Effect of Fertilization with Urea and Inhibitors on Growth, Yield and CBD Concentration of Hemp (Cannabis sativa L.)
Abstract
1. Introduction
2. Materials and Methods
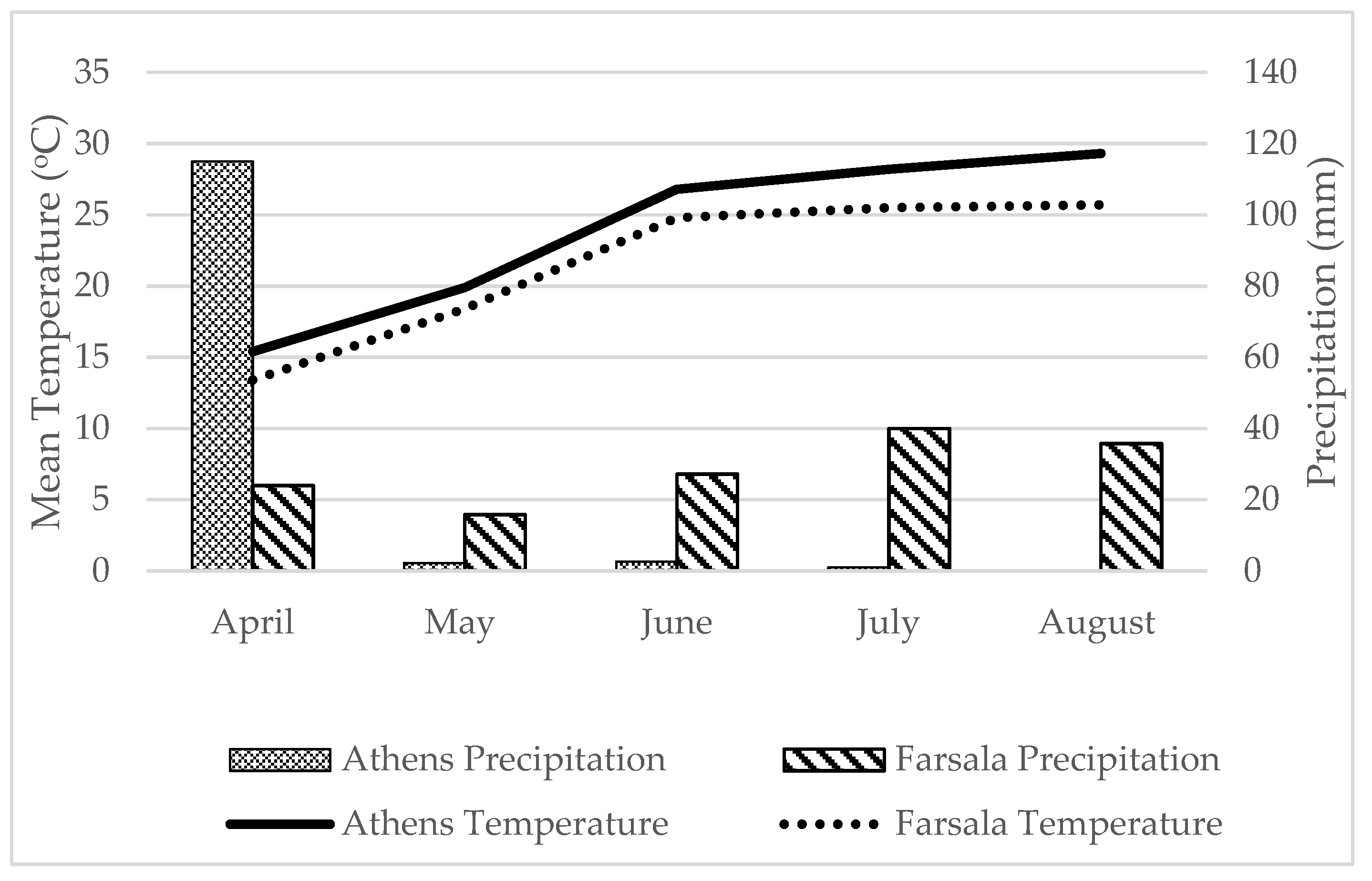
2.1. Location and Experimental Design
2.2. Plant Materials
2.3. Sampling and Methods
2.4. Calculations and Statistics
3. Results
Agronomic Characteristics
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| CBD | Cannabidiol |
| CBN | Cannabinol |
| CBG | Cannabigerol |
| DASs | Days After Sowing |
| LAI | Leaf Area Index |
| NI | Urea with nitrification Inhibitor |
| THC | Tetrahydrocannabinol |
| U | Urea |
| UI | Urea with urease Inhibitor |
References
- Rupasinghe, H.P.V.; Davis, A.; Kumar, S.K.; Murray, B.; Zheljazkov, V.D. Industrial Hemp (Cannabis sativa subsp. sativa) as an Emerging Source for Value-Added Functional Food Ingredients and Nutraceuticals. Molecules 2020, 25, 4078. [Google Scholar] [CrossRef] [PubMed]
- Carus, M.; Karst, S.; Kauffmann, A.; Hobson, J.; Bertucelli, S. The European Hemp Industry: Cultivation, Processing and Applications for Fibres, Shives and Seeds. 2013. Available online: http://eiha.org/media/attach/855/13-06_European_Hemp_Industry.alt.pdf (accessed on 5 June 2017).
