Greenhouse Gas Emissions and Life Cycle Assessment on the Black Soldier Fly (Hermetia illucens L.)
Abstract
1. Introduction
2. Methods
3. Gases Associated with Insect Production
3.1. Carbon Dioxide
3.2. Methane
3.3. Nitrous Oxide
3.4. Ammonia
3.5. Other Greenhouse Gases
4. Direct Greenhouse Gas Assessment Methods
4.1. Chamber System
| Ref. | Sampling Method | Sampling Procedure and Frequency | Study Period | CO2 | CH4 | N2O | NH3 |
|---|---|---|---|---|---|---|---|
| [5] | Closed static chamber/syringe | Three times every morning: one immediately (t = 0), at 3 min (t = 3), and at 6 min (t = 6) after sealing the jar. | 7 days | NDIR | GC–FID | – | – |
| [88] | Closed static chamber + fan/syringe | Three days: 2, 7, and 11 (5-day intervals). Samples were taken at intervals of 0, 15, 30, and 60 min. | 14 days | GC–TCD | GC–FID | GC–ECD | EC gas detector tube system |
| [90] | Closed static chamber + fan/syringe | Daily in triplicates | 13 days | – | GC–FID | GC–ECD | – |
| [87] | Closed static chamber/syringe | Every morning | 8 days | GC–FID | GC–FID | GC–ECD | Trapped in an H2SO4 solution and then measured using titration with a NaOH solution |
| [86] | Closed static chamber/syringe | Daily at 0 and 20 min after the container closure | 10 days | GC–FID | GC–FID | GC–ECD | Tapped in an H2SO4 solution and then measured using titration with a NaOH solution |
| [84] | Closed static chamber/syringe | Daily at 0 and 300 s | 12 days | GC–FID | GC–FID | GC–ECD | Trapped in an H2SO4 solution and then measured using titration with a NaOH solution |
| [85] | Closed static chamber/syringe | Every morning | 10 days | GC–FID | GC–FID | GC–ECD | Trapped in an H2SO4 solution and then measured using titration with a NaOH solution |
| [92,95] | Open chamber + fan/syringe (N2O only) | Daily. Every 9 min from the ingoing (CO2 and CH4) and outgoing airstream (CO2, CH4, and NH3). N2O samples were taken every 24 h (at noon). | 8 days | NDIR | NDIR | GC–ECD | EC gas analyser |
| [120] | Closed static chamber | Daily at 11:00 h at intervals of 0, 10, 20, 40, and 60 min. | 6 days | – | GC–FID | – | – |
| [91] | Closed dynamic chamber + fan/sampling bag and tube | Sampling was performed over a three-day period in 4 h intervals (4:00, 8:00, 12:00, 16:00, 20:00, 24:00). | 10 days | – | GC– ns | GC–ns | EC gas detector |
| [89] | Closed static chamber + fan/syringe | Samples were taken four times, three between 1 to 7 d and the last between 7–25 d depending on the BSFL composting duration (directly upon sealing the box, after 20 min, and after 1 h). | 17–35 days | EC gas detector tube system | GC–FID | GC–ECD | EC gas detector tube system |
4.2. Sampling in Containers
5. Analytical Techniques for GHG Measurement
5.1. Chromatography
5.2. Optical Gas Sensing
5.3. Electrochemical Gas Sensing
6. Life Cycle Assessment of the BSFL
6.1. System Boundaries
6.2. Functional Unit
| Index | FU | Allocation | Data Sources | LCA Method | LCA Software |
|---|---|---|---|---|---|
| 1 | 1 ton of impurity-free biodegradable waste | n.r. | literature | CML 2002 * | n.s. |
| 2 | 1 kg of dried defatted insect powder 1 kg of ready for consumption fresh product | Mass a economic b | Ecoinvent literature | ReCiPe IMPACT 2002+ | SimaPro |
| 3 | 1 ton insect meal | economic | Ecoinvent | ReCiPe | Arda Calculator |
| 4 | 1 ton of bio-digested food waste 1 kg of proteins 1 kg of lipids | Economic c | Ecoinvent literature | CML 2 baseline 2000 IPCC 2007 CED and CML 2001 | SimaPro |
| 5 | 1 ton of food waste to be treated | n.r. | Ecoinvent | CML 2 baseline 2000 CED and CML 2001 | SimaPro |
| 6 | 1 ton of biowaste (ww) | n.r. | Ecoinvent | IPCC 2013 | SimaPro |
| 7 | 1 kg of dried and pelletised organic fertilizer 1 kg of fresh BSF biomass 1 kg of protein concentrated meal 1 kg of BSF fat | economic | Agri-footprint Ecoinvent literature | IMPACT 2002+ | SimaPro |
| 8 | 0.403 g of bioplastic | mass | Ecoinvent | IMPACT 2002+ | SimaPro |
| 9 | 1 kg of fresh larvae 1 kg of larval protein | economic | Ecoinvent literature | CML | n.s. |
| 10 | 0.403 g of bioplastic | mass | Ecoinvent | IMPACT 2002+ | SimaPro |
| 11 | 1 ton of treated food waste | n.r. | Eco-invent | IMPACT 2002+ | n.s. |
| 12 | 1 ton of treated food waste | n.r. | Ecoinvent | n.s | n.s. |
6.3. Allocation Criteria
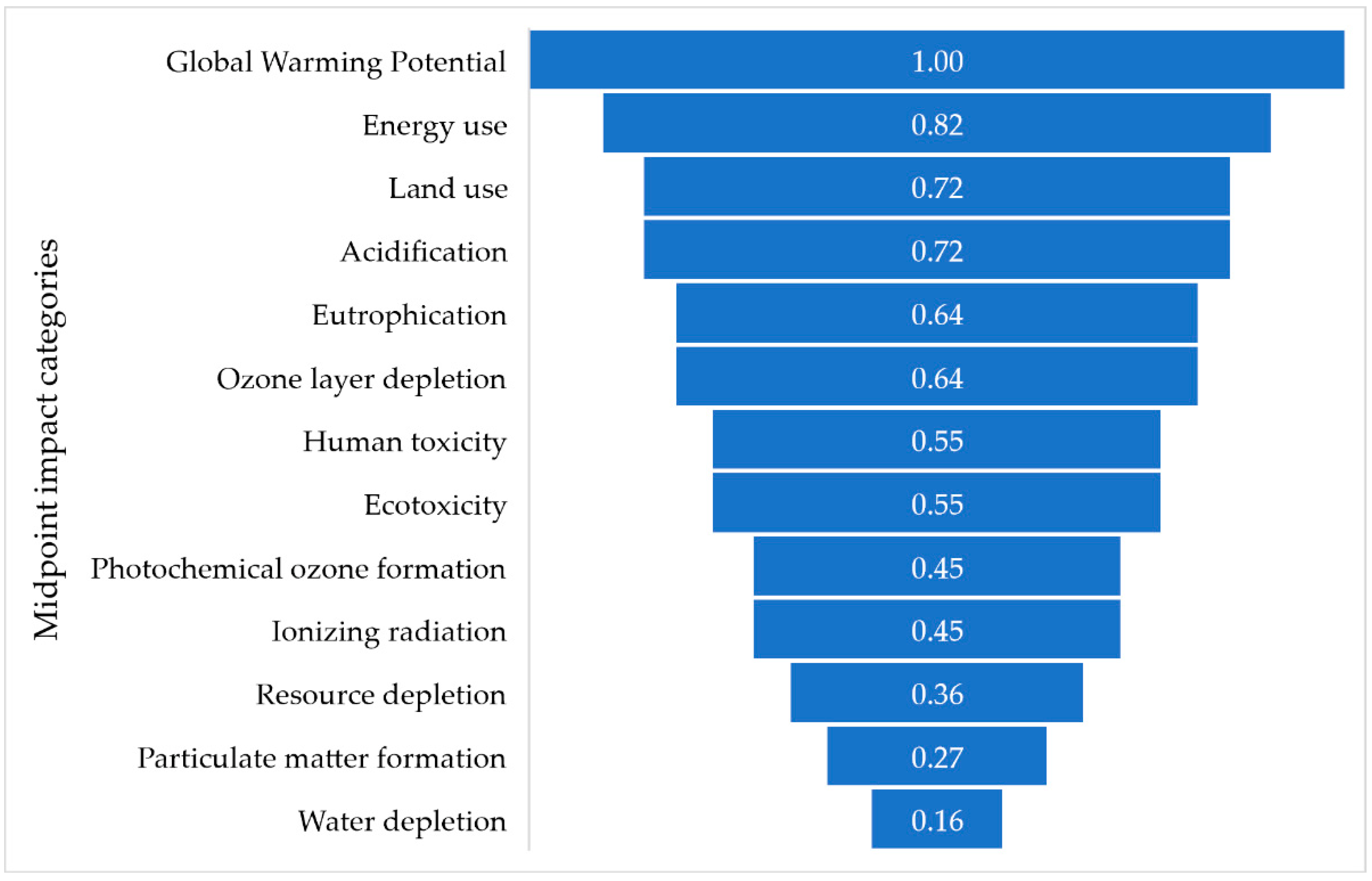
6.4. Impact Categories
6.5. LCA Methods, Databases, and Software
7. Impact Assessment
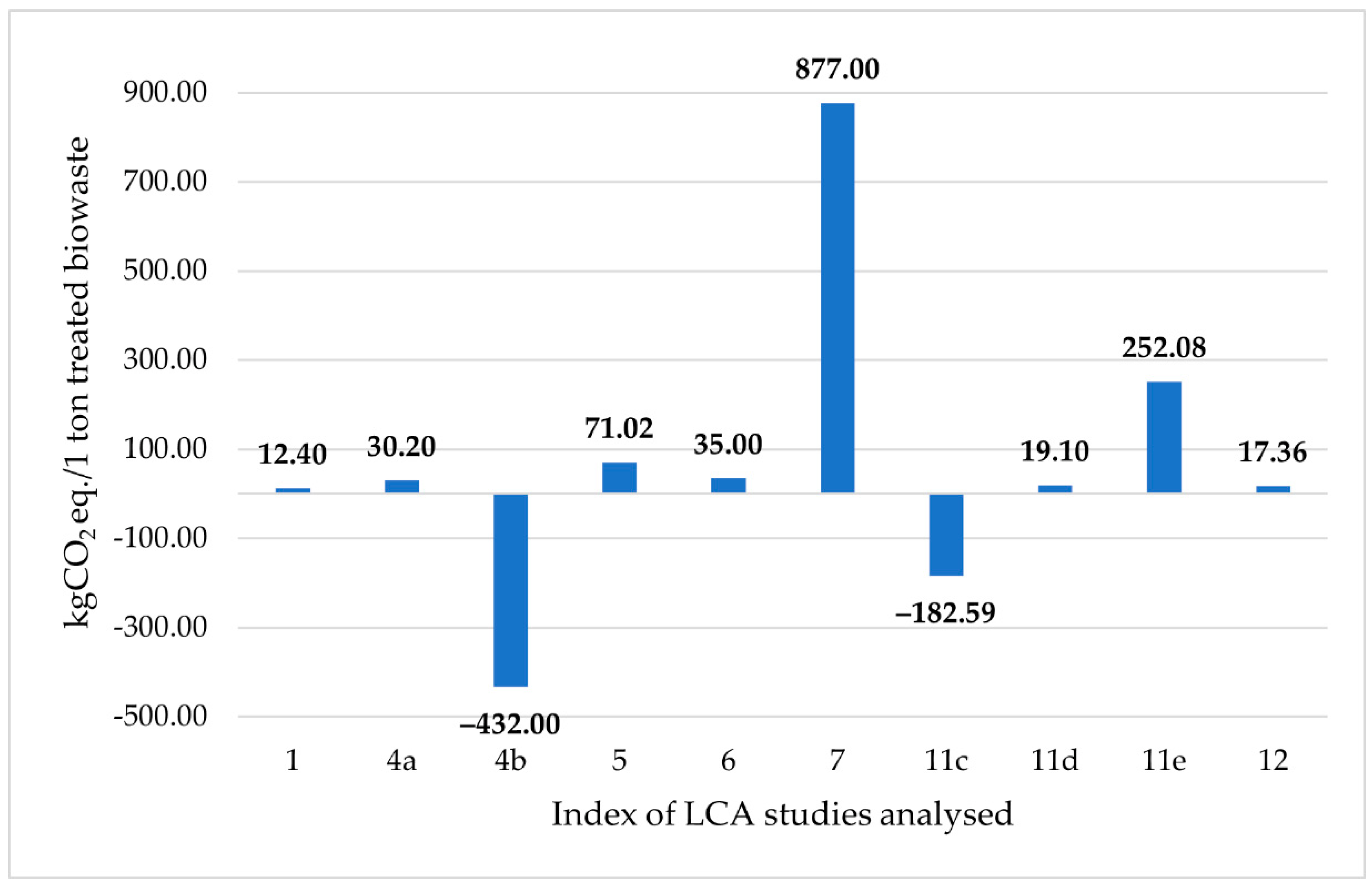
7.1. GWP for Biowaste Treatment by the BSFL
7.2. GWP for Insect Products
7.3. Energy Use
7.4. Land Use
8. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Gołasa, P.; Wysokiński, M.; Bieńkowska-Gołasa, W.; Gradziuk, P.; Golonko, M.; Gradziuk, B.; Siedlecka, A.; Gromada, A. Sources of greenhouse gas emissions in agriculture, with particular emphasis on emissions from energy used. Energies 2021, 14, 3784. [Google Scholar] [CrossRef]
- Tubiello, F.N.; Salvatore, M.; Rossi, S.; Ferrara, A.; Fitton, N.; Smith, P. The FAOSTAT database of greenhouse gas emissions from agriculture. Environ. Res. Lett. 2013, 8, 015009. [Google Scholar] [CrossRef]
- Madau, F.A.; Arru, B.; Furesi, R.; Pulina, P. Insect farming for feed and food production from a circular business model perspective. Sustainability 2020, 12, 5418. [Google Scholar] [CrossRef]
- Makkar, H.P.; Tran, G.; Heuzé, V.; Ankers, P. State-of-the-art on use of insects as animal feed. Anim. Feed Sci. Technol. 2014, 197, 1–33. [Google Scholar] [CrossRef]
- Perednia, D.A.; Anderson, J.; Rice, A. A comparison of the greenhouse gas production of black soldier fly larvae versus aerobic microbial decomposition of an organic feed material. J. Ecol. Environ. Sci. 2017, 5, 10–16. [Google Scholar]
- Surendra, K.; Tomberlin, J.K.; van Huis, A.; Cammack, J.A.; Heckmann, L.-H.L.; Khanal, S.K. Rethinking organic wastes bioconversion: Evaluating the potential of the black soldier fly (Hermetia illucens L.) (diptera: Stratiomyidae) (BSF). Waste Manag. 2020, 117, 58–80. [Google Scholar] [CrossRef] [PubMed]
- Franco, A.; Scieuzo, C.; Salvia, R.; Petrone, A.M.; Tafi, E.; Moretta, A. Lipids from Hermetia illucens, an innovative and sustainable. Source 2021, 13, 10198. [Google Scholar] [CrossRef]
- Mai, H.C.; Dao, N.D.; Lam, T.D.; Nguyen, B.V.; Nguyen, D.C.; Bach, L.G. Purification process, physicochemical properties, and fatty acid composition of black soldier fly (Hermetia illucens Linnaeus) larvae oil. J. Am. Oil Chem. Soc. 2019, 96, 1303–1311. [Google Scholar] [CrossRef]
- Soetemans, L.; Uyttebroek, M.; Bastiaens, L. Characteristics of chitin extracted from black soldier fly in different life stages. Int. J. Biol. Macromol. 2020, 165, 3206–3214. [Google Scholar] [CrossRef]
- Kawasaki, K.; Hashimoto, Y.; Hori, A.; Kawasaki, T.; Hirayasu, H.; Iwase, S.-I.; Hashizume, A.; Ido, A.; Miura, C.; Miura, T.; et al. Evaluation of black soldier fly (Hermetia illucens) larvae and pre-pupae raised on household organic waste, as potential ingredients for poultry feed. Animals 2019, 9, 98. [Google Scholar] [CrossRef]
- Spranghers, T.; Ottoboni, M.; Klootwijk, C.; Ovyn, A.; Deboosere, S.; De Meulenaer, B.; Michiels, J.; Eeckhout, M.; De Clercq, P.; De Smet, S. Nutritional composition of black soldier fly (Hermetia illucens) prepupae reared on different organic waste substrates. J. Sci. Food Agric. 2016, 97, 2594–2600. [Google Scholar] [CrossRef]
- Surendra, K.; Olivier, R.; Tomberlin, J.K.; Jha, R.; Khanal, S.K. Bioconversion of organic wastes into biodiesel and animal feed via insect farming. Renew. Energy 2016, 98, 197–202. [Google Scholar] [CrossRef]
