Abstract
Nitrogen (N) in the agricultural production system influences many aspects of agroecosystems and several critical ecosystem services widely depend on the N availability in the soil. Cumulative changes in regional ecosystem services may lead to global environmental changes. Thus, the soil N status in agriculture is of critical importance to strategize its most efficient use. Nitrogen is also one of the most susceptible macronutrients to environmental loss, such as ammonia volatilization (NH3), nitrous oxide (N2O) emissions, nitrate leaching (NO3), etc. Any form of N losses from agricultural systems can be major limitations for crop production, soil sustainability, and environmental safeguard. There is a need to focus on mitigation strategies to minimize global N pollution and implement agricultural management practices that encourage regenerative and sustainable agriculture. In this review, we identified the avenues of N loss into the environment caused by current agronomic practices and discussed the potential practices that can be adapted to prevent this N loss in production agriculture. This review also explored the N status in agriculture during the COVID-19 pandemic and the existing knowledge gaps and questions that need to be addressed.
1. Introduction
The current global population of 7.8 billion is projected to reach over 9 billion by 2050 [1]. This projected boom in population would mandate an approximately 70% increase in global agricultural production to ensure food security in the developed and nearly 100% in the developing countries [2]. To keep up with this demand, global agriculture will continue to consume more amendments in both inorganic and organic forms that can support agricultural production and simultaneously battle the food waste crisis where one-third of the annual produced food goes to waste (1.3 billion tons) [3]. Nitrogen (N) is a critical element for all living organisms and assimilation of N by both terrestrial and aquatic plants is limited by its forms in the ecosystem [4].
Over the last five decades, the global N cycle has changed significantly due to the incessant input of nitrogen fertilizers. Nitrogen cycling involves five major steps; biological N fixation, ammonification, nitrification, N assimilation into microbial biomass pool, and finally denitrification [5]. However, both chemical N fertilizers and organic manure are often applied to soil in exceeding amounts for crop growth requirements lead to N loss. Of the applied N for crop growth, only 45–50% is being incorporated into the agricultural products [6] and the remainder is subjected to substantial loss [7]. A significant amount of N in soil and may be lost to the environment as NO3, NH3, or N2O [8,9]. NO3 may also continue to undergo recycling in the soil–water–air system and convert to N2O and N2 through the denitrification process and released back to the atmosphere [10]. Particularly, N2O emission is substantial from production agriculture at the beginning stage of N fertilizer application during the cropping season [11]. Furthermore, the potential warming effect of N2O and the short-term cooling effect of NH3 and NOx both has major consequences on global human and environmental health [12,13]. Additional complications in estimating N2O arises from the nonlinear nature of N2O emission in response to N fertilizer application along with other soil and environmental controlling factors [14]. Intergovernmental Panel on Climate Change (IPCC) linear model, one of the approaches to estimating N loss in terms of N2O, which may overestimate N loss at lower N application rates while underestimating higher rates [15,16,17] (Figure 1).
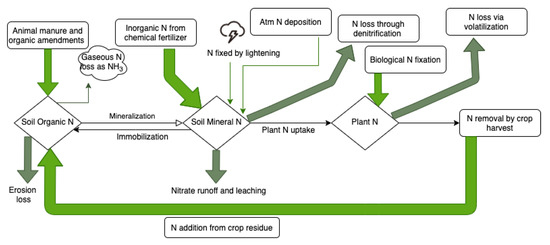
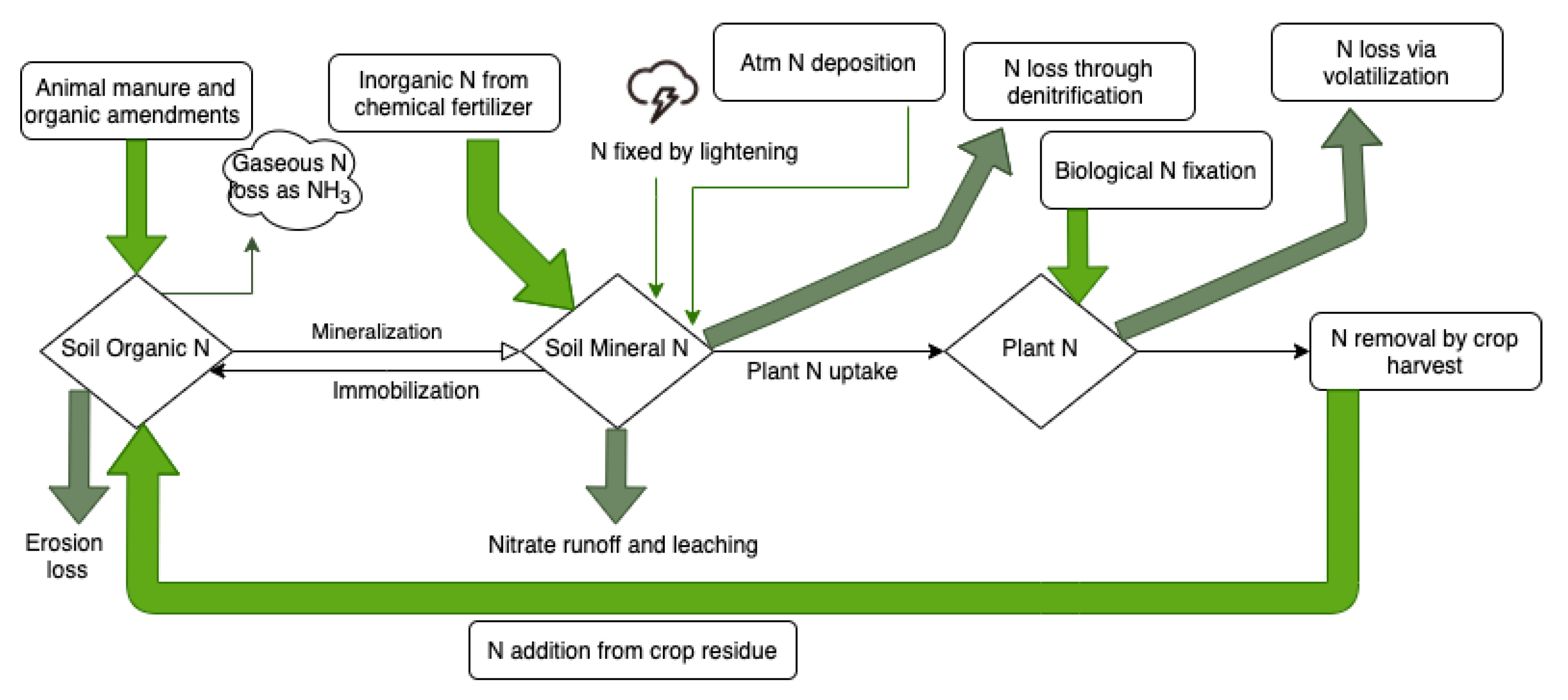
Figure 1.
The nitrogen cycle in the soil.
Agricultural contribution to global N pollution requires constant monitoring and adoption of necessary efforts at the farm, regional, and national levels to mitigate and regulate environmental degradation. Nitrogen footprint, under agricultural context, is the amount of N released from resource use in agricultural production at every step of the production line both upstream or downstream [18]. Nitrogen footprint can serve as an indicator of the usage and losses of N in the production and consumption of food and energy, thereby, an ecosystem service. The objective of this review was to explore the agricultural status of N in the current agronomic production system and possible mitigation strategies to address and minimize N loss for sustainable agriculture.
2. Nitrogen Footprint in the World
Nitrogen plays such a cardinal role in ecosystem productions and the Earth’s energy balance that any changes in the N cycle will bring about profound impacts on the global ecosystem and human health [19,20,21]. It is necessary to track the gains and losses in the N cycle and there are several tools to do just that. For instance, a quantifying tool that serves as an indicator of N losses to the environment from different stages of the N cycle ranging from production to consumption levels is called N footprint [22]. Globally, agriculture dominates N footprints [23]. Furthermore, global trades connecting different countries in importing and exporting agricultural commodities also leave a major mark on the N footprint. Nitrogen footprint can be a useful tool to help resolve the critical dilemma between the optimized N use (nitrogen use efficiency; NUE) and minimize negative impacts associated with N use. The food N footprint is the dominant component of the per capita N footprint [24,25]. On average, the per capita N footprint of ten countries from different regions of the world ranges from 15 to 47 kg N per capita per year and the principal reason behind this is the difference in the protein consumption rates and N losses during agronomic production [22]. In Asia, China has a big per capita food N footprint and it has increased almost 50% over 30 years (i.e., 1980 to 2008), however, it was still close or lower than that of countries in North America or Europe [24,26]. Of this per capita food N consumption, almost 40% is from protein, which is much lower than in other countries [26,27]. In the case of fossil fuel consumption, China leaves a lower N footprint than the USA [18] but since it has limited to no regulatory measures in place to reduce NOx emission [28], hence, China has a higher energy N footprint compared to some of the European countries like UK and Germany [24,25,26]. Australia, a country in the Asia–Pacific region, due to a high protein diet and affordable food prices has a high food N footprint and a high energy N footprint due to higher N emission from electricity generation [22]. In North America, the USA has the largest N footprint for food production where the N footprint per capita is 39 kg N. This is followed by transportation where a relatively higher N footprint is observed compared to other countries [18]. In Europe, agricultural innovation was pioneered by the industrial revolution primarily at the turn of the 18th century, and again in the early 20th century with the arrival of the Haber–Bosch process. This nitrogen-based synthetic fertilizer production dramatically increased the productivity in agricultural sector, thus, countries like UK, Netherlands, Austria, and Germany have a higher food N footprint than many parts of Asia, typically ranging from 24 to 29 kg N capita−1 year−1 [18,29]. Table 1 details per-capita nitrogen footprints from different countries adopted from Oita et al. (2016) of nations [30].

Table 1.
Ranked per-capita nitrogen footprints of nations (NFP = nitrogen footprint).
3. Nitrogen Losses in the Environment
Nitrogen contains readily converted reactive chemical (reactive nitrogen; Nr) [24] species causing a cascading effect on the environment and impacting the global ecosystems [31]. These reactive species are N2O, NO3−, nitrite (NO2−), NH3, and ammonium (NH4+) and mostly of anthropogenic origins such as fossil fuel combustion and agriculture (both legume cultivation and use of industrial fertilizers) (Figure 2).
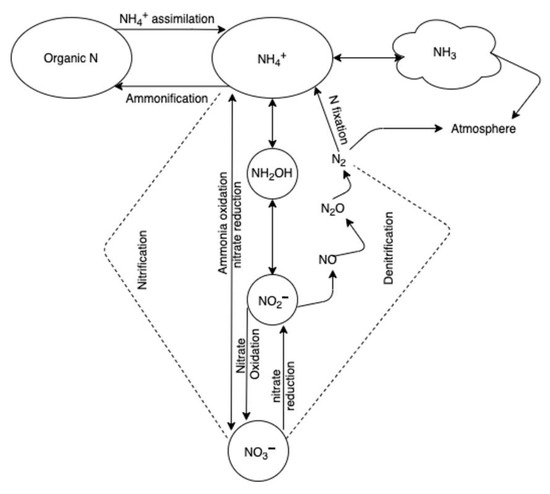
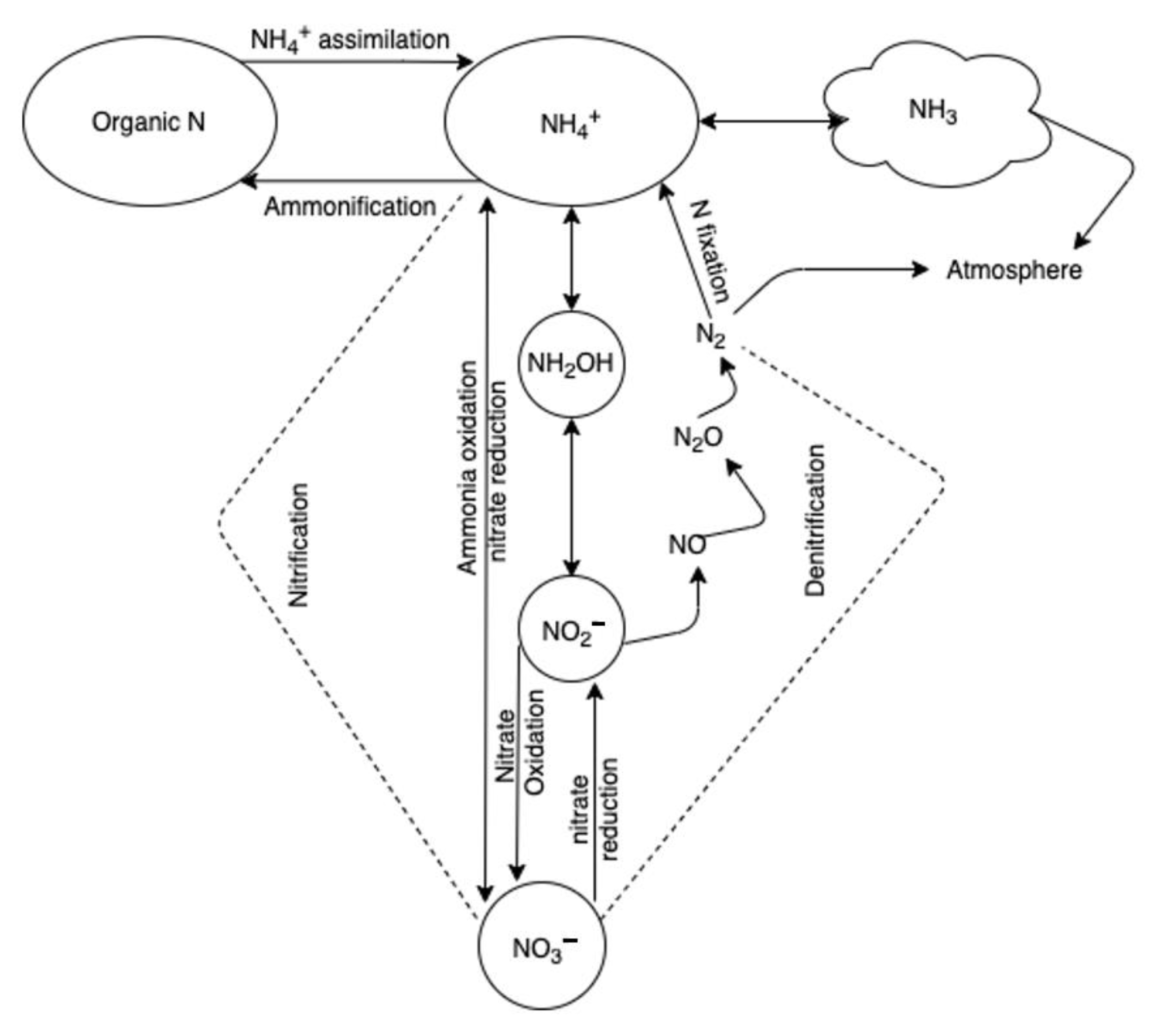
Figure 2.
