Abstract
Lactic acid is an important biochemical product. With the global pollution caused by plastics, especially marine plastics, the demand for lactic acid to produce polylactic acid has rapidly increased. However, the high costs of raw materials and fermentation–separation processes have severely limited lactic acid production. In this study, the research trend on lactic acid fermentation in recent years was analyzed by a bibliometric survey, and the latest progress in lactic acid fermentation using different biomass stocks and microorganisms is summarized. The effects of different fermentation modes and fermentation–separation coupling methods on lactic acid fermentation were analyzed. Finally, microbial strains for cooperative fermentation and polysaccharide utilization are discussed. It is meaningful to develop environmentally friendly, cost-effective in situ product removal technologies, use lactic acid as an intermediate to higher value-added products, and co-produce lactic acid and other products based on a biorefinery model.
1. Introduction
Lactic acid (LA) is an important biochemical product that had been widely used in food, beverages, household chemicals, pharmaceuticals, petrochemicals, textiles, cigarettes, antibacterial, sour, flavoring agents and preservatives [1,2]. Meanwhile, polylactic acid (PLA), a polymer of LA, is a kind of macromolecule material with good biodegradability and biocompatibility. It can be used not only for the production of disposable tableware, shopping and plastic bags, agricultural films, and hygiene and other biodegradable products but modern medical materials with good biocompatibility, such as orthopedic fixation materials, drug release materials, and surgical sutures [3,4]. Polylactic acid can be easily decomposed naturally and metabolized into carbon dioxide and water without other residues by various microorganisms and enzymes [5]. Therefore, it is considered to be a most promising biodegradable polymer material.
At present, the annual global output of petroleum based plastics is approximately 3 billion tons. If PLA could be used as a new material to replace 10–20% of petroleum-based polymers to produce plastics, then the demand for PLA would reach to 3000–6000 million tons per year. Therefore, reducing the production cost of PLA is an urgent problem to improve the competitiveness of PLA plastics. The cost of PLA is mainly derived from LA. Currently, most LA is produced through biological fermentation, in which more than 34% of the cost comes from the feedstock [6]. There are two ways to reduce the costs: finding cheap raw feedstock for fermentation and improving production technology.
The raw feedstock for industrial LA fermentative production are mainly starch-based (corn, cassava, potatoes) and sugar-based substrates (sugarcane and sugar beet), which are expensive [3]. Using renewable materials such as lignocellulosic biomass to produce LA would significantly reduce the production cost [7]. In addition, selecting highly efficient LA producing microorganisms has always been a focus of research. Most of the LA producing-microorganisms can produce L-LA, such as Lactobacillus manihotivorans LMG 18010, L. lactis, Streptococcus bovis 148, and L. plantarum A6; however, L. delbrueckii is the only known pure D-LA producer [8]. In recent years, genetically engineered bacteria have attracted considerable attention, because of their ability to be modified to meet different production requirements. Furthermore, different fermentation methods also have different effects on LA production. Especially, in situ separation technology, which can reduce product inhibition simultaneously during the fermentation process. Therefore, another present research area is to combine separation technology with the fermentation system to maximize the yield and purity of LA.
Based on a bibliometric survey, this study summarized and analyzed the latest research progress in LA production from different biomass types by various LA-producing microorganisms. It also summarized and compared the development of LA fermentation in recent years based on four aspects: raw feedstock for fermentation, microorganisms, fermentation modes, and LA separation.
2. Method of Bibliometric Analysis for Research Tendencies
In this study, keywords ((“lactic acid” or lactate) and (waste or rubbish or garbage or biomass or cellulose* or lignocellulose* or lignocellulose* or sludge or straw or “distiller’s grains” or stillage)) were used as topic search phrases to acquire all indices of articles published from 1993 to 2022 from the Web of Science database. The records of all indices were downloaded into a spreadsheet software (Microsoft Office 365) to conduct a digital logical analysis [9]. Particularly, some keywords with the same meaning, such as “poly(lactic acid)”, “polylactic acid” and “poly” were combined together in the data treatment process. After all relevant data were categorized, the tendencies of publication outputs were analyzed using four-year intervals to minimize year-to-year fluctuations [10]. After the analysis of keywords, the keyword “lactic acid” was referred the most frequently and ranked first among all keywords. Furthermore, keywords such as “poly(lactic acid)”, “cellulose”, “biomass”, “food waste”, and “rice straw” showed a sustainable growth tendency. Specific data are shown in Table 1.

Table 1.
Top 30 most used author keywords.
3. Biomass for Lactic Acid Fermentation
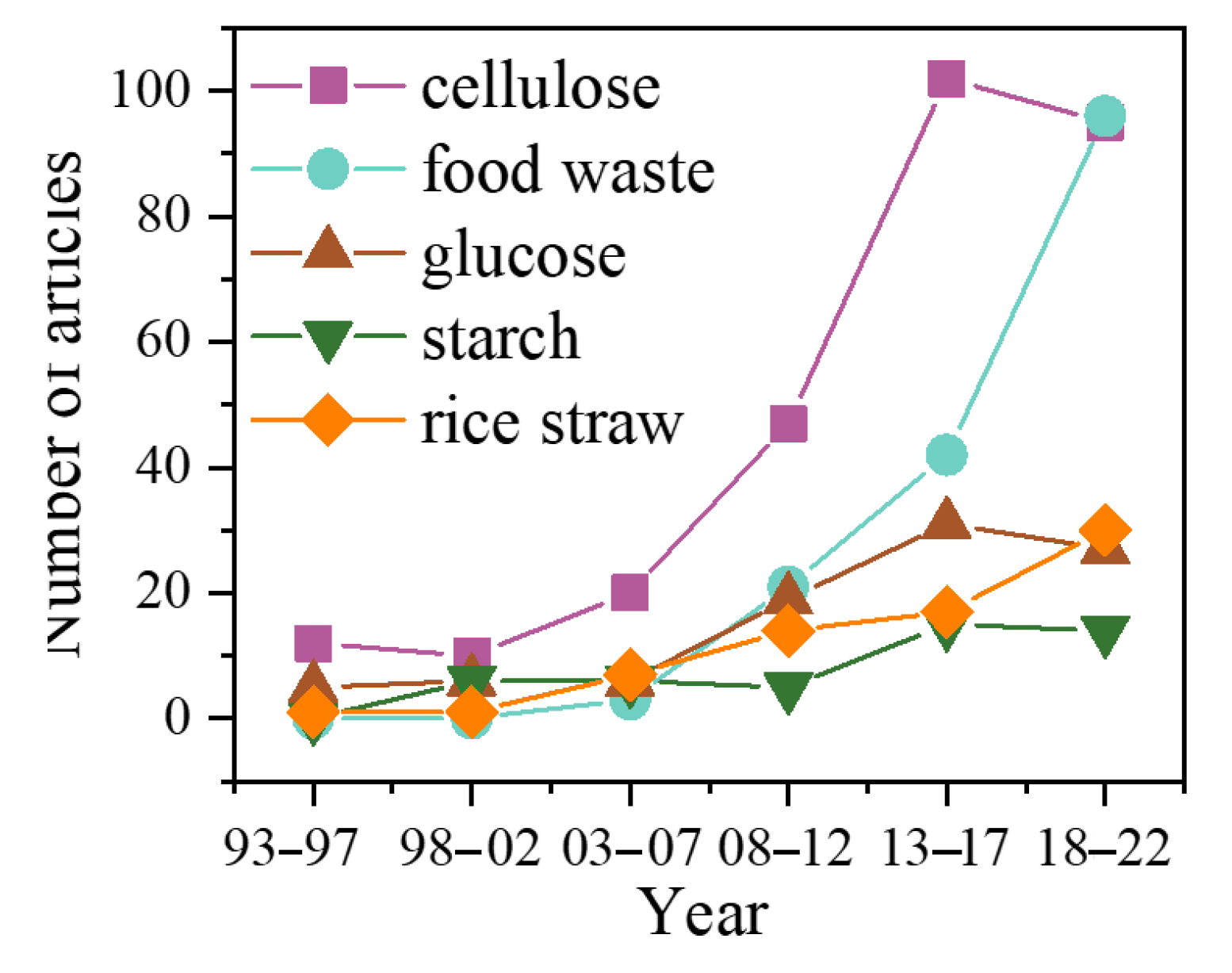
Biomass has always been the main source of energy for humans, accounting for about 14% of the world’s total energy supply. The source of biomass is diverse, which refers to all organic matter derived from organisms, such as wood, agricultural residues, animal waste, and so on [11]. From Table 1, it can be seen that the keyword ‘biomass’ had a highly used frequency. Especially during the period 2003–2022, its ranking remained around the top 10. Food waste and rice straw also rose significantly in the past 12 years, rising from 98th to 9th and 221th to 25th, respectively. However, starch’s ranking decreased from 14th to 98th, indicating that the raw materials for producing LA have been gradually replaced by other renewable, cheaper biomass waste. Figure 1 summarizes the number of articles used different substrates for LA fermentation.
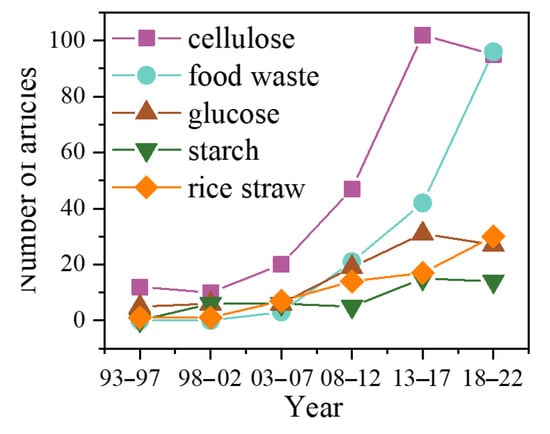
Figure 1.
The research tendency of different substrates for lactic acid fermentation.
