Following the ISO 14040 (2006) and ISO 14042 (2006) standards, the methodology used for the development of the LCA included goal and scope definition as well as the inventory analysis. The methodology followed for the Economic Analysis was developed accordingly.
2.1. Goal and Scope
This study aims to assess the environmental benefits and economic advantages generated from the implementation of an AO unit at a full-scale WWTP for the removal of PP. They were previously detected, during sampling campaigns performed by some of the authors of the present work, on the influent and effluent of the liquid fraction of the WWTP [
1,
14]. The list and concentrations of the PP detected in the studied MWW effluent are reported in
Table 1.
The full-scale WWTP selected as case study was designed for treating 37,997 m
3/day of MWW at full load, corresponding to 248,685 inhabitants-equivalent, covering the population of an area of about 880 km
2 in the center of Portugal. Currently, it is treating an average flow of 23,100 m
3/day. This WWTP receives domestic and industrial effluents, leachates coming from a municipal landfill, effluents from various hospitals and clinics, and a small flow of swine breeding wastewaters.
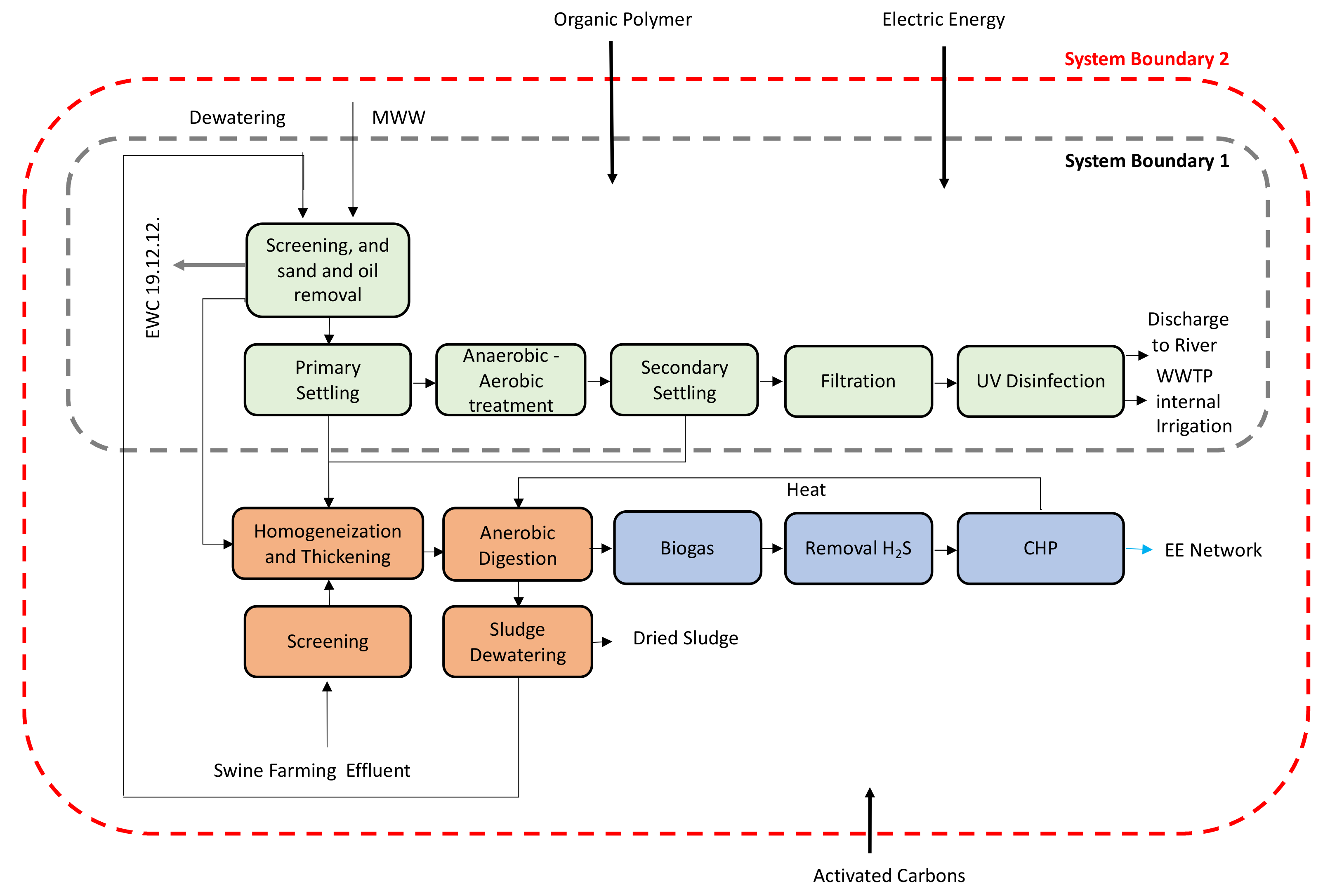
Figure 1 shows the WWTP flowsheet.
Table 2 reports the water quality parameters of the influent and effluent of the WWTP (average data of 2019) [
38].
The WWTP includes (i) liquid, (ii) solid, and (iii) gas-phase treatments (
Figure 1). The liquid phase is submitted to a medium load biological treatment by activated sludge process in three Phoredox (A/O) reactors, where the organic matter is removed. Then, it is sent to filtration and UV disinfection. The solid phase is anaerobically digested in three mesophilic digesters with swine breeding wastewaters for biogas and digestate production. The gas-phase (biogas) is burnt in a combined heat and power (CHP) engine for electricity and heat production [
39,
40].
The WWTP works for 365 d/y, 24 h/d. The produced electricity is entirely sold to the Portuguese national electric network, and the produced heat satisfies the internal need for heating the three digesters. The electricity needed for plant operation is bought from the national electric network. Currently, the WWTP produces an average of 11,941 t/y of dried sludge that is sent to a third-party company for further treatments aimed to the agricultural valorization of sludge [
38].