The major processes of the nitrogen cycle in soil.
3.1. Ammonia Volatilization
Ammonia volatilization is one of the major sources of N loss from arable farms worldwide. Different soil conditions affect the volatilization rate of NH3 from the soil. Soils with high pH are generally prone to lose significant amounts of NH3, however, neutral or acid soil may also lose NH3, especially after inorganic fertilizer application like urea or organic amendments such as urine [32,33]. Moreover, soil and atmospheric temperature greatly affect the urea hydrolysis, thereby, the NH3 (aqueous) transfer rate from the soil solution to atmospheric NH3 (gas) [34]. Furthermore, low soil moisture tends to stimulate high soil solution concentration, thus, higher loss of NH3 [35]. When NH4 containing fertilizers such as ammonium nitrate (NH4NO3) or ammonium sulfate (NH4SO4) or urea (CH4N2O) are applied to soil during different stages of crop growth [36], they are typically subjected to immediate NH3 volatilization. For instance, upon application, urea undergoes hydrolysis ensuing higher soil pH in the microsites of soil causing transformation of NH4+ to NH3 [37,38]. The greatest amount of NH3 is released through anthropogenic activities [39], which releases around 7.6 million kg ha−1 of NH3 emission is from crop and animal husbandry accounting for more than 90% of the total emission [40]. Ammonia volatilization is a major problem to human health and the environment because it can react with acidic components of the environment such as sulfate (SO42−) or NO3− and form a secondary inorganic aerosol. NH3 emissions, moreover, significantly contribute to the formation of acid rain in the atmosphere and can be an indirect source of N2O emissions, and promote eutrophication of surface water bodies [35,41]. The economic implications of NH3 emission are particularly threatening to developing countries, for instance, India, as an earlier simulation study showed that almost one-third of the applied N fertilizers or manure is lost to the atmosphere as NH3 [42,43].
3.2. Nitrous Oxide and Oxides of Nitrogen (NOx) Emissions
The simultaneous process of nitrification (aerobic condition) and denitrification (anaerobic condition) produces N2O as the common byproduct in soil [44]. During nitrification, NH4-N is microbially converted to an intermediate product, hydroxylamine (NH2OH) followed by the production of NOH, and finally NO2. During nitrification, N2O is produced both at NH2OH and NO steps, while, on the contrary, denitrification, transforms NO3 or NO to N2 or N2O [22]. Nitrous oxide is considered one of the most critical greenhouse gasses due to its prolonged atmospheric lifetime (120 years) and more potent in trapping heat than CO2. Moreover, N2O is responsible for ozone depletion by reacting with stratospheric O2 and forming nitric acid [45]. Agricultural activities are the largest anthropogenic sources of atmospheric N2O emissions [46]. An increase in fertilizer application rates, N sources, are the dominant factors of N2O production in an agroecosystem [47]. Livestock, especially, traditionally grazed ruminants on poor quality fodder in pastures also plays a significant role in N2O and methane (CH4) emissions [48].
NO2 is released to the stratosphere mostly through fossil fuel combustion from vehicles and industrial plants in addition to natural processes of nitrogen in the soil. It has been highlighted by the WHO as a potential health risk and that exposure to this pollutant should be minimized [49]. Several pulmonary and cardiovascular diseases have been linked to long-term exposure to elevated levels of NO2. Some of the most documented cases include lung function growth deficit in children and compromise lung function in adults [50,51].
3.3. Nitrate Leaching
Apart from NH4+, NO3 produced during nitrification in the aerobic condition is a preferable form of N for plant uptake. However, it is highly mobile in soil due to its anionic nature and in well-drained soil, NO3 is the first nutrient to be washed off from the soil profile. The amount and distribution of rain and irrigation affect the NO3 loss below the root zone in the soil profile [52]. Studies done in the early 90s’ suggested that depending on the soil type, almost 80% of the applied N can be lost as NO3 runoff [53]. Colder temperature contributes considerably to nitrate leaching due to greater precipitation and slower plant uptake of nitrate [54]. Earlier studies on nitrate loss in soil suggested that more than 100 kg N ha−1 year−1 of nitrate was lost from grazeland after tillage through the soil profile over the next winter [55]. Soil structure and texture may also affect nitrate movement through the soil profile. Nitrate leaching is greatest in sandy soil with poor structure and slowest in clay soil [35]. Additionally, soil macrofauna movement and plant root growth often allow rapid nitrate movement in soil [56]. Nitrate leaching and runoff lead to severely deteriorated both ground and surface water quality levels and resulting in eutrophication and algal bloom. Severe cases of NO3 contamination in human is evident by Blue-Baby Syndrome [57] where NO3 molecules in drinking water combine with blood hemoglobin and hinder blood oxygen transportation [58]. Table 2 shows the major sources, amounts, and pathways of global reactive nitrogen (Nr) emission [30].

Table 2.
Major sources, amounts, and pathways of global reactive nitrogen (Nr) emission.
4. Mitigation Strategies
4.1. Farming System Design
To establish an effective and productive resource partitioning system, a collaborative organization of the agricultural production systems, which involves the adoption and eventual adaptation of a combination of a less expansive, minimal resource extraction crop-livestock production of high-value commodities, is required. The integrated production system (IPS) is not essentially a location restrained system, rather a synergistic approach towards agricultural resource utilization with minimal external input compared to intensive systems, and IPS helps materialize the concept of integrating several on-site farming components such as crop residues and animal waste [59,60]. A recent case study from Nigeria where herdsmen and farmers exchange resources, for instance, manure (collected by farmers) and crop stubble (used by herdsmen as cattle feed) exchange helped ease societal tension and offering a limited but promising solution to an on-site N loss solution [61]. Another example of IPS has been demonstrated in the Benin Republic, West Africa where a local farm (Songhai Farm) adopted all the IPS components and successfully collaborated between the different stages of agriculture such as production, selective breeding, harvesting, product processing, product placement in the local market, and finally on-site waste recycling to reuse nutrients [62]. Thus, IPS allows all participating parties alike to place a value on the on-site waste and help in sharing spatially separated resources.
4.2. On-Farm Best Available Techniques (BATs)
Preventing the loss of ammonia can be achieved by a combination of BATs such as placing N-fertilizers subsurface or injecting, applying urea fertilizer before rainfall events and after application irrigate the crops, applying N-fertilizers with acidifying agents such as elemental sulfur (not gypsum) or urease inhibitor, etc. [35,63]. Reducing nitrate loss through the soil profile, however, requires the adoption of a wide range of management strategies, for instance, split application of N-fertilizers according to crop nitrogen requirements and if applying animal slurries as N source then according to the crop demand, spraying nitrification inhibitor, maintain a constant vegetative cover, especially over the drainage period, cultivating soil in spring rather than in fall, etc. [64,65,66,67,68,69,70,71]. Many of the mitigation strategies adopted for N2O emission overlap with the strategies for mitigating NH3 volatilization and nitrate loss with a few exceptions, for instance, using lime to increase soil pH, avoiding anaerobic pockets in soil by maintaining optimum irrigation rate, and reduce animal traffic to avoid soil compaction [35,72].
4.3. Improved Nitrogen Use Efficiency (NUE)
Achieving nitrogen use efficiency (NUE) in crop and livestock production will involve implementing the best management practices (BMPs) leading to increase recycling within the system [73,74]. Some BMPs of NUE are discussed hereunder.
4.3.1. Enhanced Efficiency of Fertilizer Material
The use of enhanced efficiency fertilizers composed of coatings of low permeable materials attached to an inhibitor (nitrification or urease inhibitor) as an additive may be used to regulate processes such as nitrification or urea hydrolysis to simultaneously reduce the N loss and increase N uptake by plant and soil microbial population [75,76,77] under both laboratory and field setup. Such examples of inhibitors are N-(n-butyl) thiophosphoric triamide (NBPT), phenylphosphorodiamidate, dicyandiamide (DCD), and 3,4-dimethyl pyrazole phosphate (DMPP). For instance, the use of NBPT coated urea compared to uncoated-urea reduced NH3 emission by 42% for sunflower in Spain [78]; from 9.5% to 1.0% of applied N for winter wheat in Australia [79], and finally in the UK, for multiple grassland and winter cereal species [80]. In New Zealand, the volatilization of NH3 in grazing pastures was reduced through the application of NBPT (18%–28%) [81]. Moreover, the application of nitrification inhibitors such as DCD and DMPP in conjunction with urea significantly reduced N2O emission from agricultural soils of Louisiana, USA by more than 76% and 67%, respectively [82].
4.3.2. Site-Specific Nutrient Management (SSNM) and Real-Time Nitrogen Management (RTNM)
Crops typically respond to applied N in varying degrees and the fate of that applied N depends vastly on the soil conditions. The traditional broad-spectrum blanket-recommendation or “one size fits all” often fails to address crop N requirements and cannot surveil N availability from all possible sources leading to severe economic and environmental drawbacks. A possible solution could be the site-specific nutrient management strategy (SSNM). The SSNM considers several factors in production agriculture, for instance, yield potential of the crop, plant nutrition, inherent soil capability to supply N, calibrated N dosage, and subsequent N recovery calculations [22]. The SSNM addresses the crop-specific nutritional needs, utilizes maximum available resources to obtain N, calculates the gap of N required to fill the nutrient deficit gap, and finally recommends optimum N application recommendation [83]. Contrary to the blanket application of N, split applications according to the crop requirements, based on the growth stage, could prove to be an important strategy to enhance N recycling in soil, minimize loss of N as NH3 and N2O. Without the destructive collection of samples, sensor-based tools have the potential to identify and correct N stress, which has already occurred during the growing season for plant production [84]. Normalized difference vegetative index (NDVI) data of the greenness of plant leaf and previous crop yield and N application data can be used to implement the splits and may lead to an increase in nitrogen use efficiency and N recovery by the plant [22]. Studies on wheat (Triticum aestivum L.) produced primarily on the Central Great Plains of the United States showed that sensor data converted to NDVI was used to formulate a response index and showed a 15% greater NUE compared to whole-field techniques [85]. An emerging technology for site-specific N application for maize is real-time fluoro-sensing for variable-rate nutrient management, especially at earlier growth stage (V2) of the plants [86]. Moreover, proximal sensor-based variable rate N management strategy was reported to achieved greater grain yield and improved NUE for maize in Colorado, USA [87]. Similarly, in the UK, applications based on real-time sensor data saved 15 kg N ha−1 that increased the NUE without any reduction in wheat grain yield [88]. In a five-year study on N recovery, approximately 25–50 greater N was recovered with RTNM in maize cropping over-scheduled N application in Punjab, India [89]. In Guangxi Zhuang, China, NUE in rice (Oryza sativa) was 75% higher in densely planted reduced and delayed N applied rice plants compared to conventional farmers practice (broadcast) [90].
4.3.3. Deep Placement of Urea Super Granules
If there is an anoxic layer overlying a reduced zone in the soil, which is very typical of low-lying flooded rice-production systems, N loss can be encouraged by nitrification and denitrification, simultaneously [22]. Deep placement of nitrogenous fertilizers, for instance, large urea granules in rice-fields has successfully prevented the conversion of NH4 to NO3 and subsequent losses. Early studies in the 90s’ showed that deep placement of urea reduced N loss by 65% and resulted in a greater rice yield by 50% in grain yield compared to split application of granulated urea [91]. Recent studies also showed that NH3 and N2O loss were significantly suppressed by 94% with the deep placement of large urea granules (≥0.7 g) in rice-growing areas [92,93]. Furthermore, policymakers across Europe have indicated a relatively new method termed as “Closed-Slot Injection Method” to reduce NH3 emissions with inorganic fertilizers or organic inputs and this technique has proven promising due to the wide-spread availability among European farmers. For instance, NH3 loss was reduced in maize by 75% for mineral fertilizer and 96% for organic amendments compared to surface broadcast [94].
4.4. Pasture and Livestock Management
Reducing N2O emissions while keeping the ruminant population at the higher end of the production spectrum if not decreasing, requires either a top-quality ruminant diet or improved yield, thus, making this a livestock nutrition issue. Ruminant excretion quality is largely dependent upon ingested feed quality and depending on soil factors such as soil moisture and temperature, the N-component of the excretion may be subject to significant losses, thus, causing environmental hazards, such as increased nitrous oxide emission and greater nitrate loss [95,96]. Apart from reducing P loss to runoff [97], to facilitate enhanced nitrogen mineralization and to reduce the loss of NO3 in the soil, strategic cattle grazing in pastures along with overseeding of susceptible areas with annual grasses may offer a sustainable solution in livestock management as opposed to traditional grazing [72]. Additional approaches, such as supplementation of animal feed and anaerobic digestion may help increasing nutrient use efficiency by recycling nutrients on-site [98]. These management strategies are particularly important for developing countries like China, India, and Brazil that are major players in the beef and dairy industries [99] where ruminant feed is mostly associated with inferior quality of feed leading to a low-efficiency animal diet [100]. On the other hand, adopting alternative approaches, such as rearing mono-gastric non-ruminants, for instance, poultry, rabbit, swine, and horses (which are typically on a better and balanced diet than beef and dairy cattle) may potentially lead to efficient nutrient use and lower nitrogen loss [98]. Additionally, supplementing animal feed with amino acids such as lysine has the benefits of reduced N loss from swine and poultry by 30% compared to traditional feeding routine [101]. In managing pasture soil health, alternative ways of adding N have been investigated. In addition to adding organic fertilizers such as poultry [102] in managed pastures, using N-fixing legumes to supply soil N is an ecologically safe practice [103]. For instance, in long term pastures in Australia and New Zealand, and organic farming in the UK, white clover (Trifolium repens) is grown with other grass species and the fixed N is released slowly to the grass once released into the soil through root exudates and dead legume tissue [104,105,106]. In more recent studies on drought-tolerant forages, inter-seeding alfalfa into established Old World bluestem (OWB) grass helped restore soil health and enhance soil microbial community complexities [107].