- Clarke, R.C.; Merlin, M.D. Cannabis: Evolution and Ethnobotany; University of California Press: Berkeley, CA, USA, 2013. [Google Scholar]
- Johnson, R. Hemp as an Agricultural Commodity. Available online: https://fas.org/sgp/crs/misc/RL32725.pdf (accessed on 23 December 2019).
- Allegret, S. Hemp: A plant with worldwide distribution. In Hemp: Industrial Production and Uses; Bouloc, P., Allegret, S., Eds.; CABI: Bar sur Aube, France, 2013; p. 1. [Google Scholar] [CrossRef]
- European Commission. Common Catalogue of Varieties of Agricultural Plant Species. Available online: https://op.europa.eu/ (accessed on 16 June 2020).
- European Commission. Directive 2012/27/Eu of the European Parliament and of the Council of 25 October 2012 on the Energy Efficiency, Amending Directive 2009/125/EC and 2010/30/EU and Repealing Directives 2004/8/EC and 2006/32/EC (Text with EEA Relevance). Available online: http://eur-lex.europa.eu (accessed on 15 July 2019).
- García-Tejero, I.; Zuazo, V.D.; Sánchez-Carnenero, C.; Hernández, A.; Ferreiro-Vera, C.; Casano, S. Seeking suitable agronomical practices for industrial hemp (Cannabis sativa L.) cultivation for biomedical applications. Ind. Crop. Prod. 2019, 139, 111524. [Google Scholar] [CrossRef]
- Bilalis, D.; Karidogianni, S.; Roussis, I.; Kouneli, V.; Kakabouki, I.; Folina, A. Cannabis sativa L.: A New Promising Crop for Medical and Industrial Use. Bull. Univ. Agric. Sci. Veter-Med. Cluj-Napoca. Hortic. 2019, 76, 145–150. [Google Scholar] [CrossRef]
- Folina, A.; Roussis, I.; Kouneli, V.; Kakabouki, I.; Karidogianni, S.; Bilalis, D. Opportunities for cultivation of medical cannabis (Cannabis sativa L.) in Greece. Sci. Pap. A. Agron. 2019, 62, 293–300. [Google Scholar]
- Amaducci, S.; Scordia, D.; Liu, F.; Zhang, Q.; Guo, H.; Testa, G.; Cosentino, S. Key cultivation techniques for hemp in Europe and China. Ind. Crop. Prod. 2015, 68, 2–16. [Google Scholar] [CrossRef]
- Rodriguez-Leyva, D.; Pierce, G.N. The cardiac and haemostatic effects of dietary hempseed. Nutr. Metab. 2010, 7, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Lukin, A.L.; Bitiutskikh, K.S. Investigation on the use of hemp flour in cookie production. Bulg. J. Agric. Sci. 2017, 23, 664–667. [Google Scholar]
- Gorelick, J.; Bernstein, N. Chemical and Physical Elicitation for Enhanced Cannabinoid Production in Cannabis. In Cannabis Sativa L.—Botany and Biotechnology; Chandra, S., Lata, H., ElSohly, M., Eds.; Springer: Cham, Switzerland, 2017. [Google Scholar]
- Bernstein, N.; Gorelick, J.; Zerahia, R.; Koch, S. Impact of N, P, K, and humic acid supplementation on the chemical profile of medical cannabis (Cannabis sativa L.). Front. Plant Sci. 2019, 10, 736. [Google Scholar] [CrossRef]
- Tang, K.; Struik, P.C.; Yin, X.; Thouminot, C.; Bjelková, M.; Stramkale, V.; Amaducci, S. Comparing hemp (Cannabis sativa L.) cultivars for dual-purpose production under contrasting environments. Ind. Crops Prod. 2016, 87, 33–44. [Google Scholar] [CrossRef]
- Folina, A.; Roussis, I.; Kouneli, V.; Kakabouki, I.; Karidogianni, S.; Bilalis, D.; Kadoglou, N. Evaluation of Woven Agrotextiles in the Development of Hemp (Cannabis sativa L.) in Greenhouse. Bull. Univ. Agric. Sci. Veter-Med. Cluj-Napoca. Hortic. 2020, 77, 53–62. [Google Scholar] [CrossRef]
- Calzolari, D.; Magagnini, G.; Lucini, L.; Grassi, G.; Appendino, G.B.; Amaducci, S. High added-value compounds from Cannabis threshing residues. Ind. Crops Prod. 2017, 108, 558–563. [Google Scholar] [CrossRef]
