Effect of Woody Biomass Gasification Process Conditions on the Composition of the Producer Gas
Abstract
1. Introduction
2. Materials and Methods
2.1. Biomass Characterization
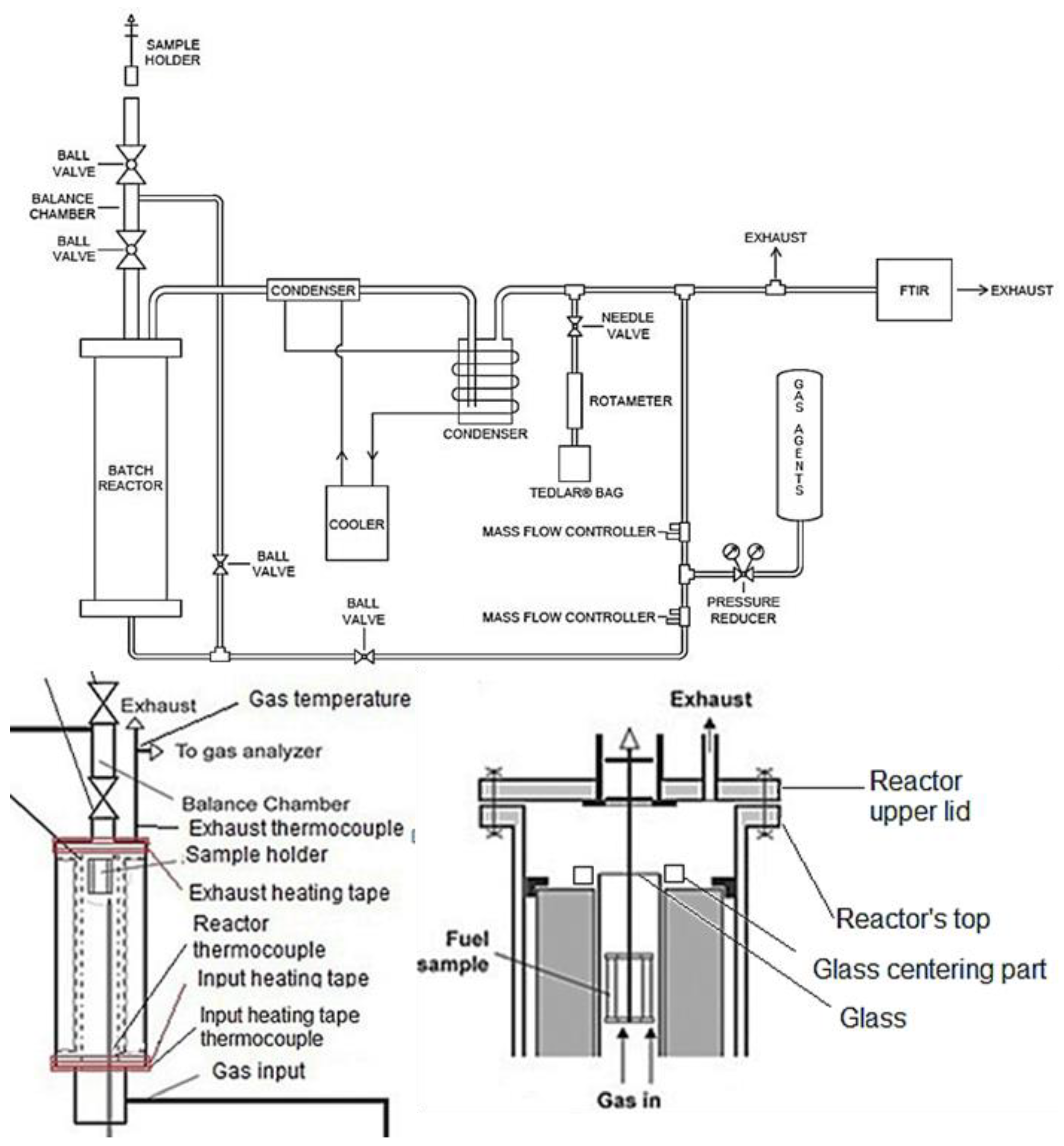
2.2. Experimental Set-Up
2.3. Equivalence Ratio and Stoichiometric Reactions
| General formula | |
| Spruce | |
| Alder | |
| Pine |
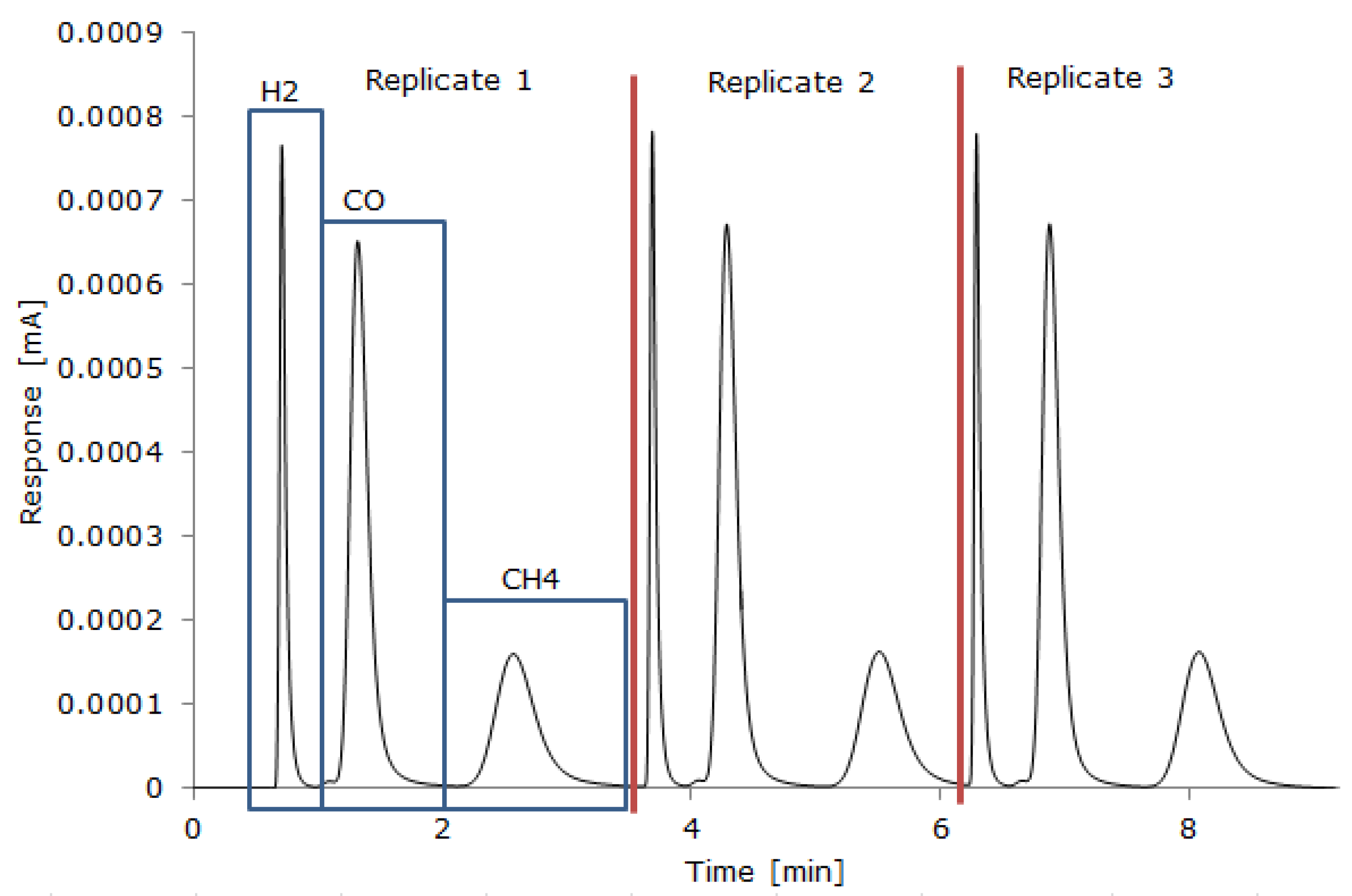
2.4. Gas Measurement
3. Results and Discussion
3.1. Woody Biomass Composition
3.2. Producer Gas Composition
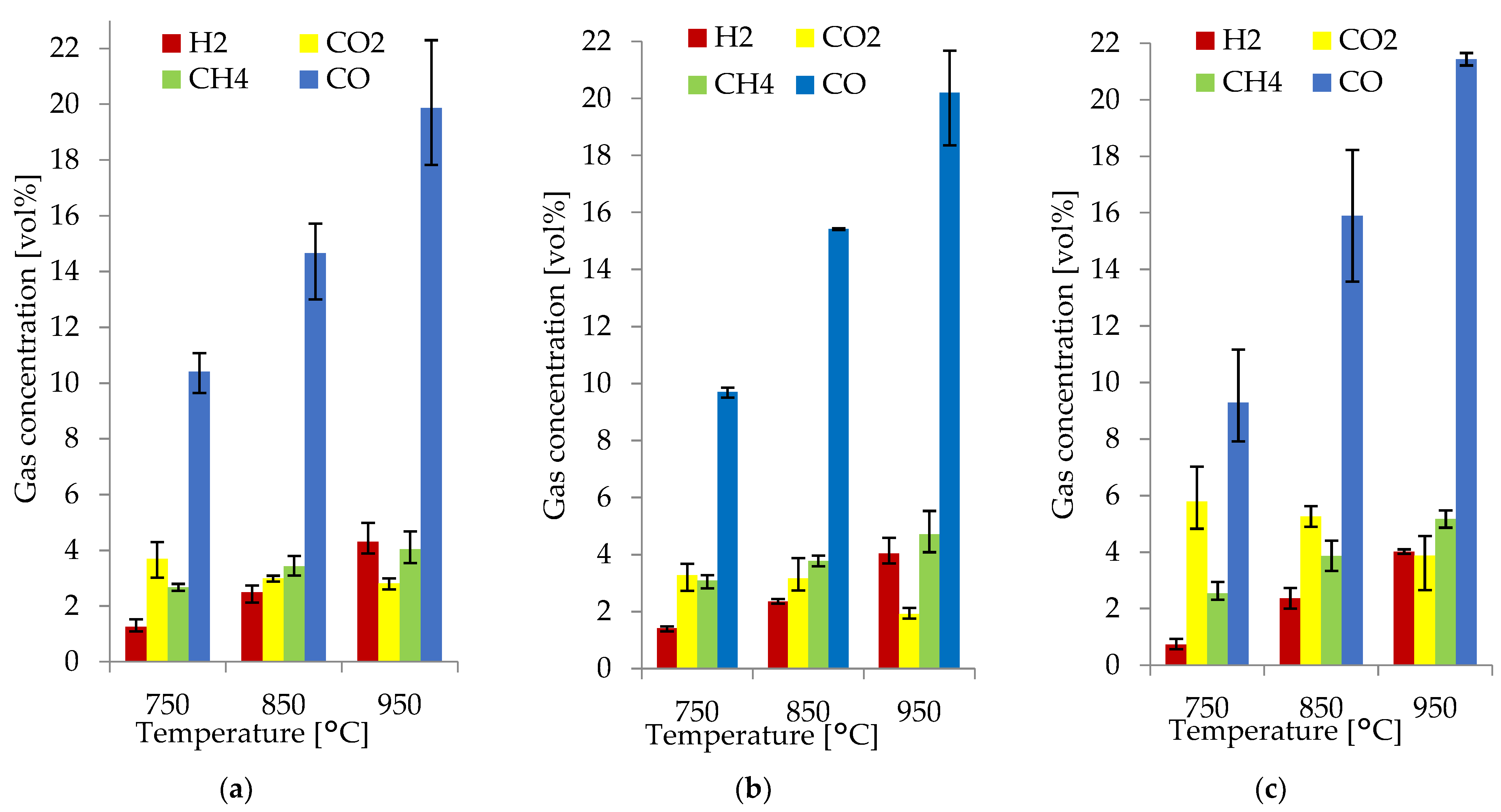
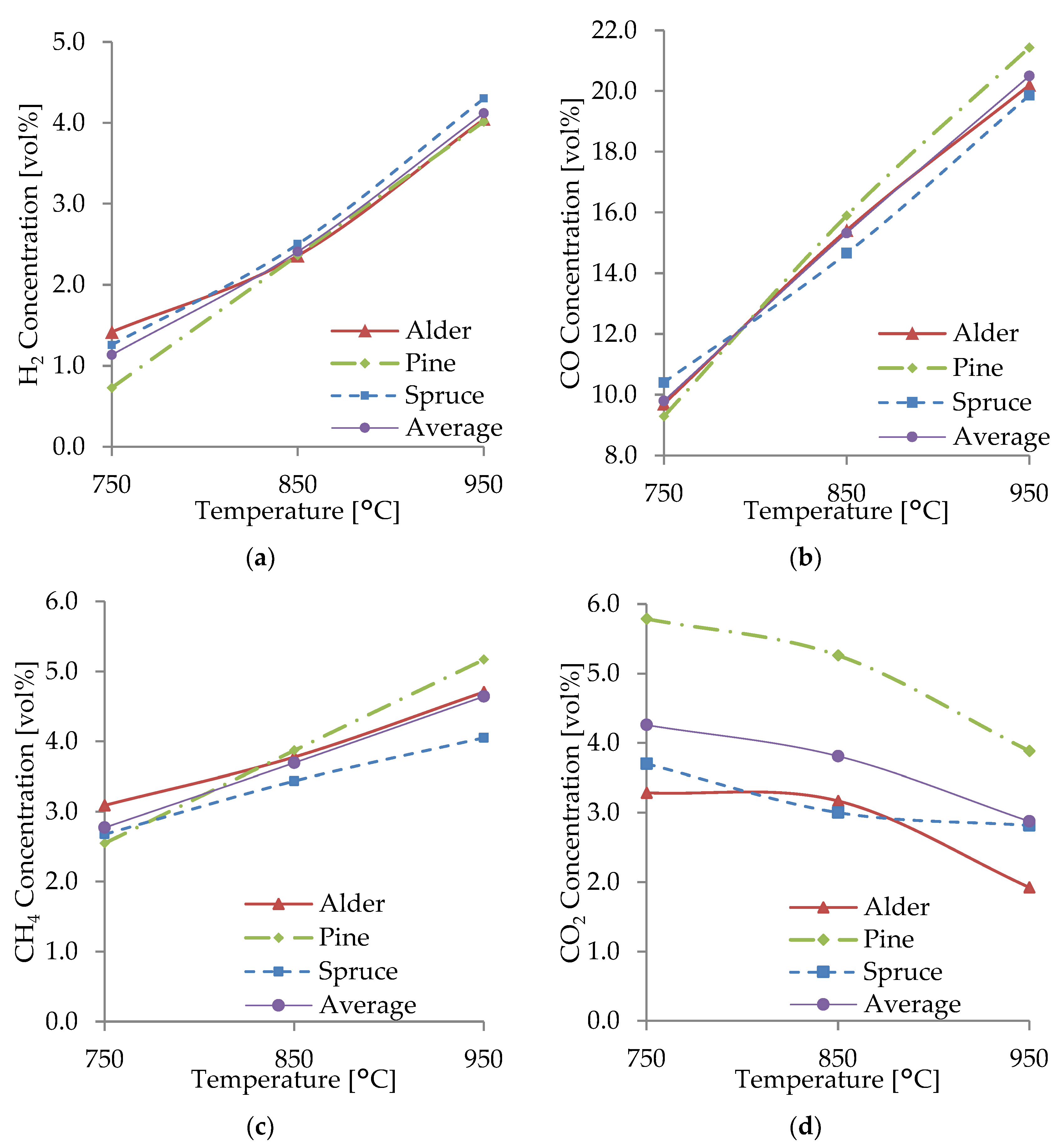
3.3. Effect of Temperature
| Primary reactions | (R1) | Boudouard | (+172.0 kJ/mol) | |
| (R2) | Water–gas | (+131.0 kJ/mol) | ||
| (R3) | Water–gas shift | (−41.0 kJ/mol) | ||
| Secondary reactions | (R4) | Methane dry reforming | (+247.0 kJ/mol) | |
| (R5) | Methane steam reforming | (+206.0 kJ/mol) | ||
| (R6) | Methane formation | (−75.0 kJ/mol) |
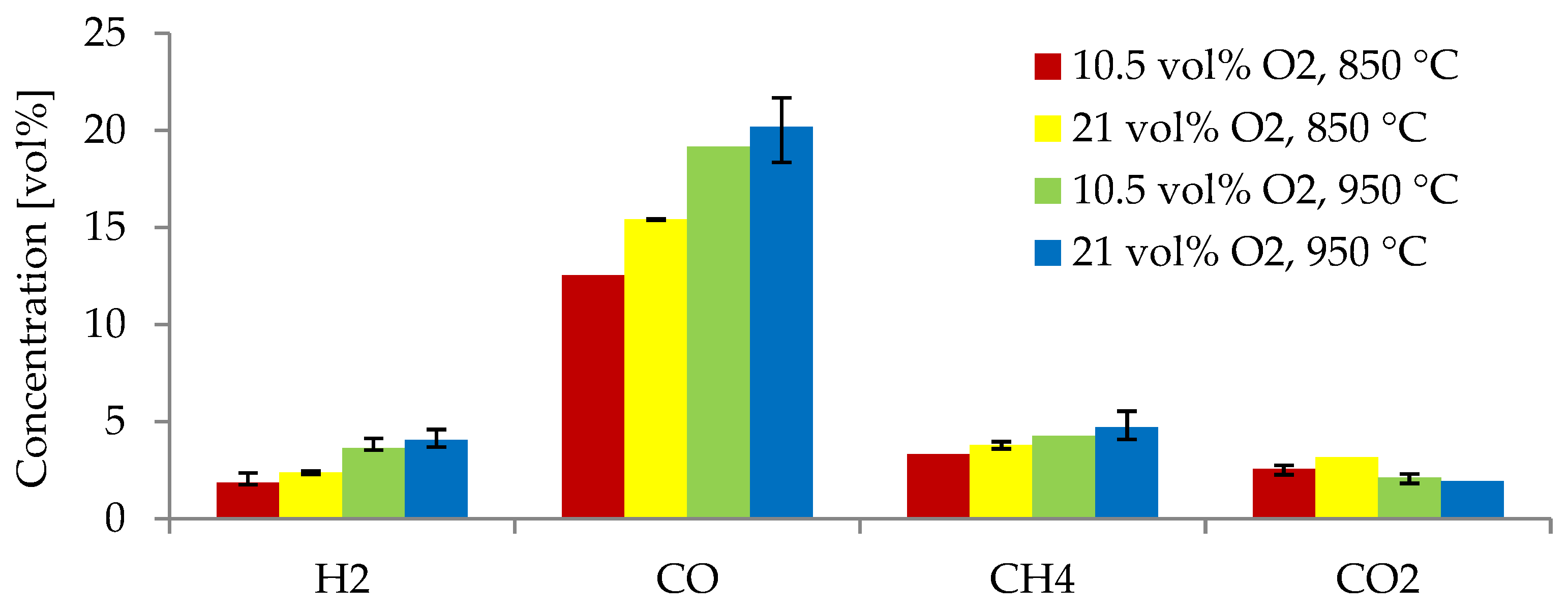
3.4. Effect of the ER
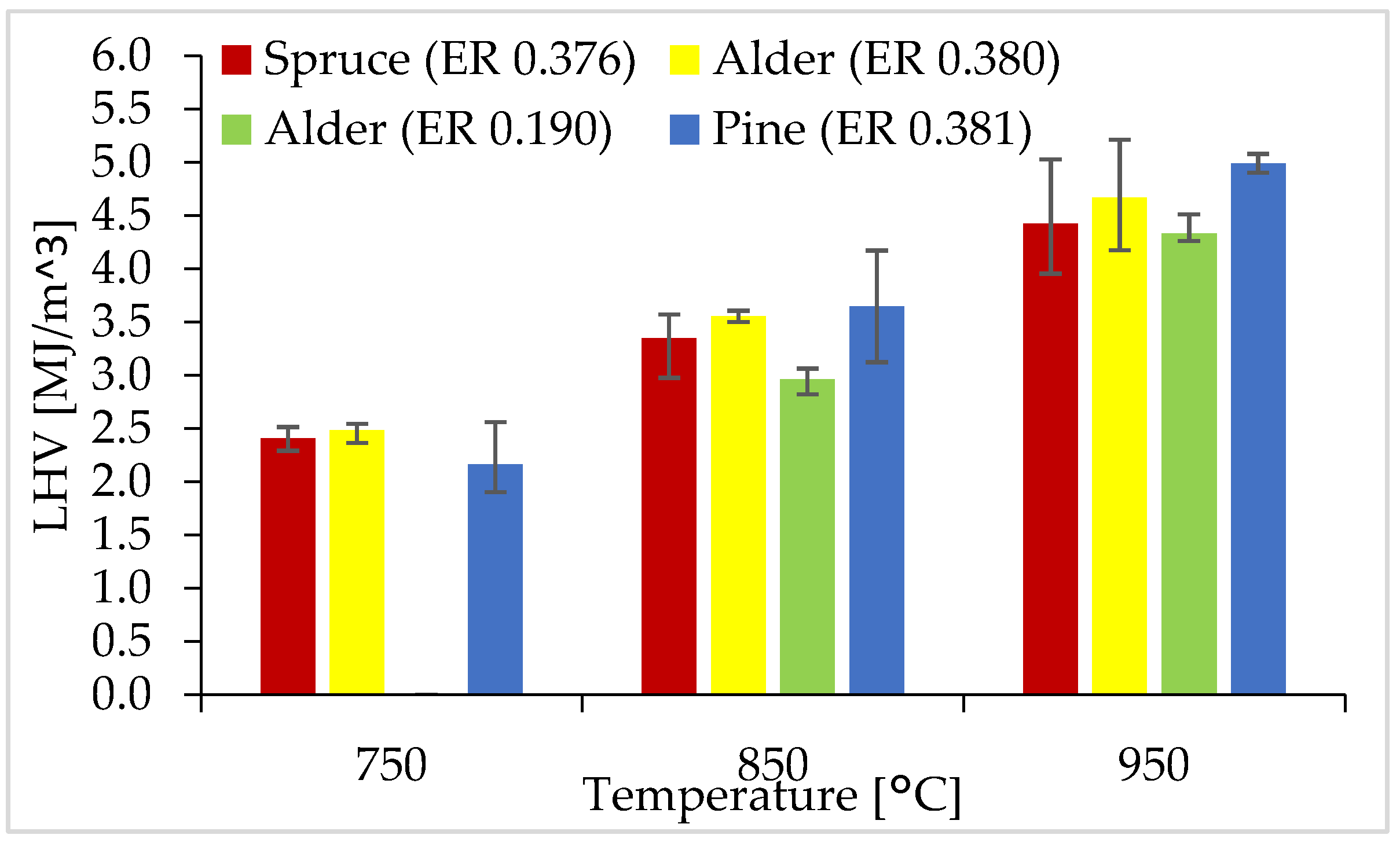
3.5. LHV
3.6. Total Organic Compounds
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Nomenclature
| A/F | Air-to-fuel ratio |
| ER | Equivalence ratio |
| FTIR | Fourier-transform infrared spectroscopy |
| GC-TCD | Gas chromatography with thermal conductivity detector |
| HHV | Gross of higher heating value |
| LHV | Net or lower heating value |
| RSD | Relative standard deviation |
| TOC | Total organic compounds |
References
- IRENA. REthinking Energy 2017: Accelerating the Global Energy Transformation; IRENA: Abu Dhabi, United Arab Emirates, 2017; Volume 55. [Google Scholar]
- IEA. Energy Atlas World Energy Statistics & World Energy Balances; IEA: Paris, France, 2020. [Google Scholar]