3.1. Lignocellulosic Biomass
Lignocellulosic biomass (e.g., agricultural straw, distiller’s grains, and garden waste) contains abundant cellulose and hemicellulose. They are characterized by a regular arrangement of cellulose molecules, forming microfibers, hemicellulose, and lignin, which created a strong binding layer, tightly packed with cellulose [12]. To hydrolyze lignocellulose to monosaccharide, biomass has to be pretreated to remove lignin, increase the porosity of the material, and expose cellulose and hemicellulose to contact with enzymes, thereby increasing hydrolysis efficiency [13]. Researchers have extensively studied the pretreatment of cellulose-based biomass wastes before LA fermentation. Novel microwave-alkali and steam-alkali coupled pretreatment processes were utilized in LA production from vinasse [14]. It suggested pretreatment enhanced LA production from raw vinasse (up to 30.32 g·L−1), which was twice that without pretreatment, and L-LA yield and optical purity increased 49.8% and 57.2%, respectively.
The LA production from residues of Sophora flavescens with reusing the NaOH-pretreated liquor as pH neutralizer was conducted in a simultaneous saccharification and fermentation (SSF) system [15]. Accordingly, the addition of NaOH-pretreated liquor did not inhibit the growth of LA bacteria, but instead increased the pH of the system and led to stable cellulase activity. When the amount of NaOH-pretreated liquor reached 50%, the LA production increased by 34.1% compared with the control group without NaOH-pretreated liquor [16]. Abdel-Rahman et al. used Enterococcus mundtii QU 25 to produce L-LA from cellobiose. This strain can realize nearly complete conversion of cellulosic substrates to L-LA without the addition of enzymes and achieve high concentration of L-LA (119 g·L−1) with high yield (0.79 g·g−1) and an extremely high optical purity (≥99.9%) [17]. From an economical point of view, by using strains without extra enzymes, the cost of LA production from cellulosic materials will be reduced due to an expected decrease in enzyme requirements, a significant cost component in the use of biomass to produce value-added products.
3.2. Food Waste
As shown in Table 1, the appearance of the keyword ‘food waste’ significantly increased from none in the period of 1993–1997 to 9th in the period of 2018–2022. This result indicated that research using food waste as fermentation feedstock has received considerable attention. Food waste with high organic matter concentrations (volatile solids/total solids [VS/TS]: 0.8–0.9) and moisture content easily spoils and stinks, and large amounts could seriously affect people’s physical health and the cityscape [18]. Presently, food waste is reused in the following processes: anaerobic digestion for biogas production, composting, bio-ethanol production, and feed production for insects and animals. Food waste contains approximately 30–60% starch, 5–10% protein, 10–40% fat and some trace elements, which are nutritious and suitable for the growth of LA bacteria [18,19,20].
To establish a highly efficient LA production system from food waste without exogenous enzyme supplementation, co-fermentation of food waste and corn straw was first performed to obtain glucoamylase (1838 U·g−1) with Aspergillus niger UV-60. Further, this kind of crude glucoamylase was found to have good acid resistance, with optimum pH of 4.2–4.9, and that relative enzyme activity at pH 3.0 can still reach 83.5% [21]. Subsequently, LA fermentation was carried out in a simultaneous saccharification fermentation mode with crude glucoamylase (142 U·g−1, dry basis) addition and 6% (v·v−1) Lactobacillus rhamnosus inoculation in food waste medium. Finally, 58.40 g·L−1 (0.58 g·g−1 food waste, dry basis) of L-LA was obtained after SSF at 38 °C for 48 h [22].
3.3. Microalgae
Microalgae can use CO2 under aerobic and bright conditions to accumulate starch and convert starch into LA, ethanol, acetic acid and other organic substances under dark and anaerobic conditions [23]. The freshwater microalga Hydrodictyon reticulum (containing 47.5% reducing sugars and 35% glucose) was used as a raw material to produce LA [24]. Lactobacillus paracasei LA104 was used as inoculum to conduct SSF at 45 °C, pH 3 and obtained 46 g·100 g−1 H. reticulum dry material with 37.11 g·L−1 LA. Therefore, microalga can be a potential feedstock for large-scale production of LA. On the other hand, Chlorella sp. GD mutant, which can overcome outdoor high-temperature inhibition and has high dissolved inorganic carbon availability and maximum photosynthetic rate [25], could quickly convert CO2 to sugar and store it. However, under anaerobic conditions, the sugars that accumulated in the body were converted into organic substances such as LA, ethanol, acetic acid, and formic acid. The harvested Chlorella was fermented with L. plantarum 23, and the conversion rate and yield of LA were 80% and 0.43 g·g−1 Chlorella biomass, respectively. Microalgae could be used as a high-efficiency raw material for large-scale LA production because of its short growth cycle and simple survival.
Table 2 compared recent research results using different biomass stocks for LA fermentation. As shown in the table, the final LA concentration obtained when fermenting with a lignocellulose-based substrate was higher than that used other substrates. Food waste had a high content of organic matter and was easily digested by microorganisms, which had an advantage in terms of LA production yield and rate. As a new substrate for LA fermentation, microalgae did not have a high final LA concentration and yield. However, the conversion rate per gram of microalgae was similar with other biomass in terms of LA production yield.

Table 2.
Effects of different biomass stocks on lactic acid fermentation.
4. Microorganism for Lactic Acid Fermentation
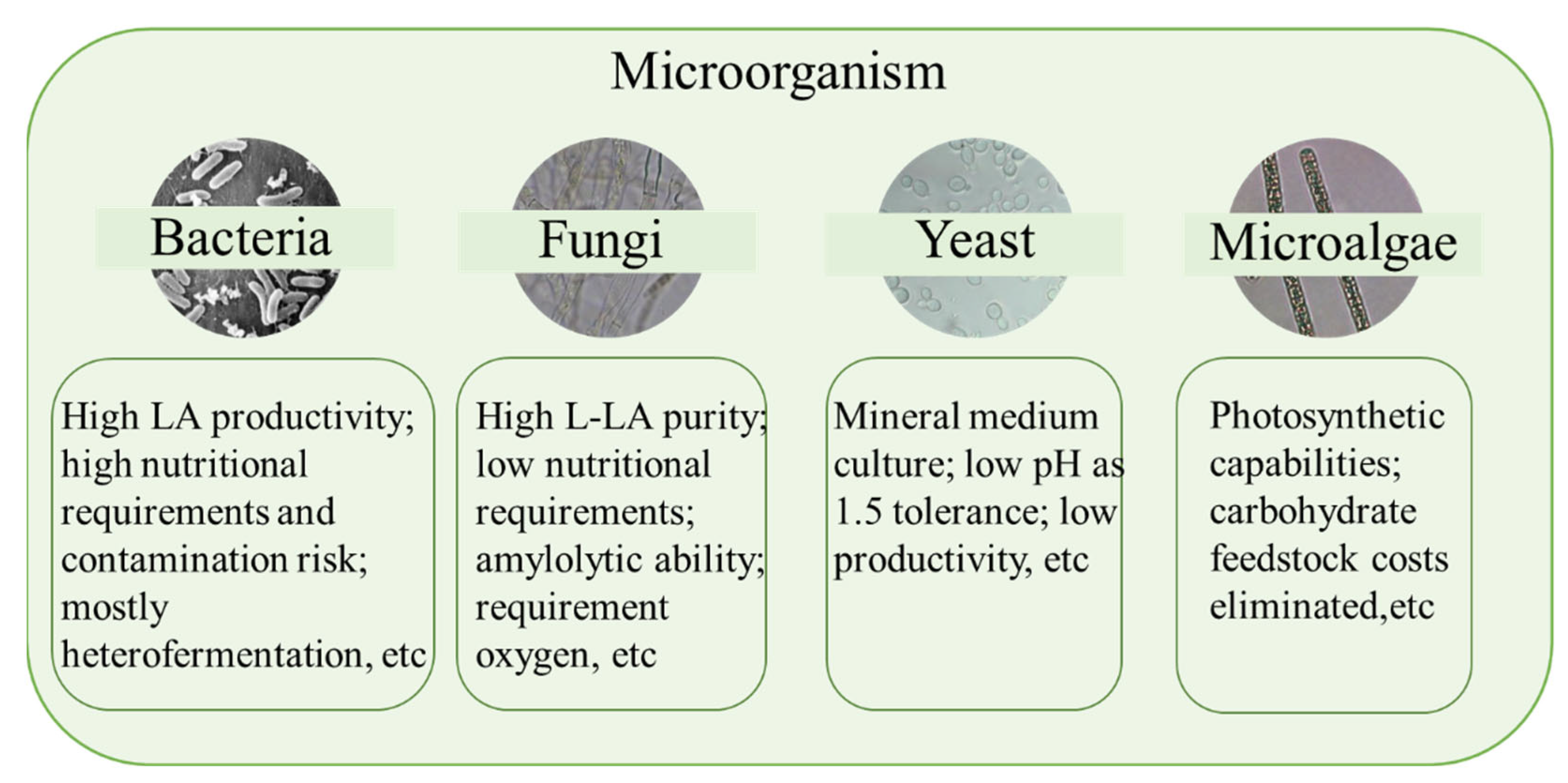
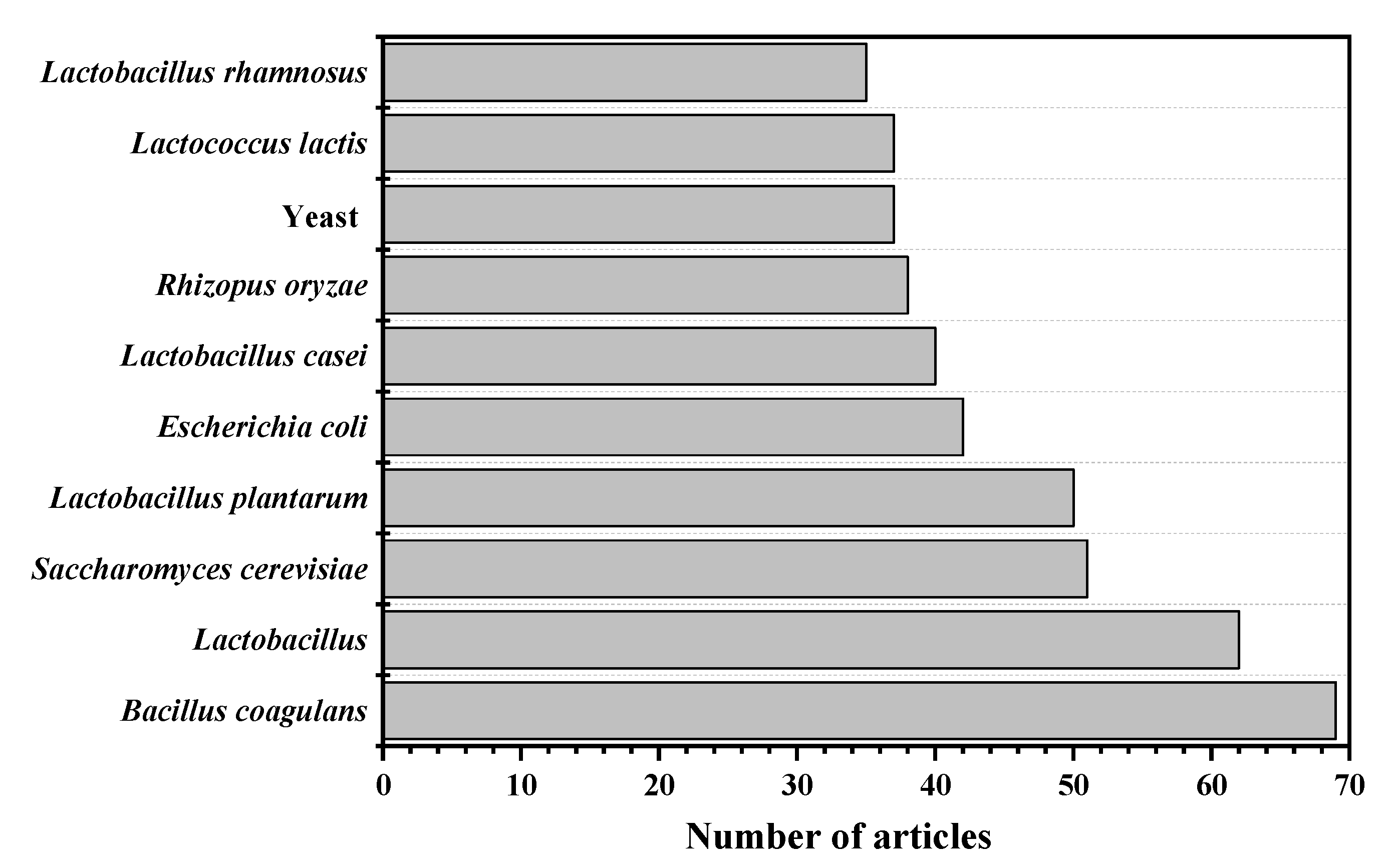
