A Review of On-Site Carwash Wastewater Treatment
Abstract
1. Introduction
2. Car Wash Wastewater Quality
3. Water Quantities Required to Wash a Car
4. Car Wash Wastewater Treatment Technique
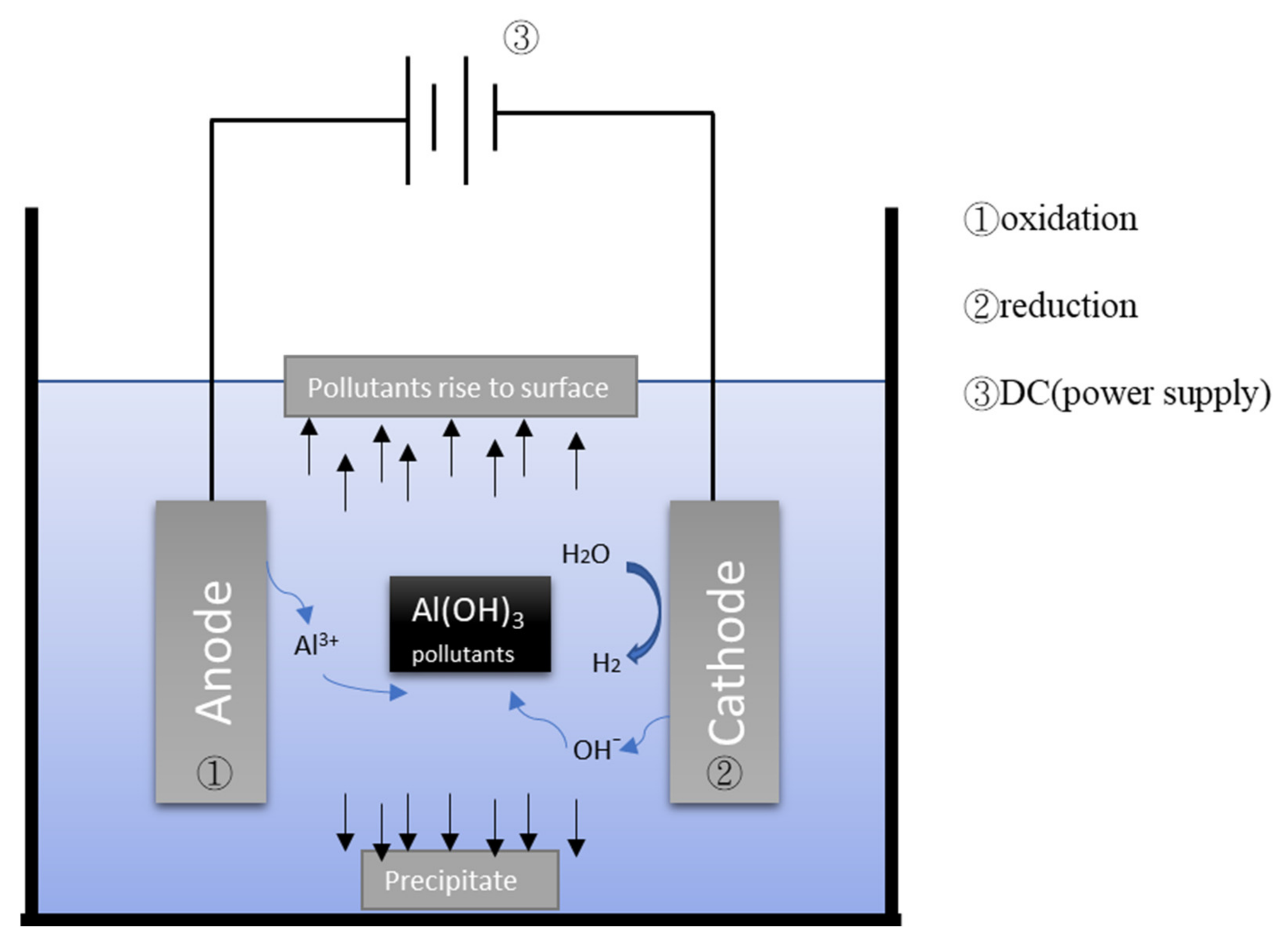
4.1. Electrocoagulation (EC)
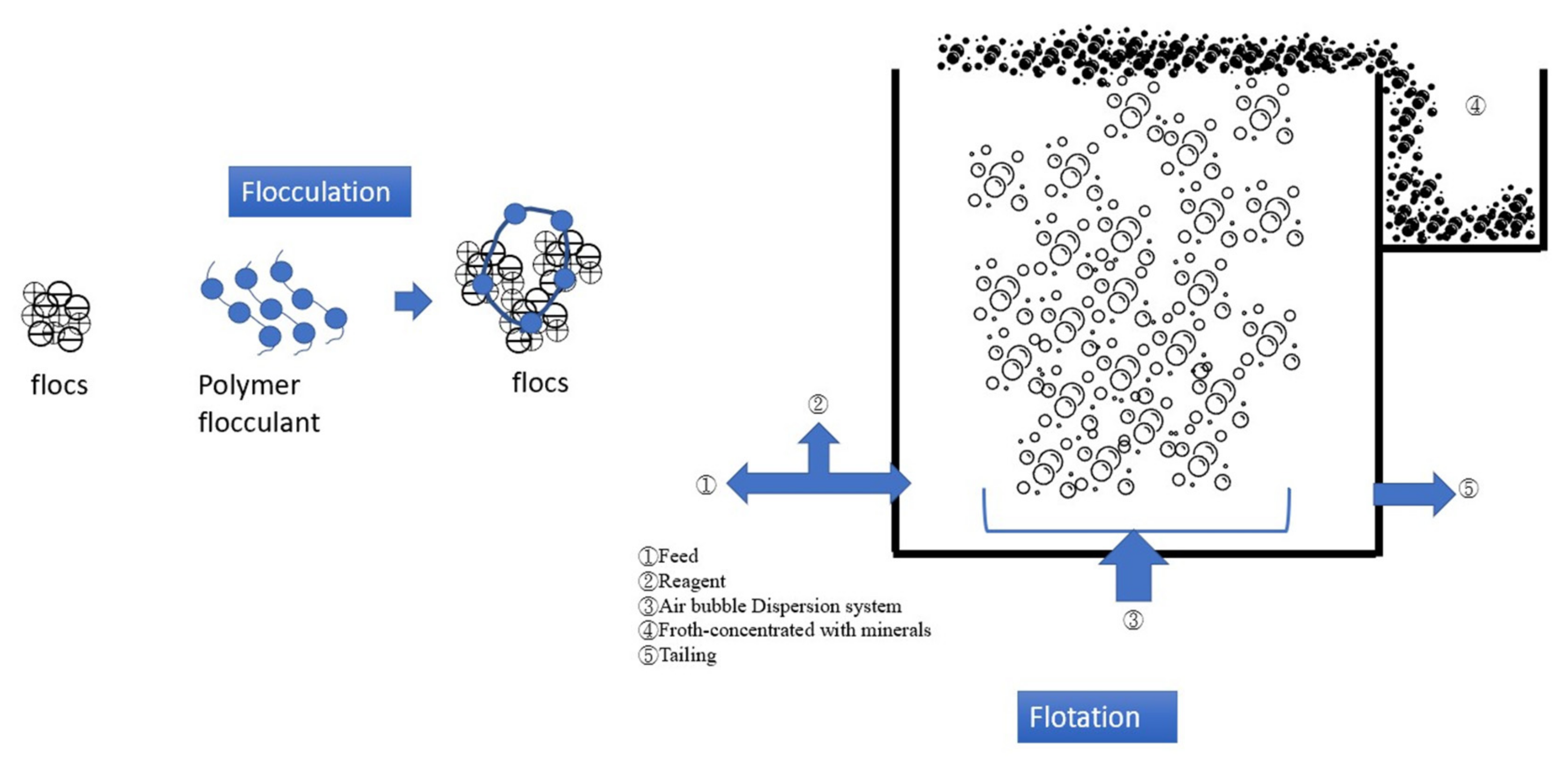
4.2. Flocculation–Flotation (FF)
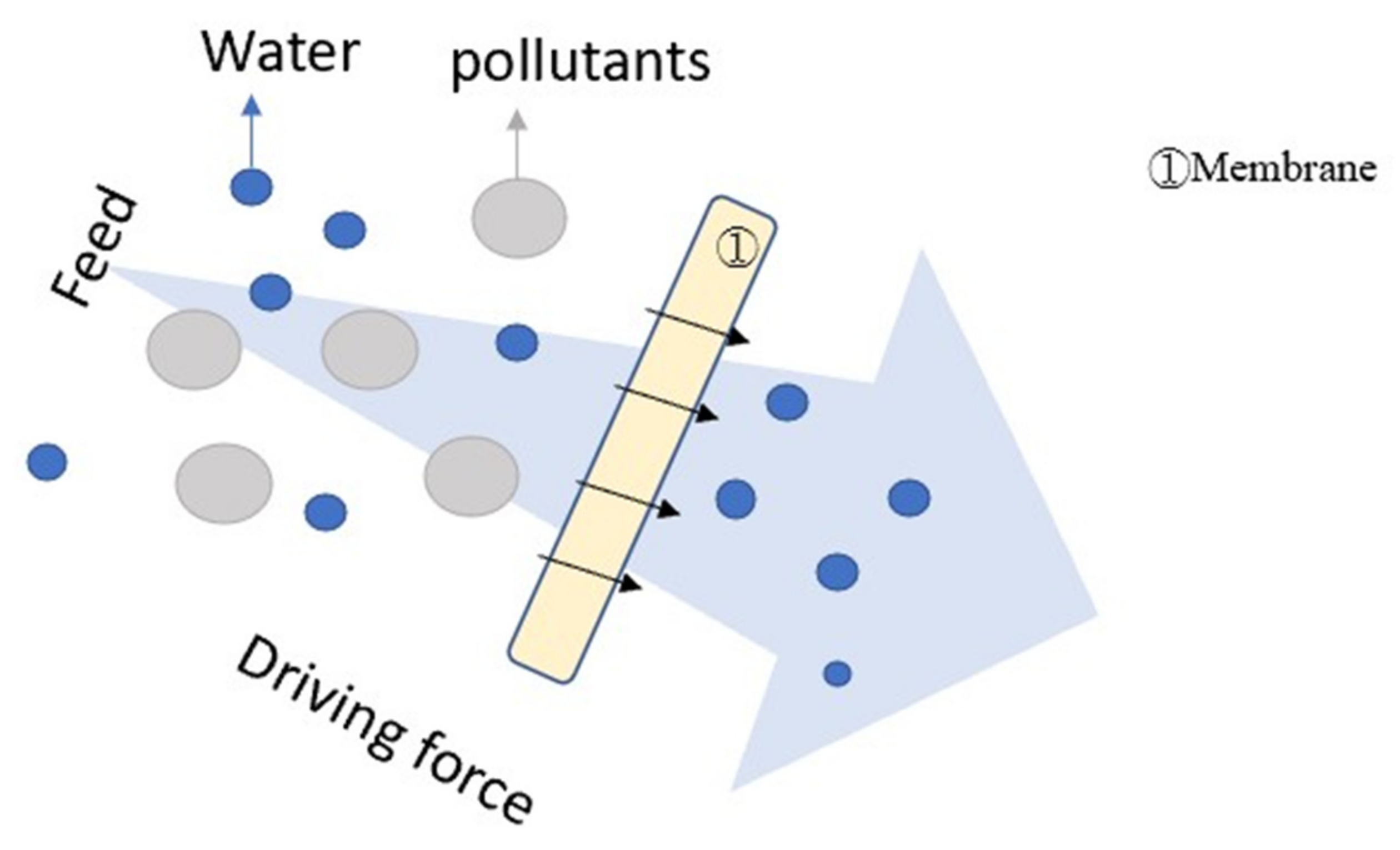
4.3. Filtration (F)
4.4. Coagulation–Flocculation (CF)
4.5. Bio-Treatment
4.6. Other Methods
5. Energy Consumption
6. Operating Cost
7. Discharge Standards
8. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| AD | adsorption |
| AS | anionic surfactant |
| Bio | biological treatment |
| COD | chemical oxygen demand |
| C | chemical coagulation |
| CF | coagulation-flocculation |
| DAF | dissolved air flotation |
| EC | electrocoagulation |
| EO | electro-oxidation |
| F | filtration |
| FF | flocculation flotation |
| M | membrane filtration |
| MBR | membrane bio-reactor |
| MF | microfiltration |
| NF | nanofiltration |
| NJDEP | New Jersey Department of Environmental Protection |
| NTU | nephelometric turbidity units |
| O&G | oil and grease |
| O | ozonation |
| RBC | rotating biological contactor |
| RO | reverse osmosis |
| SC | sand filtration and chlorination |
| SED | sedimentation |
| SF | sand filtration |
| SS | suspended solids |
| UF | ultrafiltration |
References
- Available online: https://www.statista.com/statistics/200002/international-car-sales-since-1990/ (accessed on 18 April 2022).