- International Renewable Energy Agency. National Renewably Energy Action Plan (NREAP) 2020; International Renewable Energy Agency: Abu Dhabi, United Arab Emirates, 2020. [Google Scholar]
- Republic of Estonia Ministry of Economic Affairs and Communications. National Development Plan of the Energy Sector until 2030; Republic of Estonia Ministry of Economic Affairs and Communications: Tallinn, Estonia, 2017.
- Statistics Estonia Eesti Statistika Kvartalikiri. 2/18. Quarterly Bulletin of Statistics Estonia—Statistics Estonia. 2018. Available online: https://www.stat.ee/sites/default/files/2020-07/Quarterly_Bulletin_2-2018.pdf (accessed on 1 August 2021).
- Strezov, V.; Evans, T.J. Biomass Processing Technologies; CRC Press: Boca Raton, FL, USA, 2014. [Google Scholar]
- Ahmad, A.A.; Zawawi, N.A.; Kasim, F.; Inayat, A.; Khasri, A. Assessing the gasification performance of biomass: A review on biomass gasification process conditions, optimization and economic evaluation. Renew. Sustain. Energy Rev. 2016, 53, 1333–1347. [Google Scholar] [CrossRef]
- Higman, C.; van der Burgt, M. Gasification, 2nd ed.; Gulf Professional Publishing: Burlington, MA, USA, 2008; ISBN 9780750685283. [Google Scholar]
- Sansaniwal, S.K.; Pal, K.; Rosen, M.; Tyagi, S. Recent advances in the development of biomass gasification technology: A comprehensive review. Renew. Sustain. Energy Rev. 2017, 72, 363–384. [Google Scholar] [CrossRef]
- Sepe, A.M.; Li, J.; Paul, M.C. Assessing biomass steam gasification technologies using a multi-purpose model. Energy Convers. Manag. 2016, 129, 216–226. [Google Scholar] [CrossRef]
- Zhang, L.; Xu, C.C.; Champagne, P. Overview of recent advances in thermo-chemical conversion of biomass. Energy Convers. Manag. 2010, 51, 969–982. [Google Scholar] [CrossRef]