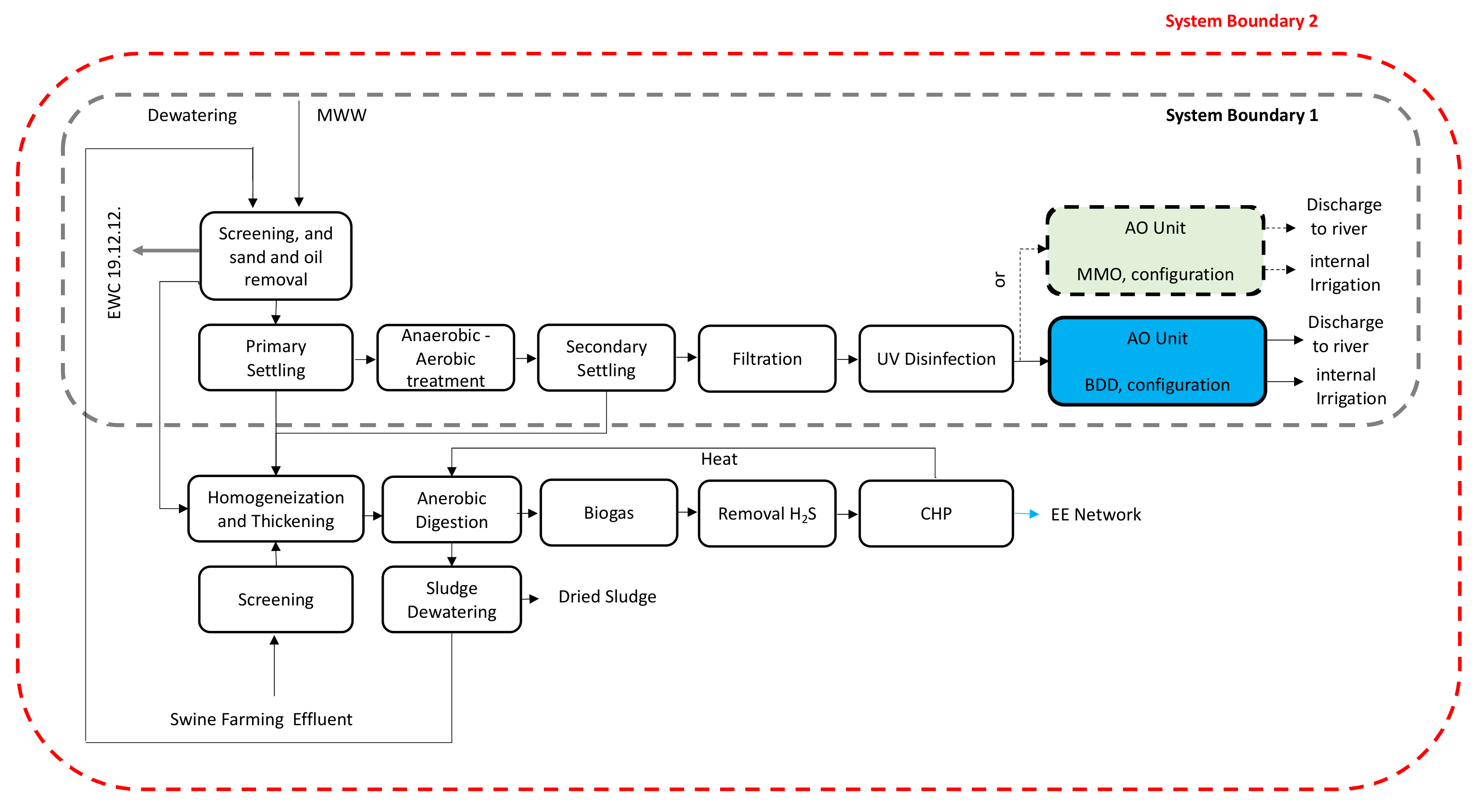
The proposed AO unit is hypothesized to be implemented as an end-line tertiary treatment before the water discharge. The AO unit includes three sheltered reinforced concrete tanks for water storage, double-lined with epoxydic resin to prevent corrosion, and equipped with 67 anode-cathodes pairs, taking into consideration two different anodes configurations, namely BDD and MMO, respectively, when cathodes are meant to be made of stainless steel (
Figure 2).
2.2. Methodology for Life Cycle Assessment
The environmental burdens associated with the presence of PP in the WWTP effluent and those associated with the implementation of the AO unit were assessed from an LCA perspective, using 1 m3 of raw wastewater (influent) as a functional unit.
For this LCA, dynamic system boundaries were considered. In a first analysis, the system boundaries were placed around the liquid flows (System boundary 1,
Figure 1). In a second analysis, to provide a comprehensive assessment on the role of the renewable electricity production in the LCA results, the treatment of the sludge (solid fraction) and the production of electricity from biogas (gas fraction) were included (System boundary 2,
Figure 1).
The flows and energy consumption of the WWTP were defined based on the 2019 operational data [
38]. These data are reported in
Tables S1 and S2 and integrated with
Figure S1. The literature suggests that PP can be adsorbed by sewage and biological sludge (solid fraction) [
11]. During the treatment of MWW (Q5,
Figure S1) and sludge dewatering stage (Q16,
Figure S1), it is expected that part of the adsorbed PP is released back into the liquid fraction, being eventually metabolized and discharged in receiving water bodies. The other part of PP remains adsorbed to the primary and secondary sludges. This study only assesses the environmental impacts associated to the presence of PP in the liquid fraction of MWW, as they were detected in the sampling campaign performed by some of the authors of this work and do not include the analysis of the environmental burdens associated to the presence of PP in the sludge (System Boundary 2,
Figure 1). No further sludge valorization as soil amendments is considered.
The implementation of the AO unit was based on the laboratory results obtained by Popescu et al. [
26] and Rosales et al. [
28] in the framework of REWATER International Research project (funded by the EU and FCT/UEFISCDI/FORMAS, under the ERA-NET Cofund WaterWorks2015 Call), and on pilot scale results obtained by Urtiaga et al. [
41,
42] on real effluents. Regarding the MMO anodes, the main efficiency parameters were retrieved from published literature [
43,
44].
This LCA takes into consideration the capital goods for AO unit implementation, including electrode manufacturing, as they are estimated in Ecoinvent v.3.5 [
45] database and integrated with literature data and personal communications provided by the manufacturers.
For the development of LCA, a consequential approach was adopted [
46]. The allocation of impacts was limited as much as possible, and when needed, it follows the system expansion methodology, known as “avoided-burden method” [
47]. In the present study, the following avoided burdens were considered: (i) the partial re-use of treated water for internal use at the WWTP, which allows sparing the equivalent amount of native “Water, unspecified natural origin, PT” and (ii) the avoided production of non-renewable electricity in the national electricity mix due to biogas cogeneration. No environmental burdens associated with the valorization of sludge, such as the partial replacement of soil amendments or use as feedstock for composting processes are allocated to the studied system.
This work was developed according to ISO 14040:2006 and 14044:2006 standards, by using SimaPro Version 9.0 software package [
48] which includes Ecoinvent v.3.5 database.
