol Fermentation of Dilute Acid-Pretreated De-oiled Rice Bran by Clostridium acetobutylicum YM1. Sci. Rep. 2019, 9, 4622. [Google Scholar] [CrossRef]
- Niglio, S.; Marzocchella, A.; Rehmann, L. Clostridial conversion of corn syrup to Acetone-Butanol-Ethanol (ABE) via batch and fed-batch fermentation. Heliyon 2019, 5, e01401. [Google Scholar] [CrossRef]
- Naleli, K. Process Modelling in Production of Biobutanol from Lignocellulosic Biomass via ABE Fermentation. Ph.D. Thesis, Stellenbosch University, Stellenbosch, South Africa, 2016; pp. 63–66. [Google Scholar]
- Kurkijärvi, A.; Lehtonen, J.; Linnekoski, J. Novel dual extraction process for acetone–butanol–ethanol fermentation. Sep. Purif. Technol. 2014, 124, 18–25. [Google Scholar] [CrossRef]
- Outram, V.; Lalander, C.A.; Lee, J.G.M.; Davies, E.; Harvey, A. Applied in situ product recovery in ABE fermentation. Biotechnol. Prog. 2017, 33, 563–579. [Google Scholar] [CrossRef]
- Cruz, G.; Pereira, A.; Ferreira, D.; Costa, W. Physical-Chemical characterization and thermal behavior of cassava harvest waste for application in thermochemical processes. J. Therm. Anal. Calorim. 2020, 143, 3611–3622. [Google Scholar] [CrossRef]
- Dincer, I. Comprehensive Energy Systems; Elsevier: Amsterdam, The Netherlands, 2018. [Google Scholar]
- Ninõ-Villalobos, A.; Puello-Yarce, J.; González-Delgado, Á.D.; Ojeda, K.A.; Sánchez-Tuirán, E. Biodiesel and Hydrogen Production in a Combined Palm and Jatropha Biomass Biorefinery: Simulation, Techno-Economic, and Environmental Evaluation. ACS Omega 2020, 5, 7074–7084. [Google Scholar] [CrossRef]
- Meramo-Hurtado, S.; Urbina-Suarez, N.; González-Delgado, Á. Computer-aided environmental and exergy analyses of a large-scale production of chitosan microbeads modified with TiO2 nanoparticles. J. Clean Prod. 2019, 273, 117804. [Google Scholar] [CrossRef]
- Smith, R.L.; Ruiz-Mercado, G.J.; Gonzalez, M.A. Using GREENSCOPE indicators for sustainable computer-aided process evaluation and design. Comput. Chem. Eng. 2015, 81, 272–277. [Google Scholar] [CrossRef]
- Arteaga-Díaz, S.J.; Meramo-Hurtado, S.I.; León-Pulido, J.; Zuorro, A.; González-Delgado, A. Environmental assessment of large scale production of magnetite (Fe3O4) nanoparticles via coprecipitation. Appl. Sci. 2019, 9, 1682. [Google Scholar] [CrossRef]
- Moreno-Sader, K.; Alarcón-Suesca, C.; González-Delgado, A.D. Application of environmental and hazard assessment methodologies towards the sustainable production of crude palm oil in North-Colombia. Sustain. Chem. Pharm. 2020, 15, 100221. [Google Scholar] [CrossRef]
- Jones, D.T. Biobutanol. In Biotechnology-Fundamentals in Biotechnology; Encyclopedia of Life Support Systems (EOLSS); EOLSS Publications: Oxford, UK, 2009; pp. 98–134. [Google Scholar]
- Lapuerta, M.; Ballesteros, R.; Barba, J. Strategies to Introduce n-Butanol in Gasoline Blends. Sustainability 2017, 9, 589. [Google Scholar] [CrossRef]
- Carpenter, N. Chemistry of Sustainable Energy; CRC Press: Boca Raton, FL, USA, 2014. [Google Scholar]
- Grace, R. Special Conditions Problems and Procedures in Well Control. In Blowout and Well Control Handbook; Gulf Professional Publishing: Houston, TX, USA, 2017; pp. 121–209. [Google Scholar]
- Janošovský, J.; Rosa, I.; Vincent, G.; Šulgan, B.; Variny, M.; Labovská, Z.; Labovský, J.; Jelemenský, L. Methodology for selection of inherently safer process design alternatives based on safety indices. Process Saf. Env. Prot. 2022, 160, 513–526. [Google Scholar] [CrossRef]
- Dickson, R.; Mancini, E.; Garg, N.; Woodley, J.; Gernaey, K.; Pinelo, M.; Liu, J.; Mansouri, S. Sustainable bio-succinic acid production: Superstructure optimization, techno-economic, and lifecycle assessment. Energy Env. Sci. 2021, 14, 3542–3558. [Google Scholar] [CrossRef]
- Moreno-Sader, K.; Meramo-Hurtado, S.I.; González-Delgado, A.D. Computer-aided environmental and exergy analysis as decision-making tools for selecting bio-oil feedstocks. Renew Sustain. Energy Rev. 2019, 112, 42–57. [Google Scholar] [CrossRef]
- Rincon, B.; Altamar, M.; Castilla, E.; Martinez-Consuegra, J.; Gonzalez-Delgado, A.D. Evaluation of Hydrothermal Liquefaction of Chlorella sp. for Biocrude Production Using Computer-aided Exergy Analysis. Contemp. Eng. Sci. 2018, 11, 1359–1366. [Google Scholar] [CrossRef][Green Version]