- La Villeta, M.; Costa, M.; Massarotti, N. Modelling approaches to biomass gasification-review. Renew. Sustain. Energy Rev. 2017, 74, 71–88. [Google Scholar] [CrossRef]
- Ruiz, J.; Juárez, M.; Morales, M.; Muñoz, P.; Mendívil, M. Biomass gasification for electricity generation: Review of current technology barriers. Renew. Sustain. Energy Rev. 2013, 18, 174–183. [Google Scholar] [CrossRef]
- Mahinpey, N.; Gomez, A. Review of gasification fundamentals and new findings: Reactors, feedstock, and kinetic studies. Chem. Eng. Sci. 2016, 148, 14–31. [Google Scholar] [CrossRef]
- Pachapur, V.L.; Kutty, P.; Pachapur, P.; Brar, S.K.; Le Bihan, Y.; Galvez-Cloutier, R.; Buelna, G. Seed Pretreatment for Increased Hydrogen Production Using Mixed-Culture Systems with Advantages over Pure-Culture Systems. Energies 2019, 12, 530. [Google Scholar] [CrossRef]
- Cao, L.; Yu, I.K.; Xiong, X.; Tsang, D.C.; Zhang, S.; Clark, J.H.; Hu, C.; Ng, Y.H.; Shang, J.; Ok, Y.S. Biorenewable hydrogen production through biomass gasification: A review and future prospects. Environ. Res. 2020, 186, 109547. [Google Scholar] [CrossRef]
- Hu, Y.; Cheng, Q.; Wang, Y.; Guo, P.; Wang, Z.; Liu, H.; Akbari, A. Investigation of Biomass Gasification Potential in Syngas Production: Characteristics of Dried Biomass Gasification Using Steam as the Gasification Agent. Energy Fuels 2019, 34, 1033–1040. [Google Scholar] [CrossRef]
- He, Q.; Guo, Q.; Umeki, K.; Ding, L.; Wang, F.; Yu, G. Soot formation during biomass gasification: A critical review. Renew. Sustain. Energy Rev. 2021, 139, 110710. [Google Scholar] [CrossRef]
- Wijayanta, A.T.; Alam, S.; Nakaso, K.; Fukai, J.; Shimizu, M. Optimized combustion of biomass volatiles by varying O2 and CO2 levels: A numerical simulation using a highly detailed soot formation reaction mechanism. Bioresour. Technol. 2012, 110, 645–651. [Google Scholar] [CrossRef]