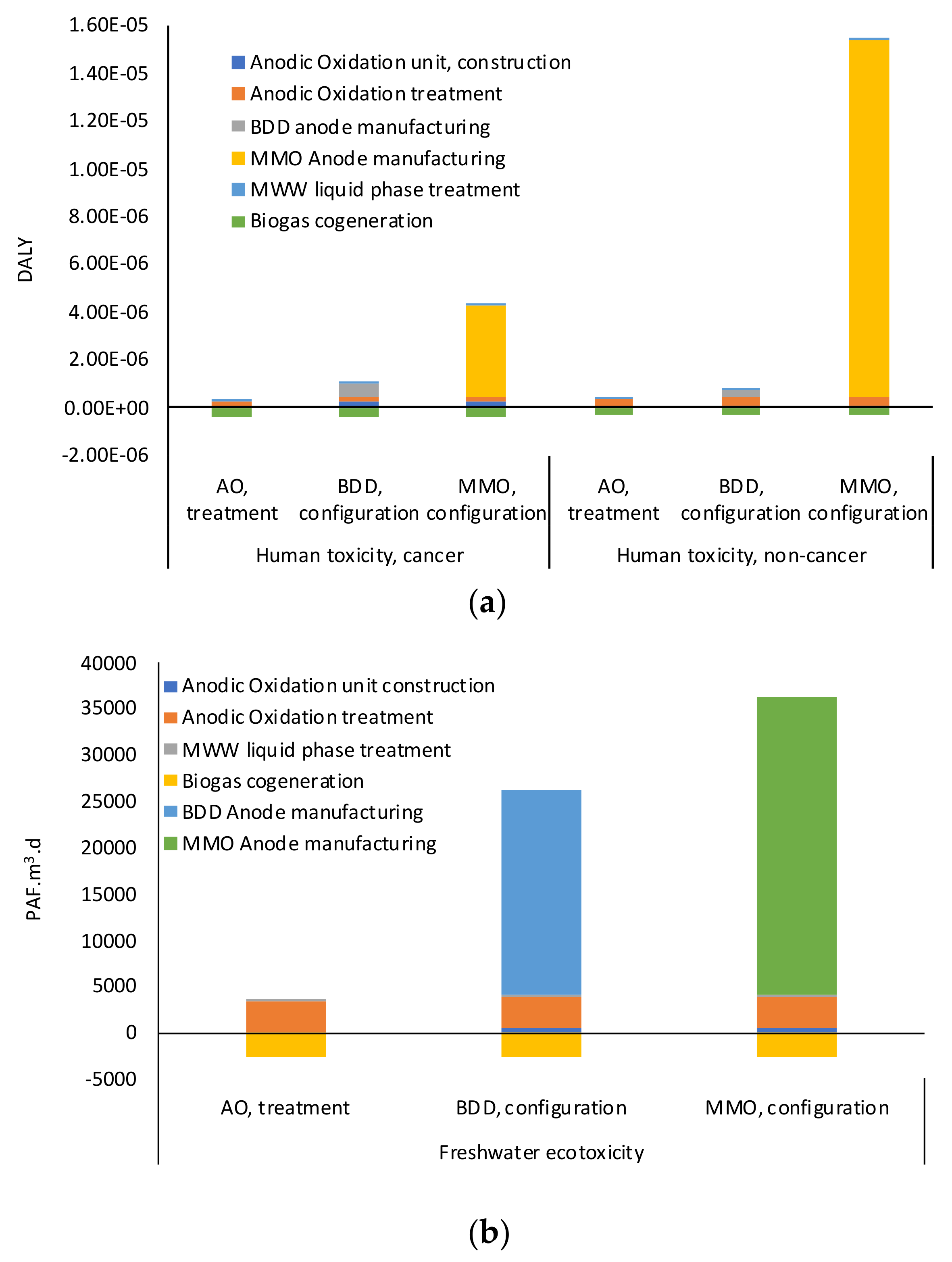
In order to provide a comprehensive and exhaustive analysis of all the environmental burdens associated with the presence of PP in the effluent of the WWTP and with the implementation of AO unit, the ReCiPe2016 Endpoint (H) v1.13 method was adopted [
49]. All the impact categories were calculated at the “Endpoint” level in human health (expressed in Disability-adjusted Life Year—DALY), ecosystem quality (expressed in species/y), and resource scarcity (expressed in US
$). “Hierarchist” was the perspective adopted, which is based on the most common policy principles with regard to timeframe and human expectations [
50]. The obtained results were interpreted both from a damage assessment perspective and through the “Weighting” function. “Weighting” allows comparing all the categories of impact through a single score (mPt) able to rank the impacts according to the importance of the effects they are able to trigger.
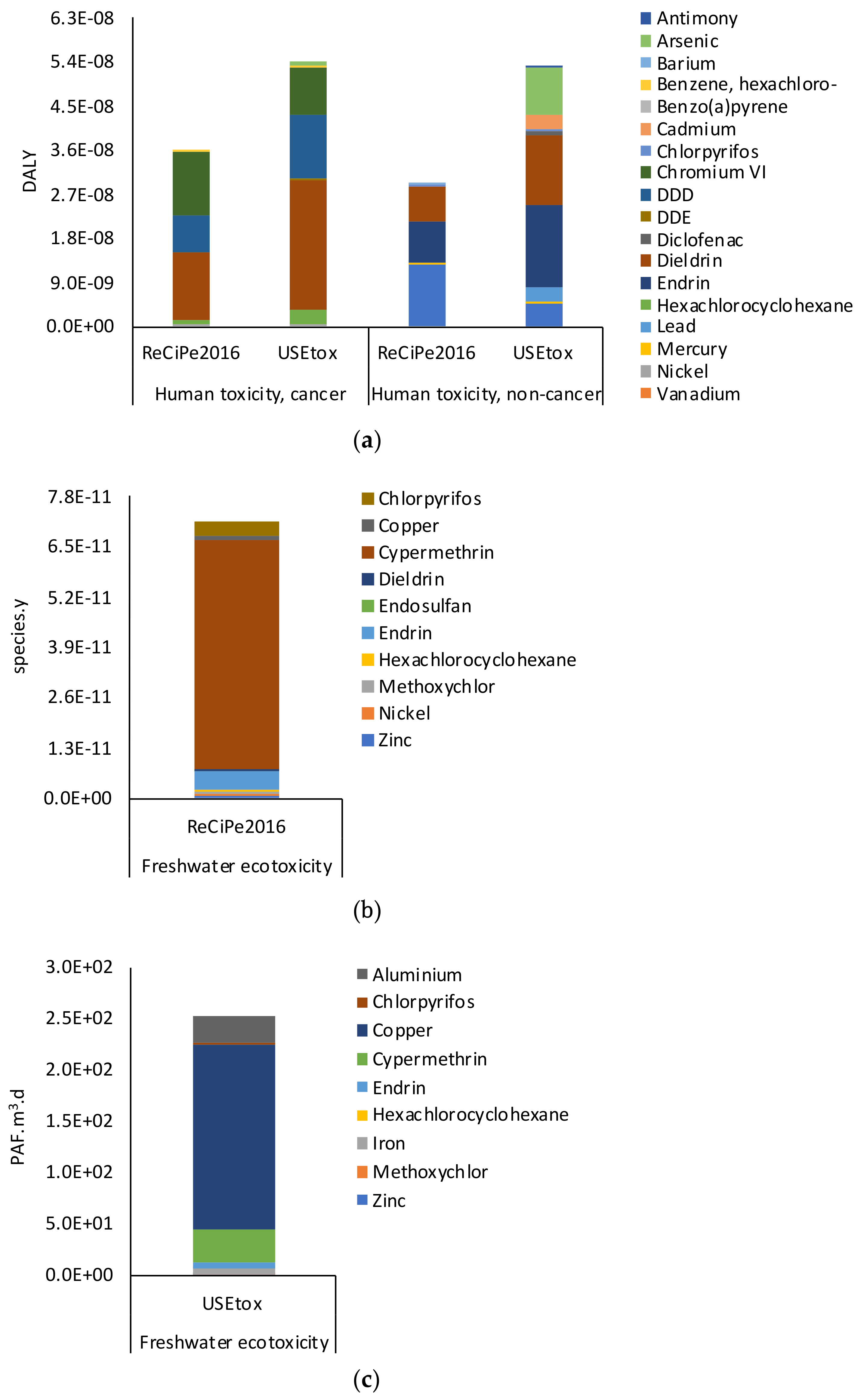