- Gao, Z.; Wang, W.; Lu, X.; Zhu, F.; Liu, W.; Wang, X.; Lei, C. Bioconversion performance and life table of black soldier fly (Hermetia illucens) on fermented maize straw. J. Clean. Prod. 2019, 230, 974–980. [Google Scholar] [CrossRef]
- Giannetto, A.; Oliva, S.; Lanes, C.F.C.; de Araújo Pedron, F.; Savastano, D.; Baviera, C.; Parrino, V.; Lo Paro, G.; Spanò, N.C.; Cappello, T.; et al. Hermetia illucens (diptera: Stratiomydae) larvae and prepupae: Biomass production, fatty acid profile and expression of key genes involved in lipid metabolism. J. Biotechnol. 2020, 307, 44–54. [Google Scholar] [CrossRef] [PubMed]
- Liland, N.S.; Biancarosa, I.; Araujo, P.; Biemans, D.; Bruckner, C.G.; Waagbø, R.; Torstensen, B.E.; Lock, E.-J. Modulation of nutrient composition of black soldier fly (Hermetia illucens) larvae by feeding seaweed-enriched media. PLoS ONE 2017, 12, e0183188. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.Y.; Wu, L.; Li, B.; Zhang, D. Reproductive potential and nutritional composition of Hermetia illucens (diptera: Stratiomyidae) prepupae reared on different organic wastes. J. Econ. Entomol. 2020, 113, 527–537. [Google Scholar] [CrossRef]
- Truzzi, C.; Giorgini, E.; Annibaldi, A.; Antonucci, M.; Illuminati, S.; Scarponi, G.; Riolo, P.; Isidoro, N.; Conti, C.; Zarantoniello, M.; et al. Fatty acids profile of black soldier fly (Hermetia illucens): Influence of feeding substrate based on coffee-waste silverskin enriched with microalgae. Anim. Feed Sci. Technol. 2020, 259, 114309. [Google Scholar] [CrossRef]
- Oonincx, D.G.A.B.; van Broekhoven, S.; van Huis, A.; van Loon, J.J.A. Feed conversion, survival and development, and composition of four insect species on diets composed of food by-products. PLoS ONE 2015, 10, e0144601. [Google Scholar] [CrossRef]
- Lalander, C.H.; Fidjeland, J.; Diener, S.; Eriksson, S.; Vinneras, B. High waste-to-biomass conversion and efficient Salmonella spp. reduction using black soldier fly for waste recycling. Agron. Sustain. Dev. 2014, 35, 261–271. [Google Scholar] [CrossRef]
- Rehman, K.U.; Rehman, A.; Cai, M.; Zheng, L.; Xiao, X.; Somroo, A.A.; Wang, H.; Li, W.; Yu, Z.; Zhang, J. Conversion of mixtures of dairy manure and soybean curd residue by black soldier fly larvae (Hermetia illucens L.). J. Clean. Prod. 2017, 154, 366–373. [Google Scholar] [CrossRef]
- Ur Rehman, K.; Ur Rehman, R.; Somroo, A.A.; Cai, M.; Zheng, L.; Xiao, X.; Ur Rehman, A.; Rehman, A.; Tomberlin, J.K.; Yu, Z.; et al. Enhanced bioconversion of dairy and chicken manure by the interaction of exogenous bacteria and black soldier fly larvae. J. Environ. Manag. 2019, 237, 75–83. [Google Scholar] [CrossRef] [PubMed]
- Gold, M.; Cassar, C.M.; Zurbrügg, C.; Kreuzer, M.; Boulos, S.; Diener, S.; Mathys, A. Biowaste treatment with black soldier fly larvae: Increasing performance through the formulation of biowastes based on protein and carbohydrates. Waste Manag. 2019, 102, 319–329. [Google Scholar] [CrossRef] [PubMed]
- Lievens, S.; Poma, G.; De Smet, J.; Van Campenhout, L.; Covaci, A.; Van Der Borght, M. Chemical safety of black soldier fly larvae (Hermetia illucens), knowledge gaps and recommendations for future research: A critical review. J. Insects Food Feed 2021, 7, 383–396. [Google Scholar] [CrossRef]
- Gligorescu, A.; Toft, S.; Hauggaard-Nielsen, H.; Axelsen, J.A.; Nielsen, S.A. Development, growth and metabolic rate of Hermetia illucens larvae. J. Appl. Entomol. 2019, 143, 875–881. [Google Scholar] [CrossRef]
- Harnden, L.M.; Tomberlin, J.K. Effects of temperature and diet on black soldier fly, Hermetia illucens (L.) (diptera: Stratiomyidae) development. Forensic Sci. Int. 2016, 266, 109–116. [Google Scholar] [CrossRef]
- Tomberlin, J.K.; Sheppard, D.C.; Joyce, J.A. Selected life-history traits of black soldier flies (diptera: Stratiomyidae) reared on three artificial diets. Ann. Entomol. Soc. Am. 2002, 95, 379–386. [Google Scholar] [CrossRef]
- Henchion, M.; Hayes, M.; Mullen, A.M.; Fenelon, M.; Tiwari, B. Future protein supply and demand: Strategies and factors influencing a sustainable equilibrium. Foods 2017, 6, 53. [Google Scholar] [CrossRef]
- Gerber, P.J.; Steinfeld, H.; Henderson, B.; Mottet, A.; Opio, C.; Dijkman, J. Tackling Climate Change through Livestock—A Global Assessment of Emissions and Mitigation Opportunities; Food and Agriculture Organization of the United Nations (FAO): Italy, Rome, 2013; Volume 14. [Google Scholar]
- Philippe, F.-X.; Nicks, B. Review on greenhouse gas emissions from pig houses: Production of carbon dioxide, methane and nitrous oxide by animals and manure. Agric. Ecosyst. Environ. 2015, 199, 10–25. [Google Scholar] [CrossRef]
- McLeod, A. World Livestock 2011 Livestock in Food Security World; Food and Agriculture Organization of the United Nations (FAO): Italy, Rome, 2011. [Google Scholar]
- Brede, A.; Wecke, C.; Liebert, F. Does the optimal dietary methionine to cysteine ratio in diets for growing chickens respond to high inclusion rates of insect meal from Hermetia illucens? Animals 2018, 8, 187. [Google Scholar] [CrossRef]
- Cullere, M.; Schiavone, A.; Dabbou, S.; Gasco, L.; Zotte, A.D. Meat quality and sensory traits of finisher broiler chickens fed with black soldier fly (Hermetia Illucens L.) larvae fat as alternative fat source. Animals 2019, 9, 140. [Google Scholar] [CrossRef]
- Dabbou, S.; Gai, F.; Biasato, I.; Capucchio, M.T.; Biasibetti, E.; Dezzutto, D. Black soldier fly defatted meal as a dietary protein source for broiler chickens: Effects on growth performance, blood traits, gut morphology and histological features. J. Anim. Sci. Biotechnol. 2018, 9, 49. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.; Kim, Y.-M.; Park, Y.-K.; Yang, Y.-C.; Jung, B.-G.; Lee, B.-J. Black soldier fly (Hermetia illucens) larvae enhances immune activities and increases survivability of broiler chicks against experimental infection of Salmonella gallinarum. J. Veter. Med. Sci. 2018, 80, 736–740. [Google Scholar] [CrossRef]
- Mbhele, F.G.T.; Mnisi, C.M.; Mlambo, V. A nutritional evaluation of insect meal as a sustainable protein source for jumbo quails: Physiological and meat quality responses. Sustainability 2019, 11, 6592. [Google Scholar] [CrossRef]
- Onsongo, V.O.; Osuga, I.M.; Gachuiri, C.K.; Wachira, A.M.; Miano, D.M.; Tanga, C.M.; Ekesi, S.; Nakimbugwe, D.; Fiaboe, K.K.M. Insects for income generation through animal feed: Effect of dietary replacement of soybean and fish meal with black soldier fly meal on broiler growth and economic performance. J. Econ. Entomol. 2018, 111, 1966–1973. [Google Scholar] [CrossRef] [PubMed]
- Pieterse, E.; Erasmus, S.W.; Uushona, T.; Hoffman, L.C. Black soldier fly (Hermetia illucens) pre-pupae meal as a dietary protein source for broiler production ensures a tasty chicken with standard meat quality for every pot. J. Sci. Food Agric. 2018, 99, 893–903. [Google Scholar] [CrossRef]
- Ruhnke, I.; Normant, C.; Campbell, D.L.; Iqbal, Z.; Lee, C.; Hinch, G.N.; Roberts, J. Impact of on-range choice feeding with black soldier fly larvae (Hermetia illucens) on flock performance, egg quality, and range use of free-range laying hens. Anim. Nutr. 2018, 4, 452–460. [Google Scholar] [CrossRef]
- Schiavone, A.; Dabbou, S.; De Marco, M.; Cullere, M.; Biasato, I.; Biasibetti, E.; Capucchio, M.T.; Bergagna, S.; Dezzutto, D.; Meneguz, M.; et al. Black soldier fly larva fat inclusion in finisher broiler chicken diet as an alternative fat source. Animal 2018, 12, 2032–2039. [Google Scholar] [CrossRef]