- Ferreiro, A.; Segurado, R.; Costa, M. Modelling soot formation during biomass gasification. Renew. Sustain. Energy Rev. 2020, 134, 110380. [Google Scholar] [CrossRef]
- Sulg, M.; Konist, A.; Järvik, O. Characterization of different wood species as potential feedstocks for gasification. Agron. Res. 2021, 19, 276–299. [Google Scholar]
- Chhiti, Y.; Salvador, S.; Commandré, J.-M.; Broust, F.; Couhert, C. Wood Bio-Oil Noncatalytic Gasification: Influence of Temperature, Dilution by an Alcohol and Ash Content. Energy Fuels 2011, 25, 345–351. [Google Scholar] [CrossRef][Green Version]
- Cascante Cirici, P. Biomass and Oil Shale Co-Pyrolysis. Master’s Thesis, Tallinn University of Technology, Tallinn, Estonia, June 2019. [Google Scholar]
- Proto, A.R.; Longo, L.; Gambella, F.; Zimbalatti, G.; Macrì, G.; Gallucci, F.; Caruso, L.; Salerno, M.; Colantoni, A. Energetic Characteristics of Syngas Obtained from Gasification of Hazelnut Prunings. Procedia—Soc. Behav. Sci. 2016, 223, 835–840. [Google Scholar] [CrossRef]
- Demirbas, M.F. Characterization of Bio-oils from Spruce Wood (Picea orientalisL.) via Pyrolysis. Energy Sources Part A Recover. Util. Environ. Eff. 2010, 32, 909–916. [Google Scholar] [CrossRef]
- Branca, C.; Di Blasi, C.; Galgano, A.; Broström, M. Effects of the Torrefaction Conditions on the Fixed-Bed Pyrolysis of Norway Spruce. Energy Fuels 2014, 28, 5882–5891. [Google Scholar] [CrossRef]
- Formowitz, B. Pyrolysis behavior of modified grey alder wood samples by TG-MS and TGA technoques. In Proceedings of the 18th European Biomass Conference and Exhibition, Lyon, France, 3–7 May 2010; Volume 3000, pp. 3–7. [Google Scholar]