Considering that the ReCiPe2016 model includes all the pesticides but only four of the 16 pharmaceuticals were detected in the post-treated wastewater, the USEtox method (interim + recommended) v1.04 [
51] was used to integrate the assessment of the ecotoxicity related categories (freshwater and human toxicities). Moreover, to provide an exhaustive description of the fate, exposure, and effects of the detected PP, the Characterization Factors (CFs) of Azithromycin, Ciprofloxacin, Citalopram, Fluoxetine, Ofloxacin, and Venlafaxine, which were not present in the USEtox method available in SimaPro Version 9.0 software package, were integrated with the CFs proposed by Li et al. [
15] and Alfonsín et al. [
52]. The results are interpreted in a “Damage assessment” perspective that evaluates the environmental impacts toward Freshwater Ecotoxicity (PAF.m
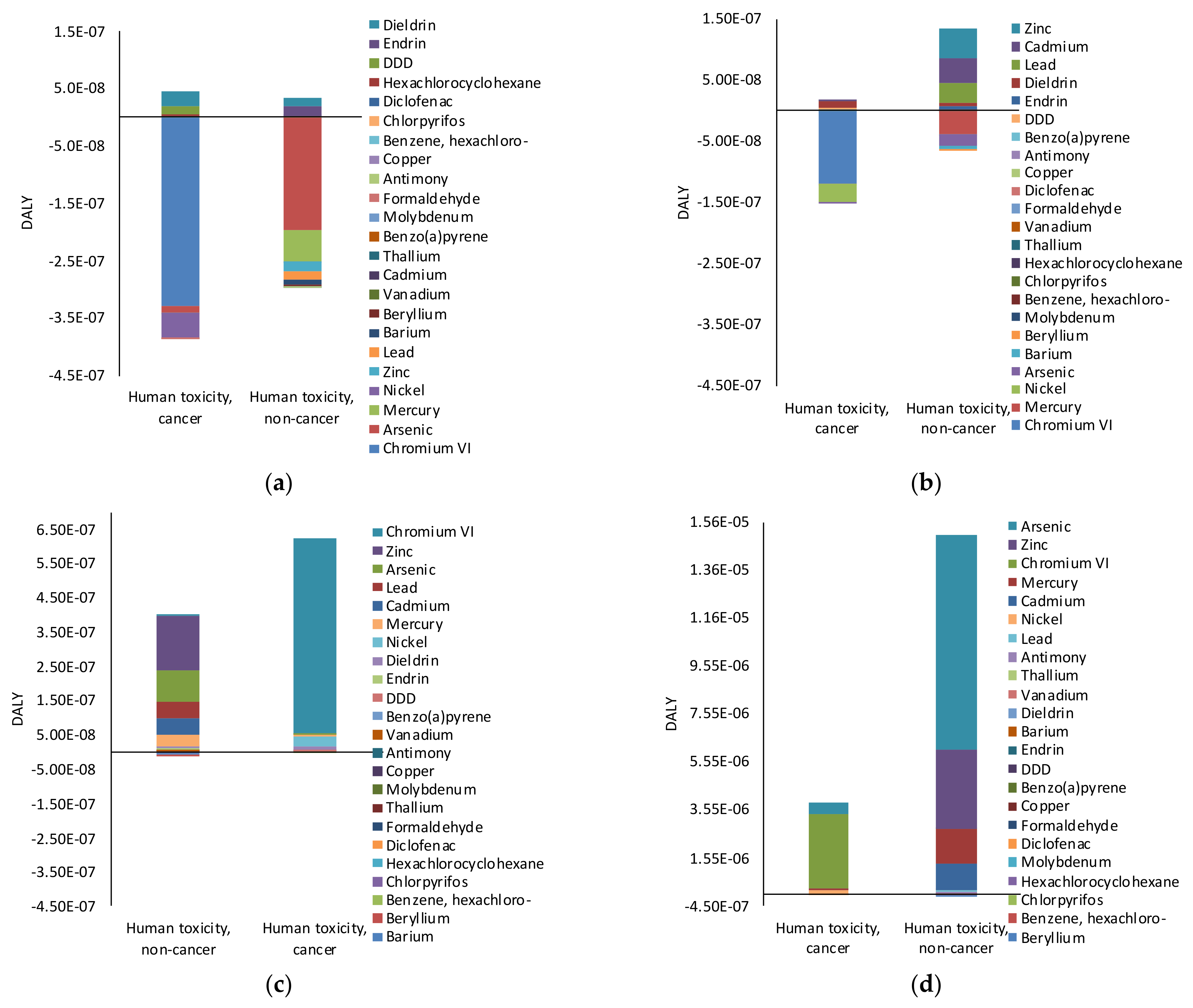
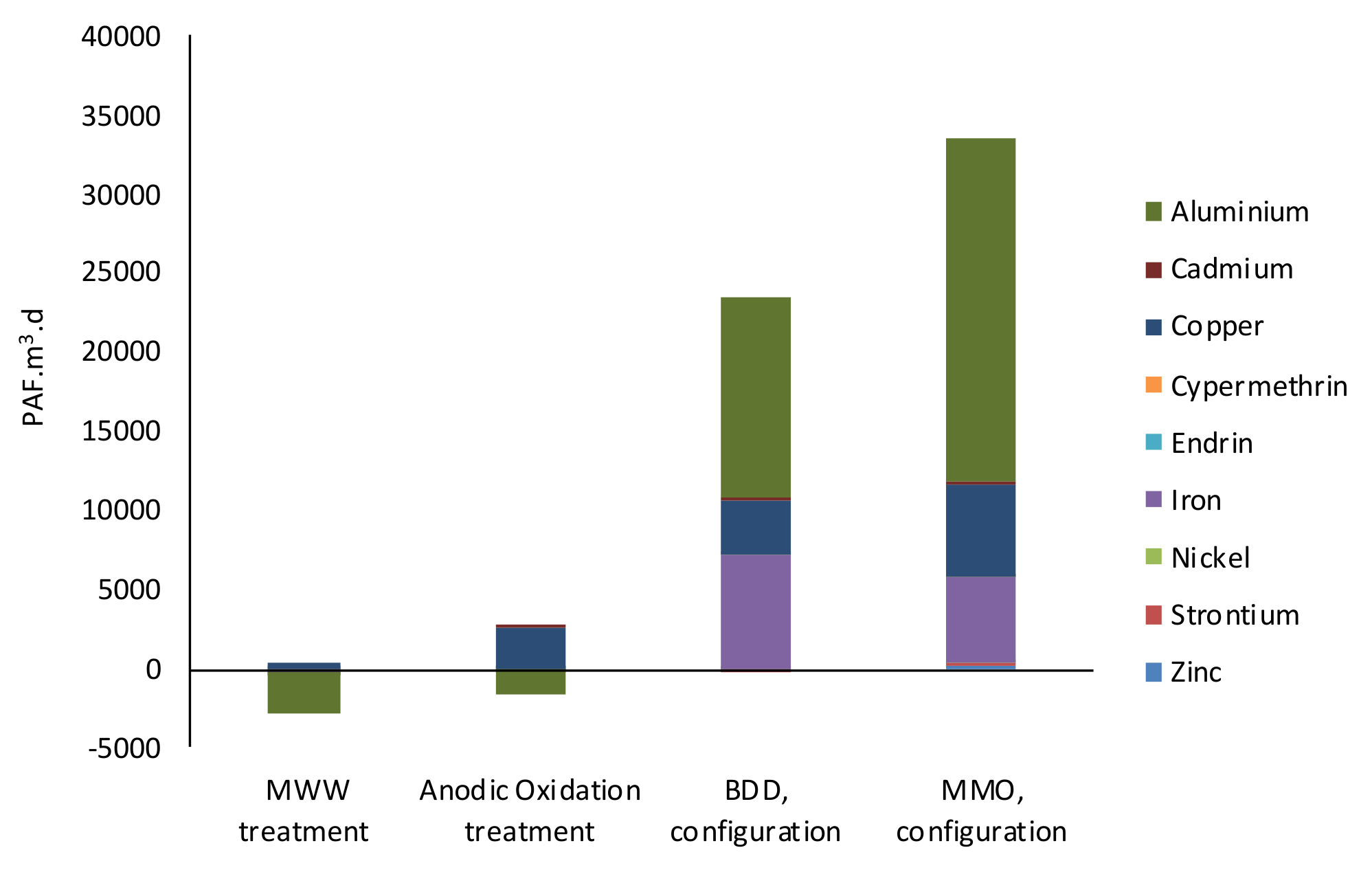
3.d) and Human Carcinogenic Toxicity and non-Carcinogenic Toxicity (DALY).
2.3. Methodology for Economic Analysis
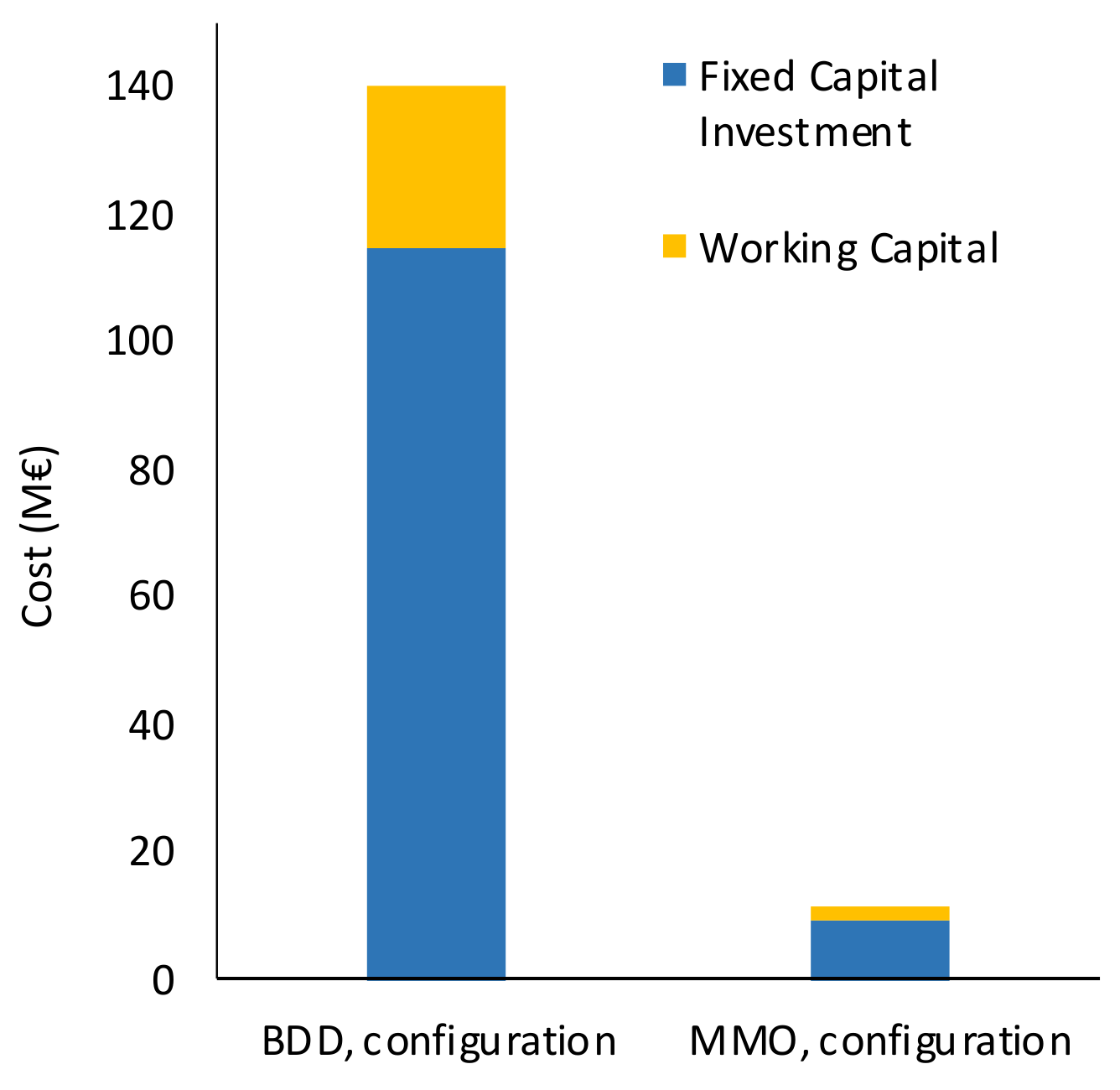
The economic analysis of the implementation of an AO unit at the existing WWTP was developed on the basis of the methodology suggested by Timmerhaus and Peters [
53]. Firstly, it consists in the assessment of (i) the Total Capital Investment (TCI), which corresponds to the sum of Fixed Capital Investment (FCI) + Working Capital (WC), from the value of the Purchase Cost of Delivered Equipment (PCDE). The PCDE is the total cost supported for acquiring all the equipment needed for the AO unit implementation and adding 10% for its delivery. The estimation of the direct cost (delivered equipment installation, purchasing, and installation of instrumentations, control and electrical systems, piping, building construction, yard improvement, and installation of service facilities), as well as the indirect costs (engineering and supervision, construction expenses, legal expenses, contractor’s fee, and contingency), were estimated as a fraction of the PCDE. The percentages used for the calculation of each of the direct and indirect sub-costs, as well as for WC are based on the delivered equipment method [
53] and are reported in
Table S3.