- Welling, M.T.; Liu, L.; Shapter, T.; Raymond, C.A.; King, G.J. Characterisation of cannabinoid composition in a diverse Cannabis sativa L. germplasm collection. Euphytica 2016, 208, 463–475. [Google Scholar] [CrossRef]
- Toth, J.A.; Stack, G.M.; Cala, A.R.; Carlson, C.H.; Wilk, R.L.; Crawford, J.L.; Viands, D.R.; Philippe, G.; Smart, C.D.; Rose, J.K.; et al. Development and validation of genetic markers for sex and cannabinoid chemotype in Cannabis sativa L. GCB Bioenergy 2020, 12, 213–222. [Google Scholar] [CrossRef]
- Ascrizzi, R.; Ceccarini, L.; Tavarini, S.; Flamini, G.; Angelini, L.G. Valorisation of hemp inflorescence after seed harvest: Cultivation site and harvest time influence agronomic characteristics and essential oil yield and composition. Ind. Crop. Prod. 2019, 139. [Google Scholar] [CrossRef]
- Baldini, M.; Ferfuia, C.; Piani, B.; Sepulcri, A.; Dorigo, G.; Zuliani, F.; Danuso, F.; Cattivello, C. The performance and potentiality of monoecious hemp (Cannabis sativa L.) cultivars as a multipurpose crop. Agronomy 2018, 8, 162. [Google Scholar] [CrossRef]
- Masclaux-Daubresse, C.; Daniel-Vedele, F.; Dechorgnat, J.; Chardon, F.; Gaufichon, L.; Suzuki, A. Nitrogen uptake, assimilation and remobilization in plants: Challenges for sustainable and productive agriculture. Ann. Bot. 2010, 105, 1141–1157. [Google Scholar] [CrossRef] [PubMed]
- Curci, P.L.; Cigliano, R.A.; Zuluaga, D.L.; Janni, M.; Sanseverino, W.; Sonnante, G. Transcriptomic response of durum wheat to nitrogen starvation. Sci. Rep. 2017, 7, 1–14. [Google Scholar] [CrossRef]
- Malceva, M.; Vikmane, M.; Stramkale, V. Changes of photosynthesis-related parameters and productivity of Cannabis sativa under different nitrogen supply. Environ. Exp. Biol. 2011, 9, 61–69. [Google Scholar]
- Ogden, M.; Hoefgen, R.; Roessner, U.; Persson, S.; Khan, G.A. Feeding the walls: How does nutrientavailability regulate cell wall composition? Int. J. Mol. Sci. 2018, 19, 2691. [Google Scholar] [CrossRef]
- Papastylianou, P.; Kakabouki, I.; Travlos, I. Effect of Nitrogen Fertilization on Growth and Yield of Industrial Hemp (Cannabis sativa L.). Not. Bot. HortiAgrobot. Cluj-Napoca 2017, 46, 197–201. [Google Scholar] [CrossRef]
- Campiglia, E.; Radicetti, E.; Mancinelli, R. Plant density and nitrogen fertilization affect agronomic performance of industrial hemp (Cannabis sativa L.) in Mediterranean environment. Ind. Crop. Prod. 2017, 100, 246–254. [Google Scholar] [CrossRef]
- Vera, C.L.; Malhi, S.S.; Raney, J.P.; Wang, Z.H. The effect of N and P fertilization on growth, seed yield and quality of industrial hemp in the Parkland region of Saskatchewan. Can. J. Plant Sci. 2004, 84, 939–947. [Google Scholar] [CrossRef]
- Rioba, N.B.; Itulya, F.M.; Saidi, M.; Dudai, N.; Bernstein, N. Effects of nitrogen, phosphorus and irrigation frequency on essential oil content and composition of sage (Salvia officinalis L.). J. Appl. Res. Med. Aromat. Plants 2015, 2, 21–29. [Google Scholar] [CrossRef]
- Cosentino, S.L.; Riggi, E.; Testa, G.; Scordia, D.; Copani, V. Evaluation of European developed fibre hemp genotypes (Cannabis sativa L.) in semi-arid Mediterranean environment. Ind. Crop. Prod. 2013, 50, 312–324. [Google Scholar] [CrossRef]