- Suttibak, S.; Sriprateep, K.; Pattiya, A. Production of Bio-oil from Pine Sawdust by Rapid Pyrolysis in a Fluidized-bed Reactor. Energy Sources Part A Recover. Util. Environ. Eff. 2015, 37, 1440–1446. [Google Scholar] [CrossRef]
- Purevsuren, B.; Dashzeveg, O.; Alyeksandr, A.; Janchig, N.; Soninkhuu, J. Pyrolysis of pine wood and characterisation of solid and liquid products. Mong. J. Chem. 2018, 19, 24–31. [Google Scholar] [CrossRef]
- Sethuraman, S.; Van Huynh, C.; Kong, S.-C. Producer Gas Composition and NOx Emissions from a Pilot-Scale Biomass Gasification and Combustion System Using Feedstock with Controlled Nitrogen Content. Energy Fuels 2011, 25, 813–822. [Google Scholar] [CrossRef]
- Inayat, M.; Sulaiman, S.A. Effect of Blending Ratio on Quality of Producer Gas From Co-Gasification of Wood and Coconut Residual. MATEC Web Conf. 2018, 225, 05005. [Google Scholar] [CrossRef]
- Joka, M.; Poskrobko, S. Methane rich gasification of wood pellets. E3S Web Conf. 2016, 10, 00031. [Google Scholar] [CrossRef]
- Poskrobko, S.; Król, D.; Gozdur, A.B. Gasification of waste wood biomass. Drewno 2016, 59, 241–248. [Google Scholar]
- Brynda, J.; Skoblia, S.; Pohořelý, M.; Beňo, Z.; Soukup, K.; Jeremiáš, M.; Moško, J.; Zach, B.; Trakal, L.; Šyc, M.; et al. Wood chips gasification in a fixed-bed multi-stage gasifier for decentralized high-efficiency CHP and biochar production: Long-term commercial operation. Fuel 2020, 281, 118637. [Google Scholar] [CrossRef]
- Chaurasia, A. Modeling, simulation and optimization of downdraft gasifier: Studies on chemical kinetics and operating conditions on the performance of the biomass gasification process. Energy 2016, 116, 1065–1076. [Google Scholar] [CrossRef]
