1. Introduction
Phthalate acid esters are micro-pollutants of great public concern due to their negative impact on the ecosystem and human health. Phthalates are dialkyl or alkyl aryl esters of phthalic acid, which are primarily synthesized from phthalic anhydride and the corresponding alcohol by the Fischer esterification in the presence of a catalyst.
Phthalate acid esters (PAEs) are used to increase a material’s flexibility, transparency, pliability, plasticity, strength, endurance and longevity [
1,
2,
3]. They are important plasticizers and additives, which, depending on the alcohol that makes up the alkyl chain, are widely used in industrial production in numerous plastic applications due to their high performance and low cost [
4]. PAEs can account for up to 40% of the final plastic product [
5]. Long-chain PAEs (di(2-ethylhexyl) phthalate (DEHP), diisononyl phthalate (DiNP), diisodecyl phthalate (DiDP)) are primarily used in plastic polymers and applications such as building and construction materials (wires and cables, electrical cords, flooring, roofing and cladding, wall coverings and floor tiles, films and sheets, paints and lacquers) and adhesives. Phthalates are also included in apparel, furniture (PVC flooring), car and public transport interiors, cosmetics, medical devices, toys and baby care products, food and packaging materials such as drinking straws, and food containers [
6,
7,
8,
9,
10,
11]. However, short-chain PAEs (dimethyl phthalate (DMP), diethyl phthalate (DEP), butyl benzyl phthalate (BBzP), di-n-butyl phthalate (DnBP), diisobutyl phthalate (DiBP)) are often used in non-PVC applications such as personal care products, adjuvants in pesticide formulations for usage in aquatic systems, paints, glue, lubricants and plastic bags [
7,
9,
12,
13,
14,
15,
16].
PAEs are not chemically bound to the host polymer and exposed to a higher temperature can easily migrate in the contact matrix, which may lead to the release from the life cycle of commercial and domestic products into the environment—air, water, soil and biota [
4,
10,
17,
18,
19,
20,
21,
22,
23,
24,
25,
26,
27].
PAEs eventually can enter the environment through multiple pathways among which are industrial and municipal wastewaters, land application of sewage sludge and leaching after the disposal of industrial and municipal solid waste [
9,
28,
29,
30,
31].
Hereby, phthalate acid esters can enter the water ecosystem through wastewater treatment plants (WWTP). Because of the high lipophilicity of PAEs, they tend to be adsorbed and concentrated to suspended organic matter and sludge and are removed through biodegradation [
3,
29,
31,
32,
33]. In addition, the use of fertilizers, pesticides and irrigation with polluted water in agriculture increase the diffusion of phthalate acid esters in the environment as well as its accumulation in plant biomass and introduction in the food chain [
34,
35,
36].
Moreover, PAEs are not persistent and readily biodegrade in the aquatic environment under aerobic and anaerobic conditions [
36,
37]. Higher molecular weight PAEs biodegrade more slowly than the lower molecular weight chemicals and may accumulate in the sediments [
3,
32,
38].
PAEs are classified as carcinogenic, toxic and endocrine-disrupting compounds (EDCs) that cause adverse effects on the developmental stage of organisms [
9,
39,
40,
41,
42,
43,
44,
45,
46,
47,
48].
Previous investigations have shown that PAEs have potential risk to human health and can cause reproductive problems, birth defects, children growth-disrupting effects, decreased testosterone levels, thyroid-disrupting effects, hormonal and endocrine disruptions, respiratory allergic symptoms, asthma and can even cause breast cancer [
2,
49,
50,
51,
52,
53,
54,
55]. Moreover, exposure to phthalate acid esters can adversely affect various human organs, such as the respiratory system, kidneys, liver as well as the endocrine system [
56].
Consequently, six PAEs (DMP, DEP, DnBP, BBzP, DEHP, di-n-octyl phthalate (DnOP)) have been included in the priority list of pollutants by the European Union and the United States Environmental Protection Agency [
41].
Effects of climate change as well as overuse and contamination put water resources under substantial pressure. The EU has some restrictions on the use of phthalates in materials and products. Therefore, industries should be prepared for and respond to the life cycle of their products. However, in Eastern European countries, many organizers of economic activity do not know what types of phthalates are released into the water and what their further qualitative and quantitative characteristics are. Taking into account the widespread use of PAEs and the evidence supported by scientific studies described in the literature, a research hypothesis was proposed. The hypothesis of this survey was that the primary level of pollution of wastewaters and their sludge by phthalates depends on domestic and industrial sources of pollution, and the higher the pollution, the more objects of economic activity are served. To clarify economic activity interactions with water pollution by PAEs in order to define their environmental impacts, the levels of DMP, DEP, DPP, DBP, DiBP, DCHP and DEHP in wastewater and sludge samples collected from domestic and industrial users were determined in 2019–2020.
Furthermore, in this paper, we present a comparative example of sewage pollution by microplastics in some Eastern European countries and Denmark that reflected the growing awareness of the concern of wastewater monitoring and European legal requirement implementation regarding the manufacture, marketing and use of PAE compounds.
2. Materials and Methods
In 2019–2020 a total of 50 water and 8 sludge samples were collected from 50 locations in Lithuania, Poland and Denmark. Experimental data were expressed as the mean of 3 results. Details of each sampling site are included in
Table S1, and sampling location maps are presented in
Figures S1 and S2.
The Standard EN ISO 18856:2005 and ISO 18856:2004 (Water quality—Determination of selected phthalates using gas chromatography/mass spectrometry) specify a method for the determination of phthalates (
Table 1) in water after solid-phase extraction and gas chromatography/mass spectrometry.
This method could be used for the determination of phthalates in groundwater, surface water, wastewater and drinking water.
The principle of the water sample analysis method is solid-phase extraction of the compounds from the water. Then the separation is accomplished using capillary columns by the gas chromatography and followed by identification and quantification of the phthalates by mass spectrometry.
However, CEN/TS 16183:2012 standard “Sludge, treated biowaste and soil-determination of selected phthalates using capillary gas chromatography with mass spectrometric detection (GC-MS)” describes a method for the determination of phthalates in sludge, treated biowaste and soil, after extraction and gas chromatographic analysis with mass spectrometric detection (
Table 1).
The principle of the sludge sample analysis method: the dried sample is extracted with ethyl acetate on the shaking device. An aliquot of the extract is cleaned with aluminum oxide (if necessary) followed by gas chromatographic separation using capillary columns and identification and quantification of the phthalates by mass spectrometry.
One-liter amber glass bottles with Teflon caps were used for sample collection to avoid exposure to light and contamination of plastic. Before taking the samples, bottles were washed with distilled water and acetone. Collected samples were extracted within 24–48 h with preservation at 4 °C [
3]. Samples were taken from the sewer hatches where the mix of pipes from various activities was coming. Further, sludge samples were collected using a grab sampler and stored in special 0.5 L glass bottles at −20 °C. Sludge sample points:
- (1)
Primary sludge—at the pumping station of the sludge gravity thickener;
- (2)
Excess sludge—from the pipeline before secondary thickening;
- (3)
Digestate—from the bottom of the sludge digester (biogas reactor);
- (4)
Dried sludge—from the bottom of the dried sludge silage/bunker.