- Bakacs, M.E.; Yergeau, S.E.; Obropt, C.C.; ASCE, P.E.M. Assessment of car wash runoff treatment using bioretention mesocosms. J. Environ. Eng. 2013, 139, 1132–1136. [Google Scholar] [CrossRef]
- Available online: https://www.slideshare.net/guest7527d21f/ss-2773696 (accessed on 18 April 2022).
- Available online: https://kknews.cc/zh-tw/agriculture/xmm99mg.html (accessed on 18 April 2022).
- Torkashvand, J.; Farzadkia, M.; Younesi, S.; Gholami, M. A systematic review on membrane technology for carwash wastewater treatment: Efficiency and limitations. Desalin. Water Treat. 2021, 210, 81–90. [Google Scholar] [CrossRef]
- Talebzadeh, F.; Valeo, C.; Gupta, R.; Constabel, C.P. Exploring the Potential in LID Technologies for Remediating Heavy Metals in Carwash Wastewater. Sustainability 2021, 13, 8727. [Google Scholar] [CrossRef]
- Gomes, A.J.; Das, K.K.; Jame, S.A.; Cocke, D.L. Treatment of truck wash water using electrocoagulation. Desalin. Water Treat. 2016, 57, 25991–26002. [Google Scholar] [CrossRef]
- Rubi-Juarez, H.; Barrera-Diaz, C.; Uena-Nunez, F. Adsorption-assisted electrocoagulation of real car wash wastewater with equilibrium and kinetic studies. Pollut. Res. 2017, 36, 175–184. [Google Scholar]
- Rubi-Juarez, H.; Barrera-Diaz, C.; Linares-Hernandez, I.; Fall, C.; Bilyeu, B. A combined electrocoagulation-electrooxidation process for carwash wastewater reclamation. Int. J. Electrochem. Sci. 2015, 10, 6754–6767. [Google Scholar]
- Zaneti, R.; Etchepare, R.; Rubio, J. More environmentally friendly vehicle washes: Water reclamation. J. Clean. Prod. 2012, 37, 115–124. [Google Scholar] [CrossRef]
- Zaneti, R.; Etchepare, R.; Rubio, J. Car wash wastewater reclamation. Full-scale application and upcoming features. Resour. Conserv. Recycl. 2011, 55, 953–959. [Google Scholar] [CrossRef]
- Subtil, E.L.; Rodrigues, R.; Hespanhol, I.; Mierzwa, J.C. Water reuse potential at heavy-duty vehicles washing facilities—The mass balance approach for conservative contaminants. J. Clean. Prod. 2017, 166, 1226–1234. [Google Scholar] [CrossRef]
- Rubio, J.; Zaneti, R.N. Treatment of washrack wastewater with water recycling by advanced flocculation–column flotation. Desalin. Water Treat. 2009, 8, 146–153. [Google Scholar] [CrossRef]
- Zaneti, R.N.; Etchepare, R.; Rubio, J. Car wash wastewater treatment and water reuse—A case study. Water Sci. Technol. 2013, 67, 82–88. [Google Scholar] [CrossRef] [PubMed]
- Ganiyu, S.O.; dos Santos, E.V.; de Araújo Costa, E.C.T.; Martínez-Huitle, C.A. Electrochemical advanced oxidation processes (EAOPs) as alternative treatment techniques for carwash wastewater reclamation. Chemosphere 2018, 211, 998–1006. [Google Scholar] [CrossRef] [PubMed]
- Pinto, A.C.S.; de Barros Grossi, L.; de Melo, R.A.C.; de Assis, T.M.; Ribeiro, V.M.; Amaral, M.C.S.; de Souza Figueiredo, K.C. Carwash wastewater treatment by micro and ultrafiltration membranes: Effects of geometry, pore size, pressure difference and feed flow rate in transport properties. J. Water Process Eng. 2017, 17, 143–148. [Google Scholar] [CrossRef]
- Etchepare, R.; Zaneti, R.; Azevedo, A.; Rubio, J. Application of flocculation–flotation followed by ozonation in vehicle wash wastewater treatment/disinfection and water reclamation. Desalin. Water Treat. 2015, 56, 1728–1736. [Google Scholar] [CrossRef]
- Boussu, K.; Kindts, C.; Vandecasteele, C.; van der Bruggen, B. Applicability of nanofiltration in the carwash industry. Sep. Purif. Technol. 2007, 54, 139–146. [Google Scholar] [CrossRef]
- Paxéus, N. Vehicle washing as a source of organic pollutants in municipal wastewater. Water Sci. Technol. 1996, 33, 1–8. [Google Scholar] [CrossRef]
- Panizza, M.; Cerisola, G. Applicability of electrochemical methods to carwash wastewaters for reuse. Part 1: Anodic oxidation with diamond and lead dioxide anodes. J. Electroanal. Chem. 2010, 638, 28–32. [Google Scholar] [CrossRef]
- Panizza, M.; Cerisola, G. Applicability of electrochemical methods to carwash wastewaters for reuse. Part 2: Electrocoagulation and anodic oxidation integrated process. J. Electroanal. Chem. 2010, 638, 236–240. [Google Scholar] [CrossRef]
- Vaccari, M.; Gialdini, F.; Collivignarelli, C. Study of the reuse of treated wastewater on waste container washing vehicles. Waste Manag. 2013, 33, 262–267. [Google Scholar] [CrossRef]
- Sablayrolles, C.; Vialle, C.; Vignoles, C.; Montrejaud-Vignoles, M. Impact of carwash discharge on stormwater quality (Toulouse, France). Water Sci. Technol. 2010, 62, 2737–2746. [Google Scholar] [CrossRef][Green Version]