The investment was intended to be made in a three-year period of time, assuming that the startup of the AO unit (time 0) at the WWTP is made 3 years after the date of the cost assessment. It is assumed that 15% of the FCI is spent in the first year (year −2), 35% is spent in the second year (year −1), and 50% is spent at the end of the third year (time 0). The 2020 Portuguese inflation rate (−0.1%) was applied to FCI after the initial estimation [
54]. It is assumed to spend all the WCI value, properly corrected with inflation rate during year 0.
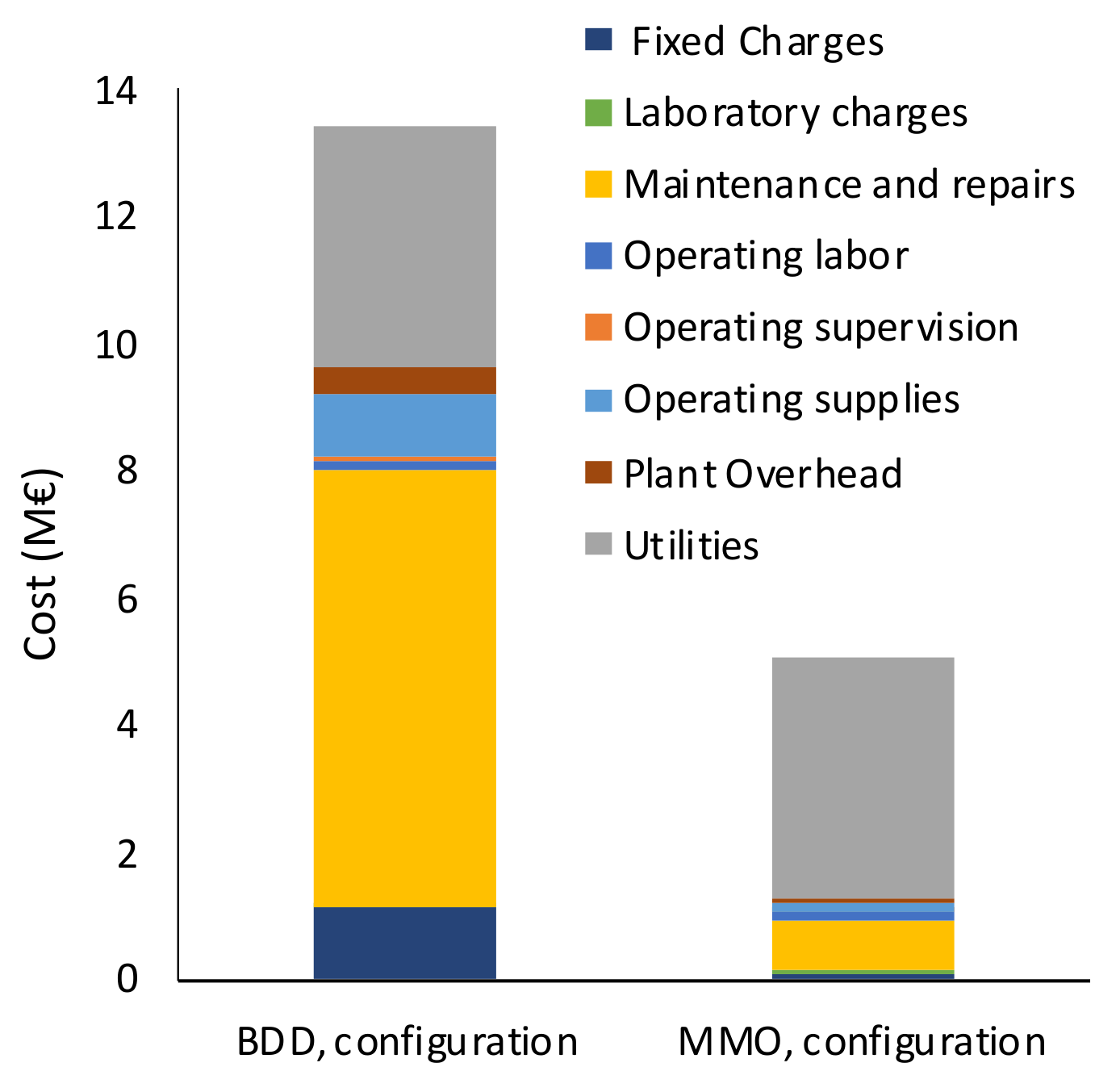
The annual Total Treatment Cost (TTC) of the AO treatment was calculated as the sum of the Variable Costs (maintenance and repair, utilities, operating supplies, laboratory charges, and chemicals), Fixed Costs (taxes, financing interest, insurance, rent), Plant Overhead, and General Expenses (administration, research, and development). The purchase cost of raw material, which in this case corresponds to wastewater, was assumed to equal to 0 €. The methodology used and fractions applied for the calculation of the annual TTC are reported in
Table S4. The depreciation of the investment was calculated hypothesizing the investment recovery along all the AO unit lifetime (20 years) according to the depreciation factors reported (1/y) in
Table S5.
For the profitability analysis of the projects, it is assumed, in a first step, that the revenues come only from the public taxes for MWW treatment and from the sale of the produced electric energy to the Portuguese network. No revenues are taken into consideration for the sale of stabilized compost nor for the potential sale of treated water for agricultural purposes.