All experimental materials were made of borosilicate glass and were used after a series of cleaning processes. They were kept in a 10% hydrochloric acid bath for 24 h, rinsed with ultrapure water and finally rinsed with methanol and dichloromethane just before use.
The phthalate standards for DMP (99%), DEP (99%), DPP (99%), DBP (99%), DiBP (99%), DCHP (99%) and DEHP (98%) were purchased from Dr. Ehrenstorfer GmbH (Germany). Organic solvents such as acetonitrile and n-hexane were of HPLC-grade and obtained from Sigma-Aldrich (USA).
The analysis was performed using a Bruker SCION 436 gas chromatograph equipped with a SCION TQ/SQ mass spectrometer detector. Chromatographic separation was achieved using a SCION-5MS capillary column (30 m, 0.25 mm i.d., 0.25 μm film thickness) with selected ion monitoring mode for quantitative determination. The flow rate of helium (a carrier gas, purity ≥ 99.999%) was 1.0 mL min−1. The temperature of the GC-MS was as follows: initial 80 °C, which was ramped to 180 °C at 20 °C min−1, 230 °C at 5 °C min−1 and 289 °C at 20 °C min−1, with an isotherm hold of 5 min. Samples of 1 μL were injected in an automatic split-less injection mode at 289 °C. Selected ion monitoring (SIM) data were acquired during the GC full scan electron MS analysis.
With every analysis regime, laboratory contaminations were adjusted by analysis of blank samples.
3. Results and Discussion
The concentrations of the DMP, DEP, DPP, DBP, DiBP, DCHP and DEHP in wastewater and sludge samples from Lithuania (Klaipeda), Poland (Goleniów) and Denmark (Rønne) sewer systems were analyzed in 2019–2020. WWTPs in Klaipeda, Goleniów and Rønne have similar mechanical and biological types of water cleaning. In order to find the common tendency of the level of the specific PAEs in sewage systems, the different pollution level sources and their cleaning at WWTPs with varied capacities were assessed. Klaipeda WWTP has the biggest capacity and serves domestic and industrial companies’ sewage, while Goleniów and Rønne treat mostly domestic sewage and just a few industrial companies’ effluents. In its turn, Rønne WWTP is similar to Goleniów but situated on the island and therefore has a direct impact on the surrounding hydro system. Such location demands specific attention to the quality of wastewater and sludge because, in the end, the ecology of the island depends on the state of the water ecosystem.
3.1. The Levels of PAEs in Wastewater and Sludge Samples Collected from Domestic and Industrial Users in Lithuania (Klaipeda).
All wastewater and sludge samples in Lithuania were taken from the places fitting to the unified Klaipeda sewage system. The Klaipeda WWTP is the main biological sewage treatment plant in Klaipeda, built in 1998, with a capacity of 305,333 population equivalent (PE). Wastewater and sludge quality reflect that it is household and industrial wastewater from cigarette production, paper mill, wood timber, wood processing, furniture as well as food production and service companies.
The highest contamination with phthalates in Lithuania (Klaipeda) was attributed to DEHP since it has the largest meaning in industrial production and consumption. Its concentration ranged from 0.05 to 159 µg/L at the water sampling points (
Table 2). Due to the low water solubility and long side chain, DEHP is stable and difficult to be biodegraded [
57,
58].
The highest DEHP concentration of 159 µg/L and 112 µg/L was observed at the truck wash and laundry sampling points, respectively. The concentrations of other PAEs at these sampling points were also high. At the truck wash water sampling point, the concentration of DiBP reached 2.2 µg/L, DBP 1.8 µg/L, DEP 0.26 µg/L and DMP 0.18 µg/L. However, at the laundry sampling point, DBP concentration reached 6.9 µg/L, DEP 4.8 µg/L, DiBP 3.3 µg/L and DMP 0.14 µg/L. PAE compounds are included in lubricants that influence the truck wash sewage’s pollution. Phthalates were detected in the content of detergents that explain the laundry’s wastewater pollution by PAEs. Moreover, according to the Environmental Working Group (EWG), fragrances are considered to be among the top five allergens and can trigger asthma attacks. Testing by the EWG also revealed that 75% of the fragrances contain phthalates [
59].
Meanwhile, near the shopping center II entity (located near several car washing and repair shops and catering companies), the concentration of DEHP was also very high—50 µg/L. Moreover, the concentration of the DEP at this sampling point reached 78.0 µg/L, DiBP 4.9 µg/L, DBP 2.6 µg/L and DMP 0.17 µg/L. Since DEHP makes up most of the production of PAEs such as plasticizers for PVC and is present in food packaging materials, kitchen utensils, gloves and cleaning products, it is easily washed off to the sewage system [
60]. Phthalates can be easily released into the environment through production, use and utilization because the chemical bond between them and main plastics is non-covalent [
61].
The results show that the concentration of DEHP at the domestic sewage sampling point reached 40 µg/L and was several times higher than at some industrial sampling points such as the bread factory (26 µg/L), furniture production (19 µg/L), wood processing factory (14 µg/L), biodiesel production plant (6.8 µg/L), tobacco factory (2.1 µg/L), food factory (1.2 µg/L), etc. In accordance with the obtained results, the DEHP concentrations in other industrial and mixed sewage pumping stations ranged from 0.7 µg/L (No. 23) to 9.7 µg/L (No. 20). Although the number of employees at each of the above-mentioned enterprises is not small (average 500 employees), the biggest effect on the water pollution by phthalates was detected by household activities provided by citizens and based on phthalates active usage as part or substitute of PVC and other resins products. This predominant effect of phthalates in domestic effluents in comparison to industrial ones can be elucidated by some industrial sector similarity to household activities, especially such as various services, catering, food, furniture and textile treatment [
62].
Finally, the concentrations of DEHP in water samples of Klaipeda and nearby city district WWTP outlets ranged from 0.07 to 0.14 µg/L. The wastewater treatment plant ensures effective phthalate precipitation at least two times per wastewater cleaning process and phthalate concentrate in the sedimentation.
It was determined that DEHP concentration in the water near the shopping center I sampling point (samples were taken from the sewer hatches where the mix of pipes from various center activities was coming) reached 14 µg/L as well as DiBP 26 µg/L, DEP 17.0 µg/L and DBP 6.1 µg/L. It can be explained by the fact that cars’ washing as well as filling stations are situated close to the shopping center. Active catering services and products’ plastic packaging also contribute to the total PAE compounds’ concentration in the sewage.
DEHP concentration was also measured in the regional dump of the Klaipeda region landfill. It was found that DEHP concentration in the water sample was up to 7.2 µg/L as well as concentrations of other PAEs ranged from less than 0.05 µg/L to 0.78 µg/L (DBP). Based on the statistics [
63], the amount of plastic waste has a tendency to increase in the world (380 million tons in 2015), and taking into account the low DEHP biodegradability in the water, the DEHP concentration is higher than other measured phthalates [
64].
Furthermore, the diversity of the DEP concentrations at industrial sampling points varied from 0.23 µg/L at the wood processing factory to 5.5 µg/L at the bread factory. Diethyl phthalate, as a low molecular weight PAE, can be found in industrial solvents, solvents in perfumes, pharmaceutical products, insecticide materials, adhesives, waxes, inks and cosmetics [
65]. DEP is quite volatile but very slowly evaporates from its aqueous solution [
66] and is expected to be degraded by microorganisms of biological WWTP.