- Freney, J.R.; Chen, D.L.; Mosier, A.R.; Rochester, I.J.; Constable, G.A.; Chalk, P.M. Use of nitrification inhibitors to increase fertilizer nitrogen recovery and lint yield in irrigated cotton. Nutr. Cycl. Agroecosystems 1993, 34, 37–44. [Google Scholar] [CrossRef]
- Kakabouki, I.; Karydogianni, S.; Zisi, C.; Folina, A. 2020 Effect of fertilization with N-inhibitors on root and crop development of flaxseed crop (Linumusitatissium L.). AGRIVITA J. Agric. Sci. 2020, 42, 411–424. [Google Scholar]
- Kokko, E.G.; Volkmar, K.M.; Gowen, B.E.; Entz, T. Determination of total root surface area in soil core samples by image analysis. Soil Tillage Res. 1993, 26, 33–43. [Google Scholar] [CrossRef]
- Bilalis, D.; Katsenios, N.; Efthimiadou, A.; Efthimiadis, P.; Karkanis, A. Pulsed electromagnetic fields effect in oregano rooting and vegetative propagation: A potential new organic method. Acta Agric. Scand. Sect. B Plant Soil Sci. 2012, 62, 94–99. [Google Scholar] [CrossRef]
- Bremner, J.M. Determination of nitrogen in soil by Kjedahl method. J. Agric. Sci. 1960, 55, 11–33. [Google Scholar] [CrossRef]
- Amaducci, S.; Zatta, A.; Raffanini, M.; Venturi, G. Characterisation of hemp (Cannabis sativa L.) roots under different growing conditions. Plant Soil 2008, 313, 227–235. [Google Scholar] [CrossRef]
- Folina, A.; Kakabouki, I.; Tourkochoriti, E.; Roussis, I.; Pateroulakis, H.; Bilalis, D. Evaluation of the Effect of Topping on Cannabidiol (CBD) Content in Two Industrial Hemp (Cannabis sativa L.) Cultivars. Bull. Univ. Agric. Sci. Veter-Med. Cluj-Napoca. Hortic. 2020, 77, 46–52. [Google Scholar] [CrossRef]
- Poiša, L.; Adamovičs, A.; Jankauskiene, Z.; Gruzdeviene, E. Industrial hemp (Cannabis sativa L.) as a biomass crop. Treatment and use of organic residues in agriculture: Challenges and opportunities towards sustainable management. In Proceedings of the 14th Ramiran International Conference of the FAO ESCORENA Network on the Recycling of Agricultural, Municipal and Industrial Resiudes in Agriculture, Lisboa, Portugal, 13–15 September 2010; Available online: http://lathemp.lv/wp-content/uploads/2010/11/Ramiran2010_0155_final_Poisa_-Adamovics.pdf (accessed on 16 June 2020).
- Izsáki, Z.; Iványi, I. Effect of Nutrient Supply on the Quality of Linseed and Hempseed. J. Nat. Fibers 2005, 1, 59–75. [Google Scholar] [CrossRef]
- Malhi, S.; Oliver, E.; Mayerle, G.; Kruger, G.; Gill, K.S. Improving effectiveness of seedrow-placed urea with urease inhibitor and polymer coating for durum wheat and canola. Commun. Soil Sci. Plant Anal. 2003, 34, 1709–1727. [Google Scholar] [CrossRef]
- Adesina, I.; Bhowmik, A.; Sharma, H.; Shahbazi, A.A. Review on the Current State of Knowledge of Growing Conditions, Agronomic Soil Health Practices and Utilities of Hemp in the United States. Agriculture 2020, 10, 129. [Google Scholar] [CrossRef]
- Tedeschi, A.; Volpe, M.G.; Polimeno, F.; Siano, F.; Maglione, G.; Di Tommasi, P.; Vasca, E.; Magliulo, V.; Vitale, L. Soil Fertilization with Urea Has Little Effect on Seed Quality but Reduces Soil N2O Emissions from a Hemp Cultivation. Agriculture 2020, 10, 240. [Google Scholar] [CrossRef]
- Beres, J.; Becka, D.; Tomasek, J.; Vasak, J. Effect of autumn nitrogen fertilization on winter oilseed rape growth and yield parameters. Plant, Soil Environ. 2019, 65, 435–441. [Google Scholar] [CrossRef]
- Chimenti, C.; Hall, A. Grain number responses to temperature during floret differentiation in sunflower. Field Crop. Res. 2001, 72, 177–184. [Google Scholar] [CrossRef]