- Tony, M.A.; Bedri, Z. Experimental design of photo-Fenton reactions for the treatment of car wash wastewater effluents by response surface methodological analysis. Adv. Environ. Chem. 2014, 2014, 958134. [Google Scholar] [CrossRef]
- El-Ashtoukhy, E.S.Z.; Amin, N.K.; Fouad, Y.O. Treatment of real wastewater produced from Mobil car wash station using electrocoagulation technique. Environ. Monit. Assess. 2015, 187, 628–638. [Google Scholar] [CrossRef] [PubMed]
- Abdelmoez, W.; Barakat, N.A.M.; Moaz, A. Treatment of wastewater contaminated with detergents and mineral oils using effective and scalable technology. Water Sci. Technol. 2013, 68, 974–981. [Google Scholar] [CrossRef] [PubMed]
- Monney, I.; Donkor, E.A.; Buamah, R. Clean vehicles, polluted waters: Empirical estimates of water consumption and pollution loads of the carwash industry. Heliyon 2020, 6, e03952. [Google Scholar] [CrossRef]
- Tekere, M.; Sibanda, T.; Maphangwa, K.W. An assessment of the physicochemical properties and toxicity potential of carwash effluents from professional carwash outlets in Gauteng Province, South Africa. Environ. Sci. Pollut. Res. 2016, 22, 12816–12828. [Google Scholar] [CrossRef] [PubMed]
- Nguegang, B.; Sibanda, T.; Tekere, M. Cultivable bacterial diversity, physicochemical profiles, and toxicity determination of car wash effluents. Environ. Monit. Assess. 2019, 191, 478. [Google Scholar] [CrossRef]
- Baddor, I.M.; Farhoud, N.; Abdel-Magid, I.M.; Alshami, S.; Hassan Ahmad, F.; Olabi, E.A. Study of car wash wastewater treatment by adsorption. In Proceedings of the International Conference of Engineering, Information Technology, and Science, Kuala Lumpur, Malaysia, 1 May 2014; pp. 2–22. [Google Scholar]
- Uçar, D. Membrane processes for the reuse of car washing wastewater. J. Water Reuse Desalin. 2018, 8, 169–175. [Google Scholar] [CrossRef]
- Hashim, N.H.; Ibrahim, M.S.S.; Awang, Z. Adsorption of anionic surfactant presence in synthetic car wash wastewater by limestone. J. Appl. Chem. Nat. Resour. 2019, 1, 1–5. [Google Scholar]
- Kara, S. Treatment of transport container washing wastewater by electrocoagulation. Environ. Prog. Sustain. Energy 2013, 32, 249–256. [Google Scholar] [CrossRef]
- Gonder, Z.B.; Balcioglu, G.; Vergili, I.; Kaya, Y. An integrated electrocoagulation-nanofiltration process for carwash wastewater reuse. Chemosphere 2020, 253, 126713. [Google Scholar] [CrossRef]
- Bazrafshan, E.; Mostafapoor, F.K.; Soori, M.M.; Mahvi, A.H. Application of combined chemical coagulation and electrocoagulation process to carwash wastewater treatment. Fresenius Environ. Bull. 2012, 21, 2694–2701. [Google Scholar]
- Mirshahghassemi, S.; Aminzadeh, B.; Torabian, A.; Afshinnia, K. Optimizing electrocoagulation and electro-Fenton process for treating car wash wastewater. Environ. Health Eng. Manag. J. 2017, 4, 37–43. [Google Scholar] [CrossRef]
- Mohammadi, M.J.; Takdastan, A.; Jorfi, S.; Neisi, A.; Farhadi, M.; Yari, A.R.; Dobaradaran, S.; Khaniabadi, Y.O. Electrocoagulation process to Chemical and Biological Oxygen Demand treatment from carwash grey water in Ahvaz megacity, Iran. Data Brief 2017, 11, 634–639. [Google Scholar] [CrossRef] [PubMed]
- Mohammadi, M.J.; Salari, J.; Takdastan, A.; Farhadi, M.; Javanmardi, P.; Yari, A.R.; Dobaradaran, S.; Almasi, H.; Rahimi, S. Removal of turbidity and organic matter from car wash wastewater by electrocoagulation process. Desalin. Water Treat. 2017, 68, 122–128. [Google Scholar] [CrossRef]
- Do, K.U.; Kim, J.H.; Chu, X.Q. Sludge characteristics and performance of a membrane bioreactor for treating oily wastewater from a car wash service station. Desalin. Water Treat. 2018, 120, 166–172. [Google Scholar] [CrossRef]
- Lau, W.J.; Ismail, A.F.; Firdaus, S. Car wash industry in Malaysia: Treatment of car wash effluent using ultrafiltration and nanofiltration membranes. Sep. Purif. Technol. 2013, 104, 26–31. [Google Scholar] [CrossRef]
- Al-Gheethi, A.A.; Mohamed, R.M.S.R.; Rahman, M.A.A.; Johari, M.R.; Kassim, A.H.M. Treatment of wastewater from car washes using natural coagulation and filtration system. IOP Conf. Ser. Mater. Sci. Eng. 2016, 136, 012046. [Google Scholar] [CrossRef]
- Radin Mohamed, R.M.S.; Abdul Rahman, N.; Mohd Kassim, A.H. Moringa Oleifera and Strychnos Potatorum seeds as natural coagulant compared with synthetic common coagulants in treating car wash wastewater: Case study 1. Asian J. Appl. Sci. 2014, 2, 693–700. [Google Scholar]