The distribution of PAEs in the water samples of Klaipeda WWTP (outlet) was as follows: DEHP > DEP > DiBP > DBP > DMP. DPP and DCHP concentrations were less than 0.05 μg/L.
In this study, the distribution of total PAEs in the sludge samples was as follows: digestate sludge > primary sludge > dried sludge > excess sludge (
Table 3). The highest concentrations of these samples were related to DEHP, where concentrations ranged from digestate sludge (95.0 mg/kg) to excess sludge (3.8 mg/kg). After sludge biodegradation in the biogas reactor, the sludge volume is decreased at least by one-third of the initial one, and the phthalates’ concentration becomes higher in digestate sludge. The analysis of the sludge contamination by PAEs after the drying process showed a significant reduction in the DEHP. The most likely mechanism to explain the loss of DEHP in the sludge during the drying process is volatilization [
67,
68]. It is reported that the rates of the DEP and DEHP degradation in the sediments are very low and depend on pH, temperature, surfactants, pollutants and microbial inhibitors [
41].
It was found that DPP and DCHP phthalate concentrations in all water and sludge (except for the Klaipeda WWTP primary sludge sample with a slightly higher concentration of 0.052 mg/kg) samples were less than 0.05 mg/kg.
3.2. The Levels of PAEs in Wastewater Samples Collected from Domestic and Industrial Users in Poland (Goleniów).
Goleniów WWTP is the biological WWTP situated in Goleniów city, West Pomeranian District, North-western Poland, built in 2004 and rebuilt in 2011, with the current capacity of 63,805 PE. Wastewater and sludge quality reflects that it is mostly household wastewater with some industrial sewage from the sectors of plastics production, meat processing, car washes and sewage delivered from septic tanks.
The highest contamination with phthalates was observed in sewage pumping station No. 6, where sewage from a supermarket and industrial area is collected before transferring it to the sewage treatment plant (
Table 4). The concentration of DMP reached 210 µg/L. Moreover, the concentrations of DEP as well as DEHP in this sampling point were high as well and reached 15 µg/L and 1.8 µg/L, respectively. The well-known method for phthalate removal from water is a biological treatment based on PAE aerobic and/or anaerobic degradation by microorganisms [
69]. The obtained PAE concentrations showed that long-chain alkyl is quite resistible and requires effective biological treatment.
However, it was determined that the total concentrations of seven PAEs in the sewage No. 1 of the industrial park was much lower. It could be explained by a variety of a smaller number of companies and their size. The DEHP (2.6 µg/L), DEP (2.2 µg/L) and DMP (1.1 µg/L) were the main phthalate compounds at this sampling point.
The results of the manhole observation No. 3, No. 4, No. 5 and No. 7 points show that the highest concentration had DEHP of up to 15 µg/L and DMP of up to 7.7 µg/L. The main source of the high concentrations of PAEs at these observation points was fast-food restaurants, hairdresser, beautician, shopping center, gas and train stations.
Wastewater treatment plants have an important role in the reduction of the risk of micropollutants, such as phthalates [
70]. The dominant PAEs in the inlet of the sewage treatment plant No. 9 were DEHP > DEP > DiBP > DBP. DMP, DPP and DCHP concentrations were less than 0.05 μg/L.
It is important to state that the concentration of DPP and DCHP at all sampling points was less than 0.05 µg/L, which is in accordance with the proposition of the other scientists [
71] and based on a small industrial/domestic sewage proportion at the wastewater treatment plant.
The distribution of PAEs in the water samples from Goleniów was as follows: DMP > DEHP > DEP > DBP and DiBP. DPP and DCHP concentrations were less than 0.05 μg/L.
3.3. The Levels of PAEs in Wastewater and Sludge Samples Collected from Domestic and Industrial Users in Denmark (Rønne)
Rønne WWTP is the largest of the seven biological WWTPs in Denmark, built in 1995, with a capacity of 60,000 PE. Wastewater and sludge quality reflects that it is mostly household wastewater without much industry, except the slaughterhouse, the hospital and a few metal processing industries in Rønne.
Sewage samples were taken at the inlet and outlet of sampling stations during the summer of 2020 in order to analyze the efficiency of the WWTP regarding phthalate reduction based on biotechnological sewage treatment.
The results of Rønne’s wastewater pollution by phthalates are presented in
Table 5. The analysis of the data showed the main trends of the PAE compound amounts’ distribution in the samples. The predominant quantity of the DEHP and following DEP concentrations are in the same proportion as in above-analyzed Lithuanian and Polish WWTPs and reflect the common tendency of sewage pollution for small and middle-sized water treatments. The positive alteration of the total amount of PAEs after sewage treatment at Rønne WWTP does not connect with special or additional sewage handling. The reduction of the total phthalate concentration at the wastewater outlet point supports the statement that the biological water treatment and its microorganisms have the main role in phthalate concentrations’ decline [
70]. These results confirm the hypothesis that sustainable maintenance of biological wastewater treatment is an essential factor of micro-organic pollutant diminishing under present conditions.
The distribution of PAEs in the water samples of Rønne WWTP was as follows: DEHP > DEP > DBP > DiBP. DMP, DPP and DCHP concentrations were less than 0.05 μg/L. A similar trend has been reported in the review by Chinese scientists [
58].