- Egli, D.B. Seed-fill duration and yield of grain crops. Adv. Agron. 2004, 83, 243–279. [Google Scholar]
- Xie, Y.; Gan, Y.; Li, Y.; Niu, J.; Gao, Y.; An, H.; Li, A. Effect of Nitrogen Fertilizer on Nitrogen Accumulation, Translocation, and Use Efficiency in Dryland Oilseed Flax. Agron. J. 2015, 107, 1931–1939. [Google Scholar] [CrossRef]
- Barreyro, R.; De La Plata, U.N.; Ringuelet, J.; Agricola, S. Nitrogen fertilization and yield in oregan (Origanum xapplii). Ciencia e Investigaciónagraria 2005, 32, 34–38. [Google Scholar] [CrossRef]
- Caplan, D.; Dixon, M.; Zheng, Y. Optimal Rate of Organic Fertilizer during the Flowering Stage for Cannabis Grown in Two Coir-based Substrates. HortScience 2017, 52, 1796–1803. [Google Scholar] [CrossRef]
- Lošák, T.; Richter, R. Split nitrogen doses and their efficiency in poppy (Papaver somniferum L.) nutrition. Plant Soil Environ. 2011, 50, 484–488. [Google Scholar] [CrossRef]
- Gagne, S.J.; Stout, J.M.; Liu, E.; Boubakir, Z.; Clark, S.M.; Page, J.E. Identification of olivetolic acid cyclase from Cannabis sativa reveals a unique catalytic route to plant polyketides. Proc. Natl. Acad. Sci. USA 2012, 109, 12811–12816. [Google Scholar] [CrossRef] [PubMed]
| Fertilization | Root Density (mm cm−3) | AMF (%) | Plant Height (cm) | LAI | DM (kg ha−1) | |||||
|---|---|---|---|---|---|---|---|---|---|---|
| Athens | ||||||||||
| “Uso31” | “Fedora 17” | “Uso31” | “Fedora 17” | “Uso31” | “Fedora 17” | “Uso31” | “Fedora 17” | “Uso31” | “Fedora 17” | |
| Control | 5.71 a | 6.12 a | 26.33 a | 27.67 a | 172.6 a | 175.0 a | 3.76 a | 3.93 a | 3.689 a | 3.729 a |
| Urea | 6.11 a | 5.95 a | 20 b | 19.67 b | 176.0 a | 182.0 a | 4.05 b | 4.18 b | 4.002 b | 3.916 b |
| Urea UI | 8.26 b | 8.14 b | 24.33 c | 25.67 c | 182.6 b | 187.6 b | 4.39 b | 4.36 b | 4.212 c | 4.148 c |
| Urea NI + UI | 9.56 c | 9.37 c | 26.33 a | 25.67 c | 190.7 c | 193.0 c | 4.71 c | 4.76 c | 4.400 d | 4.369 d |
| Farsala | ||||||||||
| Control | 5.65 a | 6.06 a | 31 a | 29.7 a | 176.6 a | 177.67 a | 4.16 a | 4.33 a | 3.30 a | 3.940 a |
| Urea | 5.91 a | 6.44 a | 22 b | 22 b | 179.6 a | 183.33 a | 4.55 b | 4.92 b | 4.186 b | 4.117 b |
| Urea UI | 7.64 b | 7.71 b | 24 c | 25 c | 186.3 b | 188.33 b | 4.82 b | 4.80 b | 4.413 c | 4.342 c |
| Urea NI + UI | 8.51 c | 8.70 c | 26.7 ac | 27 ac | 195.3 c | 197.00 c | 5.13 c | 5.12 c | 4.605 d | 4.694 d |
| FFert | 92.95 *** | 12.43 *** | 34.44 *** | 39.36 *** | 81.42 *** | |||||
| FVariety | ns | ns | 4.86 * | ns | ns | |||||
| FLocation | 4.30 * | ns | 5.13 * | 61.70 *** | 44.49 *** | |||||
| FFert × Variety | ns | ns | ns | ns | ns | |||||
| FFert × Location | ns | ns | ns | ns | ns | |||||
| FVariety × Location | ns | ns | ns | ns | ns | |||||
| FFert × Variety × Location | ns | ns | ns | ns | ns | |||||
| Fertilization | No Buds/Plant | Weight/ Bud (g) | Bud Length (cm) | Bud Compact Index (g cm−1) | ||||
|---|---|---|---|---|---|---|---|---|
| Athens | ||||||||
| “Uso 31” | “Fedora 17” | “Uso 31” | “Fedora 17” | “Uso 31” | “Fedora 17” | “Uso 31” | “Fedora 17” | |
| Control | 4.27 a | 4.47 a | 168 a | 161.67 a | 30.33 a | 28.33 a | 5.57 a | 5.70 a |
| Urea | 5.03 b | 4.77 b | 176.67 b | 171.33 b | 31.33 a | 30 a | 5.72 a | 5.78 a |
| Urea UI | 5.43 c | 5.27 c | 181.67 c | 175.33 bc | 32.33 b | 32 b | 5.68 a | 5.58 a |