- Mohamed, R.M.S.R.; Saphira, R.M.; Kutty, A.I.; Mariam, N.; Kassim, M.; Hashim, A. Efficiency of using commercial and natural coagulants in treating car wash wastewater treatment. Aust. J. Basic Appl. Sci. 2014, 8, 227–234. [Google Scholar]
- Zhang, J.K.; Yang, Y.B.; Wang, H.Y.; Dong, Z.B. CFU combined process for the treatment of oily car washing wastewater. Appl. Mech. Mater. 2013, 253–255, 999–1004. [Google Scholar] [CrossRef]
- Tan, X.; Tang, L. Application of enhanced coagulation aided by UF membrane for car wash wastewater treatment. In Proceedings of the 2008 2nd International Conference on Bioinformatics and Biomedical Engineering, Shanghai, China, 16–18 May 2008; pp. 3653–3656. [Google Scholar] [CrossRef]
- Chu, J.Y.; Li, Y.R.; Li, N.; Huang, W.H. Treatment of Car-washing Wastewater by Electrocoagulation-Ultrasound Technique for Reuse. Adv. Mater. Res. 2012, 433–440, 227–232. [Google Scholar] [CrossRef]
- Moazzem, S.; Wills, J.; Fan, L.; Roddick, F.; Jegatheesan, V. Performance of ceramic ultrafiltration and reverse osmosis membranes in treating car wash wastewater for reuse. Environ. Sci. Pollut. Res. 2018, 25, 8654–8668. [Google Scholar] [CrossRef] [PubMed]
- Boluarte, I.A.R.; Andersen, M.; Pramanik, B.K.; Chang, C.Y.; Bagshaw, S.; Farago, L.; Jegatheesan, V.; Shu, L. Reuse of car wash wastewater by chemical coagulation and membrane bioreactor treatment processes. Int. Biodeterior. Biodegrad. 2016, 113, 44–48. [Google Scholar] [CrossRef]
- Moazzem, S.; Ravishankar, H.; Fan, L.; Roddick, F.; Jegatheesan, V. Application of enhanced membrane bioreactor (eMBR) for the reuse of carwash wastewater. J. Environ. Manag. 2020, 254, 109780. [Google Scholar] [CrossRef]
- Bhatti, Z.A.; Mahmood, Q.; Raja, I.A.; Malik, A.H.; Khan, M.S.; Wu, D. Chemical oxidation of carwash industry wastewater as an effort to decrease water pollution. Phys. Chem. Earth Parts A/B/C 2011, 36, 465–469. [Google Scholar] [CrossRef]
- Syed, N.H.; Ahmad, J.; Khan, N.A.; Khan, N.; Shafiq, M.A. A low-cost wastewater treatment unit for reducing the usage of fresh water at car wash stations in Pakistan. Pak. J. Sci. Ind. Res. A Phys. Sci. 2019, 62A, 57–66. [Google Scholar] [CrossRef]
- Bhatti, S.; Siddiqui, Z.; Memon, S.; Kandhir, I.; Memon, M.A.; Mahesar, A.W. Analysis and treatment wash off water from vehicular service station in Hyderabad. Sindh Univ. Res. J. SURJ (Sci. Ser.) 2017, 49, 473–478. [Google Scholar] [CrossRef]
- Hsu, S.K.; Chen, C.H.; Chang, W.K. Reclamation of car washing wastewater by a hybrid system combining bio-carriers and non-woven membranes filtration. Desalin. Water Treat. 2011, 34, 349–353. [Google Scholar] [CrossRef]
- Tu, W.K.; Chang, C.C.; Chang, C.Y.; Ji, D.R.; Tseng, J.Y.; Chiu, C.Y.; Chen, Y.H.; Chang, C.F.; Yu, Y.H. Treatment of car wash wastewater via novel technologies for recycling and reutilization. J. Environ. Eng. Manag. 2009, 19, 49–57. [Google Scholar]
- Istirokhatun, T.; Destianti, P.; Hargianintya, A.; Oktiawan, W.; Susanto, H. Treatment of car wash wastewater by UF membranes. In Proceedings of the International Conference of Chemical and Material Engineering, Kyoto, Japan, 29 December 2015; p. 060025. [Google Scholar]
- Asha, M.N.; Chandan, K.S.; Harish, H.P.; NikhileswarReddy, S.; Sharath, K.S.; Liza, G.M. Recycling of waste water collected from automobile service station. Procedia. Environ. Sci. 2016, 35, 289–297. [Google Scholar] [CrossRef]
- Alam, J.; Farooqi, I.H. Management of grey water of an automobile workshop—A case study. In Proceedings of the International Workshop on Civil Engineering and Architecture, Istanbul, Turkey, 8–9 August 2014; pp. 133–138. [Google Scholar]
- Kiran, S.A.; Arthanareeswaran, G.; Thuyavan, Y.L.; Ismail, A.F. Influence of bentonite in polymer membranes for effective treatment of car wash effluent to protect the ecosystem. Ecotoxicol. Environmen. Saf. 2015, 121, 186–192. [Google Scholar] [CrossRef] [PubMed]
- Al-Odwani, A.; Ahmed, M.; Bou-Hamad, S. Carwash water reclamation in Kuwait. Desalination 2007, 206, 17–28. [Google Scholar] [CrossRef]
- Torkashvand, J.; Pasalari, H.; Gholami, M.; Younesi, S.; Oskoei, V.; Farzadkia, M. On-site carwash wastewater treatment and reuse: A systematic review. Int. J. Environ. Anal. Chem. 2020, 100, 1–15. [Google Scholar] [CrossRef]
- Nadzirah, Z.; Nor Haslina, H.; Rafidah, H. Removal of important parameter from car wash wastewater—A review. Appl. Mech. Mater. 2015, 773–774, 1153–1157. [Google Scholar] [CrossRef]
- Genuino, H.C.; Opembe, N.N.; Njagi, E.C.; McClain, S.; Suib, S.L. A review of hydrofluoric acid and its use in the car wash industry. J. Ind. Eng. Chem. 2012, 18, 1529–1539. [Google Scholar] [CrossRef]