It is well-known that wastewater pollution can be judged by the quality of the sludge. Such an organic pollutant as DEHP is an oily organic carcinogen, can slightly (0.27 mg/L at 25 °C) dissolve in water [
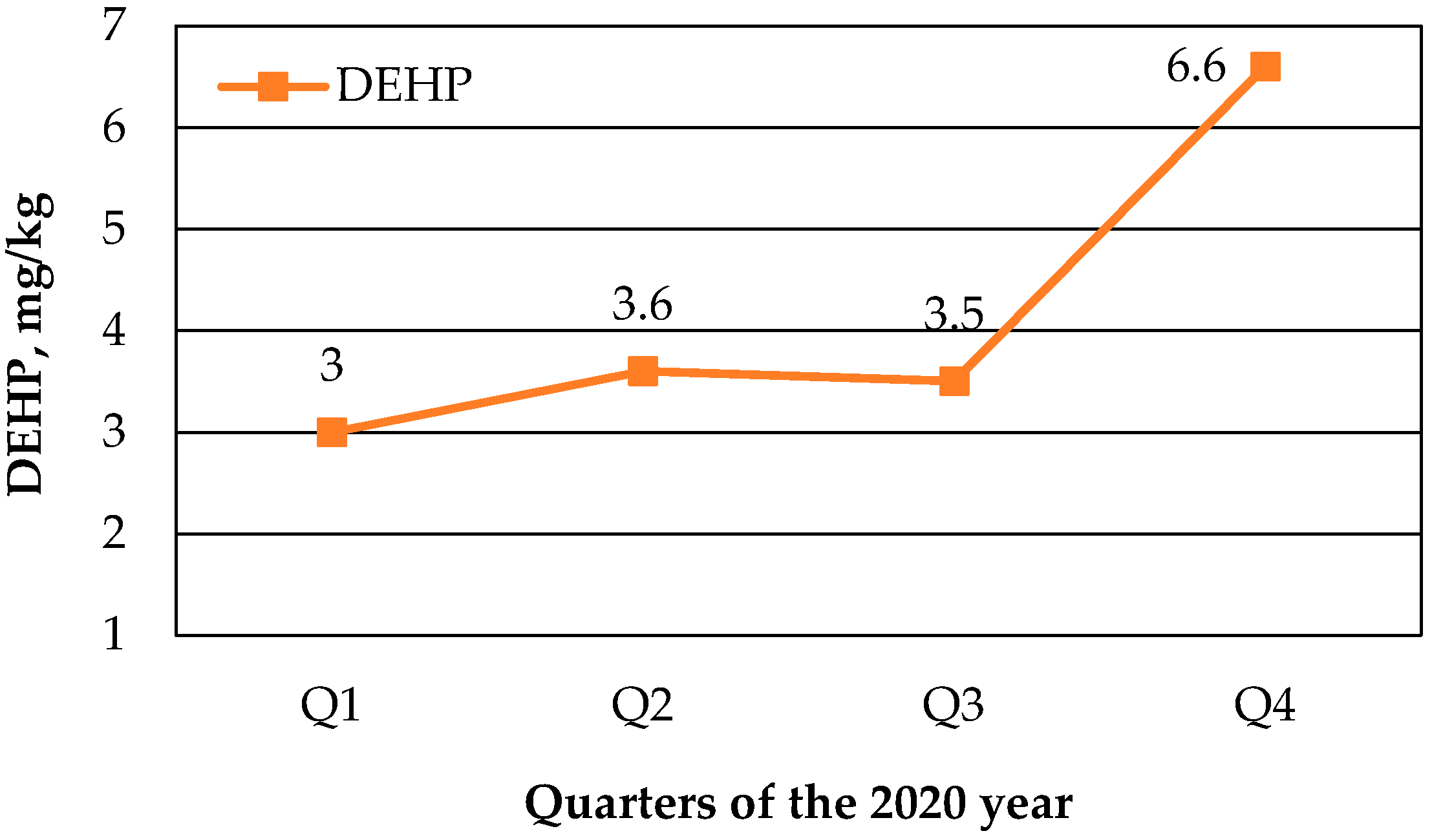
72] and has the inclination to bind to the sludge. The main samples for the determination of the Rønne wastewater quality came from the inlet and outlet of the Rønne sewage system. Besides that, the results of the samples of sludge, which were collected on-site for DEHP determination every month then mixed and analyzed every three months, were taken into account for correct evaluation of the sludge pollution by DEHP at Rønne WWTP in 2019 (
Figure 1).
The DEHP concentration increased twice during the year. The analysis of the data presented in
Figure 1 showed that the amount of phthalates in the sludge has a tendency to change during the year, but there is no seasonal relationship [
73]. The main reasons for an increase in the concentration of DEHP in sludge depend on the quality of wastewater, especially its industrial component, the type of treatment facilities and their efficiency.
3.4. Obtained Data and Conditions Comparison
The study results show that three various capacity wastewater systems, situated in three different places have common trends in the wastewater and sludge pollution by PAEs.
The most evident finding is related to the hypothesis that the level of sewage contamination depends on domestic and industrial pollution sources and is higher as more economic activity objects are served by WWTP. For instance, the bigger-capacity Klaipeda WWTP should clean the sewage of the objects whose DEHP and DEP concentrations in the sewage reached, respectively, 112 and 78 μg/L and depend on a big shopping center and combined household services. The contribution of the specific industry of biodiesel production is reflected in DBP concentration (34 μg/L). A similar pollution tendency was observed in the Goleniów sewage system. A bigger amount of DEHP and DEP are also coming with shopping centers and household services objects’ sewage (15 μg/L). The samples’ analysis done at Rønne WWTP showed that among PAE compounds, DEHP concentration in the inlet is the highest (1.2 μg/L).
Therefore, the next important output is connected to the determination of the DEHP as the predominant phthalate compound in the sewage. In Klaipeda as well as Rønne sewage, the DEHP concentrations are the highest. However, in Goleniów, the biggest amount of PAEs belongs to DMP (210 μg/L). It falls short against Klaipeda and Rønne data as well as Western Europe and China scientists’ reports [
58]. Due to the fact that, in water, the DMP loss mechanism is related to biodegradation [
3,
29,
31,
32,
33] and anaerobic condition, it could be suggested that the sampling condition or sewage biological treatment were temporarily damaged. The low DMP concentration at the Goleniów outlet point supports our assumption and confirms that the DEHP amount in the sewage is a broad issue.
It was found that although the DEHP concentration in the wastewater from different sources is high, it is still possible to significantly reduce pollution by PAEs using biological water treatment [
62,
64]. The concentration of DEHP at the outlet of Klaipeda and Rønne wastewater treatment plants is 0.07 μg/L, which is less than the Lithuanian limit for treated wastewater entering surface waters (<2 μg/L). The DEHP concentration in the outlet point of Goleniów WWTP was 8.1 μg/L, but officially there is no limit for DEHP concentration for the treated water in Poland. The evaluation of the wastewater sludge’s contamination by PAEs was available at Klaipeda and Rønne WWTPs. In both cases, sludge pollution by DMP, DEP, DPP, DBP, DiBP, DCHP and DEHP were investigated and showed the biggest pollution by DEHP. The biggest concentration of the DEHP in Rønne’s dewatered sludge was 6.6 mg/kg. Taking into consideration that Klaipeda WWTP technology includes excess sludge use for biogas production and digestate drying for further utilization, PAE pollution analysis was performed for four types of sludge. A significant amount of DEHP was found in digestate (95 mg/kg), but due to the drying process, DEHP concentration decreased six times to 16 mg/kg [
67,
68]. The comparison of the sludge pollution level in Klaipeda and Rønne was demonstrated to be effective Danish sludge quality management for its lasting utilization. Furthermore, the smallest, among three target systems, amount of the DEHP in the sewage and its sludge in Rønne demanded sustainable environmental management decisions during a long period of time in accordance with the Danish review regarding sludge operation in 1993–2003, and the amount of sludge production had a tendency to reduce [
74]. Regarding the DEHP amount in the sludge, the cut-off value for this compound (50 mg/kg d.w.) caused an important decrease of this contaminant. As a result, in 2003, more than 95% of sludge had a phthalate concentration below the cut-off criteria and was suitable for agricultural purposes [
75].
Figure 1 data also confirm the positive trend of the DEHP concentration diminishing at the Rønne WWTP sludge from 2016 to 2020. Perhaps, such result was a consequence of the active Danish Environmental Protection Agency (EPA) policy and attention to pollution sources identification as well as publishing a list of undesirable substances and other numerous actions, such as a voluntary agreement with manufacturers for the environmentally friendly alteration in products’ production and use. Total/partial prohibition of PAEs, stricter standards, emission controls, environmental labeling, green guidelines and information campaigns, as well as the guideline for manufacturers and taxation, were the main actions, which helped to reduce the PAEs in the environment [
76].