4.5. Managing Livestock Wastewater
On-site livestock wastewater may be managed with the use of microalgae in the wastewater where livestock feces and urine (high N content). Due to its high concentration of N content, this animal waste tends to produce pollutants such as NH3 and N2O, which are essential for microalgal growth [108]. In addition to inorganic P, microalgae are known to assimilate inorganic N species and transform them into organic nitrogenous compounds [109]. Nitrogen oxide emissions from the wastewater, for instance, were reduced by 80% with Chlorella sp. [110] and entirely with Gracilaria birdiae (red seaweed) [111]. Seaweed can reduce N2O emissions by assimilating and storing N in high concentration [112] and decreasing available NOx in the wastewater system [113].
4.6. Carbon-Rich Sources
In terms of regulating N loss or impacting the N cycle, manure or litter broadcast leads to enhanced N2O emission [114], whereas, the sole application of biochar or a combination of lime and biochar (livestock slurry) showed a significant reduction in NH3 emissions [115] and cumulative N2O loss [116]. In production agriculture, especially, where litter or manure is used, ammonia is the precursor of nitrous oxide emission and volatilized ammonia can travel up to 5 km from its source of origin and a minor portion (1% proportion) has the potential to be re-emitted as nitrous oxide upon redeposition [117]. The application of biochar has been shown to reduce the emission of nitrous oxide from redeposited ammonia by 69% and this was attributed to the increased aeration caused by the inherent porosity of the biochar [118]. Similar high C sources, obtained as an agro-industrial byproduct, known as char (contains 30% of total C) have shown a reduction in NH3 volatilization up to 37% under fertilized soil compared to control [119]. In current agronomic practices, sewage sludge is commonly used, however, its use is associated with a greater risk of toxic substance accumulation, for instance, heavy metals, polycyclic aromatic hydrocarbons, or polychlorinated biphenyls, phenanthrene, and pyrene [120,121]. Furthermore, upon application, sewage sludge may phyto-accumulate and can be trophic transferred in agroecosystem food webs [122]. However, in recent years, the use of sewage sludge in conjunction with carbon adsorbents like activated carbon or biochar is gaining popularity both in terms of environmental safety and soil health [123,124,125]. In a recent study, conjunctive use of sewage sludge and biochar as soil amendments showed a dramatic decrease in sludge toxicity shortly after application due to nutrient immobilization and a significant reduction in nitrate loss from the soil profile [126]. Therefore, in addition to the phyto-stabilization of heavy metals [127], the use of high C sources in regulating N loss is a potentially promising avenue for further research.
4.7. Engineering Cereal Crops for Nitrogen Fixation
For the last five decades, scientists and agronomists have been studying the prospects of N fixation in cereal crops and evidently, there has been tremendous progress in areas like the expression of nitrogenase gene in eukaryotes and the nodule formation workflow in plants [128]. These efforts have led to investigate the scope of engineering cereal crops to perform symbiotic or autonomous N fixation, however, several factors, namely, population growth and a high rate of N application in production agriculture can outwit the constant efforts of developing N fixing transgenic cereals [129]. The primary tenet behind expressing nitrogenase in cereals is to re-envisage the legume-rhizobia symbiosis leading to nodule formation. The presence of building blocks for nodule formation in cereals is indicated by the presence of several common plant hormones [130,131].
Furthermore, Nod factors (legume-rhizobia symbiosis) and Myc factors (cereals and arbuscular mycorrhizal symbiosis) are structurally similar, indicating the possibilities, although abstract but promising, to engineer cereal crops to express Nod factors and initiate the first step toward nodulation [130,132,133]. On the contrary, considering the technical challenges of engineering cereals crops for nitrogen fixation, a sustainable and potential alternative approach such as root-associated diazotrophs in fixing and supplying N to cereals may offer a solution in N management with a short turnover time [134,135]. Although less sensitive than the legume-rhizobia symbiosis, nevertheless, the association between cereal crops and the rhizosphere shares a sophisticated signal route between microbes and plant host [136,137,138,139]. A recent ground-breaking study from the N depleted areas of Oaxaca, Mexico, showed mucilage associated with the aerial roots of Sierra Mixe maize can colonize free-living diazotrophic bacteria and the estimated up to 82% of the nitrogen content of maize [140]. The known association between plant and diazotrophs may improve the growth and yield of cereals in low N soils but the performance of these microbial strains is often not reproducible in the field [141,142].
4.8. Plant Growth Promoting Microbial Consortia
Soil microbes are key players in organic matter decomposition, macro, and micronutrient cycling, and facilitating nutrient availability for plant uptake [143,144]. Microbial communities associated with plants are also capable of abating environmental pollution [145]. Rhizosphere dwelling microbes are also known as plant growth-promoting microorganisms (PGPMs) [146] because they encourage plant growth and foster soil health by N fixation, P solubilization, mineralization of macro (calcium, magnesium, and potassium)/micronutrients (zinc, iodine, and nickel) and secreting phytohormones, and finally suppressing pathogens [147,148]. Additionally, animal wastes like poultry litter or composted poultry litter is a major source of organic N in production agriculture but poultry manure can cause a high loss of nitrogen via ammonia volatilization [149]. Therefore, farmers, industries, and researchers are giving considerable attention to the formulation of these microbial communities on-farm or off-site and using them to fortify the resilience of the agroecosystems. However, the function and propagation of these concocted exogenous microbes may be limited to their ability to survive under a highly diverse, competitive, and constantly changing medium such as soil [150,151]. In Southeastern USA, recent research on the production and application of locally derived exogenous microorganisms in managing nutrient availability from animal waste and utilizing them in nutrient cycling, especially in N-cycling, has shown higher potential nitrogen mineralization in soil 0–5 and 0–15 cm depth indicating a more robust and complex microbial community composition [152,153,154]. Additionally, microbially mediated decomposition of nitrogenous compounds in soil may enhance the N availability and consequently be intercepted by plant roots and taken up [155].
4.9. Phytogenic Approach and Fungal Utilization
As discussed above, nutrients excreted by livestock reflect the diet consumed and are an indicator of N, P, and CH4 release into the environment. Tannins and saponin-rich plants such as Acacia mearnsii, Delonix regia, Enterolobium cyclocarpum, and Musa paradisiac may be used as animal feed supplementation to enhance N retention [156,157,158,159]. For instance, alfalfa silage has a higher crude protein and nitrate content, which upon feeding may lead to higher N2O emission as opposed to feeding a combination of corn silage and grass hay [160]. In some countries in Asia, for instance, China and India, rice and wheat straws are burned, thus, causing environmental havoc like, smog. However, inoculating the straws with fungal species like Aspergillus terreus may reduce the lignocellulose content and enhance the decomposition process by soil microbes, which in turn enhance the nitrogen mineralization process [98].
4.10. Organic Agriculture as a Tool in Nitrogen Pollution Remediation
The history of organic agriculture is somewhat contentious and at the turn of the 21st century, some critics portrayed organic farming as an ideologically driven inefficient food production system [161,162,163]. Nonetheless, globally, the organic farming community experienced a continuous increase in the number of organic farms, acreage of land, the consumer market for organic foods, and organic agriculture-focused research funding [164]. As of now, more than 160 countries practice organic farming and more countries continue to join the community [164]. Worldwide, nearly 2.3 million organic farmers are growing organic produce in 0.99% of the total cultivable land [165] (Table 3).

Table 3.
Regional distribution of land area (Mha and percent) under organic agricultural land.
There are some intrinsic weaknesses in organic farming practices, especially, in the timely release of nutrients that coincide with plant N demand from typical organic amendments such as compost [166,167] and consequent potential higher nutrient loss mainly as nitrate [168]. Nitrate leaching losses have been reported to be similar or minutely lower in conventional agriculture compared to organic practices in several studies [169]. Nevertheless, organically managed agricultural systems can potentially contribute to the mitigation of climate change through efficient nutrient management techniques leading to reduced emission of nitrogenous gases from the production system and sequester carbon in the soil. Probably the most powerful aspect of organic agriculture, especially in developing countries, is its capacity to compete and often attain equal or higher yields as compared to traditional farming practices [170]. Organic farming practices have been shown to safeguard water quality in rivers as well. In a simulation study, a researcher has shown that a combined effect of higher precipitation and ethanol production from common biofuel crops could cause the river N level to rise by 24%, whereas, simple practices that are common to organic farming such as cover cropping, use of legumes in crop rotations, etc., could decrease the river N level by 7% [171]. Organic cropping systems to succeed, a few factors need to be considered, for instance, the synchronicity between crop N demand and N delivery from animal waste, greater flexibility in designing crop rotations, pretreating the compost with microbial inoculum to facilitate greater and rapid N availability from compost for plant uptake, etc. [154,169].
4.10.1. Limited External Input
In organic agriculture, inputs such as synthetic fertilizers, chemical pesticides, and herbicides are strictly restricted, hence, external energy for the chemical synthesis of nitrogen or phosphorus fertilizer is not required. In a conventional wheat-growing system, typically, 56% of the energy burden falls on chemical fertilizers and 11% on pesticides [172], thereby, increased the chances of nutrient loss. Although, organic agriculture avoids this requirement of energy but often highly dependent on the use of fossil fuels, especially in mechanical weed management. A study in the UK compared crops grown on seven conventional and organic farms and realized that although there is a higher energy demand of machinery to produce foods, the energy balance still tilted toward energy savings in organic farms (indicated by a 15% lower energy demand) gained by waiving the use of synthetic fertilizers and pesticides [173].
4.10.2. Crop Diversification
Cropping of diverse assemblages of local plant varieties fosters resilience in agroecosystems to counter sudden environmental stresses such as droughts and economic volatility such as price variations [174]. Additionally, cropping diversity encourages the efficient use of soil nutrients and optimum yield [175].
4.11. Ecological Ditch
In addition to N2O emission and NH3 volatilization from agricultural fields, a major source of nitrogen pollution is agricultural drainage [176,177]. Typically, the compositional nature of agricultural runoff is complex due to the sheer number and types of nutrients, for instance, runoff often contains nitrate, ammonium, inorganic phosphorous, organic pollutants, and heavy metals [178]. These N and phosphorus (P) nutrients play a crucial role in the growth of aquatic plants, which upon a lack of regulation can lead to eutrophication in the downstream receiving aquatic systems [179]. Heavy nutrient loads from agricultural lands can potentially cause eutrophication, hypoxia, and ecological damages in nearby water bodies [180].
The “ecological ditch” (eco-ditch) is an effective component in alleviating non-point agricultural pollution. Eco-ditches are examples of best management practices and a stark contrast to the traditional agricultural drainage ditches. Eco-ditches create an exclusive ecosystem where the participating parties are aquatic plants and associated microbial communities fueled by the constant nutritional substrates [181]. Eco-ditches are designed to absorb nutrients that are otherwise lost through surface runoff and make those nutrients available for root uptake or be incorporated into microbial metabolites [182,183]. Eco-ditches designed with Leersia oryzoides and Typha latifolia reduced the load of inorganic N from 2.5% to 1.5%, accounting for more than 50% of the total reduction over 2 years in Northern Mississippi [184]. However, in designing eco-ditches, plant population diversity needs greater attention and highly efficient ditch plants should be selected. One possible constraint of these ditches could be the variability in nutrient removal capacity by plants, which is strictly dependent upon the growth stages of plants. For instance, during the growing period, plants tend to uptake more nutrients as opposed to the senesce period [185]. Plant harvesting continuously in the eco-ditches may offer a possible solution [186].
4.12. Genetic Improvement
4.12.1. Identifying Candidate Gene in Plants for Improved NUE
Nitrogen use efficiency of plants is genetically regulated; thus, nutrient use varies vastly among plant species. These variations lead to differences in several aspects in different plants including N assimilation, uptake, and remobilization capability, hence, alludes to the need to screen for potential genetic traits across these genotypes.
- a.
- Differentially expressed genes (DFEs) to validate their roles in NUE of different genotypes of crop species can be profiled globally for different genotypes under different N treatments. DFEs have four components and these are:
- i.
- Hybridization based transcriptome analysis to identify differentially expressed traits with low abundance [187];
- ii.
- Analyzing short sequence tags of individual mRNA and then linked to form long sequences and finally cloning them [188];
- iii.
- Probe-targeted hybridization of immobilized cDNA molecules to generate a large amount of data and analysis of the whole genome [189];
- iv.
- RNA sequencing involves the sequencing of every RNA molecule and subsequently profiling a particular gene expression [190].
- b.
- Functional validation of genes by mutation and transgenic studies.
Both the mutant population and natural variants can be studied to identify genes of interest in crop NUE. The steps involve propagation of the mutated population and screening for mutated phenotypes, finally followed by gene recovery through map-based cloning strategies.
4.12.2. Discovery of Genes by Mapping Studies
Biomarker-based mapping studies by biparental linkage analysis and association studies in naturally existing genotypes are two possible strategies in identifying the position of the NUE genes in crops. In low and optimum N systems, a meta-analysis of the quantitative trait loci (QTLs) for yield was mapped to discover linked markers with the gene that controls the specific trait, which in this case was N use and the study revealed a total of 22 meta-QTLs under low N [191]. Additionally, another association study in 196 accessions of wheat for yield components expressed 23 N-responsive regions, which can be exploited by breeders to develop highly N responsive varieties of wheat [192].
In the coming decades, one of the greatest challenges humanity faces is climate change, and agriculture is both a key contributor to crisis and will be immensely impacted by this problem [193]. Thus, minimizing the loss and emission of reactive N is crucial in slowing down the rate of climate change [194,195] (Table 4).

Table 4.
Mitigation strategies to prevent potential N loss.
Adapting to a combination of these mitigation strategies will enable individual growers and the farming community at large. For instance, agronomic approaches such as formulating microbial inoculum from a local source (discussed in Section 4.8) are affordable and particularly is very important for agriculture in developing countries, while molecular techniques such as N-fixing cereal crops, NUE gene identification, and mapping (discussed in Section 4.7 and Section 4.12) are time-consuming and requires a long-term research investment. Nonetheless, policy-driven and ecosystem service-oriented mitigation strategies will help combat future N losses from agriculture and offer ecological safeguard.
