Extraction of Essential Oil from River Tea Tree (Melaleuca bracteata F. Muell.): Antioxidant and Antimicrobial Properties
Abstract
1. Introduction
2. Materials and Methods
2.1. Plant Material
2.2. Extraction Method
2.3. Analysis of Tea Tree oil by GCMS
2.4. Antioxidant Analysis
2.4.1. DPPH Radical Scavenging Activity
2.4.2. Total Flavonoid Contents
2.4.3. Total Phenolic Compounds
2.4.4. Reducing Power Ability (RPA) of the Plant
2.5. Antimicrobial Analysis
2.6. Statistical Analysis
3. Results and Discussion
3.1. Essential Oil Yield
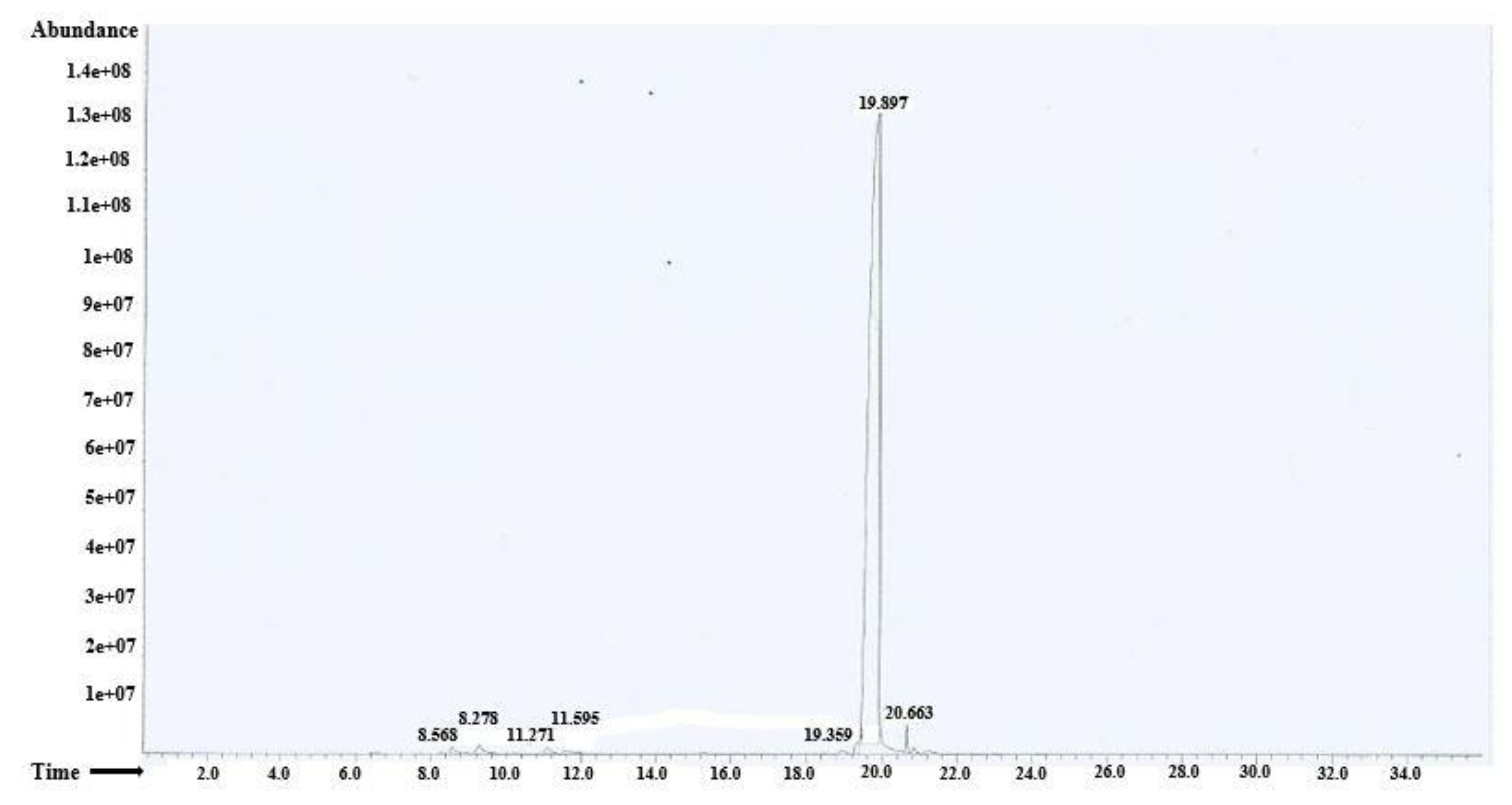
3.2. GCMS Analysis
3.3. Biochemical Assays
3.3.1. DPPH Scavenging Activity
3.3.2. Total Flavonoid Contents
3.3.3. Total Phenolic Compounds of Tea Tree Oil
3.3.4. Reducing Power Ability