From Table 1, some conclusions about the microorganisms for LA fermentation can be drawn. Regardless of the period, Bacillus coagulans was ranked in the top 24, indicating that B. coagulans has been widely used. However, with the continuous improvement of the entire process, some genetically engineered bacteria have also been widely studied. The respective characteristics of microorganism for LA fermentation are shown in Figure 2. In the past 28 years, many LA bacteria have been used for research. Among them, the research tendency of the 11 most studied LA bacteria are shown in Figure 3.
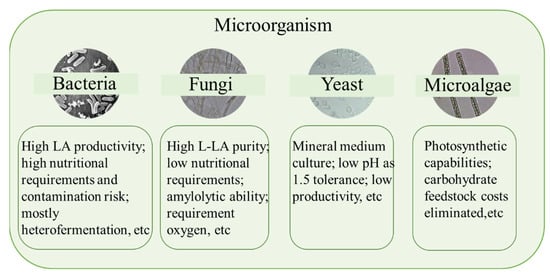
Figure 2.
Characteristics of different microorganisms for lactic acid fermentation.
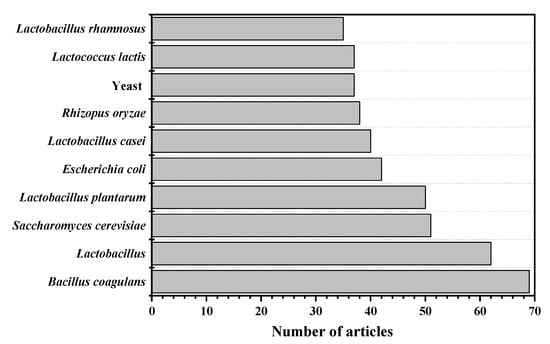
Figure 3.
The research tendency of top 10 used microorganisms for lactic acid fermentation.
4.1. Bacteria
The term LA bacteria refers to a generic group of bacteria that can produce large amounts of LA from fermentable sugars. The LA bacteria produce LA by using monosaccharides (glucose, fructose, galactose) and disaccharides (sucrose, maltose, lactose) as substrates. More than 80% of homolactic fermentation products were LA, and LA bacteria that can carry out such fermentation include L. lactis, L. delbrueckii, L. casei, and L. helveticus. Besides LA, other by-products of heterolactic acid fermentation by Leuconostoc, Lactobacillus, and Bifidobacterium included acetic acid, ethanol, and CO2 [6]. The LA bacteria are generally anaerobic or microaerobic bacteria, and their characteristics are shown in Figure 1. However, due to their rapid growth, bacteria reproduction, high LA yield, high LA productivity of LA bacteria, most researchers have been concerned with the use of LA bacteria to produce LA [16].
Bacillus coagulans 36 can utilize pentose through the pentose phosphate (PP) pathway, and its LA yield can reach up to 1.0 g·g−1 [33]. Furthermore, to develop the thermophilic characteristics of Bacillus, the open fermentation process under nonsterilized conditions is an effective and simple process to reduce the energy consumption of fermentation [34,35]. Moreover, Bacillus can effectively utilize the pentose sugar in the hydrolysate of cellulose to produce L-LA. The coagulans36 can utilize pentose sugars through the pentose-phosphate pathway, and the LA yield is up to 1.0 g·g−1 [36]. Disaccharide carbon sources such as lactose, sucrose, and maltose can also be metabolized by LA bacteria. On the other hand, Lacticaseibacillus rhamnosus was reported to metabolize cellobiose to LA directly [37]. Therefore, lignocellulose hydrolysates containing cellobiose have considerable potential to use in LA fermentation.
4.2. Fungi
The genus Rhizopus in fungi, especially R. oryzae, has attracted considerable attention because of its ability to produce high-purity L-LA [38]. Compared with LA bacteria, Rhizopus has a low requirement for nutrients and can utilize a wide range of substrates. For example, it can use lignocellulose-rich biomass materials directly to ferment and produce LA [39]. Rhizopus can also break down starches and can directly use starch to produce LA without saccharification [40]. Moreover, the filamentous structure of formed particles of the fungi facilitates the separation from the fermentation broth to reduce the cost. A new type of heat-resistant Rhizopus was isolated and characterized, which directly produced 84.3 g·L−1 and 105–119 g·L−1 of LA in batch and fed-batch fermentation, respectively, from liquefied cassava starch at pH 5.5 [41].
Furthermore, the fungi itself was a valuable by-product as organism protein. Therefore, many researchers have conducted further studies on the use of Rhizopus for fermentation of LA from renewable resources, such as molasses, lignocellulose, and raw starch [42,43,44]. However, using Rhizopus to produce LA has some deficiencies, such as the hetero-fermentation byproducts (ethanol and fumaric acid); oxygen requirements (increase energy consumption); and influence of filamentous bacteria in mass transfer, matrix mixing, and LA recovery [45].
4.3. Genetically Engineered Microorganisms
Genetically engineered strains could expand the range of substrates and increase LA production efficiency. Given the relatively clear genetic background ofyeast and Escherichia coli, and the low requirements for nutrient components, genetically engineered microorganism have become a model organism for the industrial production of LA.
Yeast can tolerate pH values as low as 1.5, so it is not necessary to neutralize the fermentation broth with alkali during the fermentation process. However, few LA was contained in the metabolites of wild yeast. Therefore, yeast capable of producing high concentrations of LA can be obtained through genetic engineering methods [46]. Osawa et al. constructed an ethanol fermentation pathway-deficient Candida boidinii by integrating bovine L-Lactate dehydrogenase-encoding gene under the control of PDC1 promoter in optimal conditions. After batch fermentation for 48 h, the LA concentration and yield reached 85.90 g·L−1 and 1.79 g·(L·h)−1 [47].
Escherichia coli transformed based on metabolic processes can use various carbon sources to conduct LA fermentation, including glucose, sucrose, and glycerol [48]. Zhao et al. constructed the engineering strain E. coli JH12 by placing the L-Lactate dehydrogenase gene of Pediococcus acidilactici into E. coli. Then, this strain was fermented with 6% xylose as a single carbon source achieving 34.73 g·L−1 LA with a purity of 98% [49]. Although the cycle of genetically modified E. coli is shorter than that of wild-type LA bacteria, the latter has advantages in terms of space–time yield, yield, and tolerance to LA. Wakai et al. constructed LA-producing Aspergillus oryzae by genetic engineering and used a pure culture of A. oryzae to produce LA from starch. Aspergillus oryzae strains innately secrete amylases. Meanwhile, A. oryzae LDHΔ871 produced approximately 30 g·L−1 of lactate from various starches, dextrin, or maltose (all at 100 g·L−1). This process was simpler than conventional methods with mixed cultures or co-utilization of enzymes [50].