- Secci, G.; Bovera, F.; Parisi, G.; Moniello, G. Quality of eggs and albumen technological properties as affected by Hermetia Illucens larvae meal in hens’ diet and hen age. Animals 2020, 10, 81. [Google Scholar] [CrossRef]
- Wallace, P.A.; Nyameasem, J.K.; Aboagye, G.A.; Affedzie-Obresi, S.; Nkegbe, K.; Murray, F.; Botchway, V.; Karbo, N.; Leschen, W.; Maquart, P.-O.; et al. Effects of replacing fishmeal with black soldier fly larval meal in the diets of grower-finishing guinea fowls reared under tropical conditions. Trop. Anim. Health Prod. 2018, 50, 1499–1507. [Google Scholar] [CrossRef]
- Xiao, X.; Jin, P.; Zheng, L.; Cai, M.; Yu, Z.; Yu, J.; Zhang, J. Effects of black soldier fly (Hermetia illucens) larvae meal protein as a fishmeal replacement on the growth and immune index of yellow catfish (Pelteobagrus fulvidraco). Aquac. Res. 2018, 49, 1569–1577. [Google Scholar] [CrossRef]
- Fawole, F.J.; Adeoye, A.A.; Tiamiyu, L.O.; Ajala, K.I.; Obadara, S.O.; Ganiyu, I.O. Substituting fishmeal with Hermetia illucens in the diets of African catfish (Clarias gariepinus): Effects on growth, nutrient utilization, haemato-physiological response, and oxidative stress biomarker. Aquaculture 2020, 518, 734849. [Google Scholar] [CrossRef]
- Bruni, L.; Belghit, I.; Lock, E.; Secci, G.; Taiti, C.; Parisi, G. Total replacement of dietary fish meal with black soldier fly (Hermetia illucens) larvae does not impair physical, chemical or volatile composition of farmed Atlantic salmon (Salmo salar L.). J. Sci. Food Agric. 2019, 100, 1038–1047. [Google Scholar] [CrossRef] [PubMed]
- Bruni, L.; Pastorelli, R.; Viti, C.; Gasco, L.; Parisi, G. Characterisation of the intestinal microbial communities of rainbow trout (Oncorhynchus mykiss) fed with Hermetia illucens (black soldier fly) partially defatted larva meal as partial dietary protein source. Aquaculture 2018, 487, 56–63. [Google Scholar] [CrossRef]
- Oncorhynchus, T.; Cardinaletti, G.; Randazzo, B.; Messina, M.; Zarantoniello, M.; Giorgini, E. Animals effects of graded dietary inclusion level of full-fat Hermetia illucens prepupae meal in practical diets. Animals 2019, 9, 251. [Google Scholar]
- Dumas, A.; Raggi, T.; Barkhouse, J.; Lewis, E.; Weltzien, E. The oil fraction and partially defatted meal of black soldier fly larvae (Hermetia illucens) affect differently growth performance, feed efficiency, nutrient deposition, blood glucose and lipid digestibility of rainbow trout (Oncorhynchus mykiss). Aquaculture 2018, 492, 24–34. [Google Scholar] [CrossRef]
- Huyben, D.; Vidaković, A.; Hallgren, S.W.; Langeland, M. High-throughput sequencing of gut microbiota in rainbow trout (Oncorhynchus mykiss) fed larval and pre-pupae stages of black soldier fly (Hermetia illucens). Aquaculture 2018, 500, 485–491. [Google Scholar] [CrossRef]
- Mancini, S.; Medina, I.; Iaconisi, V.; Gai, F.; Basto, A.; Parisi, G. Impact of black soldier fly larvae meal on the chemical and nutritional characteristics of rainbow trout fillets. Anim. Int. J. Anim. Biosci. 2018, 12, 1672–1681. [Google Scholar] [CrossRef]
- Terova, G.; Rimoldi, S.; Ascione, C.; Gini, E.; Ceccotti, C.; Gasco, L. Rainbow trout (Oncorhynchus mykiss) gut microbiota is modulated by insect meal from Hermetia illucens prepupae in the diet. Rev. Fish Biol. Fish. 2019, 29, 465–486. [Google Scholar] [CrossRef]
- Wang, G.; Peng, K.; Hu, J.; Yi, C.; Chen, X.; Wu, H.; Huang, Y. Evaluation of defatted black soldier fly (Hermetia illucens L.) larvae meal as an alternative protein ingredient for juvenile Japanese seabass (Lateolabrax japonicus) diets. Aquaculture 2019, 507, 144–154. [Google Scholar] [CrossRef]
- Biancarosa, I.; Sele, V.; Belghit, I.; Ørnsrud, R.; Lock, E.-J.; Amlund, H. Food additives & contaminants: Part A replacing fish meal with insect meal in the diet of Atlantic salmon (Salmo salar) does not impact the amount of contaminants in the feed and it lowers accumulation of arsenic in the fillet. Food Addit. Contam. Part A 2019, 36, 1191–1205. [Google Scholar] [CrossRef]
- Belghit, I.; Liland, N.S.; Waagbø, R.; Biancarosa, I.; Pelusio, N.; Li, Y.; Krogdahl, A. Potential of insect-based diets for Atlantic salmon (Salmo salar). Aquaculture 2018, 491, 72–81. [Google Scholar] [CrossRef]
- Belghit, I.; Liland, N.S.; Gjesdal, P.; Biancarosa, I.; Menchetti, E.; Li, Y. Black soldier fly larvae meal can replace fish meal in diets of sea-water phase Atlantic salmon (Salmo salar). Aquaculture 2019, 503, 609–619. [Google Scholar] [CrossRef]
- Stenberg, O.K.; Holen, E.; Piemontese, L.; Liland, N.S.; Lock, E.-J.; Espe, M.; Belghit, I. Effect of dietary replacement of fish meal with insect meal on in vitro bacterial and viral induced gene response in Atlantic salmon (Salmo salar) head kidney leukocytes. Fish Shellfish Immunol. 2019, 91, 223–232. [Google Scholar] [CrossRef] [PubMed]
- Zarantoniello, M.; Randazzo, B.; Truzzi, C.; Giorgini, E.; Marcellucci, C.; Vargas-Abúndez, J.A.; Zimbelli, A.; Annibaldi, A.; Parisi, G.; Tulli, F.; et al. A six-months study on black soldier fly (Hermetia illucens) based diets in zebrafish. Sci. Rep. 2019, 9, 8598. [Google Scholar] [CrossRef]
- Vongvichith, B.; Morioka, S.; Sugita, T.; Phousavanh, N.; Phetsanghanh, N.; Chanthasone, P.; Pommachan, P.; Nakamura, S. Evaluation of the efficacy of aquaculture feeds for the climbing perch Anabas testudineus: Replacement of fishmeal by black soldier fly Hermetia illucens prepupae. Fish. Sci. 2020, 86, 145–151. [Google Scholar] [CrossRef]
- Guerreiro, I.; Castro, C.; Antunes, B.; Coutinho, F.; Rangel, F.; Couto, A.; Serra, C.R.; Peres, H.; Pousão-Ferreira, P.; Matos, E.; et al. Catching black soldier fly for meagre: Growth, whole-body fatty acid profile and metabolic responses. Aquaculture 2020, 516, 734613. [Google Scholar] [CrossRef]
- Zhou, J.; Liu, S.; Ji, H.; Yu, H. Effect of replacing dietary fish meal with black soldier fly larvae meal on growth and fatty acid composition of Jian carp (Cyprinus carpio var. Jian). Aquac. Nutr. 2018, 24, 424–433. [Google Scholar] [CrossRef]
- Devic, E.; Leschen, W.; Murray, F.; Little, D. Growth performance, feed utilization and body composition of advanced nursing Nile tilapia (Oreochromis niloticus) fed diets containing Black Soldier Fly (Hermetia illucens) larvae meal. Aquac. Nutr. 2018, 24, 416–423. [Google Scholar] [CrossRef]
- Rimoldi, S.; Gini, E.; Iannini, F.; Gasco, L. The effects of dietary insect meal from Hermetia illucens prepupae on autochthonous gut microbiota of rainbow trout (Oncorhynchus mykiss). Animals 2019, 9, 143. [Google Scholar] [CrossRef]
- Belghit, I.; Waagbø, R.; Lock, E.-J.; Liland, N.S. Insect-based diets high in lauric acid reduce liver lipids in freshwater Atlantic salmon. Aquac. Nutr. 2019, 25, 343–357. [Google Scholar] [CrossRef]
- Altmann, B.A.; Neumann, C.; Rothstein, S.; Liebert, F.; Mörlein, D. Do dietary soy alternatives lead to pork quality improvements or drawbacks? A look into micro-alga and insect protein in swine diets. Meat Sci. 2019, 153, 26–34. [Google Scholar] [CrossRef] [PubMed]
- Biasato, I.; Renna, M.; Gai, F.; Dabbou, S.; Meneguz, M.; Perona, G. Partially defatted black soldier fly larva meal inclusion in piglet diets: Effects on the growth performance, nutrient digestibility, blood profile, gut morphology and histological features. J. Anim. Sci. Biotechnol. 2019, 10, 12. [Google Scholar] [CrossRef] [PubMed]
- Chia, S.Y.; Tanga, C.M.; Osuga, I.M.; Alaru, A.O.; Mwangi, D.M.; Githinji, M. Meal on growth performance, blood profiles and economics of growing pigs in Kenya. Animals 2019, 9, 705. [Google Scholar] [CrossRef] [PubMed]