5. Nitrogen Status in Agriculture during the COVID-19 Pandemic
On 11 March 2020, the World Health Organization (WHO) declared that the world faces a pandemic by the novel coronavirus (COVID-19) [196,197]. The tremendous disruptions across the globe caused by the COVID-19 pandemic are affecting the entire realm of human activities. A recent study investigating the relationship between long-term exposure to NO2 and coronavirus mortality through the analysis of tropospheric NO2 mapping and distribution data generated by the Sentinel-5P satellite showed that 78% of death cases were in five regions located in Northern Italy and Central Spain that displayed the highest NO2 concentration levels and low circulation of air to disperse the pollution. These findings suggest that long-term exposure to this pollutant may be a major contributor to COVID-19 death in these regions and possibly in other parts of the world [198]. However, the COVID-19 outbreak and consequent social distancing activities led to an extensive decline in traffic and allowed comparisons of air quality during and before the decline to document the impacts of COVID-19 on NO2 concentration in Florida Counties through March 2020. The results indicated a 54.07% decrease in NO2 in the atmosphere [199]. In the context of agricultural safeguard, According to World Economic Outlook, the emerging and developing nations will face extreme severity of negative growth [200], and according to the Food and Agriculture Organization (FAO) and World Food Program (WFP), there will be food insecurity at an unprecedented level [201]. In this time of global crisis, now more than ever, soil plays a pivotal role as production agriculture and bedrock of resilience in food security [202]. The impact of this global pandemic on agriculture will likely be learned in waves in the coming years. Assessing the current soil N, carbon (C), and P content as impacted by the pandemic for devising future strategies to recover from the pandemic and establish long-term sustainable goals to maintain soil health for future needs is very crucial. Likely lower livestock production and with lower fertilizer application due to the COVID-19 restrictions, global agriculture may see improved farm management scenarios resulting in reduced GHGs, like, N2O, lower NH3 emissions, and low nutrient loss to surface water. For instance, there have been limited agricultural activities in livestock production that are being reflected in the reduced greenhouse gas emission such as NOx in countries such as China and Italy; however, the same study also concluded that agricultural pollution via NH3 emission has not changed significantly compared to the pre-pandemic era [203]. This may consequently result in improved ecosystem services and higher food quality. A closer look at the examination of macronutrients such as N and P is crucial, where N limits crop growth and P fertilization plays a crucial role in crop yield. However, during this pandemic, due to lockdown, food supply chains have been massively disrupted on a regional and global scale, thus, raising the most imminent threat from the huge addition of organic waste as mass disposal from the dairy and vegetable industry. Additionally, reduced meat consumption in the USA during the earlier months of 2020 has led to the massive burial of swine and poultry in many parts the country [202]. Such an influx of surplus organic matter to the soil, especially, P in the organic matter, may result in an imbalanced soil nutrient status. Therefore, for future crop fertilization strategies, application based on N requirements of the plant may result in over-application of P, which in turn may be fixed in the soil–mineral complex, making the soil depleted in plant-available P. In this context, the long-term consequences of the massive burial of organic waste may introduce additional complication is land use, surface, and groundwater quality, soil macro and microfauna and flora diversity, critical ecosystems services, and human well-being.
6. Knowledge Gaps and Questions
The agricultural system is complex and often the feedbacks and processes are non-linear. Nitrogen loss mitigation strategies require sustainable, resilient, and redundant ways of producing and consuming that can also be adopted in climate change strategies in a broader sense [204]. Immediate attention is required to explore and interpret data from low-income countries in this regard. Although, the greatest emission of GHGs such as N2O is estimated to be higher in the low to middle-income parts of the world from food waste there exists close to no empirical studies on how to tackle these challenges [205,206]. The problem is further complicated as the mean income in these countries increases, the dietary habits change. High protein and carbohydrate diets lead to intense livestock and cereal production systems ensuing in greater GHGs emissions [207]. However, climate implication is yet to be explored in those countries as compared to developed parts (high-income) of the globe [208]. Another aspect of identifying and abating N loss is investing resources into research on post-harvest management of crops and crop residues [209]. Inclusive public policies and equitable funding mechanisms that are equitable and resilient are the two major catalysts that will lead to lower agricultural system N footprints. A major shortfall in global climate change strategies is the lack of financial allocation for tackling emission problems, especially in developing countries, and uncertainties shrouded by the lack of research to indicate whether the allocated money is being used for climate-smart agriculture. For instance, in 2015, out of $391(US) billion only $8 (US) billion was issued to address and adopt strategies to mitigate climate strategies like nitrogen use efficiency, soil health, and GHGs emission mitigation [210]. Furthermore, assigning sustainability indexes to agricultural commodities and assimilating sustainability benchmarks in dietary intake guidelines may lead to change in dietary regimens fostering healthy and low N footprint foods and low emission diets [211,212]. Countries such as Brazil, Germany, Qatar, and Sweden have taken steps towards this goal already, and the USA has thus far failed to adopt [213]. Government policies now must realize that voluntary measures are not adequate in lowering N footprints at farm level production, rather strict regulation, financial incentives, and subsidies for regenerative agriculture are obligated [214]. Now, the world is faced with an entirely new challenge of the COVID-19 pandemic, and the existing soil and agricultural management strategies must include the complex task of including the COVID-19 pandemic as a variable along with the existing ones, for instance, climate change, food insecurity, freshwater crisis, and continuously endangered and fading biodiversity. A radical shift from the dependencies on industrialized mono-agriculture to more diverse agroecology may be required in the coming days as the global population traverse through this pandemic.
7. Conclusions and Future Directions
The nitrogen challenge at its core requires societal recognition and indicates a potential opportunity to steer away from a fragmented policy approach, rather than toward rapid solutions [215]. Addressing the loss of N in agricultural production and proposing possible mitigation strategies should aim to be inclusive, realize the current shortcomings, and most importantly should be realistic to attain. From simple measures such as, the inclusion of nitrogen-fixing legumes in production agriculture as cover crops [216] to more complicated efforts that aim to materialize artificial symbioses or associative nitrogen fixation in non-legume plants, especially in cereals [128] are some of the possible ventures that may be undertaken. Moreover, undertaking efficient N management measures, for instance, controlled release of N or drip N fertilization in rice and maize, respectively, can direct the cost–benefit balance to lean toward profit [217,218]. Additionally, 4R (Right Source, Right Rate, Right Time, and Right Placement) N guidelines for corn are extremely profitable (40% increase) while decreasing the N application rate (21% reduction) [219]. Thus, connecting socioeconomic requirements with landscape potentials should be the central aspect in future N loss-related plans and policies [220]. Another key element to be explored is soil resilience and should be implemented wherever possible. Resilience will enable degraded or depleted soil to recover and possibly stop the soil from being a sink but a source. In the coming days, the soil should not be discussed as an isolated component rather it should be thought of as an essential tool of regenerative agriculture where possible answers to pollution questions can be sought after. Including soil as a resource in strategizing grass-root policies may reduce the risk of market vulnerabilities and associated risks. Finally, to summarize, while policy and funding apparatuses have been proposed amply but few have been implemented to address and mitigate the global N pollution status for regenerative and sustainable agriculture.
Author Contributions
Conceptualization, K.M.; writing—original draft preparation, K.M., D.P., A.M. (Anaas Mergoum) and A.M. (Ali Missaoui); writing—review and editing, A.M. (Anaas Mergoum), K.M., D.P. A.M. (Ali Missaoui); supervision, A.M. (Ali Missaoui); All authors have read and agreed to the published version of the manuscript.
Funding
Partial funding was provided by The Center for Bioenergy Innovation, a US Department of Energy Research Center supported by the Office of Biological and Environmental Research in the DOE Office of Science, for salary support of K.M.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Grafton, R.Q.; Daugbjerg, C.; Qureshi, M.E. Towards food security by 2050. Food Secur. 2015, 7, 179–183. [Google Scholar] [CrossRef]
- Rodriguez, A.; Sanders, I.R. The role of community and population ecology in applying mycorrhizal fungi for improved food security. ISME J. 2015, 9, 1053–1061. [Google Scholar] [CrossRef]
- FAO. Food Wastage Footprint: Impacts on Natural Resources; Summary Report; FAO: Rome, Italy, 2013. [Google Scholar]
- Gruber, N.; Galloway, J.N. An Earth-system perspective of the global nitrogen cycle. Nature 2008, 451, 293–296. [Google Scholar] [CrossRef]
- Pathak, H.; Jain, N.; Bhatia, A.; Kumar, A.; Chatterjee, D. Improved nitrogen management: A key to climate change adaptation and mitigation. Indian J. Fertil. 2016, 12, 151–162. [Google Scholar]
- Houlton, B.Z.; Almaraz, M.; Aneja, V.; Austin, A.T.; Bai, E.; Cassman, K.G.; Compton, J.E.; Davidson, E.A.; Erisman, J.W.; Galloway, J.N. A world of cobenefits: Solving the global nitrogen challenge. Earth’s Future 2019, 7, 865–872. [Google Scholar] [CrossRef]
- Xu, P.; Chen, A.; Houlton, B.Z.; Zeng, Z.; Wei, S.; Zhao, C.; Lu, H.; Liao, Y.; Zheng, Z.; Luan, S. Spatial Variation of Reactive Nitrogen Emissions from China’s Croplands Codetermined by Regional Urbanization and Its Feedback to Global Climate Change. Geophys. Res. Lett. 2020, 47, e2019GL086551. [Google Scholar] [CrossRef]
- Fageria, N.; Baligar, V. Enhancing nitrogen use efficiency in crop plants. Adv. Agron. 2005, 88, 97–185. [Google Scholar]
- Maharjan, B.; Venterea, R.T.; Rosen, C. Fertilizer and irrigation management effects on nitrous oxide emissions and nitrate leaching. Agron. J. 2014, 106, 703–714. [Google Scholar] [CrossRef]
- Mosier, A.; Kroeze, C.; Nevison, C.; Oenema, O.; Seitzinger, S.; Van Cleemput, O. Closing the global N2O budget: Nitrous oxide emissions through the agricultural nitrogen cycle. Nutr. Cycl. Agroecosyst. 1998, 52, 225–248. [Google Scholar] [CrossRef]
- Gao, W.; Yang, H.; Kou, L.; Li, S. Effects of nitrogen deposition and fertilization on N transformations in forest soils: A review. J. Soils Sediments 2015, 15, 863–879. [Google Scholar] [CrossRef]
- Fesenfeld, L.P.; Schmidt, T.S.; Schrode, A. Climate policy for short-and long-lived pollutants. Nat. Clim. Chang. 2018, 8, 933–936. [Google Scholar] [CrossRef]
- Liu, M.; Huang, X.; Song, Y.; Tang, J.; Cao, J.; Zhang, X.; Zhang, Q.; Wang, S.; Xu, T.; Kang, L. Ammonia emission control in China would mitigate haze pollution and nitrogen deposition, but worsen acid rain. Proc. Natl. Acad. Sci. USA 2019, 116, 7760–7765. [Google Scholar] [CrossRef]
- Venterea, R.T.; Halvorson, A.D.; Kitchen, N.; Liebig, M.A.; Cavigelli, M.A.; Grosso, S.J.D.; Motavalli, P.P.; Nelson, K.A.; Spokas, K.A.; Singh, B.P. Challenges and opportunities for mitigating nitrous oxide emissions from fertilized cropping systems. Front. Ecol. Environ. 2012, 10, 562–570. [Google Scholar] [CrossRef]
- Hoben, J.; Gehl, R.; Millar, N.; Grace, P.; Robertson, G. Nonlinear nitrous oxide (N2O) response to nitrogen fertilizer in on-farm corn crops of the US Midwest. Glob. Chang. Biol. 2011, 17, 1140–1152. [Google Scholar] [CrossRef]
- Shcherbak, I.; Millar, N.; Robertson, G.P. Global metaanalysis of the nonlinear response of soil nitrous oxide (N2O) emissions to fertilizer nitrogen. Proc. Natl. Acad. Sci. USA 2014, 111, 9199–9204. [Google Scholar] [CrossRef]
- Huddell, A.M.; Galford, G.L.; Tully, K.L.; Crowley, C.; Palm, C.A.; Neill, C.; Hickman, J.E.; Menge, D.N. Meta-analysis on the potential for increasing nitrogen losses from intensifying tropical agriculture. Glob. Chang. Biol. 2020, 26, 1668–1680. [Google Scholar] [CrossRef] [PubMed]
- Leach, A.M.; Galloway, J.N.; Bleeker, A.; Erisman, J.W.; Kohn, R.; Kitzes, J. A nitrogen footprint model to help consumers understand their role in nitrogen losses to the environment. Environ. Dev. 2012, 1, 40–66. [Google Scholar] [CrossRef]
- Erisman, J.W.; Galloway, J.N.; Seitzinger, S.; Bleeker, A.; Dise, N.B.; Petrescu, A.R.; Leach, A.M.; de Vries, W. Consequences of human modification of the global nitrogen cycle. Philos. Trans. R. Soc. B Biol. Sci. 2013, 368, 20130116. [Google Scholar] [CrossRef]
- Westhoek, H.; Lesschen, J.P.; Leip, A.; Rood, T.; Wagner, S.; De Marco, A.; Murphy-Bokern, D.; Pallière, C.; Howard, C.M.; Oenema, O. Nitrogen on the Table: The Influence of Food Choices on Nitrogen Emissions and the European Environment; NERC/Centre for Ecology & Hydrology: Edinburgh, UK, 2015. [Google Scholar]
- Westhoek, H.; Lesschen, J.P.; Rood, T.; Wagner, S.; de Marco, A.; Murphy-Bokern, D.; Leip, A.; van Grinsven, H.; Sutton, M.A.; Oenema, O. Food choices, health and environment: Effects of cutting Europe’s meat and dairy intake. Glob. Environ. Chang. 2014, 26, 196–205. [Google Scholar] [CrossRef]
