Abstract
Increased levels of carbon dioxide have revolutionised the Earth; higher temperatures, melting icecaps, and flooding are now more prevalent. Fortunately, renewable energy mitigates this problem by making up 20% of human energy needs. However, from a “green environment” perspective, can carbon dioxide emissions in the atmosphere be reduced and eliminated? The carbon economic circle is an ideal solution to this problem, as it enables us to store, use, and remove carbon dioxide. This research introduces the circular carbon economy (CCE) and addresses its economic importance. Additionally, the paper discusses carbon capture and storage (CCS), and the utilisation of CO2. Furthermore, it explains current technologies and their future applications on environmental impact, CO2 capture, utilisation, and storage (CCUS). Various opinions on the best way to achieve zero carbon emissions and on CO2 applications and their economic impact are also discussed. The circular carbon economy can be achieved through a highly transparent global administration that is supportive of advanced technologies that contribute to the efficient utilisation of energy sources. This global administration must also provide facilities to modernise and develop factories and power stations, based on emission-reducing technologies. Monitoring emissions in countries through a global monitoring network system, based on actual field measurements, linked to a worldwide database allows all stakeholders to track the change in greenhouse gas emissions. The process of sequestering carbon dioxide in the ocean is affected by the support for technologies and industries that adopt the principle of carbon recycling in order to maintain the balance. This includes supporting initiatives that contribute to increasing vegetation cover and preserving oceans from pollutants, especially chemicals and radioactive pollutants, which will undoubtedly affect the process of sequestering carbon dioxide in the oceans, and this will contribute significantly to maintaining carbon dioxide at acceptable levels.
Keywords:
CCE; circular carbon economy; circular economy; CO2; economic impact; zero carbon emission; CCUS 1. Introduction
Global CO2 emissions reached 36 gigatons (GT) in 2017; this translates to a loss of nearly 10 tons of carbon to the environment [1]. By 2050, there will be a profound transformation in carbon economics, spurred on by the desire to reduce carbon dioxide emissions and reuse the same in a wide range of advanced industrial processes that use modern technologies and artificial intelligence. All industries, especially those that consume a large amount of energy, must include low-carbon options, such as gaseous fuels and others [2]. However, there are two sides to this debate. One side argues that there is no relationship between global warming and CO2. They think that measuring the effect of human activity on the climate is difficult and complex, and they cannot reconcile with the notion that CO2 is a primary contributor to global warming [3]. The other side argues that CO2 is the main reason for global warming. Studies show that the carbon dioxide levels in the atmosphere have increased since the beginning of the Industrial Revolution. However, what is the rate of warming that might arise due to the increased CO2 concentration in the air? Natural experiments, based on real-world observations, indicate that global warming of no more than a few tenths of a degree can result from such an increase in carbon dioxide [4]. Recent research also shows that methane emissions outpace the impact of carbon sinks from trees in wooded wetlands [5].
In addition, there is some evidence of the contribution of greenhouse gases to the increase in global temperature, and other evidence confirms that other factors, such as solar activity and associated cosmic ray (CR) modulations, may have a more significant contribution than greenhouse gases. It is well known that solar activity and its associated geomagnetic disturbances affect the propagation of high-energy CR. During the high solar activity, the number of CRs is modulated (i.e., decreased) due to the increase in magnetic activity, which affects the rate of CRs at the top of the atmosphere. This modulation in the flux of the primary CRs affects the level of atmospheric ionisation, as CR particles are one of the main sources of atmospheric ionisation. Once the primary CR particles enter the upper atmosphere of the Earth, they interact with atmospheric molecules and produce atmospheric nuclear-electromagnetic-muon cascades. Depending on the energy of the primary particles, these cascades continue to produce more particles until they reach sea level. Due to their energies and their capabilities to cause ionisations in the Earth’s atmosphere at different atmospheric levels, CR particles have the ability to change atmospheric properties, which affect their physical and chemical characteristics. The most important consequences of CR interactions with atmospheric molecules are aerosol formations and their subsequent condensation processes (e.g., the production of ultrafine aerosols, which may act as cloud condensation nuclei).
Additionally, atmospheric aerosols are one of the significant factors affecting incoming solar radiation through scattering and absorption. Therefore, solar activity and the associated CR modulation can be considered to affect the distributions of meteorological variables directly or indirectly. Hence, their impact on climate change cannot be excluded [6,7,8].
Deforestation hurts the climate; this is evident in the worsening deforestation rates in the Amazon rainforest. In 2019, 10,129 km2 of forest cover was cut. Unfortunately, the deforestation rate continued to grow by 9.5% in 2020, compared to the previous year [9]. Carbon stocks remaining in humid tropical forests are currently at risk of human deforestation, and there is a potential for these stocks to be released due to climate change Moreover, continued tropical deforestation will play a massive role in accumulating greenhouse gas concentrations in the future [10]. Tropical forests are a significant factor in stabilising carbon dioxide in the climate. As tropical forests greatly benefit the ecosystem, it is unfortunate that they are lost for unsustainable benefits. The actual loss is when environmental degradation remains over the years. Despite the constant prohibitions against deforestation and logging, only 12% of the original tropical forest cover remains; thus, the remainder of these forests must be preserved [11].
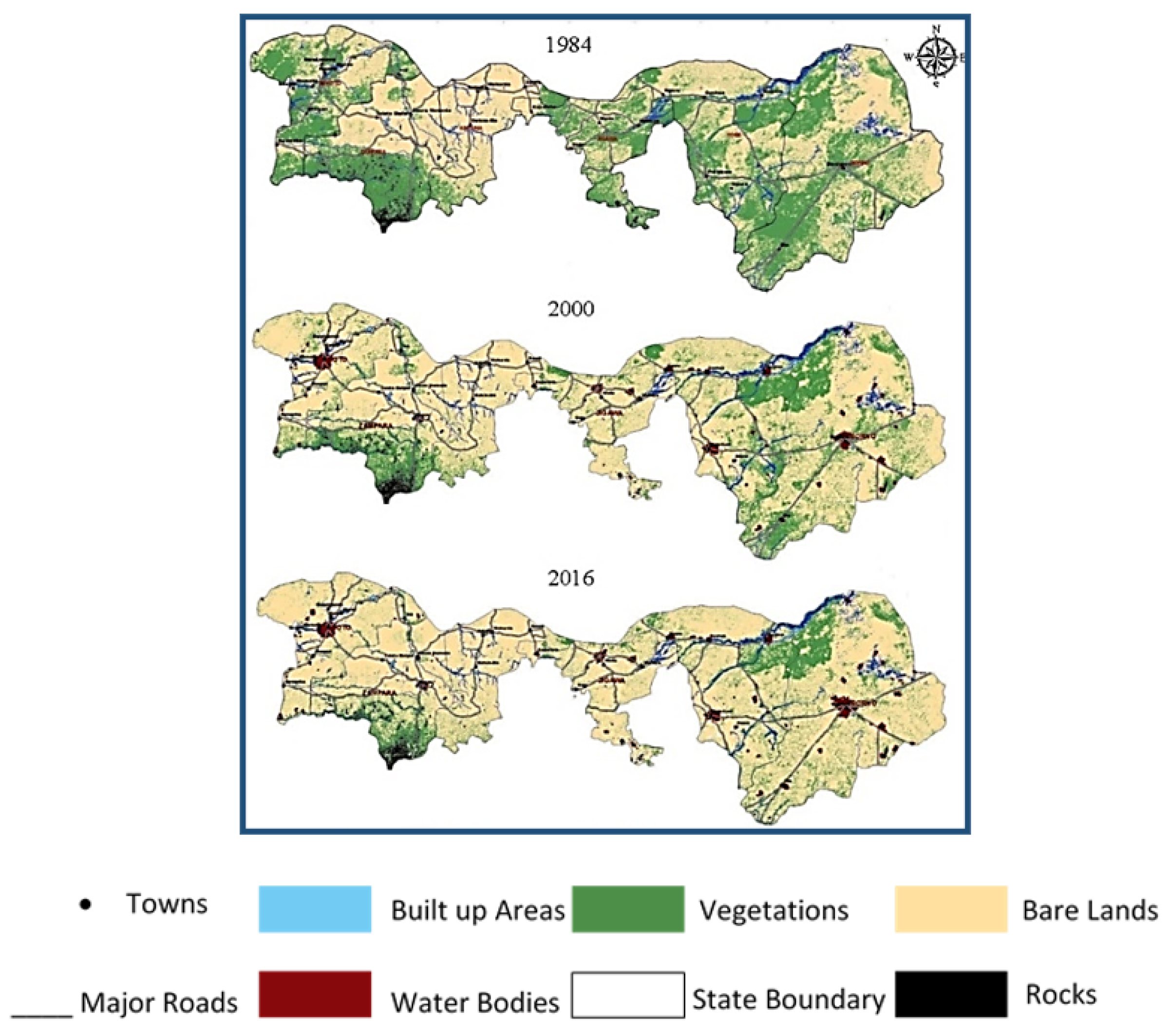
Between 1984 and 2016, 45,945.08 km2 of vegetation cover was lost in Northern Nigeria. Bare land increased by 28,293.07 km2, and the built-up area also increased by 11,422.48 km2 within the same time (Figure 1). The massive increase in the barren lands is associated with the intense creep of desert sand. Sand traps the incoming solar radiation during the day, which leads to a rise in the heating of the Earth’s surface. This, in turn, imposes extremely austere conditions that lead to the deterioration of the abundance of vegetation [12]. The circular carbon economy (CCE) is an integrated system that leans on reducing CO2 emissions and manage them through an environmental and economical integrated management system that works on modern technologies [13]. The circular carbon economy (CCE) system achieves a balance by closing the carbon circle, capturing CO2 emissions, and using them economically after their initial linear use [13]. This approach differs in that it does not focus solely on reducing or avoiding the increase in the amounts of carbon. The steady increase in carbon dioxide levels requires increased effort to mitigate their impact [14]. Carbon dioxide is sourced either naturally or through human intervention [15]. There are many uses for carbon dioxide; it can be used immediately after its capture or after being chemically reacted. These uses are considered to be the actual seizure of carbon dioxide, since they exploit carbon dioxide emissions for valuable materials. CO2 capture and storage (CCS) effectively reduce carbon dioxide emissions [16].
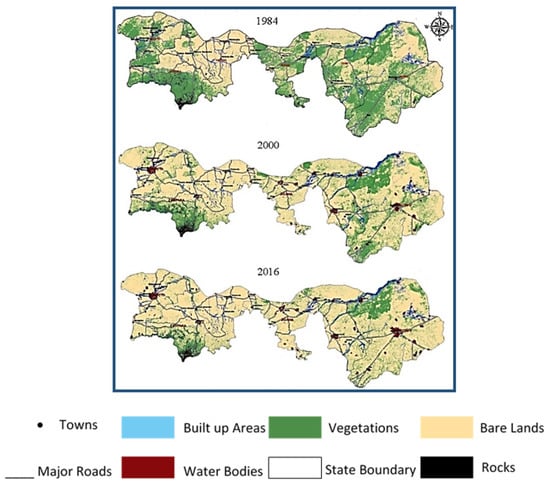
Figure 1.
Spatial variation and temporal changes in land cover are shown throughout the study from 1984 to 2016. Adapted with permission from Elsevier Ref. [12].
There are many techniques for removing carbon dioxide [17,18] that will be discussed from a technical and economic point of view, as well as the existence of masses of research methods that deal with the uptake and utilisation of carbon dioxide sequestration; however, the issue of taking advantage of carbon emissions, recycling and benefiting from them, and looking at carbon emissions from an economic perspective are topics that need more studies and research. Therefore, this article attempts to treat the subject from a holistic view and draw a roadmap for further research and studies in this field.
2. Greenhouse Effect
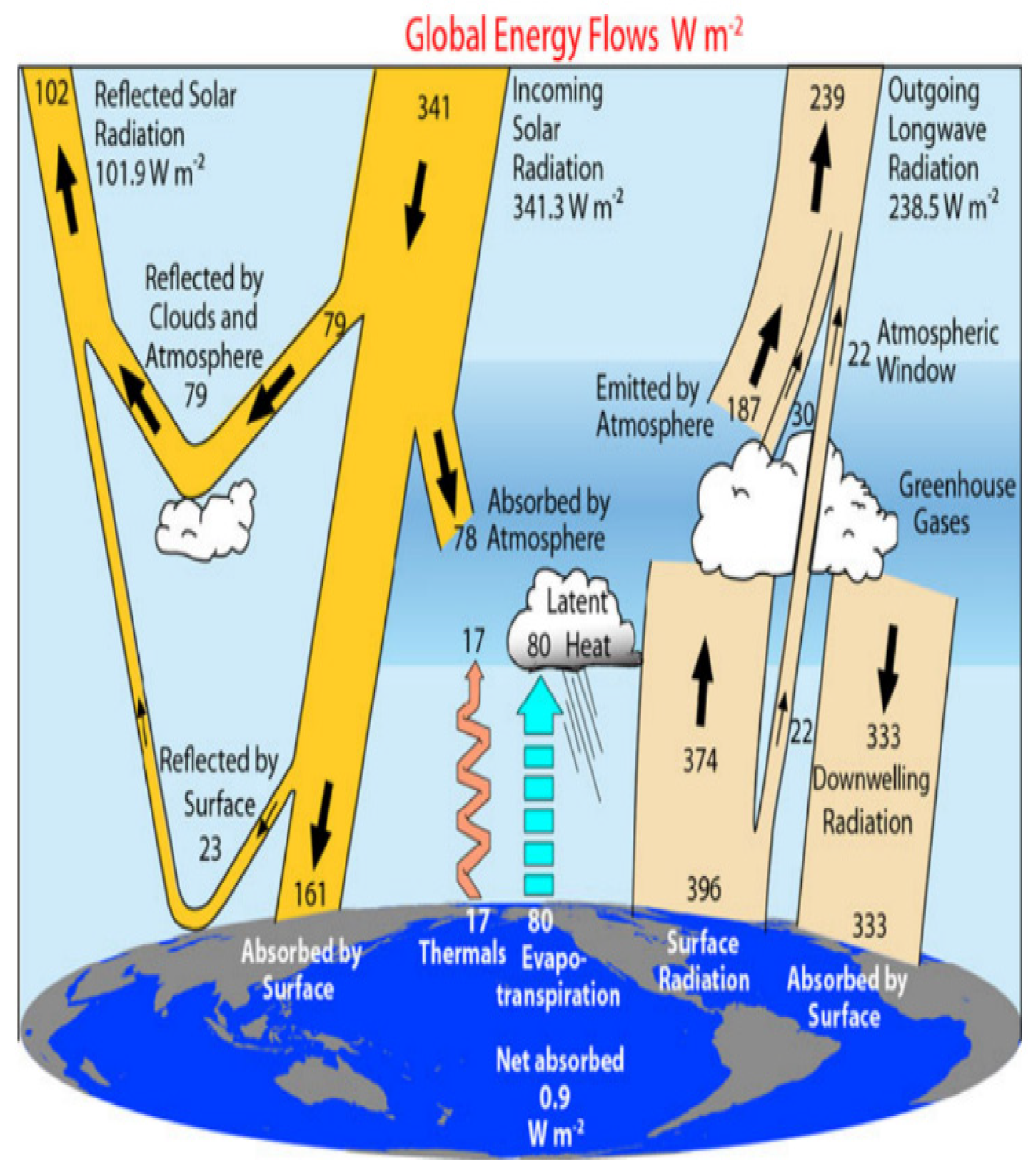
Without the greenhouse effect, life on Earth is not possible. It is a natural phenomenon that occurs when the gases in the atmosphere trap the heat coming from the sun. The fundamental physics of this process have been understood; the sun pours out large amounts of radiation across the electromagnetic spectrum. As incoming solar radiation is absorbed and reemitted back from the Earth’s surface, as infrared energy, the greenhouse gases (GHG) prevent some of this heat from escaping, instead of reflecting the energy to heat the planets [19]. Figure 2 illustrates the magnitude of sun radiation streams, which is based on the available observational data. The radiant energy flux is about 341 Wm−2 for each square meter between the Earth and the sun. About 30% of this radiation is reflected back to space by ice, deserts, and clouds, leaving 239 Wm−2 available to the climate system. The atmosphere is transparent to short-wavelength solar radiation, and only 78 Wm−2 is absorbed by it, leaving about 161 Wm−2 to be transmitted to the surface. Because of greenhouse gases and clouds, 333 Wm−2 of back radiation also warms the Earth’s surface from the atmosphere. Thus, the surface will emit heat radiation of about 396 Wm−2, which is 157 Wm−2 greater than the 239 Wm−2 that leaves the top of the atmosphere [20]. The greenhouse effect makes the Earth a comfortable place to live, warming the planet and providing all the energy that drives the climate system. Anthropogenic GHG emissions are a change in the Earth’s energy balance, between incoming solar radiation and the heat released back into space, increasing the negative impact of the greenhouse effect, and resulting in climate change [21,22].