Regarding the utilities, it is considered that the electric energy required for the WWTP operation is entirely bought from the Portuguese electric grid at the price of the feed-in tariff of the WWTP.
For economic analysis purposes, the coarser solid fractions and sands produced in the primary and secondary settlers are meant to be sent to landfill as non-hazardous wastes under the European Waste Code 19.12.12. The cost of their disposal is included in the cost of the treatment of 1 m3 crude water.
The current cost of the MWW treatment without the AO unit is included in the TTC as a fixed cost.
The profitability analysis of the project was developed using the following parameters: (i) the average Return of Investment rate (ROI) (%/y), (ii) Payback Period (PbP) (y), and Net Return (NR) of the investment (€).
The calculation of the aforementioned parameters was performed according to the Equations (1)–(3):
where
is the total Net Profit (€), TCI is the Total Capital Investment (€) assuming an income tax rate of 24.5% [
38] and a lifetime of 20 years (y);
where
is the Total Operating Cash Flow (€), calculated as
;
where
(%/.
y) is the minimum acceptable rate of return, which was fixed at 6%.
2.4. Environmental Life Cycle Inventory
Tables S6 and S7 summarise the environmental Life Cycle Inventory (LCI) data and the quantification of all the direct, indirect, and avoided burdens for the treatment of liquid and solid fractions of the MWW, respectively.
Table S8 reports all the input and output data related to the implementation of the AO treatment unit at the WWTP.
Regarding the LCI of the treatment of the liquid and solid fractions (
Tables S6 and S7), the operational data of the WWTP were integrated on the basis of the following assumptions: (i) in the absence of direct measuring at the plant, the emissions to air from primary and secondary settlers, biological reactors, water body, as well as from sludge thickener/homogenizer, digesters, and swine breeding waste screening tank were calculated according to the CFs reported in
Table S9; (ii) the emissions to soil were calculated considering that 100,619 m
3/y of the treated wastewater is internally used at the WWTP for irrigation of green yards [
38]; (iii) for H
2S removal from biogas before cogeneration, Cabot BG1 activated carbon was considered as the adsorbent material, assuming an average H
2S concentration of 500 ppm and an adsorption capacity of 0.32 kg
H2S/kg
AC [
55].
The environmental LCI of the AO treatment unit (
Table S8) is based on a BDD anode electrooxidation pilot plant as tested by Urtiaga et al. [
41] and integrated with the results obtained by Popescu et al. [
26] and Rosales et al. [
28]. A similar loading dose, Q, and current density, J, as used by Urtiaga et al. [
41], were adopted as fixed parameters for the AO unit in the present study (48.6 × 10
−6 C/kg COD
removed and 300 A/m
2, respectively). The volume,
V, of the AO reactor, was calculated taking into account the average inflow of 950 m
3/h MWW [
38] and a Hydraulic Retention Time (HRT) of 2 h. The HRT is a parameter that must be chosen depending on the concentration of the organic substances to be removed. On the basis of the laboratory results obtained by Popescu et al. [
26], Rosales et al. [
28], and Urtiaga et al. [
41] on real effluent, an HRT of 2 h guarantees a removal efficiency of at least 70% of the chemical oxygen demand (COD) by using the BDD electrode. For MMO anodes, the same removal efficiency as BDD was assumed [
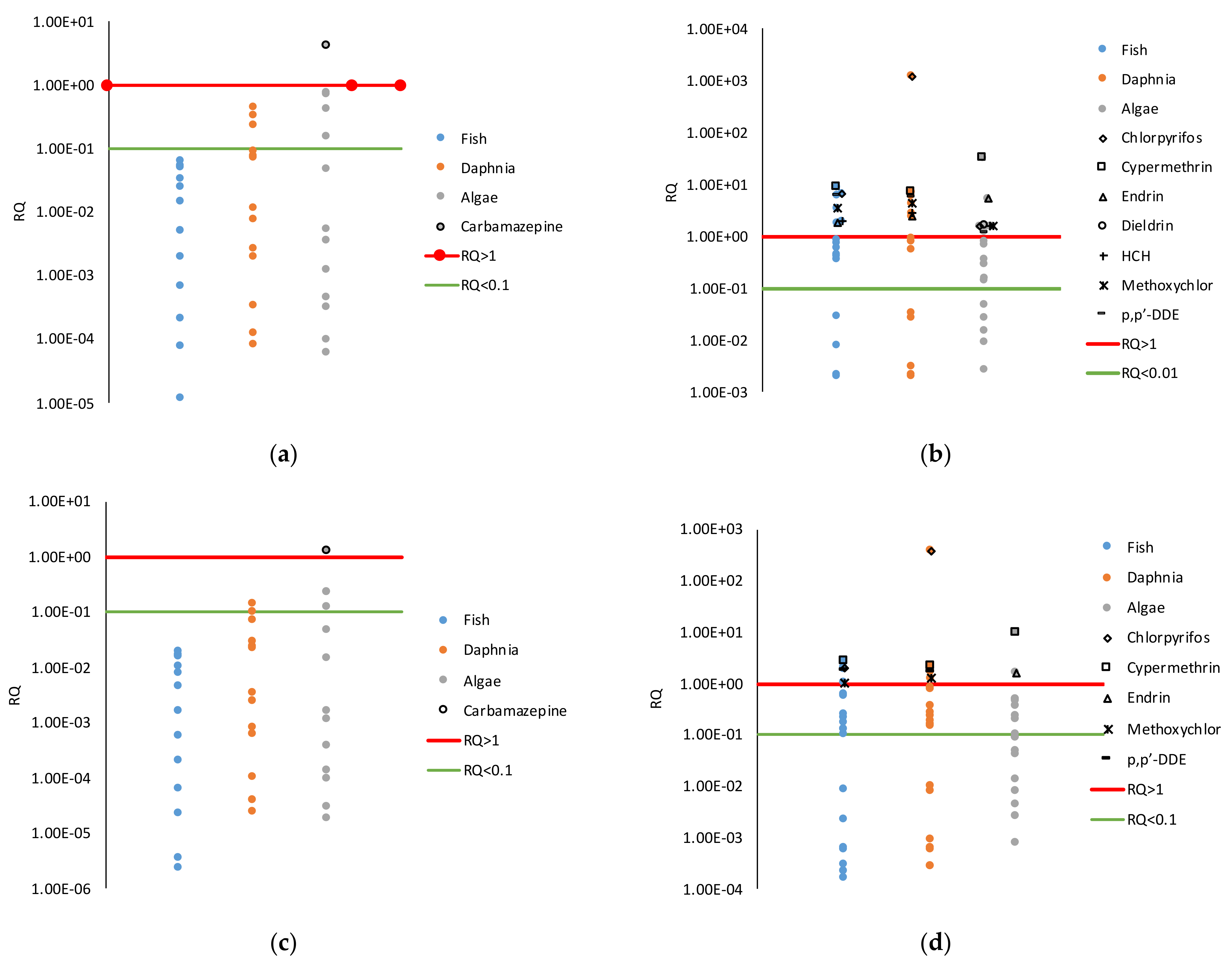
56]. For the purposes of this work, it is assumed that the concentrations of PP are included in the total COD and PP removal efficiency is equal to the removal efficiency of COD (70%). These values allow reducing the risk quotient (RQ) at a value lower than 1 for most of the PP detected. RQ represents the risk that the detected PP has on the aquatic environment at three representative trophic levels (algae,
daphnia, and fish), and it is defined as the ratio of potential exposure to the substance and the level at which no adverse effects are expected (PNEC). The methodology followed for the calculation of the RQ is reported elsewhere [
1]. The LCI of the PP, in the presence and absence of the AO unit, are reported in
Table S10.