Moreover, cyanobacteria are photosynthetic microorganisms that synthesize organic matter using only sunlight, CO2, water, and a small amount of inorganic salts. By means of metabolic engineering, the gene corresponding to L-lactate dehydrogenase was introduced into the cyanobacterial gene to obtain LA-producing cyanobacteria [51]. This was not only beneficial to the global warming effect but provided a new LA production route.
Table 3 compares and analyzes the effects of fermentation of different substrates on the production of LA by bacteria, fungi, and genetically engineered bacteria. As shown in the table, the use of bacteria for LA fermentation can achieve high yields and purity, and it is more suitable for industrial applications. In the case of LA fermentation for animal feed, it is necessary to use fungi in consideration of the toxicity.

Table 3.
Effects of different microorganisms on lactic acid fermentation.
5. Fermentation Modes
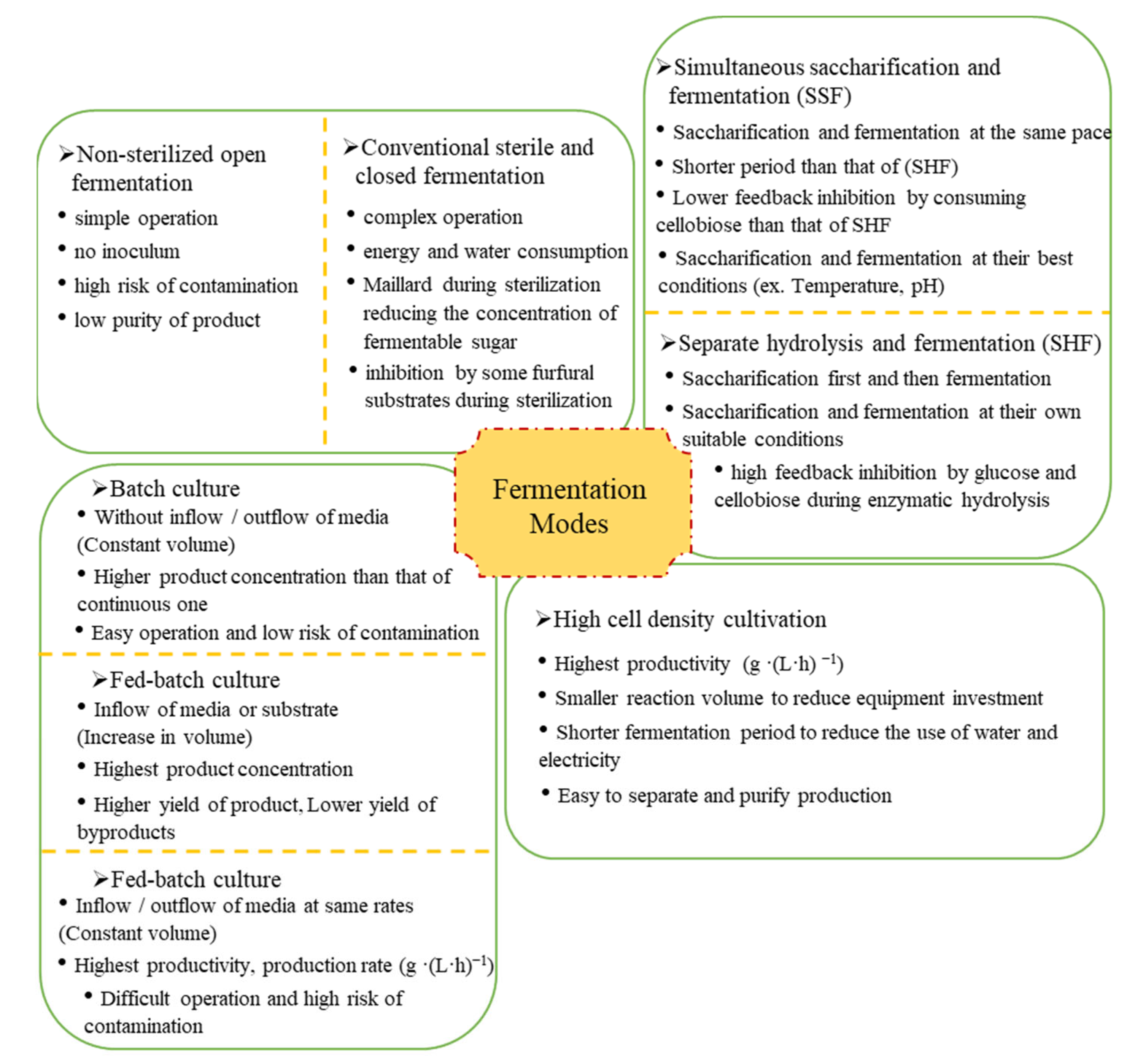
Fermentation modes include non-sterilized open fermentation, sterile closed fermentation, simultaneous saccharification fermentation (SSF), separate hydrolysis and fermentation (SHF), batch fermentation, fed-batch fermentation, semi-continuous fermentation, continuous fermentation, and high cell density fermentation. Their characteristics are shown in Figure 4.
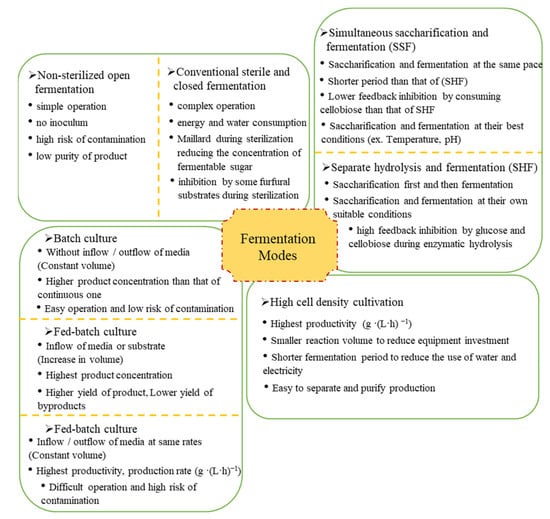
Figure 4.
Characteristics of different fermentation modes.
5.1. Non-Sterilized Open Fermentation and Conventional Sterile Closed Fermentation
As indigenous LA bacteria flora exist in food waste, most studies on open lactic fermentation use food waste as a substrate for fermentation. These bacteria groups were more competitive, and they can become dominant bacteria quickly, thereby fermenting food waste into LA. The research indicated 21.5 g·L−1 LA could be produced from food waste within 48 h without sterilization and inoculation [45]. When an unsterilized system was inoculated with LA bacteria, the inoculated bacteria would rapidly propagate to become dominant bacteria, as the organic acids and LA produced can inhibit the growth of other bacteria. Experiments showed that inoculated LA bacteria to food waste for 1–3 days would result in a drop in pH due to high LA concentration, causing the count of Staphylococcus aureus and E. coli to be 99.9% and 99.8% lower, respectively, than the control group without inoculation [53].
Although the addition of LA bacteria was beneficial to the inhibition of bacterial formation, there were still some acid-resistant hydrolytic bacteria in the fermentation process. Liu Jianguo et al. analyzed the microbial diversity of LA fermentation of food waste and found the non-sterilized open fermentation system contained abundant indigenous LA and hydrolytic bacteria. These indigenous hydrolytic bacteria could coexist with LA bacteria and degrade complex macromolecules (carbohydrates, proteins, fats) into small molecules, and further converted them into reducing sugars, thereby promoting LA production [14]. Sakai et al. obtained 86 g·L−1 of L-LA (carbon yield, 53%; optical purity, 97%) within 5 days by inoculating Bacillus coagulans NBRC12583 at pH 6.5 under 55 °C in open fermentation mode [54].
In summary, compared with traditional closed fermentation processes, non-sterilized open fermentation simplifies the production process and fermentation equipment, while reducing energy and cooling water consumption.
5.2. Simultaneous Saccharification Fermentation and Separate Hydrolysis and Fermentation
The hydrolysates of lignocellulosic biomass are mainly mixtures of hexose (glucose and mannose) and pentose (xylose and arabinose). According to the different time and method of enzymolysis and fermentation, it can be divided into simultaneous saccharification fermentation (SSF) and separate hydrolysis and fermentation (SHF).
Liu et al. inoculated Bacillus subtilis 1.420, a starch hydrolytic bacterium, into food waste to promote the degradation of starch into reducing sugars. Afterward, L. plantarum TD175 was inoculated into food waste for SHF. The maximum LA concentration was 50.77 g·L−1, and the amylase activity was 4425.92 U/g [55]. Meanwhile, 1% B. subtilis and 10% LA bacteria were simultaneously inoculated into food waste for SSF. The LA concentration reached 40.02 g·L−1 at a solid–liquid ratio of 1:1.5 at 40 °C–45 °C for 36 h. Finding glycogen- and LA-producing bacteria with optimal culture conditions was the key to improving the efficiency of SSF. Lactobacillus rhamnosus was used to ferment cassava powder for LA production in SSF mode, which produced 175 g·L−1 LA with high yield (0.71 g·g−1) and productivity (1.8 g·(L·h)−1) [56]. If SSF and SHF can be combined and then applied to practical applications, their respective advantages will create greater economic value.
5.3. Batch, Fed-Batch, Semi-Continuous, and Continuous Fermentation
According to the different feeding and discharging modes, fermentation modes can be divided into batch, fed-batch, semi-continuous, and continuous fermentation modes. In batch fermentation, without the addition and output of substrates and products, the bacterial cell, substrates, and product concentrations only changed with reaction time. This process is the simplest and most used fermentation mode at present. Liang et al. used potato peel waste to produce LA by batch fermentation and got a production yield of 0.25 g·g−1 and productivity of 125 mg·(g·d)−1 [57]. However, as fermentation progresses, almost all systems would be inhibited by the lack of substrate or high LA concentration. To alleviate product inhibition, CaCO3, NaOH, or NH4OH were added to conventional batch fermentation for acidification to ensure system stability.