- Mohanty, S.; Swain, C.K.; Kumar, A.; Nayak, A. Nitrogen Footprint: A Useful Indicator of Agricultural Sustainability. In Nutrient Dynamics for Sustainable Crop Production; Springer: Singapore, 2020; pp. 135–156. [Google Scholar]
- Oita, A.; Nagano, I.; Matsuda, H. An improved methodology for calculating the nitrogen footprint of seafood. Ecol. Indic. 2016, 60, 1091–1103. [Google Scholar] [CrossRef]
- Galloway, J.N.; Winiwarter, W.; Leip, A.; Leach, A.M.; Bleeker, A.; Erisman, J.W. Nitrogen footprints: Past, present and future. Environ. Res. Lett. 2014, 9, 115003. [Google Scholar] [CrossRef]
- Shibata, H.; Cattaneo, L.R.; Leach, A.M.; Galloway, J.N. First approach to the Japanese nitrogen footprint model to predict the loss of nitrogen to the environment. Environ. Res. Lett. 2014, 9, 115013. [Google Scholar] [CrossRef]
- Gu, B.; Leach, A.M.; Ma, L.; Galloway, J.N.; Chang, S.X.; Ge, Y.; Chang, J. Nitrogen footprint in China: Food, energy, and nonfood goods. Environ. Sci. Technol. 2013, 47, 9217–9224. [Google Scholar] [CrossRef] [PubMed]
- Gu, B.; Ju, X.; Chang, J.; Ge, Y.; Vitousek, P.M. Integrated reactive nitrogen budgets and future trends in China. Proc. Natl. Acad. Sci. USA 2015, 112, 8792–8797. [Google Scholar] [CrossRef]
- Gu, B.; Ge, Y.; Ren, Y.; Xu, B.; Luo, W.; Jiang, H.; Gu, B.; Chang, J. Atmospheric reactive nitrogen in China: Sources, recent trends, and damage costs. Environ. Sci. Technol. 2012, 46, 9420–9427. [Google Scholar] [CrossRef]
- Smil, V. Detonator of the population explosion. Nature 1999, 400, 415. [Google Scholar] [CrossRef]
- Oita, A.; Malik, A.; Kanemoto, K.; Geschke, A.; Nishijima, S.; Lenzen, M. Substantial nitrogen pollution embedded in international trade. Nat. Geosci. 2016, 9, 111–115. [Google Scholar] [CrossRef]
- San-Martín, W. Global Nitrogen in Sustainable Development: Four Challenges at the Interface of Science and Policy. Buildings 2012, 2, 300–325. [Google Scholar]
- Black, A.; Sherlock, R.; Cameron, K.; Smith, N.; Goh, K. Comparison of three field methods for measuring ammonia volatilization from urea granules broadcast on to pasture. J. Soil Sci. 1985, 36, 271–280. [Google Scholar] [CrossRef]
- Black, A.; Sherlock, R.; Smith, N.; Cameron, K.; Goh, K. Effects of form of nitrogen, season, and urea application rate on ammonia volatilisation from pastures. N. Z. J. Agric. Res. 1985, 28, 469–474. [Google Scholar] [CrossRef]
- McGarry, S.; O’Toole, P.; Morgan, M. Effects of soil temperature and moisture content on ammonia volatilization from urea-treated pasture and tillage soils. Ir. J. Agric. Res. 1987, 26, 173–182. [Google Scholar]
- Cameron, K.; Di, H.J.; Moir, J. Nitrogen losses from the soil/plant system: A review. Ann. Appl. Biol. 2013, 162, 145–173. [Google Scholar] [CrossRef]
- Acton, S.D. The Effect of Fertiliser Application Rate and Soil PH on Methane Oxidation and Nitrous Oxide Production; University of Aberdeen: Aberdeen, UK, 2007. [Google Scholar]
- Mundepi, A.; Cabrera, M.; Norton, J.; Habteselassie, M. Ammonia Oxidizers as Biological Health Indicators of Elevated Zn and Cu in Poultry Litter Amended Soil. Water Air Soil Pollut. 2019, 230, 239. [Google Scholar] [CrossRef]
- Cassity-Duffey, K.; Cabrera, M.; Franklin, D.; Gaskin, J.; Kissel, D. Effect of soil texture on nitrogen mineralization from organic fertilizers in four common southeastern soils. Soil Sci. Soc. Am. J. 2020, 84, 534–542. [Google Scholar] [CrossRef]
- Bouwman, A.; Boumans, L.; Batjes, N. Modeling global annual N2O and NO emissions from fertilized fields. Glob. Biogeochem. Cycles 2002, 16, 28-1–28-9. [Google Scholar] [CrossRef]
- Paulot, F.; Jacob, D.J.; Pinder, R.; Bash, J.; Travis, K.; Henze, D. Ammonia emissions in the United States, European Union, and China derived by high-resolution inversion of ammonium wet deposition data: Interpretation with a new agricultural emissions inventory (MASAGE_NH3). J. Geophys. Res. Atmos. 2014, 119, 4343–4364. [Google Scholar] [CrossRef]
- Sutton, M.A.; Erisman, J.W.; Dentener, F.; Möller, D. Ammonia in the environment: From ancient times to the present. Environ. Pollut. 2008, 156, 583–604. [Google Scholar] [CrossRef]
- Carlson, J.; Daehler, K.R. The refined consensus model of pedagogical content knowledge in science education. In Repositioning Pedagogical Content Knowledge in Teachers’ Knowledge for Teaching Science; Springer: Singapore, 2019; pp. 77–92. [Google Scholar]
- Pathak, H.; Bhatia, A.; Prasad, S.; Singh, S.; Kumar, S.; Jain, M.; Kumar, U. Emission of nitrous oxide from rice-wheat systems of Indo-Gangetic plains of India. Environ. Monit. Assess. 2002, 77, 163–178. [Google Scholar] [CrossRef] [PubMed]
- Caranto, J.D.; Lancaster, K.M. Nitric oxide is an obligate bacterial nitrification intermediate produced by hydroxylamine oxidoreductase. Proc. Natl. Acad. Sci. USA 2017, 114, 8217–8222. [Google Scholar] [CrossRef] [PubMed]
- Intergovernmental Panel On Climate Change. Climate change 2007: The physical science basis. Agenda 2007, 6, 333. [Google Scholar]
- Butterbach-Bahl, K.; Baggs, E.M.; Dannenmann, M.; Kiese, R.; Zechmeister-Boltenstern, S. Nitrous oxide emissions from soils: How well do we understand the processes and their controls? Philos. Trans. R. Soc. B Biol. Sci. 2013, 368, 20130122. [Google Scholar] [CrossRef]
- Syakila, A.; Kroeze, C. The global nitrous oxide budget revisited. Greenh. Gas Meas. Manag. 2011, 1, 17–26. [Google Scholar] [CrossRef]
- Sarabia, L.; Solorio, F.J.; Ramírez, L.; Ayala, A.; Aguilar, C.; Ku, J.; Almeida, C.; Cassador, R.; Alves, B.J.; Boddey, R.M. Improving the Nitrogen Cycling in Livestock Systems Through Silvopastoral Systems. In Nutrient Dynamics for Sustainable Crop Production; Springer: Singapore, 2020; pp. 189–213. [Google Scholar]
- World Health Organization. Air Quality Guidelines: Global Update 2005: Particulate Matter, Ozone, Nitrogen Dioxide, and Sulfur Dioxide; World Health Organization: Copenhagen, Denmark, 2006. [Google Scholar]
- Avol, E.L.; Gauderman, W.J.; Tan, S.M.; London, S.J.; Peters, J.M. Respiratory effects of relocating to areas of differing air pollution levels. Am. J. Respir. Crit. Med. 2001, 164, 2067–2072. [Google Scholar] [CrossRef] [PubMed]
- Bowatte, G.; Erbas, B.; Lodge, C.J.; Knibbs, L.D.; Gurrin, L.C.; Marks, G.B.; Thomas, P.S.; Johns, D.P.; Giles, G.G.; Hui, J. Traffic-related air pollution exposure over a 5-year period is associated with increased risk of asthma and poor lung function in middle age. Eur. Respir. J. 2017, 50, 1602357. [Google Scholar] [PubMed]
- Singh, B.; Sekhon, G. Nitrate pollution of groundwater from farm use of nitrogen fertilizers—A review. Agric. Environ. 1979, 4, 207–225. [Google Scholar] [CrossRef]
- Watt, W.; Tulinsky, A.; Swenson, R.P.; Watenpaugh, K.D. Comparison of the crystal structures of a flavodoxin in its three oxidation states at cryogenic temperatures. J. Mol. Biol. 1991, 218, 195–208. [Google Scholar] [CrossRef]
- Wild, A.; Cameron, K. Soil nitrogen and nitrate leaching. In Soils and Agriculture; Tinker, P.B., Ed.; Blackwell: Oxford, UK, 1980; pp. 35–70. [Google Scholar]
- Cameron, K.; Wild, A. 1984, 13, 274–278.
- Silva, R.; Cameron, K.; Di, H.; Smith, N.; Buchan, G. Effect of macropore flow on the transport of surface-applied cow urine through a soil profile. Soil Res. 2000, 38, 13–24. [Google Scholar] [CrossRef]
- Brender, J.D.; Weyer, P.J.; Romitti, P.A.; Mohanty, B.P.; Shinde, M.U.; Vuong, A.M.; Sharkey, J.R.; Dwivedi, D.; Horel, S.A.; Kantamneni, J. Prenatal nitrate intake from drinking water and selected birth defects in offspring of participants in the national birth defects prevention study. Environ. Health Perspect. 2013, 121, 1083–1089. [Google Scholar] [CrossRef]
- Knobeloch, L.; Salna, B.; Hogan, A.; Postle, J.; Anderson, H. Blue babies and nitrate-contaminated well water. Environ. Health Perspect. 2000, 108, 675–678. [Google Scholar] [CrossRef]
- Folke, C.; Kautsky, N.; Berg, H.; Jansson, Å.; Troell, M. The ecological footprint concept for sustainable seafood production: A review. Ecol. Appl. 1998, 8, S63–S71. [Google Scholar] [CrossRef]
- Little, D.; Edwards, P. Integrated Livestock-Fish Farming Systems; Food & Agriculture Organization: Rome, Italy, 2003. [Google Scholar]
- Sutton, M.A.; Bleeker, A.; Howard, C.; Erisman, J.; Abrol, Y.; Bekunda, M.; Datta, A.; Davidson, E.; De Vries, W.; Oenema, O. Our Nutrient World. The Challenge to Produce More Food & Energy with Less Pollution; Centre for Ecology & Hydrology: Edinburgh, UK, 2013. [Google Scholar]
- FAO. FAOSTAT Online Statistical Service; Food and Agriculture Organization of the United Nations (FAO): Rome, Italy, 2009. [Google Scholar]
- Horneck, D.A.; Sullivan, D.M.; Owen, J.S.; Hart, J.M. Soil Test Interpretation Guide; Oregon State University: Corvallis, OR, USA, 2011. [Google Scholar]
- Di, H.; Cameron, K. Nitrate leaching in temperate agroecosystems: Sources, factors and mitigating strategies. Nutr. Cycl. Agroecosystems 2002, 64, 237–256. [Google Scholar] [CrossRef]
- Dungait, J.A.; Cardenas, L.M.; Blackwell, M.S.; Wu, L.; Withers, P.J.; Chadwick, D.R.; Bol, R.; Murray, P.J.; Macdonald, A.J.; Whitmore, A.P. Advances in the understanding of nutrient dynamics and management in UK agriculture. Sci. Total Environ. 2012, 434, 39–50. [Google Scholar] [CrossRef] [PubMed]
- Goulding, K.; Jarvis, S.; Whitmore, A. Optimizing nutrient management for farm systems. Philos. Trans. R. Soc. B Biol. Sci. 2008, 363, 667–680. [Google Scholar] [CrossRef] [PubMed]
- Haynes, R. Mineral Nitrogen in the Plant-Soil System; Elsevier: Orlando, FL, USA, 2012. [Google Scholar]
- Kay, P.; Edwards, A.C.; Foulger, M. A review of the efficacy of contemporary agricultural stewardship measures for ameliorating water pollution problems of key concern to the UK water industry. Agric. Syst. 2009, 99, 67–75. [Google Scholar] [CrossRef]
- Monaghan, R.; De Klein, C.A.; Muirhead, R.W. Prioritisation of farm scale remediation efforts for reducing losses of nutrients and faecal indicator organisms to waterways: A case study of New Zealand dairy farming. J. Environ. Manag. 2008, 87, 609–622. [Google Scholar] [CrossRef] [PubMed]
- Monaghan, R.; Hedley, M.; Di, H.; McDowell, R.; Cameron, K.; Ledgard, S. Nutrient management in New Zealand pastures—recent developments and future issues. N. Z. J. Agric. Res. 2007, 50, 181–201. [Google Scholar] [CrossRef]
- Munoz, F.; Mylavarapu, R.; Hutchinson, C. Environmentally responsible potato production systems: A review. J. Plant Nutr. 2005, 28, 1287–1309. [Google Scholar] [CrossRef]
- Dahal, S.; Franklin, D.; Subedi, A.; Cabrera, M.; Hancock, D.; Mahmud, K.; Ney, L.; Park, C.; Mishra, D.J.S. Strategic Grazing in Beef-Pastures for Improved Soil Health and Reduced Runoff-Nitrate-A Step towards. Sustainability 2020, 12, 558. [Google Scholar] [CrossRef]
- Giller, K.E.; Chalk, P.; Dobermann, A.; Hammond, L.; Heffer, P.; Ladha, J.K.; Nyamudeza, P.; Maene, L.; Ssali, H.; Freney, J. Emerging technologies to increase the efficiency of use of fertilizer nitrogen. Agric. Nitrogen Cycle Assess. Impacts Fertil. Food Prod. Environ. 2004, 65, 35–51. [Google Scholar]
- Freney, J.R. Management practices to increase efficiency of fertilizer and animal nitrogen and minimize nitrogen loss to the atmosphere and groundwater. Tech. Bull. Food Fertil. Technol. Cent. 2011, 186, 22. [Google Scholar]
- Soares, J.R.; Cantarella, H.; de Campos Menegale, M.L. Ammonia volatilization losses from surface-applied urea with urease and nitrification inhibitors. Soil Biol. Biochem. 2012, 52, 82–89. [Google Scholar] [CrossRef]
- Verma, J.P.; Jaiswal, D.K.; Meena, V.S.; Kumar, A.; Meena, R. Issues and challenges about sustainable agriculture production for management of natural resources to sustain soil fertility and health. J. Clean. Prod. 2015, 107, 793–794. [Google Scholar] [CrossRef]
- Mitran, T.; Meena, R.S.; Lal, R.; Layek, J.; Kumar, S.; Datta, R. Role of soil phosphorus on legume production. In Legumes for Soil Health and Sustainable Management; Springer: Singapore, 2018; pp. 487–510. [Google Scholar]
- Sanz-Cobena, A.; Misselbrook, T.H.; Arce, A.; Mingot, J.I.; Diez, J.A.; Vallejo, A. An inhibitor of urease activity effectively reduces ammonia emissions from soil treated with urea under Mediterranean conditions. Agric. Ecosyst. Environ. 2008, 126, 243–249. [Google Scholar] [CrossRef]
- Turner, D.; Edis, R.; Chen, D.; Freney, J.; Denmead, O.; Christie, R. Determination and mitigation of ammonia loss from urea applied to winter wheat with N-(n-butyl) thiophosphorictriamide. Agric. Ecosyst. Environ. 2010, 137, 261–266. [Google Scholar] [CrossRef]
- Chambers, B.; Dampney, P. Nitrogen efficiency and ammonia emissions from urea-based and ammonium nitrate fertilisers. In Proceedings of Proceedings-International Fertiliser Society; Internationl Fertiliser Society: York, UK, 2009. [Google Scholar]
- Rodriguez, M.J.; Saggar, S.; Berben, P.; Palmada, T.; Lopez-Villalobos, N.; Pal, P. Use of a urease inhibitor to mitigate ammonia emissions from urine patches. Environ. Technol. 2021, 42, 20–31. [Google Scholar] [CrossRef]
- Meng, Y.; Wang, J.J.; Wei, Z.; Dodla, S.K.; Fultz, L.M.; Gaston, L.A.; Xiao, R.; Park, J.-h.; Scaglia, G. Nitrification inhibitors reduce nitrogen losses and improve soil health in a subtropical pastureland. Geoderma 2021, 388, 114947. [Google Scholar] [CrossRef]
- Dobermann, A.; Witt, C.; Dawe, D.; Abdulrachman, S.; Gines, H.; Nagarajan, R.; Satawathananont, S.; Son, T.; Tan, P.; Wang, G. Site-specific nutrient management for intensive rice cropping systems in Asia. Field Crops Res. 2002, 74, 37–66. [Google Scholar] [CrossRef]
- Tagarakis, A.C.; Ketterings, Q.M. In-season estimation of corn yield potential using proximal sensing. Agron. J. 2017, 109, 1323–1330. [Google Scholar] [CrossRef]
- Shanahan, J.; Kitchen, N.; Raun, W.; Schepers, J.S. Responsive in-season nitrogen management for cereals. Comput. Electron. Agric. 2008, 61, 51–62. [Google Scholar] [CrossRef]
- Siqueira, R.; Longchamps, L.; Dahal, S.; Khosla, R. Use of fluorescence sensing to detect nitrogen and potassium variability in maize. Remote Sens. 2020, 12, 1752. [Google Scholar] [CrossRef]
- Dahal, S.; Phillippi, E.; Longchamps, L.; Khosla, R.; Andales, A. Variable Rate Nitrogen and Water Management for Irrigated Maize in the Western US. Agronomy 2020, 10, 1533. [Google Scholar] [CrossRef]
- Havránková, J. The evaluation of ground based remote sensing systems for canopy nitrogen management in winter wheat. CIGR J. 2007. Available online: https://dspace.lib.cranfield.ac.uk/handle/1826/1711 (accessed on 23 February 2021).