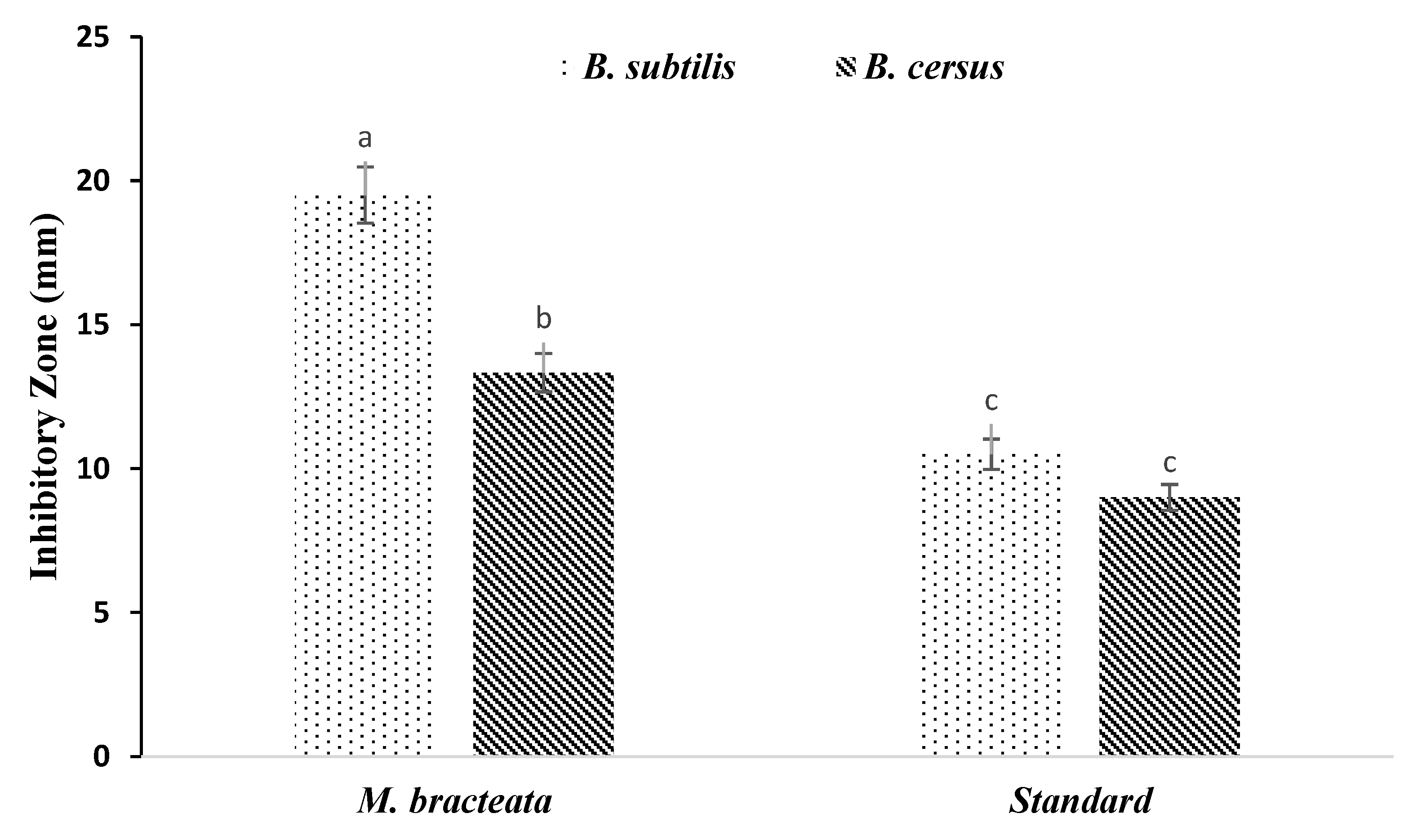
3.4. Antimicrobial Properties
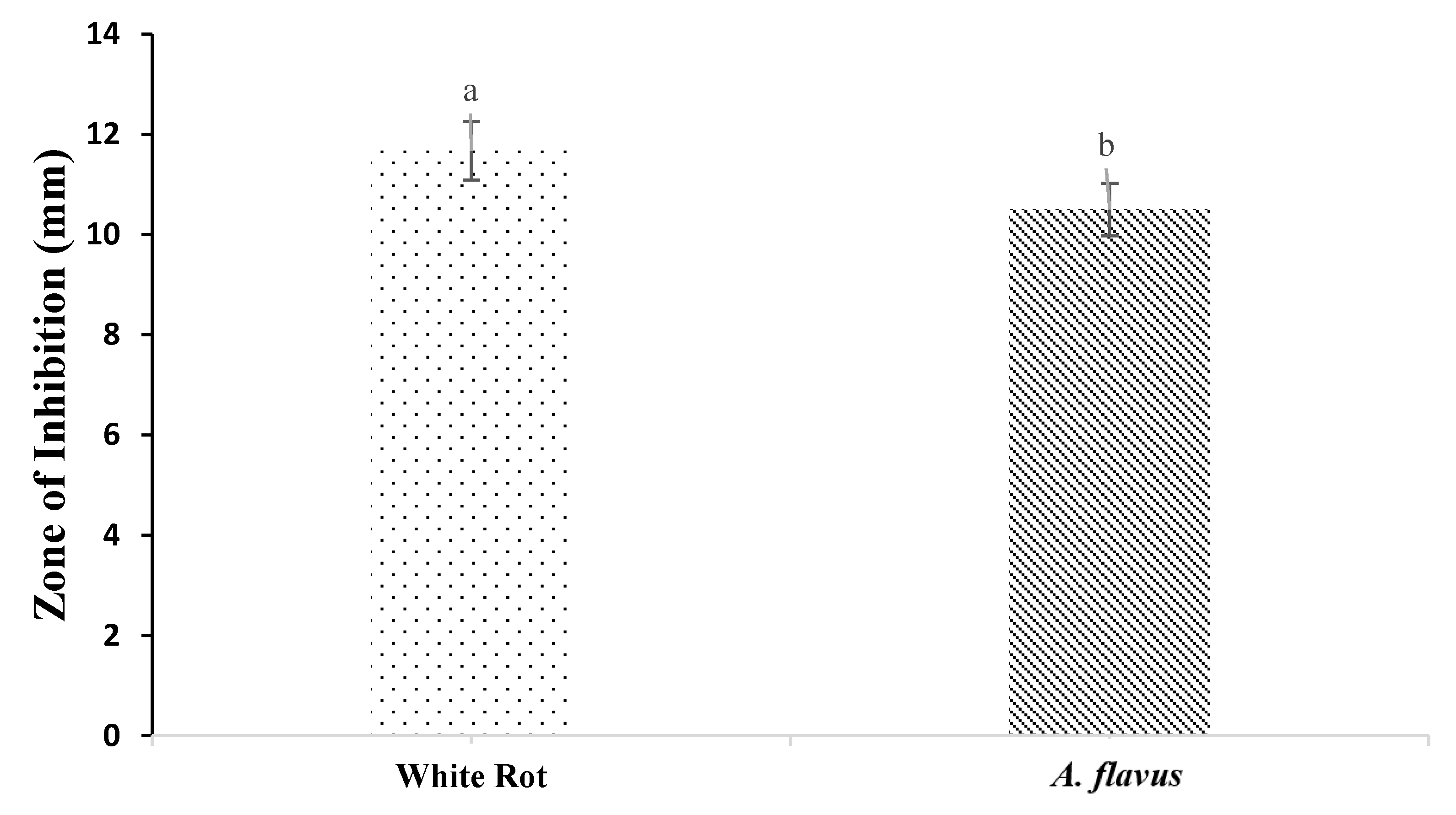
3.5. Antifungal Properties
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Bakkali, F.; Averbeck, S.; Averbeck, D.; Idaomar, M. Biological effects of essential oils—A review. Food Chem. Toxicol. 2008, 46, 446–475. [Google Scholar] [CrossRef] [PubMed]
- Andrade, L.; de Sousa, D. A review on anti-inflammatory activity of monoterpenes. Molecules 2013, 18, 1227–1254. [Google Scholar]
- Andrade, B.F.M.T.; Barbosa, L.N.; Probst, I.D.S.; Júnior, A.F. Antimicrobial activity of essential oils. J. Essent. Oil Res. 2014, 26, 34–40. [Google Scholar] [CrossRef]
- De Almeida, R.N.; Motta, S.C.; Faturi, C.D.B.; Catallani, B.; Leite, J.R. Anxiolytic-like effects of rose oil inhalation on the elevated plus-maze test in rats. Pharmacol. Biochem. Behav. 2004, 77, 361–364. [Google Scholar] [CrossRef]