- Spranghers, T.; Michiels, J.; Vrancx, J.; Ovyn, A.; Eeckhout, M.; De Clercq, P.; De Smet, S. Gut antimicrobial effects and nutritional value of black soldier fly (Hermetia illucens L.) prepupae for weaned piglets. Anim. Feed Sci. Technol. 2018, 235, 33–42. [Google Scholar] [CrossRef]
- Yu, M.; Li, Z.; Chen, W.; Rong, T.; Wang, G.; Li, J.; Ma, X. Use of Hermetia illucens larvae as a dietary protein source: Effects on growth performance, carcass traits, and meat quality in finishing pigs. Meat Sci. 2019, 158, 107837. [Google Scholar] [CrossRef]
- Fukuda, E.P.; Cox, J.R.; Wickersham, T.A.; Drewery, M.L. Evaluation of black soldier fly larvae (Hermetia illucens) as a protein supplement for beef steers consuming low-quality forage. Transl. Anim. Sci. 2022, 6, txac018. [Google Scholar] [CrossRef]
- EFSA Scientific Committee. Risk profile related to production and consumption of insects as food and feed. EFSA J. 2015, 13, 4257. [Google Scholar] [CrossRef]
- Salomone, R.; Saija, G.; Mondello, G.; Giannetto, A.; Fasulo, S.; Savastano, D. Environmental impact of food waste bioconversion by insects: Application of life cycle assessment to process using Hermetia illucens. J. Clean. Prod. 2017, 140, 890–905. [Google Scholar] [CrossRef]
- Spang, E.S.; Moreno, L.C.; Pace, S.A.; Achmon, Y.; Donis-Gonzalez, I.; Gosliner, W.A.; Jablonski-Sheffield, M.P.; Momin, A.; Quested, T.E.; Winans, K.S.; et al. Food loss and waste: Measurement, drivers, and solutions. Annu. Rev. Environ. Resour. 2019, 44, 117–156. [Google Scholar] [CrossRef]
- European Parliament. Food Waste: The Problem in the EU in Numbers [Infographic]; European Parliament: Strasbourg, France, 2017; pp. 15–17. [Google Scholar]
- European Commission. The European Green Deal. 640 Final. Eur. Comm. 2019, 53, 24. Available online: https://eur-lex.europa.eu/resource.html?uri=cellar:b828d165-1c22-11ea-8c1f-01aa75ed71a1.0002.02/DOC_1&format=PDF (accessed on 10 July 2022).
- Siddiqui, S.A.; Ristow, B.; Rahayu, T.; Putra, N.S.; Yuwono, N.W.; Nisa’, K.; Mategeko, B.; Smetana, S.; Saki, M.; Nawaz, A.; et al. Black soldier fly larvae (BSFL) and their affinity for organic waste processing. Waste Manag. 2022, 140, 1–13. [Google Scholar] [CrossRef] [PubMed]
- European Parliament and Council. Directive (EU) 2018/850 of the European parliament and of the council of 30 May 2018 amending directive 1999/31/EC on the landfill of waste. Off. J. Eur. Union 2018, 2018, 100–108. [Google Scholar]
- Chemat, F.; Vian, M.A. Larvae mediated valorization of industrial, agriculture and food wastes: Biorefinery concept and products. Processes 2020, 8, 8572020. [Google Scholar]
- Ilari, A.; Duca, D.; Toscano, G.; Pedretti, E.F. Evaluation of cradle to gate environmental impact of frozen green bean production by means of life cycle assessment. J. Clean. Prod. 2019, 236, 117638. [Google Scholar] [CrossRef]
- Ilari, A.; Toscano, G.; Boakye-Yiadom, K.A.; Duca, D.; Foppa Pedretti, E. Life cycle assessment of protected strawberry productions in central Italy. Sustainability 2021, 13, 4879. [Google Scholar] [CrossRef]
- Cooper, J.S.; Fava, J.A. Life-cycle assessment practitioner survey: Summary of results. J. Ind. Ecol. 2006, 10, 12–14. [Google Scholar] [CrossRef]
- Boakye-Yiadom, K.; Duca, D.; Pedretti, E.F.; Ilari, A. Environmental performance of chocolate produced in Ghana using life cycle assessment. Sustainability 2021, 13, 6155. [Google Scholar] [CrossRef]
- Zargar, S.; Yao, Y.; Tu, Q. A review of inventory modeling methods for missing data in life cycle assessment. J. Ind. Ecol. 2022, 1–14. [Google Scholar] [CrossRef]
- Smetana, S.; Palanisamy, M.; Mathys, A.; Heinz, V. Sustainability of insect use for feed and food: Life cycle assessment perspective. J. Clean. Prod. 2016, 137, 741–751. [Google Scholar] [CrossRef]
- Smetana, S.; Schmitt, E.; Mathys, A. Sustainable use of Hermetia illucens insect biomass for feed and food: Attributional and consequential life cycle assessment. Resour. Conserv. Recycl. 2019, 144, 285–296. [Google Scholar] [CrossRef]
- Pang, W.; Hou, D.; Nowar, E.; Chen, H.; Zhang, J.; Zhang, G.; Li, Q.; Wang, S. The influence on carbon, nitrogen recycling, and greenhouse gas emissions under different C/N ratios by black soldier fly. Environ. Sci. Pollut. Res. 2020, 27, 42767–42777. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Li, Z.; Nowar, E.E.; Chen, J.; Pang, W.; Hou, D.; Hu, R.; Jiang, H.; Zhang, J.; Li, Q. Effect of batch feeding times on greenhouse gas and NH3 emissions during meat and bone meal bioconversion by black soldier fly larvae. Waste Biomass Valorization 2021, 12, 3889–3897. [Google Scholar] [CrossRef]
- Pang, W.; Hou, D.; Chen, J.; Nowar, E.; Li, Z.; Hu, R.; Tomberlin, J.K.; Yu, Z.; Li, Q.; Wang, S. Reducing greenhouse gas emissions and enhancing carbon and nitrogen conversion in food wastes by the black soldier fly. J. Environ. Manag. 2020, 260, 110066. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Hou, D.; Pang, W.; Nowar, E.E.; Tomberlin, J.K.; Hu, R.; Chen, H.; Xie, J.; Zhang, J.; Yu, Z.; et al. Effect of moisture content on greenhouse gas and NH3 emissions from pig manure converted by black soldier fly. Sci. Total. Environ. 2019, 697, 133840. [Google Scholar] [CrossRef] [PubMed]
- Ermolaev, E.; Lalander, C.; Vinnerås, B. Greenhouse gas emissions from small-scale fly larvae composting with Hermetia illucens. Waste Manag. 2019, 96, 65–74. [Google Scholar] [CrossRef] [PubMed]
- Lindberg, L.; Ermolaev, E.; Vinnerås, B.; Lalander, C. Process efficiency and greenhouse gas emissions in black soldier fly larvae composting of fruit and vegetable waste with and without pre-treatment. J. Clean. Prod. 2022, 338, 130552. [Google Scholar] [CrossRef]
- Mertenat, A.; Diener, S.; Zurbrügg, C. Black Soldier Fly biowaste treatment—Assessment of global warming potential. Waste Manag. 2019, 84, 173–181. [Google Scholar] [CrossRef]
- Guo, H.; Jiang, C.; Zhang, Z.; Lu, W.; Wang, H. Material flow analysis and life cycle assessment of food waste bioconversion by black soldier fly larvae (Hermetia illucens L.). Sci. Total Environ. 2020, 750, 141656. [Google Scholar] [CrossRef]
- Parodi, A.; De Boer, I.J.; Gerrits, W.J.; Van Loon, J.J.; Heetkamp, M.J.; Van Schelt, J.; Bolhuis, J.E.; Van Zanten, H.H. Bioconversion efficiencies, greenhouse gas and ammonia emissions during black soldier fly rearing—A mass balance approach. J. Clean. Prod. 2020, 271, 122488. [Google Scholar] [CrossRef]
- Singh, A.; Kumari, K. An inclusive approach for organic waste treatment and valorisation using black soldier fly larvae: A review. J. Environ. Manag. 2019, 251, 109569. [Google Scholar] [CrossRef]
- Kim, C.-H.; Ryu, J.; Lee, J.; Ko, K.; Lee, J.-Y.; Park, K.Y.; Chung, H. Use of black soldier fly larvae for food waste treatment and energy production in Asian countries: A review. Processes 2021, 9, 161. [Google Scholar] [CrossRef]
- Parodi, A.; Gerrits, W.J.; Van Loon, J.J.; De Boer, I.J.; Aarnink, A.J.; Van Zanten, H.H. Black soldier fly reared on pig manure: Bioconversion efficiencies, nutrients in the residual material, greenhouse gas and ammonia emissions. Waste Manag. 2021, 126, 674–683. [Google Scholar] [CrossRef] [PubMed]
- Sánchez, A.; Artola, A.; Font, X.; Gea, T.; Barrena, R.; Gabriel, D.; Sanchez-Monedero, M.A.; Roig, A.; Cayuela, M.L.; Mondini, C. Greenhouse gas emissions from organic waste composting. Environ. Chem. Lett. 2015, 13, 223–238. [Google Scholar] [CrossRef]
- Wang, Q.; Awasthi, M.K.; Ren, X.; Zhao, J.; Li, R.; Wang, Z.; Wang, M.; Chen, H.; Zhang, Z. Combining biochar, zeolite and wood vinegar for composting of pig manure: The effect on greenhouse gas emission and nitrogen conservation. Waste Manag. 2018, 74, 221–230. [Google Scholar] [CrossRef]