| Urea NI+UI | 5.90 d | 5.80 d | 181.33 c | 181.67 c | 32 b | 30.67 a | 5.75 a | 6.02 b |
| Farsala | ||||||||
| Control | 4.53 a | 4.77 a | 165 a | 161.67 a | 30.33 a | 29.67 a | 5.46 a | 5.46 a |
| Urea | 5.27 b | 4.97 b | 172.33 b | 169 b | 32.33 a | 32.67 a | 5.36 a | 5.18 a |
| Urea UI | 5.60 c | 5.33 c | 177.33 c | 170.33 bc | 33.33 b | 35.67 b | 5.35 a | 4.79 a |
| Urea NI+UI | 6.03 d | 5.83 d | 177 c | 179.33 c | 34.67 b | 36.33 b | 5.16 a | 4.96 a |
| FFert | 64.85 *** | 11.91 *** | 3.26 * | ns | ||||
| FVariety | ns | ns | ns | ns | ||||
| FPlace | 5.75 ** | ns | 5.27 * | 7.51 ** | ||||
| FFert × Variety | ns | ns | ns | ns | ||||
| FFert × Location | ns | ns | ns | ns | ||||
| FVariety × Location | ns | ns | ns | ns | ||||
| FFert × Variety × Location | ns | ns | ns | ns | ||||
| Fertilization | 1000 Seed Weight (g) | Seed Yield (kg ha−1) | Seed Protein (%) | Protein Yield (kg ha−1) | ||||
|---|---|---|---|---|---|---|---|---|
| Athens | ||||||||
| “Uso 31” | “Fedora 17” | “Uso 31” | “Fedora 17” | “Uso 31” | “Fedora 17” | “Uso 31” | “Fedora 17” | |
| Control | 13.13 a | 13.60 a | 2335 a | 2423 a | 16.83 a | 17.07 a | 393.27 a | 412.96 a |
| Urea | 13.55 b | 13.98 b | 2514 b | 2580 b | 17.68 ab | 17.77 ab | 444.11 b | 457.66 b |
| Urea UI | 14 c | 14.23 c | 2721 c | 2703 c | 18.36 bc | 18.36 bc | 499.42 c | 495.56 c |
| Urea NI+UI | 14.32 c | 14.56 c | 2895 c | 2860 c | 18.77 c | 19.26 c | 544.15 d | 550.83 d |
| Farsala | ||||||||
| Control | 13.67 a | 14.04 a | 2149 a | 2220 a | 17 a | 17.06 a | 363.63 a | 379.12 a |
| Urea | 14.19 b | 14.59 b | 2325 b | 2385 b | 17.74 ab | 17.52 ab | 411.94 b | 417.93 b |
| Urea UI | 15.17 c | 14.94 c | 2530 c | 2512 c | 18.63 bc | 18.61 bc | 470.62 c | 467.33 c |
| Urea NI+UI | 15.65 c | 15.23 c | 2730 c | 2721 c | 19.32 c | 19.25 c | 526.14 d | 524.23 d |
| FFert | 27.42 ** | 14.48 ** | 9.47 *** | 32.32 *** | ||||
| FVariety | ns | ns | ns | ns | ||||
| FPlace | 47.12 *** | 9.69 ** | ns | 6.87 * | ||||
| FFert × Variety | ns | ns | ns | ns | ||||
| FFert × Location | ns | ns | ns | ns | ||||
| FVariety × Location | ns | ns | ns | ns | ||||
| FFert × Variety × Location | ns | ns | ns | ns | ||||
| Fertilization | CBD Content (%) | CBD Yield/Plant (g) | ||
|---|---|---|---|---|
| Athens | ||||
| “Uso 31” | “Fedora 17” | “Uso 31” | “Fedora 17” | |
| Control | 1.37 a | 1.33 a | 9.83 a | 9.59 a |
| Urea | 1.41 ab | 1.41 ab | 12.49 b | 11.55 b |
| Urea UI | 1.54 bc | 1.51 bc | 15.15 c | 13.98 c |
| Urea NI+UI | 1.66 c | 1.53 c | 17.65 d | 16.07 d |
| Farsala | ||||
| Control | 1.29 a | 1.38 a | 9.65 a | 10.61 a |
| Urea | 1.35 ab | 1.45 ab | 12.24 b | 12.15 b |
| Urea UI | 1.54 bc | 1.53 bc | 15.18 c | 13.93 c |
| Urea NI+UI | 1.69 c | 1.64 c | 18 d | 17.15 d |
| FFert | 10.23 *** | 78.037 *** | ||
| FVariety | ns | ns | ||
| FPlace | ns | ns | ||
| FFert × Variety | ns | ns | ||
| FFert × Location | ns | ns | ||
| FVariety × Location | ns | ns | ||
| FFert × Variety × Location | ns | ns | ||
| Fertilization | Oil Content (%) | Oil Yield (kg ha−1) | ||
|---|---|---|---|---|
| Athens | ||||
| “Uso 31” | “Fedora 17” | “Uso 31” | “Fedora 17” | |
| Control | 26.47 a | 24.83 a | 618.89 a | 599.38 a |
| Urea | 27.68 b | 26.83 b | 695.52 b | 691.95 b |
| Urea UI | 29.84 c | 27.66 c | 811.54 c | 748.47 c |
| Urea NI + UI | 29.90 c | 28.34 c | 865.27 d | 811.08 d |
| Farsala | ||||
| Control | 25.96 a | 25.69 a | 557.81 a | 570.31 a |