Thus, assuming a COD concentration at the AO unit inlet (COD
in) of 0.068 kg O
2/m
3 [
38] and 0.020 kg O
2/m
3 at the outlet (COD
out) (η = 70%), the total operating current I (A), calculated according to Equation (4), is 605,754 A.
where I (A) is the electrical intensity, Q (C/kg) is the loading dose, HRT (s) is the hydraulic retention time, COD
in (kg O
2/m
3) is the COD in the influent of the AO unit, COD
out (kg O
2/m
3) is the COD in the effluent, and V (m
3) is the volume of the AO reactor.
Once the total intensity I (A) and the charge density J (A/m
2) are known, the total anode surface A (m
2) necessary to eliminate the established COD with a reaction time of 2 h can be obtained through Equation (5):
The total anode surface is 2019 m2. The cathode is assumed to have the same surface as the anode.
The Electric Energy (EE) (kWh/m
3), required for the AO treatment of 1 m
3 of water, was calculated by Equation (6):
where the voltage
v (V)
, was obtained by Equation (7)
assuming the conservative value of 75 kWh/kg COD
removed, as measured by Urtiaga et al. [
41] at pilot scale on similar effluent.
The optimum electrode spacing,
d (m), for proper system operation was calculated according to Equation (8):
where
is the wastewater conductivity (µS/cm) (1500 µS/cm) [
57],
is the voltage (V), and J is the current density (A/m
2). The obtained values are 5.56 V for voltage, 3.55 kWh/m
3 for electric energy, and 0.3 cm for optimum electrode spacing.
For the environmental impact related to the construction of the AO unit, it was assumed that 3 sheltered water storage tanks (total capacity of 2500 m
3) made of steel-reinforced concrete double lined with epoxy resin are built. These tanks are equipped with 67 anode/cathode pairs of 10 m × 3 m × 0.01 m. For that purpose, the amount of normal and lean concrete as well as of reinforcing and low-alloyed steel needed for water storage tanks constructions were retrieved from the Ecoinvent 3.5 database (Water storage {RoW}| construction). The amount of epoxy resin was calculated based on the water storage tank dimension, considering two layers and a resin yield of 3 m
2/(kg.layer). A total amount of 5700 kg epoxy resin was obtained, being selected from the Ecoinvent 3.5 database under the process named as “Epoxy resin, liquid {RER}| market for epoxy resin, liquid”. Moreover, 1458 kg (200 m length) of 20 ” SCH10 chromium steel pipe (7.9 kg/m) [
58] was considered for piping connections. The wastewater coming out from the disinfection tank is pumped to the AO unit through three stainless steel pumps (one for each AO tank) of 10 kW and a head of 5 m with a flow rate of approximately 320 m
3/h.
For cathode production, 79.25 t/y of steel are considered, which were 100% allocated to “Steel, low-alloyed {GLO}| market for”, available in Ecoinvent 3.5 database.
Finally,
Table S11 reports the inventory data for the manufacturing of the two upscaled anodic configurations, based on BDD and MMO coatings.
The manufacturing process of BDD anodes was developed in SimaPro 9.0 on the basis of the information retrieved from manufacturers [
59] and on the literature [
60], considering the BDD film deposition by the Chemical Vapor Deposition (CVD) technique on titanium plates used as substrate. Titanium is one of the most used substrate materials for BDD production due to its high conductivity, corrosion resistance, and other excellent physical and mechanical properties [
37]. For BDD film deposition, methane in excess of hydrogen (1% CH
4 in H
2), doped with trimethyl boron (1–3 ppm), were used as reactive gases [
61]. In the absence of other information at pilot/industrial scales, the scale-up of the gas flows, required for BDD film production on titanium plates was obtained by adopting a geometrical scale-up factor. The gas flows were calculated by dividing the anode area needed at full-scale for the exposed electrode area for BDD film deposition used at lab-scale by Michaud et al. [
61]; a value of 1.01E+07 was obtained. For the energy consumption of the CVD process, the value proposed by Gassner et al. [
62] was upscaled accordingly. Regarding the emissions to the atmosphere during BDD film deposition, it was assumed that all the hydrogen, used in excess, is emitted entirely to the atmosphere and all methane and trimethyl boron are consumed during diamond growth.
Based on the information retrieved from the manufacturers of the electrodes [
63,
64] which was integrated with data available in the literature [
35], it was assumed that the production process of MMO anodes is performed through thermal decomposition; this consists in the painting of titanium plates by an MMO solution followed by thermal treatment at high temperature (400 °C). Assuming that the MMO solution is made of Pt oxides with Pt loading of 50 g/m
2 [
64] and yield of 1.5 kg/m
2, 101 kg of Pt oxides are used. “Platinum {GLO}| market for” available in Ecoinvent 3.5 was selected. Moreover, according to the procedure for MMO anode preparation described by Vasconcelos et al. [
35], 2518 kg of ethylene glycol (Ethylene glycol {GLO}| market for), and 2337 kg citric acid (Citric acid {GLO}| market for) must be used. The thermal treatment is intended to be carried out in an industrial furnace fueled by natural gas (NG), according to the following heating scheme: (i) 130 °C for 30 min to evaporate the water; (ii) 250 °C for 10 min to promote the adhesion of the layer to the substrate, and (iii) final calcination temperature applied for 5 min to form metal oxides and to eliminate the organic matter. Assuming a specific heat, Cp, for the titanium of 520 J/(kg.K), it was estimated that 989 MJ/y of heat are required for coating 44.6 t of MMO anode. The heat supply process retrieved from the Ecoinvent 3.5 database was the following: “Heat, district or industrial, natural gas {RER}| market group for”.