- Solgi, M.; Kafi, M.; Taghavi, T.T.; Naderi, R. Effects of Silver Nanoparticles and Essential Oils of Thyme (Thymus vulgaris) and Zattar (Zataria multiflora Boiss.) on Postharvest Qualitative Aspects of Gerbera Cut Flowers (Gerbera jamesonii L.). Ph.D Thesis, Tehran University, Tehran, Iran, 2009. [Google Scholar]
- Padalia, R.C.; Verma, R.S.; Chauhan, A.; Goswami, P.; Verma, S.K.; Darokar, M.P. Essential oil of Melaleuca linariifolia Sm. from India: A potential source of 1,8-cineole. Ind. Crops Prod. 2015, 63, 264–268. [Google Scholar] [CrossRef]
- Lis-Balchin, M.; Deans, S.G. Bioactivity of selected plant essential oils against Listeria monocytogenes. J. Appl. Microbiol. 1997, 82, 759–762. [Google Scholar] [CrossRef]
- Larson, D.; Jacob, S.E. Tea tree oil. Dermatitis 2012, 23, 48–49. [Google Scholar] [CrossRef]
- Hammer, K.A. Treatment of acne with tea tree oil (Melaleuca) products: A review of efficacy, tolerability and potential modes of action. Int. J. Antimicrob. Agents 2015, 45, 106–110. [Google Scholar] [CrossRef]
- Nogueira, M.N.M.; Aquino, S.G.; Rossa Junior, C.; Spolidorio, D.M.P. Terpinen-4-ol and alpha-terpineol (tea tree oil components) inhibit the production of IL-1b, IL-6 and IL-10 on human macrophages. Inflamm. Res. 2014, 63, 769–778. [Google Scholar] [CrossRef]
- Parker, R.N.A. Forest Flora for the Punjab with Hazara and Delhi, Superintendent; Government of Printing Press: Panaji, India, 1956; pp. 245–246.