Although Denmark, Lithuania and Poland follow the same EU regulations on phthalates, the measurement results indicated differences in phthalate amounts in the sewage and sludge of Eastern countries and Denmark. Furthermore, in Denmark, there is a special regulation about toys, which, since 1999, has banned the manufacturing, import and selling of toys and certain articles for young children with phthalates, especially for children under three years of age.
From 16 January 2007, the manufacture and import of toys and articles for children up to the age of 14 with the phthalates DEHP, DBP and BBP have been forbidden as well. In the case of a toy or a toddler article that can be put in the mouth, the ban also applies to the phthalates DINP, DIDP and DNOP. As of 16 January 2007, the sale of these products has also been banned.
Denmark successfully adopted a directive regarding harmful substance monitoring implementation on the state level (priority substances in the field of water policy). Lithuania is still under preparation for the directive’s implementation. Each middle and big-size economic unit should organize air and water pollution control, inform regional environmental protection agency on PAE compounds and their concentrations. Monitoring of the entity is carried out in Poland as well.
The recent European document, which could affect the wastewater and sludge quality, is the REACH-ECHA document: “Restrictions on the manufacture, placing on the market and use of certain dangerous substances, mixtures and articles” (XVII annex of REACH-ECHA) [
77]. It is expected that this restriction will help to reduce such PAEs as DEHP, BBP, DBP and DIBP in consumer products in the EU market and respectively diminish the air, water, soil and sediment contamination by target organic pollutants.
4. Conclusions
The current study revealed the emergence of the phthalate acid esters in the aquatic environment originating due to extensive plastics’ commercial and domestic usage.
The conducted research disclosed as well, the influence of economic activity on the pollution of the sewer system by PAEs.
It was found that the biggest PAE sources were concentrated in the vicinity of industrial parks, residential areas and roadside drains at some instances with emerging concentrations above the maximum permissible concentrations.
It was found that there is not a lot of research based on PAE analysis in Eastern countries’ sewer systems while Western Europe, USA and China sewage systems are well investigated. This comparative analysis of the level of wastewater and sludge pollution by DMP, DEP, DPP, DBP, DiBP, DCHP and DEHP of sewage systems situated in Eastern countries (Lithuania, Poland) and Denmark is a major contribution to knowledge in this field.
It was determined that the DEHP is the predominant PAE in Eastern countries and Denmark’s sewage systems as well as in Western Europe countries due to its widespread utilization and low solubility in water.
It was assessed that the Danish and the Lithuanian biological wastewater treatment facilities were more effective for lowering the concentration of DEHP and other analyzed PAEs in the outlet comparing to facilities in Poland.
It was found that during the phase of sludge drying, an effective reduction of DEHP concentration is achieved, based on the organic compound volatilization process.
It was disclosed that the investigated Danish wastewater and sludge were less polluted by DEHP and other analyzed PAEs in comparison to the Lithuanian or Polish wastewater and sludge.
It was displayed, that an active Danish Environmental Protection Agency policy and strategy grounded on numerous legal, educational and managerial actions boosts a new behavior in PAE use and disposal.
The research findings may provide reliable data for decision-makers regarding a phthalate reduction strategy’s development in Lithuania, Poland and other countries based on a precedent of successful PAE pollution prevention policy in Denmark.
Considering the risk of phthalates’ presence in the environment, more studies are needed to determine PAE; contamination dynamics in Eastern countries.
Supplementary Materials
The following are available online at
https://www.mdpi.com/2071-1050/13/2/529/s1, Figure S1: Map of the location of the sampling sites in Poland, Figure S2: Map of the location of the sampling sites in Lithuania, Table S1: Sampling site description.
Author Contributions
Conceptualization, O.A. and T.P.; methodology, O.A.; software, O.A. and T.P.; validation, O.A.; formal analysis, O.A. and T.P.; investigation, O.A. and T.P.; resources, O.A. and T.P.; data curation, O.A.; writing—original draft preparation, O.A. and T.P.; writing-review and editing, O.A. and T.P.; visualization, O.A. and T.P.; supervision, O.A.; project administration, O.A.; funding acquisition, O.A. All authors have read and agreed to the published version of the manuscript.
Funding
This research was supported by the STEP (Sludge Technological Ecological Progress—increasing the quality and reuse of sewage sludge) Interreg-V-A project, grant No. STEP-STHB.02.02.00-32-0110/17-00 of the EU South Baltic Programme 2014-2020.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
All data generated or analysed during this study are included in this published article.
Acknowledgments
The authors would like to express their gratitude to Dariusz Kozak (Technical Director) and Katarzyna Synoradzka (Environmental Protection Specialist) of “Goleniowskie Wodociągi i Kanalizacja Sp. z.o.o.”, Torben Jørgensen (Project Manager of Bornholms Energy & Supply), Kristina Bereisiene (Head of Wastewater Treatment Service) and Nijole Domarkiene (Deputy Head of Laboratory) of JSC “Klaipedos vanduo” for their valuable contribution to data compilation and analysis.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Kolarik, B.; Bornehag, C.G.; Naydenov, K.; Sundell, J.; Stavova, P.; Nielsen, O.F. The concentrations of phthalates in settled dust in Bulgarian homes in relation to building characteristic and cleaning habits in the family. Atmos. Environ. 2008, 42, 8553–8559. [Google Scholar] [CrossRef]