| Urea | 27.97 b | 27.23 b | 650.24 b | 648.25 b |
| Urea UI | 29.26 c | 28.29 c | 739.98 c | 710.50 c |
| Urea NI + UI | 29.48 c | 29.38 c | 804.45 d | 799.78 d |
| FFert | 32.53 *** | 38.73 *** | ||
| FVariety | 14.02 *** | ns | ||
| FPlace | ns | 7.71 ** | ||
| FFert × Variety | ns | ns | ||
| FFert × Location | ns | ns | ||
| FVariety × Location | ns | ns | ||
| FFert × Variety × Location | ns | ns | ||
| R Density | AMF | Plant Height | LAI | DM | 1000 Seed Weight | Seed Yield | Oil Content | Oil Yield | No Buds/Plant | Weight/Bud (g) | Bud Length (cm) | Bud Comp. Index | CBD (%) | CBD Yield/Plant | Seed Protein (%) | Protein Yield | THC Content | THC Yield/Plant (g) | Harvest Index | CBD/THC | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| R Density | 1 | 0.04 ns | 0.74 *** | 0.61 *** | 0.75 *** | 0.52 *** | 0.61 *** | 0.67 *** | 0.74 *** | 0.81 *** | 0.59 *** | 0.40 ** | −0.10 ns | 0.63 *** | 0.84 *** | 0.62 *** | 0.75 *** | −0.72 *** | −0.24 ns | 0.13 ns | 0.73 *** |
| AMF (%) | 0.04 ns | 1 | 0.11 ns | −0.04 ns | −0.06 ns | 0.10 ns | 0.04 ns | −0.22 ns | −0.05 ns | −0.16 ns | −0.24 ns | −0.31 * | 0.20 ns | 0.18 ns | −0.01 ns | −0.21 ns | −0.06 ns | −0.04 ns | −0.41 ** | 0.06 ns | 0.18 ns |
| Plant height (cm) | 0.74 *** | 0.11 ns | 1 | 0.69 *** | 0.76 *** | 0.70 *** | 0.52 *** | 0.52 *** | 0.61 *** | 0.65 *** | 0.58 *** | 0.35 * | −0.05 ns | 0.64 *** | 0.77 *** | 0.65 *** | 0.69 *** | −0.78 *** | −0.49 *** | 0.01 ns | 0.81 *** |
| LAI | 0.61 *** | −0.04 ns | 0.69 *** | 1 | 0.83 *** | 0.76 *** | 0.33 * | 0.60 *** | 0.49 *** | 0.75 *** | 0.32* | 0.55*** | −0.38** | 0.54 *** | 0.70 *** | 0.58 *** | 0.52 *** | −0.68 *** | −0.30 * | −0.23 ns | 0.66 *** |
| DM (kg ha−1) | 0.75 *** | −0.06 ns | 0.76 *** | 0.83 *** | 1 | 0.78 *** | 0.47 *** | 0.75 *** | 0.66 *** | 0.89 *** | 0.50 *** | 0.50 *** | −0.24 ns | 0.58 *** | 0.83 *** | 0.58 *** | 0.62 *** | −0.84 *** | −0.36 * | −0.21 ns | 0.80 *** |
| 1000 seed weight | 0.52 *** | 0.10 ns | 0.70 *** | 0.76 *** | 0.78 *** | 1 | 0.52 *** | 0.54 *** | 0.61 *** | 0.66 *** | 0.36 * | 0.21 ns | −0.02 ns | 0.44 ** | 0.62 *** | 0.39 ** | 0.57 *** | −0.73 *** | −0.48 *** | 0.01 ns | 0.69 *** |
| Seed Yield | 0.61 *** | 0.04 ns | 0.52 *** | 0.33 * | 0.47 *** | 0.52 *** | 1 | 0.48 *** | 0.93 ** | 0.56 *** | 0.43 ** | −0.19 ns | 0.44 ** | 0.34 * | 0.55 *** | 0.35 * | 0.90 *** | −0.71 *** | −0.49 *** | 0.77 *** | 0.61 *** |
| Oil content (%) | 0.67 *** | −0.22 ns | 0.52 *** | 0.60 *** | 0.75 *** | 0.54 *** | 0.48 *** | 1 | 0.77 *** | 0.81 *** | 0.68 *** | 0.41 ** | −0.08 ns | 0.51 *** | 0.80 *** | 0.44 ** | 0.56 *** | −0.74 *** | −0.17 ns | −0.02 ns | 0.68 *** |
| Oil Yield (kg ha−1) | 0.74 *** | −0.05 ns | 0.61 *** | 0.49 *** | 0.66 *** | 0.61 *** | 0.93 ** | 0.77 *** | 1 | 0.75 *** | 0.61 *** | 0.03 ns | 0.30 * | 0.47 *** | 0.74 *** | 0.45 *** | 0.89 *** | −0.83 *** | −0.43 ** | 0.55 *** | 0.73 *** |
| No buds/plant | 0.81 *** | −0.16 ns | 0.65 *** | 0.75 *** | 0.89 *** | 0.66 *** | 0.56 *** | 0.81 *** | 0.75 *** | 1 | 0.52 *** | 0.45 *** | −0.19 ns | 0.57 *** | 0.89 *** | 0.55 *** | 0.67 *** | −0.81 *** | −0.19 ns | −0.02 ns | 0.74 *** |
| Weight/bud (g) | 0.59 *** | −0.24 ns | 0.58 *** | 0.32 * | 0.50 *** | 0.36 * | 0.43 ** | 0.68 *** | 0.61 *** | 0.52 *** | 1 | 0.27 ns | 0.20 ns | 0.32 * | 0.62 *** | 0.49 *** | 0.55 *** | −0.55 *** | −0.04 ns | 0.09 ns | 0.53 *** |