- Yatagai, M. Allelopathy, control of plant ecosystem by plant chemical components: Chemical characteristics and activity of plant odor components. Aroma. Res. 2007, 8, 408–413. [Google Scholar]
- Adesanwo, J.K.; Shode, F.O.; Aiyelaagbe, O.O.; Rabiu, O.O.; Oyede, R.T.; Oluwole, F.S. Antisecretory and antiulcerogenic activities of the stem bark extract of Melaleuca bracteata and isolation of principles. J. Med. Plants Res. 2009, 3, 822–824. [Google Scholar]
- Kardinan, A.K.; Hidayat, P. Potency of Melaleuca bracteata and Ocimum sp. leaf extracts as fruit fly (Bactrocera dorsalis complex) attractants in guava and star fruit orchards in Bogor, West Java, Indonesia. J. Sustain. Agric. 2013, 8, 79–84. [Google Scholar]
- Bistgani, Z.E.; Siadat, S.A.; Bakhshandeh, A.; Pirbalouti, A.G.; Hashemi, M.; Maggi, F.; Morshedloo, M.R. Application of combined fertilizers improves biomass, essential oil yield, aroma profile, and antioxidant properties of Thymus daenensis Celak. Ind. Crops Prod. 2018, 121, 434–440. [Google Scholar] [CrossRef]
- Berka-Zougali, B.; Ferhat, M.A.; Hassani, A.; Chemat, F.; Allaf, K.S. Comparative study of essential oils extracted from Algerian Myrtus communis L. leaves using microwaves and hydrodistillation. Int. J. Mol. Sci. 2012, 13, 4673–4695. [Google Scholar] [CrossRef]
- Mensor, L.I.; Menezes, F.S.; Leitao, G.G.; Reis, A.S.; Santos, T.; Coube, C.S.; Leitao, S.G. Screening of Brazilian plant extracts for antioxidant activity by the use of DPPH Free Radical method. Phytother. Res. 2001, 15, 127–130. [Google Scholar] [CrossRef] [PubMed]
- Zhishen, J.; Mengcheng, T.; Jianming, W. The determination of flavonoid contents in mulberry and their scavenging effects on superoxide radicals. Food Chem. 1999, 64, 555–559. [Google Scholar] [CrossRef]
- Kujala, T.S.; Loponen, J.M.; Klika, K.D.; Pihlaja, K. Phenolics and betacyanins in red beetroot (Beta vulgaris) root: Distribution and effect of cold storage on the content of total phenolics and three individual Compounds. J. Agric. Food Chem. 2000, 48, 5338–5342. [Google Scholar] [CrossRef] [PubMed]
- Habila, J.D.; Bello, I.A.; Dzikwi, A.A.; Musa, H.; Abubakar, N. Total phenolics and antioxidant activity of Tridax procumbens Linn. Afr. J. Pharm. Pharmacol. 2010, 4, 123–126. [Google Scholar]
- Al-Abd, N.M.; Nor, Z.M.; Mansor, M.; Azhar, F.; Hasan, M.S.; Kassim, M. Antioxidant, antibacterial activity, and phytochemical characterization of Melaleuca cajuputi extract. BMC Complement. Alternat. Med. 2015, 15, 385. [Google Scholar] [CrossRef]
- Kamal, S.; Kiran, S.; Aslam, N.; Bibi, I.; Rehman, J.; Manzoor, M.; Kamal, A. Mineral profile, antioxidant and antimicrobial activities of gemm Foeniculum vulgare. Curr. Biot. 2014, 8, 111–124. [Google Scholar]
- Steel, R.G.; Torrie, J.H.; Dickey, D.A. Principles and Procedures of Statistics: A Biometrical Approach; McGraw-Hill Kogakusha Ltd.: Boston, MA, USA, 1997; Volume 2. [Google Scholar]