- Kumar, N.S.; Chauhan, M.S. Water Quality Management; Treatment of car washing unit wastewater—A review; Springer International Publishing: Singapore, 2018; pp. 247–255. [Google Scholar]
- Sarmadi, M.; Foroughi, M.; Saleh, H.N.; Sanaei, D.; Zarei, A.A.; Ghahrchi, M.; Bazrafshan, E. Efficient technologies for carwash wastewater treatment: A systematic review. Environ. Sci. Pollut. Res. 2020, 28, 34823–34839. [Google Scholar] [CrossRef]
- Hassani, A.; Malhotra, M.; Karim, A.V.; Krishnan, S.; Nidheesh, P.V. Recent progress on ultrasound-assisted electrochemical processes: A review on mechanism, reactor strategies, and applications for wastewater treatment. Environ. Res. 2022, 205, 112463. [Google Scholar] [CrossRef]
- Boussu, K.; van Baelen, G.; Colen, W.; Eelen, D.; Vanassche, S.; Vandecasteele, C.; van der Bruggen, B. Technical and economical evaluation of water recycling in the carwash industry with membrane processes. Water Sci. Technol. 2008, 57, 1131–1135. [Google Scholar] [CrossRef]
- Gonder, Z.B.; Balcioglu, G.; Vergili, I.; Kaya, Y. Electrochemical treatment of carwash wastewater using Fe and Al electrode: Techno-economic analysis and sludge characterization. J. Environ. Manag. 2017, 200, 380–390. [Google Scholar] [CrossRef]
- Gonder, Z.B.; Balcıoğlu, G.; Kaya, Y.; Vergili, I. Treatment of carwash wastewater by electrocoagulation using Ti electrode: Optimization of the operating parameters. Int. J. Environ. Sci. Technol. 2019, 16, 8041–8052. [Google Scholar] [CrossRef]
- Ghanbari, F.; Zirrahi, F.; Olfati, D.; Gohari, F.; Hassani, A. TiO2 nanoparticles removal by electrocoagulation using iron electrodes: Catalytic activity of electrochemical sludge for the degradation of emerging pollutant. J. Mol. Liq. 2020, 310, 113217. [Google Scholar] [CrossRef]
- Enoh, B.S.; Christopher, W. Adsorption of metal ions from carwash wastewater by phosphoric acid modified clay: Kinetics and thermodynamic studies. Chem. Mater. Res. 2015, 7, 278026770. [Google Scholar]
- Ghanbari, F.; Wang, Q.; Hassani, A.; Wacławek, S.; Rodríguez-Chueca, J.; Lin, K.Y.A. Electrochemical activation of peroxides for treatment of contaminated water with landfill leachate: Efficacy, toxicity and biodegradability evaluation. Chemosphere 2021, 279, 130610. [Google Scholar] [CrossRef]
- Ahmadt, A.; Zarei, M.; Hassani, A.; Ebratkhahan, M.; Olad, A. Facile synthesis of iron(II) doped carbonaceous aerogel as a three-dimensional cathode and its excellent performance in electro-Fenton degradation of ceftazidime from water solution. Sep. Purif. Technol. 2021, 278, 119559. [Google Scholar] [CrossRef]
- Zapién Serrano, L.Z.; Ortiz Lara, N.O.; Ríos Vera, R.R.; Cholico-González, D. Removal of Fe(III), Cd(II), and Zn(II) as Hydroxides by Precipitation–Flotation System. Sustainability 2021, 13, 11913. [Google Scholar] [CrossRef]
- Park, J.H.; Han, Y.S.; Ji, S.W. Investigation of Mineral-Processing Wastewater Recycling Processes: A Pilot Study. Sustainability 2018, 10, 3069. [Google Scholar] [CrossRef]
- Siddig, O.; Al-Afnan, S.; Elkatatny, S.; Bahgat, M. Novel Cake Washer for Removing Oil-Based Calcium Carbonate Filter Cake in Horizontal Wells. Sustainability 2020, 12, 3427. [Google Scholar] [CrossRef]
- Mohamed, A.; Basfar, S.; Elkatatny, S.; Al-Majed, A. Prevention of Barite Sag in Oil-Based Drilling Fluids Using a Mixture of Barite and Ilmenite as Weighting Material. Sustainability 2019, 11, 5617. [Google Scholar] [CrossRef]
- Jönsson, C.; Jönsson, A.S. The influence of degreasing agents used at car washes on the performance of ultrafiltration membranes. Desalination 1995, 100, 115–123. [Google Scholar] [CrossRef]
- Hamada, T.; Miyazaki, Y. Reuse of carwash water with a cellulose acetate ultrafiltration membrane aided by flocculation and activated carbon treatments. Desalination 2004, 169, 257–267. [Google Scholar] [CrossRef]
- Tang, L.; Tan, X.J.; Cui, F.Y.; Zhou, Q.; Yin, J. Reuse of carwash wastewater with hollow fiber membrane aided by enhanced coagulation and activated carbon treatments. Water Sci. Technol. 2007, 56, 111–118. [Google Scholar]
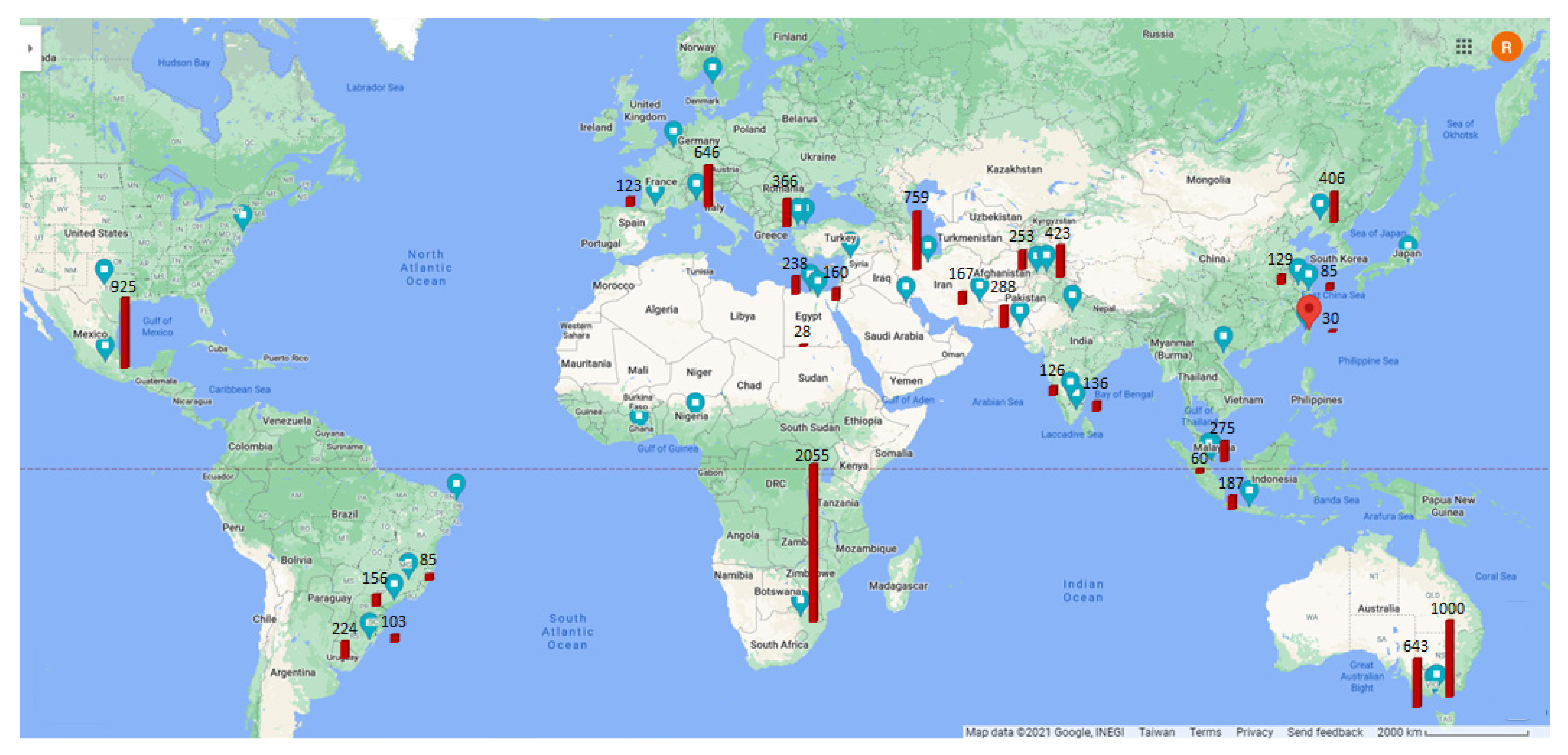


| Country | Area | Ref. | SS (mg/L) | Turbidity (NTU) | COD (mg/L) | O&G (mg/L) | AS (mg/L) |
|---|---|---|---|---|---|---|---|
| USA | New Jersey | [2] | 115 | — | — | — | 9.2 |
| USA | Texas | [7] | — | — | 260 | — | — |
| Mexico | Toluca | [8] | 538 | 925 | 1024 t, 541 s | 448 | — |