Fed-batch fermentation was conducted based on the characteristics of cell growth and initial culture medium, during which one or more specific restricted substrates were intermittently or continuously added to the bioreactor without output of products. Ding et al. added glucose and yeast extracts in an exponential manner and achieved LA concentration and production rate of 180 g·L−1 and 2.14 g·(L·h)−1, respectively. Compared with batch fermentation the two values were increased by 56.5% and 59.7%, respectively [58].
In the semi-continuous fermentation system, part of the fermentation broth containing product were periodically released and then the same volume of fresh medium was added in. This process can not only supplement nutrients and substrates but dilute metabolic harmful substances, which plays an important role in alleviating product inhibition. This system improved the culture environment of the microorganisms and helped to maintain stability of bacterial vitality, thereby contributing to the continuous synthesis of product.
Continuous fermentation involves a constant feeding, and discharge of the fermented broth at the same rate simultaneously, so that the working volume in the system is constant. This method can reduce the product inhibition of LA and keep cell, product, and substrate concentrations relatively stable. In addition, continuous fermentation does not cause system suspension due to emptying, cleaning, and sterilization of the fermenter compared with batch fermentation. However, the system is limited in its large-scale application due to the inability to make full use of the carbon source, cell loss caused by discharging, and expensive equipment.
At present, due to the high LA yield and simple transformation route, continuous fermentation is the most widely used fermentation method.
5.4. High-Cell Density Cultivation
High-cell density cultivation generally refers to the fact that the cell density in the liquid culture exceeds that of conventional culture by more than 10 times, and it includes cell recycle fermentation and immobilized fermentation, etc.
A membrane-integrated repeated batch fermentation has been developed to produce L-LA from sweet sorghum juice. To overcome the carbon catabolite repression effect, high-cell-density fermentation with the use of sugars was utilized. The results showed that a L-LA productivity of 17.55 g·(L·h)−1 was obtained [59]. The immobilized strain QU 50 could produce stable L-LA with yield of 1.01–1.02 g·g−1 in an open repeated batch fermentation using mixed sugars derived from lignocellulosic biomass [60]. The mycelium of Rhizopus oryzae NBRC 5384 was fixed in situ in sponge-like cubic particles, and CaCO3 was added to control the pH of the culture solution. After batch and fed-batch fermentation, the yields and productivities of LA were 95.0%, 92.5%, and 1.83 g·(L·h)−1, respectively. This experiment prevented the inhibition of LA products by crystallization [61]. However, some studies have showed that the production of LA using immobilized cells was lower than that of free cell fermentation, whereas the application of cell recycle technology in fermentation can usually achieve higher LA yield.
Table 4 compares the effect of different fermentation processes on LA fermentation. Fed-batch alone is superior to the batch in terms of final LA concentration. However, the feeding process may cause problems such as incomplete hydrolysis of substrate, as well as lower LA production rate and yield than those of batch fermentation. As the process continues to be optimized, combining fed-batch and batch fermentation with other processes such as cell immobilization has significantly improved LA production yield. Moreover, high-cell density cultivation has a very considerable advantage in the final yield.

Table 4.
Effects of different fermentation modes on lactic acid fermentation.
6. Extractive Fermentation of Lactic Acid
The suitable pH for LA fermentation is 5.5 to 6. When the pH is <5, the LA produced by fermentation will hinder the growth of LA bacteria and further production of LA. Combining the fermentation process with in situ product removal technology (ISPR) will greatly increase the yield and efficiency of LA. The ISPR technology currently used for LA fermentation mainly includes solvent extraction, adsorption, membrane (including dialysis, electrodialysis, hollow fiber ultrafiltration membrane, reverse osmosis membrane), adsorption, and solvent extraction.
6.1. Combination of Lactic Acid Fermentation and Electrodialysis
Compared with traditional electrodialysis, bipolar membrane electrodialysis can convert salt in fermentation broth into corresponding acid and alkali, and the generated alkali can be reused in the fermentation process to adjust the pH [66]. Min-tian et al. coupled continuous fermentation with bipolar membrane electrodialysis and obtained 2637 g of LA from 4000 g glucose with a yield of 0.69 g·g−1 and a production rate of 8.18 g·(L·h)−1 [67]. Hábová et al. used a two-stage electrodialysis with bipolar membranes to separate lactate ions from fermentation broth and achieved 175 and 151 g·L−1 lactate in the first and second ED steps, respectively. In terms of the complex characteristics of LA fermentation broth that can easily contaminate electrodialysis membranes [68], Wang et al. used a pretreatment method of flocculation–ultrafiltration before electrodialysis. They added 0.3 g·L−1 of gelatin to the fermentation broth and then stirred the broth at 50 °C for 7 min and obtained flocculation and suspended solid removal rates of 94.21% and 91.44%, respectively [69].
6.2. Combination of Lactic Acid Fermentation and Absorption
Activated carbon and ion exchange resins are commonly used as adsorbents in the adsorption and fermentation processes, respectively. Song et al. used a hyper-cross-linked meso-microporous resin HD-06 with phenolic hydroxyl functional groups to separate LA and fermentation broth with an adsorption mechanism of hydrophobic effect and size exclusion. Based on the results of model simulation, the novel HD-06 resin could be a potential adsorbent in the following continuous separation process [70]. Wang et al. integrated fermentation and separation with the use of a microfiltration membrane and then used resin adsorption to separate LA from clarified broth. The fermentation broth containing residual sugar and nutrients was then recycled back into the fermenter after LA adsorption, extending the cell growth period from 41 h to 120 h. In this integrated system, the LA concentration was 183.4 g·L−1, which is 26.1% higher than that of the control group [71]. Aljundi et al. used a zeolitic membrane as the adsorbent to separate LA from the fermentation broth in situ and the recovery rate reached 65% [72].
6.3. Combination of Lactic Acid Fermentation and Extraction
Extraction fermentation is a coupled fermentation technique that uses organic solvents to continuously extract fermentation products during fermentation process to eliminate product inhibition. Honda et al. used 40% alamine 336 diluted with oleyl alcohol and oleyl alcohol as the extractant and back-extractant to conduct continuous extractive fermentation. After 24 h, the cell concentration and LA yield were 1.25- and 1.4-fold higher than those of the control group, respectively [73]. A novel method of LA recovery from the fermentation broth via continuous ultrasonic solvent extraction was presented. The LA was successfully recovered with an overall yield of 82–84% and purity of over 98%. To remove residual amounts of ethyl acetate, the recovered LA was purified by freeze drying at −20 °C for 30 min [74].
The above-mentioned novel LA separation–extraction technology gradually replaced the traditional extraction process with its unique advantages. Although the efficiency was high, the process was relatively complicated and needed to be combined with other technologies to function better. Improving the extraction and purification technology has been the focus of LA fermentation industry.
7. Perspectives
With the continuous expansion of LA applications, researchers have begun to develop LA and its derivatives. The following aspects could be considered to improve the bioconversion efficiency and added value and reduce costs of biomass waste.
7.1. Screen for Excellent Strains and Development of New Fermentation Processes
For traditional LA fermentation, Ca(OH)2 is often used as a neutralizer. However, large amounts of byproduct gypsum are produced. To avoid or minimize the use of neutralizers, strains could be genetically engineered to make them resistant to the acidic environment and produce LA at low pH. The development of abundant microbial resources in the natural world, screening of excellent strains on the basis of metabolic and genetic engineering techniques and using various fermentation to select high-quality and high-purity LA strains were the key to future industrial fermentation [75].
Using some cellulase-producing and LA-producing bacteria to culture and directly convert cellulose into LA is an interesting research topic. Enzyme-producing bacteria instead of enzyme preparation could significantly reduce the pretreatment cost of fiber raw materials. In particular, if the production of glucoamylase, enzymatic saccharification, and LA fermentation were performed in the same reactor, it could further reduce the reactor and cost of enzyme purchase. However, developing enzyme- and LA-producing bacteria with symbiotic action and synergy is necessary. Aspergillus niger (an enzyme-producing glucoamylase) and Rhizopus oryzae (a LA-producing bacteria) are both aerobic bacteria, and they could be mixed and cultured to produce LA in the same reactor.
Using the advantage of low solubility of magnesium lactate in aqueous solution, MgO is used as a fermentation neutralizer to achieve in situ separation of magnesium lactate crystals and reduce product inhibition effects. Then, follows HCl acidification, isoamyl alcohol extraction, concentration of raffinate solution to obtain MgCl2 precipitation, and then pyrolysis to obtain MgO, to achieve reuse of MgO. The LA-containing extract was back-extracted with water, and then an aqueous solution containing LA was concentrated to obtain LA product. The back-extracted isoamyl alcohol was reused [76]. The process did not have any solid or liquid waste discharges and was simple to operate. Consequently, this method is a new in situ fermentation-separation coupled continuous batch fermentation process, with prospects for industrial applications.
7.2. Extension of High Value-Added Products
To obtain a more valuable product, the research trend of LA fermentation as an intermediate product to other products has become remarkable. Zhu et al. reported caproic acid fermentation by the Ruminococcaceae bacterium CPB6 with LA as substrate [77]. The experiments were carried out in batches, with 20 g·L−1 of LA maintained as substrate. A total of 23.4 g·L−1 of caproic acid was finally produced at pH of 6.0–6.5 and temperature of 30 ± 1 °C. At a pH of 5.5 and temperature of 30 °C, 16.6 g·L−1 of caproic acid could be produced from wastewater containing 45.1 g·L−1 of lactate [78]. Hexanoic acid is widely used in food, medicine, and chemical industry and is the main raw material for food additives, perfumes, and anticancer drugs, such as retrazin. It can also be used as an additive in surfactant productions. Therefore, studies on caproic acid converted by LA should be increasingly focused on in the future.
Some LA bacteria can also produce a new type of natural antibacterial substance: phenyl LA. Compared with bacteriocins produced by LA bacteria, phenyl LA is a small molecule substance that has a broad spectrum of inhibition and high stability and has become an effective symbol of bacteriostatic capacity of LA bacteria [79]. As a new biological preservative, phenyl LA has a broad application prospect in the dairy industry.