- Serrano, S.E.; Braun, J.; Trasande, L.; Dills, R.; Sathyanarayana, S. Phthalates and diet: A review of the food monitoring and epidemiology data. Environ. Health 2014, 13, 43. [Google Scholar] [CrossRef]
- Gao, D.; Li, Z.; Wen, Z.; Ren, N. Occurrence and fate of phthalate esters in full-scale domestic wastewater treatment plants and their impact on receiving waters along the Songhua River in China. Chemosphere 2014, 95, 24–32. [Google Scholar] [CrossRef]
- Li, X.; Liu, W.; Zhang, C.; Song, P.; Wang, J. Fate of Phthalic Acid Esters (PAEs) in Typical Greenhouse Soils of Different Cultivation Ages. Bull. Environ. Contam. Toxicol. 2020, 104, 301–306. [Google Scholar] [CrossRef]
- Wittassek, M.; Koch, H.M. Assessing exposure to phthalates—The human biomonitoring approach. Mol. Nutr. Food Res. 2011, 55, 7–31. [Google Scholar] [CrossRef]
- Ma, W.L.; Subedi, B.; Kannan, K. The occurrence of bisphenol A, phthalates, parabens and other environmental phenolic compounds in house dust: A review. Curr. Org. Chem. 2014, 18, 2182–2199. [Google Scholar] [CrossRef]
- Sopheak, N.; Sempere, R.; Delmont, A.; Paluselli, A.; Ouddane, B. Occurrence, fate, behavior and ecotoxicological state of phthalates in different environmental matrices. Environ. Sci. Technol. Am. Chem. Soc. 2015, 49, 4019–4035. [Google Scholar]
- Herrero, L.; Calvarro, S.; Fernández, M.A.; Quintanilla-López, J.E.; González, M.J.; Gómara, B. Feasibility of ultra-high-performance liquid and gas chromatography coupled to mass spectrometry for accurate determination of primary and secondary phthalate metabolites in urine samples. Anal. Chim. Acta. 2015, 853, 625–636. [Google Scholar] [CrossRef][Green Version]
- Palusellial, A.; Kim, S.K. Horizontal and vertical distribution of phthalates acid ester (PAEs) in seawater and sediment of East China Sea and Korean South Sea: Traces of plastic debris? Mar. Pollut. Bulletin. 2020, 151, 110831. [Google Scholar] [CrossRef]
- Kiralan, M.; Toptanci, I.; Yavuz, M.; Ramadan, M.F. Phthalates levels in cold-pressed oils marketed in Turkey. Environ. Sci. Pollut. Res. 2020, 27, 5630–5635. [Google Scholar] [CrossRef]
- Zhang, W.; Xiaowei, Z.; Peng, G.; Ning, W.; Zini, L.; Jian, H.; Zheng, Z. Distribution and risk assessment of phthalates in water and sediment of the Pearl River Delta. Environ. Sci. Pollut. Res. 2020, 27, 12550–12565. [Google Scholar] [CrossRef]
- Rudel, R.A.; Gray, J.M.; Engel, C.L.; Rawsthorne, T.W.; Dodson, R.E.; Ackerman, J.M.; Rizzo, J.; Nudelman, J.L.; Brody, J.G. Food packaging and bisphenol A and bis(2-ethyhexyl) phthalate exposure: Findings from a dietary intervention. Environ. Health Perspect. 2011, 119, 914–920. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.W.; Chen, C.F.; Dong, C.D. Distribution of Phthalate Esters in Sediments of Kaohsiung Harbor, Taiwan. J. Soil Contam. 2013, 22, 119–131. [Google Scholar] [CrossRef]
- Peng, X.; Feng, L.; Li, X. Pathway of diethyl phthalate photolysis in sea-water determined by gas chromatography–mass spectrometry and compound-specific isotope analysis. Chemosphere 2013, 90, 220–226. [Google Scholar] [CrossRef] [PubMed]
- Braun, J.M.; Sathyanarayana, S.; Hauser, R. Phthalate exposure and children’s health. Curr. Opin. Pediatr. 2013, 25, 247–554. [Google Scholar] [CrossRef] [PubMed]
- Abdolahnejad, A.; Gheisari, L.; Karimi, M.; Norastehfar, N.; Ebrahimpour, K.; Mohammadi, A.; Ghanbari, R.; Ebrahimi, A.; Jafari, N. Monitoring and health risk assessment of phthalate esters in household’s drinking water of Isfahan, Iran. Int. J. Environ. Sci. Technol. 2019, 16, 7409–7416. [Google Scholar] [CrossRef]
- He, H.; Hu, G.J.; Sun, C.; Chen, S.; Yang, M.N.; Li, J.; Zhao, Y.; Wang, H. Trace analysis of persistent toxic substances in the main stream of Jiangsu section of the Yangtze River, China. Environ. Sci. Pollut. Res. Int. 2011, 18, 638–648. [Google Scholar] [CrossRef]
- Zhang, Q.; Lu, X.M.; Zhang, X.L.; Sun, Y.G.; Zhu, D.M.; Wang, B.L.; Zhao, R.Z.; Zhang, Z.D. Levels of phthalate esters in settled house dust from urban dwellings with young children in Nanjing, China. Atmos. Environ. 2013, 69, 258–264. [Google Scholar] [CrossRef]
- Zota, A.R.; Calafat, A.M.; Woodruff, T.J. Temporal trends in phthalate exposures: Findings from the National Health and Nutrition Examination Survey, 2001–2010. Environ. Health Perspect. 2014, 122, 235–241. [Google Scholar] [CrossRef]
- Luo, Y.; Guo, W.; HaoNgo, H.; DucNghiem, L.; IbneyHai, F.; Zhang, J.; Liang, S.; Wang, X.C. A review on the occurrence of micropollutants in the aquatic environment and their fate and removal during wastewater treatment. Sci. Total Environ. 2014, 473, 619–641. [Google Scholar] [CrossRef]
- Moreira, M.A.; André, L.C.; Cardea, Z.L. Analysis of phthalate migration to food simulants in plastic containers during microwave operations. Int. J. Environ. Res. Public Health 2014, 11, 507–526. [Google Scholar] [CrossRef]
- Zhao, H.-M.; Du, H.; Xiang, L.; Chen, Y.-L.; Lu, L.; Li, Y.-W.; Li, H.; Cai, Q.-Y.; Mo, C.-H. Variations in phthalate ester (PAE) accumulation and their formation mechanism in Chinese flowering cabbage (Brassica parachinensis L.) cultivars grown on PAE-contaminated soils. Environ. Pollut. 2015, 206, 95–103. [Google Scholar] [CrossRef]
- Wang, J.; Chen, G.C.; Christie, P.; Zhang, M.Y.; Luo, Y.M.; Teng, Y. Occurrence and risk assessment of phthalate esters (PAEs) in vegetables and soils of suburban plastic film greenhouses. Sci. Total Environ. 2015, 523, 129–137. [Google Scholar] [CrossRef]
- Olujimi, O.O.; Aroyeun, O.A.; Akinhanmi, T.F.; Arowolo, T.A. Occurrence, removal and health risk assessment of phthalate esters in the process streams of two different wastewater treatment plants in Lagos and Ogun States, Nigeria. Environ. Monit. Assess. 2017, 189, 345. [Google Scholar] [CrossRef]
- Lu, H.X.; Mo, C.H.; Zhao, H.M.; Xiang, L.; Katsoyiannis, A.; Li, Y.W.; Cai, Q.Y.; Wong, M.H. Soil contamination and sources of phthalates and its health risk in China: A review. Environ. Res. 2018, 164, 417–429. [Google Scholar] [CrossRef]
- Yu, Q.; Xiong, X.; He, J.; Zuo, Y.; Chen, Y.; Wang, C. Photolysis of bis(2-ethylhexyl) phthalate in aqueous solutions at the presence of natural water photoreactive constituents under simulated sunlight irradiation. Environ. Sci. Pollut. Res. 2019, 26, 26797–26806. [Google Scholar] [CrossRef]