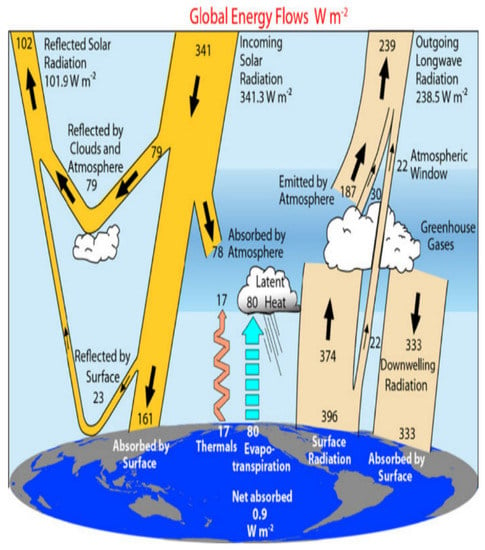
Figure 2.
The annual global energy flows of the Earth’s climate system. The broad arrows indicate the schematic flow of energy in proportion to their importance. Adapted with permission from Trenberth [21].
2.1. Greenhouse Gases
There are many greenhouse gas sources (GHGs) naturally occurring, such as water vapour, CO2, methane, and nitrous oxide. Perfluorocarbons (CF6, C2F6), hydrofluorocarbons (CHF3, CF3CH2F, CH3CHF2), and sulphur hexafluoride (SF6) are only present in the atmosphere due to industrial processes [23]. Water vapour is considered to be the most dominant GHG in the atmosphere. The concentration of water vapour is not directly dependent upon human activities [21]. However, CO2 accounted for about 78% of human activities in 2010 [24]. Global warming potentials (GWPs) show the relative effectiveness of greenhouse gases on the Earth’s heat absorption in a given time frame. Usually, CO2 is used as the reference gas, and its GWP is one [24]. For example, the 100-year GWP of CH4 is 28, indicating that its radiative effect on a mass basis is 28 times that of CO2 over the same time horizon. The mass of carbon dioxide equivalents is calculated by multiplying the emissions by the GWP of the gas [25]. Table 1 shows the primary greenhouse gases.

Table 1.
The main greenhouse gases [21,26].
2.2. Carbon Cycle
CO2 is the backbone of life on Earth. In the previous section, we see how CO2 in the atmosphere plays an essential role in maintaining the temperature of our planet. CO2 is necessary; however, this need is also entwined with one of the most severe problems facing humanity today, which is global climate change.
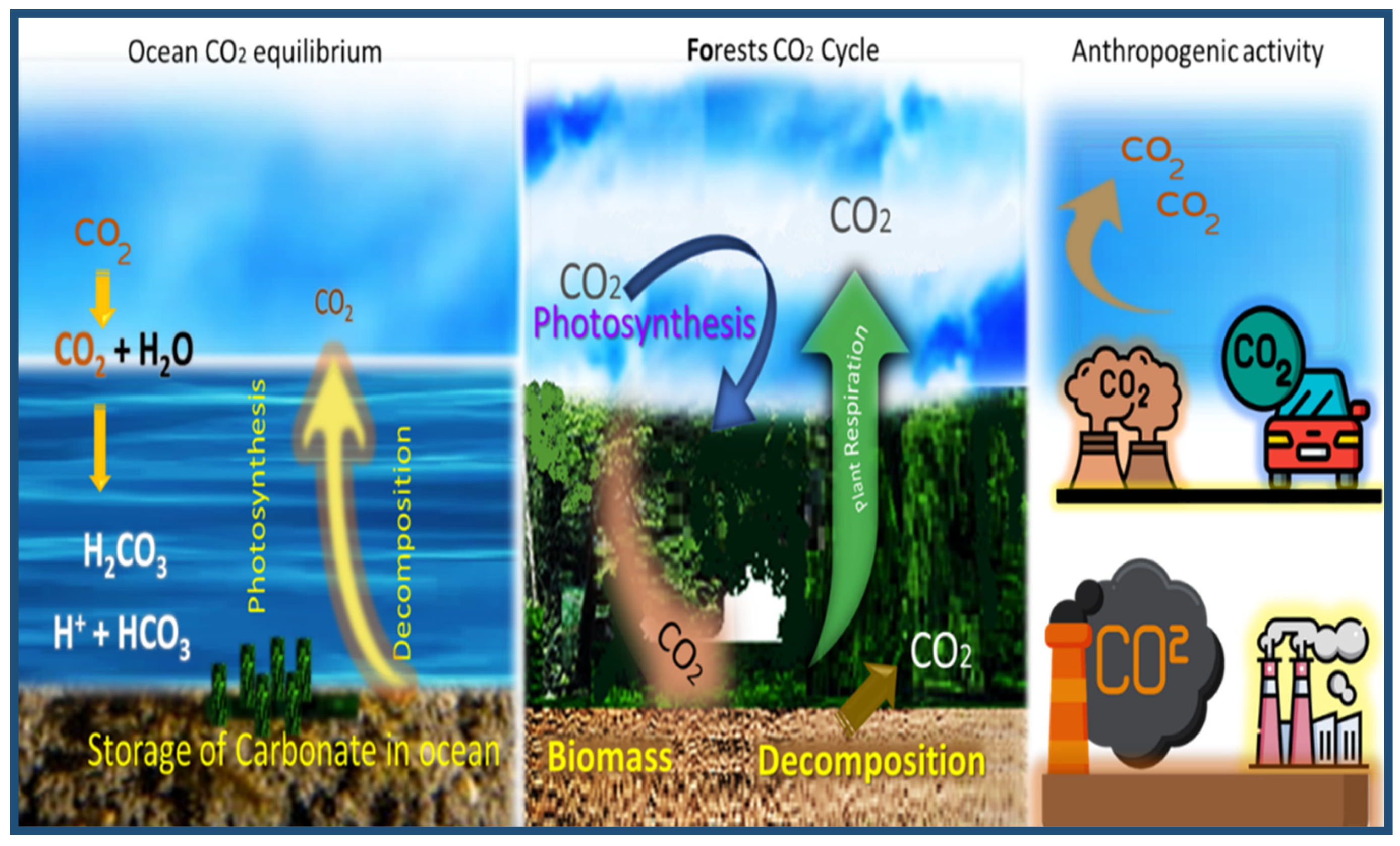
It is essential to understand the biological, geological, and chemical mechanisms that control the CO2 in the atmosphere. Figure 3 shows the various processes by which CO2 is exchanged between different regions of the Earth. The exchange of CO2 that flows between each reservoir is called the carbon cycle, which has both slow and fast components. Any cyclical change that removes carbon from one reservoir will place more CO2 in other reservoirs. As a result, changes in the emission of carbon gas into the atmosphere have caused the Earth’s temperature to rise.
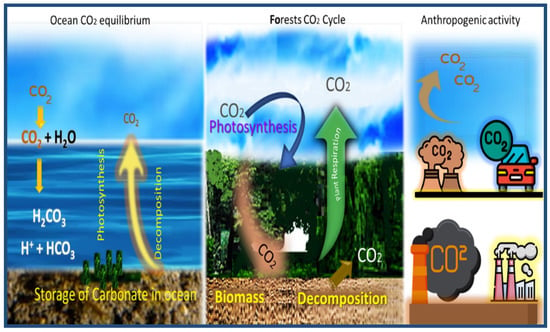
Figure 3.
Schematic diagram of the natural balance and industrial emissions in carbon dioxide. Reproduced, adapted and reprinted from Reference [27].
2.3. CO2 Sources
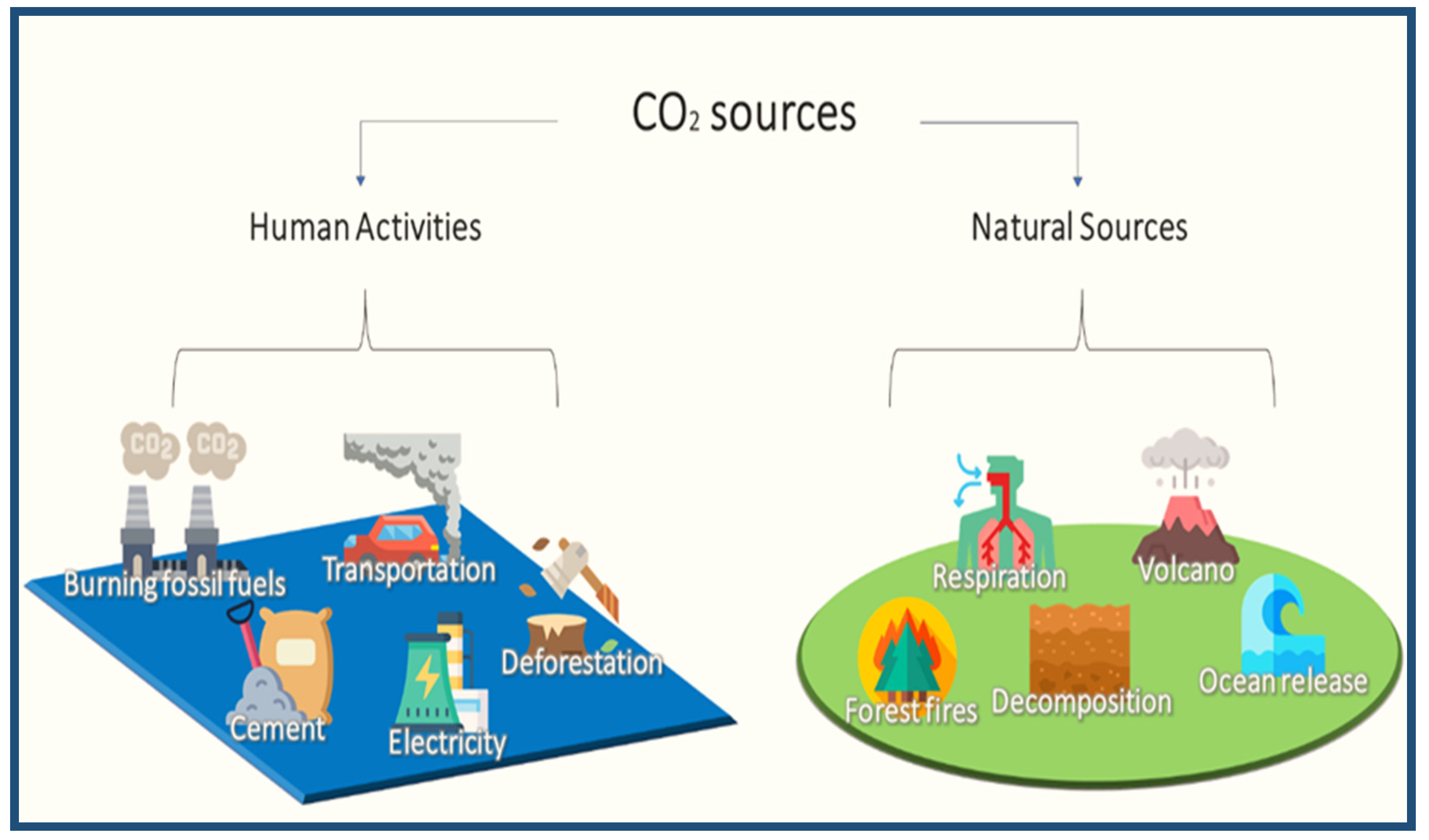
CO2 is one of the GHGs that absorbs heat and radiates it gradually over time. CO2 is naturally present in underground rocks, and is released into the atmosphere by respiration decomposition and ocean release. The release of carbon dioxide from the oceans is in equilibrium with the carbon dioxide in the atmosphere, and around 30% of CO2 is absorbed by oceans and released into the atmosphere [28]. This has made this planet warm and comfortable until recently, when global temperatures have risen, leading to a potentially perilous future. The increase in carbon dioxide in the atmosphere is one of the reasons for the rise in temperature [29]. Before humans significantly impacted the climate, there was a natural balance in the environment, in terms of its carbon dioxide content. The emissions are used for other vital activities, and carbon dioxide is constantly recycled to maintain the ecological balance. Among these activities are plant photosynthesis, ocean intake, fossil formation, soil improvement, reforestation, and the decomposition of organic matter. The increase in industrial activities did not consider the equilibrium approach to emissions; therefore, the levels of carbon dioxide emitted by those activities, such as the use of fossil fuels, electricity generation, cement production, transportation, and deforestation, compete with the existing levels of carbon dioxide emissions from natural sources, as shown in Figure 4. Carbon dioxide and other emissions have been on the rise for decades, affecting climates in general [30].
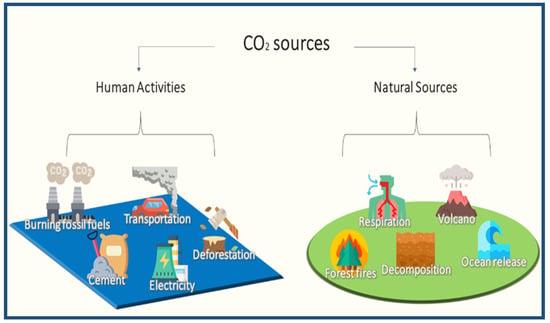
Figure 4.
Some of the sources of carbon dioxide according to the type of source.
3. The Circular Carbon Economy (CCE)
Traditional consumption methods have always been on a linear “production, consumption and discharge” model, and were maintained in an unsustainable manner. Hence, the starting point of the circular economy (CE) relies on the circular model [31]. The CE is a pattern that aims to grow the economy without using limited consumption resources, instead promoting recycling materials, and benefiting companies, society, and the environment [32]. The CCE has facilitated the avoidance of many obstacles for companies, by improving resource use and recycling efficiency [33]. In addition, the CCE framework advances the understanding of system dynamics and linkages between mitigation options. As a result, the CCE can play an essential role in reaching climate stabilisation. Ensuring that technologies are available, mature, and cost-effective in each part of the CCE is critical for achieving the climate goals at a reasonable cost.
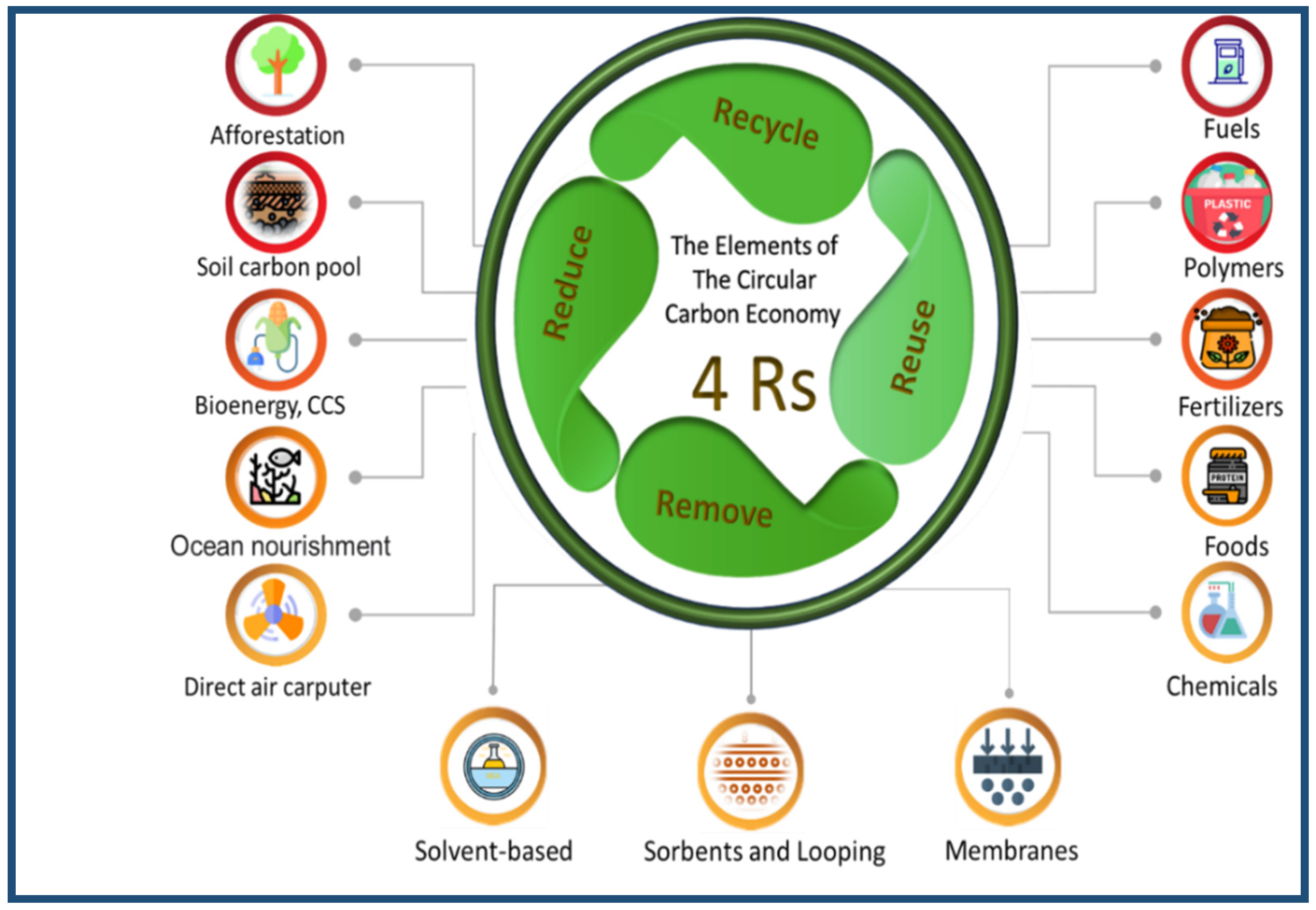
The G20 Protection and Climate Stewardship Working Group (CSWG) conference was held under the leadership of the Kingdom of Saudi Arabia. The most crucial goal of this conference was to leverage the benefits of reducing greenhouse gas emissions by using the CCE [34,35]. The objective is to limit the global temperature increase to less than 2 °C before the end of the century [36]. CCE deals with the climate problem and plays a massive role in achieving a more stable climate [32]. The CCE has 4R elements of a strategy that represent the following four organisational principles: reduce, reuse, recycle, and remove. These four elements are the cornerstones of managing CCEs. What distinguishes the CCE is the fourth element (remove) [14]. Figure 5 shows how the CCE can be expressed as a system of energy and carbon flux and represents the fundamental nature of the system.