7.3. Multiproduct Cogeneration based on Biorefinery Model
Biorefinery is considered to be an alternative to chemical refining. It uses biomass as raw material; combines biomass conversion processes and equipment; and produces fuel, electric energy, and chemical products. Future biorefinery would utilize a combination of bioconversion and chemical cracking technologies, including improved lignocellulosic grading and pretreatment methods, optimal reactor design for renewable feedstock conversion, synthesis, biocatalyst, and catalytic process improvements. Tang et al. used furfural residue, cassava residue, corn, and other mixed materials to biologically convert ethanol to LA [80]. On one hand, the joint production can reduce the nutrient input during the fermentation process and use the ethanol fermentation residue as a nitrogen source for LA fermentation. On the other hand, LA as a product can speed up the recovery of ethanol process investment. Therefore, the process efficiency and economy were improved through the joint production strategy.
In recent years, studies on the production of LA and polyglucose with xylose slag, production of L-LA and L-lysine by biological fermentation, coproduction of 1,3-propanediol and LA by crude glycerol, and coproduction of oligomers by corn cob has been conducted. Feasibility studies on xylose and LA has also been reported, realizing the biorefinery process of high value-added products. In addition, coproducing LA and protein feed from agricultural straw and LA and organic fertilizer from biomass waste fermentation has good economic and environmental benefits as it can maximize waste reduction and resource utilization.
8. Conclusions
This study not only introduced lactic acid fermentation of different biomass wastes such as starches, celluloses, and food wastes but compared the production effects of different substrates, microorganisms, and fermentation modes. Moreover, some new research directions for lactic acid fermentation in the future are introduced, including co-digestion of enzyme-producing bacterium (cellulase and glucoamylase) and lactobacillus-producing bacterium; development of strains capable of utilizing xylose or multiple substrates; in situ fermentation-separation coupling technology based on magnesium lactate crystals; extension to higher value products (hexanoic acid), and multiproduct coproduction on the basis of biorefinery (lactic acid and ethanol).
Author Contributions
Resources, conceptualization, investigation, data curation, formal analysis writing—original draft, Y.R.; resources, investigation, data curation, writing—review and editing, X.W.; investigation, data curation, Y.L.; conceptualization, writing—review and editing, Y.-Y.L.; supervision, conceptualization, writing—review and editing, Q.W. All authors have read and agreed to the published version of the manuscript.
Funding
This work was supported by the National Key R&D Program of China (Grant NO. 2019YFC1906302 and 2019YFC1906304), the National Natural Science Foundation of China (Grant NO. 51978047 and 5217100753), and the National Environmental and Energy Base for International Science and Technology Cooperation. The overseas study conducted by the first author was supported by China Scholarship Council (CSC, No. 201806460016).
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Parajuli, R.; Knudsen, M.T.; Birkved, M.; Djomo, S.N.; Corona, A.; Dalgaard, T. Environmental Impacts of Producing Bioethanol and Biobased Lactic Acid from Standalone and Integrated Biorefineries Using a Consequential and an Attributional Life Cycle Assessment Approach. Sci. Total Environ. 2017, 598, 497–512. [Google Scholar] [CrossRef] [PubMed]
- Kasmi, M.; Hamdi, M.; Trabelsi, I. Processed Milk Waste Recycling via Thermal Pretreatment and Lactic Acid Bacteria Fermentation. Environ. Sci. Pollut. Res. 2017, 24, 13604–13613. [Google Scholar] [CrossRef] [PubMed]
- Masmoudi, F.; Bessadok, A.; Dammak, M.; Jaziri, M.; Ammar, E. Biodegradable Packaging Materials Conception Based on Starch and Polylactic Acid (PLA) Reinforced with Cellulose. Environ. Sci. Pollut. Res. 2016, 23, 20904–20914. [Google Scholar] [CrossRef] [PubMed]
- Sintim, H.Y.; Bary, A.I.; Hayes, D.G.; English, M.E.; Schaeffer, S.M.; Miles, C.A.; Zelenyuk, A.; Suski, K.; Flury, M. Release of Micro- and Nanoparticles from Biodegradable Plastic during in Situ Composting. Sci. Total Environ. 2019, 675, 686–693. [Google Scholar] [CrossRef] [PubMed]
- Ingrao, C.; Tricase, C.; Cholewa-Wójcik, A.; Kawecka, A.; Rana, R.; Siracusa, V. Polylactic Acid Trays for Fresh-Food Packaging: A Carbon Footprint Assessment. Sci. Total Environ. 2015, 537, 385–398. [Google Scholar] [CrossRef] [PubMed]
- Abdel-Rahman, M.A.; Tashiro, Y.; Sonomoto, K. Lactic Acid Production from Lignocellulose-Derived Sugars Using Lactic Acid Bacteria: Overview and Limits. J. Biotechnol. 2011, 156, 286–301. [Google Scholar] [CrossRef]
- Ramírez-López, C.A.; Ochoa-Gómez, J.R.; Gil-Río, S.; Gómez-Jiménez-Aberasturi, O.; Torrecilla-Soria, J. Chemicals from Biomass: Synthesis of Lactic Acid by Alkaline Hydrothermal Conversion of Sorbitol. J. Chem. Technol. Biotechnol. 2011, 86, 867–874. [Google Scholar] [CrossRef]
- Okano, K.; Tanaka, T.; Ogino, C.; Fukuda, H.; Kondo, A. Biotechnological Production of Enantiomeric Pure Lactic Acid from Renewable Resources: Recent Achievements, Perspectives, and Limits. Appl. Microbiol. Biotechnol. 2010, 85, 413–423. [Google Scholar] [CrossRef]
- Fu, H.-Z.; Chuang, K.-Y.; Wang, M.-H.; Ho, Y.-S. Characteristics of Research in China Assessed with Essential Science Indicators. Scientometrics 2011, 88, 841–862. [Google Scholar] [CrossRef]
- Xie, S.; Zhang, J.; Ho, Y.-S. Assessment of World Aerosol Research Trends by Bibliometric Analysis. Scientometrics 2008, 77, 113–130. [Google Scholar] [CrossRef]
- Bhattacharya, S.C.; Abdul Salam, P.; Runqing, H.; Somashekar, H.I.; Racelis, D.A.; Rathnasiri, P.G.; Yingyuad, R. An Assessment of the Potential for Non-Plantation Biomass Resources in Selected Asian Countries for 2010. Biomass and Bioenergy 2005, 29, 153–166. [Google Scholar] [CrossRef]
- Hwangbo, M.; Tran, J.L.; Chu, K.-H. Effective One-Step Saccharification of Lignocellulosic Biomass Using Magnetite-Biocatalysts Containing Saccharifying Enzymes. Sci. Total Environ. 2019, 647, 806–813. [Google Scholar] [CrossRef] [PubMed]
- Guan, R.; Li, X.; Wachemo, A.C.; Yuan, H.; Liu, Y.; Zou, D.; Zuo, X.; Gu, J. Enhancing Anaerobic Digestion Performance and Degradation of Lignocellulosic Components of Rice Straw by Combined Biological and Chemical Pretreatment. Sci. Total Environ. 2018, 637–638, 9–17. [Google Scholar] [CrossRef]
- Liu, J.; Wang, Q.; Wang, S.; Sun, X.; Ma, H.; Tushiro, Y. Effects of Pretreatment on the Microbial Community and L-Lactic Acid Production in Vinasse Fermentation. J. Biotechnol. 2013, 164, 260–265. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Gao, M.; Liu, J.; Wang, Q.; Wang, C.; Yin, Z.; Wu, C. Lactic Acid Production from Sophora Flavescens Residues Pretreated with Sodium Hydroxide: Reutilization of the Pretreated Liquor during Fermentation. Bioresour. Technol. 2017, 241, 915–921. [Google Scholar] [CrossRef]
- Garde, A.; Jonsson, G.; Schmidt, A.S.; Ahring, B.K. Lactic Acid Production from Wheat Straw Hemicellulose Hydrolysate by Lactobacillus Pentosus and Lactobacillus Brevis. Bioresour. Technol. 2002, 81, 217–223. [Google Scholar] [CrossRef]
- Abdel-Rahman, M.A.; Tashiro, Y.; Zendo, T.; Hanada, K.; Shibata, K.; Sonomoto, K. Efficient homofermentative L-(+)-lactic acid production from xylose by a novel lactic acid bacterium, Enterococcus mundtii QU 25. Appl. Environ. Microbiol. 2011, 77, 1892–1895. [Google Scholar] [CrossRef]