- Siddique, S.; Parveen, Z.; Firdaus-e-Bareen; Mazhar, S. Chemical composition, antibacterial and antioxidant activities of essential oils from leaves of three Melaleuca species of Pakistani flora. Arab. J. Chem. 2020, 13, 67–74. [Google Scholar] [CrossRef]
- Li, Y.; Ye, Z.; Wang, W.; Yang, C.; Liu, J.; Zhou, L.; Shen, Y.; Wang, Z.; Chen, J.; Wu, S.; et al. Composition analysis of essential oil from Melaleuca bracteata leaves using ultrasound-assisted extraction and its antioxidative and antimicrobial activities. BioResources 2018, 13, 8488–8504. [Google Scholar] [CrossRef]
- Zhong, C.; Huang, Z.; Ling, Z.; Cheng, H. Study on chemical components of essential oil from the branches and leaves of Melaleuca bracteata. Flav. Frag. Cos. 2009, 6, 4. [Google Scholar]
- Tan, K.H.; Nishida, R. Methyl eugenol: Its occurrence, distribution, and role in nature, especially in relation to insect behavior and pollination. J. Insect. Sci. 2012, 12, 1. [Google Scholar] [CrossRef]
- Kivanc, M. Antimicrobial activity of “Cortuk” (Echinophora sibthorpiana Guss.) spice, its essential oil and methyl eugenol. Nahrung 1998, 332, 635–637. [Google Scholar] [CrossRef] [PubMed]
- Kivanc, M.; Akgul, A. Mould growth on black table olives and prevention by sorbic acid, methyl-eugenol and spice essential oil. Nahrung 1990, 34, 369–373. [Google Scholar] [CrossRef]
- Sudhakar, P.; Latha, P.; Sreenivasulu, Y.; Reddy, B.V.; Hemalaatha, T.M.; Balakrishna, M.; Reddy, K.R. Inhibition of Aspergillus flavus colonization and aflatoxin (AfB1) in peanut by methyl eugenol. Indian J. Exp. Biol. 2009, 47, 63–67. [Google Scholar]
- Ngoh, S.P.; Choo, L.E.W.; Pang, F.Y.; Huang, Y.; Kini, M.R.; Ho, S.H. Insecticidal and repellent properties of nine volatile constituents of essential oils against the American cockroach, Periplaneta americana (L.). Pestic. Sci. 1998, 54, 261–268. [Google Scholar] [CrossRef]
- Rossi, P.; Bao, L.; Luciani, A.; Panighi, J.; Desjobert, J.; Costa, J.; Casanova, J.; Bolla, J.; Berti, L. Methyl isoeugenol and elemicin: Antibaterial components of Daucus carota L. essential oil against Campylobacter jujuni. J. Agric. Food Chem. 2007, 55, 7332–7336. [Google Scholar] [CrossRef] [PubMed]
- Close, D.C.; McArthur, C. Rethinking the role of many plant phenolics-protection from photodamage not herbivores? Oikos 2002, 9, 166–172. [Google Scholar] [CrossRef]
- Treutter, D. Significance of flavonoids in plant resistance and enhancement of their biosynthesis. Plant Biol. 2005, 7, 581–591. [Google Scholar] [CrossRef]
- Sousa, A.; Ferreira, I.C.F.R.; Barros, L.; Bento, A. Pereira Antioxidant potential of traditional stoned table olives “Alcaparras”: Influence of the solvent and temperature extraction conditions. LWT Food Sci. Technol. 2008, 41, 739–745. [Google Scholar] [CrossRef]
- Joshi, S.C.; Verma, A.R.; Mathela, C.S. Antioxidant and antibacterial activities of the leaf essential oils of Himalayan Lauraceae species. J. Chem. Toxicol. 2010, 48, 37–40. [Google Scholar] [CrossRef]