| Bud length (cm) | 0.40 ** | −0.31 * | 0.35 * | 0.55 *** | 0.50 *** | 0.21 ns | −0.19 ns | 0.41 ** | 0.03 ns | 0.45 *** | 0.27 ns | 1 | −0.88 *** | 0.36 * | 0.45 *** | 0.55 *** | 0.12 ns | −0.26 ns | 0.12 ns | −0.56 *** | 0.32 * |
| Budcomp. index | −0.10 ns | 0.20 ns | −0.05 ns | −0.38 ** | −0.24 ns | −0.02 ns | 0.44 ** | −0.08 ns | 0.30 * | −0.19 ns | 0.20 ns | −0.88 *** | 1 | −0.22 ns | −0.15 ns | −0.28 ns | 0.19 ns | −0.03 ns | −0.17 ns | 0.64 *** | −0.05 ns |
| CBD (%) | 0.63 *** | 0.18 ns | 0.64 *** | 0.54 *** | 0.58 *** | 0.44 ** | 0.34 * | 0.51 *** | 0.47 *** | 0.57 *** | 0.32 * | 0.36 * | −0.22 ns | 1 | 0.86 *** | 0.37 * | 0.43 ** | −0.61 *** | −0.37 * | −0.03 ns | 0.80 *** |
| CBD yield/plant | 0.84 *** | −0.01 ns | 0.77 *** | 0.70 *** | 0.83 *** | 0.62 *** | 0.55 *** | 0.80 *** | 0.74 *** | 0.89 *** | 0.62 *** | 0.45 *** | −0.15 ns | 0.86 *** | 1 | 0.56 *** | 0.67 *** | −0.82 *** | −0.31 * | 0.01 ns | 0.89 *** |
| Seed protein (%) | 0.62 *** | −0.21 ns | 0.65 *** | 0.58 *** | 0.58 *** | 0.39 ** | 0.35 * | 0.44 ** | 0.45 *** | 0.55 *** | 0.49 *** | 0.55 *** | −0.28 ns | 0.37 * | 0.56 *** | 1 | 0.72 *** | −0.44 *** | −0.03 ns | −0.03 ns | 0.45 *** |
| Protein yield | 0.75 *** | −0.06 ns | 0.69 *** | 0.52 *** | 0.62 *** | 0.57 *** | 0.90 *** | 0.56 *** | 0.89 *** | 0.67 *** | 0.55 *** | 0.12 ns | 0.19 ns | 0.43 ** | 0.67 *** | 0.72 *** | 1 | −0.73 *** | −0.38 ** | 0.54 *** | 0.66 *** |
| THC content (%) | −0.72 *** | −0.04 ns | −0.78 *** | −0.68 *** | −0.84 *** | −0.73 *** | −0.71 *** | −0.74 *** | −0.83 *** | −0.81 *** | −0.55 *** | −0.26 ns | −0.03 ns | −0.61 *** | −0.82 *** | −0.44 ** | −0.73 *** | 1 | 0.68 *** | −0.17 ns | −0.92 ** |
| THC yield/plant | −0.24 ns | −0.41 ** | −0.49 *** | −0.30 * | −0.36 * | −0.48 *** | −0.49 *** | −0.17 ns | −0.43 ** | −0.19 ns | −0.04 ns | 0.12 ns | −0.17 ns | −0.37 * | −0.31* | −0.03 ns | −0.38 ** | 0.68 *** | 1 | −0.28 ns | −0.68 *** |
| Harvest index | 0.13 ns | 0.06 ns | 0.01 ns | −0.23 ns | −0.21 ns | 0.01 ns | 0.77 *** | −0.02 ns | 0.55 *** | −0.02 ns | 0.09 ns | −0.56 *** | 0.64 *** | −0.03 ns | 0.01 ns | −0.03 ns | 0.54 *** | −0.17 ns | −0.28 ns | 1 | 0.09 ns |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kakabouki, I.; Kousta, A.; Folina, A.; Karydogianni, S.; Zisi, C.; Kouneli, V.; Papastylianou, P. Effect of Fertilization with Urea and Inhibitors on Growth, Yield and CBD Concentration of Hemp (Cannabis sativa L.). Sustainability 2021, 13, 2157. https://doi.org/10.3390/su13042157
Kakabouki I, Kousta A, Folina A, Karydogianni S, Zisi C, Kouneli V, Papastylianou P. Effect of Fertilization with Urea and Inhibitors on Growth, Yield and CBD Concentration of Hemp (Cannabis sativa L.). Sustainability. 2021; 13(4):2157. https://doi.org/10.3390/su13042157
Chicago/Turabian StyleKakabouki, Ioanna, Angeliki Kousta, Antigolena Folina, Stella Karydogianni, Charikleia Zisi, Varvara Kouneli, and Panayiota Papastylianou. 2021. "Effect of Fertilization with Urea and Inhibitors on Growth, Yield and CBD Concentration of Hemp (Cannabis sativa L.)" Sustainability 13, no. 4: 2157. https://doi.org/10.3390/su13042157
APA StyleKakabouki, I., Kousta, A., Folina, A., Karydogianni, S., Zisi, C., Kouneli, V., & Papastylianou, P. (2021). Effect of Fertilization with Urea and Inhibitors on Growth, Yield and CBD Concentration of Hemp (Cannabis sativa L.). Sustainability, 13(4), 2157. https://doi.org/10.3390/su13042157