- Mohammed, M.; Salmiaton, A.; Azlina, W.W.; Amran, M.M.; Fakhru’L-Razi, A. Air gasification of empty fruit bunch for hydrogen-rich gas production in a fluidized-bed reactor. Energy Convers. Manag. 2011, 52, 1555–1561. [Google Scholar] [CrossRef]
- Bhatia, S. 18. Biomass Gasification. In Advanced Renewable Energy Systems; Woodhead Publishing India PVT: New Deli, India, 2014; pp. 474–490. [Google Scholar]
- Huang, F.; Jin, S. Investigation of biomass (pine wood) gasification: Experiments and Aspen Plus simulation. Energy Sci. Eng. 2019, 7, 1178–1187. [Google Scholar] [CrossRef]
- Almeida, A.; Neto, P.; Pereira, I.; Ribeiro, A.; Pilão, R. Effect of temperature on the gasification of olive bagasse particles. J. Energy Inst. 2019, 92, 153–160. [Google Scholar] [CrossRef]
- Halim, N.H.A.; Saleh, S.; Samad, N.A.F.A. Effect of Gasification Temperature on Synthesis Gas Production and Gasification Performance for Raw and Torrefied Palm Mesocarp Fibre. ASEAN J. Chem. Eng. 2020, 19, 120–129. [Google Scholar] [CrossRef]
- Yahaya, A.Z.; Somalu, M.R.; Muchtar, A.; Sulaiman, S.A.; Daud, W.R.W. Effect of particle size and temperature on gasification performance of coconut and palm kernel shells in downdraft fixed-bed reactor. Energy 2019, 175, 931–940. [Google Scholar] [CrossRef]
- Yii, B.L.S.; Ismail, W.M.S.W.; Yahya, F.N.; Rasid, R.A. The Effect of Operating Temperature and Equivalence Ratio in an Entrained Flow Gasification of EFB. Mater. Today Proc. 2019, 19, 1373–1381. [Google Scholar] [CrossRef]
- Zeng, R.; Wang, S. Study on the Characteristics of the Influence of Temperature and Excess air ratio effect on Waste Gasification Syngas. IOP Conf. Ser. Earth Environ. Sci. 2018, 199, 032094. [Google Scholar] [CrossRef]