- Thind, H.; Kumar, A.; Vashistha, M. Calibrating the leaf colour chart for need based fertilizer nitrogen management in different maize (Zea mays L.) genotypes. Field Crop. Res. 2011, 120, 276–282. [Google Scholar]
- Fu, Y.-Q.; Zhong, X.-H.; Zeng, J.-H.; Liang, K.-M.; Pan, J.-F.; Xin, Y.-F.; Liu, Y.-Z.; Hu, X.-Y.; Peng, B.-L.; Chen, R.-B. Improving grain yield, nitrogen use efficiency and radiation use efficiency by dense planting, with delayed and reduced nitrogen application, in double cropping rice in South China. J. Integr. Agric. 2021, 20, 565–580. [Google Scholar] [CrossRef]
- Chen, Y.; Fan, P.; Mo, Z.; Kong, L.; Tian, H.; Duan, M.; Li, L.; Wu, L.; Wang, Z.; Tang, X. Deep Placement of Nitrogen Fertilizer Affects Grain Yield, Nitrogen Recovery Efficiency, and Root Characteristics in Direct-Seeded Rice in South China. J. Plant. Growth Regul. 2020, 40, 379–387. [Google Scholar] [CrossRef]
- KhalilA, M. Physical and chemical manipulation of urea fertiliser for reducing the emission of gaseous nitrogen species. In Proceedings of the 19th World Congress of Soil Science: Soil Solutions for a Changing World, Brisbane, Australia, 1–6 August 2010; Congress Symposium 4: Greenhouse Gases from Soils. pp. 195–198. [Google Scholar]
- Chatterjee, D.; Mohanty, S.; Guru, P.K.; Swain, C.K.; Tripathi, R.; Shahid, M.; Kumar, U.; Kumar, A.; Bhattacharyya, P.; Gautam, P. Comparative assessment of urea briquette applicators on greenhouse gas emission, nitrogen loss and soil enzymatic activities in tropical lowland rice. Agric. Ecosyst. Environ. 2018, 252, 178–190. [Google Scholar] [CrossRef]
- Mencaroni, M.; Dal Ferro, N.; Furlanetto, J.; Longo, M.; Lazzaro, B.; Sartori, L.; Grant, B.; Smith, W.; Morari, F. Identifying N fertilizer management strategies to reduce ammonia volatilization: Towards a site-specific approach. J. Environ. Manag. 2021, 277, 111445. [Google Scholar] [CrossRef] [PubMed]
- Lessa, A.C.R.; Madari, B.E.; Paredes, D.S.; Boddey, R.M.; Urquiaga, S.; Jantalia, C.P.; Alves, B.J.J.A. Ecosystems; Environment. Bovine urine and dung deposited on Brazilian savannah pastures contribute differently to direct and indirect soil nitrous oxide emissions. Agric. Ecosyst. Environ. 2014, 190, 104–111. [Google Scholar] [CrossRef]
- Datta, R.; Kelkar, A.; Baraniya, D.; Molaei, A.; Moulick, A.; Meena, R.; Formanek, P. Enzymatic degradation of lignin in soil: A review. Sustainability 2017, 7, 1163. [Google Scholar] [CrossRef]
- Subedi, A.; Franklin, D.; Cabrera, M.; McPherson, A.; Dahal, S. Grazing Systems to Retain and Redistribute Soil Phosphorus and to Reduce Phosphorus Losses in Runoff. Soil Syst. 2020, 4, 66. [Google Scholar] [CrossRef]
- Adegbeye, M.; Reddy, P.R.K.; Obaisi, A.; Elghandour, M.; Oyebamiji, K.; Salem, A.; Morakinyo-Fasipe, O.; Cipriano-Salazar, M.; Camacho-Díaz, L. Sustainable agriculture options for production, greenhouse gasses and pollution alleviation, and nutrient recycling in emerging and transitional nations-An overview. J. Clean. Prod. 2020, 242, 118319. [Google Scholar] [CrossRef]
- Frank, S.; Havlík, P.; Stehfest, E.; van Meijl, H.; Witzke, P.; Pérez-Domínguez, I.; van Dijk, M.; Doelman, J.C.; Fellmann, T.; Koopman, J.F. Agricultural non-CO2 emission reduction potential in the context of the 1.5 °C target. Nat. Clim. Chang. 2019, 9, 66–72. [Google Scholar] [CrossRef]
- Herrero, M.; Havlík, P.; Valin, H.; Notenbaert, A.; Rufino, M.C.; Thornton, P.K.; Blümmel, M.; Weiss, F.; Grace, D.; Obersteiner, M. Biomass use, production, feed efficiencies, and greenhouse gas emissions from global livestock systems. Proc. Natl. Acad. Sci. USA 2013, 110, 20888–20893. [Google Scholar] [CrossRef]
- Takemasa, M.J.K. Nutritional strategies to reduce nutrient waste in livestock and poultry production. Food 1998, 36, 720–726. [Google Scholar]
- Stephenson, G.T. The Effects of Agricultural Waste-Based Compost Amendments in Organic Pest Management. Master’s Thesis, California Polytechnics University, San Luis Obispo, CA, USA, 2019. [Google Scholar]
- Bhandari, K.B.; West, C.P.; Acosta-Martinez, V.; Cotton, J.; Cano, A. Soil health indicators as affected by diverse forage species and mixtures in semi-arid pastures. Appl. Soil Ecol. 2018, 132, 179–186. [Google Scholar] [CrossRef]
- Peoples, M.; Baldock, J. Nitrogen dynamics of pastures: Nitrogen fixation inputs, the impact of legumes on soil nitrogen fertility, and the contributions of fixed nitrogen to Australian farming systems. Aust. J. Exp. Agric. 2001, 41, 327–346. [Google Scholar] [CrossRef]
- Stockdale, E.; Lampkin, N.; Hovi, M.; Keatinge, R.; Lennartsson, E.; Macdonald, D.; Padel, S.; Tattersall, F.; Wolfe, M.; Watson, C. Agronomic and environmental implications of organic farming systems. Agronomy 2001, 70, 261–327. [Google Scholar]
- White, J.; Hodgson, J.G. New Zealand Pasture and Crop Science; Oxford University Press: Oxford, UK, 1999. [Google Scholar]
- Bhandari, K.B.; West, C.P.; Acosta-Martinez, V. Assessing the role of interseeding alfalfa into grass on improving pasture soil health in semi-arid Texas High Plains. Appl. Soil Ecol. 2020, 147, 103399. [Google Scholar] [CrossRef]
- Mobin, S.; Alam, F. Biofuel production from algae utilizing wastewater. In Proceedings of the 19th Australasian Fluid Mechanics Conference, Melbourne, Australia, 8–11 December 2014. [Google Scholar]
- Dang, N.M.; Lee, K. Recent trends of using alternative nutrient sources for microalgae cultivation as a feedstock of biodiesel production. Appl. Chem. Eng. 2018, 29, 1–9. [Google Scholar]
- Aslan, S.; Kapdan, I.K. Batch kinetics of nitrogen and phosphorus removal from synthetic wastewater by algae. Ecol. Eng. 2006, 28, 64–70. [Google Scholar] [CrossRef]
- Marinho-Soriano, E.; Nunes, S.; Carneiro, M.; Pereira, D. Nutrients’ removal from aquaculture wastewater using the macroalgae Gracilaria birdiae. Biomass Bioenergy 2009, 33, 327–331. [Google Scholar] [CrossRef]
- He, P.; Xu, S.; Zhang, H.; Wen, S.; Dai, Y.; Lin, S.; Yarish, C. Bioremediation efficiency in the removal of dissolved inorganic nutrients by the red seaweed, Porphyra yezoensis, cultivated in the open sea. Water Res. 2008, 42, 1281–1289. [Google Scholar] [CrossRef]
- Webb, J.R.; Hayes, N.M.; Simpson, G.L.; Leavitt, P.R.; Baulch, H.M.; Finlay, K. Widespread nitrous oxide undersaturation in farm waterbodies creates an unexpected greenhouse gas sink. Proc. Natl. Acad. Sci. USA 2019, 116, 9814–9819. [Google Scholar] [CrossRef]
- Meade, G.; Pierce, K.; O’Doherty, J.; Mueller, C.; Lanigan, G.; Mc Cabe, T. Ammonia and nitrous oxide emissions following land application of high and low nitrogen pig manures to winter wheat at three growth stages. Agric. Ecosyst. Environ. 2011, 140, 208–217. [Google Scholar] [CrossRef]
- Mahmud, K.; Chowhdhury, M.; Noor, N.; Huq, S.I. Effects of different sources of biochar application on the emission of a number of gases from soil. Can. J. Pure Appl. Sci. 2014, 8, 2813–2824. [Google Scholar]
- Brennan, R.B.; Healy, M.G.; Fenton, O.; Lanigan, G.J. The effect of chemical amendments used for phosphorus abatement on greenhouse gas and ammonia emissions from dairy cattle slurry: Synergies and pollution swapping. PLoS ONE 2015, 10, e0111965. [Google Scholar] [CrossRef] [PubMed]
- Eggleston, S.; Buendia, L.; Miwa, K.; Ngara, T.; Tanabe, K. 2006 IPCC Guidelines for National Greenhouse Gas Inventories; Institute for Global Environmental Strategies: Wageningen, The Netherlands, 2006; Volume 5. [Google Scholar]
- Zhu, X.; Burger, M.; Doane, T.A.; Horwath, W.R. Ammonia oxidation pathways and nitrifier denitrification are significant sources of N2O and NO under low oxygen availability. Proc. Natl. Acad. Sci. USA 2013, 110, 6328–6333. [Google Scholar] [CrossRef]
- Panday, D.; Mikha, M.M.; Collins, H.P.; Jin, V.L.; Kaiser, M.; Cooper, J.; Malakar, A.; Maharjan, B. Optimum rates of surface-applied coal char decreased soil ammonia volatilization loss. J. Environ. Qual. 2020, 49, 256–267. [Google Scholar] [CrossRef] [PubMed]
- Oleszczuk, P.; Hollert, H. Comparison of sewage sludge toxicity to plants and invertebrates in three different soils. Chemosphere 2011, 83, 502–509. [Google Scholar] [CrossRef] [PubMed]
- Zielińska, A.; Oleszczuk, P. Evaluation of sewage sludge and slow pyrolyzed sewage sludge-derived biochar for adsorption of phenanthrene and pyrene. Bioresour. Technol. 2015, 192, 618–626. [Google Scholar] [CrossRef]
- Ramakrishnan, B.; Maddela, N.R.; Venkateswarlu, K.; Megharaj, M. Organic farming: Does it contribute to contaminant-free produce and ensure food safety? Sci. Total. Environ. 2021, 769, 145079. [Google Scholar] [CrossRef]
- Oleszczuk, P.; Hale, S.E.; Lehmann, J.; Cornelissen, G. Activated carbon and biochar amendments decrease pore-water concentrations of polycyclic aromatic hydrocarbons (PAHs) in sewage sludge. Bioresour. Technol. 2012, 111, 84–91. [Google Scholar] [CrossRef]
- Stefaniuk, M.; Oleszczuk, P. Addition of biochar to sewage sludge decreases freely dissolved PAHs content and toxicity of sewage sludge-amended soil. Environ. Pollut. 2016, 218, 242–251. [Google Scholar] [CrossRef] [PubMed]
- Oleszczuk, P.; Zielińska, A.; Cornelissen, G. Stabilization of sewage sludge by different biochars towards reducing freely dissolved polycyclic aromatic hydrocarbons (PAHs) content. Bioresour. Technol. 2014, 156, 139–145. [Google Scholar] [CrossRef] [PubMed]
- Kończak, M.; Oleszczuk, P. Application of biochar to sewage sludge reduces toxicity and improve organisms growth in sewage sludge-amended soil in long term field experiment. Sci. Total Environ. 2018, 625, 8–15. [Google Scholar] [CrossRef] [PubMed]
- Noor, N.; Mahmud, K.; Chowdhury, M.T.A.; Huq, S.I. The use of biochar as ameliorator for soil arsenic. Dhaka Univ. J. Biol. Sci. 2015, 24, 111–119. [Google Scholar] [CrossRef][Green Version]
- Mahmud, K.; Makaju, S.; Ibrahim, R.; Missaoui, A.J.P. Current Progress in Nitrogen Fixing Plants and Microbiome Research. Plants 2020, 9, 97. [Google Scholar] [CrossRef] [PubMed]
- Bloch, S.E.; Ryu, M.-H.; Ozaydin, B.; Broglie, R. Harnessing atmospheric nitrogen for cereal crop production. Curr. Opin. Biotechnol. 2020, 62, 181–188. [Google Scholar] [CrossRef]
- Rogers, C.; Oldroyd, G.E. Synthetic biology approaches to engineering the nitrogen symbiosis in cereals. J. Exp. Bot. 2014, 65, 1939–1946. [Google Scholar] [CrossRef]