- Hou, W.; Zhang, W.; Chen, G.; Lou, Y. Optimization of extraction conditions for maximal phenolic, flavonoid and antioxidant activity from Melaleuca bracteata leaves using the response surface methodology. PLoS ONE 2016, 11, e0162139. [Google Scholar] [CrossRef]
- Noumi, E.; Snoussi, M.; Hajlaoui, H.; Trabelsi, N.; Ksouri, R.; Valentin, E.; Bakhrouf, A. Chemical composition, antioxidant and antifungal potential of Melaleuca alternifolia (tea tree) and Eucalyptus globulus essential oils against oral Candida species. J. Med. Plants Res. 2011, 5, 4147–4156. [Google Scholar]
- Zhang, X.; Guo, Y.; Guo, L.; Jiang, H.; Ji, Q. In Vitro evaluation of antioxidant and antimicrobial activities of Melaleuca alternifolia essential oil. BioMed Res. Int. 2018, 3, 1–8. [Google Scholar] [CrossRef]
- Amorati, R.; Foti, M.C.; Valgimigli, L. Antioxidant activity of essential oils. J. Agric. Food Chem. 2013, 61, 10835–10847. [Google Scholar] [CrossRef] [PubMed]
- Goswami, P.; Verma, S.K.; Chauhan, A.; Venkatesha, K.T.; Verma, R.S.; Singh, R.V.; Darokar, M.P.; Chanotiya, C.S.; Padalia, R.C. Chemical composition and antibacterial activity of Melaleuca bracteata essential oil from India: A natural source of methyl eugenol. Nat. Prod. Commun. 2017, 12, 1934578X1701200633. [Google Scholar] [CrossRef]
- Griffin, S.G.; Markham, J.L.; Leach, D.N. An agar dilution method for the determination of the minimum inhibitory concentration of essential oils. J. Essent. Oil Res. 2000, 12, 249–255. [Google Scholar] [CrossRef]
- Christopher, F.; Stahl-Biskup, E.; Kaulfers, P.M. Death kinetics of Staphylococcus aureus exposed to commercial tea tree oils. J. Essent. Oil. Res. 2001, 13, 98–102. [Google Scholar] [CrossRef]
- Hammer, K.A.; Carson, C.F.; Riley, T.V. In vitro susceptibilities of lactobacilli and organisms associated with bacterial vaginosis to Melaleuca alternifolia (tea tree) oil. Antimicrob. Agents Chemother. 1999, 43, 196. [Google Scholar] [CrossRef] [PubMed]
- Shapiro, S.; Meier, A.; Guggenheim, B. The antimicrobial activity of essential oils and essential oil components towards oral bacteria. Oral Microbiol. Immunol. 1994, 9, 202–208. [Google Scholar] [CrossRef]
- Banes-Marshall, L.; Cawley, P.; Phillips, C.A. In vitro activity of Melaleuca alternifolia (tea tree) oil against bacterial and Candida spp. isolates from clinical specimens. Br. J. Biomed. Sci. 2001, 58, 139–145. [Google Scholar]
- Southwell, I.A.; Hayes, A.J.; Markham, J.; Leach, D.N. The search for optimally bioactive Australian tea tree oil. Int. Symp. Med. Aromat. Plants 1993, 344, 256–265. [Google Scholar] [CrossRef]
- Carson, C.F.; Mee, B.J.; Riley, T.V. Mechanism of action of Melaleuca alternifolia (tea tree) oil on Staphylococcus aureus determined by time-kill, lysis, leakage, and salt tolerance assays and electron microscopy. Antimicrob. Agents Chemother. 2002, 48, 1914–1920. [Google Scholar] [CrossRef] [PubMed]