- IPCC 2013. Climate change 2013: The physical science basis. In Contribution of Working Group I to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change; Cambridge University Press: Cambridge, UK, 2013. [Google Scholar]
- Meslé, M.; Dromart, G.; Oger, P. Microbial methanogenesis in subsurface oil and coal. Res. Microbiol. 2013, 164, 959–972. [Google Scholar] [CrossRef]
- Jeon, H.; Park, S.; Choi, J.; Jeong, G.; Lee, S.-B.; Choi, Y.; Lee, S.-J. The intestinal bacterial community in the food waste-reducing larvae of Hermetia illucens. Curr. Microbiol. 2011, 62, 1390–1399. [Google Scholar] [CrossRef]
- Tsutsui, H.; Fujiwara, T.; Matsukawa, K.; Funamizu, N. Nitrous oxide emission mechanisms during intermittently aerated composting of cattle manure. Bioresour. Technol. 2013, 141, 205–211. [Google Scholar] [CrossRef]
- Maeda, K.; Toyoda, S.; Shimojima, R.; Osada, T.; Hanajima, D.; Morioka, R.; Yoshida, N. Source of nitrous oxide emissions during the cow manure composting process as revealed by isotopomer analysis of and amoa abundance in betaproteobacterial Ammonia oxidizing bacteria. Appl. Environ. Microbiol. 2010, 76, 1555–1562. [Google Scholar] [CrossRef]
- Awasthi, M.K.; Wang, Q.; Huang, H.; Ren, X.; Lahori, A.H.; Mahar, A.; Ali, A.; Shen, F.; Li, R.; Zhang, Z. Influence of zeolite and lime as additives on greenhouse gas emissions and maturity evolution during sewage sludge composting. Bioresour. Technol. 2016, 216, 172–181. [Google Scholar] [CrossRef]
- Cayuela, M.L.; Sánchez-Monedero, M.A.; Roig, A.; Sinicco, T.; Mondini, C. Biochemical changes and GHG emissions during composting of lignocellulosic residues with different N-rich by-products. Chemosphere 2012, 88, 196–203. [Google Scholar] [CrossRef]
- Hao, X.; Chang, C.; Larney, F.J.; Travis, G.R. Greenhouse gas emissions during cattle feedlot manure composting. J. Environ. Qual. 2002, 31, 700. [Google Scholar] [CrossRef]
- Lv, B.; Zhang, D.; Cui, Y.; Yin, F. Effects of C/N ratio and earthworms on greenhouse gas emissions during vermicomposting of sewage sludge. Bioresour. Technol. 2018, 268, 408–414. [Google Scholar] [CrossRef]
- Nigussie, A.; Kuyper, T.W.; Bruun, S.; de Neergaard, A. Vermicomposting as a technology for reducing nitrogen losses and greenhouse gas emissions from small-scale composting. J. Clean. Prod. 2016, 139, 429–439. [Google Scholar] [CrossRef]
- Van der Heyden, C.; Demeyer, P.; Volcke, E.I.P. Mitigating emissions from pig and poultry housing facilities through air scrubbers and biofilters: State-of-the-art and perspectives. Biosyst. Eng. 2015, 134, 74–93. [Google Scholar] [CrossRef]
- Jiang, J.; Liu, X.; Huang, Y.; Huang, H. Inoculation with nitrogen turnover bacterial agent appropriately increasing nitrogen and promoting maturity in pig manure composting. Waste Manag. 2015, 39, 78–85. [Google Scholar] [CrossRef]
- Yang, F.; Li, G.; Shi, H.; Wang, Y. Effects of phosphogypsum and superphosphate on compost maturity and gaseous emissions during kitchen waste composting. Waste Manag. 2015, 39, 70–76. [Google Scholar] [CrossRef]
- Rai, R.; Rajput, M.; Agrawal, M.; Agrawal, S. Gaseous air pollutants: A review on current and future trends of emissions and impact on agriculture. J. Sci. Res. 2011, 55, 77–102. [Google Scholar]
- Yuesi, W.; Yinghong, W. Quick measurement of CH4, CO2 and N2O emissions from a short-plant ecosystem. Adv. Atmos. Sci. 2003, 20, 842–844. [Google Scholar] [CrossRef]
- Cardador, M.J.; Reyes-Palomo, C.; Díaz-Gaona, C.; Arce, L.; Rodríguez-Estévez, V. Review of the methodologies for measurement of greenhouse gas emissions in livestock farming: Pig farms as a case of study. Crit. Rev. Anal. Chem. 2020, 52, 1029–1047. [Google Scholar] [CrossRef]
- De Klein, C.A.M.; Harvey, M. Nitrous Oxide Chamber Methodology Guidelines; Ministry for Primary Industries: Wellington, New Zealand, 2012. [Google Scholar]
- Pihlatie, M.K.; Christiansen, J.R.; Aaltonen, H.; Korhonen, J.F.; Nordbo, A.; Rasilo, T.; Benanti, G.; Giebels, M.; Helmy, M.; Sheehy, J.; et al. Comparison of static chambers to measure CH4 emissions from soils. Agric. For. Meteorol. 2013, 171–172, 124–136. [Google Scholar] [CrossRef]
- Jämsén, M.; Agar, D.; Alakoski, E.; Tampio, E.; Wihersaari, M. Measurement methodology for greenhouse gas emissions from storage of forest chips—A review. Renew. Sustain. Energy Rev. 2015, 51, 1617–1623. [Google Scholar] [CrossRef]
- Oertel, C.; Matschullat, J.; Zurba, K.; Zimmermann, F.; Erasmi, S. Greenhouse gas emissions from soils—A review. Geochemistry 2016, 76, 327–352. [Google Scholar] [CrossRef]
- Smith, K.A.; Dobbie, K.E. The impact of sampling frequency and sampling times on chamber-based measurements of N2O emissions from fertilized soils. Glob. Chang. Biol. 2001, 7, 933–945. [Google Scholar] [CrossRef]
- Balogh, J.; Nagy, Z.; Fóti, S.; Pintér, K.; Czóbel, S.; Péli, E.R.; Acosta, M.; Marek, M.V.; Csintalan, Z.; Tuba, Z. Comparison of CO2 and H2O fluxes over grassland vegetations measured by the eddy-covariance technique and by open system chamber. Photosynthetica 2007, 45, 288–292. [Google Scholar] [CrossRef]
- Matos, J.S.; de Aráujo, L.P.; Allaman, I.B.; Lôbo, I.P.; de Oliva, S.T.; Tavares, T.M.; Neto, J.A.D.A. Evaluation of the reduction of methane emission in swine and bovine manure treated with black soldier fly larvae (Hermetia illucens L.). Environ. Monit. Assess. 2021, 193, 480. [Google Scholar] [CrossRef]
- Fan, Z.; Zhang, J.; Fan, C.-W.; Pennise, D.M. The MMT bag for emission source sampling: Design and evaluation. J. Air Waste Manag. Assoc. 2001, 51, 60–68. [Google Scholar] [CrossRef][Green Version]
- Papurello, D. Direct injection mass spectrometry technique for the odorant losses at ppb(v) level from nalophan™ sampling bags. Int. J. Mass Spectrom. 2018, 436, 137–146. [Google Scholar] [CrossRef]
- Rapson, T.D.; Dacres, H. Analytical techniques for measuring nitrous oxide. TrAC Trends Anal. Chem. 2014, 54, 65–74. [Google Scholar] [CrossRef]
- Ferraz-Almeida, R.; Spokas, K.A.; de Oliveira, R.C. Columns and detectors recommended in gas chromatography to measure greenhouse emission and O2 uptake in soil: A review. Commun. Soil Sci. Plant Anal. 2020, 51, 582–594. [Google Scholar] [CrossRef]
- Sudhakar, P.; Latha, P.; Reddy, P.V. (Eds.) Analytical techniques. In Phenotyping Crop Plants for Physiological and Biochemical Traits; Elsevier Inc.: Amsterdam, The Netherlands, 2016; pp. 137–149. [Google Scholar] [CrossRef]
- Mowry, C.; Jarek, R.L.; Román-Kustas, J.; Telles, A.C.; Pimentel, A.S. Gas analysis by mass spectrometry. Mater. Charact. 2020, 10, 143–152. [Google Scholar] [CrossRef]
- Gruber, B.; David, F.; Sandra, P. Capillary gas chromatography-mass spectrometry: Current trends and perspectives. TrAC Trends Anal. Chem. 2020, 124, 115475. [Google Scholar] [CrossRef]
- Hodgkinson, J.; Tatam, R.P. Optical gas sensing: A review. Meas. Sci. Technol. 2013, 24, 012004. [Google Scholar] [CrossRef]
- Aldhafeeri, T.; Tran, M.-K.; Vrolyk, R.; Pope, M.; Fowler, M. A review of methane gas detection sensors: Recent developments and future perspectives. Inventions 2020, 5, 28. [Google Scholar] [CrossRef]
- Lin, C.-H.; Grant, R.H.; Heber, A.J.; Johnston, C.T. Application of open-path fourier transform infrared spectroscopy (OP-FTIR) to measure greenhouse gas concentrations from agricultural fields. Atmos. Meas. Tech. 2019, 12, 3403–3415. [Google Scholar] [CrossRef]
- Korotcenkov, G. Handbook of Gas Sensor Materials: Properties, Advantages and Shortcomings for Applications; Springer: Berlin/Heidelberg, Germany, 2014; Volume 1. [Google Scholar]