- Chen, H.; Mao, W.; Shen, Y.; Feng, W.; Mao, G.; Zhao, T.; Yang, L.; Yang, L.; Meng, C.; Li, Y.; et al. Distribution, source, and environmental risk assessment of phthalate esters (PAEs) in water, suspended particulate matter, and sediment of a typical Yangtze River Delta City, China. Environ. Sci. Pollut. Res. 2019, 26, 24609–24619. [Google Scholar] [CrossRef]
- Milošević, N.; Milanović, M.; Sudji, J.; Živanović, D.B.; Stojanoski, S.; Vuković, B.; Milić, N.; Stojanoska, M.M. Could phthalates exposure contribute to the development of metabolic syndrome and liver disease in humans? Environ. Sci. Pollut. Res. 2020, 27, 772–784. [Google Scholar] [CrossRef]
- Paluselli, A.; Fauvelle, V.; Schmidt, N.; Galgani, F.; Net, S.; Sempéré, R. Distribution of phthalates in Marseille Bay (NW Mediterranean Sea). Sci. Total Environ. 2018, 621, 578–587. [Google Scholar] [CrossRef]
- Abtahi, M.; Dobaradaran, S.; Torabbeigi, M.; Jorfi, S.; Gholamnia, R.; Koolivand, A.; Darabi, H.; Kavousi, A.; Saeedi, R. Health risk of phthalates in water environment: Occurrence in water resources, bottled water, and tap water, and burden of disease from exposure through drinking water in Tehran, Iran. Environ. Res. 2019, 173, 469–479. [Google Scholar] [CrossRef]
- Gani, K.M.; Kazmi, A.A. Ecotoxicological risk evaluation and regulatory compliance of endocrine disruptor phthalates in a sustainable wastewater treatment scheme. Environ. Sci. Pollut. Res. 2020, 27, 7785–7794. [Google Scholar] [CrossRef] [PubMed]
- Liang, D.W.; Zhang, T.; Fang, H.H.; He, J. Phthalates biodegradation in the environment. Appl. Microbiol. Biotechnol. 2008, 80, 183–198. [Google Scholar] [CrossRef] [PubMed]
- Stasinakis, A.S. Review on the fate of emerging contaminants during sludge anaerobic digestion. Bioresour. Technol. 2012, 121, 432–440. [Google Scholar] [CrossRef] [PubMed]
- Abdel Daiem, M.M.; Rivera-Utrilla, J.; Ocampo-Pérez, R.; Méndez-Díaz, J.D.; Sánchez-Polo, M. Environmental impact of phthalic acid esters and their removal from water and sediments by different technologies—A review. J. Environ. Manag. 2012, 109, 164–178. [Google Scholar] [CrossRef] [PubMed]
- He, L.; Gielen, G.; Bolan, N.S.; Zhang, X.; Qin, H.; Huang, H.; Wang, H. Contamination and remediation of phthalic acid esters in agricultural soils in China: A review. Agron. Sustain. Dev. 2015, 35, 519–534. [Google Scholar] [CrossRef]
- Vannucchi, F.; Francini, A.; Pierattini, E.C.; Raffaelli, A.; Sebastiani, L. Populus alba dioctyl phthalate uptake from contaminated water. Environ. Sci. Pollut. Res. 2019, 26, 25564–25572. [Google Scholar] [CrossRef]
- Bartsch, P.W.; Edwards, T.M.; Brock, J.W. Prevalence of Eight Phthalate Monoesters in Water from the Okavango Delta, Northern Botswana. Bull. Environ. Contam. Toxicol. 2019, 103, 274–279. [Google Scholar] [CrossRef]
- Zhang, S.; Du, T.; Li, M.; Jia, J.; Lu, H.; Lin, X.; Yu, X. Triglyceride glucose-body mass index is effective in identifying nonalcoholic fatty liver disease in non-obese subjects. Medicine 2017, 96, 7041. [Google Scholar] [CrossRef]
- Bergé, A.; Cladière, M.; Gasperi, J.; Coursimault, A.; Tassin, B.; Moilleron, R. Meta-analysis of environmental contamination by phthalates. Environ. Sci. Pollut. Res. 2013, 20, 8057–8076. [Google Scholar] [CrossRef]
- Pant, N.; Kumar, G.; Upadhyay, A.D.; Patel, D.K.; Gupta, Y.K.; Chaturvedi, P.K. Reproductive toxicity of lead, cadmium, and phthalate exposure in men. Environ. Sci. Pollut. Res. 2014, 21, 11066–11074. [Google Scholar] [CrossRef]
- Net, S.; Delmont, A.; Sempéré, R.; Paluselli, A.; Ouddane, B. Reliable quantification of phthalates in environmental matrices (air, water, sludge, sediment and soil): A review. Sci. Total Environ. 2015, 515–516, 162–180. [Google Scholar] [CrossRef] [PubMed]
- Axelsson, J.; Rylander, L.; Rignell-Hydbom, A.; Lindh, C.H.; Jonsson, B.A.G.; Giwercman, A. Prenatal phthalate exposure and reproductive function in young men. Environ. Res. 2015, 138, 264–270. [Google Scholar] [CrossRef] [PubMed]
- Liu, R.; Jian, Z.; Yang, D.; Pu, Y.; Yin, L. Di(2-ethylhexyl) phthalate induced reproductive toxicity involved in DNA damage-dependent oocyte apoptosis using alternative caenorhabditis elegans model. Toxicol. Lett. 2016, 258, S70–S71. [Google Scholar] [CrossRef]
- Sathyanarayana, S.; Grady, R.; Barrett, E.S.; Redmon, B.; Nguyen, R.H.N.; Barthold, J.S.; Bush, N.R.; Swan, S.H. First trimester phthalate exposure and male newborn genital anomalies. Environ. Res. 2016, 151, 777–782. [Google Scholar] [CrossRef]
- Zarean, M.; Keikha, M.; Poursafa, P.; Khalighinejad, P.; Amin, M.; Kelishadi, R. A systematic review on the adverse health effects of di-2ethylhexyl phthalate. Environ. Sci. Pollut. Res. 2016, 23, 24642–24693. [Google Scholar] [CrossRef]
- Wang, G.; Wang, J.; Zhu, L.; Wang, J.; Li, H.; Zhang, Y.; Liu, W.; Gao, J. Oxidative damage and genetic toxicity induced by DBP in earthworms (Eisenia fetida). Arch. Environ. Contam. Toxicol. 2017, 74, 527–538. [Google Scholar] [CrossRef] [PubMed]
- Song, P.; Gao, J.; Li, X.; Zhang, C.; Zhu, L.; Wang, J.; Wang, J. Phthalate induced oxidative stress and DNA damage in earthworms (Eisenia fetida). Environ. Int. 2019, 129, 10–17. [Google Scholar] [CrossRef] [PubMed]
- Wei, L.; Li, Z.; Sun, J.; Zhu, L. Pollution characteristics and health risk assessment of phthalate esters in agricultural soil and vegetables in the Yangtze River Delta of China. Sci. Total Environ. 2020, 137978. [Google Scholar] [CrossRef] [PubMed]
- Casalscasas, C.; Desvergne, B. Endocrine disruptors: From endocrine to metabolic disruption. Annu. Rev. Physiol. 2011, 73, 135–162. [Google Scholar] [CrossRef]
- Foster, P.M.D.; Mylchreest, E.; Gaido, K.W.; Sar, M. Effects of phthalate esters on the developing reproductive tract of male rats. APMIS 2015, 109, S272–S277. [Google Scholar] [CrossRef]
- Katsikantami, I.; Sifakis, S.; Tzatzarakis, M.N.; Vakonaki, E.; Kalantzi, O.I.; Tsatsakis, A.M.; Rizos, A.K. A global assessment of phthalates burden and related links to health effects. Environ. Int. 2016, 97, 212–236. [Google Scholar] [CrossRef] [PubMed]