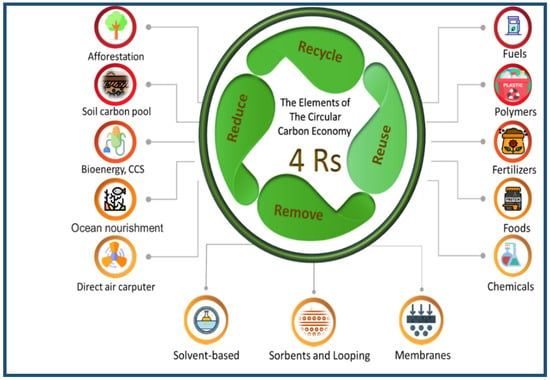
Figure 5.
Schematic of CCE elements.
Reduce: Use pathways that reduce carbon emissions. For example, energy efficiency, in terms of both supply and demand, reduces energy consumption and the associated carbon. Likewise, carbon-neutral energy supply options, such as non-biotic renewable sources and nuclear power, reduce the flow of carbon into the system as well. However, they can indirectly lead to carbon emissions during its manufacture, construction, and installation.
Reuse: Carbon capture and utilisation mean that CO2 does not chemically react to manufacture-enhanced or enhanced oil recovery material. In this context, the experience of the Saudi Basic Industries Corporation (SABIC) is considered a pioneer in storing and recycling carbon dioxide in the industry, to produce fertilisers and methanol, with 500,000 tons of carbon dioxide recycled each year [37].
Recycle: Chemical reactions conducted on carbon can convert it into new economically beneficial substances. Recycling is represented in the natural carbon cycle as natural sinks, such as plants, soil, and oceans, which pull CO2 out of the atmosphere and then rerelease it through decomposition and combustion. Thus, carbon is recycled efficiently, and the bioenergy subsystem is considered zero-emission. As a result, an equivalent of biomass grows to replace what is consumed as biological raw materials, such as wood, fuel crops, and algae bioenergy.
Remove: Remove carbon from the system and store it either directly or from natural sources. This method is the most effective. The captured carbon can be converted into raw materials in “reuse” or removed by storing it through chemical or geological methods. CO2 can be directly captured from industrial processes and combustion points, and it can also be captured directly from the air using direct air collection techniques. Also, land can be managed to become a natural sink for carbon in the atmosphere. Natural sinks, carbon capture and storage, bioenergy, and direct air harvesting can all close the loop of emissions elsewhere, in areas that may be difficult or too expensive to capture such emissions directly, such as from jet fuel combustion [11,32,33]. The economic importance of the carbon circle is limited to reducing carbon dioxide emissions and producing products with economic value.
4. Carbon Capture, Utilisation, and Storage (CCUS)
The reduced carbon dioxide scenario is based on carbon dioxide capture and storage [38]. The most effective method for mitigating carbon dioxide from the atmosphere is carbon capture technologies [39,40]. The CCUS process separates carbon dioxide in industrial production, energy sources, and other sources of significant emissions. It is then transported through pipes, until it is injected underground for geological storage, to isolate it for the long term from the atmosphere. It can also be utilised to create new products from CO2 [41,42,43,44,45,46]. When implementing a CCUS, it must look at the aspects on which it depends, including absorptivity, infrastructure, capacity, costs, especially the costs of technology transfer and localisation in developing countries. Carbon dioxide is produced and accumulated by the combustion of the following substances: fossil fuels, natural gas, or biofuel. It can be captured and reused in many industries, such as ammonia, steel, and soft drinks [42]. Carbon capture, utilisation, and storage technologies consist of the following four steps: carbon dioxide capture, carbon dioxide transport, carbon dioxide utilisation, and carbon dioxide storage [47,48,49,50]. In this aspect, it is important to evaluate each country’s ability to transfer and use these technologies in the field, and consider each country’s legislation, regulations, and environmental situation.
4.1. CO2 Capture
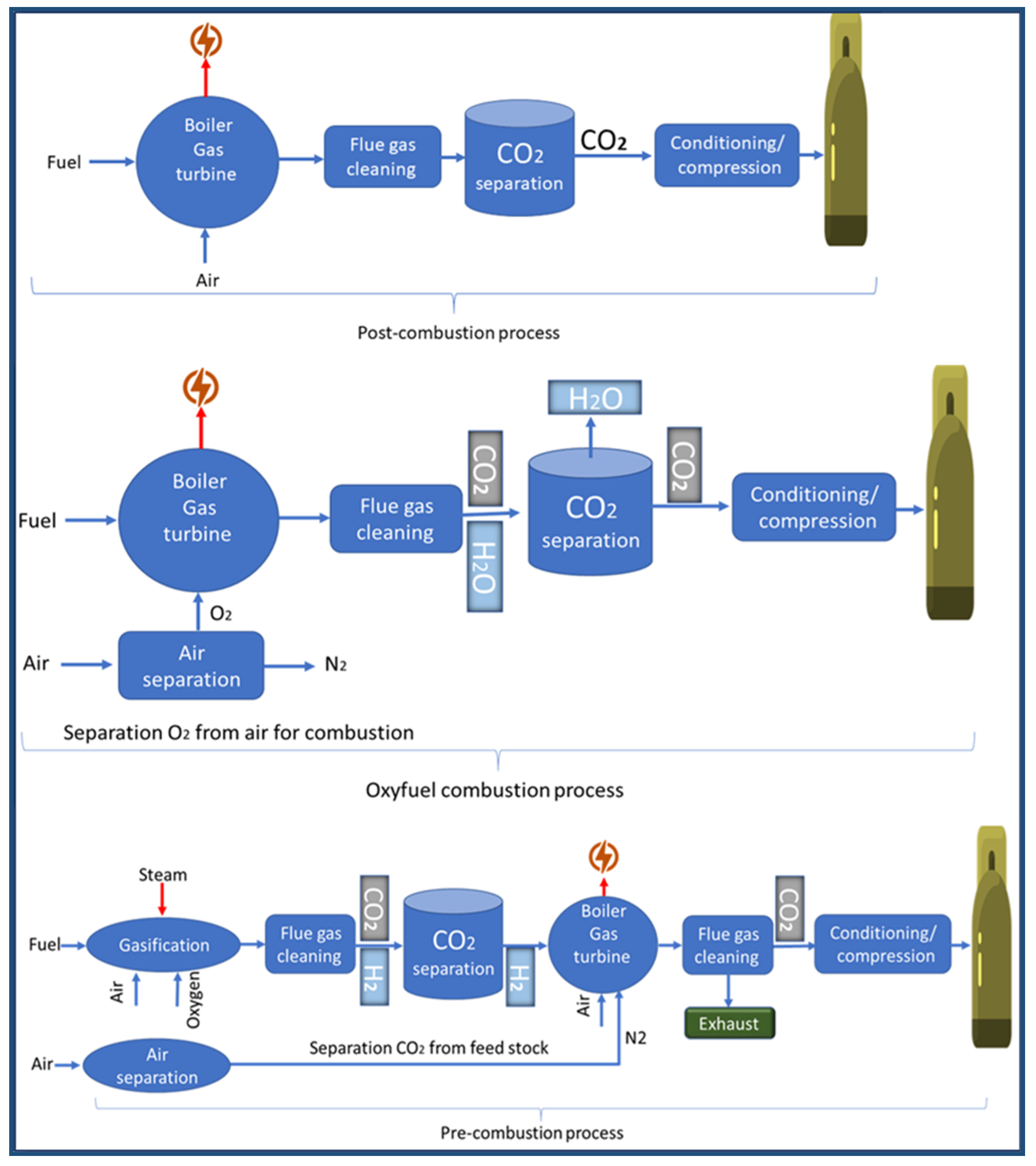
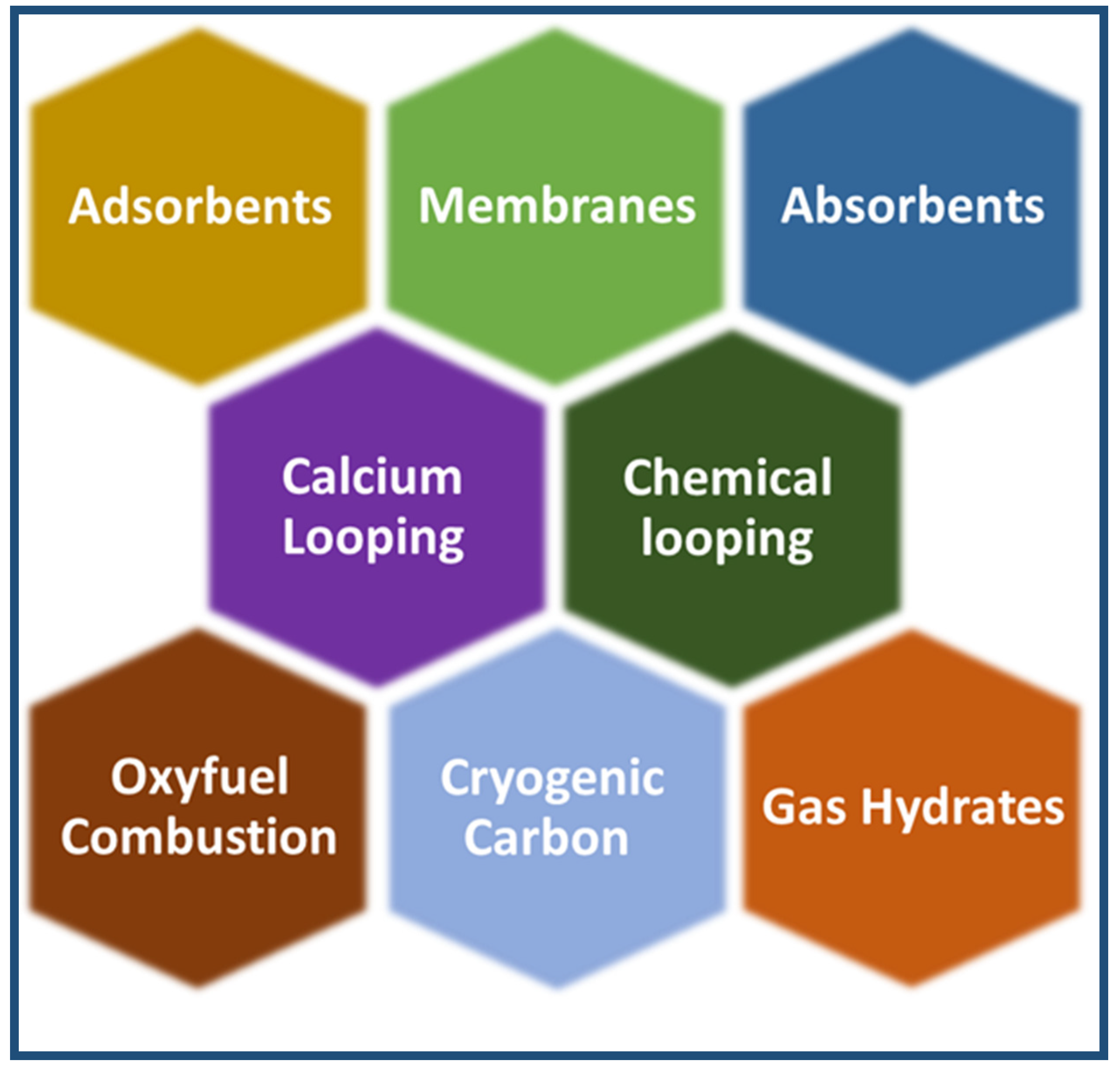
CO2 capture is considered the most expensive step in CO2 capture, utilisation, and sequestration of CCUS [51]. Carbon dioxide is captured from sources of emission, such as power plants. There are the following three carbon capture processes in power plants: pre-combustion, oxy-fuel combustion, and post-combustion [52]. Carbon dioxide capture has reached a high-tech level, as demonstrated by several commercial projects [53]. Figure 6 shows the three main carbon capture processes. Other techniques include absorbents [54], adsorbents [55,56], cryogenic distillation [57], membranes [58,59], gas hydrates [60], and chemical looping [61], as shown in Figure 7. However, during the past years, and until now, attention has been focused on the production of materials and technologies that contribute to capturing carbon dioxide and converting it into materials of economic value. These materials and technologies face many obstacles, including selectivity, stability, reuse, the cost of reuse, and overall cost. In the coming years, a revolution may occur in the field of manufacturing carbon dioxide capture materials in terms of selectivity and cost. It is expected that the cost of these materials will decrease with the increase in production quantity. It is also expected that there will be an evolution in the technical methods used, and this view is confirmed by the large jump in the amount of research and project support in the field of carbon dioxide capture [62].
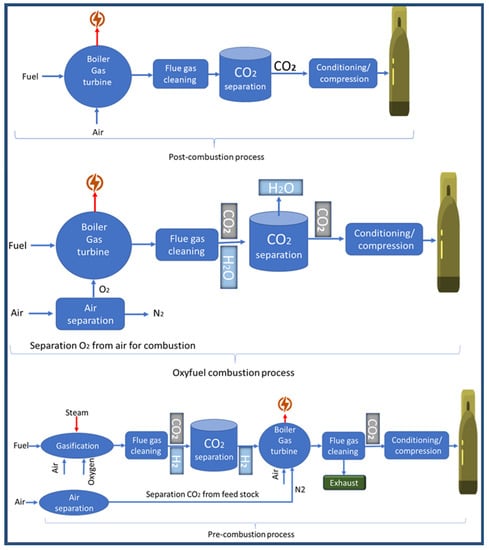
Figure 6.
The three main carbon capture processes. Adapted with permission from Elsevier Ref. [63].
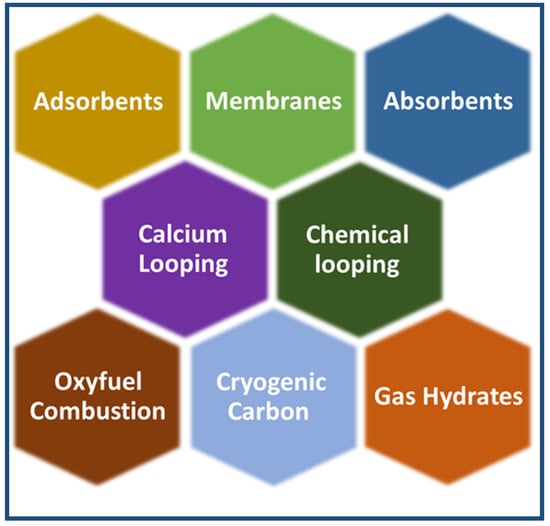
Figure 7.
Subordinate technologies in carbon dioxide capture.
4.2. CO2 Transport
After the process of capturing and compressing carbon dioxide, it can now be transported to storage. The transportation process is simple, and the potential routes of transportation are either pipelines or ships. The most mutual transportation process involves the existing oil pipelines, with a length of up to 1000 km. When carbon dioxide is stored for a longer period, it is stored similarly to liquefied natural gas (LNG) [47].
4.3. CO2 Utilisation
Carbon utilisation is a term used to describe the many ways to make use of the captured CO2, such as using or recycling the same, to produce economically valuable products. In the following sections, a list of technologies related to carbon utilisation is provided.