- Fiddler, M.N.; Begashaw, I.; Mickens, M.A.; Collingwood, M.S.; Assefa, Z.; Bililign, S. Laser spectroscopy for atmospheric and environmental sensing. Sensors 2009, 9, 10447–10512. [Google Scholar] [CrossRef]
- Popa, D.; Udrea, F. Towards integrated mid-infrared gas sensors. Sensors 2019, 19, 2076. [Google Scholar] [CrossRef]
- Cui, X.; Dong, F.; Zhang, Z.; Xia, H.; Pang, T.; Sun, P.; Wu, B.; Liu, S.; Han, L.; Li, Z.; et al. Environmental application of high sensitive gas sensors with tunable diode laser absorption spectroscopy. InTech 2018, 11, 13. [Google Scholar] [CrossRef]
- Fu, B.; Zhang, C.; Lyu, W.; Sun, J.; Shang, C.; Cheng, Y.; Xu, L. Recent progress on laser absorption spectroscopy for determination of gaseous chemical species. Appl. Spectrosc. Rev. 2020, 57, 112–152. [Google Scholar] [CrossRef]
- Gupta, V.P. Chapter 1—Introduction and overview. In Molecular and Laser Spectroscopy; Elsevier: Amsterdam, The Netherlands, 2020; pp. 1–40. [Google Scholar] [CrossRef]
- Maity, A.; Maithani, S.; Pradhan, M. Cavity Ring-Down Spectroscopy: Recent Technological Advances and Applications; Elsevier Inc.: Amsterdam, The Netherlands, 2020. [Google Scholar] [CrossRef]
- Borri, S.; Insero, G.; Santambrogio, G.; Mazzotti, D.; Cappelli, F.; Galli, I.; Galzerano, G.; Marangoni, M.; Laporta, P.; Di Sarno, V.; et al. High-precision molecular spectroscopy in the mid-infrared using quantum cascade lasers. Appl. Phys. B Lasers Opt. 2019, 125, 18. [Google Scholar] [CrossRef]
- Haas, J.; Catalán, E.V.; Piron, P.; Karlsson, M.; Mizaikoff, B. Infrared spectroscopy based on broadly tunable quantum cascade lasers and polycrystalline diamond waveguides. Analyst 2018, 143, 5112–5119. [Google Scholar] [CrossRef]
- Lambrecht, A.; Schmitt, K. Mid-Infrared Gas-Sensing Systems and Applications; Elsevier Ltd.: Amsterdam, The Netherlands, 2019. [Google Scholar] [CrossRef]
- Sigrist, M. Photoacoustic Spectroscopy and Applications, 3rd ed.; Elsevier Ltd.: Amsterdam, The Netherlands, 2017. [Google Scholar] [CrossRef]
- Sampaolo, A.; Patimisco, P.; Giglio, M.; Zifarelli, A.; Wu, H.; Dong, L.; Spagnolo, V. Quartz-enhanced photoacoustic spectroscopy for multi-gas detection: A review. Anal. Chim. Acta 2021, 1202, 338894. [Google Scholar] [CrossRef] [PubMed]
- Hussain, C.M.; Keçili, R. Electrochemical techniques for environmental analysis. Mod. Environ. Anal. Tech. Pollut. 2020, 199–222. [Google Scholar] [CrossRef]
- Simões, F.R.; Xavier, M.G. Electrochemical Sensors; Elsevier Inc.: Amsterdam, The Netherlands, 2017. [Google Scholar] [CrossRef]
- Xiong, L.; Compton, R.G. Amperometric gas detection: A review. Int. J. Electrochem. Sci. 2014, 9, 7152–7181. [Google Scholar]
- Nasri, A.; Pétrissans, M.; Fierro, V.; Celzard, A. Gas sensing based on organic composite materials: Review of sensor types, progresses and challenges. Mater. Sci. Semicond. Process. 2021, 128, 105744. [Google Scholar] [CrossRef]
- Gautam, Y.K.; Sharma, K.; Tyagi, S.; Ambedkar, A.K.; Chaudhary, M.; Singh, B.P. Nanostructured metal oxide semiconductor-based sensors for greenhouse gas detection: Progress and challenges. R. Soc. Open Sci. 2021, 8, 201324. [Google Scholar] [CrossRef] [PubMed]
- Naganowska-Nowak, A.; Konieczka, P.; Przyjazny, A.; Namieśnik, J. Development of techniques of generation of gaseous standard mixtures. Crit. Rev. Anal. Chem. 2005, 35, 31–55. [Google Scholar] [CrossRef]
- Larrey-lassalle, P.; Perfido, D.; Naneci, S. Life cycle assessment applied to nature-based solutions: Learnings, methodological challenges, and perspectives from a critical analysis of the literature. Land 2022, 11, 649. [Google Scholar] [CrossRef]
- ISO 14040; Environmental Management—Life Cycle Assessment—Principles and Framework. ISO: Geneva, Switzerland, 2006.
- ISO 14044; Environmental Management—Life Cycle Assessment—Requirements and Guidelines. ISO: Geneva, Switzerland, 2006.
- Sultana, R.; Rashedi, A.; Khanam, T.; Jeong, B.; Hosseinzadeh-Bandbafha, H.; Hussain, M. Life cycle environmental sustainability and energy assessment of timber wall construction: A comprehensive overview. Sustainability 2022, 14, 4161. [Google Scholar] [CrossRef]
- Pedretti, E.F.; Boakye-Yiadom, K.A.; Valentini, E.; Ilari, A.; Duca, D. Life cycle assessment of spinach produced in central and southern Italy. Sustainability 2021, 13, 10001. [Google Scholar] [CrossRef]
- Oonincx, D.G.A.B.; de Boer, I.J.M. Environmental impact of the production of mealworms as a protein source for humans—A life cycle assessment. PLoS ONE 2012, 7, e51145. [Google Scholar] [CrossRef]
- Miglietta, P.P.; De Leo, F.; Ruberti, M.; Massari, S. Mealworms for food: A water footprint perspective. Water 2015, 7, 6190–6203. [Google Scholar] [CrossRef]
- van Zanten, H.H.; Mollenhorst, H.; Oonincx, D.; Bikker, P.; Meerburg, B.; De Boer, I. From environmental nuisance to environmental opportunity: Housefly larvae convert waste to livestock feed. J. Clean. Prod. 2015, 102, 362–369. [Google Scholar] [CrossRef]
- Roffeis, M.; Muys, B.; Almeida, J.; Mathijs, E.; Achten, W.; Pastor, B.; Velásquez, Y.; Martinez-Sanchez, A.; Rojo, S. Pig manure treatment with housefly (Musca domestica) rearing—An environmental life cycle assessment. J. Insects Food Feed 2015, 1, 195–214. [Google Scholar] [CrossRef]
- Halloran, A.; Hanboonsong, Y.; Roos, N.; Bruun, S. Life cycle assessment of cricket farming in north-eastern Thailand. J. Clean. Prod. 2017, 156, 83–94. [Google Scholar] [CrossRef]
- EC. Regulation (EU) 1069/2009 animal by-products. Off. J. Eur. Union 2019, 53, 1689–1699. [Google Scholar]
- Setti, L.; Francia, E.; Pulvirenti, A.; De Leo, R.; Martinelli, S.; Maistrello, L.; Macavei, L.I.; Montorsi, M.; Barbi, S.; Ronga, D. Bioplastic film from black soldier fly prepupae proteins used as mulch: Preliminary results. Agronomy 2020, 10, 933. [Google Scholar] [CrossRef]
- Nuvoli, D.; Montevecchi, G.; Lovato, F.; Masino, F.; Van Der Borght, M.; Messori, M.; Antonelli, A. Protein films from black soldier fly (Hermetia illucens, diptera: Stratiomyidae) prepupae: Effect of protein solubility and mild crosslinking. J. Sci. Food Agric. 2021, 101, 4506–4513. [Google Scholar] [CrossRef]
- Rosa, R.; Spinelli, R.; Neri, P.; Pini, M.; Barbi, S.; Montorsi, M.; Maistrello, L.; Marseglia, A.; Caligiani, A.; Ferrari, A.M. Life cycle assessment of chemical vs enzymatic-assisted extraction of proteins from black soldier fly prepupae for the preparation of biomaterials for potential agricultural use. ACS Sustain. Chem. Eng. 2020, 8, 14752–14764. [Google Scholar] [CrossRef]
- Li, Q.; Zheng, L.; Cai, H.; Garza, E.; Yu, Z.; Zhou, S. From organic waste to biodiesel: Black soldier fly, Hermetia illucens, makes it feasible. Fuel 2011, 90, 1545–1548. [Google Scholar] [CrossRef]
- Wong, C.Y.; Aris, M.N.M.; Daud, H.; Lam, M.K.; Yong, C.S.; Abu Hasan, H.; Chong, S.; Show, P.L.; Hajoeningtijas, O.D.; Ho, Y.C.; et al. In-situ yeast fermentation to enhance bioconversion of coconut endosperm waste into larval biomass of Hermetia illucens: Statistical augmentation of larval lipid content. Sustainability 2020, 12, 1558. [Google Scholar] [CrossRef]
- Komakech, A.; Sundberg, C.; Jönsson, H.; Vinnerås, B. Life cycle assessment of biodegradable waste treatment systems for sub-saharan African cities. Resour. Conserv. Recycl. 2015, 99, 100–110. [Google Scholar] [CrossRef]
- Rustad, I.H. Life Cycle Assessment of Fish Feed Produced from the Black Soldier Fly (Hermetia illucens); Norwegian University of Science and Technology: Trondheim, Norway, 2016; Available online: https://ntnuopen.ntnu.no/ntnu-xmlui/bitstream/handle/11250/2413536/14657_FULLTEXT.pdf?sequence=1 (accessed on 10 July 2022).