| Mexico | Toluca | [9] | — | 898 | 1295 t, 488 s | 369 | 68.3 |
| Brazil | Porto Alegre | [10] | — | 103 ± 57 | 191~600 | — | 6.3~21 |
| Brazil | Sao Paulo | [11] | 68 ± 19 | 89 ± 16.5 | 241 ± 23.5 | 6 ± 1 | 11.7 ± 9 |
| Brazil | Sao Paulo | [12] | — | 156 ± 45 | 626 ± 125 | — | — |
| Brazil | Porto Alegre | [13] | 112 ± 21 | 139 ± 45 | 259 ± 40 | 12 ± 6 | — |
| Brazil | Porto Alegre | [14] | 89 ± 54 | 103 ± 57 | 191 ± 22 | — | — |
| Brazil | Natal | [15] | — | — | 625 ± 5 | — | — |
| Brazil | Belo Horizonte | [16] | 260 ± 20 | 85 ± 8 | 85 ± 6 | <0.1 | — |
| Brazil | Porto Alegre | [17] | 85~279 | 194~254 | 249~873 | — | 11.3~22.3 |
| Belgium | Leuven | [18] | 60~140 | — | 208~382 | — | 0.7~2.5 |
| Sweden | Goteborg | [19] | — | — | 1263~4600 | 291~550 | — |
| Italy | Genoa | [20,21] | — | — | 572 | — | 95.5 |
| Italy | Brescia | [22] | — | 559~733 | 2640~4160 | — | — |
| France | Toulouse | [23] | 46~518 | 60~152 | 539~1506 | — | ~12 |
| Egypt | South of Egypt | [24] | 55 | 28.1 | 82 | — | — |
| Egypt | Shatby | [25] | — | 90.5~386 | 282~566 | — | — |
| Egypt | Elminia | [26] | — | 160 | 1430~1649 | — | — |
| Ghana | Kumasi | [27] | 2929 ± 451 | 3649 ± 2150 | 14133 ± 237 | — | — |
| South Africa | Gauteng | [28] | — | 109~4000 | — | 12~43 | 1.4~5.8 |
| South Africa | Johannesburg | [29] | — | — | 750~1864 | 5~24 | — |
| Syria | Aleppo | [30] | 49 | — | 350~510 | 20~40 | 21~35 |
| Turkey | Istanbul | [31] | — | — | 314 ± 9.4 | — | — |
| Turkey | Istanbul | [32] | 320 | — | 500 | 120 | 290 |
| Turkey | Tekirdag | [33] | — | 312~420 | 7960~8190 | — | — |
| Turkey | Istanbul | [34] | 2300 | — | 560 | 125 | 35 |
| Iran | Zahedan | [35] | 193 ± 71.5 | 166.8 ± 51.7 | 856 ± 217 | — | 31.2 ± 18.8 |
| Iran | Tehran | [36] | — | 118~1400 | 610~2619 | — | — |
| Iran | Tehran | [37] | — | 170 ± 32.5 | 480~1560 | — | — |
| Iran | Ahvaz | [38] | — | — | 480~1560 | — | — |
| Vietnam | Hanoi | [39] | 51~110 | — | 498~808 | 37~125 | — |
| Malaysia | Johor, Skudai | [40] | — | 34.7~86 | 75~738 | — | — |
| Malaysia | Parit Raja | [41] | — | 275.1 | 220 | — | — |
| Malaysia | Parit Raja | [42] | 186 ± 56.6 | 173.7 ± 58.8 | 741 ± 316 | 1.78 ± 0.1 | — |
| Malaysia | Taman University | [43] | 202 ± 10 | 216.3 ± 21.5 | 893 ± 298 | 0.004 | — |
| China | Shenyang, Hunan | [44] | — | 362~450 | — | 5.3~13.5 | — |
| China | Shanghai | [45] | — | 70~100 | 100~160 | 5~25 | 2~5 |
| China | Zhenjiang | [46] | — | 128.7 | 155.6 | — | — |
| Australia | Melbourne | [47] | 1275 | 522~763 | 295~471.5 | — | — |
| Australia | Geelong | [48] | 4200 | 1000 | 433 | — | — |
| Australia | Melbourne | [49] | 1200 | 763 | 417.5 | — | — |
| Pakistan | Abbottabad | [50] | 110~5856 | 73~772 | 141~1019 | 1.3~83.7 | — |
| Pakistan | Peshawar | [51] | 1000 | 253 | — | 27 | — |
| Pakistan | Hyderabad | [52] | — | 82.4~493 | — | — | — |
| Taiwan | Hsinchu | [53] | 230 | — | 67 | — | — |
| Taiwan | Taipei | [54] | 30~200 | 20~40 | 50~300 | — | 3~20 |
| Indonesia | Semarang | [55] | — | 186.6 | 700 | 36 | — |
| India | Bangalore | [56] | 970~1020 | 56.3~195 | 176.23~246 | 135~190 | — |
| India | Tasveer Mahal | [57] | 242.6 | — | 79 | — | — |
| India | Trichy | [58] | — | 132~140 | 150~175 | — | — |
| Country | Area | Ref. | Technique | SS (mg/L) | Turbidity (NTU) | COD (mg/L) | O&G (mg/L) | AS (mg/L) |
|---|---|---|---|---|---|---|---|---|
| Mexico | Toluca | [8] | EC + AD | — | 92~98% | 78~94% | — | — |
| Mexico | Toluca | [9] | EC + EO | — | 98~98.4% | 76~96% | 92~100% | 81~92% |
| Italy | Genoa | [21] | EC + EO | — | — | 75~97% | — | — |
| Iran | Tehran | [36] | EC | — | 85.5% | 80.8% | — | — |
| Iran | Tehran | [38] | EC | — | — | 88% | — | — |
| Iran | Ahvaz | [37] | EC | — | — | 90% | — | — |
| USA | Texas | [7] | EC | — | — | 79% | — | — |
| Egypt | Shatby | [25] | EC | — | ~87% | ~85% | — | — |
| Turkey | Istanbul | [34] | EC | — | — | 88% | 82% | 99% |
| Turkey | Tekirdag | [33] | EC | — | 99% | 76% | — | — |
| China | Zhenjiang | [46] | EC + Ultrasound | — | 96% | 69% | — | — |
| Country | Area | Ref. | Technique | SS (mg/L) | Turbidity (NTU) | COD (mg/L) | O&G (mg/L) | AS (mg/L) |
|---|---|---|---|---|---|---|---|---|
| Brazil | Porto Alegre | [17] | FF + O | 83–99% | 89–95% 93–98% | 39–85% 81–99% | — | 78–89% 81–99% |
| Brazil | Porto Alegre | [10] | FF | — | 91-96% | — | — | 40% |
| Brazil | Sao Paulo | [11] | FF + SF | — | 87–91% | — | — | — |
| Brazil | Porto Alegre | [13] | FF | 89% | 93% | 11% | — | — |
| Brazil | Porto Alegre | [14] | FF + SC | 91–93% | 91–96% | 63–76% | — | — |