4.3.1. Carbon Dioxide—Enhanced Oil Recovery (CO2-EOR)
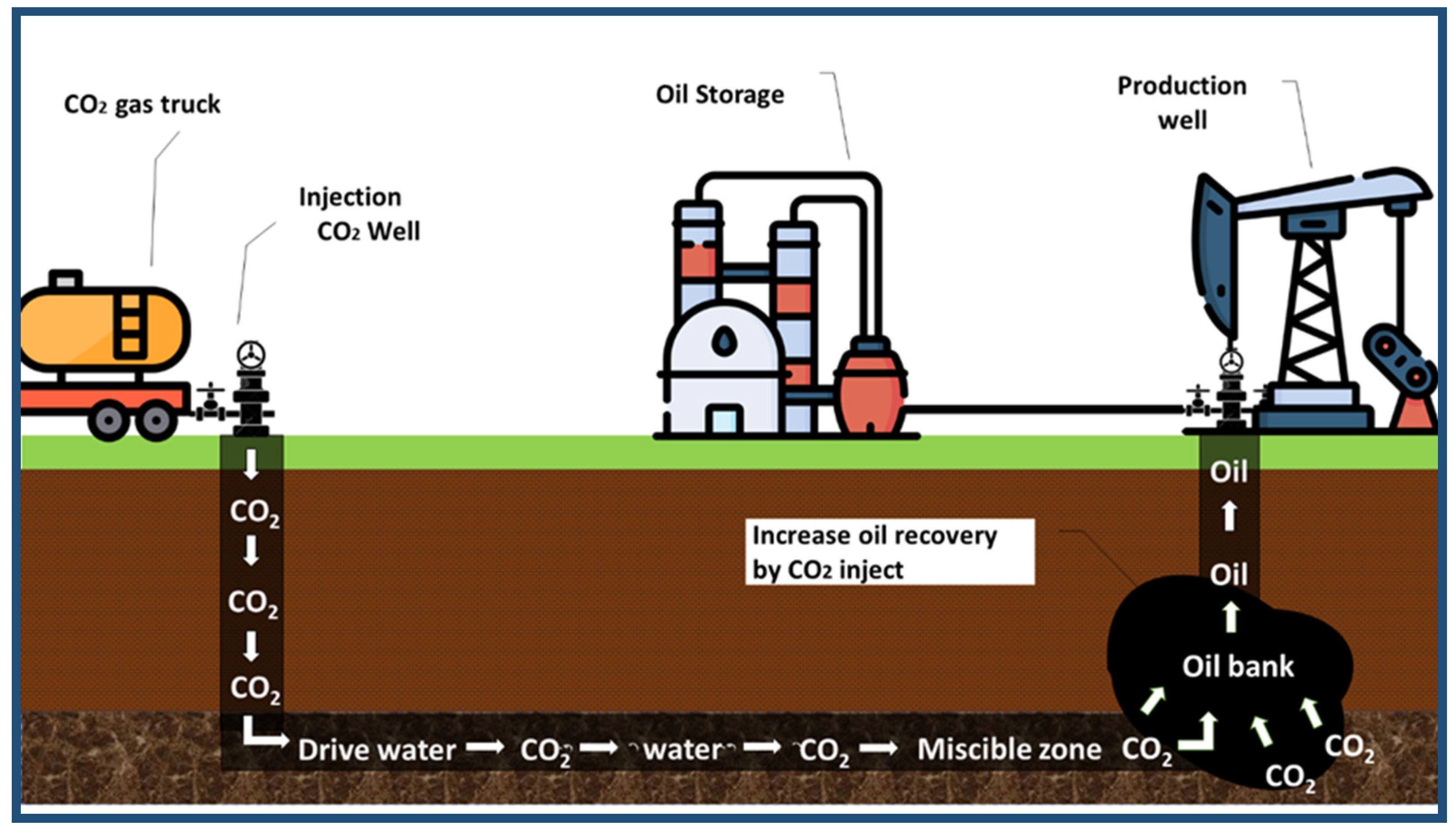
Enhanced oil recovery (EOR) using CO2, called CO2-EOR, has the following two benefits: the first is to recover additional oil, thus saving energy and additional revenues (Figure 8). The second benefit is the mitigation of climate change, brought about by a reduction in anthropogenic carbon dioxide emissions [64]. The injection of carbon dioxide into oil wells could increase oil production, as shown in Figure 8 [65]. Approximately 0.1 to 1.8 gigatons can be used and stored at projected costs, ranging from USD 40 to USD 60 per ton of CO2 per year in 2050 [66]. Aramco can capture and process 45 million standard cubic feet of CO2, then send it through an 85 km pipe to the Uthmaniyah oil field to be injected into the oil reservoir. Uthmaniyah is the Middle East’s largest carbon EOR initiative that aims to reduce emissions and improve oil extraction [67]. CO2-EOR reduces the proportion of carbon dioxide in the atmosphere to 30–40%, compared to natural sources [68].
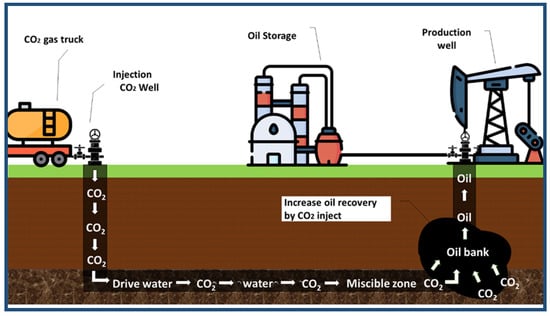
Figure 8.
Carbon dioxide-enhanced oil recovery (CO2-EOR).
4.3.2. Synthetic Fuel
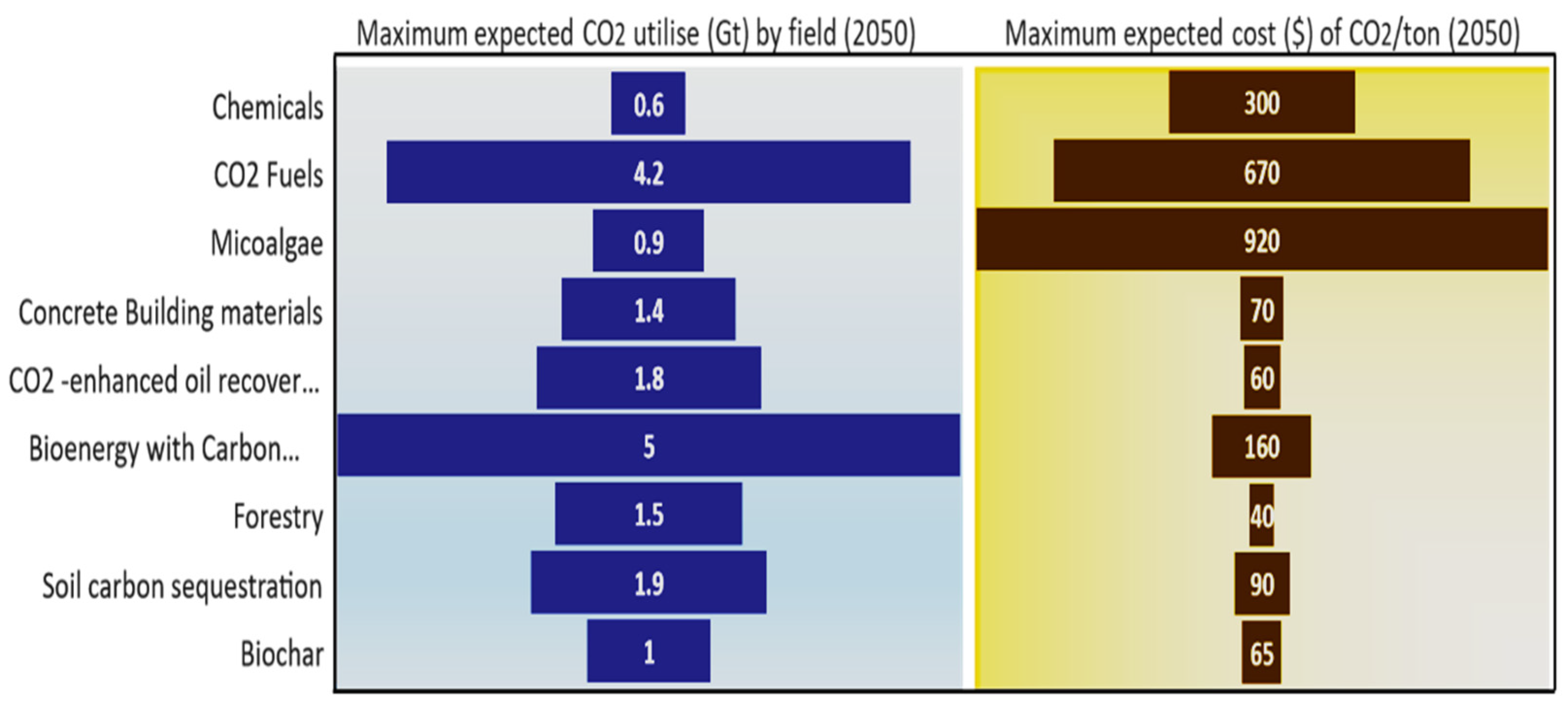
The energy stored in hydrocarbons can be converted into fuel and electricity through clean hydrocarbon technologies [69]. Hydrocarbon fuel is a combination of carbon and hydrogen; sunlight can be used to recycle carbon dioxide, along with water, into a clean hydrocarbon fuel that is compatible with our existing energy infrastructure, including methanol, synchronous fuels, and syngas. However, the current costs are so high that 1 to 4.2 gigatons of carbon dioxide could be used annually by the year 2050, and prices may reach as high as USD 670 per ton of carbon dioxide [66,70].
4.3.3. Chemical Industry
Carbon dioxide can produce many chemicals of economic value [71]. For example, it can be converted into industrially applicable chemicals to fertiliser, through catalytic chemical reactions, such as urea, as well as polymers for use in buildings or cars (for durable products) [66,72]. Also, electrical stimulation methods can convert carbon dioxide into materials of economic value, such as methane, methanol, monoxide, and other hydrocarbons [73]. Therefore, CO2 emissions will be reduced by a range of 0.3 gigatons to 0.6 gigatons per year in 2050, at a cost ranging from USD 80 to USD 300 [66]. Aramco produces polyols from carbon dioxide emissions and recycles them into clean, high-value materials with performance and cost characteristics. Polyols are utilised as industrial adhesives, food packaging, sealants and insulation, and synthetic rubber applications. They also store carbon dioxide for a long time and remove greenhouse gases from the environment [67]. It is clear that the demand for CO2 continues to increase, and that the projected average CO2 usage for each sector will increase by the year 2050, as shown in Figure 9.
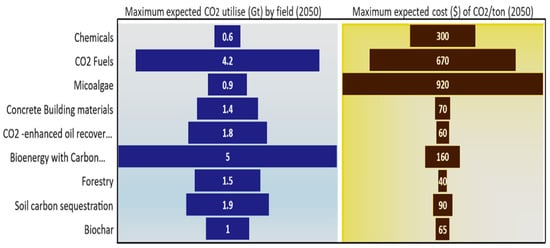
Figure 9.
The expected CO2 use and cost by field in 2050. Data were obtained from Ref. [66].
4.3.4. Concrete Building Materials
A recent pathway to capturing carbon dioxide is the natural carbonisation of concrete building materials. It is intended that where the structure of concrete is engineered, it will absorb carbon dioxide from the atmosphere slowly [74]. One of the benefits of treating concrete with carbon dioxide is the removal of carbon dioxide from industrial processes. In addition, carbon dioxide can be stored for the long time in a concrete and can be an alternative to traditional emission-intensive cement and 0.1–1.4 GtCO2 could be used and stored at the cost of approximately USD 30–70 by 2050 [66].
4.3.5. Microalgae
Microalgae require photosynthesis from sunlight, and carbon dioxide as an organic source. Also, microalgae are characterised by their ability to stabilise carbon dioxide in the following different paths: (1) they fix carbon dioxide from the air, (2) from the fumigants, as a chimney discharge, and (3) from soluble carbonates. Microalgae can carry up to 150,000 ppm of carbon dioxide from the atmosphere. In addition, algae have the capacity for the secretion of various biomolecules and fats. Therefore, it produces biofuels of great importance, such as bioethanol and biodiesel [75]. Algae is a green solution to global warming or water pollution, and may even become energy fuel in the future [76]. Microalgae have a higher production capacity than terrestrial crops in biomass, and can even be grown in unsuitable soil [46]. Microalgae can reduce carbon dioxide emissions and develop energy because they are subject to rapid growth and high energy efficiency from the sunlight. Carbon dioxide can be fixed in microalgae biomass after being processed to make fuels and chemicals [66,77], and growing microalgae has a significant impact on capturing the carbon dioxide produced in open ponds and photobioreactors. In California, 0.5 tons of carbon are extracted for every ton of biomass [46]. Microalgae could use an average of 0.2 to 0.9 gigatons of CO2 per year by 2050, at the cost of USD 230 to USD 920 per ton [66].
4.3.6. Bioenergy Carbon Capture and Storage (BECCS) Trees
BECCS is the combination of geological carbon capture and storage, which is then used to produce energy. It is considered to be one of the methods to reduce carbon dioxide [78]. Trees capture and store carbon dioxide to be used as renewable energy for the production of electricity through bioenergy and sequestered emissions. The rate of tree capture varies [66]. For example, Fagus sylvatica is one of the trees with the highest CO2 capture of 4.9 annual carbon sequestration rate (tCha−1 yr−1) [79]. By the year 2050, 0.5 to 5 gigatons of carbon dioxide are expected to be used at the cost of roughly USD 60 to USD 160 per ton [66].
4.4. CO2 Storage
Enhanced water recovery (EWR) geological storage is a way to reduce the greenhouse gas emissions of carbon dioxide. CO2-enhanced water recovery (CO2-EWR) technology improves the efficiency of carbon dioxide injection and produces saltwater. This process has potential economic value, as it stores carbon dioxide and provides cooling water for power plants. However, the sensitivity analysis of reservoir parameters, and the effects of geochemistry on CO2 transport, injection capacity, and brine production need further study [80,81].
Carbon dioxide can be safely stored in underground geological formation like in sedimentary basins. There are oil and gas reservoirs, empty fields, salt formations, and impermeable coal seams within the basins. The sites for storing carbon dioxide are inside the pores of the rocks, such as basalt rocks, organic rocks, and caves, which are in offshore or onshore geological formations [47]. Attention must be given when long-term stored CO2 starts to leak, as this is related to induce earthquakes. In addition, the injection of large amounts of carbon dioxide into geological reservoirs creates environmental risks [82].
Carbon dioxide capture could reduce the wholesale price of electricity by 15 percent in 2030, according to the analysis of a study in the Carbon Capture and Storage Association (CCSA). Carbon dioxide capture and storage could also provide many employment opportunities through the construction of CCS power plants, operating, maintenance, and associated supply chain functions [43]. Several carbon storage technologies will be mentioned in the coming sections.
In soil carbon sequestration, carbon dioxide is captured and stored in multiple places, such as forests and plants, as well as water bodies and deep geological formations, and it is held here for a very long time [83]. Carbon dioxide enhances crops, while land management techniques sequester carbon dioxide stored in the soil. Soil carbon dioxide is estimated to range from 0.9 to 1.9 gigatons per year by 2050. Costs are estimated to range from USD 20 to USD 90 per ton of carbon dioxide [66].
4.4.1. CO2 Storage in a Geological Formation
CO2 storage in geological formations involves injecting CO2 captured from emitting resources into rock formations that are deep underground. CO2 is then permanently removed from the atmosphere. Figure 10 shows the geological storage sites underground. CO2 storage has been implemented in depleted oil and gas fields [84,85]. Several projects, such as the Sleipner, Snøhvit, and In Salah projects, store 1 to 3 Mt of CO2 from gas processing each year. The underground storage of hydrocarbons has a long and successful history. In Canada, for example, around 5 Mt of acid gas has been safely stored in depleted reservoirs [86,87,88]. These examples will help in understanding the application and process for other CO2 storage projects, concerning technical issues, regulatory requirements, costs, and economic viability.

Figure 10.
Different carbon storage formation sites. Adapted from Global CCS Institute, Geological storage of CO2, 2018 [89].
4.4.2. CO2 Mineralisation
Previously, this paper discussed the geological storage of CO2. However, there are some potential issues associated with its storage in geologic formations. These include permanence, long-term monitoring, and verification; each issue comes with many unknown effects and potential risks that have yet to be determined [90,91]. An alternative to CO2 storage in geological formations is carbon mineralisation, which is a reaction between the CO2 and metal cations, such as magnesium, calcium, and iron, to form carbonate minerals.
The methods of mineralisation can be divided into the following two types: subsurface and surface mineralisation. In the subsurface method, mineralisation is a part of geologic storage. A portion of the injected CO2 and the alkaline minerals present in the formation are selected from carbon storage to form solid carbonate. In surface mineralisation, the carbonation reaction occurs above ground, within a separate reactor or industrial process. Mineral carbon dioxide storage aims to simulate the natural weathering process [92], in which calcium or magnesium silicates are transformed into carbonates via a reaction with CO2.