- Mondello, G.; Salomone, R.; Ioppolo, G.; Saija, G.; Sparacia, S.; Lucchetti, M.C. Comparative LCA of alternative scenarios for waste treatment: The case of food waste production by the mass-retail sector. Sustainability 2017, 9, 827. [Google Scholar] [CrossRef]
- Spinelli, R.; Neri, P.; Pini, M.; Barbi, S.; Montorsi, M.; Ferrari, A.M. Using black soldier flies (Hermetia illucens) to bioconvert waste from the livestock production chain: A life cycle assessment case study. WIT Trans. Ecol. Environ. 2019, 231, 47–58. [Google Scholar] [CrossRef]
- Bosch, G.; van Zanten, H.; Zamprogna, A.; Veenenbos, M.; Meijer, N.; van der Fels-Klerx, H.; van Loon, J. Conversion of organic resources by black soldier fly larvae: Legislation, efficiency and environmental impact. J. Clean. Prod. 2019, 222, 355–363. [Google Scholar] [CrossRef]
- Ites, S.; Smetana, S.; Toepfl, S.; Heinz, V. Modularity of insect production and processing as a path to efficient and sustainable food waste treatment. J. Clean. Prod. 2020, 248, 119248. [Google Scholar] [CrossRef]
- Ekvall, T. Attributional and consequential life cycle assessment. In Sustainability Assessment the 21st Century; IntechOpen: London, UK, 2020; pp. 1–21. [Google Scholar] [CrossRef]
- McLaren, S.; Berardy, A.; Henderson, A.; Holden, N.; Huppertz, T.; Jolliet, O. Integration of Environment and Nutrition in Life Cycle Assessment of Food Items: Opportunities and Challenges. Food and Agriculture Organization of the United Nations (FAO): Italy, Rome, 2021. [Google Scholar] [CrossRef]
- Schrijvers, D.L.; Loubet, P.; Sonnemann, G. Developing a systematic framework for consistent allocation in LCA. Int. J. Life Cycle Assess. 2016, 21, 976–993. [Google Scholar] [CrossRef]
- Perminova, T.; Sirina, N.; Laratte, B.; Baranovskaya, N.; Rikhvanov, L. Methods for land use impact assessment: A review. Environ. Impact Assess. Rev. 2016, 60, 64–74. [Google Scholar] [CrossRef]
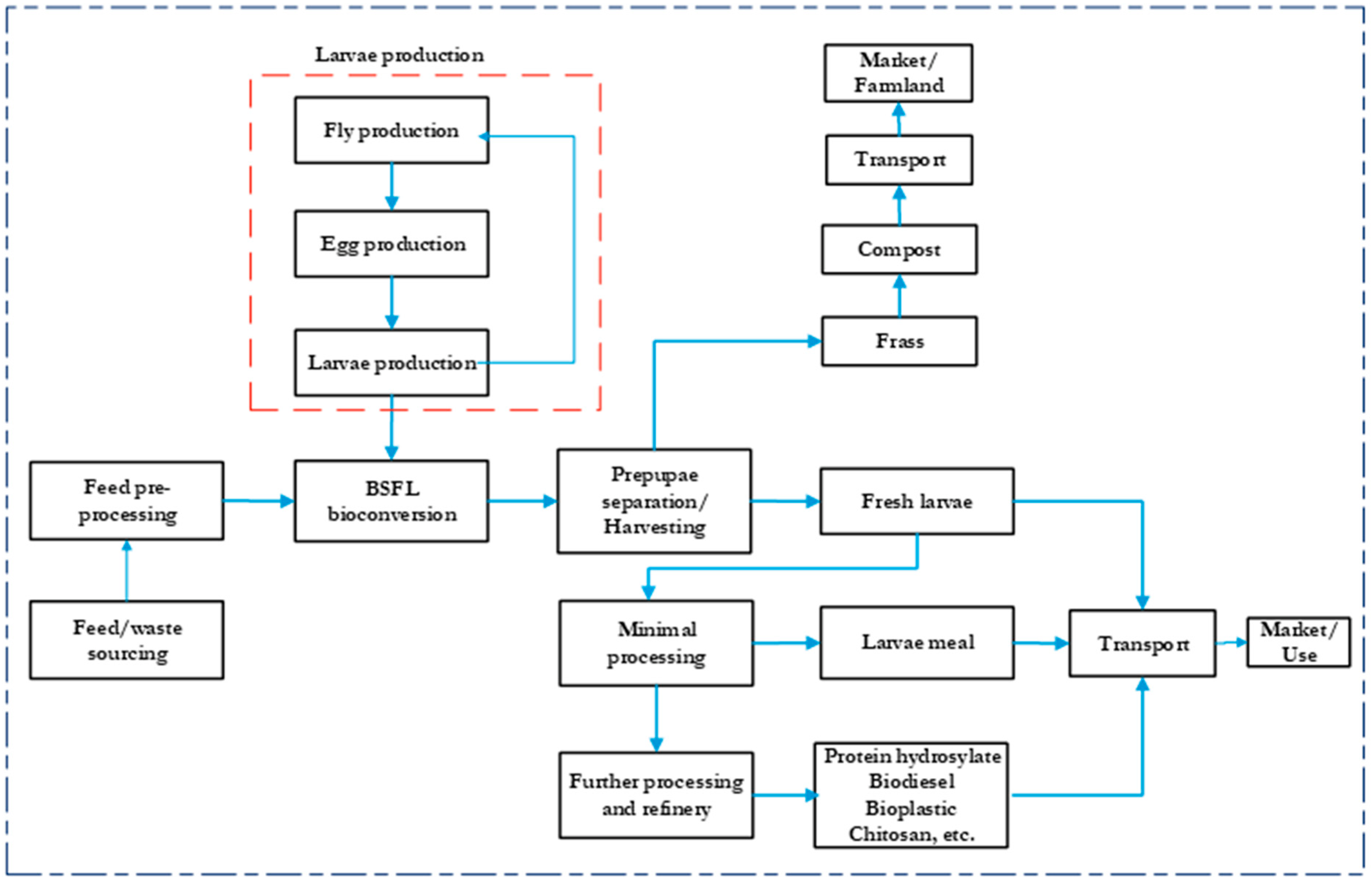
| Substrate | Larvae Age (Days) | Larvae Density (Larvae/cm2) | Feeding Rate (mg dm/Larva/Day) | CO2 (kg/kg dm BSFL) | CH4 (mg/kg dm BSFL) | N2O (mg/kg dm BSFL) | NH3 (g/kg dm BSFL) | References |
|---|---|---|---|---|---|---|---|---|
| Chicken feed | 7–10 | – | 12.61 | 2.2 ± 1.30 | Negligible | – | – | [5] |
| Food waste | 5 | 2 | 30.00 | 1.75 ± 0.17 | 49 ± 29 | 21 ± 13 | – | [88] d |
| Kitchen waste | 5 | 4 | 26.00 | – | 5.5 | 118 | – | [90] d |
| Pig manure + corn cob | 3 | 0.34 | 38.38 | 2.73 | 455.71 | 1.52 | 8.35 | [87] * |
| Food waste + rice straw | 3 | 0.64 | 23.30 | 1.39 ± 0.34 | 14 ± 6 | 7 ± 1 | – | [86] a,d,* |
| Pig manure + corn cob | 3 | 1.2 | 15.65 | 1.59 ± 0.13 | 5411.58 ± 3655 | 13.79 ± 3.46 | 7.32 ± 5.02 | [84] b,* |
| Meat and bone meal + rice bran | 3 | 1.13 | 26.67 | 3.33 ± 0.37 | 348.35 ± 390.08 | 6.52 ± 3.85 | 0.11 ± 0.02 | [85] * |
| Yeast concentrates from wheat + starch-rich byproduct | 7 | 6.6 | 16.57 | 2.75 ± 0.31 | 19 ± 10 c | 53 ± 27 | – | [92] d |
| Pig manure | 7 | 4.7 ± 0.5 | 22 ± 2 | 344 ± 43 | 10,066 ± 2652 | 6 ± 14 | – | [95] d |
| Food waste | 4 | 17.95 | 67.85 | 1.17 | 64.31 | 0.33 | 2.90 | [91] |
| Orange peels | – | 6.25 | 7.75 | 5.83–34.04 | 22.71–208.43 | 3.02–24.16 | 0.00–18.06 | [89] |
| Broccoli + cauliflower trimmings | – | 6.25 | 3.07 | 3.77–22.71 | 561–805.86 | 46.35–1904 | 4.33–125.27 | [89] |
| Food waste | – | 6.25 | 15.00 | 1.36–2.70 | 8.27–12.77 | 8.27–12.77 | 8.27–56.56 | [89] |
| Substrate Parameter | Carbon Dioxide (CO2) | Methane (CH4) | Nitrous Oxide (N2O) | Ammonia (NH3) | Reference |
|---|---|---|---|---|---|
| C/N ratio | Generally, substrates with a higher C/N ratio are rapidly degraded, increasing the respiration rate and consequent high CO2 loss during larvae growth. Thus, higher cumulative CO2 emissions were observed among treatments with higher initial C/N ratios, with 15 (108 g CO2 eq./kg substrate) being the lowest and 25 (152 g CO2 eq./kg substrate) being the highest. | C/N ratio inversely correlates with CH4 emissions due to reduced easily degradable C sources. | Generally, very low emissions were detected. However, a low C/N ratio resulted in higher NH3 emissions. BSFL activity could create a conducive aerobic environment that leads to the inhibition of denitrification and thus limits the N2O production. | A low C/N ratio resulted in higher NH3 emissions, suggesting that a high C/N ratio of substrate improves microbial assimilation and thus reduces NH3 emissions. On the other hand, a higher proportion of corncob as part of the BSFL feedstock improves the water-holding capacity, therefore partly reducing NH3 volatilization. | Pang et al. [84] |
| Moisture content | Total emissions correlated positively with increasing substrate moisture content and the overall BSFL biomass yield. | High substrate moisture content positively correlated with higher CH4 emissions due to low oxygen concentration and the formation of anaerobic zones, favouring the growth of methanogenic bacteria. | No clear relationship between moisture content and N2O emissions. | A negative correlation between moisture content and NH3 emissions was observed, mainly due to the solubility of NH3 in water, and the ability of high moisture content substrates to absorb large amounts of NH3. | Chen et al. [87] |
| pH | Emissions were lowest for extreme pH conditions (pH 3.0 and 11.0) and highest at optimum pH conditions of 5.0 and 7.0. | Decreasing the pH of the feeding material also corresponds to a decrease in CH4 production since acidic conditions inhibit methanogen activity. | Relatively higher emissions were associated with lower pH treatments, although no statistically significant differences were obtained. | Neutral to high pH (7.0–11.0) resulted in higher cumulative NH3 emissions. | Pang et al. [86] |
| Batch feeding time | Increasing the batch feeding times increased CO2 emissions and BSFL biomass. | The periodic addition of substrate led to the formation of an anoxic environment under the substrate surface, increasing CH4 emission. | No clear relationship between batch feeding times and N2O emissions, as no statistically significant difference was observed among the various treatments. | Increasing batch feeding times increased the cumulative NH3. | Zhang et al. [85] |