- Ren, Y.; Yu, M.; Wu, C.; Wang, Q.; Gao, M.; Huang, Q.; Liu, Y. A Comprehensive Review on Food Waste Anaerobic Digestion: Research Updates and Tendencies. Bioresour. Technol. 2018, 247, 1069–1076. [Google Scholar] [CrossRef]
- Zhang, S.; Ren, Y.; Ma, X.; Guan, W.; Gao, M.; Li, Y.Y.; Wang, Q.; Wu, C. Effect of Zero-Valent Iron Addition on the Biogas Fermentation of Food Waste after Anaerobic Preservation. J. Environ. Chem. Eng. 2021, 9, 106013. [Google Scholar] [CrossRef]
- Ma, X.; Gao, M.; Liu, S.; Li, Y.; Sun, X.; Wang, Q. An Innovative Approach for Reducing the Water and Alkali Consumption in the Lactic Acid Fermentation via the Reuse of Pretreated Liquid. Bioresour. Technol. 2022, 352, 127108. [Google Scholar] [CrossRef]
- Wang, X.Q.; Wang, Q.H.; Zhi Ma, H.; Yin, W. Lactic Acid Fermentation of Food Waste Using Integrated Glucoamylase Production. J. Chem. Technol. Biotechnol. 2009, 84, 139–143. [Google Scholar] [CrossRef]
- Wang, X.Q.; Wang, Q.H.; Liu, Y.Y.; Ma, H.Z. On-Site Production of Crude Glucoamylase for Kitchen Waste Hydrolysis. Waste Manag. Res. 2009, 28, 539–544. [Google Scholar] [CrossRef] [PubMed]
- Dasan, Y.K.; Lam, M.K.; Yusup, S.; Lim, J.W.; Lee, K.T. Life Cycle Evaluation of Microalgae Biofuels Production: Effect of Cultivation System on Energy, Carbon Emission and Cost Balance Analysis. Sci. Total Environ. 2019, 688, 112–128. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, C.M.; Kim, J.S.; Hwang, H.J.; Park, M.S.; Choi, G.J.; Choi, Y.H.; Jang, K.S.; Kim, J.C. Production of L-Lactic Acid from a Green Microalga, Hydrodictyon Reticulum, by Lactobacillus Paracasei LA104 Isolated from the Traditional Korean Food, Makgeolli. Bioresour. Technol. 2012, 110, 552–559. [Google Scholar] [CrossRef]
- Lee, T.-M.; Tseng, Y.-F.; Cheng, C.-L.; Chen, Y.-C.; Lin, C.-S.; Su, H.-Y.; Chow, T.-J.; Chen, C.-Y.; Chang, J.-S. Characterization of a Heat-Tolerant Chlorella Sp. GD Mutant with Enhanced Photosynthetic CO2 Fixation Efficiency and Its Implication as Lactic Acid Fermentation Feedstock. Biotechnol. Biofuels 2017, 10, 214. [Google Scholar] [CrossRef]
- Hu, J.; Zhang, Z.; Lin, Y.; Zhao, S.; Mei, Y.; Liang, Y.; Peng, N. High-Titer Lactic Acid Production from NaOH-Pretreated Corn Stover by Bacillus Coagulans LA204 Using Fed-Batch Simultaneous Saccharification and Fermentation under Non-Sterile Condition. Bioresour. Technol. 2015, 182, 251–257. [Google Scholar] [CrossRef]
- Kuo, Y.C.; Yuan, S.F.; Wang, C.A.; Huang, Y.J.; Guo, G.L.; Hwang, W.S. Production of Optically Pure L-Lactic Acid from Lignocellulosic Hydrolysate by Using a Newly Isolated and d-Lactate Dehydrogenase Gene-Deficient Lactobacillus Paracasei Strain. Bioresour. Technol. 2015, 198, 651–657. [Google Scholar] [CrossRef]
- Meng, Y.; Xue, Y.; Yu, B.; Gao, C.; Ma, Y. Efficient Production of L-Lactic Acid with High Optical Purity by Alkaliphilic Bacillus Sp. WL-S20. Bioresour. Technol. 2012, 116, 334–339. [Google Scholar] [CrossRef]
- Smerilli, M.; Neureiter, M.; Wurz, S.; Haas, C.; Frühauf, S.; Fuchs, W. Direct Fermentation of Potato Starch and Potato Residues to Lactic Acid by Geobacillus Stearothermophilus under Non-Sterile Conditions. J. Chem. Technol. Biotechnol. 2015, 90, 648–657. [Google Scholar] [CrossRef]
- Kwan, T.H.; Hu, Y.; Lin, C.S.K. Valorisation of Food Waste via Fungal Hydrolysis and Lactic Acid Fermentation with Lactobacillus Casei Shirota. Bioresour. Technol. 2016, 217, 129–136. [Google Scholar] [CrossRef]
- Mazumdar, S.; Bang, J.; Oh, M.-K. L-Lactate Production from Seaweed Hydrolysate of Laminaria Japonica Using Metabolically Engineered Escherichia Coli. Appl. Biochem. Biotechnol. 2014, 172, 1938–1952. [Google Scholar] [CrossRef] [PubMed]
- Talukder, M.M.R.; Das, P.; Wu, J.C. Microalgae (Nannochloropsis Salina) Biomass to Lactic Acid and Lipid. Biochem. Eng. J. 2012, 68, 109–113. [Google Scholar] [CrossRef]
- Maas, R.H.W.; Bakker, R.R.; Jansen, M.L.A.; Visser, D.; de Jong, E.; Eggink, G.; Weusthuis, R.A. Lactic Acid Production from Lime-Treated Wheat Straw by Bacillus Coagulans: Neutralization of Acid by Fed-Batch Addition of Alkaline Substrate. Appl. Microbiol. Biotechnol. 2008, 78, 751–758. [Google Scholar] [CrossRef] [PubMed]
- Ma, K.; Maeda, T.; You, H.; Shirai, Y. Open Fermentative Production of L-Lactic Acid with High Optical Purity by Thermophilic Bacillus Coagulans Using Excess Sludge as Nutrient. Bioresour. Technol. 2014, 151, 28–35. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Zhao, X.; Chamu, J.; Shanmugam, K.T. Isolation, Characterization and Evolution of a New Thermophilic Bacillus Licheniformis for Lactic Acid Production in Mineral Salts Medium. Bioresour. Technol. 2011, 102, 8152–8158. [Google Scholar] [CrossRef] [PubMed]
- Patel, M.A.; Ou, M.S.; Harbrucker, R.; Aldrich, H.C.; Buszko, M.L.; Ingram, L.O.; Shanmugam, K.T. Isolation and Characterization of Acid-Tolerant, Thermophilic Bacteria for Effective Fermentation of Biomass-Derived Sugars to Lactic Acid. Appl. Environ. Microbiol. 2006, 72, 3228–3235. [Google Scholar] [CrossRef]
- Castillo Martinez, F.A.; Balciunas, E.M.; Salgado, J.M.; Domínguez González, J.M.; Converti, A.; Oliveira, R.P. de S. Lactic Acid Properties, Applications and Production: A Review. Trends Food Sci. Technol. 2013, 30, 70–83. [Google Scholar] [CrossRef]
- Guo, Y.; Yan, Q.; Jiang, Z.; Teng, C.; Wang, X. Efficient Production of Lactic Acid from Sucrose and Corncob Hydrolysate by a Newly Isolated Rhizopus Oryzae GY18. J. Ind. Microbiol. Biotechnol. 2010, 37, 1137–1143. [Google Scholar] [CrossRef]
- Ruengruglikit, C.; Hang, Y.D. L(+)-Lactic Acid Production from Corncobs by Rhizopus Oryzae NRRL-395. LWT - Food Sci. Technol. 2003, 36, 573–575. [Google Scholar] [CrossRef]
- Jin, B.; Huang, L.P.; Lant, P. Rhizopus Arrhizus – a Producer for Simultaneous Saccharification and Fermentation of Starch Waste Materials to l(+)-Lactic Acid. Biotechnol. Lett. 2003, 25, 1983–1987. [Google Scholar] [CrossRef]
- Trakarnpaiboon, S.; Srisuk, N.; Piyachomkwan, K.; Yang, S.-T.; Kitpreechavanich, V. L-Lactic Acid Production from Liquefied Cassava Starch by Thermotolerant Rhizopus Microsporus: Characterization and Optimization. Process Biochem. 2017, 63, 26–34. [Google Scholar] [CrossRef]
- Amrane, A. Effect of Inorganic Phosphate on Lactate Production by Lactobacillus Helveticus Grown on Supplemented Whey Permeate. J. Chem. Technol. Biotechnol. 2000, 75, 223–228. [Google Scholar] [CrossRef]
- Saito, K.; Hasa, Y.; Abe, H. Production of Lactic Acid from Xylose and Wheat Straw by Rhizopus Oryzae. J. Biosci. Bioeng. 2012, 114, 166–169. [Google Scholar] [CrossRef]
- Zhang, Z.Y.; Jin, B.; Kelly, J.M. Production of Lactic Acid from Renewable Materials by Rhizopus Fungi. Biochem. Eng. J. 2007, 35, 251–263. [Google Scholar] [CrossRef]
- Vodnar, D.C.; Dulf, F.V.; Pop, O.L.; Socaciu, C. L (+)-Lactic Acid Production by Pellet-Form Rhizopus Oryzae NRRL 395 on Biodiesel Crude Glycerol. Microb. Cell Fact. 2013, 12, 1–9. [Google Scholar] [CrossRef]
- Ishida, N.; Saitoh, S.; Tokuhiro, K.; Nagamori, E.; Matsuyama, T.; Kitamoto, K.; Takahashi, H. Efficient production of L-lactic acid by metabolically engineered Saccharomyces cerevisiae with a genome-integrated L-lactate dehydrogenase gene. Appl. Environ. Microbiol. 2005, 71, 1964–1970. [Google Scholar] [CrossRef]
- Osawa, F.; Fujii, T.; Nishida, T.; Tada, N.; Ohnishi, T.; Kobayashi, O.; Komeda, T.; Yoshida, S. Efficient Production of L-Lactic Acid by Crabtree-Negative Yeast Candida Boidinii. Yeast 2009, 26, 485–496. [Google Scholar] [CrossRef]
- Wang, Y.; Tian, T.; Zhao, J.; Wang, J.; Yan, T.; Xu, L.; Liu, Z.; Garza, E.; Iverson, A.; Manow, R.; et al. Homofermentative Production of D-Lactic Acid from Sucrose by a Metabolically Engineered Escherichia Coli. Biotechnol. Lett. 2012, 34, 2069–2075. [Google Scholar] [CrossRef]