- Usubharatana, P.; Phungrassami, H. Life Cycle Assessment for Enhanced Efficiency of Small Power Plants by Reducing Air Input Temperature. Pol. J. Env. Stud. 2018, 27, 1781–1793. [Google Scholar] [CrossRef]
- Gonzalez-Delgado, A.D.; Parejo, V.; Herrera, T. Computer-aided environmental evaluation of bio-hydrogen production from residual biomass of palm cultivation. Contemp. Eng. Sci. 2017, 10, 773–783. [Google Scholar] [CrossRef]
- Montoya, M.; Quintero, J.; Sánchez, O.; Cardona, C. Environmental impact assessment for ethanol production process using the waste reduction algorithm. Rev. Fac. Ing. Univ. Antioq. 2006, 36, 85–95. [Google Scholar]
- Moreno-Sader, K.; Jain, P.; Tenorio, L.C.B.; Mannan, M.S.; El-Halwagi, M.M. Integrated Approach of Safety, Sustainability, Reliability, and Resilience Analysis via a Return on Investment Metric. ACS Sustain. Chem. Eng. 2019, 7, 19522–19536. [Google Scholar] [CrossRef]
- Ministerio de Agricultura y Desarrollo Rural. Producción y Rendimiento Nacional por Cultivo; Ministerio de Agricultura y Desarrollo Rural: Bogotá, Colombia, 2018. [Google Scholar]
- López-Arenas, T.; Rathi, P.; Ramírez-Jiménez, E.; Sales-Cruz, M. Factors affecting the acid pretreatment of lignocellulosic biomass: Batch and continuous process. Comput. Aided Chem. Eng. 2010, 28, 979–984. [Google Scholar]
- Wooley, R.; Ruth, M.; Sheehan, J.; Ibsen, K.; Majdeski, H.; Galvez, A. Lignocellulosic Biomass to Ethanol Process Design and Economics Utilizing Co-Current Dilute Acid Prehydrolysis and Enzymatic Hydrolysis Current and Futuristic Scenarios; National Renewable Energy Lab. (NREL): Golden, CO, USA, 1999. [Google Scholar]
- El-Naggar, N.E.A.; Deraz, S.; Khalil, A. Bioethanol production from lignocellulosic feedstocks based on enzymatic hydrolysis: Current status and recent developments. Biotechnology 2014, 13, 1–21. [Google Scholar]
- Carvajal, J.C.; Gómez, Á.; Cardona, C.A. Comparison of lignin extraction processes: Economic and environmental assessment. Bioresour. Technol. 2016, 214, 468–476. [Google Scholar] [CrossRef]
- Duque, A.; Manzanares, P.; Ballesteros, I.; Ballesteros, M. Steam Explosion as Lignocellulosic Biomass Pretreatment; Elsevier Inc.: Madrid, Spain, 2016. [Google Scholar]
- Chum, H.L.; Douglas, L.J.; Feinberg, D.A.; Schroeder, H.A. Evaluation of Pretreatments of Biomass for Enzymatic Hydrolysis of Cellulose; Solar Energy Research Inst.: Golden, CO, USA, 1985; pp. 1–64. [Google Scholar]
- Ciric, A.R.; Mumtaz, H.S.; Corbett, G.; Reagan, M.; Seider, W.D.; Fabiano, L.A.; Kolesar, D.M.; Widagdo, S. Azeotropic distillation with an internal decanter. Comput. Chem. Eng. 2000, 24, 2435–2446. [Google Scholar] [CrossRef]
- An, W.; Lin, Z.; Chen, J.; Zhu, J. Simulation and analysis of a reactive distillation column for removal of water from ethanol-water mixtures. Ind. Eng. Chem. Res. 2014, 53, 6056–6064. [Google Scholar] [CrossRef]
- Brandon, B.; Ezike, R. Ethanol and Butanol: Symbiotic partners for a modern fuel. Biofuels Digest 2015, 1. Available online: https://www.biofuelsdigest.com/bdigest/2015/06/11/ethanol-and-butanol-symbiotic-partners-for-a-modern-fuel/ (accessed on 24 September 2022).
- Anyanwu, R.C.; Rodriguez, C.; Durrant, A.; Ramadan, M.; Olabi, A.G. Micro-Macroalgae Properties and Applications. In Reference Module in Materials Science and Materials Engineering; Elsevier: Amsterdam, The Netherlands, 2018; pp. 1–28. [Google Scholar]
- Bank, D.D. Vapor Pressure of Ethanol. Available online: http://www.ddbst.com/en/EED/PCP/VAP_C11.php (accessed on 4 June 2022).
- Mejía, A.; Segura, H.; Cartes, M. Vapor-Liquid Equilibria and Interfacial Tensions of the System Ethanol + 2-Methoxy-2-methylpropane. J. Chem. Eng. Data 2010, 55, 428–434. [Google Scholar] [CrossRef]
- Majer, V.; Svoboda, V. Enthalpies of Vaporization of Organic Compounds: A Critical Review and Data Compilation; Blackwell Scientific Publications: Boston, UK, 1985; p. 300. [Google Scholar]
- Engineering Toolbox. Acetone—Thermophysical Properties. Available online: https://www.engineeringtoolbox.com/acetone-2-propanone-dimethyl-ketone-properties-d_2036.html (accessed on 4 June 2022).
- Ambrose, D.; Ellender, J.H.; Lees, E.B.; Sprake, C.H.; Townsend, R. Thermodynamic properties of organic oxygen compounds XXXVIII. Vapour pressures of some aliphatic ketones. J. Chem. Thermodyn. 1975, 7, 453–472. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).