- Madsen, L.H.; Tirichine, L.; Jurkiewicz, A.; Sullivan, J.T.; Heckmann, A.B.; Bek, A.S.; Ronson, C.W.; James, E.K.; Stougaard, J. The molecular network governing nodule organogenesis and infection in the model legume Lotus japonicus. Nat. Commun. 2010, 1, 1–12. [Google Scholar] [CrossRef]
- Maillet, F.; Poinsot, V.; André, O.; Puech-Pagès, V.; Haouy, A.; Gueunier, M.; Cromer, L.; Giraudet, D.; Formey, D.; Niebel, A. Fungal lipochitooligosaccharide symbiotic signals in arbuscular mycorrhiza. Nature 2011, 469, 58–63. [Google Scholar] [CrossRef]
- Oldroyd, G.E. Speak, friend, and enter: Signalling systems that promote beneficial symbiotic associations in plants. Nat. Rev. Microbiol. 2013, 11, 252–263. [Google Scholar] [CrossRef]
- Mus, F.; Crook, M.B.; Garcia, K.; Costas, A.G.; Geddes, B.A.; Kouri, E.D.; Paramasivan, P.; Ryu, M.-H.; Oldroyd, G.E.; Poole, P.S. Symbiotic nitrogen fixation and the challenges to its extension to nonlegumes. Appl. Environ. Microbiol. 2016, 82, 3698–3710. [Google Scholar] [CrossRef] [PubMed]
- Geddes, B.A.; Ryu, M.-H.; Mus, F.; Costas, A.G.; Peters, J.W.; Voigt, C.A.; Poole, P. Use of plant colonizing bacteria as chassis for transfer of N2-fixation to cereals. Curr. Opin. Biotechnol. 2015, 32, 216–222. [Google Scholar] [CrossRef] [PubMed]
- Rocha, F.R.; Papini-Terzi, F.S.; Nishiyama, M.Y.; Vencio, R.Z.; Vicentini, R.; Duarte, R.D.; de Rosa, V.E.; Vinagre, F.; Barsalobres, C.; Medeiros, A.H. Signal transduction-related responses to phytohormones and environmental challenges in sugarcane. BMC Genom. 2007, 8, 71. [Google Scholar] [CrossRef]
- Ibort, P.; Imai, H.; Uemura, M.; Aroca, R. Proteomic analysis reveals that tomato interaction with plant growth promoting bacteria is highly determined by ethylene perception. J. Plant Physiol. 2018, 220, 43–59. [Google Scholar] [CrossRef] [PubMed]
- Brusamarello-Santos, L.C.; Gilard, F.; Brulé, L.; Quilleré, I.; Gourion, B.; Ratet, P.; Maltempi de Souza, E.; Lea, P.J.; Hirel, B. Metabolic profiling of two maize (Zea mays L.) inbred lines inoculated with the nitrogen fixing plant-interacting bacteria Herbaspirillum seropedicae and Azospirillum brasilense. PLoS ONE 2017, 12, e0174576. [Google Scholar] [CrossRef]
- Kost, T.; Stopnisek, N.; Agnoli, K.; Eberl, L.; Weisskopf, L. Oxalotrophy, a widespread trait of plant-associated Burkholderia species, is involved in successful root colonization of lupin and maize by Burkholderia phytofirmans. Front. Microbiol. 2014, 4, 421. [Google Scholar] [CrossRef]
- Van Deynze, A.; Zamora, P.; Delaux, P.-M.; Heitmann, C.; Jayaraman, D.; Rajasekar, S.; Graham, D.; Maeda, J.; Gibson, D.; Schwartz, K.D. Nitrogen fixation in a landrace of maize is supported by a mucilage-associated diazotrophic microbiota. PLoS Biol. 2018, 16, e2006352. [Google Scholar] [CrossRef]
- Oliveira, A.L.; Santos, O.J.; Marcelino, P.R.; Milani, K.M.; Zuluaga, M.Y.; Zucareli, C.; Gonçalves, L.S. Maize inoculation with Azospirillum brasilense Ab-V5 cells enriched with exopolysaccharides and polyhydroxybutyrate results in high productivity under low N fertilizer input. Front. Microbiol. 2017, 8, 1873. [Google Scholar] [CrossRef] [PubMed]
- Compant, S.; Samad, A.; Faist, H.; Sessitsch, A. A review on the plant microbiome: Ecology, functions, and emerging trends in microbial application. J. Adv. Res. 2019, 19, 29–37. [Google Scholar] [CrossRef]
- Hartmann, M.; Frey, B.; Mayer, J.; Mäder, P.; Widmer, F. Distinct soil microbial diversity under long-term organic and conventional farming. ISME J. 2015, 9, 1177–1194. [Google Scholar] [CrossRef]
- Larsen, E.; Grossman, J.; Edgell, J.; Hoyt, G.; Osmond, D.; Hu, S. Soil biological properties, soil losses and corn yield in long-term organic and conventional farming systems. Soil Tillage Res. 2014, 139, 37–45. [Google Scholar] [CrossRef]
- Coelho, L.M.; Rezende, H.C.; Coelho, L.M.; de Sousa, P.; Melo, D.; Coelho, N. Bioremediation of polluted waters using microorganisms. Adv. Bioremedia. Wastewater Polluted Soil 2015, 10, 60770. [Google Scholar]
- Kloepper, J.W. Plant growth-promoting rhizobacteria on radishes. In Proceedings of the 4th International Conference on Plant Pathogenic Bacter, Station de Pathologie Vegetale et Phytobacteriologie, INRA, Angers, France, 27 August–2 September 1978; pp. 879–882. [Google Scholar]
- Bonkowski, M.; Villenave, C.; Griffiths, B. Rhizosphere fauna: The functional and structural diversity of intimate interactions of soil fauna with plant roots. Plant Soil 2009, 321, 213–233. [Google Scholar] [CrossRef]
- Buee, M.; De Boer, W.; Martin, F.; Van Overbeek, L.; Jurkevitch, E. The rhizosphere zoo: An overview of plant-associated communities of microorganisms, including phages, bacteria, archaea, and fungi, and of some of their structuring factors. Plant Soil 2009, 321, 189–212. [Google Scholar] [CrossRef]
- Cao, Y.; Bai, M.; Han, B.; Impraim, R.; Butterly, C.; Hu, H.; He, J.; Chen, D. Enhanced nitrogen retention by lignite during poultry litter composting. J. Clean. Prod. 2020, 277, 122422. [Google Scholar] [CrossRef]
- Morriën, E. Understanding soil food web dynamics, how close do we get? Soil Biol. Biochem. 2016, 102, 10–13. [Google Scholar] [CrossRef]
- Verbruggen, E.; van der Heijden, M.G.; Rillig, M.C.; Kiers, E.T. Mycorrhizal fungal establishment in agricultural soils: Factors determining inoculation success. New Phytol. 2013, 197, 1104–1109. [Google Scholar] [CrossRef]
- Ney, L.; Franklin, D.; Mahmud, K.; Cabrera, M.; Hancock, D.; Habteselassie, M.; Newcomer, Q. Examining trophic-level nematode community structure and nitrogen mineralization to assess local effective microorganisms’ role in nitrogen availability of swine effluent to forage crops. Appl. Soil Ecol. 2018, 130, 209–218. [Google Scholar] [CrossRef]
- Ney, L.; Franklin, D.; Mahmud, K.; Cabrera, M.; Hancock, D.; Habteselassie, M.; Newcomer, Q.; Fatzinger, B. Rebuilding Soil Ecosystems for Improved Productivity in Biosolarized Soils. Int. J. Agron. 2019, 2019. [Google Scholar] [CrossRef]
- Mahmud, K.; Franklin, D.; Ney, L.; Cabrera, M.; Habteselassie, M.; Hancock, D.; Newcomer, Q.; Subedi, A.; Dahal, S. Improving inorganic nitrogen in soil and nutrient density of edamame bean in three consecutive summers by utilizing a locally sourced bio-inocula. Org. Agric. 2021. [Google Scholar] [CrossRef]
- Li, X.; Guo, Q.; Wang, Y.; Xu, J.; Wei, Q.; Chen, L.; Liao, L. Enhancing Nitrogen and Phosphorus Removal by Applying Effective Microorganisms to Constructed Wetlands. Water 2020, 12, 2443. [Google Scholar] [CrossRef]
- Polyorach, S.; Wanapat, M.; Cherdthong, A.; Kang, S. Rumen microorganisms, methane production, and microbial protein synthesis affected by mangosteen peel powder supplement in lactating dairy cows. Trop. Anim. Health Prod. 2016, 48, 593–601. [Google Scholar] [CrossRef] [PubMed]
- Alves, T.P.; Dall-Orsoletta, A.C.; Ribeiro-Filho, H.M.N. The effects of supplementing Acacia mearnsii tannin extract on dairy cow dry matter intake, milk production, and methane emission in a tropical pasture. Trop. Anim. Health Prod. 2017, 49, 1663–1668. [Google Scholar] [CrossRef]
- Albores-Moreno, S.; Alayón-Gamboa, J.; Ayala-Burgos, A.; Solorio-Sánchez, F.; Aguilar-Pérez, C.; Olivera-Castillo, L.; Ku-Vera, J. Effects of feeding ground pods of Enterolobium cyclocarpum Jacq. Griseb on dry matter intake, rumen fermentation, and enteric methane production by Pelibuey sheep fed tropical grass. Trop. Anim. Health Prod. 2017, 49, 857–866. [Google Scholar] [CrossRef] [PubMed]
- Freitas, C.E.S.; Duarte, E.R.; Alves, D.D.; Martinele, I.; D’Agosto, M.; Cedrola, F.; de Moura Freitas, A.A.; dos Santos Soares, F.D.; Beltran, M. Sheep fed with banana leaf hay reduce ruminal protozoa population. Trop. Anim. Health Prod. 2017, 49, 807–812. [Google Scholar] [CrossRef]
- Gerlach, K.; Schmithausen, A.J.; Sommer, A.C.; Trimborn, M.; Büscher, W.; Südekum, K.-H. Cattle diets strongly affect nitrous oxide in the rumen. Sustainability 2018, 10, 3679. [Google Scholar] [CrossRef]
- Reganold, J.P.; Wachter, J.M. Organic agriculture in the twenty-first century. Nat. Plants 2016, 2, 1–8. [Google Scholar] [CrossRef]
- Trewavas, A. Urban myths of organic farming. Nature 2001, 410, 409–410. [Google Scholar] [CrossRef] [PubMed]
- Emsley, J. Going one better than nature? Nature 2001, 410, 633–634. [Google Scholar] [CrossRef]
- Willer, H.; Lernoud, J. The World of Organic Agriculture. Statistics and Emerging Trends 2019; Research Institute of Organic Agriculture FiBL and IFOAM Organics International: Frick, Switzerland; Bonn, Switzerland, 2019. [Google Scholar]
- Saffeullah, P.; Nabi, N.; Liaqat, S.; Anjum, N.A.; Siddiqi, T.O.; Umar, S. Organic Agriculture: Principles, Current Status, and Significance. In Microbiota and Biofertilizers; Springer: Singapore, 2021; pp. 17–37. [Google Scholar]
- Shi, W.; Norton, J.M. Microbial control of nitrate concentrations in an agricultural soil treated with dairy waste compost or ammonium fertilizer. Soil Biol. Biochem. 2000, 32, 1453–1457. [Google Scholar] [CrossRef]
- Habteselassie, M.Y.; Miller, B.E.; Thacker, S.G.; Stark, J.M.; Norton, J.M. Soil nitrogen and nutrient dynamics after repeated application of treated dairy-waste. Soil Sci. Soc. Am. J. 2006, 70, 1328–1337. [Google Scholar] [CrossRef]
- Cameron, K.C.; Rate, A.W.; Noonan, M.J.; Moore, S.; Smith, N.P.; Kerr, L.E. Lysimeter study of the fate of nutrients following subsurface injection and surface application of dairy pond sludge to pasture. Agric. Ecosyst. Environ. 1996, 58, 187–197. [Google Scholar] [CrossRef]
- Kirchmann, H.; Bergström, L. Do organic farming practices reduce nitrate leaching? Commun. Soil Sci. Plant Anal. 2001, 32, 997–1028. [Google Scholar] [CrossRef]
- Scialabba, N.E.-H.; Müller-Lindenlauf, M. Organic agriculture and climate change. Renew. Agric. Food Syst. 2010, 25, 158–169. [Google Scholar] [CrossRef]
- Chatterjee, R. Projecting the Future of Nitrogen Pollution 2009. [CrossRef]
- Williams, A.; Audsley, E.; Sandars, D. Determining the Environmental Burdens and Resource Use in the Production of Agricultural and Horticultural Commodities: Defra Project Report IS0205. Available online: http://randd.defra.gov.uk/Default.aspx (accessed on 27 January 2021).