| Pakistan | Hyderabad, Sindh | [52] | DAF + F | — | 97% | — | 99% | — |
| Country | Area | Ref. | Technique | SS (mg/L) | Turbidity (NTU) | COD (mg/L) | O&G (mg/L) | AS (mg/L) |
|---|---|---|---|---|---|---|---|---|
| Belgium | Leuven | [18] | UF + NF | — | — | 60~95% | — | 88~95% |
| Sweden | — | [77] | UF | — | — | 60% | — | — |
| Turkey | Istanbul | [34] | EC + NF | 99% | — | 88% | 90% | 91% |
| Malaysia | Johor, Skudai | [40] | UF + NF | — | — | 55~92% | — | — |
| Brazil | Belo Horizonte | [16] | MF + UF | — | 96.2~99.3% | 81~85% | — | — |
| Turkey | Istanbul | [31] | UF + NF | — | — | Negligible~97% | — | — |
| Indonesia | Semarang | [55] | UF | — | 100% | 91% | 83% | — |
| Japan | Tokyo | [78] | F + UF | — | 75% | 50~90% | — | — |
| China | Shanghai | [45] | C + UF | — | 85% | 80% | — | |
| Vietnam | Hanoi | [39] | MBR + F | — | — | 90% | 88% | — |
| Australia | Melbourne | [47] | UF + RO | 100% | 99.9% | 96% | — | — |
| Pakistan | Peshawar | [51] | SED + F | 80% | 99% | — | 49.2% | |
| India | Aligarh | [57] | SF | 89.2% | — | 83.5% | — | — |
| India | Trichy | [58] | UF | — | 82% | 47–60% | — | — |
| Country | Area | Ref. | Technique | SS (mg/L) | Turbidity (NTU) | COD (mg/L) | O&G (mg/L) | AS (mg/L) |
|---|---|---|---|---|---|---|---|---|
| Italy | Brescia | [22] | CF | — | 98% | 74% | — | — |
| Iran | Zahedan | [35] | C | 37% | — | 44% | — | 76% |
| Egypt | Elminia | [26] | CF + SF + O + SF | — | 100% | 88% | — | — |
| Malaysia | Parit Raja, Johor | [41] | CF | — | 97% | 35% | — | — |
| Malaysia | Parit Raja | [42] | C | — | 94% | 60% | — | — |
| Malaysia | Taman University | [43] | C | — | 90% | 60% | — | — |
| China | Shenyang | [44] | C + UF | — | 94% | — | >40% | — |
| China | Shanghai | [79] | C + M | — | 70% | — | — | — |
| India | Bangalore | [56] | CF + F | — | — | 80–90% | 92–93% | — |
| Pakistan | Abbottabad | [50] | C + H2O2 | — | 97% | 93% | 96% | — |
| Country | Area | Ref. | Technique | SS (mg/L) | Turbidity (NTU) | COD (mg/L) | O&G (mg/L) | AS (mg/L) |
|---|---|---|---|---|---|---|---|---|
| Brazil | Sao Paulo | [12] | RBC + F | — | 72~97% | 56~94% | — | — |
| USA | New Jersey | [2] | four bioretention mesocosms | 84~95% | — | — | — | 89~96% |
| Taiwan | Hsinchu | [53] | Bio + M | 95.7% | — | 70.2% | — | — |
| Australia | Geelong | [48] | C + MBR | 99.8% | 99.6% | — | — | — |
| Australia | Melbourne | [49] | enhanced MBR (eMBR) | — | 99.9% | 99.8% | 5.9~6.7 LMH | — |
| Country | Area | Ref. | Technique | SS (mg/L) | Turbidity (NTU) | COD (mg/L) | O&G (mg/L) | AS (mg/L) |
|---|---|---|---|---|---|---|---|---|
| Egypt | South of Egypt | [24] | Photo-Fenton’s process | — | — | 82~93.4% | — | — |
| Syria | Aleppo | [30] | AD | — | — | 81.6% | 86.8% | 88.3% |
| Brazil | Natal | [15] | EO | — | — | 96% | — | 83~96% |
| Technique | Operating (US $/m3) | Payback (Month) | Ref. |
|---|---|---|---|
| EC | 0.8 | - | [33] |
| EC with | - | - | [67] |
| Al electrode Fe electrode | 0.3 0.6 | ||
| EC with Titanium electrode | 9.7 | - | [68] |
| Bio + M | - | 7–15 | [51] |
| ECF | - | 15 | [53] |
| FF | 0.92 | - | [10] |
| Country | Criteria | Ref. | SS (mg/L) | Turbidity (NTU) | COD (mg/L) | O&G (mg/L) | AS (mg/L) |
|---|---|---|---|---|---|---|---|
| China | GB/T 18920-2002 | [79] | 5 | 5 | 50 | 1 | 0.5 |
| Iran | Iran Standard | [35] | 40 | 50 | 60 | — | 1.5 |
| Australia | Recycled water class A according to EPA | [47] | 5 | 2 | — | — | — |
| Belgium | — | [18] | <60 | — | <125 | — | 3 |
| France | — | [23] | 35 | — | 125 | — | — |
| Malaysia | Environmental quality Act 1974 | [41] | — | <5 | <50 | — | — |
| Syria | Syrian Standard No. 2752 | [30] | 50 | — | 75 | 5 | 5 |
| USA | NJDEP | [2] | 40 | — | — | — | — |
| Brazil | Local emission standards | [17] | 180 | — | 400 | — | 2 |
| Mexico | — | [8] | — | — | — | 15 | — |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kuan, W.-H.; Hu, C.-Y.; Ke, L.-W.; Wu, J.-M. A Review of On-Site Carwash Wastewater Treatment. Sustainability 2022, 14, 5764. https://doi.org/10.3390/su14105764
Kuan W-H, Hu C-Y, Ke L-W, Wu J-M. A Review of On-Site Carwash Wastewater Treatment. Sustainability. 2022; 14(10):5764. https://doi.org/10.3390/su14105764
Chicago/Turabian StyleKuan, Wen-Hui, Ching-Yao Hu, Li-Wei Ke, and Jung-Ming Wu. 2022. "A Review of On-Site Carwash Wastewater Treatment" Sustainability 14, no. 10: 5764. https://doi.org/10.3390/su14105764
APA StyleKuan, W.-H., Hu, C.-Y., Ke, L.-W., & Wu, J.-M. (2022). A Review of On-Site Carwash Wastewater Treatment. Sustainability, 14(10), 5764. https://doi.org/10.3390/su14105764