4.4.3. Ocean Storage of CO2
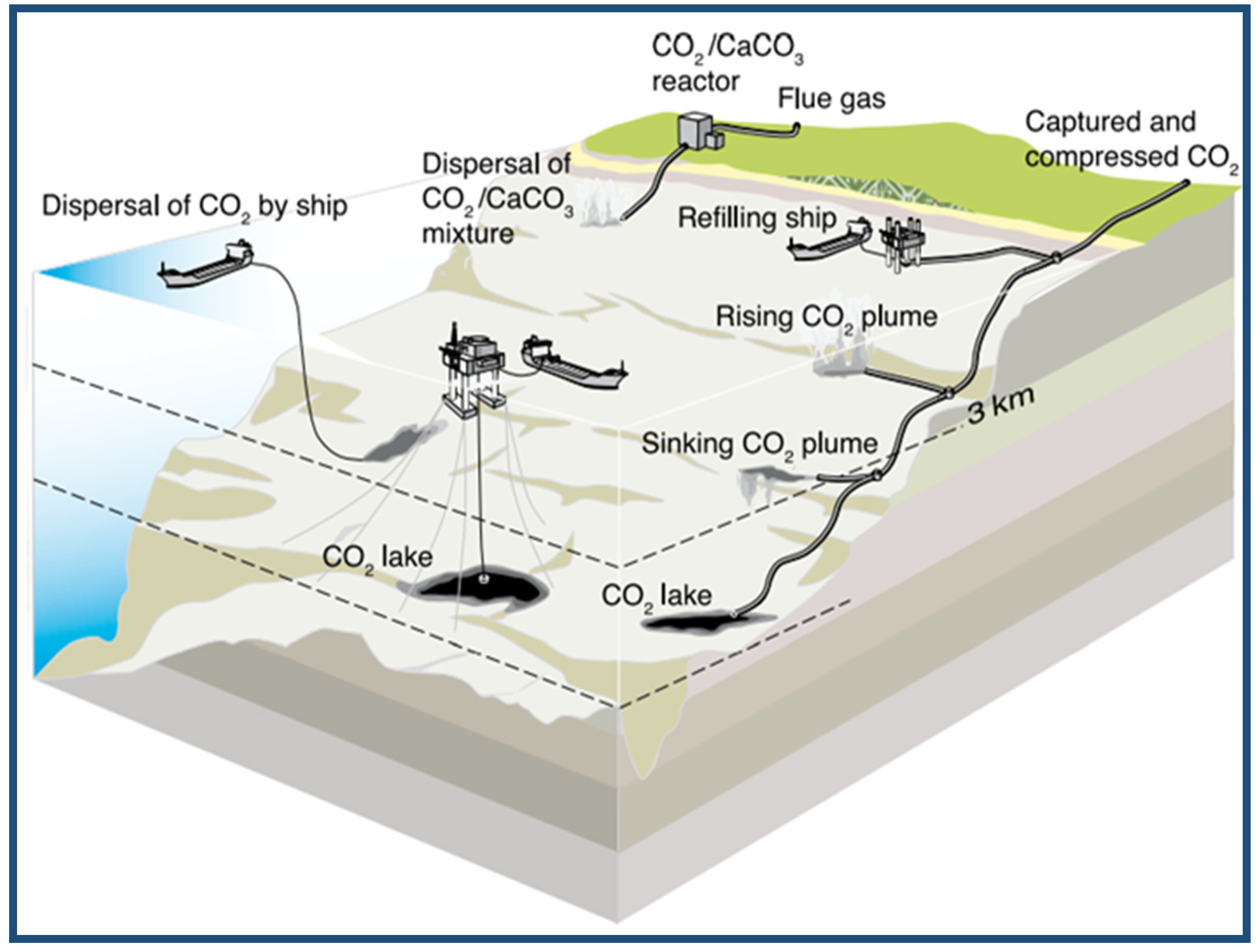
The captured carbon dioxide can be injected into the ocean, where it is stored out of the atmosphere for centuries. The CO2 can be transported via a pipeline or ship for release into the ocean. There have been many small-scale field experiments and over 25 years of studies of CO2 ocean storage, but it has not yet been implemented or completely evaluated [93]. However, for this technology to be deployed widely, it should be cost-effective, the environmental impacts must be considered, the techniques must be politically and legally feasible, and scientific certainty must be increased as to the effectiveness of the oceans to sequester CO2 safely [94]. Figure 11 shows an illustration of some ocean storage strategies [93].
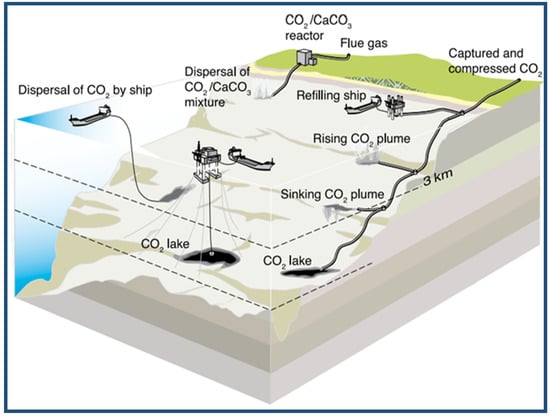
Figure 11.
Schematic of several ocean storage strategies. IPCC Special Report on carbon dioxide Capture and Storage. Adapted from Sean Goddard [95].
5. Current and Future Technology
It is uncertain whether the current and future technology will achieve the goal of zero carbon emissions. To have zero carbon emissions requires little to no carbon dioxide emissions to exist in the Earth’s atmosphere, as referred to in the circular carbon economy [96]. Next, we will mention some of the technologies that will help achieve the goal of zero carbon emissions. With concerted efforts from all countries aiming to responsibly reduce emissions and support research into affordable technologies, the researchers believe that this desired goal will be reached by the year 2050.
5.1. Fossil Fuel Alternatives
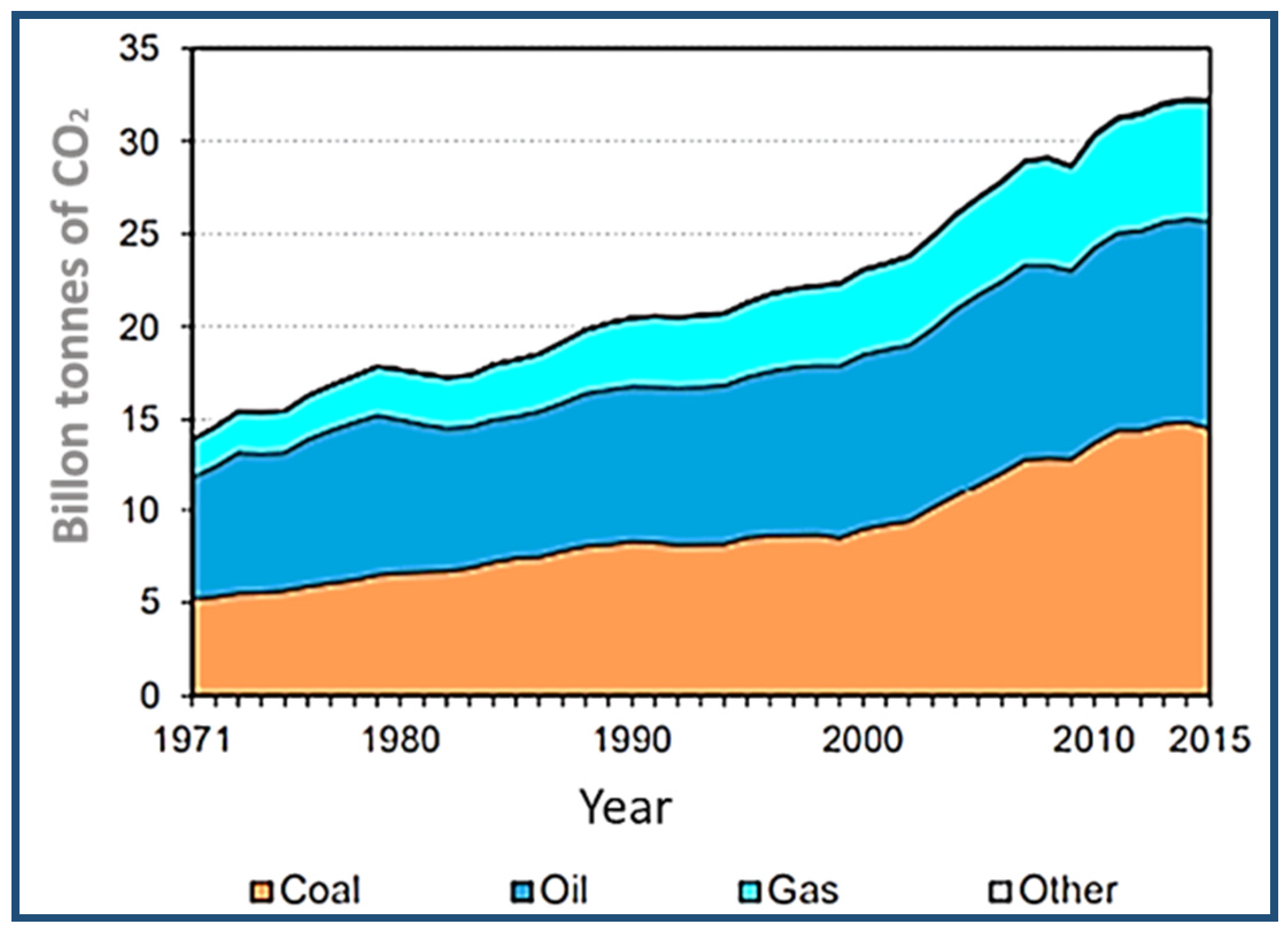
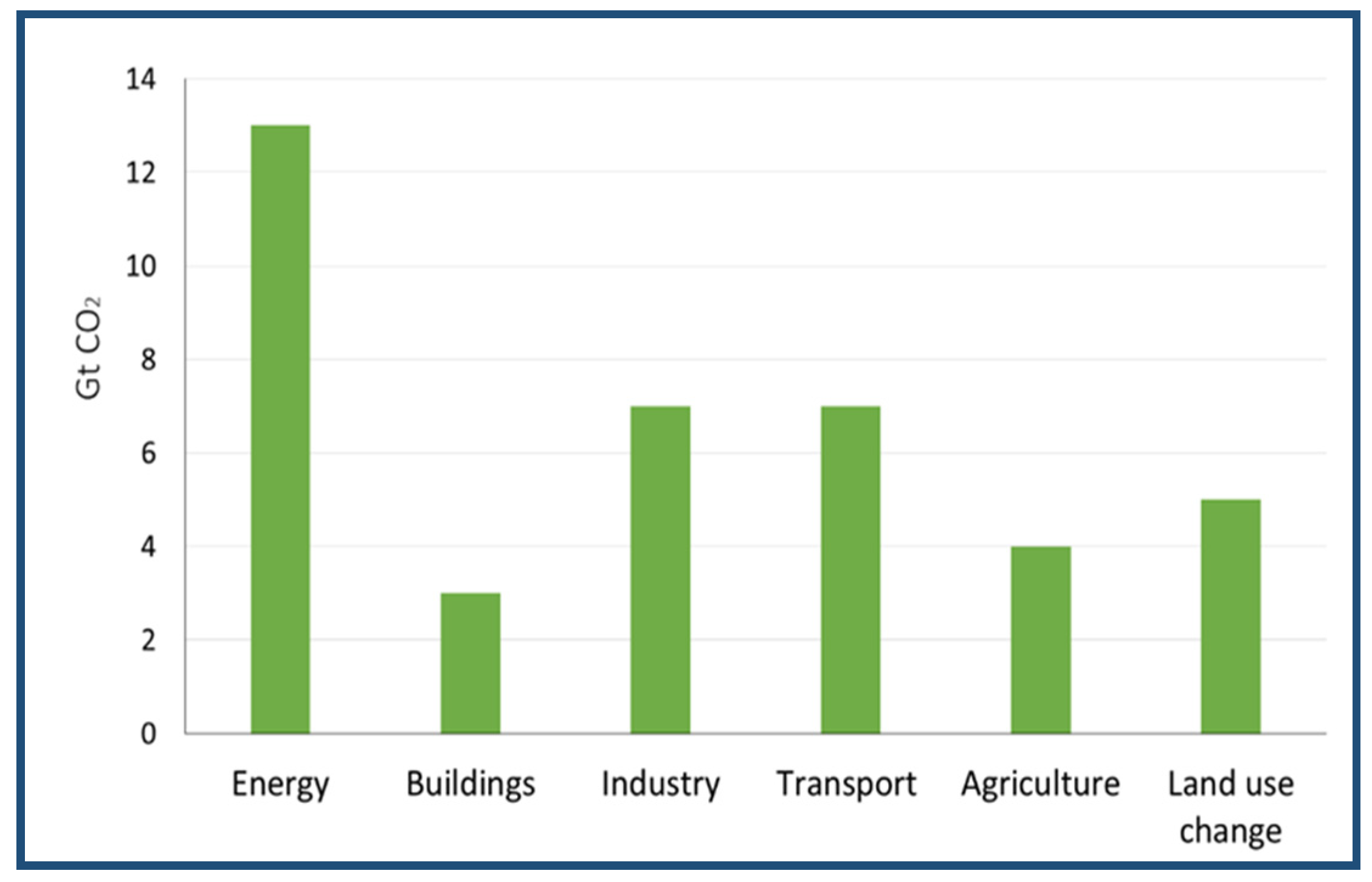
The options leading to the reduction in carbon dioxide emissions must first be defined, and then measures to reduce these emissions must be established [97]. Figure 12 shows the world’s sources of fuel-emitted carbon dioxide [98]. The CO2 emissions increased to 164.4%, and, based on IPCC estimates, the main carbon dioxide emission source over the 2008–2017 period is the energy sector [66], as illustrated in Figure 13. Given the urgent need to begin the shift towards greater emission reductions, it is imperative to define how mitigation measures are tailored to the mid-2050s, to see which measurements can be applied at present and which measures require longer lead times to be implemented. This problem could be partially solved by using clean energy sources, such as natural gas [99], geothermal power [100], solar power [101], wave energy [101], wind energy [102], biofuels [103], nuclear power [103], and by improving the quality of oil-based energy sources and reducing carbon dioxide emissions.
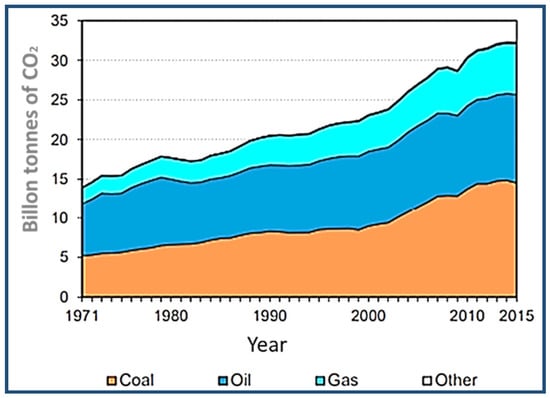
Figure 12.
World CO2 emissions by fuel type. Adapted from Reference [98].
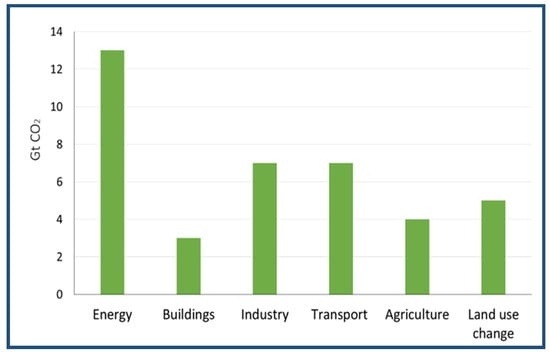
Figure 13.
The most emitting sources of carbon dioxide, based on IPCC estimates. The data are adapted from Reference [49].
5.2. Processing Building Materials
The options include some reuse and recycling measures that reduce emissions, with the aim of reaching a zero-carbon emission [104]. The figures in the following sections indicate the carbon dioxide emission reduction rate.
5.2.1. Steel Production and Use
Bio-based integrated steel plant fuel reduces carbon emissions by 9–90%. The integrated steel plant is on top of gas recycling, with a CCS of 50–60%. Hydrogen as a steel reduction agent 54–60%, while reinforcement steel from recycled steel 5–25%. Reinforcement steel produced with low-emission electricity 27%. Bio-based secondary steel heating oven fuel 6–27%. Finally, bio-based steel metallurgy fuel 8–12% [97].
5.2.2. Cement and Concrete Production and Use
An 8–28% reduction can be expected for bio-based cement plant fuel. Waste as cement plant, 1–20%. Traditional cement clinker substitutes, 9–41%. Conventional cement clinker substitutes are outside the current standards, 36–65%. Non-traditional waste-based cement clinker substitutes, 11–43%. Natural cement clinker substitutes, 20–46%, and advanced concrete use of an optimised concrete recipe, 9–85%. Design optimisation/use of precast concrete has a 4–9% reduction rate. Cement plant CCS, 32–100%. Cement plant electrification, 32–54%. An alternative bridge construction material, 9–48% [97].
5.2.3. Asphalt Production and Paving
Bio-based asphalt plant fuel, 33–47%; low-tempered warm asphalt (WMA), 5–19%; cold asphalt emulsion mixes, 40–68%, and asphalt recycling and reuse, 5–35%. The 3–15% aggregate moisture content, waste-based pavement substitutes, 5–6%, bio-fuelled/electric aggregate production, 4–6%, and bio-based bitumen 16–30% [97].
5.2.4. Heavy Transport
Bio-based heavy transport fuel is constructed at 35–90%. Hybrid heavy transports make up an 11–24% reduction. Fuel-celled/plug-in heavy trucks reduce emissions by 25–60%. The optimisation of logistics and road freight operations reduce emissions by 10–34% [97].
5.2.5. Construction Process
Bio-based construction equipment fuel constitutes a 35–90% reduction. Hybrid construction equipment is constructed at 10–50%. Fuel-celled/plug-in hybrid construction equipment, 50–60%. Electrified construction equipment, 66–95%. The optimisation of mass handling equipment has a reduction of 5–22%. Bio-fuelled electric rock crushing plants can reduce emissions by 16–96%. In-pit crushing and conveying constitute a 2–22% reduction, while recycled aggregates and base layers reduce emissions by 29–65% [97].