- Zhao, J.; Xu, L.; Wang, Y.; Zhao, X.; Wang, J.; Garza, E.; Manow, R.; Zhou, S. Homofermentative Production of Optically Pure L-Lactic Acid from Xylose by Genetically Engineered Escherichia Coli B. Microb. Cell Fact. 2013, 12, 1–6. [Google Scholar] [CrossRef]
- Wakai, S.; Yoshie, T.; Asai-Nakashima, N.; Yamada, R.; Ogino, C.; Tsutsumi, H.; Hata, Y.; Kondo, A. L-Lactic Acid Production from Starch by Simultaneous Saccharification and Fermentation in a Genetically Engineered Aspergillus Oryzae Pure Culture. Bioresour. Technol. 2014, 173, 376–383. [Google Scholar] [CrossRef]
- Angermayr, S.A.; Paszota, M.; Hellingwerf, K.J. Engineering a Cyanobacterial Cell Factory for Production of Lactic Acid. Appl. Environ. Microbiol. 2012, 78, 7098–7106. [Google Scholar] [CrossRef] [PubMed]
- Abdel-Rahman, M.A.; Tashiro, Y.; Zendo, T.; Sakai, K.; Sonomoto, K. Enterococcus Faecium QU 50: A Novel Thermophilic Lactic Acid Bacterium for High-Yield l-Lactic Acid Production from Xylose. FEMS Microbiol. Lett. 2015, 362. [Google Scholar] [CrossRef] [PubMed]
- Zhao, N.; Yu, M.; Wang, Q.; Song, N.; Che, S.; Wu, C.; Sun, X. Effect of Ethanol and Lactic Acid Pre-Fermentation on Putrefactive Bacteria Suppression, Hydrolysis, and Methanogenesis of Food Waste. Energy and Fuels 2016, 30, 2982–2989. [Google Scholar] [CrossRef]
- Sakai, K.; Ezaki, Y. Open L-Lactic Acid Fermentation of Food Refuse Using Thermophilic Bacillus Coagulans and Fluorescence in Situ Hybridization Analysis of Microflora. J. Biosci. Bioeng. 2006, 101, 457–463. [Google Scholar] [CrossRef]
- Liu, Y.Y.; Wang, Q.H.; Chen, L.W.; Wang, X.Q.; Wang, J. Optimization of Lactic Acid Production from Food Waste by the Saccharification of Bacillus Subtili. Adv. Mater. Res. 2010, 113–116, 1080–1083. [Google Scholar] [CrossRef]
- Wang, L.; Zhao, B.; Liu, B.; Yang, C.; Yu, B.; Li, Q.; Ma, C.; Xu, P.; Ma, Y. Efficient Production of L-Lactic Acid from Cassava Powder by Lactobacillus Rhamnosus. Bioresour. Technol. 2010, 101, 7895–7901. [Google Scholar] [CrossRef]
- Liang, S.; McDonald, A.G.; Coats, E.R. Lactic Acid Production from Potato Peel Waste by Anaerobic Sequencing Batch Fermentation Using Undefined Mixed Culture. Waste Manag. 2015, 45, 51–56. [Google Scholar] [CrossRef]
- Ding, S.; Tan, T. L-Lactic Acid Production by Lactobacillus Casei Fermentation Using Different Fed-Batch Feeding Strategies. Process Biochem. 2006, 41, 1451–1454. [Google Scholar] [CrossRef]
- Wang, Y.; Meng, H.; Cai, D.; Wang, B.; Qin, P.; Wang, Z.; Tan, T. Improvement of L-Lactic Acid Productivity from Sweet Sorghum Juice by Repeated Batch Fermentation Coupled with Membrane Separation. Bioresour. Technol. 2016, 211, 291–297. [Google Scholar] [CrossRef]
- Tan, J.; Abdel-Rahman, M.A.; Numaguchi, M.; Tashiro, Y.; Zendo, T.; Sakai, K.; Sonomoto, K. Thermophilic Enterococcus Faecium QU 50 Enabled Open Repeated Batch Fermentation for L-Lactic Acid Production from Mixed Sugars without Carbon Catabolite Repression. RSC Adv. 2017, 7, 24233–24241. [Google Scholar] [CrossRef]
- Yamane, T.; Tanaka, R. Highly Accumulative Production of l(+)-Lactate from Glucose by Crystallization Fermentation with Immobilized Rhizopus Oryzae. J. Biosci. Bioeng. 2013, 115, 90–95. [Google Scholar] [CrossRef] [PubMed]
- Abdel-Rahman, M.A.; Xiao, Y.; Tashiro, Y.; Wang, Y.; Zendo, T.; Sakai, K.; Sonomoto, K. Fed-Batch Fermentation for Enhanced Lactic Acid Production from Glucose/Xylose Mixture without Carbon Catabolite Repression. J. Biosci. Bioeng. 2015, 119, 153–158. [Google Scholar] [CrossRef] [PubMed]
- Abdel-Rahman, M.A.; Tashiro, Y.; Zendo, T.; Sonomoto, K. Improved Lactic Acid Productivity by an Open Repeated Batch Fermentation System Using Enterococcus Mundtii QU 25. RSC Adv. 2013, 3, 8437–8445. [Google Scholar] [CrossRef]
- Ou, M.S.; Ingram, L.O.; Shanmugam, K.T. L(+)-Lactic Acid Production from Non-Food Carbohydrates by Thermotolerant Bacillus Coagulans. J. Ind. Microbiol. Biotechnol. 2011, 38, 599–605. [Google Scholar] [CrossRef] [PubMed]
- Qin, J.; Zhao, B.; Wang, X.; Wang, L.; Yu, B.; Ma, Y.; Ma, C.; Tang, H.; Sun, J.; Xu, P. Non-Sterilized Fermentative Production of Polymer-Grade L-Lactic Acid by a Newly Isolated Thermophilic Strain Bacillus Sp. 2–6. PLoS One 2009, 4, e4359. [Google Scholar] [CrossRef]
- Franken, T. Bipolar Membrane Technology and Its Applications. Membr. Technol. 2000, 2000, 8–11. [Google Scholar] [CrossRef]
- Min-tian, G.; Koide, M.; Gotou, R.; Takanashi, H.; Hirata, M.; Hano, T. Development of a Continuous Electrodialysis Fermentation System for Production of Lactic Acid by Lactobacillus Rhamnosus. Process Biochem. 2005, 40, 1033–1036. [Google Scholar] [CrossRef]
- Hábová, V.; Melzoch, K.; Rychtera, M.; Sekavová, B. Electrodialysis as a Useful Technique for Lactic Acid Separation from a Model Solution and a Fermentation Broth. Desalination 2004, 162, 361–372. [Google Scholar] [CrossRef]
- Wang, L.; Wang, Q.; Li, Y.; Cao, D.; Sun, T. Flocculation of a Lactic Acid Fermentation Broth by Gelatin. Int. J. Environ. Pollut. 2013, 51, 255–268. [Google Scholar] [CrossRef]
- Song, M.; Jiao, P.; Qin, T.; Jiang, K.; Zhou, J.; Zhuang, W.; Chen, Y.; Liu, D.; Zhu, C.; Chen, X.; et al. Recovery of Lactic Acid from the Pretreated Fermentation Broth Based on a Novel Hyper-Cross-Linked Meso-Micropore Resin: Modeling. Bioresour. Technol. 2017, 241, 593–602. [Google Scholar] [CrossRef]
- Wang, C.; Li, Q.; Wang, D.; Xing, J. Improving the Lactic Acid Production of Actinobacillus Succinogenes by Using a Novel Fermentation and Separation Integration System. Process Biochem. 2014, 49, 1245–1250. [Google Scholar] [CrossRef]
- Aljundi, I.H.; Belovich, J.M.; Talu, O. Adsorption of Lactic Acid from Fermentation Broth and Aqueous Solutions on Zeolite Molecular Sieves. Chem. Eng. Sci. 2005, 60, 5004–5009. [Google Scholar] [CrossRef]
- Honda, H.; Toyama, Y.; Takahashi, H.; Nakazeko, T.; Kobayashi, T. Effective Lactic Acid Production by Two-Stage Extractive Fermentation. J. Ferment. Bioeng. 1995, 79, 589–593. [Google Scholar] [CrossRef]
- Hu, Y.; Kwan, T.H.; Daoud, W.A.; Lin, C.S.K. Continuous Ultrasonic-Mediated Solvent Extraction of Lactic Acid from Fermentation Broths. J. Clean. Prod. 2017, 145, 142–150. [Google Scholar] [CrossRef]
- Upadhyaya, B.P.; DeVeaux, L.C.; Christopher, L.P. Metabolic Engineering as a Tool for Enhanced Lactic Acid Production. Trends Biotechnol. 2014, 32, 637–644. [Google Scholar] [CrossRef]
- Wang, Y.; Cai, D.; Chen, C.; Wang, Z.; Qin, P.; Tan, T. Efficient Magnesium Lactate Production with in Situ Product Removal by Crystallization. Bioresour. Technol. 2015, 198, 658–663. [Google Scholar] [CrossRef]
- Zhu, X.; Tao, Y.; Liang, C.; Li, X.; Wei, N.; Zhang, W.; Zhou, Y.; Yang, Y.; Bo, T. The Synthesis of N-Caproate from Lactate: A New Efficient Process for Medium-Chain Carboxylates Production. Sci. Rep. 2015, 5, 14360. [Google Scholar] [CrossRef]
- Zhu, X.; Zhou, Y.; Wang, Y.; Wu, T.; Li, X.; Li, D.; Tao, Y. Production of High-Concentration n-Caproic Acid from Lactate through Fermentation Using a Newly Isolated Ruminococcaceae Bacterium CPB6. Biotechnol. Biofuels 2017, 10, 102. [Google Scholar] [CrossRef]
- Le Lay, C.; Mounier, J.; Vasseur, V.; Weill, A.; Le Blay, G.; Barbier, G.; Coton, E. In Vitro and in Situ Screening of Lactic Acid Bacteria and Propionibacteria Antifungal Activities against Bakery Product Spoilage Molds. Food Control 2016, 60, 247–255. [Google Scholar] [CrossRef]
- Tang, Y.; Dou, X.; Jiang, J.; Lei, F.; Liu, Z. Yield-Determining Components in High-Solid Integrated First and Second Generation Bioethanol Production from Cassava Residues, Furfual Residues and Corn. RSC Adv. 2016, 6, 50373–50383. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).