| Index | Author(s) | Year | Country | LCA Model | The Goal of the Study |
|---|---|---|---|---|---|
| 1 | Komakech et al. [165] | 2015 | Uganda | Attributional | Comparing the environmental impacts of different biowaste treatment technologies |
| 2 | Smetana et al. [82] | 2016 | Germany | Attributional | LCA of insect production and processing at an industrial scale |
| 3 | Rustad [166] | 2016 | Norway | Attributional | Comparing the environmental impacts of insect meal with fish meal |
| 4 | Salomone et al. [70] | 2017 | Italy | Attributional | LCA on the mass-rearing and food waste bioconversion by BSFL in a pilot plant |
| 5 | Mondello et al. [167] | 2017 | Italy | Attributional | Comparing the environmental impacts of different food waste treatment scenarios |
| 6 | Mertenat et al. [90] | 2019 | Switzerland | Attributional | LCA to compare the global warming potential (GWP) of BSF biowaste treatment and composting |
| 7 | Smetana et al. [83] | 2019 | Germany | Attributional | LCA on production stages of insect-based products and its comparison to benchmarks |
| Consequential | Identifying the environmental consequences of production and consumption choices toward insect-based feed and food | ||||
| 8 | Spinelli et al. [168] | 2019 | Italy | Attributional | LCA on laboratory-scale production of innovative bioplastics made from biopolymers derived from BSF proteins |
| 9 | Bosch et al. [169] | 2019 | Netherlands | Attributional | LCA on the production of fresh BSF larvae reared on different organic biomass resources |
| 10 | Rosa et al. [162] | 2020 | Italy | Attributional | LCA of BSF protein-derived bioplastic and comparison of two protein extraction protocols |
| 11 | Ites et al. [170] | 2020 | Germany | Attributional | Determining the environmental impact of using insects to treat food waste in a modular system |
| 12 | Guo et al. [91] | 2021 | China | Attributional | Revealing the environmental impact of a BSFL bioconversion plant |
| Index | Fs | Tr | Fp | Lp | Bb | Lsh | Mp | Fpr | Fm | Wm |
|---|---|---|---|---|---|---|---|---|---|---|
| 1 | – | – | x | – | x | x | – | – | x | – |
| 2 | x | – | x | x | x | x | x | x | x | x |
| 3 | x | x | – | – | x | x | x | – | x | – |
| 4 | – | x | x | x | x | x | x | – | x | x |
| 5 | x | x | x | – | x | x | x | – | – | – |
| 6 | – | – | x | x | x | x | x | – | x | – |
| 7 | x | x | x | x | x | x | x | x | x | x |
| 8 | x | x | x | x | x | x | x | x | x | x |
| 9 | x | – | x | x | x | x | – | – | – | – |
| 10 | x | x | x | x | x | x | x | x | x | x |
| 11 | x | – | x | x | x | x | x | – | x | x |
| 12 | – | – | – | x | x | x | x | – | x | – |
| Index | GW | E | LU | EU | A | OD | HT | ET | POF | IR | RD | WD | PMF |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | x | x | – | x | – | – | – | – | – | – | – | – | – |
| 2 | x | x | x | x | x | x | x | x | x | x | x | x | x |
| 3 | x | x | x | x | x | x | x | x | x | x | x | – | x |
| 4 | x | x | x | x | x | x | x | x | x | – | x | – | – |
| 5 | x | x | x | x | x | x | x | x | x | – | x | – | – |
| 7 | x | x | x | – | x | x | x | x | – | x | – | x | – |
| 8 | x | – | – | – | – | – | – | – | – | – | – | – | – |
| 9 | x | x | x | – | – | – | – | – | – | – | – | – | – |
| 10 | x | x | x | x | x | x | x | x | x | x | – | – | – |
| 11 | x | x | x | – | x | x | – | – | – | – | – | – | – |
| 12 | x | – | – | x | x | – | – | – | – | – | – | – | x |
| Index | GWP, kg CO2 eq. | Functional Unit |
|---|---|---|
| 2 | 1.36–15.1 | 1 kg of insect protein meal |
| 3 | 0.17 | 1 kg of insect meal (wet weight) |
| 4 | 2.1 | 1 kg of insect protein (dried larvae) |
| 2.9 | 1 kg of insect lipid (dried larvae) | |
| 7 | 5.3 | 1 kg of insect meal (defatted protein concentrate) |
| 1.16 | 1 kg of insect puree (fresh insect production) | |
| 8 | 1100 | 1 kg of bioplastic obtained from BSFL protein |
| 9 | 1 | 1 kg of fresh larvae |
| 19 | 1 kg of protein (food) a | |
| 3 | 1 kg of protein (feed) a | |
| 6 | 1 kg of protein (residue) a | |
| 10 | 698 | 1 kg of extracted protein (chemical extraction) |
| 1884 | 1 kg of extracted protein (enzyme-assisted extraction) |
| Index | MJ | m2 Arable | FU |
|---|---|---|---|
| 1 | 15.12 | – | 1 ton of food waste |
| 2 | 21.20–99.60 | 0.03–7.03 | 1 kg of insect protein meal |
| 4 | 215.30 | 0.67 | 1 ton of food waste treated |
| 15.10 | 0.47 | 1 kg of protein (dried larvae) | |
| 20.80 | 1.38 | 1 kg of lipid (dried larvae) | |
| 5 | 772.62 | 0.89 | 1 ton of food waste to be treated |
| 7 | 13.00 | 0.47 | 1 kg of insect fertilizer (dried and pelletized) |
| 61.29 | 1.38 | 1 kg of lipid | |
| 17.90 | 0.48 | 1 kg of puree (fresh insect production) | |
| 9 | 8.00 | 2.00 | 1 kg of fresh larvae |
| 174.00* | 67.00 | 1 kg of protein (food) a | |
| 84.00* | 3.00 | 1 kg of protein (feed) a | |
| 26.00* | 0.00 | 1 kg of protein (residue) a | |
| 10 | 15.06 | 0.12 | 1 g of extracted protein (chemical extraction) |
| 40.26 | 0.33 | 1 g of extracted protein (enzyme-assisted extraction) | |
| 11 | −3067.66 * | −477.20 | 1 ton of brewery grains treated |
| 2846.94 * | −1.16 | 1 ton of potato peels treated | |
| −23.49 * | −0.61 | 1 ton of expired food treated |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Boakye-Yiadom, K.A.; Ilari, A.; Duca, D. Greenhouse Gas Emissions and Life Cycle Assessment on the Black Soldier Fly (Hermetia illucens L.). Sustainability 2022, 14, 10456. https://doi.org/10.3390/su141610456
Boakye-Yiadom KA, Ilari A, Duca D. Greenhouse Gas Emissions and Life Cycle Assessment on the Black Soldier Fly (Hermetia illucens L.). Sustainability. 2022; 14(16):10456. https://doi.org/10.3390/su141610456
Chicago/Turabian StyleBoakye-Yiadom, Kofi Armah, Alessio Ilari, and Daniele Duca. 2022. "Greenhouse Gas Emissions and Life Cycle Assessment on the Black Soldier Fly (Hermetia illucens L.)" Sustainability 14, no. 16: 10456. https://doi.org/10.3390/su141610456
APA StyleBoakye-Yiadom, K. A., Ilari, A., & Duca, D. (2022). Greenhouse Gas Emissions and Life Cycle Assessment on the Black Soldier Fly (Hermetia illucens L.). Sustainability, 14(16), 10456. https://doi.org/10.3390/su141610456