5.3. Hydrogen Technologies and Fuel Cells
Hydrogen technologies are expected to be successful in the future, and have many unique and beneficial advantages, including the significant reduction in greenhouse gas emissions [105]. An alternative and recommended solution for the future of decarbonised energy is hydrogen and fuel cells. Electrochemical converters and oxygen convert hydrogen or hydrogen-containing energy sources into direct electricity. It is used not only for transportation, but also as a way to store renewable energy from solar energy and wind farms. In addition, fuel and hydrogen technologies are used in home heating, where hydrogen is mixed in natural gas supplies and is targeted at decarbonising industrial processes. Hydrogen technologies have begun to play a significant role in the future, towards a carbon-free path, as they are the key to a future of renewable and sustainable energy. Strategies of hydrogen and fuel cell technologies include the following:
- (I)
- Hydrogen is used as a secondary value of energy. It uses the excess electrical energy generated from renewable energy sources to produce hydrogen via electrolysis, which also works in storing renewable energy.
- (II)
- The employment of fuel cells as significant energy providers, for transport and fixed energy sources.
- (III)
- Natural gas networks may be converted to hydrogen-based networks, dramatically reducing carbon dioxide emissions into the atmosphere [106].
5.4. Planting Trees and Mangrove Forests
Afforestation contributes to the release of oxygen, keeps the air from being polluted, and balances the ecosystem. Tree planting and afforestation may help reduce global warming, by taking in carbon dioxide and removing carbon dioxide from the atmosphere, when its use is properly planned. However, afforestation may not be a definitive solution to averting climate change. Rather, it can be used in combination with other greenhouse emission reduction methods [107]. Mangrove forests play an important role in mitigating the effects of climate change, as they provide an important environmental role for CO2 uptake. One of these roles is their ability to absorb carbon dioxide and store it in the form of “blue carbon” in the sediments of coastal areas [108]. Mangroves are able to store 15,514,807,200 tons of carbon dioxide [109]. Mangroves trees have an important role in sequestering CO2, as they can do so at a rate that is 100 times faster than the rate of terrestrial forests. Aramco’s mangrove initiative has seen the planting of more than two million mangrove seedlings in the Kingdom of Saudi Arabia, thus removing carbon dioxide and protecting sensitive areas of biodiversity [67]. The environmental benefits were not limited to carbon dioxide reduction and biodiversity protection; these trees can also protect the coast from wind and erosion, and provide shelter and food for some marine animals and birds. These benefits lead to the conclusion that mangroves are a green option [110].
5.5. Electric Transport
The second leading source of CO2 emissions and air pollution is road transport [111]. Electric transportation is a promising ideal solution, and is even more efficient than the use of fuel engines, as electric cars and electric buses are 3.5 and 2.5 times more efficient than cars and buses that run on fuel combustion, respectively. On the other hand, electric power may not be suitable in the case of long-distance sea and air transportation and heavy transportation. Therefore, it is better to use alternatives to fossil fuels in this case, such as biofuels and hydrogen [112].
In this sense, there is a departure from traditional combustion and a pivot towards electric cars that are powered by battery technology, such as electric vehicle technology and lithium-ion battery technology [113].
6. The Application That Involves CO2 and Its Economic Impact
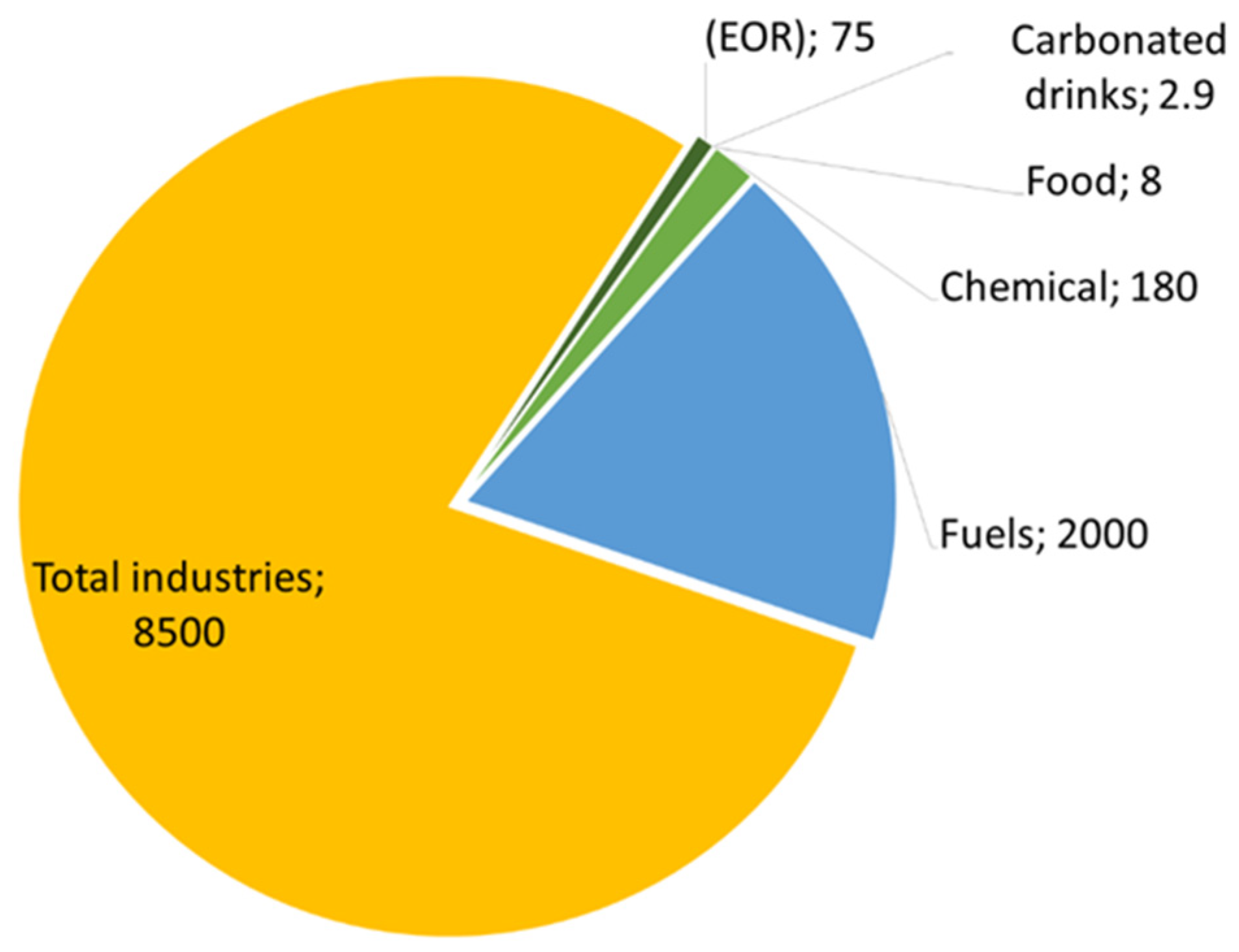
Carbon capture, utilisation, and storage (CCUS) emissions of chemical plants will eliminate 3.5 gigatons of carbon dioxide annually by 2030 [114]. It is possible to use CO2 in many products, with economic and beneficial returns in various chemical and biological fields, such as in food and other technologies. This will achieve the required climate change goals in a valuable and intelligent way [73,115,116]. For example, carbon dioxide is consumed in oil and gas manufacturing, enhanced oil recovery, food production, plant growth stimulation, manufacturing processes, etc. [115]. The use of carbon dioxide in these technologies reduces carbon dioxide emissions by 13% when compared to use in energy, fuel, and industrial conversion, as demonstrated in Figure 14. The production of chemicals, cement, and steel accounts for 60% of the total industry sector’s production [117].
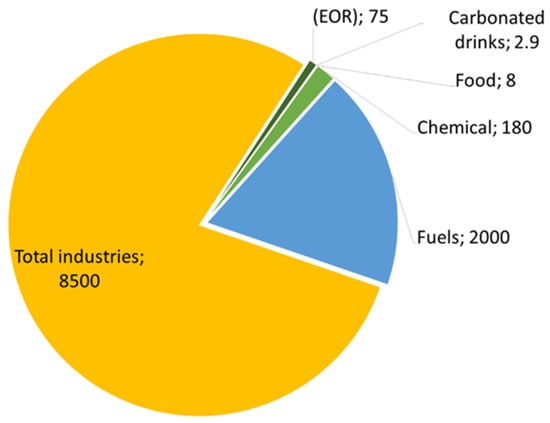
Figure 14.
Ratios of carbon dioxide consumption to its uses.
Carbon dioxide can be used in the production of many chemicals. Table 2 shows the amount of carbon dioxide consumed in producing some chemicals, along with their economic cost. The urea industry is the largest consumer of carbon dioxide in chemical production, consuming 130 gigatons of a total of 230 megatons of carbon dioxide per year [117]. Carbon dioxide reacts with ammonia at temperatures between 185 and 195 °C, to produce urea, which is distinguished as an agricultural fertiliser involved in the creation of some medicines and chemicals [118]. Many value-added materials can be produced on the market in fuel production, such as methane, methanol, gasoline, and engine fuel [115]. Two thousand gigatons of carbon dioxide per year are consumed in fuel production; this is considered a very high percentage [119]. In the production of enhanced oil recovery, carbon dioxide is used immediately after capture. As a result, 70–80 gigatons per year of CO2 are consumed.
There are several uses of carbon dioxide in food production, one of which is as a coolant to preserve food and drinks, while another is the production of dry ice, which has a greater cooling capacity than water ice [120]. In addition, carbon dioxide has recently been used as a packaging gas in the food industry, preserving fresh products and freshness for meat, fish, and pre-packaged food products, by controlling their biochemical metabolism [121]. Carbon dioxide production from ammonia was found to be higher than any other chemicals that use carbon dioxide, as demonstrated in Table 3.

Table 2.
The costs and amount of CO2 use in a year.
Table 2.
The costs and amount of CO2 use in a year.
| Pathway | Utilisation Potential (Mt of CO2 per Year) | The Cost of CO2 Utilisation (USD) per Year | Ref. |
|---|---|---|---|
| Total industries | 8500 | 3,544,500 million | [117] |
| Chemicals | 180 | 75,060 million | [119] |
| Fuels | 2000 | 834 billion | [119] |
| (EOR) | 70–80 | 29,190–33,360 million | [115] |
| Food | 8 | 3336 million | [122] |
| Carbonated drinks | 2.9 | 12,093.3 million | [123] |

Table 3.
Costs and amount of use of carbon dioxide for chemicals.
Table 3.
Costs and amount of use of carbon dioxide for chemicals.
| Pathway | Utilisation Potential CO2 per a Year | The Cost of CO2 Utilisation (USD) | Ref. |
|---|---|---|---|
| Urea | 130 Mt | 54,210 million | [115] |
| Methanol | 10.0 Mt | 4170 million | [123] |
| Cyclic carbonates | 0.04 Mt | 16,680 thousand | [124] |
| Dimethyl ether | 5.00 Mt | 2085 million | [123] |
| Ethylene carbonate | 0.04 Mt | 16,680 thousand | [124] |
| Di-methyl carbonate | 0.04 Mt | 16,680 thousand | [124] |
| Copolymers | 0.04 Mt | 16,680 thousand | [124] |
| Polymers | 1.50 Mt | 6255 million | [123] |
| Fine chemicals | 0.04 Mt | 16,680 thousand | [124] |
| Salicylic acid | 0.03 Mt | 12,510 thousand | [124] |
| Formaldehyde | 0.90 Mt | 3753 million | [123] |
| Formic acid | 5.00 Mt | 2085 million | [123] |
7. Socioeconomic Impact
It is known that the rise in pollutants, in general, affects public health and increases the financial burden on countries, due to the cost of treating conditions that may require long periods of medical attention. However, in this aspect, we address the relationship between carbon dioxide and the socioeconomic system. A study conducted in China [125] indicated that carbon dioxide emissions are increasing, due to an increase in traditional agricultural activity. The study was conducted from 1991 to 2018, and found a complementary relationship between economic growth and carbon emissions from an agricultural source. This study attributed this complementary relationship to emissions resulting from five sources involved in the agricultural field that produce carbon dioxide, with the most influential source identified as diesel. The study confirmed a steady proportion between economic growth and the increase in carbon dioxide levels. The study finally concluded that improving agricultural methods and using technologies that reduce carbon emissions will contribute to improving the socioeconomic situation.
In another study, it was shown that, despite the decline in the costs of renewable energy, such as wind and solar energy, in recent years, humankind is still reliant on energy produced from the burning of fossil fuels [126]. This is most evident in transportation, and the study argues that the removal of carbon from the transport and industry sector will face many challenges, leading to continued emissions of greenhouse gases. According to the authors, this will result in detrimental effects on global welfare and decreases in economic productivity. Thus, reducing economic productivity will reduce greenhouse gases, which will, in turn, lead to a reduction in global warming. The study indicates that the net effect of carbon feedback on socioeconomic impact is of about the same magnitude. It found that the levels of greenhouse gases in the atmosphere, predicted for the year 2100, are about the same as if the feedback from natural socioeconomic or biophysical sources were active.
Thus, there may be impacts on the socioeconomic situation, due to the increase in greenhouse gas rates. The effect of this may vary according to the economic activity of each country, the quality of the resources that the country depends on for energy production, and the extent to which the country keeps pace with modern technologies that benefit from all energy sources, and the country’s reliance on modern technologies that reduce emissions in general and greenhouse gases in particular. A comprehensive view of the use of all energy sources and the improvement of related technologies will undoubtedly lead to an improvement in the quality of socioeconomic life.
8. The Future of CCUS
There are the following two ways of decarbonising the future: capturing and storing carbon dioxide for natural gas plants, or removing natural gas as a fuel source by around 2030. Given that the latter possibility is unlikely, CCUS is ideal to preserve the climate [127]. Moving forward, CCUS is set on achieving its vision of zero emissions by 2050 [128,129]. CCUS technologies are currently considered immature, and when used in the future, with other reduction methods, such as renewable energy sources, they may solve climate and environmental problems [130]. The goal of the G20 agreement is to limit the global temperature rise to below 2 °C [131]. Achieving this goal requires significant effort to reduce and exploit carbon dioxide emissions in future years. With the current rapid pace of technological progress, we can reach a stable climate eventually.
Social tipping elements (STEs) and social tipping interventions (STIs) are paved paths towards zero carbon emissions in the future [128]. One study showed that the expected changes in 2050 would decrease oil consumption by at least 70% in the coming years. Reliance on renewable energy should be at about 30% to 40%, owing to the rapid adoption in electric transport [132]. There is also the following less expensive alternative technology to the non-biological methods of CCUS: the mineralisation or mineral carbonation (MC) process [133]. Even so, the use of fossil fuels may persist after 2050. Although renewable energy sources are the best solution to achieve zero emissions, they may also cause environmentally harmful waste. When the materials that make up some of the applications used to reduce emissions are disposed of, they may cause harm to the environment if they are not disposed of properly. Thus, it is important to discuss future environmental damage. For example, when solar panels contain lead and cadmium, these chemicals damage public health and are difficult to get rid of. It is therefore essential to encourage alternative energy industries that use environmentally friendly materials. Also, the batteries used in transportation devices contain lithium, mercury, and other substances that cause environmental hazards. Therefore, the modern battery industry must consider recycling and the use of environmentally friendly materials that do not harm human health.
9. Conclusions
This work demonstrates the importance of the economics of circular carbon and the importance of considering carbon dioxide as an essential chemical molecule. When there are inexpensive techniques to capture CO2 with high purity and transformational economic techniques, this will lead to the prosperity and growth of carbon economies.
Carbon dioxide can be used as a feedstock to produce urea, salicylic acid, and plastics that have already been produced routinely. Supercritical carbon dioxide also has many applications in food, chemicals, and micro-pharmaceuticals.
It is evident that there is an increase in the rates of carbon dioxide due to pollution from various industries, such as transportation. The most significant pollutants are electricity and heat production energy sources, as they were measured to have increased by 39–164.4% in the past decade. The worsening of this problem, coupled with the apparent decline in the rate of green cover due to deforestation and logging, which certainly plays a considerable role in the accumulation of greenhouse gas concentrations, is expected to continue in the future. The technologies to capture, use, and store (CCUS) carbon dioxide achieve a significant and profound goal of reducing carbon dioxide emissions. With the help of technologies that reduce emissions to achieve a zero-carbon target, approximately 3.5 gigatons of emissions will be reduced by 2030.
There are significant economies based on CO2; the most important is the industrial sector, which produces 8.5 gigatons of carbon dioxide. Therefore, using and storing carbon dioxide is a process of very high economic benefit. The interest in increasing green cover and preserving forests is essential in maintaining the balance in carbon dioxide levels.
In summary, oil and natural gas will remain the main source of energy, and with improved CO2 capture and sequestration, it will be the right choice for decades, with the importance of adopting circular carbon economy policies for existing and modern companies.
In addition, all modern means and necessary measures must be taken into account when constructing new electrical plants or factories. Furthermore, supporting clean energy research is of paramount importance, as it must be taken into account that these technologies and their products rely on environmentally and human-friendly materials, while setting specifications and laws that limit the use of materials harmful to humans and the environment.
All this must be conducted before adopting any technologies for new energy sources to avoid any undesirable effects that may result from them due to the type of waste produced, which may be difficult to dispose of and pose a real threat to the environment, human health, and safety.