- Taba, L.E.; Irfan, M.F.; Daud, W.A.M.W.; Chakrabarti, M.H. The effect of temperature on various parameters in coal, biomass and CO-gasification: A review. Renew. Sustain. Energy Rev. 2012, 16, 5584–5596. [Google Scholar] [CrossRef]
- Franco, C.; Pinto, F.; Gulyurtlu, I.; Cabrita, I. The study of reactions influencing the biomass steam gasification process. Fuel 2003, 82, 835–842. [Google Scholar] [CrossRef]
- Sikarwar, V.S.; Zhao, M.; Clough, P.; Yao, J.; Zhong, X.; Memon, M.Z.; Shah, N.; Anthony, E.J.; Fennell, P.S. An overview of advances in biomass gasification. Energy Environ. Sci. 2016, 9, 2939–2977. [Google Scholar] [CrossRef]
- Wang, L.; Weller, C.L.; Jones, D.D.; Hanna, M.A. Contemporary issues in thermal gasification of biomass and its application to electricity and fuel production. Biomass Bioenergy 2008, 32, 573–581. [Google Scholar] [CrossRef]
- Couto, N.; Rouboa, A.; Silva, V.; Monteiro, E.; Bouziane, K. Influence of the Biomass Gasification Processes on the Final Composition of Syngas. Energy Procedia 2013, 36, 596–606. [Google Scholar] [CrossRef]
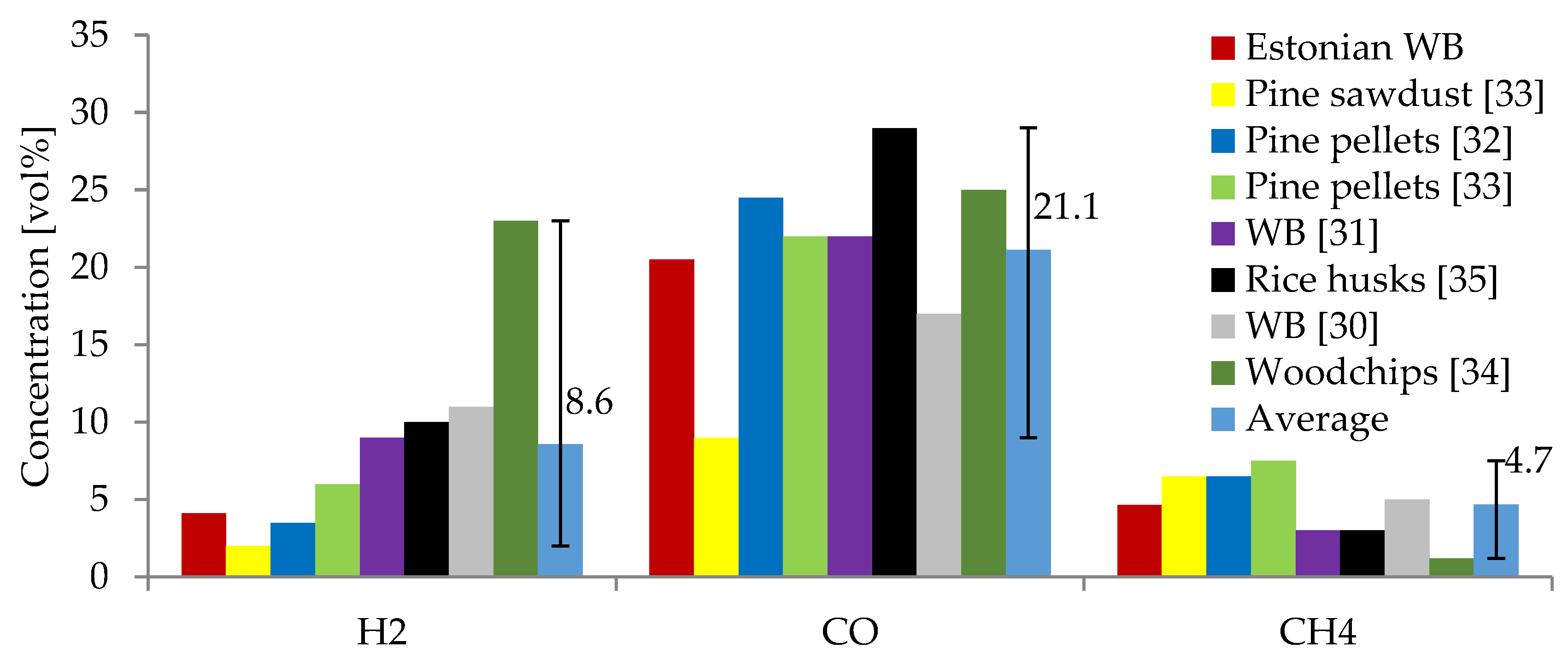

| Parameter | WB | |||
|---|---|---|---|---|
| Spruce | Alder | Pine | ||
| Stoichiometric A/F | Oxy/fuel, g of O2 per g biomass | 1.44 | 1.42 | 1.43 |
| Air/fuel, g of air per g of biomass | 6.87 | 6.78 | 6.80 | |
| Operational A/F 1 1 | Oxy/fuel, g of O2 per g biomass | 0.54 | 0.54 | 0.54 |
| Air/fuel, g of air per g of biomass | 2.58 | 2.58 | 2.58 | |
| Operational A/F 2 2 | Oxy/fuel, g of O2 per g biomass | - | 0.27 | - |
| Air/fuel, g of air per g of biomass | - | 1.29 | - | |
| ER 1 1 | 0.376 | 0.380 | 0.381 | |
| ER 2 2 | - | 0.190 | - | |
| Composition | Feedstock Used [21] | Literature | |||||
|---|---|---|---|---|---|---|---|