- Rahmann, G.; Ardakani, M.R.; Bàrberi, P.; Boehm, H.; Canali, S.; Chander, M.; David, W.; Dengel, L.; Erisman, J.W.; Galvis-Martinez, A.C. Organic Agriculture 3.0 is innovation with research. Org. Agric. 2017, 7, 169–197. [Google Scholar] [CrossRef]
- Smith, J.B.; Lenhart, S.S. Climate change adaptation policy options. Clim. Res. 1996, 6, 193–201. [Google Scholar] [CrossRef]
- Zhang, F.; Li, L. Using competitive and facilitative interactions in intercropping systems enhances crop productivity and nutrient-use efficiency. Plant Soil 2003, 248, 305–312. [Google Scholar] [CrossRef]
- Tilman, D. Global environmental impacts of agricultural expansion: The need for sustainable and efficient practices. Proc. Natl. Acad. Sci. USA 1999, 96, 5995–6000. [Google Scholar] [CrossRef]
- Vymazal, J.; Březinová, T.D. Removal of nutrients, organics and suspended solids in vegetated agricultural drainage ditch. Ecol. Eng. 2018, 118, 97–103. [Google Scholar] [CrossRef]
- Xia, Y.; Zhang, M.; Tsang, D.C.; Geng, N.; Lu, D.; Zhu, L.; Igalavithana, A.D.; Dissanayake, P.D.; Rinklebe, J.; Yang, X. Recent advances in control technologies for non-point source pollution with nitrogen and phosphorous from agricultural runoff: Current practices and future prospects. Appl. Biol. Chem. 2020, 63, 1–13. [Google Scholar] [CrossRef]
- Conley, D.J.; Paerl, H.W.; Howarth, R.W.; Boesch, D.F.; Seitzinger, S.P.; Havens, K.E.; Lancelot, C.; Likens, G.E. Controlling eutrophication: Nitrogen and phosphorus. Science 2009, 323, 1014–1015. [Google Scholar] [CrossRef] [PubMed]
- Howarth, R.; Chan, F.; Conley, D.J.; Garnier, J.; Doney, S.C.; Marino, R.; Billen, G. Coupled biogeochemical cycles: Eutrophication and hypoxia in temperate estuaries and coastal marine ecosystems. Front. Ecol. Environ. 2011, 9, 18–26. [Google Scholar] [CrossRef]
- Kumwimba, M.N.; Meng, F.; Iseyemi, O.; Moore, M.T.; Zhu, B.; Tao, W.; Liang, T.J.; Ilunga, L. Removal of non-point source pollutants from domestic sewage and agricultural runoff by vegetated drainage ditches (VDDs): Design, mechanism, management strategies, and future directions. Sci. Total Environ. 2018, 639, 742–759. [Google Scholar] [CrossRef] [PubMed]
- Dollinger, J.; Dagès, C.; Bailly, J.-S.; Lagacherie, P.; Voltz, M. Managing ditches for agroecological engineering of landscape: A review. Agron. Sustain. Dev. 2015, 35, 999–1020. [Google Scholar] [CrossRef]
- Nsenga Kumwimba, M.; Dzakpasu, M.; Zhu, B.; Wang, T.; Ilunga, L.; Kavidia Muyembe, D. Nutrient removal in a trapezoidal vegetated drainage ditch used to treat primary domestic sewage in a small catchment of the upper Yangtze River. Water Environ. J. 2017, 31, 72–79. [Google Scholar] [CrossRef]
- Kröger, R.; Holland, M.; Moore, M.; Cooper, C. Hydrological variability and agricultural drainage ditch inorganic nitrogen reduction capacity. J. Environ. Qual. 2007, 36, 1646–1652. [Google Scholar] [CrossRef]
- Menon, R.; Holland, M.M. Phosphorus release due to decomposition of wetland plants. Wetlands 2014, 34, 1191–1196. [Google Scholar] [CrossRef]
- Yu, H.; Yang, Z.; Xiao, R.; Zhang, S.; Liu, F.; Xiang, Z. Absorption capacity of nitrogen and phosphorus of aquatic plants and harvest management research. Acta Prataculturae Sin. 2013, 22, 294–299. [Google Scholar]
- Rounsley, S.D.; Glodek, A.; Sutton, G.; Adams, M.D.; Somerville, C.R.; Venter, J.C.; Kerlavage, A.R. The construction of Arabidopsis expressed sequence tag assemblies (a new resource to facilitate gene identification). Plant Physiol. 1996, 112, 1177–1183. [Google Scholar] [CrossRef] [PubMed]
- Velculescu, V.E.; Zhang, L.; Vogelstein, B.; Kinzler, K.W. Serial analysis of gene expression. Science 1995, 270, 484–487. [Google Scholar] [CrossRef]
- Katagiri, F.; Glazebrook, J. Overview of mRNA expression profiling using DNA microarrays. Curr. Protoc. Mol. Biol. 2009, 85, 22.4.1–22.4.13. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Gerstein, M.; Snyder, M. RNA-Seq: A revolutionary tool for transcriptomics. Nat. Rev. Genet. 2009, 10, 57–63. [Google Scholar] [CrossRef]
- Liu, R.; Zhang, H.; Zhao, P.; Zhang, Z.; Liang, W.; Tian, Z.; Zheng, Y. Mining of candidate maize genes for nitrogen use efficiency by integrating gene expression and QTL data. Plant Mol. Biol. Report. 2012, 30, 297–308. [Google Scholar] [CrossRef]
- Bordes, J.; Ravel, C.; Jaubertie, J.; Duperrier, B.; Gardet, O.; Heumez, E.; Pissavy, A.; Charmet, G.; Le Gouis, J.; Balfourier, F. Genomic regions associated with the nitrogen limitation response revealed in a global wheat core collection. Theor. Appl. Genet. 2013, 126, 805–822. [Google Scholar] [CrossRef]
- Robertson, G.P.; Bruulsema, T.W.; Gehl, R.J.; Kanter, D.; Mauzerall, D.L.; Rotz, C.A.; Williams, C.O. Nitrogen–climate interactions in US agriculture. Biogeochemistry 2013, 114, 41–70. [Google Scholar] [CrossRef]
- Suddick, E.C.; Whitney, P.; Townsend, A.R.; Davidson, E.A. The role of nitrogen in climate change and the impacts of nitrogen–climate interactions in the United States: Foreword to thematic issue. Biogeochemistry 2013, 114, 1–10. [Google Scholar] [CrossRef]
- Townsend, A.R.; Vitousek, P.M.; Houlton, B.Z. The climate benefits of better nitrogen and phosphorus management. Issues Sci. Technol. 2012, 28, 85–91. [Google Scholar]
- Sohrabi, C.; Alsafi, Z.; O’Neill, N.; Khan, M.; Kerwan, A.; Al-Jabir, A.; Iosifidis, C.; Agha, R. World Health Organization declares global emergency: A review of the 2019 novel coronavirus (COVID-19). Int. J. Surg. 2020, 76, 71–76. [Google Scholar] [CrossRef]
- Jebril, N. World Health Organization declared a pandemic public health menace: A systematic review of the coronavirus disease 2019 “COVID-19”, up to 26th March 2020. SSRN . 2020. [CrossRef]
- Ogen, Y. Assessing nitrogen dioxide (NO2) levels as a contributing factor to the coronavirus (COVID-19) fatality rate. Sci. Total Environ. 2020, 726, 138605. [Google Scholar] [CrossRef]
- Karaer, A.; Balafkan, N.; Gazzea, M.; Arghandeh, R.; Ozguven, E.E. Analyzing COVID-19 Impacts on Vehicle Travels and Daily Nitrogen Dioxide (NO2) Levels among Florida Counties. Energies 2020, 13, 6044. [Google Scholar] [CrossRef]
- Dasgupta, D.; Rajeev, M. Paradox of a Supply Constrained Keynesian Equilibrium. Econ. Political Week. 2020, 55, 23. [Google Scholar]
- Sampath, P.V.; Jagadeesh, G.S.; Bahinipati, C.S. Sustainable Intensification of Agriculture in the Context of the COVID-19 Pandemic: Prospects for the Future. Water 2020, 12, 2738. [Google Scholar] [CrossRef]
- Lal, R.; Brevik, E.C.; Dawson, L.; Field, D.; Glaser, B.; Hartemink, A.E.; Hatano, R.; Lascelles, B.; Monger, C.; Scholten, T. Managing soils for recovering from the COVID-19 pandemic. Soil Syst. 2020, 4, 46. [Google Scholar] [CrossRef]
- Le Quéré, C.; Jackson, R.B.; Jones, M.W.; Smith, A.J.; Abernethy, S.; Andrew, R.M.; De-Gol, A.J.; Willis, D.R.; Shan, Y.; Canadell, J.G. Temporary reduction in daily global CO2 emissions during the COVID-19 forced confinement. Nat. Clim. Chang. 2020, 10, 647–653. [Google Scholar] [CrossRef]
- Tirado, M.C.; Crahay, P.; Mahy, L.; Zanev, C.; Neira, M.; Msangi, S.; Brown, R.; Scaramella, C.; Coitinho, D.C.; Müller, A. Climate change and nutrition: Creating a climate for nutrition security. Food Nutr. Bull. 2013, 34, 533–547. [Google Scholar] [CrossRef]
- Porter, S.D.; Reay, D.S.; Higgins, P.; Bomberg, E. A half-century of production-phase greenhouse gas emissions from food loss & waste in the global food supply chain. Sci. Total Environ. 2016, 571, 721–729. [Google Scholar] [PubMed]
- Nemecek, T.; Jungbluth, N.; i Canals, L.M.; Schenck, R. Environmental impacts of food consumption and nutrition: Where are we and what is next. Int. J. Life Cycle Assess. 2016, 5, 607–620. [Google Scholar] [CrossRef]
- Clonan, A.; Roberts, K.E.; Holdsworth, M. Socioeconomic and demographic drivers of red and processed meat consumption: Implications for health and environmental sustainability. Proc. Nutr. Soc. 2016, 75, 367–373. [Google Scholar] [CrossRef]
- Jones, A.D.; Hoey, L.; Blesh, J.; Miller, L.; Green, A.; Shapiro, L.F. A systematic review of the measurement of sustainable diets. Adv. Nutr. 2016, 7, 641–664. [Google Scholar] [CrossRef]
- Tirado, M.; Hunnes, D.; Cohen, M.; Lartey, A. Climate change and nutrition in Africa. J. Hunger Environ. Nutr. 2015, 10, 22–46. [Google Scholar] [CrossRef]
- Niles, M.T.; Ahuja, R.; Barker, T.; Esquivel, J.; Gutterman, S.; Heller, M.C.; Mango, N.; Portner, D.; Raimond, R.; Tirado, C. Climate change mitigation beyond agriculture: A review of food system opportunities and implications. Renew. Agric. Food Syst. 2018, 33, 297–308. [Google Scholar] [CrossRef]
- Fischer, C.G.; Garnett, T. Plates, Pyramids, and Planets: Developments in National Healthy and Sustainable Dietary Guidelines: A State of Play Assessment; Food and Agriculture Organization of the United Nations: Rome, Italy, 2016. [Google Scholar]
- Rockström, J.; Stordalen, G.A.; Horton, R. Acting in the anthropocene: The EAT–Lancet Commission; Lancet: London, UK, 2016. [Google Scholar]
- Marinangeli, C.P.; Mansilla, W.D.; Shoveller, A.-K. Navigating protein claim regulations in North America for foods containing plant-based proteins. Cereal Foods World 2018, 63, 207–216. [Google Scholar]
- Springmann, M.; Mason-D’Croz, D.; Robinson, S.; Wiebe, K.; Godfray, H.C.J.; Rayner, M.; Scarborough, P. Mitigation potential and global health impacts from emissions pricing of food commodities. Nat. Clim. Chang. 2017, 7, 69–74. [Google Scholar] [CrossRef]
- Sutton, M.A.; Howard, C.M.; Kanter, D.R.; Lassaletta, L.; Móring, A.; Raghuram, N.; Read, N. The nitrogen decade: Mobilizing global action on nitrogen to 2030 and beyond. One Earth 2021, 4, 10–14. [Google Scholar] [CrossRef]
- De Notaris, C.; Olesen, J.E.; Sørensen, P.; Rasmussen, J. Input and mineralization of carbon and nitrogen in soil from legume-based cover crops. Nutr. Cycl. Agroecosyst. 2020, 116, 1–18. [Google Scholar] [CrossRef]
- Lu, J.; Hu, T.; Zhang, B.; Wang, L.; Yang, S.; Fan, J.; Yan, S.; Zhang, F. Nitrogen fertilizer management effects on soil nitrate leaching, grain yield and economic benefit of summer maize in Northwest China. Agric. Water Manag. 2021, 247, 106739. [Google Scholar] [CrossRef]
- Lyu, Y.; Yang, X.; Pan, H.; Zhang, X.; Cao, H.; Ulgiati, S.; Wu, J.; Zhang, Y.; Wang, G.; Xiao, Y. Impact of fertilization schemes with different ratios of urea to controlled release nitrogen fertilizer on environmental sustainability, nitrogen use efficiency and economic benefit of rice production: A study case from Southwest China. J. Clean. Prod. 2021, 293, 126198. [Google Scholar] [CrossRef]
- De Laporte, A.; Banger, K.; Weersink, A.; Wagner-Riddle, C.; Grant, B.; Smith, W. Economic and environmental consequences of nitrogen application rates, timing and methods on corn in Ontario. Agric. Syst. 2021, 188, 103018. [Google Scholar] [CrossRef]
- Wiggering, H.; Dalchow, C.; Glemnitz, M.; Helming, K.; Müller, K.; Schultz, A.; Stachow, U.; Zander, P. Indicators for multifunctional land use—Linking socio-economic requirements with landscape potentials. Ecol. Indic. 2006, 6, 238–249. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).