Author Contributions
L.M.A., collecting data writing up the manuscript; A.S.A., collecting data; N.B.A., producing the figures, and editing; N.H.K., editing and review. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
No humans or animals involved in this research.
Informed Consent Statement
There is no patient in this study.
Data Availability Statement
All data and articles are available.
Acknowledgments
The authors thank King Abdulaziz City for Science and Technology (KACST) for technical support for this study.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Grim, R.G.; Huang, Z.; Guarnieri, M.T.; Ferrell, J.R.; Tao, L.; Schaidle, J.A. Transforming the carbon economy: Challenges and opportunities in the convergence of low-cost electricity and reductive CO2 utilisation. Energy Environ. Sci. 2020, 13, 472–494. [Google Scholar] [CrossRef]
- Rizos, V.; Egenhofer, C.; Elkerbout, M. Circular Economy for Climate Neutrality: Setting the Priorities for the EU. CEPS Policy Brief 2019. Available online: https://papers.ssrn.com/sol3/papers.cfm?abstract_id=3493573 (accessed on 1 September 2021).
- DiChristopher, T. EPA Chief Scott Pruitt Says Carbon Dioxide Is Not a Primary Contributor to Global Warming. CNBC 2017. Available online: https://www.cnbc.com/2017/03/09/epa-chief-scott-pruitt.html (accessed on 1 September 2021).
- Res, C.; April, P.; Idso, S.B. CO2-induced global warming: A skeptic’s view of potential climate change. Clim. Res. 1998, 10, 69–82. [Google Scholar]
- Covey, K.; Soper, F.; Pangala, S.; Bernardino, A.; Pagliaro, Z.; Basso, L.; Cassol, H.; Fearnside, P.; Navarrete, D.; Novoa, S.; et al. Carbon and beyond: The biogeochemistry of climate in a rapidly changing Amazon. Front. For. Glob. Chang. 2021, 4, 11. [Google Scholar]
- Maghrabi, A.; Almutairi, M.; Aldosari, A.; Altilasi, M.; Alshehri, A. Cosmic rays detection in Saudi Arabia: Review of the facilities and preliminarily results. J. King Saud Univ. Sci. 2021, 33, 101495. [Google Scholar] [CrossRef]
- Seppälä, A.; Matthes, K.; Randall, C.E.; Mironova, I.A. What is the solar influence on climate? Overview of activities during CAWSES-II. Eur. J. Nucl. Med. Mol. Imaging 2014, 1, 1–12. [Google Scholar] [CrossRef] [Green Version]
- Gray, L.J.; Beer, J.; Geller, M.; Haigh, J.D.; Lockwood, M.; Matthes, K.; Cubasch, U.; Fleitmann, D.; Harrison, G.; Hood, L.; et al. Solar influences on climate. Rev. Geophys. 2010, 48. [Google Scholar] [CrossRef]
- Junior, C.H.L.S.; Pessôa, A.C.M.; Carvalho, N.S.; Reis, J.B.C.; Anderson, L.O.; Aragão, L.E.O.C. The Brazilian Amazon deforestation rate in 2020 is the greatest of the decade. Nat. Ecol. Evol. 2020, 5, 144–145. [Google Scholar] [CrossRef]
- Cramer, W.; Bondeau, A.; Schaphoff, S.; Lucht, W.; Smith, B.; Sitch, S. Tropical forests and the global carbon cycle: Impacts of atmospheric carbon dioxide, climate change and rate of deforestation. Philos. Trans. R. Soc. B Biol. Sci. 2004, 359, 331–343. [Google Scholar] [CrossRef] [Green Version]
- Gomes, V.H.F.; Vieira, I.C.G.; Salomão, R.P.; Ter Steege, H. Amazonian tree species threatened by deforestation and climate change. Nat. Clim. Chang. 2019, 9, 547–553. [Google Scholar] [CrossRef]
- Nwilo, P.C.; Olayinka, D.N.; Okolie, C.J.; Emmanuel, E.I.; Orji, M.J.; Daramola, O.E. Impacts of land cover changes on deserti fi cation in northern Nigeria and implications on the Lake Chad Basin. J. Arid Environ. 2020, 181, 104190. [Google Scholar] [CrossRef]
- Seven Messages about the Circular Economy and Climate Change. World Resour. Forum 2019. Available online: https://www.ovam.be/ (accessed on 1 September 2021).
- Williams, E. Achieving Climate Goals by Closing the Loop in a Circular Carbon Economy; KAPSARC: Riyadh, Saudi Arabia, 2019; pp. 1–13. [Google Scholar]
- Djuričin, S.; Pataki, D.E.; Xu, X. A comparison of tracer methods for quantifying CO2 sources in an urban region. J. Geophys. Res. Space Phys. 2010, 115, 1–13. [Google Scholar] [CrossRef]
- Schmelz, W.J.; Hochman, G.; Miller, K.G. Total cost of carbon capture and storage implemented at a regional scale: Northeastern and midwestern United States: Total Cost of Carbon Capture and Storage. Interface Focus 2020, 10, 20190065. [Google Scholar] [CrossRef]
- Kenarsari, S.D.; Yang, D.; Jiang, G.; Zhang, S.; Wang, J.; Russell, A.G.; Wei, Q.; Fan, M. Review of recent advances in carbon dioxide separation and capture. RSC Adv. 2013, 3, 22739–22773. [Google Scholar] [CrossRef]
- Terlouw, T.; Bauer, C.; Rosa, L.; Mazzotti, M. Life cycle assessment of carbon dioxide removal technologies: A critical review. Energy Environ. Sci. 2021, 14, 1701–1721. [Google Scholar] [CrossRef]
- Zhong, W.; Haigh, J.D. The greenhouse effect and carbon dioxide. Weather 2013, 68, 100–105. [Google Scholar] [CrossRef]
- Trenberth, K.E.; Fasullo, J.T. Tracking Earth’s energy: From El Niño to Global Warming. Surv. Geophys. 2012, 33, 413–426. [Google Scholar] [CrossRef] [Green Version]
- Trenberth, K.E.; Fasullo, J.; Kiehl, J. Earth’s global energy budget. Bull. Am. Meteorol. Soc. 2009, 90, 311–324. [Google Scholar] [CrossRef]
- Morgan, M.R. Climate change 2001. Weather 2004, 59, 217–223. [Google Scholar] [CrossRef]
- Intergovernmental Panel on Climate Change. Climate Change 2014: Mitigation of Climate Change; Cambridge University Press: Cambridge, UK, 2014. [Google Scholar] [CrossRef] [Green Version]
- Renewables, V. Inventory of U.S. greenhouse gas emissions and sinks: 1990–2009. Fed. Regist. 2011, 76, 10026. [Google Scholar]
- WMO; GTW. The State of Greenhouse Gases in the Atmosphere. Greenh. Gas Bull. 2020, 16, 5–8. [Google Scholar]
- Graber, J.; Amthor, J.; Dahlman, R.; Drell, D.; Weatherwax, S. Carbon Cycling and Biosequestration Integrating Biology and Climate through Systems Science Report from the March 2008 Workshop. Available online: https://doi.org/10.2172/948438 (accessed on 1 September 2021).
- Sunquist, E.T.; Broecker, W.S. The carbon cycle and atmospheric CO2. Eos Trans. Am. Geophys. Union 1986, 67, 191. [Google Scholar] [CrossRef]
- Seinberh, M. History of CO2 greenhouse gas mitigation technologies. Energy Convers. Manag. 1992, 33, 311–315. [Google Scholar]
- Ebhota, W.S.; Tabakov, P.Y. Development of domestic technology for sustainable renewable energy in a zero-carbon emission -driven economy. Int. J. Environ. Sci. Technol. 2020, 18, 1253–1268. [Google Scholar] [CrossRef]
- Korhonen, J.; Honkasalo, A.; Seppälä, J. Circular economy: The concept and its limitations. Ecol. Econ. 2018, 143, 37–46. [Google Scholar] [CrossRef]
- Ellen MacArthur Foundation. Completing the Picture: How the Circular Economy Tackles Climate Change; Ellen MacArthur Foundation: Cowes, UK, 2019; pp. 1–62. [Google Scholar]
- Ma, S.; Hu, S.; Chen, D.; Zhu, B. A case study of a phosphorus chemical firm’s application of resource efficiency and eco-efficiency in industrial metabolism under circular economy. J. Clean. Prod. 2015, 87, 839–849. [Google Scholar] [CrossRef]
- Arabia, S. G20 Drives Joint Efforts to Safeguard the Planet. 2020. Available online: https://www.g20.org/ (accessed on 1 September 2021).
- Arabia, S. G20 Statement on the Circular Carbon Economy; G20 Engagement Groups: New York, NY, USA, 2020; p. 20. [Google Scholar]
- Gas, I.; March, U. G20 Saudi Arabia the Role of Gas Technologies in the Circular Carbon Economy; International Gas Union: Vevey, Switzerland, 2020. [Google Scholar]
- Annual Report 2018: Growing through Transformation. 2018. Available online: https://www.sabic.com/assets/zh/Images/SABIC-AR-English-2018_tcm11-18629.pdf (accessed on 1 September 2021).
- Wich, T.; Lueke, W.; Deerberg, G.; Oles, M. Carbon2Chem®-CCU as a step toward a circular economy. Front. Energy Res. 2020, 7, 162. [Google Scholar] [CrossRef] [Green Version]
- Farabi-Asl, H.; Itaoka, K.; Chapman, A.; Kato, E.; Kurosawa, A. Key factors for achieving emission reduction goals cognizant of CCS. Int. J. Greenh. Gas Control. 2020, 99, 103097. [Google Scholar] [CrossRef]
- Wilberforce, T.; Olabi, A.; Sayed, E.T.; Elsaid, K.; Abdelkareem, M.A. Progress in carbon capture technologies. Sci. Total Environ. 2020, 761, 143203. [Google Scholar] [CrossRef]
- Netto, A.L.A.; Câmara, G.; Rocha, E.; Silva, A.L.; Andrade, J.C.S.; Peyerl, D.; Rocha, P. A first look at social factors driving CCS perception in Brazil: A case study in the Recôncavo Basin. Int. J. Greenh. Gas Control 2020, 98, 103053. [Google Scholar] [CrossRef]
- Mathieu, P. The IPCC special report on carbon dioxide capture and storage. ECOS 2006. In Proceedings of the 19th International Conference on Efficiency, Cost, Optimisation, Simulation and Environmental Impact of Energy Systems, Aghia Pelagia, Greece, 12–14 July 2006. [Google Scholar]
- CCSA; TUC. The Economic Benefits of Carbon Capture and Storage in the UK. Trades Union Congr. 2014. Available online: https://www.tuc.org.uk/sites/default/files/carboncapturebenefits_1.pdf (accessed on 1 September 2021).
- Olfe-Kräutlein, B. Advancing CCU technologies pursuant to the SDGs: A challenge for policy Making. Front. Energy Res. 2020, 8, 1–16. [Google Scholar] [CrossRef]
- Ravanchi, M.T.; Sahebdelfar, S. Catalytic conversions of CO2 to help mitigate climate change: Recent process developments. Process Saf. Environ. Prot. 2021, 145, 172–194. [Google Scholar] [CrossRef]
- Styring, P.; Jansen, D.; de Coninck, H.; Reith, H.; Armstrong, K. Carbon Capture and Utilisation in the Green Economy; Centre for Low Carbon Futures: New York, NY, USA, 2011. [Google Scholar]
- Goel, M. Carbon capture and storage technology for sustainable energy. OPEC Bull. 2010, 39, 20–23. [Google Scholar]
- Bui, M.; Adjiman, C.S.; Bardow, A.; Anthony, E.J.; Boston, A.; Brown, S.; Fennell, P.S.; Fuss, S.; Galindo, A.; Hackett, L.A.; et al. Carbon capture and storage (CCS): The way forward. Energy Environ. Sci. 2018, 11, 1062–1176. [Google Scholar] [CrossRef] [Green Version]
- Hepburn, C.; Adlen, E.; Beddington, J.; Carter, E.A.; Fuss, S.; Mac Dowell, N.; Minx, J.C.; Smith, P.; Williams, C.K. The technological and economic prospects for CO2 utilisation and removal. Nature 2019, 575, 87–97. [Google Scholar] [CrossRef] [Green Version]
- Global CO2 Initiative. Global Roadmap for Implementing CO2 Utilization; University of Michigan: Ann Arbor, MI, USA, 2016. [Google Scholar]
- Hasan, M.M.F.; First, E.L.; Boukouvala, F.; Floudas, C.A. A multi-scale framework for CO2 capture, utilisation, and sequestration: CCUS and CCU. Comput. Chem. Eng. 2015, 81, 2–21. [Google Scholar] [CrossRef] [Green Version]
- Creamer, A.E.; Gao, B. Carbon Dioxide Capture: An Effective Way to Combat Global Warming; Springer: Cham, Switzerland, 2015. [Google Scholar]
- Koytsoumpa, E.I.; Bergins, C.; Kakaras, E. The CO2 economy: Review of CO2 capture and reuse technologies. J. Supercrit. Fluids 2018, 132, 3–16. [Google Scholar] [CrossRef]
- Rosli, A.; Ahmad, A.L.; Lim, J.K.; Low, S.C. Advances in liquid absorbents for CO2 capture: A review. J. Phys. Sci. 2017, 28, 121–144. [Google Scholar] [CrossRef] [Green Version]
- Wang, Q.; Luo, J.; Zhong, Z.; Borgna, A. CO2 capture by solid adsorbents and their applications: Current status and new trends. Energy Environ. Sci. 2011, 4, 42–55. [Google Scholar] [CrossRef]
- Khdary, N.H.; Ghanem, M.A.; Merajuddine, M.G.; Bin Manie, F.M. Incorporation of Cu, Fe, Ag, and Au nanoparticles in mercapto-silica (MOS) and their CO2 adsorption capacities. J. CO2 Util. 2014, 5, 17–23. [Google Scholar] [CrossRef]
- Baxter, L.; Baxter, A.; Burt, S. Cryogenic CO2 capture as a cost-effective CO2 capture process. Int. J. Hydrog. Energy 2011, 36, 10355–10365. [Google Scholar]
- Castel, C.; Bounaceur, R.; Favre, E. Membrane processes for direct carbon dioxide capture from air: Possibilities and limitations. Front. Chem. Eng. 2021, 3, 17. [Google Scholar] [CrossRef]
- Khdary, N.H.; Abdelsalam, M.E. Polymer-silica nanocomposite membranes for CO2 capturing. Arab. J. Chem. 2020, 13, 557–567. [Google Scholar] [CrossRef]
- Zhou, X.; Zang, X.; Long, Z.; Liang, D. Multiscale analysis of the hydrate based carbon capture from gas mixtures containing carbon dioxide. Sci. Rep. 2021, 11, 9197. [Google Scholar] [CrossRef] [PubMed]
- Hu, J.; Galvita, V.V.; Poelman, H.; Marin, G.B. Advanced chemical looping materials for CO2 utilisation: A review. Materials 2018, 11, 1187. [Google Scholar] [CrossRef] [Green Version]
- National Research Council. Carbon Management: Implications for R&D in the Chemical Sciences and Technology; The National Academies Press: Washington, DC, USA, 2001. [Google Scholar]
- Song, C.; Liu, Q.; Deng, S.; Li, H.; Kitamura, Y. Cryogenic-based CO2 capture technologies: State-of-the-art developments and current challenges. Renew. Sustain. Energy Rev. 2019, 101, 265–278. [Google Scholar] [CrossRef]
- Eide, L.I.; Batum, M.; Dixon, T.; Elamin, Z.; Graue, A.; Hagen, S.; Hovorka, S.; Nazarian, B.; Nøkleby, P.H.; Olsen, G.I.; et al. Enabling large-scale carbon capture, utilisation, and storage (CCUS) using offshore carbon dioxide (CO2) infrastructure developments—A review. Energies 2019, 12, 1945. [Google Scholar] [CrossRef] [Green Version]
- Jia, B.; Tsau, J.-S.; Barati, R. A review of the current progress of CO2 injection EOR and carbon storage in shale oil reservoirs. Fuel 2019, 236, 404–427. [Google Scholar] [CrossRef]
- Adlen, E.; Hepburn, C. Carbon Capture methods compared: Costs, scalability, permanence, cleanness related posts. Energy Post 2019, 10, 1–8. Available online: https://energypost.eu/10-carbon-capture-methods-compared-costs-scalability-permanence-cleanness/ (accessed on 1 September 2021).