- Shin, S. Anti-Aspergillus activities of plant essential oils and their combination effects with ketoconazole or amphotericin B. Arch. Pharmacol. Res. 2003, 26, 389–393. [Google Scholar] [CrossRef] [PubMed]
- Siddique, S.; Parveen, Z.; Butt, A.; Chaudhary, M.N.; Akram, M. The essential oil of Melaleuca armillaris (Sol. ex Gaertn.) Sm. leaves from Pakistan: A potential source of eugenol methyl ether. Inds. Crops Prod. 2017, 109, 912–917. [Google Scholar] [CrossRef]
- Homeyer, D.C.; Sanchez, C.J.; Mende, K.; Beckius, M.L.; Murray, C.K.; Wenke, J.C.; Akers, K.S. In vitro activity of Melaleuca alternifolia (tea tree) oil on filamentous fungi and toxicity to human cells. Med. Mycol. 2015, 53, 285–294. [Google Scholar] [CrossRef] [PubMed]
| Batch No. | % Age Yield |
|---|---|
| 1 | 0.25 ± 0.017 |
| 2 | 0.2 ± 0.017 |
| 3 | 0.15 ± 0.015 |
| Peak No. | Identified Compounds | Retention Time (min) | % of Total |
|---|---|---|---|
| 1 | alpha-phellendrene | 8.568 | 0.49% |
| 2 | p-Mentha-2,8-diene-1-ol | 9.278 | 0.92% |
| 3 | Terpinolene | 11.071 | 0.64% |
| 4 | Linalool | 11.595 | 0.50% |
| 5 | Methyl cinnamate | 19.359 | 0.77% |
| 6 | Methyl eugenol | 19.897 | 96.02% |
| 7 | Germacrene D | 20.663 | 0.67% |
| Replication No. | TFC Value (mm/g) | TPC Value (mm/g) | RPA Value (mm/g) | DPPH %Age |
|---|---|---|---|---|
| R1 | 955.67 | 108.945 | 300.79 | 89.13 |
| R2 | 942.16 | 108.945 | 314.88 | 86.95 |
| R3 | 955.67 | 112.145 | 307.81 | 84.45 |
| Mean | 951.16 | 110.01 | 307.83 | 86.84 |
| S.D | 7.80 | 1.84 | 7.04 | 4.22 |
| S.E | 4.50 | 2.60 | 4.06 | 2.43 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yasin, M.; Younis, A.; Ramzan, F.; Javed, T.; Shabbir, R.; Noushahi, H.A.; Skalicky, M.; Ondrisik, P.; Brestic, M.; Hassan, S.; et al. Extraction of Essential Oil from River Tea Tree (Melaleuca bracteata F. Muell.): Antioxidant and Antimicrobial Properties. Sustainability 2021, 13, 4827. https://doi.org/10.3390/su13094827
Yasin M, Younis A, Ramzan F, Javed T, Shabbir R, Noushahi HA, Skalicky M, Ondrisik P, Brestic M, Hassan S, et al. Extraction of Essential Oil from River Tea Tree (Melaleuca bracteata F. Muell.): Antioxidant and Antimicrobial Properties. Sustainability. 2021; 13(9):4827. https://doi.org/10.3390/su13094827
Chicago/Turabian StyleYasin, Mursleen, Adnan Younis, Fahad Ramzan, Talha Javed, Rubab Shabbir, Hamza Armghan Noushahi, Milan Skalicky, Peter Ondrisik, Marian Brestic, Sabry Hassan, and et al. 2021. "Extraction of Essential Oil from River Tea Tree (Melaleuca bracteata F. Muell.): Antioxidant and Antimicrobial Properties" Sustainability 13, no. 9: 4827. https://doi.org/10.3390/su13094827
APA StyleYasin, M., Younis, A., Ramzan, F., Javed, T., Shabbir, R., Noushahi, H. A., Skalicky, M., Ondrisik, P., Brestic, M., Hassan, S., & EL Sabagh, A. (2021). Extraction of Essential Oil from River Tea Tree (Melaleuca bracteata F. Muell.): Antioxidant and Antimicrobial Properties. Sustainability, 13(9), 4827. https://doi.org/10.3390/su13094827