| Norway Spruce | Grey Alder | Scots Pine | Spruce [25,26] | Alder [27] | Pine [28,29] | ||
| Proximate analysis (wt%) | Ash content | 0.3 | 0.3 | 0.3 | 0.2–0.6 | 0.7 | 0.3–0.6 |
| Moisture | 6.9 | 7.6 | 8.5 | 7.6 | 7.3 | 7.6 | |
| Fixed carbon | 14.2 | 14.0 | 14.5 | 12.8–28.3 | 12.5 | 14.2–18.9 | |
| Volatile matter | 85.5 | 85.7 | 85.2 | 70.2–87.0 | 87.4 | 77.9–90.3 | |
| Elemental analysis (wt%) | C | 50.3 | 49.9 | 50.1 | 49.5–51.7 | 49.9 | 44.7–56.9 |
| H | 6.6 | 6.6 | 6.6 | 6.1–6.2 | 5.1 | 6.3–6.5 | |
| N | 0.1 | 0.2 | 0.19 | 0.1–1.0 | 0.5 | 0.25 | |
| S | n.d. 2 | n.d. | n.d. | <0.1 | 0.3 | <0.1 | |
| O 1 | 42.7 | 43.0 | 43.1 | 41.2–43.2 | 43.4 | 42.9–46.9 | |
| Calorific value (MJ/kg) | LHV | 18.6 | 18.5 | 18.4 | 19.0 | 19.0 | 16.3–19.2 |
| HHV | 20.0 | 19.9 | 19.8 | 19.7–20.3 | 19.0 | 17.6–20.6 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lyons Cerón, A.; Konist, A.; Lees, H.; Järvik, O. Effect of Woody Biomass Gasification Process Conditions on the Composition of the Producer Gas. Sustainability 2021, 13, 11763. https://doi.org/10.3390/su132111763
Lyons Cerón A, Konist A, Lees H, Järvik O. Effect of Woody Biomass Gasification Process Conditions on the Composition of the Producer Gas. Sustainability. 2021; 13(21):11763. https://doi.org/10.3390/su132111763
Chicago/Turabian StyleLyons Cerón, Alejandro, Alar Konist, Heidi Lees, and Oliver Järvik. 2021. "Effect of Woody Biomass Gasification Process Conditions on the Composition of the Producer Gas" Sustainability 13, no. 21: 11763. https://doi.org/10.3390/su132111763
APA StyleLyons Cerón, A., Konist, A., Lees, H., & Järvik, O. (2021). Effect of Woody Biomass Gasification Process Conditions on the Composition of the Producer Gas. Sustainability, 13(21), 11763. https://doi.org/10.3390/su132111763