- Ahmad, O. Khowaiter, the Circular Carbon Economy; Saudi Aramco: Dhahran, Saudi Arabia, 2020. [Google Scholar]
- Farajzadeh, R.; Eftekhari, A.A.; Dafnomilis, G.; Lake, L.; Bruining, J. On the sustainability of CO2 storage through CO2—Enhanced oil recovery. Appl. Energy 2020, 261, 114467. [Google Scholar] [CrossRef]
- Alshammari, Y.M. Achieving climate targets via the circular carbon economy: The case of Saudi Arabia. C J. Carbon Res. 2020, 6, 54. [Google Scholar] [CrossRef]
- Sorcar, S.; Hwang, Y.; Lee, J.; Kim, H.; Grimes, K.M.; Grimes, C.A.; Jung, J.-W.; Cho, C.-H.; Majima, T.; Hoffmann, M.R.; et al. CO2, water, and sunlight to hydrocarbon fuels: A sustained sunlight to fuel (Joule-to-Joule) photoconversion efficiency of 1%. Energy Environ. Sci. 2019, 12, 2685–2696. [Google Scholar] [CrossRef] [Green Version]
- Mikhelkis, L.; Govindarajan, V. Techno-economic and partial environmental analysis of carbon capture and storage (CCS) and carbon capture, utilisation, and storage (CCU/S): Case study from proposed waste-fed district-heating incinerator in Sweden. Sustainability 2020, 12, 5922. [Google Scholar] [CrossRef]
- Pérez-Fortes, M.; Bocin-Dumitriu, A.; Tzimas, E. CO2 utilisation pathways: Techno-economic assessment and market opportunities. Energy Procedia 2014, 63, 7968–7975. [Google Scholar] [CrossRef]
- Zhang, Z.; Pan, S.-Y.; Li, H.; Cai, J.; Olabi, A.G.; Anthony, E.J.; Manovic, V. Recent advances in carbon dioxide utilisation. Renew. Sustain. Energy Rev. 2020, 125, 109799. [Google Scholar] [CrossRef]
- Zhang, N.; Duan, H.; Miller, T.R.; Tam, V.W.; Liu, G.; Zuo, J. Mitigation of carbon dioxide by accelerated sequestration in concrete debris. Renew. Sustain. Energy Rev. 2020, 117, 109495. [Google Scholar] [CrossRef]
- Tang, D.Y.Y.; Yew, G.Y.; Koyande, A.K.; Chew, K.W.; Vo, D.V.N.; Show, P.L. Green technology for the industrial production of biofuels and bioproducts from microalgae: A review. Environ. Chem. Lett. 2020, 18, 1967–1985. [Google Scholar] [CrossRef]
- Arun, J.; Gopinath, K.P.; SundarRajan, P.; Felix, V.; JoselynMonica, M.; Malolan, R. A conceptual review on microalgae biorefinery through thermochemical and biological pathways: Bio-circular approach on carbon capture and wastewater treatment. Bioresour. Technol. Rep. 2020, 11, 100477. [Google Scholar] [CrossRef]
- Cheng, J.; Yang, Z.; Huang, Y.; Huang, L.; Hu, L.; Xu, D.; Zhou, J.; Cen, K. Improving growth rate of microalgae in a 1191m2 raceway pond to fix CO2 from flue gas in a coal-fired power plant. Bioresour. Technol. 2015, 190, 235–241. [Google Scholar] [CrossRef]
- Langholtz, M.; Busch, I.; Kasturi, A.; Hilliard, M.R.; McFarlane, J.; Tsouris, C.; Mukherjee, S.; Omitaomu, O.A.; Kotikot, S.M.; Allen-Dumas, M.R.; et al. The economic accessibility of CO2 sequestration through bioenergy with carbon capture and storage (BECCS) in the US. Land 2020, 9, 299. [Google Scholar] [CrossRef]
- Santori, G.; Charalambous, C.; Ferrari, M.; Brandani, S. Adsorption arti fi cial tree for atmospheric carbon dioxide capture, puri fi cation and compression. Energy 2018, 162, 1158–1168. [Google Scholar] [CrossRef] [Green Version]
- Yang, Z.; Xu, T.; Wang, F.; Diao, Y.; Li, X.; Ma, X.; Tian, H. A study on the CO2-enhanced water recovery efficiency and reservoir Pressure Control Strategies. Geofluids 2019. [Google Scholar] [CrossRef] [Green Version]
- Yang, Z.; Dai, Z.; Xu, T.; Wang, F.; Jia, S. Effects of Dip-Angle on the CO2-Enhanced Water Recovery Efficiency and Reservoir Pressure Control Strategies. 2021. Available online: https://doi.org/10.5194/egusphere-egu2020-13028 (accessed on 1 September 2021).
- Araújo, O.D.Q.F.; De Medeiros, J.L. Carbon capture and storage technologies: Present scenario and drivers of innovation. Curr. Opin. Chem. Eng. 2017, 17, 22–34. [Google Scholar] [CrossRef]
- Pattnaik, R.; Sethi, K.; Das, D. Soil carbon sequestration through various agronomic practices: A way to mitigate climate change impacts. J. Pharmacogn. Phytochem. 2020, 9, 755–761. [Google Scholar]
- IEAGHG. Case Studies of CO2 Storage in Depleted Oil and Gas Fields; IEAGHG: Cheltenham, UK, 2017. [Google Scholar]
- Hannis, S.; Lu, J.; Chadwick, A.; Hovorka, S.; Kirk, K.; Romanak, K.; Pearce, J. CO2 storage in depleted or depleting oil and gas fields: What can we learn from existing projects? Energy Procedia 2017, 114, 5680–5690. [Google Scholar] [CrossRef]
- Kolenković, I.; Saftić, B. Geological storage of carbon dioxide. Rudarsko Geolosko Naftni Zbornik 2014, 28, 9–22. [Google Scholar]
- Maldal, T.; Tappel, I. CO2 underground storage for Snøhvit gas field development. Energy 2004, 29, 1403–1411. [Google Scholar] [CrossRef]
- Mathieson, A.; Midgely, J.; Wright, I.; Saoula, N.; Ringrose, P. In Salah CO2 storage JIP: CO2 sequestration monitoring and verification technologies applied at Krechba, Algeria. Energy Procedia 2011, 4, 3596–3603. [Google Scholar] [CrossRef] [Green Version]
- Global CCS Institute. Geological Storage of CO2: Safe, Permanent, and Abundant; Global CCS Institute: Washington, DC, USA, 2018. [Google Scholar]
- Lackner, K.S.; Brennan, S. Envisioning carbon capture and storage: Expanded possibilities due to air capture, leakage insurance, and C-14 monitoring. Clim. Chang. 2009, 96, 357–378. [Google Scholar] [CrossRef]
- Lackner, K.S.; Brennan, S.; Matter, J.M.; Park, A.H.A.; Wright, A.; Van der Zwaan, B. The urgency of the development of CO2 capture from ambient air. Proc. Natl. Acad. Sci. USA 2012, 109, 13156–13162. [Google Scholar] [CrossRef] [Green Version]
- IEAGHG. Steve Goldthorpe, Carbonation and Enhanced; Report:2013/TR6; IEAGHG: Cheltenham, UK, 2013. [Google Scholar]
- Rackley, S.A. Ocean storage. Carbon Capture Storage 2010. [Google Scholar] [CrossRef]
- Heinrich, J. Legal Implications of CO2 Ocean Storage; Laboratory for Energy and the Environment: Cambridge, UK, 2002; p. 17. [Google Scholar]
- Zhang, Y.; Lu, X.; Ji, X. Carbon dioxide capture. Deep Eutectic Solvents Synth. Prop. Appl. 2019, 297–319. [Google Scholar] [CrossRef]
- Preparing Your Business for the Zero-Carbon Circular Economy. Available online: http://www.lowcarbonlivingcrc.com.au/sites/all/files/publications_file_attachments/lclguide_buildingsmes_web.pdf (accessed on 1 September 2021).
- Karlsson, I.; Rootzén, J.; Johnsson, F. Reaching net-zero carbon emissions in construction supply chains—Analysis of a Swedish road construction project. Renew. Sustain. Energy Rev. 2019, 120, 109651. [Google Scholar] [CrossRef]
- Chen, H.M. Additional resources and final remarks. Libr. Technol. Rep. 2017, 53, 28–30. [Google Scholar]
- IEA. Global Energy Review 2020: The Impacts of the COVID-19 Crisis on Global Energy Demand and CO2 Emissions; OECD Publishing: Paris, France, 2020. [Google Scholar] [CrossRef]
- Mostafaeipour, A.; Dehshiri, S.H.; Dehshiri, S.H.; Jahangiri, M.; Techato, K. A thorough analysis of potential geothermal project locations in afghanistan. Sustainability 2020, 12, 8397. [Google Scholar] [CrossRef]
- Kihlström, V.; Elbe, J. Constructing markets for solar energy—a review of literature about market barriers and government responses. Sustainability 2021, 13, 3273. [Google Scholar] [CrossRef]
- Darwish, A.S.; Al-Dabbagh, R. Wind energy state of the art: Present and future technology advancements. Renew. Energy Environ. Sustain. 2020, 5, 7. [Google Scholar] [CrossRef]
- Brown, A.; Waldheim, L.; Landalv, I.; Saddler, J.; Ebadian, M.; McMillan, J.D.; Bonomi, A.; Klein, B. Advanced Biofuels—Potential for Cost Reduction. IEA Bioenergy 2020. Available online: https://www.ieabioenergy.com/wp-content/uploads/2020/02/T41_CostReductionBiofuels-11_02_19-final.pdf (accessed on 1 September 2021).
- Pacheco-Torgal, F.; Labrincha, J.A. The future of construction materials research and the seventh un Millennium Development Goal: A few insights. Constr. Build. Mater. 2013, 40, 729–737. [Google Scholar] [CrossRef]
- Iida, S.; Sakata, K. Hydrogen technologies and developments in Japan. Clean Energy 2019, 3, 105–113. [Google Scholar] [CrossRef] [Green Version]
- Thomas, J.M.; Edwards, P.P.; Dobson, P.J.; Owen, G.P. Decarbonising energy: The developing international activity in hydrogen technologies and fuel cells. J. Energy Chem. 2020, 51, 405–415. [Google Scholar] [CrossRef]
- Waring, B.; Neumann, M.; Prentice, I.C.; Adams, M.; Smith, P.; Siegert, M. What role can forests play in tackling climate change? Grantham Inst. Discuss. Pap. 2020, 6, 1–8. [Google Scholar]
- Eid, E.M.; Arshad, M.; Shaltout, K.H.; El-Sheikh, M.A.; Alfarhan, A.H.; Picó, Y.; Barcelo, D. Effect of the conversion of mangroves into shrimp farms on carbon stock in the sediment along the southern Red Sea coast, Saudi Arabia. Environ. Res. 2019, 176, 108536. [Google Scholar] [CrossRef] [PubMed]
- Jakovac, C.C.; Latawiec, A.E.; Lacerda, E.; Lucas, I.L.; Korys, K.A.; Iribarrem, A.; Malaguti, G.A.; Turner, R.K.; Luisetti, T.; Strassburg, B.B.N. Costs and Carbon Benefits of Mangrove Conservation and Restoration: A Global Analysis. Ecol. Econ. 2020, 176, 106758. [Google Scholar] [CrossRef]
- Shaltout, K.H.; Ahmed, M.T.; Alrumman, S.A.; Ahmed, D.A.; Eid, E.M. Evaluation of the carbon sequestration capacity of arid mangroves along nutrient availability and salinity gradients along the Red Sea coastline of Saudi Arabia. Oceanologia 2019, 62, 56–69. [Google Scholar] [CrossRef]
- Bonsu, N.O. Towards a circular and low-carbon economy: Insights from the transitioning to electric vehicles and net zero economy. J. Clean. Prod. 2020, 256, 120659. [Google Scholar] [CrossRef]
- Kätelhön, A.; Meys, R.; Deutz, S.; Suh, S.; Bardow, A. Climate change mitigation potential of carbon capture and utilisation in the chemical industry. Proc. Natl. Acad. Sci. USA 2019, 116, 11187–11194. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Manthiram, A. An outlook on lithium ion battery technology. ACS Cent. Sci. 2017, 3, 1063–1069. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kaiser, S.; Bringezu, S. Use of carbon dioxide as raw material to close the carbon cycle for the German chemical and polymer industries. J. Clean. Prod. 2020, 271, 122775. [Google Scholar] [CrossRef] [PubMed]
- International Energy Agency. Putting CO2 to use. Energy Rep. 2019. Available online: https://www.oecd-ilibrary.org/energy/putting-co2-to-use_dfeabbf4-en (accessed on 1 September 2021).
- Zimmermann, A.W.; Wunderlich, J.; Müller, L.; Buchner, G.A.; Marxen, A.; Michailos, S.; Armstrong, K.; Naims, H.; Mccord, S.; Styring, P.; et al. Techno-economic assessment guidelines for CO2 utilisation. Front. Energy Res. 2020, 8, 1–23. [Google Scholar] [CrossRef] [Green Version]
- IEA. Exploring Clean Energy Pathways: The Role of CO2 Storage; IEA: Paris, France, 2019. [Google Scholar] [CrossRef]
- Ghiat, I.; Al-Ansari, T. A review of carbon capture and utilisation as a CO2 abatement opportunity within the EWF nexus. J. CO2 Util. 2021, 45, 101432. [Google Scholar] [CrossRef]
- Meunier, N.; Chauvy, R.; Mouhoubi, S.; Thomas, D.; De Weireld, G. Alternative production of methanol from industrial CO2. Renew. Energy 2020, 146, 1192–1203. [Google Scholar] [CrossRef]
- Song, C. CO2 Conversion and Utilisation: An Overview. ACS Symp. Ser. 2016. Available online: https://pubs.acs.org/doi/pdf/10.1021/bk-2002-0809.ch001 (accessed on 1 September 2021).
- Gulzar, A.; Ansari, M.B.; He, F.; Gai, S.; Yang, P. Carbon dioxide utilisation: A paradigm shift with CO2 economy. Chem. Eng. J. Adv. 2020, 3, 100013. [Google Scholar] [CrossRef]
- Esposito, E.; Dellamuzia, L.; Moretti, U.; Fuoco, A.; Giorno, L.; Jansen, J.C. Simultaneous production of biomethane and food grade CO2 from biogas: An industrial case study. Energy Environ. Sci. 2018, 12, 281–289. [Google Scholar] [CrossRef]
- Kamkeng, A.D.N.; Wang, M.; Hu, J.; Du, W.; Qian, F. Transformation technologies for CO2 utilisation: Current status, challenges and future prospects. Chem. Eng. J. 2021, 409, 128138. [Google Scholar] [CrossRef]
- Alper, E.; Orhan, O.Y. CO2 utilisation: Developments in conversion processes. Petroleum 2017, 3, 109–126. [Google Scholar] [CrossRef]
- Long, D.J.; Tang, L. The impact of socio-economic institutional change on agricultural carbon dioxide emission reduction in China. PLoS ONE 2021, 16, e0251816. [Google Scholar] [CrossRef]
- Caldeira, K.; Brown, P.T. Reduced emissions through climate damage to the economy. Proc. Natl. Acad. Sci. USA 2018, 116, 714–716. [Google Scholar] [CrossRef] [Green Version]
- Righetti, T.; Richardson, J.; Koski, K.; Taylor, S. The Carbon Storage Future of Public Lands. Pace Environ. Law Rev. 2020. Available online: https://digitalcommons.pace.edu/cgi/viewcontent.cgi?article=1847&context=pelr (accessed on 1 September 2021).
- Otto, I.M.; Donges, J.F.; Cremades, R.; Bhowmik, A.; Hewitt, R.; Lucht, W.; Rockström, J.; Allerberger, F.; McCaffrey, M.; Doe, S.S.P.; et al. Social tipping dynamics for stabilising Earth’s climate by 2050. Proc. Natl. Acad. Sci. USA 2020, 117, 2354–2365. [Google Scholar] [CrossRef] [Green Version]
- Mitchell-Larson, E.; Heidug, W. Achieving Net-Zero in the G20: A Novel Supply-Side Climate Policy to Value Carbon Sinks; Climate Change, Sustainable Energy and Environment: Berlin, Germany, 2020; Available online: https://www.g20-insights.org/wp-content/uploads/2020/11/achieving-net-zero-in-the-g20-a-novel-supply-side-climate-policy-to-value-carbon-sinks-1606065435.pdf (accessed on 1 September 2021).
- Tapia, J.F.D.; Lee, J.Y.; Ooi, R.E.; Foo, D.C.; Tan, R.R. Review article A review of optimisation and decision-making models for the planning of CO2 capture, utilisation and storage (CCUS) systems. Sustain. Prod. Consum. 2017, 13, 1–15. [Google Scholar]
- Popa, A. Why capturing and using or storing carbon dioxide is vital for the world’s energy future. Proc. Inst. Civ. Eng. Civ. Eng. 2021, 174, 8. [Google Scholar]
- Tsiropuolus, I.; Nijs, W.; Tarvydas, D.; Ruiz Castello, P. Towards Net-Zero Emissions in the EU Energy System by 2050; Technical Report EUR 29981 EN; Publications Office of the European Union: Luxembourg, 2020. [Google Scholar] [CrossRef]
- Bhagat, C.; Dudhagara, P.; Tank, S. Trends, application and future prospectives of microbial carbonic anhydrase mediated carbonation process for CCUS. J. Appl. Microbiol. 2017, 124, 316–335. [Google Scholar] [CrossRef] [Green Version]
- Chowdhury, M.S.; Rahman, K.S.; Chowdhury, T.; Nuthammachot, N.; Techato, K.; Akhtaruzzaman, M.; Tiong, S.K.; Sopian, K.; Amin, N. An overview of solar photovoltaic panels’ end-of-life material recycling. Energy Strateg. Rev. 2020, 27, 100431. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).