- Faraji, M.; Taherkhani, A.; Nemati, S.; Mohammadi, A. Challenges in the use of polyethylene terephthalate bottles for the packaging of drinking water. Biomed. Health 2017, 2, 224–229. [Google Scholar]
- Wenzel, A.G.; Brock, J.W.; Bloom, M.; Newman, R.B.; Kucklick, J.R.; Cruze, L.; Somerville, S.; Wineland, R.; Butts, C.; Unal, E. Influence of race on prenatal phthalate exposure and anogenital measurements among boys and girls. Environ. Int. 2017, 110, 61–70. [Google Scholar] [CrossRef] [PubMed]
- Hosney, H.; Nadiem, B.; Ashour, I.; Mustafa, I.; El-Shibiny, A. Epoxidized vegetable oil and bio-based materials as PVC plasticizer. J. Appl. Polym. Sci. 2018, 135, 46270. [Google Scholar] [CrossRef]
- Li, Y.; Lu, J.; Yin, X.; Liu, Z.; Tong, Y.; Zhou, L. Indoor phthalate concentrations in residences in Shihezi, China: Implications for preschool children’s exposure and risk assessment. Environ. Sci. Pollut. Res. 2019, 26, 19785–19794. [Google Scholar] [CrossRef] [PubMed]
- Zaki, G.; Shoeib, T. Concentrations of several phthalates contaminants in Egyptian bottled water: Effects of storage conditions and estimate of human exposure. Sci. Total Environ. 2018, 618, 142–150. [Google Scholar] [CrossRef] [PubMed]
- Staples, C.A.; Peterson, D.R.; Parkerton, T.F.; Adams, W.J. The environmental fate of phthalate esters: A Literature review. Chemosphere 1997, 35, 667–749. [Google Scholar] [CrossRef]
- Gao, D.; Li, Z.; Wang, H.; Liang, H. An overview of phthalate acid ester pollution in China over the last decade: Environmental occurrence and human exposure. Sci. Total Environ. 2018, 645, 1400–1409. [Google Scholar] [CrossRef]
- Steinemann, A.C. Fragranced consumer products and undisclosed ingredients. Environ. Impact Assess. Rev. 2009, 29, 32–38. [Google Scholar] [CrossRef]
- Green, R.; Hauser, R.; Calafat, A.M.; Weuve, J.; Schettler, T.; Set, R. Use of di(2-ethylhexyl) phthalate-containing medical products and urinary levels of mono(2-ethylhexyl) phthalate in neonatal intensive care unit infants. Environ. Health Perspect. 2005, 113, 1222–1225. [Google Scholar] [CrossRef]
- Schecter, A.; Lorber, M.; Guo, Y.; Wu, Q.; Yun, S.H.; Kannan, K. Phthalate concentrations and dietary exposure from food purchased in New York State. Environ. Health Perspect. 2013, 121, 473–479. [Google Scholar] [CrossRef] [PubMed]
- Bergé, A.; Gasperi, J.; Rocher, V.; Gras, L.; Coursimault, A. Phthalates and alkylphenols in industrial and domestic effluents: Case of Paris conurbation (France). Sci. Total Environ. 2014, 488–489, 26–35. [Google Scholar] [CrossRef] [PubMed]
- Ritchie, H.; Roser, M. Plastic Pollution. Available online: https://ourworldindata.org/plastic-pollution (accessed on 1 September 2018).
- Marttinen, S.K.; Kettunen, R.H.; Sormunen, K.M.; Rintala, J.A. Removal of bis(2-ethylhexyl) phthalate at a sewage treatment plant. Water Res. 2003, 37, 1385–1393. [Google Scholar] [CrossRef]
- Koniecki, D.; Wang, R.; Moody, R.P.; Zhu, J. Phthalates in cosmetic and personal care products: Concentrations and possible dermal exposure. Environ. Res. 2011, 111, 329–336. [Google Scholar] [CrossRef]
- Cousins, I.; Mackay, D. Correlating the physical-chemical properties of phthalate esters using the ‘three solubility’ approach. Chemosphere 2000, 41, 1389–1399. [Google Scholar] [CrossRef]
- Gibson, R.W.; Wa, M.J.; Padgett, E.; Lopez-Real, J.M.; Beck, A.J. Impact of drying and composting procedures on the concentrations of 4-nonylphenols, di-(2-ethylhexyl) phthalate and polychlorinated biphenyls in anaerobically digested sewage sludge. Chemosphere 2007, 68, 1352–1358. [Google Scholar] [CrossRef]
- Bagó, B.; Martín, Y.; Mejía, G.; Broto-Puig, F.; Díaz-Ferrero, J.; Agut, M.; Comellas, L. Di-(2-ethylhexyl)phthalate in sewage sludge and post-treated sludge: Quantitative determination by HRGC-MS and mass spectral characterization. Chemosphere 2005, 59, 1191–1195. [Google Scholar] [CrossRef]
- Adhoum, N.; Monser, L. Removal of phthalate on modified activated carbon: Application to the treatment of industrial wastewater. Sep. Purif. Technol. 2004, 38, 233–239. [Google Scholar] [CrossRef]
- Matamoros, V.; Rodríguez, Y.; Albaigés, J. A comparative assessment of intensive and extensive wastewater treatment technologies for removing emerging contaminants in small communities. Water Res. 2016, 88, 777–785. [Google Scholar] [CrossRef]
- Menga, X.Z.; Wanga, Y.; Xianga, N.; Chena, L.; Liub, Z.; Wub, B.; Daib, X.; Zhangc, Y.H.; Xied, Z.; Ebinghausd, R. Flow of sewage sludge-borne phthalate esters (PAEs) from human release to human intake: Implication for risk assessment of sludge applied to soil. Sci. Total Environ. 2014, 476–477, 242–249. [Google Scholar] [CrossRef]
- Defoe, D.L.; Holcombe, G.W.; Hammermeister, D.E.; Biesinger, K.E. Solubility and toxicity of eight phthalate esters to four aquatic organisms. Environ. Toxicol. Chem. 1990, 9, 623–636. [Google Scholar] [CrossRef]
- Nøgletal for Miljøfarlige Stoffer i Spildevand fra Renseanlæg—På Baggrund af Data fra det Nationale Overvågningsprogram for Punktkilder 1998–2009/Key Figures for Environmentally Hazardous Substances in Wastewater from Wastewater Treatment Plants—Based on Data from the National Point Source Monitoring Program 1998–2009; Nature Agency, Ministry of the Environment: Copenhagen, Denmark, 2011; p. 64. ISBN 978-87-7279-285-9.
- Çifci, D.I.; Osman, K.K.; Arikan, A. Occurrence of phthalates in sewage sludge from three wastewater treatment plants in Istanbul, Turkey. Clean Soil Air Water 2013, 41, 851–855. [Google Scholar] [CrossRef]
- Jensen, J.; Jepsen, S.E. The production, use and quality of sewage sludge in Denmark. Waste Manag. 2005, 25, 239–247. [Google Scholar] [CrossRef] [PubMed]
- List of Undesirable Substances 2000; Environmental Review No. 9. EPA; Miljøstyrelsen. Miljøministeriet: Copenhagen, Denmark, 2001.
- Restrictions on the Manufacture, Placing on the Market and Use of Certain Dangerous Substances, Mixtures and Articles (XVII Annex of REACH/ ECHA). Available online: https://echa.europa.eu/lt/substances-restricted-under-reach (accessed on 28 September 2020).
| Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).